 Open Access
Open Access
ARTICLE
Prognostic Value of Neutrophil-to-Lymphocyte Ratio in Locally Advanced Rectal Cancer Treated with Neoadjuvant Concurrent Chemoradiotherapy and Robotic-Assisted Resection
1 Graduate Institute of Clinical Medicine, College of Medicine; Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
2 Division of Colorectal Surgery, Department of Surgery, Kaohsiung Medical University Hospital, Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
3 Department of Surgery, Faculty of Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
4 Department of Surgery, Faculty of Post-Baccalaureate Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
5 Division of Trauma and Surgical Critical Care, Department of Surgery, Kaohsiung Medical University Hospital, Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
6 Department of Emergency Medicine, Faculty of Post-Baccalaureate Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
7 Graduate Institute of Injury Prevention and Control, College of Public Health, Taipei Medical University, Taipei, 11031, Taiwan
8 Division of Colorectal Surgery, Department of Surgery, Kaohsiung Medical University Gangshan Hospital, Kaohsiung, 820, Taiwan
9 Department of Surgery, Division of Colorectal Surgery, Kaohsiung Municipal Hsiaokang Hospital, Kaohsiung, 812, Taiwan
10 Graduate Institute of Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
11 Center for Cancer Research, Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
12 Center for Disease Multi-omincs Research, Kaohsiung Medical University, Kaohsiung, 80708, Taiwan
* Corresponding Author: Jaw-Yuan Wang. Email:
(This article belongs to the Special Issue: Advances and Innovations in Colorectal Cancer Research and Treatment)
Oncology Research 2026, 34(3), 17 https://doi.org/10.32604/or.2025.069397
Received 22 June 2025; Accepted 31 October 2025; Issue published 24 February 2026
Abstract
Background: The long-term outcomes of robotic-assisted surgery and the prognostic significance of the pretreatment neutrophil-to-lymphocyte ratio (NLR) in locally advanced rectal cancer (LARC) remain uncertain. This study aimed to assess the long-term outcomes of patients with LARC undergoing robotic-assisted surgery and to determine the prognostic value of pretreatment NLR. Methods: We retrospectively reviewed 252 patients with LARC who were treated at a single medical center in Taiwan between January 2012 and January 2023. All patients underwent neoadjuvant concurrent chemoradiotherapy (CRT) followed by robotic-assisted surgery with total mesorectal excision (TME). Patients were stratified into four groups on the basis of pretreatment NLRs and carcinoembryonic antigen (CEA) levels. Univariate and multivariate analyses were conducted to identify prognostic indicators for overall survival (OS) and disease-free survival (DFS). Results: Patients with a pretreatment NLR of ≥3.2 exhibited significantly worse OS and DFS compared with those with an NLR of <3.2 (OS: 94.4 vs. 116.5 months, p = 0.001; DFS: 78.8 vs. 101.7 months, p = 0.003). Group A exhibited the poorest prognosis, whereas Group D had the most favorable outcomes. Multivariate analysis revealed NLR ≥ 3.2 as an independent predictor of poor OS (hazard ratio [HR] = 2.306, 95% CI: 1.149–3.747; p = 0.001) and DFS (HR = 2.055, 95% CI: 1.341–3.148; p = 0.001). Conclusion: Neoadjuvant concurrent CRT followed by robotic-assisted TME is an effective treatment strategy for LARC. A higher pretreatment NLR (≥3.2) independently predicted worse OS and DFS. Stratification using the NLR in combination with CEA levels may enhance prognostic accuracy for patients undergoing robotic-assisted surgery for LARC.Keywords
Supplementary Material
Supplementary Material FileNeoadjuvant concurrent chemoradiotherapy (CRT) is widely used in the treatment of locally advanced rectal cancer (LARC) [1–3]. Surgical resection of LARC remains technically demanding because of the narrow pelvic space and complex anatomy [4–6]. Furthermore, local recurrence rates following surgery can reach 15%–31% [6,7]. In consideration of these challenges, neoadjuvant radiotherapy was introduced as a means of reducing tumor volume and improving local recurrence control [5,8]. When combined with total mesorectal excision (TME), the local recurrence rate associated with neoadjuvant radiotherapy can be significantly reduced to 6%–7% [9,10]. Advances in surgical technology have further improved outcomes; robotic systems have been developed that offer high-definition, three-dimensional magnified visualization, along with multidirectional instruments capable of rotation at various angles. These features enable more precise resections and have been associated with improved oncologic outcomes in rectal cancer surgery [5,11,12].
Although the aforementioned developments have improved the local recurrence rates associated with radiotherapy, the effects of such therapy on overall survival (OS) in rectal cancer remain a topic of debate [6]. A randomized controlled trial (RCT) in Germany involving 421 patients with T3 or T4 tumors or node-positive LARC revealed that preoperative CRT improved local control but had no advantage over postoperative CRT in terms of OS [13]. Additionally, a multicenter Dutch RCT that followed up 1861 patients with resectable rectal cancer for 12 years reported a significantly lower local recurrence rate in the preoperative radiotherapy group compared with that in the surgery alone group (5% vs. 11%, p < 0.0001); notably, no difference in OS was noted between the two groups [14]. The PROSPECT Trial (Alliance N1048), which compared neoadjuvant CRT with neoadjuvant chemotherapy alone in LARC, demonstrated comparable 5-year disease-free survival (DFS) and OS between the groups, with no clear OS benefit being associated with CRT [15].
Prognostic factors in rectal cancer have long been a subject of clinical interest. Variables such as race, sex, age, pathologic T and N stage, tumor grade, pretreatment and peritreatment carcinoembryonic antigen (CEA) levels, and postoperative chemotherapy have been identified as independent prognostic indicators [16,17]. Achieving pathological complete response (pCR) after CRT, observed in 15%–31% of LARC cases, is also associated with better survival [1,18,19]. Recently, the neutrophil-to-lymphocyte ratio (NLR), an indicator of systemic inflammation, has emerged as another potential prognostic biomarker [20,21]. Our previous study demonstrated that an NLR of ≥3.2 is an independent predictor of poor OS and DFS in patients with LARC who received neoadjuvant CRT and achieved pCR [9].
In consideration of the aforementioned findings, we investigated effective prognostic markers for OS in LARC and elucidated the role of neoadjuvant CRT in LARC. We retrospectively reviewed 252 patients with LARC who underwent neoadjuvant concurrent CRT followed by robotic low anterior resection and TME. Real-world data, including patient characteristics, laboratory results, pathological findings, and long-term survival outcomes, were analyzed.
We retrospectively reviewed 695 patients who underwent robotic colorectal cancer resection at Kaohsiung Medical University Hospital, a tertiary academic medical center, between January 2012 and January 2023. Patients with colon cancer, stage IV disease, non-adenocarcinoma histology, or incomplete clinical information were excluded. Ultimately, 252 patients with LARC who received neoadjuvant concurrent CRT followed by robotic low anterior resection were included in the study (Supplementary Fig. S1). Clinical data were obtained from medical records, with these data including patient demographics, routine laboratory results, serum CEA levels, pathological findings, local recurrence, distant metastasis, and survival status. The study protocol was approved by the Institutional Review Board of Kaohsiung Medical University Hospital (KMUHIRB-E(II)-20250271).
The diagnosis of adenocarcinoma in patients with LARC was confirmed through histopathologic examination. Treatment strategies—including surgical resection, chemotherapy, and chemoradiotherapy—were determined through multidisciplinary team cancer conferences, following institutional clinical practice guidelines [8]. LARC was defined on the basis of the following computed tomography (CT) criteria: (1) T3 tumors with >5 mm extramural extension, (2) clinical T4-stage tumors, or (3) CT-detected regional lymph node metastases. The neoadjuvant chemotherapy regimen comprised biweekly FOLFOX (folinic acid, 5-fluorouracil, and oxaliplatin) administered concurrently with long-course radiotherapy (total dose of 5000 cGy delivered in 25 fractions). Following completion of radiotherapy, chemotherapy was continued biweekly until 2–3 weeks prior to surgery. Robotic surgery was performed using the single-docking technique, as described in our previous study [5]. The other aspects of chemotherapy dosing followed protocols established through our institutional experience with colorectal cancer treatment [5,8].
After surgery, the adjuvant chemotherapy with FOLFOX was administered every 2 weeks for a total of 12 cycles. Patients who achieved pCR received fluoropyrimidine-based chemotherapy for up to 6 months, with close follow-up [8]. For those who developed local recurrence or distant metastasis, treatment was guided by current consensus recommendations, including the use of chemotherapy or targeted therapy [22,23].
Patients were stratified according to pretreatment CEA levels (≥5 ng/mL vs. <5 ng/mL) for evaluation of survival outcomes [8]. The pretreatment NLR was also categorized (≥3.2 vs. <3.2) because this threshold was previously identified as a poor prognostic factor in our earlier studies [9]. To further assess the combined prognostic impact of CEA and the NLR, patients were classified into four groups: Group A: NLR ≥ 3.2, CEA ≥ 5 ng/mL; Group B: NLR ≥ 3.2, CEA < 5 ng/mL; Group C: NLR < 3.2, CEA ≥ 5 ng/mL; and Group D: NLR < 3.2, CEA < 5 ng/mL.
Descriptive statistics, including medians, means, and proportions, were used to summarize patient demographics and gene alteration profiles. The end of the follow-up period was defined as the date of death, the last follow-up visit, or 31 December 2024, whichever occurred first. OS was defined as the interval from the date of LARC diagnosis to death from any cause, last follow-up, or the study endpoint. DFS was defined as the time from treatment initiation to the occurrence of local recurrence or distant metastasis or to the date of the last follow-up.
DFS and OS were evaluated using the Kaplan–Meier method, and survival curves were compared using the log-rank test. Univariate and multivariate analyses were performed using the Cox proportional-hazards regression model to identify prognostic factors for OS and DFS. Multicollinearity among variables was assessed using the variance inflation factor (VIF), and variables with a VIF <2 were considered acceptable. The proportional hazards assumption for the Cox regression model was verified using Schoenfeld residuals, and no violations were observed. A two-sided p value of <0.05 was considered significant. All statistical analyses were performed using SPSS software, version 20.0 (IBM Corp., Armonk, NY, USA), consistent with our previous studies [8,23].
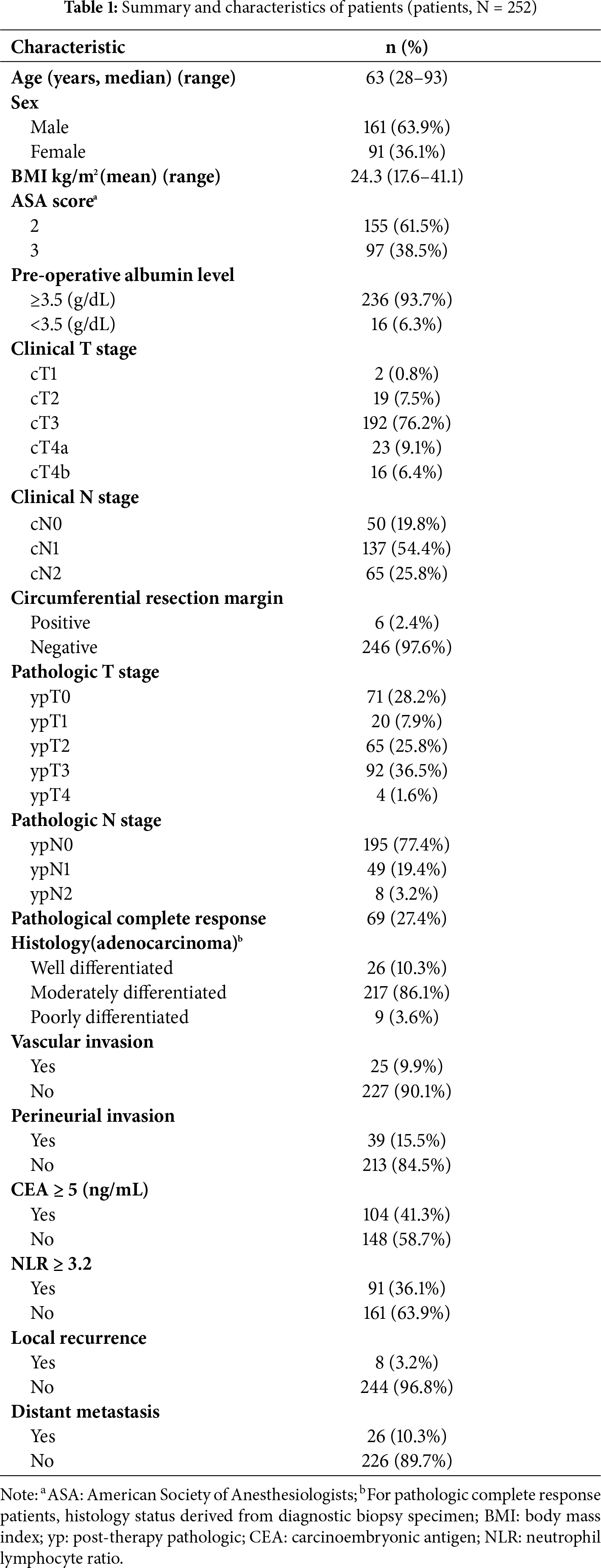
Among the 252 patients with LARC, the median age was 63 years, and 63.9% were male. Most patients had American Society of Anesthesiologists (ASA) scores of 2–3, and 93.7% had adequate preoperative nutritional status (albumin >3.5 g/dL). Clinically, 76.2% had T3 disease, 54.4% had N1, and 25.8% had N2 stage tumors.
After surgery, a free circumferential resection margin was achieved in 97.6% of patients. pCR (ypT0N0) was observed in 27.4% of cases, whereas 25.8% and 36.5% had ypT2 and ypT3 disease, respectively. Most tumors were moderately differentiated adenocarcinomas (86.1%), with vascular and perineural invasion observed in 9.9% and 15.5% of patients, respectively.
Before treatment, elevated CEA levels (≥5 ng/mL) were found in 41.3% of patients, and a pretreatment NLR ≥3.2 in 36.1%. During follow-up, local recurrence and distant metastasis occurred in 3.2% and 10.3% of patients, respectively. Detailed clinicopathologic characteristics are summarized in Table 1.
3.2 Survival and Treatment Outcomes
The median follow-up duration was 61.2 months (range, 7.4–146.7 months). For all patients, the median OS and DFS were 109.2 and 93.4 months, with estimated 5-year OS and DFS rates of 72.5% and 62.6%, respectively (Fig. 1A,B).
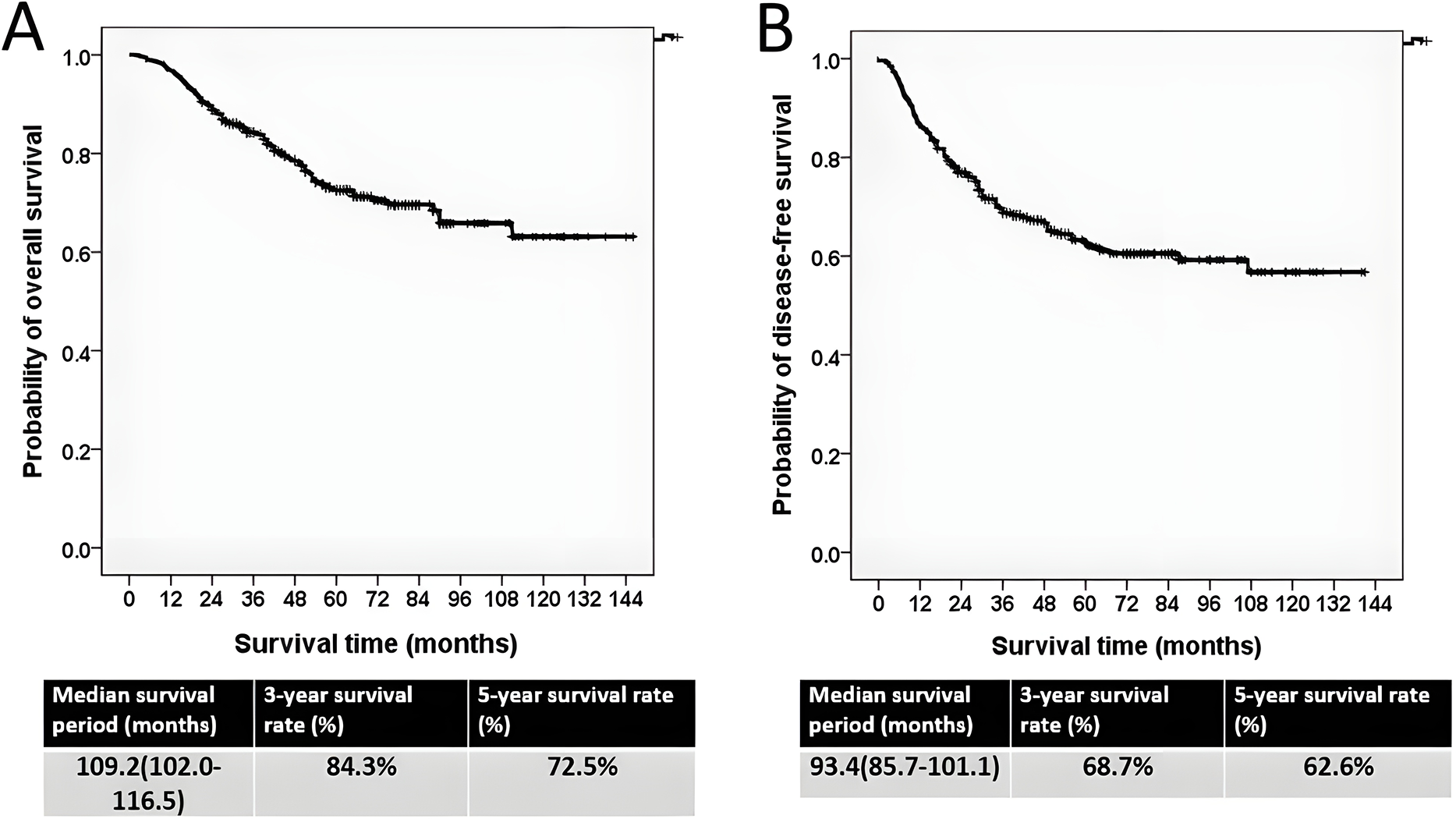
Figure 1: The Kaplan–Meier survival curve. (A) Overall survival curve. (B) Disease-free survival curve
The patients were stratified into two groups on the basis of the pretreatment NLR: NLR ≥ 3.2 and NLR < 3.2. Patients with NLR ≥ 3.2 showed significantly poorer survival than those with NLR < 3.2. Median OS was 94.4 vs. 116.5 months (p = 0.001), and median DFS was 78.8 vs. 101.7 months (p = 0.003) (Fig. 2A,B).
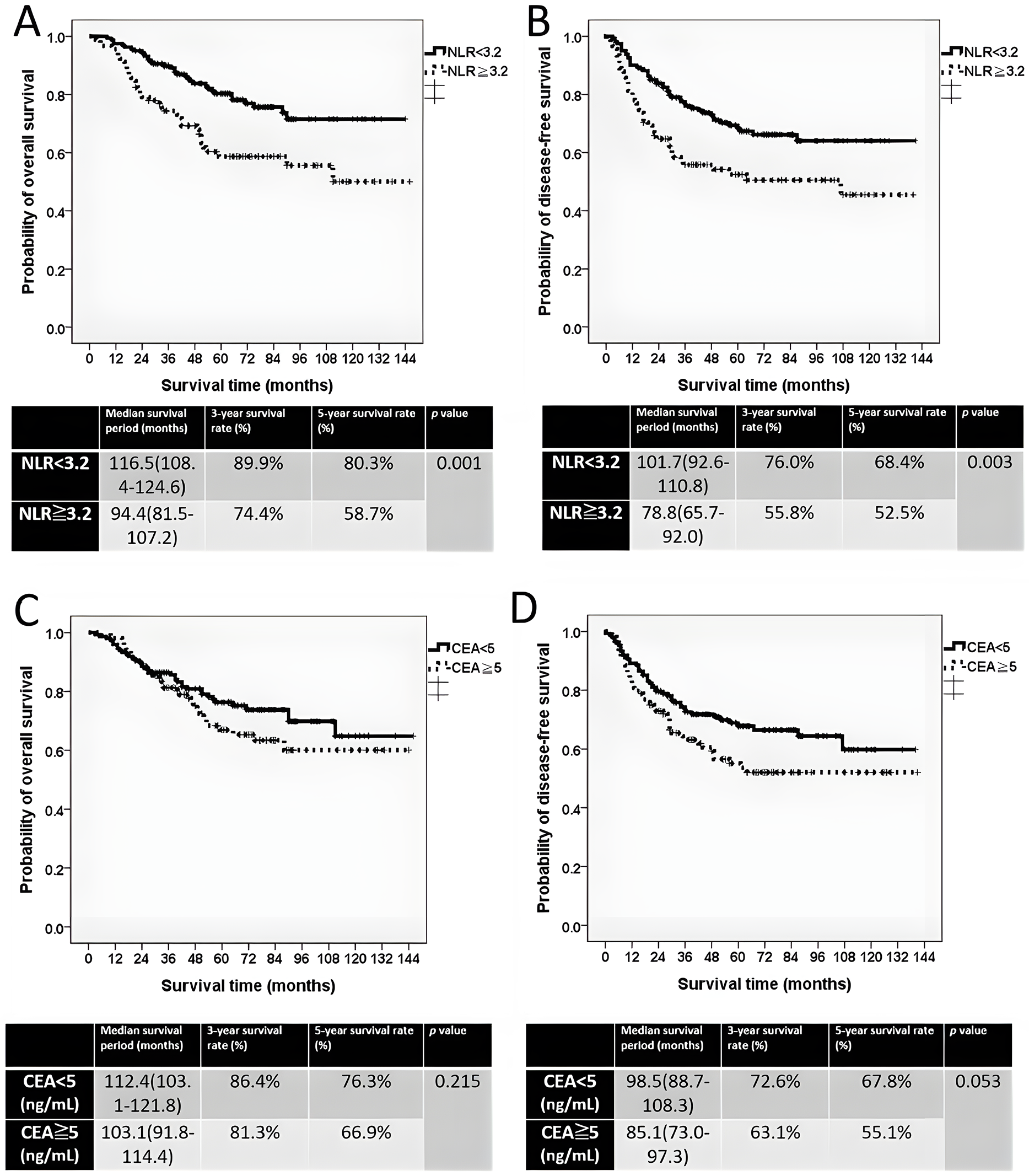
Figure 2: The Kaplan–Meier survival curve of two groups. (A) Overall survival curve stratified by pretreatment NLR ≥ 3.2 and NLR < 3.2. (B) Disease-free survival curve stratified by pretreatment NLR ≥ 3.2 and NLR < 3.2. (C) Overall survival curve stratified by pretreatment CEA ≥ 5 ng/mL and CEA < 5 ng/mL. (D) Disease-free survival curve stratified by pretreatment CEA ≥ 5 ng/mL and CEA < 5 ng/mL
Pretreatment CEA levels (≥5 ng/mL vs. <5 ng/mL) were used to stratify patients for survival analysis. In contrast, CEA levels were not significantly associated with survival. Median OS was 112.4 months for patients with CEA < 5 ng/mL and 103.1 months for those with CEA ≥ 5 ng/mL (p = 0.215), whereas median DFS was 98.5 vs. 85.1 months (p = 0.053) (Fig. 2C,D).
Patients were divided into four subgroups according to pretreatment NLR and CEA levels:
Group A: NLR ≥ 3.2 and CEA ≥ 5 ng/mL (13.9%),
Group B: NLR ≥ 3.2 and CEA < 5 ng/mL (22.2%),
Group C: NLR < 3.2 and CEA ≥ 5 ng/mL (27.4%), and
Group D: NLR < 3.2 and CEA < 5 ng/mL (36.5%).
For OS: Group A demonstrated the poorest survival (median OS, 73.6 months), whereas Group C demonstrated the longest median OS, at 114.9 months. However, Group D exhibited the highest estimated 3-year and 5-year OS rates, which were 91.2% and 80.3%, respectively (Fig. 3A, p = 0.001). Among patients with elevated CEA, those with NLR < 3.2 (Group C) had significantly longer OS than those with NLR ≥ 3.2 (Group A) (p = 0.001; Fig. 3C). In the CEA < 5 ng/mL subgroup, OS tended to be longer in Group D than in Group B, although the difference was not significant (p = 0.111; Fig. 3D).
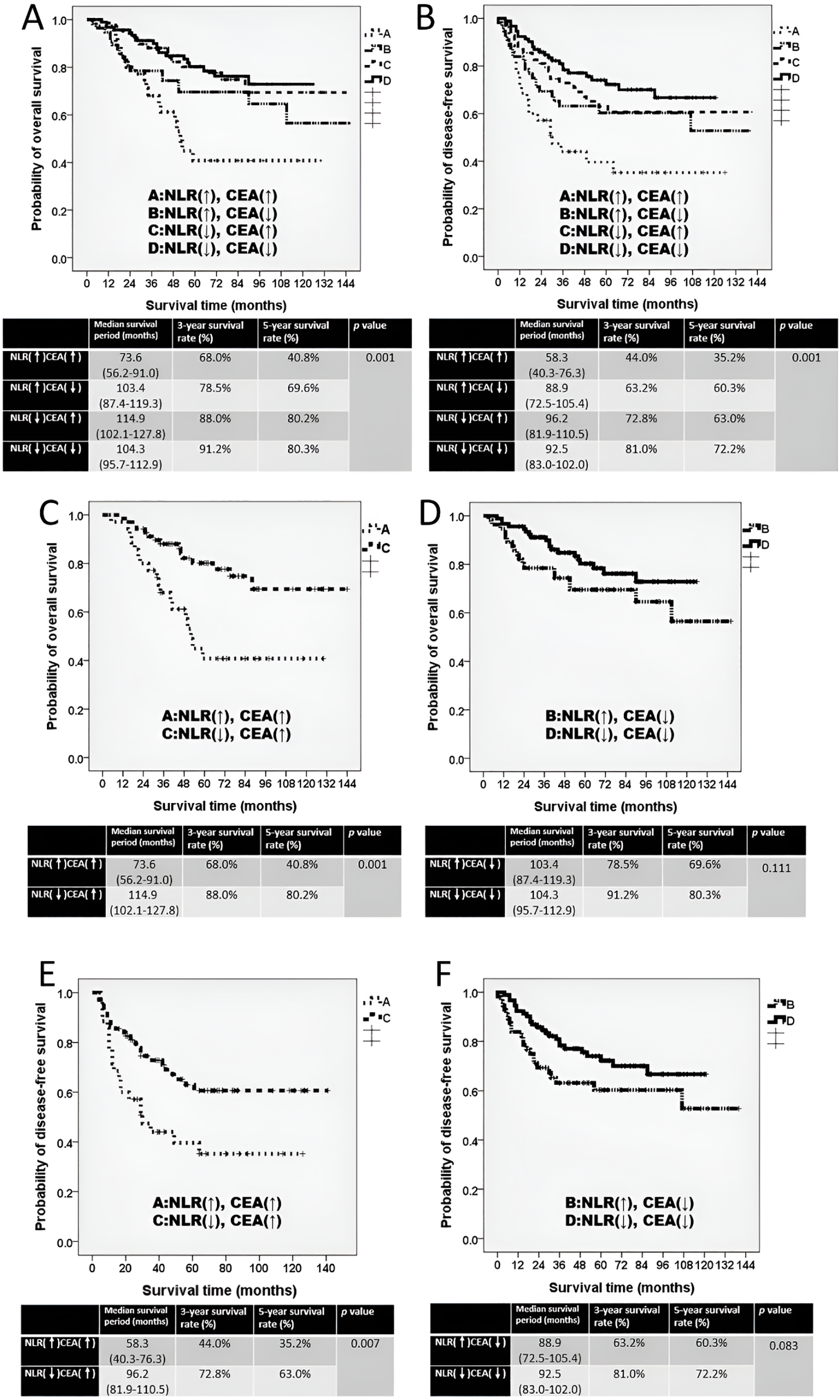
Figure 3: The Kaplan–Meier survival curve of four subgroups. (A) Overall survival curve for four subgroups. (B) Disease-free survival curve for four subgroups. (C) Overall survival curve comparing groups A and C. (D) Overall survival curve comparing groups B and D. (E) Disease-free survival curve comparing groups A and C. (F) Disease-free survival curve comparing groups B and D. The four subgroups were defined as follows: Group A, NLR ≥ 3.2 and CEA ≥ 5 ng/mL (NLR↑, CEA↑); Group B, NLR ≥ 3.2 and CEA < 5 ng/mL (NLR↑, CEA↓); Group C, NLR < 3.2 and CEA ≥ 5 ng/mL (NLR↓, CEA↑); and Group D, NLR < 3.2 and CEA < 5 ng/mL (NLR↓, CEA↓). ↑ indicates high values (≥ cutoff); ↓ indicates low values (<cutoff)
For DFS: The DFS pattern was consistent with OS findings. Group A showed the shortest DFS (median 58.3 months). Group C exhibited the longest median DFS, at 96.2 months, whereas Group D exhibited the highest estimated 3-year and 5-year DFS rates at 81.0% and 72.2%, respectively (Fig. 3B, p = 0.001). Among patients with CEA ≥ 5 ng/mL, those with NLR < 3.2 (Group C) had significantly superior DFS compared with Group A (p = 0.007; Fig. 3E). In patients with CEA < 5 ng/mL, DFS was higher in Group D than in Group B, but without statistical significance (p = 0.083; Fig. 3F).
3.3 Prognostic Factors for Patient Survival
Prognostic factors for OS and DFS were analyzed using Cox regression models (Table 2). On univariable analysis, age ≥ 65 years, pathologic T3–T4 stage, lymph-node metastasis, perineural invasion, and pretreatment NLR ≥ 3.2 were associated with worse OS, whereas pCR predicted better outcomes. In the multivariable model, age ≥ 65 years (HR = 1.820; p = 0.018) and NLR ≥ 3.2 (HR = 2.306; p = 0.001) remained independent adverse prognostic factors. The NLR and CEA interaction was not significant (p = 0.572).
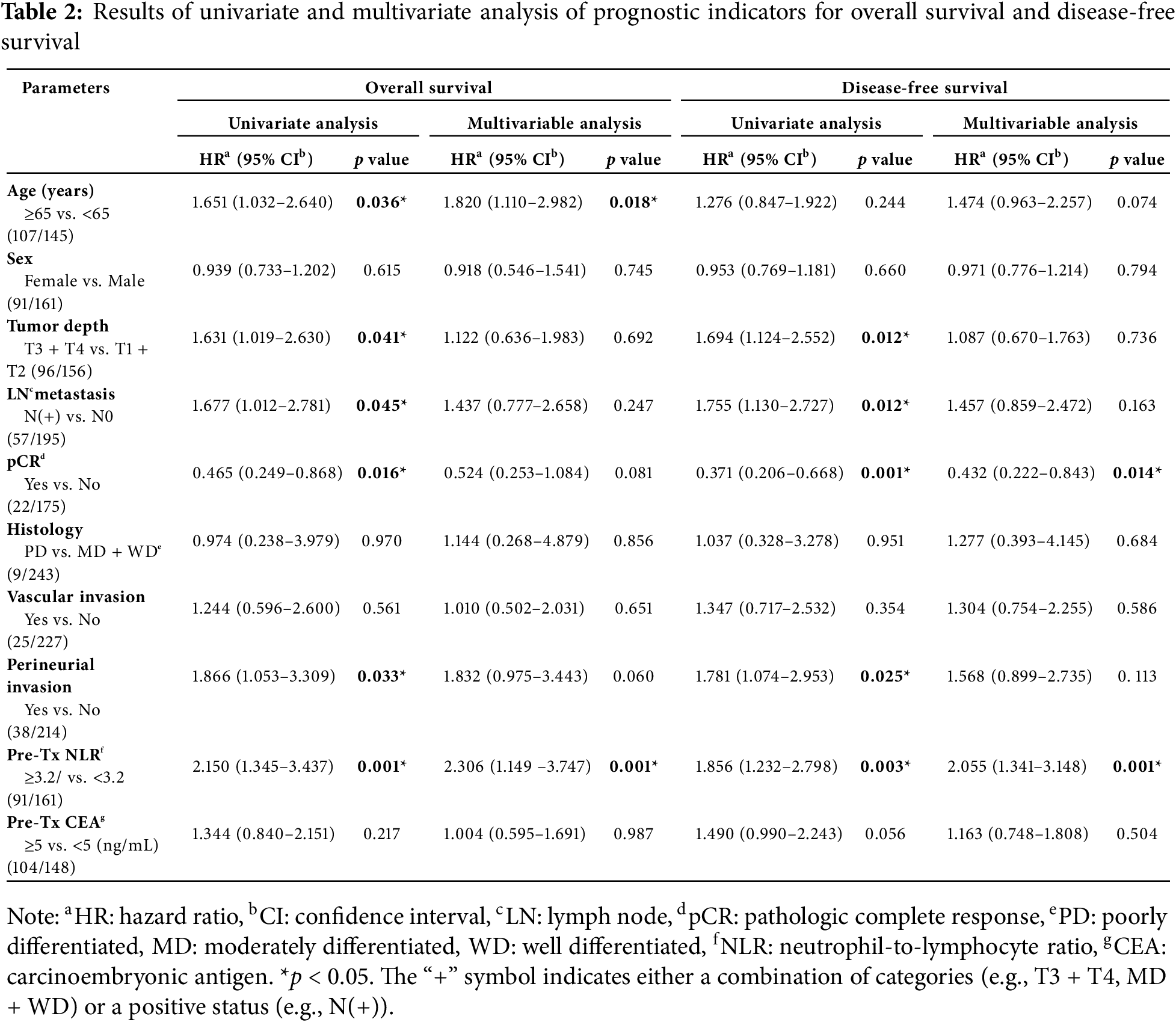
Similarly, for DFS, pathologic T3–T4 stage, lymph-node metastasis, perineural invasion, and NLR ≥ 3.2 were linked to poorer outcomes, whereas pCR was protective. Multivariate analysis confirmed NLR ≥ 3.2 (HR = 2.055; p = 0.001) as an independent poor prognostic factor and pCR (HR = 0.432; p = 0.014) as an independent favorable factor. The NLR and CEA interaction was not significant (p = 0.639).
No multicollinearity was detected (all VIF < 2), and proportional-hazards assumptions were satisfied (all p > 0.05).
This study presents long-term real-world data on the treatment outcomes of LARC in our institution. The estimated 3-year OS and DFS rates were 84.3% and 68.7%, respectively, which are consistent with our previously published findings [1]. The present study had a longer follow-up period and larger sample size than the previous study. Furthermore, the current study estimated 5-year OS and DFS rates of 72.5% and 62.6%, respectively, outcomes that align with those reported in robotic-assisted rectal cancer surgery research [24]. The similarity in OS and DFS rates between the previous and current research supports the representativeness of the current cohort and suggests that the findings are reflective of real-world clinical practice rather than a selected patient subgroup. Moreover, the current findings of median OS and DFS durations of 109.2 and 93.4 months, respectively, indicate that neoadjuvant concurrent CRT followed by robotic-assisted surgery is an effective treatment strategy for LARC.
This study exclusively analyzed patients with LARC who underwent robotic surgery. In our clinical practice, we have established a standardized treatment protocol involving neoadjuvant concurrent CRT followed by robotic-assisted surgery [1,4]. The smooth and stable motion of the robotic arm enables precise lymph node dissection [12,25]. Although rectal cancer surgery can also be performed through laparoscopy or traditional laparotomy, the current study focused solely on robotic surgery to minimize variability arising from different surgical approaches. Consequently, we are confident that the observed differences in survival outcomes are primarily attributable to patients’ pretreatment condition rather than to the surgical technique. Another strength of this study is its extended follow-up duration, with survival outcomes reported over a period exceeding 10 years. Although the retrospective and single-center design of the current study led to it having certain limitations, its use of a homogenous patient cohort and long-term follow-up enhanced the reliability and interpretability of the findings.
Numerous prognostic factors for LARC treatment, such as tumor distance from the anal verge, preoperative and postoperative CEA levels, tumor size and depth, tumor regression grade, lymph node metastasis, distal resection margin, vascular invasion, and perineural invasion, were identified in our previous study [1]. However, in that study, we were unable to establish a simple yet effective prognostic tool for predicting treatment outcomes. In the present study, we proposed the NLR as a potential solution. Research has indicated that NLR values between 2.5 and 5 may predict treatment outcome in patients with LARC [26–28]. For patients with LARC undergoing neoadjuvant CRT followed by radical resection, Colloca et al. reported through a systematic review that an NLR greater than 3 may serve as a reliable prognostic factor [29]. Similarly, our previous study of 478 patients with LARC identified NLR ≥ 3.2 as an independent adverse prognostic factor based on receiver operating characteristic (ROC) analysis [9]. Consistent with these findings, the current study demonstrated that patients with NLR < 3.2 exhibited significantly better OS and DFS compared with those of patients with NLR ≥ 3.2 (Fig. 2A,B). Multivariate analysis confirmed that an NLR of ≥3.2 is an independent predictor of poor survival. Notably, the prognostic value of NLR was not affected by CEA levels.
The NLR is a widely recognized biomarker for predicting cancer prognosis [30,31]. However, the underlying mechanism linking an elevated NLR to poor outcomes in LARC remains unclear [9]. The most commonly proposed explanation involves the interplay between inflammation and immune response [32,33]. Neutrophils reflect systemic inflammation, which can promote tumor cell proliferation and angiogenesis, thereby facilitating cancer progression and metastasis [33]. Conversely, lymphocytes represent the host’s antitumor immune response within the tumor microenvironment; a reduced peripheral lymphocyte count may indicate compromised immune surveillance, contributing to unfavorable treatment outcomes [34]. High NLR reflects a state of systemic inflammation and immune dysregulation. Neutrophils can secrete various pro-tumorigenic cytokines, such as interleukin-6 and vascular endothelial growth factor, which promote tumor growth, angiogenesis, and metastasis. Meanwhile, a relative decrease in lymphocyte count indicates impaired immune surveillance, reducing the host’s ability to mount an effective anti-tumor response [30,31,35]. Neutrophil-derived cytokines, particularly interleukin-8, have been implicated in colorectal cancer progression and treatment response. Elevated interleukin-8 promotes epithelial–mesenchymal transition and chemotherapy resistance, and high serum interleukin-8 levels are associated with poor survival in patients with colorectal liver metastases [36,37]. Overall, the NLR is believed to reflect the balance between tumorigenesis and antitumor immunity. A high NLR is associated with poor prognosis in LARC treatment [9]. In the present study, we confirmed our previous finding that pretreatment NLR ≥ 3.2 is a meaningful threshold for predicting LARC treatment outcomes [9].
CEA is a well-established prognostic biomarker in colorectal cancer management [16,17]. However, as demonstrated in our previous study, a pretreatment CEA level of ≥5 ng/mL cannot serve as an independent prognostic factor in every situation [8]. Typically, additional clinical or pathological features are necessary to accurately evaluate treatment outcomes in LARC [9]. Notably, in the present study, patients with CEA < 5 ng/mL exhibited better OS and DFS than did those with CEA ≥ 5 ng/mL, although these differences did not reach statistical significance. Notably, when CEA levels were analyzed in conjunction with the NLR, more nuanced prognostic distinctions emerged. Reports have suggested that combining the NLR and CEA can more effectively predict colorectal cancer outcomes than either alone can [28]. In our analysis, although Group C (NLR < 3.2, CEA ≥ 5 ng/mL) had the longest median OS and DFS, Group D (NLR < 3.2, CEA < 5 ng/mL) exhibited the highest estimated 3-year and 5-year OS and DFS rates. These outcome differences among the four subgroups become increasingly pronounced when the follow-up exceeded 5 years. We also observed that among patients with CEA ≥ 5 ng/mL, the survival differences between group A and C were particularly marked, which aligns with previous findings linking elevated CEA with an increased recurrence risk and poor prognosis [38]. In such high-risk cases, the pretreatment NLR may be a critical factor in assessing treatment outcomes in LARC.
In this study, we exclusively used the pretreatment NLR to predict LARC outcomes rather than assessing the preoperative or postoperative NLR. Although several studies have identified the NLR as a prognostic biomarker in CRC treatment [39–41], most of the studies have focused on metastatic CRC. For example, Nemoto et al. retrospectively analyzed 71 patients with metastatic CRC undergoing first-line chemotherapy and found that a decreased NLR after 3 months was significantly associated with improved OS and PFS [39]. Liu et al. retrospectively analyzed 128 patients with metastatic CRC who received FOLFOX and targeted therapies (bevacizumab or cetuximab) and reported that a decline in the posttreatment NLR was correlated with longer OS and PFS, whereas an increased posttreatment NLR was associated with worse OS and PFS [40]. Notably, these findings are not directly applicable to our cohort, which exclusively comprised patients with LARC without distant metastasis. Although all patients included in this study received 10–12 weeks of chemotherapy before surgery [1], no association was observed between changes in the NLR and survival outcomes. Moreover, the majority of the patients in our study exhibited sustained NLR elevation after neoadjuvant concurrent CRT, likely due to chemotherapy-induced bone marrow suppression and lymphopenia [42]. Additionally, because of the retrospective design of this study, some preoperative and postoperative NLR data were unavailable. To better determine the prognostic utility of the NLR in LARC, future studies with larger sample sizes and more comprehensive laboratory data are warranted.
Radiotherapy is a key component in the treatment of colorectal cancer, particularly for locally advanced tumors [2,8]. In clinical practice, neoadjuvant concurrent CRT often results in substantial tumor shrinkage, which can facilitate tumor resection [1,4]. However, strong evidence has yet to be obtained that supports radiotherapy offering a survival benefit—specifically, prolonged OS—in patients with LARC [13–15]. Although this is understandable for stage IV rectal cancer, where systemic therapy is the primary treatment [43,44], it is paradoxical in stage III LARC, where surgical resection is the main therapeutic approach [2]. It seems contradictory that radiotherapy facilitates surgical resection but fails to improve OS. Our results provide a potential explanation for this: neoadjuvant concurrent CRT can confer survival benefits only in select subgroups of patients with LARC. Specifically, only patients with a pretreatment NLR of <3.2 demonstrate a favorable response to preoperative radiotherapy. Thus, the NLR may primarily reflect the baseline systemic condition of patients with LARC. To validate this, a study must be conducted that uses comparative data from patients who did not receive preoperative radiotherapy. Given the absence of a control group not receiving CRT, our retrospective study design does not allow for the assessment of the predictive value of NLR in relation to treatment benefit. Instead, our findings highlight the significant prognostic value of pretreatment NLR in the management of patients with LARC.
In addition to the NLR, hematological biomarkers such as the platelet-to-lymphocyte ratio (PLR) have been reported to be effective prognostic indicators in rectal cancer [32]. The underlying mechanism of the PLR as a prognostic indicator is similar to that of the NLR. Platelets contribute not only to hemostasis and thrombosis but also to inflammatory processes and immune regulation through cytokine secretion [45], rendering the PLR a valuable biomarker in cancer prognosis [26]. Ergen et al. conducted a retrospective study involving 53 patients with LARC who underwent neoadjuvant CRT and revealed that patients with PLR ≥ 131 had significantly lower OS and DFS rates than did those with PLR < 131. Furthermore, a multivariate analysis in their study revealed that PLR ≥ 131 was an independent poor prognostic factor DFS in LARC [27]. The relevant literature supports the prognostic utility of both the NLR and the PLR in LARC [27,32]. Hence, the PLR can be considered a promising target for future research. Nevertheless, at present, there are no established treatment guidelines for colorectal cancer patients with elevated NLR or PLR. Based on current evidence, these patients may be considered at higher risk for poor outcomes, and closer follow-up with timely intervention upon early signs of disease progression is suggested [46,47].
This study has several notable limitations. First, its retrospective design and limited sample size may affect the generalizability of the findings. In addition, the investigated patient characteristics were primarily pathological features, whereas key preoperative variables such as pelvic magnetic resonance imaging (MRI) image, tumor size, tumor location (e.g., lower vs. mid-rectum), and patients’ comorbidities were not considered. Additionally, perioperative data, including blood loss, operation time, and surgical complications, were not investigated. This missing information may have introduced selection bias, as patients with missing data might have different clinical characteristics or outcomes compared to those with complete data, potentially affecting the precision and generalizability of our findings. Moreover, we focused on the NLR, selecting this ratio on the basis of findings from our previous research [9], and we did not examine other potentially relevant biomarkers, such as the PLR. Furthermore, changes in the NLR throughout the treatment course, such as postoperative values or those following adjuvant chemotherapy, were not analyzed. Although we proposed a hypothesis regarding the differential effect of radiotherapy on LARC outcomes, no direct evidence was presented to support this claim. In our clinical practice, most patients with LARC received neoadjuvant radiotherapy, limiting our ability to compare outcomes in patients treated with surgery alone. Our proposed NLR + CEA model demonstrated promising prognostic value in patients with LARC undergoing neoadjuvant CRT followed by surgery. However, due to the retrospective and single-center nature of our study, external validation was not feasible. Further prospective trials, such as multicenter programs with larger-scale data collection, standardized laboratory testing schedules, and the use of electronic medical record systems, are warranted to better elucidate the prognostic role of NLR in the treatment of LARC.
This study provides real-world evidence of long-term oncologic outcomes in patients with LARC treated with neoadjuvant CRT followed by robotic-assisted tumor resection. The study findings support the effectiveness of this treatment strategy. A pretreatment NLR of ≥3.2 was identified as a reliable and independent prognostic factor for both OS and DFS. Furthermore, considering the combination of pretreatment NLRs and CEA levels enhanced prognostic stratification. Further research is warranted to elucidate the potential interaction between neoadjuvant CRT and systemic inflammatory markers such as the NLR in the management of LARC.
Acknowledgement: Not applicable.
Funding Statement: This work was supported by grants through funding from the National Science and Technology Council (NSTC 112-2314-B-037-050-MY3, NSTC 114-2314-B-037-103-MY3, NSTC 114-2321-B-037-003) and the Ministry of Health and Welfare (MOHW113-TDU-B-222-134014) and funded by the health and welfare surcharge of on tobacco products, and the Kaohsiung Medical University Hospital (KMUH113-3R31, KMUH113-3R32, KMUH113-3R33, KMUH113-3M58, KMUH113-3M59, KMUH-S11412, KMUH-SH11403), Kaohsiung Medical University Research Center Grant (KMU-TC113A04) and National Tsing Hua University-Kaohsiung Medical University Joint Research Project (NTHU-KMU-KT114P008). In addition, this study was supported by the Grant of Taiwan Precision Medicine Initiative and Taiwan Biobank, Academia Sinica, Taiwan.
Author Contributions: Yen-Cheng Chen, Tsung-Kun Chang, Wei-Chih Su, and Jaw-Yuan Wang designed the study. Yen-Cheng Chen, Tsung-Kun Chang, Wei-Chih Su, Yung-Sung Yeh, Po-Jung Chen, Tzu-Chieh Yin, Ching-Chun Li, Ching-Wen Huang, Hsiang-Lin Tsai, and Jaw-Yuan Wang extracted and collected data. Yen-Cheng Chen, Tsung-Kun Chang, Wei-Chih Su, Yung-Sung Yeh, Po-Jung Chen, Tzu-Chieh Yin, Ching-Chun Li, Ching-Wen Huang, Hsiang-Lin Tsai, and Jaw-Yuan Wang analyzed and interpreted the data. Yen-Cheng Chen, Tsung-Kun Chang, and Jaw-Yuan Wang drafted the manuscript. Yen-Cheng Chen, Tsung-Kun Chang, Wei-Chih Su, and Jaw-Yuan Wang critically revised the manuscript. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: All relevant data and information can be obtained from the corresponding author upon reasonable request.
Ethics Approval: The present study protocol was approved by the Institutional Ethics Committee of Kaohsiung Medical University Hospital (KMUHIRB-E(II)-20250271). A declaration regarding consent to participate is not applicable because of the retrospective study design.
Conflicts of Interest: The authors declare no competing interests.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/or.2025.069397/s1.
Abbreviations
| ASA | American Society of Anesthesiologists |
| CEA | Carcinoembryonic antigen |
| CI | Confidence interval |
| CRT | Chemoradiotherapy |
| CT | Computed tomography |
| DFS | Disease-free survival |
| FOLFOX | Folinic acid, 5-fluorouracil, and oxaliplatin |
| HR | Hazard ratio |
| LARC | Locally advanced rectal cancer |
| MRI | Magnetic resonance imaging |
| NLR | Neutrophil-to-lymphocyte ratio |
| OS | Overall survival |
| pCR | Pathological complete response |
| PLR | Platelet-to-lymphocyte ratio |
| RCT | Randomized controlled trial |
| ROC | Receiver operating characteristic |
| TME | Total mesorectal excision |
| VIF | Variance inflation factor |
References
1. Huang CW, Su WC, Yin TC, Chen PJ, Chang TK, Chen YC, et al. Time interval between the completion of radiotherapy and robotic-assisted surgery among patients with stage I-III rectal cancer undergoing preoperative chemoradiotherapy. PLoS One. 2020;15(10):e0240742. doi:10.1371/journal.pone.0240742. [Google Scholar] [PubMed] [CrossRef]
2. Network NCC. Clinical Practice Guidelines in Oncology (NCCN Guidelines®)–Rectal Cancer (Version 1.2024); 2024 [cited 2025 Jan 1]. Available from: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1461. [Google Scholar]
3. Wang Y, Shen L, Wan J, Zhang H, Wu R, Wang J, et al. Neoadjuvant chemoradiotherapy combined with immunotherapy for locally advanced rectal cancer: a new era for anal preservation. Front Immunol. 2022;13:1067036. doi:10.3389/fimmu.2022.1067036. [Google Scholar] [PubMed] [CrossRef]
4. Huang MY, Huang CW, Wang JY. Surgical treatment following neoadjuvant chemoradiotherapy in locally advanced rectal cancer. Kaohsiung J Med Sci. 2020;36(3):152–9. [Google Scholar] [PubMed]
5. Huang CW, Tsai HL, Yeh YS, Su WC, Huang MY, Huang CM, et al. Robotic-assisted total mesorectal excision with the single-docking technique for patients with rectal cancer. BMC Surg. 2017;17(1):126. doi:10.1186/s12893-017-0315-x. [Google Scholar] [PubMed] [CrossRef]
6. Zhao F, Wang J, Yu H, Cheng X, Li X, Zhu X, et al. Neoadjuvant radiotherapy improves overall survival for T3/4N+M0 rectal cancer patients: a population-based study of 20300 patients. Radiat Oncol. 2020;15(1):49. doi:10.1186/s13014-020-01497-4. [Google Scholar] [PubMed] [CrossRef]
7. Li Y, Wang J, Ma X, Tan L, Yan Y, Xue C, et al. A review of neoadjuvant chemoradiotherapy for locally advanced rectal cancer. Int J Biol Sci. 2016;12(8):1022–31. doi:10.7150/ijbs.15438. [Google Scholar] [PubMed] [CrossRef]
8. Chen YC, Tsai HL, Li CC, Huang CW, Chang TK, Su WC, et al. Critical reappraisal of neoadjuvant concurrent chemoradiotherapy for treatment of locally advanced colon cancer. PLoS One. 2021;16(11):e0259460. [Google Scholar] [PubMed]
9. Huang CM, Huang MY, Tsai HL, Huang CW, Su WC, Chang TK, et al. Pretreatment neutrophil-to-lymphocyte ratio associated with tumor recurrence and survival in patients achieving a pathological complete response following neoadjuvant chemoradiotherapy for rectal cancer. Cancers. 2021;13(18):4589. doi:10.3390/cancers13184589. [Google Scholar] [PubMed] [CrossRef]
10. Chen PC, Yang AS, Fichera A, Tsai MH, Wu YH, Yeh YM, et al. Neoadjuvant radiotherapy vs up-front surgery for resectable locally advanced rectal cancer. JAMA Netw Open. 2025;8(5):e259049. doi:10.1001/jamanetworkopen.2025.9049. [Google Scholar] [PubMed] [CrossRef]
11. Lam J, Tam MS, Retting RL, McLemore EC. Robotic versus laparoscopic surgery for rectal cancer: a comprehensive review of oncological outcomes. Perm J. 2021;25(4):1–9. doi:10.7812/TPP/21.050. [Google Scholar] [PubMed] [CrossRef]
12. Su WC, Huang CW, Ma CJ, Chen PJ, Tsai HL, Chang TK, et al. Feasibility of robot-assisted surgery in elderly patients with rectal cancer. J Minimal Access Surg. 2021;17(2):165–74. doi:10.4103/jmas.jmas_154_19. [Google Scholar] [PubMed] [CrossRef]
13. Sauer R, Becker H, Hohenberger W, Rödel C, Wittekind C, Fietkau R, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. New Engl J Med. 2004;351(17):1731–40. doi:10.1056/nejmoa040694. [Google Scholar] [PubMed] [CrossRef]
14. van Gijn W, Marijnen CA, Nagtegaal ID, Kranenbarg EM, Putter H, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol. 2011;12(6):575–82. doi:10.1016/s1470-2045(11)70097-3. [Google Scholar] [PubMed] [CrossRef]
15. Schrag D, Shi Q, Weiser MR, Gollub MJ, Saltz LB, Musher BL, et al. PROSPECT: a randomized phase III trial of neoadjuvant chemoradiation versus neoadjuvant FOLFOX chemotherapy with selective use of chemoradiation, followed by total mesorectal excision (TME) for treatment of locally advanced rectal cancer (LARC) (Alliance N1048). J Clin Oncol. 2023;41(17_suppl):LBA2. doi:10.1200/jco.2023.41.17_suppl.lba2. [Google Scholar] [CrossRef]
16. Lin Y. A prognostic nomogram for stage II/III rectal cancer patients treated with neoadjuvant chemoradiotherapy followed by surgical resection. BMC Surg. 2022;22(1):256. doi:10.1186/s12893-022-01710-z. [Google Scholar] [PubMed] [CrossRef]
17. Song J, Chen Z, Huang D, Wu Y, Lin Z, Chi P, et al. Nomogram predicting overall survival of resected locally advanced rectal cancer patients with neoadjuvant chemoradiotherapy. Cancer Manag Res. 2020;12:7375–82. doi:10.2147/cmar.s255981. [Google Scholar] [PubMed] [CrossRef]
18. Lefèvre JH, Mineur L, Cachanado M, Denost Q, Rouanet P, de Chaisemartin C, et al. Does A longer waiting period after neoadjuvant radio-chemotherapy improve the oncological prognosis of rectal cancer? Three years’ follow-up results of the greccar-6 randomized multicenter trial. Ann Surg. 2019;270(5):747–54. doi:10.1097/sla.0000000000003530. [Google Scholar] [PubMed] [CrossRef]
19. Ryan JE, Warrier SK, Lynch AC, Ramsay RG, Phillips WA, Heriot AG. Predicting pathological complete response to neoadjuvant chemoradiotherapy in locally advanced rectal cancer: a systematic review. Color Dis Off J Assoc Coloproctol Great Br Irel. 2016;18(3):234–46. doi:10.1111/codi.13207. [Google Scholar] [PubMed] [CrossRef]
20. Song Y, Yang Y, Gao P, Chen X, Yu D, Xu Y, et al. The preoperative neutrophil to lymphocyte ratio is a superior indicator of prognosis compared with other inflammatory biomarkers in resectable colorectal cancer. BMC Cancer. 2017;17(1):744. doi:10.1186/s12885-017-3752-0. [Google Scholar] [PubMed] [CrossRef]
21. Kocak MZ, Coban S, Araz M, Eryilmaz MK, Artac M. Prognostic biomarkers in metastatic colorectal cancer: delta prognostic nutritional index, delta neutrophil to lymphocyte ratio, and delta platelet to lymphocyte ratio. Support Care Cancer. 2023;31(6):357. doi:10.1007/s00520-023-07829-w. [Google Scholar] [PubMed] [CrossRef]
22. Chen HH, Ke TW, Huang CW, Jiang JK, Chen CC, Hsieh YY, et al. Taiwan society of colon and rectal surgeons consensus on mCRC treatment. Front Oncol. 2021;11:764912. doi:10.3389/fonc.2021.764912. [Google Scholar] [PubMed] [CrossRef]
23. Chen YC, Chang TK, Su WC, Yeh YS, Chen PJ, Huang PJ, et al. Impact on survival benefits of asymptomatic primary tumor resection after bevacizumab plus FOLFIRI as first-line therapy for patients with metastatic colorectal cancer with synchronous unresectable metastasis. Int J Color Dis. 2024;39(1):171. doi:10.21203/rs.3.rs-4516245/v1. [Google Scholar] [CrossRef]
24. Huang CW, Wei PL, Chen CC, Kuo LJ, Wang JY. Clinical safety and effectiveness of robotic-assisted surgery in patients with rectal cancer: real-world experience over 8 years of multiple institutions with high-volume robotic-assisted surgery. Cancers. 2022;14(17):4175. doi:10.3390/cancers14174175. [Google Scholar] [PubMed] [CrossRef]
25. Yin TC, Chen YC, Su WC, Chen PJ, Chang TK, Huang CW, et al. Low ligation plus high dissection versus high ligation of the inferior mesenteric artery in sigmoid colon and rectal cancer surgery: a meta-analysis. Front Oncol. 2021;11:774782. doi:10.7759/cureus.39406. [Google Scholar] [CrossRef]
26. Hamid HKS, Emile SH, Davis GN. Prognostic significance of lymphocyte-to-monocyte and platelet-to-lymphocyte ratio in rectal cancer: a systematic review, meta-analysis, and meta-regression. Dis Colon Rectum. 2022;65(2):178–87. doi:10.1097/dcr.0000000000002291. [Google Scholar] [PubMed] [CrossRef]
27. Ergen ŞA, Barlas C, Yıldırım C, Öksüz D. Prognostic role of peripheral Neutrophil-Lymphocyte Ratio (NLR) and Platelet-Lymphocyte Ratio (PLR) in patients with rectal cancer undergoing neoadjuvant chemoradiotherapy. J Gastrointest Cancer. 2022;53(1):151–60. doi:10.1007/s12029-020-00578-7. [Google Scholar] [PubMed] [CrossRef]
28. Tsai PL, Su WJ, Leung WH, Lai CT, Liu CK. Neutrophil-lymphocyte ratio and CEA level as prognostic and predictive factors in colorectal cancer: a systematic review and meta-analysis. J Cancer Res Ther. 2016;12(2):582–9. doi:10.1002/ijc.28536. [Google Scholar] [CrossRef]
29. Colloca G, Venturino A, Guarneri D. Neutrophil-to-lymphocyte ratio predicts survival of patients with rectal cancer receiving neo-adjuvant chemoradiation followed by radical resection: a meta-analysis. Expert Rev Anticancer Ther. 2023;23(4):421–9. doi:10.1080/14737140.2023.2194635. [Google Scholar] [PubMed] [CrossRef]
30. Guthrie GJ, Charles KA, Roxburgh CS, Horgan PG, McMillan DC, Clarke SJ. The systemic inflammation-based neutrophil-lymphocyte ratio: experience in patients with cancer. Crit Rev Oncol Hematol. 2013;88(1):218–30. doi:10.1016/j.critrevonc.2013.03.010. [Google Scholar] [PubMed] [CrossRef]
31. Templeton AJ, McNamara MG, Šeruga B, Vera-Badillo FE, Aneja P, Ocaña A, et al. Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: a systematic review and meta-analysis. J Natl Cancer Inst. 2014;106(6):dju124. doi:10.1093/jnci/dju124. [Google Scholar] [PubMed] [CrossRef]
32. Portale G, Bartolotta P, Azzolina D, Gregori D, Fiscon V. Prognostic role of platelet-to-lymphocyte ratio, neutrophil-to-lymphocyte, and lymphocyte-to-monocyte ratio in operated rectal cancer patients: systematic review and meta-analysis. Langenbeck’s Arch Surg. 2023;408(1):85. doi:10.1007/s00423-023-02786-8. [Google Scholar] [PubMed] [CrossRef]
33. Cupp MA, Cariolou M, Tzoulaki I, Aune D, Evangelou E, Berlanga-Taylor AJ. Neutrophil to lymphocyte ratio and cancer prognosis: an umbrella review of systematic reviews and meta-analyses of observational studies. BMC Med. 2020;18(1):360. doi:10.1186/s12916-020-01817-1. [Google Scholar] [PubMed] [CrossRef]
34. Fogar P, Sperti C, Basso D, Sanzari MC, Greco E, Davoli C, et al. Decreased total lymphocyte counts in pancreatic cancer: an index of adverse outcome. Pancreas. 2006;32(1):22–8. doi:10.1097/01.mpa.0000188305.90290.50. [Google Scholar] [PubMed] [CrossRef]
35. Coffelt SB, Wellenstein MD, de Visser KE. Neutrophils in cancer: neutral no more. Nat Rev Cancer. 2016;16(7):431–46. doi:10.1038/nrc.2016.52. [Google Scholar] [PubMed] [CrossRef]
36. Bazzichetto C, Milella M, Zampiva I, Simionato F, Amoreo CA, Buglioni S, et al. Interleukin-8 in colorectal cancer: a systematic review and meta-analysis of its potential role as a prognostic biomarker. Biomedicines. 2022;10(10):2631. doi:10.3390/biomedicines10102631. [Google Scholar] [PubMed] [CrossRef]
37. Pecqueux M, Brückner F, Oehme F, Hempel S, Baenke F, Riediger C, et al. Preoperative IL-8 levels as prognostic indicators of overall survival: an extended follow-up in a prospective cohort with colorectal liver metastases. BMC Cancer. 2024;24(1):90. doi:10.1186/s12885-023-11787-1. [Google Scholar] [PubMed] [CrossRef]
38. Huang SH, Tsai WS, You JF, Hung HY, Yeh CY, Hsieh PS, et al. Preoperative carcinoembryonic antigen as a poor prognostic factor in stage I-III colorectal cancer after curative-intent resection: a propensity score matching analysis. Ann Surg Oncol. 2019;26(6):1685–94. doi:10.1245/s10434-019-07184-3. [Google Scholar] [PubMed] [CrossRef]
39. Nemoto T, Endo S, Isohata N, Takayanagi D, Nemoto D, Aizawa M, et al. Change in the neutrophil-to-lymphocyte ratio during chemotherapy may predict prognosis in patients with advanced or metastatic colorectal cancer. Mol Clin Oncol. 2021;14(5):107. [Google Scholar] [PubMed]
40. Liu Q, Xi Y, He G, Li X, Zhan F. Dynamics of neutrophil-to-lymphocyte ratio predict outcomes of metastatic colorectal carcinoma patients treated by FOLFOX. J Gastrointest Oncol. 2021;12(6):2846–53. doi:10.21037/jgo-21-716. [Google Scholar] [PubMed] [CrossRef]
41. Chua W, Charles KA, Baracos VE, Clarke SJ. Neutrophil/lymphocyte ratio predicts chemotherapy outcomes in patients with advanced colorectal cancer. Br J Cancer. 2011;104(8):1288–95. doi:10.1038/bjc.2011.100. [Google Scholar] [PubMed] [CrossRef]
42. Hong C, Peng J, Wei Y, Peng H, Yang H, Zhao C, et al. The impact of chemotherapy-associated neutrophil/ lymphocyte counts on prognosis of adjuvant chemotherapy in colorectal cancer. BMC Cancer. 2013;13(1):177. doi:10.1186/1471-2407-13-177. [Google Scholar] [PubMed] [CrossRef]
43. Yin TC, Chen PJ, Yeh YS, Li CC, Chen YC, Su WC, et al. Efficacy of concurrent radiotherapy in patients with locally advanced rectal cancer and synchronous metastasis receiving systemic therapy. Front Oncol. 2023;13:1099168. [Google Scholar] [PubMed]
44. Chen CC, Chang SC, Chang YY, Lin BW, Chen HH, Hsieh YY, et al. Survival benefit of metastasectomy in first-line cetuximab therapy in patients with RAS wild-type metastatic colorectal cancer: a nationwide registry. Am J Cancer Res. 2023;13(12):6333–45. doi:10.62347/qzfd8646. [Google Scholar] [CrossRef]
45. Chen Y, Zhong H, Zhao Y, Luo X, Gao W. Role of platelet biomarkers in inflammatory response. Biomark Res. 2020;8(1):28. doi:10.1186/s40364-020-00207-2. [Google Scholar] [PubMed] [CrossRef]
46. Ortíz-López D, Marchena-Gómez J, Sosa-Quesada Y, Artiles-Armas M, Arencibia-Pérez B, Gil-García J, et al. Prognostic impact of persistent postoperative neutrophil-to-lymphocyte ratio elevation 1 year after colorectal cancer surgery. Updates Surg. 2025;77(8):2331–42. doi:10.1007/s13304-025-02286-y. [Google Scholar] [PubMed] [CrossRef]
47. Ma CJ, Hu WH, Huang MC, Chiang JM, Hsieh PS, Wang HS, et al. Taiwan society of colon and rectum surgeons (TSCRS) consensus for anti-inflammatory nutritional intervention in colorectal cancer. Front Oncol. 2021;11:819742. doi:10.3389/fonc.2021.819742. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools