 Open Access
Open Access
REVIEW
Propolis as a potential modulator of aryl hydrocarbon receptor signaling in inflammation
1 Graduate Program in Food, Nutrition and Health—Institute of Nutrition, State University of Rio de Janeiro (UERJ), Rio de Janeiro, Brazil
2 Graduate Program in Biological Sciences—Physiology, Federal University of Rio de Janeiro (UFRJ), Rio de Janeiro, Brazil
3 Graduate Program in Medical Sciences, Fluminense Federal University (UFF), Rio de Janeiro, Brazil
4 Graduate Program in Nutrition Sciences, Fluminense Federal University (UFF), Rio de Janeiro, Brazil
* Corresponding Author: Denise Mafra. Email:
Current Address: Hospital Universitário Antônio Pedro, Universidade Federal Fluminense (UFF), Rua Marquês do Paraná n° 303, 4° Andar, Niterói, Rio de Janeiro, Brazil
European Cytokine Network 2026, 37(1), 1-11. https://doi.org/10.32604/ecn.2026.0ECN78096
Received 24 December 2025; Accepted 24 February 2026; Issue published 13 April 2026
Abstract
The aryl hydrocarbon receptor (AhR) is a ligand-activated transcription factor that exhibits antagonistic pleiotropy, mediating both protective and detrimental cellular effects depending on the ligand and context. AhR can be activated by a variety of endogenous and exogenous stimuli, including environmental pollutants, UVB radiation, heme, arachidonic acid metabolites, gut microbiota–derived compounds, and xenobiotics. Upon activation, AhR translocates to the nucleus, where it dimerizes with the aryl hydrocarbon receptor nuclear translocator (ARNT) and binds to xenobiotic response elements, inducing the expression of genes involved in xenobiotic metabolism, oxidative stress responses, and inflammatory signaling. In addition to these classical pathways, AhR also modulates immune function by regulating cytokine networks, including IL-17, IL-22, and IL-10, which play key roles in barrier integrity and immune homeostasis. Bioactive dietary compounds, particularly polyphenols such as flavonoids, have emerged as potential modulators of AhR signaling. Propolis, a complex bee-derived product rich in flavonoids and other phenolic compounds, has demonstrated antioxidant and anti-inflammatory effects across multiple experimental models. However, the specific mechanisms through which propolis interacts with AhR signaling remain poorly understood. This mini-review summarizes current evidence on the potential role of propolis as an AhR modulator, discusses its implications for immune regulation, barrier function, and inflammation control, and highlights areas for future research.Keywords
The aryl hydrocarbon receptor (AhR) is a transcription factor activated by specific agonists. Activation of the AhR leads to nuclear translocation and transcriptional regulation of genes associated with xenobiotic metabolism, immune regulation, oxidative stress control, and cellular differentiation. By integrating environmental, dietary, and microbial-derived signals, AhR influences critical cellular responses, including cytokine production, immune cell polarization, epithelial barrier integrity, and metabolic adaptation [1,2]. Dysregulation of these pathways has been implicated in chronic inflammatory conditions, metabolic disorders, and chronic kidney disease [3,4].
AhR is expressed in various tissues and plays multiple roles, regulating the physiological functions of the cardiovascular, gastrointestinal, nervous, immune, and circadian systems [5]. Several studies have already described the role of the xenobiotic pollutant 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) as a potent AhR agonist. In addition, non-xenobiotic AhR ligands have been identified, including tryptophan metabolites from microbial metabolism, heme metabolites, and indole metabolites [3].
Given this diversity of endogenous and exogenous ligands, increasing attention has been directed toward understanding how nutrients and bioactive dietary compounds modulate AhR signaling. Advances in nutritional and molecular sciences have revealed that specific bioactive compounds can interact with the AhR pathway, influencing downstream cellular responses. In this context, polyphenols, particularly plant-derived flavonoids, have been identified as biologically relevant AhR ligands [6,7].
Polyphenols, the most prevalent antioxidants in the human diet, are found in a wide range of foods, including vegetables, fruits, teas, wines, and chocolate. They play a role in disease prevention [8]. Polyphenols can be classified into families based on their distinct chemical structures, with flavonoids, lignans, stilbenes, and phenolic acids representing the primary families [9]. Among flavonoids, the main subclasses reported to modulate the AhR are flavones, flavonols, flavanones, and isoflavones [7]. The precise mechanisms and implications of flavonoid-AhR interactions are actively investigated in nutritional science and toxicology, shedding light on the multifaceted health benefits of these bioactive compounds.
The AhR signaling pathway is a multistep process that begins with ligand binding and involves conformational changes, interactions with multiple cofactors, nuclear translocation, heterodimerization with the aryl hydrocarbon receptor nuclear translocator (ARNT), and subsequent binding of the AhR–ARNT complex to specific DNA response elements. Through these sequential events, polyphenols may modulate AhR activity at multiple regulatory levels, including nuclear translocation, receptor–cofactor interactions, chromatin remodeling, and the recruitment of transcriptional co-regulators, ultimately influencing gene expression outcomes [10].
Among the sources of polyphenols is propolis, a complex mixture produced by bees and composed of resinous and balsamic plant materials, along with bee saliva and enzymes [8,9]. Due to its rich composition and salutary properties, mainly antioxidant and anti-inflammatory, propolis has shown great potential to improve health and help treat several diseases [11]. A study in hepatocellular carcinoma cells showed that propolis and its flavonoids (chrysin, pinocembrin, galangin, and pinobanksin) exhibit potent antioxidant and anti-inflammatory effects, activating the nuclear factor erythroid 2–related factor 2 (Nrf2) pathway and inhibiting the nuclear factor kappa B (NF-kB) pathway in a dose-dependent manner [12]. Studies have shown crosstalk between the AhR, NF-kB, and Nrf2 pathways [13]. Additionally, studies in patients with chronic kidney disease [11], rheumatoid arthritis [14], diabetes [12], and COVID-19 [15] have shown the anti-inflammatory effects of propolis [16,17].
Despite our limited understanding of the molecular mechanisms underlying this signaling pathway, AhR modulators, such as polyphenols in propolis, may significantly impact health. This mini review explores the potential role of propolis as an AhR modulator.
The AhR is a ligand-activated transcription factor encoded by a gene located on chromosome 7p15. It is part of the period (PER)-ARNT-single-minded protein (SIM) (PER-ARNT-SIM) superfamily of transcription factors [3]. AhR is considered a sensor that integrates endogenous and exogenous signals within the cell, employing complex mechanisms to trigger responses crucial to homeostasis [3]. In general, the regulation of the AhR-ARNT heterodimer is complex and involves multiple biological processes, including xenobiotic metabolism, cell-cycle regulation, and immune responses [3].
The AhR typically exists in an inactive form in the cytoplasm, as part of a stable protein complex that includes two heat shock proteins (HSPs), an AHR-interacting protein (AIP, also known as XAP2), a co-chaperone p23, and a protein kinase SRC [3]. Some endogenous AhR ligands include tryptophan-derived metabolites such as kynurenine, microbiota-derived indole-3-acetate (IAA), and 6-formylindolo[3,2-b]carbazole (FICZ). Environmental pollutants such as dioxins e.g. TCDD (2,3,7,8-tetrachlorodibenzo-p-dioxin) and polycyclic aromatic hydrocarbons (benzo[a]pyrene) are considered exogenous AhR ligands. Additionally, diet can provide ligands, including flavonoids (e.g., quercetin and resveratrol), carotenoids, and glucosinolates [3].
The biological effects of AhR activation are highly context-dependent and determined primarily by the nature of the ligand, the signaling pathway engaged, and the cellular environment.
In the canonical pathway, upon binding to specific ligands, AhR is activated and translocates to the nucleus, where it heterodimerizes with ARNT. The AhR–ARNT complex binds to xenobiotic or dioxin response elements (XREs/DREs) in the regulatory regions of target genes, inducing the transcription of phase I enzymes, such as cytochrome P450 1A1 (CYP1A1), and phase II detoxification and antioxidant enzymes, including glutathione S-transferase-α (GST-α) and NAD(P)H quinone reductase 1 (NQO1) [3,18]. CYP1A1 expression is therefore widely used as a hallmark of AhR activation [19].
AhR can activate noncanonical pathways by interacting with alternative DNA response elements, regulatory RNAs, or other transcription factors, particularly nuclear factor NF-κB. Through both indirect mechanisms, such as suppressor of cytokine signaling 2 (SOCS2), and direct protein–protein interactions, AhR can modulate NF-κB–dependent transcriptional programs [3,18]. To date, however, experimental studies directly addressing the integrated AhR–SOCS2–NF-κB signaling axis remain scarce, and most available evidence supporting this mechanism derives from studies examining pairwise interactions between AhR and NF-κB or from SOCS-mediated modulation of NF-κB signaling [20,21]. In this context, Kim et al. (2019) reported that indoxyl sulfate is associated with the modulation of TNF-α expression in human macrophages through an axis involving AhR, NF-κB, and SOCS2 [22]. IS-mediated AhR activation was shown to promote interaction with the NF-κB p65 subunit and to induce SOCS2, which in turn negatively modulates NF-κB signaling. These findings suggest a non-canonical regulatory mechanism by which a uremic toxin may influence inflammatory responses. This bidirectional crosstalk establishes a regulatory feedback loop in which AhR and NF-κB mutually influence each other’s expression and activity [10,23].
Importantly, although classical AhR signaling is primarily associated with xenobiotic detoxification and antioxidant responses, alternative AhR-mediated pathways may promote proinflammatory signaling, depending on ligand specificity and cellular context. Endogenous regulatory mechanisms further control this functional duality. The AhR repressor (AhRR) negatively regulates AhR transcriptional activity by competing for ARNT binding, whereas hypoxia-inducible factor 1α (HIF-1α) also competes with AhR for ARNT, thereby modulating AhR-dependent gene expression [1].
Termination of AhR signaling occurs through nuclear export of the receptor, followed by proteasomal degradation in the cytoplasm, ensuring tight temporal control of transcriptional responses [18].
In addition to these regulatory mechanisms, AhR activity can be directly activated or inhibited by natural dietary compounds, including tryptophan-derived metabolites, carotenoids, and flavonoids. These bioactive compounds represent important diet-derived AhR modulators, alongside environmental xenobiotics, further highlighting the integrative role of AhR as a sensor of dietary and environmental signals [6].
1.2 Aryl Hydrocarbon Receptors and Polyphenols
Polyphenols are a vast group of phytochemicals ubiquitously present in the plant kingdom and produced as a reaction to stress provoked by reactive oxygen and nitrogen species, pathogens, parasites, plant predators, and ultraviolet radiation [15]. At least 10,000 polyphenolic compounds are distributed in various plant species. They can be classified as flavonoids, the most abundant family of polyphenols, distributed into flavones, flavandiols, flavonols, flavanonols, flavanones, anthocyanidins, catechins, and isoflavones, and non-flavonoids, namely stilbenes, phenolic acids, and lignans. Polyphenols’ defining feature is at least one aromatic ring and one or more hydroxyl groups [15–17].
In recent years, numerous studies have investigated dietary polyphenols and their impact on various metabolic diseases, including non-alcoholic fatty liver disease, chronic kidney disease, diabetes mellitus, and cardiovascular and neurodegenerative diseases. These effects are primarily attributed to their anti-inflammatory, antioxidant, insulin-resistance-modulating, and antiproliferative properties, as well as to their ability to inhibit specific enzyme pathways [24–27]. In the human diet, polyphenols are among the most abundant antioxidant compounds, primarily derived from plant-based foods and products, including fruits, vegetables, tea, wine, cocoa, dark chocolate, coffee, cereals, and propolis [27,28].
Polyphenols can act as AhR ligands (agonists and antagonists), playing a significant role in their signaling pathways (table 1). Many flavonoids act as selective AhR modulators, capable of activating or inhibiting AhR signaling depending on ligand competition, concentration, and cellular context. Importantly, AhR agonism or antagonism alone does not reliably predict inflammatory outcomes. While some AhR agonists elicit anti-inflammatory or cytoprotective effects, these responses are highly context dependent. Moreover, most studies have focused on AhR activation assessed by reporter assays or target gene expression, with limited evaluation of cytokine or functional inflammatory endpoints. Thus, inflammatory readouts provide a more informative framework than receptor activation alone [6,29].
Ashida (2000) reported that multiple flavones and flavonols suppressed AhR activation by TCDD, which is associated with high toxicity [30]. Palermo et al. (2003) investigated the effects of epigallocatechin gallate (EGCG) and epigallocatechin (EGC), the primary polyphenols present in green tea extracts (GTE), on AhR gene induction in mouse hepatoma cells. Both compounds modulated AhR transcription and were responsible for the most pronounced AhR-antagonistic effects observed with GTE [31].
In contrast, Fukuda et al. (2007) did not observe competitive or allosteric inhibition of AhR by EGCG in cytosolic fractions from rat liver. These findings suggest that catechins may indirectly inhibit AhR activation by targeting partner proteins within the AhR complex rather than AhR itself. In the same study, only specific flavonoid subclasses (namely flavones, flavonols, and flavanones) acted as direct competitive AhR antagonists [29]. A review by Amakura et al. (2008) corroborates these findings, reporting that specific flavones (e.g., apigenin), flavonols (e.g., quercetin), and anthraquinones (e.g., emodin) inhibit TCDD-induced AhR activation in vitro reporter gene assay using mouse hepatoma cells [32].
Furthermore, in a mouse AhR study, baicalin (a flavone glycoside) attenuated the myocardial injury and inflammatory response by inhibiting model expression [33]. Similarly, tangeretin (an O-polymethoxylated flavone) has shown a normalizing effect on AhR hepatic expression, reducing the DNA damage triggered by 7, 12-dimethylbenz[a]anthracene (DMBA) [34].
The modulatory effects of flavonoids on AhR signaling are highly context-dependent and influenced by parameters such as exposure time, dose, species, and tissue type. Van der Heiden et al. (2009) systematically investigated these variables using multiple AhR-responsive reporter cell lines and demonstrated that flavonoids can exert both agonistic and antagonistic activities depending on the experimental context. For instance, genistein exhibited agonistic activity in rat hepatoma cells after short-term exposure (6 h), but not after prolonged exposure (24 h). In human breast tumor cells, flavonoids displayed time-dependent agonistic or antagonistic effects, whereas in human hepatoma cells, only antagonistic activity was observed. These findings highlight that flavonoid–AhR interactions are dynamic and may shift over time, underscoring the importance of cellular context in interpreting AhR-mediated responses [35].
A study by Ronnekleiv-Kelly et al. (2016) using various human colorectal cancer cell lines demonstrated that treatment with chrysin (a flavone) induced apoptosis through an AhR-dependent pathway [36]. Nevertheless, baicalin and chrysin have been reported to exhibit both ligand-like properties, displaying agonist and antagonist effects, along with luteolin, flavone, and apigenin [30,37–40].
In a rat model, resveratrol treatment was associated with enhanced antioxidant capacity, reduced lipid peroxidation, and upregulation of AhR gene expression. In contrast, selective AhR inhibition led to increased oxidative stress, DNA fragmentation, and downregulation of AhR signaling. These findings highlight the essential physiological role of AhR in redox homeostasis and tissue integrity and reinforce the concept that natural AhR modulators may exert protective effects distinct from those of AhR antagonists or toxic ligands [41]. In an experimental model, a polyphenolic compound containing a caffeic acid moiety (acteoside) ameliorated renal dysfunction and fibrosis in adenine-induced chronic kidney disease (CKD) rats, whereas its structural isomer, isoacteoside, in which the caffeic acid group is repositioned, showed limited efficacy. Mechanistically, acteoside inhibited AhR nuclear translocation and downregulated AhR-dependent gene expression, thereby attenuating NF-κB–mediated inflammatory signaling and modulating the Nrf2 antioxidant pathway. These findings suggest that selective AhR antagonists containing bioactive caffeic acid structures may represent promising antifibrotic agents targeting the AhR–NF-κB/Nrf2 axis in CKD [42].
In cardiomyocytes, kynurenine (KYN) activated AhR-dependent mitogen-activated protein kinase (MAPK) signaling, promoting apoptosis, whereas resveratrol attenuated cell death and enhanced protective autophagy by modulating AhR signaling. Pharmacological inhibition of AhR or p38 MAPK further reduced apoptotic responses, underscoring AhR as a key mediator of KYN-induced cardiotoxicity and a potential target of dietary polyphenols in CKD [43].
In silico and in vitro analyses demonstrated that several hydroxyflavonoids, including chrysin, apigenin, quercetin, and naringenin, exhibit high AhR binding affinity and dose-dependently inhibit AhR activation in the presence of toxic ligands. Notably, naringenin was identified as an AhR antagonist. These findings highlight the structure-dependent modulation of AhR by flavonoids and support their potential therapeutic relevance as AhR-targeting agents [44].
From the flavonol class, galangin, quercitrin [45], and isorhamnetin [45] have only presented antagonistic effects on AhR, while kaempferol, fisetin, quercetin [39,45], rutin [38], myricetin, morin [39], tamarixetin [30,45] and 3,6,2′,4′-tetrahydroxyflavone [39] have shown both agonist and antagonist activity. In the flavanone group, such duplicity is found in naringin [30,38], flavanone [30,45], and hesperetin [38,46], whereas eriodyctiol [15] is only related to the antagonistic effects on AhR. Among the isoflavones, genistein and daidzein exhibit both antagonist and antagonist activities [47,48].
While the structure of each flavonoid subclass does not allow conclusive inferences upon the respective effect (agonist/antagonist) on AhR modulation, flavonoids are broadly present in the human diet, and many studies have defined them as natural AhR modulators. As previously mentioned, some polyphenols appear to activate and inhibit the AhR, leading to conflicting results in the literature, which may be attributed to differences in cell types, tissues, and assay conditions [49]. Therefore, further studies are needed to deepen the understanding of the role of polyphenols in the AhR signaling pathway, their antagonist effects, and their therapeutic use in clinical practice.
Propolis is a resinous substance produced by bees from various plant exudates collected from different parts of plants, including twigs, flowers, pollen, and buds. Its chemical composition depends on the region’s flora. Therefore, this substance has a variable composition, and the combination of its various chemical components determines its broad biological activity. Bioactive compounds, such as terpenes, lignans, amino acids, phenolic acids, flavonoids (flavones, flavanones, flavonols, dihydroflavonols, and chalcones), fatty acids, sugars, vitamins, and minerals, have been identified in propolis [55–57].
Propolis has been used for centuries for medicinal purposes, and current research has confirmed its biological effects, including anticancer, antidiabetic, antimicrobial, antioxidant, immunomodulatory, and anti-inflammatory activities [56]. Research has shown that propolis, with its wide range of bioactive compounds, interacts with diverse molecular targets, either directly or by modulating gene expression. Recently, the interaction between propolis compounds and AhR has been explored, elucidating this transcription factor’s role in mediating propolis’s effects on human health. In this regard, propolis has been investigated as a potential modulator of the AhR pathway [57].
Park et al. (2004) examined the effects of dried Brazilian propolis extract on rat hepatic cytosolic fractions and demonstrated that ethanolic propolis extracts, rich in flavonoid aglycones, dose-dependently suppressed AhR-mediated transformation induced by TCDD [50].
As previously mentioned, the CYP1A1 gene is a sensitive marker of AhR activation. CYP1A1 is involved in the production of reactive oxygen species (ROS), such as superoxide anion and hydrogen peroxide, and in the metabolic activation of procarcinogens [19]. Pinostrobin, a bioactive dihydroxyflavone isolated from propolis and plants, has been suggested as an AhR antagonist, negatively modulating the AhR/CYP1A1 signaling pathway, as shown by Zhao et al. (2023) in human coronavirus OC43 (HCoV-OC43) infected cells, elucidating the potential beneficial effect of this bioactive compound [58].
An in vitro and in vivo study demonstrated that pinocembrin (PCB), a natural flavonoid from honey and propolis, alleviated the toxicity of benzo(a)pyrene, a carcinogen, by attenuating the CYP1A1 expression and DNA damage due to suppression of the AhR/Src/ERK pathways in lung epithelial cells. Also, PCB reduced the activity of CYP1A1 and Src in the lung tissues of mice [53].
Wang et al. (2021) [54] evaluated the chemoprotective effect of galangin, a flavonol found in propolis, against benzo(a)pyrene-induced stomach carcinogenesis in Swiss albino mice. The authors found that pretreatment with galangin prevented lipid peroxidation and restored the loss of cellular antioxidant status. In addition, galangin inhibited the expression of cytochrome P450 isoform genes, AhR system (AhR, ARNT), transcriptional activators (NF-kB), and other tumor growth factors in the stomach tissue of mice.
Ye et al. (2020) [59] investigated the potential effects of three representative flavonoid compounds in Chinese propolis (pinocembrin, galangin, and chrysin) on mitigating ethanol-induced injuries in HepG2 cells. The authors reported that flavonoids could suppress AhR nuclear translocation and CYP1A1 expression, suggesting a protective effect against ethanol-induced injury by inhibiting the AhR-CYP1A1 pathway. These results provide promising evidence for the use of pinocembrin, galangin, and chrysin in preventing and treating ethanol-induced fatty liver disease.
On the other hand, tectochrysin present in propolis extract showed marked AhR binding activity at high concentrations in mouse hepatoma (H1L6.1c2) cells, suggesting an agonist AhR response to propolis extract related to this flavone [32]. Kalthoff & Strassburg (2019) conducted a study to clarify the mechanism underlying the protective antioxidant effects of propolis [60]. Propolis tincture (62% ethanol) significantly decreased hydrogen peroxide levels in human esophageal squamous cell carcinoma (KYSE70). Hydrogen peroxide is a ROS generated during various biochemical processes, such as the activation of xenobiotic compounds by cytochrome P450 enzymes (phase I metabolism). The inactivation and detoxification of this reactive metabolite are mediated by phase II enzymes, such as UDP-glucuronosyltransferase 1A (UGT1A), which act as indirect antioxidants. Different nuclear receptors and transcription factors transcriptionally regulate the UGT1A gene family. In this experiment, propolis was a potential activator of UGT1A transcription and enzyme activity, mediated by AhR and Nrf2 signaling [60].
Hypothetically, another route of interaction between propolis and AhR may involve the gut microbiota. The structural complexity of polyphenols in propolis may affect gut microbiota composition, enabling these compounds to reach the large intestine, where they can modify the intestinal microbiota through prebiotic-like effects and antimicrobial actions [56,61]. On the other hand, the processing of polyphenols can vary with interindividual differences in intestinal microbiota profiles, thereby influencing propolis’s biological activity. In this context, propolis has been reported to affect intestinal barrier function, gut microbiota composition, and gut microbiota metabolites. In vivo and in vitro studies have shown that propolis increases tight junction protein expression (Claudin, Occludin, and Zonula Occludens Protein 1), increases the abundance of salutogenic bacteria, and increases the production of short-chain fatty acids (SCFA: acetic acid, propionic acid, and butyric acid) [56,62]. Interestingly, intestinal AhR ligands have been shown to mitigate gut inflammation and protect intestinal barrier integrity [51,56,58–60].
Furthermore, studies have demonstrated AhR’s involvement in communication with the gut microbiota and in regulating intestinal metabolic homeostasis. However, the specific mechanism by which AhR exerts its effects in this process remains unclear. Notably, certain commensal bacteria produce metabolites that act as AhR agonists. Germ-free mice exhibited lower levels of AhR and cytochrome P450 gene expression than mice with a normal microbiota. Conversely, administration of SCFA derived from microbiota-driven anaerobic fermentation restored the expression of AhR target genes to normal levels. Taken together, this information suggests a role for the gut microbiota in mediating propolis’s communication with the AhR [5,63].
Figure 1 summarizes the possible mechanisms of interaction of the compounds present in propolis with the AhR pathway.
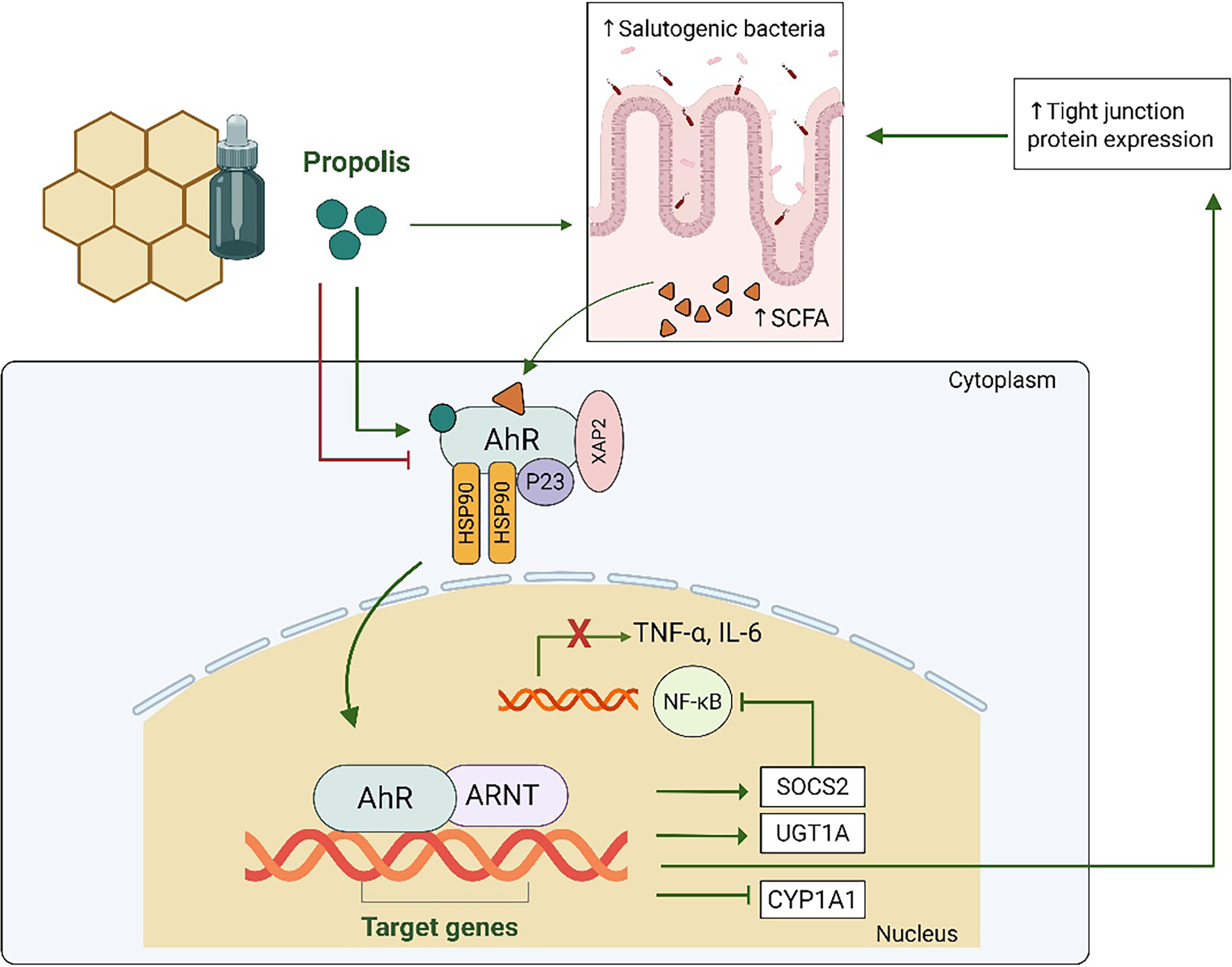
Figure 1: The aryl hydrocarbon receptor (AhR) signaling pathway involves ligand binding, AhR nuclear translocation, dimerization with the AhR Nuclear Translocator (ARNT), and binding to target genes, thereby triggering a range of pleiotropic cellular actions. Polyphenols from propolis may exert both agonist and antagonist effects on the AhR, modulating this pathway through diverse mechanisms. Direct interactions between propolis compounds and AhR may attenuate cytochrome P450 (CYP1A1) expression and upregulate the suppressor of cytokine signaling 2 (SOCS2) and UDP-glucuronosyltransferase 1A (UGT1A) transcription, thereby inhibiting the nuclear factor-κB (NF-κB) pathway. Additionally, polyphenols in propolis may indirectly modulate inflammation via prebiotic-like effects that promote a healthy gut microbiota. This microbiota produces metabolites that interact with AhR, further contributing to propolis’s anti-inflammatory properties. Created by BioRender.com. HSP90: Heat shock protein 90; IL-6: Interleukin-6; P23: p23 co-chaperone protein; SCFA: short-chain fatty acids; Tumor Necrosis Factor Alpha (TNF-α): XAP2: X-associated protein 2. Created by Biorender.com.
Potential Involvement of AhR–Cytokine Signaling in the Immunomodulatory Effects of Propolis
While growing evidence supports the interaction between propolis flavonoids and AhR signaling, experimental studies directly assessing cytokine modulation through AhR-dependent mechanisms in response to propolis remain limited. Nevertheless, AhR activation or inhibition is known to regulate cytokine production in immune and epithelial cells, including tumor necrosis factor alpha (TNF-α), IL-6, interleukin-1 beta (IL-1β), IL-10, IL-17, and IL-22, with outcomes that vary according to the nature of the ligand, the cellular context, and the inflammatory microenvironment [64–68]. Emerging experimental data further indicate that propolis and its flavonoids reduce pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β, while promoting anti-inflammatory mediators, effects commonly associated with NF-κB inhibition and redox regulation [69–72]. Given the established crosstalk between AhR, NF-κB, and Nrf2 pathways, it is biologically plausible that the immunomodulatory effects of propolis are mediated, at least in part, by context-dependent AhR–cytokine signaling.
Within this framework, mechanistic studies using propolis-derived flavonoids provide additional support for this hypothesis. Chrysin has been shown to function as an AhR ligand, inducing AhR-dependent transcriptional activity and TNF-α/TNF-β expression in human colorectal cancer cells, effects that are abolished upon AhR silencing [36]. More recent evidence indicates that flavones, such as chrysin, apigenin, and luteolin, may also function as AhR antagonists. In endothelial cells exposed to the uremic toxin indoxyl sulfate, these compounds attenuated AhR activation by inhibiting nuclear translocation and reducing the expression of classical AhR-responsive genes, including CYP1A1 and AhRR, concomitantly suppressing pro-inflammatory mediators such as IL-6, MCP-1, COX-2, NOX-4, and ICAM-1 [73].
Quercetin acts as a weak AhR agonist, reducing pro-inflammatory cytokines such as TNF-α, IL-6, and IL-17 in neutrophils and colonic tissue while promoting antioxidant responses via NQO1/reactive oxygen species (NQO1/ROS) pathways [74]. In addition, quercetin has been reported to activate AhR signaling in human dendritic cells, leading to reduced production of interleukin-12p70 (IL-12p70) and the acquisition of an immunoregulatory phenotype [75]. Apigenin, a flavonoid present in propolis, has been shown to act as a functional antagonist of the kynurenine/AhR pathway in MED12-mutant uterine leiomyoma cells by inhibiting kynurenine-induced AhR activation, reducing tryptophan 2,3-dioxygenase (TDO2) expression, and blocking TDO2 upregulation mediated by the pro-inflammatory cytokines TNF and IL-1β [76]. Additionally, in a model of eosinophilic airway inflammation, apigenin decreased AhR expression and downregulated type 2 cytokines, including IL-4, IL-5, and IL-13 [77]. Collectively, these findings support the biological plausibility that propolis flavonoids modulate cytokine production through ligand- and context-specific AhR-dependent mechanisms, contributing to their anti-inflammatory effects.
Evidence from diverse disease models indicates that AhR-mediated cytokine regulation is context-dependent rather than uniform across diseases. In CKD, AhR signaling has been implicated in the modulation of multiple pro-inflammatory cytokines as part of the uremic milieu, and the fibrosis/inflammation network, with ligand-dependent effects on NF-κB and downstream inflammatory mediators reported in experimental and clinical settings [78,79]. In contrast, in autoimmune conditions such as rheumatoid arthritis, experimental studies show that AhR activation can contribute to disease pathogenesis by promoting pro-inflammatory cytokine production, including IL-1β and IL-6, and by influencing Th17 cell differentiation, rather than primarily suppressing IL-6 expression [80,81]. These findings suggest that the cytokine profile modulated by AhR varies with disease context and ligand milieu.
In addition to its interactions with NF-κB, the AhR has been increasingly recognized as a modulator of T helper cell–derived cytokine networks, including IL-17, IL-22, and IL-10. AhR activation influences the differentiation and function of Th17 and Th22 cells, which produce IL-17 and IL-22, respectively, with IL-22 playing a key role in maintaining barrier integrity and host defense in mucosal tissues; experimental models demonstrate that AhR deficiency reduces IL-22 production and compromises immune homeostasis, whereas AhR activation enhances IL-22 expression via coordinated interactions with transcription factors including RelB and RORγt at the IL-22 promoter [68,82]. Beyond the IL-17/IL-22/IL-10 axis, other cytokines such as IL-33 are emerging as important modulators of immune responses in complex tissue microenvironments [83]. Proinflammatory mediators, including TNF-α and IL-1β, can compromise epithelial barrier integrity, underscoring the potential of natural compounds, such as propolis flavonoids, to restore cytokine balance and preserve barrier function, potentially through context-dependent modulation of AhR signaling [84]. While direct evidence linking propolis–AhR interactions to shifts between pro-inflammatory IL-17 and barrier-protective IL-22/IL-10 remains limited, propolis extracts have been shown to modulate Th17-associated cytokines such as IL-17 in immune models, consistent with broader immunoregulatory effects on T cell-driven networks [85]. In the context of SARS-CoV-2–associated cytokine storm, hyperactivation of NF-κB drives excessive production of pro-inflammatory mediators, including IL-6, TNF-α, IL-1β, and IL-17, which are linked to acute respiratory distress; modulating these pathways via AhR-dependent suppression of NF-κB and promotion of regulatory/repair cytokines such as IL-22 and IL-10 may therefore represent a mechanism by which AhR ligands could contribute to limiting hyperinflammation [86,87].
The AhR exhibits antagonistic pleiotropy, mediating both harmful and beneficial effects depending on the ligand and context. Toxic signaling, such as persistent activation by TCDD, induces CYP1A1 expression and ROS generation, leading to oxidative stress and cellular damage. In contrast, dietary and microbial ligands, including propolis flavonoids, may favor salutogenic pathways, promoting IL-22–mediated barrier integrity and tissue protection. These findings suggest that propolis could modulate AhR signaling in a context-dependent manner [86,87].
1.4 Limitations and Future Prospects
Although growing evidence supports the AhR’s role as a key interface among dietary compounds, immune regulation, and inflammation, most data on polyphenols and propolis come from in vitro and animal studies that use heterogeneous preparations and experimental conditions. In addition, direct mechanistic evidence linking propolis-induced cytokine modulation specifically to AhR-dependent pathways remains limited, and AhR signaling is highly context dependent. Future studies using standardized propolis preparations, cytokine-specific analyses, and AhR-targeted experimental approaches, including translational human studies, are needed to clarify the therapeutic relevance of propolis as a dietary modulator of the AhR–cytokine axis.
Although initially characterized as a transcription factor involved in xenobiotic detoxification, the AhR is now recognized as a multifunctional regulator with broad implications for human homeostasis. Accumulating evidence indicates that endogenous and diet-derived AhR ligands elicit signaling responses distinct from those triggered by toxic xenobiotics, often promoting regulatory and protective effects. In this context, bioactive compounds present in propolis emerge as relevant modulators of AhR signaling. Both agonistic and antagonistic mechanisms appear to contribute to propolis’s biological effects, suggesting a fine-tuned modulation of the AhR axis rather than simple activation or inhibition. Through direct or indirect interactions with this pathway, propolis-derived polyphenols may represent a promising nutritional strategy to attenuate oxidative stress and inflammation.
Although the available evidence remains limited, recent studies have begun to elucidate the molecular mechanisms underlying the interaction between propolis and the AhR signaling pathway. Further well-designed experimental and clinical investigations are warranted to fully characterize this complex pathway and define the therapeutic potential of propolis as an AhR modulator.
Acknowledgement: The authors thank the institutions and graduate programs to which they are affiliated for their support.
Funding Statement: This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (n. 407814/2024-8) and Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) (n. E-26/200.063/2026).
Author Contributions: Conceptualization: Denise Mafra and Natália Alvarenga Borges; literature search and data curation: Natália Alvarenga Borges, Larissa Manhães, Ludmilla Dias de Santana e Santana, Larissa Fonseca, Ludmila F. M. F. Cardozo and Jessyca Sousa de Brito; writing—original draft preparation: Natália Alvarenga Borges, Larissa Manhães, Ludmilla Dias de Santana e Santana, Ludmila F. M. F. Cardozo, Larissa Fonseca and Jessyca Sousa de Brito; writing—review and editing: all authors; supervision: Denise Mafra. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable. This study is a narrative review based exclusively on previously published literature.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Bahman F, Choudhry K, Al-Rashed F, Al-Mulla F, Sindhu S, Ahmad R. Aryl hydrocarbon receptor: current perspectives on key signaling partners and immunoregulatory role in inflammatory diseases. Front Immunol 2024;15:1421346. doi:10.3389/fimmu.2024.1421346. [Google Scholar] [PubMed] [CrossRef]
2. Coumoul X, Barouki R, Esser C, et al. The aryl hydrocarbon receptor: structure, signaling, physiology and pathology. Signal Transduct Target Ther 2026;11(1):20. doi:10.1038/s41392-025-02500-8. [Google Scholar] [PubMed] [CrossRef]
3. Rothhammer V, Quintana FJ. The aryl hydrocarbon receptor: an environmental sensor integrating immune responses in health and disease. Nat Rev Immunol 2019;19(3):184–97. doi:10.1038/s41577-019-0125-8. [Google Scholar] [PubMed] [CrossRef]
4. Xie H, Yang N, Yu C, Lu L. Uremic toxins mediate kidney diseases: the role of aryl hydrocarbon receptor. Cell Mol Biol Lett 2024;29(1):38. doi:10.1186/s11658-024-00550-4. [Google Scholar] [PubMed] [CrossRef]
5. Kou Z, Dai W. Aryl hydrocarbon receptor: its roles in physiology. Biochem Pharmacol 2021;185(3):114428. doi:10.1016/j.bcp.2021.114428. [Google Scholar] [PubMed] [CrossRef]
6. Stejskalova L, Dvorak Z, Pavek P. Endogenous and exogenous ligands of aryl hydrocarbon receptor: current state of art. Curr Drug Metab 2011;12(2):198–212. doi:10.2174/138920011795016818. [Google Scholar] [PubMed] [CrossRef]
7. Goya-Jorge E, Jorge Rodríguez ME, Veitía MS, Giner RM. Plant occurring flavonoids as modulators of the aryl hydrocarbon receptor. Molecules 2021;26(8):2315. doi:10.3390/molecules26082315. [Google Scholar] [PubMed] [CrossRef]
8. Bié J, Sepodes B, Fernandes PCB, Ribeiro MHL. Polyphenols in health and disease: gut microbiota, bioaccessibility, and bioavailability. Compounds 2023;3(1):40–72. doi:10.3390/compounds3010005. [Google Scholar] [CrossRef]
9. Rana A, Samtiya M, Dhewa T, Mishra V, Aluko RE. Health benefits of polyphenols: a concise review. J Food Biochem 2022;46(10):e14264. doi:10.1111/jfbc.14264. [Google Scholar] [PubMed] [CrossRef]
10. Xue Z, Li D, Yu W, et al. Mechanisms and therapeutic prospects of polyphenols as modulators of the aryl hydrocarbon receptor. Food Funct 2017;8(4):1414–37. doi:10.1039/c6fo01810f. [Google Scholar] [PubMed] [CrossRef]
11. Zullkiflee N, Taha H, Usman A. Propolis: its role and efficacy in human health and diseases. Molecules 2022;27(18):6120. doi:10.3390/molecules27186120. [Google Scholar] [PubMed] [CrossRef]
12. Xu W, Lu H, Yuan Y, Deng Z, Zheng L, Li H. The antioxidant and anti-inflammatory effects of flavonoids from propolis via Nrf2 and NF-κB pathways. Foods 2022;11(16):2439. doi:10.3390/foods11162439. [Google Scholar] [PubMed] [CrossRef]
13. Celik-Turgut G, Olmez N, Koc T, et al. Role of AHR, NF-kB and CYP1A1 crosstalk with the X protein of Hepatitis B virus in hepatocellular carcinoma cells. Gene 2023;853(1):147099. doi:10.1016/j.gene.2022.147099. [Google Scholar] [PubMed] [CrossRef]
14. Maddahi M, Nattagh-Eshtivani E, Jokar M, et al. The effect of propolis supplementation on cardiovascular risk factors in women with rheumatoid arthritis: a double-blind, placebo, controlled randomized clinical trial. Phytother Res 2023;37(12):5424–34. doi:10.1002/ptr.7996. [Google Scholar] [PubMed] [CrossRef]
15. Silveira MAD, de Alencar Menezes M, de Souza SP, et al. Standardized Brazilian green propolis extract (EPP-AF®) in COVID-19 outcomes: a randomized double-blind placebo-controlled trial. Sci Rep 2023;13(1):18405. doi:10.1038/s41598-023-43764-w. [Google Scholar] [PubMed] [CrossRef]
16. Baptista BG, Fanton S, Ribeiro M, et al. The effect of Brazilian Green Propolis extract on inflammation in patients with chronic kidney disease on peritoneal dialysis: a randomised double-blind controlled clinical trial. Phytomedicine 2023;114(2):154731. doi:10.1016/j.phymed.2023.154731. [Google Scholar] [PubMed] [CrossRef]
17. Chermut TR, Fonseca L, Figueiredo N, et al. Effects of propolis on inflammation markers in patients undergoing hemodialysis: a randomized, double-blind controlled clinical trial. Complement Ther Clin Pract 2023;51(2):101732. doi:10.1016/j.ctcp.2023.101732. [Google Scholar] [PubMed] [CrossRef]
18. Shivanna B, Chu C, Moorthy B. The aryl hydrocarbon receptor (AHRa novel therapeutic target for pulmonary diseases? Int J Mol Sci 2022;23(3):1516. doi:10.3390/ijms23031516. [Google Scholar] [PubMed] [CrossRef]
19. Coelho NR, Pimpão AB, Correia MJ, et al. Pharmacological blockage of the AHR-CYP1A1 axis: a call for in vivo evidence. J Mol Med 2022;100(2):215–43. doi:10.1007/s00109-021-02163-2. [Google Scholar] [PubMed] [CrossRef]
20. Vogel CFA, Khan EM, Leung PSC, et al. Cross-talk between aryl hydrocarbon receptor and the inflammatory response: a role for nuclear factor-κB. J Biol Chem 2014;289(3):1866–75. doi:10.1074/jbc.M113.505578. [Google Scholar] [PubMed] [CrossRef]
21. Paul I, Batth TS, Iglesias-Gato D, et al. The ubiquitin ligase Cullin5SOCS2 regulates NDR1/STK38 stability and NF-κB transactivation. Sci Rep 2017;7(1):42800. doi:10.1038/srep42800. [Google Scholar] [PubMed] [CrossRef]
22. Kim HY, Yoo TH, Cho JY, Kim HC, Lee WW. Indoxyl sulfate-induced TNF-α is regulated by crosstalk between the aryl hydrocarbon receptor, NF-κB, and SOCS2 in human macrophages. FASEB J 2019;33(10):10844–58. doi:10.1096/fj.201900730R. [Google Scholar] [PubMed] [CrossRef]
23. Riaz F, Pan F, Wei P. Aryl hydrocarbon receptor: the master regulator of immune responses in allergic diseases. Front Immunol 2022;13:1057555. doi:10.3389/fimmu.2022.1057555. [Google Scholar] [PubMed] [CrossRef]
24. Abenavoli L, Larussa T, Corea A, et al. Dietary polyphenols and non-alcoholic fatty liver disease. Nutrients 2021;13(2):494. doi:10.3390/nu13020494. [Google Scholar] [PubMed] [CrossRef]
25. Ashkar F, Bhullar KS, Wu J. The effect of polyphenols on kidney disease: targeting mitochondria. Nutrients 2022;14(15):3115. doi:10.3390/nu14153115. [Google Scholar] [PubMed] [CrossRef]
26. Da Porto A, Cavarape A, Colussi G, Casarsa V, Catena C, Sechi LA. Polyphenols rich diets and risk of type 2 diabetes. Nutrients 2021;13(5):1445. doi:10.3390/nu13051445. [Google Scholar] [PubMed] [CrossRef]
27. Tangney CC, Rasmussen HE. Polyphenols, inflammation, and cardiovascular disease. Curr Atheroscler Rep 2013;15(5):324. doi:10.1007/s11883-013-0324-x. [Google Scholar] [PubMed] [CrossRef]
28. Zhu L, Zhang J, Yang H, et al. Propolis polyphenols: a review on the composition and anti-obesity mechanism of different types of propolis polyphenols. Front Nutr 2023;10:1066789. doi:10.3389/fnut.2023.1066789. [Google Scholar] [PubMed] [CrossRef]
29. Fukuda I, Mukai R, Kawase M, Yoshida KI, Ashida H. Interaction between the aryl hydrocarbon receptor and its antagonists, flavonoids. Biochem Biophys Res Commun 2007;359(3):822–7. doi:10.1016/j.bbrc.2007.05.199. [Google Scholar] [PubMed] [CrossRef]
30. Ashida H. Suppressive effects of flavonoids on dioxin toxicity. Biofactors 2000;12(1–4):201–6. doi:10.1002/biof.5520120132. [Google Scholar] [PubMed] [CrossRef]
31. Palermo CM, Martin Hernando JI, Dertinger SD, Kende AS, Gasiewicz TA. Identification of potential aryl hydrocarbon receptor antagonists in green tea. Chem Res Toxicol 2003;16(7):865–72. doi:10.1021/tx025672c. [Google Scholar] [PubMed] [CrossRef]
32. Amakura Y, Tsutsumi T, Sasaki K, Nakamura M, Yoshida T, Maitani T. Influence of food polyphenols on aryl hydrocarbon receptor-signaling pathway estimated by in vitro bioassay. Phytochemistry 2008;69(18):3117–30. doi:10.1016/j.phytochem.2007.07.022. [Google Scholar] [PubMed] [CrossRef]
33. Xue Y, Shui X, Su W, et al. Baicalin inhibits inflammation and attenuates myocardial ischaemic injury by aryl hydrocarbon receptor. J Pharm Pharmacol 2015;67(12):1756–64. doi:10.1111/jphp.12484. [Google Scholar] [PubMed] [CrossRef]
34. Arivazhagan L, Sorimuthu Pillai S. Tangeretin, a citrus pentamethoxyflavone, exerts cytostatic effect via p53/p21 up-regulation and suppresses metastasis in 7,12-dimethylbenz(α)anthracene-induced rat mammary carcinoma. J Nutr Biochem 2014;25(11):1140–53. doi:10.1016/j.jnutbio.2014.06.007. [Google Scholar] [PubMed] [CrossRef]
35. Van der Heiden E, Bechoux N, Muller M, et al. Food flavonoid aryl hydrocarbon receptor-mediated agonistic/antagonistic/synergic activities in human and rat reporter gene assays. Anal Chim Acta 2009;637(1–2):337–45. doi:10.1016/j.aca.2008.09.054. [Google Scholar] [PubMed] [CrossRef]
36. Ronnekleiv-Kelly SM, Nukaya M, Díaz-Díaz CJ, et al. Aryl hydrocarbon receptor-dependent apoptotic cell death induced by the flavonoid chrysin in human colorectal cancer cells. Cancer Lett 2016;370(1):91–9. doi:10.1016/j.canlet.2015.10.014. [Google Scholar] [PubMed] [CrossRef]
37. Zhu W, Chen X, Yu J, et al. Baicalin modulates the Treg/Teff balance to alleviate uveitis by activating the aryl hydrocarbon receptor. Biochem Pharmacol 2018;154(9):18–27. doi:10.1016/j.bcp.2018.04.006. [Google Scholar] [PubMed] [CrossRef]
38. Kaur M, Badhan RKS. Phytochemical mediated-modulation of the expression and transporter function of breast cancer resistance protein at the blood-brain barrier: an in-vitro study. Brain Res 2017;1654(Pt A):9–23. doi:10.1016/j.brainres.2016.10.020. [Google Scholar] [PubMed] [CrossRef]
39. Jin UH, Park H, Li X, et al. Structure-dependent modulation of aryl hydrocarbon receptor-mediated activities by flavonoids. Toxicol Sci 2018;164(1):205–17. doi:10.1093/toxsci/kfy075. [Google Scholar] [PubMed] [CrossRef]
40. Ashida H, Harada K, Mishima S, Mitani T, Yamashita Y, Matsumura F. Luteolin suppresses TCDD-induced wasting syndrome in a cultured adipocyte model. Pestic Biochem Physiol 2015;120:14–20. doi:10.1016/j.pestbp.2014.11.005. [Google Scholar] [PubMed] [CrossRef]
41. Bustani GS, Alghetaa HFK. Aryl hydrocarbon receptor signaling in male fertility: protective role of resveratrol and disruptive effects of CH223191 in adult male rats. Vet World 2025;18(5):1274–87. doi:10.14202/vetworld.2025.1274-1287. [Google Scholar] [PubMed] [CrossRef]
42. Wang YN, Wu X, Shan QY, et al. Acteoside-containing caffeic acid is bioactive functional group of antifibrotic effect by suppressing inflammation via inhibiting AHR nuclear translocation in chronic kidney disease. Acta Pharmacol Sin 2025;46(11):2975–88. doi:10.1038/s41401-025-01598-4. [Google Scholar] [PubMed] [CrossRef]
43. Mohiti S, Alizadeh E, Bisgaard LS, Ebrahimi-Mameghani M, Christoffersen C. The AhR/P38 MAPK pathway mediates kynurenine-induced cardiomyocyte damage: the dual role of resveratrol in apoptosis and autophagy. Biomed Pharmacother 2025;186:118015. doi:10.1016/j.biopha.2025.118015. [Google Scholar] [PubMed] [CrossRef]
44. de Santana MR, Dos Santos YB, Santos KS, et al. Differential interactions of flavonoids with the aryl hydrocarbon receptor in silico and their impact on receptor activity in vitro. Pharmaceuticals 2024;17(8):980. doi:10.3390/ph17080980. [Google Scholar] [PubMed] [CrossRef]
45. Ashida H, Fukuda I, Yamashita T, Kanazawa K. Flavones and flavonols at dietary levels inhibit a transformation of aryl hydrocarbon receptor induced by dioxin. FEBS Lett 2000;476(3):213–7. doi:10.1016/s0014-5793(00)01730-0. [Google Scholar] [PubMed] [CrossRef]
46. Tan YQ, Chiu-Leung LC, Lin SM, Leung LK. The citrus flavonone hesperetin attenuates the nuclear translocation of aryl hydrocarbon receptor. Comp Biochem Physiol C Toxicol Pharmacol 2018;210(3):57–64. doi:10.1016/j.cbpc.2018.05.007. [Google Scholar] [PubMed] [CrossRef]
47. Dunlap TL, Howell CE, Mukand N, et al. Red clover aryl hydrocarbon receptor (AhR) and estrogen receptor (ER) agonists enhance genotoxic estrogen metabolism. Chem Res Toxicol 2017;30(11):2084–92. doi:10.1021/acs.chemrestox.7b00237. [Google Scholar] [PubMed] [CrossRef]
48. Bialesova L, Novotna A, Macejova D, Brtko J, Dvorak Z. Agonistic effect of selected isoflavones on arylhydrocarbon receptor in a novel AZ-AhR transgenic gene reporter human cell line. Gen Physiol Biophys 2015;34(3):331–4. doi:10.4149/gpb_2015008. [Google Scholar] [PubMed] [CrossRef]
49. Karasová M, Procházková J, Tylichová Z, et al. Inhibition of aryl hydrocarbon receptor (AhR) expression disrupts cell proliferation and alters energy metabolism and fatty acid synthesis in colon cancer cells. Cancers 2022;14(17):4245. doi:10.3390/cancers14174245. [Google Scholar] [PubMed] [CrossRef]
50. Park YK, Fukuda I, Ashida H, et al. Suppression of dioxin mediated aryl hydrocarbon receptor transformation by ethanolic extracts of propolis. Biosci Biotechnol Biochem 2004;68(4):935–8. doi:10.1271/bbb.68.935. [Google Scholar] [PubMed] [CrossRef]
51. Koper JEB, Loonen LMP, Wells JM, Troise AD, Capuano E, Fogliano V. Polyphenols and tryptophan metabolites activate the aryl hydrocarbon receptor in an in vitro model of colonic fermentation. Mol Nutr Food Res 2019;63(3):1800722. doi:10.1002/mnfr.201800722. [Google Scholar] [PubMed] [CrossRef]
52. Lakshmi A, Subramanian S. Chemotherapeutic effect of tangeretin, a polymethoxylated flavone studied in 7, 12-dimethylbenz(a)anthracene induced mammary carcinoma in experimental rats. Biochimie 2014;99:96–109. doi:10.1016/j.biochi.2013.11.017. [Google Scholar] [PubMed] [CrossRef]
53. Alzahrani AM, Rajendran P. Pinocembrin attenuates benzo(a)pyrene-induced CYP1A1 expression through multiple pathways: an in vitro and in vivo study. J Biochem Mol Toxicol 2021;35(4):e22695. doi:10.1002/jbt.22695. [Google Scholar] [PubMed] [CrossRef]
54. Wang L, Xue J, Wei F, Zheng G, Cheng M, Liu S. Chemopreventive effect of galangin against benzo(a)pyrene-induced stomach tumorigenesis through modulating aryl hydrocarbon receptor in Swiss albino mice. Hum Exp Toxicol 2021;40(9):1434–4. doi:10.1177/0960327121997979. [Google Scholar] [PubMed] [CrossRef]
55. Alqarni AM, Niwasabutra K, Sahlan M, et al. Propolis exerts an anti-inflammatory effect on PMA-differentiated THP-1 cells via inhibition of purine nucleoside phosphorylase. Metabolites 2019;9(4):75. doi:10.3390/metabo9040075. [Google Scholar] [PubMed] [CrossRef]
56. Alvarenga L, Cardozo LFMF, Borges NA, et al. To bee or not to bee? The bee extract propolis as a bioactive compound in the burden of lifestyle diseases. Nutrition 2021;83:111094. doi:10.1016/j.nut.2020.111094. [Google Scholar] [PubMed] [CrossRef]
57. Oršolić N. Allergic inflammation: effect of propolis and its flavonoids. Molecules 2022;27(19):6694. doi:10.3390/molecules27196694. [Google Scholar] [PubMed] [CrossRef]
58. Zhao L, Yao L, Chen R, et al. Pinostrobin from plants and propolis against human coronavirus HCoV-OC43 by modulating host AHR/CYP1A1 pathway and lipid metabolism. Antivir Res 2023;212(1):105570. doi:10.1016/j.antiviral.2023.105570. [Google Scholar] [PubMed] [CrossRef]
59. Ye M, Xu M, Ding M, et al. Gender associated effects of the ethanolic extracts of Chinese propolis on the hepatic transcriptome in ethanol-treated mice. Iran J Basic Med Sci 2019;22(10):1211–7. doi:10.22038/ijbms.2019.37348.8886. [Google Scholar] [PubMed] [CrossRef]
60. Kalthoff S, Strassburg CP. Contribution of human UDP-glucuronosyltransferases to the antioxidant effects of propolis, artichoke and silymarin. Phytomedicine 2019;56:35–9. doi:10.1016/j.phymed.2018.08.013. [Google Scholar] [PubMed] [CrossRef]
61. Mafra D, Borges N, Alvarenga L, et al. Dietary components that may influence the disturbed gut microbiota in chronic kidney disease. Nutrients 2019;11(3):496. doi:10.3390/nu11030496. [Google Scholar] [PubMed] [CrossRef]
62. Xue M, Liu Y, Xu H, et al. Propolis modulates the gut microbiota and improves the intestinal mucosal barrier function in diabetic rats. Biomed Pharmacother 2019;118(3):109393. doi:10.1016/j.biopha.2019.109393. [Google Scholar] [PubMed] [CrossRef]
63. Postler TS, Ghosh S. Understanding the holobiont: how microbial metabolites affect human health and shape the immune system. Cell Metab 2017;26(1):110–30. doi:10.1016/j.cmet.2017.05.008. [Google Scholar] [PubMed] [CrossRef]
64. Jensen BA, Leeman RJ, Schlezinger JJ, Sherr DH. Aryl hydrocarbon receptor (AhR) agonists suppress interleukin-6 expression by bone marrow stromal cells: an immunotoxicology study. Environ Health 2003;2(1):16. doi:10.1186/1476-069X-2-16. [Google Scholar] [PubMed] [CrossRef]
65. Plé C, Fan Y, Ait Yahia S, et al. Polycyclic aromatic hydrocarbons reciprocally regulate IL-22 and IL-17 cytokines in peripheral blood mononuclear cells from both healthy and asthmatic subjects. PLoS One 2015;10(4):e0122372. doi:10.1371/journal.pone.0122372. [Google Scholar] [PubMed] [CrossRef]
66. Jacob A, Tomkiewicz-Raulet C, Jamet C, et al. Aryl hydrocarbon receptor upregulates IL-1β expression in hCMEC/D3 human cerebral microvascular endothelial cells after TCDD exposure. Toxicology in Vitro 2017;41:200–4. doi:10.1016/j.tiv.2017.03.001. [Google Scholar] [PubMed] [CrossRef]
67. Zhu J, Luo L, Tian L, et al. Aryl hydrocarbon receptor promotes IL-10 expression in inflammatory macrophages through src-STAT3 signaling pathway. Front Immunol 2018;9:2033. doi:10.3389/fimmu.2018.02033. [Google Scholar] [PubMed] [CrossRef]
68. Ishihara Y, Kado SY, Bein KJ, et al. Aryl hydrocarbon receptor signaling synergizes with TLR/NF-κB-signaling for induction of IL-22 through canonical and non-canonical AhR pathways. Front Toxicol 2022;3:787360. doi:10.3389/ftox.2021.787360. [Google Scholar] [PubMed] [CrossRef]
69. Wu Z, Zhu A, Takayama F, et al. Brazilian green propolis suppresses the hypoxia-induced neuroinflammatory responses by inhibiting NF-κB activation in microglia. Oxid Med Cell Longev 2013;2013:906726. doi:10.1155/2013/906726. [Google Scholar] [PubMed] [CrossRef]
70. Wang K, Ping S, Huang S, et al. Molecular mechanisms underlying the in vitro anti-inflammatory effects of a flavonoid-rich ethanol extract from Chinese propolis (poplar type). Evid Based Complement Altern Med 2013;2013(1):127672. doi:10.1155/2013/127672. [Google Scholar] [PubMed] [CrossRef]
71. Alanazi S, Alenzi N, Fearnley J, Harnett W, Watson DG. Temperate propolis has anti-inflammatory effects and is a potent inhibitor of nitric oxide formation in macrophages. Metabolites 2020;10(10):413. doi:10.3390/metabo10100413. [Google Scholar] [PubMed] [CrossRef]
72. Gholami A, Dinarvand N, Hariri M. Propolis supplementation can reduce serum level of interleukin-6, C-reactive protein, and tumor necrosis factor-α: an updated systematic review and dose-response meta-analysis on randomized clinical trials. J Health Popul Nutr 2024;43(1):119. doi:10.1186/s41043-024-00600-9. [Google Scholar] [PubMed] [CrossRef]
73. Iwashima T, Takemura Y, Kishimoto Y, Ono C, Watanabe A, Iida K. Natural antagonistic flavones for AhR inhibit indoxyl sulfate-induced inflammatory gene expression in vitro and renal pathological damages in vivo. Food Nutr Res 2024;68:10032. doi:10.29219/fnr.v68.10032. [Google Scholar] [PubMed] [CrossRef]
74. Wei Q, Jiang H, Zeng J, et al. Quercetin protected the gut barrier in ulcerative colitis by activating aryl hydrocarbon receptor. Phytomedicine 2025;140(1):156633. doi:10.1016/j.phymed.2025.156633. [Google Scholar] [PubMed] [CrossRef]
75. Michalski J, Deinzer A, Stich L, Zinser E, Steinkasserer A, Knippertz I. Quercetin induces an immunoregulatory phenotype in maturing human dendritic cells. Immunobiology 2020;225(4):151929. doi:10.1016/j.imbio.2020.151929. [Google Scholar] [PubMed] [CrossRef]
76. Iizuka T, Zuberi A, Wei H, et al. Therapeutic targeting of the tryptophan-kynurenine-aryl hydrocarbon receptor pathway with apigenin in MED12-mutant leiomyoma cells. Cancer Gene Ther 2025;32(4):393–402. doi:10.1038/s41417-025-00881-0. [Google Scholar] [PubMed] [CrossRef]
77. Kim SH, Quoc QL, Park HS, Shin YS. The effect of apigenin, an aryl hydrocarbon receptor antagonist, in Phthalate-Exacerbated eosinophilic asthma model. J Cell Mol Med 2023;27(13):1900–10. doi:10.1111/jcmm.17804. [Google Scholar] [PubMed] [CrossRef]
78. Curran CS, Kopp JB. Aryl hydrocarbon receptor mechanisms affecting chronic kidney disease. Front Pharmacol 2022;13:782199. doi:10.3389/fphar.2022.782199. [Google Scholar] [PubMed] [CrossRef]
79. Lotfollahzadeh S, Vazirani A, Sellinger IE, et al. Aryl hydrocarbon receptor pathway augments peritoneal fibrosis in a murine CKD model exposed to peritoneal dialysate. Kidney360 2024;5(9):1238–50. doi:10.34067/KID.0000000000000516. [Google Scholar] [PubMed] [CrossRef]
80. Nguyen NT, Nakahama T, Kishimoto T. Aryl hydrocarbon receptor and experimental autoimmune arthritis. Semin Immunopathol 2013;35(6):637–44. doi:10.1007/s00281-013-0392-6. [Google Scholar] [PubMed] [CrossRef]
81. Xi X, Ye Q, Fan D, et al. Polycyclic aromatic hydrocarbons affect rheumatoid arthritis pathogenesis via aryl hydrocarbon receptor. Front Immunol 2022;13:797815. doi:10.3389/fimmu.2022.797815. [Google Scholar] [PubMed] [CrossRef]
82. Nehmar R, Fauconnier L, Alves-Filho J, et al. Aryl hydrocarbon receptor (Ahr)-dependent Il-22 expression by type 3 innate lymphoid cells control of acute joint inflammation. J Cell Mol Med 2021;25(10):4721–31. doi:10.1111/jcmm.16433. [Google Scholar] [PubMed] [CrossRef]
83. Zhang F, Lin M, Jiang Y, Zhou F. The role of IL-33 in immunotherapy for breast cancer: targets and signalling pathways. Eur Cytokine Netw 2025;36(1):1–5. doi:10.1684/ecn.2025.0500. [Google Scholar] [PubMed] [CrossRef]
84. Gryka-Marton M, Grabowska A, Szukiewicz D. Effect of proinflammatory cytokines on blood-brain barrier integrity. Eur Cytokine Netw 2024;35(3):38–47. doi:10.1684/ecn.2024.0498. [Google Scholar] [PubMed] [CrossRef]
85. Okamoto Y, Tanaka M, Fukui T, Masuzawa T. Brazilian propolis inhibits the differentiation of Th17 cells by inhibition of interleukin-6-induced phosphorylation of signal transducer and activator of transcription 3. Immunopharmacol Immunotoxicol 2012;34(5):803–9. doi:10.3109/08923973.2012.657304. [Google Scholar] [PubMed] [CrossRef]
86. Modipane N, Mbambara S, Serite T, Sathekge M, Kgatle M. Classification and regulatory interactions of key transcription factors in COVID-19. Front Cell Infect Microbiol 2025;15:1645333. doi:10.3389/fcimb.2025.1645333. [Google Scholar] [PubMed] [CrossRef]
87. Aksakal S, Gorgun S. The protective role of IL-17 and IL-22 in COVID-19 infection. Iran J Immunol 2024;21(3):225–33. doi:10.22034/iji.2024.100909.2721. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF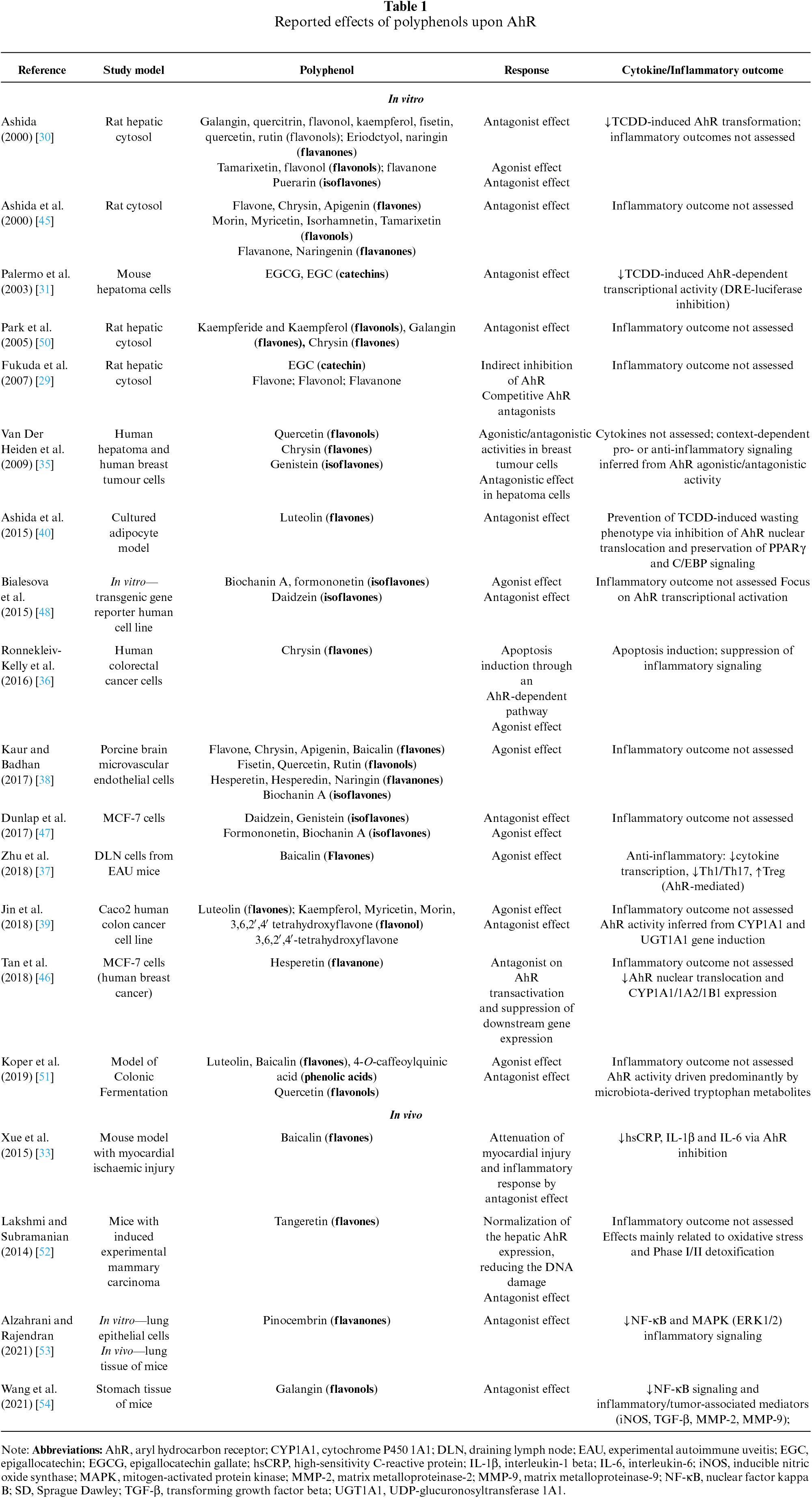
 Downloads
Downloads
 Citation Tools
Citation Tools