 Open Access
Open Access
REVIEW
Photoreforming of Organic Waste into Hydrogen: Catalyst Design, Feedstock Valorization, and Future Perspectives
1 Department of Mechanical Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, Perak, Darul Ridzuan, Malaysia
2 Center of Renewable Intelligent Sustainable Power (CRISP), Universiti Teknologi PETRONAS, Bandar Seri Iskandar, Perak, Darul Ridzuan, Malaysia
* Corresponding Author: Mirna Omar. Email:
(This article belongs to the Special Issue: Green Hydrogen Technologies)
Energy Engineering 2026, 123(3), 3 https://doi.org/10.32604/ee.2026.072583
Received 30 August 2025; Accepted 08 January 2026; Issue published 27 February 2026
Abstract
Photoreforming is an emerging photocatalytic process that converts organic waste into hydrogen H2 using solar energy, offering a dual solution for waste valorization and sustainable fuel production. This review comprehensively examines the fundamental mechanisms of photoreforming, emphasizing the critical role of photocatalyst design in optimizing hydrogen evolution. Key criteria for effective photocatalysts including suitable band edge positions, broad spectrum solar absorption, and photostability are systematically analyzed alongside advances in heterojunction engineering and defect modulation. The review further explores diverse waste-derived feedstocks, such as biomass: alcohols, saccharides, lignin and plastics: PET, PLA, polyolefins, highlighting substrate, specific challenges and pretreatment strategies. Despite progress, challenges like catalyst deactivation, limited visible-light utilization, and scalability persist. Future directions advocate for robust photocatalyst engineering, mechanistic insights into charge dynamics, and scalable reactor designs to realize photoreforming’s potential as a sustainable hydrogen production technology.Graphic Abstract
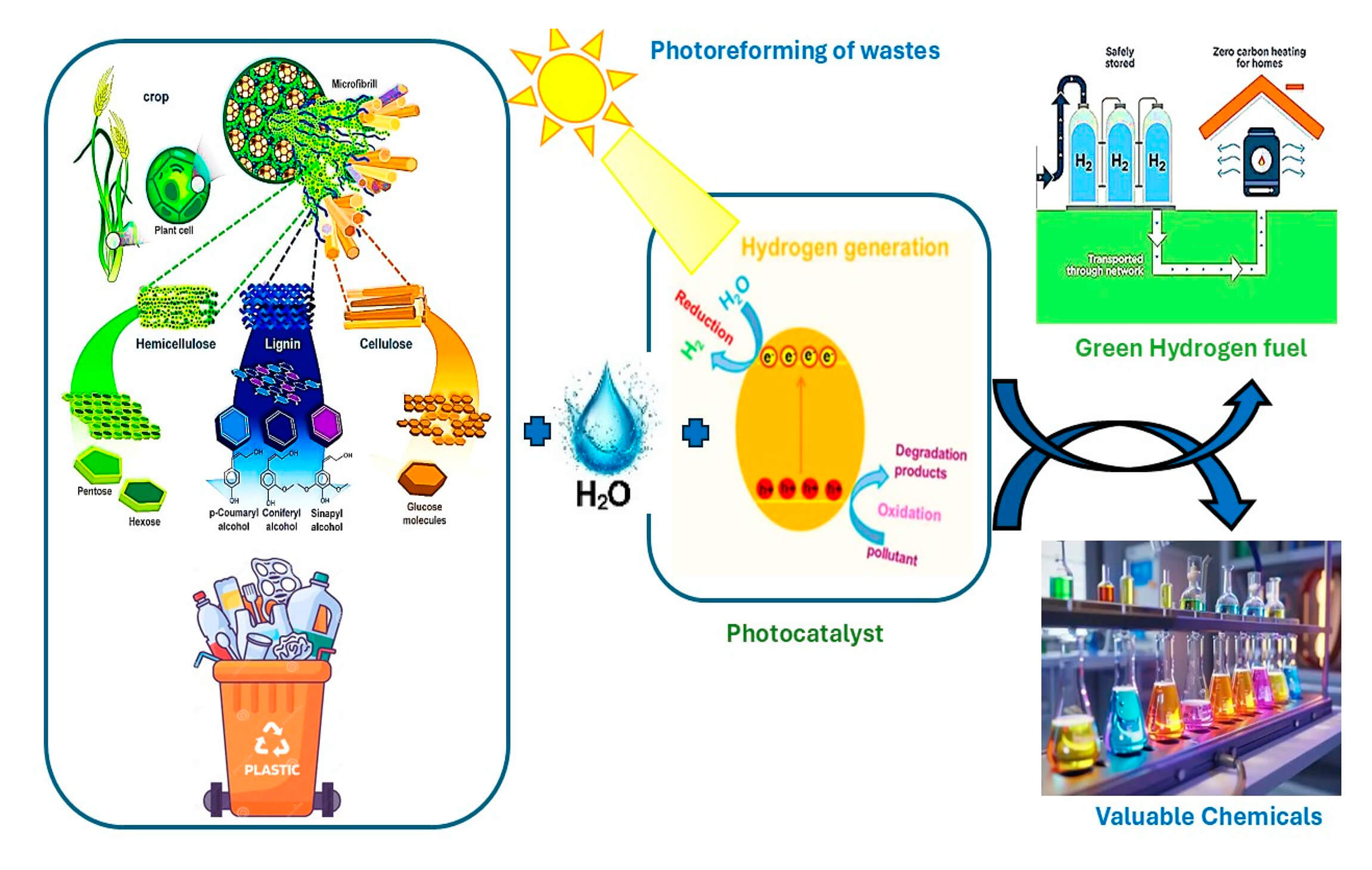
Keywords
The rising urgency to mitigate climate change and reduce dependence on fossil fuels has propelled the global pursuit of clean and sustainable energy technologies. Hydrogen, with its high energy density and zero carbon emissions upon combustion, has emerged as a pivotal energy carrier in the transition towards a low-carbon economy [1]. As governments and industries increasingly commit to hydrogen-based energy systems, the demand for cost-effective, environmentally benign, and scalable hydrogen production methods is intensifying [2]. Among the diverse hydrogen generation pathways, solar-driven photocatalytic water splitting has attracted considerable attention as a renewable and clean process harnessing abundant sunlight [3]. However, conventional pure water splitting often faces fundamental limitations, including sluggish oxygen evolution kinetics, high overpotentials, and low overall solar-to-hydrogen efficiency, which pose practical challenges for widespread adoption. To overcome these barriers, innovative approaches such as photoreforming have emerged, which utilize organic waste streams as sacrificial donors or hole scavengers to replace the demanding water oxidation half-reaction [4].
Photoreforming exploits semiconductor photocatalysts to mediate the simultaneous oxidation of organic waste substrates and the reduction of protons into molecular hydrogen under solar irradiation. The organic wastes serve multiple vital functions: acting as electron donors to consume photogenerated holes, thus significantly mitigating charge recombination losses; providing a feedstock for valorization through conversion into simpler, often valuable, chemical species; and contributing to environmental remediation by degrading pollutants and problematic wastes [5]. This dual functionality positions photoreforming not just as a green hydrogen production strategy but also as a promising waste-to-energy technology, aligning with circular economy and sustainability goals. The diversity of organic wastes applicable to photoreforming is staggering, spanning renewable biomass feedstocks (including alcohols, carbohydrates, lignin derivatives, and agricultural residues), recalcitrant plastic wastes, and complex industrial wastewater effluents containing dyes, pharmaceuticals, and other contaminants [6]. This complexity poses both an opportunity and a challenge: tailoring photocatalyst design and reaction conditions to suit distinct substrate chemistries and reaction pathways is essential for optimizing hydrogen yield, selectivity, and long-term catalyst stability [7]. Recent years have witnessed substantial progress in photocatalyst engineering aimed at improving solar light absorption, charge separation, and catalytically active sites. Strategies such as bandgap modulation through doping, creation of homo and heterojunctions, defect engineering, and hybridization with co-catalysts have collectively advanced photoreforming efficiency and stability [8]. Moreover, fundamental mechanistic insights into substrate-specific oxidation pathways and charge transfer dynamics have illuminated the critical factors that govern reaction performance and guide rational catalyst development. Nonetheless, several scientific and technical challenges remain [9]. These include overcoming catalyst deactivation caused by surface fouling or photocorrosion, broadening optical absorption to capture a greater portion of the solar spectrum, achieving efficient charge separation under real operational conditions, and managing the complexity of mixed and variable waste feedstocks [10]. Additionally, scalability and economic viability hinge upon the use of earth-abundant, non-toxic materials amenable to facile synthesis and sustainable lifecycle management [11]. This review delves into the fundamentals of photoreforming, focusing on photocatalyst design principles, bandgap engineering, heterojunction construction, and defect modulation to optimize charge carrier dynamics. We then evaluate waste-derived feedstocks, comparing biomass (e.g., methanol, glucose, lignin) and plastic waste (e.g., PET, PLA) in terms of reactivity, pretreatment needs, and H2 yields. Despite advancements, challenges like catalyst stability, solar spectrum utilization, and scalability hinder large-scale adoption. By addressing these gaps through innovative materials design and mechanistic studies, photoreforming could become a cornerstone of the circular economy.
2 Fundamentals of Photoreforming
Basic Mechanism of Photoreforming
At its essence, photoreforming is a photocnwac011e conversion of organic waste substrates into hydrogen gas H2. This process is built upon the interaction of light with semiconductor materials, which act as the catalysts for these transformations. When a photocatalyst absorbs photons that possess energy greater than or equal to its unique bandgap, it triggers a fundamental electronic excitation. This excitation propels electrons from the semiconductor’s valence band to its conduction band, simultaneously creating positively charged holes in the valence band. These photogenerated charge carriers migrate to the surface of the photocatalyst, where they engage in two simultaneous half-reactions; reduction-oxidation reactions [7]. The excited electrons e− in the conduction band reduce protons H+, which are essentially hydrogen ions present in water or the organic substrate itself, to generate molecular hydrogen H2 (Fig. 1A,B) [12]. Concurrently, the photogenerated holes H+ oxidize organic substrates such as those found in biomass, plastics, or other industrial residues, into simpler compounds, often carbon dioxide CO2, organic acids, or intermediates like ethylene glycol, depending on the specific conditions and catalyst design [6]. A significant advantage of photoreforming, especially when compared to traditional water splitting, is that the oxidation half-reaction is often kinetically and energetically more favourable than the oxidation of water [13].
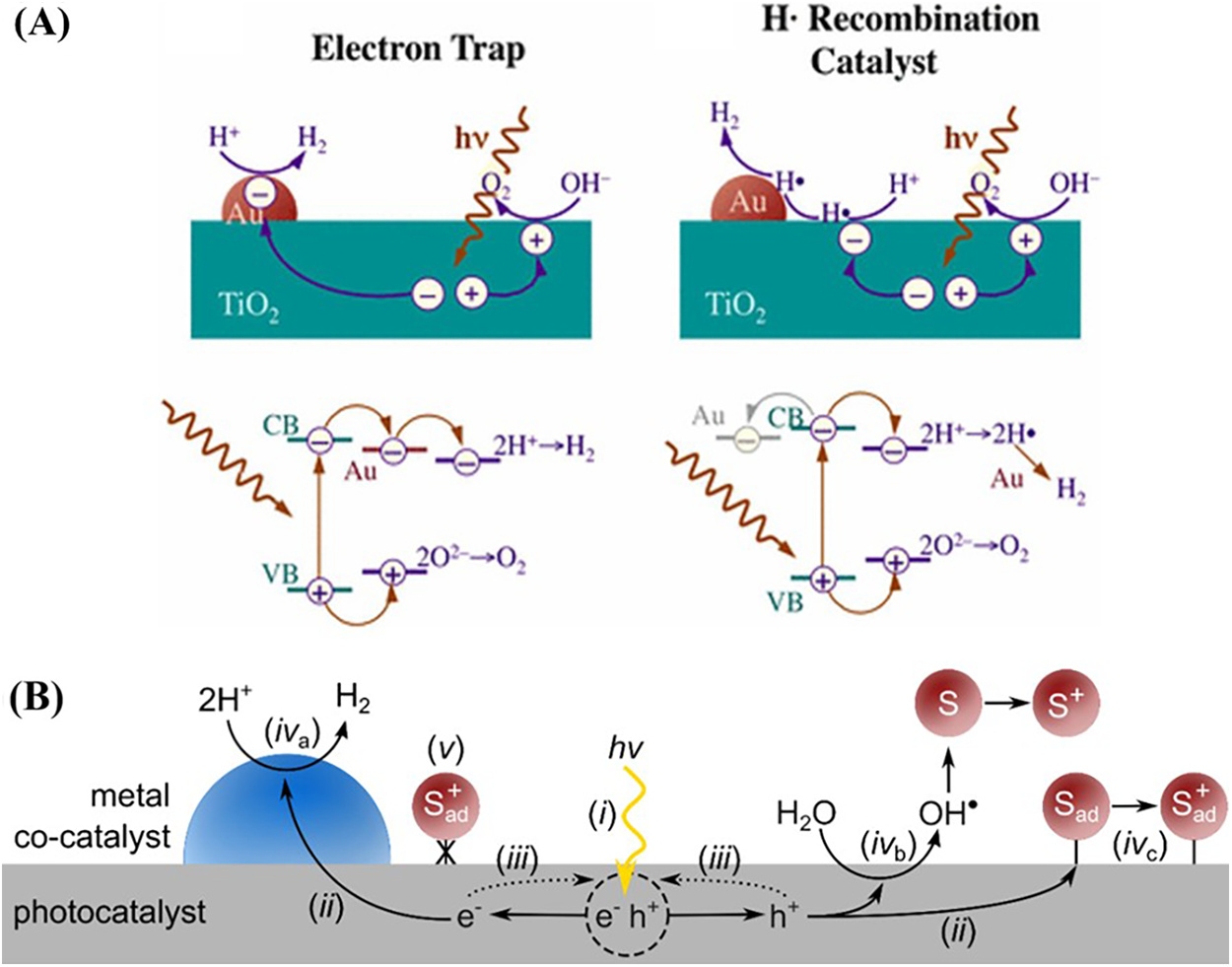
Figure 1: (A) Schematic representation of the role of Au in TiO2-based photocatalytic water splitting, illustrating electron trapping by Au and hydrogen evolution via interfacial charge transfer. Adapted from Ref. [14]. (B) General photocatalytic redox process showing light absorption, charge separation, and surface redox reactions. Adapted from Ref. [15].
3 The Role of Photocatalysts on Photoreforming H2 Generation
The selection and design of the photocatalyst are paramount to the success and efficiency of the photoreforming process. These materials must fulfil several critical criteria to effectively convert light energy into chemical energy such as Suitable Band Edge Positions, Broad Solar Spectrum Absorption, Photostability, availability and safety (Summarized in Table 1).
3.1 Suitable Band Edge Positions
For a photocatalyst to effectively facilitate hydrogen evolution reactions (HER), it must exhibit a bandgap and corresponding band edge positions that are meticulously tailored to enable the simultaneous oxidation of sacrificial donors and the reduction of protons [16]. The alignment of these energy levels governs the overall viability of the redox processes involved. For HER in photocatalytic systems, the photocatalyst’s band gap energy must surpass the minimum energy threshold required for water reduction (1.23 eV) [17]. In practical scenarios, however, it is crucial to consider energy losses due to overpotentials and inherent thermodynamic inefficiencies, which increases the demand for a band gap usually exceeding 1.8 eV. Nevertheless, materials with excessively large band gaps exhibit poor visible light utilization [18]. Consequently, the desirable theoretical band gap for photocatalysts involved in HER generally falls within the range of 1.8 to 3.2 eV [6]. Given that the absorption profile of a material is dictated by its band gap, it is essential to match the illumination source to the semiconductor’s optical properties during actual photoreforming operations. Furthermore, apart from the band gap magnitude, the precise positions of the conduction and valence bands play a pivotal role in HER activity. The conduction band (CB) must be positioned more negatively relative to the hydrogen reduction potential (Eo = −0.41 V vs. standard hydrogen electrode at pH = 7), while the valence band (VB) should be sufficiently positive to promote the oxidation of the organic substrate or sacrificial agent. Notably, the lower energy, longer-wavelength components of sunlight, such as visible and infrared, can be more effectively harnessed for hydrogen generation in waste photoreforming than in pure water splitting processes [19]. Despite attaining an optimal band gap and band edge configuration, photocatalyst performance may still be hampered by rapid recombination of photogenerated electron-hole pairs and a scarcity of active catalytic sites [17]. Addressing these challenges necessitates structural band gap engineering, which can be achieved via element doping, constructing heterojunctions or homojunctions, and incorporating targeted surface functionalization. Such band gap engineering strategies have been exemplified in the development of Zn1−XCdXS homojunction photocatalysts for biomass photoreforming. Zhao et al. reported a rationally designed Zn1−XCdXS solid solution homojunction photocatalyst with a pseudo-periodic twinning superlattice composed of cubic zinc blende (ZB) and hexagonal wurtzite (WZ) phases [20] (Entry 1 in Table 1). The optimized Zn0.6Cd0.4S photocatalyst exhibited a finely tuned bandgap and excellent photocatalytic performance under visible light irradiation without the need for any co-catalyst. It achieved a hydrogen evolution rate of 690 ± 27.6 μmol·h−1·g·cat−1, along with approximately 90% glucose conversion and a lactic acid selectivity of around 87%. The periodic WZ/ZB interfaces within the twinning superlattice facilitated efficient charge separation, while photogenerated superoxide radicals (.O2−) and holes primarily drove glucose oxidation and water oxidation, respectively. The construction of the homojunction markedly improved the separation of photoexcited charge carriers, as illustrated in (Fig. 2A). A notable advantage of this design is the capability to precisely adjust the band gap energy by varying the Zn-to-Cd molar ratio. As depicted in (Fig. 2B), the band gap gradually increases with rising Zn content in Zn1−XCdXS solid solutions, spanning from 2.25 eV for CdS up to 3.55 eV for ZnS. At x = 0.6, an optimized homojunction forms in Zn0.6Cd0.4S (Fig. 1A), which exhibits the highest hydrogen evolution rate of 0.69 mmol·g−1·h−1. This study highlights that precise band structure engineering and phase modulation within Zn1−XCdxS homojunctions effectively promote the simultaneous production of hydrogen and value-added chemicals from biomass substrates, offering a promising route for integrated solar to chemical energy conversion.
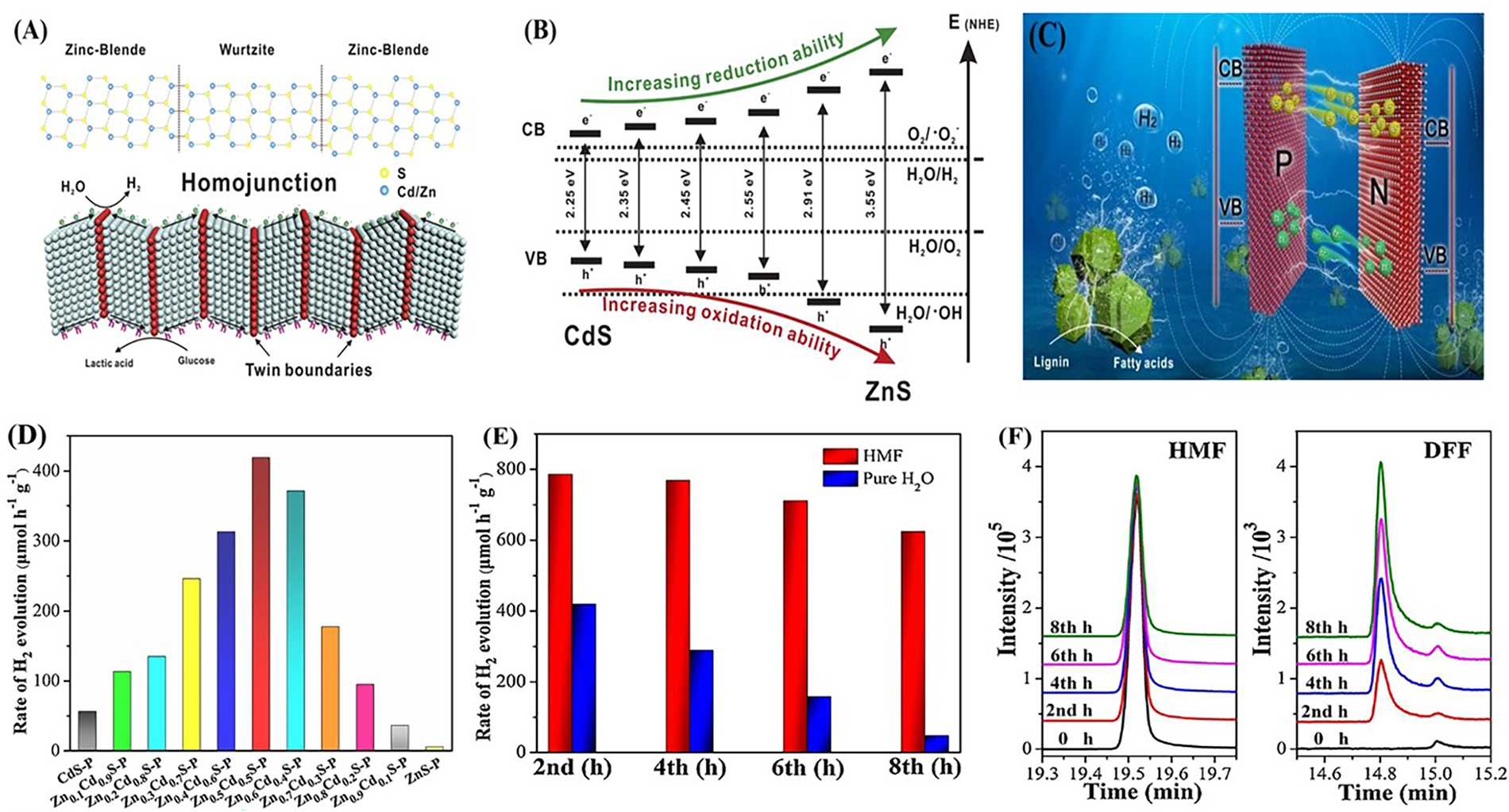
Figure 2: (A) Structural model of Zn1−XCdXS homojunction showing ZB/WZ twinning interfaces and charge migration pathways; (B) Bandgap energies and relative band edge positions of Zn1−XCdXS compositions. Adapted from Ref. [20]. (C) Schematic of the TiO2-NiO n-p heterojunction illustrating interfacial charge separation and photocatalytic redox reactions. Adapted from Ref. [21]. (D) Evaluation of hydrogen via photoreforming using P-doped ZnxCd1−XS; (E) Time-dependent H2 evolution in pure water and aqueous 5-HMF; (F) GC-MS analysis of 5-HMF oxidation products over Zn0.5Cd0.5S-P. Adapted from Ref [22].
The development of heterojunction and doped semiconductor systems for enhanced photocatalytic activity are echoed in band structure engineering. For instance, the integration of n-type and p-type semiconductors in an ultrafine core-shell heterojunction has proven to be an effective approach for promoting charge carrier separation and improving overall quantum efficiency. Zhao et al. established a TiO2-NiO core-shell structure wherein a uniform interface was achieved between the two phases, offering an efficient pathway for electron-hole separation [21] (Entry 2 in Table 1). Importantly, minimizing interfacial defects at the n-p junction introduces a strong internal electric field at the interface, substantially enhancing the separation and migration of photogenerated carriers. This internal field effectively drives the migration of electrons and holes to their respective reaction sites, thereby boosting the efficiency of redox processes during photocatalysis (Fig. 2C). Unlike conventional approaches where incompact interfacial contact leads to rapid carrier recombination, the tailored TiO2-NiO nanocomposite with a precise 3.25 wt% NiO loading exhibited excellent photocatalytic performance. Specifically, this system achieved a notable hydrogen generation rate of 23.5 mmol·h−1·g−1 during water splitting, while in lignin photoreforming, it delivered H2 production at 0.45 mmol·h−1·g−1 along with cogeneration of CH4 and a select amount of fatty acids (palmitic and stearic acid). The superior activity is credited to the synergy between the high crystallinity of TiO2 nanoparticles, the structural benefits of the core-shell interface, and the formation of an effective n-p heterojunction with NiO nanoclusters. Notably, the conduction and valence band positions of TiO2 (−0.92 eV, 2.54 eV) and NiO (−0.46 eV, 6.46 eV) were rationally aligned, ensuring favorable energetics for both water reduction and organic oxidation reactions. This work not only underlines the importance of band edge alignment and crystallinity but also demonstrates a scalable, noble-metal free strategy for coupling hydrogen evolution and value-added biomass conversion.
Further extending the utility of band gap and band edge modulation, recent advances in tailored solid solutions have enabled sacrificial-agent-free hydrogen production under visible light irradiation. Ye et al. reported the synthesis of P-doped ZnxCd1−XS solid solutions with abundant sulfur vacancies as an efficient photocatalyst for water splitting and coupled oxidation of 5-hydroxymethylfurfural (HMF) [22] (Entry 3 in Table 1). Here, interstitial phosphorus doping and the engineered S-vacancy defects markedly prolonged the lifetime of charge carriers and enhanced their separation, thus boosting the hydrogen evolution rate to 419 μmol·h−1·g−1 in pure water-substantially outperforming both ZnS-P and CdS-P counterparts by factors of 72 and 7.5, respectively (Fig. 2D). The strategic incorporation of HMF as a biomass-derived substrate not only facilitated additional charge transfer, further amplifying the HER (Fig. 2E), but also allowed for the concurrent production of valuable HMF oxidation derivatives (Fig. 2F). This example underscores the impact of electronic structure and defect engineering in broadening spectrum absorption, optimizing charge dynamics, and enabling simultaneous solar-driven hydrogen production and chemical valorization from renewable feedstocks.
Although substantial advances have been achieved in designing photocatalysts with suitable band structures for efficient hydrogen evolution, the development of catalysts that can effectively balance both reduction and oxidation potentials remains a critical challenge. Ensuring the valence band position is adequately matched to the oxidation of sacrificial substrates or biomass is essential not only for driving target reactions but also for preventing the buildup of undesirable byproducts that can deactivate the photocatalyst and reduce long-term stability [23]. Moreover, understanding the interplay between oxidation pathways and hydrogen generation efficiency across different photocatalytic systems is vital for optimizing overall performance. Future research should therefore focus on fine-tuning band edge alignments and charge carrier dynamics to realize catalysts that not only produce high hydrogen yields but also maintain selectivity and durability in diverse photocatalytic processes.
For practical and sustainable photocatalytic applications, effective utilization of the solar spectrum is paramount. Ideal photocatalysts should harvest not just ultraviolet (UV) light, but also visible and near-infrared (NIR) regions, which together constitute the majority of incident solar energy [18]. Traditional photocatalysts such as titanium dioxide (TiO2), despite being widely studied for their chemical stability, low toxicity, and affordability, are restricted by a wide band gap (about 3.2 eV for anatase) that primarily allows absorption in the UV region representing only a minor portion of sunlight. This intrinsic limitation curtails TiO2’s efficiency in photocatalytic processes, including hydrogen evolution, since solar-driven activity is hampered by limited absorption and facile recombination of photoinduced electron-hole pairs. To overcome these challenges, recent research has emphasized the modification of TiO2’s band structure in order to expand its optical response into the visible region and ultimately enhance solar utilization. Strategies such as doping, formation of heterojunctions, and defect engineering have been extensively explored. Among these, integrating TiO2 with suitable narrow-band-gap materials has shown notable promise in achieving broader spectral absorption and improved photocatalytic performance. An illustrative example is provided by Endo-Kimura et al. [24], who reported the fabrication of Ag2O/TiO2 heterojunctions (Entry 4 in Table 1) by a straightforward grinding technique, combining silver oxide with various titania photocatalysts. Extensive characterization, including diffuse-reflectance spectroscopy, confirmed that introducing a small amount of Ag2O (as low as 1 wt%) enables significant visible light response, even though the basic crystalline phases of the components remain unchanged. Photocatalytic testing revealed that these hybrid composites exhibit markedly enhanced activity under both UV/visible and pure visible-light irradiation. Under UV/vis irradiation, the Ag2O/TiO2 heterojunctions promoted the dehydrogenation of methanol and the oxidation of acetic acid, while under visible light only, they facilitated the breakdown of organic pollutants such as phenol and 2-propanol. The improved photocatalytic performance was attributed to more efficient charge separation, which may occur through either a Z-scheme mechanism or a classic p-n heterojunction, depending on the specific titania polymorph present. Notably, rutile containing composites demonstrated particularly strong visible-light activity, likely due to the more negative conduction band level of rutile enhancing interfacial electron transfer from Ag2O. In addition to their photocatalytic properties, these Ag2O/TiO2 materials displayed robust antimicrobial activities across bacterial and fungal strains, with visible light exposure further amplifying their effects. This enhanced antimicrobial action was linked to favorable surface charge interactions between the positively charged Ag2O (post electron transfer) and the negatively charged microbial cells. See (Fig. 3A,B) for the Photocatalytic efficiency of Ag2O/TiO2 composites with distinct titania frameworks under UV/vis illumination in acetic acid oxidation, accompanied by photographs of the samples after 45 min of irradiation.
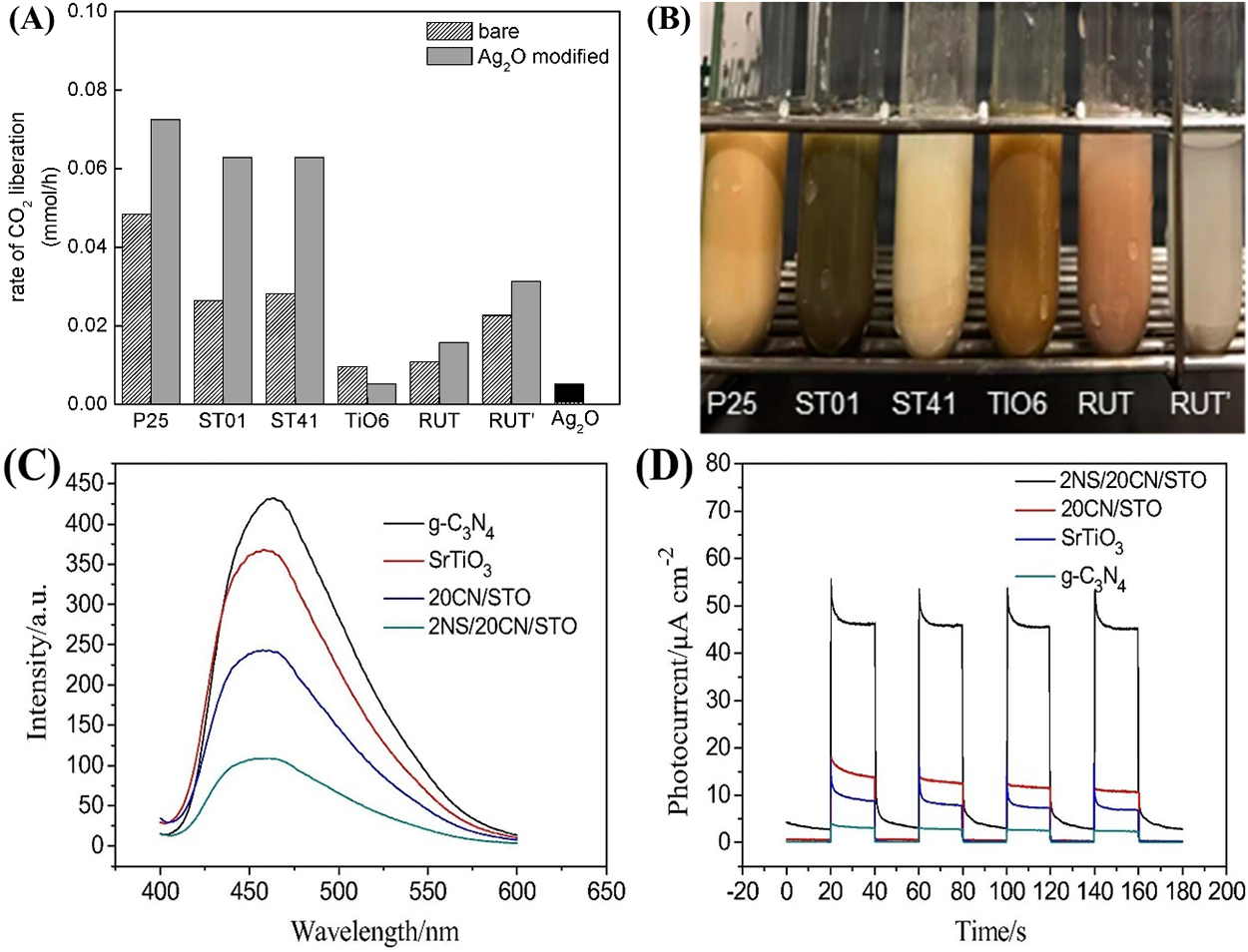
Figure 3: (A) Photocatalytic performance of Ag2O/TiO2 composites with varying titania matrices in acetic acid degradation under UV/vis light, and (B) visual appearance of the samples after 45 min of irradiation. Reproduced from Ref [24] copyright 2019 MDPI. (C) Photoluminescence PL emission characteristics and (D) photoelectrochemical I-t curves demonstrating the charge-carrier dynamics of SrTiO3, 20CN/STO, g-C3N4, and 2NS/20CN/ST. Reproduced from Ref. [25], Copyright 2018 Elsevier.
In addition, the development of multicomponent semiconductor composites has emerged as a promising approach for maximizing solar energy utilization. Luo et al. reported the synthesis of NiS/g-C3N4/SrTiO3 (NS/CN/STO) nanocomposites [25] (Entry 5 in Table 1) by a straightforward hydrothermal route. The unique assembly of these materials brings together the visible-light harvesting ability of C3N4, the UV-responsive SrTiO3, and the narrow band gap of NiS (0.5 eV), which functions as a potent cocatalyst for hydrogen evolution. The NS/CN/STO system demonstrates a remarkable enhancement in photocatalytic hydrogen production under UV-visible irradiation, achieving a hydrogen evolution rate of 1722.7 μmol·h−1·g−1 for the optimized 2%NiS/20%g-C3N4/SrTiO3 composite. This performance is vastly superior by factors of 32.8, 8.9, and 4.2 to those of pristine C3N4, SrTiO3, and the C3N4/SrTiO3 heterojunction, respectively. Comprehensive characterization including PL, and PC studies confirmed that both the heterojunction between SrTiO3 and C3N4 and the presence of NiS are responsible for promoting efficient charge separation and transfer (Fig. 3C,D). Notably, the observed catalytic enhancement is ascribed primarily to electronic effects rather than differences in surface area, underscoring the importance of smart band-alignment and interfacial engineering for solar-driven photocatalysis. The synergy achieved in this multicomponent system not only enables broader solar absorption spanning from the UV response of SrTiO3 to the visible-light sensitivity of C3N4 and NiS but also leads to a substantial extension of charge carrier lifetimes and a boost in overall hydrogen production efficiency.
3.3 Photostability, Availability and Safety
For practical and large-scale applications of photocatalysts, factors such as photostability, material availability, and safety are of paramount importance. A photocatalyst must maintain its structural integrity and catalytic performance over extended periods of light exposure to ensure long-term efficiency and economic viability. Furthermore, the widespread availability of precursor materials and the simplicity of synthesis routes greatly influence the scalability of photocatalyst production. Safety considerations, including environmental and health impacts, cannot be overlooked, especially for materials intended for sustainable energy and environmental applications, where minimizing toxicity and preventing secondary pollution are critical.
Within this context, graphitic carbon nitride g-C3N4 has emerged as a highly promising photocatalyst platform. Synthesized from abundant and inexpensive precursors such as urea, melamine, or dicyandiamide, g-C3N4 benefits from commercial availability and cost-effectiveness. Its synthesis pathway is notably straightforward and amenable to large-scale production, making it accessible for various practical uses. Importantly, g-C3N4 is metal-free and non-toxic, presenting an environmentally benign alternative to many traditional photocatalysts that contain toxic heavy metals or rare elements. This aspect significantly reduces potential environmental hazards and health risks associated with photocatalyst deployment. In addition to its favorable safety profile and synthesis advantages, g-C3N4 is widely recognized for its excellent photostability under visible light irradiation. It resists photodegradation, maintaining its catalytic activity during prolonged light exposure, a critical feature for sustainable photocatalytic operations such as hydrogen evolution, water splitting, and waste photoreforming. Moreover, heterojunction engineering has proven to enhance the performance of g-C3N4 in photocatalytic applications. Combining g-C3N4 with complementary semiconductors has led to improvements in hydrogen evolution efficiency by enhancing visible light absorption, prolonging charge carrier lifetime, and increasing the density of active catalytic sites. Notably, g-C3N4/perovskite type oxide heterojunctions have attracted significant interest due to their synergistic properties and scalability. A study by Wang et al. demonstrated the successful synthesis of a Z-scheme LaCoO3/g-C3N4 heterostructure [26] (Entry 6 in Table 1) through simple solvothermal methods, utilizing LaCoO3 prepared via an accessible sol-gel process. This system leverages the abundant and low-cost nature of both g-C3N4 (derived from inexpensive precursors) and LaCoO3, a non-noble metal oxide known for its tunable band gap and robust photocorrosion resistance. Hydrogen production rates of LaCoO3, g-C3N4, and their composites with various mass ratios were systematically measured, with each sample tested in triplicate to ensure data reliability. The results clearly demonstrate distinct photocatalytic performances among the samples. Pure LaCoO3 and g-C3N4 showed hydrogen evolution rates of 298.17 and 732.65 μmol·h−1·g−1, respectively. Notably, the LaCoO3/g-C3N4 composite containing 20 wt% g-C3N4 exhibited the highest activity, achieving an average hydrogen generation rate of 1046.15 μmol·h−1·g−1 approximately 3.5 and 1.4 times greater than those of LaCoO3 and g-C3N4 alone (Fig. 4A). The photocatalyst also demonstrated excellent stability, as evidenced by negligible decline in hydrogen production after four consecutive cycles, confirming its durability under full-spectrum xenon lamp irradiation (Fig. 4B). Another study reported by Liu et al. explored the integration of MoS2 quantum dots (MSQDs) with g-C3N4 nanosheets to form a 0D/2D heterojunction photocatalyst, further enhancing photostability and safety while improving catalytic performance [27] (Entry 7 in Table 1). The MSQDs were synthesized via a facile one-pot hydrothermal process, yielding uniformly dispersed, water-stable quantum dots that decorated the g-C3N4 surface to form intimate p-n heterojunction interfaces. The enhanced performance, including a significant increase in hydrogen evolution rates over pristine g-C3N4, is detailed in (Fig. 4C). Recycling experiments over three consecutive 12-h runs revealed only marginal decreases in activity, which were attributed primarily to the gradual photo-oxidation of MoS2 rather than degradation of the composite structure itself (Fig. 4D). Structural characterization by X-ray diffraction and FT-IR spectroscopy confirmed the preservation of the g-C3N4 framework after repeated photocatalytic cycles, underscoring the protective role of g-C3N4 in mitigating photocorrosion of the sulfide component. The synergy enables a photocatalyst free from precious or hazardous metals an attractive feature for scalable, green hydrogen production technologies. Continued advancements in material engineering and heterojunction optimization are expected to further refine the functionality of g-C3N4-based photocatalysts, paving the way for their broader application in renewable energy conversion and storage technologies.
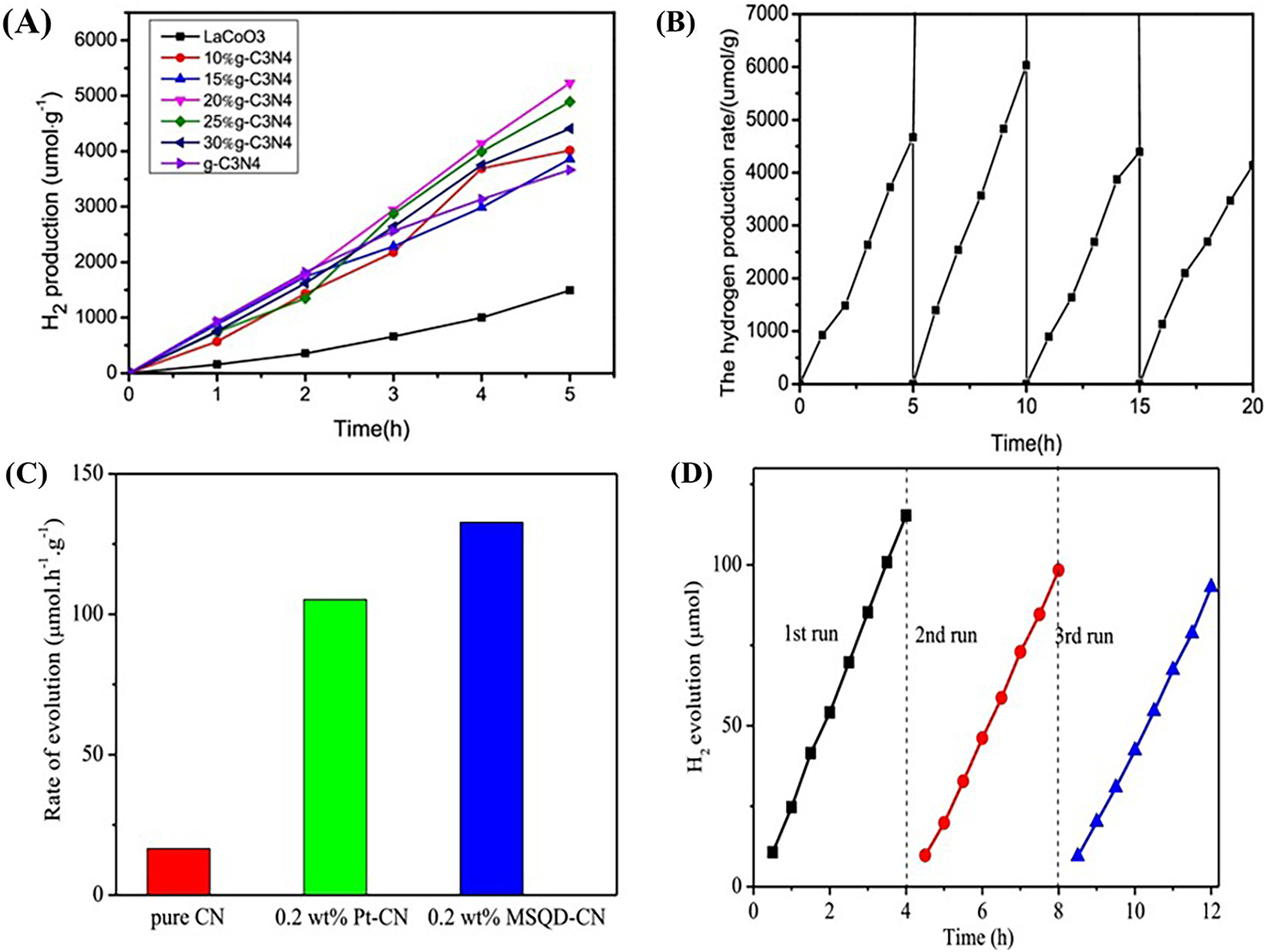
Figure 4: (A) Time-dependent hydrogen evolution over LaCoO3, g-C3N4, and LaCoO3/g-C3N4 composites under full-spectrum irradiation; (B) Long-term stability of LaCoO3/20% g-C3N4 during a 20 h photocatalytic run. Adapted from Ref [26]. (C) Hydrogen evolution rates of g-C3N4 modified with different co-catalysts; (D) Cycling stability of 5 wt% MSQD-CN in methanol-water solution. Adapted from Ref [27].
4 Waste-Derived Feedstocks for Photoreforming
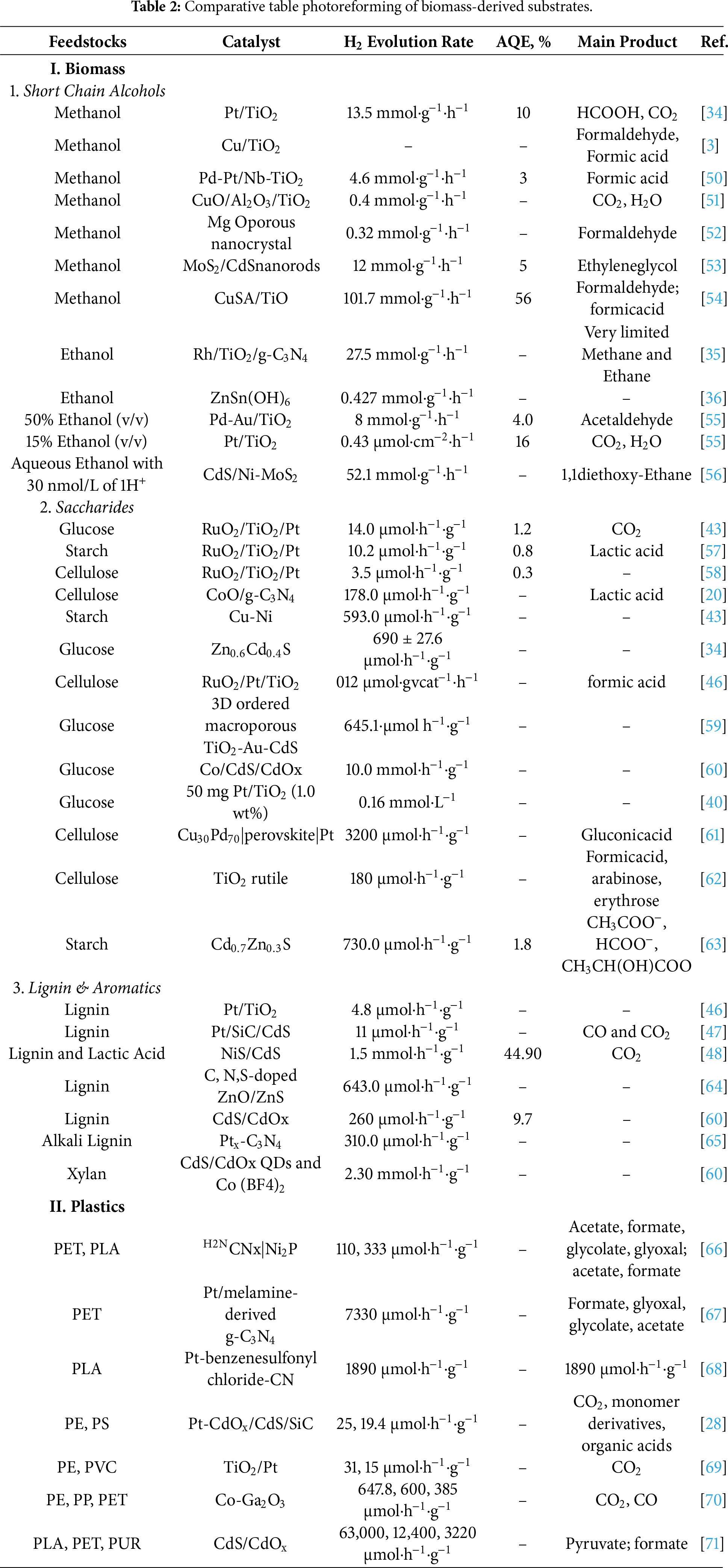
A central advantage of photoreforming lies in its ability to valorize a broad spectrum of organic waste streams as sacrificial electron donors, thereby coupling waste remediation with sustainable hydrogen production [4]. These waste substrates not only provide the carbon backbone for oxidation but also act as efficient hole scavengers, reacting preferentially with photogenerated valence band holes to suppress charge carrier recombination and enhance the net electron flux toward hydrogen evolution [28]. The kinetics and thermodynamics of this oxidation process are often more favorable than water oxidation, making organic-rich waste particularly effective in boosting H2 yields [29]. First, biomass-derived substrates (Table 2), including short-chain alcohols (methanol, ethanol), saccharides, and lignin derivatives (Fig. 5A), have been widely studied due to their renewable origin and rich carbon-hydrogen content [30]. Second, synthetic plastic wastes (Table 2) such as polyethylene terephthalate (PET), polyethylene (PE), and polypropylene (PP) are gaining attention owing to their environmental persistence and carbon-rich structure, making them promising candidates for photoreforming-based valorization [31,32]. These categories offer distinct challenges and mechanistic pathways, requiring tailored catalyst designs and reaction strategies for effective hydrogen production [33]. The following subsections provide an in-depth analysis of these waste categories, drawing on recent literature to elucidate substrate-specific photoreforming mechanisms, efficiency metrics, and catalytic system design.
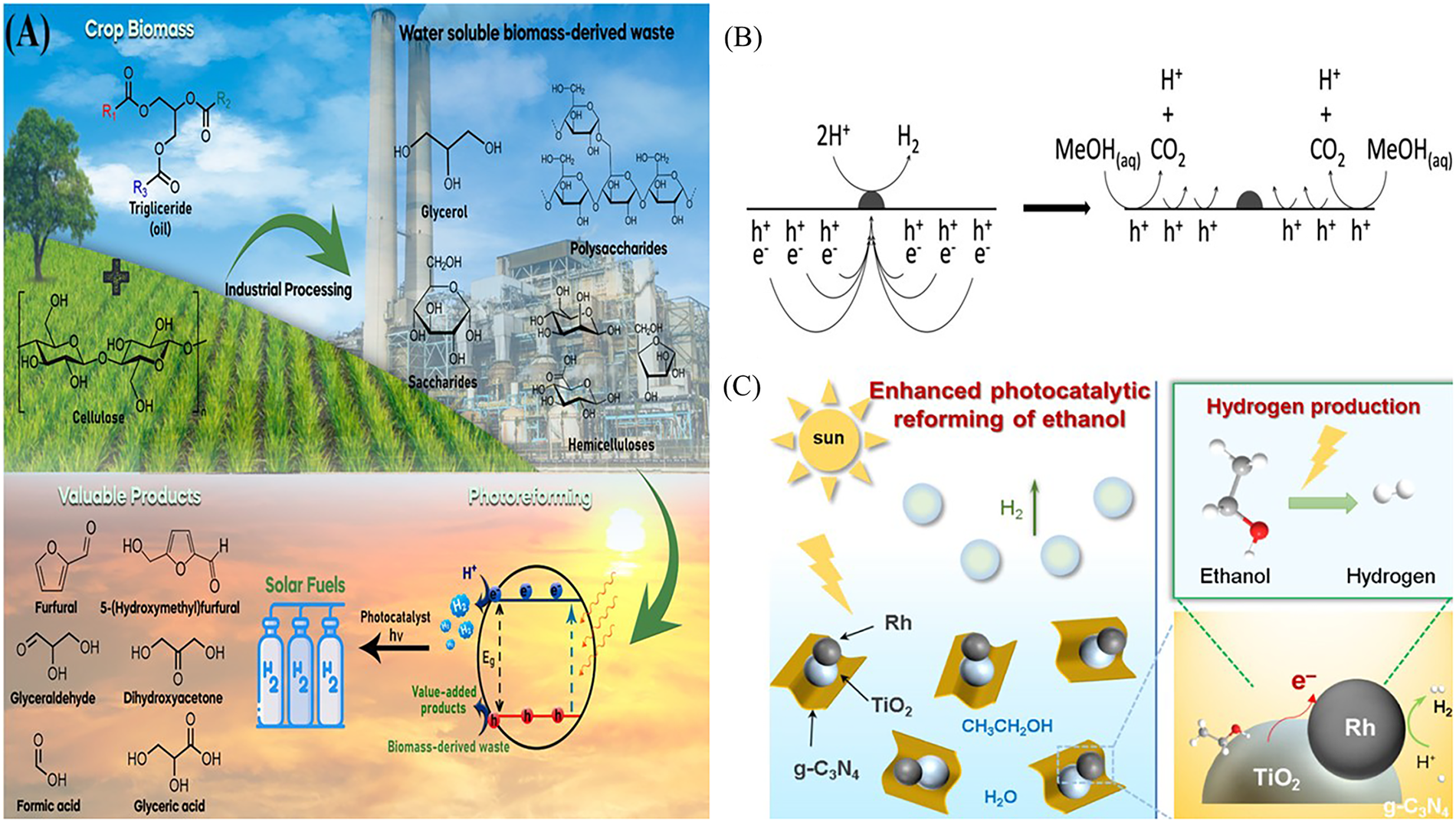
Figure 5: (A) Heterogeneous photocatalytic reforming of biomass-derived compounds. Copyright 2024 Elsevier [37]. (B) Methanol photoreforming. Copyright 2019 Elsevier [38]. (C) Catalytic photoreforming of Ethanol with a Rh/TiO2/g-C3N4 ternary system. Copyright 2024 ACS Applied Materials & Interfaces [35].
4.1 Biomass-Derived Substrates
Biomass photoreforming leverages a wide variety of organic substrates as both renewable feedstocks and effective hole scavengers. The nature of these substrates, short-chain alcohols, saccharides, and lignin derivatives, not only governs the efficiency of hydrogen production but also shapes the mechanistic pathways and design requirements for advanced catalytic systems. Below, recent literature and studies provide detailed comparisons of hydrogen evolution from various biomass feeds, highlighting mechanistic insights, efficiency metrics, and guiding principles for substrate-specific catalytic design.
4.1.1 Short Chain Alcohols: Methanol and Ethanol
Simple C1–C2 alcohols have high hydrogen to carbon ratios and are readily oxidized by photogenerated holes. In water, methanol and ethanol are oxidized to CO2 (and/or intermediate organics) while producing H2 from water reduction. Methanol is widely regarded as the standard for high yield hydrogen production in photoreforming. Its simple molecular structure (CH3OH) permits rapid, efficient oxidative removal of photogenerated holes, greatly suppressing electron-hole recombination. Consequently, methanol-based systems often achieve the highest H2 yields among biomass derivatives. Its low ionization energy and high-water miscibility further enhance its reactivity. In methanol photoreforming, holes oxidize CH3OH to CH2O (formaldehyde) and then to HCOOH and CO2, while electrons reduce protons to H2 (Fig. 5B). For example, methanol photoreforming on TiO2 yields CO2 and 3H2 per mol CH3OH (ΔG° = +8 kJ/mol) [34]. In practice, noble-metal or Cu-modified TiO2 catalysts are used to enhance charge separation and selectivity. On Pt/TiO2 or Cu/TiO2, methanol photoreforming achieve high quantum yields (e.g., =10% QE at 365 nm) [3] with essentially only H2 as the gaseous product; carbon remains as liquid organics (formaldehyde/formic acid) when Cu/TiO2 is used [3]. By contrast, ethanol (C2H5OH), being larger, typically gives slower H2 generation. During ethanol photoreforming, photo-oxidation proceeds via: CH3CH2OH → CH3CHO (acetaldehyde) → CH3COOH (acetic acid) → CO2, with each step coupled to H2 release. Byproducts of alcohol can accumulate and block active sites or require downstream separation. Photocatalysts used are usually UV-active oxides (anatase/rutile TiO2), though some studies employ visible light absorbers (e.g., Au or Rh on TiO2, carbon nitrides) to harvest more of the solar spectrum. For example, with Rh/TiO2/g-C3N4 composite (Fig. 5C), ethanol photoreforming achieved about 27.5 mmol·h−1·g−1 [35], and a ZnSn (OH)6 nanoparticle catalyst gave about 0.427 mmol·h−1·g−1 [36].
4.1.2 Saccharides (Monosaccharides and Polysaccharides)
Saccharides, especially monosaccharides like glucose and disaccharides, yield moderate hydrogen quantities in photoreforming. Polysaccharides (cellulose, starch) present greater challenges due to their highly polymerized C–C backbones. Efficient photoreforming of saccharides absolutely requires catalysts capable of promoting stepwise C–C bond cleavage and subsequent oxidation of the derived intermediates [28,39]. Under UV or visible light, TiO2-based photocatalysts oxidatively cleave C–C bonds in sugar chains (Fig. 6A). A well-established mechanism is sequential α-scission at the C1–C2 bond: e.g., glucose (C6) is oxidized to arabinose (C5) + formic acid in the first step [34]. In this way, photoreforming of a hexose yields successively smaller sugars (C5, C4, …) and formic acid, ultimately giving CO2 and H2. A key finding is that H2 is produced via dehydrogenation coupled with C–C cleavage [2]. In situ spectroscopy shows formic acid/formate strongly adsorbed on TiO2; Pt-loaded TiO2 (Fig. 6B) can oxidize these, boosting net H2 (since TiO2 alone cannot fully convert adsorbed formate) [34,40,41]. Thus, platinum co-catalysts tend to increase H2 selectivity in sugar reforming by handling the formic acid step [42]. Reaction kinetics are profoundly impacted by substrate solubility, biomass pretreatment, and the catalyst’s ability to facilitate mass transfer and surface reactions. Saccharide reforming rates are generally lower than for alcohols. Classic work reported H2 formation rates under UV: 14.0 μmol·h−1·g−1 from glucose, 10.2 μmol·h−1·g−1 from starch, but only 3.5 μmol·h−1·g−1 from cellulose (all using RuO2/TiO2/Pt) [34,43]. This shows that simple sugars yield more H2 than polymeric cellulose under comparable conditions. Modern studies similarly find that monomeric sugars give higher H2 rates per gram than bulk biomass. The complexity of polysaccharides slows reaction (requiring depolymerization to glucose first) and produces carbon-rich intermediates that tie up holes. Thermodynamically, saccharide reforming can be favorable (e.g., ΔG° of sucrose photoreforming = −84.7 kJ/mol) [4], but multiple steps limit practical yields [44]. Reported apparent quantum efficiencies for glucose reforming on Pt/TiO2 [45] are typically a few percent under UV (lower than methanol case).
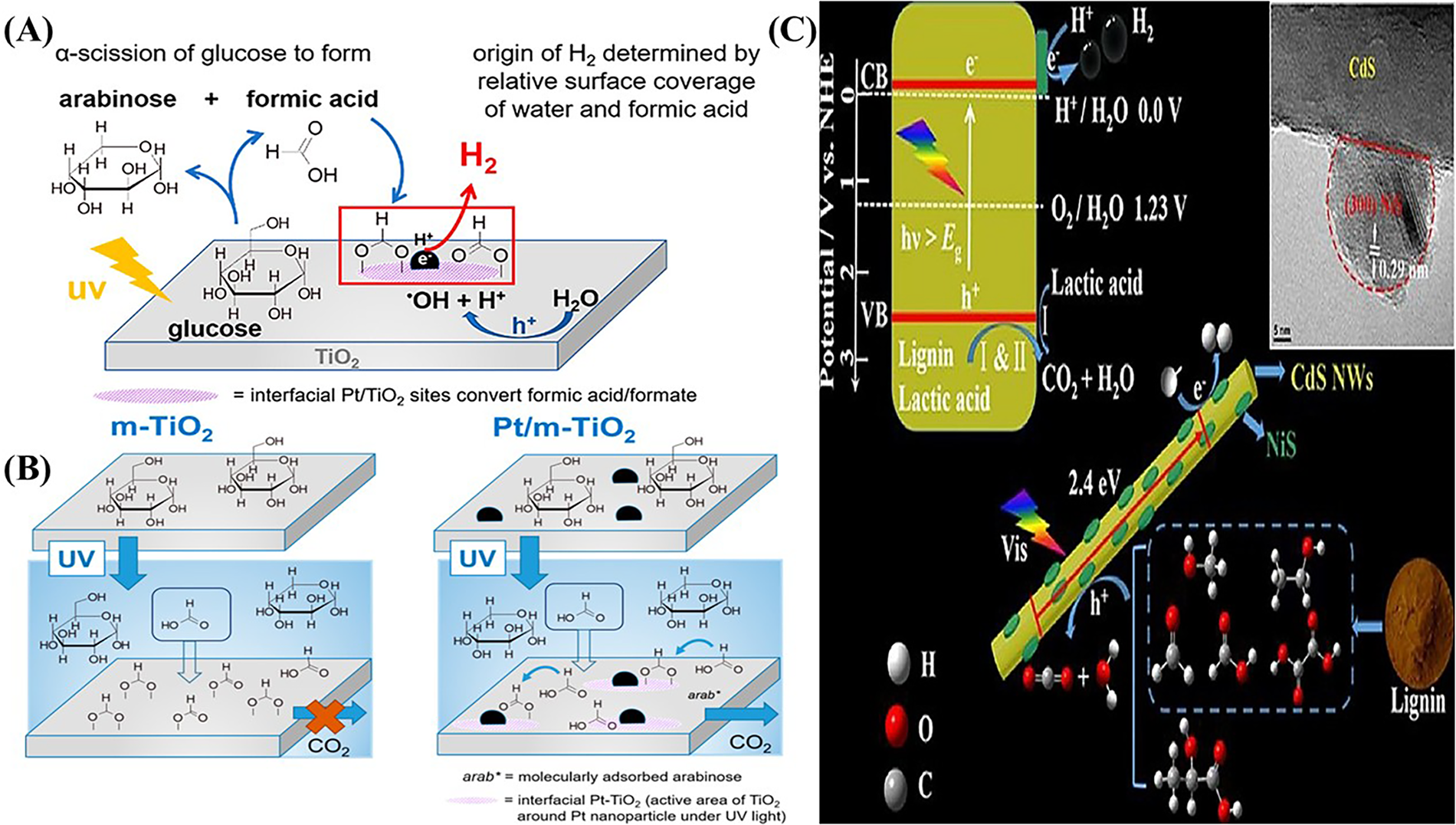
Figure 6: (A) In situ ATR-IR spectra showing accumulation of formic acid/formate during early-stage glucose photoreforming; (B) Proposed reaction pathway on m-TiO2 and Pt/m-TiO2 highlighting preferential formic acid adsorption and transformation at Pt-Ti interfacial sites. Adapted from Ref. [34]. (C) Schematic of lignin and lactic acid photoreforming over CdS nanowires/NiS under visible light, illustrating charge generation and H2/CO2 formation; inset shows HRTEM of CdS nanowires. Adapted from Ref [48].
Lignin-derived substrates are the most challenging. Lignin (a complex aromatic polymer) is highly stable, strongly absorbs light, and yields recalcitrant byproducts. Photoreforming requires breaking aromatic C–C and C–O bonds (such as β–O–4 linkages), which produce phenolic compounds (phenol, catechol, guaiacol, etc.) [46]. These aromatic intermediates tend to adsorb strongly on catalysts and even repolymerize, greatly hindering H2 formation. For example, photoreforming of phenol (a lignin model) over TiO2 produces catechol/hydroquinone, but strong adsorption and charge trapping on the catalyst drastically reduce H2 yield. Lignin yields orders of magnitude less H2 than sugars. In one study, organosolv lignin photoreforming on 0.1% Pt/TiO2 (P25) under UVA gave only 4.8 μmol·h−1·g−1. Under the same conditions, cellulose produced about 62.8 μmol·h−1·g−1 [46]. Another catalyst Pt/SiC/CdS yielded just 11 μmol·h−1·g−1 from technical lignin, whereas cellulose or grass gave >100 μmol·h−1·g−1 [47]. Thus, lignin’s H2 yield is typically <5% of a comparable sugar substrate. The low activity is attributed to severe catalyst poisoning and poor hole utilization. Photogenerated holes attack lignin aromatics, but fragmentation is inefficient; many holes are consumed inside reactions. This study overcame some limitations by alternating anaerobic/aerobic illumination to regenerate Pt sites, tripling lignin H2 output. In another study, hybrid sulfide systems like NiS/CdS (Fig. 6C) have been used (sometimes with co-feeders like lactic acid) to boost electron transfer; one NiS/CdS catalyst produced about 1.5 mmol·h−1·g−1 when oxidizing lignin-containing mixtures [48]. However, these often require sacrificial conditions and face stability issues. Emerging approaches include single-atom catalysts and S-scheme heterojunctions on carbon nitrides or oxides to enhance the difficult aromatic oxidation steps [49]. In lignin photoreforming tests, the main byproducts are smaller aromatics and CO2; often a char or polymeric residues can form, indicating partial dehydrogenation without complete mineralization [12]. In summary, lignin photoreforming gives lower H2 yields than sugars (often by orders of magnitude) and tends to generate a mix of oxygenated organics, therefore catalyst regeneration and product separation are major challenges.
Plastic photoreforming has emerged as a light driven route to convert polymer waste into hydrogen fuel and value-added organics, complementing thermal and biological recycling. Unlike conventional pyrolysis or gasification, which break polymers at high temperature into complex mixtures (and even hazardous byproducts like HCl or Cl2 from PVC), photoreforming uses sunlight and catalysts to oxidize plastics under milder and green conditions [8]. In essence, photogenerated holes attack the polymer backbone while photogenerated electrons reduce water to H2, offering a combined waste treatment and fuel production pathway. This strategy builds on lessons from biomass photoreforming and promises a circular approach: waste plastics become feedstock for hydrogen and commodity chemicals [4]. Common plastic wastes span several polymer families: Thermoplastic polyesters include PET polyethylene terephthalate (used in bottles and fibers) and bioplastics like PLA polylactic acid. Polyolefins dominate by volume: PE (polyethylene, including LDPE/HDPE) and PP (polypropylene) account for about 60% of production. Other classes include PS (polystyrene) and halogenated PVC. For example, PET alone constitutes near 10% of annual plastics production (82 Mt/yr) yet <20% is recycled [66]. All these polymers persist in the environment (hundreds of years for PET). In photoreforming, the polymer category dictates reactivity: polyesters (PET, PLA) are hydrophilic ester linkages enable chemical hydrolysis. PET yields terephthalate and ethylene glycol (EG) monomers; PLA yields lactate/lactic acid under alkali. Whereas polyolefins (PE, PP) are Saturated C–C polymers and very inert. They typically require oxidative degradation (e.g., acid oxidizers, thermal treatment) to generate shorter acids or oligomers. Polystyrene (PS) few photoreforming reports exist because under photooxidation its aromatic backbone can produce benzaldehyde or benzoic acid fragments rather than being efficiently converted into H2 or other desired products. (PVC) Chlorinated chains pose challenges; dehydrochlorination yields HCl or chlorinated byproducts that corrode catalysts. Very limited photoreforming work is reported due to these issues. Bulk plastics exhibit limited surface area and robust bonds, necessitating size reduction into soluble intermediates. Pretreatments optimize polymer reactivity and control photoreforming pathways [72]. In PET studies, NaOH (e.g., 2 M at 40°C for 24 h) converts much of PET to ethylene glycol (EG) and terephthalic acid [71]. For PLA, mild KOH produces lactic acid, while very concentrated base (5–10 M) can yield lactate salts [73]. In contrast, acidic or oxidative pretreatment is used for polyolefins. Strong HNO3 or H2SO4 oxidation of PE or PP chains generates short diacids (e.g., succinic, glutaric, formic) [74]. For example, 6%–7% HNO3 at 160°C–180°C oxidizes PE into nearly 40% succinic and about 20% glutaric acids [74]; these serve as soluble feedstocks for photocatalysis. In practice, pretreatment often involves combinations of mechanical milling, heat/oxidant, and acid/base steps. Overall, pretreatment tailors the substrate: it increases surface area and changes thermodynamics/kinetics (monomer vs. polymer), thus strongly affecting which products form.
Photoreforming PET usually begins with hydrolysis-derived EG. In one example, Reisner and coworkers used a carbon nitride/Ni2P photocatalyst under 430 nm light (CNx/Ni2P), obtaining visible light-driven hydrogen evolution [66]. As illustrated in (Fig. 7A) [75] EG is oxidized stepwise by photogenerated holes: glycolaldehyde → glyoxal → glycolate → glyoxylate oxalate → format, and eventually CO32−. The dominant products are thus small C1/C2 acids; for example, EG photoreforms mainly to formate, with minor glyoxal, glycolate, or acetate. These fragmentations liberate electrons that reduce protons to H2 [75]. Reported H2 evolution rates vary: Nguyen et al. achieved 7.3 mmol·h−1·g−1 with a Pt-g-C3N4 catalyst on PET-derived EG [67], whereas more recent catalysts (e.g., CdS/NiS heterojunctions) reach tens of mmol·h−1·g−1 [76]. Notably, product selectivity depends strongly on pretreatment and catalyst: under highly alkaline pre-hydrolysis, ethanol (from EG hydrogenation) can form and oxidize to acetate, explaining observed acetate fractions [77]. In all cases, aromatic terephthalate (TPA) is largely inert under photocatalysis, so the focus is on the soluble glycol fragment [78]. In summary, PET photoreforming yields hydrogen alongside organic acids (formate, glyoxylate, etc.) with efficiencies (H2 rates and quantum yields) that remain an active research area [67].
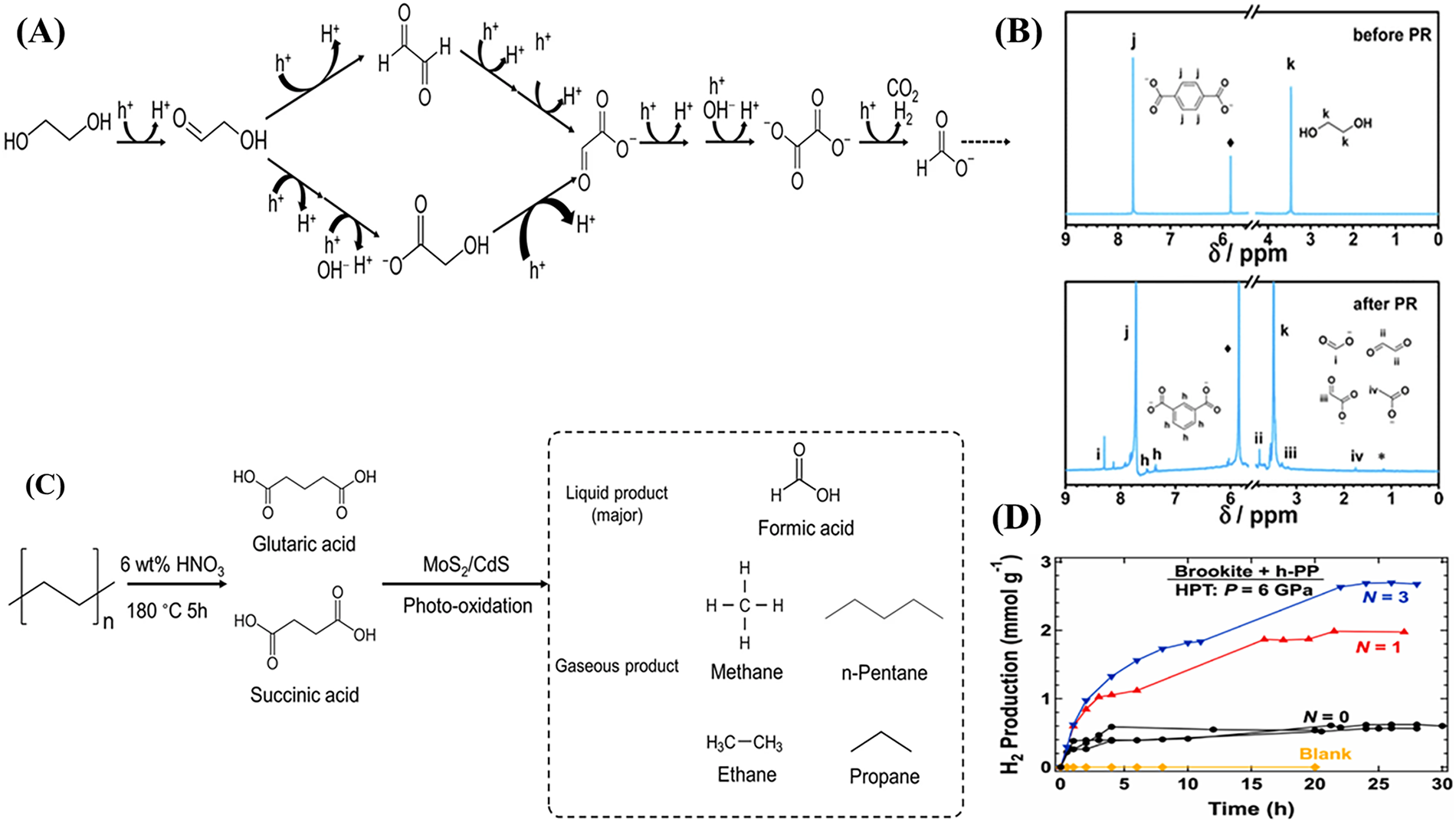
Figure 7: (A) Schematic of the three stages of photocatalytic redox: light absorption, charge separation, and surface redox reactions. Adapted from Ref [75]. (B) 1H NMR spectra of pristine PET and PET after photoreforming. Adapted from Ref [81]. Copyright Elsevier 2024. (C) Schematic of PE oxidation to carboxylic acids and subsequent photoreforming over MoS2/CdS. Adapted from Ref [79]. Copyright RSC 2022. (D) Hydrogen evolution from PP photoreforming over brookite-PP systems, comparing untreated and HPT-processed samples; triplicate blank tests confirm reproducibility. Adapted from Ref [82].
Polylactic acid (PLA) a bio-based polyester behaves similarly under base hydrolysis. Mild KOH predominantly yields lactic acid (LA) [73]. Photocatalytic oxidation of lactic acid then produces acetate, formate and other C1-C2 products [68]. For instance, a NiCoP/rGO/g-C3N4 catalyst photoreformed LA to primarily acetate and formate [79]. Under stronger alkali (5–10 M KOH), PLA yields sodium lactate, which is oxidized to pyruvate (PA) before further cleavage [80]. In one report, a Ni2P/ZnIn2S4 catalyst produced PA with 90% selectivity from lactate, giving H2 at 0.781 mmol·h−1·g−1 [80]. Under very high KOH, Du et al. and Zhu et al. found that lactate dominated and photoreforming of lactate over CdS/NiS delivered H2 up to 62.9 mmol·h−1·g−1 [75]. As shown in the literature, more aggressive pretreatment (10 M KOH) greatly boosts H2 yield (Fig. 7B) [75]. In all cases, the co-produced organics tend to be C1–C3 oxygenates (formate, pyruvate, acetaldehyde, acetate, methanol). For example, a g-C3N4/CdS/NiS catalyst achieved 25–30 mmol·h−1·g−1 H2 from photoreforming PLA-derived lactate in seawater, concurrently oxidizing lactate to formate, acetate and pyruvate [76]. These examples illustrate that tailoring PLA pretreatment (LA vs. lactate) tunes the photochemical pathway and efficiency.
Polyolefins are more recalcitrant; acidic pretreatment breaks PE/PP into small acids (succinic, glutaric, formic). The photoreforming mechanism then proceeds via decarboxylation and hydrogenolysis. For example, PE treated with HNO3 is oxidized mainly to succinic and glutaric acids. On photocatalysts (e.g., Pd single-atom TiO2), these diacids produce C2 hydrocarbons: ethylene and ethane [74]. The PE’s two-step conversion (Fig. 7C) [79] HNO3 oxidation then leads to the formation of succinate/glutarate. A recent study found that severe plastic deformation (high-pressure torsion) of PP before photoreforming on brookite TiO2 greatly enhanced H2 production (4–5× higher) [82]. In addition, triplicate experiments conducted for PP mixed with brookite in the absence of HPT treatment (Fig. 7D) exhibit a relative deviation below 10%, demonstrating the good reproducibility of the photoreforming results. In general, polyolefin photoreforming tends to generate small alkanes/alkenes (via decarboxylation) rather than multi-carbon organics [82]. In summary, plastic waste photoreforming represents a promising avenue for simultaneous environmental remediation and sustainable hydrogen production. By tailoring pretreatment strategies to polymer type and optimizing photocatalyst design, significant progress has been made in converting diverse plastic streams, particularly PET and PLA, into hydrogen and valuable organics under mild conditions. However, challenges remain in improving efficiency for more inert polymers like PE and PP, enhancing catalyst durability, and scaling the process for real-world applications. Continued innovation in materials engineering, mechanistic understanding, and reactor integration will be crucial to realizing the full potential of this waste-to-energy approach.
5 Challenges and Future Perspective
Despite significant advances in photocatalyst design and photoreforming processes for sustainable hydrogen production, several key challenges remain before large-scale practical implementation can be realized [83]. Future research must address the fundamental link between catalyst structure and reaction dynamics to rationally optimize activity and selectivity, especially under realistic waste feedstock conditions, which are often complex and variable [84]. Improving the photostability and long-term durability of photocatalysts under continuous solar irradiation and harsh reaction environments remains critical. Current systems frequently suffer from catalyst deactivation due to surface fouling, photocorrosion, or accumulation of byproducts, requiring robust surface engineering and self-cleaning mechanisms [85]. Enhancing the solar spectrum utilization via broadband absorption, particularly in the visible and near-infrared regions, is another pressing challenge [2]. This entails further advancement in band structure engineering, including defect modulation and heterojunction integration, while balancing recombination rates and charge carrier lifetimes. From a practical standpoint, scalability and cost-effectiveness must be prioritized by utilizing earth-abundant, non-toxic, and easily synthesized materials that align with circular economy principles. Development of sustainable synthesis routes and photocatalyst recycling/regeneration strategies will be vital to reduce environmental impact and promote industrial viability [6]. Finally, gaining deeper mechanistic insights into substrate-specific oxidation pathways and electron transfer dynamics within heterogeneous waste matrices will facilitate tailored catalyst systems with improved efficiency and selectivity [86]. Integrating advanced operando characterization techniques and computational modeling will accelerate this understanding. Addressing these scientific and engineering challenges through interdisciplinary approaches will be essential to harness the full potential of waste photoreforming as a clean, renewable, and economically feasible hydrogen production technology.
In conclusion, waste photoreforming represents a promising and sustainable approach for addressing two major global challenges: plastic waste management and green hydrogen production. By utilizing abundant waste feedstocks such as biomass-derived substrates, and synthetic plastics, photoreforming allows for the conversion of complex organic materials into valuable chemical products while simultaneously generating clean hydrogen. Biomass-derived feedstocks, including short chain alcohols, saccharides, and lignin derivatives, offer significant potential due to their renewable nature, high hydrogen to carbon ratios, and carbon-rich structure. These materials, however, require specific catalyst designs and pretreatment strategies to efficiently break down their complex structures, optimize hydrogen production, and minimize the formation of unwanted byproducts. In the case of synthetic plastic waste, photoreforming provides an innovative way to recycle non-biodegradable polymers such as PET, PE, and PP, which are otherwise difficult to degrade. The conversion of these plastics into hydrogen and small organics not only reduces plastic pollution but also transforms waste into a valuable energy resource. However, challenges remain, particularly for more inert polymers like PE and PP, where surface activation and effective charge transfer are critical for enhancing reaction rates. The development of advanced photocatalysts, such as metal-modified TiO2, g-C3N4 composites, and heterojunction systems, plays a pivotal role in maximizing the efficiency of these photoreforming processes. Overall, photoreforming provides a versatile platform for the sustainable valorization of diverse waste streams, offering a circular economy solution that ties together waste management and clean energy production. Moving forward, advancements in catalyst design, reactor efficiency, and process scalability will be essential for realizing the full potential of photoreforming technologies. As research continues to evolve, it is likely that photoreforming will become an integral part of global strategies for waste valorization and renewable energy generation, driving forward a more sustainable and cleaner future.
Acknowledgement: The authors would like to express their sincere gratitude to Universiti Teknologi PETRONAS (UTP) and the Institute of Technology PETRONAS Sdn. Bhd. (ITPSB) for the financial support provided through the Graduate Assistantship Scheme. We also acknowledge the guidance and facilities provided by the Department of Mechanical Engineering at UTP, which significantly contributed to the successful completion of this work.
Funding Statement: This work was financially supported by Universiti Teknologi PETRONAS and the Institute of Technology PETRONAS Sdn. Bhd. (ITPSB) through the Graduate Assistantship Scheme.
Author Contributions: Mirna Omar: writing—original draft, conceptualized the research idea, conducted the literature review, and prepared the original draft of the manuscript. Sarah Omar: contributed to data collection, figure design, and assisted in manuscript editing. Kamaruzzaman Sopian: provided supervision, validation, and overall project guidance. Taib Iskandar Mohamad: supervision, review & editing, resources, project administration, funding acquisition, formal analysis, data curation, conceptualization. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: No new data were created or analyzed in this study. Data sharing is not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The author declares no conflicts of interest.
References
1. Supramaniam L, Lam SM, Sin JC, Lahijani P, Mohamed AR. Recent advances in hydrogen production by solar photoreforming of plastic feedstocks using nanocatalytic materials: a review. Int J Hydrogen Energy. 2025;145(11):95–118. doi:10.1016/j.ijhydene.2025.06.010. [Google Scholar] [CrossRef]
2. Rossetti I. Hydrogen production by photoreforming of renewable substrates. Int Sch Res Not. 2012;2012(1):964936. doi:10.5402/2012/964936. [Google Scholar] [CrossRef]
3. Shelake SP, Sutar DN, Abraham BM, Banerjee T, Sainath AVS, Pal U. Emerging photoreforming process to hydrogen production: a future energy. Adv Funct Mater. 2024;34(40):2403795. doi:10.1002/adfm.202403795. [Google Scholar] [CrossRef]
4. Ashraf M, Ullah N, Khan I, Tremel W, Ahmad S, Tahir MN. Photoreforming of waste polymers for sustainable hydrogen fuel and chemicals feedstock: waste to energy. Chem Rev. 2023;123(8):4443–509. doi:10.1021/acs.chemrev.2c00602. [Google Scholar] [PubMed] [CrossRef]
5. Niu F, Wu Z, Chen D, Huang Y, Ordomsky VV, Khodakov AY, et al. State-of-the-art and perspectives of hydrogen generation from waste plastics. Chem Soc Rev. 2025;54(10):4948–72. doi:10.1039/d4cs00604f. [Google Scholar] [PubMed] [CrossRef]
6. Shi C, Kang F, Zhu Y, Teng M, Shi J, Qi H, et al. Photoreforming lignocellulosic biomass for hydrogen production: optimized design of photocatalyst and photocatalytic system. Chem Eng J. 2023;452:138980. doi:10.1016/j.cej.2022.138980. [Google Scholar] [CrossRef]
7. Toe CY, Tsounis C, Zhang J, Masood H, Gunawan D, Scott J, et al. Advancing photoreforming of organics: highlights on photocatalyst and system designs for selective oxidation reactions. Energy Environ Sci. 2021;14(3):1140–75. doi:10.1039/d0ee03116j. [Google Scholar] [CrossRef]
8. Zhang X, Zu W, Lee LYS. Crucial role of pre-treatment in plastic photoreforming for precision upcycling. npj Mater Sustain. 2025;3(1):3. doi:10.1038/s44296-024-00045-5. [Google Scholar] [CrossRef]
9. Hisatomi T, Wang Q, Zhang F, Ardo S, Reisner E, Nishiyama H, et al. Photocatalytic water splitting for large-scale solar-to-chemical energy conversion and storage. Front Sci. 2024;2:1411644. doi:10.3389/fsci.2024.1411644. [Google Scholar] [CrossRef]
10. Mokhtar B, Ahmed MG, Alqahtani HS, Kandiel TA. Biomass and plastic photoreforming for hydrogen and valuable chemicals production. In: The handbook of environmental chemistry. Berlin/Heidelberg, Germany: Springer; 2024. p. 1–31. doi:10.1007/698_2024_1150. [Google Scholar] [CrossRef]
11. Ay H, Sen F. Metal, metal oxides, and metal sulfide roles in fuel cell. In: Metal, metal-oxides and metal sulfides for batteries, fuel cells, solar cells, photocatalysis and health sensors. Cham, Switzerland: Springer; 2021. p. 115–45. [Google Scholar]
12. Cheng X, Palma B, Zhao H, Zhang H, Wang J, Chen Z, et al. Photoreforming for lignin upgrading: a critical review. ChemSusChem. 2023;16(23):e202300675. doi:10.1002/cssc.202300675. [Google Scholar] [PubMed] [CrossRef]
13. Augustin A, Chuaicham C, Shanmugam M, Vellaichamy B, Rajendran S, Hoang TKA, et al. Recent development of organic-inorganic hybrid photocatalysts for biomass conversion into hydrogen production. Nanoscale Adv. 2022;4(12):2561–82. doi:10.1039/d2na00119e. [Google Scholar] [PubMed] [CrossRef]
14. Joo JB, Dillon R, Lee I, Yin Y, Bardeen CJ, Zaera F. Promotion of atomic hydrogen recombination as an alternative to electron trapping for the role of metals in the photocatalytic production of H2. Proc Natl Acad Sci U S A. 2014;111(22):7942–7. doi:10.1073/pnas.1405365111. [Google Scholar] [PubMed] [CrossRef]
15. Uekert T. Plastic and mixed waste as feedstocks for solar-driven H2 production [master’s thesis]. Cambridge, UK: University of Cambridge Repository; 2021. [Google Scholar]
16. Lin WC, Jayakumar J, Chang CL, Ting LY, Elsayed MH, Abdellah M, et al. Effect of energy bandgap and sacrificial agents of cyclopentadithiophene-based polymers for enhanced photocatalytic hydrogen evolution. Appl Catal B Environ. 2021;298:120577. doi:10.1016/j.apcatb.2021.120577. [Google Scholar] [CrossRef]
17. Ling GZS, Ng SF, Ong WJ. Tailor-engineered 2D cocatalysts: harnessing electron-hole redox center of 2D g-C3N4 photocatalysts toward solar-to-chemical conversion and environmental purification. Adv Funct Mater. 2022;32(29):2111875. doi:10.1002/adfm.202111875. [Google Scholar] [CrossRef]
18. Nawaz A, Kuila A, Mishra NS, Leong KH, Sim LC, Saravanan P, et al. Challenges and implication of full solar spectrum-driven photocatalyst. Rev Chem Eng. 2021;37(4):533–60. doi:10.1515/revce-2018-0069. [Google Scholar] [CrossRef]
19. Puga A. Photocatalytic hydrogen production for sustainable energy. Hoboken, NJ, USA: John Wiley & Sons, Inc.; 2023. doi:10.1002/9783527835423. [Google Scholar] [CrossRef]
20. Zhao H, Li CF, Yong X, Kumar P, Palma B, Hu ZY, et al. Coproduction of hydrogen and lactic acid from glucose photocatalysis on band-engineered Zn1−xCdxS homojunction. iScience. 2021;24(2):102109. doi:10.1016/j.isci.2021.102109. [Google Scholar] [PubMed] [CrossRef]
21. Zhao H, Li CF, Liu LY, Palma B, Hu ZY, Renneckar S, et al. n-p heterojunction of TiO2-NiO core-shell structure for efficient hydrogen generation and lignin photoreforming. J Colloid Interface Sci. 2021;585:694–704. doi:10.1016/j.jcis.2020.10.049. [Google Scholar] [PubMed] [CrossRef]
22. Ye HF, Shi R, Yang X, Fu WF, Chen Y. P-doped ZnxCd1−xS solid solutions as photocatalysts for hydrogen evolution from water splitting coupled with photocatalytic oxidation of 5-hydroxymethylfurfural. Appl Catal B Environ. 2018;233:70–9. doi:10.1016/j.apcatb.2018.03.060. [Google Scholar] [CrossRef]
23. Chang L, Yong ST, Chai SP, Putri LK, Tan LL, Mohamed AR. A review of methanol photoreforming: elucidating the mechanisms, photocatalysts and recent advancement strategies. Mater Today Chem. 2023;27:101334. doi:10.1016/j.mtchem.2022.101334. [Google Scholar] [CrossRef]
24. Endo-Kimura M, Janczarek M, Bielan Z, Zhang D, Wang K, Markowska-Szczupak A, et al. Photocatalytic and antimicrobial properties of Ag2O/TiO2 heterojunction. ChemEngineering. 2019;3(1):3. doi:10.3390/chemengineering3010003. [Google Scholar] [CrossRef]
25. Luo XL, He GL, Fang YP, Xu YH. Nickel sulfide/graphitic carbon nitride/strontium titanate (NiS/g-C3N4/SrTiO3) composites with significantly enhanced photocatalytic hydrogen production activity. J Colloid Interface Sci. 2018;518:184–91. doi:10.1016/j.jcis.2018.02.038. [Google Scholar] [PubMed] [CrossRef]
26. Wang R, Ye C, Wang H, Jiang F. Z-scheme LaCoO3/g-C3N4 for efficient full-spectrum light-simulated solar photocatalytic hydrogen generation. ACS Omega. 2020;5(47):30373–82. doi:10.1021/acsomega.0c03318. [Google Scholar] [PubMed] [CrossRef]
27. Liu Y, Zhang H, Ke J, Zhang J, Tian W, Xu X, et al. 0D (MoS2)/2D (g-C3N4) heterojunctions in Z-scheme for enhanced photocatalytic and electrochemical hydrogen evolution. Appl Catal B Environ. 2018;228:64–74. doi:10.1016/j.apcatb.2018.01.067. [Google Scholar] [CrossRef]
28. Nagakawa H, Nagata M. Photoreforming of organic waste into hydrogen using a thermally radiative CdOx/CdS/SiC photocatalyst. ACS Appl Mater Interfaces. 2021;13(40):47511–9. doi:10.1021/acsami.1c11888. [Google Scholar] [PubMed] [CrossRef]
29. Edirisooriya EMNT, Senanayake PS, Ahasan T, Xu P, Wang H. Comprehensive insights into photoreforming of waste plastics for hydrogen production. Catalysts. 2025;15(5):453. doi:10.3390/catal15050453. [Google Scholar] [CrossRef]
30. Lakshmana Reddy N, Cheralathan KK, Durga Kumari V, Neppolian B, Muthukonda Venkatakrishnan S. Photocatalytic reforming of biomass derived crude glycerol in water: a sustainable approach for improved hydrogen generation using Ni(OH)2 decorated TiO2 nanotubes under solar light irradiation. ACS Sustain Chem Eng. 2018;6(3):3754–64. doi:10.1021/acssuschemeng.7b04118. [Google Scholar] [CrossRef]
31. Gong X, Tong F, Ma F, Zhang Y, Zhou P, Wang Z, et al. Photoreforming of plastic waste poly (ethylene terephthalate) via in-situ derived CN-CNTs-NiMo hybrids. Appl Catal B Environ Energy. 2022;307(38):121143. doi:10.1016/j.apcatb.2022.121143. [Google Scholar] [CrossRef]
32. Al-Fatesh AS, AL-Garadi NYA, Osman AI, Al-Mubaddel FS, Ibrahim AA, Khan WU, et al. From plastic waste pyrolysis to Fuel: impact of process parameters and material selection on hydrogen production. Fuel. 2023;344(1):128107. doi:10.1016/j.fuel.2023.128107. [Google Scholar] [CrossRef]
33. Su K, Liu H, Zhang C, Wang F. Photocatalytic conversion of waste plastics to low carbon number organic products. Chin J Catal. 2022;43(3):589–94. doi:10.1016/S1872-2067(21)63885-8. [Google Scholar] [CrossRef]
34. Lan L, Daly H, Sung R, Tuna F, Skillen N, Robertson PKJ, et al. Mechanistic study of glucose photoreforming over TiO2-based catalysts for H2 production. ACS Catal. 2023;13(13):8574–87. doi:10.1021/acscatal.3c00858. [Google Scholar] [PubMed] [CrossRef]
35. Wang J, Lu Z, Gao K, Kang X, Zhu C, Qiao F, et al. Photocatalytic reforming of ethanol in the liquid phase using a ternary composite of Rh/TiO2/g-C3N4 as a catalyst. ACS Appl Mater Interfaces. 2024;16(37):49371–9. doi:10.1021/acsami.4c09188. [Google Scholar] [PubMed] [CrossRef]
36. Fu X, Leung DYC, Wang X, Xue W, Fu X. Photocatalytic reforming of ethanol to H2 and CH4 over ZnSn(OH)6 nanocubes. Int J Hydrogen Energy. 2011;36(2):1524–30. doi:10.1016/j.ijhydene.2010.10.090. [Google Scholar] [CrossRef]
37. Nascimento LL, Carvalho Souza RA, Zacour Marinho J, Wang C, Patrocinio AOT. Light-driven conversion of biomass-derived compounds into green fuels and chemicals. J Clean Prod. 2024;449(8):141709. doi:10.1016/j.jclepro.2024.141709. [Google Scholar] [CrossRef]
38. Mills A, Bingham M, O’Rourke C, Bowker M. Modelled kinetics of the rate of hydrogen evolution as a function of metal catalyst loading in the photocatalysed reforming of methanol by Pt (or Pd)/TiO2. J Photochem Photobiol A Chem. 2019;373:122–30. doi:10.1016/j.jphotochem.2018.12.039. [Google Scholar] [CrossRef]
39. Ren P, Gao Z, Montini T, Zhao Z, Ta N, Huang Y, et al. Stepwise photoassisted decomposition of carbohydrates to H2. Joule. 2023;7(2):333–49. doi:10.1016/j.joule.2023.01.002. [Google Scholar] [CrossRef]
40. Fu X, Long J, Wang X, Leung DYC, Ding Z, Wu L, et al. Photocatalytic reforming of biomass: a systematic study of hydrogen evolution from glucose solution. Int J Hydrogen Energy. 2008;33(22):6484–91. doi:10.1016/j.ijhydene.2008.07.068. [Google Scholar] [CrossRef]
41. Li Z, Li W, Wang J, Jing L, Li P, Zhang H, et al. Roles of cocatalysts in biomass photo (electro) refining. Adv Energy Mater. 2024;14(42):2401838. doi:10.1002/aenm.202401838. [Google Scholar] [CrossRef]
42. Pargoletti E, Rimoldi L, Meroni D, Cappelletti G. Photocatalytic removal of gaseous ethanol, acetaldehyde and acetic acid: from a fundamental approach to real cases. Int Mater Rev. 2022;67(8):864–97. doi:10.1080/09506608.2021.2017390. [Google Scholar] [CrossRef]
43. Kawai T, Sakata T. Conversion of carbohydrate into hydrogen fuel by a photocatalytic process. Nature. 1980;286(5772):474–6. doi:10.1038/286474a0. [Google Scholar] [CrossRef]
44. Uekert T, Pichler CM, Schubert T, Reisner E. Solar-driven reforming of solid waste for a sustainable future. Nat Sustain. 2021;4(5):383–91. doi:10.1038/s41893-020-00650-x. [Google Scholar] [CrossRef]
45. Luu XC, Phan Thi LA, Raizada P, Singh P, Nguyen LH, Ghotekar S, et al. Solar-driven reforming of lignocellulosic biomass to renewable biohydrogen: a review. ChemCatChem. 2024;16(22):e202401287. doi:10.1002/cctc.202401287. [Google Scholar] [CrossRef]
46. Aljohani M, Daly H, Lan L, Mavridis A, Lindley M, Haigh SJ, et al. Enhancing hydrogen production from the photoreforming of lignin. ChemPlusChem. 2024;89(2):e202300411. doi:10.1002/cplu.202300411. [Google Scholar] [PubMed] [CrossRef]
47. Nagakawa H, Nagata M. Photoreforming of lignocellulosic biomass into hydrogen under sunlight in the presence of thermally radiative CdS/SiC composite photocatalyst. ACS Appl Energy Mater. 2021;4(2):1059–62. doi:10.1021/acsaem.0c02530. [Google Scholar] [CrossRef]
48. Li C, Wang H, Naghadeh SB, Zhang JZ, Fang P. Visible light driven hydrogen evolution by photocatalytic reforming of lignin and lactic acid using one-dimensional NiS/CdS nanostructures. Appl Catal B Environ. 2018;227:229–39. doi:10.1016/j.apcatb.2018.01.038. [Google Scholar] [CrossRef]
49. Kasap H, Achilleos DS, Huang A, Reisner E. Photoreforming of lignocellulose into H2 using nanoengineered carbon nitride under benign conditions. J Am Chem Soc. 2018;140(37):11604–7. doi:10.1021/jacs.8b07853. [Google Scholar] [PubMed] [CrossRef]
50. Caudillo-Flores U, Muñoz-Batista MJ, Kubacka A, Fernández-García M. Pd-Pt bimetallic Nb-doped TiO2 for H2 photo-production: gas and liquid phase processes. Mol Catal. 2020;481:110240. doi:10.1016/j.mcat.2018.11.011. [Google Scholar] [CrossRef]
51. Miwa T, Kaneco S, Katsumata H, Suzuki T, Ohta K, Chand Verma S, et al. Photocatalytic hydrogen production from aqueous methanol solution with CuO/Al2O3/TiO2 nanocomposite. Int J Hydrogen Energy. 2010;35(13):6554–60. doi:10.1016/j.ijhydene.2010.03.128. [Google Scholar] [CrossRef]
52. Liu Z, Yin Z, Cox C, Bosman M, Qian X, Li N, et al. Room temperature stable COx-free H2 production from methanol with magnesium oxide nanophotocatalysts. Sci Adv. 2016;2(9):e1501425. doi:10.1126/sciadv.1501425. [Google Scholar] [PubMed] [CrossRef]
53. Xie S, Shen Z, Deng J, Guo P, Zhang Q, Zhang H, et al. Visible light-driven C−H activation and C-C coupling of methanol into ethylene glycol. Nat Commun. 2018;9(1):1181. doi:10.1038/s41467-018-03543-y. [Google Scholar] [PubMed] [CrossRef]
54. Zhang Y, Zhao J, Wang H, Xiao B, Zhang W, Zhao X, et al. Single-atom Cu anchored catalysts for photocatalytic renewable H2 production with a quantum efficiency of 56%. Nat Commun. 2022;13(1):58. doi:10.1038/s41467-021-27698-3. [Google Scholar] [PubMed] [CrossRef]
55. Gallo A, Montini T, Marelli M, Minguzzi A, Gombac V, Psaro R, et al. H2 production by renewables photoreforming on Pt-Au/TiO2 catalysts activated by reduction. ChemSusChem. 2012;5(9):1800–11. doi:10.1002/cssc.201200085. [Google Scholar] [PubMed] [CrossRef]
56. Chao Y, Zhang W, Wu X, Gong N, Bi Z, Li Y, et al. Visible-light direct conversion of ethanol to 1, 1-diethoxyethane and hydrogen over a non-precious metal photocatalyst. Chemistry. 2019;25(1):189–94. doi:10.1002/chem.201804664. [Google Scholar] [PubMed] [CrossRef]
57. Wu X, Zhao H, Khan MA, Maity P, Al-Attas T, Larter S, et al. Sunlight-driven biomass photorefinery for coproduction of sustainable hydrogen and value-added biochemicals. ACS Sustain Chem Eng. 2020;8(41):15772–81. doi:10.1021/acssuschemeng.0c06282. [Google Scholar] [CrossRef]
58. Speltini A, Gualco F, Maraschi F, Sturini M, Dondi D, Malavasi L, et al. Photocatalytic hydrogen evolution assisted by aqueous (waste) biomass under simulated solar light: oxidized g-C3N4 vs. P25 titanium dioxide. Int J Hydrogen Energy. 2019;44(8):4072–8. doi:10.1016/j.ijhydene.2018.12.126. [Google Scholar] [CrossRef]
59. Aguirre AR, Reina AC, Pérez JP, Colon G, Malato S. Catalysts and photoreactors for photocatalytic solar hydrogen production: fundamentals and recent developments at pilot scale. In: Photocatalytic hydrogen production for sustainable energy. Hoboken, NJ, USA: John Wiley & Sons, Inc.; 2023. p. 275–303. [Google Scholar]
60. Wakerley DW, Kuehnel MF, Orchard KL, Ly KH, Rosser TE, Reisner E. Solar-driven reforming of lignocellulose to H2 with a CdS/CdOx photocatalyst. Nat Energy. 2017;2(4):17021. doi:10.1038/nenergy.2017.21. [Google Scholar] [CrossRef]
61. Bhattacharjee S, Andrei V, Pornrungroj C, Rahaman M, Pichler CM, Reisner E. Reforming of soluble biomass and plastic derived waste using a bias-free Cu30Pd70 |Perovskite| Pt photoelectrochemical device. Adv Funct Mater. 2022;32(7):2109313. doi:10.1002/adfm.202109313. [Google Scholar] [CrossRef]
62. Pichler CM, Uekert T, Reisner E. Photoreforming of biomass in metal salt hydrate solutions. Chem Commun. 2020;56(43):5743–6. doi:10.1039/d0cc01686a. [Google Scholar] [PubMed] [CrossRef]
63. Kurenkova AY, Medvedeva TB, Gromov NV, Bukhtiyarov AV, Gerasimov EY, Cherepanova SV, et al. Sustainable hydrogen production from starch aqueous suspensions over a Cd0.7Zn0.3S-based photocatalyst. Catalysts. 2021;11(7):870. doi:10.3390/catal11070870. [Google Scholar] [CrossRef]
64. Kadam SR, Mate VR, Panmand RP, Nikam LK, Kulkarni MV, Sonawane RS, et al. A green process for efficient lignin (biomass) degradation and hydrogen production via water splitting using nanostructured C, N, S-doped ZnO under solar light. RSC Adv. 2014;4(105):60626–35. doi:10.1039/c4ra10760h. [Google Scholar] [CrossRef]
65. Wang E, Mahmood A, Chen SG, Sun W, Muhmood T, Yang X, et al. Solar-driven photocatalytic reforming of lignocellulose into H2 and value-added biochemicals. ACS Catal. 2022;12(18):11206–15. doi:10.1021/acscatal.2c02624. [Google Scholar] [CrossRef]
66. Uekert T, Kasap H, Reisner E. Photoreforming of nonrecyclable plastic waste over a carbon nitride/nickel phosphide catalyst. J Am Chem Soc. 2019;141(38):15201–10. doi:10.1021/jacs.9b06872. [Google Scholar] [PubMed] [CrossRef]
67. Nguyen TKA, Trần-Phú T, Ta XMC, Truong TN, Leverett J, Daiyan R, et al. Understanding structure-activity relationship in Pt-loaded g-C3N4 for efficient solar-photoreforming of polyethylene terephthalate plastic and hydrogen production. Small Meth. 2024;8(2):2300427. doi:10.1002/smtd.202300427. [Google Scholar] [PubMed] [CrossRef]
68. Sun DW, Chen KL, Huang JH. Benzenesulfonyl chloride-incorporated g-C3N4 for photocatalytic hydrogen generation by using the hydrolysate of poly(lactic acid) as sacrificial reagent. Appl Catal A Gen. 2021;628:118397. doi:10.1016/j.apcata.2021.118397. [Google Scholar] [CrossRef]
69. Kawai T, Sakata T. Photocatalytic hydrogen production from water by the decomposition of poly-vinylchloride, protein, algae, dead insects, and excrement. Chem Lett. 1981;10(1):81–4. doi:10.1246/cl.1981.81. [Google Scholar] [CrossRef]
70. Xu J, Jiao X, Zheng K, Shao W, Zhu S, Li X, et al. Plastics-to-syngas photocatalysed by co-Ga2O3 nanosheets. Natl Sci Rev. 2022;9(9):nwac011. doi:10.1093/nsr/nwac011. [Google Scholar] [PubMed] [CrossRef]
71. Uekert T, Kuehnel MF, Wakerley DW, Reisner E. Plastic waste as a feedstock for solar-driven H2 generation. Energy Environ Sci. 2018;11(10):2853–7. doi:10.1039/c8ee01408f. [Google Scholar] [CrossRef]
72. Yao Y, Zhang J, Shi L, Wang S, Duan X. Solar fuels production from plastics and biomass photoreforming. Energy Fuels. 2025;39(30):14455–82. doi:10.1021/acs.energyfuels.5c02800. [Google Scholar] [CrossRef]
73. Yan JQ, Sun DW, Huang JH. Synergistic poly(lactic acid) photoreforming and H2 generation over ternary NixCo1−xP/reduced graphene oxide/g-C3N4 composite. Chemosphere. 2022;286(Pt 3):131905. doi:10.1016/j.chemosphere.2021.131905. [Google Scholar] [PubMed] [CrossRef]
74. Zhang S, Xia B, Qu Y, Jing L, Jaroniec M, Ran J, et al. Photocatalytic production of ethylene and propionic acid from plastic waste by titania-supported atomically dispersed Pd species. Sci Adv. 2023;9(49):eadk2407. doi:10.1126/sciadv.adk2407. [Google Scholar] [PubMed] [CrossRef]
75. Zhu C, Wang J, Lv J, Zhu Y, Huang Q, Sun C. Solar-driven reforming of waste polyester plastics into hydrogen over CdS/NiS catalyst. Int J Hydrogen Energy. 2024;51(7):91–103. doi:10.1016/j.ijhydene.2023.08.064. [Google Scholar] [CrossRef]
76. Yin Z, Chen H, Wang Q, Wang Z, Yu G, Tang B, et al. Construction of an interface interaction in a g-C3N4/CdS/NiS for photoreforming of plastic and clean hydrogen regeneration. J Colloid Interface Sci. 2024;675:218–25. doi:10.1016/j.jcis.2024.06.214. [Google Scholar] [PubMed] [CrossRef]
77. Karayannidis GP, Chatziavgoustis AP, Achilias DS. Poly(ethylene terephthalate) recycling and recovery of pure terephthalic acid by alkaline hydrolysis. Adv Polym Technol. 2002;21(4):250–9. doi:10.1002/adv.10029. [Google Scholar] [CrossRef]
78. Al-Sabagh AM, Yehia FZ, Eshaq G, Rabie AM, ElMetwally AE. Greener routes for recycling of polyethylene terephthalate. Egypt J Petrol. 2016;25(1):53–64. doi:10.1016/j.ejpe.2015.03.001. [Google Scholar] [CrossRef]
79. Du M, Zhang Y, Kang S, Guo X, Ma Y, Xing M, et al. Trash to treasure: photoreforming of plastic waste into commodity chemicals and hydrogen over MoS2-tipped CdS nanorods. ACS Catal. 2022;12(20):12823–32. doi:10.1021/acscatal.2c03605. [Google Scholar] [CrossRef]
80. Liu CX, Shi R, Ma W, Liu F, Chen Y. Photoreforming of polyester plastics into added-value chemicals coupled with H2 evolution over a Ni2P/ZnIn2S4 catalyst. Inorg Chem Front. 2023;10(15):4562–8. doi:10.1039/d3qi00914a. [Google Scholar] [CrossRef]
81. Nguyen TT, Edalati K. Impact of high-pressure torsion on hydrogen production from photodegradation of polypropylene plastic wastes. Int J Hydrogen Energy. 2024;81:411–7. doi:10.1016/j.ijhydene.2024.07.306. [Google Scholar] [CrossRef]
82. Guo S, Huang Y, Li D, Xie Z, Jia Y, Wu X, et al. Visible-light-driven photoreforming of poly(ethylene terephthalate) plastics via carbon nitride porous microtubes. Chem Commun. 2023;59(50):7791–4. doi:10.1039/d3cc02012f. [Google Scholar] [PubMed] [CrossRef]
83. Lei D, Wang L, Lv Y, Luo N, Wang Z. A comprehensive review of solar photocatalysis & photothermal catalysis for hydrogen production from biomass: from material characteristics to engineering application. Chemistry. 2024;30(47):e202401486. doi:10.1002/chem.202401486. [Google Scholar] [PubMed] [CrossRef]
84. Rossetti I, Bahadori E, Villa A, Prati L, Ramis G. Hydrogen production by photoreforming of organic compounds. J Technol Innov Renew Energy. 2018;7:55–9. doi:10.6000/1929-6002.2018.07.07. [Google Scholar] [CrossRef]
85. Bhattacharjee S, Guo C, Lam E, Holstein JM, Rangel Pereira M, Pichler CM, et al. Chemoenzymatic photoreforming: a sustainable approach for solar fuel generation from plastic feedstocks. J Am Chem Soc. 2023;145(37):20355–64. doi:10.26434/chemrxiv-2023-m5rzw. [Google Scholar] [CrossRef]
86. Davis KA, Yoo S, Shuler EW, Sherman BD, Lee S, Leem G. Photocatalytic hydrogen evolution from biomass conversion. Nano Converg. 2021;8(1):6. doi:10.1186/s40580-021-00256-9. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF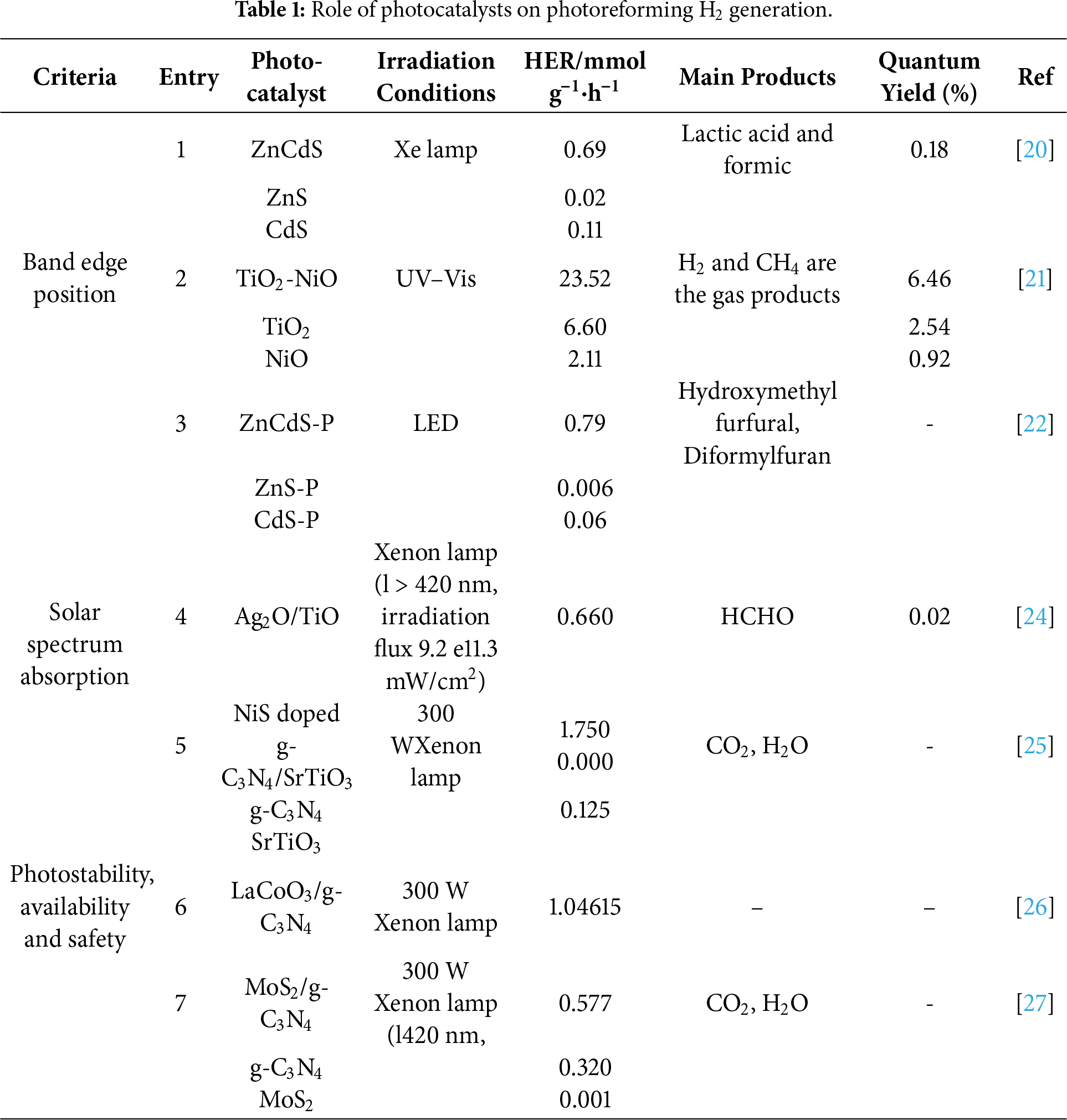
 Downloads
Downloads
 Citation Tools
Citation Tools