 Open Access
Open Access
REVIEW
Cellular and Molecular Insights into the Pathophysiology of Obesity-Related Asthma
1 Department of Pediatric Laboratory, Affiliated Children’s Hospital of Jiangnan University, Wuxi Children’s Hospital, Wuxi Key Laboratory of Genetic and Metabolic Diseases in Children, Wuxi, 214023, China
2 State Key Laboratory of Food Science and Resources, School of Food Science and Technology, Jiangnan University, Wuxi, 214122, China
3 Department of Pediatrics, Affiliated Children’s Hospital of Jiangnan University, Wuxi Children’s Hospital, Wuxi, 214023, China
* Corresponding Authors: Jiahui Zhang. Email: ; Zhiwei Yu. Email:
BIOCELL 2026, 50(3), 5 https://doi.org/10.32604/biocell.2025.073989
Received 29 September 2025; Accepted 19 November 2025; Issue published 23 March 2026
Abstract
Obesity-related asthma is a distinct clinical phenotype, characterized by severe respiratory symptoms, reduced responsiveness to conventional glucocorticoid therapy, and a significantly increase in disease burden. With the rising global prevalence of obesity, the number of individuals affected by obesity-related asthma is steadily growing, presenting a pressing public health issue. The pathogenesis of obesity-related asthma is multifactorial, involving a complex interplay of metabolic and immune pathways. Key mechanisms include dysregulated T-cell differentiation, pro-inflammatory macrophage polarization, oxidative stress, and altered cytokines and adipokines secretion, all contributing to airway inflammation and remodeling. Additionally, metabolic factors, such as adiposity and adipokine imbalance, further complicated disease progression. A major clinical challenge is developing targeted therapies to address the substantial heterogeneity in this patient population. Current treatment approaches, largely focused on corticosteroids, often fail to achieve satisfactory outcomes, emphasizing the need for novel, tailored therapies that target the specific pathophysiological features of obesity-related asthma. This review systematically explores the cellular and molecular mechanisms driving obesity-related asthma, focusing on how obesity-associated factors such as adipokines and airway remodeling influence disease progression. The review also evaluates emerging therapeutic interventions and highlights the ongoing challenges in clinical diagnosis and management. By synthesizing recent research, this study aims to provide insights into potential strategies for improving treatment and clinical outcomes for patients with obesity-related asthma.Keywords
Asthma is a heterogeneous respiratory disorder characterized by chronic airway inflammation, airway hyperresponsiveness (AHR), and reversible airflow limitation, predominantly affecting the lower respiratory tract. It presents clinically with recurrent episodes of wheezing, dyspnea, chest tightness, and cough, often exacerbated at night or in the early morning, triggered by allergens, respiratory infections, cold air, exercise, and other environmental factors [1,2].
Obesity has been firmly established as an independent risk factor for asthma development [3]. Consistent with global epidemiological trends, asthma prevalence has risen in parallel with the obesity epidemic, and accumulating evidence highlights a strong association between the two conditions [4–7]. In a large cohort of 60,639 individuals aged 25–74 years, 30.8% of incident asthma cases in women and 19.2% in men were attributable to overweight and obesity [8]. The rising prevalence of obesity globally has paralleled the increase in asthma cases, and childhood obesity in particular has become a significant concern. According to the World Health Organization, nearly 35 million children under five years of age were overweight or obese in 2024, with over 390 million school-aged children affected in 2020. This alarming trend highlights the growing challenge of obesity-related asthma. This distinct and treatment-refractory phenotype is characterized by poor disease control, reduced quality of life, and an increased risk of acute exacerbations. Despite its increasing clinical recognition, targeted therapeutic strategies for this phenotype remain limited [9].
Obesity contributes to both the initiation and progression of asthma through several interrelated mechanisms, primarily involving mechanical, inflammatory, and metabolic pathways. First, mechanical constraints from excessive thoracic adiposity increase chest wall load and airway resistance, thereby reducing lung volumes and impairing pulmonary function [10]. Prolonged exposure to mechanical stress can independently trigger airway remodeling. In vitro studies show that mechanical compression induces goblet cell hyperplasia, enhances extracellular matrix deposition, and promotes release of mediators such as endothelin-1 and chitinase-3-like protein 1, which drive airway smooth muscle proliferation [11]. Clinically, the decline in the forced expiratory volume in the first second (FEV1)/forced vital capacity (FVC) ratio is a critical marker of airflow obstruction, and has been closely related to asthma incidence, with studies showing a strong association between obesity and airflow limitation in both children and adults [12,13].
Second, immuno-inflammatory pathways play a central role in obesity-related asthma. Visceral adipose tissue in obesity individuals becomes enriched with macrophages that polarize toward the pro-inflammatory M1 phenotype [14–16]. These macrophages release cytokines such as Interleukin-1 Beta (IL-1β), Tumor Necrosis Factor Alpha (TNF-α), and Interleukin (IL)-8, which disrupt the pulmonary immune microenvironment, promote neutrophil chemotaxis, and skew T-cell differentiation, driving Th1/Th2/Th17 imbalance and amplifying airway inflammation [17,18].
Third, obesity induces metabolic dysfunction characterized by altered adipokine secretion and oxidative stress, further exacerbating airway pathology [19]. Elevated leptin levels correlate with asthma onset and severity [20,21], while reduced adiponectin weakens its anti-inflammatory effects [22,23]. Additionally, resistin overproduction promotes insulin resistance and systemic inflammation, further aggravating airway inflammation in asthma [24,25]. In parallel, excessive generation of reactive oxygen species impaired antioxidant defenses, promoted lipid peroxidation, and directly injured the airway epithelium, thereby driving airway remodeling and hyperresponsiveness [26].
This review aims to systematically investigate the multifactorial mechanisms through which obesity contributes to asthma, with a particular focus on mechanical constraints, immune-inflammatory pathways, and endocrine-metabolic dysregulation. By synthesizing recent advances, this study seeks to clarify the intricate interactions between metabolic and immune factors that exacerbate asthma pathogenesis. Additionally, the review evaluates the clinical challenges and diagnostic complexities associated with obesity-related asthma, emphasizing the need for more precise and tailored therapeutic strategies, as well as improved diagnostic tools, to better manage this increasingly prevalent condition. By providing an integrated perspective on its pathophysiology, this review aims to inform the development of emerging therapeutic strategies, potentially enabling precision-based interventions to improve outcomes for affected individuals.
2 From Immune Cells and Cytokines to Molecular Pathways and Tissue Crosstalk
2.1.1 Macrophage Polarization and Its Implications in the Pathogenesis of Obesity-Related Asthma
Macrophages are abundant immune cells in the lungs and play a central role in maintaining tissue homeostasis [27]. Increasing evidence highlights their significant role in asthma pathogenesis. Data from the U-BIOPRED cohort, which analyzed sputum samples from 104 asthmatic patients and 16 healthy controls, revealed that macrophage counts were significantly lower in patients with severe asthma compared to mild-to-moderate cases and healthy controls [28]. Similarly, in obese asthmatic mice, reduced macrophage proportions were observed in bronchoalveolar lavage fluid, alongside more severe airway inflammation [29]. These findings suggest that asthma progression may be linked to macrophage immunodeficiency, although the exact mechanisms are yet to be fully elucidated. Macrophages exhibit two principal activation states: M1 (classically activated) and M2 (alternatively activated). In obesity-related asthma patients, the M1/M2 ratio in visceral adipose tissue is elevated, accompanied by altered macrophage morphology and function [30,31]. M1 polarization drives the release of pro-inflammatory cytokines, contributing to systemic low-grade inflammation and metabolic dysfunction. Studies show that obesity-related asthma patients display increased circulating IL-6 levels, further supporting the role of macrophage-driven inflammation in asthma [32,33]. In addition to systemic effects, M1 polarization profoundly influences airway inflammation. The proportion of M1 macrophages in sputum correlates with neutrophil counts, highlighting the close relationship between macrophage polarization and airway neutrophilia. Furthermore, M1 macrophages promote Th1 and Th17 differentiation by secreting pro-inflammatory cytokines such as IL-12 and IL-23, thus amplifying neutrophil-driven inflammation [34].
2.1.2 Immunological Dysregulation of T Cells and B Cells in the Pathogenesis of Obesity-Related Asthma
CD4+ T cells are central to the immunopathology of asthma. Although asthma was once regarded primarily as an allergic disease mediated by Th2 cells, this view is now recognized as overly simplistic. Asthma pathogenesis is complex and can be broadly classified into allergic and non-allergic phenotypes, or alternatively into “type 2-high” and “type 2-low” asthma, based on underlying immune mechanisms [35]. The former is driven predominantly by type 2 inflammation, often triggered by allergen exposure, and involves not only Th2 cells but also other immune cells that release type 2 cytokines. In contrast, type 2-low asthma is mediated by non-type 2 inflammatory pathways, the mechanisms of which remain incompletely understood but seem to be influenced by factors such as obesity and environmental pollution [2]. The prevalence of these phenotypes also varies with age, with allergic asthma most common in childhood, whereas non-allergic asthma tends to increase with aging [36]. Obesity-related asthma is considered a prototypical type 2-low phenotype, marked by enhanced Th1/Th17 polarization of CD4+ T cells [37–39]. In this context, CD4+ T-cell subsets undergo significant reprogramming, which includes the upregulation of cell division cycle 42 expression, enhancement of Th1-driven inflammatory responses, and downregulation of the glucocorticoid receptor gene (NR3C1), all contributing to the distinct obesity-related asthma phenotype [40,41]. Clinical studies further support this: randomized controlled trials in children show that dexamethasone-induced FK506 binding protein (FKBP) 51 expression in CD4+ T cells strongly correlates with poorer asthma control in obese patients [42]. Similarly, transcriptomic analyses of obesity-related asthma have revealed upregulation of several molecular signatures, such as IL-7R, IL-32, poly (ADP-ribose) polymerase-1, FKBP5, elevated IL-6 levels, and activation of the Rho signaling pathway [43]. At the cellular level, peripheral CD4+ T-cell subsets in obese children with asthma show a skewed distribution, characterized by an increased proportion of memory T cells and a decreased proportion of naïve T cells. Notably, the Rho-GTPase signaling pathway is enriched in Th cells from this population [44]. The role of CD8+ T cells in asthma is less well understood, though emerging evidence suggests that certain CD8+ T-cell subsets may contribute to airway pathology by mediating tissue inflammation and recurrent airway damage via effector molecules such as granzyme K [45].
B cells play a pivotal role in allergic asthma by producing IgE antibodies upon allergens exposure [46]. In addition to IgE production, B cells contribute to immune regulation through IL-10 secretion, which has dual effects: it suppresses activation of certain immune cells and stimulates lung epithelial cells to produce the chemokine C-C motif chemokine ligand 20 (CCL-20). CCL-20 facilitates dendritic cells recruitment to the lungs, thereby amplifying Th2-mediated immune responses and worsening airway hyperresponsiveness and mucus secretion [47]. In obesity, however, the proportion of regulatory B cells is reduced, resulting in low IL-10 secretion, impaired immune tolerance, and a skewing toward Th1/Th17-driven inflammation [48,49]. Clinical evidence supports this: a cohort of 168 children with asthma showed that those in the obese/overweight subgroup had significantly lower serum IL-10 levels compared to children with asthma alone (8.69 ± 2.76 pg/mL vs. 15.32 ± 6.28 pg/mL), highlighting the role of obesity in aggravating immune dysregulation in asthma [50].
2.1.3 The Role of Neutrophils in Obesity-Related Asthma
Excessive lipid accumulation in adipose tissue induces adipocyte stress and dysfunction, which trigger inflammatory responses. Neutrophils, the most abundant white blood cells in circulation, are the first immune cells to infiltrate adipose tissue, where they further activate the immune system by releasing inflammatory mediators and recruiting macrophages and other leukocytes [51]. A meta-analysis of 4475 participants found that both absolute neutrophil count and the neutrophil-to-lymphocyte ratio were elevated in obese individuals compared to normal-weight controls. Notably, the absolute neutrophil count showed greater divergence from controls, lower heterogeneity among obese subjects, and a strong positive correlation with body mass index (BMI) [52]. Beyond systemic circulation, obesity is also linked with gut microbiota dysbiosis and impaired intestinal barrier integrity, allowing translocation of live bacteria, bacterial DNA, lipopolysaccharides, and pro-inflammatory cytokines into the bloodstream and adipose tissue. These changes contribute to metabolic inflammation, further modulate neutrophil activation, migration, and mediator release. Clinically, adult-onset asthma (first onset >15 years) with non-type 2 inflammatory features, such as obesity and elevated blood neutrophils, has been strongly associated with poor asthma control and increased requirements for inhaled corticosteroids (ICS) [53]. Consistent with these findings, obesity-related asthma patients exhibit a ~5% higher proportion of sputum neutrophils compared to non-obese counterparts [54]. Animal models further corroborate that obesity exacerbates asthma development and progression through non-Th2 inflammatory pathways characterized by neutrophil infiltration [29,39]. A schematic diagram summarizing the proposed mechanism is presented in Fig. 1.
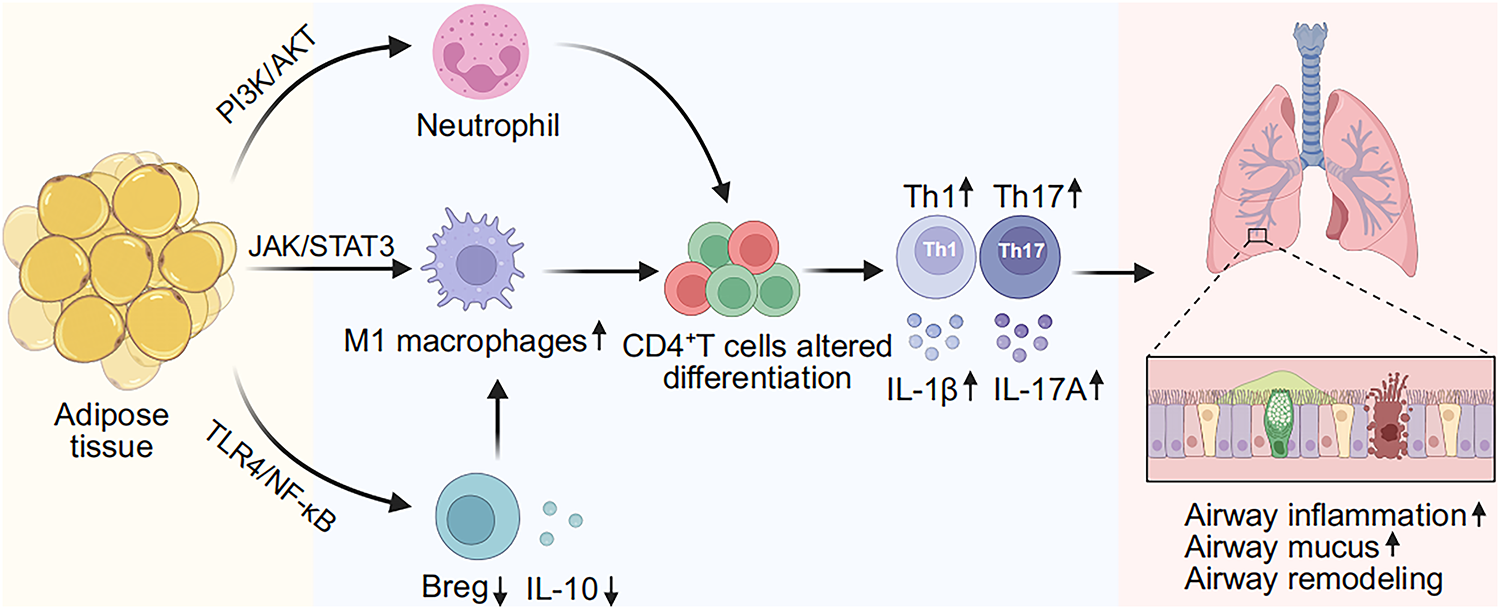
Figure 1: Adipose tissue-mediated immune dysregulation in obesity-related asthma. Adipose tissue–driven immune dysregulation in obesity-related asthma, characterized by increased M1 macrophages, enhanced neutrophil infiltration, and reduced regulatory B cells/IL-10, contributes to impaired CD4+ T cells differentiation and the worsening of airway inflammation, mucus hypersecretion, and airway remodeling. IL-10, interleukin-10. IL-1β, interleukin-1β. IL-17A, interleukin-17A. Breg, regulatory B cell. Th1, T helper 1 cell. Th17, T helper 17 cell, PI3K, phosphoinositide 3-kinase. AKT, protein kinase B. JAK, Janus kinase. STAT3, signal transducer and activator of transcription 3. TLR4, toll-like receptor 4. NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells. Figures were created with biorender.com
2.2 Cytokines and Signaling Pathways
In addition to immune cell reprogramming, cytokine networks and signaling pathways further amplify obesity-related airway inflammation. The NF-κB signaling pathway is regulated by the Rel protein family, which includes c-Rel, NF-κB1 (p105/p50), NF-κB2 (p100/p52), RelA (p65), and RelB, forming transcriptionally active complexes through dimerization [55]. Under basal conditions, NF-κB is inactive in the cytoplasm, bound to the inhibitor of kappa B (IκB). Upon cellular stimulation, IκB undergoes phosphorylation and degradation, releasing NF-κB to translocate to the nucleus and initiate transcription of downstream target genes [56]. Notably, selective activation of NF-κB alone has been shown to replicate key pathological features of allergic airway disease, such as AHR and smooth muscle thickening [57]. In obesity-related asthma, oxidative stress and reactive oxygen species-mediated cellular injury activate p38 mitogen-activated protein kinase (MAPK) and c-Jun N-terminal kinase (JNK), driving NF-κB overactivation and persistent production of inflammatory mediators such as IL-1β, TNF-α, and IL-6 [58–60]. These cytokines amplify the inflammatory cascade by promoting M1 macrophage polarization and Th17 differentiation in lung tissues [61,62]. As a central transcription factor in the TLR4 signaling pathway, NF-κB not only enhances pro-inflammatory mediator production but also establishes a positive feedback loop with NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome-mediated pyroptosis [63]. Experimental studies suggest that blocking this axis confers therapeutic benefit; for example, Involucrasin B has been shown to alleviate airway inflammation in obesity-related asthma models by inhibiting the TLR4/NF-κB/NLRP3 pathway [64]. Clinical evidence supports these findings, demonstrating that activation of the TLR4/MyD88/NF-κB pathway facilitates NLRP3 inflammasome assembly, thereby exacerbating neutrophil infiltration and pyroptosis-associated airway injury in asthma patients [65]. As a transcriptional hub in the TLR4 signaling axis, NF-κB also amplifies inflammation through crosstalk with the NLRP3 inflammasome, contributing to neutrophil infiltration and pyroptosis-associated airway injury.
Building on this, the hallmark of obesity-related asthma is a chronic, low-grade inflammatory state sustained by abnormal adipose tissue accumulation and overactivation of cytokines, particularly IL-6 and TNF-α [66,67]. This process involves three interconnected mechanisms. First, hypertrophic adipose tissue releases excess free fatty acids (FFAs), activating the TLR4/NF-κB signaling axis, inducing M1 macrophage polarization, and stimulating cytokine secretion [68,69]. Second, an elevated leptin-to-adiponectin ratio enhances macrophage activity via the JAK-STAT3-PI3K/AKT pathway, promotes C-X-C motif chemokine ligand 2 (CXCL2)-driven neutrophil recruitment, and diminishes the anti-inflammatory role of adiponectin [29,70,71].
Third, hypoxic stress and the induction of hypoxia-inducible factor 1-alpha amplify cytokine release, futher intensifying systemic inflammation. These pro-inflammatory mediators collectively disrupt epithelial barrier integrity, increase mucus hypersecretion (MUC5AC) expression, promote type I and III collagen deposition, and accelerate airway remodeling [72,73]. Importantly, IL-6 and TNF-α synergize to promote Th17 differentiation and IL-17A release, sustaining neutrophilic inflammation and tissue injury [74–78]. Clinically, elevated serum and sputum IL-6 and TNF-α levels in obesity-related asthma patients correlate with accelerated lung function decline and glucocorticoid resistance [50,79–81]. Systemic inflammatory patterns, similar to those observed during respiratory crises at the population level, contribute to disease exacerbation and reduced therapeutic responses due to immune dysregulation. In the context of COVID-19, Nazari et al. emphasized the importance of immune-inflammatory control strategies in managing respiratory crises [82]. Their findings highlight the critical role of effective immune modulation in preventing exacerbations and improving therapeutic outcomes, offering valuable insights that could inform the management of obesity-related asthma.
2.3 Crosstalk between Adipose Tissue and Airway Inflammation
In obesity, adipocyte hypertrophy and dysfunction drive macrophage recruitment and polarization toward a pro-inflammatory state, contributing to distal airway injury [16,19]. Central to this “adipose-lung axis” is the ability of adipose-derived factors to modulate the pulmonary immune microenvironment and structural cells via systemic circulation or local paracrine signaling. This section outlines the molecular mechanisms, effector targets, and potential therapeutic strategies of adipose secretion-mediated airway inflammation (see Table 1).
3 Clinical Manifestations and Diagnostic Challenges in Obesity-Related Asthma
Obesity-related asthma is recognized as a distinct phenotype characterized by elevated BMI, severe symptoms, and unique inflammatory features, highlighting heterogeneity within the asthma spectrum [90]. Unlike classical allergic asthma, this phenotype shows weak associations with atopy and often presents as non-allergic asthma [3,91]. Symptom burden and airflow limitation correlate with obesity severity and may persist despite minimal airway inflammation.
Standard anti-inflammatory therapies, such as ICS, often provide suboptimal control, underscoring the influence of weight-related factors [53]. Pathogenesis is multifactorial: beyond airway inflammation and bronchoconstriction, excess adiposity imposes mechanical constraints that reduce lung volumes and ventilatory capacity, leading to refractory airflow obstruction poorly responsive to bronchodilators [90,92]. Inflammatory profiles also differ. Classical asthma is typically type 2–high with eosinophilia and elevated IL-4, IL-5, and IL-13 [93], whereas obesity-related asthma is commonly neutrophilic inflammation accompanied by low-grade inflammation and increased IL-6 and TNF-α [29,70]. Adipokines imbalance further promotes airway inflammation and impairs responses to conventional therapies [70]. Management therefore, requires integrated approaches. Weight reduction and metabolic control improve symptoms and airflow limitation [94], while biologics targeting IL-6 and TNF-α represent potential strategies for addressing non-T2 inflammation [9]. Overall, the distinct clinical manifestations, non-T2 predominance, and reduced responsiveness to conventional therapy emphasize the need for combined weight management and individualized anti-inflammatory interventions to optimize patient outcomes and quality of life.
The heterogeneous manifestations of obesity-related asthma make its diagnosis a significant clinical challenge. Its symptoms often overlap with other asthma phenotypes, and obesity-induced mechanical constraints and metabolic abnormalities further complicate recognition. Olejnik AE and Kuźnar-Kamińska B reported that reduced chest wall compliance, decreased lung volumes, and systemic metabolic inflammation in obesity contribute to atypical airflow limitation and worsened clinical outcomes, particularly evident in pulmonary function [95]. Reyes Noriega et al. noted that lung function tests often reveal obstruction not fully explained by airway inflammation but strongly influenced by the mechanical burden of obesity, limiting the diagnostic accuracy of conventional spirometry [96]. Another challenge is inflammatory heterogeneity: unlike classical asthma, which is typically T2-high, obesity-related asthma often presents with a systemic pro-inflammatory response. Picado et al. demonstrated that elevated IL-6 and TNF-α are closely associated with this phenotype [70], while experimental models have revealed enhanced non-T2 pathways such as IL-17, TNF-α, and transforming growth factor beta (TGF-β) with neutrophilic infiltration, but without increases in classical T2 cytokines like IL-5 and IL-13, highlighting its distinctive inflammatory profile [39]. These differences reduce the utility of conventional biomarkers and single inflammatory markers.
Diagnostic complexity is further compounded by comorbidities such as metabolic syndrome, diabetes, and hypertension, where chronic low-grade inflammation can obscure or exaggerate asthma symptoms, reducing the sensitivity of current criteria. As emphasized by Picado et al., systemic inflammation induced by obesity may mimic asthma and alter disease progression and therapeutic responses, rendering traditional diagnostic frameworks insufficient. Advances in omics offer new opportunities: Björkander et al. reported that plasma metabolite and inflammatory marker profiles in obesity-related asthma differ markedly from those in non-obese individuals, supporting the use of metabolomics for subtype identification [33]. Popović-Grle et al. highlighted that future precision diagnosis will require integration of clinical features, lung function, biomarkers, metabolomics, and emerging imaging techniques to establish personalized diagnostic standards [97]. As mechanistic insights grow, novel biomarkers and advanced imaging technologies hold strong potential to improve diagnostic accuracy and enhance precision medicine for this complex phenotype.
4 Current Clinical Management of Obesity-Related Asthma
4.1 Conventional Pharmacological Therapy
ICS remain the cornerstone of asthma management, with strong evidence supporting their ability to suppress airway inflammation, improve lung function, and reduce exacerbations [98]. However, patients with obesity-related asthma exhibit a consistently reduced response to ICS. Even at high doses, ICS provides less clinical benefit in this subgroup compared to non-obese patients [94]. This reduced efficacy is linked to the low-T2 inflammatory profile characteristic of obesity-related asthma, which is defined by reduced eosinophil counts, low fractional exhaled nitric oxide (FeNO), absence of allergic drivers, and a decreased need for maintenance oral corticosteroids (OCS) [99]. Furthermore, Li et al. emphasized that obesity-induced mechanical load and metabolic dysregulation significantly influence airway obstruction, both of which may diminish ICS effectiveness [100].
Beta2-agonists are commonly used to provide rapid relief of acute asthma symptoms [101]. However, their efficacy is reduced in obesity. Elevated plasma FFAs, a common feature in obesity, activate G protein-coupled receptors (GPCRs) such as GPCR40 and GPCR120, triggering inflammatory cascades that enhance airway hyperresponsiveness and reduce bronchial smooth muscle responsiveness to beta2-agonists [102]. Additionally, the mechanical load caused by obesity promotes structural remodeling of airway smooth muscle, further impairing its sensitivity to bronchodilators. Obesity-associated low-grade inflammation, marked by elevated IL-6 levels, may also exacerbate airway dysfunction and reduce the effectiveness of bronchodilators [81].
4.1.2 Leukotriene Receptor Antagonists (LTRAs)
LTRAs, such as montelukast, target the CysLT1 receptor and block leukotriene-mediated pathways, including airway constriction, mucus secretion, and inflammatory cell infiltration. These agents have been used to treat conditions like exercise-induced bronchoconstriction, allergic rhinitis, and aspirin-exacerbated respiratory disease [103]. In obesity, excessive leptin secretion from adipose tissue leads to elevated circulating leptin levels [29]. Experimental studies show that leptin enhances cysteinyl leukotriene production in rat alveolar macrophages [104]. Additionally, obesity is associated with increased expression of 5-lipoxygenase and its activating 5-lipoxygenase-activating protein, which boosts leukotriene synthesis, particularly leukotriene B4, within adipose tissue. This amplifies both local and systemic inflammatory responses [105]. These findings support the use of LTRAs in obesity-related asthma. Indeed, Farzan et al. reported that montelukast treatment significantly improved Asthma Control Test (ACT) scores and asthma control in patients with early-onset atopic obesity-related asthma [106]. However, while these results suggest potential benefits, further randomized controlled trials are needed to confirm the efficacy of LTRAs in this pehenotype.
4.2 Future Perspectives on Targeted Therapy
4.2.1 Bruton’s Tyrosine Kinase (BTK) Inhibitors
BTK is a key signaling molecule expressed in B cells and myeloid cells such as macrophages, mast cells, and neutrophils. It regulates cellular activation and inflammatory mediators release [107]. BTK inhibitors block signaling downstream of the B-cell receptor (BCR), Toll-like receptors (TLRs), and Fc receptors, thereby suppressing excessive immune responses. These agents show therapeutic potential in autoimmune, allergic, and inflammatory diseases, including asthma [108]. In a murine mixed-granulocytic asthma model, ibrutinib suppressed both neutrophilic and eosinophilic inflammation, reduced Th2 cytokine production, and lowered immune activation markers [109]. In obesity-related asthma, airway tissues show increased eosinophil and neutrophil infiltration, suggesting a complex mixed-granulocytic phenotype [110,111]. In a high-fat-diet-induced obese mouse model, ibrutinib attenuated NF-κB and NLRP3 inflammasome-driven inflammation, lowered fasting glucose, and improved glucose tolerance, indicating additional benefits in obesity-associated metabolic dysregulation [112]. Taken together, these findings provide a mechanistic rationale for BTK inhibitors as potential therapeutics in obesity-related asthma. However, current evidence is preliminary, and further studies and clinical trials are required [113].
Anti-IgE monoclonal antibodies, such as omalizumab, block free IgE from binding to its receptor FcεRI, preventing mast cells and basophils activation [114]. In two randomized controlled trials, omalizumab reduced asthma exacerbations by 71.9% in obese patients (BMI ≥ 30 kg/m2), with efficacy comparable to non-obese groups [115]. A prospective study also reported that 12 months of omalizumab therapy improved asthma control and, for the first time, was linked to better lung function in obese patients [116]. Similar results were observed by Pilkington et al., confirming favorable clinical responses [117]. Collectively, these findings support anti-IgE therapy as an effective treatment option for obesity-related asthma.
4.2.3 IL-5 Inhibitors and Related Biologic Therapies
IL-5 drives eosinophil differentiation and activation, making it central in type 2-high asthma. Monoclonal antibodies targeting IL-5 reduce eosinophil counts in the circulation and airways, improving lung function and reducing exacerbation [118,119]. IL-13 also contributes to airway remodeling by promoting goblet cell hyperplasia, mucus hypersecretion, smooth muscle hypertrophy, and fibrosis. Blocking IL-13 or its downstream pathways can reduce these changes and potentially improve long-term outcomes [120]. In obesity-related asthma, elevated IL-5, IL-17A, and IL-25 expression suggests multiple inflammatory cascades contribute to disease [121]. Although no dedicated trials have yet assessed the efficacy of anti-IL-5 or anti-IL-13 antibodies in obesity-related asthma, mechanistic evidence supports their therapeutic potential, especially in patients with eosinophilic or type 2 inflammatory features.
4.3 Obesity-Targeted and Lifestyle Interventions
Recent studies show that patients with obesity-related asthma often present with T2-low or neutrophil-dominant inflammatory phenotypes. These are characterized by elevated levels of IL-6, IL-17, and TNF-α, along with excessive NLRP3 inflammasome activation. This ultimately drives AHR and corticosteroid resistance [70,122,123]. Weight reduction therapy has been shown to ameliorate this inflammatory state through multiple mechanisms. Weight loss significantly lowers systemic inflammatory markers, such as C-reactive protein, and reduces airway inflammation [94]. Long-term follow-up after bariatric surgery consistently demonstrates sustained asthma control improvements, with reductions in pro-inflammatory adipokines like leptin and resistin, and a decrease in adipose tissue inflammation [124]. Lifestyle interventions combining dietary modification with physical exercise have also been shown to decrease circulating IL-6 and TNF-α levels, improving asthma control and lung function in a relatively short period [125].
In addition to lifestyle changes, pharmacological interventions show promise. Glucagon-like peptide-1 receptor agonists (GLP-1RAs) provide dual benefits by promoting weight reduction and exerting independent anti-inflammatory effects. In a murine model of obesity-related asthma, liraglutide significantly reduced AHR and eosinophilic inflammation, while also suppressing NLRP3 inflammasome activation and IL-1β expression in bronchial tissues. It also led to marked reductions in IL-4, IL-5, and IL-33 levels in bronchoalveolar lavage fluid [126]. Similarly, Kaplan et al. found that patients receiving GLP-1RAs had fewer asthma exacerbations. The abundant expression of GLP-1 receptors in lung tissues supports the anti-inflammatory activity of GLP-1RAs [127]. Overall, weight reduction improves chest wall compliance through mechanical unloading and induces immunometabolic reprogramming, significantly alleviating airway inflammation in obesity-related asthma. This reinforces the role of weight management as a cornerstone of comprehensive disease management.
5 Conflicting Evidence and Unresolved Mechanistic Controversies
5.1 Inconsistent Associations between BMI and Asthma Phenotypes across Ethnic Groups
The relationship between BMI and asthma risk varies significantly across populations, both in the strength and shape of the association (linear, U-shaped, or threshold effects). For example, data from China and the United States show distinct patterns. In elderly populations in China (CHARLS), a U-shaped relationship is observed, where both low and high BMI correlate with increased asthma risk. In contrast, the United States (NHANES) shows a nonlinear “threshold-after-increase” relationship in the same age group, suggesting a different optimal BMI for asthma risk across populations [128]. In pediatric populations, analysis of NHANES data from 1999 to 2020 reveals a nonlinear positive association between BMI and asthma. However, stratified analyses demonstrate variations in this relationship based on racial and socioeconomic subgroups, indicating that population background may influence the strength of this association [129]. A real-world study from the Singapore Severe Asthma Registry, which includes multiple ethnic groups (Chinese, Malay, and Indian), found higher obesity-related comorbidities among non-Chinese patients, with poorer asthma control. Phenotypic clustering analysis revealed ethnic differences, indicating that the “obesity-related asthma” burden is ethnicity-specific [130]. Furthermore, BMI alone may obsure population differences. Studies based on NHANES data and bidirectional Mendelian randomization suggest a J-shaped relationship between fat distribution and adult asthma risk, with causal effects showing significant variance between populations [6]. Obesity-related asthma involves various immune and metabolic pathways and is associated with a non-eosinophilic inflammation phenotype. This phenotype is particularly pronounced in certain populations, women, and individuals with metabolic comorbidities. These differences contribute to the inconsistencies in the relationship between BMI and asthma phenotypes across populations [9].
5.2 Divergent Findings Regarding IL-17A’s Role in Obesity-Related Asthma
Recent studies on IL-17A’s role in obesity-related asthma have produced inconsistent findings. Al Heialy et al. demonstrated that IL-17A stimulation in adipocytes from obesity-related asthma patients led to an imbalance in the GR-α/GR-β ratio, suggesting that IL-17A may contribute to steroid resistance by regulating glucocorticoid receptor expression [131]. Conversely, Leija-Martínez et al. found reduced promoter methylation of the IL-17A and TNFA genes in obesity-related asthma, indicating epigenetic regulation of IL-17A expression [132]. Additionally, Buabraig et al. proposed that IL-17F, rather than IL-17A, may play a more prominent role in obesity-related asthma. The differential roles of IL-17A and IL-17F could vary across individuals, underlining the need for further research into their specific mechanisms in different populations [133]. In conclusion, IL-17A’s role in obesity-related asthma remains complex, influenced by IL-17F, epigenetic factors, and individual variability. Future research should aim to clarify IL-17A’s specific role and its mechanisms, to develop more precise and targeted therapies.
5.3 Mixed Clinical Efficacy of Biologics in Non-T2 Asthma
Recent clinical studies have shown considerable heterogeneity in the efficacy of biologics for non-T2 obesity-related asthma. A retrospective study found that omalizumab reduced severe asthma exacerbations by approximately 60%–70% in obese patients who experienced weight loss, though the treatment effect was more pronounced in non-obese patients. This suggests that body weight may influence biologic efficacy [134]. A review by Pilkington et al. found that biologics such as dupilumab, mepolizumab, omalizumab, and tezepelumab were effective for obesity-related asthma, especially tezepelumab, which showed therapeutic benefits in patients with low-type T2 asthma [117]. However, the efficacy of biologics for non-T2 asthma remains contentious. For instance, while azithromycin and tezepelumab are available treatment options for non-T2 asthma, their efficacy and indications require further investigation [135]. Overall, the response to biologics in non-T2 obesity-related asthma is highly variable, influenced by body weight, inflammation phenotype, and comorbidities. Future research should focus on the phenotypic characteristics of obesity-related asthma and explore personalized treatment strategies to improve biologic efficacy and safety in this population.
6 Challenges and Future Directions
6.1 Personalized Treatment Needs
Tiotiu et al. categorized obesity-related asthma into two major phenotypes: an early-onset, allergic phenotype with more severe disease, and a late-onset, non-allergic phenotype where obesity-related chest wall loading impairs lung function. Both phenotypes often respond poorly to standard therapies, highlighting the need for individualized treatment [136]. Hudler et al. proposed that BMI should be a central factor in evaluating these patients and advocated for incorporating obesity as a treatable trait in asthma management. Defining target BMI values and promoting sustained weight reduction could improve disease control and reduce the overall burden [137]. From a precision medicine perspective, identifying obesity-related asthma endotypes should rely on an integrated comprehensive assessment of clinical manifestations, pulmonary function, inflammatory biomarkers (peripheral blood neutrophil-to-eosinophil ratios), and metabolic status [138].
6.2 Emerging Pharmacological Agents and Therapeutic Strategies
In recent years, the treatment landscape for obesity-related asthma has envolved, moving beyond traditional anti-inflammatory and bronchodilator therapies toward more diversified and precise approaches. Among the most promising strategies, cell-based therapies have gained significant attention. Mesenchymal stem cells and their extracellular vesicles are increasingly recognized for their ability to modulate abnormal inflammatory responses and promote tissue repair, offering novel therapeutic opportunities [139]. Additionally, notable advances have been made in immunomodulatory and biologic therapies. Tezepelumab, a monoclonal antibody targeting the epithelial cytokine thymic stromal lymphopoietin, has shown broad anti-inflammatory effects across various asthma phenotypes in clinical trials, suggesting potential therapeutic benefits for severe asthma complicated by obesity [140]. Natural compounds and small-molecule immunomodulators, such as celastrol, γ-secretase inhibitors, and C-X-C chemokine receptor type 2 (CXCR2) antagonists, have demonstrated preclinical efficacy in reducing AHR by dampening Th17-driven responses and neutrophilic inflammation [141]. Furthermore, emerging gene therapies, along with single-cell transcriptomic analyses, reveal mechanisms in obesity-related asthma, such as CDC42 upregulation, NLRP3 inflammasome activation, and NR3C1 downregulation in CD4+ T cells. These insights highlight potential molecular targets for future precision therapies [40]. Together, these developments expand the therapeutic options and provide a framework for individualized management in obesity-related asthma.
6.3 Translational Challenges in Clinical Practice
Obesity-related asthma presents significant therapeutic challenges. Emerging evidence suggests that face multiple therapeutic barriers in clinical practice. One major barrier is the lack of phenotype-specific recommendations in current asthma management guidelines, hindering the integration of individualized treatments into routine care [142]. A substantial body of evidence also indicates that obese patients show reduced responsiveness to ICS, highlighting the limited efficacy of conventional anti-inflammatory therapies in this subgroup [143]. Beyond pharmacological challenges, obesity exacerbates airway inflammation and structural remodeling, contributing to progressive lung function decline and increased risks of exacerbations and hospitalizations, further complicating management [121]. Of particular concern, many patients with obesity-related asthma present with an atypical T2-low inflammatory phenotype, which is generally resistant to standard treatments, and no specific therapies for this endotype are available [144]. Additionally, certain phenotypic subsets, such as those with more severe symptoms, higher prevalence among women, and poorer disease control, highlight the clinical heterogeneity and the need for precision-based approaches [33]. The primary challenges in managing obesity-related asthma involve heterogeneous drug responses, obesity-driven airway remodeling, and the lack of personalized treatment strategies. Overcoming these barriers requires a “treatable traits” framework that integrates metabolic modulation, weight management, and next-generation biologics for more precise and effective interventions.
Obesity-related asthma is increasingly recognized as a distinct clinical phenotype, often characterized by non-T2 inflammation and airway remodeling, where ICS efficiency is limited. This endotype is driven by obesity-associated adipokines such as leptin, which sustain systemic inflammation, TGF-β-mediated airway remodeling, and the mechanical burden of excess weight on lung mechanics. Emerging evidence suggests that GLP-1RAs, like semaglutide, may reduce symptoms and exacerbation risk through mechanisms beyond weight reduction. Lifestyle modifications and weight-loss interventions have also shown consistent improvements in asthma control.
Future research should focus on the immunometabolic-airway axis and rigorously evaluate GLP-1Ras, alongside other innovative therapies, such as microbiome-targeted and receptor-specific treatment. Cross-disciplinary collaboration involving respiratory, endocrine, nutritional, and immunological disciplines is essential to advance precision management tailored to asthma endotypes. Looking ahead, future studies should prioritize the integration of omics technologies, such as genomics, proteomics, and metabolomics, to provide a more comprehensive understanding of the molecular mechanisms underlying obesity-related asthma. This integrated approach will facilitate the identification of novel biomarkers for more accurate diagnosis and personalized strategies.
Further exploration into the microbiome-adipose tissue crosstalk is crucial, as it may reveal how gut microbiota influences adipose tissue function and inflammation, offering new insights into the pathogenesis of obesity-related asthma. Additionally, utilizing AI-based phenotyping could improve patient stratification, identifying specific subtypes of obesity-related asthma based on clinical, genetic, and environmental factors. By applying advanced data analytics, we can refine therapeutic approaches and optimize outcomes for this heterogeneous patient population. These research directions hold the potential to significantly enhance our understanding of obesity-related asthma and pave the way for more effective, personalized therapies. A proposed future research roadmap is presented in Fig. 2.
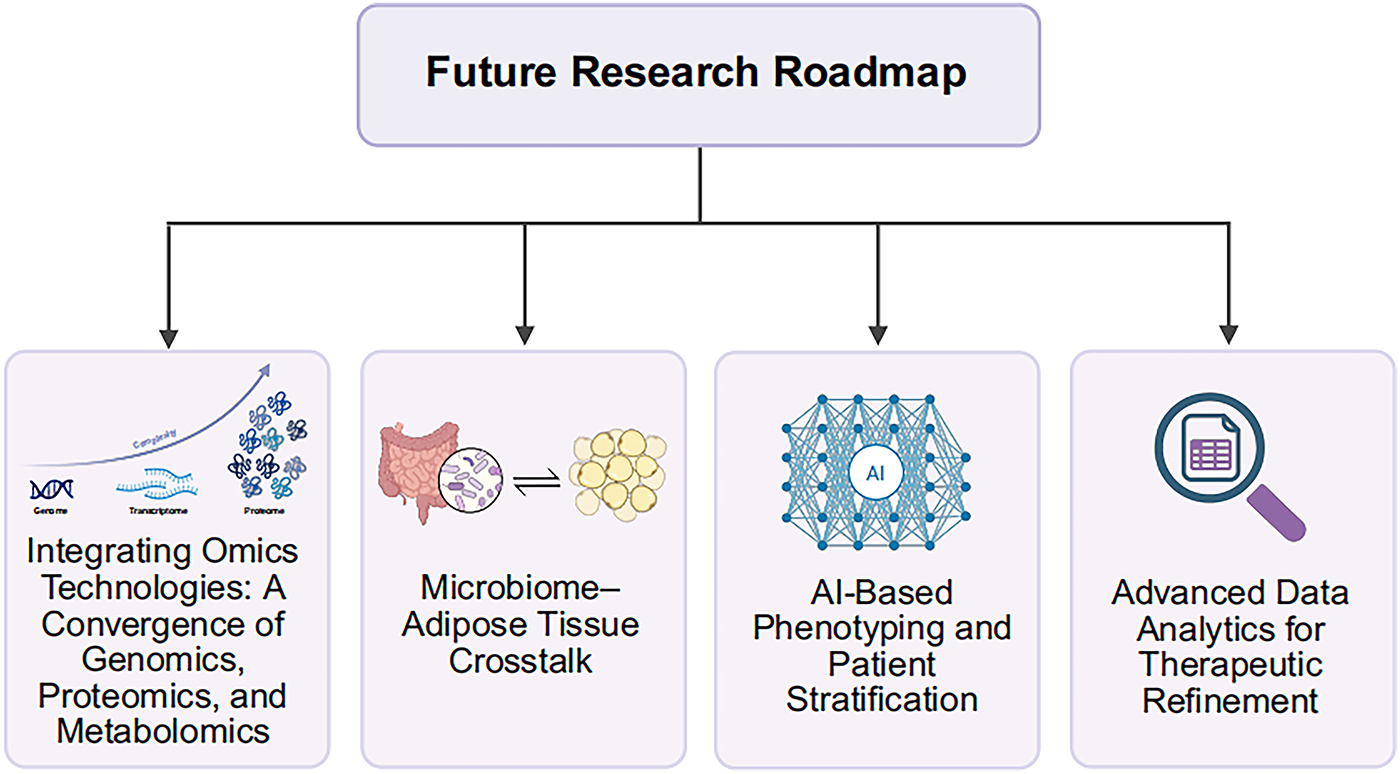
Figure 2: Future research roadmap for obesity-related asthma. This schematic illustrates key research directions, including the integration of omics technologies, investigation of microbiome-adipose tissue interactions, AI-based phenotyping for patient stratification, and the use of advanced data analytics to optimize therapeutic approaches. These efforts are aimed at improving the understanding and treatment of obesity-related asthma. AI, artificial intelligence. Figures created with biorender.com
Acknowledgement: None.
Funding Statement: This work was supported by the Pediatric Medicine Phase II Scientific Research Special Fund of the Jiangsu Medical Association (Grant No. SYH-32034-0106 [2024010]) and the Wuxi Science and Technology Development Fund (Grant No. K20241001).
Author Contributions: All authors made substantial contributions to the development of this manuscript. Conceptualization and study design: Huan Zhou, Jiami Jiang, Yuqing Zou. Manuscript drafting: Huan Zhou. Critical revision and editing: Jiahui Zhang, Zhiwei Yu. Visualization and figure preparation: Jiahui Zhang. Supervision and project oversight: Zhiwei Yu. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
References
1. Mims JW. Asthma: definitions and pathophysiology. Int Forum Allergy Rhinol. 2015;5(S1):S2–6. doi:10.1002/alr.21609. [Google Scholar] [PubMed] [CrossRef]
2. Gans MD, Gavrilova T. Understanding the immunology of asthma: pathophysiology, biomarkers, and treatments for asthma endotypes. Paediatr Respir Rev. 2020;36(2):118–27. doi:10.1016/j.prrv.2019.08.002. [Google Scholar] [PubMed] [CrossRef]
3. Miethe S, Karsonova A, Karaulov A, Renz H. Obesity and asthma. J Allergy Clin Immunol. 2020;146(4):685–93. doi:10.1016/j.jaci.2020.08.011. [Google Scholar] [PubMed] [CrossRef]
4. Zhu Z, Guo Y, Shi H, Liu CL, Panganiban RA, Chung W, et al. Shared genetic and experimental links between obesity-related traits and asthma subtypes in UK Biobank. J Allergy Clin Immunol. 2020;145(2):537–49. doi:10.1016/j.jaci.2019.09.035. [Google Scholar] [PubMed] [CrossRef]
5. Conrad LA, Nandalike K, Rani S, Rastogi D. Associations between sleep, obesity, and asthma in urban minority children. J Clin Sleep Med. 2022;18(10):2377–85. doi:10.5664/jcsm.10114. [Google Scholar] [PubMed] [CrossRef]
6. Wang K, Chen Z, Wei Z, He L, Gong L. Association between body fat distribution and asthma in adults: results from the cross-sectional and bidirectional Mendelian randomization study. Front Nutr. 2024;11:1432973. doi:10.3389/fnut.2024.1432973. [Google Scholar] [PubMed] [CrossRef]
7. Larsson SC, Burgess S. Causal role of high body mass index in multiple chronic diseases: a systematic review and meta-analysis of Mendelian randomization studies. BMC Med. 2021;19(1):320. doi:10.1186/s12916-021-02188-x. [Google Scholar] [PubMed] [CrossRef]
8. Vartiainen VA, Jousilahti P, Tuomilehto J, Laatikainen T, Vartiainen E. Body mass index and the risk of adult-onset asthma: a prospective observational study among 59,668 middle-aged men and women in Finland. Nutrients. 2024;16(15):2515. doi:10.3390/nu16152515. [Google Scholar] [PubMed] [CrossRef]
9. Althoff MD, Gaietto K, Holguin F, Forno E. Obesity-related asthma: a pathobiology-based overview of existing and emerging treatment approaches. Am J Respir Crit Care Med. 2024;210(10):1186–200. doi:10.1164/rccm.202406-1166SO. [Google Scholar] [PubMed] [CrossRef]
10. Anderson MR, Shashaty MGS. Impact of obesity in critical illness. Chest. 2021;160(6):2135–45. doi:10.1016/j.chest.2021.08.001. [Google Scholar] [PubMed] [CrossRef]
11. Veerati PC, Mitchel JA, Reid AT, Knight DA, Bartlett NW, Park JA, et al. Airway mechanical compression: its role in asthma pathogenesis and progression. Eur Respir Rev. 2020;29(157):190123. doi:10.1183/16000617.0123-2019. [Google Scholar] [PubMed] [CrossRef]
12. De Jong A, Wrigge H, Hedenstierna G, Gattinoni L, Chiumello D, Frat JP, et al. How to ventilate obese patients in the ICU. Intensive Care Med. 2020;46(12):2423–35. doi:10.1007/s00134-020-06286-x. [Google Scholar] [PubMed] [CrossRef]
13. Arismendi E, Bantulà M, Perpiñá M, Picado C. Effects of obesity and asthma on lung function and airway dysanapsis in adults and children. J Clin Med. 2020;9(11):3762. doi:10.3390/jcm9113762. [Google Scholar] [PubMed] [CrossRef]
14. Wensveen FM, Valentić S, Šestan M, Turk Wensveen T, Polić B. The Big Bang in obese fat: events initiating obesity-induced adipose tissue inflammation. Eur J Immunol. 2015;45(9):2446–56. doi:10.1002/eji.201545502. [Google Scholar] [PubMed] [CrossRef]
15. Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW Jr. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112(12):1796–808. doi:10.1172/JCI19246. [Google Scholar] [PubMed] [CrossRef]
16. Luo M, Zhao F, Cheng H, Su M, Wang Y. Macrophage polarization: an important role in inflammatory diseases. Front Immunol. 2024;15:1352946. doi:10.3389/fimmu.2024.1352946. [Google Scholar] [PubMed] [CrossRef]
17. Mancuso P. Obesity and lung inflammation. J Appl Physiol. 2010;108(3):722–8. doi:10.1152/japplphysiol.00781.2009. [Google Scholar] [PubMed] [CrossRef]
18. Jiang Z, Zhu L. Update on the role of alternatively activated macrophages in asthma. J Asthma Allergy. 2016;9:101–7. doi:10.2147/JAA.S104508. [Google Scholar] [PubMed] [CrossRef]
19. Ahmed B, Sultana R, Greene MW. Adipose tissue and insulin resistance in obese. Biomed Pharmacother. 2021;137:111315. doi:10.1016/j.biopha.2021.111315. [Google Scholar] [PubMed] [CrossRef]
20. Obradovic M, Sudar-Milovanovic E, Soskic S, Essack M, Arya S, Stewart AJ, et al. Leptin and obesity: role and clinical implication. Front Endocrinol. 2021;12:585887. doi:10.3389/fendo.2021.585887. [Google Scholar] [PubMed] [CrossRef]
21. Muñoz-Cofré R, Rojas-Mancilla E, Lizana PA, Escobar-Cabello M, García-Herrera C, Conei D, et al. Relationship between plasma leptin levels and airflow limitation in the small and medium airways in young adults. J Clin Med. 2025;14(5):1624. doi:10.3390/jcm14051624. [Google Scholar] [PubMed] [CrossRef]
22. Achari AE, Jain SK. Adiponectin, a therapeutic target for obesity, diabetes, and endothelial dysfunction. Int J Mol Sci. 2017;18(6):1321. doi:10.3390/ijms18061321. [Google Scholar] [PubMed] [CrossRef]
23. Sood A, Qualls C, Schuyler M, Thyagarajan B, Steffes MW, Smith LJ, et al. Low serum adiponectin predicts future risk for asthma in women. Am J Respir Crit Care Med. 2012;186(1):41–7. doi:10.1164/rccm.201110-1767OC. [Google Scholar] [PubMed] [CrossRef]
24. Ren Y, Zhao H, Yin C, Lan X, Wu L, Du X, et al. Adipokines, hepatokines and myokines: focus on their role and molecular mechanisms in adipose tissue inflammation. Front Endocrinol. 2022;13:873699. doi:10.3389/fendo.2022.873699. [Google Scholar] [PubMed] [CrossRef]
25. Larochelle J, Freiler J, Dice J, Hagan L. Plasma resistin levels in asthmatics as a marker of disease state. J Asthma. 2007;44(7):509–13. doi:10.1080/02770900701495785. [Google Scholar] [PubMed] [CrossRef]
26. Michaeloudes C, Abubakar-Waziri H, Lakhdar R, Raby K, Dixey P, Adcock IM, et al. Molecular mechanisms of oxidative stress in asthma. Mol Aspects Med. 2022;85:101026. doi:10.1016/j.mam.2021.101026. [Google Scholar] [PubMed] [CrossRef]
27. Lugg ST, Scott A, Parekh D, Naidu B, Thickett DR. Cigarette smoke exposure and alveolar macrophages: mechanisms for lung disease. Thorax. 2022;77(1):94–101. doi:10.1136/thoraxjnl-2020-216296. [Google Scholar] [PubMed] [CrossRef]
28. Tiotiu A, Zounemat Kermani N, Badi Y, Pavlidis S, Hansbro PM, Guo YK, et al. Sputum macrophage diversity and activation in asthma: role of severity and inflammatory phenotype. Allergy. 2021;76(3):775–88. doi:10.1111/all.14535. [Google Scholar] [PubMed] [CrossRef]
29. Wang Y, Wan R, Hu C. Leptin/obR signaling exacerbates obesity-related neutrophilic airway inflammation through inflammatory M1 macrophages. Mol Med. 2023;29(1):100. doi:10.1186/s10020-023-00702-w. [Google Scholar] [PubMed] [CrossRef]
30. Periyalil HA, Wood LG, Wright TA, Karihaloo C, Starkey MR, Miu AS, et al. Obese asthmatics are characterized by altered adipose tissue macrophage activation. Clin Exp Allergy. 2018;48(6):641–9. doi:10.1111/cea.13109. [Google Scholar] [PubMed] [CrossRef]
31. Reichenbach A, O’Brien W, Duran S, Authelet KJ, Freishtat RJ, Nadler EP, et al. Immune profile of adipose tissue from youth with obesity and asthma. Pediatr Obes. 2024;19(1):e13078. doi:10.1111/ijpo.13078. [Google Scholar] [PubMed] [CrossRef]
32. Khramova RN, Eliseeva TI, Ovsyannikov DY, Karpenko MA, Tush EV, Krasilnikova SV, et al. Effect of overweight and obesity on serum interleukin-6 in children and adolescents with bronchial asthma. J Biomed Res. 2025;39(6):648. doi:10.7555/jbr.38.20240381. [Google Scholar] [PubMed] [CrossRef]
33. Björkander S, Klevebro S, Hernandez-Pacheco N, Kere M, Ekström S, Sparreman Mikus M, et al. Obese asthma phenotypes display distinct plasma biomarker profiles. Clin Transl Allergy. 2023;13(3):e12238. doi:10.1002/clt2.12238. [Google Scholar] [PubMed] [CrossRef]
34. Chang C, Chen G, Wu W, Chen D, Chen S, Gao J, et al. Exogenous IL-25 ameliorates airway neutrophilia via suppressing macrophage M1 polarization and the expression of IL-12 and IL-23 in asthma. Respir Res. 2023;24(1):260. doi:10.1186/s12931-023-02557-5. [Google Scholar] [PubMed] [CrossRef]
35. Fainardi V, Esposito S, Chetta A, Pisi G. Asthma phenotypes and endotypes in childhood. Minerva Med. 2022;113(1):94–105. doi:10.23736/s0026-4806.21.07332-8. [Google Scholar] [PubMed] [CrossRef]
36. Pakkasela J, Ilmarinen P, Honkamäki J, Tuomisto LE, Andersén H, Piirilä P, et al. Age-specific incidence of allergic and non-allergic asthma. BMC Pulm Med. 2020;20(1):9. doi:10.1186/s12890-019-1040-2. [Google Scholar] [PubMed] [CrossRef]
37. Rastogi D, Canfield SM, Andrade A, Isasi CR, Hall CB, Rubinstein A, et al. Obesity-associated asthma in children: a distinct entity. Chest. 2012;141(4):895–905. doi:10.1378/chest.11-0930. [Google Scholar] [PubMed] [CrossRef]
38. Bapat SP, Whitty C, Mowery CT, Liang Y, Yoo A, Jiang Z, et al. Obesity alters pathology and treatment response in inflammatory disease. Nature. 2022;604(7905):337–42. doi:10.1038/s41586-022-04536-0. [Google Scholar] [PubMed] [CrossRef]
39. Mohamed MME, Amrani Y. Obesity enhances non-Th2 airway inflammation in a murine model of allergic asthma. Int J Mol Sci. 2024;25(11):6170. doi:10.3390/ijms25116170. [Google Scholar] [PubMed] [CrossRef]
40. Thompson DA, Wabara YB, Duran S, Reichenbach A, Chen L, Collado K, et al. Single cell analysis identifies distinct CD4+ T cells associated with the pathobiology of pediatric obesity related asthma. Sci Rep. 2025;15(1):6844. doi:10.1038/s41598-025-88423-4. [Google Scholar] [PubMed] [CrossRef]
41. Yon C, Thompson DA, Jude JA, Panettieri RA Jr, Rastogi D. Crosstalk between CD4+ T cells and airway smooth muscle in pediatric obesity-related asthma. Am J Respir Crit Care Med. 2023;207(4):461–74. doi:10.1164/rccm.202205-0985OC. [Google Scholar] [PubMed] [CrossRef]
42. Tejwani V, McCormack A, Suresh K, Woo H, Xu N, Davis MF, et al. Dexamethasone-induced FKBP51 expression in CD4+ T-lymphocytes is uniquely associated with worse asthma control in obese children with asthma. Front Immunol. 2021;12:744782. doi:10.3389/fimmu.2021.744782. [Google Scholar] [PubMed] [CrossRef]
43. Tejwani V, Wang R, Villabona-Rueda A, Suresh K, Wu TD, Adcock IM, et al. Distinct single-cell transcriptional profile in CD4+ T-lymphocytes among obese children with asthma. Am J Physiol Lung Cell Mol Physiol. 2025;328(3):L372–8. doi:10.1152/ajplung.00270.2024. [Google Scholar] [PubMed] [CrossRef]
44. Rastogi D, Johnston AD, Nico J, Loh LN, Jorge Y, Suzuki M, et al. Functional genomics of the pediatric obese asthma phenotype reveal enrichment of rho-GTPase pathways. Am J Respir Crit Care Med. 2020;202(2):259–74. doi:10.1164/rccm.201906-1199OC. [Google Scholar] [PubMed] [CrossRef]
45. Lan F, Li J, Miao W, Sun F, Duan S, Song Y, et al. GZMK-expressing CD8+ T cells promote recurrent airway inflammatory diseases. Nature. 2025;638(8050):490–8. doi:10.1038/s41586-024-08395-9. [Google Scholar] [PubMed] [CrossRef]
46. Kliem CV, Schaub B. The role of regulatory B cells in immune regulation and childhood allergic asthma. Mol Cell Pediatr. 2024;11(1):1. doi:10.1186/s40348-023-00174-2. [Google Scholar] [PubMed] [CrossRef]
47. Qian G, Jiang W, Sun D, Sun Z, Chen A, Fang H, et al. B-cell-derived IL-10 promotes allergic sensitization in asthma regulated by Bcl-3. Cell Mol Immunol. 2023;20(11):1313–27. doi:10.1038/s41423-023-01079-w. [Google Scholar] [PubMed] [CrossRef]
48. Wang P, Yang X, Zhang L, Sha S, Huang J, Peng J, et al. Tlr9 deficiency in B cells leads to obesity by promoting inflammation and gut dysbiosis. Nat Commun. 2024;15(1):4232. doi:10.1038/s41467-024-48611-8. [Google Scholar] [PubMed] [CrossRef]
49. Li M, Wang H, Ni Y, Li C, Xu X, Chang H, et al. Helminth-induced CD9+ B-cell subset alleviates obesity-associated inflammation via IL-10 production. Int J Parasitol. 2022;52(2–3):111–23. doi:10.1016/j.ijpara.2021.08.009. [Google Scholar] [PubMed] [CrossRef]
50. Jiang WY, Jiao RH, Ma SL, Dai JS, Zhu HF, Wu MY, et al. Serum inflammatory factors, vitamin D levels, and asthma severity in children with comorbid asthma and obesity/overweight: a comparative study. Front Pediatr. 2025;13:1439841. doi:10.3389/fped.2025.1439841. [Google Scholar] [PubMed] [CrossRef]
51. Uribe-Querol E, Rosales C. Neutrophils actively contribute to obesity-associated inflammation and pathological complications. Cells. 2022;11(12):1883. doi:10.3390/cells11121883. [Google Scholar] [PubMed] [CrossRef]
52. Gomez-Casado G, Jimenez-Gonzalez A, Rodriguez-Muñoz A, Tinahones FJ, González-Mesa E, Murri M, et al. Neutrophils as indicators of obesity-associated inflammation: a systematic review and meta-analysis. Obes Rev. 2025;26(3):e13868. doi:10.1111/obr.13868. [Google Scholar] [PubMed] [CrossRef]
53. Winsa-Lindmark S, Stridsman C, Sahlin A, Hedman L, Stenfors N, Myrberg T, et al. Severity of adult-onset asthma—a matter of blood neutrophils and severe obesity. Respir Med. 2023;219(10122):107418. doi:10.1016/j.rmed.2023.107418. [Google Scholar] [PubMed] [CrossRef]
54. Scott HA, Ng SH, McLoughlin RF, Valkenborghs SR, Nair P, Brown AC, et al. Effect of obesity on airway and systemic inflammation in adults with asthma: a systematic review and meta-analysis. Thorax. 2023;78(10):957–65. doi:10.1136/thorax-2022-219268. [Google Scholar] [PubMed] [CrossRef]
55. Alharbi KS, Fuloria NK, Fuloria S, Rahman SB, Al-Malki WH, Javed Shaikh MA, et al. Nuclear factor-kappa B and its role in inflammatory lung disease. Chem Biol Interact. 2021;345(Suppl 3):109568. doi:10.1016/j.cbi.2021.109568. [Google Scholar] [PubMed] [CrossRef]
56. Zhang T, Ma C, Zhang Z, Zhang H, Hu H. NF-κB signaling in inflammation and cancer. MedComm. 2021;2(4):618–53. doi:10.1002/mco2.104. [Google Scholar] [PubMed] [CrossRef]
57. Pantano C, Ather JL, Alcorn JF, Poynter ME, Brown AL, Guala AS, et al. Nuclear factor-κB activation in airway epithelium induces inflammation and hyperresponsiveness. Am J Respir Crit Care Med. 2008;177(9):959–69. doi:10.1164/rccm.200707-1096oc. [Google Scholar] [PubMed] [CrossRef]
58. do Nascimento Camargo L, Righetti RF, de Almeida FM, Dos Santos TM, Fukuzaki S, Martins NAB, et al. Modulating asthma-COPD overlap responses with IL-17 inhibition. Front Immunol. 2023;14:1271342. doi:10.3389/fimmu.2023.1271342. [Google Scholar] [PubMed] [CrossRef]
59. Nani A, Murtaza B, Khan AS, Khan NA, Hichami A. Antioxidant and anti-inflammatory potential of polyphenols contained in Mediterranean diet in obesity: molecular mechanisms. Molecules. 2021;26(4):985. doi:10.3390/molecules26040985. [Google Scholar] [PubMed] [CrossRef]
60. Akhter N, Wilson A, Arefanian H, Thomas R, Kochumon S, Al-Rashed F, et al. Endoplasmic reticulum stress promotes the expression of TNF-α in THP-1 cells by mechanisms involving ROS/CHOP/HIF-1α and MAPK/NF-κB pathways. Int J Mol Sci. 2023;24(20):15186. doi:10.3390/ijms242015186. [Google Scholar] [PubMed] [CrossRef]
61. Luo J, Wang J, Zhang J, Sang A, Ye X, Cheng Z, et al. Nrf2 deficiency exacerbated CLP-induced pulmonary injury and inflammation through autophagy- and NF-κB/PPARγ-mediated macrophage polarization. Cells. 2022;11(23):3927. doi:10.3390/cells11233927. [Google Scholar] [PubMed] [CrossRef]
62. Liu L, Guo H, Song A, Huang J, Zhang Y, Jin S, et al. Progranulin inhibits LPS-induced macrophage M1 polarization via NF-кB and MAPK pathways. BMC Immunol. 2020;21(1):32. doi:10.1186/s12865-020-00355-y. [Google Scholar] [PubMed] [CrossRef]
63. Yang J, Zhang M, Luo Y, Xu F, Gao F, Sun Y, et al. Protopine ameliorates OVA-induced asthma through modulatingTLR4/MyD88/NF-κB pathway and NLRP3 inflammasome-mediated pyroptosis. Phytomedicine. 2024;126(4):155410. doi:10.1016/j.phymed.2024.155410. [Google Scholar] [PubMed] [CrossRef]
64. Yang Z, Li X, Wei L, Bao L, Hu H, Liu L, et al. Involucrasin B suppresses airway inflammation in obese asthma by inhibiting the TLR4-NF-κB-NLRP3 pathway. Phytomedicine. 2024;132:155850. doi:10.1016/j.phymed.2024.155850. [Google Scholar] [PubMed] [CrossRef]
65. Liu L, Zhou L, Wang L, Mao Z, Zheng P, Zhang F, et al. MUC1 attenuates neutrophilic airway inflammation in asthma by reducing NLRP3 inflammasome-mediated pyroptosis through the inhibition of the TLR4/MyD88/NF-κB pathway. Respir Res. 2023;24(1):255. doi:10.1186/s12931-023-02550-y. [Google Scholar] [PubMed] [CrossRef]
66. Shailesh H, Noor S, Hayati L, Belavendra A, Van Panhuys N, Abou-Samra AB, et al. Asthma and obesity increase inflammatory markers in children. Front Allergy. 2025;5:1536168. doi:10.3389/falgy.2024.1536168. [Google Scholar] [PubMed] [CrossRef]
67. Fitzpatrick AM, Mutic AD, Mohammad AF, Stephenson ST, Grunwell JR. Obesity is associated with sustained symptomatology and unique inflammatory features in children with asthma. J Allergy Clin Immunol Pract. 2022;10(3):815–26.e2. doi:10.1016/j.jaip.2021.10.020. [Google Scholar] [PubMed] [CrossRef]
68. Vassiliou E, Farias-Pereira R. Impact of lipid metabolism on macrophage polarization: implications for inflammation and tumor immunity. Int J Mol Sci. 2023;24(15):12032. doi:10.3390/ijms241512032. [Google Scholar] [PubMed] [CrossRef]
69. Wang N, Li C, Zhang Z. Arctigenin ameliorates high-fat diet-induced metabolic disorders by reshaping gut microbiota and modulating GPR/HDAC3 and TLR4/NF-κB pathways. Phytomedicine. 2024;135:156123. doi:10.1016/j.phymed.2024.156123. [Google Scholar] [PubMed] [CrossRef]
70. Bantulà M, Roca-Ferrer J, Arismendi E, Picado C. Asthma and obesity: two diseases on the rise and bridged by inflammation. J Clin Med. 2021;10(2):169. doi:10.3390/jcm10020169. [Google Scholar] [PubMed] [CrossRef]
71. Russjan E. The role of peptides in asthma-obesity phenotype. Int J Mol Sci. 2024;25(6):3213. doi:10.3390/ijms25063213. [Google Scholar] [PubMed] [CrossRef]
72. Hara S, Tojima I, Shimizu S, Kouzaki H, Shimizu T. 17,18-epoxyeicosatetraenoic acid inhibits TNF-α-induced inflammation in cultured human airway epithelium and LPS-induced murine airway inflammation. Am J Rhinol Allergy. 2022;36(1):106–14. doi:10.1177/19458924211027682. [Google Scholar] [PubMed] [CrossRef]
73. Vieira CP, de Oliveira LP, Da Silva MB, Majolli Andre D, Tavares EBG, Pimentel ER, et al. Role of metalloproteinases and TNF-α in obesity-associated asthma in mice. Life Sci. 2020;259(5):118191. doi:10.1016/j.lfs.2020.118191. [Google Scholar] [PubMed] [CrossRef]
74. Adair D, Bagheri A, Yosef M, Khalatbari S, Lewis T, Mohan A, et al. High interleukin (IL)-6 is associated with lower lung function and increased likelihood of metabolic dysfunction in asthma. Pulm Ther. 2025;11(1):41–54. doi:10.1007/s41030-024-00281-z. [Google Scholar] [PubMed] [CrossRef]
75. Stancil IT, Michalski JE, Hennessy CE, Hatakka KL, Yang IV, Kurche JS, et al. Interleukin-6-dependent epithelial fluidization initiates fibrotic lung remodeling. Sci Transl Med. 2022;14(654):eabo5254. doi:10.1126/scitranslmed.abo5254. [Google Scholar] [PubMed] [CrossRef]
76. Kyler KE, Jones BL. The chicken or the egg: the role of IL-6 in pediatric obese and allergen-exposed asthma. J Allergy Clin Immunol. 2023;152(6):1420–2. doi:10.1016/j.jaci.2023.09.037. [Google Scholar] [PubMed] [CrossRef]
77. Guo M, Liu H, Yu Y, Zhu X, Xie H, Wei C, et al. Lactobacillus rhamnosus GG ameliorates osteoporosis in ovariectomized rats by regulating the Th17/Treg balance and gut microbiota structure. Gut Microbes. 2023;15(1):2190304. doi:10.1080/19490976.2023.2190304. [Google Scholar] [CrossRef]
78. Biesemann N, Margerie D, Asbrand C, Rehberg M, Savova V, Agueusop I, et al. Additive efficacy of a bispecific anti-TNF/IL-6 nanobody compound in translational models of rheumatoid arthritis. Sci Transl Med. 2023;15(681):eabq4419. doi:10.1126/scitranslmed.abq4419. [Google Scholar] [PubMed] [CrossRef]
79. Baltieri L, Cazzo E, Oliveira Modena DA, Gobato Rentel RC, Martins LC, Chaim EA. Correlation between levels of adipokines and inflammatory mediators with spirometric parameters in individuals with obesity and symptoms of asthma: cross-sectional study. Pulmonology. 2022;28(2):105–12. doi:10.1016/j.pulmoe.2020.04.003. [Google Scholar] [PubMed] [CrossRef]
80. Xu C, Rafique A, Potocky T, Paccaly A, Nolain P, Lu Q, et al. Differential binding of sarilumab and tocilizumab to IL-6Rα and effects of receptor occupancy on clinical parameters. J Clin Pharmacol. 2021;61(5):714–24. doi:10.1002/jcph.1795. [Google Scholar] [PubMed] [CrossRef]
81. Guerrero SC, Panettieri RA Jr, Rastogi D. Mechanistic links between obesity and airway pathobiology inform therapies for obesity-related asthma. Paediatr Drugs. 2023;25(3):283–99. doi:10.1007/s40272-022-00554-7. [Google Scholar] [PubMed] [CrossRef]
82. Nazari J, Amini S, Amiresmaili M. Lessons learned to control COVID-19 to prevent a crisis: the case of Iran. Open Public Health J. 2020;13(1):529–30. doi:10.2174/1874944502013010529. [Google Scholar] [CrossRef]
83. Chen W, Chen H, Yang ZT, Mao EQ, Chen Y, Chen EZ. Free fatty acids-induced neutrophil extracellular traps lead to dendritic cells activation and T cell differentiation in acute lung injury. Aging. 2021;13(24):26148–60. doi:10.18632/aging.203802. [Google Scholar] [PubMed] [CrossRef]
84. Vollmer CM, Dias ASO, Lopes LM, Kasahara TM, Delphim L, Silva JCC, et al. Leptin favors Th17/Treg cell subsets imbalance associated with allergic asthma severity. Clin Transl Allergy. 2022;12(6):e12153. doi:10.1002/clt2.12153. [Google Scholar] [PubMed] [CrossRef]
85. Vollmer C, Dias A, Sales M, Sacramento PM, Silva JC, Oyamada HAA, et al. Leptin favors imbalance of antigen-specific CD4+ T-cells associated with severity of cat allergy. Front Immunol. 2023;14:1290740. doi:10.3389/fimmu.2023.1290740. [Google Scholar] [PubMed] [CrossRef]
86. Ramos-Ramírez P, Malmhäll C, Tliba O, Rådinger M, Bossios A. Adiponectin/AdipoR1 axis promotes IL-10 release by human regulatory T cells. Front Immunol. 2021;12:677550. doi:10.3389/fimmu.2021.677550. [Google Scholar] [PubMed] [CrossRef]
87. Ramos-Ramírez P, Malmhäll C, Johansson K, Adner M, Lötvall J, Bossios A. Lung regulatory T cells express adiponectin receptor 1: modulation by obesity and airway allergic inflammation. Int J Mol Sci. 2020;21(23):8990. doi:10.3390/ijms21238990. [Google Scholar] [PubMed] [CrossRef]
88. Duan J, Yin Y, Cui J, Yan J, Zhu Y, Guan Y, et al. Chikusetsu saponin IVa ameliorates cerebral ischemia reperfusion injury in diabetic mice via adiponectin-mediated AMPK/GSK-3β pathway in vivo and in vitro. Mol Neurobiol. 2016;53(1):728–43. doi:10.1007/s12035-014-9033-x. [Google Scholar] [CrossRef]
89. Liu H, Wu X, Luo J, Zhao L, Li X, Guo H, et al. Adiponectin peptide alleviates oxidative stress and NLRP3 inflammasome activation after cerebral ischemia-reperfusion injury by regulating AMPK/GSK-3β. Exp Neurol. 2020;329:113302. doi:10.1016/j.expneurol.2020.113302. [Google Scholar] [PubMed] [CrossRef]
90. Tooba R, Wu TD. Obesity and asthma: a focused review. Respir Med. 2022;204(1):107012. doi:10.1016/j.rmed.2022.107012. [Google Scholar] [PubMed] [CrossRef]
91. Shailesh H, Bhat AA, Janahi IA. Obesity-associated non-T2 mechanisms in obese asthmatic individuals. Biomedicines. 2023;11(10):2797. doi:10.3390/biomedicines11102797. [Google Scholar] [PubMed] [CrossRef]
92. Bradding P, Porsbjerg C, Côté A, Dahlén SE, Hallstrand TS, Brightling CE. Airway hyperresponsiveness in asthma: the role of the epithelium. J Allergy Clin Immunol. 2024;153(5):1181–93. doi:10.1016/j.jaci.2024.02.011. [Google Scholar] [PubMed] [CrossRef]
93. Reza MI, Ambhore NS. Inflammation in asthma: mechanistic insights and the role of biologics in therapeutic frontiers. Biomedicines. 2025;13(6):1342. doi:10.3390/biomedicines13061342. [Google Scholar] [PubMed] [CrossRef]
94. Foer D, Forno E, Holguin F, Cahill KN. Weight loss interventions for adults with obesity-related asthma. J Allergy Clin Immunol Pract. 2024;12(4):840–7. doi:10.1016/j.jaip.2023.12.041. [Google Scholar] [PubMed] [CrossRef]
95. Olejnik AE, Kuźnar-Kamińska B. Association of obesity and severe asthma in adults. J Clin Med. 2024;13(12):3474. doi:10.3390/jcm13123474. [Google Scholar] [PubMed] [CrossRef]
96. Reyes Noriega N, Del-Río-Navarro BE, Berber A, de Jesús Romero Tapia S, Molina Díaz DJM. Effect of obesity on lung function in the pediatric and adult populations with asthma: a review. J Clin Med. 2023;12(16):5385. doi:10.3390/jcm12165385. [Google Scholar] [PubMed] [CrossRef]
97. Popović-Grle S, Štajduhar A, Lampalo M, Rnjak D. Biomarkers in different asthma phenotypes. Genes. 2021;12(6):801. doi:10.3390/genes12060801. [Google Scholar] [PubMed] [CrossRef]
98. Henderson I, Caiazzo E, McSharry C, Guzik TJ, Maffia P. Why do some asthma patients respond poorly to glucocorticoid therapy? Pharmacol Res. 2020;160(1):105189. doi:10.1016/j.phrs.2020.105189. [Google Scholar] [PubMed] [CrossRef]
99. Witte JA, Braunstahl GJ, Blox WJB, van’t Westeinde SC, In’t Veen JCCM, Kappen JH, et al. STOP: an open label crossover trial to study ICS withdrawal in patients with a combination of obesity and low-inflammatory asthma and evaluate its effect on asthma control and quality of life. BMC Pulm Med. 2022;22(1):53. doi:10.1186/s12890-022-01843-0. [Google Scholar] [PubMed] [CrossRef]
100. Li W, Marx N, Yang Q, Fang D, Zhang Y. Obesity: next game changer of allergic airway diseases? Clin Transl Med. 2025;15(5):e70316. doi:10.1002/ctm2.70316. [Google Scholar] [PubMed] [CrossRef]
101. Baggott C, Hardy JK, Sparks J, Sabbagh D, Beasley R, Weatherall M, et al. Epinephrine (adrenaline) compared to selective beta-2-agonist in adults or children with acute asthma: a systematic review and meta-analysis. Thorax. 2022;77(6):563–72. doi:10.1136/thoraxjnl-2021-217124. [Google Scholar] [PubMed] [CrossRef]
102. Mizuta K, Matoba A, Shibata S, Masaki E, Emala CW Sr. Obesity-induced asthma: role of free fatty acid receptors. Jpn Dent Sci Rev. 2019;55(1):103–7. doi:10.1016/j.jdsr.2019.07.002. [Google Scholar] [PubMed] [CrossRef]
103. Marcello C, Carlo L. Asthma phenotypes: the intriguing selective intervention with Montelukast. Asthma Res Pract. 2016;2(1):11. doi:10.1186/s40733-016-0026-6. [Google Scholar] [PubMed] [CrossRef]
104. Mancuso P, Canetti C, Gottschalk A, Tithof PK, Peters-Golden M. Leptin augments alveolar macrophage leukotriene synthesis by increasing phospholipase activity and enhancing group IVC iPLA2 (cPLA2gamma) protein expression. Am J Physiol Lung Cell Mol Physiol. 2004;287(3):L497–502. doi:10.1152/ajplung.00010.2004. [Google Scholar] [PubMed] [CrossRef]
105. Liu X, Tang Y, Luo Y, Gao Y, He L. Role and mechanism of specialized pro-resolving mediators in obesity-associated insulin resistance. Lipids Health Dis. 2024;23(1):234. doi:10.1186/s12944-024-02207-9. [Google Scholar] [PubMed] [CrossRef]
106. Farzan S, Coyle T, Coscia G, Rebaza A, Santiago M. Clinical characteristics and management strategies for adult obese asthma patients. J Asthma Allergy. 2022;15:673–89. doi:10.2147/JAA.S285738. [Google Scholar] [PubMed] [CrossRef]
107. Alu A, Lei H, Han X, Wei Y, Wei X. BTK inhibitors in the treatment of hematological malignancies and inflammatory diseases: mechanisms and clinical studies. J Hematol Oncol. 2022;15(1):138. doi:10.1186/s13045-022-01353-w. [Google Scholar] [PubMed] [CrossRef]
108. Ringheim GE, Wampole M, Oberoi K. Bruton’s tyrosine kinase (BTK) inhibitors and autoimmune diseases: making sense of BTK inhibitor specificity profiles and recent clinical trial successes and failures. Front Immunol. 2021;12:662223. doi:10.3389/fimmu.2021.662223. [Google Scholar] [PubMed] [CrossRef]
109. Choi Y, Kim SH, Shin SJ, Park HS, Shin YS. The effect of Bruton’s tyrosine kinase (BTK) inhibitor in the eosinophilic asthma model of mouse. Int Immunopharmacol. 2024;132(17):111903. doi:10.1016/j.intimp.2024.111903. [Google Scholar] [PubMed] [CrossRef]
110. Kong J, Yang F, Bai M, Zong Y, Li Z, Meng X, et al. Airway immune response in the mouse models of obesity-related asthma. Front Physiol. 2022;13:909209. doi:10.3389/fphys.2022.909209. [Google Scholar] [PubMed] [CrossRef]
111. Zouein J, Que LG, Ingram JL. Obesity-driven airway eosinophilia and neutrophilia in asthma. J Asthma. 2025;62(9):1472–82. doi:10.1080/02770903.2025.2505464. [Google Scholar] [PubMed] [CrossRef]
112. Chan DY, Barra NG, Fang H, Rodrigues e-Lacerda R, Schertzer JD. Bruton’s tyrosine kinase (BTK) inhibitors alter blood glucose and insulin in obese mice but reduce inflammation independent of BTK. Am J Physiol Endocrinol Metab. 2024;327(3):E271–8. doi:10.1152/ajpendo.00205.2024. [Google Scholar] [PubMed] [CrossRef]
113. Zhu S, Gokhale S, Jung J, Spirollari E, Tsai J, Arceo J, et al. Multifaceted immunomodulatory effects of the BTK inhibitors ibrutinib and acalabrutinib on different immune cell subsets—beyond B lymphocytes. Front Cell Dev Biol. 2021;9:727531. doi:10.3389/fcell.2021.727531. [Google Scholar] [PubMed] [CrossRef]
114. Chinthrajah RS, Galli SJ. Omalizumab in “non-IgE-mediated” diseases. J Allergy Clin Immunol. 2021;147(4):1207–8. doi:10.1016/j.jaci.2020.10.033. [Google Scholar] [PubMed] [CrossRef]
115. Geng B, Dixon AE, Ko J, Janampally P, Haselkorn T, Holweg CTJ, et al. Impact of body mass index on omalizumab response in adults with moderate-to-severe allergic asthma. Ann Allergy Asthma Immunol. 2022;128(5):553–60. doi:10.1016/j.anai.2022.01.025. [Google Scholar] [PubMed] [CrossRef]
116. Oliveira MJ, Vieira M, Coutinho D, Ladeira I, Pascoal I, Ferreira J, et al. Severe asthma in obese patients: improvement of lung function after treatment with omalizumab. Pulmonology. 2019;25(1):15–20. doi:10.1016/j.pulmoe.2018.01.005. [Google Scholar] [PubMed] [CrossRef]
117. Pilkington AW 4th, Buragamadagu B, Johnston RA. Weighted breaths: exploring biologic and non-biologic therapies for co-existing asthma and obesity. Curr Allergy Asthma Rep. 2024;24(7):381–93. doi:10.1007/s11882-024-01153-x. [Google Scholar] [PubMed] [CrossRef]
118. Principe S, Porsbjerg C, Bolm Ditlev S, Kjærsgaard Klein D, Golebski K, Dyhre-Petersen N, et al. Treating severe asthma: targeting the IL-5 pathway. Clin Exp Allergy. 2021;51(8):992–1005. doi:10.1111/cea.13885. [Google Scholar] [PubMed] [CrossRef]
119. Antosz K, Batko J, Błażejewska M, Gawor A, Sleziak J, Gomułka K. Insight into IL-5 as a potential target for the treatment of allergic diseases. Biomedicines. 2024;12(7):1531. doi:10.3390/biomedicines12071531. [Google Scholar] [PubMed] [CrossRef]
120. Sahnoon L, Bajbouj K, Mahboub B, Hamoudi R, Hamid Q. Targeting IL-13 and IL-4 in asthma: therapeutic implications on airway remodeling in severe asthma. Clin Rev Allergy Immunol. 2025;68(1):44. doi:10.1007/s12016-025-09045-2. [Google Scholar] [PubMed] [CrossRef]
121. Listyoko AS, Okazaki R, Harada T, Inui G, Yamasaki A. Impact of obesity on airway remodeling in asthma: pathophysiological insights and clinical implications. Front Allergy. 2024;5:1365801. doi:10.3389/falgy.2024.1365801. [Google Scholar] [PubMed] [CrossRef]
122. Williams EJ, Negewo NA, Baines KJ. Role of the NLRP3 inflammasome in asthma: relationship with neutrophilic inflammation, obesity, and therapeutic options. J Allergy Clin Immunol. 2021;147(6):2060–2. doi:10.1016/j.jaci.2021.04.022. [Google Scholar] [PubMed] [CrossRef]
123. Tashiro H, Kurihara Y, Kuwahara Y, Takahashi K. Impact of obesity in asthma: possible future therapies. Allergol Int. 2024;73(1):48–57. doi:10.1016/j.alit.2023.08.007. [Google Scholar] [PubMed] [CrossRef]
124. Maniscalco M, Zamparelli AS, Vitale DF, Faraone S, Molino A, Zedda A, et al. Long-term effect of weight loss induced by bariatric surgery on asthma control and health related quality of life in asthmatic patients with severe obesity: a pilot study. Respir Med. 2017;130(2):69–74. doi:10.1016/j.rmed.2017.06.010. [Google Scholar] [PubMed] [CrossRef]
125. Freitas PD, Ferreira PG, Silva AG, Stelmach R, Carvalho-Pinto RM, Fernandes FLA, et al. The role of exercise in a weight-loss program on clinical control in obese adults with asthma. A randomized controlled trial. Am J Respir Crit Care Med. 2017;195(1):32–42. doi:10.1164/rccm.201603-0446OC. [Google Scholar] [PubMed] [CrossRef]
126. Mitchell PD, Salter BM, Oliveria JP, El-Gammal A, Tworek D, Smith SG, et al. Glucagon-like peptide-1 receptor expression on human eosinophils and its regulation of eosinophil activation. Clin Exp Allergy. 2017;47(3):331–8. doi:10.1111/cea.12860. [Google Scholar] [PubMed] [CrossRef]
127. Kaplan AG, Kim JW. Asthma exacerbations and glucagon-like peptide-1 receptor agonists: a review of the current evidence. Pulm Ther. 2022;8(4):343–58. doi:10.1007/s41030-022-00203-x. [Google Scholar] [PubMed] [CrossRef]
128. Kong W, Zhang X, Gu H, Chen M, Li M, Zhang X, et al. Association between BMI and asthma in adults over 45 years of age: analysis of Global Burden of Disease 2021, China Health and Retirement Longitudinal Study, and National Health and Nutrition Examination Survey data. eClinicalMedicine. 2025;82(4):103163. doi:10.1016/j.eclinm.2025.103163. [Google Scholar] [PubMed] [CrossRef]
129. Fang C, Jiang Z, Su X, Fan W. The association between body mass index and asthma in children: a cross-sectional study from NHANES 1999 to 2020. Sci Rep. 2025;15(1):9448. doi:10.1038/s41598-025-92619-z. [Google Scholar] [PubMed] [CrossRef]
130. Tay TR, Pang PH, Liew MF, Tiew PY, Tun MH, Jaggi TK, et al. Ethnic variation in South East Asian patients with severe asthma: a first look at the Singapore Severe Asthma Registry (SSAR) and implications on clinical practice. Respir Med. 2025;245(10):108199. doi:10.1016/j.rmed.2025.108199. [Google Scholar] [PubMed] [CrossRef]
131. Al Heialy S, Gaudet M, Ramakrishnan RK, Mogas A, Salameh L, Mahboub B, et al. Contribution of IL-17 in steroid hyporesponsiveness in obese asthmatics through dysregulation of glucocorticoid receptors α and β. Front Immunol. 2020;11:1724. doi:10.3389/fimmu.2020.01724. [Google Scholar] [PubMed] [CrossRef]
132. Leija-Martínez JJ, Giacoman-Martínez A, Del-Río-Navarro BE, Sanchéz-Muñoz F, Hernández-Diazcouder A, Muñoz-Hernández O, et al. Promoter methylation status of RORC, IL17A, and TNFA in peripheral blood leukocytes in adolescents with obesity-related asthma. Heliyon. 2022;8(12):e12316. doi:10.1016/j.heliyon.2022.e12316. [Google Scholar] [PubMed] [CrossRef]
133. Buabraig A, Karakuş R. The roles of interleukin-17A/F and IL-36Ra in obese asthmatic children. J Gazi Univ Health Sci Inst. 2022;4(1):10–7. [Google Scholar]
134. Garg D, Que LG, Ingram JL. Effects of biological therapies on patients with Type-2 high asthma and comorbid obesity. Front Pharmacol. 2024;14:1315540. doi:10.3389/fphar.2023.1315540. [Google Scholar] [PubMed] [CrossRef]
135. Adrish M, Akuthota P. Approach to non-type 2 asthma. Respir Med. 2023;216(7933):107327. doi:10.1016/j.rmed.2023.107327. [Google Scholar] [PubMed] [CrossRef]
136. Tiotiu A, Labor M, Nedeva D, Novakova S, Oguzulgen IK, Mihaicuta S, et al. How to apply the personalized medicine in obesity-associated asthma? Expert Rev Respir Med. 2020;14(9):905–15. doi:10.1080/17476348.2020.1780123. [Google Scholar] [PubMed] [CrossRef]
137. Hudler AC, Díaz IRR, Sharma S, Holguin F. Gaps and future directions in clinical research on obesity-related asthma. Pulm Ther. 2023;9(3):309–27. doi:10.1007/s41030-023-00230-2. [Google Scholar] [PubMed] [CrossRef]
138. Muzaffar N, Baber M, Iqra Malik H. Role of precision medicine on different endotypes of asthma. Explor Asthma Allergy. 2025;3:100984. doi:10.37349/eaa.2025.100984. [Google Scholar] [CrossRef]
139. Cereta AD, Oliveira VR, Costa IP, Afonso JPR, Fonseca AL, de Souza ART, et al. Emerging cell-based therapies in chronic lung diseases: what about asthma? Front Pharmacol. 2021;12:648506. doi:10.3389/fphar.2021.648506. [Google Scholar] [PubMed] [CrossRef]
140. D’Amato M, Pastore D, Lupia C, Candia C, Bruni A, Garofalo E, et al. Biologic therapy in severe asthma: a phenotype-driven and targeted approach. J Clin Med. 2025;14(13):4749. doi:10.3390/jcm14134749. [Google Scholar] [PubMed] [CrossRef]
141. Qin Z, Yang H, Liu J, Li D, Wang Y, Chen Y, et al. Obesity alters inflammatory response in the pathology of asthma (Review). Int J Mol Med. 2023;52(1):63. doi:10.3892/ijmm.2023.5266. [Google Scholar] [PubMed] [CrossRef]
142. Mendes FC, Garcia-Larsen V, Moreira A. Obesity and asthma: implementing a treatable trait care model. Clin Exp Allergy. 2024;54(11):881–94. doi:10.1111/cea.14520. [Google Scholar] [PubMed] [CrossRef]
143. Mazzotta C, Barkai L. Obesity and asthma in children-coexistence or pathophysiological connections? Biomedicines. 2025;13(5):1114. doi:10.3390/biomedicines13051114. [Google Scholar] [PubMed] [CrossRef]
144. Ricciardolo FLM, Sprio AE, Baroso A, Gallo F, Riccardi E, Bertolini F, et al. Characterization of T2-low and T2-high asthma phenotypes in real-life. Biomedicines. 2021;9(11):1684. doi:10.3390/biomedicines9111684. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF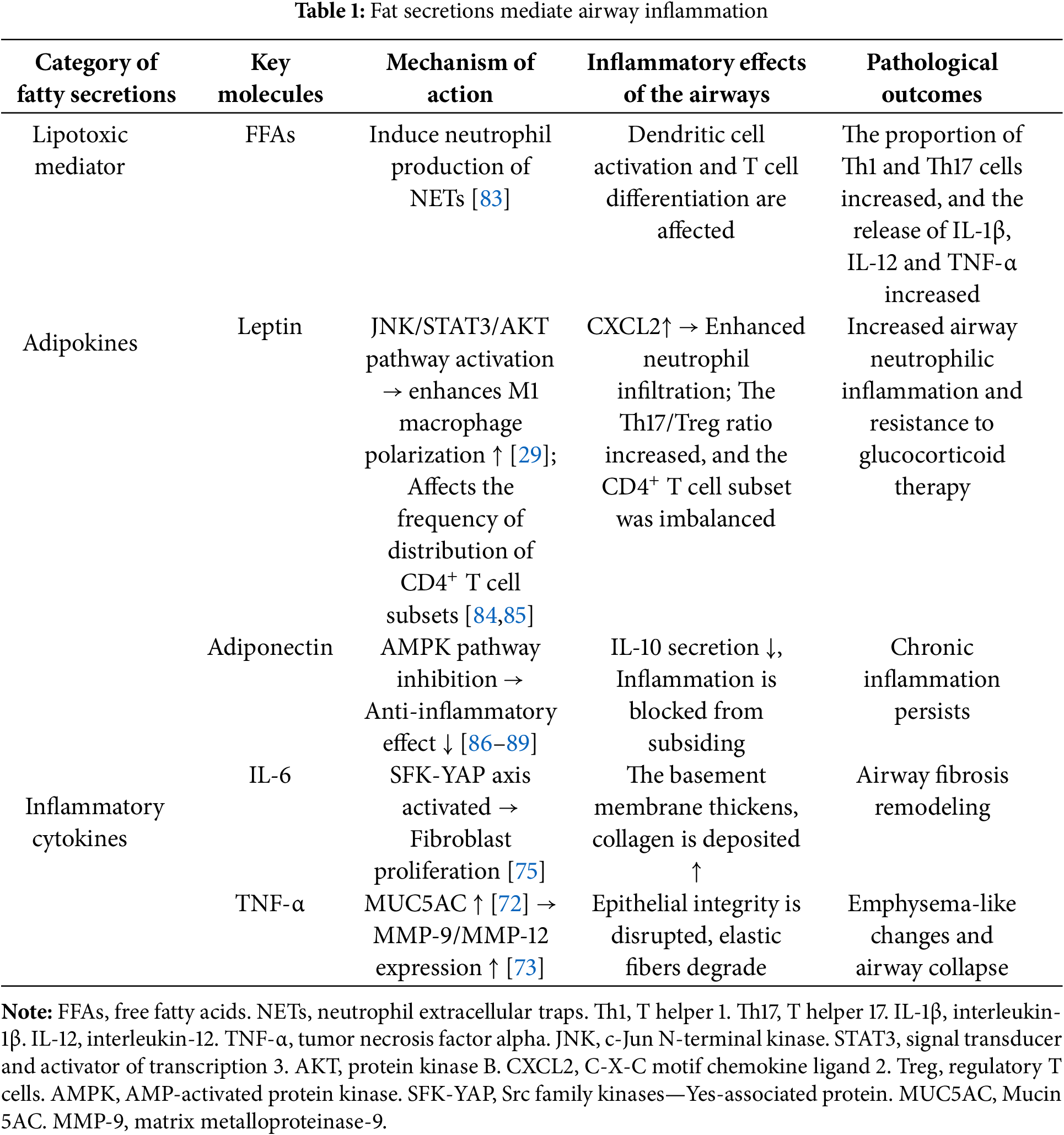
 Downloads
Downloads
 Citation Tools
Citation Tools