 Open Access
Open Access
REVIEW
Opportunities and challenges of CD47-targeted therapy in cancer immunotherapy
1 Key Laboratory for Translational Medicine, The First Affiliated Hospital, Huzhou University School of Medicine, Huzhou, 313000, China
2 Department of Hematology, Puyang Youtian General Hospital, Puyang, 457001, China
3 Department of Medicine, Moores Cancer Center, Sanford Stem Cell Institute, University of California San Diego, La Jolla, San Diego, 92093, USA
* Corresponding Authors: Qiuqiang Chen, ; Wenxue Ma,
(This article belongs to the Special Issue: Advances in Cancer Immunotherapy)
Oncology Research 2024, 32(1), 49-60. https://doi.org/10.32604/or.2023.042383
Received 28 May 2023; Accepted 09 August 2023; Issue published 15 November 2023
Abstract
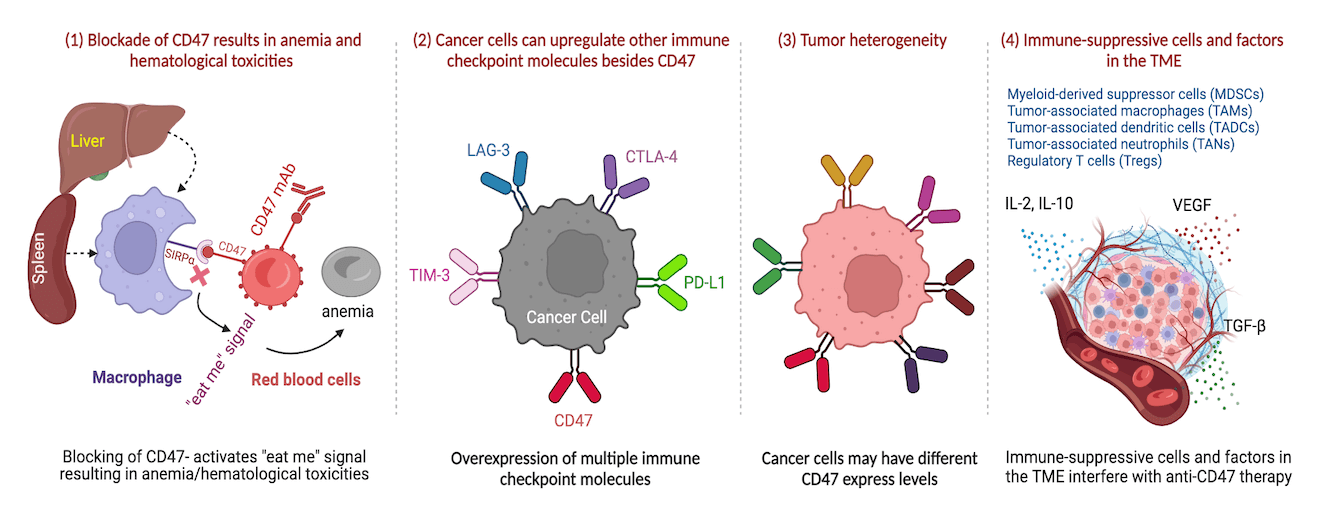
Cancer immunotherapy has emerged as a promising strategy for the treatment of cancer, with the tumor microenvironment (TME) playing a pivotal role in modulating the immune response. CD47, a cell surface protein, has been identified as a crucial regulator of the TME and a potential therapeutic target for cancer therapy. However, the precise functions and implications of CD47 in the TME during immunotherapy for cancer patients remain incompletely understood. This comprehensive review aims to provide an overview of CD47’s multifaced role in TME regulation and immune evasion, elucidating its impact on various types of immunotherapy outcomes, including checkpoint inhibitors and CAR T-cell therapy. Notably, CD47-targeted therapies offer a promising avenue for improving cancer treatment outcomes, especially when combined with other immunotherapeutic approaches. The review also discusses current and potential CD47-targeted therapies being explored for cancer treatment and delves into the associated challenges and opportunities inherent in targeting CD47. Despite the demonstrated effectiveness of CD47-targeted therapies, there are potential problems, including unintended effects on healthy cells, hematological toxicities, and the development if resistance. Consequently, further research efforts are warranted to fully understand the underlying mechanisms of resistance and to optimize CD47-targeted therapies through innovative combination approaches, ultimately improving cancer treatment outcomes. Overall, this comprehensive review highlights the significance of CD47 as a promising target for cancer immunotherapy and provides valuable insight into the challenges and opportunities in developing effective CD47-targeted therapies for cancer treatment.Graphic Abstract
Keywords
Cite This Article
 Copyright © 2024 The Author(s). Published by Tech Science Press.
Copyright © 2024 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools