 Open Access
Open Access
REVIEW
Bacterial Biorefineries: Transforming Agro-Industrial Waste into Sustainable Solutions for a Circular Bioeconomy
1 Institute of Climate Adaptation and Marine Biotechnology (ICAMB), Universiti Malaysia Terengganu, Kuala Nerus, 21030, Terengganu, Malaysia
2 Ocular Infections and Antimicrobials Research Group, Singapore Eye Research Institute, The Academia, 20 College Road, Discovery Tower, Singapore, 169856, Singapore
3 Faculty of Science and Marine Environment, Universiti Malaysia Terengganu, Kuala Nerus, 21030, Terengganu, Malaysia
4 BioSES Research Interest Group, Faculty of Science and Marine Environment, Universiti Malaysia Terengganu, Kuala Nerus, 21030, Terengganu, Malaysia
5 Biomaterials and Product Development laboratory, Department of Biotechnology, Kalasalingam Academy of Research and Education, Anand Nagar, Krishnankoil, 626126, Tamil Nadu, India
6 School of Applied Science, Faculty of Integrated Life Sciences, Quest International University, Ipoh, 34020, Perak, Malaysia
* Corresponding Author: Sevakumaran Vigneswari. Email:
(This article belongs to the Special Issue: Valorization of Lignocellulosic Biomass for Functional Materials)
Journal of Renewable Materials 2026, 14(3), 6 https://doi.org/10.32604/jrm.2025.02025-0148
Received 27 July 2025; Accepted 09 October 2025; Issue published 25 March 2026
Abstract
The escalating accumulation of agro-industrial waste—exceeding 350 million tons annually from post-harvest residues, food processing, and aquaculture—poses serious environmental threats, including greenhouse gas emissions, groundwater contamination, and excessive landfill usage. Although conventional treatment methods such as composting, incineration, and recycling offer partial mitigation, they often fall short of delivering scalable, circular solutions. Microbial biorefineries have emerged as a transformative approach, enabling the conversion of diverse biomass streams into high-value renewable materials. Through microbial fermentation, agricultural and municipal waste can be repurposed into functional outputs such as nanocellulose, biochar, and biocompatible compounds with applications in packaging, pharmaceuticals, and cosmetics. This review critically examines the role of microbial systems in agro-waste valorization, highlighting recent advances in strain engineering, process integration, and bioproduct development. It also explores the regulatory and techno-economic frameworks required to scale these innovations within a biomass-driven circular economy, positioning microbial biorefineries as key enablers of sustainable material transitions.Graphic Abstract
Keywords
Agricultural waste refers to surplus materials generated during the cultivation, harvesting, and processing of crops, including fruits, vegetables, dairy, poultry, meat, and other agricultural products [1]. Priya et al. [2] noted that substantial biomass waste arises from post-harvest and post-processing operations. The rapidly growing global population has transformed the food and agro-industrial sectors, significantly boosting productivity and, as a result, producing vast quantities of agro-industrial waste [3]. Currently, this waste is estimated at 1.03 billion tons annually, with most being burned or dumped in landfills [4]. Consequently, greenhouse gas (GHG) emissions have surged and are projected to increase by 60% by 2030. Blasi et al. [5] reported that such disposal methods release toxic compounds, contributing to pollution, health hazards, and climate change.
Agricultural processing generates a wide range of solid waste, including rice husks, wheat straw, leaves, stems, jute fiber residues, rusk, livestock waste, post-harvest leftovers, fruit and vegetable scraps, peels, skins, shells, edible oil waste, cooking oil, vegetable oil residues, poultry byproducts, feathers, egg discards, meat scraps, and forestry debris [6]. Agro-industrial waste, primarily composed of lignocellulosic biomass, has attracted growing interest in material and chemical engineering due to its mechanical strength, thermal stability, renewability, abundance, non-toxicity, affordability, and biodegradability [6]. These solid wastes can be transformed into valuable products through microbial biorefineries [7]. Microbial biorefineries offer sustainable methods for converting agricultural waste into marketable products. They focus on utilizing agro-industrial byproducts as feedstock to produce biofuels, bioenergy, biopolymers, bioactive compounds, and other high-value materials, promoting environmental sustainability [8]. This approach supports the development of bio-based industries by reducing pollution and conserving natural resources. The linear economy has contributed to excessive agro-industrial pollution [9,10], while the circular economy offers a promising solution for converting waste into useful products.
However, several challenges and future directions must be addressed to ensure successful implementation and advancement of this technology [1]. These include overcoming technical barriers and promoting environmental protection, resource efficiency, food security, and modern agricultural practices [10,11].
This review explores the sustainable use of agro-industrial waste through bacteria-driven biorefineries and evaluates current industrial efforts to produce valuable products via microbial conversion. It provides a comprehensive overview of the complexity of agro-industrial waste and the pretreatment methods needed to enhance microbial access. By identifying challenges and research gaps, the review emphasizes the need for greater visibility and integration of circular bioeconomy principles. It introduces a novel framework that combines microbial pathways, feedstock diversity, and industrial scalability—an approach often overlooked in previous studies. Additionally, the review addresses key questions such as how bacterial systems can be optimized for various waste streams, which pretreatment strategies best balance efficiency and sustainability, and how microbial biorefineries can be integrated into existing industrial systems. It also examines the socio-economic and policy factors influencing adoption, offering a strategic roadmap for advancing microbial biorefineries as scalable, low-carbon solutions for waste valorization.
2 Various Sources of Agro-Industrial Waste
Agriculture plays a vital role in contributing to the global economy across various resource sectors [12]. According to Bala et al. [13], unprocessed agro-industrial waste generates significant greenhouse gas emissions, which in turn intensify climate change through multiple pathways. Additionally, this results in the release of unwanted gaseous byproducts. Agricultural waste primarily originates from farming activities; however, it is not confined to production alone, as food processing also contributes to waste generation. Biorefineries can function as multipurpose facilities, particularly in waste management, by converting biomass waste into valuable products such as bioplastics, biofuels, organic acids, and chemicals [14,15]. The main sources of biomass waste include agricultural residues, food industry byproducts, and municipal waste streams [16]. Agricultural waste can be further categorized into lignocellulosic materials and green biomass [16], while food industry waste may consist of filtered grains and fibers, molasses, and shells. Fig. 1 below presents a broad classification of agricultural waste based on its various sources.
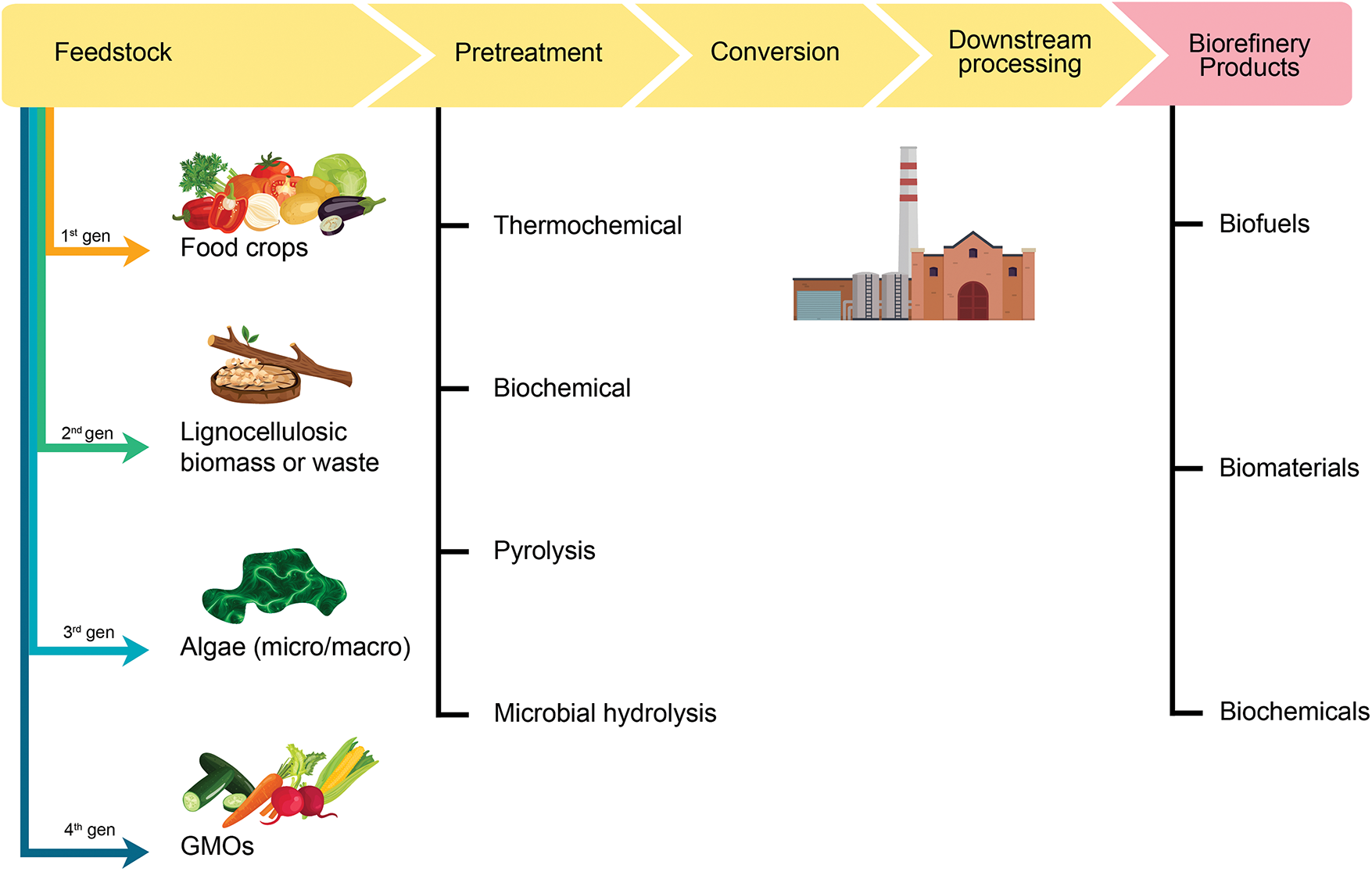
Figure 1: General schematic of different types of pre-treatment process of biorefineries of agricultural waste (Created using Adobe Illustrator)
Post-harvest waste refers to losses occurring throughout the entire food supply chain, beginning from the moment crops are harvested until their final consumption [17]. These losses can be categorized into weight reduction due to rotting, quality deterioration, nutritional depletion, reduced seed viability, and commercial loss. Waste generated from the agricultural industry contributes to GHG emissions and has several adverse environmental impacts. Like other production enterprises, the aquaculture industry requires various inputs to produce goods and materials [18]. Kumar and colleagues [18] noted that such wastes often hold little or no economic value and frequently act as environmental pollutants. Additionally, chemical usage in farming can be harmful to ecosystems. Hazardous substances such as pesticides, herbicides, and insecticides applied to crops may leach into groundwater and soil. Unused and leftover chemicals are considered toxic waste, posing significant risks to environmental health.
3 Common Practices in Agro-Industrial Waste Management
Identifying effective strategies for managing agricultural residues has become a critical priority in the agricultural sector, emerging as one of the most urgent challenges of the 21st century [19]. Conventional methods such as composting, recycling, landfilling, and incineration offer moderate reductions in carbon emissions. For instance, composting emits approximately from 30–400 kg CO2 equivalent per metric ton of biowaste (Fig. 2), whereas microbial biorefineries processing agro-waste can emit as little as 15–35 g CO2 equivalent per ton, depending on process design and post-treatment [20]. Biorefineries offer a more transformative solution by converting agro-waste into high-value products such as biofuels, bioplastics, biosurfactants, and pharmaceutical precursors. Life-cycle assessments indicate that optimized biorefinery systems can reduce emissions by up to 80%–90% compared to fossil-based production, with some advanced configurations achieving net-negative carbon balances through carbon capture and biochar generation. This not only enhances carbon savings by displacing fossil fuels but also improves resource efficiency and economic viability. Unlike conventional waste treatment, biorefineries enable cascading valorization, extracting maximum utility from biomass at each stage, in alignment with green chemistry and sustainability principles. While agro-waste management is locally scalable and accessible, biorefineries offer broader systemic impact, positioning them as strategic platforms for climate-smart innovation and sustainable industrial development (Fig. 2). Among waste management practices, landfilling remains the most widely used, with over 71% of municipal solid waste in the U.S. disposed of in landfill sites annually [21]. The United States Environmental Protection Agency categorizes landfills into five types: municipal solid waste landfills, bioreactor landfills, industrial waste landfills, hazardous waste landfills, and polychlorinated biphenyl landfills [22]. Landfills involve burying toxic waste in engineered pits, allowing slow decomposition. This method can be effective for farm or agro-industrial disposal if managed properly within a bioreactor landfill. However, a major concern is the degradation of soil and water quality during landfilling [23]. Potential hazards include the emission of gases and leachate. Moreover, poorly managed landfill sites can deteriorate into open dumps. Despite these risks, landfills can serve as a source of conventional energy if waste is carefully treated within the landfill environment [21].

Figure 2: Some of the common examples of agro-waste management globally and the carbon footprint (Created using Canva.com)
Composting is a green technology that transforms organic matter into a humidified and stable product through microbial activity. For effective composting, key parameters such as temperature, pH, moisture content, and the carbon-to-nitrogen ratio must be properly regulated [24]. Various strategies have been developed to compost organic waste. Chang et al. [25] utilized black soldier fly larvae to assist in composting, co-processing organic waste with agricultural residues to produce biofertilizers and larval biomass, which was subsequently used as animal feed. The larval biomass is nutritionally rich and suitable for feeding fish and poultry. Similarly, Peng et al. [26] demonstrated that co-composting kitchen waste with agricultural and forestry residues on an industrial scale results in an efficient composting process.
Vermicomposting is another method for processing organic waste. This biotechnology technique converts organic matter into compost through the combined action of red worms and bacteria [24]. Numerous studies have investigated composting and vermicomposting of food processing waste to ensure safe disposal and promote environmental sustainability. Recent reports suggest that vermicomposting is more effective than traditional composting in reducing greenhouse gas emissions and enhancing the quality of the final product [27]. It is also efficient in breaking down lignocellulose-rich materials, improving compost quality [28]. However, vermicomposting of municipal excess sludge has been associated with increased microplastic traces [27].
Traditional agro-industrial waste management practices such as landfilling, composting, and incineration present notable environmental and economic drawbacks compared to biorefinery-based solutions. Landfilling contributes significantly to methane emissions and groundwater contamination, with life cycle assessments indicating high global warming potential and ecotoxicity [29]. Incineration reduces waste volume but emits toxic gases and requires high capital and operational costs. Composting is more environmentally friendly but offers limited scalability and economic return, even with advanced systems, which still fall short of full valorization [30].
In contrast, biorefineries represent a circular economy model by converting waste into high-value products such as biofuels, bioplastics, and enzymes, thereby improving sustainability and reducing net emissions. Although biorefineries demand substantial initial investment, their long-term cost-effectiveness is supported by diversified revenue streams and reduced environmental liabilities. Bio-waste refineries offer dual benefits: they process waste from local communities and refine it into valuable bio-products. Usmani et al. [31] described biorefinery sustainability as a balanced coexistence between bioprocessing industries and the environment, meeting socioeconomic needs without compromising product quality. This includes utilizing bio-resources, especially bio-waste, to manufacture commodities economically. However, transitioning to this system may be gradual and vary across countries [32]. Sustainability assessments can aid in evaluating techno-economic feasibility and life cycle impacts [31]. To enhance biorefinery efficiency, Sukhla and Khan [33] discussed the use of microbial electrosynthesis (MES) to decarbonize carbon sources and support a green circular economy. Yet, challenges in stack design, anodic reaction efficiency, and reliable extraction methods can hinder full operational potential. Resolving these issues could significantly improve technology performance and feedstock conversion [34]. Additionally, De Buck et al. (2020) proposed refinery designs and supply chain models to improve sustainability and economic viability. Given the diverse and extensive composition of waste for these refineries, variability in feedstock size and type poses a major challenge. Therefore, a stable supply chain and logistics network are essential for successful biorefinery operations [35].
In another perspective, Regueira et al. [36] highlighted the restricted and inefficient use of microbes, suggesting flexible resource allocation models to address substrate utilization inefficiencies. Moreover, biowaste generated within biorefineries can be repurposed as a biosorbent for removing synthetic methylene blue dye from water, promoting sustainable practices in the biorefining industry [37].
Biorefinery is a widely adopted concept that enables the comprehensive and integrated utilization of biomass often generated from microbes into a diverse array of bio-based products and bioenergy sources [20]. From conventional biorefineries to more advanced systems employing various processing technologies, such as thermochemical and biochemical methods, the full potential of biorefineries remains to be fully explored and optimized [38]. Fig. 3 illustrates the transformation of agro-industrial waste into valuable products.
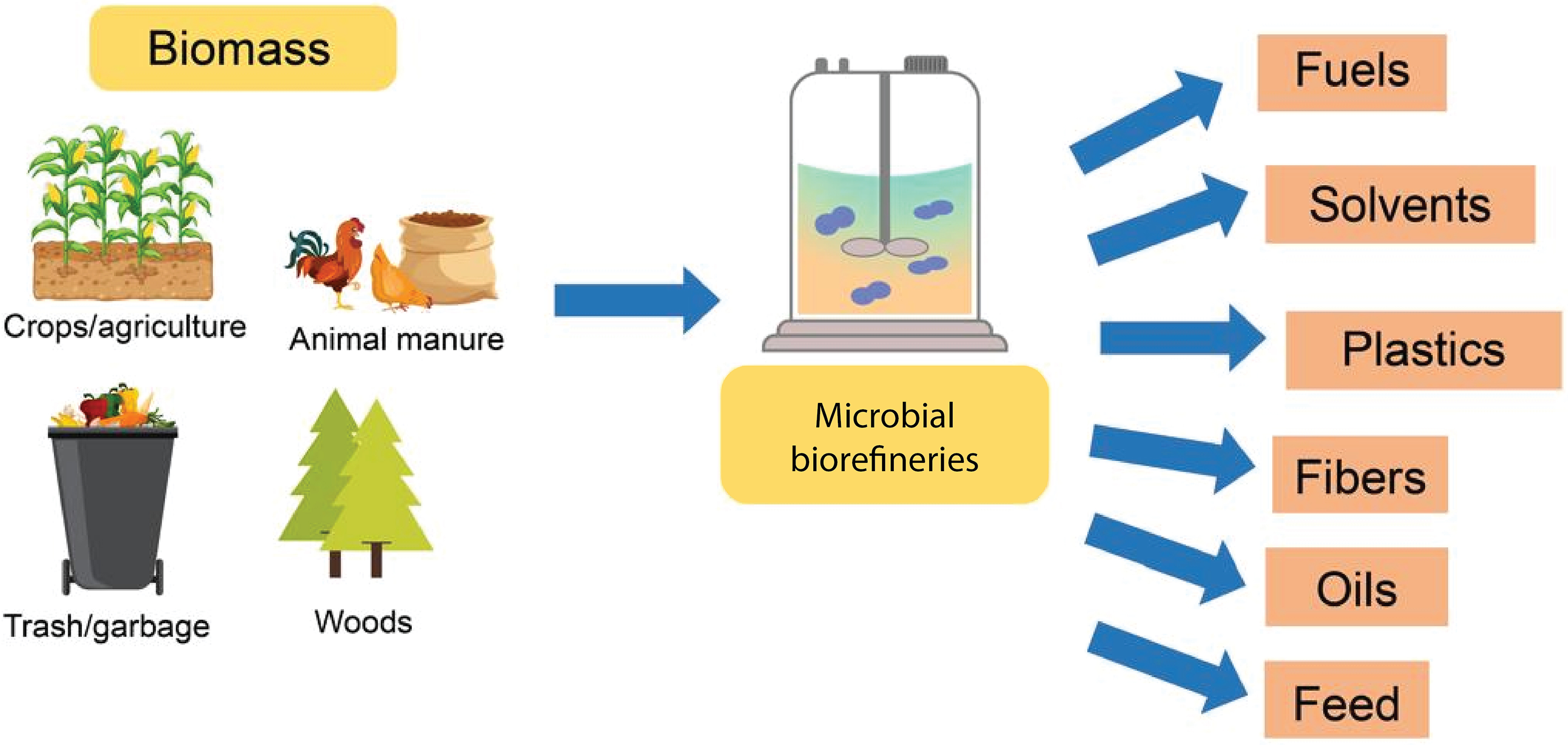
Figure 3: Sustainable processing of biomass into marketable products (Created using Adobe Illustrator)
Galbe and Wallberg [39] defined biorefineries as a sustainable counterpart to petroleum refineries, with the key distinction being the source of raw materials. Their study also emphasized that biomass can be refined into a wide spectrum of chemicals and energy products, contributing to the advancement of the circular economy. Among the most promising feedstocks is lignocellulosic biomass an abundant and renewable resource that has emerged as a viable alternative to fossil carbon for producing value-added chemicals. However, due to its complex structure, along with that of other agricultural wastes, lignocellulosic biomass is not easily broken down [40]. To address this challenge, biomass must undergo specific processing steps. Awogbemi and Von Kallon [40] noted that pretreatment methods are essential operations applied to biomass to overcome its initial resistance to direct conversion. These methods facilitate the breakdown of structural barriers, enabling more efficient transformation into desired bio-products.
Microbial communities [41,42] or specific enzymes such as esterases, lyases [43], and β-mannanases [44] are commonly employed to convert biowaste into value-added products (Fig. 4). Most biorefineries utilize thermophilic or extremotolerant microorganisms to enhance biomass conversion, particularly for lignocellulosic biomass. This type of biomass, derived from plant material, is notoriously difficult to break down due to its dense crystalline structure and strong glycosidic bonds [45]. The low yield from such substrates often results in commercially unviable processes. Therefore, pretreatment of biowaste is essential to facilitate enzymatic hydrolysis [35]. Bedoić et al. [16] identified wet biomass from garden waste—such as cuttings, grasses, and cover crops—as another excellent source of biowaste, which would otherwise be discarded or used as compost. Lignocellulosic biomass primarily consists of biopolymers like cellulose, hemicellulose, and lignin, with trace amounts of inorganic minerals and organic extractives [45]. Its composition varies depending on the source and origin [46]. The estimated annual production of lignocellulosic biomass is approximately 181.5 billion tons. Table 1 presents the classification of predominant bacterial types into two general categories.
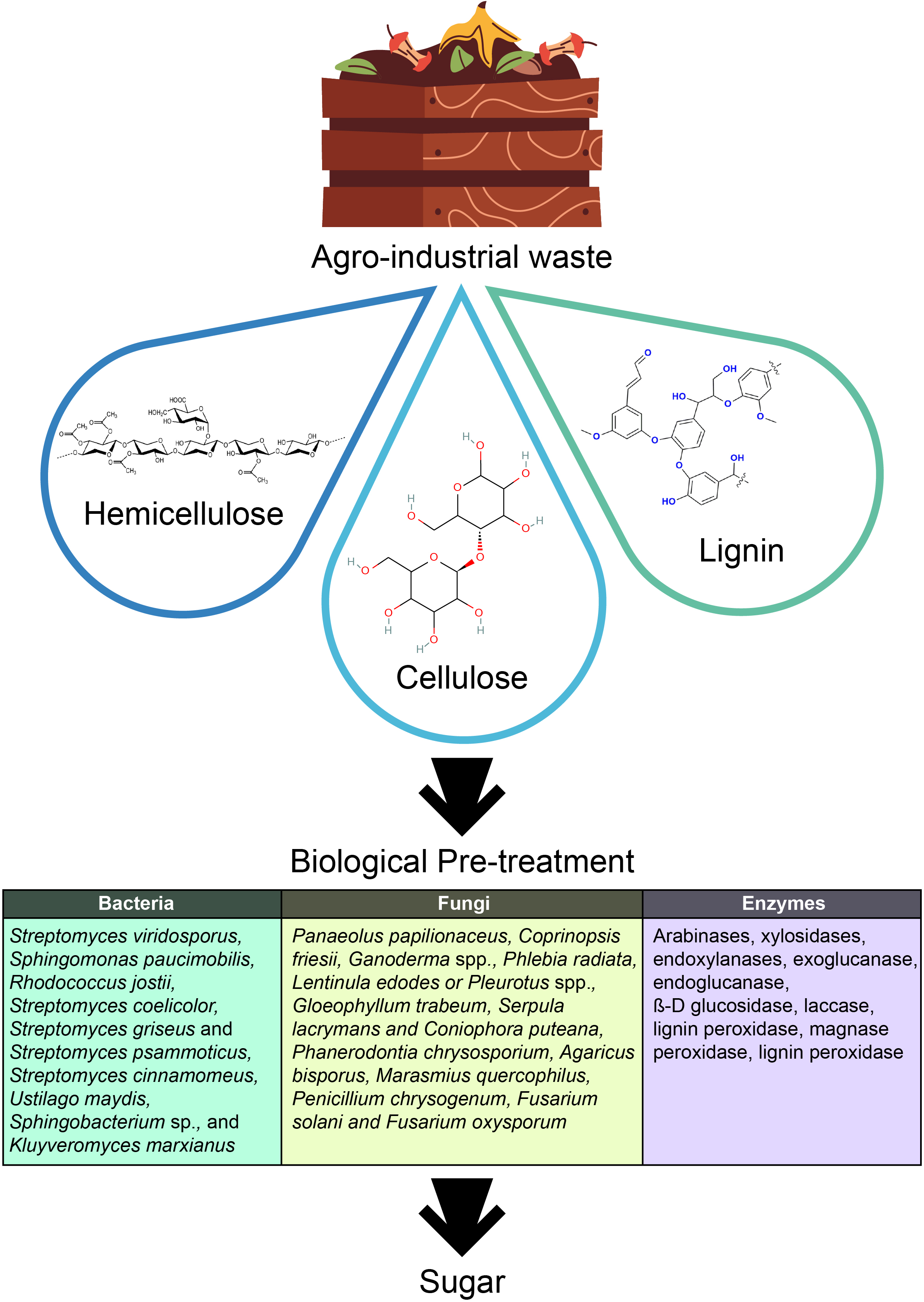
Figure 4: Biological pre-treatment of the agro-industrial waste requires the use of bacteria, fungi and enzymes to convert the agro-waste into simple sugars to be utilized for the bioproduction of valuable products (Created using Adobe Illustrator)

The dominant bacterial type significantly influences the characteristics of the treatment system by utilizing the organic components in the waste. These include species capable of metabolizing a wide range of organic materials and reproducing rapidly. Once the organic substrate is depleted, most bacteria die and lyse, releasing their biological components, which in turn support the growth of other microbial populations. Secondary predominance often arises from the overdesign of biological treatment systems for safety purposes. Beyond metabolic traits, the ability of bacteria to flocculate is a critical feature. For complete stabilization, all aerobic biological waste treatment systems rely on the separation of microorganisms from the liquid phase through flocculation [47].
Microorganisms have played a vital role in helping society address various environmental challenges. They have been successfully applied in the treatment of municipal and industrial waste, genetic engineering, and human and animal health. Microbial applications have enabled feasible and cost-effective solutions that would be difficult to achieve through chemical or physical engineering methods [47]. Moreover, microbial technologies have proven effective in tackling a wide range of environmental issues, particularly those related to waste management.
4.1 Bacterial Cellulose (BC) Biosynthesis
Bacterial cellulose (BC) is a highly pure, biodegradable biopolymer synthesized by aerobic bacteria. Unlike plant-derived cellulose, BC lacks lignin and hemicellulose, resulting in exceptional crystallinity, tensile strength, and biocompatibility qualities that make it ideal for biomedical, food, and textile applications [48,49]. Its nanofibrillar network provides high water retention, elasticity, and mechanical integrity, supporting cell adhesion and proliferation in tissue engineering contexts [49]. Despite these advantages, BC production faces economic and scalability challenges. A major limitation is the high cost of the fermentation medium, which accounts for 50%–65% of total production expenses [50]. Additionally, conventional static culture methods yield low quantities of BC, making industrial-scale manufacturing inefficient [50]. To overcome these constraints, researchers have emphasized the need for low-cost fermentation strategies that optimize media composition for maximum yield. Extensive efforts have focused on replacing expensive synthetic media with renewable nutrient sources, particularly agro-industrial waste streams such as dairy wastewater, winery residues, and textile effluents [51]. This approach not only reduces production costs but also promotes environmental sustainability by mitigating industrial waste disposal and pollution [52]. Recent advancements in fed-batch cultivation and dynamic bioreactor systems have further improved BC yield and quality, offering scalable solutions for biomedical and industrial applications [53]. These integrated strategies highlight the importance of techno-economic assessments and circular bioeconomy models to bridge the gap between laboratory innovation and commercial viability.
One accessible agro-industrial waste is banana peel, which has been used as a carbon source in BC biosynthesis [54]. A promising concentration of BC of 19.46 g/L was reported after 15 days of fermentation. Other agricultural wastes such as coconut water and fruit juices showed similar results when fermented by Acetobacter xylinum [54]. Additionally, whey protein contains various nutritional compounds that serve as a nitrogen source. Revin et al. [55] investigated the use of dairy and winery industry waste for cost-effective BC biosynthesis by Gluconacetobacter sp., successfully improving BC yield after just three days of fermentation. These findings suggest that whey products are a viable low-cost nitrogen source for BC production. Cotton textiles also hold potential as a sustainable resource for BC synthesis while addressing environmental concerns. A study by Kuo et al. [56] demonstrated that BC yield from discolored hydrolysate was higher than from colored hydrolysate. This may be due to the removal of colored reducing sugars via chitosan adsorption, which otherwise inhibits the metabolic activity of Gluconacetobacter sp. during BC biosynthesis.
Soemphol and colleagues [57] found that BC production improved when crude glycerol was used as an alternative substrate fermented by Gluconacetobacter sp. However, increasing glycerol concentrations beyond a certain threshold reduced BC biosynthesis, likely due to pollutants in crude glycerol affecting bacterial activity. Another study indicated that adding pineapple peel extract to crude glycerol enhanced BC biosynthesis. In addition to boosting BC production, utilizing these biofuel industry wastes helps reduce environmental impact and lowers reliance on non-renewable energy sources [57].
A recent study by Quiñones-Cerna and colleagues [58] employed Komagataeibacter rhaeticus, cultivated using asparagus waste. Additionally, agro-industrial wastes such as palm date, fig, and sugarcane molasses have been utilized for cost-effective BC production, either supplementing or replacing the widely used Hestrin-Schramm (HS) medium [59]. The researchers successfully synthesized highly crystalline BC (81.89%) with an ideal fiber size of 178 nm. Further investigations explored the use of various agro-waste substrates to assess BC yield. Residues from cucumber, tomato, apple, melon, and kiwi have been processed into hydrolysates for fermentation [60], while hydrolysates from mandarin fruit, lemon, grapefruit, and orange have also been used to produce BC [61]. Industrial waste products are increasingly recognized as promising candidates for BC production. However, their practical viability depends on the affordability of raw materials, which is influenced by factors such as geographic location and climate [51]. Consequently, agro-industrial by-products from agricultural export zones present a viable option for BC production substrates (Fig. 5). In microbial refineries focused on BC synthesis from lignocellulosic biomass, several bacterial taxa exhibit distinct metabolic pathways and substrate specificities that enhance their effectiveness. Komagataeibacter species, particularly K. xylinus and K. sucrofermentans, are well-known for producing high-purity BC via the pentose phosphate pathway and gluconeogenesis. These strains show strong specificity for glucose and xylose derived from fruit peels, sugarcane bagasse, and detoxified lignocellulosic hydrolysates [55,62]. In natural kombucha or yeast co-cultures, Komagataeibacter spp. work synergistically with saccharolytic partners to convert complex polysaccharides into fermentable sugars. This metabolic collaboration expands their substrate range to include starch-rich agro-residues such as cassava peels, rice bran, and sweet potato waste, which are first degraded by yeasts before being transformed into cellulose or acids by Komagataeibacter [63,64]. Gluconacetobacter spp., now largely reclassified under Komagataeibacter, share similar metabolic traits and can produce BC under acidic conditions using sugar-rich waste streams [65]. Acetobacter spp., traditionally associated with acetic acid fermentation, have also demonstrated BC synthesis when cultivated on ethanol or glucose-based lignocellulosic substrates, especially under optimized aeration and pH conditions [65]. Enterobacter spp., though not classical BC producers, have shown potential under tailored conditions due to their ability to metabolize both hexoses and pentoses via the Embden-Meyerhof-Parnas pathway. Their facultative anaerobic nature offers flexibility in fermentation setups [65]. Lactiplantibacillus plantarum, primarily known for lactic acid fermentation, has recently been explored for BC production through co-culturing or genetic modification. It utilizes hexoses from lignocellulosic hydrolysates and tolerates inhibitory compounds such as furfural [62]. Notably, Achromobacter spp. have emerged as promising BC producers, with strains capable of utilizing pectin-rich substrates like mango peel waste (MPW). Their metabolic versatility includes strong pectinase activity and efficient sugar uptake, resulting in enhanced BC yield and crystallinity compared to conventional media [65]. Collectively, these bacterial taxa represent a metabolically diverse and substrate-adapted toolkit for converting complex lignocellulosic feedstocks into high-value BC (Table 2).
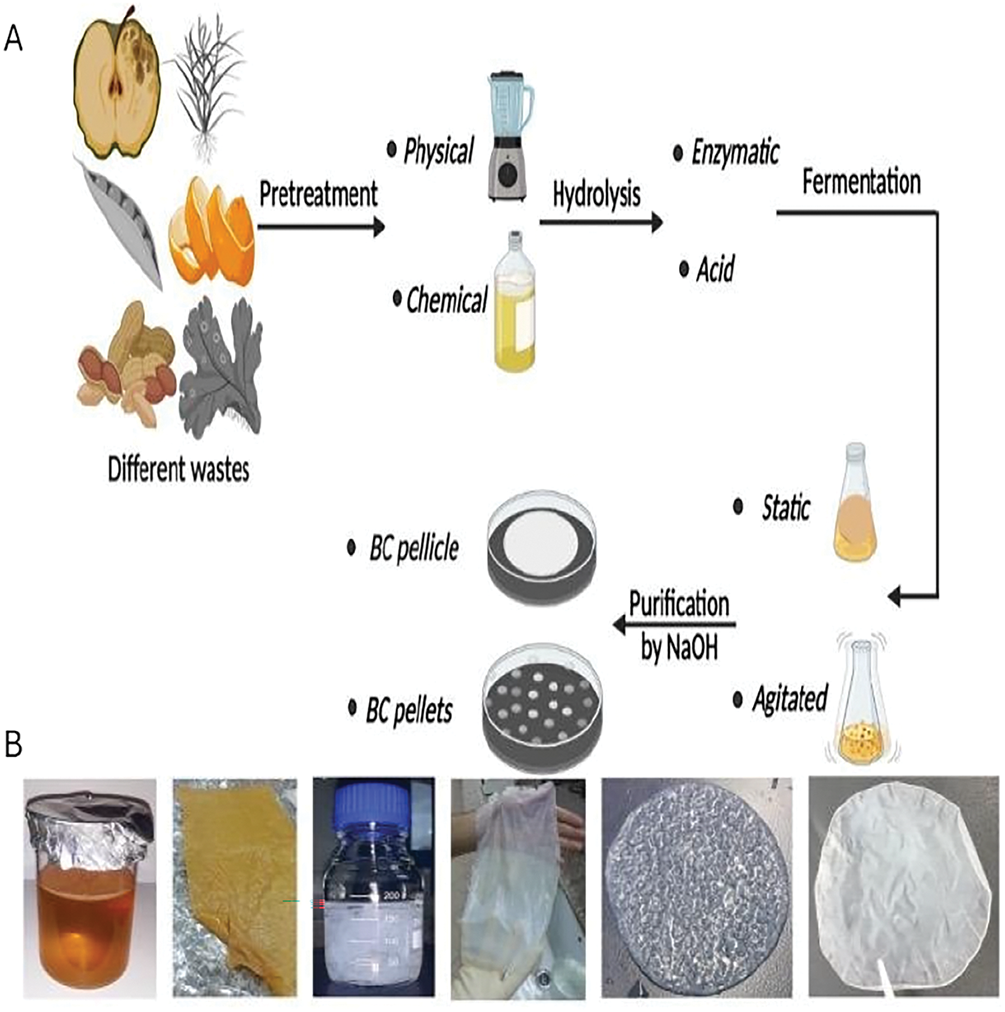
Figure 5: (A) Schematic illustration of the production of BC using low-cost effective media (Adapted under the terms of the Creative Commons Attribution 4.0 International License from Ref. [68], Copyright 2022, Springer Nature SharedIt); (B) Production and extraction of BC using the culture of K. Saccharivorans MD1 and the BC film (Adapted under the terms of the Creative Commons Attribution 4.0 International License from Ref. [59], Copyright 2020, Springer Nature SharedIt)
4.2 Bio Textiles from Lignocellulose
One of the major drawbacks of both the fashion and agriculture industries is their significant carbon footprint. As a result, bio-textiles have emerged as a potential solution to the environmental challenges posed by these sectors. Jayaprakash and colleagues [82] reported that millions of people worldwide, particularly in developing countries, are engaged in natural fiber production, spanning both small- and large-scale operations. In recent years, the bioprocessing of cellulosic agricultural biomass has gained considerable attention. Biomass derived from palm oil, corn stalks, bamboo, banana, pineapple, and rice husk is increasingly recognized for its benefits and is considered a secondary source of fiber [82].
Bio-textiles made from microbial cellulose involve the bioengineering and bio-fabrication of specific microbes such as Acetobacter xylinus to produce textiles for the clothing and footwear industries. Agro-waste is often converted into bacterial nanocellulose, which is then processed for use in textile manufacturing. Compared to conventional material production, bio-fabrication requires fewer chemicals, less water, and less energy, resulting in a smaller carbon footprint. Notably, the “Bio Couture” textile brand was founded as a research initiative exploring bacterial cellulose through Kombucha fermentation. Since then, extensive research has focused on producing nanocellulose using A. xylinus under aerobic conditions (Fig. 6). These bacteria biosynthesize extracellular cellulose nanofibrils that self-assemble into a highly organized, crystalline 3D structure [49].
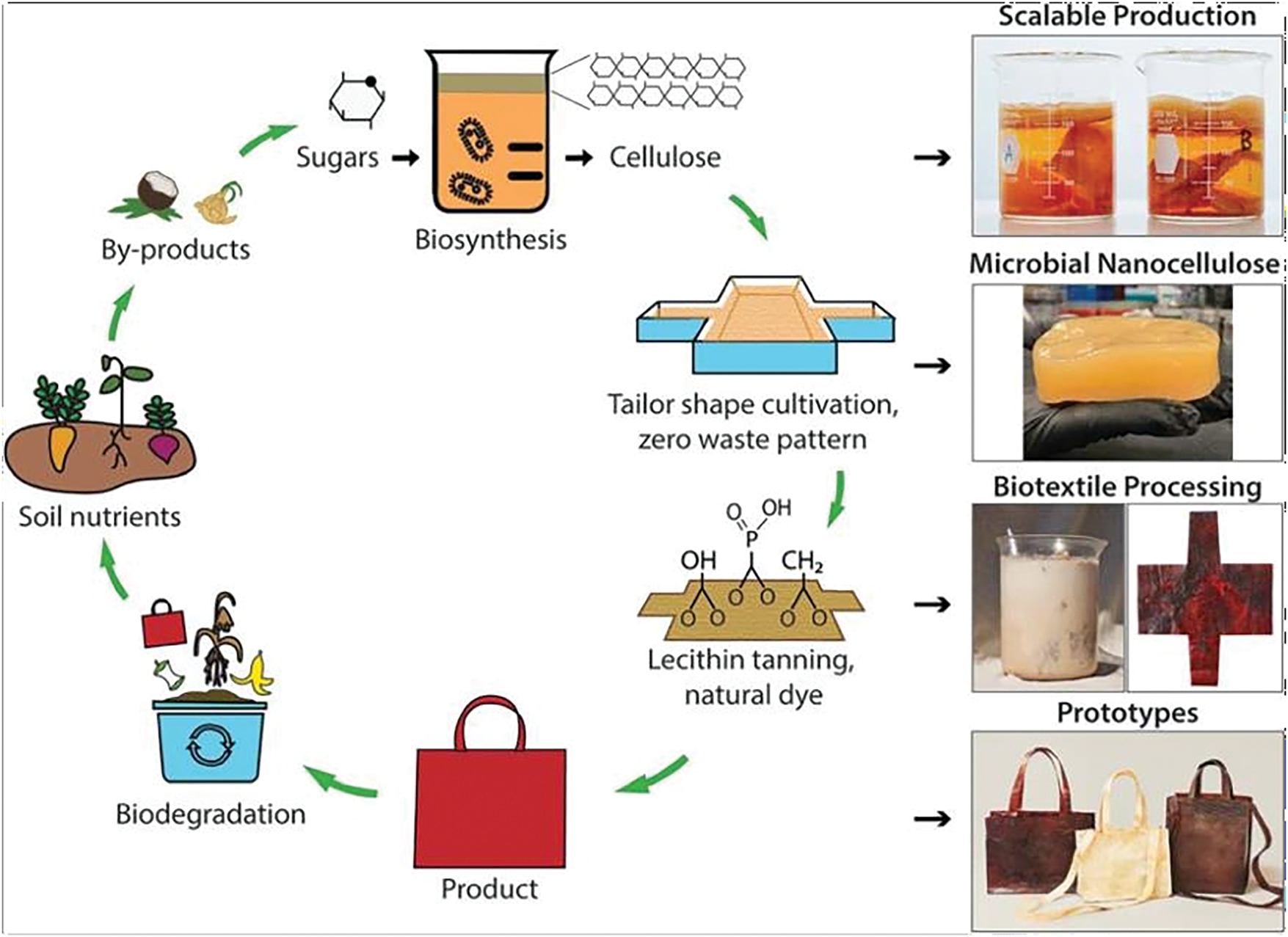
Figure 6: Microbial biofabrication from agro-industrial waste and development of bio-textiles from microbial cellulose (Adapted under the terms of the Common Creative BY-NC 3.0 license from [83], Copyright 2022, Royal Society of Chemistry)
A study by Stenton and colleagues [84] reported that the fashion sector contributes approximately 10 percent of global GHG emissions, including 4 percent of worldwide carbon dioxide emissions, with projections indicating a rise to 25 percent by 2050. Currently, only 65 percent of fibers used in the textile industry originate from natural sources, while a substantial amount of natural fiber is wasted in the form of food excess. For instance, the banana industry alone generates an estimated 270 million tons of waste annually [85]. In 2018, the biotechnology company Bolt Threads developed a plant-based synthetic leather called “Mylo,” suitable for use in clothing. Mylo is derived from mycelium, the root-like structure found in mushrooms, and serves as an eco-friendly alternative to conventional vegan leathers. Agrowaste fabric is another animal-free textile that offers a promising vegan substitute, with a texture and appearance similar to real silk [86]. Banana fibers present an environmentally friendly option to silk and cotton, holding significant potential for commercial applications in the textile and fiber industries. Enhancing the mechanical properties of banana fibers through softening treatments could expand their usability across a broader range of textile products. Bacterial cultures such as Bacillus aryabhattai, Bacillus licheniformis, and Bacillus subtilis have been employed to soften banana fibers for the development of sustainable bio-textiles [87]. These fibers possess a natural sheen and extremely fine inner strands within the stalk, enabling them to replicate the texture of silk.
Biofuels have gained significant attention as environmentally friendly sources of green energy and are considered promising alternatives to fossil fuels due to their economic and ecological benefits. They are sustainable and emit up to ten times fewer harmful gases than fossil fuels during consumption [40]. The biofuel industry is expected to benefit substantially from agricultural waste. Lignocellulosic biomass, the most abundant, sustainable, and cost-effective form of biomass, has become a central focus for generating not only thermal and electrical energy but also biofuels such as ethanol and biodiesel. Research has demonstrated the potential of this feedstock, including its use by Escherichia coli to produce ethanol, 1,2-propanediol, and L-lactate. Additionally, novel microbes capable of converting agro-waste into biofuel have been discovered near oil-contaminated sites, with Pseudomonas sp. and Ochrobactrum sp. being notable examples [40]. Pseudomonas aeruginosa WD23, isolated from petroleum refinery effluent, was recently found to degrade up to 27% of crude oil in seawater with minimal supplementation [88], paving the way for biofuel production from agro-waste.
The Melle-Boinot process is the conventional method for bioethanol production via batch fermentation. Saccharomyces cerevisiae is the most commonly used microorganism for fermenting sugar-rich feedstocks. This yeast has long been employed in ethanol production. Schizosaccharomyces pombe is also utilized due to its ability to tolerate high osmotic pressures and solid content [89]. In response to growing interest, researchers have explored other microorganisms for bioethanol production, including Zymomonas mobilis, Klebsiella oxytoca, Escherichia coli, Thermoanaerobacter ethanolicus, Pichia stipitis, Candida shehatae, and Mucor indicus. Kluyveromyces marxianus is considered a promising alternative to S. cerevisiae, although further research is needed to optimize its performance at industrial scale [89] (Fig. 7).
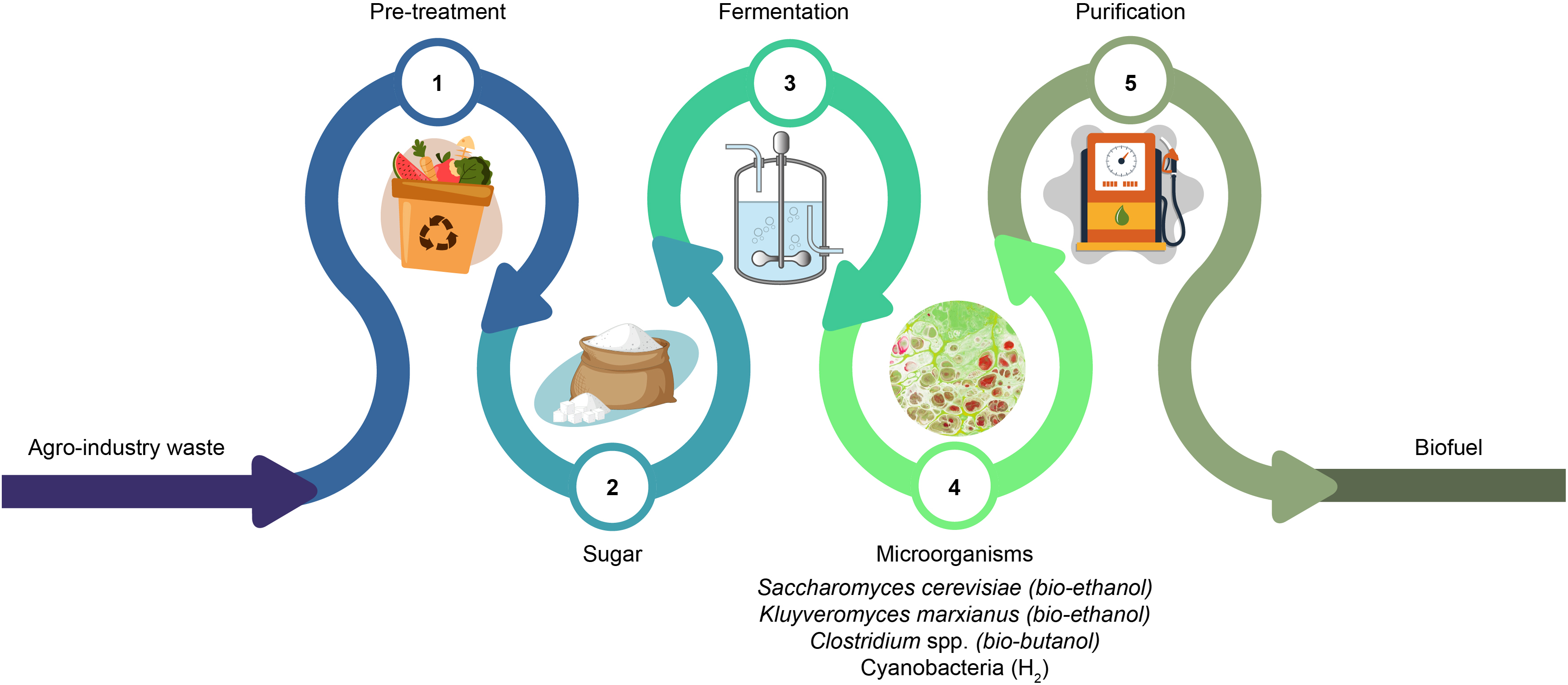
Figure 7: Summary of methods and technologies involved in the production of biofuel from agro-industry waste (Created using the Adobe Illustrator)
In addition to ethanol, biomass fermentation can produce n-butanol using microorganisms from the genus Clostridium, a process known as Acetone-Butanol-Ethanol (ABE) fermentation or the Weizmann process [90]. This method has been implemented at industrial scale, with facilities producing multiple tonnes per day. Hydrogen (H2) gas has also attracted global interest as a highly promising, renewable, and eco-friendly energy source [91]. H2 is versatile and can be produced by various organisms under specific conditions. For example, microalgae use light energy to split water molecules and generate hydrogen, while cyanobacteria typically consume carbohydrates to store energy from photosynthesis and produce H2 from water molecules [92].
4.4 Conversion into Biosurfactants
Biosurfactants are increasingly recognized as eco-friendly alternatives to synthetic surfactants due to their biodegradability, non-toxicity, and wide-ranging applications in biotechnology, healthcare, and agriculture [93]. However, their commercial viability is often limited by high production costs, particularly those associated with fermentation media and downstream processing. A promising solution lies in the valorization of agro-industrial waste, which provides a cost-effective, renewable, and abundant substrate for microbial biosurfactant synthesis. Waste materials from fruit and vegetable processing, oil refining, starch and sugar industries, and dairy effluents have been successfully repurposed to support biosurfactant production, while simultaneously addressing environmental pollution and waste disposal challenges [94]. This approach not only lowers overall production costs but also aligns with circular bioeconomy principles by converting low-value residues into high-value biomolecules. Additionally, biosurfactant production from agro-waste promotes waste minimization, improves resource efficiency, and supports sustainable industrial practices [93]. As such, agro-industrial waste emerges as a strategic enabler for scaling biosurfactant technologies while reinforcing environmental stewardship.
Microorganisms such as bacteria, fungi, and yeasts are capable of producing various biosurfactants that can replace synthetic chemical compounds [95]. These biosurfactants possess both hydrophilic and hydrophobic properties, consisting of distinct moieties that reduce surface tension and facilitate the formation of emulsions or micelles [96]. The nonpolar hydrophobic tail typically comprises hydrocarbon chains of varying lengths and complexities, while the polar hydrophilic head includes carbohydrates, amino acids, peptides, alcohols, or phosphate carboxylic acids. Despite their advantages, the high cost of biosurfactant production particularly due to fermentation media and downstream processing poses a challenge for large-scale manufacturing. To address this, recent developments have focused on utilizing industrial agricultural waste as renewable raw substrates for biosurfactant synthesis [97]. These include waste from fruit and vegetable processing, starch residues, sugar industry byproducts, and distillery effluents (Table 3). Microbes from the genera Bacillus, Candida, and Pseudomonas are commonly known for biosurfactant production, typically as metabolic byproducts during fermentation. Optimizing fermentation conditions such as temperature, pH, and oxygen levels is essential for maximizing yield [98]. Biosurfactants are classified based on their chemical structure, microbial origin, and functional characteristics. Common types include glycolipids, lipopeptides, fatty acids, ionic, and polymeric biosurfactants [99]. Notably, biosurfactants can be co-produced with other valuable products such as biofuels and enzymes during biorefinery processes (Fig. 8).
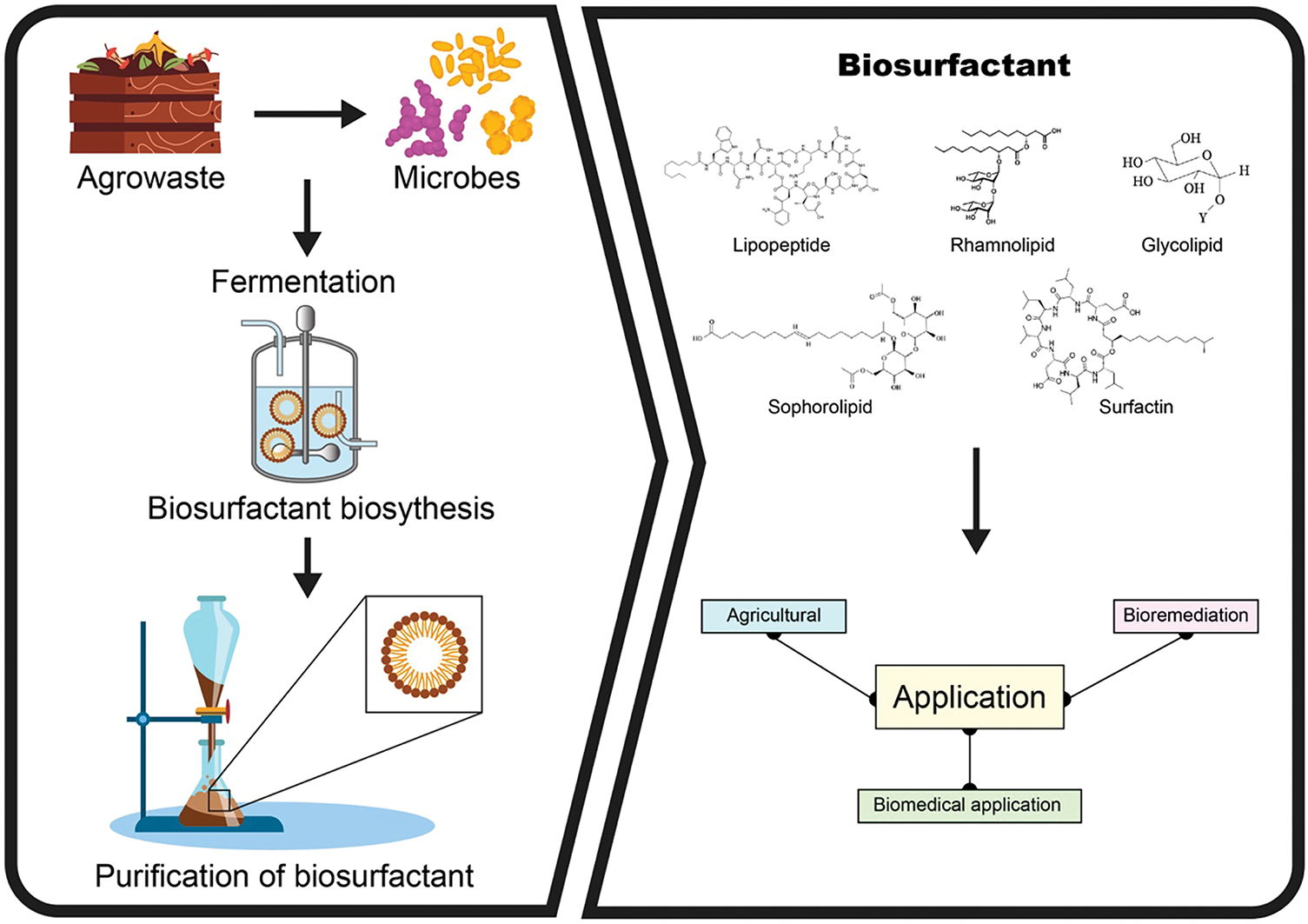
Figure 8: Schematic diagram of the biosynthesis of biosurfactant agro-industrial waste using microbial fermentation (Created using BioRender.com)
Bioplastics currently account for just 1% of the 335 million tonnes of plastic produced annually, but the market is expanding rapidly as new biopolymers, applications, and products emerge [100]. The widespread use of conventional plastics has significant environmental consequences, prompting increased interest in biodegradable alternatives [101]. Bioplastics are bio-based, biodegradable, or compostable materials that serve as substitutes for fossil-based polymers [102]. Key types include poly(butylene succinate) (PBS), poly(butylene adipate-co-terephthalate) (PBAT), polycaprolactone (PCL), polylactic acid (PLA), and polyhydroxyalkanoates (PHA) [103]. Microbial fermentation remains one of the most innovative approaches for bioplastic production.
PLA and PHA are bacterial-based bioplastics [104], synthesized and polymerized by microorganisms such as Azotobacter beijernickii, Cupriavidus necator, and strains from Bacillus, Pseudomonas, Rhodococcus, and Lactobacillus genera. PHA is biosynthesized in response to excess carbon and nutrient-limited conditions [105], while PLA is produced through microbial conversion of substrates into lactic acid [106]. Despite their promise, high production costs remain a barrier to industrial-scale commercialization [107]. Recent research highlights the potential of agro-waste and food byproducts to reduce costs in bacterial bioplastic production [105]. These bioplastics are manufactured using renewable biomass sources such as corn, rice, palm fiber, potatoes, and wood cellulose, in contrast to conventional plastics derived from petroleum and natural gas [108] (Fig. 9). Careful selection of microorganisms and carbon substrates can significantly lower production costs. Utilizing agricultural and industrial waste as renewable carbon sources not only addresses waste management challenges but also supports a sustainable future [109]. Replacing synthetic plastics with biodegradable alternatives could reduce global greenhouse gas emissions by up to 800 million tons annually [110].
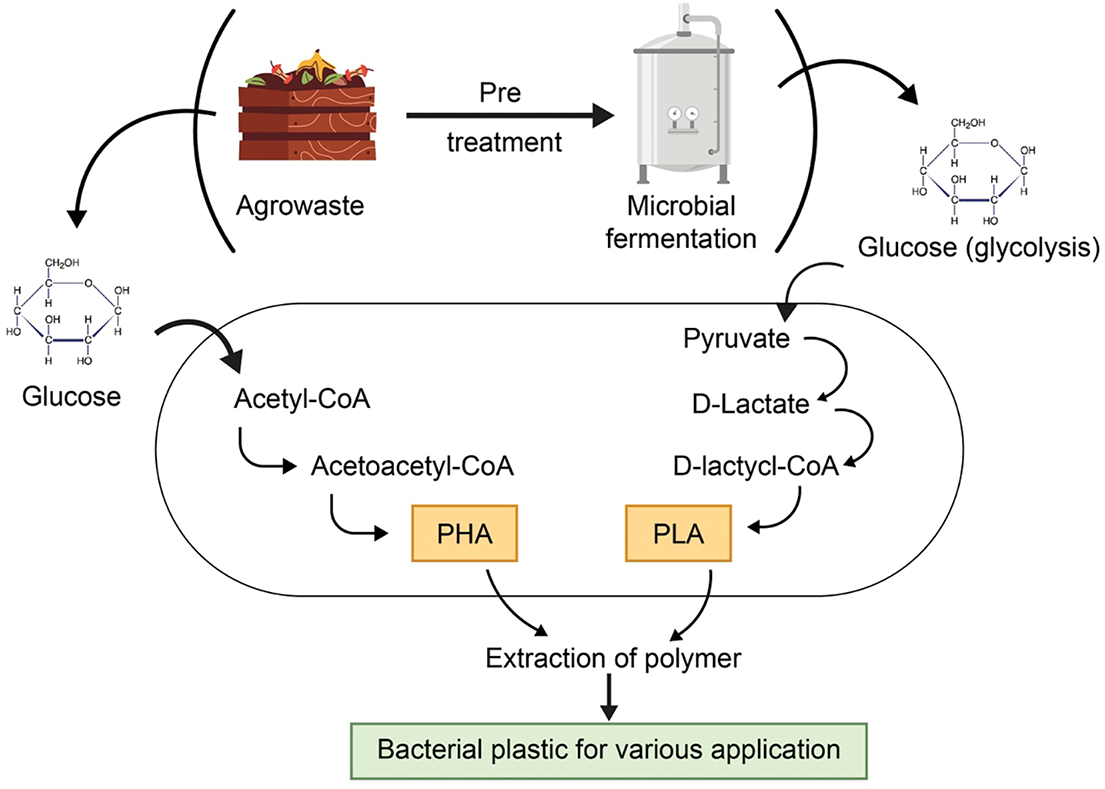
Figure 9: Schematic diagram of the production of bacterial based bioplastic PLA and PHA biosynthesized from agro-industrial waste (Created using BioRender.com)
5 Challenges and Future Direction of Sustainable Agro-Waste Conversion Using Microbial Biorefineries
Agro-industrial waste, once considered a disposal challenge, is now increasingly recognized as a valuable resource for sustainable innovation and economic development [47]. To fully unlock the potential of bacterial biorefineries in valorizing such waste, supportive policy models and regulatory frameworks are essential. National bioeconomy strategies should prioritize microbial biorefineries through mission-oriented R&D funding, public–private partnerships, and innovation clusters that help de-risk early-stage technologies [119]. Regulatory frameworks must evolve to include tiered biosafety assessments for microbial strains, streamlined approval pathways for non-pathogenic and genetically optimized organisms, and waste-to-resource certification schemes that legitimize agro-waste as industrial feedstock [1]. Extended Producer Responsibility (EPR) policies can further incentivize industries to adopt biodegradable inputs and invest in microbial valorization platforms. Technologically, scaling bacterial biorefineries requires advanced genetic tools such as CRISPR-Cas systems, gene shuffling, and in situ mutagenesis algorithms to enhance strain performance and unlock novel metabolic pathways [120–122]. The integration of multi-omics, whole genome sequencing, and bioinformatics platforms will optimize enzyme efficiency and metabolite yield [120]. Additionally, modular bioreactor designs, real-time biosensors, and AI-driven techno-economic modeling—including digital twins—are critical for simulating, monitoring, and scaling production systems efficiently [123]. Together, these policy and technological strategies can transform bacterial biorefineries into cornerstone platforms for a circular bioeconomy and climate-resilient manufacturing (Fig. 10).

Figure 10: Schematic representation on the improved production involved in sustainably employing various microbial systems to develop bio-based products
Environmental concerns, resource scarcity, and the growing demand for renewable and bio-based products are the key drivers behind the sustainable development paradigm in agro-industry conversion [124]. However, converting agro-waste into useful products faces several major challenges, including the absence of robust policy or legal frameworks, limited resources, technological constraints in processing diverse agro-industrial waste sustainably, and a lack of financial support, public awareness, and transportation infrastructure (Fig. 11). In order to overcome these barriers, future research efforts must focus on developing integrated biorefineries capable of simultaneously processing various types of agro-waste into valuable products [125]. Agro-waste conversion also holds significant potential for environmental remediation and effective waste management. Overall, the future of sustainable agro-waste conversion is promising, offering opportunities for bioeconomy development, production of high-value goods, bioremediation, and improved waste handling [126]. These advancements could pave the way for a more sustainable and circular agricultural system, promoting greener and more efficient resource use [127,128]. By adopting innovative technologies and sustainable practices, we can transform agro-waste into a valuable resource, generate new employment opportunities, and contribute to a cleaner, more resource-efficient planet [129].
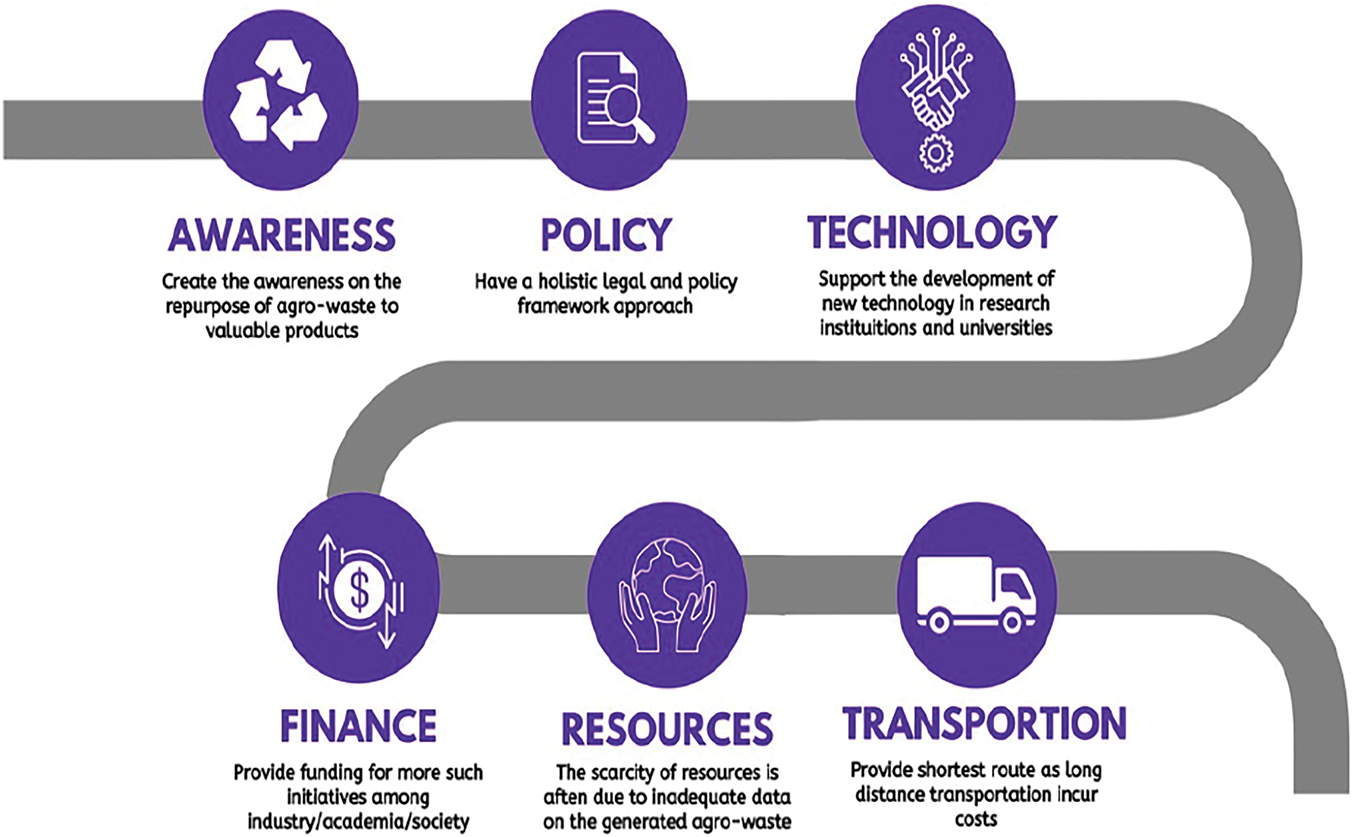
Figure 11: Schematic illustration on the challenges faced in the sustainable agro-waste conversion using microbial biorefineries (Created using Canva.com)
The agricultural sector generates substantial quantities of agro-industrial processing waste, animal waste, pesticides, and fertilizers, all of which contribute significantly to environmental pollution. This review highlights the potential of converting agro-waste into valuable secondary metabolites, bioactive compounds, bioenergy, and sustainable food packaging materials. The global market for products derived from agricultural waste is experiencing rapid and exponential growth. Beyond reducing production costs, these innovations offer promising solutions to urgent environmental challenges. Microbial transformation of agro-industrial waste enhances the sustainability of industrial processes by promoting resource efficiency and circularity. This review also explores strategies to maximize the yield of high-value products and investigates novel types of agro-industrial waste as viable substrates for bioconversion. However, challenges persist in scaling up production and addressing existing knowledge gaps. These gaps can be bridged through targeted research, unlocking vast opportunities in the valorization of agro-industrial residues. In order to advance this field, research efforts must focus on optimizing pretreatment methods and downstream recovery processes. The objective is to develop efficient, scalable technologies for producing sustainable products that are safe for both human health and the environment. A multidisciplinary approach is essential to ensure the long-term viability of these processes. By integrating expertise across scientific and industrial domains, we can minimize waste, reduce environmental impact, and foster the development of eco-friendly products and technologies that benefit both society and the planet.
Acknowledgement: Not applicable.
Funding Statement: The authors would like to thank the Postgraduate Research Grant (PGRG) by Universiti Malaysia Terengganu [UMT/PGRG/2024/ 55527] for the financial support.
Author Contributions: Conceptualization: Sevakumaran Vigneswari; Lakshiminarayanan Rajamani. Writing—original draft preparation: Sevakumaran Vigneswari; Muhammad Shahrul Md Noor; Fazilah Ariffin; Azila Adnan; Amirah Alias; Lakshmanan Muthulakshmi; Nurul Nadhirah Ruzelan; Hemalatha Murugaiah; Nor Omaima Harun. Writing—review and editing: Sevakumaran Vigneswari; Azila Adnan. Visualization: Sevakumaran Vigneswari. Supervision: Sevakumaran Vigneswari; Lakshiminarayanan Rajamani. Project administration: Sevakumaran Vigneswari. Funding acquisition: Sevakumaran Vigneswari. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable (This article does not involve data availability, and this section is not applicable).
Ethics Approval: Not applicable (This study does not involve humans or animals).
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
References
1. El-Ramady H, Domokos-Szabolcsy É, Abdalla N. Agro-waste management and circular bioeconomy. Sustainability. 2022;14(9):4872. doi:10.3390/su14094872. [Google Scholar] [CrossRef]
2. Priya AK, Alagumalai A, Balaji D, Song H. Bio-based agricultural products: a sustainable alternative to agrochemicals for promoting a circular economy. RSC Sustain. 2023;1(4):746–62. doi:10.1039/d3su00075c. [Google Scholar] [CrossRef]
3. Nguyen TTU, Nguyen PT, Nguyen TT, Nguyen TPT, Nguyen HT. Rice leaves or ricegrass—available biomaterial with potential biological activities for different industrial applications: a review. Discov Food. 2024;4(1):106. doi:10.1007/s44187-024-00187-4. [Google Scholar] [CrossRef]
4. Capanoglu E, Nemli E, Tomas-Barberan F. Novel approaches in the valorization of agricultural wastes and their applications. J Agric Food Chem. 2022;70(23):6787–804. doi:10.1021/acs.jafc.1c07104. [Google Scholar] [PubMed] [CrossRef]
5. Blasi A, Verardi A, Lopresto CG, Siciliano S, Sangiorgio P. Lignocellulosic agricultural waste valorization to obtain valuable products: an overview. Recycling. 2023;8(4):61. doi:10.3390/recycling8040061. [Google Scholar] [CrossRef]
6. Hasanin MS, Abdelraof M, Hashem AH, El Saied H. Sustainable bacterial cellulose production by Achromobacter using mango peel waste. Microb Cell Fact. 2023;22(1):24. doi:10.1186/s12934-023-02031-3. [Google Scholar] [PubMed] [CrossRef]
7. Benatti ALT, de Lourdes Teixeira de Moraes Polizeli M. Lignocellulolytic biocatalysts: the main players involved in multiple biotechnological processes for biomass valorization. Microorganisms. 2023;11(1):162. doi:10.3390/microorganisms11010162. [Google Scholar] [PubMed] [CrossRef]
8. de Dieu Marcel Ufitikirezi J, Filip M, Ghorbani M, Zoubek T, Olšan P, Bumbálek R, et al. Agricultural waste valorization: exploring environmentally friendly approaches to bioenergy conversion. Sustainability. 2024;16(9):3617. doi:10.3390/su16093617. [Google Scholar] [CrossRef]
9. Kataya G, Cornu D, Bechelany M, Hijazi A, Issa M. Biomass waste conversion technologies and its application for sustainable environmental development—a review. Agronomy. 2023;13(11):2833. doi:10.3390/agronomy13112833. [Google Scholar] [CrossRef]
10. Duan Y, Tarafdar A, Kumar V, Ganeshan P, Rajendran K, Shekhar Giri B, et al. Sustainable biorefinery approaches towards circular economy for conversion of biowaste to value added materials and future perspectives. Fuel. 2022;325(19):124846. doi:10.1016/j.fuel.2022.124846. [Google Scholar] [CrossRef]
11. Prado-Acebo I, Cubero-Cardoso J, Lu-Chau TA, Eibes G. Integral multi-valorization of agro-industrial wastes: a review. Waste Manag. 2024;183:42–52. doi:10.1016/j.wasman.2024.05.001. [Google Scholar] [PubMed] [CrossRef]
12. Sharma V, Tsai ML, Nargotra P, Chen CW, Kuo CH, Sun PP, et al. Agro-industrial food waste as a low-cost substrate for sustainable production of industrial enzymes: a critical review. Catalysts. 2022;12(11):1373. doi:10.3390/catal12111373. [Google Scholar] [CrossRef]
13. Bala S, Garg D, Sridhar K, Inbaraj BS, Singh R, Kamma S, et al. Transformation of agro-waste into value-added bioproducts and bioactive compounds: micro/nano formulations and application in the agri-food-pharma sector. Bioengineering. 2023;10(2):152. doi:10.3390/bioengineering10020152. [Google Scholar] [PubMed] [CrossRef]
14. Chojnacka K. Valorization of biorefinery residues for sustainable fertilizer production: a comprehensive review. Biomass Convers Biorefin. 2023;13(16):14359–88. doi:10.1007/s13399-023-04639-2. [Google Scholar] [CrossRef]
15. Prabha S, Vijay AK, Paul RR, George B. Cyanobacterial biorefinery: towards economic feasibility through the maximum valorization of biomass. Sci Total Environ. 2022;814(2):152795. doi:10.1016/j.scitotenv.2021.152795. [Google Scholar] [PubMed] [CrossRef]
16. Bedoić R, Čuček L, Ćosić B, Krajnc D, Smoljanić G, Kravanja Z, et al. Green biomass to biogas—a study on anaerobic digestion of residue grass. J Clean Prod. 2019;213:700–9. doi:10.1016/j.jclepro.2018.12.224. [Google Scholar] [CrossRef]
17. Ozlu E, Arriaga FJ, Bilen S, Gozukara G, Babur E. Carbon footprint management by agricultural practices. Biology. 2022;11(10):1453. doi:10.3390/biology11101453. [Google Scholar] [PubMed] [CrossRef]
18. Kumar R, Saurabh K, Kumawat N, Sundaram PK, Mishra JS, Singh DK, et al. Sustaining productivity through integrated use of microbes in agriculture. In: Role of microbial communities for sustainability. Singapore: Springer; 2021. p. 109–45. doi:10.1007/978-981-15-9912-5_5. [Google Scholar] [CrossRef]
19. Hodge D, Singh A, Kumar A. Life cycle assessment of anaerobic digestion systems for organic waste treatment. Renew Sustain Energ Rev. 2022;158(3):112093. doi:10.1016/j.rser.2022.112093. [Google Scholar] [CrossRef]
20. Kamm B, Kamm M. Biorefineries-Status quo and future perspectives. Biofuels Bioprod Biorefin. 2023;17(1):3–20. doi:10.1002/bbb.2405. [Google Scholar] [CrossRef]
21. Yaashikaa PR, Kumar PS, Nhung TC, Hemavathy RV, Jawahar MJ, Neshaanthini JP, et al. A review on landfill system for municipal solid wastes: insight into leachate, gas emissions, environmental and economic analysis. Chemosphere. 2022;309(Pt 1):136627. doi:10.1016/j.chemosphere.2022.136627. [Google Scholar] [PubMed] [CrossRef]
22. Jagaba AH, Kutty SM, Lawal IM, Abubakar S, Hassan I, Zubairu I, et al. Sequencing batch reactor technology for landfill leachate treatment: a state-of-the-art review. J Environ Manage. 2021;282(6):111946. doi:10.1016/j.jenvman.2021.111946. [Google Scholar] [PubMed] [CrossRef]
23. Vyas S, Prajapati P, Shah AV, Kumar Srivastava V, Varjani S. Opportunities and knowledge gaps in biochemical interventions for mining of resources from solid waste: a special focus on anaerobic digestion. Fuel. 2022;311:122625. doi:10.1016/j.fuel.2021.122625. [Google Scholar] [CrossRef]
24. Guo HN, Wu SB, Tian YJ, Zhang J, Liu HT. Application of machine learning methods for the prediction of organic solid waste treatment and recycling processes: a review. Bioresour Technol. 2021;319:124114. doi:10.1016/j.biortech.2020.124114. [Google Scholar] [PubMed] [CrossRef]
25. Chang CT, Negi S, Rani A, Hu AH, Pan SY, Kumar S. Food waste and soybean curd residue composting by black soldier fly. Environ Res. 2022;214(Pt 1):113792. doi:10.1016/j.envres.2022.113792. [Google Scholar] [PubMed] [CrossRef]
26. Peng L, Ma R, Jiang S, Luo W, Li Y, Wang G, et al. Co-composting of kitchen waste with agriculture and forestry residues and characteristics of compost with different particle size: an industrial scale case study. Waste Manag. 2022;149(50):313–22. doi:10.1016/j.wasman.2022.06.029. [Google Scholar] [PubMed] [CrossRef]
27. Cui G, Lü F, Hu T, Zhang H, Shao L, He P. Vermicomposting leads to more abundant microplastics in the municipal excess sludge. Chemosphere. 2022;307(Pt 3):136042. doi:10.1016/j.chemosphere.2022.136042. [Google Scholar] [PubMed] [CrossRef]
28. Gong X, Li S, Carson MA, Chang SX, Wu Q, Wang L, et al. Spent mushroom substrate and cattle manure amendments enhance the transformation of garden waste into vermicomposts using the earthworm Eisenia fetida. J Environ Manage. 2019;248(5):109263. doi:10.1016/j.jenvman.2019.109263. [Google Scholar] [PubMed] [CrossRef]
29. Kamaruddin MA, Yusoff MS, Aziz HA. Environmental performance analysis of landfilling, composting, and EP-50 composting. Circular Economy and Sustainability. [cited 2025 Oct 1]. Available from: https://link.springer.com/article/10.1007/s43621-024-00614-7. [Google Scholar]
30. Zulkifli NA, Hassan MA, Rahman NA. Comparative life cycle assessment of landfilling vs sustainable waste management methods. Environments. 2024;11(11):248. doi:10.3390/environments11110248. [Google Scholar] [CrossRef]
31. Usmani Z, Sharma M, Awasthi AK, Sivakumar N, Lukk T, Pecoraro L, et al. Bioprocessing of waste biomass for sustainable product development and minimizing environmental impact. Bioresour Technol. 2021;322(2):124548. doi:10.1016/j.biortech.2020.124548. [Google Scholar] [PubMed] [CrossRef]
32. Gírio F, Marques S, Pinto F, Oliveira AC, Costa P, Reis A. Biorefineries in the world. In: Biorefineries: targeting energy, high value products and waste valorisation. Cham, Switzerland: Springer International Publishing; 2017. p. 227–81. [Google Scholar]
33. Shukla S, Khan R. Sustainable waste management approach: a paradigm shift towards zero waste into landfills. In: Advanced organic waste management. Amsterdam, The Netherlands: Elsevier; 2022. p. 381–95. doi:10.1016/b978-0-323-85792-5.00006-x. [Google Scholar] [CrossRef]
34. Supreeth M. Enhanced remediation of pollutants by microorganisms-plant combination. Int J Environ Sci Technol. 2022;19(5):4587–98. doi:10.1007/s13762-021-03354-7. [Google Scholar] [PubMed] [CrossRef]
35. De Buck V, Polanska M, Van Impe J. Modeling biowaste biorefineries: a review. Front Sustain Food Syst. 2020;4:11. doi:10.3389/fsufs.2020.00011. [Google Scholar] [CrossRef]
36. Regueira A, Lema JM, Mauricio-Iglesias M. Microbial inefficient substrate use through the perspective of resource allocation models. Curr Opin Biotechnol. 2021;67:130–40. doi:10.1016/j.copbio.2021.01.015. [Google Scholar] [PubMed] [CrossRef]
37. Bankar A, Patil S, Shinde M, Shinde S, Kowligi B. Potential of microbial extremophiles for biotechnological applications: an overview. In: Microbial extremozymes. Amsterdam, The Netherlands: Elsevier; 2022. p. 89–109. doi:10.1016/b978-0-12-822945-3.00016-6. [Google Scholar] [CrossRef]
38. Tsegaye B, Jaiswal S, Jaiswal AK. Food waste biorefinery: pathway towards circular bioeconomy. Foods. 2021;10(6):1174. doi:10.3390/foods10061174. [Google Scholar] [PubMed] [CrossRef]
39. Galbe M, Wallberg O. Pretreatment for biorefineries: a review of common methods for efficient utilisation of lignocellulosic materials. Biotechnol Biofuels. 2019;12(1):294. doi:10.1186/s13068-019-1634-1. [Google Scholar] [PubMed] [CrossRef]
40. Awogbemi O, Von Kallon DV. Valorization of agricultural wastes for biofuel applications. Heliyon. 2022;8(10):e11117. doi:10.1016/j.heliyon.2022.e11117. [Google Scholar] [PubMed] [CrossRef]
41. Firmino FC, Porcellato D, Cox M, Suen G, Broadbent JR, Steele JL. Characterization of microbial communities in ethanol biorefineries. J Ind Microbiol Biotechnol. 2020;47(2):183–95. doi:10.1007/s10295-019-02254-7. [Google Scholar] [PubMed] [CrossRef]
42. Jiang LL, Zhou JJ, Quan CS, Xiu ZL. Advances in industrial microbiome based on microbial consortium for biorefinery. Bioresour Bioprocess. 2017;4(1):11. doi:10.1186/s40643-017-0141-0. [Google Scholar] [PubMed] [CrossRef]
43. Husarcíková J, Voß H, de María PD, Schallmey A. Microbial β-etherases and glutathione lyases for lignin valorisation in biorefineries: current state and future perspectives. Appl Microbiol Biotechnol. 2018;102(13):5391–401. doi:10.1007/s00253-018-9040-3. [Google Scholar] [PubMed] [CrossRef]
44. Dawood A, Ma K. Applications of microbial β-mannanases. Front Bioeng Biotechnol. 2020;8:598630. doi:10.3389/fbioe.2020.598630. [Google Scholar] [PubMed] [CrossRef]
45. Cao MK, Guo HT, Zheng GD, Chen TB, Cai L. Microbial succession and degradation during kitchen waste biodrying, highlighting the thermophilic phase. Bioresour Technol. 2021;326(7–8):124762. doi:10.1016/j.biortech.2021.124762. [Google Scholar] [PubMed] [CrossRef]
46. Dong X, Song J, Duan H. Prioritizing countries for implementing waste recycling under socioeconomic supports. J Environ Manag. 2022;322(3):116158. doi:10.1016/j.jenvman.2022.116158. [Google Scholar] [CrossRef]
47. de Oliveira Silva S, Mafra AKC, Pelissari FM, Rodrigues de Lemos L, Molina G. Biotechnology in agro-industry: valorization of agricultural wastes, by-products and sustainable practices. Microorganisms. 2025;13(8):1789. doi:10.3390/microorganisms13081789. [Google Scholar] [PubMed] [CrossRef]
48. Betlej I, Zakaria S, Chia CH, Lee SH. Bacterial cellulose—properties and its potential application. Sains Malays. 2021;50(2):395–404. [Google Scholar]
49. Yanti NA, Sari RM, Sari N. Characterization of bacterial cellulose produced by Acetobacter xylinum using sago liquid waste. Pak J Biol Sci. 2021;24(5):335–44. doi:10.3923/pjbs.2021.335.344. [Google Scholar] [PubMed] [CrossRef]
50. Zhong C. Industrial-scale production and applications of bacterial cellulose. Front Bioeng Biotechnol. 2020;8:605374. doi:10.3389/fbioe.2020.605374. [Google Scholar] [PubMed] [CrossRef]
51. Kadier A, Ilyas RA, Huzaifah MM, Harihastuti N, Sapuan SM, Harussani MM, et al. Use of industrial wastes as sustainable nutrient sources for bacterial cellulose (BC) production: mechanism, advances, and future perspectives. Polymers. 2021;13(19):3365. doi:10.3390/polym13193365. [Google Scholar] [PubMed] [CrossRef]
52. Taokaew S. Bacterial nanocellulose produced by cost-effective and sustainable methods and its applications: a review. Fermentation. 2024;10(6):316. doi:10.3390/fermentation10060316. [Google Scholar] [CrossRef]
53. Chua GK, Yang Kae S, Yang Kae S. Development of fed-batch strategy to produce bacteria cellulose. ASM Sci J. 2023;18:1–14. doi:10.32802/asmscj.2023.1473. [Google Scholar] [CrossRef]
54. Khami S, Khamwichit W, Suwannahong K, Sanongraj W. Characteristics of bacterial cellulose production from agricultural wastes. Adv Mater Res. 2014;931–932:693–7. doi:10.4028/www.scientific.net/amr.931-932.693. [Google Scholar] [CrossRef]
55. Revin VV, Liyas’kina EV, Sapunova NB, Bogatyreva AO. Isolation and characterization of the strains producing bacterial cellulose. Microbiology. 2020;89(1):86–95. doi:10.1134/s0026261720010130. [Google Scholar] [CrossRef]
56. Kuo CH, Lin PJ, Lee CK. Enzymatic saccharification of dissolution pretreated waste cellulosic fabrics for bacterial cellulose production by Gluconacetobacter xylinus. J Chem Technol Biotechnol. 2010;85(10):1346–52. doi:10.1002/jctb.2439. [Google Scholar] [CrossRef]
57. Soemphol W, Hongsachart P, Tanamool V. Production and characterization of bacterial cellulose produced from agricultural by-product by Gluconacetobacter strains. Mater Today Proc. 2018;5(5):11159–68. doi:10.1016/j.matpr.2018.01.036. [Google Scholar] [CrossRef]
58. Quiñones-Cerna C, Rodríguez-Soto JC, Barraza-Jáuregui G, Huanes-Carranza J, Cruz-Monzón JA, Ugarte-López W, et al. Bioconversion of agroindustrial Asparagus waste into bacterial cellulose by Komagataeibacter rhaeticus. Sustainability. 2024;16(2):736. doi:10.3390/su16020736. [Google Scholar] [CrossRef]
59. Abol-Fotouh D, Hassan MA, Shokry H, Roig A, Azab MS, Kashyout AEB. Bacterial nanocellulose from agro-industrial wastes: low-cost and enhanced production by Komagataeibacter saccharivorans MD1. Sci Rep. 2020;10(1):3491. doi:10.1038/s41598-020-60315-9. [Google Scholar] [PubMed] [CrossRef]
60. Güzel F, Sayğılı G. Utilization of fruit and vegetable residues (cucumber, tomato, apple, melon, and kiwi) for the production of enzymatic hydrolysates with potential antiox-idant activity. J Environ Chem Eng. 2020;8(4):104002. doi:10.1016/j.jece.2020.104002. [Google Scholar] [CrossRef]
61. Ciecholewska-Juśko D, Broda M, Żywicka A, Styburski D, Sobolewski P, Gorący K, et al. Potato juice, a starch industry waste, as a cost-effective medium for the biosynthesis of bacterial cellulose. Int J Mol Sci. 2021;22(19):10807. doi:10.3390/ijms221910807. [Google Scholar] [PubMed] [CrossRef]
62. Cannazza P, Rissanen AJ, Guizelini D, Losoi P, Sarlin E, Romano D, et al. Characterization of Komagataeibacter isolate reveals new prospects in waste stream valorization for bacterial cellulose production. Microorganisms. 2021;9(11):2230. doi:10.3390/microorganisms9112230. [Google Scholar] [PubMed] [CrossRef]
63. Moradi M, Jacek P, Farhangfar A, Guimarães JT, Forough M. The role of genetic manipulation and in situ modifications on production of bacterial nanocellulose: a review. Int J Biol Macromol. 2021;183(5):635–50. doi:10.1016/j.ijbiomac.2021.04.173. [Google Scholar] [PubMed] [CrossRef]
64. Gorgieva S, Jančič U, Cepec E, Trček J. Production efficiency and properties of bacterial cellulose membranes in a novel grape pomace hydrolysate by Komagataeibacter melomenusus AV436T and Komagataeibacter xylinus LMG 1518. Int J Biol Macromol. 2023;244(25):125368. doi:10.1016/j.ijbiomac.2023.125368. [Google Scholar] [PubMed] [CrossRef]
65. Mohd Razin SN, Oslan SN, Muhd Noor ND. Physical parameters optimization of bacterial cellulose from Komagataeibacter sucrofermentans. Pertanika J Sci Technol. 2025;33(2):893–912. doi:10.47836/pjst.33.2.16. [Google Scholar] [CrossRef]
66. Jozala AF, Pértile RAN, dos Santos CA, de Carvalho Santos-Ebinuma V, Seckler MM, Gama FM, et al. Bacterial cellulose production by Gluconacetobacter xylinus by employing alternative culture media. Appl Microbiol Biotechnol. 2015;99(3):1181–90. doi:10.1007/s00253-014-6232-3. [Google Scholar] [PubMed] [CrossRef]
67. Hungund BS, Gupta SG. Production of bacterial cellulose from Enterobacter amnigenus GH-1 isolated from rotten apple. World J Microbiol Biotechnol. 2010;26(10):1823–8. doi:10.1007/s11274-010-0363-1. [Google Scholar] [CrossRef]
68. El-Gendi H, Salama A, El-Fakharany EM, Saleh AK. Optimization of bacterial cellulose production from prickly pear peels and its ex situ impregnation with fruit byproducts for antimicrobial and strawberry packaging applications. Carbohydr Polym. 2023;302:120383. doi:10.1016/j.carbpol.2022.120383. [Google Scholar] [PubMed] [CrossRef]
69. Toda K, Asakura T, Fukaya M, Entani E, Kawamura Y. Cellulose production by acetic acid-resistant Acetobacter xylinum. J Ferment Bioeng. 1997;84(3):228–31. doi:10.1016/s0922-338x(97)82059-4. [Google Scholar] [CrossRef]
70. Cheng Z, Yang R, Liu X, Liu X, Chen H. Green synthesis of bacterial cellulose via acetic acid pre-hydrolysis liquor of agricultural corn stalk used as carbon source. Bioresour Technol. 2017;234:8–14. doi:10.1016/j.biortech.2017.02.131. [Google Scholar] [PubMed] [CrossRef]
71. Goelzer FDE, Faria-Tischer PCS, Vitorino JC, Sierakowski MR, Tischer CA. Production and characterization of nanospheres of bacterial cellulose from Acetobacter xylinum from processed rice bark. Mater Sci Eng C. 2009;29(2):546–51. doi:10.1016/j.msec.2008.10.013. [Google Scholar] [CrossRef]
72. Yanti NA, Ahmad SW, Ahmad Nur Ramadhan LO, Jamili M, Walhidayah T, et al. Properties and application of edible modified bacterial cellulose film based sago liquid waste as food packaging. Polymers. 2021;13(20):3570. doi:10.3390/polym13203570. [Google Scholar] [PubMed] [CrossRef]
73. Tyagi N, Suresh S. Production of cellulose from sugarcane molasses using Gluconacetobacter intermedius SNT-1: optimization & characterization. J Clean Prod. 2016;112:71–80. doi:10.1016/j.jclepro.2015.07.054. [Google Scholar] [CrossRef]
74. Lin D, Lopez-Sanchez P, Li R, Li Z. Production of bacterial cellulose by Gluconacetobacter hansenii CGMCC, 3917 using only waste beer yeast as nutrient source. Bioresour Technol. 2014;151:113–9. doi:10.1016/j.biortech.2013.10.052. [Google Scholar] [PubMed] [CrossRef]
75. Akintunde MO, Adebayo-Tayo BC, Ishola MM, Zamani A, Horváth IS. Bacterial cellulose production from agricultural residues by two Komagataeibacter sp. Strains. Bioengineered. 2022;13(4):10010–25. doi:10.1080/21655979.2022.2062970. [Google Scholar] [PubMed] [CrossRef]
76. Liu Z, Wang Y, Guo S, Liu J, Zhu P. Preparation and characterization of bacterial cellulose synthesized by kombucha from vinegar residue. Int J Biol Macromol. 2024;258(Pt 1):128939. doi:10.1016/j.ijbiomac.2023.128939. [Google Scholar] [PubMed] [CrossRef]
77. Yilmaz M, Goksungur Y. Optimization of bacterial cellulose production from waste figs by Komagataeibacter xylinus. Fermentation. 2024;10(9):466. doi:10.3390/fermentation10090466. [Google Scholar] [CrossRef]
78. Ma X, Yuan H, Wang H, Yu H. Coproduction of bacterial cellulose and pear vinegar by fermentation of pear peel and pomace. Bioprocess Biosyst Eng. 2021;44(11):2231–44. doi:10.1007/s00449-021-02599-3. [Google Scholar] [PubMed] [CrossRef]
79. Tsouko E, Pilafidis S, Kourmentza K, Gomes HI, Sarris G, Koralli P, et al. A sustainable bioprocess to produce bacterial cellulose (BC) using waste streams from wine distilleries and the biodiesel industry: evaluation of BC for adsorption of phenolic compounds, dyes and metals. Biotechnol Biofuels Bioprod. 2024;17(1):40. doi:10.1186/s13068-024-02488-3. [Google Scholar] [PubMed] [CrossRef]
80. Tsouko E, Pilafidis S, Dimopoulou M, Kourmentza K, Sarris D. Bioconversion of underutilized brewing by-products into bacterial cellulose by a newly isolated Komagataeibacter rhaeticus strain: a preliminary evaluation of the bioprocess environmental impact. Bioresour Technol. 2023;387(4):129667. doi:10.1016/j.biortech.2023.129667. [Google Scholar] [PubMed] [CrossRef]
81. Efthymiou MN, Tsouko E, Pateraki C, Papagiannopoulos A, Tzamalis P, Pispas S, et al. Property evaluation of bacterial cellulose nanostructures produced from confectionery wastes. Biochem Eng J. 2022;186:108575. doi:10.1016/j.bej.2022.108575. [Google Scholar] [CrossRef]
82. Jayaprakash K, Osama A, Rajagopal R, Goyette B, Karthikeyan OP. Agriculture waste biomass repurposed into natural fibers: a circular bioeconomy perspective. Bioengineering. 2022;9(7):296. doi:10.3390/bioengineering9070296. [Google Scholar] [PubMed] [CrossRef]
83. Schiros TN, Antrobus R, Farías D, Chiu YT, Joseph CT, Esdaille S, et al. Microbial nanocellulose biotextiles for a circular materials economy. Env Sci Adv. 2022;1(3):276–84. doi:10.1039/d2va00050d. [Google Scholar] [PubMed] [CrossRef]
84. Stenton M, Kapsali V, Blackburn RS, Houghton JA. From clothing rations to fast fashion: utilising regenerated protein fibres to alleviate pressures on mass production. Energies. 2021;14(18):5654. doi:10.3390/en14185654. [Google Scholar] [CrossRef]
85. Springwise. Circular systems: sustainable innovation in textiles; 2018. [cited 2025 Oct 1]. Available from: https://www.circular-systems.com. [Google Scholar]
86. Gupta N, Mahur BK, Izrayeel AMD, Ahuja A, Rastogi VK. Biomass conversion of agricultural waste residues for different applications: a comprehensive review. Environ Sci Pollut Res. 2022;29(49):73622–47. doi:10.1007/s11356-022-22802-6. [Google Scholar] [PubMed] [CrossRef]
87. Patel BY, Patel HK. Retting of banana pseudostem fibre using Bacillus strains to get excellent mechanical properties as biomaterial in textile & fiber industry. Heliyon. 2022;8(9):e10652. doi:10.1016/j.heliyon.2022.e10652. [Google Scholar] [PubMed] [CrossRef]
88. Goveas LC, Krishna A, Salian A, Menezes J, Alva M, et al. Isolation and characterization of bacteria from refinery effluent for degradation of petroleum crude oil in seawater. J Pure Appl Microbiol. 2020;14(1):473–84. doi:10.22207/jpam.14.1.49. [Google Scholar] [CrossRef]
89. Bilal M, Ji L, Xu Y, Xu S, Lin Y, Iqbal HMN, et al. Bioprospecting Kluyveromyces marxianus as a robust host for industrial biotechnology. Front Bioeng Biotechnol. 2022;10:851768. doi:10.3389/fbioe.2022.851768. [Google Scholar] [PubMed] [CrossRef]
90. Pfromm PH, Amanor-Boadu V, Nelson R, Vadlani P, Madl R. Bio-butanol vs. bio-ethanol: a technical and economic assessment for corn and switchgrass fermented by yeast or Clostridium acetobutylicum. Biomass Bioenergy. 2010;34(4):515–24. doi:10.1016/j.biombioe.2009.12.017. [Google Scholar] [CrossRef]
91. Chandrasekhar K, Lee YJ, Lee DW. Biohydrogen production: strategies to improve process efficiency through microbial routes. Int J Mol Sci. 2015;16(4):8266–93. doi:10.3390/ijms16048266. [Google Scholar] [PubMed] [CrossRef]
92. Ahmed SF, Kabir M, Mehjabin A, Oishi FTZ, Ahmed S, Mannan S, et al. Waste biorefinery to produce renewable energy: bioconversion process and circular bioeconomy. Energy Rep. 2023;10:3073–91. doi:10.1016/j.egyr.2023.09.137. [Google Scholar] [CrossRef]
93. Sundaram T, Govindarajan RK, Vinayagam S, Krishnan V, Nagarajan S, Gnanasekaran GR, et al. Advancements in biosurfactant production using agro-industrial waste for industrial and environmental applications. Front Microbiol. 2024;15:1357302. doi:10.3389/fmicb.2024.1357302. [Google Scholar] [PubMed] [CrossRef]
94. Shaimerdenova U, Kaiyrmanova G, Lewandowska W, Bartoszewicz M, Swiecicka I, Yernazarova A. Biosurfactant and biopolymer producing microorganisms from West Kazakhstan oilfield. Sci Rep. 2024;14(1):2294. doi:10.1038/s41598-024-52906-7. [Google Scholar] [PubMed] [CrossRef]
95. Gaur VK, Sharma P, Sirohi R, Varjani S, Taherzadeh MJ, Chang J, et al. Production of biosurfactants from agro-industrial waste and waste cooking oil in a circular bioeconomy: an overview. Bioresour Technol. 2022;343:126059. doi:10.1016/j.biortech.2021.126059. [Google Scholar] [PubMed] [CrossRef]
96. Glatter O, Salentinig S. Inverting structures: from micelles via emulsions to internally self-assembled water and oil continuous nanocarriers. Curr Opin Colloid Interface Sci. 2020;49:82–93. doi:10.1016/j.cocis.2020.05.003. [Google Scholar] [CrossRef]
97. Domínguez Rivera Á, Martínez Urbina MÁ, López y López VE. Advances on research in the use of agro-industrial waste in biosurfactant production. World J Microbiol Biotechnol. 2019;35(10):155. doi:10.1007/s11274-019-2729-3. [Google Scholar] [PubMed] [CrossRef]
98. Sankhyan S, Kumar P, Pandit S, Kumar S, Ranjan N, Ray S. Biological machinery for the production of biosurfactant and their potential applications. Microbiol Res. 2024;285(5):127765. doi:10.1016/j.micres.2024.127765. [Google Scholar] [PubMed] [CrossRef]
99. Rusu AV, Trif M, Rocha JM. Microbial secondary metabolites via fermentation approaches for dietary supplementation formulations. Molecules. 2023;28(16):6020. doi:10.3390/molecules28166020. [Google Scholar] [PubMed] [CrossRef]
100. Visco A, Scolaro C, Facchin M, Brahimi S, Belhamdi H, Gatto V, et al. Agri-food wastes for bioplastics: european prospective on possible applications in their second life for a circular economy. Polymers. 2022;14(13):2752. doi:10.3390/polym14132752. [Google Scholar] [PubMed] [CrossRef]
101. Longo A, Fanelli F, Villano M, Montemurro M, Rizzello CG. Bioplastic production from agri-food waste through the use of Haloferax mediterranei: a comprehensive initial overview. Microorganisms. 2024;12(6):1038. doi:10.3390/microorganisms12061038. [Google Scholar] [PubMed] [CrossRef]
102. Niveditha SS, Ranjana S, Sangeetha M. Preparation of bioplastic from agro waste. J Emerg Technol Innov Res. 2022;9(7):a5–22. [cited 2025 Oct 1]. Available from: http://www.jetir.org/papers/JETIR2207002.pdf. [Google Scholar]
103. Saravanan K, Umesh M, Kathirvel P. Microbial polyhydroxyalkanoates (PHAsa review on biosynthesis, properties, fermentation strategies and its prospective applications for sustainable future. J Polym Environ. 2022;30(12):4903–35. doi:10.1007/s10924-022-02562-7. [Google Scholar] [CrossRef]
104. Oonkhanond B, Jonglertjunya W, Srimarut N, Bunpachart P, Tantinukul S, Nasongkla N, et al. Lactic acid production from sugarcane bagasse by an integrated system of lignocellulose fractionation, saccharification, fermentation, and ex-situ nanofiltration. J Environ Chem Eng. 2017;5(3):2533–41. doi:10.1016/j.jece.2017.05.004. [Google Scholar] [CrossRef]
105. Sawant SS, Bhapkar SR, Song J, Seo HJ. Sustainable bioplastics: harnessing pear agro-industrial waste for polyhydroxyalkanoates production: a review. Int J Environ Sci Technol. 2024;21(10):7341–52. doi:10.1007/s13762-024-05493-z. [Google Scholar] [CrossRef]
106. Margalho LP, Jorge GP, Noleto DAP, Silva CE, Abreu JS, Piran MVF, et al. Biopreservation and probiotic potential of a large set of lactic acid bacteria isolated from Brazilian artisanal cheeses: from screening to in product approach. Microbiol Res. 2021;242:126622. doi:10.1016/j.micres.2020.126622. [Google Scholar] [PubMed] [CrossRef]
107. Chan JX, Wong JF, Hassan A, Zakaria Z. Bioplastics from agricultural waste. In: Biopolymers and biocomposites from agro-waste for packaging applications. Amsterdam, The Netherlands: Elsevier; 2021. p. 141–69. doi:10.1016/b978-0-12-819953-4.00005-7. [Google Scholar] [CrossRef]
108. Vigneswari S, Noor MSM, Amelia TSM, Balakrishnan K, Adnan A, Bhubalan K, et al. Recent advances in the biosynthesis of polyhydroxyalkanoates from lignocellulosic feedstocks. Life. 2021;11(8):807. doi:10.3390/life11080807. [Google Scholar] [PubMed] [CrossRef]
109. Aliotta L, Seggiani M, Lazzeri A, Gigante V, Cinelli P. A brief review of poly (butylene succinate) (PBS) and its main copolymers: synthesis, blends, composites, biodegradability, and applications. Polymers. 2022;14(4):844. doi:10.3390/polym14040844. [Google Scholar] [PubMed] [CrossRef]
110. Atiwesh G, Mikhael A, Parrish CC, Banoub J, Le TT. Environmental impact of bioplastic use: a review. Heliyon. 2021;7(9):e07918. doi:10.1016/j.heliyon.2021.e07918. [Google Scholar] [PubMed] [CrossRef]
111. Solaiman DKY, Ashby RD, Zerkowski JA, Foglia TA. Simplified soy molasses-based medium for reduced-cost production of sophorolipids by Candida bombicola. Biotechnol Lett. 2007;29(9):1341–7. doi:10.1007/s10529-007-9407-5. [Google Scholar] [PubMed] [CrossRef]
112. Ribeiro BG, Santos MDM, Silva IA, Meira HM, Medeiros AOD, Guerra JCM, et al. Study of the biosurfactant production by saccharomyces cerevisiae Urm 6670 using agroindustrial waste. Chem Eng Trans. 2020;79:61–6. [Google Scholar]
113. Nasir MS, Mohd Yahya AR, Md Noh NA. Agro-industrial wastes as potential substrates for rhamnolipid production by Pseudomonas aeruginosa USM-AR2. Trop Life Sci Res. 2024;35(1):33–47. doi:10.21315/tlsr2024.35.1.3. [Google Scholar] [PubMed] [CrossRef]
114. Andreolli M, Villanova V, Zanzoni S, D’Onofrio M, Vallini G, Secchi N, et al. Characterization of trehalolipid biosurfactant produced by the novel marine strain Rhodococcus sp. SP1d and its potential for environmental applications. Microb Cell Fact. 2023;22(1):126. doi:10.1186/s12934-023-02128-9. [Google Scholar] [PubMed] [CrossRef]
115. Vieira IMM, Santos BLP, Silva LS, Ramos LC, de Souza RR, Ruzene DS, et al. Potential of pineapple peel in the alternative composition of culture media for biosurfactant production. Environ Sci Pollut Res Int. 2021;28(48):68957–71. doi:10.1007/s11356-021-15393-1. [Google Scholar] [PubMed] [CrossRef]
116. Umar A, Zafar A, Wali H, Siddique MP, Qazi MA, Naeem AH, et al. Low-cost production and application of lipopeptide for bioremediation and plant growth by Bacillus subtilis SNW3. AMB Express. 2021;11(1):165. doi:10.1186/s13568-021-01327-0. [Google Scholar] [PubMed] [CrossRef]
117. Rastogi S, Tiwari S, Ratna S, Kumar R. Utilization of agro-industrial waste for biosurfactant production under submerged fermentation and its synergistic application in biosorption of Pb2+. Bioresour Technol Rep. 2021;15(2):100706. doi:10.1016/j.biteb.2021.100706. [Google Scholar] [CrossRef]
118. Nogueira IB, Rodríguez DM, da Silva Andradade RF, Lins AB, Bione AP, da Silva IGS, et al. Bioconversion of agroindustrial waste in the production of bioemulsifier by Stenotrophomonas maltophilia UCP, 1601 and application in bioremediation process. Int J Chem Eng. 2020;2020(1):9434059. doi:10.1155/2020/9434059. [Google Scholar] [CrossRef]
119. Soni SK, Soni R. Policy and regulatory frameworks for green biorefinery: driving sustainable waste valorization. In: Green biorefinery solutions. Singapore: Springer Nature Singapore; 2025. p. 315–49. doi:10.1007/978-981-96-9013-8_10. [Google Scholar] [CrossRef]
120. Chemier JA, Fowler ZL, Koffas MAG, Leonard E. Trends in microbial synthesis of natural products and biofuels. Adv Enzymol Relat Areas Mol Biol. 2009;76:151–217. doi:10.1002/9780470392881.CH4. [Google Scholar] [PubMed] [CrossRef]
121. Yadav AN, Suyal DC, Kour D, Rajput VD, Rastegari AA, Singh J. Bioremediation and waste management for environmental sustainability. J App Biol Biotech. 2022:1–5. doi:10.7324/jabb.2022.10s201. [Google Scholar] [CrossRef]
122. Yrjälä K, Ramakrishnan M, Salo E. Agricultural waste streams as resource in circular economy for biochar production towards carbon neutrality. Curr Opin Environ Sci Health. 2022;26:100339. doi:10.1016/j.coesh.2022.100339. [Google Scholar] [CrossRef]
123. Gaur S, Kaur M, Kalra R, Rene ER, Goel M. Application of microbial resources in biorefineries: current trend and future prospects. Heliyon. 2024;10(8):e28615. doi:10.1016/j.heliyon.2024.e28615. [Google Scholar] [PubMed] [CrossRef]
124. Calvo-Flores FG, Martin-Martinez FJ. Biorefineries: achievements and challenges for a bio-based economy. Front Chem. 2022;10:973417. doi:10.3389/fchem.2022.973417. [Google Scholar] [PubMed] [CrossRef]
125. Makepa DC, Chihobo CH. Barriers to commercial deployment of biorefineries: a multi-faceted review of obstacles across the innovation chain. Heliyon. 2024;10(12):e32649. doi:10.1016/j.heliyon.2024.e32649. [Google Scholar] [PubMed] [CrossRef]
126. Pranaw K, Drewniak L, Nain L, Singh S. Editorial: waste to wealth: a sustainable circular bioeconomy approach. Front Bioeng Biotechnol. 2022;10:1010811. doi:10.3389/fbioe.2022.1010811. [Google Scholar] [PubMed] [CrossRef]
127. Decker SR, Brunecky R, Yarbrough JM, Subramanian V. Perspectives on biorefineries in microbial production of fuels and chemicals. Front Ind Microbiol. 2023;1:1202269. doi:10.3389/finmi.2023.1202269. [Google Scholar] [CrossRef]
128. de Melo E Silva J, da Silva Martins LH, Moreira DKT, do Prado Silva L, de Paula Menezes Barbosa P, Komesu A, et al. Microbial lipid based biorefinery concepts: a review of status and prospects. Foods. 2023;12(10):2074. doi:10.3390/foods12102074. [Google Scholar] [PubMed] [CrossRef]
129. Goyal D, Mishra S, Dantu PK. Microbial technologies for biorefineries: current research and future applications. In: Biofuels production—sustainability and advances in microbial bioresources. Cham, Switzerland: Springer International Publishing; 2020. p. 229–50. doi:10.1007/978-3-030-53933-7_11. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF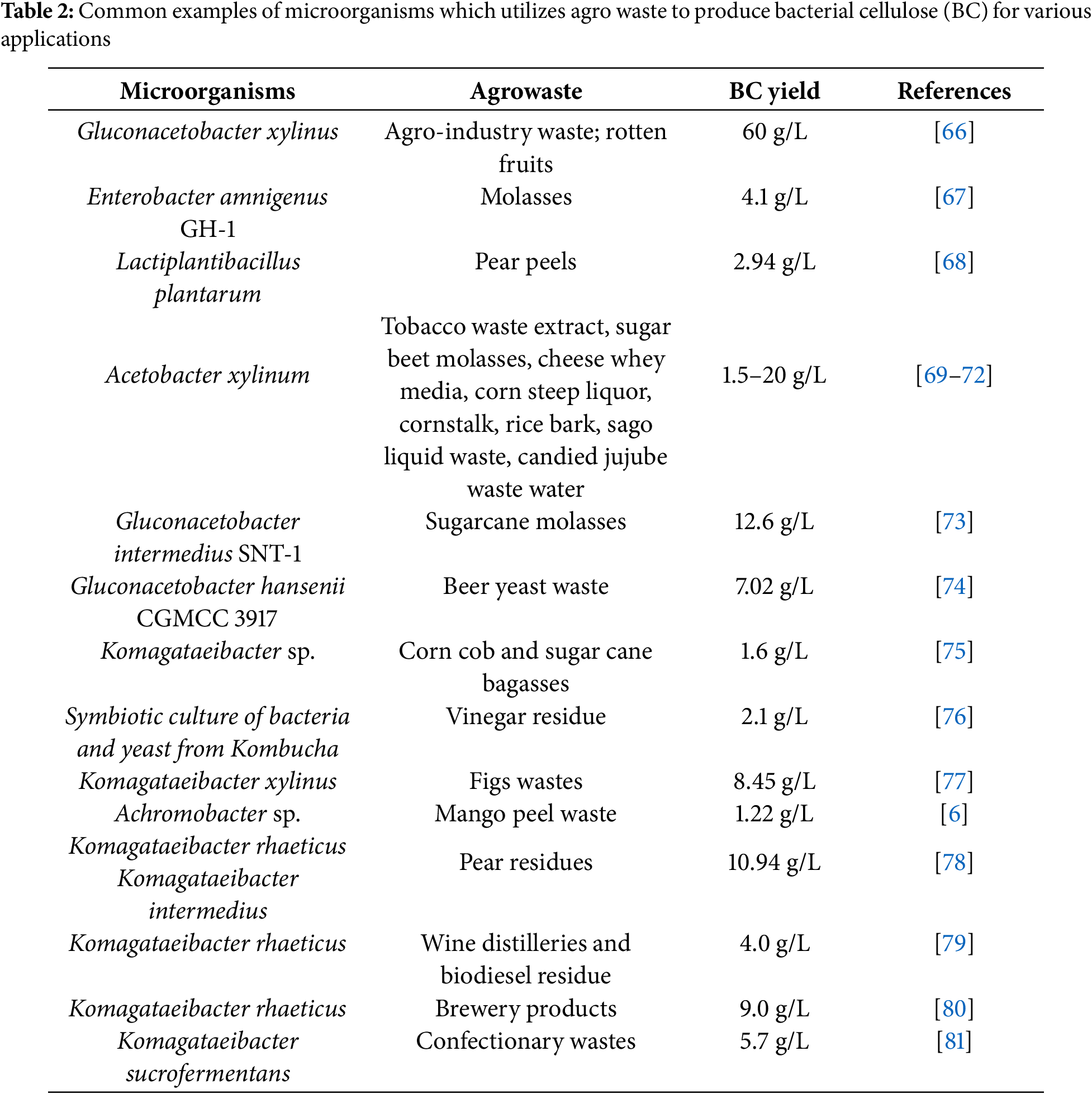
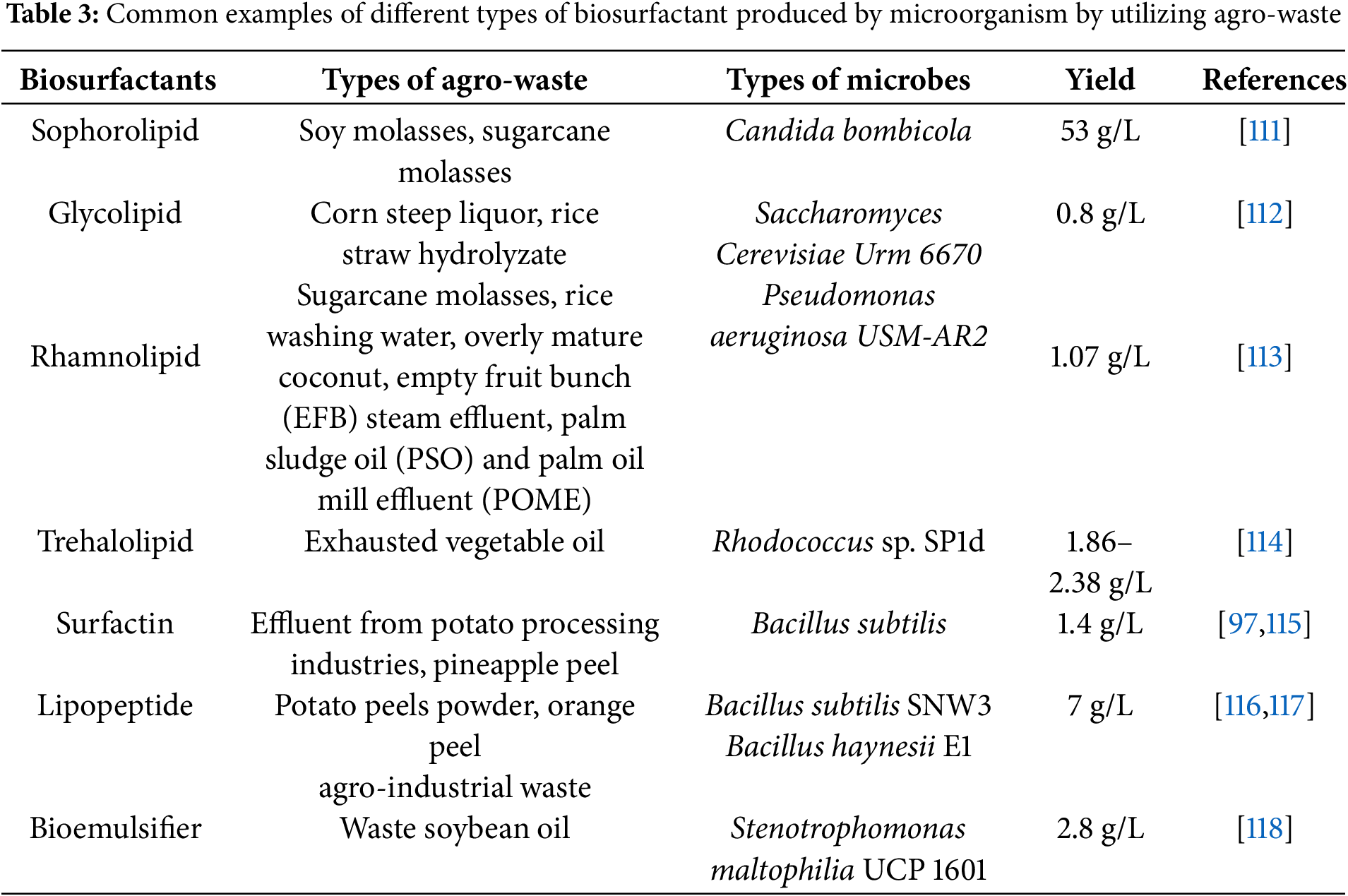
 Downloads
Downloads
 Citation Tools
Citation Tools