 Open Access
Open Access
ARTICLE
Zein Membrane Barrier for Bone Guided Regeneration
1 University of Araraquara—UNIARA, Rua Carlos Gomes, 1217, São Paulo, 14801-340, Brazil
2 Department of Materials Engineering, São Carlos School of Engineering, University of São Paulo, São Paulo, 13563-120, Brazil
* Corresponding Author: Eliane Trovatti. Email:
Journal of Renewable Materials 2026, 14(3), 1 https://doi.org/10.32604/jrm.2025.02025-0166
Received 22 August 2025; Accepted 23 October 2025; Issue published 25 March 2026
Abstract
Materials from natural sources have been studied to replace the conventional synthetic or animal-derived products as a safer alternative to be used in the healthcare field. In dentistry, guided bone regeneration (GBR) relies on barrier membranes, predominantly from animals or synthetic materials, to improve osteogenesis by avoiding undesired soft tissue cells from defect sites. In this study, membranes were prepared from zein, a corn-derived protein, using a simple extraction and casting method, followed by optional formaldehyde cross-linking to evaluate their behavior for application in GBR. The membranes were characterised by FTIR, DSC, TGA, tensile strength analysis, and in vitro biological assays. Cross-linked membranes exhibited improved mechanical strength (~5 MPa) and slower degradation (~43% mass loss over 30 days), while non-cross-linked membranes disintegrated more rapidly. Cytotoxicity assays using GM07492 fibroblasts confirmed biocompatibility, and cell migration studies demonstrated effective barrier function. These results indicated that zein membranes, particularly in their cross-linked form, combine biodegradability, mechanical integrity, and cellular safety, suggesting significant potential as sustainable GBR materials. This work introduces, for the first time, zein membranes prepared from corn crude extract for GBR in dentistry, paving the way for eco-friendly alternatives to animal-derived products.Graphic Abstract
Keywords
Periodontal diseases have increasingly affected the global population in recent decades, a trend largely attributed to lifestyle changes and rising life expectancy. These epidemiological shifts have fostered the development of advanced clinical procedures, high-performance materials, and innovative treatment strategies targeting periodontal tissues. Since the 1990s, tissue engineering has emerged as a promising field for regenerating lost tissues, introducing the concept of guided tissue regeneration in periodontology [1,2].
GBR, a variant of guided tissue regeneration, is particularly relevant to improving conditions for osteogenesis during the healing of alveolar bone defects. In this approach, a biocompatible membrane functions as a selective barrier, preventing the invasion of undesired epithelial and connective tissue cells into the defect site, thereby enabling bone-forming cells to repopulate the area [3]. Since epithelial tissues migrate significantly faster than connective or bone tissues, the concept—first proposed by Bjorn in 1961—emphasized the need to physically exclude these faster-proliferating cell types to facilitate effective tissue regeneration [4,5].
The use of membranes in GBR is a standard clinical practice. First-generation membranes, composed of non-resorbable expanded polytetrafluoroethylene (e-PTFE), were biocompatible and mechanically stable, but required surgical removal after healing. The development of second-generation membranes, based on biodegradable polymers, addressed this limitation by eliminating the need for a second surgical procedure [6]. However, inappropriate degradation rates and mechanical weaknesses in some bioresorbable materials have led to inconsistent clinical outcomes, emphasizing the importance of materials that balance structural integrity with resorption kinetics [7]. Among current materials, collagen-based membranes, especially those derived from porcine or bovine sources (e.g., Geistlich Bio-Gide®), remain the gold standard due to their excellent biocompatibility, barrier function, and capability to promote tissue integration while inducing minimal inflammatory response [8]. Synthetic membranes made from poly(lactic acid), polyglactin, and poly(lactic-co-glycolic acid) also offer controlled degradation and structural versatility [5]. Nevertheless, concerns regarding cost, residues from processing that can eventually cause immunogenicity, and environmental impact have stimulated interest in natural polymers derived from renewable sources.
Natural polymers show several advantages, including lower risk of contamination by prions or allergens derived from animal sources, environmental sustainability, and a range of mechanical and chemical properties suitable for biomedical applications [9]. Among them, zein, a class of hydrophobic prolamin proteins extracted from corn endosperm, has gained attention because of its biodegradability, biocompatibility, and excellent film-forming capability [10]. Zein consists predominantly of α-zein, which accounts for approximately 75%–85% of its total protein content. Its solubility in aqueous ethanol and ability to form stable films under physiological conditions make it a compelling candidate for the design of bioactive membranes in tissue engineering.
Recent studies have shown the potential of zein-based materials in supporting osteogenic differentiation and promoting bone regeneration. For instance, nanofibrous membranes based on zein/gelatin/nanohydroxyapatite have shown biocompatible features and induced osteogenic differentiation in human periodontal ligament stem cells [11]. Similarly, electrospun poly(aspartic acid)-modified zein nanofibers have been shown to enhance bone regeneration by promoting the differentiation of osteoblast precursor cells [12]. The osteoinductive potential of zein-based scaffolds has also been confirmed. Cross-linked zein scaffolds support the osteogenic differentiation of MC3T3-E1 cells even in the absence of additional induction factors [13]. Furthermore, the inclusion of inorganic components such as hydroxyapatite into zein matrices has been found to improve stimulate osteoblastic activity, as evidenced by bone-related gene expression [14]. Freeze-dried zein/chitosan/nanohydroxyapatite scaffold showed favorable properties for bone tissue engineering, including porosity and mechanical strength [15]. Zein/nano-hydroxyapatite 3D scaffolds have shown good biodegradability, mechanical performance and support for osteogenesis in vitro [14]. Zein/whitlockite composites loaded with levofloxacin showed antimicrobial activity alongside bone regenerative capacity, offering multifunctionality for GBR applications [16].
Zein properties have been enhanced by blending with other natural polymers, surface modification, or incorporation of bioactive agents and composites for several applications, however, there is no description of its clinical translation. In addition, there is limited evidence regarding the use of pristine zein membranes without modification. Its use as a membrane for GBR, prepared from its pristine crude extract directly from corn, without mixing with other materials, was not described in the literature, to the best of our knowledge, and can be seen as an alternative material to the biomaterials industry. Thus, this study aims to develop and characterize novel bioresorbable membranes based on zein for application in GBR, contributing to the advancement of sustainable biomaterials in periodontal therapy and bone tissue engineering. The novelty of our work relies on systematically characterizing pristine zein as a GBR candidate before introducing functional modifications.
Zein was extracted from the dry-milled corn purchased from Agrosolo (Agro Pintadão, Barretos, Brazil). Isopropyl alcohol (88% v aqueous solution) was purchased from Manipulart (Barretos, Brazil), hexane from Qhemis (São Paulo, Brazil), ethanol (70% v) from Ciclo Farma (São Paulo, Brazil) and formaldehyde solution (4% v solution) from Exodo (São Paulo, Brasil), resazurin hydrochloride was purchased from Sigma Aldrich (Saint Louis, MO, USA), Fetal Bovine Serum from Gibco (FBS, Burlington, Canada), DMEM culture medium (Dulbecco’s modified Eagle’s medium-Sigma Aldrich, Saint Louis, MO, USA), trypsin-ethylenediaminetetraacetic acid (trypsin-EDTA, Gibco, Saint Louis, MO, USA), trypan blue exclusion solution (Sigma Aldrich, Saint Louis, MO, USA), GM07492 cell line, a non-transforming fibroblastic type, was purchased from the Laboratory of Mycobacteriology and the Laboratory of Mutagenesis at FCF-UNESP, Araraquara.
2.2.1 Zeins Extraction and Purification
Dried milled corn (100 g) was soaked into 800 mL of isopropyl alcohol solution (88% vol). The mixture was homogenized using a magnetic stirrer, at 65°C for 40 min. The sample was filtered, the liquid fraction was poured into the glass Petri dish, and kept at −18°C for 24 h for protein precipitation. The resulting precipitate was mixed with hexane (1:1 mass ratio) and stirred at room temperature for five minutes to remove the lipids. This step was repeated three times and the resulting sample was dried in the Petri dish at room temperature, giving rise to the film [17,18]. The yield of the extraction process was determined using the mass of the raw material and the mass of the purified product, calculated according to the equation: yield (%) = (Product mass × 100)/raw material mass.
2.2.2 Preparation of the Membranes
Two types of membranes were prepared, (i) non-cross-linked membranes and the (ii) cross-linked membranes. For the preparation of non-cross-linked membranes, the extracted purified zein (0.5 g) was solubilized into 5 mL of a 70% (v) ethanol solution, poured into the Petri dish and dried at room temperature. The dried sample was removed from the Petri dish, washed in deionized water in order to remove residual ethanol, and kept at room temperature for drying. The dried membrane was hot pressed at 1 t at 73°C for 2 min, using a hydraulic hot press (SL10-SOLAB), with metal plates. For preparing the cross-linked membranes, the extracted purified zein (0.5 g) was solubilized into 5 mL of a 70% ethanol solution, poured into a Petri dish and dried at room temperature. After drying, the sample was removed from the Petri dish and immersed into 10 mL of 4% (v) of formaldehyde. These membranes remained in solution for 30 min and after this procedure they were washed with deionized water, dried at room temperature and pressed in a hydraulic press, following the procedure described for non-cross-linked membranes. Formaldehyde mass was estimated using the mass gain on the membrane cross-linking procedure, which was about 0.002 g/g of zein film.
Zein was characterized by Fourier Transform Infrared Spectroscopy (FTIR) using a Perkin-Elmer Spectrometer (Spectrum 100, Shelton, WA, USA) equipped with an attenuated total reflectance (ATR) zinc selenide crystal device. Measurements were performed on zein films (about 100 μm thick) with a resolution of 4 cm−1, with 16 scans for each run, ranging from 4.000 to 650 cm−1.
Thermogravimetric analysis (TGA) was performed on a Perkin-Elmer Pyris 1, equipped with a platinum cell. The samples (~5 mg) were heated at a constant rate of 10°C·min−1 from room temperature to 800°C under a nitrogen flow of 20 mL·min−1. For differential scanning calorimetry (DSC), the samples were cut into a circular shape with approximately 10 mg and analyzed in a Perkin-Elmer DSC 8000 calorimeter calibrated with indium. The analyses were carried out under nitrogen atmosphere at a 50 mL∙min−1 flow rate. The heating and cooling cycles ranged from −25°C to 150°C at a scan rate of 10°C min−1 using 40 μL aluminum standard pans.
Tensile tests were performed on Perkin Elmer’s Model 8000 DMA equipment, in traction mode, at 25°C, following ASTM D882. The load used in the test was 10 N at a rate of 0.5 N/min. The specimen’s dimensions were 7.5 mm × 25 mm × 0.1 mm. Three specimens of each sample were tested and the results represent the average and standard deviation for each sample set.
Non-cross-linked and cross-linked membranes with dimensions 1.6 cm × 1.0 cm were immersed into a beaker containing 0.9% saline solution (Sonobiol-9 g/L, pH 7.0), and removed after periods of 0, 7, 15 and 30 days. The membranes were washed in deionized water, dried at room temperature to constant mass (24 h) and weighed in an analytical balance (SHIMADZU, Kyoto, Japan, model AY-220—220 g × 0.1 mg capacity) to determine their mass loss. Three specimens of each sample were tested and the results represent the average and standard deviation for each sample set.
Cytotoxicity assay
The cytotoxicity assay was performed using resazurin hydrochloride as the revealing substance, which has redox (oxidation-reduction) potential, with colorimetric change and fluorescence indicator in response to cell metabolism, following ISO10993-12.
Sample preparation
Membranes (1.25 cm2) were sterilized using ultraviolet light for 30 min on each surface, inserted into 0.5 mL of DMEM culture medium supplemented with 10% vol FBS, and kept for 24 h at 37°C under agitation in order to prepare the eluate. The eluates (named 100%) were used to perform the cytotoxicity assays.
Cell culture and cytotoxicity assay (cell viability)
The GM07492 cells were seeded into 75 cm2 culture bottles, using DMEM culture medium supplemented with FBS and incubated at 37°C, 5% CO2 atmosphere for 24 h. Cells were treated with 3 mL of 0.25% trypsin-EDTA, centrifuged at 1500 rpm for 4 min and counted in a Newbauer chamber using 0.004% trypan blue exclusion solution. The cells (1.5 × 104 cells/well) were seeded into the 96-well microplates and incubated at 37°C, 5% CO2 atmosphere for 24 h to allow cells to adhere. The samples (eluates) were diluted (from 100% to 0.78%) into the microplate wells and incubated at the standard conditions for 24 h. The culture medium was removed, and 50 μL of 0.4 mm resazurin hydrochloride was added to each plate-well. Fluorescence reading was performed in a microplate reader on excitation and emission filters at wavelengths of 530 and 590 nm, respectively. All the tests were performed in triplicate in three independent experiments. The test was carried out in triplicate with three independent experiments. The results represent the average and standard deviation for each sample set.
Cell migration and invasion assays
The membranes were sterilized using ultraviolet light (UV) for 30 min on each surface. The cells were incubated into 75 cm2 bottles, filled with DMEM supplemented with 10% vol FBS at 37°C, 5% CO2 atmosphere for 24 h. The cells were collected from the culture bottle using 3 mL of 0.25% trypsin-EDTA, centrifuged at 1500 rpm for 4 min and counted in a Newbauer chamber, using a 0.004% trypan blue exclusion solution. To verify cell migration, the membrane was inserted in the apical compartment of the Transwell® device, followed by the deposition of the cell suspension on the top of the membrane, in order to follow their migration across the membrane. The cell concentration was 4.5 × 104 cells/well into the 24-well plate. The culture medium in the well was supplemented with fetal bovine serum (FBS) and the culture medium in the cells compartment was free of FBS. The well loaded only with the cells and culture medium was used as the control sample for cell growth. The plate was incubated for 24 h in an oven with 5% CO2 atmosphere, at 37°C. Cell invasion was evaluated by examining the presence or absence of cells on the lower surface of the membrane and at the bottom of the well, using a DM2500 transmission/reflection optical microscope (Leica Microsystems). Migration was confirmed by the presence of cells adhered to the lower surface of the membrane. The test was carried out in triplicate and the results represent the average and standard deviation for each sample set.
The experiments were performed in replicates as described in each respective section above, and the results represent the average and standard deviation for each sample set.
3.1 Extraction of Zeins and the Preparation of the Membranes
The process of zein extraction from dry milled corn and the products of each step are shown in Fig. 1, as follows: the dried milled corn (a), the mixture with the isopropyl alcohol for solubilizing the protein from corn (b), followed by paper filtration (c). The filtrate was cooled (d) leading the protein to precipitate and separated from the solvent (e). The protein was then dissolved in ethanol (f), dried (g), hot pressed (h) and cut for the tests. The extraction process was relatively easy and the yield was 5.11 (±0.13)% wt, value in agreement with the literature [17]. Zein membrane and its cross-linked counterpart displayed similar appearance.
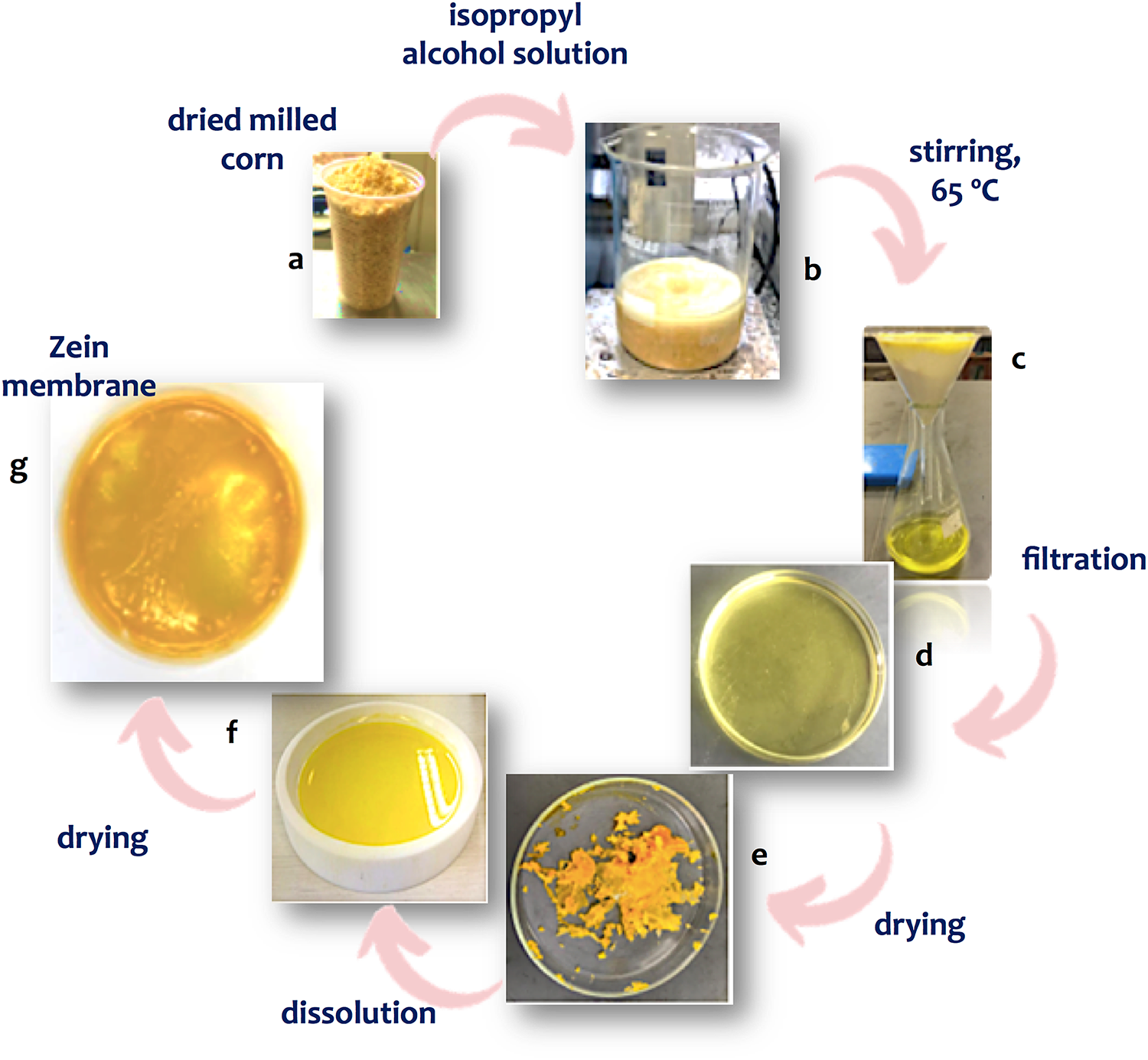
Figure 1: Pictures of zein extraction process, showing the dried milled corn, the mixture with the isopropyl alcohol, filtrated zein solution, precipitated protein, protein dissolved in ethanol and the dried hot-pressed film
3.2 Characterization of Cross-Linked and Non-Cross-Linked Membranes by FTIR
The FTIR spectrum of the cross-linked and non-cross-linked membranes are shown in Fig. 2. In the spectrum referring to the non-cross-linked sample, a typical corn prolamine profile can be observed in the bands. The broad band at 3300 cm−1 and the strong bands at about 1100 cm−1 correspond to the vibration of hydroxyl groups (O–H), and the bands in the range of 2924 to 2851 cm−1 indicate the aliphatic groups CH2 and CH3. The bands at1600 cm−1 indicate the presence of the carboxyl group (C=O) and those in the range of 1540 to 1450 cm−1 correspond to the vibration of the amide bonds, typical of proteins. These bands, particularly, are associated with the C=O elongation vibration, directly related to the conformation of the main chemical structure of the protein. The amide in the sample is composed of many absorption bands, which suggests a high proportion of α-zeins. In the spectrum of cross-linked membranes, in addition to the typical protein bands, that at about 1743 cm−1, indicating the C=O group from the imide bonds, indicating the success of the cross-linking reaction. The band at 1380 cm−1 corresponds to the methylene bridges (C2H4O) resulting from the cross-linking and the band at 1160 cm−1 corresponds to C–C stretching vibration, also suggesting the bridge formation by the cross-linking. In addition, the band at 1100 cm−1 almost disappears, and that band at 3300 cm−1 displayed decreased intensity, also indicating the cross-linking reaction. The FTIR spectra indicated the success of zein extraction, as well as the cross-linking reaction.
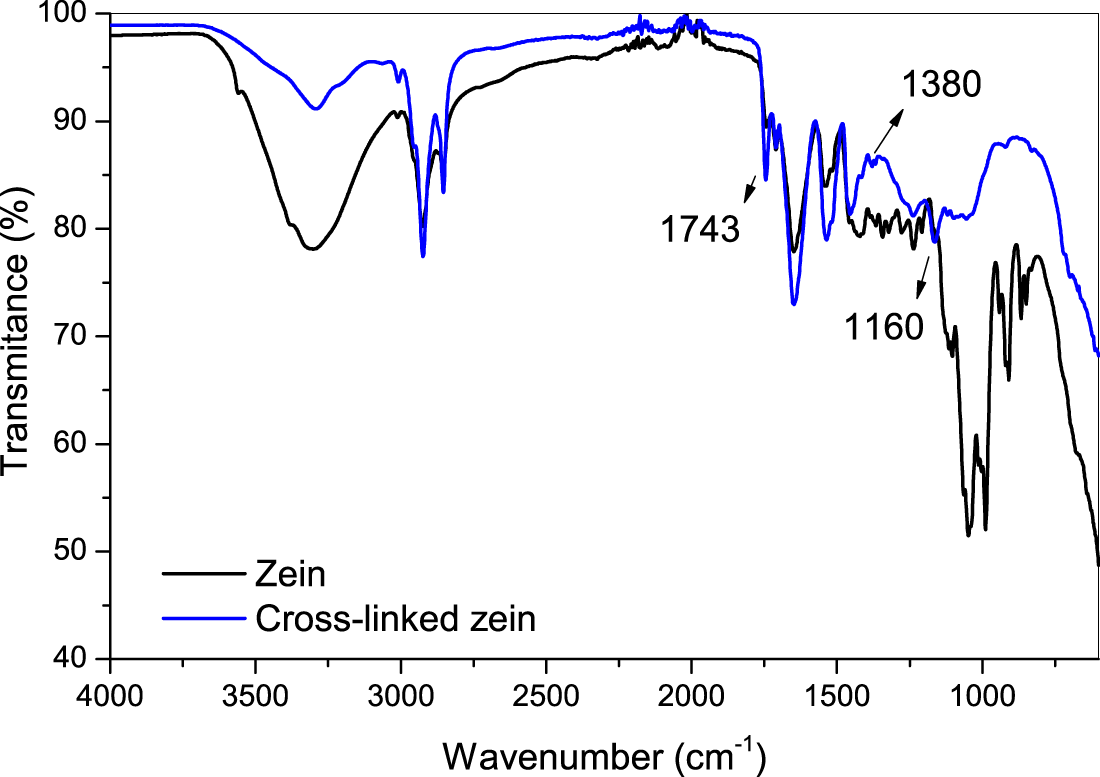
Figure 2: FTIR spectrum of zein and cross-linked zein samples
Thermal properties
TGA and DSC results are shown in Fig. 3. The results of TGA, Fig. 3A, indicate for both samples the weight loss is related to the water evaporation at the temperatures ranging from 25°C to 120°C, and the zein thermal degradation in the range of 230°C to 415°C. For both samples, the main weight loss (Tonset) was at 350°C, which can be attributed to the zein main chain depolymerization. The solid residual was about 15 and 20% wt, at 600°C, for zein and cross-linked zein, respectively. The mass loss process at 150°C–270°C, corresponds possibly to the loss of the isopropanol which was kept entrapped within the zein structure even after drying and is lost at lower temperatures than zein because of its lower molecular weight. This peak is much more evident for the cross-linked sample. DSC profiles in Fig. 3B indicated, for both samples, no transition temperature (Tg) until 145°C. This result is in agreement with the sample’s nature, i.e., a protein, in which their amino acids side chains interact with each other, forming a strong hydrogen bonding network that is not affected by heating (at the tested conditions). These results indicated the stability of the membrane at the body temperature.
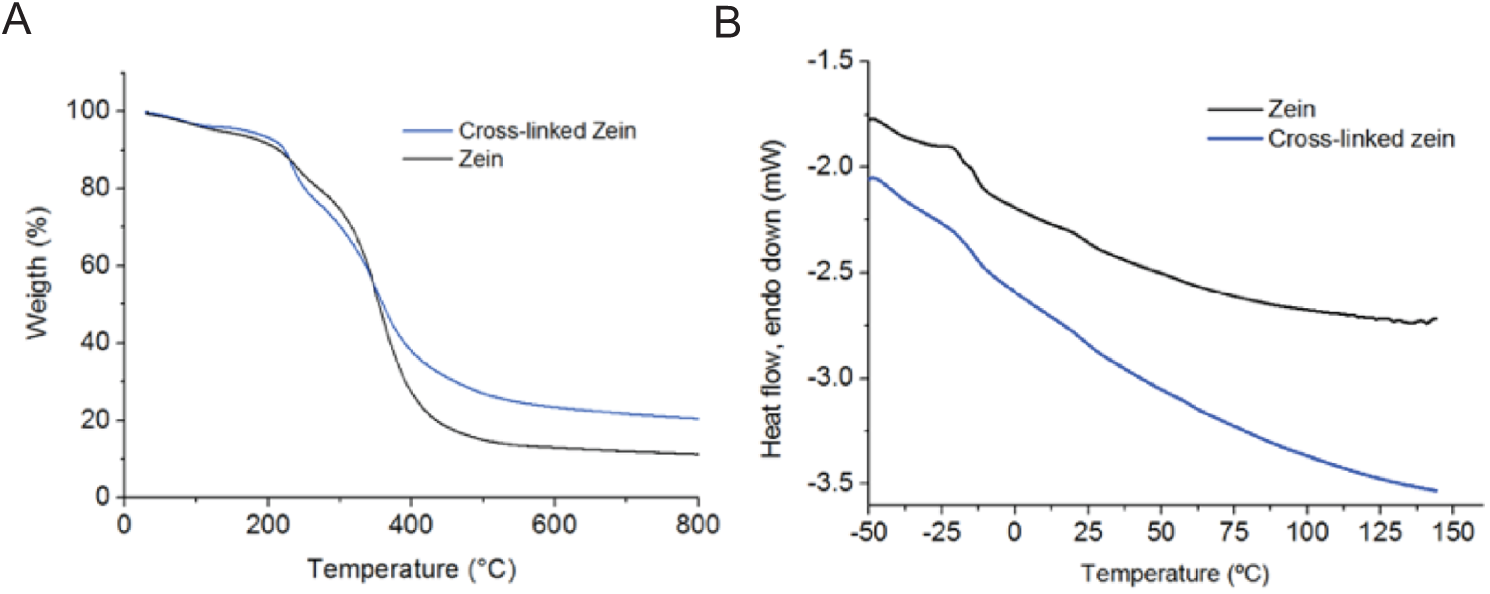
Figure 3: TGA and DSC profile of zein and cross-linked zein membranes
The results of the mechanical tests for tensile strength and deformation of cross-linked and non-cross-linked membranes are shown in Fig. 4 and Table 1. The cross-linked membrane displayed higher tensile strength, about 5 MPa, when compared to the non cross-linked counterpart, about 2.5 MPa, as expected. The greater tensile strength is the response of the interconnected macromolecules by the cross-linking process, to the applied mechanical forces. The deformation rate results, about three times lower for the cross-linked sample (0.03%), when compared to the non cross-linked counterpart (0.09%), can be also explained by the cross-liking effect on the macromolecules network.
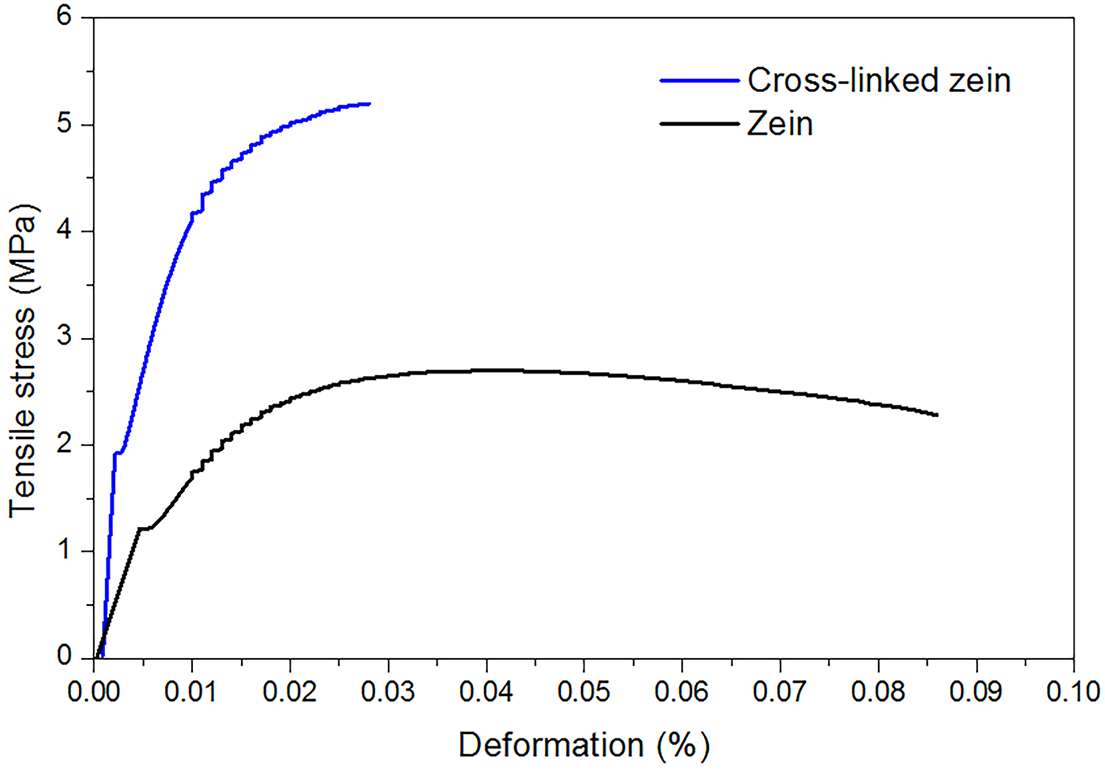
Figure 4: Tensile strength and deformation rate of zein and cross-linked zein samples
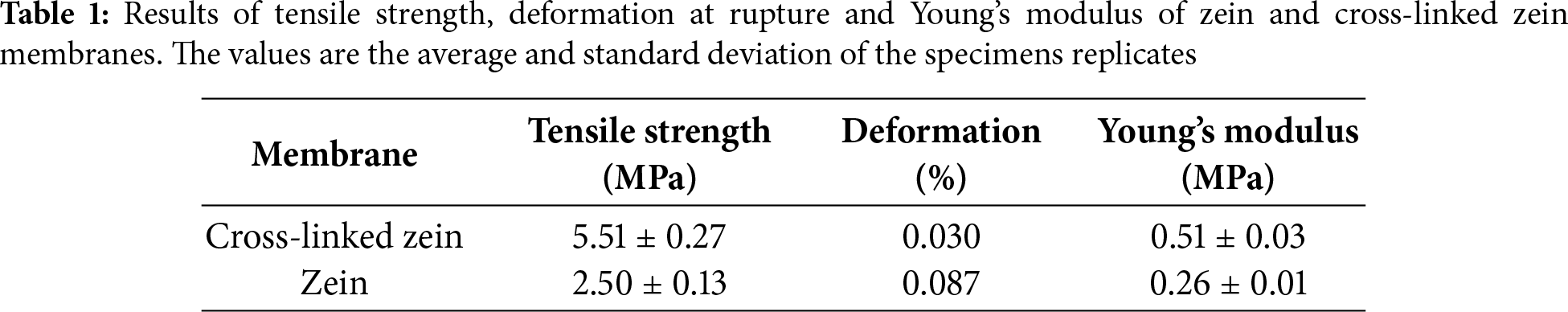
Non-cross-linked and cross-linked membranes were submitted to the hydrolytic degradation tests in physiological medium, and the results are shown in Fig. 5. Mass loss was followed for seven days for non-cross-linked samples. Considering their initial masses as 100% wt, after removal of the membranes from the physiological medium, the mass loss was approximately 5%, however, the membranes clearly disintegrated, indicating that they are not ideal for the proposed application. Cross-linked samples’ mass loss was followed for a period of time of 30 days, with measurements at 0-, 15- and 30-day time points. Considering its initial masses as 100%, after removal from the physiological medium, the samples lost approximately 4, 7 and 43% of their mass in 7, 15 and 30 days time points, respectively. After 30 days time point, the membrane started to disintegrate and the test was stopped. This result shows that the cross-linked membrane is acceptable for the proposal of this work, based on the disintegration time in physiological in vitro conditions and on its loss of mass in a period of time compatible with bone repair [7].
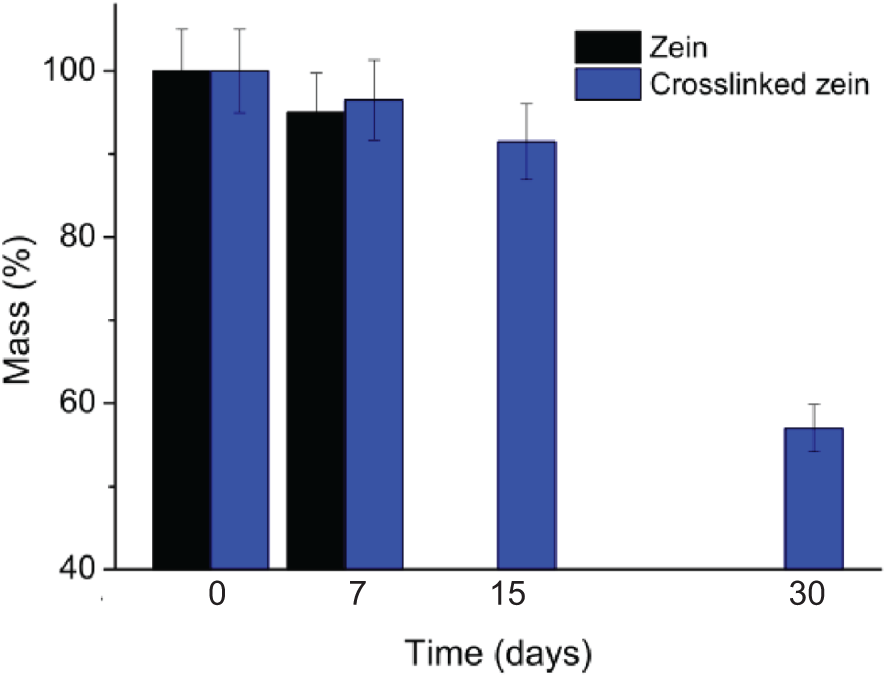
Figure 5: Results of the in vitro hydrolytic degradation of the samples
The results of cell viability assessment are shown in Fig. 6. The cell viability is expressed as the percentage of viable GM07492 cells treated with different concentrations of membrane eluates. The concentrations evaluated did not induce a statistically significant reduction (p ≤ 0.05) in cell viability compared to the negative control. ISO 10993-5 establishes that a substance is cytotoxic if it kills more than 30% of the cells. Here, the results are much lower, and no significant values were found, when compared to the negative control sample, which means a cell viability of about 100%, and about zero cytotoxic effect. The results showed a statistically significant reduction in cell viability of cultures treated with 20% DMSO (positive control), ensuring the sensitivity of the test system (25% cell viability). In general, these results indicated that zein did not show toxicity under the tested conditions. In addition, the results indicated that the formaldehyde used for cross-linking zein did not leach and did not lead to any toxic effect on the cells. This result aligns with expectations, since it refers to a protein of natural origin, extracted from a food product [19,20].
Figure 6: Cell viability assay using GM07492 cells treated with zein cross-linked membrane. Values are shown as mean and standard deviation. NC: negative control (DMEM culture medium, supplemented with 10% fetal bovine serum, 100% cell viability); PC: Positive control (20% DMSO; 25% cell viability), *: Significantly different from the negative control (p < 0.05)
3.6 In Vitro Cell Migration and Invasion Assay
The capability of the membranes to act as a physical barrier was assessed using a transwell migration and invasion assay, to investigate if they were capable of inhibiting the cell migration, once the purpose of the work is to prepare a barrier for bone grafts. GM07492 fibroblasts were seeded in the upper chamber, with the zein membranes positioned as the separating barrier between the cells and the chemoattractant-containing lower chamber. After 24 h of incubation, the cells were adhered to the bottom in the control sample (Fig. 7A) and no cells were observed for cross-linked zein sample (Fig. 7B) as confirmed by optical microscopy. These results demonstrate that the cross-linked zein membranes effectively prevented cell migration, fulfilling one of the essential requirements for GBR membranes, which is to inhibit the early invasion of fibroblasts and epithelial cells into the defect site. By maintaining this barrier function, the membranes create a protected environment for osteogenic cells to repopulate and support bone healing. Fibroblasts were used in this test because in therapeutic applications, the main aim of the membrane is to avoid the migration of soft tissue into the hard tissue.

Figure 7: Optical microscopy images of cell migration and invasion assays showing the bottom of culture plate from the positive control (A) and cross-linked zein membrane (B)
The health field’s growing demand for research and development of new biomaterials based on natural sources to replace synthetic and animal counterparts encourages the search for new bioactive, biodegradable, and non-toxic polymers for clinical applications. Here we showed the preparation of zein-based membranes, from pristine corn-derived zein as a renewable and biocompatible alternative for GBR. Several studies have explored zein composites for bone tissue engineering, but none describe pure or pristine zein membranes used directly as GBR barrier membranes, as proposed in this work. The goal of this work was to evaluate if zein membranes, without blending with other polymers or chemical modification, could provide the key properties required of a GBR membrane: mechanical strength, degradation in physiological conditions, cytocompatibility, and effectiveness as a physical barrier against undesirable cell migration. The results demonstrated that cross-linked zein membranes meet many of these criteria, indicating their promising potential for future translational applications.
The extraction process yielded zein with an efficiency of 5.11 ± 0.13% wt, consistent with reported yields for corn prolamins [1,17]. Fourier-transform infrared spectroscopy (FTIR) confirmed the preservation of the protein’s structural motifs and verified cross-linking through the emergence of imide-associated carbonyl bands. In addition, the bands at about 2800–2900 cm−1, related to the CH2 chemical group corresponding to the amino acids side chain keep stable in size. Such band can also result from the vibrations of methylene after formaldehyde cross-linking, thus suggesting it small contribution to the zein structure. Cross-linking proved pivotal for enhancing the membrane’s structural properties. Mechanical testing revealed that tensile strength doubled from approximately 2.5 MPa in non-cross-linked membranes to 5 MPa in cross-linked ones, while deformation decreased threefold. This result is in the range of the gold standard collagen membrane [21], thus, indicating its mechanically adequate performance for the suggested application. TGA and DSC data indicated the membranes’ thermal stability well above physiological temperatures, with major degradation only occurring above 270°C and an onset degradation temperature (Tonset) of 350°C. This behaviour is consistent with zein’s tightly packed hydrophobic amino acid network [10] and confirms that these membranes would not prematurely lose their structural function in vivo. The differences in thermal stability between zein and cross-linked zein were modest, however, the shift in degradation onset suggests improved structural stability, which could translate into slightly prolonged membrane integrity under physiological conditions, in agreement with the results of the hydrolytic degradation tests.
Hydrolytic degradation tests further differentiated the performance of the two formulations. Non-cross-linked membranes disintegrated rapidly, losing structural integrity within seven days, which would be insufficient for clinical GBR timelines. By contrast, cross-linked membranes exhibited a gradual and controlled resorption, losing 4%, 7%, and 43% of their mass at 7, 15, and 30 days, respectively. This resorption profile aligns with the properties of natural source-based GBR, however, the membranes must act as a barrier during the early phase of bone regeneration, which is normally longer. In general, this behaviour of the natural source-based GBR represents a limitation of these materials, including zein [7]. Tensile strength measurements were not performed during this study. To strengthen this point, we added a note on the importance of future studies to correlate degradation with mechanical retention and functional barrier performance.
The biological evaluation reinforced zein’s suitability as a GBR material. Cytotoxicity assays showed no reduction in cell viability for any tested membrane, confirming that zein is inherently biocompatible, consistent with its status as a dietary protein [20]. Furthermore, in vitro transwell migration assays demonstrated that the membranes effectively prevented cell passage, validating their functionality as physical barriers that could prevent epithelial and fibroblastic cell infiltration into bone defects—a critical feature first recognised in the conceptualisation of GBR by Bjorn [4] and later demonstrated by Kunrath et al. [5]. The use of formaldehyde presents safety concerns due to its carcinogenicity, which can limit its clinical applicability. Here, formaldehyde was used because it is a classical and effective cross-linker, widely employed in early-stage studies of protein-based biomaterials, and thus provides a useful baseline for assessing how cross-linking can alter zein properties, intended as a proof of concept. More biocompatible cross-linkers, such as glyceraldehyde and genipin, may achieve similar improvements in stability while offering a significantly safer profile for biomedical use.
When compared to the current gold standard, collagen membranes [8], zein membranes offer several compelling advantages. Being plant-derived, they eliminate the risks of zoonotic pathogen transmission, prion contamination, and immunogenicity associated with animal products, while also addressing environmental concerns linked to animal-derived and petroleum-based materials [22,23]. Although further optimisation is required, particularly regarding mechanical tuning and degradation rates, the present findings demonstrate that pristine zein membranes show considerable promise as sustainable, safe, and functional alternatives for GBR [9,24]. The reduced animal dependence and the reduction of risks [9] represent important positive aspects of zein as a biomaterial, however, the production scalability and life cycle analyses [10,25] remain as the main challenge for the development of commercial market products from zein. In general, by demonstrating the feasibility of pristine zein membranes for GBR, this study opens a new avenue in the field of biodegradable and plant-derived biomaterials for periodontal therapy. The findings indicate that cross-linked zein membranes display potential to meet clinical requirements while addressing environmental concerns based on their renewable source. However, tests such as enzymatic degradation and analysis of mechanical property retention, extended cytotoxicity tests, ultrastructure (electronic microscopy and pore size determination), wettability and the dimensional stability as well as stability under industrial sterilization techniques ethylene oxide, gamma/e-beam can be useful to reinforce translation to GBR. The interest in GBR membranes is high, thus several approaches have been also tested to improve the quality of collagen-based membranes [26,27].
This study showed the successful development and characterization of pristine corn-derived zein membranes, for potential application in GBR. The preparation method generated homogeneous, and flexible membranes. FTIR confirmed the preservation of zein’s protein structure and verified the formation of imide bonds in the cross-linked samples. Thermal analysis (TGA/DSC) revealed good thermal stability up to ~270°C, suggesting structural robustness under physiological conditions. The cross-linked membranes exhibited superior mechanical performance, with tensile strength nearly double that of non-cross-linked counterparts (5 MPa vs. 2.5 MPa), and lower deformation rates, which are desirable features as barrier membranes during healing. In vitro degradation tests showed that cross-linked membranes degraded gradually, losing ~43% of their mass in 30 days, a timeframe compatible with early stages of bone regeneration, while non-cross-linked membranes disintegrated rapidly, indicating that cross-linking is crucial for clinical applicability. Biological assays confirmed the biocompatibility of the membranes and the migration and invasion assays in the Transwell® system showed that the membranes effectively prevented cell migration, fulfilling their barrier function. Collectively, these results demonstrate that zein membranes meet several key requirements for GBR, including biocompatibility, barrier integrity, tunable degradation, and adequate mechanical stability.
Acknowledgement: This work was supported by the São Paulo Research Foundation, FAPESP facilities [research project funding 2019-25318-0] and Conselho Nacional de Desenvolvimento Científico e Tecnológico-CNPq, grant number 305518/2023-2.
Funding Statement: This research was funded by São Paulo Research Foundation, FAPESP [research project funding 2019-25318-0 and 2017-18782-6], and Conselho Nacional de Desenvolvimento Científico e Tecnológico-CNPq, grant number 305518/2023-2.
Author Contributions: Cristiane Michele Alves de Oliveira, Formal analysis, Investigation, Methodology; Bruna Carolina Dorm, Methodology, Writing—original draft; Antonio José Felix Carvalho, Visualization, Writing—review & editing; Deliane da Silva Cabral, Formal analysis, Methodology; Flávia Aparecida Resende Nogueira, Methodology, Writing—review & editing; Nádia Andrade Aleixo, Data curation, Investigation, Methodology; Mônica Rosas Costa Iemma, Investigation, Methodology; Eliane Trovatti, Conceptualization, Funding acquisition, Supervision, Writing—review & editing. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Data available on request from the authors.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
References
1. Langer R, Vacanti JP. Tissue engineering. Science. 1993;260(5110):920–6. [Google Scholar] [PubMed]
2. Dabra S, Dasgupta S, Joshi V. Tissue engineering in periodontology: a future perspective. J Indian Soc Periodontol. 2012;16(3):329–34. [Google Scholar]
3. Sharma R, Durga K, Wadhwa H, Gupta SK. Methodological appraisal and evidence-based insights on guided tissue regeneration in endodontic surgery: an umbrella review of systematic reviews and meta-analyses. Aust Endod J. 2025;54(4):536. doi:10.1111/aej.12967. [Google Scholar] [PubMed] [CrossRef]
4. Dimitriou R, Mataliotakis GI, Calori GM, Giannoudis PV. The role of barrier membranes for guided bone regeneration and restoration of large bone defects: current experimental and clinical evidence. BMC Med. 2012;10:81. doi:10.1186/1741-7015-10-81. [Google Scholar] [PubMed] [CrossRef]
5. Kunrath MF, Magrin GL, Zorzo CS, Rigotto I, Aludden H, Dahlin C. Membranes for periodontal and bone regeneration: everything you need to know. J Periodontal Res. 2025;31(3):265. doi:10.1111/jre.70005. [Google Scholar] [PubMed] [CrossRef]
6. Hämmerle CHF, Jung RE, Feloutzis A. A systematic review of the survival of implants in bone sites augmented with barrier membranes (guided bone regeneration) in partially edentulous patients. J Clin Periodontol. 2003;29:226–31. doi:10.1034/j.1600-051X.29.s3.14.x. [Google Scholar] [PubMed] [CrossRef]
7. Wang D, Zhou X, Cao H, Zhang H, Wang D, Guo J, et al. Barrier membranes for periodontal guided bone regeneration: a potential therapeutic strategy. Front Mater. 2023;10:1220420. doi:10.3389/fmats.2023.1220420. [Google Scholar] [CrossRef]
8. Anderson TJ, Lamsal BP. Development of new method for extraction of α-zein from corn gluten meal using different solvents. Cereal Chem. 2011;88:356–62. doi:10.1094/CCHEM-08-10-0117. [Google Scholar] [CrossRef]
9. Manivannan RK, Sharma N, Kumar V, Jayaraj I, Vimal S, Umesh M. A comprehensive review on natural macromolecular biopolymers for biomedical applications: recent advancements, current challenges, and future outlooks. Carbohydr Polym Technol Appl. 2024;8:100536. doi:10.1016/j.carpta.2024.100536. [Google Scholar] [CrossRef]
10. Shukla R, Cheryan M. Zein: the industrial protein from corn. Ind Crops Prod. 2001;13(3):171–92. doi:10.1016/s0926-6690(00)00064-9. [Google Scholar] [CrossRef]
11. Ou Q, Miao Y, Yang F, Lin X, Zhang L, Wang Y. Zein/gelatin/nanohydroxyapatite nanofibrous scaffolds are biocompatible and promote osteogenic differentiation of human periodontal ligament stem cells. Biomater Sci. 2019;7:1973–83. doi:10.1039/c8bm01653d. [Google Scholar] [PubMed] [CrossRef]
12. Liu Y, Miao Y, Qin F, Cao C, Yu X, Wu Y, et al. Electrospun poly(aspartic acid)-modified zein nanofibers for promoting bone regeneration. Int J Nanomed. 2019;14:9497–512. doi:10.2147/IJN.S224265. [Google Scholar] [PubMed] [CrossRef]
13. Turner JC, Collins G, Blaber EA, Almeida EAC, Arinzeh TL. Evaluating the cytocompatibility and differentiation of bone progenitors on electrospun zein scaffolds. J Tissue Eng Regen Med. 2020;14(1):173–85. doi:10.1002/term.2984. [Google Scholar] [PubMed] [CrossRef]
14. Zaersabet M, Salehi Z, Hadavi M, Talesh SS, Rastgoo NF. Development and evaluation of bioactive 3D zein and zein/nano-hydroxyapatite scaffolds for bone tissue engineering application. J Eng Med. 2022;236(6):785–93. doi:10.1177/09544119221090726. [Google Scholar] [PubMed] [CrossRef]
15. Shahbazarab Z, Teimouri A, Chermahini A, Azadi M. Fabrication and characterization of nanobiocomposite scaffold of zein/chitosan/nanohydroxyapatite prepared by freeze-drying method for bone tissue engineering. Int J Biol Macromol. 2017;108:1017–27. doi:10.1016/j.ijbiomac.2017.11.017. [Google Scholar] [PubMed] [CrossRef]
16. Lin X, Wang Y, Liu L, Du X, Wang W, Guo S, et al. Enhanced bone regeneration by osteoinductive and angiogenic zein/whitlockite composite scaffolds loaded with levofloxacin. RSC Adv. 2024;14:14470–9. doi:10.1039/d4ra00772g. [Google Scholar] [PubMed] [CrossRef]
17. Carter R, Reck DR, inventors; Nutrilite Products Inc., assignee. Low temperature solvent extraction process for producing high purity zein. United States patent US 3,535,305. 1970 Oct 20. [Google Scholar]
18. Tan H, Zhou H, Guo T, Zhou Y, Wang S, Zhang Y, et al. Investigating the effects of zein preparation conditions on its structure, functional properties and zearalenone contamination. Food Control. 2024;159(1):110297. doi:10.1016/j.foodcont.2024.110297. [Google Scholar] [CrossRef]
19. Arbade GK, Hanuman S, Nune M, Katiki AB, Arbade AF, Gajanan K, et al. Zein-based polymeric biomaterials: an overview for applications in soft tissue engineering. Int J Polym Mater Polym Biomater. 2024;74(9):812–28. doi:10.1080/00914037.2024.2374381. [Google Scholar] [CrossRef]
20. Bisharat L, Berardi A, Perinelli DR, Bonacucina G, Casettari L, Cespi M, et al. Aggregation of zein in aqueous ethanol dispersions: effect on cast film properties. Int J Biol Macromol. 2017;106:360–8. doi:10.1016/j.ijbiomac.2017.08.024. [Google Scholar] [PubMed] [CrossRef]
21. Raz P, Brosh T, Ronen G, Tal H. Tensile properties of three selected collagen membranes. Biomed Res Int. 2019;2019:5163603. doi:10.1155/2019/5163603. [Google Scholar] [PubMed] [CrossRef]
22. Moutasim K, Nystrom M, Thomas G. Cell migration and invasion assays. Methods Mol Biol. 2011;731:333–43. doi:10.1007/978-1-61779-080-5_27. [Google Scholar] [PubMed] [CrossRef]
23. Rahman M, Dip TM, Haase T, Truong YB, Le TC, Houshyar S. Fabrication of zein-based fibrous scaffolds for biomedical applications—a review. Macromol Mater Eng. 2023;308:2300175. doi:10.1002/mame.202300175. [Google Scholar] [CrossRef]
24. Tivano F, Chiono V. Zein as a renewable material for the preparation of green nanoparticles for drug delivery. Front Biomater Sci. 2023;2:1–21. doi:10.3389/fbiom.2023.1156403. [Google Scholar] [CrossRef]
25. Tortorella S, Maturi M, Vetri Buratti V, Vozzolo G, Locatelli E, Sambri L, et al. Zein as a versatile biopolymer: different shapes for different biomedical applications. RSC Adv. 2021;11(62):39004–26. doi:10.1039/d1ra07424e. [Google Scholar] [PubMed] [CrossRef]
26. Takallu S, Karimi Z, Khorshidi H, Bazargani A, Bigham-Sadegh A, Nezhad ST, et al. Multifunctional three-layer collagen-based membrane for periodontal guided tissue regeneration. ACS Biomater Sci Eng. 2025;3(5):281. doi:10.1021/acsbiomaterials.5c00613. [Google Scholar] [PubMed] [CrossRef]
27. Valamvanos TF, Dereka X, Katifelis H, Gazouli M, Lagopati N. Recent advances in scaffolds for guided bone regeneration. Biomimetics. 2024;9(3):153. doi:10.3390/biomimetics9030153. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools