 Open Access
Open Access
ARTICLE
Determination of Bioactive Potential in a Semi-domesticated Population of Oregano (Lippia graveolens Kunth) with Organic Fertilization
1 Facultad de Agricultura y Zootecnia, Universidad Juárez del Estado de Durango, Carretera Gómez Palacio-Tlahualilo Km. 32. Venecia, Gómez Palacio, C.P. 35111, Dgo, Mexico
2 Instituto Tecnológico Superior de Lerdo, Av. Tecnológico N° 1555 Sur. Periférico Lerdo Km. 14.5, Placido Domingo, Ciudad Lerdo, C.P. 35150, Durango, Mexico
3 Centro de Investigación para los Recursos Naturales, Antigua Normal Rural SN, Saláices, C.P. 33941, Chihuahua, Mexico
* Corresponding Author: José Luis García-Hernández. Email:
Phyton-International Journal of Experimental Botany 2026, 95(2), 2 https://doi.org/10.32604/phyton.2026.074379
Received 10 October 2025; Accepted 24 December 2025; Issue published 28 February 2026
Abstract
Mexican oregano (Lippia graveolens Kunth) is an aromatic species of high culinary and medicinal relevance. In Mexico, more than 40 taxa with characteristic aroma and flavor are commercially recognized as oregano, with L. graveolens being the most widely distributed and economically important. Despite its relevance, few domesticated or semi-domesticated cultivars exist, and wild populations remain the main source of raw material, raising concerns regarding sustainability and quality standardization. The essential oil and oleoresins of L. graveolens possess recognized bioactivity, including antioxidant, antifungal, antibacterial, and anti-inflammatory properties, largely attributed to phenolic compounds such as thymol and carvacrol. Given the increasing global demand for natural bioactives and functional foods, optimizing cultivation practices is essential to enhance both the yield and phytochemical quality of this species. This study evaluated the effect of organic fertilization (0, 5, 10, and 15 t ha−1 of vermicompost) on the phenolic profile, flavonoid content, and antioxidant capacity of oregano oleoresin obtained from a semi-domesticated population across three harvests. The highest phenolic concentration (≈500 mg GAE g−1 extract) and greatest antioxidant activity (ABTS > 3.5 × 105 µmol TE g−1 extract) were observed at the 5 t ha−1 dose during the second harvest. Flavonoid content peaked in the third harvest (480–620 mg QE g−1 extract), whereas the unfertilized control exhibited the highest DPPH activity in the first harvest. Overall, the results indicate that vermicompost dosage and harvest timing substantially influence the functional quality of L. graveolens. Moderate organic fertilization, particularly 5 t ha−1, enhances the biosynthesis of bioactive secondary metabolites, underscoring its potential for sustainable production systems.Keywords
Oregano is an aromatic–medicinal plant widely known and used worldwide in food preparation due to its aromatic and organoleptic qualities [1]. There are four major species (also referred to as groups) of oregano studied globally: Turkish (Origanum onites L.), Spanish (Coridohymus capitatus L.), Greek (O. vulgare L.), and Mexican oregano (Lippia graveolens Kunth) [2]. L. graveolens grows extensively in the wild, mainly in arid and semiarid regions [3] of different countries such as the United States, Guatemala, Mexico, and others [4]. In Mexico, around 40 related subspecies or ecotypes with characteristic smell and flavor are recognized as oregano; one of the most well-known and used is L. graveolens, distributed across more than 15 states [5,6].
Mexico is the second-largest oregano exporter worldwide, surpassed only by Turkey. Remarkably, the Codex Alimentarius Commission (2019) identified Mexico as the second-largest source of L. graveolens, making it the species with the broadest geographic distribution and highest economic importance in the country [7,8]. Oregano plants are primarily used to obtain essential oils (EOs) and various types of extracts composed of terpenes, which give oregano its strong aroma and flavor [9,10]. Currently, a large number of plants are known and valued for their EO content, and oregano is among the most widely used [11].
In recent years, the characterization of biologically active compounds in EOs has been widely investigated for use in the food and pharmaceutical industries [12,13]. Moreover, the demand for bioactive compounds is increasing; the EO market was estimated to have reached $3226.2 million in 2019 [14]. The essential oil of Mexican oregano has been reported to have high nutritional and therapeutic value, composed mainly of thymol and carvacrol, as well as phenolic compounds associated with antioxidant capacity [15], antifungal and antibacterial properties [16], food supplementation [17], insecticide production [18], and inhibition of microorganisms of industrial relevance, representing an ecological alternative for various applications [19]. It also shows high potential as a functional food due to the diversity of applications and benefits provided by its phytochemical profile [20].
Numerous factors influence the phytochemical and organoleptic properties of oregano. These characteristics vary according to chemotype, geographic and climatic factors, and agronomic management [4,21]. However, information on the ecophysiology of oregano remains scarce in both basic and applied aspects [22]. In Mexico, there are few cultivated varieties of L. graveolens, with wild populations representing the primary source of raw material [23]. One of the main limitations of this production system is the variability in chemical composition, which hinders commercialization, as the market demands homogeneity of its main components [4,24]. Cultivated oregano production has been considered unattractive to farmers because it is classified as an informal economy and due to insufficient knowledge dissemination about crop production systems. Nevertheless, the superiority of cultivated oregano in all studied variables compared to wild populations has been reported [25].
Cultivated oregano presents several advantages over wild oregano, particularly regarding agronomic control, phytochemical standardization, and sustainability. Several studies have shown that cultivation conditions allow optimization of essential oil yield [26]. Controlled cultivation enables a more stable chemical composition between production cycles, which is crucial for industrial applications requiring reproducible phytochemical profiles, such as the pharmaceutical and food industries [27]. A recent study conducted in Sicily showed that organic oregano production supplemented with organic amendments improves both biomass yield and the concentration of compounds of interest [28]. Furthermore, intensive harvesting of wild oregano can pose an ecological threat, whereas cultivated oregano offers a sustainable alternative [29].
Therefore, it is highly important to generate information and promote sustainable technification for this type of crop to mitigate ecosystem impacts and the decline of native populations, as well as to ensure resource availability to meet current market demand [30]. The main objectives of this study were to characterize the oleoresin of Mexican oregano and to evaluate the content of total phenolic compounds, flavonoids, and antioxidant activity in a semi-domesticated oregano population under different organic fertilization doses.
The experiment was conducted in 2024 at an experimental site belonging to the Center for Research on Natural Resources (CIRENA), located at 27° 54′ 09″ North and 105° 12′ 22″ West, in the community of Salaices, municipality of López, in the state of Chihuahua, at an elevation of 1500 m above sea level. The climate is extremely semi-arid, with a maximum temperature of 41.7°C and a minimum of –14.1°C. The annual average temperature is 18.3°C. The average annual rainfall is 363.9 mm, with an annual average of 61 rainy days and a relative humidity of 48%.
2.2 Treatments and Experimental Design
The oregano plot at the Experimental Station had not been cultivated for 5 years. During this time, no conventional crop management practices were applied; for this reason, the plot is considered not wild but a semi-domesticated population. The total plot area was 90 m2. Soil analyses were conducted to determine the initial conditions of the soil, and final analyses were performed to assess the possible effects of organic fertilization on its basic physicochemical properties (Table 1).
Table 1: Initial and final physicochemical characteristics of the soil in the experiment.
| Initial Characteristics | Final Characteristics | |
|---|---|---|
| E.C. (mScm−1) | 2.29 | 1.51 |
| C.E.C. (mEq/100 g) | 27.07 | 29.83 |
| pH | 7.94 | 7.95 |
| O.M. (%) | 2.66 | 2.61 |
| N-NO3 (%) | 5.98 | 9.44 |
| P (ppm) | 13.73 | 11.36 |
| K (ppm) | 567.75 | 458.66 |
The biofertilizer used was obtained from GAIA Agro Orgánicos, which holds several certifications such as OMRI (OMRI Listed®) for use in organic agriculture under USDA National Organic Program (NOP) standards, product code (aeh-19640) [31]. The product characteristics are shown in Table 2.
Table 2: Characteristics of biofertilizer (worm humus).
| Nutritional Composition | |||
|---|---|---|---|
| Organic matter | 40% | Iron Fe | 76,000 PPM* |
| Nitrogen N | 1.8% | Zinc ZN | 435 PPM |
| Phosphorus P | 1% | Magnesium Mg | 0.9% |
| Potassium K | 2% | Manganese Mn | 650 PPM |
| Calcium Ca | 3% | Total organic acids | 6.5% |
| Phytobeneficialmicroorganisms | |||
| Aerobic bacteria | 1.5 × 106 | Trichoderma sp. | 553 propagules/g |
| Anaerobic bacteria | 3.0 × 104 | Aspergillus sp. | 275 propagules/g |
| Nitrifying bacteria | 1.9 × 105 | Actinomycetes | 3.1 × 104 |
| Bacillus sp. | 1.0 × 105 | ||
| Physical-Chemical Characteristics | |||
| Carbon-nitrogen ratio | less than 15 | Greasy grain size | <5 mm |
| pH | 7.5 | Density | 0.98 g/cm3 |
| Electrical conductivity ratio | (1:5) 3.7 | Humidity % (gravimetric method) | 24.5% |
The experiment followed a randomized block design with four treatments and three replications, totaling 12 experimental units (EU). On average, six plants were sampled per EU. Treatments consisted of different doses of organic fertilizer. Although few studies mention optimal doses for this species under open-field cultivation, several studies on crops such as pepper [32] and related species such as Salvia officinalis L. [33] were used as references to determine fertilization doses. The treatments consisted of soil application of earthworm humus at the following rates: T1 = 5 t ha−1, T2 = 10 t ha−1, T3 = 15 t ha−1, and T4 = 0 t ha−1 (control).
The experiment began on 15 May 2024, with field clearing, site measurements, and setup of the experimental design.
For harvest, a complete row was selected from each EU by choosing the most uniform row. An initial collection of plants from each selected row was conducted as the first crop cutting (C1). Afterward, all plants in the plot were harvested at 15 cm above ground level to ensure uniformity in the experiment.
On 16 May 2024, the fertilizer doses were applied to each EU. Two additional cuttings were carried out 60 (15 July 2024) and 140 (4 October 2024) days after fertilization. At 60 days, plants showed vigorous growth and height similar to—or even greater than—those harvested in C1. Irrigation was supplied via drip irrigation, with an average of six hours per week.
2.3 Sample Preparation and Obtaining Oregano Oleoresin (Passive Maceration)
All samples were washed with distilled water, and excess moisture was removed using brown paper. Drying was carried out at room temperature (25 ± 2°C) for 15 days. After dehydration, samples were ground into a fine powder [34].
Five grams of dried plant material were placed in a 100 mL beaker with 50 mL (1:10) of solvent (99.9% ethanol). The mixture was stirred using a magnetic plate (AC-CUPLATE™ ANALOG HOTPLATE STIRRER Labnet) at room temperature for 24 h [35].
The macerate was filtered through Whatman No. 4 filter paper into an Erlenmeyer flask. The solvent was recovered using a conventional distiller adapted to a water bath for better temperature control. Once the solvent was fully removed, the oleoresin was stored in 5 mL amber vials until analysis.
2.4 Quantification of Total Phenolic Compounds
Total phenolic content was quantified using the method by Singleton et al. [36], with modifications for microplates. This analysis is based on the reaction of phenolic compounds with the Folin–Ciocalteu reagent in an alkaline medium, generating a blue coloration proportional to phenol concentration, measured at 740 nm. The reaction mixture consisted of 30 µL of extract (prepared at 0.125 mg oleoresin/mL solvent), 150 µL of Folin–Ciocalteu reagent, and 120 µL of 7.5% sodium carbonate solution (pH 10). The mixture was incubated for 30 min at 25°C in the dark, after which absorbance was recorded in a microplate reader (VeloScan™ LUX, Thermo Scientific). Results were expressed as milligrams of gallic acid equivalents per gram of extract (mg GAE/g), using a calibration curve with gallic acid (0–0.4 mg/mL).
2.5 Quantification of Total Flavonoids
Total flavonoids were quantified following the colorimetric method described by Zhishen et al. [37], with modifications for microplate reading. This method involves the reaction of flavonoids with sodium nitrite (NaNO2) and subsequently with aluminum chloride (AlCl3) under alkaline conditions provided by sodium hydroxide (NaOH). Hydroxyl groups of flavonoids form stable complexes with aluminum ions, producing an intense yellow color.
The Samples were incubated for 15 min in the dark at room temperature, and absorbance was measured at 510 nm using a multimode microplate reader (VeloScan™ LUX, Thermo Scientific). Flavonoid concentration was calculated using a quercetin calibration curve, and results were expressed as milligrams of quercetin equivalents per gram of extract (mg QE/g).
2.6 Antioxidant Capacity Assessment
Antioxidant capacity was evaluated using the ABTS+ and DPPH• assays with a VeloScan™ LUX microplate reader (Thermo Scientific) and 96-well plates. Absolute ethanol was used as the blank in both assays.
For the ABTS+ assay, adapted from Re et al. [38], antioxidants in the extract neutralize the ABTS•+ cation radical generated by oxidation with potassium persulfate. The reaction was initiated by mixing 20 µL extract with 280 µL ABTS•+ solution, followed by incubation for 7 min at 25°C in darkness, and absorbance was measured at 734 nm.
For the DPPH method, adapted from Palafox-Carlos et al. [39], 20 µL extract were mixed with 280 µL DPPH• solution in methanol (0.1 mM), incubated 30 min in darkness at room temperature, and absorbance was measured at 515 nm. Results were expressed as Trolox equivalents per gram of extract: mg TE/g for ABTS+ and µmol TE/g for DPPH.
Data are expressed as mean ± SD from three replicates. Normality and homogeneity of variance were verified before ANOVA. Correlations among parameters were evaluated with Pearson’s correlation test (p < 0.05). Statistical Analysis Software (SAS) version 9.0 [40] was used with the ANOVA procedure (two-way). LSD tests (p < 0.05) were used to determine differences between treatments (fertilization rate/cut). Figures were created with SigmaPlot version 12.0 [41].
Quantification of total phenolic compounds in oregano oleoresin showed significant differences among treatments with different doses (0, 5, 10, and 15 t ha−1) and among the three crop cuttings (C1, C2, and C3) (Fig. 1).
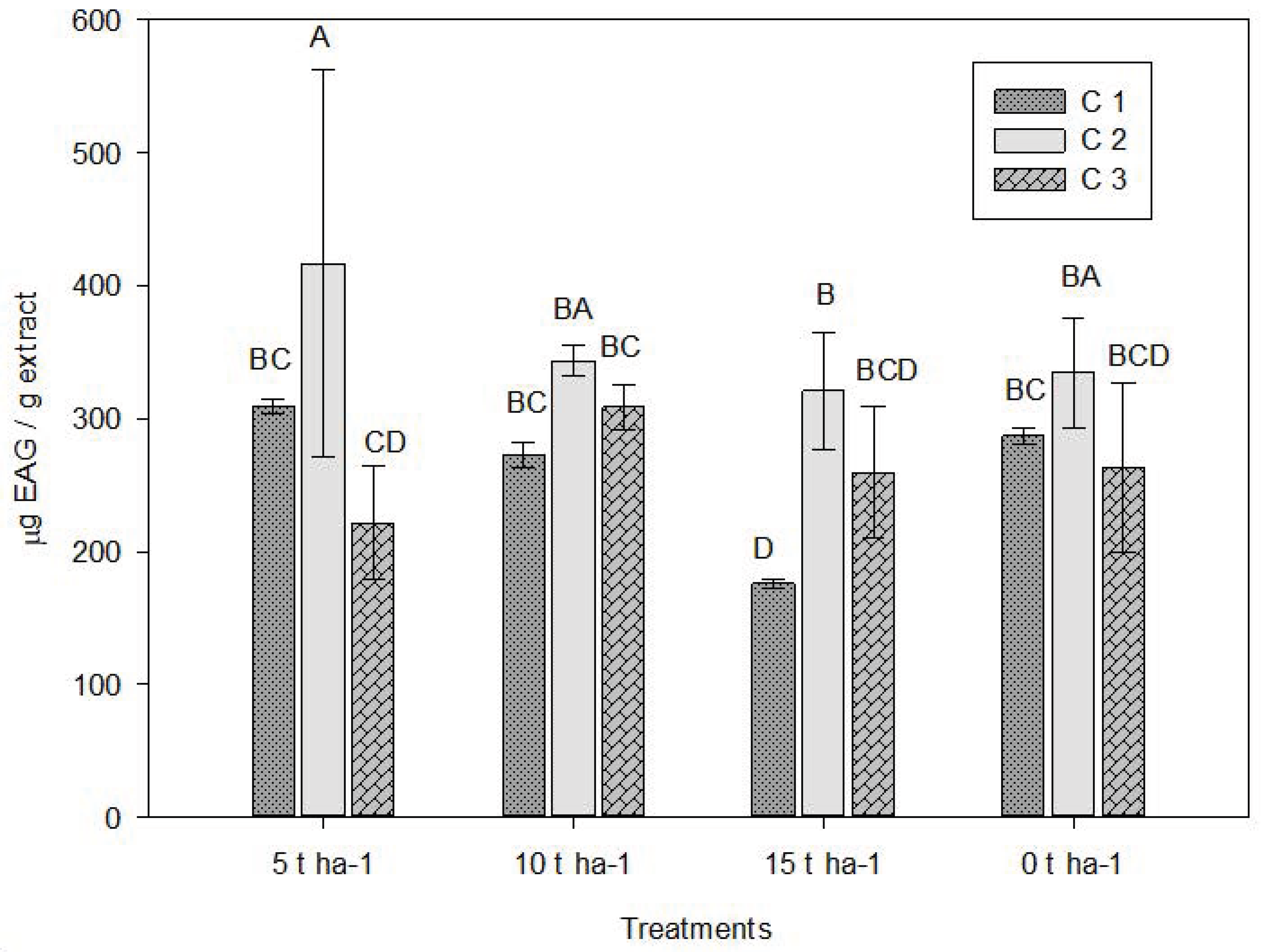
Figure 1: Average values obtained for total phenolics compounds. C1: Crop cutting one; C2: Crop cutting two; C3: Crop cutting three. Values presented are the means ± 1 S.D. Different letters above histograms indicate statistically significant differences using the least significant difference (LSD) test at p ≤ 0.05 among the mean values of the different treatments at the 3 sampling dates.
The highest phenolic content was observed in the 5 t ha−1 treatment under C2, reaching approximately 500 mg GAE/g extract (Table 3). Similar values were reported by Ríos et al. [42] in Mexican oregano essential oils obtained from different regions. Overall, the 5 t ha−1 treatment promoted higher phenolic compound concentrations compared to the other treatments. These values exceeded those reported by Fukalova et al. [43] in a comparative study of oregano morphotypes, as well as those reported by Aarland et al. [44].
Table 3: Content of total phenolic compounds and flavonoids in the different treatments.
| Code | Treatment | Total Phenolic Compounds µg EAG/g of Extract | Total Flavonoids µg QT/g of Extract |
|---|---|---|---|
| C1T1 | 5 t ha−1 | 309.3735 ± 5.7896 BC | 276.4552 ± 11.7702 EDF |
| C1T2 | 10 t ha−1 | 273.0368 ± 9.5492 BC | 189.0659 ± 46.2562 EF |
| C1T3 | 15 t ha−1 | 175.8697 ± 3.2149 D | 157.5087 ± 10.2794 F |
| C1T4 | 0 t ha−1 | 287.0332 ± 6.1535 BC | 172.4204 ± 80.1101 EF |
| C2T1 | 5 t ha−1 | 416.8584 ± 145.2637 A | 411.1225 ± 247.0545 BDC |
| C2T2 | 10 t ha−1 | 343.7364 ± 12.0743 BA | 231.7202 ± 128.3597 EDF |
| C2T3 | 15 t ha−1 | 320.8577 ± 44.1861 B | 340.4568 ± 191.9727 EDC |
| C2T4 | 0 t ha−1 | 334.4055 ± 41.8707 BA | 414.6481 ± 64.5826 BDC |
| C3T1 | 5 t ha−1 | 221.6271 ± 42.1913 CD | 606.2456 ± 109.0222 A |
| C3T2 | 10 t ha−1 | 308.5660 ± 17.5513 BC | 507.5281 ± 13.1427 BAC |
| C3T3 | 15 t ha−1 | 259.3993 ± 49.8917 BCD | 596.8824 ± 36.7344 A |
| C3T4 | 0 t ha−1 | 262.8984 ± 63.6276 BCD | 542.6687 ± 117.0320 BA |
Values presented are the means ± standard deviation of n = 36. Values followed by different letters in the same column indicate statistically significant differences using the least significant difference (LSD) test at p ≤ 0.05.
The 10 t ha−1 treatment yielded intermediate values, while 15 t ha−1 produced the lowest phenolic content, particularly under C1. The control (0 t ha−1) also exhibited relatively low levels. Results suggest that applying 5 t ha−1 of vermicompost favors biosynthesis or accumulation of phenolic compounds in oregano oleoresin, especially in C2. This may result from moderate stimulation of secondary metabolism promoted by improved nutrient availability or optimal physiological conditions [45].
In contrast, the highest dose (15 t ha−1) may cause nutrient excess or imbalance that inhibits synthesis of secondary metabolites, negatively affecting phytochemical qualities of the extract. Differences among cuttings may be attributed to biotic or abiotic factors [46].
The According to the LSD test (p ≤ 0.05), there were statistically significant differences among treatments (Table 2). In all four treatments, the third crop cutting (C3) consistently showed the highest total flavonoid values, ranging from 480–620 mg QE/g extract. C2 showed intermediate values, while C1 presented the lowest.
Although no clear linear trend was observed, the highest peaks in C3 occurred at 5 t ha−1 and 15 t ha−1 (610 and 620 mg QE/g extract), exceeding the values (25.8–180.7 mg CE/g extract) reported in a study of alcoholic extracts from Mexican oregano residues [47]. The intermediate dose (10 t ha−1) showed a slight decrease, suggesting possible inhibition at medium doses. The control treatment showed moderate values. Based on the results, there appears to be an increasing trend in flavonoid accumulation over time (Fig. 2).
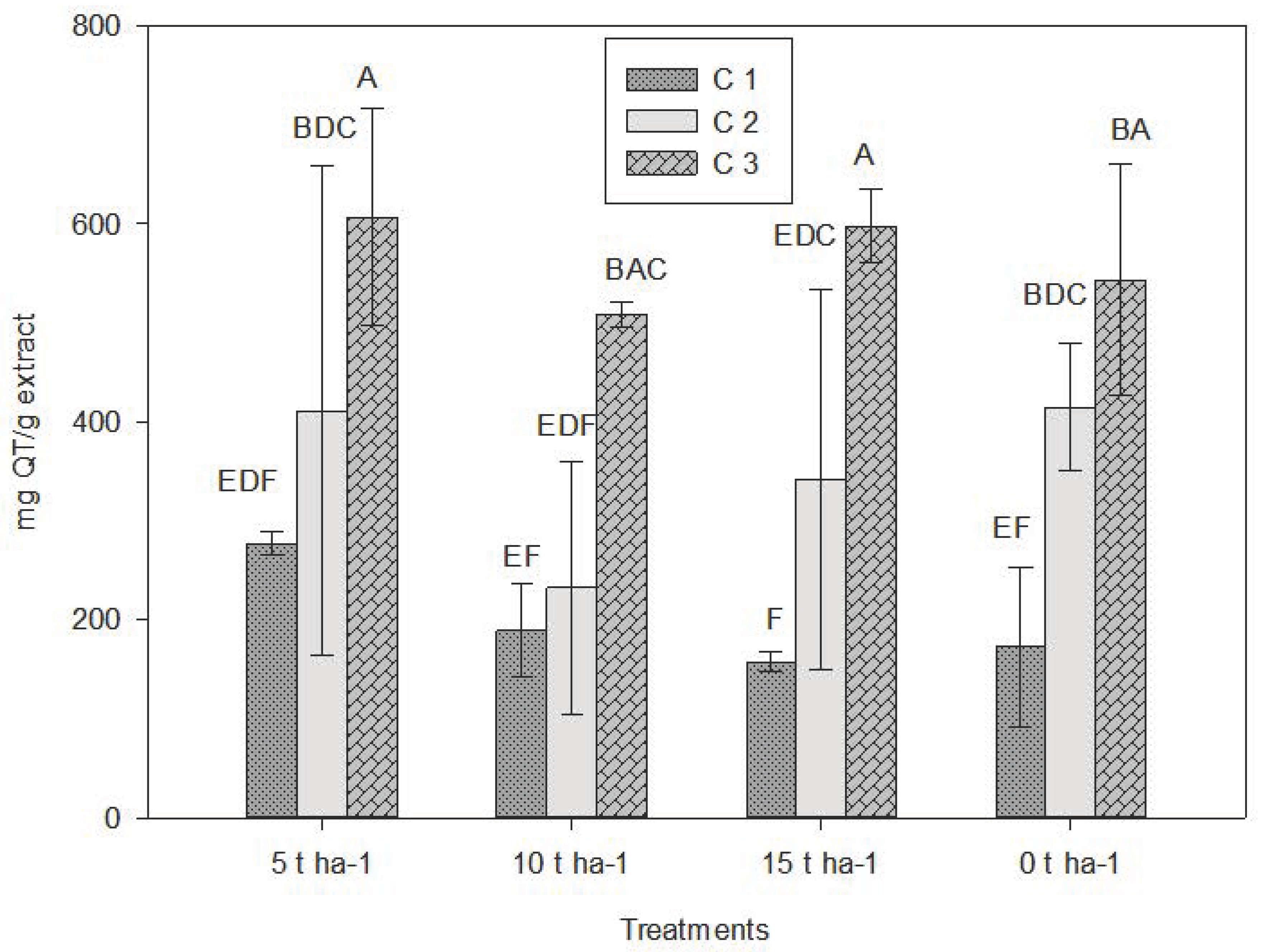
Figure 2: Average values obtained for total flavonoid. C1: Crop cutting one; C2: Crop cutting two; C3: Crop cutting three. Values presented are the means ± 1 S.D. Different letters above histograms indicate statistically significant differences using the least significant difference (LSD) test at p ≤ 0.05 among the mean values of the different treatments at the 3 sampling dates.
The progressive increase from C1 to C3 suggests that secondary metabolites such as flavonoids continue to accumulate over time. This may be related to biotic and abiotic stresses that activate biosynthetic pathways promoting flavonoid synthesis [48].
The treatment showing the highest values (5 t ha−1) may indicate that this dose induces moderate stress and nutritional conditions that favor flavonoid synthesis [49]. This supports the “optimal moderate stress” theory, which states that suboptimal conditions stimulate secondary metabolite production as an adaptive mechanism [50]. In this study, hormesis induced under the lowest fertilization dose likely increased flavonoid synthesis [51,52]. Studies on aromatic plants report that mild water deficit, high radiation, or moderate salinity can increase essential oils, phenolic compounds, and flavonoids [53,54].
Thus, harvest timing and fertilization dose must be considered together. Optimal crop management combined with fertilization can maximize oregano’s bioactive compounds. This aligns with findings by Bettaieb et al. [55], who showed that phenological stage and nutritional conditions influence the phenolic profile of Origanum spp. and other aromatic species.
Results showed significant statistical differences among treatments (Table 4). The 5 t ha−1 vermicompost dose promoted the highest antioxidant capacity, especially in C2, exceeding 3.5 × 10⁵ µmol TE g−1 extract. These values were higher than those reported by Uskutoğlu T. [56] in a study on three oregano species, where 997 µg TE/g dry extract were recorded.
Table 4: Values obtained for antioxidant capacity by the ABTS and DPPH methods.
| Code | Treatment | ABTS Inhibition µmol TE/g of Extract | DPPH Inhibition µmol TE/g of Extract |
|---|---|---|---|
| C1T1 | 5 t ha−1 | 188,797.6979 ± 4708.6835 B | 1009.3136 ± 54.9782 B |
| C1T2 | 10 t ha−1 | 193,439.1796 ± 1792.6377 B | 904.0366 ± 44.6918 CB |
| C1T3 | 15 t ha−1 | 173,132.6972 ± 2160.4921 CBD | 1035.8401 ± 63.1674 B |
| C1T4 | 0 t ha−1 | 184,852.4385 ± 6557.2052 CB | 1512.2017 ± 65.9021 A |
| C2T1 | 5 t ha−1 | 312,472.4978 ± 50,387.5836 A | 871.2493 ± 220.9848 CB |
| C2T2 | 10 t ha−1 | 139,366.4216 ± 8772.5680 EFD | 1014.7353 ± 65.3527 B |
| C2T3 | 15 t ha−1 | 131,150.1553 ± 20,805.7391 EF | 934.5876 ± 106.8487 CB |
| C2T4 | 0 t ha−1 | 147,092.8011 ± 18,599.2714 CEBD | 910.6925 ± 99.6810 CB |
| C3T1 | 5 t ha−1 | 120,665.3226 ± 4657.8201 F | 760.4711 ± 293.8250 C |
| C3T2 | 10 t ha−1 | 169,409.8006 ± 40,318.4049 CEBD | 1027.7705 ± 70.5968 B |
| C3T3 | 15 t ha−1 | 128,933.7730 ± 20,078.7612 F | 874.5957 ± 184.3167 CB |
| C3T4 | 0 t ha−1 | 141,130.1368 ± 20,782.3387 EFD | 1046.7305 ± 313.3746 B |
Values presented are the means ± standard deviation of n = 36. Values followed by different letters in the same column indicate statistically significant differences using the least significant difference (LSD) test at p ≤ 0.05. The antioxidant activity of DPPH and ABTS was performed by the inhibition curve.
Conversely, higher doses (10 and 15 t ha−1) were associated with progressive decreases in antioxidant capacity. C2 generally showed the highest antioxidant values compared to C1 and C3, indicating that harvest timing plays a critical role in the accumulation of secondary metabolites (Fig. 3).
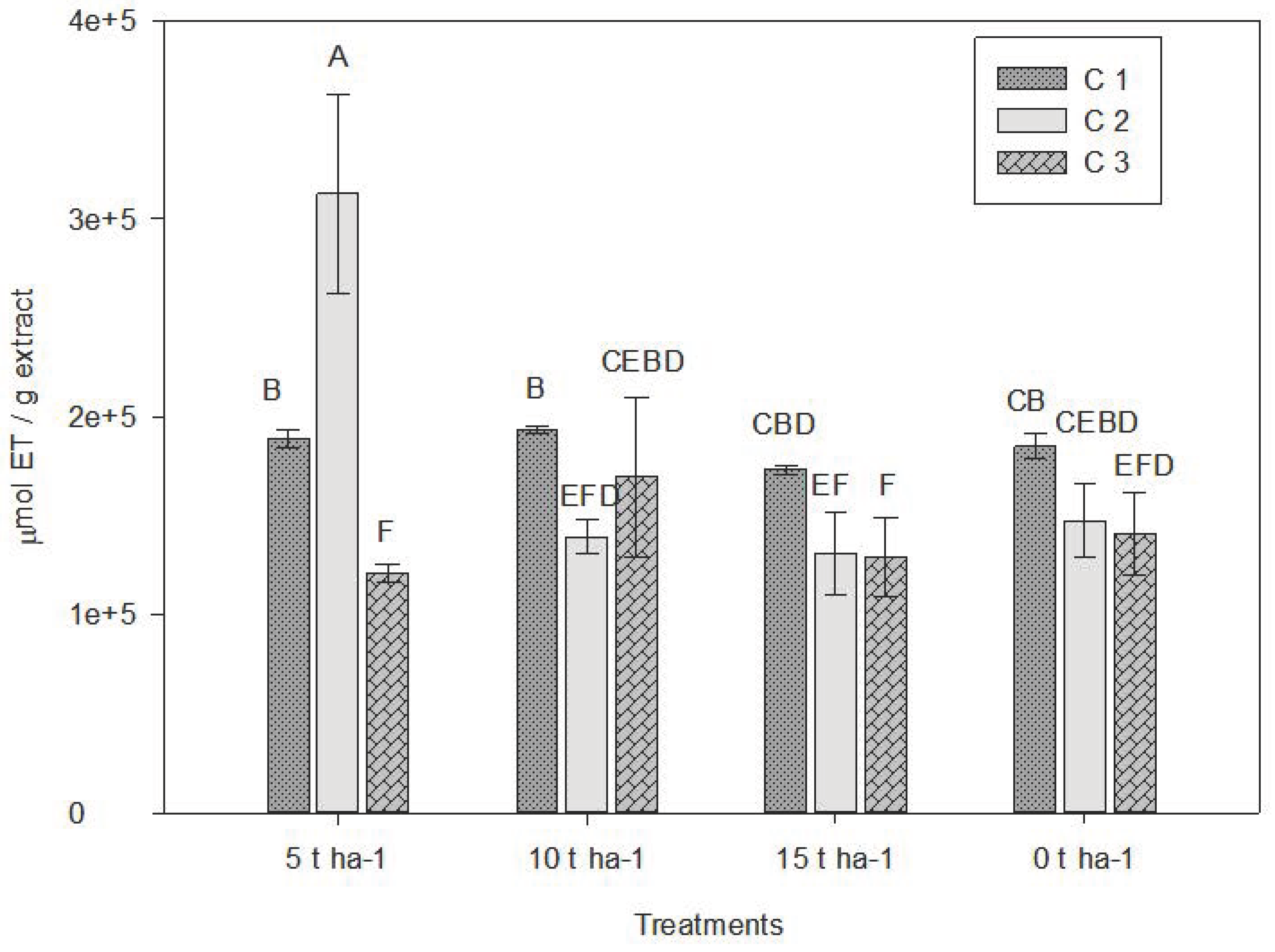
Figure 3: Average values obtained for antioxidant capacity by the ABTS method. C1: Crop cutting one; C2: Crop cutting two; C3: Crop cutting three. Values presented are the means ± 1 S.D. Different letters above histograms indicate statistically significant differences using the least significant difference (LSD) test at p ≤ 0.05 among the mean values of the different treatments at the 3 sampling dates.
Several studies have shown that the chemical composition of spices varies with harvest time, processing method, and postharvest handling, all of which significantly affect medicinal properties [57]. The timing of C2 coincided with the peak flowering stage, suggesting that this phenological phase is critical for maximizing antioxidant potential of L. graveolens, as also reported by Uskutoğlu [56].
Overall, For the DPPH method, significant statistical differences were also observed (Table 4). In C1, the highest antioxidant value (1500 µmol TE/g) was found in the control (0 t ha−1), suggesting that lack of fertilization induced moderate nutritional stress, increasing antioxidant compound synthesis and radical scavenging activity [58]. Additionally, fertilization may alter both the quantity and the phenolic profile—affecting relative proportions of compounds reacting with DPPH. Differences between control and treated samples may be linked to differences in free radical neutralization [59].
Variability between ABTS and DPPH values especially in the control may result from differences in solubility, reaction mechanisms (hydrogen vs. electron transfer), kinetics, and compound sensitivity. Comparative studies show that ABTS generally yields higher antioxidant values in plant extracts due to greater solubility in both aqueous and organic media [38,60]. As fertilization dose increased (from 5 to 15 t ha−1), antioxidant capacity generally decreased (Fig. 4).
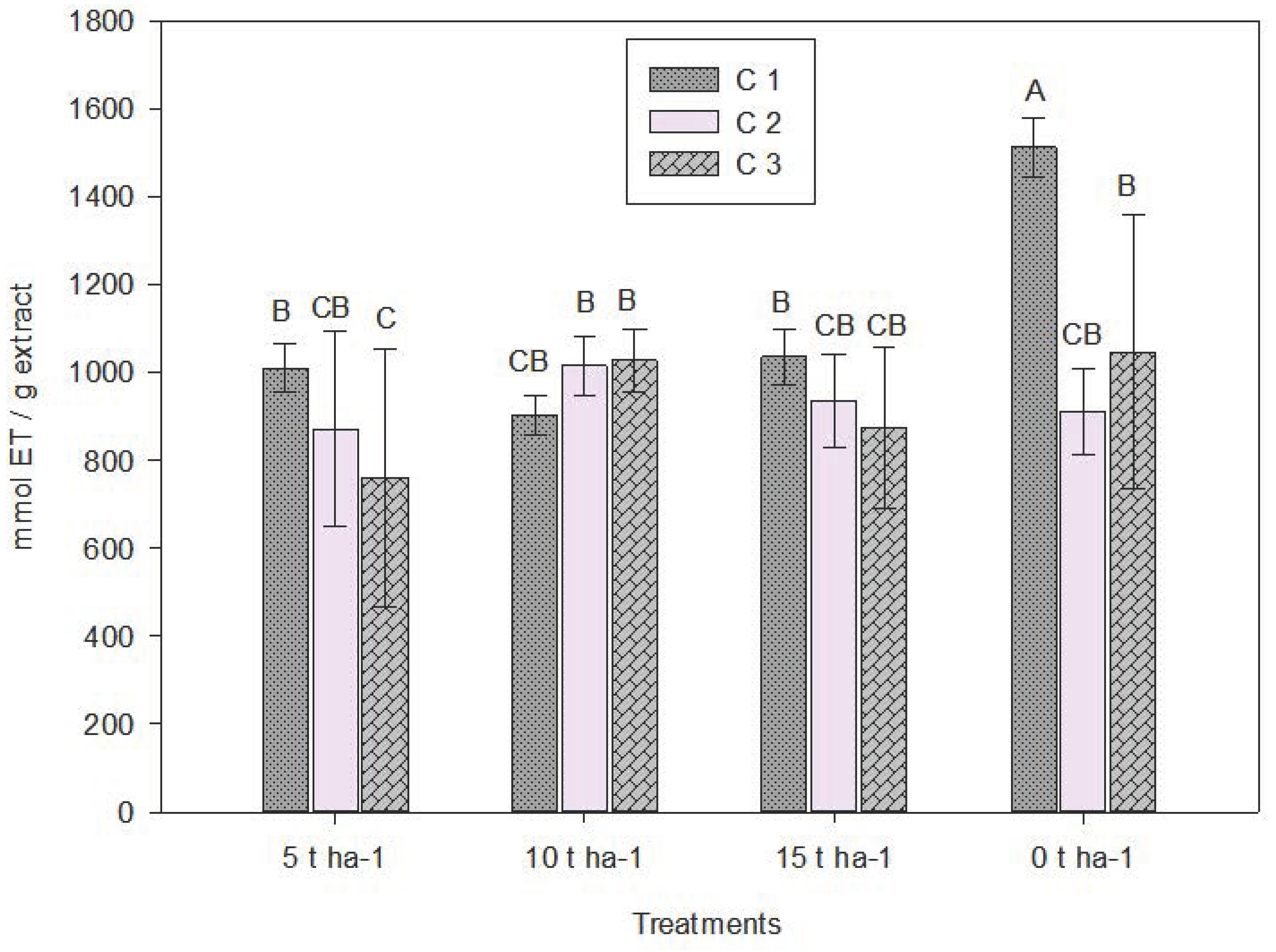
Figure 4: Average values for antioxidant capacity by the DPPH method. C1: Crop cutting one; C2: Crop cutting two; C3: Crop cutting three. Values presented are the means ± 1 S.D. Different letters above histograms indicate statistically significant differences using the least significant difference (LSD) test at p ≤ 0.05 among the mean values of the different treatments at the 3 sampling dates.
This aligns with the hypothesis that moderate stress (such as low nutrients) stimulates secondary metabolite production. Studies on oregano have also shown that high fertilization doses reduce antioxidant capacity [61]. Overall, C1 exhibited higher values than C2 and C3 in most treatments, especially in the control. These results may stem from fertilization reducing secondary metabolism activity, thereby diluting compound concentrations [58].
Findings suggest that moderate vermicompost doses may promote antioxidant synthesis, possibly by improving nutrient availability and stimulating soil microbial activity, as noted by Manzoor [62]. Furthermore, the type and dose of organic fertilization influence the enhancement or reduction of phenolic compounds depending on dose and soil matrix, leading to varied responses among treatments [63].
Conversely, higher doses were associated with decreased antioxidant values, likely due to nutritional imbalance or overfertilization stress, effects previously reported in aromatic species exposed to excessive organic fertilization. These results align with those of Novák et al. [64], who reported that phenological stage and environmental conditions significantly influence phenolic and volatile compound content in oregano.
The results of this study demonstrate that both vermicompost dosage and harvest timing are key factors for optimizing the functional quality of L. graveolens. Findings confirm that organic amendments not only improve vegetative growth and yield but also enhance the biosynthesis of nutraceutical secondary metabolites such as phenolics and antioxidants. Vermicompost may act as a modulator of plant metabolism by improving nutrient availability and stimulating physiological processes associated with stress adaptation.
Among the treatments evaluated, the application of 5 t ha−1 prior to the second harvest produced the highest concentrations of phenolic compounds and antioxidant activity. These results indicate that moderate organic fertilizer doses, paired with appropriate harvest scheduling, can promote both growth and functional quality in oregano. Similar patterns have been reported in other aromatic plants under organic fertilization, where intermediate nutrient availability enhances secondary metabolism without reducing biomass. Thus, combining organic management with optimal harvesting schedules represents a viable strategy to increase the added value of L. graveolens and strengthen its agro-industrial, medicinal, and culinary potential.
Furthermore, these results contribute valuable information for developing sustainable production systems aimed at balancing agricultural yield and phytochemical quality. Adopting organic fertilization practices such as vermicompost application may reduce reliance on synthetic inputs, promote soil health, and improve the nutraceutical profile of oregano biomass.
However, some limitations should be noted. This experiment was conducted in a single location and soil type, which may limit extrapolation to different edaphoclimatic conditions. Additionally, only a limited number of harvests and fertilizer doses were evaluated, and the study was carried out in a small experimental plot, which may not reflect the variability observed in commercial production systems. These factors should be considered when interpreting results and making recommendations.
Acknowledgement:
Funding Statement: The authors received no specific funding for this study.
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, José Luis García-Hernández and Mercedes Georgina Ramírez-Aragón; methodology, Priscilla Yamilhet Montes-Orona; software, Jared Ceniceros-García; validation, Urbano Nava-Camberos, Isaela Villalpando-De La Torre and José Luis García-Hernández; formal analysis, Mercedes Georgina Ramírez-Aragón. investigation, Priscilla Yamilhet Montes-Orona; resources, José Luis García-Hernández and Mercedes Georgina Ramírez-Aragón; writing—original draft preparation, Priscilla Yamilhet Montes-Orona; writing—review and editing, José Luis García-Hernández; visualization, UNC; supervision, Mercedes Georgina Ramírez-Aragón; project administration, José Luis García-Hernández; funding acquisition, José Luis García-Hernández and Mercedes Georgina Ramírez-Aragón. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: The data that support the findings of this study are available from the Corresponding Author, [José Luis García-Hernández], upon reasonable request.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
References
1. Soto-Armenta LC , Sacramento-Rivero JC , Acereto-Escoffié PO , Peraza-González EE , Reyes-Sosa CF , Rocha-Uribe JA . Extraction yield of essential oil from Lippia graveolens leaves by steam distillation at laboratory and pilot scales. J Essent Oil Bear Plants. 2017; 20( 3): 610– 21. doi:10.1080/0972060X.2017.1331756. [Google Scholar] [CrossRef]
2. Cid-Pérez TS , Torres-Muñoz JV , Nevárez-Moorillón GV , Palou E , López-Malo A . Chemical characterization and antifungal activity of Poliomintha longiflora Mexican oregano. J Essent Oil Res. 2016; 28( 2): 157– 65. doi:10.1080/10412905.2015.1083490. [Google Scholar] [CrossRef]
3. Orona-Castillo IAJ , Salvador-Almazán JJ , Espinoza-Arellano JJ , Vázquez C . Harvesting and marketing of oregano (Lippia spp.) in the Mexican semi-desert: Sierra Municipal Ecological Reserve. Rev Mex Agronegocios. 2017; 41( 1): 684– 95. (In Spanish). [Google Scholar]
4. Calvo-Irabién LM , Parra-Tabla V , Acosta-Arriola V , Escalante-Erosa F , Díaz-Vera L , Dzib GR , et al. Phytochemical diversity of the essential oils of Mexican Oregano (Lippia graveolens Kunth) populations along an Edapho-climatic gradient. Chem Biodivers. 2014; 11( 7): 1010– 21. doi:10.1002/cbdv.201300389. [Google Scholar] [CrossRef]
5. León DD , González-Álvarez M , Guzmán-Lucio MA , Núñez-Guzmán GR , Moreno-Limón S . The oregano of the genus Lippia (Verbenaceae) and Poliomintha (Lamiaceae) in the State of Nuevo León, Mexico. Polibotánica. 2020;( 50): 1– 18. doi:10.18387/polibotanica.50.1. [Google Scholar] [CrossRef]
6. Criollo-Mendoza MS , Heredia JB , Contreras-Angulo LA , García-Aguiar I , Gutiérrez-Grijalva EP . Apoptotic potential of polyphenol extract of Mexican oregano Lippia graveolens kunth on breast cancer cells MDA-MB-231. Appl Sci. 2025; 15( 12): 6393. doi:10.3390/app15126393. [Google Scholar] [CrossRef]
7. Galván Calamaco Z , Ramos Clamont Montfort G , Marszalek JE , Vargas González G . Revisión sobre el orégano mexicano Lippia graveolens HBK. (Sinonimia Lippia berlandieri Schauer) y su aceite esencial. Investig Y Desarrollo En Cienc Y Tecnología De Aliment. 2023; 8( 1): 861– 71. doi:10.29105/idcyta.v8i1.109. [Google Scholar] [CrossRef]
8. Reyes-Jurado F , Cervantes-Rincón T , Bach H , López-Malo A , Palou E . Antimicrobial activity of Mexican oregano (Lippia berlandieri), thyme (Thymus vulgaris), and mustard (Brassica nigra) essential oils in gaseous phase. Ind Crops Prod. 2019; 131: 90– 5. doi:10.1016/j.indcrop.2019.01.036. [Google Scholar] [CrossRef]
9. Vârban D , Zăhan M , Crișan I , Pop CR , Gál E , Ștefan R , et al. Phytochemistry and bioactivities of organic oregano (Origanum vulgare L.) essential oil: volatile profile, terpene composition and biological potentials. Plants. 2023; 12: 4017. doi:10.3390/plants12234017. [Google Scholar] [CrossRef]
10. Arias J , Mejía J , Córdoba Y , Martínez JR , Stashenko E , del Valle JM . Optimization of flavonoids extraction from Lippia graveolens and Lippia origanoides chemotypes with ethanol-modified supercritical CO2 after steam distillation. Ind Crops Prod. 2020; 146: 112170. doi:10.1016/j.indcrop.2020.112170. [Google Scholar] [CrossRef]
11. Leyva-López N , Gutiérrez-Grijalva EP , Vazquez-Olivo G , Heredia JB . Essential oils of oregano: biological activity beyond their antimicrobial properties. Molecules. 2017; 22( 6): 989. doi:10.3390/molecules22060989. [Google Scholar] [CrossRef]
12. Contini A , Di Bello D , Azzarà A , Giovanelli S , D’Urso G , Piaggi S , et al. Assessing the cytotoxic/genotoxic activity and estrogenic/antiestrogenic potential of essential oils from seven aromatic plants. Food Chem Toxicol. 2020; 138: 111205. doi:10.1016/j.fct.2020.111205. [Google Scholar] [CrossRef]
13. Calo JR , Crandall PG , O’Bryan CA , Ricke SC . Essential oils as antimicrobials in food systems—a review. Food Control. 2015; 54: 111– 9. doi:10.1016/j.foodcont.2014.12.040. [Google Scholar] [CrossRef]
14. Yan MR , Wang CH , Jose Cruz Flores N , Su YY . Targeting open market with strategic business innovations: a case study of growth dynamics in essential oil and aromatherapy industry. J Open Innov Technol Mark Complex. 2019; 5( 1): 7. doi:10.3390/joitmc5010007. [Google Scholar] [CrossRef]
15. Torrenegra Alarcon M , Hernández Santos R , León Ménde G , Granados Conde C , De La Espriella Angarita S . Especies vegetales con potencial antioxidante en la innovación de productos cárnicos procesados: una revisión. @limentech. 2024; 22( 1): 295– 311. doi:10.24054/limentech.v22i1.3185. [Google Scholar] [CrossRef]
16. Bautista-Hernández I , Aguilar CN , Martínez-Ávila GCG , Torres-León C , Ilina A , Flores-Gallegos AC , et al. Mexican oregano (Lippia graveolens Kunth) as source of bioactive compounds: a review. Molecules. 2021; 26( 17): 5156. doi:10.3390/molecules26175156. [Google Scholar] [CrossRef]
17. Ortega-Ramirez LA , Rodriguez-Garcia I , Silva-Espinoza BA , Ayala-Zavala JF . Oregano (Origanum spp.) oils. In: Essential oils in food preservation, flavor and safety. Amsterdam, The Netherlands: Elsevier; 2016. p. 625– 31. doi:10.1016/b978-0-12-416641-7.00071-7. [Google Scholar] [CrossRef]
18. Peschiutta ML , Arena JS , Ramirez Sanchez A , Gomez Torres E , Pizzolitto RP , Merlo C , et al. Effectiveness of Mexican oregano essential oil from the Dominican Republic (Lippia graveolens) against maize pests (Sitophilus zeamais and Fusarium verticillioides). AgriScientia. 2016; 33( 2): 89– 97. doi:10.31047/1668.298x.v33.n2.16576. [Google Scholar] [CrossRef]
19. Cid-Pérez TS , Ávila-Sosa R , Ochoa-Velasco CE , Rivera-Chavira BE , Nevárez-Moorillón GV . Antioxidant and antimicrobial activity of Mexican oregano (Poliomintha longiflora) essential oil, hydrosol and extracts from waste solid residues. Plants. 2019; 8( 1): 22. doi:10.3390/plants8010022. [Google Scholar] [CrossRef]
20. Gutiérrez-Grijalva EP , Antunes-Ricardo M , Acosta-Estrada BA , Gutiérrez-Uribe JA , Heredia JB . Cellular antioxidant activity and in vitro inhibition of α-glucosidase, α-amylase and pancreatic lipase of oregano polyphenols under simulated gastrointestinal digestion. Food Res Int. 2019; 116: 676– 86. doi:10.1016/j.foodres.2018.08.096. [Google Scholar] [CrossRef]
21. Pascual ME , Slowing K , Carretero E , Mata DS , Villar A . Lippia: traditional uses, chemistry and pharmacology: a review. J Ethnopharmacol. 2001; 76( 3): 201– 14. doi:10.1016/s0378-8741(01)00234-3. [Google Scholar] [CrossRef]
22. Irina LT , Javier BP , Teresa CM , Eurídice LA , Luz María del Carmen CI . Integrating ecological and socioeconomic criteria in a GIS-based multicriteria-multiobjective analysis to develop sustainable harvesting strategies for Mexican oregano Lippia graveolens Kunth, a non-timber forest product. Land Use Policy. 2019; 81: 668– 79. doi:10.1016/j.landusepol.2018.11.038. [Google Scholar] [CrossRef]
23. Martínez-Natarén DA , Parra-Tabla V , Ferrer-Ortega MM , Calvo-Irabién LM . Genetic diversity and genetic structure in wild populations of Mexican oregano (Lippia graveolens H.B.K.) and its relationship with the chemical composition of the essential oil. Plant Syst Evol. 2014; 300( 3): 535– 47. doi:10.1007/s00606-013-0902-y. [Google Scholar] [CrossRef]
24. Torroba MUL , Bravo SL , Aguilera LG . Technical assistance and transfer of Origanum × applii plant material obtained through biotechnological methods to microentrepreneurs. Fytotron (fytotronica). 2004; 81: 23– 34. (In Spanish). [Google Scholar]
25. Reyes CJ , Ortega RS . Use, management, and cultivation of oregano in the Lagunera Region (Bro-chure for Producers No. 6). Torreón, Coahuila, México: SAGARPA-INIFAP-CIRNOC-CELALA; 2002. 21 p. (In Spanish). [Google Scholar]
26. Ilić Z , Stanojević L , Milenković L , Šunić L , Milenković A , Stanojević J , et al. The yield, chemical composition, and antioxidant activities of essential oils from different plant parts of the wild and cultivated oregano (Origanum vulgare L.). Horticulturae. 2022; 8( 11): 1042. doi:10.3390/horticulturae8111042. [Google Scholar] [CrossRef]
27. Mora-Zúñiga AE , Treviño-Garza MZ , Amaya Guerra CA , Galindo Rodríguez SA , Castillo S , Martínez-Rojas E , et al. Comparison of chemical composition, physicochemical parameters, and antioxidant and antibacterial activity of the essential oil of cultivated and wild Mexican oregano Poliomintha longiflora gray. Plants. 2022; 11( 14): 1785. doi:10.3390/plants11141785. [Google Scholar] [CrossRef]
28. Farruggia D , Di Miceli G , Licata M , Urso G , Leto C , Novak J . Seaweed extract and fulvic acid application affect the biomass performance, the essential oil yield and composition of Sicilian oregano grown in an organic agricultural system. Ind Crops Prod. 2024; 222: 119790. doi:10.1016/j.indcrop.2024.119790. [Google Scholar] [CrossRef]
29. Asri FÖ . The effects of organic and conventional fertilization on oregano (Origanum onites L.) yield and quality factors. Folia Hortic. 2023; 35( 1): 209– 19. doi:10.2478/fhort-2023-0016. [Google Scholar] [CrossRef]
30. Huerta C . Mexican oregano: vegetable gold. CONABIO. 1997; 15: 8– 13. (In Spanish). [Google Scholar]
31. Organic Materials Review Institute (OMRI). WORM HUMUS Organic Soil Conditioner Concentrated Solution [Internet]. Eugene, OR, USA: OMRI; 2024 [cited 2025 Nov 14]. Available from: https://www.omri.org/omri-search?page=1&query=aeh-19640&exactMatch=false. (In Spanish). [Google Scholar]
32. Zhang M , Liu Y , Wei Q , Liu L , Gu X , Gou J , et al. Ameliorative effects of vermicompost application on yield, fertilizer utilization, and economic benefits of continuous cropping pepper in karst areas of southwest China. Agronomy. 2023; 13( 6): 1591. doi:10.3390/agronomy13061591. [Google Scholar] [CrossRef]
33. El-Haddad ME , Zayed MS , El-Sayed GAM , Abd EL-Safer AM . Efficiency of compost and vermicompost in supporting the growth and chemical constituents of Salvia officinalis L. cultivated in sand soil. Int J Recycl Org Waste Agric. 2020; 9( 1): 49– 59. doi:10.30486/IJROWA.2020.671209. [Google Scholar] [CrossRef]
34. Salas-Pérez L , Moncayo-Luján MR , Borroel-García VJ , Guzmán-Silos TL , Ramírez-Aragón MG . Phytochemical composition and antioxidant activity in three varieties of basil under the effect of different solvents. Rev Mex Cienc Agric. 2022; 13( spe28): 113– 23. (In Spanish). doi:10.29312/remexca.v13i28.3267. [Google Scholar] [CrossRef]
35. Moghrovyan A , Sahakyan N , Babayan A , Chichoyan N , Petrosyan M , Trchounian A . Essential oil and ethanol extract of oregano (Origanum vulgare L.) from Armenian flora as a natural source of terpenes, flavonoids and other phytochemicals with antiradical, antioxidant, metal chelating, tyrosinase inhibitory and antibacterial activity. Curr Pharm Des. 2019; 25( 16): 1809– 16. doi:10.2174/1381612825666190702095612. [Google Scholar] [CrossRef]
36. Singleton VL , Rossi JA Jr . Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic. 1965; 16( 3): 144– 58. doi:10.5344/ajev.1965.16.3.144. [Google Scholar] [CrossRef]
37. Jia Z , Tang M , Wu J . The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999; 64( 4): 555– 9. doi:10.1016/s0308-8146(98)00102-2. [Google Scholar] [CrossRef]
38. Re R , Pellegrini N , Proteggente A , Pannala A , Yang M , Rice-Evans C . Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med. 1999; 26( 9–10): 1231– 7. doi:10.1016/s0891-5849(98)00315-3. [Google Scholar] [CrossRef]
39. Palafox-Carlos H , Yahia EM , González-Aguilar GA . Identification and quantification of major phenolic compounds from mango (Mangifera indica, cv. Ataulfo) fruit by HPLC–DAD–MS/MS-ESI and their individual contribution to the antioxidant activity during ripening. Food Chem. 2012; 135( 1): 105– 11. doi:10.1016/j.foodchem.2012.04.103. [Google Scholar] [CrossRef]
40. SAS Statistical Analysis System. SAS software versión 9.1. Cary, NC, USA: SAS Institute Inc.; 2009. [Google Scholar]
41. Systat Software Inc. SigmaPlot for Windows, version 12.0. San Jose, CA, USA: Systat Software Inc.; 2011. [Google Scholar]
42. Ríos-Saucedo JC , Rosales-Serna R , Velazquez-Quiñones SE , López JA , Herrera MD . Phenolic compounds and antioxidant capacity of essential oregano oil from two federal entities in Mexico. Biotecnol Sustentabilidad. 2024; 9( 1): 12– 20. (In Spanish). doi:10.57737/469gwb37. [Google Scholar] [CrossRef]
43. Fukalova Fukalova T , Brito GB , Novoa CT , Sagñay TE , Samaniego MI , Tacan PM , et al. Análisis comparativo de Las propiedades fisicoquímicas y capacidad antioxidante de un morfotipo de orégano (Origanum vulgare L.) cultivado en dos localidades de la sierra ecuatoriana. Siembra. 2021; 8( 1): e2289. doi:10.29166/siembra.v8i1.2289. [Google Scholar] [CrossRef]
44. Aarland R , Castellanos-Hernandez O , Rodriguez-Sahagun A , Acevedo-Hernandez G . Efecto del estrés salino sobre la morfología y fitoquímica de orégano mexicano (Lippia graveolens Kunth) cultivado in vitro. Biotecnia. 2020; 22( 3): 131– 7. doi:10.18633/biotecnia.v22i3.1223. [Google Scholar] [CrossRef]
45. Matłok N , Stępień AE , Gorzelany J , Wojnarowska-Nowak R , Balawejder M . Effects of organic and mineral fertilization on yield and selected quality parameters for dried herbs of two varieties of oregano (Origanum vulgare L.). Appl Sci. 2020; 10( 16): 5503. doi:10.3390/app10165503. [Google Scholar] [CrossRef]
46. Verma N , Shukla S . Impact of various factors responsible for fluctuation in plant secondary metabolites. J Appl Res Med Aromat Plants. 2015; 2( 4): 105– 13. doi:10.1016/j.jarmap.2015.09.002. [Google Scholar] [CrossRef]
47. Frías-Zepeda ME , Rosales-Castro M . Efecto de Las condiciones de extracción sobre la concentración de compuestos fenólicos en residuos de orégano mexicano (Lippia graveolens Kunth). Rev Chapingo Ser Cienc Y Del Ambiente. 2021; 27( 3): 367– 81. doi:10.5154/r.rchscfa.2020.10.066. [Google Scholar] [CrossRef]
48. Ghassemi-Golezani K , Javadzadeh A , Zehtab-Salmasi S , Moghaddam M . Changes in essential oil content of peppermint under water stress. Am Eurasian J Agric Environ Sci. 2011; 10( 1): 70– 3. doi:10.1016/j.scienta.2017.12.011. [Google Scholar] [CrossRef]
49. Patil JR , Mhatre KJ , Yadav K , Yadav LS , Srivastava S , Nikalje GC . Flavonoids in plant-environment interactions and stress responses. Discov Plants. 2024; 1( 1): 68. doi:10.1007/s44372-024-00063-6. [Google Scholar] [CrossRef]
50. Selmar D , Kleinwächter M . Influencing the product quality by deliberately applying drought stress during the cultivation of medicinal plants. Ind Crops Prod. 2013; 42: 558– 66. doi:10.1016/j.indcrop.2012.06.020. [Google Scholar] [CrossRef]
51. Agathokleous E , Kitao M , Calabrese EJ . Hormesis: a compelling platform for sophisticated plant science. Trends Plant Sci. 2019; 24( 4): 318– 27. doi:10.1016/j.tplants.2019.01.004. [Google Scholar] [CrossRef]
52. Erofeeva EA . Plant hormesis: the energy aspect of low and high-dose stresses. Plant Stress. 2024; 14: 100628. doi:10.1016/j.stress.2024.100628. [Google Scholar] [CrossRef]
53. Isah T . Stress and defense responses in plant secondary metabolites production. Biol Res. 2019; 52( 1): 39. doi:10.1186/s40659-019-0246-3. [Google Scholar] [CrossRef]
54. Laftouhi A , Eloutassi N , Ech-Chihbi E , Rais Z , Abdellaoui A , Taleb A , et al. The impact of environmental stress on the secondary metabolites of medicinal and aromatic plants. Plants. 2023; 12( 2): 350. doi:10.3390/su15107842. [Google Scholar] [CrossRef]
55. Bettaieb I , Zakhama N , Wannes WA , Kchouk ME , Marzouk B . Water deficit effects on Salvia officinalis fatty acids and essential oils composition. Sci Hortic. 2009; 120( 2): 271– 5. doi:10.1016/j.scienta.2008.10.016. [Google Scholar] [CrossRef]
56. Uskutoğlu T . Effect of harvest on the agronomic, mineral and antioxidant profile of three oregano species (Origanum onites L., Origanum vulgare L. ssp. hirtum, and Origanum acutidens (Hand.-Mazz.) Ietswaart). PeerJ. 2025; 13: e20223. doi:10.7717/peerj.20223. [Google Scholar] [CrossRef]
57. Hazrati S , Mousavi Z , Nicola S . Harvest time optimization for medicinal and aromatic plant secondary metabolites. Plant Physiol Biochem. 2024; 212: 108735. doi:10.1016/j.plaphy.2024.108735. [Google Scholar] [CrossRef]
58. Lattanzio V , Cardinali A , Ruta C , Fortunato I , Lattanzio V , Linsalata V , et al. Relationship of secondary metabolism to growth in oregano (Origanum vulgare L.) shoot cultures under nutritional stress. Environ Exp Bot. 2009; 65( 1): 54– 62. doi:10.1016/j.envexpbot.2008.09.002. [Google Scholar] [CrossRef]
59. Król B , Sęczyk Ł , Kołodziej B , Paszko T . Biomass production, active substance content, and bioaccessibility of Greek oregano (Origanum vulgare ssp. hirtum (Link) Ietswaart) following the application of nitrogen. Ind Crops Prod. 2020; 148: 112271. doi:10.1016/j.indcrop.2020.112271. [Google Scholar] [CrossRef]
60. Floegel A , Kim DO , Chung SJ , Koo SI , Chun OK . Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods. J Food Compos Anal. 2011; 24( 7): 1043– 8. doi:10.1016/j.jfca.2011.01.008. [Google Scholar] [CrossRef]
61. Sęczyk Ł , Król B , Kołodziej B . In vitro bioaccessibility and activity of Greek oregano (Origanum vulgare L. ssp. hirtum (link) Ietswaart) compounds as affected by nitrogen fertilization. J Sci Food Agric. 2020; 100( 6): 2410– 7. doi:10.1002/jsfa.10252. [Google Scholar] [CrossRef]
62. Manzoor A , Naveed MS , Ali RMA , Naseer MA , UL-Hussan M , Saqib M , et al. Vermicompost: a potential organic fertilizer for sustainable vegetable cultivation. Sci Hortic. 2024; 336: 113443. doi:10.1016/j.scienta.2024.113443. [Google Scholar] [CrossRef]
63. Yusof Z , Ramasamy S , Mahmood NZ , Yaacob JS . Vermicompost supplementation improves the stability of bioactive anthocyanin and phenolic compounds in Clinacanthus nutans lindau. Molecules. 2018; 23( 6): 1345. doi:10.3390/molecules23061345. [Google Scholar] [CrossRef]
64. Novák I , Sipos L , Kókai Z , Szabó K , Pluhár Z , Sárosi S . Effect of the drying method on the composition of Origanum vulgare L. subsp. hirtum essential oil analysed by GC-MS and sensory profile method. Acta Aliment. 2011; 40( Supplement 1): 130– 8. doi:10.1556/aalim.40.2011.suppl.13. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools