 Open Access
Open Access
ARTICLE
Tailoring the Properties of Balsa and Pulai Wood Sponge through Dual-Stages Delignification as a Potential Bio-Based Adsorbent
1 Study Program of Forest Products Science and Technology, Department of Forest Products, Faculty of Forestry and Environment, Post-Graduate School (SPs) IPB University, Bogor, Indonesia
2 Forest Products Department, Faculty of Forestry and Environment, IPB University, Bogor, Indonesia
3 Research Center for Biomass and Bioproducts, National Research and Innovation Agency (BRIN), Serpong, South Tangerang, Indonesia
4 Faculty of Forestry, Universitas Tanjungpura, Pontianak, West Kalimantan, Indonesia
* Corresponding Authors: Imam Wahyudi. Email: ; Sarah Augustina. Email:
(This article belongs to the Special Issue: Advances in Eco-friendly Wood-Based Composites: Design, Manufacturing, Properties and Applications – Ⅱ)
Journal of Renewable Materials 2026, 14(3), 7 https://doi.org/10.32604/jrm.2026.02025-0161
Received 14 August 2025; Accepted 27 January 2026; Issue published 25 March 2026
Abstract
The increasing need for efficient, sustainable, and environmentally friendly adsorbent materials has driven interest in bio-based alternatives. Conventional silica-based adsorbents, while effective, are often brittle and energy-intensive to produce. In contrast, wood offers a renewable and low-energy option with natural porosity suitable for adsorption. This study investigated the fabrication of wood sponge from tropical balsa (Ochroma bicolor) and pulai (Alstonia scholaris) wood through a dual-stage delignification process as a novel bio-based adsorbent. The process involved alkaline treatment using sodium sulfite (Na2SO3) and sodium hydroxide (NaOH) at 100°C for 8, 9, and 10 h, followed by bleaching process using hydrogen peroxide (H2O2) at 100°C for 1, 2, and 3 h. The treated samples were then rinsed to neutral pH, frozen at −20°C for 24 h, and freeze-dried at −50°C for 48 h. The results revealed a notable reduction in density and specific gravity, accompanied by increased weight loss and pore diameter size with prolonged delignification process. Optical changes showed increased translucency and layered structures, particularly in balsa wood. FTIR analysis confirmed a reduction in lignin and hemicellulose content, validating the chemical modification within the treated samples. The resulting wood sponges exhibited good porosity and adsorption capacity, ranging from 1.3 to 5.7 g/g. The optimal treatment—10 h of alkaline delignification followed by 3 h of H2O2 bleaching—demonstrated the highest performance, highlighting the potential of tropical wood species as efficient, biodegradable, and eco-friendly adsorbent materials.Graphic Abstract

Keywords
The growing demand for efficient, sustainable, and environmentally benign adsorbent materials has spurred significant interest in the development of bio-based alternatives to replace conventional adsorbents. Bio-based adsorbents are particularly advantageous due to their biodegradability and eco-friendly manufacturing processes, beyond cost-effectiveness, and do not cause any secondary effects from their by-products [1]. According to previous research, bio-based adsorbents come from different materials such as agricultural waste, including crop and forest residue [2], animal waste materials [3], fish scales [4], crab shells [5], chicken feathers [6], as well as algae [7]. These materials have been utilized particularly for the removal of pharmaceuticals and dyes [8], water pollutants [9], as well as carbon capture and storage purposes [10]. The performance of such bio-based adsorbents is influenced by various factors, including porosity, adsorption/desorption kinetics, holding capacity, operational temperature range, regeneration capability, and resistance to degradation [11–13]. Among the strategies for fabricating bio-based adsorbents, the bottom-up method offers advantages in precise structural design and functional customization for targeted applications [14]. However, bottom-up approaches are often associated with drawbacks such as extended reaction time, safety concerns during synthesis, and higher production cost [15,16]. In contrast, the top-down approach has gained more traction for large-scale production due to its simplicity and lower economic cost. In particular, wood-based materials employed in the top-down method offer present significant advantages. Wood possesses a natural hierarchical porous structure that facilitates adsorption without requiring extensive chemical or structural modifications. Additionally, its abundance and renewability make wood a highly suitable and cost-effective raw material for bio-adsorbent development [17,18].
Wood is a complex three-dimensional composite composed primarily of biopolymers, cellulose, hemicellulose, lignin, and various extractives and inorganic materials. Its natural porosity makes wood an excellent candidate for the development of lignocellulosic-based adsorbents, which are innovative materials designed to enhance the functional value of renewable resources. Wood-based adsorbents can be engineered into advanced porous structures such as wood sponges and aerogels. These forms offer improved surface functionality and porosity, thus significantly enhancing their adsorption capabilities [19]. In particular, wood sponges are gaining considerable attention as robust alternatives due to their superior mechanical strength, resilience, and reusability. They have been successfully applied in various fields, such as oil–water separation, heavy metal ion detection [20], and energy harvesting systems [21]. Each application requires a different fabrication method, tailored to achieve the desired microstructure and performance. Specific methods are selected based on the target properties to maximize the adsorption efficiency, improve mechanical durability, or produce unique porous architectures suitable for multifunctional applications [22]. For instance, sengon (Falcataria moluccana) wood has been utilized to fabricate wood-based sponges with the capability to adsorb oil and isopropyl alcohol as model liquid pollutants [23]. Other studies have developed compressible and reusable wood sponges suitable for multiple adsorption–desorption cycles [24]. Given the importance of wood characteristics in adsorption performance, optimizing the fabrication process for wood-based sponges with high porosity remains a critical research priority. Such optimization will further enhance their efficiency and broaden their applicability in environmental and industrial sectors.
Generally, the fabrication of wood sponges with optimal porosity largely depends on the lignin removal process. Lignin is an amorphous aromatic polymer that works as a natural glue, binding hemicellulose and cellulose together to give plant cell walls their strength and rigidity [25]. It is a heterogeneous and aromatic macromolecule polymer, which is mainly comprised of three kinds of component units: syringyl (S), guaiacyl (G), and p-hydroxyphenyl (H) units [26]. These units are interconnected, forming strong bonds that contribute to the rigidity of the wood structure. Therefore, effective lignin removal is essential to facilitate the development of a highly porous material, particularly wood sponge [25]. Previous research on the lignin removal process by Horikawa [25] employed single-step delignification using a strong oxidative mixture, which simplified wood sponge preparation while preserving the desirable structure; however, the resulting porosity and pore size were lower compared to those obtained using the dual-stages method [23]. Dual-stages delignification method, using chemical alkaline method (CAM) enables more effective lignin and hemicellulose elimination, thus improving its porosity, mechanical stability, and functional characteristics [27].
Furthermore, exploring combinations of NaOH/Na2SO3 with H2O2, along with variations in treatment duration, offers significant potential for optimizing the delignification process. This approach could facilitate the development of high-performance bio-based adsorbents with superior adsorption capabilities. Li et al. [28] employed a combined treatment using NaOH and Na2SO3, followed by hot pressing, to produce transparent wood with optimized porosity and mechanical strength. The mechanism during alkali treatment (NaOH/Na2SO3) primarily involves nucleophilic reactions between the alkaline solution and the wood cell structure due to the deprotonation of the phenolic OH-group and the cleavage of lignin-carbohydrate bonds and β-O-4 linkage, leading to the massive destruction and fragmentation of side chains of lignin macromolecules, enhancing their hydrophilicity, and thereby effectively removing lignin from or in the wood structure [18,29]. The addition of H2O2 can effectively promote the delignification process by inducing oxidation, hydrolysis, and degradation of lignin through aromatic ring cleavage and the disruption of unsaturated bonds in its side chains [29]. This procedure can significantly increase the wood porosity by creating new pores and enlarging existing ones, by enhancing the void volume, and by widening the gaps between cell walls. Vitas et al. [30] reported that delignification could increase the amounts of new voids, change the pore distribution and relative pore volume into larger radii (>30–100 μm) due to the disassembly of the wood cells, particularly vessel and middle lamella. In addition, the usage of NaOH could transform the cellulose I into cellulose II structures and cross-link the cellulose to produce an aerogel with a porous structure by generating nano-scale voids and enlarging existing pores [31].
Previous findings underscore the importance of selecting appropriate wood species and fine-tuning the process parameters to achieve the desired structural and functional outcomes. As tropical wood species native to Indonesia, both balsa and pulai exhibit promising characteristics as raw materials for the fabrication of wood sponges. Balsa wood is often chosen due to its low density, large pore diameter, and excellent pore distribution, which make it highly suitable for sponge-like structures. Although less extensively studied, pulai wood is emerging as a promising alternative. Despite its relatively low dimensional stability and mechanical strength, it offers lightweight and flexible fibers, which are advantageous for non-timber applications such as bio-based sponges or adsorption [32]. Therefore, this study investigated the fabrication of wood sponges from balsa and pulai woods using a dual-stages delignification method. This research aims to provide comprehensive insights into the physical properties (density, specific gravity, weight loss, and color changes), anatomical structure (porosity, pore diameter, absorption capacity), and chemical characteristics (lignin content and functional group changes) of these materials. Given that the application of wood sponges as bio-based adsorbents remains a developing and relatively novel research area, further investigations into suitable wood species and optimal treatment durations are essential to enhance fabrication efficiency and overall material performance.
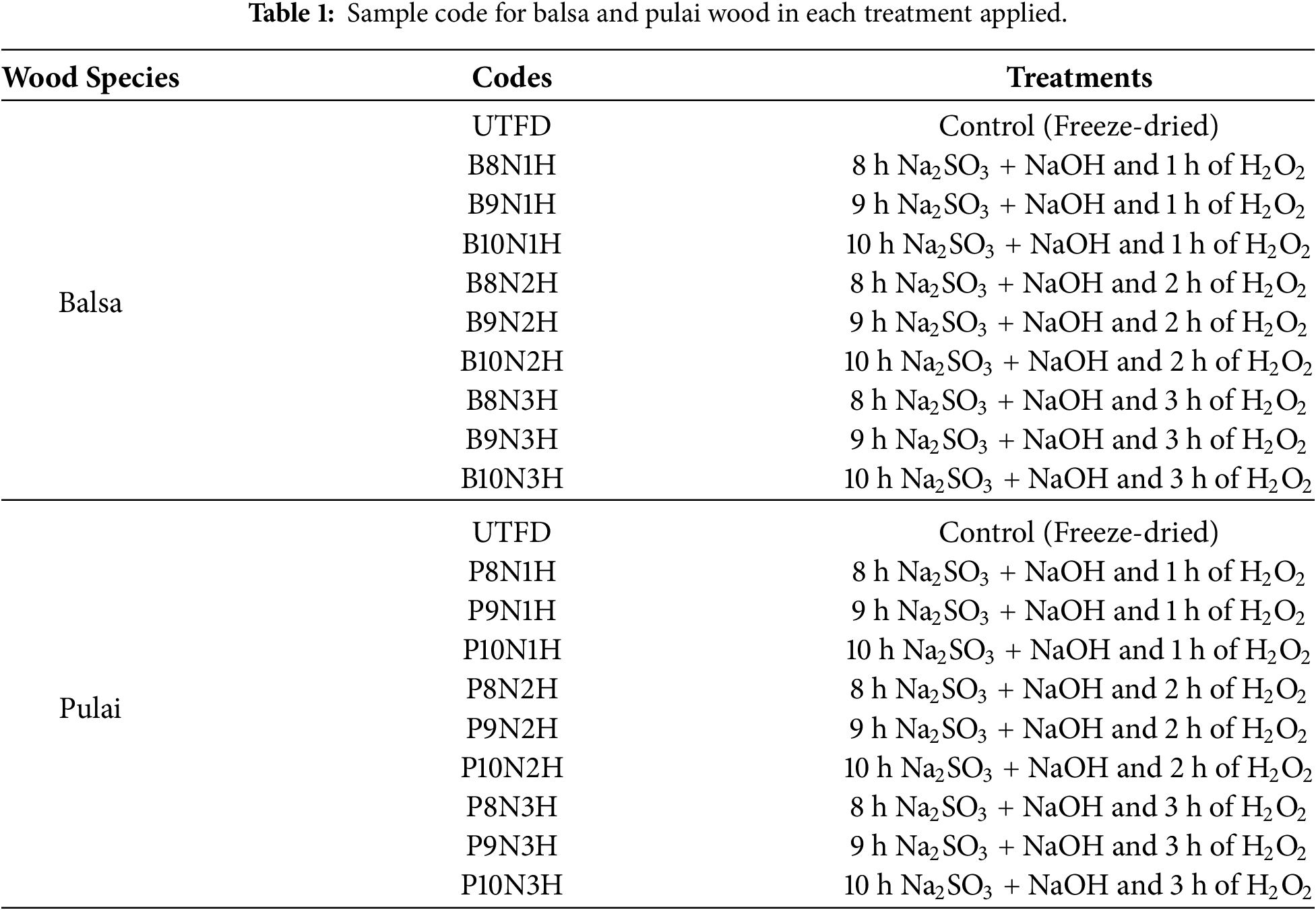
Balsa (Ochroma bicolor) and pulai (Alstonia scholaris) wood around 7 years old, sourced from a community forest area near Dramaga subdistrict in Bogor, West Java, were taken from defect-free middle trunks, with a diameter of approximately 18 cm and a length of ±100 cm. The logs were cross-cut into box-shaped specimens measuring of 2 cm × 2 cm × 1 cm3 (length × width × thickness, based on cross-section area), with 10 samples per treatment. The wood samples were oven-dried at 103 ± 2°C for 24 h before chemical treatment. For dual-stages delignification, sodium sulfite (Na2SO3, >98%) and sodium hydroxide (NaOH, >98%) were used, followed by treatment with hydrogen peroxide (H2O2, 30%). These chemicals were procured from Sigma-Aldrich and verified suppliers via e-commerce platforms. For adsorption performance analysis, isopropyl alcohol (C3H7OH) and commercial engine oil (AHM Oil MPX2) were selected as model liquid pollutants. The experimental setup included standard laboratory equipment such as beakers, containers, magnetic stirrers with hot plates, thermometer, ovens, refrigeration units, freeze-dry machines, digital calipers, and analytical balances.
2.2.1 Wood Sponge Manufacture via Dual-Stages Delignification
The delignification consisted of dual-stages processes through combined chemical treatments. Before delignification, a solution was prepared by dissolving sodium sulfite and sodium hydroxide separately in distilled water at a 1:1 ratio (wt%), by mixing 15 g of Na2SO3 with 750 mL of distilled water and 15 g of NaOH with 750 mL of distilled water. The first delignification stage involved immersing the oven-dried wood samples in the mixture solution, followed by heating at 100°C for 8, 9, and 10 h. Following this, the samples were then subjected to a second delignification stage by submerging the samples in the 6% hydrogen peroxide solution (100 mL of H2O2 and 400 mL of distilled water) at 100°C for 1, 2, and 3 h. After each delignification stage, the samples were thoroughly rinsed with distilled water to remove residual chemicals. The samples were post-treated via pre-freezing at −20°C for 24 h and then dried via freeze-drying at −50°C for 48 h to produce the final porous wood sponge structure. The flow chart of the process is presented in Fig. 1, while sample code for each treatment is presented in Table 1.
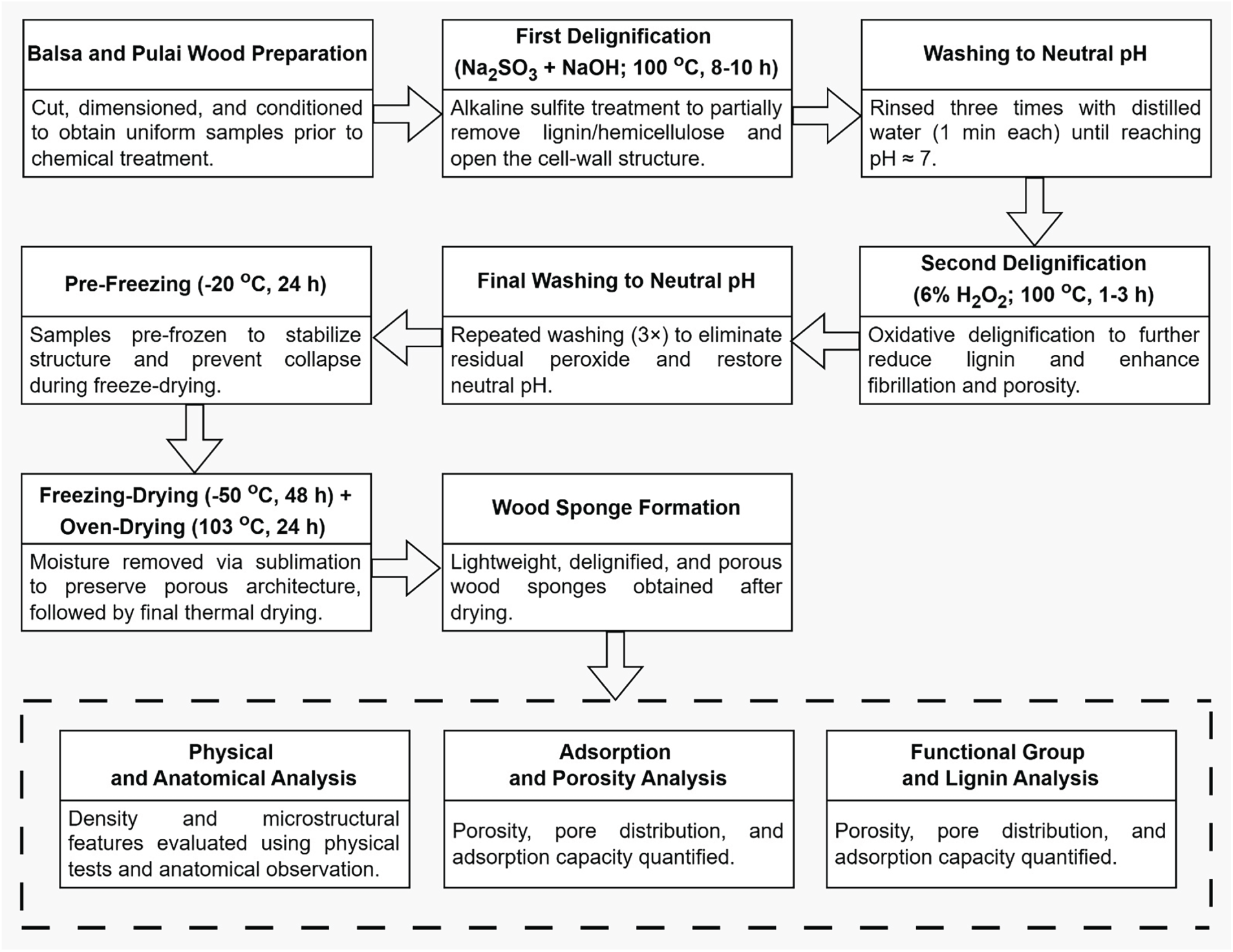
Figure 1: Flow chart of wood sponge manufacture via dual-stages delignification.
In this study, physical properties of wood such as density, specific gravity, and weight loss were tested. The samples were prepared with dimensions of 2 cm × 2 cm × 1 cm3 based on the modified British Standard 373–1957 [33]. The physical properties of the wood samples were calculated using the following formulas:
where, DA is the sample’s weight in air-dried condition (g); OD is samples weight in oven-dried condition (g); VA is samples volume in air-dried conditions (cm3); while ρ water = 1 g/cm3.
The color measurement involved the use of a colorimeter (Konica Minolta model), which was carefully calibrated to ensure accuracy. After calibration, the color values were systematically recorded. The lightness values and overall color changes, represented by ΔE, were then calculated using Eqs. (4) and (5). The classification of color changes degree is presented in Table 2, as adopted from Valverde and Moya [34]:
where, ΔE is the accumulation of color changes before and after treatment; ΔL is the difference between L (lightness) values before and after treatment; Δa is the difference between the a (red-green) value before and after treatment; while Δb is the difference between the b (yellow-blue) value before and after treatment.

The anatomical structure of balsa and pulai wood species was analyzed using a direct observation approach with a USB light microscope at 50× magnification. Cross-sectional samples were examined to observe the effects of the dual-stages delignification process on pore diameter and pore distribution. The pore diameter was determined from the lumen cell measurement. The images obtained were further analyzed using the Image-J software, focusing on parameters such as average pore diameter and pore distribution [35]. The pore distribution was calculated using the following equation:
2.2.5 The Changes in Chemical Components
Fourier Transform Infrared Spectroscopy (FTIR) analysis was conducted to examine changes in chemical components, particularly functional groups associated with lignin, cellulose, and hemicellulose, during the dual-stages delignification process. The successful removal of lignin can be verified by the disappearance or significant reduction of characteristic absorption peaks corresponding to lignin functional groups. In this study, FTIR analysis was carried out using a Universal Attenuated Total Reflectance (UATR) accessory with a diamond crystal plate (PerkinElmer Inc., Waltham, MA, USA). Powdered samples (2 mg, 30–40 mesh) in both untreated and wood sponge samples were analyzed in the spectral range of 400–4000 cm−1 at room temperature, with a resolution of 4 cm−1.
Quantification of lignin content is essential to assess the efficiency of the dual-stages delignification process in removing lignin from the wood structure. In this study, lignin content was measured on both untreated and wood sponge samples, using particles sieved to a size of 40–60 mesh. The procedure followed a modified version of the TAPPI T-203 standard method [36]. The lignin percentage was then calculated using the following formula:
where, OD is the oven-dried of crucible with samples (g); and DA is the air-dried of crusible without samples (g).
Evaluation of solvent adsorption capability in both untreated and wood sponge samples was observed by immersing the samples within 10 mL of isopropyl alcohol and engine oil at room temperature for 24 h. The sample’s weight was measured before and after the immersion process. In addition, porosity analysis was observed by immersing the samples within organic solvent, i.e., isopropyl alcohol. The adsorption capability and porosity formulas were calculated using the following formulas [37]:
where, mt is the adsorbed sample weight (g); mo is samples weight before adsorption (g); M is the adsorption capability (g/g); P is the porosity of wood sponge (%); Pb is the bulk density (g/cm3); Ps is the value of wood and microfibril cellulose density based on previous studies which is 1.49 g/cm3.
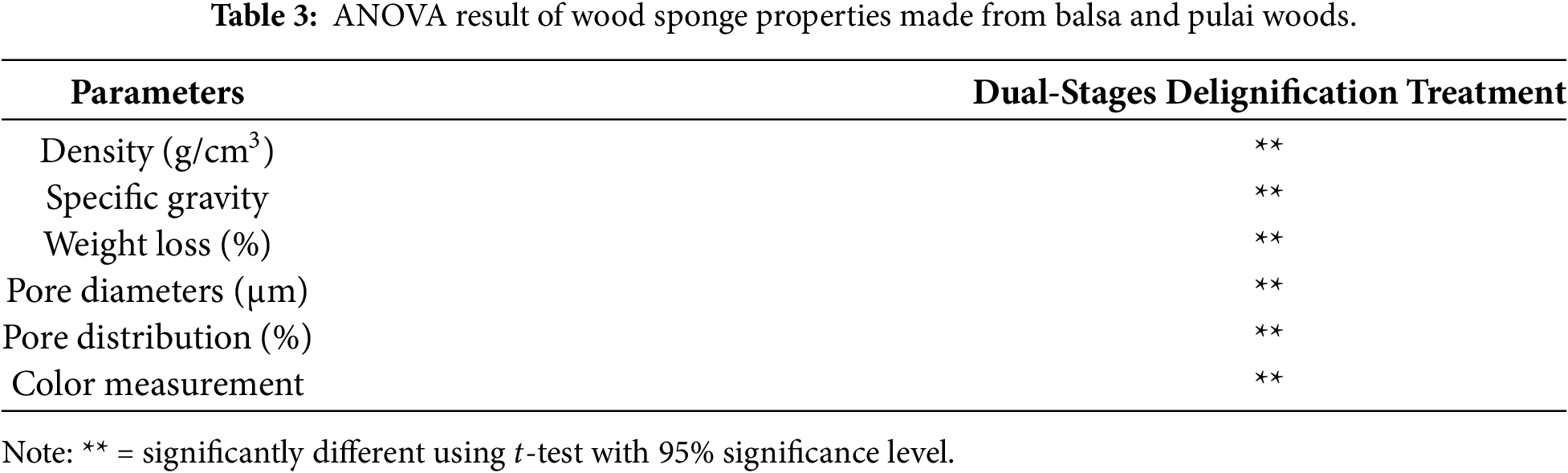
This research focused on the differences in wood species and treatment durations, specifically during dual-stages delignification treatment utilizing Na2SO3 + NaOH and H2O2. Analysis of variance (ANOVA) was conducted as the main method to determine the statistical significance of the differences among treatments. If the ANOVA result shows a significant difference at the 95% confidence level (p < 0.05), Duncan multiple range test was performed as a post-hoc analysis to compare pairwise differences of each treatment. All statistical analyses were performed using IBM SPSS Statistics 23.0 (SPSS Inc., Chicago, IL, USA).
3.1 Density, Specific Gravity, and Weight Loss
Wood density is defined as the ratio of wood mass to its volume at the same conditions. A higher density is generally associated with greater mechanical strength [35]. In general, it is highly dependent on the intrinsic characteristics of the species. Fig. 2 illustrates the density variation of balsa and pulai wood before and after the dual-stages delignification treatment. The comparison shows that both species experienced a decrease in density with increasing treatment duration [38]. This trend suggests that prolonged delignification reduces the structural components such as lignin, hemicellulose, and cellulose, leading to a decrease in density and potentially compromising the strength and stiffness of the resulting wood sponge [39]. This finding is supported by the ANOVA results (Table 3). The density of balsa wood sponge decreased significantly, with the reductions ranging from 22.4% to 47.2% (average decrease is 29.6%) (Fig. 2). In contrast, pulai wood sponge showed a slightly smaller reduction in density, ranging from 4.1% to 14.7% with an average of around 17.9%. Specifically, the density of balsa wood dropped from 0.13 to 0.7 g/cm3, while pulai wood decreased from 0.21 to 0.17 g/cm3. These two values are in accordance with previous studies [26].
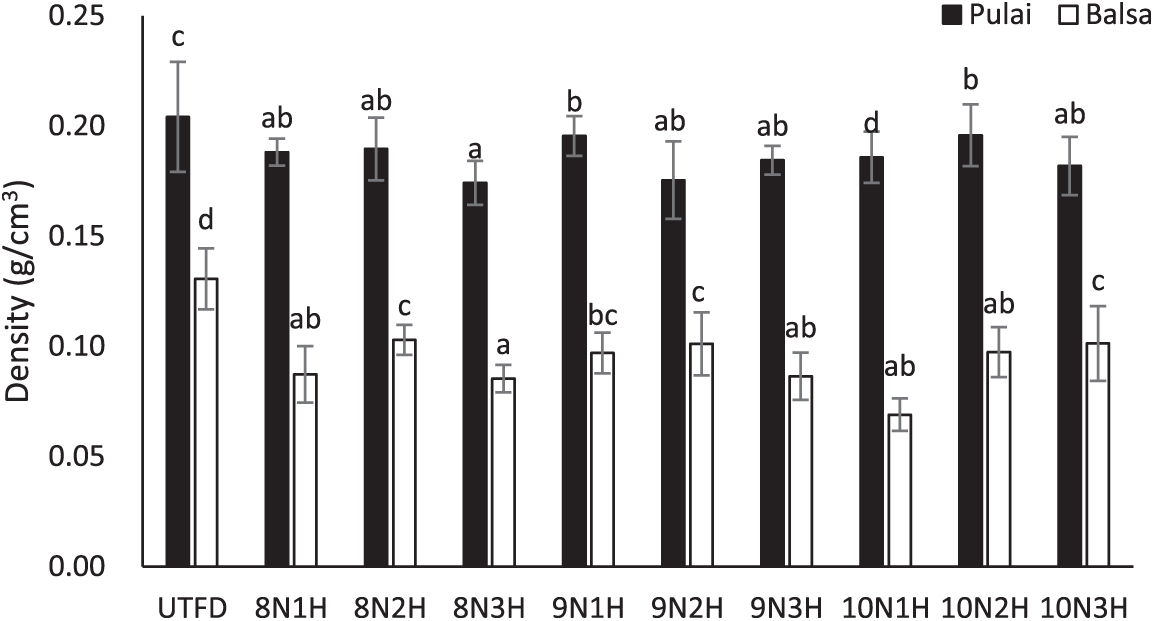
Figure 2: Density variation of balsa and pulai wood species in each treatment applied. Note: Letters above the graph show significant differences according to Duncan’s 5% significance level test.
A previous study also reported similar findings on delignified wood, where dual-stages delignification applied to poplar and Eurasian aspen resulted in a density reduction of approximately 10%–20% [40]. This reduction is primarily attributed to the effective removal of lignin during the process, which leads to a loosening of the wood’s structural matrix and a corresponding increase in porosity [41]. Although the removal of lignin softens the structure, it does not necessarily compromise its performance. According to Zhu et al. [42], the decrease in density can actually contribute to improved mechanical properties, as the delignification process facilitates the expansion and realignment of cellulose fibers. This expansion forms a more flexible and interconnected network, which enhances certain mechanical characteristics, particularly in low-density wood sponge applications.
Additional insights confirm the correlation between the reduction in wood density and lignin loss due to the dual-stages delignification process [42,43]. The application of NaOH and Na2SO3 in the alkaline phase of delignification significantly removes lignin and hemicellulose. Alkaline delignification enhances structural fragmentation by cleaving lignin–carbohydrate complexes and β-O-4 ether bonds [18], resulting in greater exposure of hydroxyl groups on the cellulose surface. In order to facilitate complete delignification, bleaching stage using hydrogen peroxide was applied. This phase promotes lignin degradation through a series of oxidative chain reactions involving peroxide ions (HOO−), superoxide (O2−) radicals, and hydroxyl (HO•) radicals. Among these, HOO− is the principal bond responsible for breaking the linkages between aromatic rings in lignin [44]. Specifically, HOO− ions cleave bonds between aromatic rings and side chains, disrupt unsaturated bonds in lignin side chains, and generate intermediates that are further oxidized into carbonyl and carboxyl compounds. Other reactive sites, such as HO ions and O2 radicals, also contribute to oxidation and degradation of lignin [29]. Therefore, H2O2 treatment is crucial for residual lignin collapse and achieving complete delignification of the wood cell wall structure [24]. Delignification using H2O2 leads to total collapse of residual lignin on the cell walls and complete delignification of wood; therefore, determining the concentration of H2O2 is an important process [45]. H2O2 delignification mainly operates through oxidative cleavage of lignin, generating reactive oxygen and attacking the lignin aromatic rings and side chains, leading to breaking of ether and carbon bonds, converting lignin into smaller water-soluble fragments [44]. Usually, acidic delignification itself could create density around 0.24–0.12 g/cm3, indicating a removal of lignin and hemicellulose, thus substantially reduces the wood density when no further processing is applied [46]. During dual-stages delignification, wood itself would react to the chemical, leading to covalent bond formation [47], crosslinking [44], or surface modification [24]. However, the efficiency of this process is highly dependent on parameters such as concentration, time, and temperature. Previous studies have shown that insufficient temperature results in high lignin residue [48], whereas optimal delignification efficiency is achieved by increasing temperature [49].
Another important indicator is specific gravity (SG), which reflects wood quality. A higher SG value often indicates a stronger and more durable wood material [50,51]. SG is defined as the ratio of wood density to water density and can also distinguish between hardwoods and softwoods in terms of mechanical performance [52]. Fig. 3 illustrates the changes in SG values for balsa and pulai wood in each treatment applied. A clear declining trend is observed, with balsa wood showing a decrease ranging from 0.07 to 0.12 (average: 0.09), while pulai wood declined from approximately 0.15 to 0.21 (average: 0.17). ANOVA results confirmed significant differences between all treatment groups and the control sample, with specific gravity decreasing as delignification time increased (Table 3). These findings align with previous results by Lin et al. [53] who observed the delignification process combined with freeze-drying can reduce both the moisture content and specific gravity of the wood sponge. Nonetheless, pulai wood exhibited a relatively lower reduction in SG compared to balsa wood, which may be attributed to its high extractive content [54]. These extractives can hinder the penetration of delignification chemicals, necessitating longer exposure durations or higher chemical concentrations to achieve comparable effects [55].
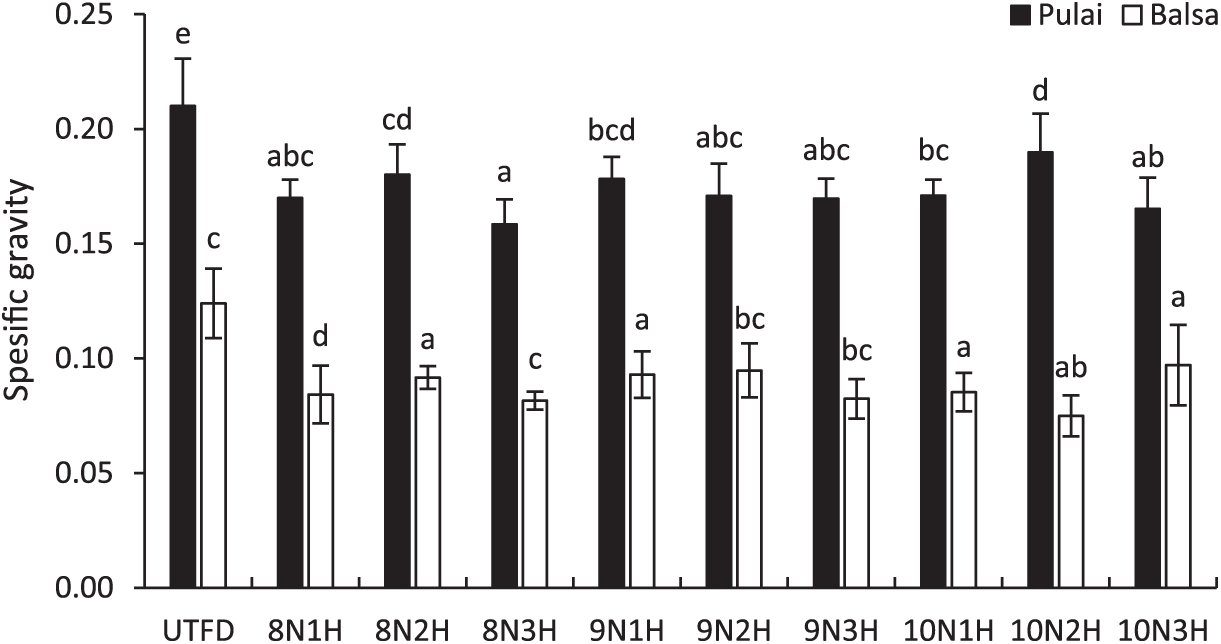
Figure 3: Specific gravity variation of balsa and pulai wood species in each treatment applied. Note: Letters above the graph show significant differences according to Duncan’s 5% significance level test.
Weight loss refers to the percentage reduction in mass after the delignification process relative to the original dry weight of the wood. This parameter serves as an indirect indicator of the extent of lignin and hemicellulose removal. A significant difference in weight loss was observed between balsa and pulai wood (Fig. 4). Balsa wood exhibited a higher weight loss, ranging from 15.30% to 36.47%, with an average of 24.64%. In contrast, pulai wood showed a more moderate reduction, ranging from 13.06% to 20.98%, with an average of 15.57%. Weight loss of balsa and pulai woods showed fluctuating results, because of the extreme impact of dual-stages delignification and freeze-dry process. Delignification process can significantly reduce partly hemicellulose and lignin content within the wood structure, thus subsequently increasing the weight loss of the wood sponge samples. Moreover, freeze-dry process can effectively remove the water and decrease the weight content of samples through sublimation, however, this reduction occurs only due to water loss, as freeze drying does not cause significant volume reduction or shrinkage of wood, unlike conventional drying methods [23]. These findings suggest that balsa wood is more susceptible to weight loss during the delignification process, potentially due to its lower density and looser fiber structure, which allows easier chemical penetration. Among the treatment combinations, the highest weight loss for balsa and pulai woods was recorded under the 10N3H treatment condition (Fig. 4).
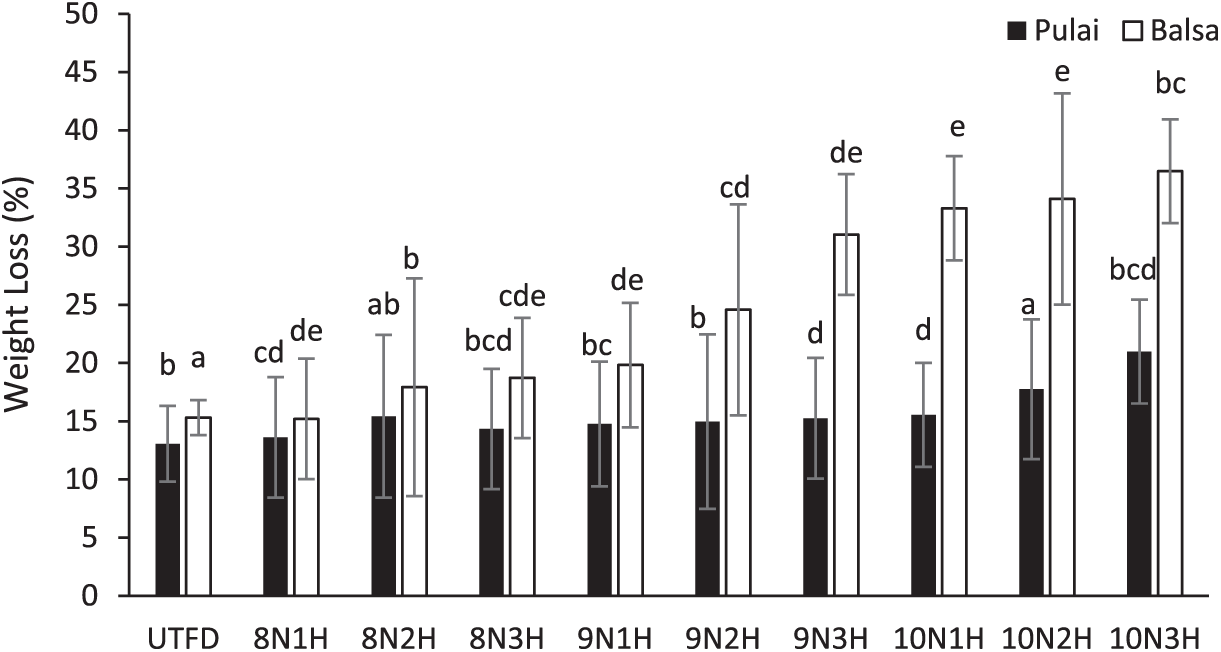
Figure 4: Weight loss variation of balsa and pulai wood species in each treatment applied. Note: Letters above the graph show significant differences according to Duncan’s 5% significance level test.
Since both balsa and pulai woods are classified as hardwood, their structural composition are dominated by fibers (ranging from 0.2–1.2 mm in length) with relatively small lumina, which primarily provide mechanical support [18]. Differences in fiber morphology and anatomical characteristics may contribute to the varying delignification responses between the two species. The lower weight loss observed in pulai wood suggests that it is more resistant to chemical degradation during the delignification process. The fibers in pulai wood may possess a unique arrangement or composition that enhances compactness and provides greater protection to lignin, particularly within the middle lamella and cell corners. These regions contain densely packed lignin that are less accessible to delignifying agents such as alkali or acid treatments, thereby limiting chemical penetration and resulting in lower weight loss [54]. Moreover, compared to balsa wood, pulai wood has greater density, higher resistance to brittleness, and a higher proportion of cellulose and hemicellulose relative to lignin, resulting in a less rigid structure with hollow lumina that enhances overall flexibility [55,56]. These factors collectively contribute to pulai wood’s increased resistance to chemical degradation during delignification [29,56].
This finding aligns with the results reported by Owoyemi et al. [57], who noted that higher-density wood species tend to exhibit lower weight loss following chemical treatments due to their denser and more compact structure. Supporting this conclusion, the anatomical structure of balsa wood is characterized by wider pore diameters and lower pore distribution, which facilitates deeper chemical penetration and, consequently, greater component loss. In contrast, pulai wood has narrower pores but with higher pore distribution, which contributes to greater stiffness and structural integrity [58,59]. These anatomical differences likely affect not only the degree of chemical accessibility but also the extent of lignin and hemicellulose removal during dual-stages delignification.
Wood treated through a dual-stages delignification process tends to retain its naturally porous structure while forming a hierarchical pore system (Fig. 5). This treatment significantly reduces lignin content and increases pore distribution by generating nanopores within the cell walls. The morphological differences between balsa and pulai wood after delignification can be identified through light microscopy, which allows observation of anatomical changes such as vessel expansion, wall thinning, and pore structure [60]. The removal of major structural compounds such as lignin and hemicellulose results in an increase in both the number and size of voids within the wood matrix. As lignin is removed, the overall void volume increases, which in turn leads to a rise in through-pore porosity and an observable widening of gaps between adjacent cell walls as these walls become thinner and more fragile [61]. Moreover, fibers and vessels often experience collapse due to the loss of intracellular support components, leading to reduced pore size uniformity and microstructural instability. In the preparation of wood sponges, the use of hydrogen peroxide not only promotes complete lignin degradation but also causes noticeable color changes in the wood structure, which is typically associated with the breakdown of chromophore lignin units [62]. Similar result by Li et al. [27] who reported that delignified wood cell structures exhibit a disordered and hollow morphology, particularly in the parenchyma tissues.

Figure 5: Morphological structure of balsa wood in each treatment applied; (a) Untreated/UTFD; (b) 8N1H; (c) 8N2H; (d) 8N3H; (e) 9N1H; (f) 9N2H; (g) 9N3H; (h) 10N1H; (i) 10N2H; (j) 10N3H.
According to Figs. 5 and 6, the comparison of balsa and pulai wood morphologies highlights several distinctive anatomical features, and reassures the location on the cell wall. Balsa wood is characterized by its solitary pores, a mixed vessel arrangement, and a clearly visible wood ray structure, despite the parenchyma cells being less observable [58]. It also exhibits simple perforation plates. In contrast, pulai wood displays a more complex pore structure, including vessel fusion, a clearer visibility of both rays and parenchyma, and the presence of diffuse apotracheal and ribbon-like parenchyma, with regularly aligned intercellular spacing [59]. These observations support the idea that dual-stages delignification not only removes lignin but also significantly alters the microstructure, leading to the formation of nano- and mesopores. According to previous findings, the removal of lignin facilitates the formation of nano-sized voids, which in turn contribute to the development of mesoporous networks within the wood matrix [61]. Consistent with this, Li et al. [27] reported that both microscale and nanoscale voids emerge within the cell walls after lignin removal, further enhancing the porous architecture of delignified wood.
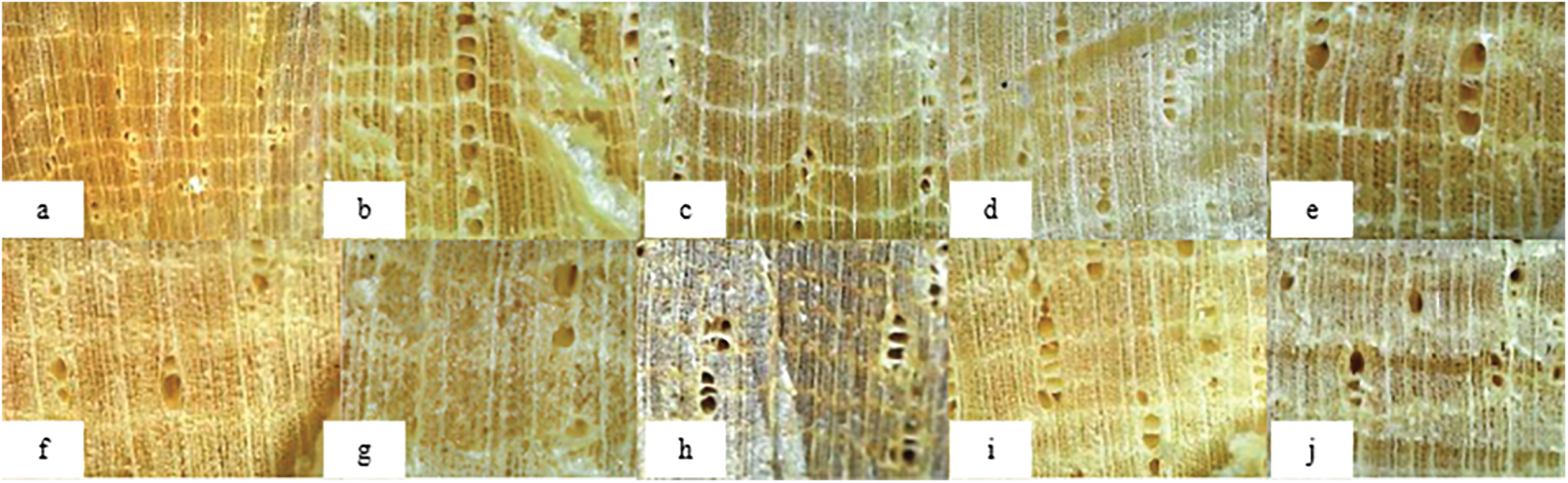
Figure 6: Morphological structure of pulai wood in each treatment applied; (a) Untreated/UTFD; (b) 8N1H; (c) 8N2H; (d) 8N3H; (e) 9N1H; (f) 9N2H; (g) 9N3H; (h) 10N1H; (i) 10N2H; (j) 10N3H.
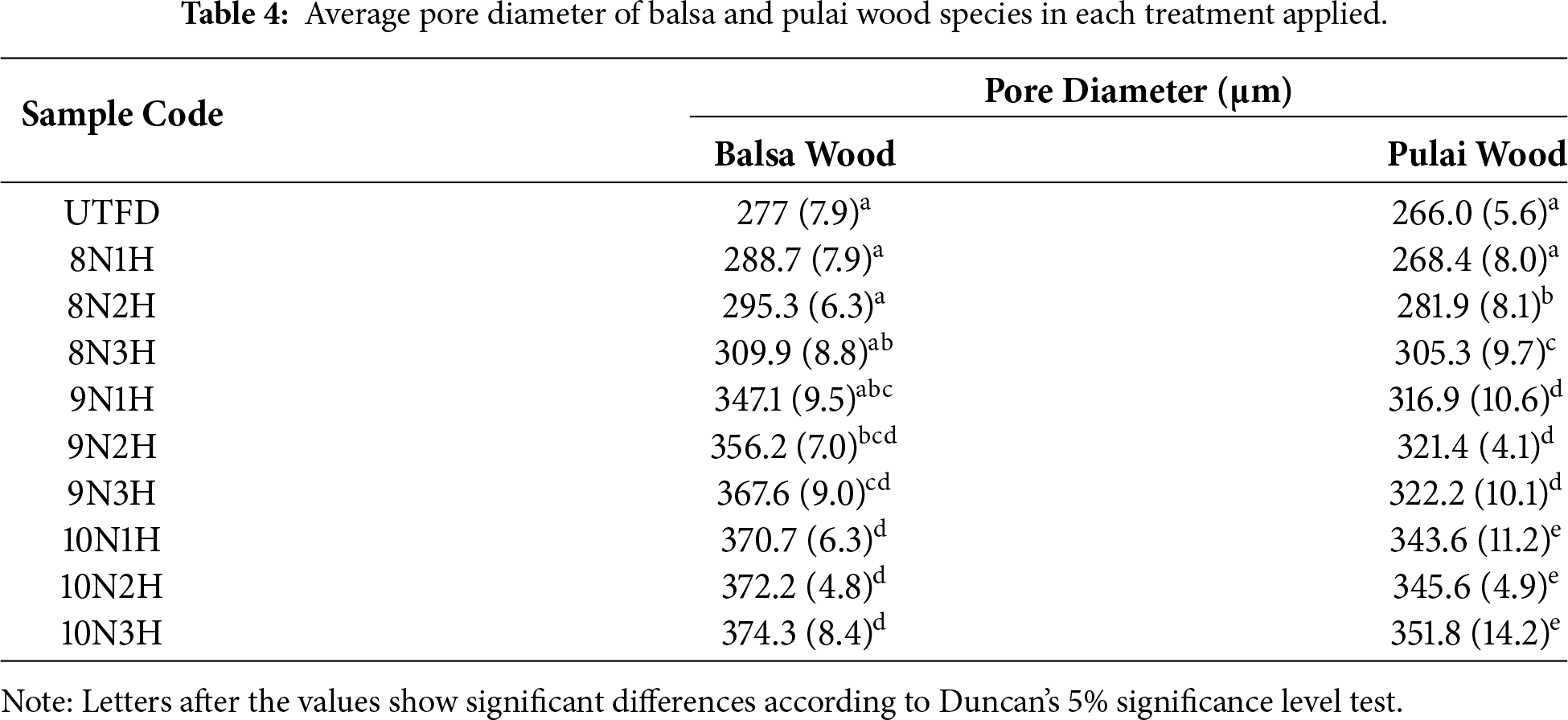
Based on the results presented in Table 4, all treatments applied to both balsa and pulai wood led to an increase in pore diameter. The most significant increase was observed in the 10N3H treatment, with balsa wood reaching a pore diameter of approximately 374.3 μm and pulai wood around 351.8 μm. This observation is consistent with previous findings which reported that fully delignified wood sponges treated for six hours exhibited a highly mesoporous structure, as evidenced by the resulting pore size distribution. Furthermore, Li et al. [27] state that the increase in pore size and porosity distribution is primarily attributed to the expansion of void volume following lignin removal. The delignification process significantly increases the internal porosity; while simultaneously improving accessibility by widening the pore diameter through the removal of extractives. This phenomenon results in the observed enhancement of adsorption capabilities.
Pore distribution in wood is typically categorized into macropores, mesopores, and micropores. The results indicate a clear enhancement in pore distribution, as illustrated in Fig. 7. A progressive increase in pore distribution was observed from the UTFD samples to those of treated samples with the longest delignification duration. The ANOVA results confirmed a significant difference among treatments, with longer delignification times resulting in increased pore diameter and distribution (Table 3). Specifically, in balsa wood, pore distribution increased from 55% to 88%, with an average improvement of approximately 79%. Meanwhile, pulai wood exhibited a more pronounced increase, ranging from 48% to 82%, with an average of around 68%. The best treatment outcomes in terms of pore distribution were recorded at 88% for balsa wood and 82% for pulai wood. This increase in pore distribution is attributed to the effective removal of lignin during the dual-stages delignification process. These findings align with prior research by Fu et al. [63], which reported that enhanced pore distribution results from dual-stages delignification, accompanied by an increase in specific surface area from 9 to 41 m2/g. Interestingly, despite the structural changes at the microscopic level, the overall morphology of the wood remained relatively unchanged. Li et al. [64], suggested that such processes primarily remove chromophore components of lignin while preserving bulk lignin, maintaining the general morphology of the wood structure.
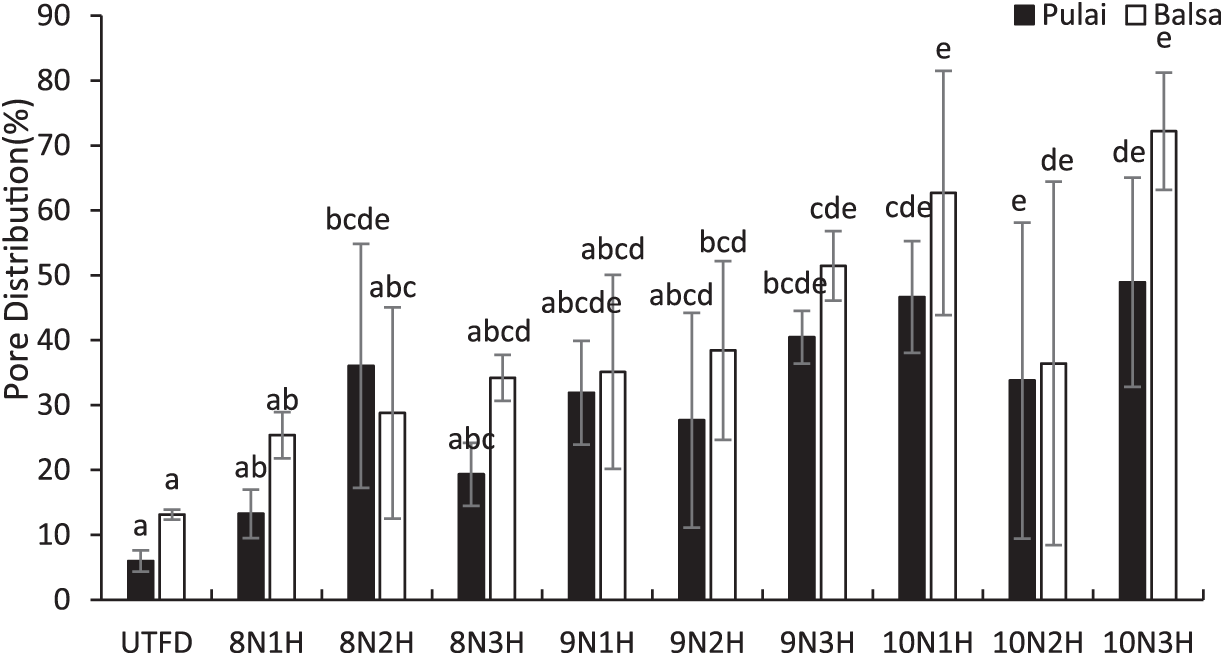
Figure 7: Pore distribution of balsa and pulai wood species in each treatment applied. Note: Letters above the graph show significant differences according to Duncan’s 5% significance level test.
After delignification, the wood cell wall exhibited increased porosity and an expanded cell lumen area. This structural transformation effectively opened the native cellular network, enhancing internal passageways within the wood [18]. The significant changes in pore diameter observed after dual-stages delignification also correspond with previous findings on wood sponges, which emphasize that their hierarchical pore structures arise from a combination of nanoscale cross-linked chemical networks and micrometer-scale intercellular channels [28].
Based on the results, the dual-stages delignification process effectively enhances the brightness and whiteness of the wood, as indicated by a significant increase in the L (lightness) values and ∆E (total color difference) values according to the CIE Lab color system. In this system, a parameter represents the red-green axis, while b indicates the yellow-blue axis. As shown in Fig. 8, both balsa and pulai woods exhibit fluctuating color changes throughout the treatments. Balsa wood tends to be more easily delignified, resulting in more pronounced changes in brightness and color, whereas pulai wood shows a more resistant behavior toward delignification, likely due to its intrinsic anatomical and chemical characteristics. This resistance influences the extent of color change after treatment. These findings are supported by Bradai et al. [65], who reported that wood with lower lignin content could enhance light penetration. Lignin contributes to the natural brownish coloration of wood due to its phenolic structure. Upon delignification, the removal of lignin leads to a cellulose-rich matrix that becomes more translucent, although the structural shape of the wood remains intact. The degree of color change varied significantly between treatments. In pulai wood, distinct total color changes occurred in the first treatment applied, the ∆E value increased from 6.15 (UTFD) to 41.85 (8N1H), whereas for balsa wood, the ∆E value increased from 10.15 (UTFD) to 33.68 (10N3H).
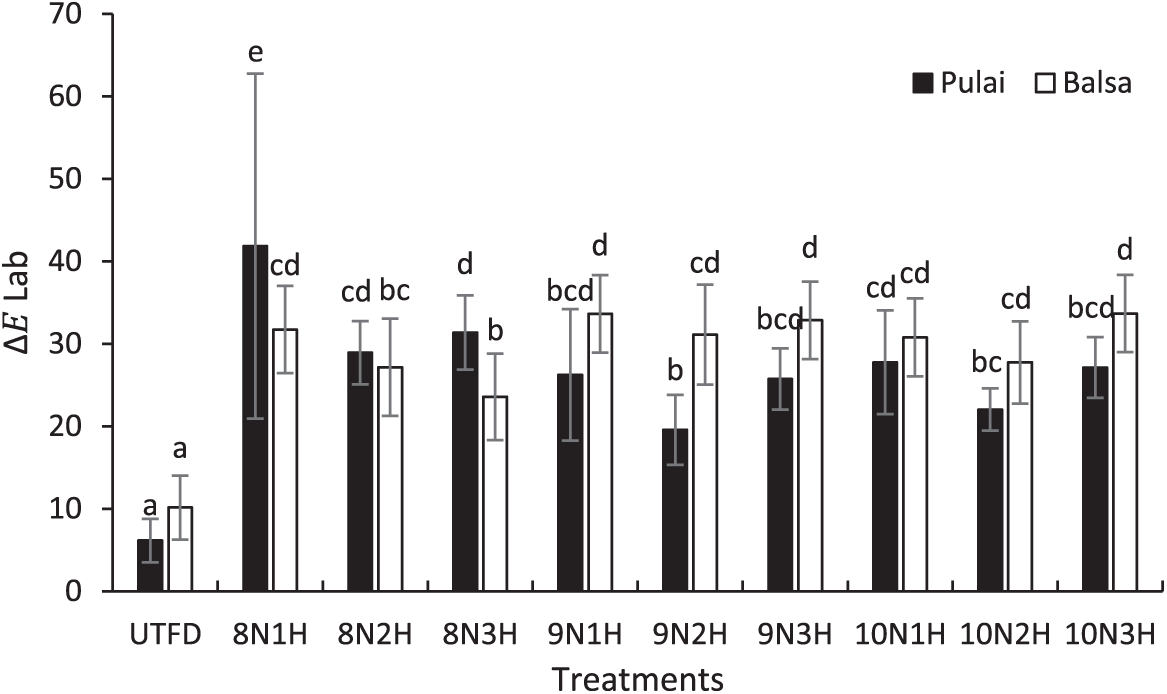
Figure 8: Color changes of balsa and pulai wood species in each treatment applied. Note: Letters above the graph show significant differences according to Duncan’s 5% significance level test.
The lightness (L) values in each wood species and treatment applied were presented in Fig. 9. For pulai wood, the highest increase in lightness was observed in the 8N3H treatment, where the L value increased significantly from 54.64 (UTFD) to 81.50. Meanwhile, for balsa wood, the 9N1H treatment showed the greatest escalation, from 63.67 (UTFD) to 87.71. This considerable improvement in brightness is primarily attributed to the delignification process, which removes lignin and extractives from the wood. Lignin and certain extractive compounds are known to cause significant light absorption and scattering within the visible spectrum due to their complex phenolic structures. Their removal enhances the wood’s ability to reflect light, resulting in a brighter and whiter appearance [66]. As delignification progresses, the reduction in these chromophore compounds contributes to the increased lightness observed, reinforcing the role of chemical composition, particularly lignin content, in determining wood color response to treatment [67].
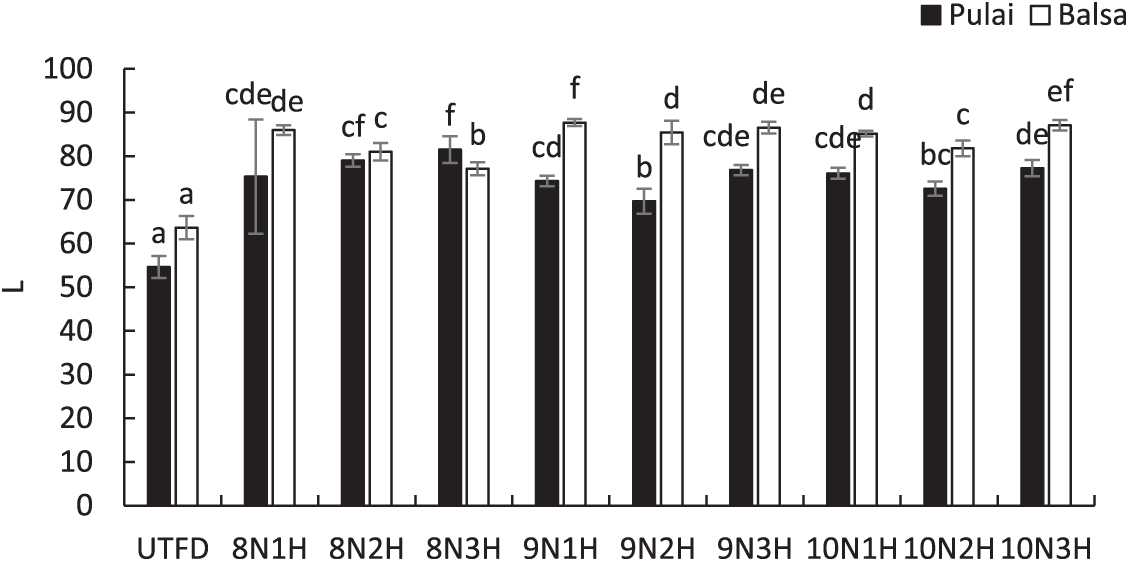
Figure 9: Lightness values of balsa and pulai wood species in each treatment applied. Note: Letters above the graph show significant differences according to Duncan’s 5% significance level test.
According to Table 5, the results of converting the CIELAB color values into the RGB spectrum indicate that brightness escalation occurred in all treatments, with balsa wood exhibiting a higher degree of translucency compared to pulai wood (Fig. 10). Although pulai wood is more resistant to delignification, it still showed a consistent increase in brightness across the treatments (Fig. 10). This finding shows similarity with previous research that underlines the effect of delignification. The impact of lignin removal could result in a white, cellulose-rich scaffold of the wood skeleton [67]. Furthermore, the delignification process led to a noticeable discoloration in both wood types. Based on the classification of color variation, all treatments demonstrated ΔE values greater than 12.0, indicating a “very distinct” or “clearly noticeable” color change [68]. This significant change is strongly associated with lignin removal, particularly during longer delignification durations, which contributes to enhanced visibility and the translucent appearance of the resulting wood sponges.
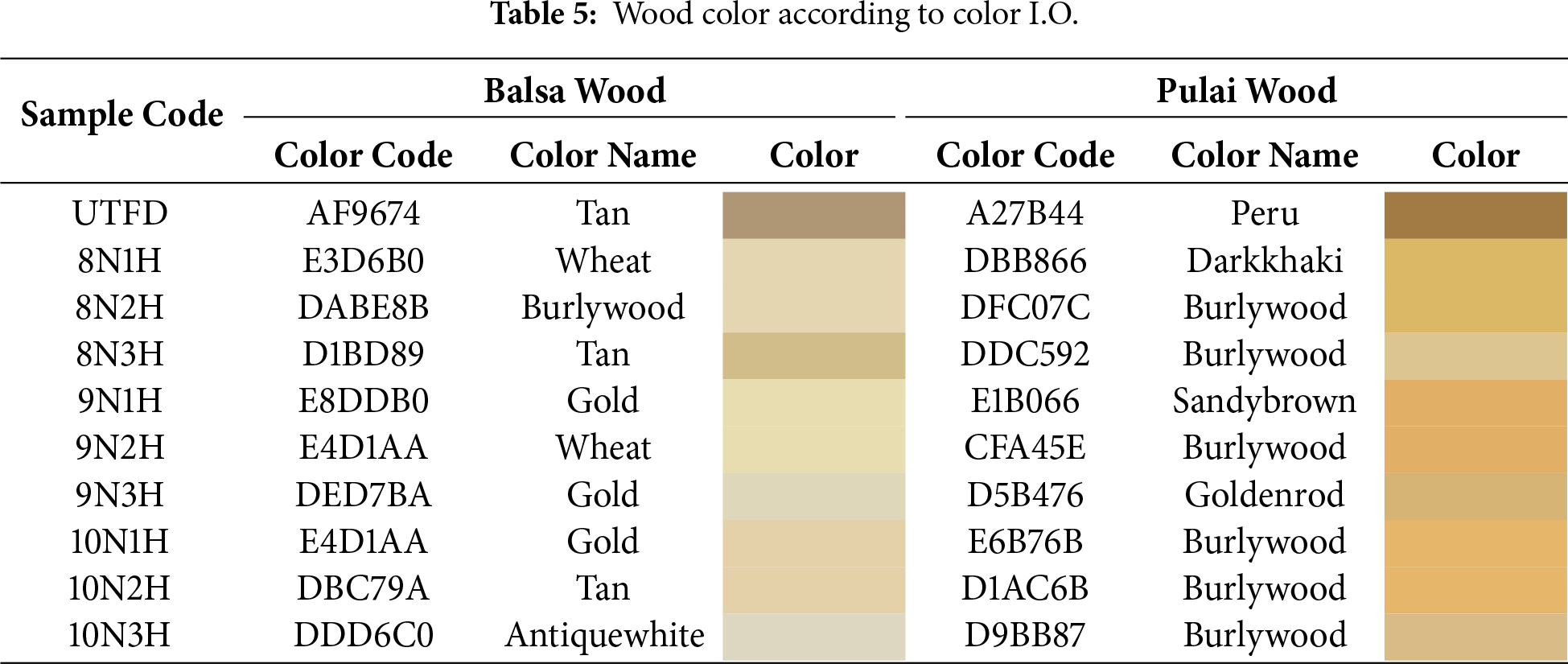

Figure 10: Colour appearance of balsa (A) and pulai (B) wood species in each treatment applied; (a) UTFD/Untreated; (b) 8N1H; (c) 8N2H; (d) 8N3H; (e) 9N1H; (f) 9N2H; (g) 9N3H; (h) 10N1H; (i) 10N2H; (j) 10N3H.
3.4 The Changes in Chemical Component
Fourier Transform Infrared Spectroscopy (FTIR) analysis was conducted to examine the chemical component changes in wood sponges from each treatment, focusing on the effectiveness of lignin removal while preserving cellulose integrity. As shown in Fig. 11A, significant spectral differences were observed in the range of 1940–3420 cm−1. These variations indicate a decrease in C–O bond intensity, which is commonly associated with the presence of lignin and hemicellulose. Notably, balsa wood exhibited a distinct peak at 1055 cm−1 and a noticeable shift at 1252 cm−1, suggesting C–O bond stretching. These spectral shifts imply that the delignification process primarily targeted lignin and hemicellulose, while cellulose remained largely unaffected. This observation aligns with previous findings that the C–O stretching vibration absorption peak at around 1232 cm−1 originates mainly from lignin and hemicellulose components. Moreover, the gradual weakening of the C=O carbonyl stretching peak at 1733 cm−1 further confirms the partial removal of hemicellulose through hydrogen peroxide treatment. Declining slope in the region of 1780–2290 cm−1 indicates the presence of carboxyl group stretching, while a distinct signal at 2335 cm−1 suggests degradation of lignin and hemicellulose. Additionally, a broad absorption band between 3051–3589 cm−1, especially around 3340 cm−1, corresponds to O–H stretching vibrations, highlighting further breakdown of hydroxyl-containing compounds such as hemicellulose and lignin. These spectral changes clearly demonstrate that the hydrogen peroxide-assisted delignification effectively reduces lignin and hemicellulose content while preserving the cellulose framework.
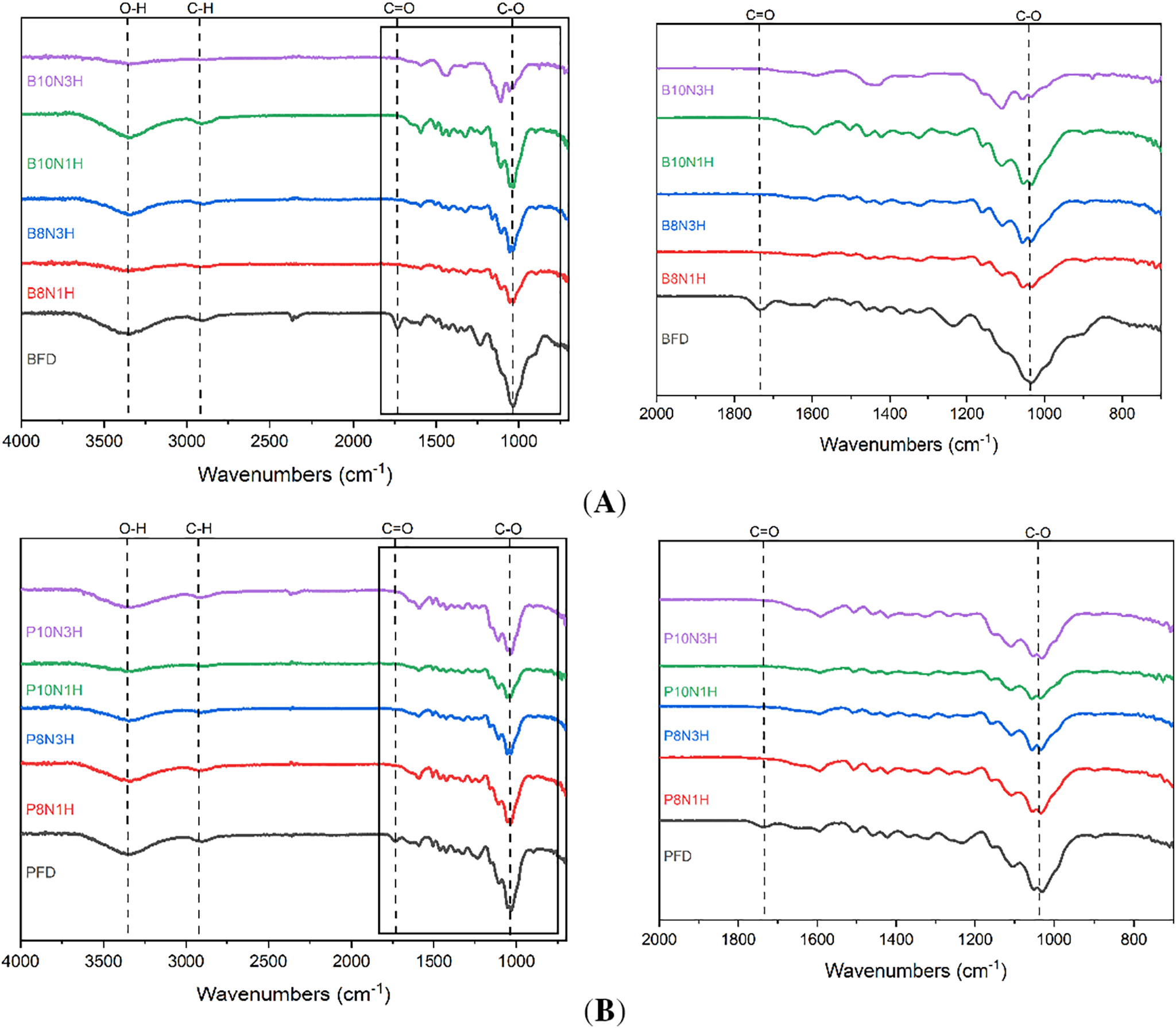
Figure 11: FTIR observation of balsa (A) and pulai (B) wood species in each treatment applied.
The results shown in Fig. 11B illustrate the FTIR spectral analysis of pulai wood before and after the delignification process. Stretching vibrations were observed across the range of 866–3645 cm−1, with a notable broad peak at 3339 cm−1 corresponding to O–H stretching. This indicates that the delignification process was effective in disrupting hydroxyl-containing components, particularly lignin. These findings are in line with previous research, which stated that untreated wood typically exhibits a distinct peak around 1734 cm−1—commonly associated with carbonyl groups in hemicellulose and lignin—and that the reduction or disappearance of this peak reflects successful removal of these components. Further confirmation is provided by the appearance of stretching at 2355 cm−1, which corresponds to C–H stretching, a marker of residual lignin presence. The persistence of this peak suggests that pulai wood is more resistant to delignification compared to other wood types, likely due to its anatomical and chemical characteristics. Pulai wood is classified as a hardwood species, yet it is lightweight and possesses relatively fine fibers, typically ranging from 0.2 to 1.22 mm in length, and smaller fiber pits, making chemical penetration more difficult. These hardwood fibers contribute significantly to pulai wood’s mechanical strength, thereby complicating the delignification process [18]. Nonetheless, Table 6 FTIR data clearly show that even in pulai wood, the delignification process induces considerable chemical changes, particularly in lignin and hemicellulose removal, although to a lesser extent than in balsa wood. In addition, pulai wood is one of the hardwoods that have a low density with a special characteristic, having the length of fibers about 0.2–1.22 mm with smaller pits, and the extractive content (flavonoid and alkaloid), as well as a packed lignin composition that is more resistant to chemical degradation by alkali or acid treatment [29,69].
Based on the data presented in Table 6, the delignification process affected absorption within the wavenumber ranging from 1125–1736 cm−1, particularly at the C–O stretching bands, which are indicative of hemicellulose and lignin structures. The observed spectral changes confirm the progressive dissolution of these components. In balsa wood, a distinct peak at 1055 cm−1 and a shift observed at 1252 cm−1 suggest the reduction of lignin and hemicellulose content, along with a weakening band at 1433 cm−1, which further supports the removal of hemicellulose. In addition, balsa wood showed a flatter pattern around wavenumbers of 3555 cm−1 (hydroxyl group/–OH), 1650–1750 cm−1 (carboxyl (–COOH) and C=O groups stretching) as a prolonged delignification process (10N3H) due to lignin and hemicellulose bound breaking with wood structure [23,42,75,76]. On the other hand, pulai wood exhibited a comparable pattern, with a notable peak at 1734 cm−1—characteristic of lignin—and stretching around 2355 cm−1 (C–H group), which reflects the partial degradation of lignin and hemicellulose. reduced accessibility to chemical agents, ultimately impacting the efficiency of lignin degradation [40]. Even though, pulai wood showed a similar/comparable pattern, however, it remains more difficult to delignify in the same condition with balsa wood. From these findings, it can be concluded that the most effective treatment for both balsa and pulai wood, in terms of lignin and hemicellulose removal as identified through FTIR analysis, was the 10N3H treatment.
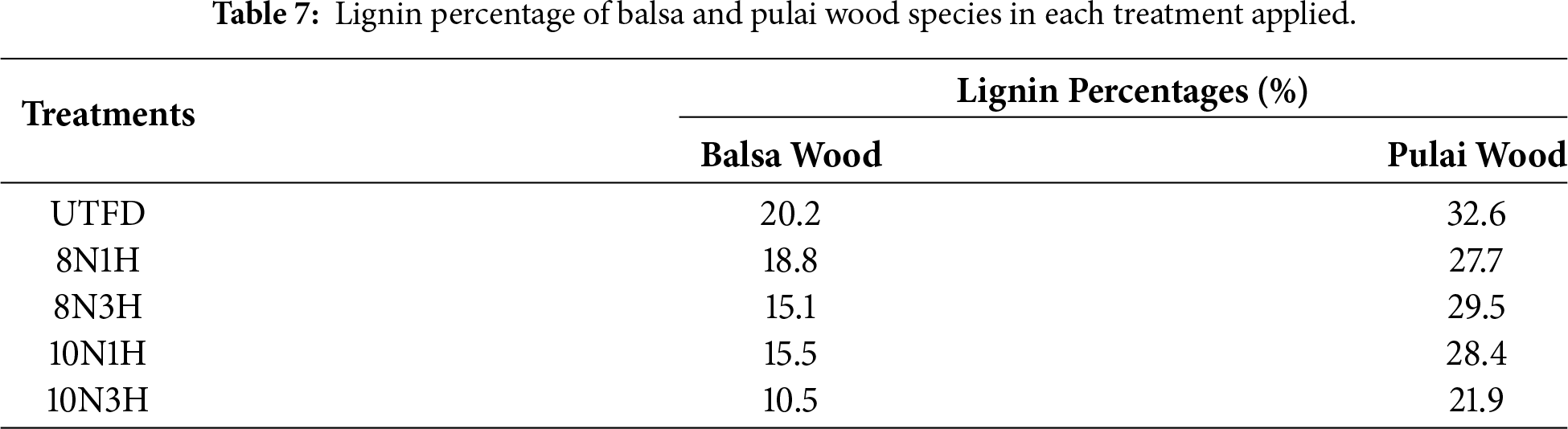
The lignin extraction method applied in this study followed a modified TAPPI protocol 1990 [36], which involves sequential extraction steps from finely ground mesh samples. The obtained data support the conclusion that extended durations in the dual-stages delignification process significantly enhance lignin removal. During delignification, the complex structure of lignin is chemically degraded and becomes soluble in the applied solution. This approach, which aims to remove lignin and hemicellulose while preserving the cellulose framework, is a key strategy in developing advanced wood-based sponge materials with improved functional properties. As shown in Table 7, balsa wood exhibited the lowest lignin content after 10N3H treatment, at approximately 10.5%, whereas the highest lignin content was observed in the untreated (UTFD) samples, reaching 18.8%. In contrast, pulai wood demonstrated more variation, with the lowest lignin content also observed in the 10N3H treatment at 21.9%, while the highest was found in the 8N3H treatment at 29.5%. These findings are consistent with prior studies, which reported significant reductions in lignin content following similar delignification. Further supporting evidence from literature confirms that lignin can be effectively removed by sequential cooking in NaOH and Na2SO3 solutions, followed by hydrogen peroxide (H2O2) treatment, which enhances the breakdown and solubilization of lignin compounds [63].
3.5 Adsorption Capacity and Porosity
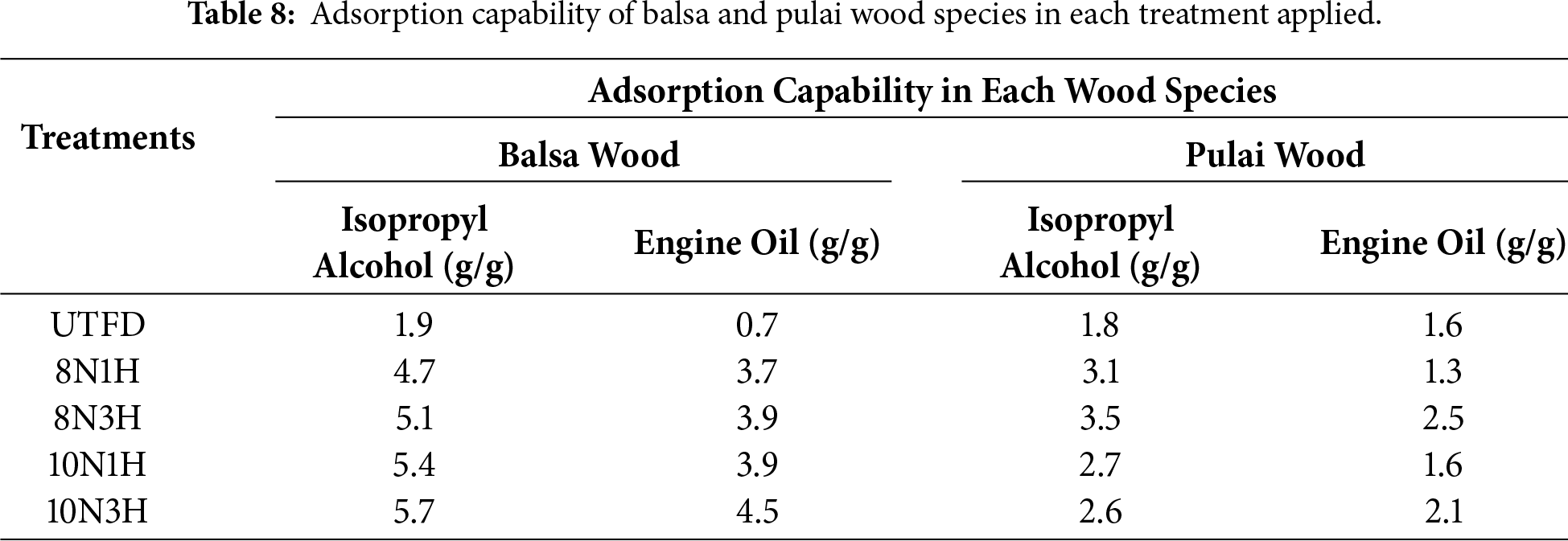
Adsorption capability was evaluated using isopropyl alcohol and engine oil by submerging the delignified wood sponge samples in both liquids and measuring their adsorption capability. Adsorption plays a crucial role, especially considering that contaminants such as oils and alcohols—which tend to adsorb strongly onto soil and water are often recalcitrant to biodegradation [77]. As shown in Table 8, there is a significant difference between balsa and pulai wood in terms of their adsorption performance. Pulai wood exhibited fluctuating adsorption values, with the optimal performance at treatment 8N3H, reaching 3.5 g/g for isopropyl alcohol and 2.5 g/g for engine oil. In contrast, balsa wood demonstrated higher adsorption values overall, with the best results observed at 10N3H—5.7 g/g for isopropyl alcohol and 4.5 g/g for engine oil. This indicates that balsa wood, due to its lower density and more open pore structure, is more suitable as an adsorbent material compared to pulai wood. These findings also confirm the effectiveness of the dual-stages delignification using H2O2 which appears to enhance the porous structure and increase surface area available for adsorption. The use of an alkaline solution (NaOH + Na2SO3) in the first stage helps break down lignin, and the subsequent peroxide treatment likely removes remaining matrix components, further opening up the wood structure for fluid absorption.
The primary factor influencing the adsorption capability of balsa and pulai wood lies in their cell wall structure and resulting porosity. The collapse of porous structures during drying has been well documented, yet several studies have reported that delignification significantly enhances wood porosity, leading to the formation of an open, interconnected porous system in fully delignified wood. Structurally, the wood cell wall is composed of multiple layers built from oriented cellulose microfibrils, which are closely associated with hemicellulose and embedded within a matrix formed by lignin. Lignin primarily acts as an adhesive, contributing to the rigidity and cohesion of the cell wall, especially within the middle lamella that binds adjacent wood cells together [70]. During the dual-stages delignification process—initially using NaOH + Na2SO3 followed by hydrogen peroxide treatment—the lignin and a portion of hemicellulose are removed, leaving behind a more exposed and accessible cellulose framework. This transformation results in a layered, sponge-like architecture with increased surface area and porosity. As reflected in the adsorption data, balsa wood demonstrated a superior capability for adsorbing oil and organic solvents compared to pulai wood. This difference is attributed to the more open and layered microstructure developed in balsa wood after delignification, which facilitates greater fluid uptake [78]. These findings align with previous reports indicating that dual-stages chemical delignification enhances the adsorption behavior of lignocellulosic materials by improving pore accessibility and interconnectivity [43].
It can be concluded that the higher porosity tends to give more storage space for adsorbing more liquid spills and enhancing capillary action, wood sponges with higher porosity (often >80%) and exhibit a significantly larger adsorption performance [23], directly boosting the capability of wood sponges, and creating a more larger and more interconnected pores [37].
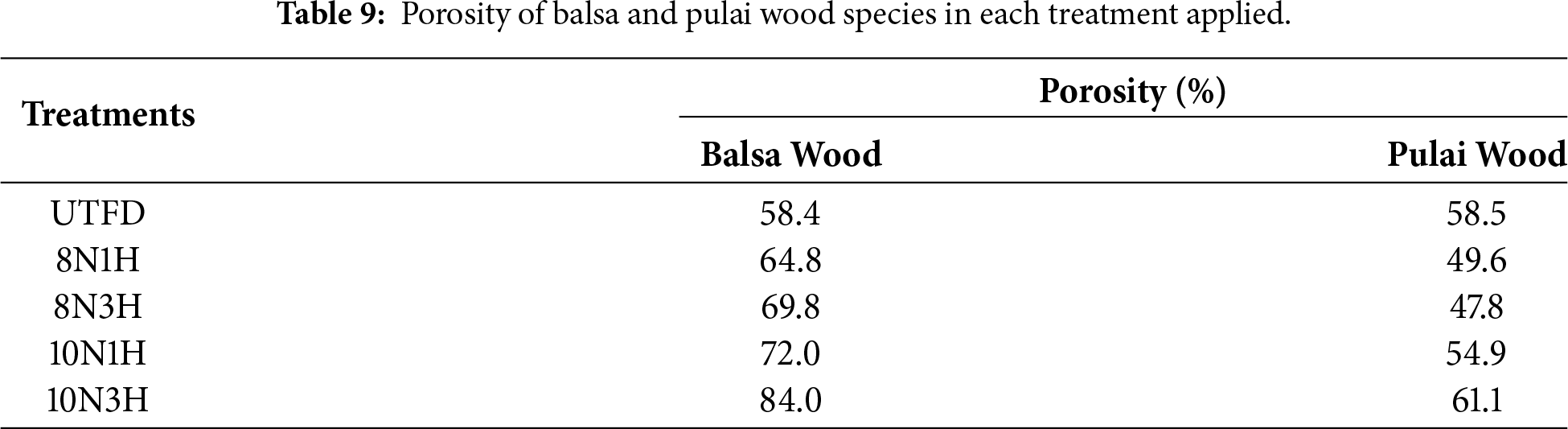
Wood porosity refers to the amount and distribution of void spaces or pores within the wood structure. The formation of a mesoporous structure, particularly through delignification, significantly enhances the specific surface area, which is a key factor in determining the adsorption behavior and overall performance of wood-based materials such as aerogels. As shown in Table 9, the porosity values of balsa and pulai wood after being submerged in isopropyl alcohol exhibit a similar trend to their adsorption capability. Balsa wood demonstrated the highest porosity value of 84% with the 10N3H treatment when tested with isopropyl alcohol, while pulai wood reached a porosity of 61.1% under the same treatment. These findings confirm that the dual-stages delignification process substantially increases the porosity compared to the untreated/control samples. This increase is attributed to the removal of lignin and hemicellulose, which creates additional voids and enlarges existing lumens within the wood structure. A study by Vitas et al. [30] supports this observation, stating that delignification can increase the number and size of ray cell voids, typically ranging from 20 to 50 μm. Furthermore, literature indicates that ideal porosity levels for highly functional absorbent materials range from 90% to 98% [79]. However, even porosity levels above 80%—as observed in balsa wood—are sufficient to produce a dense network of micropores and mesopores, which provide storage capacity for liquids and enhance the capillary-driven adsorption behavior of wood sponges.
Based on Fig. 12, wood sponges with higher porosity (often >80%) and larger pore diameter exhibit a significant adsorption performance [23]. A positive trend was observed between pore diameter and adsorption capability on balsa wood (y = 0.0194x − 2.4619; R2 = 0.3038) and pulai wood (y = 0.0183x − 3.187; R2 = 0.331). However, pulai wood has a lower R2 than balsa wood due to a smaller pore diameter, which could reflect its lower adsorption capability. On the other hand, the characteristics of the liquid play a major role. The absorption capability during engine oil immersion is lower than isopropyl alcohol due to differences in their characteristics. After treatment, wood sponge exhibits enhanced hydrophilic and could offer an eco-friendly, sustainable, efficient solution for oil-separation and absorption for non-polar substances such as alcohol [80]. Another factor is the formation of mesopores during the dual-stages delignification process, as noted by Liang et al. [79]. This contributes to the high adsorption of isopropyl solution, since delignification enhances the capillary action and pore structure of wood, favoring the uptake of less viscous liquids like isopropyl alcohol [23,37]. This process directly boosts the capability of wood sponges, and creates larger and interconnected pores.
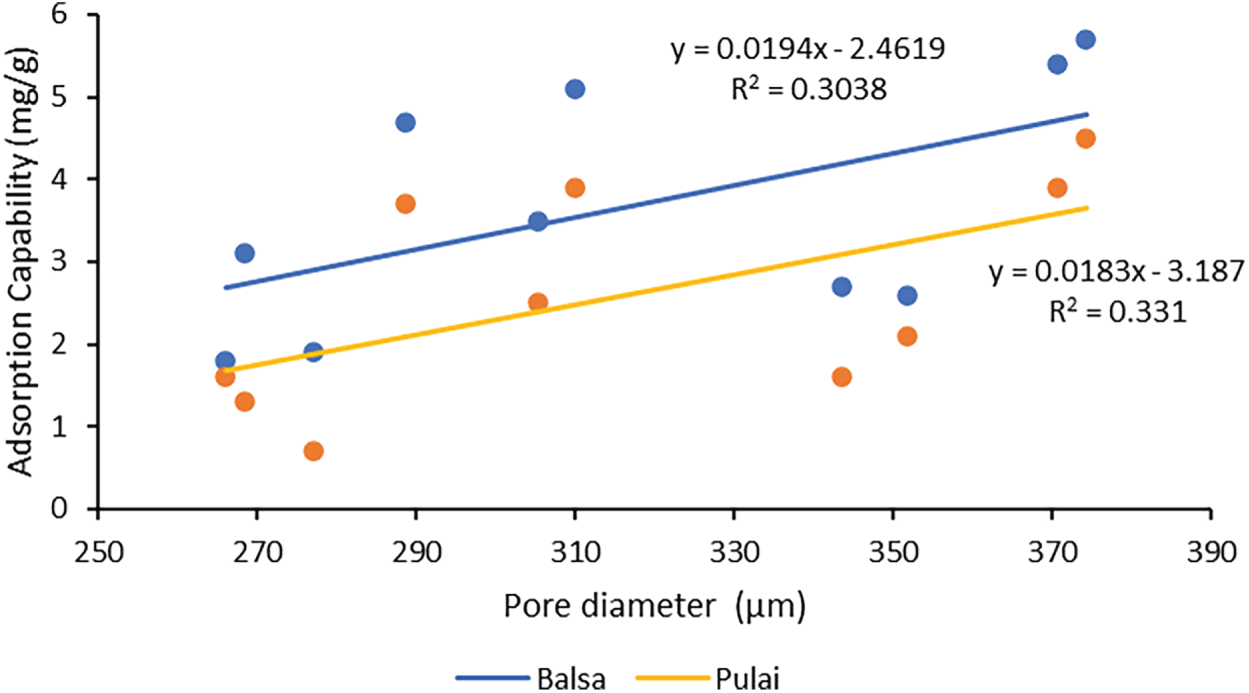
Figure 12: Correlation between pore diameter and adsorption capability for each wood sponge.
The dual-stages delignification process notably enhanced the physical and structural properties of balsa and pulai wood sponges. Color analysis showed a clear shift to brighter tones—particularly under the 10N3H treatment—indicating the removal of chromophore compounds like lignin and extractives. Key physical properties, such as density, specific gravity, and weight loss, also decreased, supporting the development of a more porous, lightweight, and layered cell wall structure favorable for adsorption. FTIR spectra confirmed lignin and hemicellulose degradation, with balsa wood showing strong activity at 1940–3420 cm−1 and pulai wood at 1780–2290 cm−1. These findings aligned with lignin content analysis, which revealed a significant reduction in balsa wood, especially at 10N3H. Pulai wood exhibited more variability, suggesting greater resistance to delignification due to its denser anatomy and higher lignin content. Adsorption studies further supported these trends. Balsa wood achieved peak adsorption for both isopropyl alcohol and engine oil at 10N3H, correlating with its higher porosity (84%). In contrast, pulai wood performed best at 8N3H, with reduced performance at longer treatment durations, likely due to compromised structural integrity. Porosity measurements mirrored this pattern, with pulai wood reaching 61.1% at 10N3H. Overall, the 10N3H treatment—10 h of NaOH + Na2SO3 followed by 3 h of H2O2—proved most effective for enhancing porosity and adsorption capability. While both species responded to treatment, balsa wood consistently outperformed pulai wood, making it a more suitable candidate for bio-based adsorbent applications.
Acknowledgement: The authors would like to thank the National Research and Innovation Agency and IPB University for supporting this study.
Funding Statement: This work was supported by Riset dan Inovasi untuk Indonesia Maju (RIIM) Kompetisi scheme (Grant number: 48/II.7/HK/2025), RP ORNM 2025, National Collaborative Research/RiNa (No. 499/2023), and Penelitian Dosen Pemula grant from the Ministry of Higher Education, Science and Technology, Republic of Indonesia (1483az/IT9.2.1/PT.01.03/2025).
Author Contributions: Hapidh Alaudin Syalsabil: Investigation; Methodology; Visualization; Writing—Original Draft. Imam Wahyudi: Conceptualization; Writing—Review & Editing. Deazy Rachmi Trisatya: Investigation. Sarah Augustina: Resources; Investigation; Methodology; Visualization; Writing—Review & Editing. Sari Delviana Marbun: Writing—Review & Editing. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The authors confirm that the data supporting the findings of this study are available within the article.
Ethnics Approval: The article contains no studies involving human or animal subjects.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Dubey SP, Gopal K, Bersillon JL. Utility of adsorbents in the purification of drinking water: a review of characterization, efficiency and safety evaluation of various adsorbents. J Environ Biol. 2009;30(3):327–32. [Google Scholar] [PubMed]
2. Osman AI, El-Monaem EMA, Elgarahy AM, Aniagor CO, Hosny M, Farghali M, et al. Methods to prepare biosorbents and magnetic sorbents for water treatment: a review. Environ Chem Lett. 2023;21(4):2337–98. doi:10.1007/s10311-023-01603-4. [Google Scholar] [CrossRef]
3. Ngueagni PT, Hefnawy M, Ofudje EA, El Gamal A, Akande JA, Bin Emran T. Cellulose-based adsorbent of animal waste for the adsorption of lead and phenol. BioResources. 2025;20(2):3923–52. doi:10.15376/biores.20.2.3923-3952. [Google Scholar] [CrossRef]
4. Teshale F, Karthikeyan R, Sahu O. Synthesized bioadsorbent from fish scale for chromium (III) removal. Micron. 2020;130:102817. doi:10.1016/j.micron.2019.102817. [Google Scholar] [PubMed] [CrossRef]
5. Dai L, Zhu W, He L, Tan F, Zhu N, Zhou Q, et al. Calcium-rich biochar from crab shell: an unexpected super adsorbent for dye removal. Bioresour Technol. 2018;267:510–6. doi:10.1016/j.biortech.2018.07.090. [Google Scholar] [PubMed] [CrossRef]
6. Pradhan P, Bajpai A. Preparation and characterization of films from chicken feathers for dye adsorption. Mater Today Proc. 2020;29:1204–12. doi:10.1016/j.matpr.2020.05.433. [Google Scholar] [CrossRef]
7. Al-Homaidan AA, Al-Qahtani HS, Al-Ghanayem AA, Ameen F, Ibraheem IBM. Potential use of green algae as a biosorbent for hexavalent chromium removal from aqueous solutions. Saudi J Biol Sci. 2018;25(8):1733–8. doi:10.1016/j.sjbs.2018.07.011. [Google Scholar] [PubMed] [CrossRef]
8. Rehman MU, Taj MB, Carabineiro SAC. Biogenic adsorbents for removal of drugs and dyes: a comprehensive review on properties, modification and applications. Chemosphere. 2023;338:139477. doi:10.1016/j.chemosphere.2023.139477. [Google Scholar] [PubMed] [CrossRef]
9. Xiang Z, Tang N, Jin X, Gao W. Fabrications and applications of hemicellulose-based bio-adsorbents. Carbohydr Polym. 2022;278:118945. doi:10.1016/j.carbpol.2021.118945. [Google Scholar] [PubMed] [CrossRef]
10. Shao L, Sang Y, Liu N, Liu J, Zhan P, Huang J, et al. Selectable microporous carbons derived from poplar wood by three preparation routes for CO2 capture. ACS Omega. 2020;5(28):17450–62. doi:10.1021/acsomega.0c01918. [Google Scholar] [PubMed] [CrossRef]
11. Choi HS, Suh M. Highly selective CO2 capture in flexible 3D coordination polymer networks. Angew Chem Int Ed. 2009;48(37):6865–9. doi:10.1002/anie.200902836. [Google Scholar] [PubMed] [CrossRef]
12. Liu S, Lin J, Chen Q, Liu Z, Gui L, Chen L, et al. A strategy of liquid-grafted slippery sponges with simultaneously enhanced absorption and desorption performances for crude oil spill remediation. Macromol Mater Eng. 2021;306(10):2170038. doi:10.1002/mame.202170038. [Google Scholar] [CrossRef]
13. Kukkar D, Rani A, Kumar V, Younis SA, Zhang M, Lee SS, et al. Recent advances in carbon nanotube sponge-based sorption technologies for mitigation of marine oil spills. J Colloid Interface Sci. 2020;570:411–22. doi:10.1016/j.jcis.2020.03.006. [Google Scholar] [PubMed] [CrossRef]
14. Saleh TA. Development and synthesis of nanoparticles and nanoadsorbents. Interface Sci Technol. 2022;34:127–65. doi:10.1016/b978-0-12-849876-7.00004-x. [Google Scholar] [CrossRef]
15. Singh S, Naik TSSK, Shehata N, Aguilar-Marcelino L, Dhokne K, Lonare S, et al. Novel insights into graphene oxide-based adsorbents for remediation of hazardous pollutants from aqueous solutions: a comprehensive review. J Mol Liq. 2023;369:120821. doi:10.1016/j.molliq.2022.120821. [Google Scholar] [CrossRef]
16. Cao M, Liu BW, Zhang L, Peng ZC, Zhang YY, Wang H, et al. Fully biomass-based aerogels with ultrahigh mechanical modulus, enhanced flame retardancy, and great thermal insulation applications. Compos Part B Eng. 2021;225:109309. doi:10.1016/j.compositesb.2021.109309. [Google Scholar] [CrossRef]
17. Moud AA. Advanced cellulose nanocrystals (CNC) and cellulose nanofibrils (CNF) aerogels: bottom-up assembly perspective for production of adsorbents. Int J Biol Macromol. 2022;222(Pt A):1–29. doi:10.1016/j.ijbiomac.2022.09.148. [Google Scholar] [PubMed] [CrossRef]
18. Kumar A, Jyske T, Petrič M. Delignified wood from understanding the hierarchically aligned cellulosic structures to creating novel functional materials: a review. Adv Sustain Syst. 2021;5(5):2000251. doi:10.1002/adsu.202000251. [Google Scholar] [CrossRef]
19. Mo L, Pang H, Lu Y, Li Z, Kang H, Wang M, et al. Wood-inspired nanocellulose aerogel adsorbents with excellent selective pollutants capture, superfast adsorption, and easy regeneration. J Hazard Mater. 2021;415:125612. doi:10.1016/j.jhazmat.2021.125612. [Google Scholar] [PubMed] [CrossRef]
20. Jiang J, Shi Y, Ma NL, Ye H, Verma M, Ng HS, et al. Utilizing adsorption of wood and its derivatives as an emerging strategy for the treatment of heavy metal-contaminated wastewater. Environ Pollut. 2024;340:122830. doi:10.1016/j.envpol.2023.122830. [Google Scholar] [PubMed] [CrossRef]
21. Sun J, Guo H, Ribera J, Wu C, Tu K, Binelli M, et al. Sustainable and biodegradable wood sponge piezoelectric nanogenerator for sensing and energy harvesting applications. ACS Nano. 2020;14(11):14665–74. doi:10.1021/acsnano.0c05493. [Google Scholar] [PubMed] [CrossRef]
22. Alqrinawi H, Ahmed B, Wu Q, Lin H, Kameshwar S, Shayan M. Effect of partial delignification and densification on chemical, morphological, and mechanical properties of wood: structural property evolution. Ind Crops Prod. 2024;213:118430. doi:10.1016/j.indcrop.2024.118430. [Google Scholar] [CrossRef]
23. Darajat AZ, Wahyudi I, Bahanawan A, Augustina S. Characteristics of wood sponge from sengon (Falcataria moluccana) wood manufacturing through a multistage delignification process. J Renew Mater. 2025;13(8):1661–81. doi:10.32604/jrm.2025.02024-0081. [Google Scholar] [CrossRef]
24. Meng J, Guan H, Dai X, Wang X. Amino-functionalized wood aerogel for efficient removal of copper ions from water. Int J Polym Sci. 2021;2021(1):4913226. doi:10.1155/2021/4913226. [Google Scholar] [CrossRef]
25. Horikawa Y. Structural diversity of natural cellulose and related applications using delignified wood. J Wood Sci. 2022;68(1):54. doi:10.1186/s10086-022-02061-2. [Google Scholar] [CrossRef]
26. Yu Q, Sun X, Liu F, Yang Z, Wei S, Wang C, et al. Eco-friendly method for wood aerogel preparation with efficient catalytic reduction of 4-nitrophenol. Gels. 2023;9(12):978. doi:10.3390/gels9120978. [Google Scholar] [PubMed] [CrossRef]
27. Li Y, Vasileva E, Sychugov I, Popov S, Berglund L. Optically transparent wood: recent progress, opportunities, and challenges. Adv Opt Mater. 2018;6(14):1800059. doi:10.1002/adom.201800059. [Google Scholar] [CrossRef]
28. Li Y, Fu Q, Yu S, Yan M, Berglund L. Optically transparent wood from a nanoporous cellulosic template: combining functional and structural performance. Biomacromolecules. 2016;17(4):1358–64. doi:10.1021/acs.biomac.6b00145. [Google Scholar] [PubMed] [CrossRef]
29. Li J, Chen C, Zhu JY, Ragauskas AJ, Hu L. In situ wood delignification toward sustainable applications. Acc Mater Res. 2021;2(8):606–20. doi:10.1021/accountsmr.1c00075. [Google Scholar] [CrossRef]
30. Vitas S, Segmehl JS, Burgert I, Cabane E. Porosity and pore size distribution of native and delignified beech wood determined by mercury intrusion porosimetry. Materials. 2019;12(3):416. doi:10.3390/ma12030416. [Google Scholar] [PubMed] [CrossRef]
31. Fauziyah M, Widiyastuti W, Balgis R, Setyawan H. Production of cellulose aerogels from coir fibers via an alkali-urea method for sorption applications. Cellulose. 2019;26(18):9583–98. doi:10.1007/s10570-019-02753-x. [Google Scholar] [CrossRef]
32. Dayadi I. Retention of borax preservative and intensity of blue stain fungus attack on pulai (Alstonia scholaris R.Br.) based on preservation method. Int J Integr Sci. 2025;4(3):605–16. doi:10.55927/ijis.v4i3.120. [Google Scholar] [CrossRef]
33. Susdiyanti T, Meiganati KB, Lidiawati I, Abiksak A. Struktur anatomi dan sifat fisik kayu jati (Tectona grandis L.f.) unggul nusantara trubusan pada umur 8 tahun. J Hutan Lestari. 2023;11(1):28. doi:10.26418/jhl.v11i1.55115. [Google Scholar] [CrossRef]
34. Valverde JC, Moya R. Correlation and modeling between color variation and quality of the surface between accelerated and natural tropical weathering in Acacia mangium, Cedrela odorata and Tectona grandis wood with two coating. Color Res Appl. 2014;39(5):519–29. doi:10.1002/col.21826. [Google Scholar] [CrossRef]
35. Haeri M, Haeri M. ImageJ plugin for analysis of porous scaffolds used in tissue engineering. J Open Res Softw. 2015;3(1):1–4. doi:10.5334/jors.bn. [Google Scholar] [CrossRef]
36. TAPPI. TAPPI T-203: alfa, beta and gamma cellulose in pulp. Atlanta, GA, USA: TAPPI Press; 2022. [Google Scholar]
37. Wang Z, Lin S, Li X, Zou H, Zhuo B, Ti P, et al. Optimization and absorption performance of wood sponge. J Mater Sci. 2021;56(14):8479–96. doi:10.1007/s10853-020-05547-w. [Google Scholar] [CrossRef]
38. Larjavaara M, Muller-Landau HC. Rethinking the value of high wood density. Funct Ecol. 2010;24(4):701–5. doi:10.1111/j.1365-2435.2010.01698.x. [Google Scholar] [CrossRef]
39. Mania P, Kupfernagel C, Curling S. Densification of delignified wood: influence of chemical composition on wood density, compressive strength, and hardness of Eurasian aspen and Scots pine. Forests. 2024;15(6):892. doi:10.3390/f15060892. [Google Scholar] [CrossRef]
40. Zhu M, Li T, Davis CS, Yao Y, Dai J, Wang Y, et al. Transparent and haze wood composites for highly efficient broadband light management in solar cells. Nano Energy. 2016;26:332–9. doi:10.1016/j.nanoen.2016.05.020. [Google Scholar] [CrossRef]
41. Wang J, Liu J, Li J, Zhu JY. Characterization of microstructure, chemical, and physical properties of delignified and densified poplar wood. Materials. 2021;14(19):5709. doi:10.3390/ma14195709. [Google Scholar] [PubMed] [CrossRef]
42. Zhu M, Song J, Li T, Gong A, Wang Y, Dai J, et al. Highly anisotropic, highly transparent wood composites. Adv Mater. 2016;28(26):5181–7. doi:10.1002/adma.201600427. [Google Scholar] [PubMed] [CrossRef]
43. Wang J, Minami E, Asmadi M, Kawamoto H. Effect of delignification on thermal degradation reactivities of hemicellulose and cellulose in wood cell walls. J Wood Sci. 2021;67(1):19. doi:10.1186/s10086-021-01952-0. [Google Scholar] [CrossRef]
44. Zhang J, Ying Y, Yi X, Han W, Yin L, Zheng Y, et al. H2O2 solution steaming combined method to cellulose skeleton for transparent wood infiltrated with cellulose acetate. Polymers. 2023;15(7):1733. doi:10.3390/polym15071733. [Google Scholar] [PubMed] [CrossRef]
45. Wang Q, Xiao S, Shi SQ, Cai L. Effect of light-delignification on mechanical, hydrophobic, and thermal properties of high-strength molded fiber materials. Sci Rep. 2018;8:955. doi:10.1038/s41598-018-19623-4. [Google Scholar] [PubMed] [CrossRef]
46. Kilumets C, Kallakas H, Ralph S, Zhu JY, Hunt CG, Rohumaa A, et al. Effect of lignin on veneer densification and set-recovery. Constr Build Mater. 2024;451:138795. doi:10.1016/j.conbuildmat.2024.138795. [Google Scholar] [CrossRef]
47. Meng D, Long W, Sun J, Li H, Wang Z, Gu X, et al. Eco-friendly fabrication of a delignified wood-calcium alginate aerogel with improved mechanical properties for efficient thermal insulation and flame retardancy. Int J Biol Macromol. 2025;287:138561. doi:10.1016/j.ijbiomac.2024.138561. [Google Scholar] [PubMed] [CrossRef]
48. Surianegara I, Lemmens RHMJ. Timber trees: minor commercial timbers. In: Plant resources of South-East-Asia. Leiden, The Netherlands: Backhuys Publisher; 1995. doi:10.2307/1224176. [Google Scholar] [CrossRef]
49. Li H, Guo X, He Y, Zheng R. A green steam-modified delignification method to prepare low-lignin delignified wood for thick, large highly transparent wood composites. J Mater Res. 2019;34(6):932–40. doi:10.1557/jmr.2018.466. [Google Scholar] [CrossRef]
50. Marsoem SN, Prasetyo VE, Sulistyo J, Sudaryono S, Lukmandaru G. Study of teak wood quality in gunungkidul community forest III. Phys Prop of Wood J Ilmu Kehutan. 2014;8(2):75–88. (In Indonesia). [Google Scholar]
51. Uslinawaty Z, Pujirahayu N, Hadjar N, Kabe A, Hamzah N. Uji Kadar air titik jenuh serat beberapa jenis kayu perdagangan di kabupaten konawe selatan. Jurnal Celebica. 2024;5(1):75–82. doi:10.33772/jc.v5i1.83. [Google Scholar] [CrossRef]
52. Armstrong JP, Skaar C, DeZeeuw C. The effect of specific gravity on several mechanical properties of some world woods. Wood Sci Technol. 1984;18(2):137–46. doi:10.1007/BF00350472. [Google Scholar] [CrossRef]
53. Lin M, Guo X, Xu Y, Zhang X, Hu D. A top-down approach to the fabrication of flame-retardant wood aerogel with in situ-synthesized borax and zinc borate. Materials. 2024;17(11):2638. doi:10.3390/ma17112638. [Google Scholar] [PubMed] [CrossRef]
54. Chen HL. A note on the effect of alcohol-benzene extractives on juvenile wood specify gravity in red pine. Wood Fiber Sci. 1985;18(3):376–81. [Google Scholar]
55. Pari G, Roliadi H, Setiawan D, Saepuloh S. Komponen Kimia sepuluh jenis kayu tanaman dari jawa barat. J Penelit Has Hutan. 2006;24(2):89–101. doi:10.20886/jphh.2006.24.2.89-101. [Google Scholar] [CrossRef]
56. Anggraini R, Marwoto M, Khabibi J, Zaini B. Peningkatan kayu pulai (Alstonia scholaris) dengan metode densifikasi waktu pengukusan. J Silva Trop. 2024;8(2):130–40. (In Indonesia). doi:10.22437/jurnalsilvatropika.v8i2.37797. [Google Scholar] [CrossRef]
57. Owoyemi JM, Olaniran OS. Natural resistance of ten selected Nigerian wood species to subterranean termites’ attack. Int J Biol Sci Appl. 2014;1(2):35–9. [Google Scholar]
58. Borrega M, Ahvenainen P, Serimaa R, Gibson L. Composition and structure of balsa (Ochroma pyramidale) wood. Wood Sci Technol. 2015;49(2):403–20. doi:10.1007/s00226-015-0700-5. [Google Scholar] [CrossRef]
59. Andianto A. Perbandingan ciri anatomi kayu dan kulit 3 jenis pulai (Alstonia sp.). J Penelit Has Hutan. 2011;29(4):356–68. doi:10.20886/jphh.2011.29.4.356-368. [Google Scholar] [CrossRef]
60. Grönquist P, Frey M, Keplinger T, Burgert I. Mesoporosity of delignified wood investigated by water vapor sorption. ACS Omega. 2019;4(7):12425–31. doi:10.1021/acsomega.9b00862. [Google Scholar] [PubMed] [CrossRef]
61. Jang ES, Kang CW. Delignification effects on Indonesian momala (Homalium foetidum) and Korean red toon (Toona sinensis) hardwood pore structure and sound absorption capabilities. Materials. 2021;14(18):5215. doi:10.3390/ma14185215. [Google Scholar] [PubMed] [CrossRef]
62. Kumar S, Nautiyal I, Shukla S. Some physical properties of delignified and compressed Melia dubia wood. Bois For Trop. 2019;341:71–7. doi:10.19182/bft2019.341.a31758. [Google Scholar] [CrossRef]
63. Fu Q, Medina L, Li Y, Carosio F, Hajian A, Berglund LA. Nanostructured wood hybrids for fire-retardancy prepared by clay impregnation into the cell wall. ACS Appl Mater Interfaces. 2017;9(41):36154–63. doi:10.1021/acsami.7b10008. [Google Scholar] [PubMed] [CrossRef]
64. Li Y, Fu Q, Rojas R, Yan M, Lawoko M, Berglund L. Lignin-retaining transparent wood. ChemSusChem. 2017;10(17):3445–51. doi:10.1002/cssc.201701089. [Google Scholar] [PubMed] [CrossRef]
65. Bradai H, Koubaa A, Zhang J, Demarquette NR. Effect of wood species on lignin-retaining high-transmittance transparent wood biocomposites. Polymers. 2024;16(17):2493. doi:10.3390/polym16172493. [Google Scholar] [PubMed] [CrossRef]
66. Wu Y, Zhou J, Huang Q, Yang F, Wang Y, Wang J. Study on the properties of partially transparent wood under different delignification processes. Polymers. 2020;12(3):661. doi:10.3390/polym12030661. [Google Scholar] [PubMed] [CrossRef]
67. Zhong L, Zhao W, Yao X, Zhang Y, Wu Y, Zhou X. Preparation and physicochemical properties of colored transparent wood. Mater Today Commun. 2025;48:113387. doi:10.1016/j.mtcomm.2025.113387. [Google Scholar] [CrossRef]
68. Han X, Wang Z, Ding L, Chen L, Wang F, Pu J, et al. Water molecule-induced hydrogen bonding between cellulose nanofibers toward highly strong and tough materials from wood aerogel. Chin Chem Lett. 2021;32(10):3105–8. doi:10.1016/j.cclet.2021.03.044. [Google Scholar] [CrossRef]
69. Syarifuddin RN, Trisnawaty AR, Nurwidah A. Identifikasi senyawa kimia pada tanaman pulai (Alstonia scholaris) sebagai pestisida nabati untuk pengendali hama. J Galung Trop. 2021;10(1):40–7. doi:10.31850/jgt.v10i1.701. [Google Scholar] [CrossRef]
70. Ruan C, Shen M, Ren X, Ai K, Lu L. A versatile and scalable approach toward robust superhydrophobic porous materials with excellent absorbency and flame retardancy. Sci Rep. 2016;6:31233. doi:10.1038/srep31233. [Google Scholar] [PubMed] [CrossRef]
71. Song J, Chen C, Yang Z, Kuang Y, Li T, Li Y, et al. Highly compressible, anisotropic aerogel with aligned cellulose nanofibers. ACS Nano. 2018;12(1):140–7. doi:10.1021/acsnano.7b04246. [Google Scholar] [PubMed] [CrossRef]
72. Garemark J, Perea-Buceta JE, Rico Del Cerro D, Hall S, Berke B, Kilpeläinen I, et al. Nanostructurally controllable strong wood aerogel toward efficient thermal insulation. ACS Appl Mater Interfaces. 2022;14(21):24697–707. doi:10.1021/acsami.2c04584. [Google Scholar] [PubMed] [CrossRef]
73. Wang Y, Yang Y, Qu Y, Zhang J. Selective removal of lignin with sodium chlorite to improve the quality and antioxidant activity of xylo-oligosaccharides from lignocellulosic biomass. Bioresour Technol. 2021;337:125506. doi:10.1016/j.biortech.2021.125506. [Google Scholar] [PubMed] [CrossRef]
74. Jin Y, Tian F, She Y, Xu H, Zhu C, Wu Y, et al. Thermal insulation properties of delignified Balsa and Paulownia wood foams with polylactic acid coverings. Forests. 2023;14(12):2339. doi:10.3390/f14122339. [Google Scholar] [CrossRef]
75. Savy D, Verrillo M, Cangemi S, Cozzolino V. Lignin nanoparticles from hydrotropic fractionation of giant reed and eucalypt: structural elucidation and antibacterial properties. Int J Biol Macromol. 2024;262(Pt 2):129966. doi:10.1016/j.ijbiomac.2024.129966. [Google Scholar] [PubMed] [CrossRef]
76. Hidayat Sihotang S. Sintesis poliol lignin isolat Dari serbuk kayu jati sebagai bahan dasar pembentukan poliuretan Dan karakterisasi menggunakan ft-ir (Fourier transform infrared spectroscopy). J Ilm Keperawatan Imelda. 2018;4(1):32–7. doi:10.52943/jikeperawatan.v4i1.281. [Google Scholar] [CrossRef]
77. Ren X, Zeng G, Tang L, Wang J, Wan J, Liu Y, et al. Sorption, transport and biodegradation—an insight into bioavailability of persistent organic pollutants in soil. Sci Total Environ. 2018;610–611:1154–63. doi:10.1016/j.scitotenv.2017.08.089. [Google Scholar] [PubMed] [CrossRef]
78. Guan H, Cheng Z, Wang X. Highly compressible wood sponges with a spring-like lamellar structure as effective and reusable oil absorbents. ACS Nano. 2018;12(10):10365–73. doi:10.1021/acsnano.8b05763. [Google Scholar] [PubMed] [CrossRef]
79. Liang R, Zhu YH, Wen L, Zhao WW, Kuai BB, Zhang YL, et al. Exploration of effect of delignification on the mesopore structure in poplar cell wall by nitrogen absorption method. Cellulose. 2020;27(4):1921–32. doi:10.1007/s10570-019-02921-z. [Google Scholar] [CrossRef]
80. Zhang C, Cai T, Ge-Zhang S, Mu P, Liu Y, Cui J. Wood sponge for oil-water separation. Polymers. 2024;16(16):2362. doi:10.3390/polym16162362. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF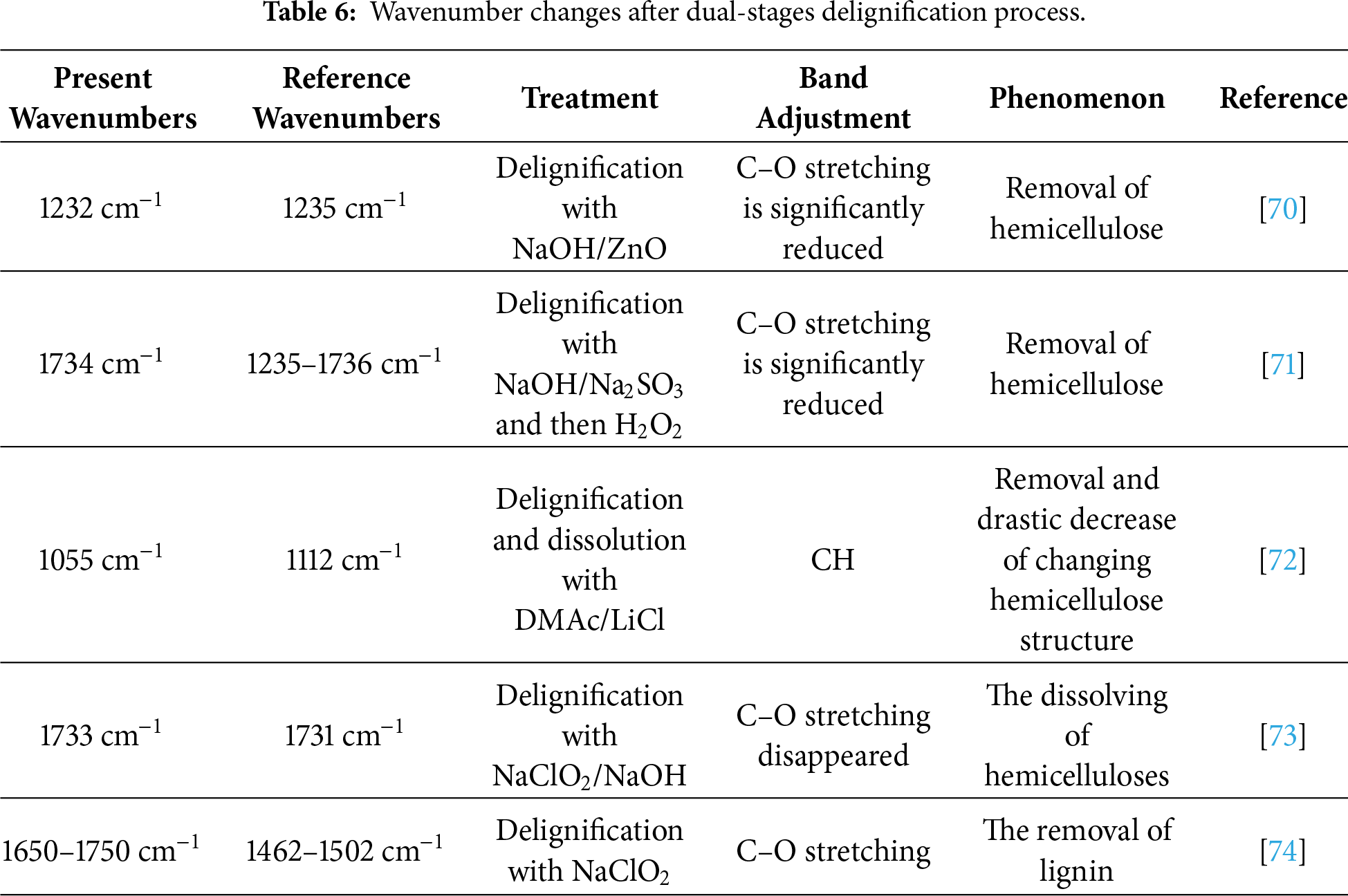
 Downloads
Downloads
 Citation Tools
Citation Tools