 Open Access
Open Access
REVIEW
Targeting Protein Arginine Deiminases in Rheumatoid Arthritis: Pathophysiology and Therapeutic Progress
1 Division of Allergy, Immunology and Rheumatology, Department of Pediatrics, Taichung Veterans General Hospital, Taichung, 407, Taiwan
2 Department of Post-Baccalaureate Medicine, College of Medicine, National Chung Hsing University, Taichung, 402, Taiwan
3 Doctoral Program in Translational Medicine, National Chung Hsing University, Taichung, 402, Taiwan
4 Division of Pulmonary and Critical Care Medicine, Department of Internal Medicine, China Medical University Hospital, Taichung, 402, Taiwan
5 School of Medicine, College of Medicine, China Medical University, Taichung, 402, Taiwan
6 Department of Material Science and Engineering, National Chiao Tung University, Hsinchu, 30010, Taiwan
7 Institute of Bioinformatics and Structural Biology & Department of Medical Sciences, National Tsing Hua University, Hsinchu, 300, Taiwan
8 Department of Medical Research, China Medical University Hospital, Taichung, 402, Taiwan
9 Department of Medical Research, Taichung Veterans General Hospital, Taichung, 402, Taiwan
10 Department of Pharmacology, College of Medicine, Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
11 Institute of Biomedical Science, National Chung-Hsing University, Taichung, 402, Taiwan
* Corresponding Authors: Tzu-Ting Chen. Email: ; Chi-Chen Lin. Email:
(This article belongs to the Special Issue: Natural and Synthetic Small Molecules in the Regulation of Immune Cell Functions)
BIOCELL 2026, 50(4), 3 https://doi.org/10.32604/biocell.2025.072732
Received 02 September 2025; Accepted 02 December 2025; Issue published 21 April 2026
Abstract
Protein arginine deiminases (PADs) are key enzymes in the development of rheumatoid arthritis (RA), catalyzing the conversion of arginine to citrulline in a process called citrullination. This post-translational modification is crucial to RA pathogenesis as it creates neo-antigens that trigger the production of anti-citrullinated protein antibodies (ACPAs). These ACPAs are highly specific to RA and often appear before clinical symptoms, making them valuable biomarkers for diagnosis and prognosis. Beyond ACPA production, PADs, particularly PAD4, play a vital role in forming neutrophil extracellular traps (NETs). NETs contribute to inflammation and joint damage, further highlighting the importance of PAD activity in the disease. The dysregulation of PADs is not limited to joint inflammation but also contributes to systemic complications of RA, such as cardiovascular and pulmonary disorders like interstitial lung disease (ILD). Citrullination of proteins in various tissues may contribute to these complications by generating autoantigens or disrupting tissue structures. Targeting PADs with small-molecule inhibitors or monoclonal antibodies shows promise in preclinical studies, with compounds like Cl-amidine and isozyme-selective inhibitors like GSK199 demonstrating efficacy in animal models. However, challenges remain in drug development, including limited isozyme specificity, off-target effects, and the complexity of citrullination networks, which hinder clinical translation. Overcoming these obstacles will require a multidisciplinary approach to optimize drug design and validate PADs as effective therapeutic targets for improving RA management and mitigating its systemic effects. The purpose of this review is to summarize the current understanding of the multifaceted roles of PADs in RA, from pathogenesis and systemic complications to the status and challenges of therapeutic development.Graphic Abstract
Keywords
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by persistent inflammation of the synovium, which leads to the degeneration of bone and cartilage [1]. The complex and multifaceted pathophysiology of RA involves a combination of environmental triggers, dysregulated immune responses, and genetic predisposition [1–3]. The family of enzymes known as protein arginine deiminases (PADs) is a major contributor to this complex process [4,5]. This review examines the current understanding of PADs’ roles in RA, with particular attention to their function in disease pathophysiology, its consequences, and the ongoing challenges in developing effective therapeutics that target PADs.
2 PADs and Citrullination in RA Pathogenesis
PADs constitute a family of calcium-dependent enzymes responsible for an irreversible post-translational modification known as citrullination, or deamination [6,7]. This enzymatic reaction involves the hydrolysis of the guanidinium group of peptidylarginine residues, converting them into citrulline [8]. The resulting citrullinated proteins act as neo-antigens, prompting the production of ACPAs. ACPAs are highly specific to RA and frequently appear before the onset of clinical symptoms, making them valuable biomarkers for diagnosis and prognosis [9]. Their presence often precedes overt disease, suggesting their involvement in disease initiation or exacerbation [10].
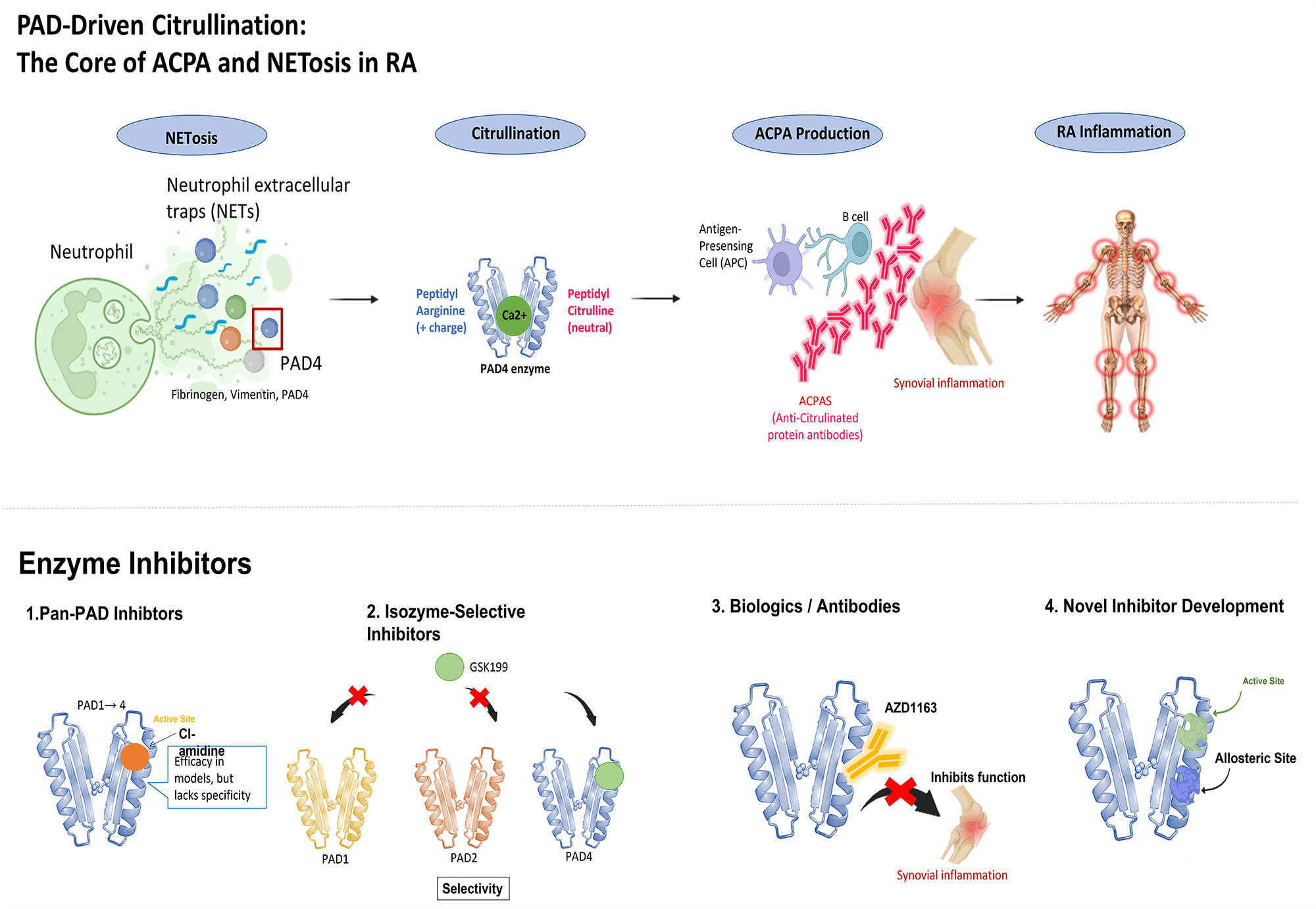
ACPAs are not merely biomarkers; they can actively contribute to disease pathogenesis. By interacting with citrullinated proteins [11], they can activate immune cells and promote inflammation in the synovial joint. Moreover, ACPAs can directly interact with synovial lining cells, including fibroblast-like synoviocytes (FLSs), altering their activity and causing joint degradation [11]. Specifically, ACPAs can bind to citrullinated antigens expressed on the surface of FLSs. This interaction triggers intracellular signaling cascades, particularly the phosphorylation of Focal Adhesion Kinase (FAK) and the subsequent activation of the PI3K/Akt pathway, which ultimately enhances FLS migration and promotes synovial invasion [11]. The substantial correlation between the existence and levels of ACPAs and the prognosis and severity of the disease demonstrates their key role in RA pathophysiology [12,13]. Fig. 1 illustrates the central role of PAD4-driven citrullination in initiating this pathogenic cycle, highlighting how it simultaneously drives ACPA production and NETosis to amplify RA inflammation.

Figure 1: Protein arginine deiminase (PAD)-driven citrullination: the core of anti-citrullinated protein antibodie (ACPA) and neutrophil extracellular traps (NET) osis in rheumatoid arthritis (RA)
PAD4 functions as the core enzymatic engine in RA pathogenesis, catalyzing the conversion of arginine to citrulline, which drives two major intertwined pathways. Firstly, PAD4 creates neo-antigens (e.g., citrullinated fibrinogen), leading to the production of ACPAs that precipitate synovial inflammation and joint damage. Secondly, PAD4 translocates into the neutrophil nucleus to citrullinate histones, driving NETosis. The released NETs—rich in citrullinated proteins—further amplify the inflammatory response and ACPA production, establishing a pathogenic vicious cycle.
3 Multifaceted Contributions of PADs to RA Pathogenesis
Protein citrullination by PADs significantly influences RA pathophysiology beyond being merely a consequence of inflammation. Citrullination generates neo-antigens from self-proteins [14,15], which the immune system recognizes as foreign targets. PADs themselves, particularly PAD4, can also serve as immune targets [16], leading to ACPA production. Certain HLA-DR alleles, particularly those carrying the “shared epitope” such as HLA-DRB1*04:01, have been linked to the occurrence of anti-PAD4 antibodies in RA patients, potentially via a hapten carrier mechanism where the PAD4 enzyme itself is presented as an autoantigen [7,17]. In this specific context, it is hypothesized that the PAD4 enzyme itself functions as the carrier protein. According to the hapten-carrier model, the PAD4 enzyme functions as a carrier protein that forms complexes with citrullinated substrates (acting as haptens). In this scenario, autoreactive T cells recognize PAD4 peptides presented by HLA-DR molecules and provide help to B cells that have internalized the PAD4-citrullinated protein complex, thereby driving the production of both ACPAs and anti-PAD4 antibodies [7,17]. The specific interaction between autoreactive T cells and B cells via this hapten-carrier mechanism, leading to the production of dual autoantibodies, is schematically represented in Fig. 2. Furthermore, PAD gene polymorphisms have been associated with RA in existing literature [18–20].

Figure 2: The proposed mechanism by which B cells are activated to produce two distinct types of autoantibodies: ACPAs and anti-PAD4 antibodies. Step 1: Antigen capture and internalization. An autoreactive B cell recognizes and captures a complex formed by the PAD4 enzyme (carrier) bound to a citrullinated protein (hapten) via its surface B-cell receptor (BCR). The B cell subsequently internalizes and processes this complex. Step 2: Peptide presentation. The B cell processes the internalized complex and loads a specific peptide fragment derived from the PAD4 enzyme onto the MHC class II molecule (Human Leukocyte Antigen DR [HLA-DR]). This PAD4 peptide is then presented on the B cell surface. Step 3: T cell recognition and B Cell activation. An autoreactive T cell recognizes and binds to the PAD4 peptide presented by the MHC class II molecule. This recognition event activates the T cell, which in turn provides the necessary co-stimulatory signals to fully activate the B cell. Step 4: Dual autoantibody production. Upon activation, the B cell differentiates into plasma cells and generates two distinct types of autoantibodies: (1) ACPAs: targeting the citrullinated protein (hapten). (2) Anti-PAD4 antibodies: targeting the PAD4 enzyme (carrier) itself
Beyond ACPA production, PADs contribute to RA pathogenesis through other mechanisms. PAD enzymes are crucial for the development of NETs [5,21,22]. NETs are web-like structures composed of DNA, histones, and other granule proteins released by activated neutrophils [23,24]. While NETs participate in host defense against infections, their overproduction in inflammatory diseases like RA causes tissue damage and inflammation [25]. Notably, PAD4’s citrullination of histones is essential for NET formation, highlighting the direct link between PAD activity and NET-mediated inflammation in RA [23]. Mechanistically, upon neutrophil activation by stimuli such as calcium influx, PAD4 translocates to the nucleus [26]. There, it catalyzes the hypercitrullination of histones (particularly H3), which reduces their positive charge and weakens DNA-histone binding; this enzymatic step is crucial for driving chromatin decondensation and the subsequent release of NETs [26,27].
Several publications emphasize the involvement of PAD4, a specific PAD isozyme, in RA pathophysiology [2,16,28]. Neutrophils, as key inflammatory cells in the RA synovium, exhibit significant PAD4 expression [16,28–30]. PAD4 activity facilitates the production of NETs; specifically, PAD4-mediated citrullination of histones is necessary for chromatin decondensation, a crucial step in NET formation [22,23]. Since NETs can amplify inflammation and aggravate joint injury, PAD4 inhibition can reduce NET production, thereby attenuating inflammation. Strong evidence from animal models, such as the collagen-induced arthritis (CIA) model, also supports the role of PADs in RA pathogenesis. Studies showing a considerable reduction in disease severity using pan-PAD inhibitors, such as Cl-amidine, underscore the therapeutic potential of targeting PADs [31,32].
4 Structure, Function, and Regulation of PAD Enzymes
The PADs family comprises calcium-dependent enzymes that catalyze the post-translational conversion of peptidyl-arginine residues to peptidyl-citrulline [4,5]. This process, known as citrullination, alters protein structure and function, potentially leading to the generation of neo-epitopes that trigger autoimmunity [14]. The citrullination of various proteins, including fibrinogen and vimentin [1,14], is linked to the formation of ACPAs in RA.
The human genome encodes five PAD isozymes (PAD1–4 and PAD6) [21,33], each displaying distinct tissue distribution, subcellular localization, and substrate specificity [34]. While all PAD isozymes catalyze the citrullination of arginine residues, their specific roles in RA pathogenesis vary. The prominence of PAD4 in RA pathogenesis, compared to the more ubiquitously expressed PAD2, is thought to stem from several key differences. Critically, PAD4 possesses a unique nuclear localization signal [35,36], allowing it to enter the nucleus and citrullinate histones—the requisite step for the formation of pro-inflammatory NETs. In contrast, other isozymes like PAD2 are primarily cytoplasmic [37]. The exact contributions of other PAD isozymes, such as PAD2 and PAD3, to RA pathophysiology remain unclear compared to PAD4, despite their documented involvement in citrullination processes in the RA synovium [21,38]. Furthermore, intriguing clinical data suggest that while anti-PAD4 antibodies correlate with severe disease, anti-PAD2 antibodies are paradoxically associated with milder outcomes, implying distinct and non-redundant roles for these enzymes [39]. Further research is necessary to fully elucidate the interactions between various PAD isozymes and their overall effects on RA onset and progression.
The catalytic mechanism of PAD enzymes involves a unique reverse protonation mechanism, distinct from other hydrolases [34,40]. This mechanism critically involves an active site cysteine residue and a histidine residue, functioning as a nucleophile and general acid-base catalyst, respectively. PAD activity is tightly regulated, primarily by calcium ions [34,41]. An increase in intracellular calcium levels, often observed in inflammatory conditions like RA, is a critical determinant for PAD activation [23]. Calcium binding induces a series of conformational changes in PADs, leading to the formation of a catalytically competent active site [42]. However, PAD activity regulation extends beyond calcium. Recent studies highlight the role of bicarbonate as a pH-independent regulator of PAD activity. Bicarbonate, at physiological concentrations, enhances PAD activity independently of calcium levels and pH, suggesting a more complex regulatory network than previously appreciated. Further research is crucial to fully elucidate the interplay among calcium, bicarbonate, and other potential regulatory factors in controlling PAD activity within the context of RA. Understanding these regulatory mechanisms is vital for developing effective PAD inhibitors with enhanced specificity and therapeutic potential. The precise mechanisms controlling PAD isozyme expression and activity in the inflamed synovium of RA patients remain an active area of investigation, necessitating further studies to define the specific contributions of each PAD isozyme to RA pathogenesis and identify potential targets for selective inhibition.
5 The Contradictory Roles of Autoantibodies against PADs
Autoantibodies against PADs were discovered more than two decades ago [35]. Among them, autoantibodies against PAD4 (anti-PAD4 Abs) are the most well-documented. Anti-PAD4 Abs can exist before the clinical diagnosis of RA, and these antibodies correlate with the development of ACPAs.
The role of autoantibodies against PADs, particularly anti-PAD4 antibodies, represents one of the most significant ongoing debates in RA research. The evidence is highly contradictory, leading to the ‘foes or friends’ question. On one hand, a large body of evidence positions these antibodies as ‘foes’. Multiple studies have convincingly linked anti-PAD4 antibodies to more severe disease [43–45]. Furthermore, their presence has been associated with persistent disease activity and greater radiographic progression [46–49]. Furthermore, recent studies have provided direct mechanistic evidence for this pathogenic role, demonstrating that anti-PAD4 autoantibodies from RA patients can activate monocytes and exacerbate inflammatory arthritis [50].
In stark contrast, other studies present a paradoxical picture. A recent study focusing on RA-associated ILD found that anti-PAD4 antibodies were associated with less lung fibrosis and better overall survival, suggesting a context-dependent or even protective role in certain extra-articular manifestations [51].
This apparent contradiction is a critical area of investigation. The most likely explanation is that the anti-PAD4 antibody response is not uniform. These antibodies are heterogeneous; it is hypothesized that antibodies targeting different epitopes on the PAD4 enzyme may have opposing functions—some may activate the enzyme, while others may inhibit it [49,52].
Furthermore, the complexity is highlighted by other PAD isozymes. For instance, anti-PAD2 antibodies have also been identified, and they are associated with less disease severity, directly challenging the simple narrative that all anti-PAD autoimmunity is pathogenic [39]. In clinical practice, resolving this heterogeneity is crucial. Anti-PAD4 Abs may serve as a potential biomarker for diagnosis and prognosis in the future [35], but larger cohorts and standardized assays are necessary for validation.
6 PADs and RA Clinical Complications
The dysregulation of PAD activity in RA is not limited to arthritis or joint inflammation. Emerging evidence suggests a link between PADs and various RA complications. RA is a systemic autoimmune disease that can affect multiple organs and systems, leading to a range of extra-articular manifestations [53]. These extra-articular manifestations (EAMs) affect multiple organ systems, are significant predictors of mortality, and include major complications such as interstitial lung disease (ILD) and cardiovascular disease (CVD), where PAD involvement is an active area of research [54].
Interstitial lung disease (ILD) is a serious extra-articular complication of RA, characterized by inflammation and fibrosis of the lung tissue [1,53]. Several studies suggest a potential link between PAD activity and the development of RA-associated ILD [53,55]. Increased citrullination of lung proteins may contribute to inflammation and fibrosis [56], potentially through the generation of autoantigens or the disruption of normal lung tissue architecture. The presence of anti-PAD antibodies has been associated with a higher risk of ILD in RA patients [53], suggesting a direct involvement of PADs in the pathogenesis of this complication.
RA patients have a significantly increased risk of cardiovascular disease (CVD) [1,57]. This association is not fully explained by traditional risk factors and is strongest in autoantibody-positive patients. PAD enzymes and citrullination appear to play a multifaceted role in this process. PADs are known to citrullinate fibrinogen, which may promote aberrant clot formation. Mechanistically, citrullination alters the structural properties of fibrinogen. This modification leads to the formation of denser fibrin clots that are inherently resistant to plasmin-mediated fibrinolysis. As depicted in Fig. 3, this structural alteration results in the formation of denser fibrin clots that are resistant to plasmin-mediated lysis, thereby contributing to the pro-thrombotic state observed in RA patients. Consequently, this impairment in clot dissolution contributes to a persistent pro-thrombotic state, which has been clinically correlated with the severity of coronary artery disease in RA patients [58,59].

Figure 3: Effect of PAD-mediated fibrinogen citrullination on fibrin clotting and fibrinolysis. In normal physiology, healthy fibrinogen forms porous clots that are readily degraded by plasmin, ensuring efficient fibrinolysis and vascular patency. Conversely, in the pathological RA state, the PAD enzyme converts fibrinogen into citrullinated fibrinogen, which polymerizes into denser fibrin clots. These dense clots exhibit resistance to fibrinolysis by plasmin, a failure that drives two major systemic outcomes: a pro-thrombotic state and atherosclerosis. CVD: Cardiovascular diseases
However, the impact of PADs extends beyond thrombosis; there is growing evidence for their involvement in accelerating atherosclerosis. Immunohistochemistry has confirmed the presence of the PAD4 enzyme and various citrullinated proteins within human atherosclerotic plaques. While Western blot analysis has confirmed citrullinated fibrinogen (cFb) is present in these plaques, other proteins are also targeted. For instance, antibodies against citrullinated vimentin (cVim) have been associated with an increased plaque burden in RA patients, suggesting it is a relevant autoantigen in the vascular wall as well as the joint [60].
The mechanism linking autoantibodies to CVD is complex. Regarding the contribution of autoantibodies, the mechanism is complex. Regarding ACPA pathogenicity, rather than a direct cytotoxic effect, the link may be primarily mediated by the chronic inflammatory state that ACPAs signify [61]. Systemic inflammation itself is a primary driver of endothelial dysfunction [62,63], which is one of the earliest steps in CVD pathogenesis. This concept is supported by animal models [64], which have shown that PAD4 inhibition may prevent inflammation-mediated heart failure by reducing neutrophil activation.
7 Current Status and Challenges in PAD-Targeted Drug Development
The critical role of PADs in RA pathogenesis has driven significant efforts to develop PAD inhibitors as therapeutic agents [33,65]. Various classes of PAD inhibitors have been identified, including irreversible inactivators and reversible inhibitors [33]. Irreversible inactivators, such as Cl-amidine and F-amidine, covalently modify the active site cysteine residue of PAD enzymes, leading to irreversible inhibition [33]. These compounds have shown promise in preclinical models of RA, demonstrating a significant reduction in disease severity [31,32]. However, their irreversible nature raises concerns about potential off-target effects and toxicity. Reversible inhibitors, such as streptomycin and minocycline, bind to PADs non-covalently and are less likely to cause off-target effects [29]. However, their potency and selectivity may be lower compared to irreversible inactivators.
A significant challenge in PAD inhibitor development is achieving isozyme selectivity [34,65]. PADs exhibit distinct tissue distribution and substrate specificity, and inhibiting all isozymes simultaneously may lead to undesirable side effects. Therefore, the development of potent and selective inhibitors targeting specific PAD isozymes, particularly PAD4, is a major focus of current research efforts [32]. Significant progress has been made in designing PAD inhibitors with improved selectivity and potency. Structure-activity relationship (SAR) studies have been instrumental in optimizing the chemical structures of PAD inhibitors to enhance their affinity and selectivity for specific PAD isozymes [33,65]. For instance, the development of PAD2-selective inhibitors is of significant interest, considering the role of PAD2 in multiple sclerosis and other autoimmune diseases [65].
The use of high-throughput screening (HTS) and activity-based protein profiling (ABPP) has enabled the identification of novel PAD inhibitors with unique mechanisms of action [66]. These advanced techniques have significantly accelerated the drug discovery process and facilitated the identification of lead compounds for further optimization. A recent database study using in silico molecular docking simulations on the PAD4 enzyme found several potential FDA-approved medications [67], but further in vivo validation is crucial for clinical applications. A summary of these potential therapeutic agents is provided in Table 1.
The involvement of PADs in RA pathogenesis and complications has fueled significant interest in developing PAD-targeted therapies [4,74]. Several approaches to PAD inhibition are currently under investigation. Fig. 4 summarizes the distinct mechanisms of action for these therapeutic strategies, categorizing them into pan-PAD inhibitors, isozyme-selective inhibitors, biologics, and novel allosteric modulators.

Figure 4: Inhibitors targeting PAD enzymes utilize diverse mechanisms of action. 1. Pan-PAD inhibitors (e.g., Cl-amidine) act directly at the active site, demonstrating efficacy in preclinical models but generally lacking specificity. 2. Isozyme-selective inhibitors (e.g., GSK199) achieve precision by binding specifically to a target enzyme, such as PAD4, while sparing other isoforms (PAD1, PAD2). 3. Biologics/antibodies (e.g., AZD1163) employ large-molecule approaches, typically binding to the enzyme’s exterior to allosterically inhibit its function. 4. Novel inhibitor development strategy focuses on molecules that bind to an allosteric site (non-active site), inducing a conformational change that renders the enzyme inactive
Early research focused on the development of pan-PAD inhibitors, which target all PAD isozymes [4,33]. Cl-amidine is a well-studied example of a pan-PAD inhibitor that has shown efficacy in preclinical models of RA [31,33]. However, pan-PAD inhibitors lack isozyme specificity, potentially leading to off-target effects and limited therapeutic benefits [21,38]. Dakin and colleagues described a strategy of targeting a Ca2+-dependent allosteric binding site, which led to a more thorough inhibition of protein citrullination [75].
7.2 Isozyme-Selective Inhibitors
More recent efforts have focused on developing isozyme-selective inhibitors [21,38,65]. This approach aims to improve therapeutic efficacy and minimize adverse effects by targeting specific PAD isozymes implicated in RA pathogenesis, such as PAD4 [30]. Several promising PAD4-selective inhibitors have been identified, including GSK199, GSK484, and JBI-589 [30,32,69]. AFM-30a is a highly selective PAD-2 inhibitor that might also be a promising drug candidate [76]. These inhibitors have demonstrated efficacy in preclinical models of RA, providing hope for the development of more effective and safer therapies [30,32]. No significant toxicity was found in cell models [76]. Nevertheless, since PADs play important roles in host defense, such as NETosis, there is concern that these selective PAD inhibitors could increase bacterial survival [77].
7.3 Novel Inhibitor Development
Researchers are exploring various strategies to improve the efficacy and selectivity of PAD inhibitors [28]. These include the development of photo-switchable inhibitors [28], which can be activated by light at the target site, thereby improving spatial and temporal control over PAD inhibition [28]. Some researchers are investigating D-amino acid analogs of existing PAD inhibitors [38], which may exhibit enhanced bioavailability and stability. Bertran and colleagues used cyclic peptide screening and found several potential inhibiting peptides against PAD4 [78]. Li and colleagues have screened traditional Chinese medicine for PAD inhibition in silico, and they reported that salvianolic acid A from Salvia miltiorrhiza was the most potent compound in their study [71].
7.4 Potential Clinical Implications and Anticipated Effects of PAD Inhibition
To this date, no PAD inhibitor has proceeded to a human clinical trial, according to the clinicaltrials.gov website. Therefore, the discussion of their clinical implications is necessarily based on extrapolations from preclinical models and the known biological functions of PAD enzymes.
A primary clinical implication, and a significant safety concern, for the development of PAD inhibitors stems directly from their mechanism of action. Based on preclinical studies, a major anticipated risk is target-based toxicity, specifically related to immunosuppression [22].
PAD enzymes, particularly PAD4, are known to play a crucial role in host defense against infections with the formation of NETs. While the overproduction of NETs is a key driver of inflammation and joint damage in RA, their fundamental physiological role is in host defense. Therefore, the anticipated negative clinical effect of inhibiting PAD4 is the potential impairment of this critical defense mechanism. There is a concern, as noted in cell-based studies, that selective PAD inhibitors, by blocking NETosis, could inadvertently increase bacterial survival and lead to a heightened susceptibility to infections in patients [22,77]. This represents a significant clinical challenge. The very mechanism that makes PAD inhibitors therapeutically promising (reducing NET-mediated inflammation) is also the source of their greatest potential risk (impairing infection control). Consequently, any future human clinical trials for PAD inhibitors in RA must incorporate rigorous monitoring for infectious complications as a primary safety endpoint.
A significant potential clinical implication of PAD inhibition is the opportunity to treat not only the articular inflammation of RA, but also its systemic, extra-articular complications. Current preclinical evidence suggests that the therapeutic benefits of PAD inhibitors may extend beyond the joints [75,79]. For example, given the link between PAD activity and the pathogenesis of interstitial lung disease (ILD) [79], PAD inhibitors could represent a novel therapeutic strategy for this serious complication. Furthermore, PADs are implicated in the increased risk of CVD in RA patients [64], partly through the citrullination of proteins like fibrinogen. Animal models have indeed shown that PAD4 inhibition may prevent inflammation-mediated heart failure [80].
7.5 Challenges and Future Directions in PAD-Targeted Therapy for RA
Despite the encouraging preclinical results, several challenges remain in translating PAD-targeted therapies into effective clinical treatments for RA. These challenges include achieving sufficient potency, selectivity, bioavailability, and in vivo stability of PAD inhibitors [33,65,81]. The complexity of PAD regulation and the potential for off-target effects are also major hurdles [42]. Furthermore, the heterogeneity of RA and the involvement of multiple pathways in disease pathogenesis pose further complexities. To overcome these challenges, several research directions are being pursued. A deeper understanding of PAD regulation is crucial for the development of more effective inhibitors [42]. Further investigation into the interplay between calcium, bicarbonate, and other regulatory factors is necessary to identify potential points of intervention. The development of novel drug delivery systems to improve bioavailability and tissue penetration is also essential. The use of advanced screening technologies, such as HTS and ABPP, continues to play a vital role in identifying and optimizing new PAD inhibitors. The integration of proteomic analyses with PAD inhibition studies is crucial. The study of protein citrullination using modern mass spectrometry (MS) has become a vital approach for dissecting the molecular mechanisms of autoimmune diseases and cancer [11,14,82,83]. According to the latest MS strategies [84], current methods include: (a) the diagnostic marker approach for citrullination validation; (b) data search-assisted analysis; (c) enrichment strategies for specific modified peptides; (d) total proteome analysis; and (e) targeted fragmentation. These advanced techniques allow researchers to more accurately identify modification sites, enhance detection sensitivity, and reveal the functional roles of citrullination in disease. On a clinical and translational level, the identification of the “citrullinome” has critical value. For instance, ACPAs are the key serological markers for RA diagnosis and patient subtyping. This is not limited to RA; increased citrullination is seen in various cancers and other autoimmune diseases, suggesting its potential as a broader biomarker. The sensitivity of these techniques is high; for example, the use of biotin-PG chemical labeling successfully identified over 50 intracellular citrullinated proteins [85]. Furthermore, these proteomic approaches provide tangible, disease-specific insights that can directly guide clinical practice [86]. A study using LC-MALDI-TOF/TOF mass spectrometry identified 182 citrullinated peptides (at 200 sites) in the synovial fluid (SF) of RA patients. In stark contrast, only 3 peptides (at 4 sites) were detected in osteoarthritis (OA) synovial fluid [83]. This significant difference strongly supports the potential of using specific citrullinated peptides as disease-specific molecular biomarkers to differentiate RA from other arthritides (i.e., identify new disease subtypes) or to monitor treatment response. In summary, the integration of high-resolution MS strategies with chemical labeling and enrichment techniques is driving the systematic identification of citrullinated proteins. This provides a critical foundation for elucidating disease mechanisms and, most importantly, for discovering and validating the next generation of biomarkers essential for drug development and clinical practice.
Some experts proposed a modulating strategy of screening autoantibodies with allosteric binding sites adjacent to the PAD catalytic pocket [52]. Compared to small molecules, antibodies are more likely to inhibit PAD activity in the extracellular space without interfering with the normal activities of intracellular PADs. Moreover, a better understanding of the specific roles of different PAD isozymes in RA pathogenesis will aid in the development of isozyme-selective inhibitors, reducing potential off-target effects.
Given the involvement of PADs in other autoimmune diseases and even cancer, the development of effective and safe PAD inhibitors holds broad therapeutic potential beyond RA. Clinical trials are essential to evaluate the safety and efficacy of PAD inhibitors in RA patients and to determine their potential for improving patient outcomes [32]. The investigation of bacterial PADs, such as those from Porphyromonas gingivalis [87] and Helicobacter pylori [88], and their potential role in initiating or exacerbating RA is a promising area of research [15,89,90]. Understanding the interplay between bacterial and host PADs could lead to novel therapeutic strategies.
Despite significant progress, several challenges remain in the development of effective PAD-targeted therapies. These include:
Isozyme selectivity: Achieving high selectivity for specific PAD isozymes is crucial to minimize off-target effects and improve therapeutic efficacy [21,38].
Bioavailability and pharmacokinetics: Many PAD inhibitors, for example, have poor bioavailability and rapid clearance, limiting their therapeutic potential [38]. A prime example is the first-generation pan-PAD inhibitor, Cl-amidine. This compound is built on a benzoyl-L-arginine amide (BAA) scaffold, which mimics the structure of arginine. This design makes it highly susceptible to hydrolysis and inactivation by endogenous proteases, such as trypsin-like enzymes. Consequently, Cl-amidine suffers from very low in vivo stability and rapid metabolism. Indeed, studies have reported its serum half-life to be only approximately 15 min in mouse plasma, a significant hurdle that has limited its clinical translation and driven the development of second-generation inhibitors with improved stability [5,68].
In vivo efficacy: Demonstrating robust in vivo efficacy in relevant animal models is essential before proceeding to clinical trials [30,32].
Toxicity and safety: Ensuring the safety and tolerability of PAD inhibitors is critical for their clinical translation [4].
PADs are central to the pathogenesis of rheumatoid arthritis (RA) and its complications, contributing to both joint inflammation and extra-articular manifestations. Specifically, PADs, particularly PAD4, facilitate the generation of ACPAs and NET formation, alongside potentially other disease manifestations. Significant progress has been made in developing PAD inhibitors, with several promising candidates demonstrating efficacy in preclinical models [56,57]. However, the development of effective PAD-targeted therapies still faces significant challenges. These include achieving optimal potency, high isozyme selectivity, improved bioavailability, enhanced in vivo stability, and ensuring overall safety and tolerability [15]. Future research is crucial to fully elucidate the complex roles of PADs in RA, understand their intricate regulation, and identify novel drug targets. This includes focusing on PAD4-selective inhibitors and thoroughly investigating the role of PADs in extra-articular manifestations. Advancements in drug design, sophisticated screening methodologies, and innovative drug delivery strategies hold promise for future PAD-targeted therapies. The continued exploration of drug repositioning strategies, exemplified by the investigation of Saquinavir’s potential as a PAD4 inhibitor, also presents a viable path towards effective and cost-efficient RA treatments [67]. Furthermore, a deeper understanding of the complex interplay between PADs, the immune system, and the microbiome (particularly Porphyromonas gingivalis and its PPAD enzyme), along with the potential role of bacterial PADs, will be crucial for developing truly effective and personalized therapies [15,91,92]. Investigating additional regulatory factors like bicarbonate could also pave the way for novel therapeutic strategies [42]. Ultimately, success in translating the promise of PAD-targeted therapy into effective clinical treatments for RA and related autoimmune disorders will hinge on a comprehensive understanding of these complex interactions and the development of more sophisticated biomarkers [82,83,89]. Ultimately, overcoming these challenges is essential for therapeutic progress rather than academic interest alone. The successful translation of PAD-targeted strategies into clinical practice would introduce a first-in-class mechanism in RA treatment, with the potential to benefit patients who are refractory to current therapies and those burdened by extra-articular disease.
Acknowledgement: None.
Funding Statement: This research was funded by the grant TCVGH-1144401B from Taichung Veterans General Hospital. This work was financially supported by the National Science and Technology Council (Ministry of Science and Technology (MOST) 110-2313-B-005-042-MY3; NSTC 113-2320-B-005-010-MY3).
Author Contributions: The authors confirm their contribution to the paper as follows: study conception and design: Yung-Chieh Huang; draft manuscript preparation: Wen-Chien Cheng; review and editing: Ya-Hsuan Chao; visualization: Tzu-Ting Chen; supervision: Chi-Chen Lin. All authors reviewed the content and approved the final version of the manuscript.
Availability of Data and Materials: Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
References
1. Gottenberg JE. Gammaglobulins, autoantibodies, therapeutic antibodies and anti-drug antibodies in rheumatoid arthritis. Clin Exp Immunol. 2014;178 Suppl 1:115–7. doi:10.1111/cei.12534. [Google Scholar] [PubMed] [CrossRef]
2. Martinez-Prat L, Aure MA, Bentow C, Lucia D, Lopez-Hoyos M, Mahler M. OP0118 deciphering the anti-protein-arginine deiminase (PAD) response identifies PAD1 and PAD6 as novel autoantigens in rheumatoid arthritis. Ann Rheum Dis. 2020;79:78–9. doi:10.1136/annrheumdis-2020-eular.2853. [Google Scholar] [CrossRef]
3. Yu SF, Cheng TT, Huang GK, Hsu CY, Kao YH, Chung YH. Targeting FGFR3 is a useful therapeutic strategy for rheumatoid arthritis treatment. Curr Mol Pharmacol. 2024;17:e18761429261684. doi:10.2174/0118761429261684231002062505. [Google Scholar] [PubMed] [CrossRef]
4. Dreyton CJ, Jones JE, Knuckley BA, Subramanian V, Anderson ED, Brown SJ, et al. Optimization and characterization of a pan protein arginine deiminase (PAD) inhibitor. In: Probe reports from the NIH molecular libraries program. Bethesda, MD, USA: National Center for Biotechnology Information (US); 2010. [Google Scholar]
5. Bicker KL, Thompson PR. The protein arginine deiminases: structure, function, inhibition, and disease. Biopolymers. 2013;99(2):155–63. doi:10.1002/bip.22127. [Google Scholar] [PubMed] [CrossRef]
6. Yu K, Dillemans L, Gouwy M, Bessa H, Metzemaekers M, Martens E, et al. Novel method to quantify peptidylarginine deiminase activity shows distinct citrullination patterns in rheumatoid and juvenile idiopathic arthritis. Front Immunol. 2023;14:1111465. doi:10.3389/fimmu.2023.1111465. [Google Scholar] [PubMed] [CrossRef]
7. Auger I, Balandraud N, Massy E, Hemon MF, Peen E, Arnoux F, et al. Peptidylarginine deiminase autoimmunity and the development of anti-citrullinated protein antibody in rheumatoid arthritis: the hapten-carrier model. Arthritis Rheumatol. 2020;72(6):903–11. doi:10.1002/art.41189. [Google Scholar] [PubMed] [CrossRef]
8. Slade DJ, Fang P, Dreyton CJ, Zhang Y, Fuhrmann J, Rempel D, et al. Protein arginine deiminase 2 binds calcium in an ordered fashion: implications for inhibitor design. ACS Chem Biol. 2015;10(4):1043–53. doi:10.1021/cb500933j. [Google Scholar] [PubMed] [CrossRef]
9. Joshua V, Loberg Haarhaus M, Hensvold A, Wahamaa H, Gerstner C, Hansson M, et al. Rheumatoid arthritis-specific autoimmunity in the lung before and at the onset of disease. Arthritis Rheumatol. 2023;75(11):1910–22. doi:10.1002/art.42549. [Google Scholar] [PubMed] [CrossRef]
10. Krishnamurthy A. Molecular studies on the autoantibody-mediated bone destruction in rheumatoid arthritis [dissertation]. Stockholm, Sweden: Karolinska Institutet; 2024. [Google Scholar]
11. Sun M, Rethi B, Krishnamurthy A, Joshua V, Circiumaru A, Hensvold AH, et al. Anticitrullinated protein antibodies facilitate migration of synovial tissue-derived fibroblasts. Ann Rheum Dis. 2019;78(12):1621–31. doi:10.1136/annrheumdis-2018-214967. [Google Scholar] [PubMed] [CrossRef]
12. Gudmann NS, Hansen NU, Jensen AC, Karsdal MA, Siebuhr AS. Biological relevance of citrullinations: diagnostic, prognostic and therapeutic options. Autoimmun. 2015;48(2):73–9. doi:10.3109/08916934.2014.962024. [Google Scholar] [PubMed] [CrossRef]
13. Turesson C, Ronnelid J, Kastbom A. Autoantibodies as prognostic markers in rheumatoid arthritis. J Transl Autoimmun. 2025;10:100291. doi:10.1016/j.jtauto.2025.100291. [Google Scholar] [PubMed] [CrossRef]
14. Tilvawala R, Nguyen SH, Maurais AJ, Nemmara VV, Nagar M, Salinger AJ, et al. The rheumatoid arthritis-associated citrullinome. Cell Chem Biol. 2018;25(6):691–704 e6. doi:10.1016/j.chembiol.2018.03.002. [Google Scholar] [PubMed] [CrossRef]
15. Hitchon CA, Chandad F, Ferucci ED, Willemze A, Ioan-Facsinay A, van der Woude D, et al. Antibodies to porphyromonas gingivalis are associated with anticitrullinated protein antibodies in patients with rheumatoid arthritis and their relatives. J Rheumatol. 2010;37(6):1105–12. doi:10.3899/jrheum.091323. [Google Scholar] [PubMed] [CrossRef]
16. Martinez-Prat L, Palterer B, Vitiello G, Parronchi P, Robinson WH, Mahler M. Autoantibodies to protein-arginine deiminase (PAD) 4 in rheumatoid arthritis: immunological and clinical significance, and potential for precision medicine. Expert Rev Clin Immunol. 2019;15(10):1073–87. doi:10.1080/1744666x.2020.1668778. [Google Scholar] [PubMed] [CrossRef]
17. Balandraud N, Auger I, Roudier J. Do RA associated HLA-DR molecules bind citrullinated peptides or peptides from PAD4 to help the development of RA specific antibodies to citrullinated proteins? J Autoimmun. 2021;116:102542. doi:10.1016/j.jaut.2020.102542. [Google Scholar] [PubMed] [CrossRef]
18. Bagheri-Hosseinabadi Z, Mirzaei MR, Esmaeili O, Asadi F, Ahmadinia H, Shamsoddini B, et al. Implications of peptidyl arginine deiminase 4 gene transcription and polymorphisms in susceptibility to rheumatoid arthritis in an Iranian population. BMC Med Genom. 2023;16(1):104. doi:10.1186/s12920-023-01532-9. [Google Scholar] [PubMed] [CrossRef]
19. Suzuki A, Yamada R, Chang X, Tokuhiro S, Sawada T, Suzuki M, et al. Functional haplotypes of PADI4, encoding citrullinating enzyme peptidylarginine deiminase 4, are associated with rheumatoid arthritis. Nat Genet. 2003;34(4):395–402. doi:10.1038/ng1206. [Google Scholar] [PubMed] [CrossRef]
20. Lee YH, Rho YH, Choi SJ, Ji JD, Song GG. PADI4 polymorphisms and rheumatoid arthritis susceptibility: a meta-analysis. Rheumatol Int. 2007;27(9):827–33. doi:10.1007/s00296-007-0320-y. [Google Scholar] [PubMed] [CrossRef]
21. Subramanian V, Knight JS, Parelkar S, Anguish L, Coonrod SA, Kaplan MJ, et al. Design, synthesis, and biological evaluation of tetrazole analogs of Cl-amidine as protein arginine deiminase inhibitors. J Med Chem. 2015;58(3):1337–44. doi:10.1021/jm501636x. [Google Scholar] [PubMed] [CrossRef]
22. Chamardani TM, Amiritavassoli S. Inhibition of NETosis for treatment purposes: friend or foe? Mol Cell Biochem. 2022;477(3):673–88. doi:10.1007/s11010-021-04315-x. [Google Scholar] [PubMed] [CrossRef]
23. Zhou Y, An LL, Chaerkady R, Mittereder N, Clarke L, Cohen TS, et al. Evidence for a direct link between PAD4-mediated citrullination and the oxidative burst in human neutrophils. Sci Rep. 2018;8(1):15228. doi:10.1038/s41598-018-33385-z. [Google Scholar] [PubMed] [CrossRef]
24. Chapman EA, Lyon M, Simpson D, Mason D, Beynon RJ, Moots RJ, et al. Caught in a trap? Proteomic analysis of neutrophil extracellular traps in rheumatoid arthritis and systemic lupus erythematosus. Front Immunol. 2019;10:423. doi:10.3389/fimmu.2019.00423. [Google Scholar] [PubMed] [CrossRef]
25. Liu X, Arfman T, Wichapong K, Reutelingsperger CPM, Voorberg J, Nicolaes GAF. PAD4 takes charge during neutrophil activation: impact of PAD4 mediated NET formation on immune-mediated disease. J Thromb Haemost. 2021;19(7):1607–17. doi:10.1111/jth.15313. [Google Scholar] [PubMed] [CrossRef]
26. Thiam HR, Wong SL, Wagner DD, Waterman CM. Cellular mechanisms of NETosis. Annu Rev Cell Dev Biol. 2020;36:191–218. doi:10.1146/annurev-cellbio-020520-111016. [Google Scholar] [PubMed] [CrossRef]
27. Masucci MT, Minopoli M, Del Vecchio S, Carriero MV. The emerging role of neutrophil extracellular traps (NETs) in tumor progression and metastasis. Front Immunol. 2020;11:1749. doi:10.3389/fimmu.2020.01749. [Google Scholar] [PubMed] [CrossRef]
28. Mondal S, Parelkar SS, Nagar M, Thompson PR. Photochemical control of protein arginine deiminase (PAD) activity. ACS Chem Biol. 2018;13(4):1057–65. doi:10.1021/acschembio.8b00053. [Google Scholar] [PubMed] [CrossRef]
29. Doring Y, Soehnlein O, Weber C. Neutrophils cast NETs in atherosclerosis: employing peptidylarginine deiminase as a therapeutic target. Circ Res. 2014;114(6):931–4. doi:10.1161/circresaha.114.303479. [Google Scholar] [PubMed] [CrossRef]
30. Gajendran C, Fukui S, Sadhu NM, Zainuddin M, Rajagopal S, Gosu R, et al. Alleviation of arthritis through prevention of neutrophil extracellular traps by an orally available inhibitor of protein arginine deiminase 4. Sci Rep. 2023;13(1):3189. doi:10.1038/s41598-023-30246-2. [Google Scholar] [PubMed] [CrossRef]
31. Willis VC, Gizinski AM, Banda NK, Causey CP, Knuckley B, Cordova KN, et al. N-alpha-benzoyl-N5-(2-chloro-1-iminoethyl)-L-ornithine amide, a protein arginine deiminase inhibitor, reduces the severity of murine collagen-induced arthritis. J Immunol. 2011;186(7):4396–404. doi:10.4049/jimmunol.1001620. [Google Scholar] [PubMed] [CrossRef]
32. Willis VC, Banda NK, Cordova KN, Chandra PE, Robinson WH, Cooper DC, et al. Protein arginine deiminase 4 inhibition is sufficient for the amelioration of collagen-induced arthritis. Clin Exp Immunol. 2017;188(2):263–74. doi:10.1111/cei.12932. [Google Scholar] [PubMed] [CrossRef]
33. Causey CP, Jones JE, Slack JL, Kamei D, Jones LE, Subramanian V, et al. The development of N-alpha-(2-carboxyl)benzoyl-N(5)-(2-fluoro-1-iminoethyl)-l-ornithine amide (o-F-amidine) and N-alpha-(2-carboxyl)benzoyl-N(5)-(2-chloro-1-iminoethyl)-l-ornithine amide (o-Cl-amidine) as second generation protein arginine deiminase (PAD) inhibitors. J Med Chem. 2011;54(19):6919–35. doi:10.1021/jm2008985. [Google Scholar] [PubMed] [CrossRef]
34. Knuckley B, Causey CP, Jones JE, Bhatia M, Dreyton CJ, Osborne TC, et al. Substrate specificity and kinetic studies of PADs 1, 3, and 4 identify potent and selective inhibitors of protein arginine deiminase 3. Biochemistry. 2010;49(23):4852–63. doi:10.1021/bi100363t. [Google Scholar] [PubMed] [CrossRef]
35. Curran AM, Naik P, Giles JT, Darrah E. PAD enzymes in rheumatoid arthritis: pathogenic effectors and autoimmune targets. Nat Rev Rheumatol. 2020;16(6):301–15. doi:10.1038/s41584-020-0409-1. [Google Scholar] [PubMed] [CrossRef]
36. Li P, Li M, Lindberg MR, Kennett MJ, Xiong N, Wang Y. PAD4 is essential for antibacterial innate immunity mediated by neutrophil extracellular traps. J Exp Med. 2010;207(9):1853–62. doi:10.1084/jem.20100239. [Google Scholar] [PubMed] [CrossRef]
37. Zhou Y, Chen B, Mittereder N, Chaerkady R, Strain M, An LL, et al. Spontaneous secretion of the citrullination enzyme PAD2 and cell surface exposure of PAD4 by neutrophils. Front Immunol. 2017;8:1200. doi:10.3389/fimmu.2017.01200. [Google Scholar] [PubMed] [CrossRef]
38. Bicker KL, Anguish L, Chumanevich AA, Cameron MD, Cui X, Witalison E, et al. D-amino acid based protein arginine deiminase inhibitors: synthesis, pharmacokinetics, and in cellulo efficacy. ACS Med Chem Lett. 2012;3(12):1081–5. doi:10.1021/ml300288d. [Google Scholar] [PubMed] [CrossRef]
39. Darrah E, Giles JT, Davis RL, Naik P, Wang H, Konig MF, et al. Autoantibodies to peptidylarginine deiminase 2 are associated with less severe disease in rheumatoid arthritis. Front Immunol. 2018;9:2696. doi:10.3389/fimmu.2018.02696. [Google Scholar] [PubMed] [CrossRef]
40. Knuckley B, Bhatia M, Thompson PR. Protein arginine deiminase 4: evidence for a reverse protonation mechanism. Biochemistry. 2007;46(22):6578–87. doi:10.1021/bi700095s. [Google Scholar] [PubMed] [CrossRef]
41. Knuckley B, Causey CP, Pellechia PJ, Cook PF, Thompson PR. Haloacetamidine-based inactivators of protein arginine deiminase 4 (PAD4evidence that general acid catalysis promotes efficient inactivation. Chembiochem. 2010;11(2):161–5. doi:10.1002/cbic.200900698. [Google Scholar] [PubMed] [CrossRef]
42. Zhou Y, Mittereder N, Sims GP. Perspective on protein arginine deiminase activity-bicarbonate is a pH-independent regulator of citrullination. Front Immunol. 2018;9:34. doi:10.3389/fimmu.2018.00034. [Google Scholar] [PubMed] [CrossRef]
43. Halvorsen EH, Pollmann S, Gilboe IM, van der Heijde D, Landewe R, Odegard S, et al. Serum IgG antibodies to peptidylarginine deiminase 4 in rheumatoid arthritis and associations with disease severity. Ann Rheum Dis. 2008;67(3):414–7. doi:10.1136/ard.2007.080267. [Google Scholar] [PubMed] [CrossRef]
44. Harris ML, Darrah E, Lam GK, Bartlett SJ, Giles JT, Grant AV, et al. Association of autoimmunity to peptidyl arginine deiminase type 4 with genotype and disease severity in rheumatoid arthritis. Arthritis Rheum. 2008;58(7):1958–67. doi:10.1002/art.23596. [Google Scholar] [PubMed] [CrossRef]
45. Martinez-Prat L, Martinez-Taboada V, Santos C, Lopez-Hoyos M, Mahler M. Anti-protein-arginine deiminase 4 IgG and IgA delineate severe rheumatoid arthritis. Diagnostics. 2022;12(9):2187. doi:10.3390/diagnostics12092187. [Google Scholar] [PubMed] [CrossRef]
46. Navarro-Millan I, Darrah E, Westfall AO, Mikuls TR, Reynolds RJ, Danila MI, et al. Association of anti-peptidyl arginine deiminase antibodies with radiographic severity of rheumatoid arthritis in African Americans. Arthritis Res Ther. 2016;18(1):241. doi:10.1186/s13075-016-1126-7. [Google Scholar] [PubMed] [CrossRef]
47. Cappelli LC, Konig MF, Gelber AC, Bingham COIII, Darrah E. Smoking is not linked to the development of anti-peptidylarginine deiminase 4 autoantibodies in rheumatoid arthritis. Arthritis Res Ther. 2018;20(1):59. doi:10.1186/s13075-018-1533-z. [Google Scholar] [PubMed] [CrossRef]
48. Darrah E, Yu F, Cappelli LC, Rosen A, O’Dell JR, Mikuls TR. Association of baseline peptidylarginine deiminase 4 autoantibodies with favorable response to treatment escalation in rheumatoid arthritis. Arthritis Rheumatol. 2019;71(5):696–702. doi:10.1002/art.40791. [Google Scholar] [PubMed] [CrossRef]
49. Demoruelle MK, Wang H, Davis RL, Visser A, Hoang J, Norris JM, et al. Anti-peptidylarginine deiminase-4 antibodies at mucosal sites can activate peptidylarginine deiminase-4 enzyme activity in rheumatoid arthritis. Arthritis Res Ther. 2021;23(1):163. doi:10.1186/s13075-021-02528-5. [Google Scholar] [PubMed] [CrossRef]
50. Won T, Naik P, Wood MK, Wang H, Talor MV, Shi J, et al. Anti-peptidylarginine deiminase 4 autoantibodies derived from patients with rheumatoid arthritis exert pathogenic effects by activating monocytes and exacerbating inflammatory arthritis. Arthritis Rheumatol. 2025;77(9):1150–65. doi:10.1002/art.43168. [Google Scholar] [PubMed] [CrossRef]
51. Wilson TM, Solomon JJ, Humphries SM, Swigris JJ, Ahmed F, Wang H, et al. Serum antibodies to peptidylarginine deiminase-4 in rheumatoid arthritis associated-interstitial lung disease are associated with decreased lung fibrosis and improved survival. Am J Med Sci. 2023;365(6):480–7. doi:10.1016/j.amjms.2023.03.003. [Google Scholar] [PubMed] [CrossRef]
52. Zhou X, Kong S, Maker A, Remesh SG, Leung KK, Verba KA, et al. Antibody discovery identifies regulatory mechanisms of protein arginine deiminase 4. Nat Chem Biol. 2024;20(6):742–50. doi:10.1038/s41589-023-01535-8. [Google Scholar] [PubMed] [CrossRef]
53. Palterer B, Vitiello G, Del Carria M, D’Onofrio B, Martinez-Prat L, Mahler M, et al. Anti-protein arginine deiminase antibodies are distinctly associated with joint and lung involvement in rheumatoid arthritis. Rheumatology. 2023;62(7):2410–7. doi:10.1093/rheumatology/keac667. [Google Scholar] [PubMed] [CrossRef]
54. Cojocaru M, Cojocaru IM, Silosi I, Vrabie CD, Tanasescu R. Extra-articular manifestations in rheumatoid arthritis. Maedica. 2010;5(4):286–91. [Google Scholar] [PubMed]
55. Nava-Quiroz KJ, Lopez-Flores LA, Perez-Rubio G, Rojas-Serrano J, Falfan-Valencia R. Peptidyl arginine deiminases in chronic diseases: a focus on rheumatoid arthritis and interstitial lung disease. Cells. 2023;12(24):2829. doi:10.3390/cells12242829. [Google Scholar] [PubMed] [CrossRef]
56. Baka Z, Barta P, Losonczy G, Krenacs T, Papay J, Szarka E, et al. Specific expression of PAD4 and citrullinated proteins in lung cancer is not associated with anti-CCP antibody production. Int Immunol. 2011;23(6):405–14. doi:10.1093/intimm/dxr026. [Google Scholar] [PubMed] [CrossRef]
57. Bezuidenhout JA, Pretorius E. The central role of acute phase proteins in rheumatoid arthritis: involvement in disease autoimmunity, inflammatory responses, and the heightened risk of cardiovascular disease. Semin Thromb Hemost. 2020;46(4):465–83. doi:10.1055/s-0040-1709475. [Google Scholar] [PubMed] [CrossRef]
58. Varju I, Toth E, Farkas AZ, Farkas VJ, Komorowicz E, Feller T, et al. Citrullinated fibrinogen forms densely packed clots with decreased permeability. J Thromb Haemost. 2022;20(12):2862–72. doi:10.1111/jth.15875. [Google Scholar] [PubMed] [CrossRef]
59. Bezuidenhout JA, Venter C, Roberts TJ, Tarr G, Kell DB, Pretorius E. Detection of citrullinated fibrin in plasma clots of rheumatoid arthritis patients and its relation to altered structural clot properties, disease-related inflammation and prothrombotic tendency. Front Immunol. 2020;11:577523. doi:10.3389/fimmu.2020.577523. [Google Scholar] [PubMed] [CrossRef]
60. Sokolove J, Brennan MJ, Sharpe O, Lahey LJ, Kao AH, Krishnan E, et al. Brief report: citrullination within the atherosclerotic plaque: a potential target for the anti-citrullinated protein antibody response in rheumatoid arthritis. Arthritis Rheum. 2013;65(7):1719–24. doi:10.1002/art.37961. [Google Scholar] [PubMed] [CrossRef]
61. Montecucco F, Mach F. Common inflammatory mediators orchestrate pathophysiological processes in rheumatoid arthritis and atherosclerosis. Rheumatology. 2009;48(1):11–22. doi:10.1093/rheumatology/ken395. [Google Scholar] [PubMed] [CrossRef]
62. Yang X, Chang Y, Wei W. Endothelial dysfunction and inflammation: immunity in rheumatoid arthritis. Mediators Inflamm. 2016;2016:6813016. [Google Scholar] [PubMed]
63. Dijkshoorn B, de Mooij GW, Blanken AB, Popa CD, Nurmohamed MT. Endothelial cell dysfunction in RA: biomarkers IL-8, E-selectin, VCAM-1 and MCP-1 correlated with PET/CT. Rheumatology. 2025;64(9):4968–75. doi:10.1093/rheumatology/keaf208. [Google Scholar] [PubMed] [CrossRef]
64. Heger LA, Schommer N, Fukui S, Van Bruggen S, Sheehy CE, Chu L, et al. Inhibition of protein arginine deiminase 4 prevents inflammation-mediated heart failure in arthritis. Life Sci Alliance. 2023;6(10):e202302055. doi:10.26508/lsa.202302055. [Google Scholar] [PubMed] [CrossRef]
65. Muth A, Subramanian V, Beaumont E, Nagar M, Kerry P, McEwan P, et al. Development of a selective inhibitor of protein arginine deiminase 2. J Med Chem. 2017;60(7):3198–211. doi:10.1021/acs.jmedchem.7b00274. [Google Scholar] [PubMed] [CrossRef]
66. Knuckley B, Jones JE, Bachovchin DA, Slack J, Causey CP, Brown SJ, et al. A fluopol-ABPP HTS assay to identify PAD inhibitors. Chem Commun. 2010;46(38):7175–7. doi:10.1039/c0cc02634d. [Google Scholar] [PubMed] [CrossRef]
67. Thirugnanasambandham I, Jupudi S, Roychowdhury P, Karri V, Madhunapantula SRV, Singh SK, et al. Revamped role for approved drug: integrative computational and biophysical analysis of saquinavir’s peptidyl arginine deiminase 4 inhibition for rheumatoid arthritis. Biochem J. 2024;481(20):1379–93. doi:10.1042/bcj20240366. [Google Scholar] [PubMed] [CrossRef]
68. Knight JS, Subramanian V, O’Dell AA, Yalavarthi S, Zhao W, Smith CK, et al. Peptidylarginine deiminase inhibition disrupts NET formation and protects against kidney, skin and vascular disease in lupus-prone MRL/lpr mice. Ann Rheum Dis. 2015;74(12):2199–206. doi:10.1136/annrheumdis-2014-205365. [Google Scholar] [PubMed] [CrossRef]
69. Lewis HD, Liddle J, Coote JE, Atkinson SJ, Barker MD, Bax BD, et al. Inhibition of PAD4 activity is sufficient to disrupt mouse and human NET formation. Nat Chem Biol. 2015;11(3):189–91. doi:10.1038/nchembio.1735. [Google Scholar] [PubMed] [CrossRef]
70. Alturki MS, Gomaa MS, Tawfeeq N, Al Khzem AH, Shaik MB, Alshaikh Jafar M, et al. A Multifaceted computational approach to identify PAD4 inhibitors for the treatment of rheumatoid arthritis (RA). Metabolites. 2025;15(3):156. doi:10.3390/metabo15030156. [Google Scholar] [PubMed] [CrossRef]
71. Li Y, Gao C, Zhao J, Zhao Z, Xie B, Zuo H, et al. Screening of peptidyl arginine deiminase 4 inhibitors in traditional herbal medicines. Fitoterapia. 2024;177:106095. doi:10.1016/j.fitote.2024.106095. [Google Scholar] [PubMed] [CrossRef]
72. Sims DA, Andrews J, Kuriakose T, Scaletta L, Rajasinghe L, Sigurdardottir A, et al. OP0110 AZD1163—first-in-class anti-PAD2/4 bi-specific antibody to inhibit autoantigen formation in rheumatoid arthritis. Ann Rheum Dis. 2024;83:54. doi:10.1136/annrheumdis-2024-eular.3510. [Google Scholar] [CrossRef]
73. Chen B, Sun H, Ding N, Zhan C, Chen A. POS1017 A novel PAD2/4 bispecific antibody blocks pads activity in vitro and delays disease progress in cia-PADS model. Ann Rheum Dis. 2023;82:825. doi:10.1136/annrheumdis-2023-eular.2894. [Google Scholar] [CrossRef]
74. Kumar S. Highly efficient cell-penetrating probes of protein arginine deiminases for functional proteomics. Chembiochem. 2018;19(17):1806–9. doi:10.1002/cbic.201800257. [Google Scholar] [PubMed] [CrossRef]
75. Dakin LA, Xing L, Hall J, Ding W, Vajdos FF, Pelker JW, et al. Inhibiting peptidylarginine deiminases (PAD1-4) by targeting a Ca2+ dependent allosteric binding site. Nat Commun. 2025;16(1):4579. doi:10.1038/s41467-025-59919-4. [Google Scholar] [PubMed] [CrossRef]
76. Martin Monreal MT, Rebak AS, Massarenti L, Mondal S, Senolt L, Odum N, et al. Applicability of small-molecule inhibitors in the study of peptidyl arginine deiminase 2 (PAD2) and PAD4. Front Immunol. 2021;12:716250. doi:10.3389/fimmu.2021.716250. [Google Scholar] [PubMed] [CrossRef]
77. Baird R, Yusuf A, Forde L, Pohl K, Kavanagh K, Fitzpatrick F, et al. The vacuolar anti-Pseudomonal activity of neutrophil primary granule peptidyl-arginine deiminase enzymes. Front Immunol. 2024;15:1452393. doi:10.3389/fimmu.2024.1452393. [Google Scholar] [PubMed] [CrossRef]
78. Bertran MT, Walmsley R, Cummings T, Valle Aramburu I, Benton DJ, Assalaarachchi J, et al. A cyclic peptide toolkit reveals mechanistic principles of peptidylarginine deiminase IV regulation. Nat Commun. 2024;15(1):9746. doi:10.1101/2023.12.12.571217. [Google Scholar] [CrossRef]
79. Tsoyi K, Esposito AJ, Sun B, Bowen RG, Xiong K, Poli F, et al. Syndecan-2 regulates PAD2 to exert antifibrotic effects on RA-ILD fibroblasts. Sci Rep. 2022;12(1):2847. doi:10.1038/s41598-022-06678-7. [Google Scholar] [PubMed] [CrossRef]
80. Holthaus M, Xiong X, Eghbalzadeh K, Grossmann C, Geissen S, Piontek F, et al. Loss of peptidylarginine deiminase 4 mitigates maladaptive cardiac remodeling after myocardial infarction through inhibition of inflammatory and profibrotic pathways. Transl Res. 2025;280:1–16. doi:10.1016/j.trsl.2025.04.003. [Google Scholar] [PubMed] [CrossRef]
81. Mansouri P, Mansouri P, Behmard E, Najafipour S, Kouhpayeh SA, Farjadfar A. Peptidylarginine deiminase (PADa promising target for chronic diseases treatment. Int J Biol Macromol. 2024;278(Pt 3):134576. doi:10.1016/j.ijbiomac.2024.134576. [Google Scholar] [PubMed] [CrossRef]
82. Clancy KW, Weerapana E, Thompson PR. Detection and identification of protein citrullination in complex biological systems. Curr Opin Chem Biol. 2016;30:1–6. doi:10.1016/j.cbpa.2015.10.014. [Google Scholar] [PubMed] [CrossRef]
83. Wang F, Chen FF, Gao WB, Wang HY, Zhao NW, Xu M, et al. Identification of citrullinated peptides in the synovial fluid of patients with rheumatoid arthritis using LC-MALDI-TOF/TOF. Clin Rheumatol. 2016;35(9):2185–94. doi:10.1007/s10067-016-3247-4. [Google Scholar] [PubMed] [CrossRef]
84. Rebak AS, Hendriks IA, Nielsen ML. Characterizing citrullination by mass spectrometry-based proteomics. Philos Trans R Soc Lond B Biol Sci. 2023;378(1890):20220237. doi:10.1098/rstb.2022.0237. [Google Scholar] [PubMed] [CrossRef]
85. Lewallen DM, Bicker KL, Subramanian V, Clancy KW, Slade DJ, Martell J, et al. Chemical proteomic platform to identify citrullinated proteins. ACS Chem Biol. 2015;10(11):2520–8. doi:10.1021/acschembio.5b00438. [Google Scholar] [PubMed] [CrossRef]
86. Zhang L, Yang H, Liu J, Wang K, Cai X, Xiao W, et al. Metabolomics-based approach to analyze the therapeutic targets and metabolites of a synovitis ointment for knee osteoarthritis. Curr Pharm Anal. 2023;19:222–34. doi:10.2174/1573412919666221223152915. [Google Scholar] [CrossRef]
87. Oliveira SR, de Arruda JAA, Schneider AH, Bemquerer LM, de Souza RMS, Barbim P, et al. Neutrophil extracellular traps in rheumatoid arthritis and periodontitis: contribution of PADI4 gene polymorphisms. J Clin Periodontol. 2024;51(4):452–63. doi:10.1111/jcpe.13921. [Google Scholar] [PubMed] [CrossRef]
88. Wu H, Yuan H, Zhang J, He T, Deng Y, Chen Y, et al. Helicobacter pylori upregulates PAD4 expression via stabilising HIF-1alpha to exacerbate rheumatoid arthritis. Ann Rheum Dis. 2024;83(12):1666–76. doi:10.1136/ard-2023-225306. [Google Scholar] [PubMed] [CrossRef]
89. Xibillé-Friedmann DX, Martínez-Rivera JI, la Garza-Ramos De MA, González-Christen J, Carrillo-Vázquez S-M, Montiel-Hernandez J-L. FRI0052 Salivary peptidyl-arginine deiminase and tannerella forsythia are associated with clinical activity of rheumatoid arthritis. Ann Rheum Dis. 2017;76:498. doi:10.1136/annrheumdis-2017-eular.3894. [Google Scholar] [CrossRef]
90. Montgomery AB, Kopec J, Shrestha L, Thezenas ML, Burgess-Brown NA, Fischer R, et al. Crystal structure of Porphyromonas gingivalis peptidylarginine deiminase: implications for autoimmunity in rheumatoid arthritis. Ann Rheum Dis. 2016;75(6):1255–61. doi:10.1136/annrheumdis-2015-207656. [Google Scholar] [PubMed] [CrossRef]
91. Desclos-Theveniau M, Bonnaure-Mallet M, Meuric V. Protein arginine deiminase of oral microbiome plays a causal role in the polyarthritis rheumatoid initiating. Med Sci. 2020;36(5):465–71. [Google Scholar]
92. Hulzebos J. Rheumatoid arthritis & periodontitis: the role of neutrophils and porphyromonas gingivalis [master’s thesis]. Groningen, The Netherlands: University of Groningen; 2015. [Google Scholar]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF
 Downloads
Downloads
 Citation Tools
Citation Tools