 Open Access
Open Access
ARTICLE
Sustainable Biopolymer Packaging Utilizing Non-Food-Competing Starches Derived from Rubber Cassava and Banana Wastes
1 Master’s Program in Nanotechnology, Graduate School, Institut Teknologi Bandung, Bandung, 40132, Indonesia
2 Research Center for Electronics, National Research and Innovation Agency, Bandung, 40135, Indonesia
3 Research Center for Environmental and Clean Technology, National Research and Innovation Agency, Bandung, 40135, Indonesia
4 Research Center for Nanoscience and Nanotechnology, Institut Teknologi Bandung, Bandung, 40132, Indonesia
5 School of Pharmacy, Institut Teknologi Bandung, Bandung, 40132, Indonesia
6 Research Center for Food Technology and Processing, National Research and Innovation Agency, Yogyakarta, 55861, Indonesia
7 Research Collaboration Center for Electrochemistry, BRIN-UNDIP, Semarang, 50275, Indonesia
* Corresponding Author: Athanasia Amanda Septevani. Email:
(This article belongs to the Special Issue: Biomass-based Thermoset and Thermoplastic Polymers for Biomass-based Composites)
Journal of Renewable Materials 2026, 14(2), 5 https://doi.org/10.32604/jrm.2025.02025-0118
Received 23 July 2025; Accepted 30 October 2025; Issue published 25 February 2026
Abstract
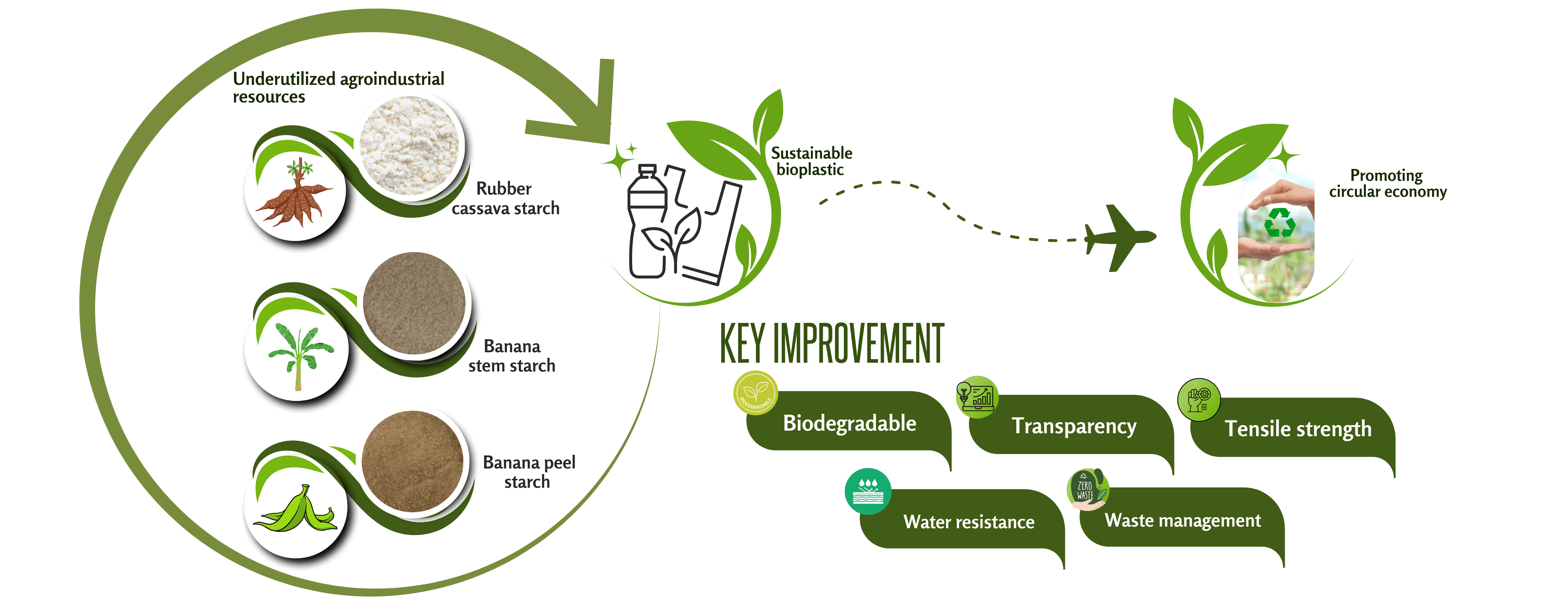
This study investigates the potential of starch extracted from underutilized agro-industrial resources as non-food-competing raw materials for the development of flexible bioplastics for food packaging applications. Starch was extracted from three biomass sources: rubber cassava (Manihot glaziovii), banana stem, and banana peel from Ambonese banana (Musa acuminata L.). Rubber cassava starch (SRC) exhibited the highest starch yield (50.68 ± 0.28%), significantly surpassing banana stem (SBS, 14.20 ± 0.25%) and banana peel (SBP, 3.07 ± 0.15%). The amylose contents of SRC, SBS, and SBP were 28.18%, 52.80%, and 56.57%, respectively, while their amylopectin contents were 71.83%, 47.20%, and 43.43%. FTIR spectra confirmed the absence of cyanogenic groups in SRC, indicating its safety for packaging applications. XRD analysis revealed that PSRC films were predominantly amorphous, while PSBS and PSBP showed higher crystallinity. The enhancement of mechanical properties, specifically PSBS, showed the highest tensile strength at 16.04 ± 0.56 MPa, whereas PSRC demonstrated the highest elongation at break at 23.57 ± 0.40%, which could be attributed to the inherent characteristics of the starch sources. Additionally, PSRC film exhibited the highest transparency at 60.2%, the greatest water solubility at 34.92%, and the lowest water contact angle at 41.58°, confirming its more hydrophilic nature compared to other films. This work highlights the potential of low-cost, sustainable, and non-food agro-industrial starch sources as promising candidates for the development of flexible, eco-friendly bioplastics.Graphic Abstract
Keywords
Plastic pollution has emerged as one of the most critical global environmental challenges, largely driven by the extensive use of petroleum-based plastics in everyday products such as packaging, consumer products, and textiles. These materials are inherently non-biodegradable, leading to their persistent accumulation in landfills and marine ecosystems, where they pose significant threats to environmental and ecological health [1,2]. The long-term presence of plastic waste in terrestrial and aquatic environments contributes to both physical pollution and microplastic contamination, as well as the release of greenhouse gases, such as methane and ethylene, further exacerbating global environmental issues [3]. These days, upwards of 300 million metric tons of plastic waste are generated yearly, yet a minimal fraction is recycled properly. As of the year 2017, it was believed that a total of 8.3 billion metric tons of plastic had been created around the globe, leading to 6.3 billion metric tons of waste [4–6]. Alarmingly, merely 9% of this waste underwent recycling, with 12% subjected to incineration, while the remaining 79% was either deposited in landfills or released into the natural environment [5]. In the event that current tendencies remain unchanged, it is expected that the international total of plastic will increase to 12 billion metric tons by 2050 [5,7–9].
These concerns have driven a growing interest in developing sustainable and biodegradable alternatives to conventional plastics. Among the most promising are bioplastics derived from renewable natural polymers, which offer potential environmental advantages due to their biodegradability and reduced carbon footprint. Starch-based bioplastics have been widely studied due to their high availability, low cost, and compatibility with conventional processing methods [10]. Various starch sources, such as corn, potato, cassava, and wheat, have been investigated using chemical and biological processing techniques, including hydrolysis, microbial fermentation, and acidification [2]. For instance, de Azêvedo et al. [10] and Kowser et al. [2] developed starch-based bioplastics using corn and potato starch, while Arifin et al. [6] explored the potential of elephant foot yam starch for similar applications.
However, the use of edible starch sources for bioplastic production raises sustainability concerns due to competition with food resources. To address this issue, non-food starch sources are being explored as more sustainable alternatives. One such candidate is Manihot glaziovii (rubber cassava), a non-edible cassava variety reported to contain a high starch content ranging from 77%–81% [11]. Despite its high starch content, it also contains elevated levels of cyanogenic compounds, rendering it unsuitable for human consumption [12]. In addition, underutilized agro-industrial residues such as banana stems and peels (from Musa acuminata L. or Ambonese banana) have shown potential as starch sources owing to their rich polysaccharide content, with reported starch yields of approximately 10%–20% from banana stems and 18%–25% from banana peels. Utilizing these residues for bioplastic production not only avoids competition with the food supply chain but also adds value to agricultural waste streams, aligning with circular economy principles [2,10]. This approach also contributes to several United Nations Sustainable Development Goals (SDGs), including responsible production and consumption, climate action, and innovation in sustainable materials.
Several studies have explored the development of starch-based bioplastics using underutilized agro-industrial residues as alternative feedstocks. For instance, starch extracted from various biomass sources has been utilized in the fabrication of bioplastic films, such as corn starch [2,13], potato starch [8], banana pseudostem [14,15], and banana peel [16]. However, most reported works have primarily focused on specific types of resources rather than exploring a broader range of potential feedstocks. A comparative study to explore potential resources for bioplastic production was conducted by Ishara et al. [17], who attempted to compare biocomposites derived from sweet cassava starch, banana peel, and banana stem. Nevertheless, the resulting materials exhibited relatively low tensile strengths in the range of 5.3–11.8 MPa. In addition, starch derived from rubber cassava has also been processed into a starch-based thermoplastic. Although the resulting material demonstrated a relatively high tensile strength of 11.94 MPa, it exhibited very low elongation at break of only 2.82%, indicating its limited flexibility and ductility for flexible packaging [18].
Despite these advances in bioplastic production using underutilized agro-industrial residues, the existing research remains fragmented with minimal comprehensive analysis to meet the specifications of commercial flexible biopackaging performances. Moreover, most of the studies were conducted using specific resources, utilizing different botanical origins, preparation methods, formulations, and processing conditions, thereby limiting direct comparability and comprehensive evaluation of their relative performances. Consequently, a systematic and comparative investigation of multiple non-food starch sources under a unified processing framework has yet to be conducted. To address this critical research gap, the present study provides the first direct comparative assessment of bioplastic films derived from rubber cassava, banana stem, and banana peel (from Musa acuminata L., Ambonese banana). Such a comparative approach is crucial for identifying the most promising raw material among diverse agro-industrial residues and for advancing the development of sustainable, high-performance bioplastics.
Starch sources, including rubber cassava, banana stem, and banana peel from Ambonese bananas, were collected from West Java, Indonesia. Glycerol (85%, p.a), sodium hydroxide (NaOH), potassium iodide (KI), hydrochloric acid (HCl), amylose standard, and acetic acid (glacial) were purchased from Merck, while technical grade of sodium bisulfate (NaHSO4) was supplied from a local chemical store in West Java, Indonesia. Distilled water was used throughout all experimental procedures. Unless otherwise specified, all chemicals were used as received without further purification.
2.2 Extraction of Starch from Banana Stems, Banana Peels, and Rubber Cassava
Starch was extracted from banana stems, banana peels, and rubber cassava using a modified wet extraction method adapted from Abera et al. [19]. The raw materials were first thoroughly cleaned and chopped into small pieces. They were then homogenized with distilled water containing 0.25 g/L NaHSO4 at a 1:10 (w/v) ratio for each source. The resulting mixtures were stored at 6°C for 24 h to facilitate starch release. After cold incubation, the slurries were filtered using muslin cloth to remove coarse fibers, and the filtrates were centrifuged at 3000 rpm for 15 min to separate the starch. The collected wet starch was dried in an oven at 40°C overnight, ground into a fine powder, and sieved using a 100 μm mesh to obtain uniform starch particles. Each starch extract produced from rubber cassava, banana stem, and banana peel was denoted as SRC, SBS, and SBP, respectively.
2.3 Characterization of Starch
The mass of the extracted starch was measured after the drying process. The percentage yield was then calculated using the following formula (Eq. (1)):
This calculation quantifies the efficiency of the extraction method, indicating the proportion of starch successfully recovered from the initial biomass.
Bulk density was determined by placing 1.0 g of the starch sample in a 25 mL graduated cylinder. The cylinder was gently tapped ten times from a height of 5 cm. After tapping, the sample volume was recorded. The bulk density was calculated as the ratio of the sample weight to the tapped volume [20].
The amylose and amylopectin contents of the starch were determined using a UV-Vis spectrophotometer [21]. For amylose stock solution preparation, 100 mg of amylose was dispersed in 1 mL of absolute ethanol. Subsequently, 9 mL of 1 M NaOH solution was added. The solution was placed in a boiling water bath for 15 min with frequent shaking to obtain a clear solution. The total volume was then adjusted to 100 mL with distilled water to yield the amylose stock solution. To generate the absorption spectrum, 4 mL of the amylose stock solution was added to 30 mL of distilled water. The pH was then adjusted to 3.6 using 0.5 M HCl. Subsequently, 0.5 mL of iodine-potassium iodide (I2-KI) solution (2 g/L I2, 20 g/L KI) was added, and the total volume was adjusted to 50 mL with distilled water. In this reaction (Eq. (2)), molecular iodine (I2) reacts with iodide ions (I−) to form triiodide (I3−), which intercalates into the amylose helical structure to form a dark blue amylose-triiodide inclusion complex (Eq. (3)), as shown below:
The reaction mixture was thoroughly mixed, allowed to stand at room temperature for 30 min, and then scanned using a spectrophotometer at 600 nm. For the amylose standard curve, the amylose stock solution was diluted to prepare reaction solutions with final concentrations of 10, 20, 30, 40, 50, 60, and 70 µg/mL, respectively [19]. Based on this amylose standard calibration curve, the maximum amylose content in starches was determined using the following formula (Eq. (4)) [19].
Amylopectin content was then calculated as the remaining percentage (Eq. (5))
Lignin content in the extracted starch was analyzed following the NREL (National Renewable Energy Laboratory) standard method [22]. The analysis involved subjecting the starch sample to a two-stage acid hydrolysis process. After hydrolysis, the total lignin content was calculated by summing the acid-insoluble lignin and acid-soluble lignin fractions.
The functional groups of starch were characterized using a Thermo Scientific Nicolet iS5 in the ATR method, within a wavenumber range of 4000–500 cm−1, at the BJ Habibie Science and Technology Area-BRIN.
Crystallinity was analyzed using an X-ray diffractometer (D8 Advance, Bruker) equipped with a Cu Kα radiation source. Measurements were performed at scattering angles (2θ) ranging from 4° to 90° at the Samaun Samadikun Science and Technology Area-BRIN.
The morphology and elemental composition of the starch samples were examined using a scanning electron microscope (SEM) coupled with energy-dispersive X-ray spectroscopy (EDX). Analysis was performed using a JEOL JSM-IT300LV SEM located at the Samaun Samadikun Science and Technology Area-BRIN.
2.4 Preparation of Bioplastic Film
Bioplastic samples were prepared based on a modified method from Abdullah et al. [23]. The preparation process consisted of two main stages: mixing and hot pressing, as further described. In the mixing stage, starch (60% w/w), glycerol (20% w/w), and acetic acid (20% w/w) were blended. The mixture was stirred using a mechanical mixer until a homogeneous, solid-like bioplastic paste was formed. The resulting paste was then transferred onto a Teflon surface and covered with a mold in preparation for hot pressing. In the hot pressing stage, the molded mixture was subjected to compression molding using a hot press at a temperature of 120°C and a pressure of 40 kgf/cm2 for approximately 38 min. The resulting bioplastic films derived from SRC, SBS, and SBP are denoted as PSRC, PSBS, and PSBP, respectively. For comparison, bioplastics derived from commercial sweet cassava starch were also prepared using the same method and denoted as PSCM.
2.5 Characterization and Performance of Bioplastic Film
The functional groups of the starch-based bioplastics were characterized using a Thermo Scientific Nicolet iS5 Fourier-transform infrared (FTIR) spectrometer in ATR mode. Spectra were recorded within a wavenumber range of 4000–500 cm−1 at the Samaun Samadikun Science and Technology Area-BRIN.
Crystallinity was analyzed using an X-ray diffractometer (D8 Advance, Bruker) equipped with a Cu Kα radiation source. Measurements were performed at scattering angles (2θ) ranging from 4° to 90° at the Samaun Samadikun Science and Technology Area-BRIN.
The morphology of the starch-based bioplastic samples was examined using a scanning electron microscope (SEM). Analysis was performed using a JEOL JSM-IT300LV SEM located at the Samaun Samadikun Science and Technology Area-BRIN.
Water solubility of bioplastic film was determined following the procedure described by Oblitas et al. [24] with slight modifications. Briefly, a known mass (Wi) of dried bioplastic film was immersed in 50 mL of distilled water and maintained at room temperature for 6 h under static conditions. The undissolved residue was then recovered by filtration using Whatman No. 40 filter paper and dried in an oven at 60°C until a constant weight (Wf) was obtained. Water solubility was calculated using Eq. (6):
where Wi is the initial dry weight of the bioplastic film, and Wf is the final dry weight of the undissolved residue.
The hydrophilicity of the bioplastic film was evaluated by measuring the water contact angle (WCA) using the sessile drop method. A synthesized bioplastic film was cut into 1 × 1 cm pieces, and a single drop of distilled water was deposited on the surface at room temperature using a syringe. The contact angle formed between the water droplet and the bioplastic surface was recorded and analyzed using the DropSnake plugin in ImageJ software. Lower contact angle values indicate higher hydrophilicity of the material surface.
Tensile strength specimens were prepared and conditioned according to ASTM D882 standard for thin plastic sheeting. Testing was performed using a universal testing machine (UTM) equipped with a 500 N load cell at a crosshead speed of 10 mm/min. From the developed plastic sheets, dumbbell-shaped film strips with uniform dimensions were cut for analysis [1]. The tensile strength and elongation at break of each sample were determined using Eqs. (7) and (8) below:
The transparency of the bioplastic films was determined using a UV-Vis spectrophotometer at a wavelength of 600 nm. Prior to measurement, the films were cut into uniform rectangular pieces and gently wiped clean. The transmittance of each film was recorded, and its opacity was calculated using the following equation (Eq. (9)):
A higher opacity value indicates a less transparent film. Each measurement was performed in triplicate to ensure reproducibility.
3.1 Properties of Starch Derived from Banana Stems, Banana Peels, and Rubber Cassava
Starch isolation from underutilized agro-industrial resources was carried out via the wet extraction method. This method was selected over the dry extraction approach due to its advantages in obtaining starch with higher purity and minimal contamination from proteins, lipids, and fibers [25]. Moreover, the wet method allows better preservation of starch granule morphology, which is essential for subsequent applications, particularly in material development. While the wet method has the drawback of promoting microbial growth during processing, sodium bisulfate was employed as both an antimicrobial and antioxidant agent. It effectively suppressed microbial growth and inhibited oxidative reactions by scavenging dissolved oxygen and reactive oxygen species (Eq. (10)). In addition, its acidic nature likely contributed to the denaturation of amylase enzymes, thereby preventing enzymatic hydrolysis of glycosidic bonds within the starch backbone (Eq. (11)) [19,26].
The yield, density, amylose-amylopectin, and lignin content of the extracted starches from different biomass sources are presented in Table 1.
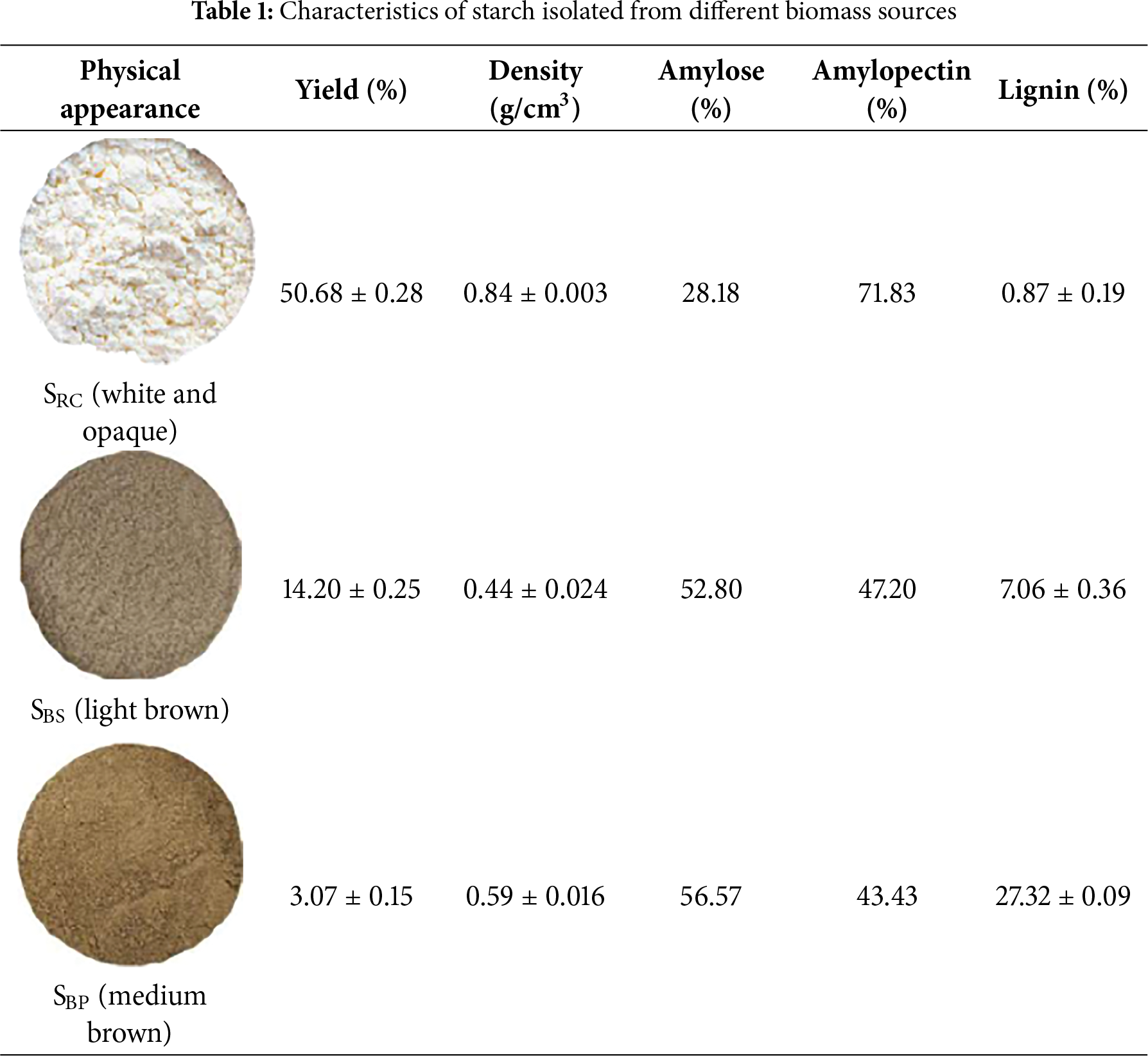
Among the tested samples (Table 1), SRC exhibited the highest starch yield (50.68 ± 0.28% dry basis), which is expected as a major carbohydrate source, compared to SBS and SBP. This result falls within the typical starch content range reported for rubber cassava (40–70% dry basis) [11]. SBS and SBP resulted in significantly lower starch yields compared to SRC, at 14.20 ± 0.25% and 3.07 ± 0.15%, respectively, reflecting the predominantly structural and protective roles of banana stem and peel tissues [27,28]. These tissues possess a high lignocellulosic content that forms rigid matrices, impeding starch release. In particular, SBP exhibits the highest lignin content at 27.32 ± 0.09% followed by SBs at 7.06 ± 0.36%, while SRC shows the lowest lignin content at 0.87 ± 0.19%. The significant presence of lignin, particularly in SBP and SBS, can physically entrap starch granules within fibrous cell walls, thereby reducing limiting starch extraction efficiency and resulting in notably lower starch yields, accordingly [29]. Further, lignin content also correlates with color characteristics, where higher lignin levels contribute to the darker appearance of SBS and SBP. Hence, SRC containing very low lignin content, exhibited white and opaque indicating higher starch purity.
Interestingly, the SBS yield obtained in this study exceeded previous studies from similar banana pseudostem matrices, including 5% reported in Puwalu, Sri Lanka [14], and 10.35 ± 0.76% as reported by Mohd Ali et al. [15]. Meanwhile, the SBP yielded less starch than previous studies, such as 7.1% from green banana peel [16] and 28.24 ± 0.68% from industrial peel waste in Chennai, India [30]. These disparities may arise from a complex interplay of biological and methodological factors. Genetic diversity within Musa spp., agroclimatic conditions, harvesting maturity, and post-harvest storage practices can significantly influence starch accumulation and localization. Moreover, the physical form and pretreatment of the biomass, such as particle size, soaking duration, extraction solvent, and drying method, further affect yield. In particular, fruit ripening shifts the starch–sugar equilibrium, influencing extractable starch levels [26,27,29,31].
Further, the bulk density and amylose–amylopectin composition of the extracted starches are critical determinants of their suitability for bioplastic production. SRC showed the highest bulk density (0.84 ± 0.003 g/cm3), suggesting a compact granule structure with low interparticle voids, which is favorable for uniform film casting and consistent material packing. In contrast, SBS (0.44 ± 0.024 g/cm3) and SBP (0.59 ± 0.016 g/cm3) exhibited lower densities, likely due to porous or irregular granule morphology, which could compromise mechanical integrity during film formation.
The amylose content varied markedly among samples. SBP and SBS contained higher amylose levels (56.57% and 52.80%, respectively), whereas SRC had a lower amylose content (28.18%) and correspondingly higher amylopectin (71.83%). Given amylose’s linear structure and stronger intermolecular interactions, this suggests that SBP and SBS starches are likely to produce more rigid and mechanically strong bioplastics. Conversely, the high amylopectin content in SRC (71.83%) implies a more branched polymer network that typically imparts greater flexibility but lower strength. These compositional features suggest different mechanical profiles among the resulting bioplastics, which will be further validated through morphological characterization of the starch via SEM and mechanical testing of bioplastic films.
The structural characteristics of starch samples, SRC, SBS, SBP, and SCM, were investigated using Fourier transform infrared (FTIR) spectroscopy (Fig. 1). All samples exhibited a broad absorption band centered around 3300–3400 cm−1, which is attributed to O–H stretching vibrations arising from inter- and intramolecular hydrogen bonding within the starch matrix. Additionally, C–H stretching vibrations near 2920 cm−1 were consistently observed across the spectra, corresponding to the aliphatic –CH and –CH2 groups of the glucopyranose rings, as typically reported in both native and modified starches [32,33].
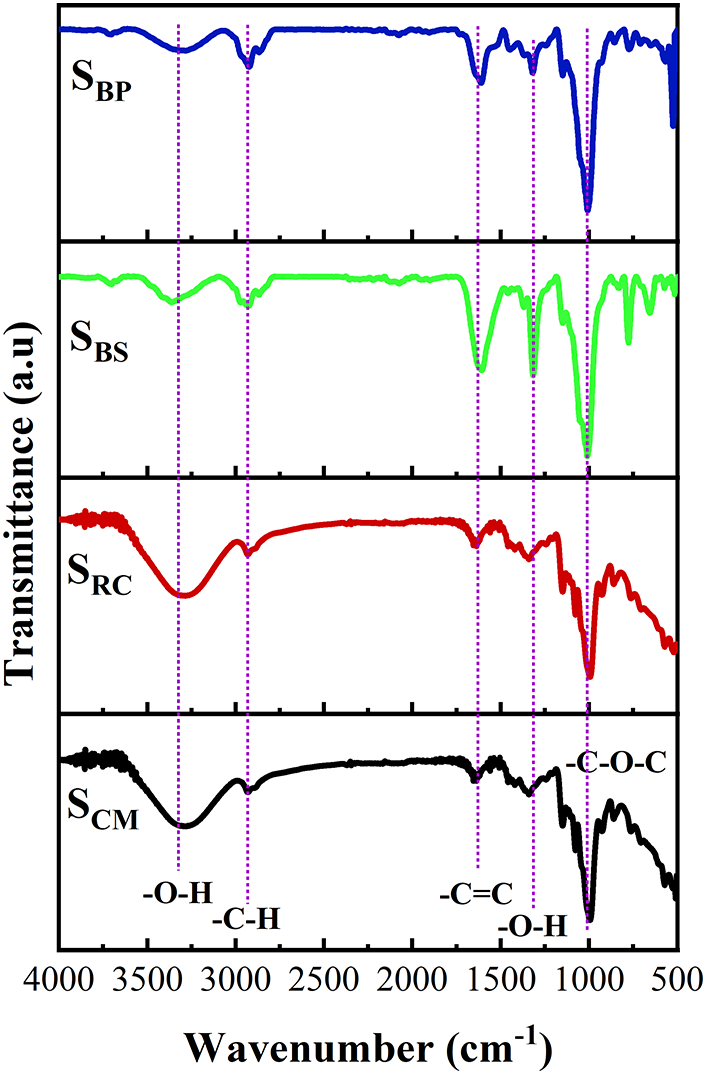
Figure 1: FTIR spectra of starch
A distinct absorption band near 1600 cm−1 was observed in the SBS and SBP samples, which was assigned to aromatic C=C stretching vibrations, suggesting the presence of residual lignin or phenolic compounds as evidenced by the higher content of lignin correspondingly (Table 1). In contrast, this band was considerably less intense in the spectra of SRC and SCM, indicating a higher chemical purity of cassava-derived starches. The persistence of lignin-related bands in SBS and SBP implies the co-extraction of non-starch polysaccharides, phenolic residues, or other plant matrix components. This can be attributed to the intrinsic structural complexity of pseudostem and peel tissues, coupled with limited extraction or pretreatment efficiency. The presence of these aromatic moieties also correlates with the light brown and medium brown coloration observed in the SBS and SBP powders, respectively (Table 1). These observations are consistent with the findings of Silva et al. [34], who demonstrated that blanching of banana peel prior to film production significantly reduced browning by inactivating polyphenol oxidase, thereby minimizing the enzymatic oxidation of phenolics into quinones and melanin pigments. Such pretreatment strategies could enhance the purity and optical properties of banana-based biopolymers.
Moreover, all starch samples displayed strong and broad absorption bands in the region of 1020–1030 cm−1, corresponding to C–O–C and C–O stretching vibrations in polysaccharides, commonly regarded as the fingerprint region for glycosidic bonds in starch [35,36]. The spectra of SRC, in particular, appeared relatively clean and well-defined, indicative of successful starch isolation from low-lignin biomass. This aligns with the high yield and low mineral content observed for SRC, in contrast to the chemically more heterogeneous SBS and SBP, as further supported by SEM and EDX analysis (Section 3.1.3).
Starch is primarily composed of two polysaccharide macromolecules, amylose and amylopectin, which are responsible for its amorphous and crystalline domains, respectively. To investigate the crystalline structure and phase composition of starches obtained from various agro-industrial sources, XRD analysis was performed and compared to the diffraction pattern of commercial cassava starch. As shown in Fig. 2, starch extracted from rubber cassava (SRC) exhibited diffraction patterns closely resembling that of the commercial cassava starch (SCM), with prominent reflections at 2θ = 14.9°, 16.9°, 17.9°, and 22.9°, along with weaker peaks at 2θ = 11.2° and 26.4°, all characteristic of an A-type crystalline polymorph [37]. These profiles are typical of starches derived from cereals and tuberous crops, consistent with the botanical origins of Manihot glaziovii (SRC) and Manihot esculenta Crantz (SCM).
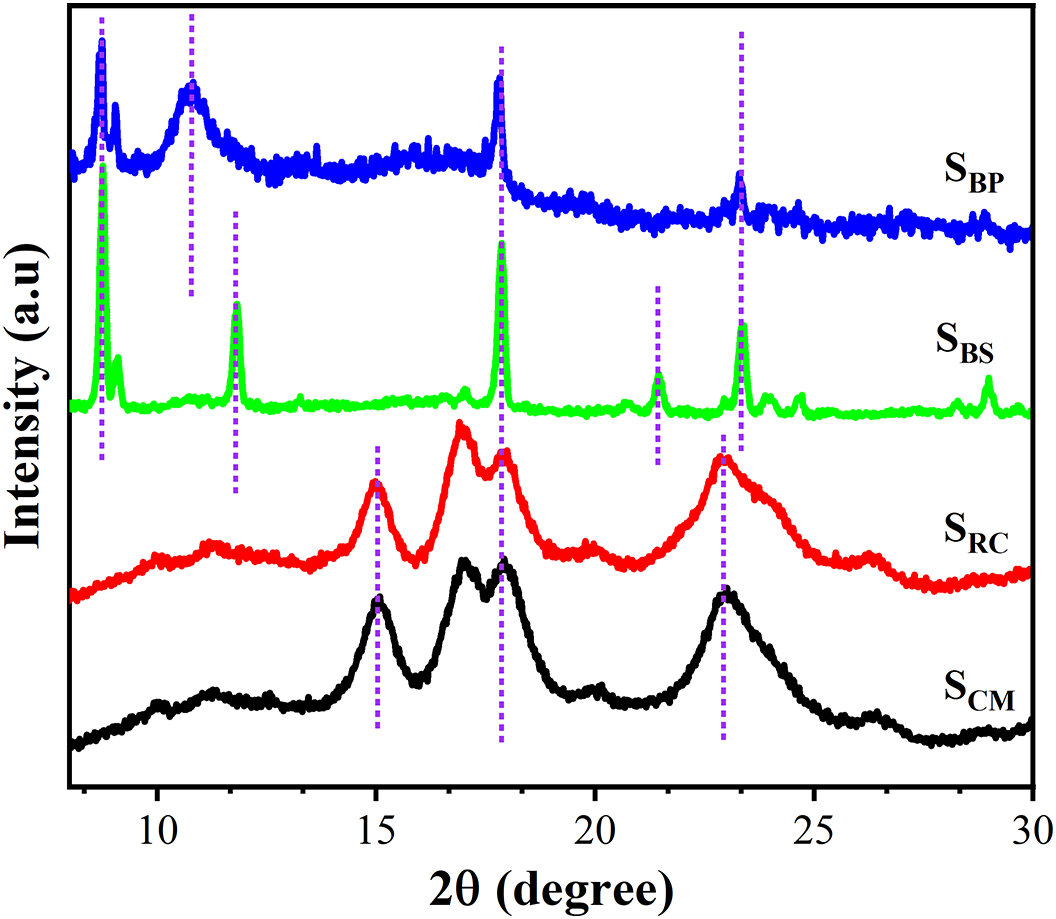
Figure 2: XRD pattern of various starch samples
In contrast, starches extracted from banana stem (SBS) and banana peel (SBP) displayed diffraction patterns indicative of a C-type crystalline structure, as evidenced by additional peaks at 2θ = 8.7° and 10.7° for SBP, and 8.7° and 11.8° for SBS, along with a distinct reflection at 2θ = 17.9° [38]. Notably, the SBS sample exhibited more intense and well-defined crystalline peaks, resembling those typically associated with α-amylose [39], which aligns with its higher amylose content (Table 1), suggesting a higher degree of crystallinity compared to the other samples.
3.1.3 Morphology Analysis of Starch
The surface morphology of the isolated starch granules was examined via SEM to elucidate the structural characteristics and purity of starches. The differences in granule morphology observed across the samples provide valuable insights into their extraction efficiency, molecular composition, and potential processability in biopolymer applications. SRC exhibited well-defined, smooth, and predominantly oval to round granules with relatively uniform size distribution (Fig. 3). This regular morphology reflects the efficient isolation of starch from tuberous cassava tissue, which is characterized by low lignin content and minimal fibrous obstruction. The high bulk density, as presented in Table 1, further supports the notion that uniform and compact starch granules in SRC were efficiently obtained with minimal structural disruption during extraction. Additionally, the SEM-EDX results show that the SRC indicates a high chemical purity with minimal mineral contamination.

Figure 3: Morphological analysis and elemental composition of (a,b) SRC; (c,d) SBS; and (e,f) SBP
In contrast, SBS exhibited irregular, fragmented structures with disrupted or partially gelatinized granules embedded in a fibrous matrix. This disordered morphology is consistent with the low starch yield and bulk density (Table 1), suggesting that the complex lignocellulosic framework of banana pseudostem tissue hinders efficient starch release. These morphological features indicate that a significant portion of starch may remain entrapped within cellulose–lignin networks. The elevated amylose content (Table 1) of SBS may also contribute to partial collapse or retrogradation of granules during processing, given amylose’s tendency to recrystallize and aggregate. FTIR analysis of starches supports these morphological findings (Section 3.1.1), which indicate residual hemicellulose and lignin, respectively. These components likely interfered with starch granule liberation and compromised the structural uniformity observed under SEM. Similarly, SBP demonstrated a highly heterogeneous surface morphology characterized by flattened or ruptured particles intermixed with amorphous residues. This is consistent with the extremely low starch yield and moderate density (Table 1), suggesting limited starch content.
3.2 Properties of Bioplastic Films
3.2.1 FTIR Analysis of Bioplastic Films
The structural characteristics of the fabricated starch-based bioplastics, PSRC, PSBS, PSBP, and PSCM were characterized using FTIR spectroscopy (Fig. 4). All bioplastic films exhibited broad absorption bands around 3200–3300 cm−1, corresponding to O–H stretching vibrations from inter- and intramolecular hydrogen bonding. Compared to native starches, where the –OH band appeared at 3300–3400 cm−1, there is a shift to lower wavenumbers indicating stronger hydrogen-bonding interactions in the bioplastic films. This shift indicates the formation of a new hydrogen-bonding network between starch and glycerol, which contributes to enhanced elasticity and flexibility, resulting in the development of thermoplastic starch (TPS) [40]. Additionally, C–H stretching vibrations near 2920 cm−1 were consistently observed, associated with the aliphatic –CH and –CH2 groups in the glucopyranose units [32,33].
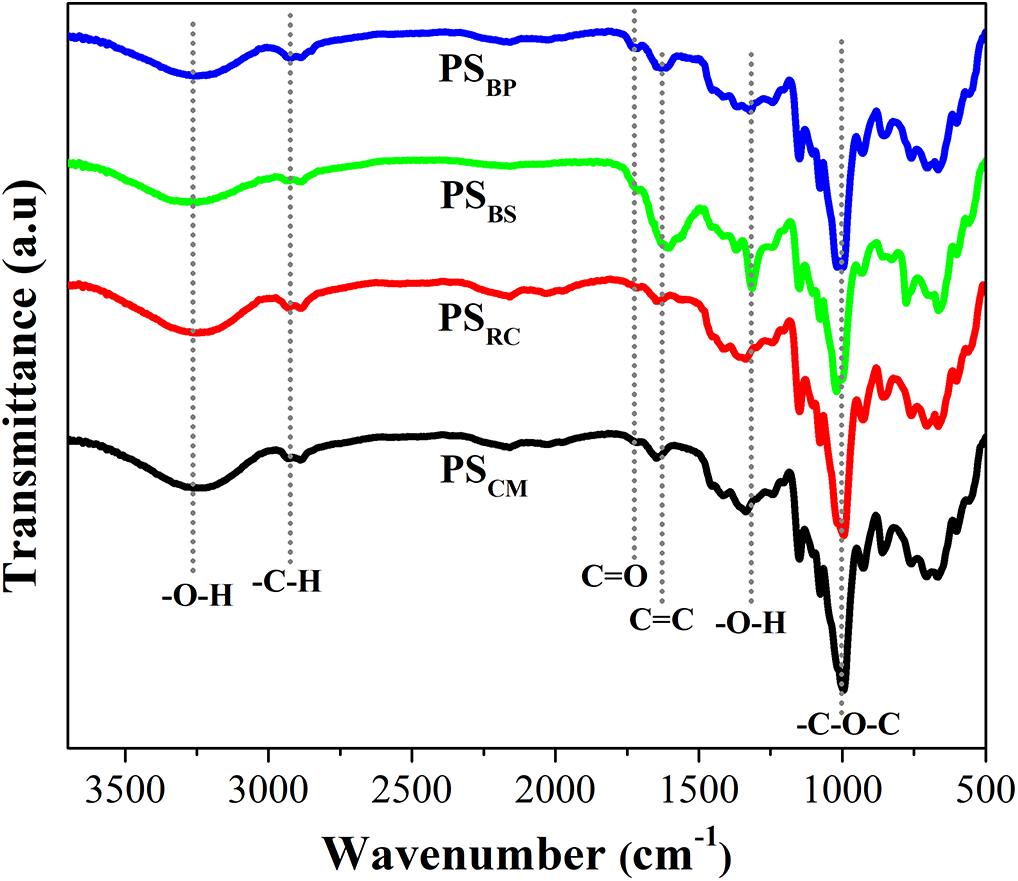
Figure 4: FTIR analysis of starch-based bioplastics
A notable absorption band near 1730 cm−1 emerged with significantly higher intensity in PSBS and PSBP compared to PSRC and PSCM. This band is attributed to C=O stretching vibrations, which in the context of these bioplastics may arise from two possible sources: ester linkages introduced via crosslinking with acetic acid during film formation [41,42] and residual carboxylic groups or aldehydes associated with lignin or hemicellulose, suggesting a greater contribution from lignocellulosic impurities in the banana-derived films [43]. Supporting this interpretation, both PSBS and PSBP also exhibited an additional band near 1600 cm−1, which corresponds to aromatic C=C stretching vibrations, a spectral feature also prominently observed in the SBS and SBP starches [43–45]. These observations indicate the presence of non-starch polysaccharides, phenolic compounds, or mineral contaminants that were retained throughout the bioplastic fabrication process.
Despite the relatively high amylose content in SBS and SBP (Table 1), which typically promotes the formation of ordered starch granules, the prominent spectral contributions from non-starch components suggest that the granule integrity may have been disrupted during thermal processing. The elevated intensities of both O–H and C=O bands may also reflect the presence of oxidized degradation products, likely derived from incomplete washing or thermal decomposition of peel constituents, which contribute to poor granule cohesion and compositional heterogeneity in the final films.
In contrast, the PSRC and PSCM films revealed fewer interfering bands, with no pronounced signals in the carbonyl or aromatic regions, indicating higher chemical purity. Moreover, all bioplastic samples retained a strong and broad absorption band between 1020 and 1030 cm−1, attributed to C–O–C and C–O stretching vibrations typical of glycosidic linkages in starch-based polysaccharides [35,36]. Importantly, although rubber cassava is known to contain cyanogenic compounds, the FTIR spectra of PSRC showed no absorption peaks associated with CN groups, confirming the absence of such toxic residue in the bioplastics. This finding is particularly significant for ensuring material safety, supporting the utilization of rubber cassava as an alternative, non-food-competing starch source for packaging, including potential use in food-contact and other advanced applications.
3.2.2 XRD Analysis of Bioplastic Film
The X-ray diffraction patterns of starch-based bioplastics derived from banana peel (PSBP), banana stem (PSBS), and rubber cassava (PSRC) are presented in Fig. 5. Among these, the PSBS sample exhibits the most prominent and well-defined diffraction peaks, particularly at 2θ ≈ 17°, 22°, and 30°, indicative of a highly crystalline phase, which is consistent with the inherent crystallinity of its native starch (Fig. 2). In contrast, PSBP shows a broad halo centered at 2θ ≈ 18°–20°, with barely noticeable crystalline signals, suggesting a semi-crystalline to predominantly amorphous structure. Similarly, the PSRC sample displays a broad, diffuse pattern with a hump around 2θ ≈ 19°–20°, but with lower diffraction intensity compared to PSBP, indicating a predominantly amorphous structure. The XRD patterns of PSBP and PSRC are similar to those of corn starch bioplastics prepared by Kowser et al. [2].
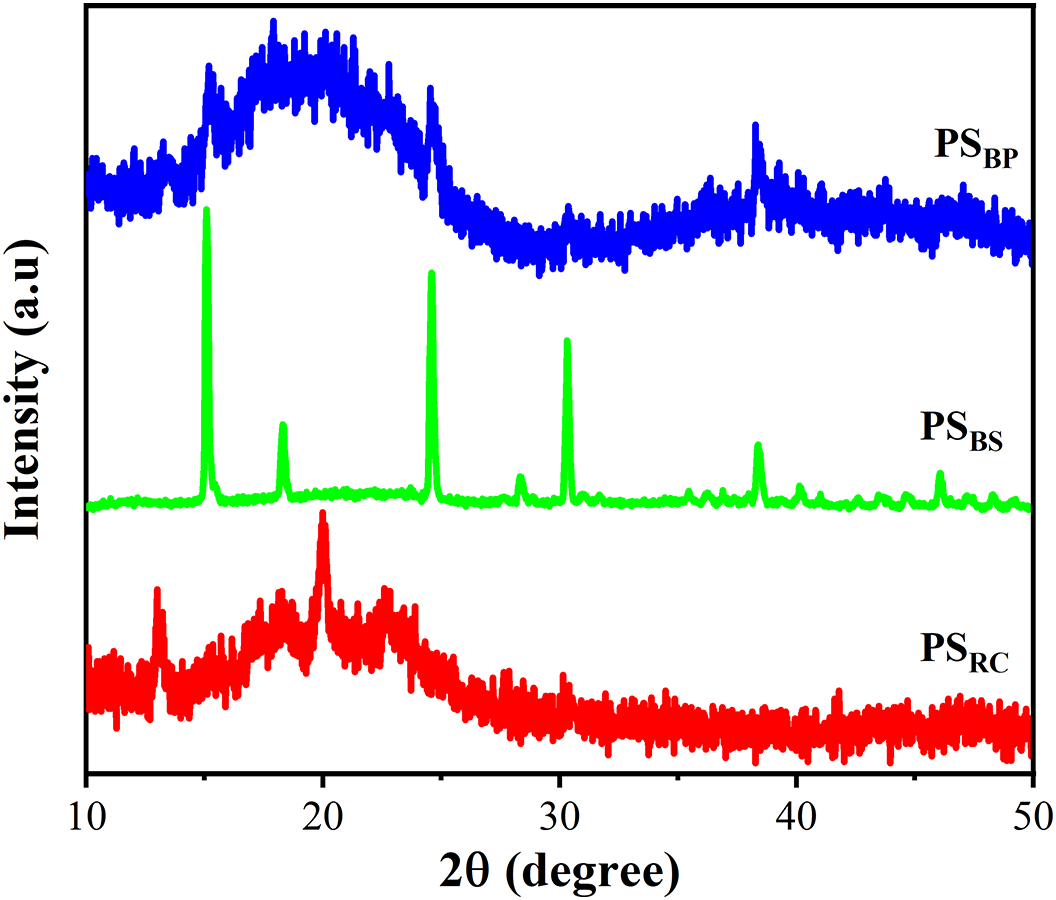
Figure 5: XRD pattern of starch-based bioplastics
These variations in crystalline structure are reflected in the mechanical and barrier properties of the bioplastics. The higher crystallinity observed in PSBS is correlated with enhanced mechanical strength and reduced water solubility, as will be discussed in detail in the following section. This behavior suggests a more compact and less permeable polymer matrix.
3.2.3 Morphology Analysis of Bioplastic Film
The surface morphologies of starch-based bioplastics are shown in the SEM images at 250× and 1000× magnifications in Fig. 6. The PSRC film exhibits a relatively uniform surface compared to the others, simply because the well-defined and predominantly oval to round granules with relatively uniform size distribution of raw starch SRC (Fig. 3). In contrast, the PSBS film displayed a rough and densely textured surface, with rod-like structures becoming more apparent at higher magnification. The PSBP film showed a relatively smooth surface at 250× magnification; however, at 1000× magnification, a partially dense, layered structure with observable surface cracks was evident. The fractured surface of the PSBP film revealed characteristics of brittle failure, whereas such features were less pronounced in the PSBS film.

Figure 6: Morphology analysis of bioplastic film from rubber cassava (a), banana stem (b), and banana peel (c)
3.2.4 Water Solubility and Wettability Analysis
Water solubility analysis was conducted to evaluate the interaction between the starch-based bioplastics and water, particularly their susceptibility to water-induced disintegration. The results are summarized in Table 2. Among the tested samples, PSRC exhibited the highest water solubility at 34.92%, while PSBS and PSBP showed lower solubility values of 24.30% and 25.00%, respectively. These results suggest that PSRC films are more prone to water uptake and dissolution.
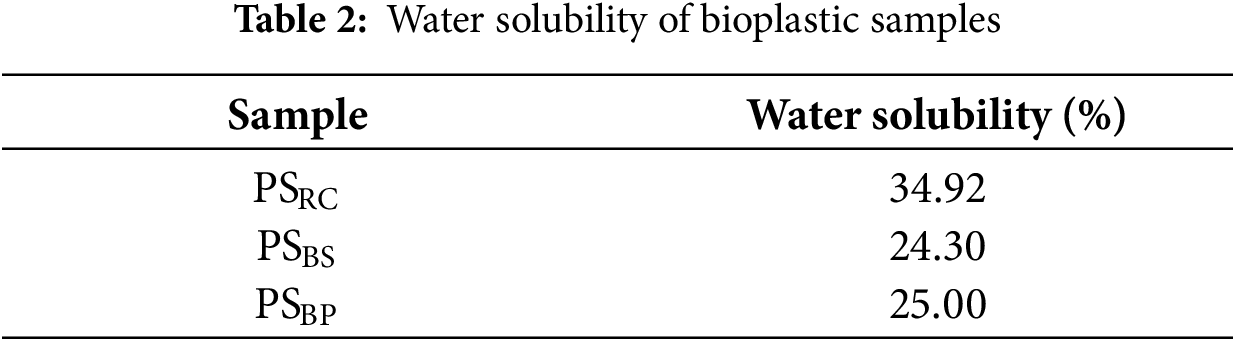
The relatively high solubility of PSRC can be attributed to the intrinsic properties of starch, a hydrophilic polysaccharide composed of numerous hydroxyl groups capable of forming hydrogen bonds with water molecules [19]. In particular, the amorphous structure of PSRC likely enhances water penetration and increases polymer chain mobility, thereby facilitating the dissolution process. This interpretation is supported by the corresponding XRD data, which revealed broad and diffuse peaks for the rubber cassava-based film, indicative of low crystallinity [46]. In contrast, PSBS and PSBP exhibited sharper and more defined diffraction peaks, reflecting a higher degree of crystallinity. These ordered structures typically hinder water diffusion and restrict polymer chain relaxation, ultimately reducing water solubility [46]. The water solubility values obtained in this study are consistent with those reported by Singh et al. [47], who developed bioplastics from sago and potato starch, reporting solubility values ranging from 26% to 33%.
To further elucidate the surface wettability of the bioplastics, WCA measurements were performed, with the results presented in Fig. 7. Consistent with the solubility data, PSBP films showed the highest contact angle, followed by PSBS and PSRC. The contact angle serves as a quantitative indicator of surface hydrophilicity, where values below 90° generally reflect hydrophilic surfaces, while values above 90° are associated with hydrophobic behaviour [48,49]. The inverse relationship observed between contact angle and water solubility supports the hypothesis that the more hydrophilic and amorphous nature of PSRC films facilitates water uptake and dissolution. Meanwhile, the higher crystallinity and lower wettability of PSBS and PSBP films limit water diffusion and promote structural stability.

Figure 7: Water contact angle (WCA) of (a) PSRC, (b) PSBS, and (c) PSBP
These findings on solubility and wettability also provide insight into the potential biodegradability of the bioplastics. Starch, widely used in bioplastic production due to its abundance and low cost, is known to degrade effectively under composting and soil burial conditions, with degradation rates influenced by environmental factors such as temperature, moisture, pH, and microbial activity. The relatively high water solubility suggests that the material may be more readily accessible to microbial colonization in an aqueous environment. This aligns with the findings of Lai et al. [50], who reported that bioplastic-based zein protein amyloid fibrils and methylcellulose with high hydrophobicity and wet stability required long-term burial for complete biodegradation.
Previous studies have also examined the biodegradability of starch-based bioplastic in various environments, including soil, seawater, and freshwater systems [51]. For example, Ghasemlou et al. [52] reported that corn starch-based bioplastic films achieved approximately 70% degradation after 120 days of soil burial. Conversely, Accinelli et al. [53] observed only 1.5% degradation of starch-based bioplastics under marine and freshwater conditions at 25∘C after 90 days, highlighting the slower degradation rates in aquatic settings. Nevertheless, other studies have shown significantly higher degradation rates in marine sediments. The results showed that starch-based shopping bags degraded by 69% (weight loss) within 236 days when exposed to seawater and sediment, likely due to both the material characteristics and the environmental conditions [54]. To this end, starch-based bioplastics can degrade readily, with rates varying across different environments. Yet, the mechanical strength should comply with retaining adequate performance during their intended service life, as discussed in the following section.
3.2.5 Mechanical Properties of Bioplastic Films
The mechanical performance of PSRC, PSBS, and PSBP starches was evaluated and compared with a reference film made from commercial cassava starch (PSCM). The tensile strength and elongation at break data (Fig. 8) reveal distinct mechanical profiles for each material, which correlate closely with the physicochemical composition and microstructural characteristics of the starch sources.
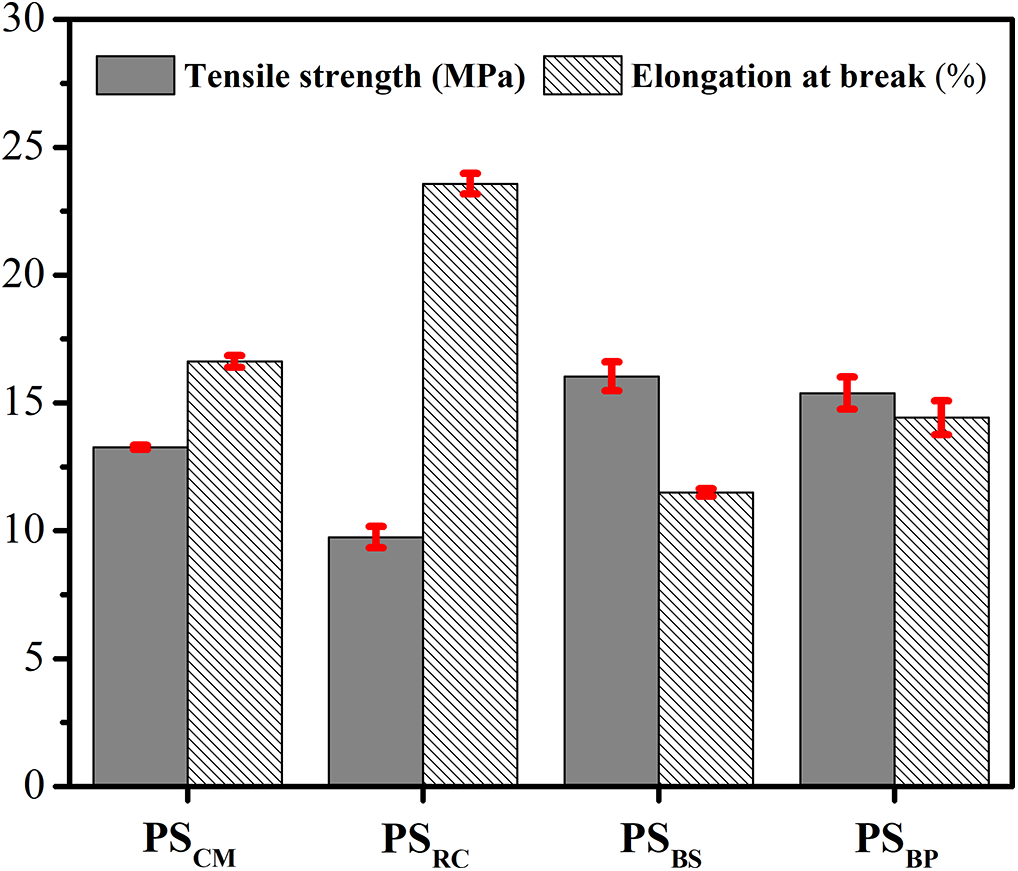
Figure 8: Mechanical properties of starch-based bioplastic
Among all samples, PSBS exhibited the highest tensile strength (16.04 MPa), indicating a highly rigid matrix, albeit with the lowest elongation at break (11.5%). This mechanical behavior is primarily attributed to its high amylose content (52.80%), which promotes dense chain packing and extensive intermolecular hydrogen bonding, contributing to high film stiffness. The SEM micrograph of SBS confirmed a fragmented and fibrous surface morphology, likely resulting from incomplete removal of lignocellulosic components during extraction. EDX analysis further supported this interpretation by detecting a substantial presence of inorganic ions, including Ca (3.28%) and Mg (0.81%), both of which are known to interact ionically with starch hydroxyl groups. These interactions likely served as physical crosslinks, reinforcing the starch matrix and enhancing tensile strength while simultaneously restricting chain mobility and reducing ductility.
In contrast, PSRC presented the lowest tensile strength (9.75 MPa) but the highest elongation at break (23.57%), indicating a more flexible and extensible bioplastic. This material was predominantly composed of amylopectin (71.83%), whose branched molecular structure disrupts crystallinity and facilitates polymer chain slippage under stress. The SEM image of SRC showed well-defined, spherical, and homogeneously distributed starch granules, which are favorable for uniform gelatinization and plasticization. Furthermore, the EDX spectrum revealed minimal presence of interfering mineral elements, indicating high chemical purity that supports the formation of a clean and thermoplastic film network.
The transformation into thermoplastic starch occurs during the mixing stage, where starch granules interact with glycerol and acetic acid. Glycerol functions as a plasticizer by forming hydrogen bonds with starch hydroxyl groups, thereby replacing strong starch–starch interactions and reducing rigidity. Acetic acid facilitates gelatinization and enhances molecular mobility. During hot pressing at 120∘C and 40 kgf/cm², the combined effects of heat and shear disrupt the crystalline regions, leading to gelatinization and loss of granular structure. These molecular rearrangements, together with starch–glycerol interactions, promote the formation of homogeneous, flexible, and continuous thermoplastic starch films, as also evidenced by the FTIR results (Fig. 4) [40,55].
PSBP demonstrated an intermediate mechanical profile, with a tensile strength of 15.38 MPa and elongation at break of 14.42%. Despite possessing the highest amylose content (56.57%), PSBP did not achieve the tensile performance observed in PSBS. This discrepancy may be explained by the highly irregular and aggregated granule morphology visible in the SEM image, which may compromise the continuity and stress distribution of the film. Additionally, while EDX results indicated a relatively clean elemental profile (C = 60.64%, O = 38.87%, Ca = 0.5%), the low starch yield (3.07%) could suggest the presence of residual fiber or non-starch polysaccharides that limit structural coherence at the macro scale. Thus, although chemically suited for high-strength films, PSBP may require further refinement to achieve its full mechanical potential.
Overall, the mechanical performance of the bioplastics derived from these underutilized agro-industrial resources is comparable to that of the control film (PSCM), which recorded a tensile strength of 13.26 MPa and elongation at break of 16.62%. Notably, these values surpass those reported in previous studies. For instance, bioplastics developed from banana peel reinforced with pseudostem cellulosic fibers only achieved a tensile strength of 7.23 MPa [19]. Other studies reported even lower strengths for films based on cassava (4.5 MPa), corn starch (3.59 MPa), and potato starch (4.87 MPa) [56]. Another study also reported lower tensile strength for bioplastic-based corn starch (5.62 MPa) [13]. In terms of flexibility, the elongation at break obtained in this study significantly exceeded previous findings, such as 5.46% for banana peel-based film reinforced with pseudostem fibers, 8.69% for antimicrobial plastic films [56], and 4.32% for bioplastics developed from corn starch reinforced with bagasse-derived fibers [57].
3.2.6 Physical Properties of Bioplastic Films
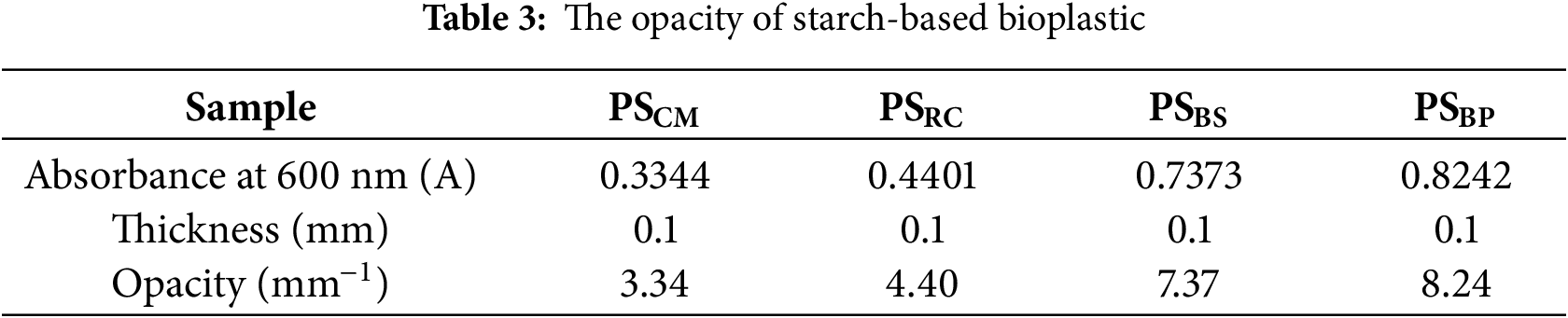
The optical transparency of starch-based bioplastics is a critical functional property, particularly for packaging applications where product visibility is a desired attribute. The visible light transmittance spectra from PSCM, PSRC, PSBS, and PSBP are shown in Fig. 9. Opacity values (Table 3), derived from the absorbance at 600 nm standardized to a 0.1 mm film thickness, were determined to be 3.34 (PSCM), 4.40 (PSRC), 7.37 (PSBS), and 8.24 mm−1 (PSBP). These values reflect an inverse relationship between opacity and optical clarity, consistent with previously reported definitions [13,58].
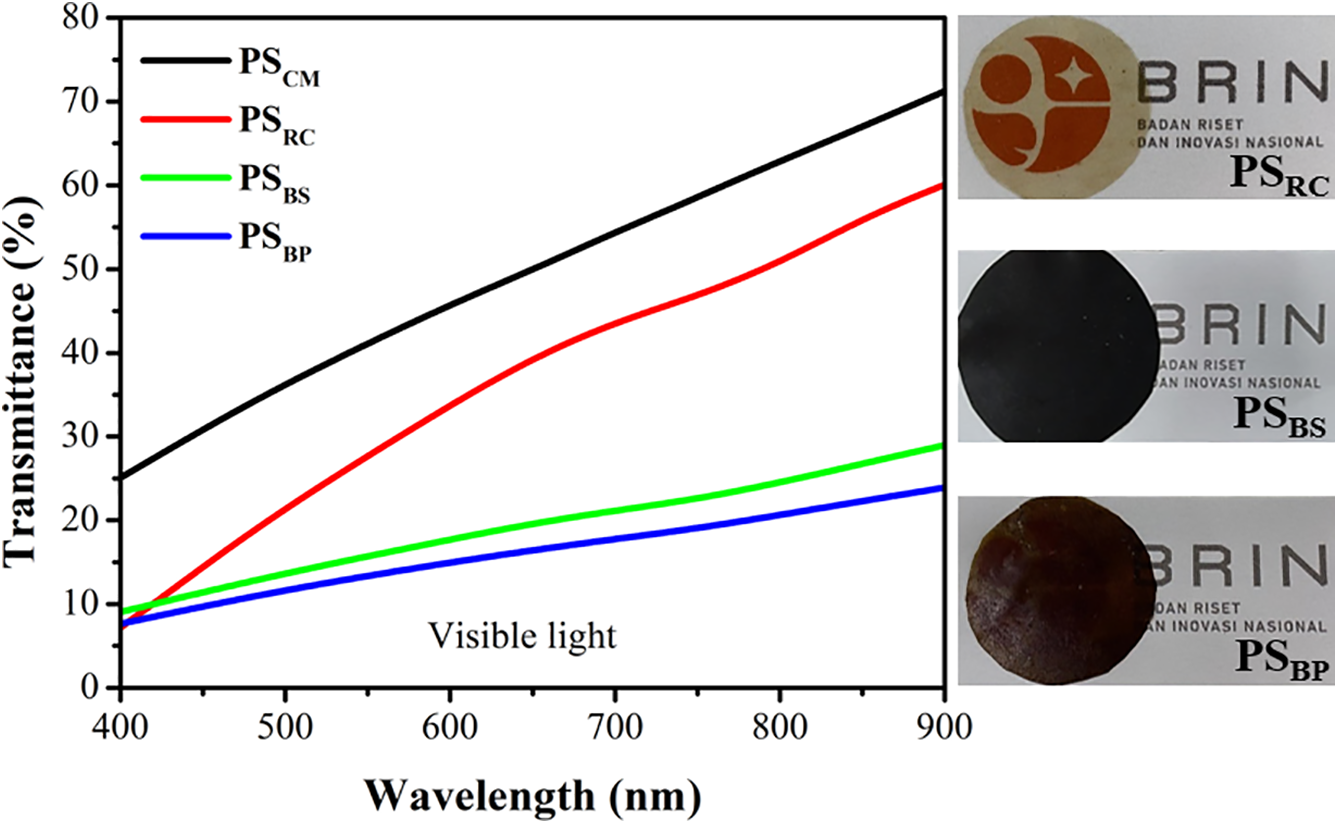
Figure 9: Transmittance spectra of starch-based bioplastic films and their corresponding visual appearance
Among the samples, the bioplastic derived from commercial food-grade cassava starch (PSCM) exhibited the highest visible light transmittance across the visible spectrum (400–900 nm), followed closely by PSRC, while PSBS and PSBP demonstrated significantly lower transmittance and higher opacity values. This is primarily due to the fact that food-grade sweet cassava generally contains fewer impurities compared to non-food-grade cassava sources, resulting in higher optical clarity in the PSCM film [59].
Interestingly, the SRC-based film (PSRC) displayed optical properties that were comparable to PSCM, exhibiting moderate opacity (4.40 mm−1) and a visibly translucent appearance despite being derived from non-commercial starch. This favourable behaviour is attributed to several factors. First, the native starch color of SRC was observed to be white and opaque, whereas SBS and SBP starches appeared light brown and medium brown, respectively (Table 1). The darker appearance of banana-derived starches is likely due to the presence of residual lignin, as evidenced by their higher lignin contents (Table 1), and the appearance of characteristic aromatic C=C vibrations around 1600 cm−1 in FTIR spectra (Fig. 1), which reflect the lignin chemical structure and may indicate the oxidation of phenolic compounds [34].
Second, SEM and EDX analyses confirmed that SRC retained lower levels of inorganic and lignocellulosic residues and displayed a homogeneous, spherical granule morphology. These traits promote uniform film formation and minimize internal structural heterogeneity, both of which are critical for reducing optical scattering. Furthermore, the high amylopectin content in SRC (71.83%) likely promotes an amorphous and continuous film network, facilitating more efficient internal light transmission and reducing scattering. In contrast, films with higher amylose content, such as PSBS and PSBP, tend to exhibit more heterogeneous surface and cross-sectional morphologies due to crystallization tendencies of linear amylose chains [58,60]. Heterogeneity enhances light scattering and diminishes transmittance. Additionally, crystalline regions as reflected by higher content of amylose observed in SBS and SBP are known to exhibit higher refractive indices than amorphous domains; as a result, more light is reflected and scattered at the air–polymer interface, further reducing film transparency [61]. Moreover, the higher opacity values of PSBS and PSBP are compounded by the thermal oxidation of retained phenolics during hot pressing. At elevated temperatures (120∘C), polyphenolic compounds and lignocellulosic residues can undergo thermal oxidation or participate in Maillard-type reactions, forming chromophoric products such as quinones or melanoidin-like compounds [62,63], which absorb visible light and cause film browning. EDX spectra further supported these findings by showing substantial levels of Ca, Mg, Cl, and Si in PSBS and PSBP films, indicative of residual mineral and lignocellulosic matter derived from the complex structure of banana pseudostem and peel. These thermally reactive components not only alter the visual properties of the films but also significantly impact their optical performance by increasing opacity and reducing transmittance (Fig. 9, right panel). As a result, PSBS and PSBP films appeared visibly darker and more opaque, confirming the role of botanical origin and extractive purity in governing film clarity.
The moderate opacity value of the PSRC film (4.40 mm−¹), close to that reported for corn starch-based bioplastics (3.04 mm−¹) by Yang et al. [13], highlights the feasibility of rubber cassava starch as a functional biopolymer matrix for semi-transparent packaging materials. However, further optimization may be needed for lignocellulose-derived starches to achieve comparable optical quality. In summary, the optical appearance and transmittance of starch-based bioplastics are governed by a complex interplay of factors, including starch botanical origin, residual phenolic and inorganic content, the amylose-to-amylopectin ratio, and the thermal chemistry of the processing method.
This study comprehensively evaluated starch-based bioplastics derived from underutilized agro-industrial residues, namely, banana stem (PSBS), banana peel (PSBP), and rubber cassava (PSRC), in comparison with bioplastics fabricated from commercially available cassava starch (PSCM). The findings highlight the significant influence of starch botanical origin and associated non-starch components on the final bioplastic performance.
Among the tested sources, PSRC emerged as the most promising candidate. It exhibited a well-balanced mechanical profile with the highest elongation at break (23.57%) and adequate tensile strength (9.75 MPa), while also achieving the greatest transparency (60.2%), highest water solubility (34.92%), and lowest water contact angle (41.58°). These properties indicate a more hydrophilic and amorphous structure, as confirmed by XRD and FTIR analyses, which is advantageous for packaging applications requiring flexibility, clarity, and biodegradability.
By contrast, PSBS and PSBP showed higher crystallinity and strength but limited ductility and optical quality, making them less suitable for flexible packaging applications. Overall, the findings highlight rubber cassava starch (SRC) as a sustainable, non-food, and economically viable feedstock for bioplastic production, offering competitive performance compared to food-grade starch. This work provides valuable insights for tailoring starch-based bioplastics through source selection and processing strategies, thereby advancing the development of flexible, eco-friendly packaging materials.
Acknowledgement: Fitra Yurid expresses sincere gratitude for the scholarship received through the Degree by Research program—BRIN. The authors would like to thank Asri Maelani for her technical assistance in starch and bioplastic preparation, as well as the E-Layanan Sains (ELSA)—BRIN for analytical support. The authors also wish to express their appreciation to Nasim Amiralian under collaboration BRIN-KONEKSI Project for providing valuable feedback and insightful discussions.
Funding Statement: This research was financially supported by the RIIM BRIN and LPDP Grants, grant number B-2880/II.7.5/KS.00/4/2025 dan B-7930/III.6/TK.01.03/4/2025 under the scheme BRIN-KONEKSI Joint Call for Proposals with the theme “Indonesia‘s Bioeconomy: Maximising Sustainable Marine Biodiversity Utilisation 2024” No 6/II.7/HK/2025.
Author Contributions: Fitra Yurid: writing—original draft; data curation; formal analysis; investigation; methodology; visualization. Nadiatus Silmi; writing—original draft; data curation; investigation; validation. Heni Rachmawati: writing—review & editing; supervision; validation. Nanda Nagara: supervision; validation. Riyanti Ekafitri: resources; validation. Athanasia Amanda Septevani: writing—review & editing; conceptualization; funding acquisition; methodology; project administration; resources; supervision; validation. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Data can be made available upon request.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
Nomenclature
| SCM | Commercial Starch |
| SRC | Rubber Cassava Starch |
| SBS | Banana Pseudostem Starch |
| SBP | Banana Peel Starch |
| PSCM | Commercial Starch-based Bioplastic |
| PSRC | Rubber Cassava Starch-based Bioplastic |
| PSBS | Banana Pseudostem Starch-based Bioplastic |
| PSBP | Banana Peel Starch-based Bioplastic |
References
1. Rahayoe S, Novianto TD, Sedayu BB. Enhancing the properties of biodegradable food packaging films derived from Agar and Porang-Glucomannan (Amorphophallus oncophyllus) blends. J Renew Mater. 2025;13(2):385–400. doi:10.32604/jrm.2024.057313. [Google Scholar] [CrossRef]
2. Kowser MA, Mahmud H, Chowdhury MA, Hossain N, Mim JJ, Islam S. Fabrication and characterization of corn starch based bioplastic for packaging applications. Results Mater. 2025;25(10):100662. doi:10.1016/j.rinma.2025.100662. [Google Scholar] [CrossRef]
3. Fredi G, Dorigato A. Recycling of bioplastic waste: a review. Adv Ind Eng Polym Res. 2021;4(3):159–77. doi:10.1016/j.aiepr.2021.06.006. [Google Scholar] [CrossRef]
4. Atiwesh G, Mikhael A, Parrish CC, Banoub J, Le TAT. Environmental impact of bioplastic use: a review. Heliyon. 2021;7(9):e07918. doi:10.1016/j.heliyon.2021.e07918. [Google Scholar] [PubMed] [CrossRef]
5. Gurunathan MK, Navasingh RJH, Selvam JDR, Čep R. Development and characterization of starch bioplastics as a sustainable alternative for packaging. Sci Rep. 2025;15(1):15264. doi:10.1038/s41598-025-00221-0. [Google Scholar] [PubMed] [CrossRef]
6. Arifin HR, Angelica YC, Nurhadi B, Marta H, Nissa RC. Elephant foot yam starch-NCC bionanocomposite film incorporated with virgin coconut oil and monoglyceride for hydrophobic and biodegradable packaging. J Renew Mater. 2025;13(3):617–35. doi:10.32604/jrm.2025.057812. [Google Scholar] [CrossRef]
7. Geyer R, Jambeck JR, Law KL. Production, use, and fate of all plastics ever made. Sci Adv. 2017;3(7):e1700782. doi:10.1126/sciadv.1700782. [Google Scholar] [PubMed] [CrossRef]
8. Thakkar A, Patel B, Sahu SK, Yadav VK, Patel R, Sahoo DK, et al. Potato starch bioplastic films reinforced with organic and inorganic fillers: a sustainable packaging alternative. Int J Biol Macromol. 2025;306:141630. doi:10.1016/j.ijbiomac.2025.141630. [Google Scholar] [PubMed] [CrossRef]
9. Dmitrenko M, Pasquini D, Bernardo MP, de Lima Alves JM, Kuzminova A, Dzhakashov I, et al. Bio-composite films from carrageenan/starch reinforced with nanocellulose for active edible food packaging: development and optimization. J Renew Mater. 2025;13(6):1139–68. doi:10.32604/jrm.2025.02024-0023. [Google Scholar] [CrossRef]
10. de Azêvedo LC, Rovani S, Santos JJ, Dias DB, Nascimento SS, Oliveira FF, et al. Study of renewable silica powder influence in the preparation of bioplastics from corn and potato starch. J Polym Environ. 2021;29(3):707–20. doi:10.1007/s10924-020-01911-8. [Google Scholar] [CrossRef]
11. Gesase LE, King’ondu CK, Jande YAC. Manihot glaziovii-bonded and bioethanol-infused charcoal dust briquettes: a new route of addressing sustainability, ignition, and food security issues in briquette production. Bioenergy Res. 2020;13(1):378–86. doi:10.1007/s12155-019-10076-9. [Google Scholar] [CrossRef]
12. Hawashi M, Surya Ningsih T, Bias Tri Cahyani S, Tri Widjaja K, Gunawan S. Optimization of the fermentation time and bacteria cell concentration in the starter culture for cyanide acid removal from wild cassava (Manihot glaziovii). MATEC Web Conf. 2018;156:01004. doi:10.1051/matecconf/201815601004. [Google Scholar] [CrossRef]
13. Yang J, Dong X, Wang J, Ching YC, Liu J, Chunhui L, et al. Synthesis and properties of bioplastics from corn starch and citric acid-epoxidized soybean oil oligomers. J Mater Res Technol. 2022;20(5):373–80. doi:10.1016/j.jmrt.2022.07.119. [Google Scholar] [CrossRef]
14. Samarasekara AMPB, Chamikara AWC, Wijesundara WWHP. Extraction and usage of starch from banana pseudostem to develop biodegradable polymer composites. In: Proceedings of the 18th International Forestry and Environment Symposium; 2013 Jan 10–11; Kalutara, Sri Lanka. 18 p. [Google Scholar]
15. Mohd Ali NA, Abdullah N, Jong SH, Muhammad N, Tan MC. Effect of different banana pseudostem parts on their starch yield, morphology and thermal properties. IOP Conf Ser Earth Environ Sci. 2021;736(1):012038. doi:10.1088/1755-1315/736/1/012038. [Google Scholar] [CrossRef]
16. Francis A, Shukla RN, Munjaji KS, Kumar Singh A. Isolation, modification and utilization of starch from green banana peel as a fruit waste. Asian J Microbiol Biotechnol Environ Sci. 2023;25(2):355–9. doi:10.53550/AJMBES.2023.v25i02.028. [Google Scholar] [CrossRef]
17. Ishara GKK, Koliyabandara PA, Samarakoon G. Eco-friendly bio-composite sheets: a study on the utilization of banana peels, cassava starch, and banana stem fibers. Front Sustain. 2024;5:1410986. doi:10.3389/frsus.2024.1410986. [Google Scholar] [CrossRef]
18. Arfiathi A, Sumirat R, Syamani FA, Lubis MAR, Filianty F, Nurhamiyah Y. Effect of citric acid on the properties of thermoplastic bitter cassava starch plasticized with isosorbide. Polym from Renew Resour. 2024;15(1):43–59. doi:10.1177/20412479231202591. [Google Scholar] [CrossRef]
19. Abera WG, Kasirajan R, Majamo SL. Synthesis and characterization of bioplastic film from banana (Musa Cavendish species) peel starch blending with banana pseudo-stem cellulosic fiber. Biomass Convers Biorefinery. 2024;14(17):20419–40. doi:10.1007/s13399-023-04207-8. [Google Scholar] [CrossRef]
20. Oladebeye AO, Oladebeye AA, Arawande JO. Physicochemical properties of wild yam (Dioscorea villosa) starch. Hashemi Gahruie H, editor. Int J Food Sci. 2023;2023:1–9. doi:10.1155/2023/8868218. [Google Scholar] [PubMed] [CrossRef]
21. Subroto E. Review on the analysis methods of starch, amylose, amylopectinin food and agricultural products. Int J Emerg Trends Eng Res. 2020;8(7):3519–24. doi:10.30534/ijeter/2020/103872020. [Google Scholar] [CrossRef]
22. Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton DC. Determination of structural carbohydrates and lignin in biomass. Golden, CO, USA: National Renewable Energy Laboratory; 2008. Technical Report. NREL/TP-510-42618. [Google Scholar]
23. Abdullah AHD, Putri OD, Sugandi WW. Effects of starch-glycerol concentration ratio on mechanical and thermal properties of cassava starch-based bioplastics. Indones J Mater Sci. 2019;20(4):162–7. doi:10.17146/jsmi.2019.20.4.5505. [Google Scholar] [CrossRef]
24. Oblitas R, Quispe-Sanchez L, Guadalupe G, Diaz EH, Oliva S, Diaz-Valderrama JR, et al. Physicochemical properties of bioactive bioplastics based on cellulose from coffee and cocoa by-products. Results Chem. 2025;15:102201. doi:10.1016/j.rechem.2025.102201. [Google Scholar] [CrossRef]
25. Dorantes-Fuertes MG, López-Méndez MC, Martínez-Castellanos G, Meléndez-Armenta RÁ, Jiménez-Martínez HE. Starch extraction methods in tubers and roots: a systematic review. Agronomy. 2024;14(4):865. doi:10.3390/agronomy14040865. [Google Scholar] [CrossRef]
26. Zhu F. Composition, structure, physicochemical properties, and modifications of cassava starch. Carbohydr Polym. 2015;122:456–80. doi:10.1016/j.carbpol.2014.10.063. [Google Scholar] [PubMed] [CrossRef]
27. Pablo J, Niño C, Mina H. Potential uses of musaceae wastes: case of application in the development of bio-based composites. Polymers. 2021;13(11):1–34. doi:10.3390/polym13111844. [Google Scholar] [PubMed] [CrossRef]
28. Kumar V, Chakraborty P, Janghu P, Umesh M, Sarojini S, Pasrija R, et al. Potential of banana based cellulose materials for advanced applications: a review on properties and technical challenges. Carbohydr Polym Technol Appl. 2023;6(5):100366. doi:10.1016/j.carpta.2023.100366. [Google Scholar] [CrossRef]
29. Weligama Thuppahige VT, Moghaddam L, Welsh ZG, Wang T, Xiao HW, Karim A. Extraction and characterisation of starch from cassava (Manihot esculenta) agro-industrial wastes. LWT. 2023;182(4):114787. doi:10.1016/j.lwt.2023.114787. [Google Scholar] [CrossRef]
30. Chandrasekar CM, Krishnamachari H, Farris S, Romano D. Development and characterization of starch-based bioactive thermoplastic packaging films derived from banana peels. Carbohydr Polym Technol Appl. 2023;5(21):100328. doi:10.1016/j.carpta.2023.100328. [Google Scholar] [CrossRef]
31. Benesi IR, Labuschagne MT, Dixon AG, Mahungu NM. Stability of native starch quality parameters, starch extraction and root dry matter of cassava genotypes in different environments. J Sci Food Agric. 2004;84(11):1381–8. doi:10.1002/jsfa.1734. [Google Scholar] [CrossRef]
32. Olad A, Doustdar F, Gharekhani H. Fabrication and characterization of a starch-based superabsorbent hydrogel composite reinforced with cellulose nanocrystals from potato peel waste. Colloids Surf A Physicochem Eng Asp. 2020;601(5):124962. doi:10.1016/j.colsurfa.2020.124962. [Google Scholar] [CrossRef]
33. Zou W, Yu L, Liu X, Chen L, Zhang X, Qiao D, et al. Effects of amylose/amylopectin ratio on starch-based superabsorbent polymers. Carbohydr Polym. 2012;87(2):1583–8. doi:10.1016/j.carbpol.2011.09.060. [Google Scholar] [CrossRef]
34. Silva RD, Pacheco TF, de Santi AD, Manarelli F, Bozzo BR, Brienzo M, et al. From bulk banana peels to active materials: slipping into bioplastic films with high UV-blocking and antioxidant properties. J Clean Prod. 2024;438:140709. doi:10.1016/j.jclepro.2024.140709. [Google Scholar] [CrossRef]
35. Zhang B, Li X, Liu J, Xie F, Chen L. Supramolecular structure of A- and B-type granules of wheat starch. Food Hydrocoll. 2013;31(1):68–73. doi:10.1016/j.foodhyd.2012.10.006. [Google Scholar] [CrossRef]
36. Lu H, Ma R, Chang R, Tian Y. Evaluation of starch retrogradation by infrared spectroscopy. Food Hydrocoll. 2021;120(1):106975. doi:10.1016/j.foodhyd.2021.106975. [Google Scholar] [CrossRef]
37. Pozo C, Rodríguez-Llamazares S, Bouza R, Barral L, Castaño J, Müller N, et al. Study of the structural order of native starch granules using combined FTIR and XRD analysis. J Polym Res. 2018;25(12):266. doi:10.1007/s10965-018-1651-y. [Google Scholar] [CrossRef]
38. Marta H, Cahyana Y, Djali M, Pramafisi G. The properties, modification, and application of banana starch. Polymers. 2022;14(15):3092. doi:10.3390/polym14153092. [Google Scholar] [PubMed] [CrossRef]
39. Todica M, Nagy EM, Niculaescu C, Stan O, Cioica N, Pop CV. XRD investigation of some thermal degraded starch based materials. J Spectrosc. 2016;2016(1):1–6. doi:10.1155/2016/9605312. [Google Scholar] [CrossRef]
40. Stelescu MD, Oprea OC, Sonmez M, Ficai A, Motelica L, Ficai D, et al. Structural and thermal characterization of some thermoplastic starch mixtures. Polysaccharides. 2024;5(4):504–22. doi:10.3390/polysaccharides5040032. [Google Scholar] [CrossRef]
41. Chakraborty I, Pooja N, Banik S, Govindaraju I, Das K, Mal SS, et al. Synthesis and detailed characterization of sustainable starch-based bioplastic. J Appl Polym Sci. 2022;139(39):e52924. doi:10.1002/app.52924. [Google Scholar] [CrossRef]
42. Yang J, Ching YC, Chuah CH, Hai ND, Singh R, Nor ARM. Preparation and characterization of starch-based bioplastic composites with treated oil palm empty fruit bunch fibers and citric acid. Cellulose. 2021;28(7):4191–210. doi:10.1007/s10570-021-03816-8. [Google Scholar] [CrossRef]
43. Li X, Wei Y, Xu J, Xu N, He Y. Quantitative visualization of lignocellulose components in transverse sections of moso bamboo based on FTIR macro- and micro-spectroscopy coupled with chemometrics. Biotechnol Biofuels. 2018;11(1):263. doi:10.1186/s13068-018-1251-4. [Google Scholar] [PubMed] [CrossRef]
44. Liu Y, Hu T, Wu Z, Zeng G, Huang D, Shen Y, et al. Study on biodegradation process of lignin by FTIR and DSC. Environ Sci Pollut Res. 2014;21(24):14004–13. doi:10.1007/s11356-014-3342-5. [Google Scholar] [PubMed] [CrossRef]
45. Maceda A, Soto-Hernández M, Peña-Valdivia CB, Trejo C, Terrazas T. Characterization of lignocellulose of Opuntia (Cactaceae) species using FTIR spectroscopy: possible candidates for renewable raw material. Biomass Convers Biorefin. 2022;12(11):5165–74. doi:10.1007/s13399-020-00948-y. [Google Scholar] [CrossRef]
46. Figueroa-Lopez KJ, Villabona-Ortíz Á, Ortega-Toro R. Sustainable starch-based films from cereals and tubers: a comparative study on cherry tomato preservation. Polymers. 2024;16(20):2913. doi:10.3390/polym16202913. [Google Scholar] [PubMed] [CrossRef]
47. Singh DAK, Sharma P, Shrivastav M, Chaudhary R, Chauhan P, Sanskriti, et al. Production of biodegradable plastic film from potato and sago starch. J Microbiol Biotechnol Food Sci. 2025;14(4):e11143. doi:10.55251/jmbfs.11143. [Google Scholar] [CrossRef]
48. Wrona M, Cran MJ, Nerín C, Bigger SW. Development and characterisation of HPMC films containing PLA nanoparticles loaded with green tea extract for food packaging applications. Carbohydr Polym. 2017;156(2):108–17. doi:10.1016/j.carbpol.2016.08.094. [Google Scholar] [PubMed] [CrossRef]
49. Mukaila T, Adeniyi A, Bello I, Sarker NC, Monono E, Hammed A. Optimizing film mechanical and water contact angle properties via PLA/starch/lecithin concentrations. Clean Circ Bioeconomy. 2024;8:100095. doi:10.1016/j.clcb.2024.100095. [Google Scholar] [CrossRef]
50. Lai YR, Ho TL, Yang YH, Lin TH, Wang SSS. Development of sustainable bioplastic films for food packaging using zein protein-derived amyloid fibrils: characterization and functional properties. Int J Biol Macromol. 2025;318(26):144817. doi:10.1016/j.ijbiomac.2025.144817. [Google Scholar] [PubMed] [CrossRef]
51. Ahsan WA, Hussain A, Lin C, Nguyen MK. Biodegradation of different types of bioplastics through composting—a recent trend in green recycling. Catalysts. 2023;13(2):294. doi:10.3390/catal13020294. [Google Scholar] [CrossRef]
52. Ghasemlou M, Daver F, Murdoch BJ, Ball AS, Ivanova EP, Adhikari B. Biodegradation of novel bioplastics made of starch, polyhydroxyurethanes and cellulose nanocrystals in soil environment. Sci Total Environ. 2022;815:152684. doi:10.1016/j.scitotenv.2021.152684. [Google Scholar] [PubMed] [CrossRef]
53. Accinelli C, Saccà ML, Mencarelli M, Vicari A. Deterioration of bioplastic carrier bags in the environment and assessment of a new recycling alternative. Chemosphere. 2012;89(2):136–43. doi:10.1016/j.chemosphere.2012.05.028. [Google Scholar] [PubMed] [CrossRef]
54. Tosin M, Weber M, Siotto M, Lott C, Degli Innocenti F. Laboratory test methods to determine the degradation of plastics in marine environmental conditions. Front Microbiol. 2012;3:18–32. doi:10.3389/fmicb.2012.00225. [Google Scholar] [PubMed] [CrossRef]
55. Pooja N, Ahmed NY, Mal SS, Bharath PAS, Zhuo GY, Noothalapati H, et al. Assessment of biocompatibility for citric acid crosslinked starch elastomeric films in cell culture applications. Sci Rep. 2025;15(1):6427. doi:10.1038/s41598-025-90933-0. [Google Scholar] [PubMed] [CrossRef]
56. Sujuthi M, Liew. Properties of bioplastic sheets made from different types of starch incorporated with recycled newspaper pulp. Trans Sci Technol. 2016;3(2):257–64. [Google Scholar]
57. Motelica L, Ficai D, Ficai A, Oprea OC, Kaya DA, Andronescu E. Biodegradable antimicrobial food packaging: trends and perspectives. Foods. 2020;9(10):1438. doi:10.3390/foods9101438. [Google Scholar] [PubMed] [CrossRef]
58. Song HG, Choi I, Lee JS, Chung MN, Yoon CS, Han J. Comparative study on physicochemical properties of starch films prepared from five sweet potato (Ipomoea batatas) cultivars. Int J Biol Macromol. 2021;189(10):758–67. doi:10.1016/j.ijbiomac.2021.08.106. [Google Scholar] [PubMed] [CrossRef]
59. Montagnac JA, Davis CR, Tanumihardjo SA. Processing techniques to reduce toxicity and antinutrients of cassava for use as a staple food. Compr Rev Food Sci Food Saf. 2009;8(1):17–27. doi:10.1111/j.1541-4337.2008.00064.x. [Google Scholar] [CrossRef]
60. Jiménez A, Fabra MJ, Talens P, Chiralt A. Effect of re-crystallization on tensile, optical and water vapour barrier properties of corn starch films containing fatty acids. Food Hydrocoll. 2012;26(1):302–10. doi:10.1016/j.foodhyd.2011.06.009. [Google Scholar] [CrossRef]
61. Lin Y, Bilotti E, Bastiaansen CWM, Peijs T. Transparent semi-crystalline polymeric materials and their nanocomposites: a review. Polym Eng Sci. 2020;60(10):2351–76. doi:10.1002/pen.25489. [Google Scholar] [CrossRef]
62. Chen Y, Tshabalala MA, Gao J, Stark NM, Fan Y. Color and surface chemistry changes of extracted wood flour after heating at 120°C. Wood Sci Technol. 2014;48(1):137–50. doi:10.1007/s00226-013-0582-3. [Google Scholar] [CrossRef]
63. Yao GY, Chen XP, Long ZY, Du XB, Liang JZ, Wei XJ, et al. The non-enzymatic browning of pine bark during thermal treatment: color and chemical changes, color kinetics and insights into mechanisms. Ind Crops Prod. 2023;204(3):117289. doi:10.1016/j.indcrop.2023.117289. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools