 Open Access
Open Access
REVIEW
Navigating the Labyrinth of Hepatocellular Carcinoma: Leveraging AI/ML for Precision Oncology
1 Department of Molecular Science and Technology, Ajou University, Suwon, Republic of Korea
2 Department of Herbal Pharmacology, College of Korean Medicine, Gachon University, 1342 Seongnamdaero, Sujeong-gu, Seongnam-si, Republic of Korea
* Corresponding Author: Sidra Ilyas. Email:
(This article belongs to the Special Issue: Molecular Targets and Combinatorial Therapeutics of Liver Cancer)
Oncology Research 2026, 34(5), 9 https://doi.org/10.32604/or.2026.074185
Received 05 October 2025; Accepted 16 January 2026; Issue published 22 April 2026
Abstract
Hepatocellular carcinoma (HCC) remains a significant global health challenge, with therapeutic efficacy in advanced stages often limited by underlying liver dysfunction and adaptive resistance. In this review, the evolving landscape of molecular targets and combinatorial strategies is critically examined, with a particular focus on the transition from preclinical discovery to clinical application. While traditional molecular heterogeneity is acknowledged, the aim is to elucidate how emerging computational paradigms are redefining target discovery and therapeutic stratification in HCC. The primary purpose is to evaluate the role of Artificial Intelligence (AI) and Machine Learning (ML) as integrative tools for translating high-dimensional multi-omics data into clinically actionable insights for HCC management. Special attention is given to the capacity of AI-driven frameworks to analyze complex datasets derived from genomics, transcriptomics, proteomics, metabolomics, and epigenomics, thereby enabling the identification of novel predictive biomarkers, patient subgroups, and rational drug combinations. By synthesizing recent preclinical and clinical evidence, this review highlights how AI-guided approaches can accelerate biomarker validation and optimize therapeutic decision-making. Furthermore, the convergence of AI with spatial transcriptomics, digital pathology, and single-cell technologies is discussed as a transformative infrastructure for decoding tumor–microenvironment interactions and spatial heterogeneity. These integrative strategies provide unprecedented resolution into tumor evolution, immune landscapes, and resistance mechanisms. Collectively, the evidence reviewed supports the conclusion that AI-enabled, multi-omics–driven approaches are instrumental in advancing HCC treatment toward a new era of adaptive, spatially informed, and precision-based personalized medicine.Graphic Abstract
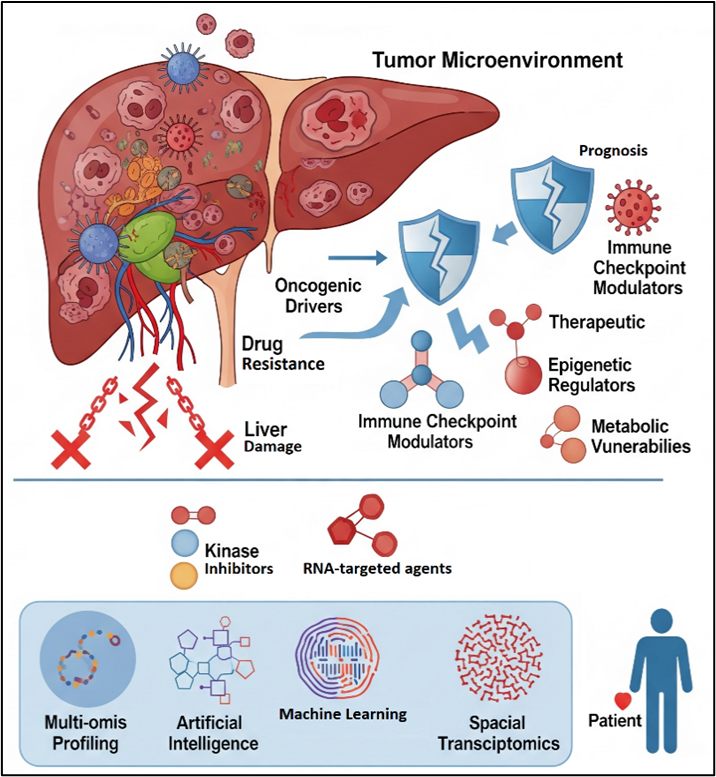
Keywords
Hepatocellular carcinoma (HCC) represents a formidable public health challenge, constituting the third leading cause of cancer related mortality globally. With an estimated 906,000 new diagnoses and 830,000 deaths in 2022 alone, its incidence is projected to rise by 55% by 2040, underscoring the urgent need for transformative therapeutic strategies [1,2]. Accounting for over 80% of all liver cancer cases, HCC is frequently diagnosed at intermediate or advanced stages, resulting in limited curative options and poor long-term survival. HCC is a biologically and clinically heterogeneous disease arising from diverse etiologies, including chronic hepatitis B virus (HBV) and hepatitis C virus (HCV) infections, alcohol-associated liver disease, metabolic dysfunction-associated steatotic liver disease (MASLD/NASH), and environmental carcinogens such as aflatoxin. These etiological factors shape distinct molecular, immunological, and metabolic tumor landscapes, contributing to substantial inter- and intra-tumoral heterogeneity [3]. This heterogeneity underlies variable therapeutic responses, frequent treatment resistance, and high recurrence rates, posing major challenges for effective disease management.
Despite remarkable progress in surveillance, diagnostic modalities, and systemic therapy, clinical management of advanced HCC remains suboptimal. Multi-kinase inhibitors, anti-angiogenic agents, and immune checkpoint inhibitors (ICIs) have modestly extended survival in select patient populations; however, intrinsic and acquired drug resistance, profound tumor heterogeneity, and the underlying fragility of diseased livers continue to restrict durable outcomes. Extensive genomic profiling has catalogued recurrent mutations in canonical drivers and chromatin remodeling, yet the functional interplay of these alterations and their ultimate impact on therapeutic vulnerability remain incompletely understood.
Precision oncology has therefore emerged as a critical framework for advancing HCC treatment by aligning therapeutic decisions with tumor-specific molecular and biological characteristics. Unlike conventional staging systems, precision approaches aim to integrate molecular alterations, TME features, and host factors to enable individualized therapy selection. However, the clinical implementation of precision medicine in HCC is challenged by limited tumor tissue availability, spatial and temporal heterogeneity, and the complexity of interpreting high-dimensional biological data [4]. Recent advances in high-throughput multi-omics technologies including genomics, transcriptomics, proteomics, epigenomics, and metabolomics have provided unprecedented insights into the molecular architecture of HCC [5]. When combined with spatial transcriptomics, these approaches allow detailed characterization of tumor microenvironment (TME) interactions and immune landscapes. Nevertheless, the scale and complexity of these datasets exceed the capacity of conventional analytical methods.
Artificial intelligence (AI) and machine learning (ML) have consequently emerged as indispensable tools for integrating multi-modal data and extracting clinically actionable insights [6]. In HCC, AI-driven models are increasingly applied to radiomics, digital pathology, multi-omics integration, prognostic modeling, and therapeutic response prediction [5,7]. These approaches hold promise for improving early diagnosis, refining risk stratification, guiding treatment selection, and enabling adaptive therapeutic strategies. This review synthesizes current advances in HCC treatment through the lens of precision oncology, with a particular focus on the integration of multi-omics profiling, spatial transcriptomics, and AI-driven computational models. We critically examine emerging molecular targets, combination therapeutic strategies, and AI-enabled clinical decision support systems, while highlighting translational challenges, unmet needs, and future directions for personalized HCC management.
2 Overcoming Therapeutic Limitations: Tumor Heterogeneity and Drug Resistance
The biological complexity of HCC presents formidable obstacles to effective therapeutic intervention. Two intertwined challenges are profound tumor heterogeneity and the rapid emergence of drug resistance, which account for the historically limited success of systemic therapies in advanced disease.
2.1 Tumor Heterogeneity as a Central Barrier
HCC is characterized by inter-patient heterogeneity arising from diverse etiological factors leaving distinct mutational and epigenetic imprints [8,9]. Host specific variables, including gut microbiome, immune landscape, and germline background, add additional layers of heterogeneity [10]. At the molecular level, oxidative stress, recurrent alteration in oncogenes (CTNNB1, c-Myc, c-Met, TERT, PIK3CA), tumor suppressors (AXIN, RB, PTEN, TP53, KEAP1), and chromatin regulators (ARID1A, ARID2), drive distinct phenotypes, such as the “proliferation” vs. “non-proliferation” subclasses, each carrying unique prognostic implications [11,12].
Equally critical is intra-tumoral heterogeneity (ITH), reflecting the coexistence of genetically and phenotypically distinct subclones within individual tumors. ITH drives clonal evolution, metastasis, recurrence, and therapeutic resistance [13]. Under therapeutic pressure, resistant subclones with survival advantages expand a Darwinian process that underlies the modest benefits of multi-kinase inhibitors and fosters multi-drug resistance. Beyond genetic diversity, ITH drives functional divergence, including differences in proliferative capacity, invasiveness, angiogenic signaling, and immune evasion (Fig. 1). Clinically, this diversity complicates tumor sampling, as a single biopsy rarely captures the complete genomic and immune architecture of a tumor, necessitating multi-regional or liquid biopsy approaches, which remain technically and clinically challenging [14].

Figure 1: Schematic representation of the drivers and consequences of tumor heterogeneity in hepatocellular carcinoma (HCC). Exposures (HBV/HCV, alcohol, MASLD/NASH, aflatoxin) and host specific factors (immune system, microbiome, germline variants) converge with tumor intrinsic alterations (mutations, epigenetic changes, metabolic rewiring) to generate profound inter-patient and intra-tumoral heterogeneity. This diversity fuels clonal evolution, adaptive resistance, and immune evasion, ultimately limiting the efficacy of targeted therapies and immunotherapies. Emerging strategies such as liquid biopsy, spatial multi-omics, and adaptive combination therapies may offer the potential to convert descriptive molecular classification into actionable stratification, guiding dynamic and personalized treatment for HCC. HCC: Hepatocellular Carcinoma; HBV: Hepatitis B Virus; HCV: Hepatitis C Virus; MASLD: Metabolic Dysfunction-Associated Steatotic Liver Disease; NASH: Nonalcoholic Steatohepatitis; TKI: Tyrosine Kinase Inhibitor; ICI: Immune Checkpoint Inhibitor.
2.2 Towards Adaptive Therapeutic Strategies and Actionable Stratification
Addressing this biological reality requires a shift from static to adaptive therapeutic strategies. Rational combinations—whether simultaneous or sequential—must target multiple vulnerabilities to prevent clonal escape. While recent molecular classifications have categorized HCC by oncogenic pathways (e.g., Wnt/β-catenin) and immune phenotypes (e.g., “inflamed” vs. “excluded”), these frameworks remain largely descriptive. To achieve clinical impact, they must evolve into actionable stratification models that link subtypes to specific biomarkers and therapeutic vulnerabilities. An ideal clinical workflow functions as a dynamic decision tree (Fig. 2), where longitudinal monitoring via liquid biopsies and spatial multi-omics guides real-time adjustments in treatment, shifting from empirical selection to mechanism-based precision medicine [13].

Figure 2: Dynamic decision tree for personalized HCC management. The workflow emphasizes a personalized, adaptive, and systems level approach, aiming to translate molecular understanding into actionable clinical strategies, ultimately overcoming heterogeneity and resistance in HCC. CT: Computed Tomography; MRI; Magnetic Resonance Imaging; AFP: Alpha-fetoprotein; PIVKA-II: Protein Induced by Vitamin K Antagonist-II; ICI: Immune Checkpoint Inhibitor; TME: Tumor Microenvironment; ML: Machine Learning; HCC: Hepatocellular Carcinoma.
2.3 Mechanisms of Intrinsic and Acquired Drug Resistance in HCC
HCC frequently exhibits insensitivity to conventional chemotherapy agents and a propensity to develop multidrug resistance (MDR) rapidly during treatment, leading to reduced survival and poor prognosis. This resistance arises from a complex interplay of molecular and cellular mechanisms (Table 1). Overexpression of ATP-binding cassette (ABC) transporter proteins such as P-glycoprotein (P-gp/ABCB1), Breast Cancer Resistance Protein (BCRP/ABCG2), and multidrug resistance-associated proteins (MRPs/ABCC), actively pump anticancer drugs (adriamycin, paclitaxel, 5-FU, sorafenib) out of tumor cells, thereby diminishing the drug’s therapeutic efficacy and inducing MDR [15].

Dysregulation of programmed cell death (PCD) pathways is a hallmark of MDR. This includes TP53 mutations and the upregulation of anti-apoptotic proteins like Bcl-2 and MCL-1 [16]. PANoptosis is an emerging inflammatory PCD pathway that integrates molecular features of pyroptosis, apoptosis, and necroptosis through the formation of multi-protein PANoptosome complexes. Disruption of PANoptosis related genes can influence tumor progression, immune evasion, and therapy resistance in multiple malignancies, and that PANoptosis linked signatures have potential as biomarkers of prognosis and immunotherapeutic responsiveness in HCC [17]. Furthermore, enhanced DNA repair capabilities (e.g., via XRCC4-like factor in the non-homologous end-joining (NHEJ) pathway allow HCC cells to survive the genomic stress induced by conventional cytotoxic agents [18].
Reversible epigenetic modifications can silence tumor suppressors or activate pro-survival genes. Similarly, while basal autophagy is suppressive, activated autophagy in established tumors helps cells survive metabolic and therapeutic stress.
TME is a complex ecosystem comprising various normal cells (e.g., fibroblasts, endothelial cells, immune cells) and the extracellular matrix (ECM), acts as a protective niche. Specific metabolites (e.g., 27-hydroxycholesterol) or ECM stiffness can impede drug delivery and foster a pro-survival signalling. Furthermore, cancer stem cells (CSCs) (marked by EpCAM or CD133) contribute to recurrence and metastasis due to their inherent resistance and capacity for dormancy (Table 2).
These resistance mechanisms rarely operate in isolation; they form a redundant network where targeting a single node often triggers compensatory activation of another. For example, epigenetic changes may upregulate ABC transporters, while an immunosuppressive TME protects resistant CSCs. This inherent redundancy necessitates multi-target combination therapies designed to disrupt several nodes concurrently. By identifying master regulators or common vulnerabilities (Table 2), clinicians can move toward highly personalized, mechanism-based strategies that delay resistance and improve long-term outcomes.
3 Molecular Drivers and Therapeutic Targets in HCC
The complex landscape of HCC, marked by significant heterogeneity and drug resistance, necessitates the identification of novel molecular targets and the exploitation of therapeutic vulnerabilities to develop interventions that are more effective. Research efforts are increasingly focused on unraveling the intricate oncogenic signaling pathways and identifying unique tumor-associated antigens and targets (Table 3). Major pathways and molecular mechanisms implicated in HCC include:
3.1 Major Pathways and Molecular Mechanisms
3.1.1 Receptor Tyrosine Kinase (RTK) Driven Pathways
The molecular pathogenesis of HCC is initiated by aberrant activation of signaling networks, with receptor tyrosine kinases (RTKs) representing a critical upstream driver. RTKs such as epidermal growth factor receptor (EGFR), fibroblast growth factor receptor (FGFR), hepatocyte growth factor receptor (c-MET), and vascular endothelial growth factor receptor 8(VEGFR) are frequently dysregulated through sustained growth factor stimulation (Fig. 3). This hyperactivation promotes malignant transformation and drives tumor progression. Downstream of RTKs, two key oncogenic cascades dominate: the RAS/RAF/MEK/ERK pathway, which regulates proliferation, angiogenesis, and metastasis, and the PI3K/AKT/mTOR pathway, which enhances proliferation, invasion, metabolic reprogramming (via the Warburg effect) and is associated with aggressive tumor biology and adverse clinical outcomes.

Figure 3: HCC cell survival proliferation and differentiation occur through the activation of interconnected signaling pathways. Inhibitors (red circles) (e.g., lenvatinib, sorafenib, atezolizumab, bevacizumab) target interdependent intracellular signaling pathways such as EGFR, FGFR, IGFR, TGFR, and VEGFR can alter several targets to exhibit anti-HCC effects (Figure generated by Biorender.com; https://www.biorender.com/). LRP: Low Density Lipoprotein Receptor-Related Protein; Frizzled: Frizzled Class Receptor; CKIα: Casein Kinase 1 Alpha; GSK-3β: Glycogen Synthase Kinase 3 Beta; Axin: Axis Inhibition Protein; APC: Adenomatous Polyposis Coli; TCF/LEF: T-Cell Factor/Lymphoid Enhancer-Binding Factor; TGFR: Transforming Growth Factor-Beta Receptor; Smad2/3: Mothers Against Decapentaplegic Homolog 2/3; Smad4: Mothers Against Decapentaplegic Homolog 4; MAPK: Mitogen-Activated Protein Kinase; SRC: Proto-Oncogene Tyrosine-Protein Kinase Src; RAS: Rat Sarcoma Viral Oncogene Homolog; RAF: Rapidly Accelerated Fibrosarcoma Kinase; MEK: MAPK/ERK Kinase; ERK: Extracellular Signal-Regulated Kinase; VEGFR: Vascular Endothelial Growth Factor Receptor; HGF: Hepatocyte Growth Factor; c-MET: Mesenchymal-Epithelial Transition Factor Receptor; PI3K: Phosphoinositide 3-Kinase; AKT: Protein Kinase; mTOR: Mechanistic Target of Rapamycin; EGFR: Epidermal Growth Factor Receptor; IGFR: Insulin-Like Growth Factor Receptor; ROS: Reactive Oxygen Species; NRF2: Nuclear Factor Erythroid 2-Related Factor 2; p53: Tumor Protein p53; TERT: Telomerase Reverse Transcriptase; c-Myc: MYC Proto-Oncogene.
3.1.2 Wnt/β-Catenin Pathway and Other Pathways
Dysregulation of Wnt/β-catenin signaling, found in ~30%–40% of HCC cases, contributes to transcriptional reprogramming, tumor progression, and the establishment of an immune cold phenotype. The β-catenin pathway can modulate the immune microenvironment, promoting immune evasion. The nuclear accumulation of β-catenin, the pathway’s key effector, is strongly associated with activating mutations in CTNNB1 (encoding β-catenin) and loss-of-function mutations in AXIN1 [33]. Other pathways, such as JAK/STAT and Hippo, also play significant roles. The JAK/STAT pathway, specifically through the activation of STAT3 by pro-inflammatory cytokines, influences gene expression and the behavior of activated hepatic stellate cells, which are crucial in liver fibrosis and injury response [34]. The Hippo pathway participates in HCC by regulating cell proliferation, apoptosis, and stem cell self-renewal. While these pathways may appear to function independently, a fundamental principle of HCC pathogenesis is their profound interconnectedness. The primary pathways are not isolated entities but are organized in a clear hierarchical manner, with RTKs serving as a central command-and-control hub that initiates multiple, parallel downstream cascades [35]. The binding of a single ligand to a receptor can trigger both major proliferation and survival pathways simultaneously, illustrating a powerful, convergent point of dysregulation where a singular insult can have broad, pleiotropic effects [36,37].
3.1.3 PI3K/AKT/mTOR and Wnt/β-Catenin Pathways
Extensive functional interplay exists between the PI3K/AKT/mTOR and Wnt/β-catenin signaling pathways, both of which are central to cellular growth and metabolism. A key enzyme is glycogen synthase kinase-3β (GSK-3β), a component of the β-catenin destruction complex. Under physiological conditions, GSK-3β phosphorylates β-catenin, targeting it for degradation. However, PI3K/AKT signaling inhibits GSK-3β, stabilizing β-catenin and promoting its nuclear accumulation, where it interacts with LEF/TCF transcription factors to activate oncogenic gene expression. This direct linkage allows PI3K/AKT signaling to reinforce Wnt activity, promoting stemness, metastasis, and resistance to therapy (28).
3.1.4 Notch Pathway and Wnt/β-Catenin Signaling
The Notch signaling pathway, highly conserved and frequently upregulated in HCC, adds another dimension to tumor pathogenesis. Ligand induced cleavage of Notch receptors releases the Notch intracellular domain (NICD), which translocates to the nucleus to regulate gene transcription. Importantly, NICD acts synergistically with Wnt/β-catenin signaling, amplifying transcriptional outputs that drive proliferation, tumor progression, and therapeutic resistance.
3.1.5 Oncofetal Markers: Glypican-3 (GPC3) and Alpha-Fetoprotein (AFP)
Oncofetal proteins play pivotal roles in HCC pathogenesis and clinical management. GPC3, a cell surface heparan sulfate proteoglycan, is overexpressed in ~70% of HCC cases and correlates with poor prognosis [38]. Functionally, it promotes tumor growth by modulating FGF2 activity and activating canonical Wnt signaling. AFP, a fetal glycoprotein that undergoes aberrantly re-expressed during hepatocarcinogenesis. While it is widely utilized as a gold-standard diagnostic and prognostic serum biomarker, AFP also functions as a direct mediator of oncogenesis. It contributes to a multifaceted pathogenic network by stimulating angiogenesis, enhancing metastatic potential, and further suppressing anti-tumor immune responses.
3.1.6 TERT Reactivation and Cell Cycle Dysregulation
Telomerase reverse transcriptase (TERT) promoter mutations are among the earliest and most prevalent genomic alterations in HCC, enabling replicative immortality. Concurrent dysfunction of cell-cycle regulators, including CDK4/6, CCND1, and RB1, further accelerates uncontrolled proliferation. These alterations establish cell-cycle disruption as a central oncogenic dependency in HCC.
Collectively, these findings highlight how RTK driven cascades, Wnt/β-catenin, PI3K/AKT/mTOR, Notch, telomerase reactivation, and oncofetal proteins converge into an interconnected oncogenic network. Their extensive crosstalk and feedback loops amplify malignant signaling, fuel immune evasion, and reinforce a pro-malignant phenotype. This complexity underscores both the challenges and opportunities in HCC therapy, pointing toward biomarker guided strategies, rational pathway specific inhibitors, and combinatorial regimens as essential approaches to improving clinical outcomes.
3.2 Tumor Microenvironment (TME) and Immune Checkpoints
The TME is a highly dynamic ecosystem composed of malignant cells, stroma, extracellular matrix (ECM), and immune populations that actively orchestrates tumor survival, angiogenesis, and immune evasion (Fig. 4). Central to this immunosuppressive landscape is the upregulation of inhibitory checkpoints that induce T cell exhaustion and promote immune tolerance. The PD-1/PD-L1 axis is the most clinically validated pathway; tumor cells exploit PD-L1 expression to deactivate effector T cells directly within the TME (Fig. 4). CTLA-4 (Cytotoxic T-Lymphocyte-Associated protein 4) functions as a critical immune brake primarily during the early priming phase of T cell activation in the lymph nodes, distinct from the peripheral activity of PD-1. LAG-3 impairs CD4+ T cell activation via MHC-II trans-endocytosis and enhances Treg-mediated suppression [39]. TIM-3 receptor, expressed on exhausted CD8+ T cells, interacts with galectin-9 to inhibit interferon-γ secretion and drive macrophages toward a pro-tumorigenic, immunosuppressive phenotype.

Figure 4: A multi-layered, precision medicine framework for HCC classification and therapy. The molecular and immune classification is composed of proliferative (Wnt/β-Catenin activated, proliferative/MAPK activated and metabolic syndrome related) and immune phenotypes (Inflamed, Excluded, and Immunosuppressive). Key biomarkers (e.g., CTNNB1 mutation, high PD-L1/CD8+ T cell density, FGF19 amplification) are the specific evidence used for definitive subtyping. Associated therapeutic strategies provides subtype-specific treatment recommendations: immunotherapy (anti-PD-1/L1/anti-VEGF/CTLA-4) is prioritized for immune hot/suppressive subtypes, targeted therapies (FGFR4 inhibitors, MDM2 inhibitors) for proliferative subtypes, and novel combinations (locoregional and immunotherapy) are proposed to convert immune cold tumors to hot phenotypes. HCC: Hepatocellular Carcinoma; TKI: Tyrosine kinase inhibitor; ICI: Immune Checkpoint Inhibitor; MAPK: Mitogen-Activated Protein Kinase; CTNNB1: Catenin Beta 1; TERT: Telomerase Reverse Transcriptase; TP53: Tumor Protein p53; FGF19: Fibroblast Growth Factor 19; CCND1: Cyclin D1; AFP: Alpha-Fetoprotein; PNPLA3: Patatin-Like Phospholipase Domain-Containing Protein 3; TM6SF2: Transmembrane 6 Superfamily Member 2; PD-L1: Programmed Death-Ligand 1; VEGFR: Vascular Endothelial Growth Factor Receptor; PD-1: Programmed Cell Death Protein 1; CTLA-4: Cytotoxic T-Lymphocyte-Associated Protein 4; FoxP3: Forkhead Box P3; Tregs: Regulatory T Cells; CD163: Cluster of Differentiation 163; CD8+: Cluster of Differentiation 8–Positive T Cells; Wnt: Wingless/Integrated Signaling Pathway; FGFR4: Fibroblast Growth Factor Receptor 4; MDM2: Mouse Double Minute 2 Homolog.
3.3 Metabolic Reprogramming and Stress Adaptation
Metabolic plasticity is a defining feature of HCC, reflecting both tumor-intrinsic alterations and adaptation to the diseased liver microenvironment. HCC cells exhibit enhanced glycolysis, altered lipid metabolism, increased glutamine dependency, and dysregulated bile acid signaling [40]. In MASLD/NASH related HCC, lipid accumulation, oxidative stress, and chronic inflammation further complicate tumor metabolism and immune responses. These metabolic states can directly influence sensitivity to systemic therapies, including immunotherapy, underscoring the need for etiology aware treatment strategies [41].
3.4 Epigenetic Regulation and Non-Coding RNAs
Aberrant DNA methylation silences tumor suppressor genes such as CDKN2A, while deregulated histone modifications, alter chromatin accessibility and oncogenic transcriptional programs. These changes not only promote tumor initiation but also blunt antitumor immunity by repressing immune-stimulatory gene expression. Emerging data highlighted a mechanosensitive checkpoint Osr2 recruits HDAC3 to suppress cytotoxic T cell gene programs, directly connecting biomechanical stress with immune exhaustion [33].
4 Emerging Combination Strategies in HCC
The marked molecular heterogeneity and adaptive capacity of HCC impose fundamental limitations on monotherapy-based treatment paradigms. Clinical resistance frequently emerges through compensatory pathway activation, immune escape, and metabolic or phenotypic plasticity, resulting in transient or incomplete responses. Consequently, contemporary HCC management has shifted toward rational combination strategies designed to simultaneously target complementary oncogenic processes, remodel the TME, and enhance the durability of therapeutic responses. Building on the molecular vulnerabilities, this section focuses on how these targets are therapeutically integrated to overcome resistance and improve clinical outcomes.
4.1 Integration of Kinase Inhibitors and Immune-Based Therapies
Tyrosine kinase inhibitors (TKIs) were the first systemic agents to demonstrate survival benefit in advanced HCC; however, their efficacy is limited by rapid adaptive resistance. In parallel, ICIs have transformed HCC treatment but exhibit modest response rates when used alone, largely due to an immunosuppressive TME. These complementary limitations have driven the development of TKI-ICI combination regimens, which now form the backbone of first-line systemic therapy (Table 4). The mechanistic rationale underlying this approach lies in the ability of anti-angiogenic TKIs to normalize tumor vasculature, reduce hypoxia, and attenuate immunosuppressive cytokine signaling, thereby facilitating immune cell infiltration and function. When combined with ICI, this vascular and immunological reprogramming enhances T cell-mediated anti-tumor responses and mitigates intrinsic immune resistance (Fig. 5). Clinical validation of this strategy was achieved with the IMbrave150 trial, which established atezolizumab plus bevacizumab as the global first-line standard for unresectable HCC (can’t be removed by surgery). Beyond this regimen, multiple combinations pairing ICI with multi-targeted TKI are under active investigation, reflecting a sustained effort to refine efficacy while balancing toxicity profiles.

Figure 5: The Evolution of HCC management: A timeline from locoregional intervention to immunotherapy driven systemic care. ICI: Immune checkpoint inhibitor; CRISPR: Clustered Regularly Interspaced Short Palindromic Repeats; PD-1: Programmed Cell Death Protein 1; PD-L1: Programmed Death-Ligand 1; CTLA-4: Cytotoxic T-Lymphocyte-Associated Protein 4; VEGF: Vascular Endothelial Growth Factor; anti-PD-1: Anti-Programmed Cell Death Protein 1; anti-PD-L1: Anti-Programmed Death-Ligand 1; anti-VEGF: Anti-Vascular Endothelial Growth Factor.
Multiple clinical trials are evaluating the combination of ICI with TKI that possess anti-angiogenic activity. These regimens aim to exploit synergistic effects by concurrently inhibiting oncogenic signaling, suppressing angiogenesis, and reinvigorating anti-tumor immunity. Clinical studies involving lenvatinib plus pembrolizumab, cabozantinib plus atezolizumab, and similar combinations has demonstrated encouraging objective response rates and progression-free survival benefits, supporting continued development in both frontline and later-line settings (Table 5) [70]. Importantly, the success of these combinations has underscored the need for biomarker-driven patient selection, as not all tumors derive equal benefit from immune-angiogenic modulation [71].

An alternative strategy to enhance immune activation involves the simultaneous blockade of multiple immune checkpoints. Dual inhibition of PD-1/PD-L1 and CTLA-4 targets distinct phases of the T cell response, with CTLA-4 blockade enhancing early T cell priming and PD-1/PD-L1 inhibition sustaining effector function within the tumor [72]. This approach has demonstrated clinical efficacy in advanced HCC, with durable responses observed in subsets of patients. While immune-related adverse events remain a concern, optimized dosing schedules and patient stratification have improved the therapeutic index of dual ICI regimens, positioning them as viable options for selected patients.
4.1.3 ICI and MAPK Pathway Inhibitors Combinations
Given the frequent activation of the MAPK signaling cascade in HCC, combinations incorporating MEK or RAF inhibitors with ICI have emerged as a strategy to counteract immune resistance driven by oncogenic signaling. MAPK pathway inhibition may reduce tumor-induced immune suppression and enhance antigen presentation, thereby sensitizing tumors to immune checkpoint blockade. Although still largely in early-phase clinical development, this approach represents a rational extension of pathway-informed combination therapy, particularly for tumors exhibiting MAPK-driven resistance phenotypes.
4.1.4 Locoregional Therapy (LRT) and Systemic Therapy Combinations
Locoregional therapies (LRT), including transarterial chemoembolization (TACE), radiofrequency ablation (RFA), and radioembolization, remain central to HCC management across disease stages. Increasing evidence suggests that LRT can induce immunogenic tumor cell death, releasing tumor antigens that prime systemic immune responses [82]. Combining LRT with systemic agents particularly ICI and anti-angiogenic therapies leverages this immunogenic effect to enhance systemic disease control. Ongoing trials evaluating TACE or Y90 radioembolization in combination with ICI and TKI have reported improvements in progression-free survival, supporting the concept of locoregional-systemic synergy in intermediate-stage disease [83].
4.1.5 RNA Targeted Therapeutics
Despite advances in targeted and immune-based therapies, directly targeting core genetic drivers of HCC remains challenging. RNA-based therapeutics, including small interfering RNAs (siRNAs) and microRNAs (miRNAs), offer a powerful modality to modulate oncogenic gene expression post-transcriptionally [84,85]. Preclinical studies have demonstrated that multi-target RNA strategies can suppress multiple oncogenic pathways simultaneously, mirroring the rationale of multi-drug therapy. When integrated with TKI or ICI, RNA-based agents have the potential to sensitize tumors, overcome resistance, and enhance therapeutic durability. Advances in delivery technologies particularly lipid nanoparticles and hepatocyte-targeted conjugates have significantly improved the translational feasibility of these approaches.
4.2 Future Directions: Toward Tri-Modal and Integrated Therapy
The future of HCC treatment lies in integrated, multi-modal strategies that combine molecular targeting, immune modulation, and microenvironmental reprogramming. A conceptual framework involves a sequenced approach:
• An RNA agent (e.g., siRNA) silences a primary resistance driver (e.g., CTNNB1).
• A TKI/anti-angiogenic agent remodels the TME.
• An ICI is administered to unleash a potent and durable immune response against the now-sensitized tumor.
While such regimens offer substantial promise, their complexity introduces challenges related to toxicity, treatment sequencing, and cost. Addressing these barriers will require the development of predictive biomarkers, optimized dosing strategies, and adaptive trial designs. Ultimately, the successful implementation of integrated combination therapies will depend on precision medicine approaches that align therapeutic intensity with tumor biology and patient-specific risk profiles. The vast majority of trials combine ICI, TKI, anti-VEGF agents, and locoregional therapy (Table 6).

A significant number of trials are investigating the combinations in the neoadjuvant (pre-surgery) and adjuvant (post-curative treatment) settings to reduce recurrence rates (Table 6). Many trials are specifically focused on treating patients who have progressed on the current first-line standard of care. Emerging agents target new pathways such as TIGIT (Tiragolumab), LAG-3 (Relatlimab), TIM-3 (Cobolimab), CTLA-4 with enhanced Fc function (BMS-986218), and cellular therapies (CAR-T, CAR-Macrophages). The integration and timing of TACE, TARE (transarterial radioembolization), SBRT (Stereotactic Body Radiation Therapy), and HAIC (Hepatic Artery Infusion Chemotherapy) with systemic therapy is a major area of active investigation (Table 7).

4.3 Etiology-Specific Molecular Signatures and Therapeutic Implications
HCC arises from diverse etiologies can reshape tumor biology and therapeutic responsiveness (Table 8). HBV-associated HCC is characterized by viral DNA integration, HBx-mediated malfunction of oncogenic signaling pathways, TERT promoter and TP53 mutations, AKT pathway activation, and epigenetic silencing of tumor suppressor genes, generating a relatively “inflamed” TME with T-cell infiltration and often enhanced responsiveness to ICI [86]. In contrast, HCV-associated HCC arises mainly through chronic inflammation, oxidative stress, and immune disruption rather than direct genomic integration, with frequent CTNNB1 and AXIN1 alterations promoting a WNT/β-catenin driven “immune-excluded” TME and resistance to anti-PD-1 therapy. MASLD/NASH-related HCC exhibits pronounced metabolic reprogramming, lipotoxicity, IL-6/JAK/STAT pathway activation, immune exhaustion, and characteristic genetic variants such as PNPLA3, TM6SF2, and MBOAT7, resulting in a metabolically inflamed, immunosuppressive TME that reduces ICI efficacy but highlights potential vulnerabilities to metabolic and cytokine-targeted therapies [87,88]. Similarly, ALD-associated HCC shows overlapping features with MASLD-HCC, including IL-6/JAK/STAT activation and immunosuppressive TME shaped by chronic alcoholic injury, alongside acetaldehyde-induced DNA damage, oxidative stress, and aberrant methylation patterns that accelerate hepatocarcinogenesis. Finally, aflatoxin-related HCC is defined by a high-frequency TP53 R249S mutation caused by aflatoxin B1 exposure, often synergizing with HBV infection, and generating a genotoxin-driven TME with vulnerabilities in DNA damage repair and cell cycle control [89].

While ICI, particularly combinations such as atezolizumab plus bevacizumab, have redefined first-line therapy for advanced HCC, critical gaps remain that limit their universal efficacy [90]. Notably, chronic viral HCC tends to be “hot” and inflamed, favoring ICI responsiveness, whereas MASH/NASH-HCC often exhibits a “cold” microenvironment, potentially rendering ICI ineffective or even detrimental; however, pivotal trials have largely failed to stratify patients by etiology [89]. Moreover, current studies exclude patients with compromised liver function (Child-Pugh B/C), leaving their safety and efficacy profiles uncertain. High-frequency alterations in the TERT promoter (up to 60%) and Wnt/β-catenin pathway (CTNNB1 mutations, up to 30%) remain largely undruggable, highlighting the need for next-generation targeted therapies, including small-molecule inhibitors or gene-editing approaches, potentially in combination with ICI, to achieve durable responses across HCC subtypes [91]. Collectively, these etiological subtypes illustrate that HCC is not a uniform disease entity; integrating molecular, immunological, and metabolic profiling into precision oncology frameworks enables etiology-stratified biomarker development, individualized immunotherapeutic and metabolic interventions, and rationalized clinical trial design [91].
5 Advancing Precision Oncology in Hepatocellular Carcinoma: Multi-Omics, AI, and Spatial Technologies
The therapeutic landscape of HCC is undergoing a fundamental transition toward precision oncology, a paradigm that tailors interventions to the molecular, spatial, and immunological architecture of individual tumors. This shift reflects the recognition that HCC is not a single disease entity but a spectrum of biologically distinct malignancies shaped by diverse etiologies, evolutionary trajectories, and TME states. The convergence of multi-omics profiling, spatial transcriptomics (ST) and artificial intelligence (AI) now enables a systems-level understanding of HCC that surpasses traditional clinic-pathologic stratification. Collectively, these technologies support earlier diagnosis, robust prediction of therapeutic response, and rational design of individualized treatment strategies.
5.1 Multi-Omics Data Resources for Precision HCC Research
Comprehensive multi-omics profiling is essential for capturing the molecular complexity of HCC. Large-scale public consortia have generated foundational datasets integrating genomics, transcriptomics, epigenomics, proteomics, and clinical annotation. The Cancer Genome Atlas-Liver Hepatocellular Carcinoma (TCGA-LIHC) cohort provides matched genomic, transcriptomic, epigenomic, and proteomic data for approximately 360–400 primary HCC tumors, predominantly derived from patients with viral hepatitis (HBV/HCV) or metabolic liver disease (NAFLD/MASH). Complementing TCGA, the International Cancer Genome Consortium (ICGC) incorporates ethnically and etiologically diverse cohorts from multiple geographic regions, expanding the generalizability of molecular discoveries, as highlighted in Table 9. Transcriptomic datasets from the Gene Expression Omnibus (GEO) and European Nucleotide Archive (ENA) further support validation studies, particularly for TME characterization and drug resistance mechanisms. Proteogenomic data from the Clinical Proteomic Tumor Analysis Consortium (CPTAC) provide direct insights into functional protein states and post-translational modifications linked to prognosis and treatment response.

5.2 Deciphering HCC Complexity through Integrated Multi-Omics Analysis
Single-layer molecular analyses are insufficient to explain HCC pathogenesis or therapeutic resistance. Integrated multi-omics approaches overcome this limitation by combining genomics, transcriptomics, proteomics, epigenomics, and metabolomics to construct a comprehensive molecular portrait of the tumor. Genomic analyses reveal recurrent alterations in oncogenic and tumor suppressor pathways, including Wnt/β-catenin, PI3K/AKT/mTOR, MAPK/ERK, and TP53, which inform patient stratification for targeted therapies and immunotherapy. Transcriptomic profiling captures dynamic gene expression programs associated with proliferation, immune exclusion, metastasis, and drug resistance, while also identifying non-coding RNAs (lncRNAs and miRNAs) as emerging biomarkers and therapeutic targets. Proteomics provides a functional readout of tumor biology by quantifying signaling proteins, immune checkpoint expression, and angiogenic mediators, often outperforming mRNA-based predictors. Epigenomic and metabolomic analyses further uncover regulatory and metabolic rewiring, including aberrant DNA methylation and enhanced glycolysis, fatty acid oxidation, and glutaminolysis.
Crucially, integrated multi-omics signatures outperform single biomarkers in predicting prognosis and therapeutic response. Systems-level analyses have identified robust prognostic programs, including PANoptosis-related gene signatures, as well as actionable vulnerabilities in treatment-refractory HCC, underscoring the translational value of multi-modal integration (Fig. 6) [69].

Figure 6: The multi-omics to AI pipeline for HCC precision oncology. Conceptual framework illustrating the integration of heterogeneous clinical, molecular, and imaging data through advanced AI/ML architectures to generate actionable, personalized clinical guidance for HCC management. BCLC: Barcelona Clinic Liver Cancer; WES: Whole Exome Sequencing; WGS: Whole Genome Sequencing; CT: Computed Tomography; MRI: Magnetic Resonance Imaging; CNN: Convolutional Neural Network; DNN: Deep Neural Network; GNN: Graph Neural Network; AE: Autoencoder; ICI: Immune Checkpoint Inhibitor; TKI: Tyrosine Kinase Inhibitor; AFP: Alpha-Fetoprotein.
5.3 Artificial Intelligence Architectures for Precision Oncology in HCC
The high dimensionality and heterogeneity of multi-omics, imaging, and clinical datasets necessitate advanced AI and machine-learning (ML) frameworks capable of extracting clinically actionable insights. Deep learning architectures, particularly convolutional neural networks (CNN) for imaging and deep neural networks (DNNs) for multimodal data fusion now form the computational backbone of precision oncology in HCC (Table 10).
5.3.1 AI-Driven Radiomics and Imaging Analytics
Radiomics leverages AI to extract quantitative features from contrast-enhanced CT and MRI that are imperceptible to visual inspection. CNN-based radiomics models are most extensively validated for predicting microvascular invasion (MVI), recurrence risk, and treatment response. Three-dimensional CNN architectures trained across arterial, portal venous, and delayed phases implicitly learn features related to tumor margin irregularity, intra-tumoral heterogeneity, and peritumoral enhancement. Meta-analyses report pooled AUC values exceeding 0.85 for MVI prediction and recurrence risk stratification, supporting radiomics as a reliable non-invasive adjunct [96–99].
5.3.2 AI for Multi-Omics Integration and Therapeutic Response Prediction
DNNs excel at integrating heterogeneous data streams, including genomic alterations (e.g., CTNNB1, TERT, TP53), transcriptomic immune signatures, proteomic markers, imaging features, and clinical variables. Modality-specific embedding layers followed by fusion architectures enable synergistic learning across data types and consistently outperform single-modality models. Autoencoders (AE) are commonly used to compress high-dimensional transcriptomic data into biologically meaningful latent representations. In HCC, such models have demonstrated strong performance in predicting response to ICI based therapies and locoregional treatments, with pooled AUROC values approaching 0.89 in internal validation and ~0.81 in external cohorts (Table 10) [100,101].
5.4 Clinical Performance, Validation, and Generalizability
AI models in HCC have achieved clinically meaningful performance across diagnostic, prognostic, and therapeutic domains (Table 11). Radiomics-based classifiers differentiate HCC from benign or cirrhotic nodules with pooled AUCs of approximately 0.86, while survival prediction models achieve C-indices ranging from 0.73 to 0.78 in external validation cohorts [86,102,103]. Integrated multi-omics models further enhance survival prediction, reporting 1-year survival AUCs approaching 0.98 in TCGA-based analyses and improved long-term prognostic accuracy when incorporating key driver genes (FBN1, MAP1B) [104,105].

Despite these advances, generalizability remains a major barrier. Models trained in HBV-predominant populations frequently underperform in MASLD-dominant cohorts, reflecting true biological heterogeneity rather than technical bias [86]. Addressing this challenge requires federated learning, multinational collaboration, and harmonization of imaging protocols, radiomics pipelines, and omics preprocessing.
5.5 Toward Adaptive Precision Oncology in HCC
Static treatment algorithms are insufficient to address tumor evolution and microenvironmental plasticity. Adaptive precision oncology reframes treatment as a dynamic, data-driven process in which therapy evolves in response to longitudinal molecular and immunological changes. This framework integrates baseline molecular stratification, AI-guided therapy selection, serial liquid biopsy surveillance, and dynamic therapeutic adaptation.
AI-driven platforms integrating multi-omics, radiomics, and clinical data can anticipate resistance mechanisms (often preceding radiographic progression) and recommend timely therapeutic modification. Reinforcement learning (RL) models and graph-based frameworks further enable simulation of tumor evolution under different treatment pressures, informing optimal sequencing strategies [86]. Collectively, these approaches redefine HCC management as an anticipatory and personalized process, with AI serving as the analytical backbone for next-generation precision oncology (Fig. 7).

Figure 7: Adaptive precision oncology framework in HCC. The cyclic process of adaptive precision oncology, where initial profiling guides therapy, and continuous AI-powered monitoring of tumor evolution dictates real-time treatment modifications to overcome resistance. RNA-Seq: RNA Sequencing; CT: Computed Tomography; AI: Artificial Intelligence; ICI: Immune Checkpoint Inhibitor; TKI: Tyrosine Kinase Inhibitor; ctDNA: Circulating Tumor DNA.
Algorithms such as YOLOv8 and frameworks (MONAI) extract quantitative features from medical images to enable earlier HCC detection, non-invasive characterization of tumor grade and vascular invasion, and more accurate monitoring of disease progression (Table 12). Furthermore, AI tools (Visiopharm and Aiforia) provide quantitative analysis of the TME from digital pathology images, elucidating immune cell infiltration patterns and spatial relationships that are critical for predicting immunotherapy outcomes. The development of generative AI models promises to create sophisticated in silico simulations of tumor evolution. By integrating the full spectrum of omics data, these models aim to reconstruct the dynamic interplay within the TME, predict responses to novel interventions, and ultimately guide the development of truly personalized, adaptive therapeutic strategies for every HCC patient.

5.6 Spatial Transcriptomics (ST): Mapping TME for the Therapeutic Precision
While multi-omics profiling reveals what molecular alterations are present in a tumor, it often homogenizes the sample, obscuring the critical architectural context of where these alterations occur. ST addresses this fundamental limitation by mapping gene expression with cellular-to-subcellular resolution directly within the native tissue architecture [106]. This transformative technology provides an unprecedented view of the TME, enabling the deconstruction of the intricate cellular ecosystems, signaling networks, and metabolic niches that collectively govern HCC progression, immune evasion, and therapeutic response [107]. In HCC, a malignancy defined by profound intra-tumoral heterogeneity, ST is particularly powerful. It moves beyond bulk analysis to delineate distinct tumor subclones coexisting within a single lesion, each with unique molecular features. For instance, it can reveal regions characterized by high expression of angiogenic markers (e.g., VEGF) adjacent to zones expressing immune checkpoint proteins (e.g., PD-L1). This granular mapping is essential for understanding the functional consequences of tumor evolution and for designing therapies that can address the tumor’s multifaceted biology. Furthermore, this technology is revolutionizing our understanding of the immunosuppressive landscape of HCC. By spatially resolving the locations and gene expression profiles of diverse cell populations, researchers can precisely map the “battlegrounds” within the TME. This includes identifying immunosuppressive hubs enriched with regulatory T cells (Tregs), myeloid derived suppressor cells (MDSCs), tumor associated macrophages (TAMs), and visualizing the physical barriers that lead to immune exclusion zones, where cytotoxic T cells are prevented from infiltrating the tumor core. Understanding these spatial dynamics is critical for developing next-generation immunotherapies, such as CAR-T cell therapies or novel checkpoint inhibitors, designed to reprogram the TME and overcome resistance.
The synergy between ST and AI is essential for unlocking the full potential of this data rich technology. The sheer complexity of spatial datasets necessitates sophisticated computational methods for analysis. AI algorithms, particularly deep learning models like convolutional neural networks (CNNs), are being deployed to automate the identification of histological features, quantify the density and spatial distribution of tumor infiltrating lymphocytes (TILs), and define recurrent cellular neighborhoods that correlate with clinical outcomes. Moreover, AI is enabling the development of 3D spatial reconstructions of the TME, providing multi-dimensional insights into the interactions between tumor, stromal, and immune cells. This powerful combination is not only elucidating fundamental mechanisms of immune evasion but is also identifying novel, spatially defined biomarkers and therapeutic targets. Integrating these spatial insights with multi-omics data has moved us towards a new era of precision oncology where therapeutic strategies are tailored not only to the molecular profile of a tumor but also to its unique spatial architecture.
Practical Constraints on Spatial Transcriptomics (ST)
While techniques such as ST and multi-modal AI modeling represent the scientific apex of precision oncology, their clinical implementation faces significant practical barriers. Despite its revolutionary power to map cellular interactions within the TME, the clinical utility of ST is currently hampered by high per-sample cost, low throughput compared to bulk sequencing, and the lack of standardized computational pipelines. These factors restrict its use primarily to specialized research institutions, delaying its deployment as a routine clinical tool. Although many AI models show high performance in silico, integrating them into diverse clinical settings is challenging. Deployment requires significant IT infrastructure investment, data harmonization efforts across multiple imaging systems, and ongoing regulatory approval and maintenance. This high initial and operational cost often creates a disparity, limiting the availability of these advanced predictive tools to large academic medical centers, thereby potentially increasing existing health disparities. The foundational data for multi-omics and ST requires high-quality tissue. HCC often presents in patients with cirrhosis, making repeated, invasive tumor biopsies risky and limiting the amount of available tissue. This constraint forces reliance on less precise liquid biopsy methods or radiomics, highlighting a fundamental physical limitation of data acquisition in this disease setting.
6 Translational Insights and Future Directions
Rapid advances in molecular profiling, spatial biology, and computational analytics are refurbishing the conceptual framework of HCC research. The central challenge now lies in converting these biological insights into clinically actionable strategies that enable durable, patient-specific benefit. Bridging discovery science with therapeutic implementation will define the next phase of progress in HCC management.
6.1 The Translation Gap and Preclinical Limitations
Despite an expanding catalog of oncogenic drivers, immune regulators, epigenetic modifiers, and metabolic dependencies in HCC, only a limited number of discoveries have translated into approved therapies. This translational gap largely reflects the limitations of preclinical models (like standard cell lines and basic CDX models) that inadequately recapitulate the complexity of human TME, including tumor heterogeneity, immune contexture, vascular remodeling, and underlying liver dysfunction [108].
Successful examples, such as the clinical validation of immune checkpoint inhibition combined with anti-angiogenic therapy, illustrate the importance of aligning mechanistic rationale with clinically representative models and biomarker-driven trial design [109]. Future translational research must prioritize advanced preclinical systems such as patient-derived organoids, spatially resolved co-culture models, and humanized mouse platforms that faithfully mimic the TME and chronic liver disease context [110]. These models are essential for improving therapeutic predictability and reducing late-stage clinical attrition.
6.2 Personalized Therapeutic Strategies and Resistance Mitigation
The intrinsic molecular diversity of HCC and the inevitability of adaptive resistance necessitate a transition from uniform treatment algorithms to individualized therapeutic strategies. Limited tissue availability and spatial heterogeneity remain major barriers to precision medicine; however, the expanding use of liquid biopsy offers a scalable solution for non-invasive, longitudinal tumor profiling. Circulating tumor DNA and RNA analyses enable real-time monitoring of clonal evolution, emergent resistance mechanisms, and immune dynamics, supporting adaptive treatment modification [111]. The clinical utility of liquid biopsy will depend on the development of robust biomarkers capable of guiding therapeutic selection and response assessment. AI-driven analytical frameworks are poised to play a pivotal role by integrating multi-omics, imaging, and clinical data to enhance signal detection, reduce noise, and identify composite molecular signatures that define clinically relevant HCC subtypes. Such approaches move beyond single biomarkers toward multidimensional predictors with greater clinical robustness.
Overcoming therapeutic resistance remains a critical objective. As a single pathway rarely drives resistance in HCC; rather, compensatory signaling networks enable tumor persistence under selective pressure. This complexity underscores the need for rationally designed combination strategies that target shared regulatory nodes or convergent vulnerabilities. Emerging RNA-targeted therapeutics provide a promising orthogonal modality by enabling direct modulation of oncogenic transcripts and resistance-associated regulators [112,113].
6.3 PANoptosis and Multi-Target Therapeutic Convergence
PANoptosis is recognized as a bridge between inflammatory signaling and cell death. Its imbalance can suppress antigen presentation, alter cytokine production, and create an immunosuppressive TME. Multi-omics analyses have identified PANoptosis associated regulators and gene signatures linked to aggressive, treatment-refractory HCC phenotypes. Therapeutically activating PANoptosis has the potential to convert “cold” tumors into “hot” immunogenic lesions, thereby enhancing tumor immunogenicity, sensitize tumors to immune checkpoint blockade, and synergize with kinase inhibitors [114]. Integrating PANoptosis associated biomarkers into immunotherapy stratification models may refine patient selection and help mitigate the heterogeneity in treatment outcomes. This strategy exemplifies how systems-level biological insights can inform mechanism based therapeutic innovation.
6.4 Etiology and AI-Guided Precision Oncology
Precision oncology in HCC therefore requires a shift from histology- and stage-based treatment toward etiology- and molecularly stratified decision-making. AI and ML are essential enablers of this transition. By integrating etiological background, multi-omics profiles, imaging features, and longitudinal clinical data, AI models can stratify patients into biologically coherent subgroups and predict optimal therapeutic sequencing [88]. Future clinical trials must adopt stratified designs that evaluate therapeutic efficacy within defined etiological-molecular subtypes, rather than treating HCC as a homogeneous entity [95]. Such trial paradigms will be critical for maximizing the benefit of emerging systemic and combination therapies.
Hepatocellular carcinoma remains a major global health challenge, characterized by high mortality, biological complexity, and limited long-term therapeutic success in advanced disease. Although combination immunotherapy has redefined the treatment landscape and improved outcomes for selected patients, durable benefit remains elusive for many, largely due to profound tumor heterogeneity and adaptive resistance. The shifting epidemiology of HCC from viral hepatitis toward metabolic and alcohol-associated etiologies further underscores the need for updated screening strategies, tailored therapeutic approaches, and renewed pharmaceutical investment. The future of HCC management lies in the convergence of multi-omics profiling, artificial intelligence, and spatially resolved technologies to enable biologically informed, adaptive treatment strategies. The therapeutic paradigm is evolving from monotherapy toward intelligently sequenced, multi-modal regimens that integrate immunotherapy, kinase inhibition, and RNA-targeted approaches. Achieving this vision will require rigorous biomarker development, innovative clinical trial designs, and close integration of computational models into clinical decision-making. By accelerating the translation of systems-level insights into personalized interventions, precision oncology has the potential to transform HCC from a largely lethal disease into a manageable and ultimately curable condition.
Acknowledgement: None.
Funding Statement: The authors received no specific funding.
Author Contributions: The authors confirm contribution to the paper as follows: study conception and design: Abdul Manan, Sidra Ilyas; data collection: Abdul Manan, Sidra Ilyas; draft manuscript preparation: Abdul Manan, Sidra Ilyas. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Data sharing is not applicable to this article.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49. doi:10.3322/caac.21660. [Google Scholar] [PubMed] [CrossRef]
2. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229–63. doi:10.3322/caac.21834. [Google Scholar] [PubMed] [CrossRef]
3. Rivera-Esteban J, Muñoz-Martínez S, Higuera M, Sena E, Bermúdez-Ramos M, Bañares J, et al. Phenotypes of metabolic dysfunction-associated steatotic liver disease-associated hepatocellular carcinoma. Clin Gastroenterol Hepatol. 2024;22(9):1774–89.e8. doi:10.1016/j.cgh.2024.03.028. [Google Scholar] [PubMed] [CrossRef]
4. Galun D, Mijac D, Filipovic A, Bogdanovic A, Zivanovic M, Masulovic D. Precision medicine for hepatocellular carcinoma: clinical perspective. J Pers Med. 2022;12(2):149. doi:10.3390/jpm12020149. [Google Scholar] [PubMed] [CrossRef]
5. Rajak D, Nema P, Sahu A, Vishwakarma S, Kashaw SK. Advancement in hepatocellular carcinoma research: biomarkers, therapeutics approaches and impact of artificial intelligence. Comput Biol Med. 2025;198(Pt A):111120. doi:10.1016/j.compbiomed.2025.111120. [Google Scholar] [PubMed] [CrossRef]
6. Grignaffini F, Barbuto F, Troiano M, Piazzo L, Simeoni P, Mangini F, et al. The use of artificial intelligence in the liver histopathology field: a systematic review. Diagnostics. 2024;14(4):388. doi:10.3390/diagnostics14040388. [Google Scholar] [PubMed] [CrossRef]
7. Rawat A, Rajan, Malhi HK, Chaudhary K. Artificial intelligence using multiomics/genetic tools and application in liver disease. In: Artificial intelligence, machine learning, and deep learning in precision medicine in liver diseases. Amsterdam, The Netherlands: Elsevier; 2023. p. 109–31. doi:10.1016/b978-0-323-99136-0.00015-5. [Google Scholar] [CrossRef]
8. Blach S, Zeuzem S, Manns M, Altraif I, Duberg AS, Muljono DH, et al. Global prevalence and genotype distribution of hepatitis C virus infection in 2015: a modelling study. Lancet Gastroenterol Hepatol. 2017;2(3):161–76. doi:10.1016/S2468-1253(16)30181-9. [Google Scholar] [PubMed] [CrossRef]
9. El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365(12):1118–27. doi:10.1056/nejmra1001683. [Google Scholar] [PubMed] [CrossRef]
10. Lawal G, Xiao Y, Rahnemai-Azar AA, Tsilimigras DI, Kuang M, Bakopoulos A, et al. The immunology of hepatocellular carcinoma. Vaccines. 2021;9(10):1184. doi:10.3390/vaccines9101184. [Google Scholar] [PubMed] [CrossRef]
11. Harding JJ, Nandakumar S, Armenia J, Khalil DN, Albano M, Ly M, et al. Prospective genotyping of hepatocellular carcinoma: clinical implications of next-generation sequencing for matching patients to targeted and immune therapies. Clin Cancer Res. 2019;25(7):2116–26. doi:10.1158/1078-0432.CCR-18-2293. [Google Scholar] [PubMed] [CrossRef]
12. Hoshida Y, Toffanin S, Lachenmayer A, Villanueva A, Minguez B, Llovet JM. Molecular classification and novel targets in hepatocellular carcinoma: recent advancements. Semin Liver Dis. 2010;30(1):35–51. doi:10.1055/s-0030-1247131. [Google Scholar] [PubMed] [CrossRef]
13. Dagogo-Jack I, Shaw AT. Tumour heterogeneity and resistance to cancer therapies. Nat Rev Clin Oncol. 2018;15(2):81–94. doi:10.1038/nrclinonc.2017.166. [Google Scholar] [PubMed] [CrossRef]
14. Yang C, Zhang S, Cheng Z, Liu Z, Zhang L, Jiang K, et al. Multi-region sequencing with spatial information enables accurate heterogeneity estimation and risk stratification in liver cancer. Genome Med. 2022;14(1):142. doi:10.1186/s13073-022-01143-6. [Google Scholar] [PubMed] [CrossRef]
15. Gao L, Morine Y, Yamada S, Saito Y, Ikemoto T, Tokuda K, et al. Nrf2 signaling promotes cancer stemness, migration, and expression of ABC transporter genes in sorafenib-resistant hepatocellular carcinoma cells. PLoS One. 2021;16(9):e0256755. doi:10.1371/journal.pone.0256755. [Google Scholar] [PubMed] [CrossRef]
16. De Fromentel CC, Levrero M. p53 functional loss, stemness and hepatocellular carcinoma. Hepatoma Res. 2020;6:80. doi:10.20517/2394-5079.2020.77. [Google Scholar] [CrossRef]
17. Li Y, Li J, Chen Y. PANoptosis-associated genes exhibit significant potential in the diagnosis of hepatocellular carcinoma. J Gastrointest Oncol. 2025;16(3):1105–14. doi:10.21037/jgo-2025-356. [Google Scholar] [PubMed] [CrossRef]
18. Tang S, Bai C, Yang P, Chen X. 14-3-3ε boosts bleomycin-induced DNA damage response by inhibiting the drug-resistant activity of MVP. J Proteome Res. 2013;12(6):2511–24. doi:10.1021/pr301085c. [Google Scholar] [PubMed] [CrossRef]
19. Mukherjee N, Sheetz J, Shellman YG. Targeting the BCL2 family: advances and challenges in BH3 mimetic-based therapies. Int J Mol Sci. 2025;26(20):9859. doi:10.3390/ijms26209859. [Google Scholar] [PubMed] [CrossRef]
20. Wu Y, Zhang J, Li Q. Autophagy, an accomplice or antagonist of drug resistance in HCC? Cell Death Dis. 2021;12(3):266. doi:10.1038/s41419-021-03553-7. [Google Scholar] [PubMed] [CrossRef]
21. Sperandio LP, Lins IVF, Erustes AG, Leão AHFF, Antunes F, Morais IBM, et al. Blocking autophagy by the two-pore channels antagonist tetrandrine improves sorafenib-induced death of hepatocellular carcinoma cells. Toxicol in Vitro. 2023;90(2):105603. doi:10.1016/j.tiv.2023.105603. [Google Scholar] [PubMed] [CrossRef]
22. Zhou L, Liu C, Adoro SA, Chen L, Ramirez D, Jin G. Leukemia-associated HSC vascular niche is negatively regulated by PERK of unfolded protein response (UPR). Blood. 2019;134(Supplement_1):2486. doi:10.1182/blood-2019-127607. [Google Scholar] [CrossRef]
23. Chipurupalli S, Ganesan R, Martini G, Mele L, Reggio A, Esposito M, et al. Cancer cells adapt FAM134B/BiP mediated ER-phagy to survive hypoxic stress. Cell Death Dis. 2022;13(4):357. doi:10.1038/s41419-022-04813-w. [Google Scholar] [PubMed] [CrossRef]
24. Mortezaee K. WNT/β-catenin regulatory roles on PD-(L)1 and immunotherapy responses. Clin Exp Med. 2024;24(1):15. doi:10.1007/s10238-023-01274-z. [Google Scholar] [PubMed] [CrossRef]
25. Fu S, Debes JD, Boonstra A. DNA methylation markers in the detection of hepatocellular carcinoma. Eur J Cancer. 2023;191(12):112960. doi:10.1016/j.ejca.2023.112960. [Google Scholar] [PubMed] [CrossRef]
26. Scheller T, Hellerbrand C, Moser C, Schmidt K, Kroemer A, Brunner SM, et al. mTOR inhibition improves fibroblast growth factor receptor targeting in hepatocellular carcinoma. Br J Cancer. 2015;112(5):841–50. doi:10.1038/bjc.2014.638. [Google Scholar] [PubMed] [CrossRef]
27. Rimassa L, Finn RS, Sangro B. Combination immunotherapy for hepatocellular carcinoma. J Hepatol. 2023;79(2):506–15. doi:10.1016/j.jhep.2023.03.003. [Google Scholar] [PubMed] [CrossRef]
28. Zhou G, Sprengers D, Boor PPC, Doukas M, Schutz H, Mancham S, et al. Antibodies against immune checkpoint molecules restore functions of tumor-infiltrating T cells in hepatocellular carcinomas. Gastroenterology. 2017;153(4):1107–19.e10. doi:10.1053/j.gastro.2017.06.017. [Google Scholar] [PubMed] [CrossRef]
29. Hou PP, Luo LJ, Chen HZ, Chen QT, Bian XL, Wu SF, et al. Ectosomal PKM2 promotes HCC by inducing macrophage differentiation and remodeling the tumor microenvironment. Mol Cell. 2020;78(6):1192–206.e10. doi:10.1016/j.molcel.2020.05.004. [Google Scholar] [PubMed] [CrossRef]
30. Wang Z, Zhang L, Zhang D, Sun R, Wang Q, Liu X. Glycolysis inhibitor 2-deoxy-D-glucose suppresses carcinogen-induced rat hepatocarcinogenesis by restricting cancer cell metabolism. Mol Med Rep. 2015;11(3):1917–24. doi:10.3892/mmr.2014.2945. [Google Scholar] [PubMed] [CrossRef]
31. Chan SL, Cheng PNM, Liu AM, Chan LL, Li L, Chu CM, et al. A phase II clinical study on the efficacy and predictive biomarker of pegylated recombinant arginase on hepatocellular carcinoma. Investig New Drugs. 2021;39(5):1375–82. doi:10.1007/s10637-021-01111-8. [Google Scholar] [PubMed] [CrossRef]
32. Liu M, Zhang L, Li H, Hinoue T, Zhou W, Ohtani H, et al. Integrative epigenetic analysis reveals therapeutic targets to the DNA methyltransferase inhibitor guadecitabine (SGI-110) in hepatocellular carcinoma. Hepatology. 2018;68(4):1412–28. doi:10.1002/hep.30091. [Google Scholar] [PubMed] [CrossRef]
33. Liang B, Wang H, Qiao Y, Wang X, Qian M, Song X, et al. Differential requirement of Hippo cascade during CTNNB1 or AXIN1 mutation-driven hepatocarcinogenesis. Hepatology. 2023;77(6):1929–42. doi:10.1002/hep.32693. [Google Scholar] [PubMed] [CrossRef]
34. Zhang C, Hu S, Yin C, Wang G, Liu P. STAT3 orchestrates immune dynamics in hepatocellular carcinoma: a pivotal nexus in tumor progression. Crit Rev Oncol Hematol. 2025;207(1):104620. doi:10.1016/j.critrevonc.2025.104620. [Google Scholar] [PubMed] [CrossRef]
35. Montor WR, Salas AROSE, de Melo FHM. Receptor tyrosine kinases and downstream pathways as druggable targets for cancer treatment: the current arsenal of inhibitors. Mol Cancer. 2018;17(1):55. doi:10.1186/s12943-018-0792-2. [Google Scholar] [PubMed] [CrossRef]
36. Huang Y, Peng M, Yu W, Li H. Activation of Wnt/β-catenin signaling promotes immune evasion via the β-catenin/IKZF1/CCL5 axis in hepatocellular carcinoma. Int Immunopharmacol. 2024;138:112534. doi:10.1016/j.intimp.2024.112534. [Google Scholar] [PubMed] [CrossRef]
37. Xu C, Xu Z, Zhang Y, Evert M, Calvisi DF, Chen X. β-Catenin signaling in hepatocellular carcinoma. J Clin Investig. 2022;132(4):e154515. doi:10.1172/JCI154515. [Google Scholar] [PubMed] [CrossRef]
38. Zheng X, Liu X, Lei Y, Wang G, Liu M. Glypican-3: a novel and promising target for the treatment of hepatocellular carcinoma. Front Oncol. 2022;12:824208. doi:10.3389/fonc.2022.824208. [Google Scholar] [PubMed] [CrossRef]
39. Sangro B, Yau T, Harding JJ, Acosta Rivera M, Kazushi N, El-Khoueiry AB, et al. RELATIVITY-106: a phase 1/2 trial of nivolumab (NIVO) + relatlimab (RELA) in combination with bevacizumab (BEV) in first-line (1L) hepatocellular carcinoma (HCC). J Clin Oncol. 2023;41(4_suppl):TPS636. doi:10.1200/jco.2023.41.4_suppl.tps636. [Google Scholar] [CrossRef]
40. Liang B, Jiang Y, Song S, Jing W, Yang H, Zhao L, et al. ASPP2 suppresses tumour growth and stemness characteristics in HCC by inhibiting Warburg effect via WNT/β-catenin/HK2 axis. J Cell Mol Med. 2023;27(5):659–71. doi:10.1111/jcmm.17687. [Google Scholar] [PubMed] [CrossRef]
41. Li L, Nie L, Jordan A, Cai Q, Liu Y, Li Y, et al. Targeting glutaminase is therapeutically effective in ibrutinib-resistant mantle cell lymphoma. Haematologica. 2023;108(6):1616–27. doi:10.3324/haematol.2022.281538. [Google Scholar] [PubMed] [CrossRef]
42. Chu H, Wu C, Zhao Q, Sun R, Yang K, Zhao B, et al. Quantitative proteomics identifies FOLR1 to drive sorafenib resistance via activating autophagy in hepatocellular carcinoma cells. Carcinogenesis. 2021;42(5):753–61. doi:10.1093/carcin/bgab019. [Google Scholar] [PubMed] [CrossRef]
43. Yu Y, Harring A, Volckova E, Savage RE, Schwartz B. Abstract C076: In vitro and in vivo combination of ARQ 751 with PARP inhibitors, CDK4/6 inhibitors, Fulvestrant and Paclitaxel. Mol Cancer Ther. 2019;18(12_Suppl):C076. doi:10.1158/1535-7163.targ-19-c076. [Google Scholar] [CrossRef]
44. Tao Z, Cui Y, Xu X, Han T. FGFR redundancy limits the efficacy of FGFR4-selective inhibitors in hepatocellular carcinoma. Proc Natl Acad Sci U S A. 2022;119(40):e2208844119. doi:10.1073/pnas.2208844119. [Google Scholar] [PubMed] [CrossRef]
45. Gadaleta-Caldarola G, Rizzo A, Dadduzio V, Lombardi L, Gadaleta-Caldarola A, Infusino S, et al. Pemigatinib in intrahepatic cholangiocarcinoma: a work in progress. Curr Oncol. 2022;29(10):7925–31. doi:10.3390/curroncol29100626. [Google Scholar] [PubMed] [CrossRef]
46. Han P, Li H, Jiang X, Zhai B, Tan G, Zhao D, et al. Dual inhibition of Akt and c-Met as a second-line therapy following acquired resistance to sorafenib in hepatocellular carcinoma cells. Mol Oncol. 2017;11(3):320–34. doi:10.1002/1878-0261.12039. [Google Scholar] [PubMed] [CrossRef]
47. Qin S, Chan SL, Sukeepaisarnjaroen W, Han G, Choo SP, Sriuranpong V, et al. A phase II study of the efficacy and safety of the MET inhibitor capmatinib (INC280) in patients with advanced hepatocellular carcinoma. Ther Adv Med Oncol. 2019;11:1758835919889001. doi:10.1177/1758835919889001. [Google Scholar] [PubMed] [CrossRef]
48. Hsieh FS, Chen YL, Hung MH, Chu PY, Tsai MH, Chen LJ, et al. Palbociclib induces activation of AMPK and inhibits hepatocellular carcinoma in a CDK4/6-independent manner. Mol Oncol. 2017;11(8):1035–49. doi:10.1002/1878-0261.12072. [Google Scholar] [PubMed] [CrossRef]
49. Rimini M, Rimassa L, Ueshima K, Burgio V, Shigeo S, Tada T, et al. Atezolizumab plus bevacizumab versus lenvatinib or sorafenib in non-viral unresectable hepatocellular carcinoma: an international propensity score matching analysis. ESMO Open. 2022;7(6):100591. doi:10.1016/j.esmoop.2022.100591. [Google Scholar] [PubMed] [CrossRef]
50. Graepler F, Nissler V, Scharpf M, Vonthein R, Heidenreich R, Wehrmann M, et al. T2016 the angiogenesis inhibitor VEGF trap (aflibercepta soluble VEGF decoy receptor, inhibits tumor growth in a human HCC xenograft model. Gastroenterology. 2009;136(5):A-620. doi:10.1016/S0016-5085(09)62862-6. [Google Scholar] [CrossRef]
51. Chen YH, Tsai CH, Chen YY, Wang CC, Wang JH, Hung CH, et al. Real-world comparison of pembrolizumab and nivolumab in advanced hepatocellular carcinoma. BMC Cancer. 2023;23(1):810. doi:10.1186/s12885-023-11298-z. [Google Scholar] [PubMed] [CrossRef]
52. Psilopatis I, Damaskos C, Garmpi A, Sarantis P, Koustas E, Antoniou EA, et al. FDA-approved monoclonal antibodies for unresectable hepatocellular carcinoma: what do we know so far? Int J Mol Sci. 2023;24(3):2685. doi:10.3390/ijms24032685. [Google Scholar] [PubMed] [CrossRef]
53. Takagi H, Kaji K, Nishimura N, Ishida K, Ogawa H, Takaya H, et al. The angiotensin II receptor blocker losartan sensitizes human liver cancer cells to lenvatinib-mediated cytostatic and angiostatic effects. Cells. 2021;10(3):575. doi:10.3390/cells10030575. [Google Scholar] [PubMed] [CrossRef]
54. Song Y, Kim JS, Choi EK, Kim J, Kim KM, Seo HR. TGF-β-independent CTGF induction regulates cell adhesion mediated drug resistance by increasing collagen I in HCC. Oncotarget. 2017;8(13):21650–62. doi:10.18632/oncotarget.15521. [Google Scholar] [PubMed] [CrossRef]
55. Toh TB, Lim JJ, Hooi L, Rashid MBMA, Chow EK. Targeting Jak/Stat pathway as a therapeutic strategy against SP/CD44+ tumorigenic cells in Akt/β-catenin-driven hepatocellular carcinoma. J Hepatol. 2020;72(1):104–18. doi:10.1016/j.jhep.2019.08.035. [Google Scholar] [PubMed] [CrossRef]
56. Cao G, Zhang G, Liu M, Liu J, Wang Q, Zhu L, et al. GPC3-targeted CAR-T cells secreting B7H3-targeted BiTE exhibit potent cytotoxicity activity against hepatocellular carcinoma cell in the in vitro assay. Biochem Biophys Rep. 2022;31:101324. doi:10.1016/j.bbrep.2022.101324. [Google Scholar] [PubMed] [CrossRef]
57. Cheng J, Huang T, Li Y, Guo Y, Zhu Y, Wang Q, et al. AMP-activated protein kinase suppresses the in vitro and in vivo proliferation of hepatocellular carcinoma. PLoS One. 2014;9(4):e93256. doi:10.1371/journal.pone.0093256. [Google Scholar] [PubMed] [CrossRef]
58. Yoo JJ, Yu SJ, Na J, Kim K, Cho YY, Bin Lee Y, et al. Hexokinase-II inhibition synergistically augments the anti-tumor efficacy of sorafenib in hepatocellular carcinoma. Int J Mol Sci. 2019;20(6):1292. doi:10.3390/ijms20061292. [Google Scholar] [PubMed] [CrossRef]
59. Wright T, Wang Y, Bedford MT. The role of the PRMT5-SND1 axis in hepatocellular carcinoma. Epigenomes. 2021;5(1):2. doi:10.3390/epigenomes5010002. [Google Scholar] [PubMed] [CrossRef]
60. Zhou H, Cai Y, Liu D, Li M, Sha Y, Zhang W, et al. Pharmacological or transcriptional inhibition of both HDAC1 and 2 leads to cell cycle blockage and apoptosis via p21(Waf1/Cip1) and p19(INK4d) upregulation in hepatocellular carcinoma. Cell Prolif. 2018;51(3):e12447. doi:10.1111/cpr.12447. [Google Scholar] [PubMed] [CrossRef]
61. Pan L, Feng F, Wu J, Fan S, Han J, Wang S, et al. Demethylzeylasteral targets lactate by inhibiting histone lactylation to suppress the tumorigenicity of liver cancer stem cells. Pharmacol Res. 2022;181(10127):106270. doi:10.1016/j.phrs.2022.106270. [Google Scholar] [PubMed] [CrossRef]
62. Liu Y, Guo Q, Yang H, Zhang XW, Feng N, Wang JK, et al. Allosteric regulation of IGF2BP1 as a novel strategy for the activation of tumor immune microenvironment. ACS Cent Sci. 2022;8(8):1102–15. doi:10.1021/acscentsci.2c00107. [Google Scholar] [PubMed] [CrossRef]
63. Abbastabar M, Sarfi M, Golestani A, Khalili E. lncRNA involvement in hepatocellular carcinoma metastasis and prognosis. EXCLI J. 2018;17:900–13. doi:10.17179/excli2018-1541. [Google Scholar] [PubMed] [CrossRef]
64. Tat Trung N, Duong DC, Van Tong H, Hien TTT, Hoan PQ, Bang MH, et al. Optimisation of quantitative miRNA panels to consolidate the diagnostic surveillance of HBV-related hepatocellular carcinoma. PLoS One. 2018;13(4):e0196081. doi:10.1371/journal.pone.0196081. [Google Scholar] [PubMed] [CrossRef]
65. Tong M, Fung TM, Luk ST, Ng KY, Lee TK, Lin CH, et al. ANXA3/JNK signaling promotes self-renewal and tumor growth, and its blockade provides a therapeutic target for hepatocellular carcinoma. Stem Cell Rep. 2015;5(1):45–59. doi:10.1016/j.stemcr.2015.05.013. [Google Scholar] [PubMed] [CrossRef]
66. Xie M, Sun M, Ji X, Li D, Chen X, Zhang B, et al. Overexpression of BACH1 mediated by IGF2 facilitates hepatocellular carcinoma growth and metastasis via IGF1R and PTK2. Theranostics. 2022;12(3):1097–116. doi:10.7150/thno.65775. [Google Scholar] [PubMed] [CrossRef]
67. Zeng L, Fan X, Wang X, Deng H, Zhang X, Zhang K, et al. Involvement of NEK2 and its interaction with NDC80 and CEP250 in hepatocellular carcinoma. BMC Med Genomics. 2020;13(1):158. doi:10.1186/s12920-020-00812-y. [Google Scholar] [PubMed] [CrossRef]
68. Liu F, Liao Z, Qin L, Zhang Z, Zhang Q, Han S, et al. Targeting VPS72 inhibits ACTL6A/MYC axis activity in HCC progression. Hepatology. 2023;78(5):1384–401. doi:10.1097/HEP.0000000000000268. [Google Scholar] [PubMed] [CrossRef]
69. Liu J, Chen Z, Zhao P, Li W. Prognostic and immune regulating roles of YIF1B in Pan-Cancer: a potential target for both survival and therapy response evaluation. Biosci Rep. 2020;40(7):BSR20201384. doi:10.1042/BSR20201384. [Google Scholar] [PubMed] [CrossRef]
70. Qin S, Chen M, Cheng AL, Kaseb AO, Kudo M, Lee HC, et al. Atezolizumab plus bevacizumab versus active surveillance in patients with resected or ablated high-risk hepatocellular carcinoma (IMbrave050a randomised, open-label, multicentre, phase 3 trial. Lancet. 2023;402(10415):1835–47. doi:10.1016/S0140-6736(23)01796-8. [Google Scholar] [PubMed] [CrossRef]
71. Salazar R, Tafuto S, Krogh M, Teule A, Garcia-Carbonero R, Klumpen HJ, et al. LBA45 Randomized open label phase III study comparing the efficacy and safety of everolimus followed by chemotherapy (CT) with streptozotocin (STZ)-5FU upon progression or the reverse sequence, in advanced progressive panNETs: the SEQTOR study (GETNE 1206). Ann Oncol. 2022;33:S1412. doi:10.1016/j.annonc.2022.08.044. [Google Scholar] [CrossRef]
72. Yau T, Galle PR, Decaens T, Sangro B, Qin S, da Fonseca LG, et al. Nivolumab plus ipilimumab versus lenvatinib or sorafenib as first-line treatment for unresectable hepatocellular carcinoma (CheckMate 9DWan open-label, randomised, phase 3 trial. Lancet. 2025;405(10492):1851–64. doi:10.1016/S0140-6736(25)00403-9. [Google Scholar] [PubMed] [CrossRef]
73. Kudo M. Scientific rationale for combination immunotherapy of hepatocellular carcinoma with anti-PD-1/PD-L1 and anti-CTLA-4 antibodies. Liver Cancer. 2019;8(6):413–26. doi:10.1159/000503254. [Google Scholar] [PubMed] [CrossRef]
74. Nakagawa T, Tohyama O, Yamaguchi A, Matsushima T, Takahashi K, Funasaka S, et al. E7050: a dual c-Met and VEGFR-2 tyrosine kinase inhibitor promotes tumor regression and prolongs survival in mouse xenograft models. Cancer Sci. 2010;101(1):210–5. doi:10.1111/j.1349-7006.2009.01343.x. [Google Scholar] [PubMed] [CrossRef]
75. Cheng AL, Hsu C, Chan SL, Choo SP, Kudo M. Challenges of combination therapy with immune checkpoint inhibitors for hepatocellular carcinoma. J Hepatol. 2020;72(2):307–19. doi:10.1016/j.jhep.2019.09.025. [Google Scholar] [PubMed] [CrossRef]
76. Wang W, Sun H, Gong Y, Liu X, Liu X, Wang M, et al. Ratiometric co-delivery of hydroxychloroquine and calculated low-dose paclitaxel efficiently suppresses tumor growth in hepatocellular carcinoma mouse models in vivo. Nano Today. 2022;44(15):101446. doi:10.1016/j.nantod.2022.101446. [Google Scholar] [CrossRef]
77. Himmelsbach V, Koch C, Trojan J, Finkelmeier F. Systemic drugs for hepatocellular carcinoma: what do recent clinical trials reveal about sequencing and the emerging complexities of clinical decisions? J Hepatocell Carcinoma. 2024;11:363–72. doi:10.2147/JHC.S443218. [Google Scholar] [PubMed] [CrossRef]
78. Hu L, Shi W, Liu K, Ma D, Xin Q, Wang Z, et al. EGFR bypass activation mediates acquired resistance to regorafenib in hepatocellular carcinoma. Front Med. 2024;11:1464610. doi:10.3389/fmed.2024.1464610. [Google Scholar] [PubMed] [CrossRef]
79. Zhang Z, Zhang W, Wang Y, Wan T, Hu B, Li C, et al. Construction and validation of a ferroptosis-related lncRNA signature as a novel biomarker for prognosis, immunotherapy and targeted therapy in hepatocellular carcinoma. Front Cell Dev Biol. 2022;10:792676. doi:10.3389/fcell.2022.792676. [Google Scholar] [PubMed] [CrossRef]
80. Bang J, Jun M, Lee S, Moon H, Ro SW. Targeting EGFR/PI3K/AKT/mTOR signaling in hepatocellular carcinoma. Pharmaceutics. 2023;15(8):2130. doi:10.3390/pharmaceutics15082130. [Google Scholar] [PubMed] [CrossRef]
81. Sun W, Cabrera R. Systemic treatment of patients with advanced, unresectable hepatocellular carcinoma: emergence of therapies. J Gastrointest Cancer. 2018;49(2):107–15. doi:10.1007/s12029-018-0065-8. [Google Scholar] [PubMed] [CrossRef]
82. Llovet J, Finn RS, Ren Z, Guo Y, Han G, Lin H, et al. LBA3 Transarterial chemoembolization (TACE) with or without lenvatinib (len) + pembrolizumab (pembro) for intermediate-stage hepatocellular carcinoma (HCCphase III LEAP-012 study. Ann Oncol. 2024;35:S1229. doi:10.1016/j.annonc.2024.08.2277. [Google Scholar] [CrossRef]
83. Villalobos A, Dabbous HH, Little O, Gbolahan OB, Akce M, Lilly MA, et al. Safety and efficacy of concurrent atezolizumab/bevacizumab or nivolumab combination therapy with yttrium-90 radioembolization of advanced unresectable hepatocellular carcinoma. Curr Oncol. 2023;30(12):10100–10. doi:10.3390/curroncol30120734. [Google Scholar] [PubMed] [CrossRef]
84. Li T, Xue Y, Wang G, Gu T, Li Y, Zhu YY, et al. Multi-target siRNA: therapeutic strategy for hepatocellular carcinoma. J Cancer. 2016;7(10):1317–27. doi:10.7150/jca.15157. [Google Scholar] [PubMed] [CrossRef]
85. Wu YY, Chen L, Wang GL, Zhang YX, Zhou JM, He S, et al. Inhibition of hepatocellular carcinoma growth and angiogenesis by dual silencing of NET-1 and VEGF. J Mol Histol. 2013;44(4):433–45. doi:10.1007/s10735-012-9480-5. [Google Scholar] [PubMed] [CrossRef]
86. Harding-Theobald E, Louissaint J, Maraj B, Cuaresma E, Townsend W, Mendiratta-Lala M, et al. Systematic review: radiomics for the diagnosis and prognosis of hepatocellular carcinoma. Aliment Pharmacol Ther. 2021;54(7):890–901. doi:10.1111/apt.16563. [Google Scholar] [PubMed] [CrossRef]
87. Lin HY, Jeon AJ, Chen K, Lee CJM, Wu L, Chong SL, et al. The epigenetic basis of hepatocellular carcinoma-mechanisms and potential directions for biomarkers and therapeutics. Br J Cancer. 2025;132(10):869–87. doi:10.1038/s41416-025-02969-8. [Google Scholar] [PubMed] [CrossRef]
88. Fujiwara N, Matsushita Y, Tempaku M, Tachi Y, Kimura G, Izuoka K, et al. AI-based phenotyping of hepatic fiber morphology to inform molecular alterations in metabolic dysfunction-associated steatotic liver disease. Hepatology. 2025. doi:10.1097/HEP.0000000000001360. [Google Scholar] [PubMed] [CrossRef]
89. Maki A, Kono H, Gupta M, Asakawa M, Suzuki T, Matsuda M, et al. Predictive power of biomarkers of oxidative stress and inflammation in patients with hepatitis C virus-associated hepatocellular carcinoma. Ann Surg Oncol. 2007;14(3):1182–90. doi:10.1245/s10434-006-9049-1. [Google Scholar] [PubMed] [CrossRef]
90. Li J, Liang YB, Wang QB, Luo WL, Chen XM, Lakang Y, et al. Rechallenge with immune checkpoint inhibitors in patients with hepatocellular carcinoma: a narrative review. Liver Cancer. 2025:1–20. doi:10.1159/000549355. [Google Scholar] [PubMed] [CrossRef]
91. Wu S, Anand N, Guo Z, Li M, Santiago Figueroa M, Jung L, et al. Bridging immune evasion and vascular dynamics for novel therapeutic frontiers in hepatocellular carcinoma. Cancers. 2025;17(11):1860. doi:10.3390/cancers17111860. [Google Scholar] [PubMed] [CrossRef]
92. Yin L, Liu R, Li W, Li S, Hou X. Deep learning-based CT radiomics predicts prognosis of unresectable hepatocellular carcinoma treated with TACE-HAIC combined with PD-1 inhibitors and tyrosine kinase inhibitors. BMC Gastroenterol. 2025;25(1):24. doi:10.1186/s12876-024-03555-7. [Google Scholar] [PubMed] [CrossRef]
93. Jiang YQ, Cao SE, Cao S, Chen JN, Wang GY, Shi WQ, et al. Preoperative identification of microvascular invasion in hepatocellular carcinoma by XGBoost and deep learning. J Cancer Res Clin Oncol. 2021;147(3):821–33. doi:10.1007/s00432-020-03366-9. [Google Scholar] [PubMed] [CrossRef]
94. Xu J, Wang T, Li J, Wang Y, Zhu Z, Fu X, et al. A multimodal fusion system predicting survival benefits of immune checkpoint inhibitors in unresectable hepatocellular carcinoma. npj Precis Oncol. 2025;9(1):185. doi:10.1038/s41698-025-00979-6. [Google Scholar] [PubMed] [CrossRef]
95. Zhang JX, Yan HT, Fan WL, Wei J, Liu S, Shi HB, et al. An explainable machine learning model for predicting response to targeted therapy and immunotherapy in advanced hepatocellular carcinoma: a multicentric study. Res Square. 2024;74(3):229. doi:10.21203/rs.3.rs-5304318/v1. [Google Scholar] [CrossRef]
96. Liu P, Tan XZ, Zhang T, Gu QB, Mao XH, Li YC, et al. Prediction of microvascular invasion in solitary hepatocellular carcinoma ≤5 cm based on computed tomography radiomics. World J Gastroenterol. 2021;27(17):2015–24. doi:10.3748/wjg.v27.i17.2015. [Google Scholar] [PubMed] [CrossRef]
97. Zhou HY, Cheng JM, Chen TW, Zhang XM, Ou J, Cao JM, et al. CT radiomics for prediction of microvascular invasion in hepatocellular carcinoma: a systematic review and meta-analysis. Clinics. 2023;78(10127):100264. doi:10.1016/j.clinsp.2023.100264. [Google Scholar] [PubMed] [CrossRef]
98. Dai H, Lu M, Huang B, Tang M, Pang T, Liao B, et al. Considerable effects of imaging sequences, feature extraction, feature selection, and classifiers on radiomics-based prediction of microvascular invasion in hepatocellular carcinoma using magnetic resonance imaging. Quant Imaging Med Surg. 2021;11(5):1836–53. doi:10.21037/qims-20-218. [Google Scholar] [PubMed] [CrossRef]
99. Wang QB, Luo WL, Li YK, Li J, Yang ZS, Zhao K, et al. Tumor compression of the hepatic or portal vein predicts the presence of microvascular invasion and satellite nodules in hepatocellular carcinoma: a retrospective study. J Hepatocell Carcinoma. 2025;12:2055–67. doi:10.2147/JHC.S544589. [Google Scholar] [PubMed] [CrossRef]
100. Tran KA, Kondrashova O, Bradley A, Williams ED, Pearson JV, Waddell N. Deep learning in cancer diagnosis, prognosis and treatment selection. Genome Med. 2021;13(1):152. doi:10.1186/s13073-021-00968-x. [Google Scholar] [PubMed] [CrossRef]
101. Kiani I, Razeghian I, Valizadeh P, Esmaeilian Y, Jannatdoust P, Khosravi B. Performance of artificial intelligence models in predicting responsiveness of hepatocellular carcinoma to transarterial chemoembolization (TACEa systematic review and meta-analysis. J Am Coll Radiol. 2026;23(1):76–88. doi:10.1016/j.jacr.2025.08.028. [Google Scholar] [PubMed] [CrossRef]
102. Oselio B, Singal AG, Zhang X, Van T, Liu B, Zhu J, et al. Reinforcement learning evaluation of treatment policies for patients with hepatitis C virus. BMC Med Inform Decis Mak. 2022;22(1):63. doi:10.1186/s12911-022-01789-7. [Google Scholar] [PubMed] [CrossRef]
103. Maung ST, Tangkijvanich P, Chaiteerakij R. Magnetic resonance imaging radiomics for predicting hepatocellular carcinoma recurrence following resection or ablation: a systematic review and meta-analysis. Abdom Radiol Forthcoming. 2025;76(3):681. doi:10.1007/s00261-025-05184-4. [Google Scholar] [PubMed] [CrossRef]
104. Han Y, Zeng A, Liang X, Jiang Y, Wang F, Song L. Multi-omics analyses develop and validate the optimal prognostic model on overall survival prediction for resectable hepatocellular carcinoma. J Gastrointest Oncol. 2025;16(2):628–49. doi:10.21037/jgo-24-710. [Google Scholar] [PubMed] [CrossRef]
105. Wang KD, Guan MJ, Bao ZY, Shi ZJ, Tong HH, Xiao ZQ, et al. Radiomics analysis based on dynamic contrast-enhanced MRI for predicting early recurrence after hepatectomy in hepatocellular carcinoma patients. Sci Rep. 2025;15(1):22240. doi:10.1038/s41598-025-02291-6. [Google Scholar] [PubMed] [CrossRef]
106. Naveed M, Jamil H, Aziz T, Makhdoom SI, Alamri AS, Alhomrani M, et al. Spatially resolved transcriptomics reveals local invasion-related genes in liver hepatocellular carcinoma: exploring the therapeutic potential of a chimeric protein targeting glypican-3. J Comput Biophys Chem. 2024;23(3):321–32. doi:10.1142/s2737416523500643. [Google Scholar] [CrossRef]
107. Binnewies M, Roberts EW, Kersten K, Chan V, Fearon DF, Merad M, et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med. 2018;24(5):541–50. doi:10.1038/s41591-018-0014-x. [Google Scholar] [PubMed] [CrossRef]
108. Safri F, Nguyen R, Zerehpooshnesfchi S, George J, Qiao L. Heterogeneity of hepatocellular carcinoma: from mechanisms to clinical implications. Cancer Gene Ther. 2024;31(8):1105–12. doi:10.1038/s41417-024-00764-w. [Google Scholar] [PubMed] [CrossRef]
109. Zarlashat Y, Ghaffar A, Guerra F, Picca A. Immunological landscape and molecular therapeutic targets of the tumor microenvironment in hepatocellular carcinoma. Int J Mol Sci. 2025;26(16):7836. doi:10.3390/ijms26167836. [Google Scholar] [PubMed] [CrossRef]
110. Qu S, Xu R, Yi G, Li Z, Zhang H, Qi S, et al. Patient-derived organoids in human cancer: a platform for fundamental research and precision medicine. Mol Biomed. 2024;5(1):6. doi:10.1186/s43556-023-00165-9. [Google Scholar] [PubMed] [CrossRef]
111. Vargas-Accarino E, Higuera M, Bermúdez-Ramos M, Soriano-Varela A, Torrens M, Pons M, et al. Harnessing plasma biomarkers to predict immunotherapy outcomes in hepatocellular carcinoma: the role of cfDNA, ctDNA, and cytokines. Int J Mol Sci. 2025;26(6):2794. doi:10.3390/ijms26062794. [Google Scholar] [PubMed] [CrossRef]
112. Duangchan K, Limjunyawong N, Rodponthukwaji K, Ittiudomrak T, Thaweesuvannasak M, Kunwong N, et al. Development of small interfering RNA loaded cationic lipid nanoparticles for the treatment of liver cancer with elevated α-fetoprotein expression. ACS Bio Med Chem Au. 2025;5(1):78–88. doi:10.1021/acsbiomedchemau.4c00061. [Google Scholar] [PubMed] [CrossRef]
113. Hou B, Qin L, Huang L. Liver cancer cells as the model for developing liver-targeted RNAi therapeutics. Biochem Biophys Res Commun. 2023;644:85–94. doi:10.1016/j.bbrc.2023.01.007. [Google Scholar] [PubMed] [CrossRef]
114. Zhu J, Huang Q, Peng X, Luo C, Liu Z, Liu D, et al. Identification of molecular subtypes based on PANoptosis-related genes and construction of a signature for predicting the prognosis and response to immunotherapy response in hepatocellular carcinoma. Front Immunol. 2023;14:1218661. doi:10.3389/fimmu.2023.1218661. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF



 Downloads
Downloads
 Citation Tools
Citation Tools