 Open Access
Open Access
ARTICLE
Study on the Relationship between Flowering Characteristics and Fruit Setting of Oil Tree Peony
1 College of Agriculture/Tree Peony, Henan University of Science & Technology, Luoyang, China
2 College of Horticulture and Plant Protection, Henan University of Science & Technology, Luoyang, China
3 College of Urban Construction, Luoyang Vocational & Technical College, Luoyang, China
* Corresponding Author: Xiaogai Hou. Email:
(This article belongs to the Special Issue: Integrated Approaches to Crop Yield Enhancement)
Phyton-International Journal of Experimental Botany 2026, 95(3), 11 https://doi.org/10.32604/phyton.2026.078506
Received 01 January 2026; Accepted 02 March 2026; Issue published 31 March 2026
Abstract
This study investigated the association between floral and fruiting traits in oil tree peony to inform elite breeding and hybrid programmes. Floral and fruiting metrics of Paeonia ostii ‘Fengdanbai’ and Paeonia rockii ‘Zibanbai’ were recorded under natural conditions; data were analysed using independent-samples t-tests, Pearson correlations, stepwise multiple linear regression and membership functions. Paeonia ostii ‘Fengdanbai’ produced more pollen number per flower, but fewer ovules and seeds per fruit than Paeonia rockii ‘Zibanbai’ (p < 0.05). Pollen viability and stigma receptivity followed similar unimodal temporal curves in both taxa. Peak pollen viability occurred on day 3 after anthesis in P. ostii ‘Fengdanbai’ (86.95 ± 1.41%) and on day 2 in P. rockii ‘Zibanbai’ (85.91 ± 2.99%). Optimal stigma receptivity spanned days 3–5 and 2–5 post-anthesis, respectively, yielding a 3-day overlap favourable for synchronous pollination. Seed number per fruit correlated positively with petal number per flower (r = 0.49), ovule number per fruit (r = 0.68) and seed setting rate (r = 0.70, p < 0.01); carpel number also contributed (r = 0.31, p < 0.05). Stepwise multiple regression identified ovule number per fruit and seed setting rate as the most significant predictors of seed number per fruit (p < 0.001), together explaining over 99% of the variation in the model (adjusted R2 = 0.992). Membership function analysis, based on pollen viability, ovule number per fruit, and seed setting rate, suggested that ‘Fengdanbai’ exhibits superior comprehensive performance, though selection indices should be validated across broader germplasm. We propose ovule number per fruit and seed setting rate as primary selection indices, with pollen viability as a supplementary indicator, for consideration in breeding high-yield cultivars.Keywords
Oil tree peony has recently been domesticated as a novel woody oilseed crop in China for the commercial production of edible oil. In 2011, the former Ministry of Health of the People’s Republic of China formally approved peony seed oil as a novel food resource [1]. This oil is recognised as safe, non-toxic and highly nutritious [1,2,3], containing more than 90% unsaturated fatty acids, with α-linolenic acid-an essential human nutrient-contributing up to 40% of total fatty acids [4,5]. To satisfy increasing consumer demand for safe and nutritious edible oils, large-scale cultivation of tree peony has been actively promoted across China. The crop is currently grown in over 20 provincial-level administrative regions, including Shandong, Henan, Gansu, Anhui, Hubei, Qinghai, and Tibet [6,7]. Among these, Paeonia ostii ‘Fengdan’ and P. rockii are the principal cultivars under commercial cultivation [8]. Flowers are large, white, fragrant, and pollen-abundant, with bright yellow, cockscomb-type stigmas lacking nectaries; although self-compatible, seed set is extremely low [9,10,11]. Recent expansion into heterogeneous environments has intensified intraspecific variation and resulted in chronically low and unstable seed yield, thereby limiting oil output and constraining industrial development [12,13,14].
Flowering marks the initiation of sexual reproduction, whereas fruiting constitutes a critical phase for plant propagation and regeneration. A comprehensive understanding of floral and fruiting traits is therefore essential to stabilize both yield quantity and quality [15]. Pollen viability and stigma receptivity directly govern the efficacy of double fertilization and, consequently, fruit set [16,17]. Cai et al. demonstrated that both pollen viability and stigma receptivity in passion fruit were significantly correlated with fruit set [18]. In orchids, Yang et al. revealed that stronger pollen viability and stigma receptivity increased pod set, whereas weaker activity reduced it [19]. Germplasm collections of oil tree peony provide the foundational resources for genetic improvement and fundamental research. Previous studies have predominantly examined pairwise correlations among vegetative, fruit, and yield traits, whereas comprehensive multivariate analyses remain scarce [20,21,22,23]. The primary yield components in oil tree peony are fruits per plant, seeds per fruit, and individual seed weight [24,25]. Nevertheless, the relationships among floral phenology, pollen viability, stigma receptivity, and seed set remain uncharacterised in oil tree peony.
In this study, P. ostii ‘Fengdan’ and P. rockii served as the experimental materials. Floral morphology, pollen viability, stigma receptivity, and fruiting parameters of both taxa were systematically assessed using in field tagging, macro-photography, and light microscopy. Multivariate statistical analyses were subsequently employed to elucidate the relationships among floral traits, pollen viability, stigma receptivity, and seed set. These findings are expected to provide a theoretical basis for elite cultivar development and accelerated breeding of oil tree peony.
Field experiments were conducted at the experimental farm at the Kaiyuan campus of Henan University of Science and Technology, Luoyang, China (112°28′36.34″ E, 34°39′30.34″ N, altitude: 120 m). The site is situated in western Henan Province, south of the lower Yellow River. The climate is classified as warm-temperate, semi-humid, continental monsoon, with hot, humid summers and cold, dry winters. Mean annual precipitation is 580 mm, and mean annual temperature is 13.5°C. Uniform agronomic practices were applied throughout the experimental plots. Weeds were manually removed as required during the growing season. Fertiliser was top-dressed two to three times annually; irrigation was synchronised with fertilisation to maintain optimal soil moisture and nutrient status. The plant materials used in this study were all cultivated varieties grown at the farm of the College of Agriculture, Henan University of Science and Technology. The specific cultivated varieties are P. ostii ‘Fengdanbai’ and the P. rockii ‘Zibanbai’, the varieties picture is shown in Fig. 1.

Figure 1: Oil tree peony plant strains. (a) P. ostii ‘Fengdanbai’, (b) P. rockii ‘Zibanbai’.
For each cultivar (P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’), a total of 20 distinct, randomly selected plants served as biological replicates. From each of these plants, three flower (or three fruit at maturity) was sampled for trait measurement. This design guaranteed that each data point originated from a unique genetic individual, satisfying the assumption of independence for subsequent statistical analyses. Specific sample sizes for time-series measurements (e.g., pollen viability) are detailed in the respective subsections below.
2.2.1 Observation on Single Flower Longevity and Flowering Dynamics of P. ostii ‘Fengdan’ and P. rockii
The experiment commenced at 08:00 on 6 April 2023. Ten newly opened flowers of P. ostii ‘Fengdanbai’ were randomly selected from the experimental farm of Henan University of Science and Technology for dynamic observation of the individual floral anthesis period. Each flower was photographed and documented. The initial opening was defined as follows: petals slightly parted, maintaining a bud-like shape, with visible but unsplit stigma and neatly arranged, glossy stamens.
On sunny days with high temperature and low humidity, the rate of floral opening was observed to accelerate. Ten newly opened flowers were randomly selected from 10 individual plants (three flower per plant to ensure independence) were labeled as FD-1, FD-2, …, FD-i, where FD-i denotes the i-th flower of P. ostii ‘Fengdanbai’. To accurately monitor the flowering dynamics, observations commenced on 6 April 2023. Observations were conducted hourly from 08:00 to 18:00 on the first day, with each flower photographed for subsequent comparative analysis. Subsequently, daily observations and photographic documentation were performed at 08:00 from 7 to 15 April 2023. The same observation protocol was applied to P. rockii ‘Zibanbai’, with recordings initiated on 10 April 2023.
2.2.2 Determination of Floral Traits
To ensure independence of observations, all measurements were conducted on distinct individual plants with one flower per plant (n = 20 plants per cultivar for floral traits; n = 20 plants per cultivar for fruiting traits), avoiding pseudoreplication from multiple samples per plant.
Twenty individual plants per cultivar were randomly selected, with one flower per plant (n = 20 flowers from 20 plants) used for measurements. To measure the corolla diameter, the number of petals per flower and the number of anthers per flower, and the amount of pollen per flower was measured.
Completely blooming peony flowers were randomly selected in the experimental site, The diameter of corolla was measured by cross method (steel ruler: 1 mm). The determination of single flower pollen amount was referred to the method of Shen et al. [26], with modifications following recent protocols for oil crops [27]. Six anthers of the inner, middle and outer wheels of a peony flower were randomly selected in a centrifuge tube, 1000 μL 20% sodium hexametaphosphate solution was added, and the pollen solution was prepared by mixing and centrifugation. After the completion, 800 μL of the supernatant was sucked away, and 300 μL of distilled water was added to the remaining solution to mix evenly. Take 1 μL of the above solution on the slide, observe and count under the microscope, repeat 5 times, and calculate the number of single flower pollen:
2.2.3 Determination of Pollen Viability
Pollen viability was measured on the 1st day (8:00 a.m., 9:00 a.m., 10:00 a.m., 12:00 a.m., 14:00 p.m., 18:00 p.m.), 2nd days (8:00 a.m.), 3rd days (8:00 a.m.), 4th days (8:00 a.m.), 5th days (8:00 a.m.), 6th days (8:00 a.m.), 7th days (8:00 a.m.) after anthesis. The pollen viability was determined by the TTC method of Bao et al. [28]. One to two drops of TTC solution containing pollen were placed on a glass slide. After the reaction of pollen and TTC solution for 10–20 min, the staining of pollen was observed under a microscope. The staining effect of TTC staining on the determination of pollen viability is shown in Fig. 2. Measurements were taken on flowers from distinct plants (one flower per plant, n = 10 plants per time point) to avoid pseudoreplication.
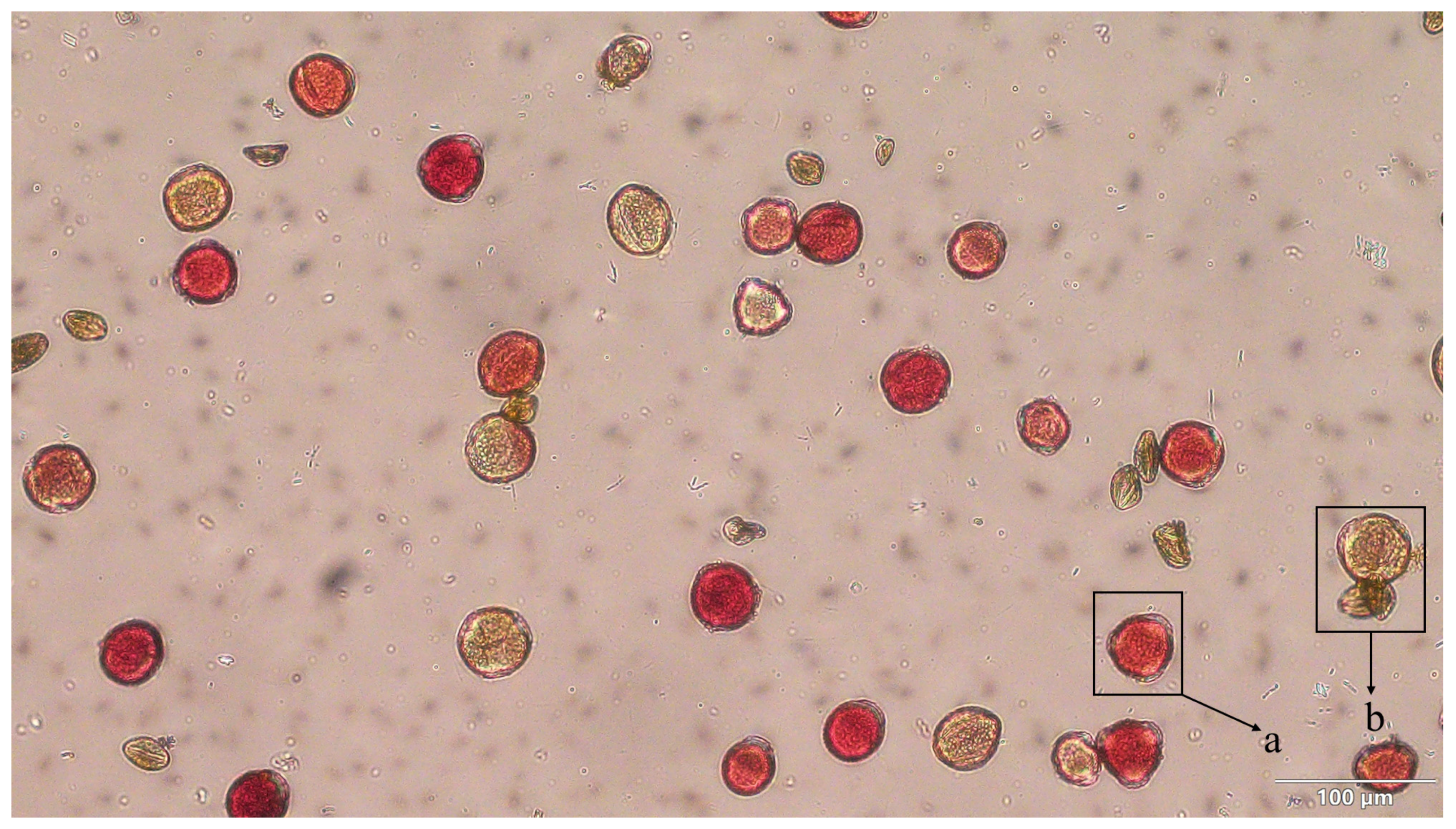
Figure 2: Staining effect of TTC staining for determination of pollen viability. Note: a: denotes viable pollen, and the darker the color, the stronger the pollen viability; b: represents not inactive pollen. Bar = 100 μm.
2.2.4 Determination of Stigma Receptivity
The stigmas of the 1st day (8:00 a.m., 9:00 a.m., 10:00 a.m., 12:00 a.m., 14:00 p.m., 18:00 p.m.), 2nd d (8:00 a.m.), 3rd d (8:00 a.m.), 4th d (8:00 a.m.), 5th d (8:00 a.m.), 6th d (8:00 a.m.), 7th d (8:00 a.m.), 8th d (8:00 a.m.), 9th d (8:00 a.m.), 10th d (8:00 a.m.) after anthesis were measured for receptivity. Stigma receptivity measurement and results grading standards refer to Luo et al. [29] benzidine-hydrogen peroxide method).
Stigma receptivity was scored based on morphological criteria and reaction criteria observred within 30 s of reagent application: ‘+’ (stigma pale yellow, slightly swollen, minimal bubble production); ‘++’ (stigma bright yellow, fully expanded, moderate bubbles); ‘+++’ (stigma deep yellow, glossy surface with sticky exudate, abundant bubbles); ‘++++’ (stigma orange-yellow, papillae fully turgid, profuse bubbles). Fig. 3 provides definitive reference images corresponding to each receptivity score (‘+’ to ‘++++’), which were used as the visual standard for all assessments. All scoring was conducted by the same trained researcher to ensure consistency.
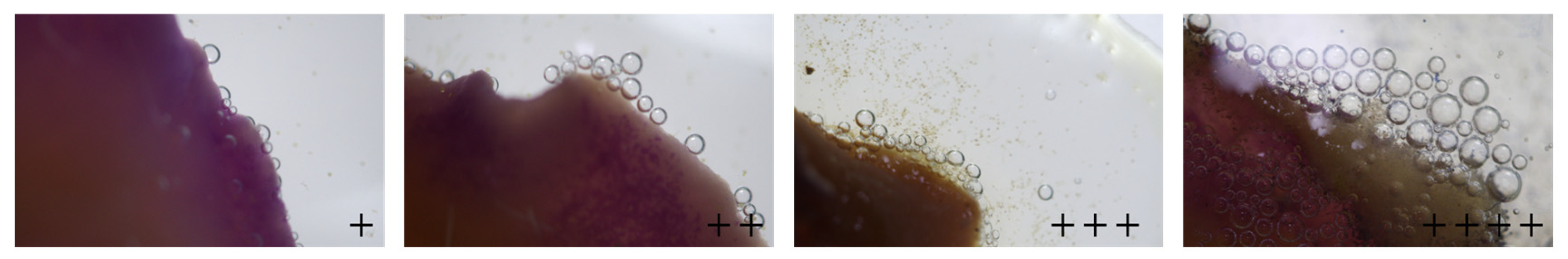
Figure 3: Visual reference standards for scoring stigma receptivity in oil tree peony. ‘+’: Stigmas with receptivity, the more ‘+’, the higher the receptivity.
2.2.5 Determination of Fruiting Traits
After 15 days of the end of the peony flowering period, 20 peony plants with all the petals falling off were randomly selected in the experimental site. Carpel number per fruit, ovule number per fruit and seed number per fruit were counted, and the seed setting rate was calculated. The calculation formula was:
Twenty individual plants per cultivar were tagged; one fruit per plant was harvested at maturity (n = 20 fruits). Ovule number per fruit refers to the total number of ovules in the aggregate fruit, while Ovule number per fruit was counted from representative individual fruits.
Excel software was used to analyze the data of each index. SPSS 20.0 software was used for independent sample t test, correlation analysis and regression analysis. Regression assumptions were checked through residual diagnostics. Origin 2022 software was used for mapping. The indicators that significantly affect the number of seeds per fruit, as determined by regression analysis and path analysis, were selected to conduct a comprehensive evaluation of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ cultivars using the membership function method [20]. The calculation formula for the membership function value is:
(U) is the membership function value; (X) is the measured value of a certain indicator for all tested varieties; (Xmax) is the maximum measured value of a certain indicator among all tested varieties; (Xmin) is the minimum measured value of a certain indicator among all tested varieties. The cumulative membership function value (Utotal) is calculated as (Utotal = U1 + U2 + … + Un). This standardization allows comparison of traits with different units, though it assumes equal importance of all traits.
3.1 Observation of Single Flower Duration and Flowering Dynamics in P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’
Through field observation in the field, it was found that the first flowering time of P. ostii ‘Fengdanbai’ was April 6, 2023, and the first flowering time of P. rockii ‘Zibanbai’ was April 10, 2023; the wilting time of P. ostii ‘Fengdanbai’ is 14 April 2023, and the decline time of P. rockii ‘Zibanbai’ is 16 April 2023. The flowering time of the two is quite different.
Through the t-test of the single flower flowering period of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’, the results showed that the average single flower flowering period of P. ostii ‘Fengdanbai’ was (7.0 ± 0.21) d, and the average single flower flowering period of P. rockii ‘Zibanbai’ was (4.9 ± 0.18) d, the difference between the two was significant (t = 7.58, F = 0.036, p < 0.05).
Through the observation of the flowering dynamics of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’, it was found that the whole flowering dynamics can be divided into three processes: initial opening period, full opening period and decay period. The initial opening period of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ was 1 d. The full-bloom period of P. ostii ‘Fengdanbai’ was about 1 d longer than that of P. rockii ‘Zibanbai’. When the peony of P. ostii ‘Fengdanbai’ decayed, the petals fell slowly and experienced 3–4 days; however, it only takes 1–2 days for all petals of P. rockii ‘Zibanbai’ to fall off. The morphological characteristics of tree peony flowers changed as follows: petals changed from flat to wrinkled, and finally fell; the anther changed from orderly and glossy to disordered and dull, and finally fell down. The stigma changed from strong and full to dark and loose (Fig. 4).

Figure 4: Observation of Single Flower Duration and Flowering Dynamics in P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’. (a–c) in the picture is the initial opening period of P. ostii ‘Fengdanbai’, the first day after anthesis; (d–h) is the full-bloom stage, 2nd–4th day after anthesis; (i–p) is the decay period, 5th–10th day after anthesis. (A–D) is the initial opening period of P. rockii ‘Zibanbai’, and the first day after anthesis; (E–H) is the full-bloom stage, 2nd–3rd day after anthesis; (I–P) is the decay period, 4th–10th day after anthesis.
3.2 Floral Traits and Fruiting Traits of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’
The flower traits and fruiting traits of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ were tested by independent sample t-test. The results showed that there were significant differences between P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ in the number of pollen grains per flower, the number of ovules per fruit and the number of seeds per fruit (p < 0.05) (Table 1).
Table 1: Descriptive statistics of floral traits and fruit traits in P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’.
| Trait | Mean | SE | Maximum | Minimum | t-Test | F-Value | p-Value |
|---|---|---|---|---|---|---|---|
| F-X1 | 15.44 | 0.31 | 17.85 | 12.35 | 0.915 | 4.365 | 0.37 |
| Z-X1 | 15.11 | 0.19 | 16.70 | 13.55 | |||
| F-X2 | 12.90 | 0.40 | 16.00 | 10.00 | 0.983 | 2.068 | 0.33 |
| Z-X2 | 12.40 | 0.31 | 15.00 | 10.00 | |||
| F-X3 | 257.95 | 11.25 | 328.00 | 176.00 | 1.473 | 5.331 | 0.15 |
| Z-X3 | 228.55 | 16.49 | 353.00 | 124.00 | |||
| F-X4 | 781.30 | 13.05 | 1065.90 | 597.60 | −12.501 | 8.408 | 0.00 |
| Z-X4 | 1160.77 | 77.47 | 1277.83 | 1056.47 | |||
| F-X5 | 82.43 | 1.64 | 90.70 | 59.38 | 1.062 | 0.310 | 0.30 |
| Z-X5 | 79.87 | 1.76 | 90.32 | 58.06 | |||
| F-X6 | 2.53 | 1.20 | 4.00 | 1.33 | −0.870 | 0.331 | 0.39 |
| Z-X6 | 2.85 | 1.13 | 4.00 | 1.50 | |||
| F-X7 | 5.20 | 0.14 | 7.00 | 4.00 | 0.237 | 0.289 | 0.81 |
| Z-X7 | 5.25 | 0.16 | 7.00 | 4.00 | |||
| F-X8 | 92.95 | 4.05 | 136.00 | 49.00 | 2.253 | 1.405 | 0.03 |
| Z-X8 | 82.15 | 2.56 | 100.00 | 64.00 | |||
| F-X9 | 54.52 | 2.35 | 75.93 | 30.00 | 1.115 | 0.085 | 0.27 |
| Z-X9 | 50.94 | 2.18 | 71.76 | 36.25 | |||
| F-X10 | 50.80 | 3.39 | 83.00 | 27.00 | 2.454 | 6.009 | 0.02 |
| Z-X10 | 41.40 | 1.77 | 61.00 | 29.00 |
3.3 Comparison of Pollen Viability and Stigma Receptivity between P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’
The pollen viability of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ was determined. The results showed in Fig. 5 that: During the whole flowering period, the pollen viability of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ increased first and then decreased. On the first day of flowering, the pollen viability of P. ostii ‘Fengdanbai’ (81.39%3 ± 2.40%) and P. rockii ‘Zibanbai’ (81.65% ± 4.77%) was the strongest at 4th h after anthesis, and there was no significant difference between them (t = −0.33, F = 2.28, p > 0.05). The pollen viability of P. ostii ‘Fengdanbai’ reached the strongest during anthesis stage on the 3rd day, with an average of 86.95% ± 1.41%. The pollen viability of P. rockii ‘Zibanbai’ reached the strongest during anthesis stage on the 2nd day, with an average of 85.91% ± 2.99%. The difference between the two is not significant (t = 0.91, F = 2.42, p > 0.05).
The stigma receptivity of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ was determined. The results showed in Fig. 6 that: During the whole flowering period, the change trend of stigma receptivity was first enhanced and then weakened; On the first day of flowering, the stigmas of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ began to have weak receptivity. The stigma receptivity of P. ostii ‘Fengdanbai’ was the strongest at 3rd–5th day after anthesis, and the stigma receptivity of P. rockii ‘Zibanbai’ was the strongest at 2nd–5th day after anthesis. The total overlap period between pollen viability and stigma receptivity was 7 days, whereas the overlap period during peak pollen viability (>65%) and highest stigma receptivity (++++) was 3 days.
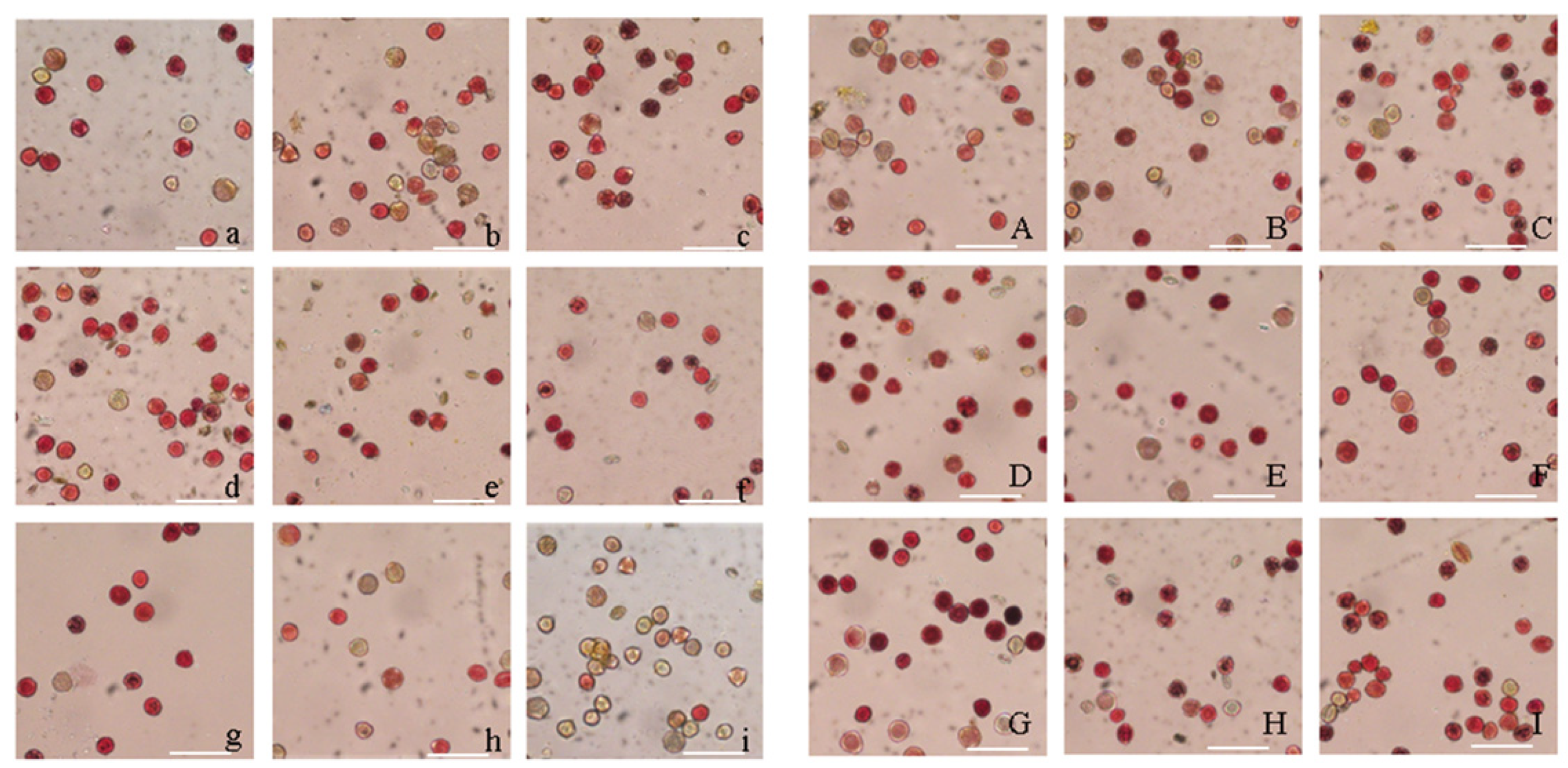
Figure 5: Results of the determination of pollen viability of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ (scale bars: 100 μm.) In the figure, (a–c) represents the activity determination results of P. ostii ‘Fengdanbai’ pollen at 0 h, 4th h and 8th h after anthesis, and (d–i) represents the activity determination results at 2nd–7th d after anthesis. (A–C) represents the activity determination results of P.rockii ‘Zibanbai’ pollen at 0 h, 4th h and 8th h after anthesis, and (D–I) represents the activity determination results at 2nd–7th d after anthesis.
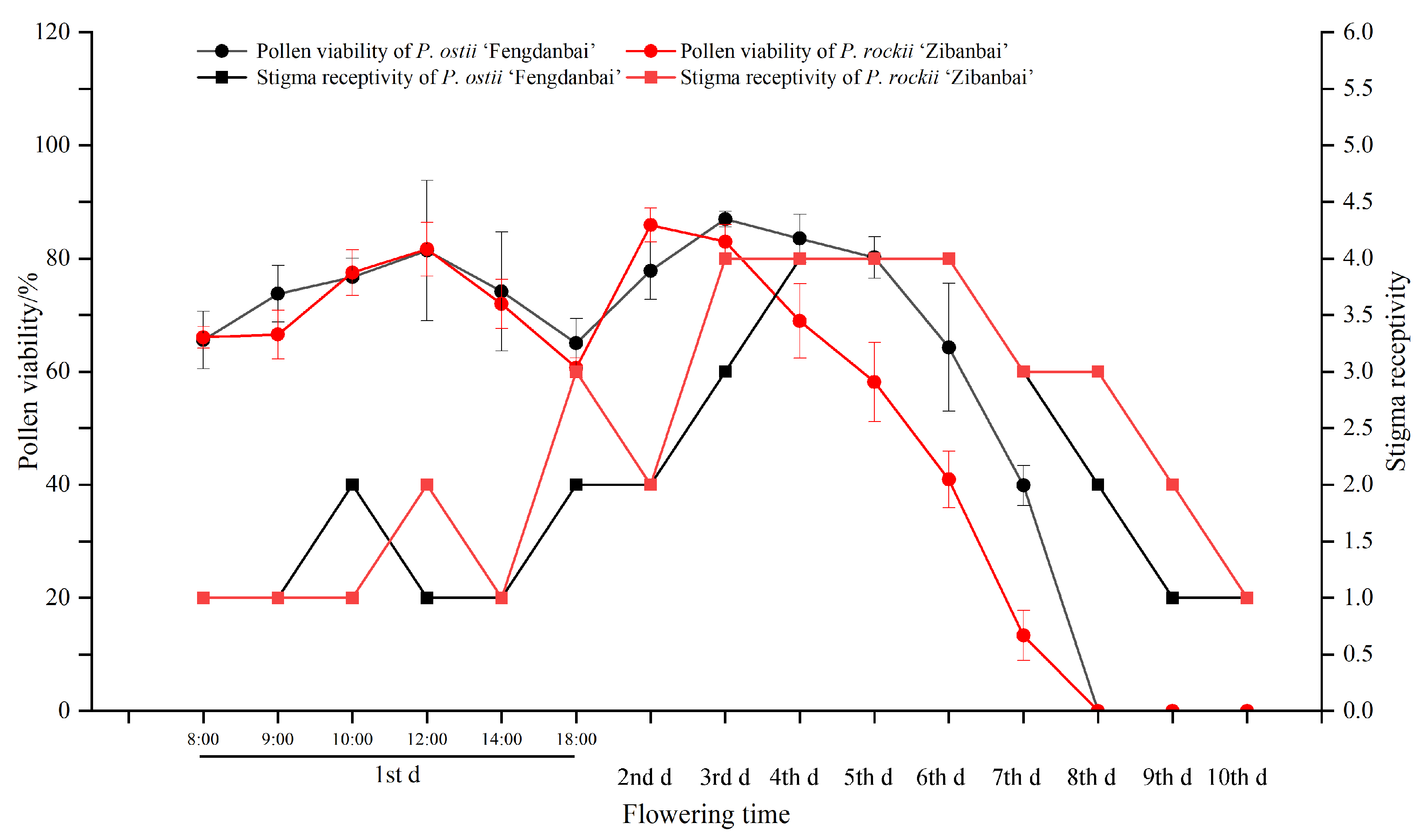
Figure 6: The temporal dynamic changes of pollen viability and stigma receptivity showed that the best pollination window period was 2nd–5th days after anthesis.
3.4 Correlation Analysis of Various Traits of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’
Pearson correlation analysis was used to analyze the correlation between corolla diameter, petal number per flower, anther number per flower, pollen number per flower, pollen viability, carpel number per fruit, number of ovule number per fruit, ovule number per fruit, seed setting rate and seed number per fruit (Fig. 7). The results showed that the correlation between the traits of oil tree peony was complex. There was a significant or extremely significant correlation between seed number per fruit and the five traits. It was significantly associated with petal number per flower, ovule number per fruit and the seed setting rate (p < 0.01). There was a significant positive association with carpel number per fruit (p < 0.05). In addition, the seed setting rate was significantly correlated with the petal number per flower (p < 0.01) and anther number per flower (p < 0.05). Ovule number per fruit was significantly positively correlated with ovule number per fruit and carpel number per fruit (p < 0.01), and significantly positively correlated with pollen viability (p < 0.05). There was a significant negative correlation between carpel number per fruit and anther number per flower (p < 0.05). Each traits restricts each other, and the patterns require experimental validation only by the correlation coefficient between yield and each trait.
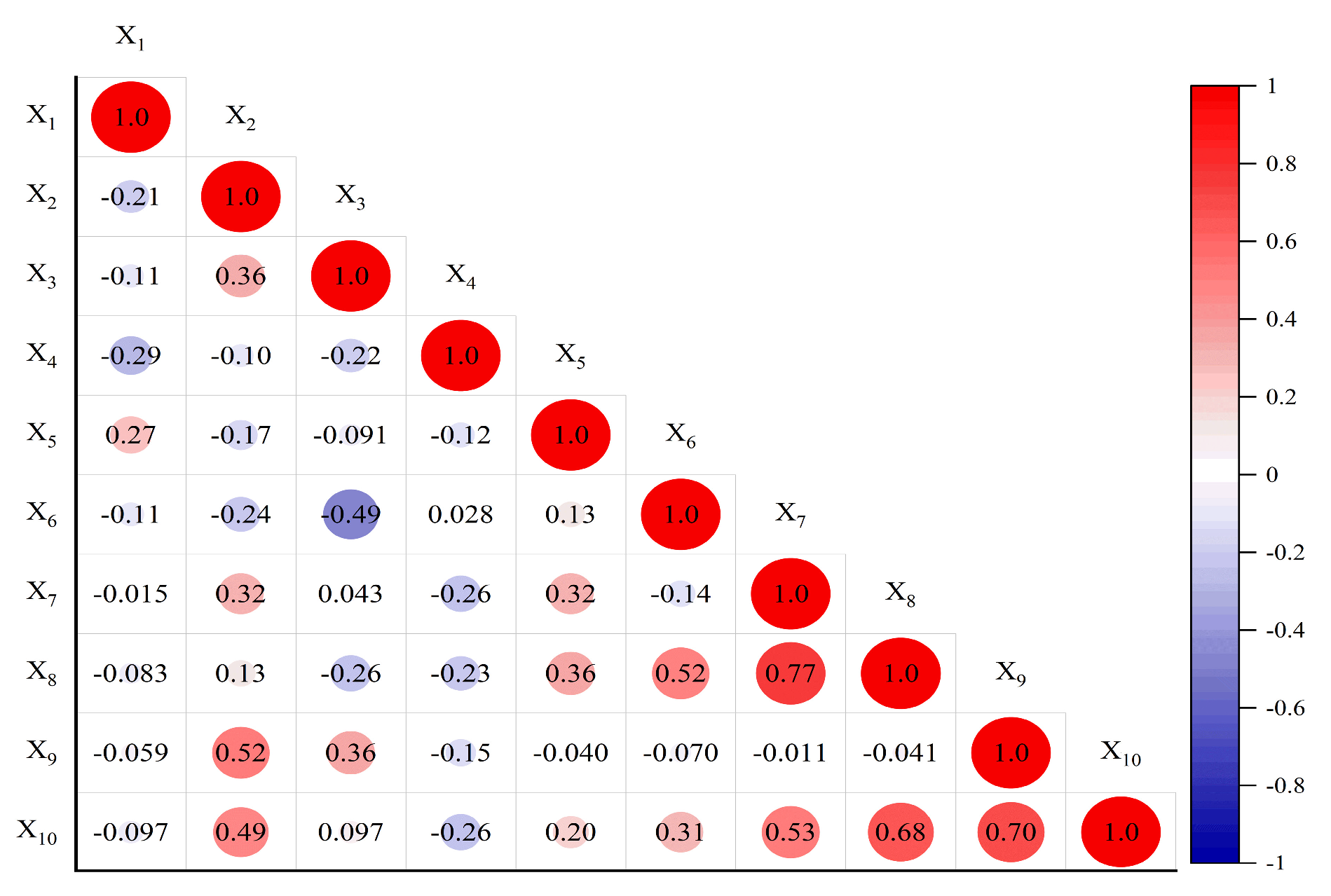
Figure 7: Correlation analysis heat map of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ traits. The indicators expressed in Xi are the same as in Table 1. Based on cultivar means (n = 2); limited degrees of freedom.
3.5 Linear Regression Analysis and Path Analysis of Seed Setting Characteristics
Statistical Note: The stepwise multiple regression model yielded a high coefficient of determination (R2 = 0.994, Adjusted R2 = 0.992). Model diagnostics were conducted on the residuals. The residuals were approximately normally distributed (mean = 0.00, standard deviation = 1.00), and a plot of standardized residuals against predicted values showed no systematic pattern, supporting the assumptions of normality and homoscedasticity.
Stepwise linear regression retained two significant predictors (Table 2): Y = −45.52 + 0.73X8 + 91.81X9 (R2 = 0.994, Adjusted R2 = 0.992, F = 547.97, df = 2, 37, p < 0.001).
Summary analysis showed the strongest associations with seed number per fruit and may serve as reference indicators for cultivar evaluation (Table 2).
Table 2: Linear regression analysis of seed number of seeds per fruit with each traits.
| B | SE | Beta | t | p | |
|---|---|---|---|---|---|
| const | −45.52 | 9.62 | - | −4.734 | 0.000** |
| X1 | 0.30 | 0.19 | 0.026 | 1.587 | 0.123 |
| X2 | −0.00 | 0.16 | −0.000 | −0.003 | 0.998 |
| X3 | 0.005 | 0.004 | 0.026 | 1.373 | 0.180 |
| X4 | 0.00 | 0.00 | 0.027 | 1.609 | 0.118 |
| X5 | −5.89 | 2.84 | −0.035 | −2.072 | 0.047* |
| X6 | −2.08 | 1.60 | −0.11 | −1.296 | 0.205 |
| X7 | −0.75 | 0.53 | −0.15 | −1.416 | 0.167 |
| X8 | 0.73 | 0.10 | 0.90 | 7.215 | 0.000** |
| X9 | 91.81 | 2.28 | 0.73 | 40.262 | 0.000** |
| R2 | 0.994 | ||||
| Adjusted R2 | 0.992 | ||||
| F | F(9, 30) = 547.97, p < 0.05 | ||||
| D | 2.465 | ||||
Path analysis of 7 traits showed that: Among the seven independent variables, the number of ovules per fruit had the greatest direct association with the number of seeds per fruit. The order of the direct path coefficient of each trait to the number of seeds per fruit is: Single fruit ovule number > seed setting rate > corolla diameter > anther number per flower > pollen viability > single fruit carpel number > Ovule number per fuit. Among them, corolla diameter, anther number per flower, ovule number per fruit and seed setting rate had positive effects on seed number per fruit, while other traits had negative effects. The order of the indirect path coefficients of each trait to the number of seeds per fruit is: Ovule number per fruit > single carpel number > pollen viability > anther number per flower > seed setting rate > corolla diameter > single fruit ovule number (Table 3). The indirect effects of anther number per flower, pollen viability, carpel number per fruit and ovule number per fruit were higher than the direct effects. It shows that these traits can indirectly affect the yield. Causal interpretation is limited by the observational design.
Table 3: Path analysis of traits significantly influencing number of seeds per fruit.
| Trait | Direct Effects P (X → Y) | Indirect Effects P (Xi → Xj → Y) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | X1 → Y | X3 → Y | X5 → Y | X6 → Y | X7 → Y | X8 → Y | X9 → Y | ||
| X1 | 0.026 | −0.12 | −0.0029 | −0.0095 | 0.0117 | 0.0023 | −0.075 | −0.043 | |
| X3 | 0.026 | 0.065 | −0.0029 | 0.0032 | 0.052 | −0.0065 | −0.24 | 0.26 | |
| X5 | −0.035 | 0.24 | 0.007 | −0.0024 | −0.014 | −0.050 | 0.33 | −0.029 | |
| X6 | −0.106 | 0.46 | −0.0029 | 0.013 | −0.0046 | 0.021 | 0.47 | −0.051 | |
| X7 | −0.151 | 0.69 | −0.0004 | 0.0011 | −0.012 | 0.015 | 0.69 | −0.008 | |
| X8 | 0.904 | −0.23 | −0.0021 | −0.0068 | −0.013 | −0.055 | −0.12 | −0.029 | |
| X9 | 0.727 | 0.014 | −0.0015 | 0.0094 | 0.0014 | 0.0074 | 0.0017 | −0.0037 | |
3.6 Membership Function Analysis of Key Indexes of Different Oil Tree Peony Varieties
Select the indicators that have a significant impact on the number of single fruit seeds obtained by regression analysis and path analysis: Pollen viability, number of ovules per fruit and seed setting rate were compared by membership function method to comprehensively compare the seed setting potential of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’. Membership function analysis of the key indicators revealed that P. ostii ‘Fengdanbai’ had a higher cumulative membership function value (Utotal = 1.775) than P. rockii ‘Zibanbai’ (Utotal = 1.594) (Table 4), suggesting a relatively better comprehensive performance based on the selected traits under the experimental conditions. We acknowledge that this ranking assumes equal weights for all traits and should be validated with additional germplasm.
Table 4: Affiliation function values of key indicators for P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’.
| Varieties | Membership Function Value of Pollen Viability | Membership Function Value of Number of Ovules Per Fruit | Membership Function Value of Seed Setting Rate | Total | Ranking |
|---|---|---|---|---|---|
| P. ostii ‘Fengdanbai’ | 0.736 | 0.505 | 0.534 | 1.775 | 1 |
| P. rockii ‘Zibanbai’ | 0.676 | 0.504 | 0.414 | 1.594 | 2 |
Ten floral and fruiting traits of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’were subjected to statistical analysis; subsequent comparison revealed significant inter-cultivar differences in pollen number per flower, ovule number per fruit, ovules per fruit, and seeds per fruit. Conversely, all remaining evaluated traits did not differ significantly between cultivars.
Yield is one of the important indexes to evaluate the value of oil tree peony. Pollen and stigma are essential structures mediating plant sexual reproduction. Quantifying pollen viability and stigma receptivity, together with their temporal overlap, enhances fruit set and pollination efficiency, providing critical insights for commercial production and hybrid breeding [18]. Pollen viability ranged from 39.9% to 87.0% in P. ostii ‘Fengdanbai’ and from 14.4% to 85.9% in P. rockii ‘Zibanbai’, peaking on day 3 and day 2 post-anthesis, respectively. Pollen remained viable for 7 d, while stigma receptivity persisted for 9 d in both cultivars. The functional overlap between pollen viability and stigma receptivity lasted 5 d; periods of peak viability and receptivity coincided for 4 d in P. ostii ‘Fengdanbai’ and 5 d in P. rockii ‘Zibanbai’. These temporal patterns vary among species, cultivars, and even developmental stages [30,31,32]. Beyond pollen longevity, the two cultivars differed significantly in pollen number per flower. This variation is attributable to both genetic factors [33,34] and environmental cues such as light, temperature, and humidity [35]. Large, showy flowers with abundant, bright-yellow pollen and expansive stigmatic surfaces enhance pollinator attraction and facilitate efficient insect-mediated pollination [36].
Yield-related traits of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’ were evaluated using correlation, linear regression, path, and membership function analyses. Among ten traits examined, seven were yield-related; four exhibited extremely significant differences (p < 0.01): petal number per flower, ovule number per fruit, ovule number per fruit, and seed-setting rate. This is basically consistent with the research results of Cui et al. [21] using multiple peony germplasm resources as materials, and is also basically consistent with the research results in Zea mays L. and Glycine max (L.) Merr. [37,38]. Studies have reported that stigma receptivity can significantly associated with the fruit setting rate of Passiflora edulis Sims and Vaccinium spp. [18,19]. However, there was a positive correlation between the stigma receptivity and the number of single fruit carpels on oil peony, but the effect was not significant. The reason for this phenomenon may be related to the specific reproductive mode of tree peony: tree peony is open at day and close at night, self-compatible, and requires cross-pollination by pollinators [10,39,40]. The specific effects of pollen viability and stigma receptivity on seed setting of oil peony need further research and analysis. Linear regression indicated that ovule number per fruit and seed-setting rate were the principal predictors of yield variation, corroborating earlier studies Path analysis confirmed that these two traits exhibited the greatest direct and indirect associations with yield, consistent with Cui et al. [21]. Membership function analysis ranked P. ostii ‘Fengdanbai’ superior to P. rockii ‘Zibanbai’ under identical environmental conditions, though this integration method assumes equal trait importance. In summary, pollen viability, number of ovules per fruit, and seed setting rate are significantly related to the single fruit yield of P. ostii ‘Fengdanbai’ and P. rockii ‘Zibanbai’. In the high-yield cultivation and variety breeding of oil tree peony, they should be used as the primary indicators. And P. ostii ‘Fengdanbai’ is a hybrid breeding resource superior to P. rockii ‘Zibanbai’.
Acknowledgement:
Funding Statement: This work was supported by the Innovation Scientists and Technicians Troop Construction Projects of Henan Province (212101510003).
Author Contributions: Xiangnan He and Xinyu Gao designed and executed the study; Xiangnan He and Xinyu Gao analyzed the data and wrote the first draft of the paper; Yuying Li, Kaiyue Zhang, Dongfeng Wei and Chunling He designed and analyzed the experimental results; Yuying Li and Xiangnan He wrote and revised the manuscript of the thesis; Xiaogai Hou initiated and led the project, guiding the design, analysis, and writing and revision of the paper. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The authors confirm that the data supporting the findings of this study are available within the article.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. National Health Commission of the People's Republic of China . Announcement on the Acer truncatum and Peony Seed Oil as New Resource Foods (No.9 of 2011) [Internet]. [cited 2025 Jan 1]. Available from: https://www.nhc.gov.cn/sps/c100088/201103/e8cb209a5d0b49eabc7d0e16756a82de.shtml [Google Scholar]
2. Li Y , Wang X , Wu S , Mei X , Song W , Qing S . Metabolic pathway for evaluation of polyunsaturated fatty acids content variation and regulation of its biosynthesis in Paeonia ostii through abscisic acid and ABSCISIC ACID-INSENSITIVE 5 (PoABI5). Ind Crops Prod. 2025; 227: 120758. doi:10.1016/j.indcrop.2025.120758. [Google Scholar] [CrossRef]
3. Yang X , Zhang D , Song LM , Xu Q , Li H , Xu H . Chemical profile and antioxidant activity of the oil from peony seeds (Paeonia suffruticosa Andr.). Oxid Med Cell Longev. 2017; 2017: 9164905. doi:10.1155/2017/9164905. [Google Scholar] [CrossRef]
4. He C , Zhang K , Hou X , Han D , Wang S . Foraging behavior and pollination efficiency of Apis mellifera L. on the oil tree peony ‘Feng Dan’ (Paeonia ostii T. Hong et J.X. Zhang). Insects. 2019; 10( 4): 116. doi:10.3390/insects10040116. [Google Scholar] [CrossRef]
5. Zhang K , He C , Wang S , Hou X . Influence of pollination methods on fruit development, fruit yield and oil quality in oil tree peony. Sci Hortic. 2022; 295: 110877. doi:10.1016/j.scienta.2022.110877. [Google Scholar] [CrossRef]
6. Peng LP , Cheng FY , Hu XG , Mao JF , Xu XX , Zhong Y , et al. Modelling environmentally suitable areas for the potential introduction and cultivation of the emerging oil crop Paeonia ostii in China. Sci Rep. 2019; 9( 1): 3213. doi:10.1038/s41598-019-39449-y. [Google Scholar] [CrossRef]
7. Gao J , Niu M , Lei Y , Wang X , Zhang Z , Zuo D , et al. PomiR164 targets PoLBD25 to promote early flowering in tree peony. Hortic Plant J. 2025. doi:10.1016/j.hpj.2025.05.015. [Google Scholar] [CrossRef]
8. Wang H , Wei S , He Y , Wang X , Li Y , Wei D , et al. Characterization of agronomic and seed oil features for different cultivars of tree peony. Plants. 2023; 12( 17): 3112. doi:10.3390/plants12173112. [Google Scholar] [CrossRef]
9. Xie L , Niu L , Zhang Y , Jin M , Ji D , Zhang X . Pollen sources influence the traits of seed and seed oil in Paeonia ostii ‘Feng Dan’. HortScience. 2017; 52( 5): 700– 5. doi:10.21273/hortsci11803-17. [Google Scholar] [CrossRef]
10. Niu T , Zhang C , Wang C , Xue X , Zhang Y , Guo L , et al. Identification of floral fragrance components in an F1 population derived from Paeonia ostii ‘Fengdan’ × P. suffruticosa ‘Chunguihuawu’ Cross. Ornam Plant Res. 2022; 2( 1): 1– 9. doi:10.48130/OPR-2022-0020. [Google Scholar] [CrossRef]
11. Zhang K , Wang X , Bao J , He X , Lei Y , He C , et al. Bumblebee pollination ensures the stability of both yield and quality of the woody oil crop Paeonia ostii ‘Fengdan’. Basic Appl Ecol. 2024; 79: 38– 45. doi:10.1016/j.baae.2024.03.001. [Google Scholar] [CrossRef]
12. Han C , Wang Q , Zhang H , Wang S , Song H , Hao J , et al. Light shading improves the yield and quality of seed in oil-seed peony (Paeonia ostii Feng Dan). J Integr Agric. 2017; 17( 7): 1631– 40. doi:10.1016/S2095-3119(18)61979-3. [Google Scholar] [CrossRef]
13. Kwek E , Zhu H , Ding H , He Z , Hao W , Liu J , et al. Peony seed oil decreases plasma cholesterol and favorably modulates gut microbiota in hypercholesterolemic hamsters. Eur J Nutr. 2022; 61( 5): 2341– 56. doi:10.1007/s00394-021-02785-9. [Google Scholar] [CrossRef]
14. Deng R , Gao J , Yi J , Liu P . Peony seeds oil by-products: Chemistry and bioactivity. Ind Crops Prod. 2022; 187( PA): 115333. doi:10.1016/j.indcrop.2022.115333. [Google Scholar] [CrossRef]
15. Iftikhar J , Lyu M , Liu Z , Mehmood N , Munir N , Ahmed MAA , et al. Sugar and hormone dynamics and the expression profiles of SUT/SUC and SWEET sugar transporters during flower development in Petunia axillaris. Plants. 2020; 9( 12): 1770. doi:10.3390/plants9121770. [Google Scholar] [CrossRef]
16. Wei S , Ma L . Comprehensive insight into tapetum-mediated pollen development in Arabidopsis thaliana. Cells. 2023; 12( 2): 247. doi:10.3390/cells12020247. [Google Scholar] [CrossRef]
17. Ait Aabd N , Tahiri A , Qessaoui R , Mimouni A , Bouharroud R . Self- and cross-pollination in argane tree and their implications on breeding programs. Cells. 2022; 11( 5): 828. doi:10.3390/cells11050828. [Google Scholar] [CrossRef]
18. Cai ZY , Dong L , Wang HQ , Qiu WW , Su WQ , Ren H , et al. Pollen viability, stigma receptivity and their effect on fruit set of passionfruit at different flower developmental stages. J Fruit Sci. 2023; 40( 5): 969– 77. (In Chinese). doi:10.13925/j.cnki.gsxb.20220523. [Google Scholar] [CrossRef]
19. Yang X , Li Z , Wang J , Liu Y , Chen G , Liu D , et al. Effects of pollen viability and stigma receptivity on interbreeding rate of orchid. Jiangsu Agric Sci. 2019; 47( 10): 166– 72. (In Chinese). [Google Scholar]
20. Shi XL , Liu W , Zhang L , Liu GX , Liu P , Hou XG . Comprehensive evaluation of seed yield and seed oil quality of Paeonia rockii. J Henan Univ Sci Technol Nat Sci. 2020; 41( 6): 73– 88. (In Chinese). doi:10.15926/j.cnki.issn1672-6871.2020.06.012. [Google Scholar] [CrossRef]
21. Cui H , Chen C , Huang N , Cheng F . Association Analysis of Yield, Oil and Fatty Acid Content, and Main Phenotypic Traits in Paeonia Rockii as an Oil Crop. J Hortic Sci Biotechnol. 2018; 93( 4): 425– 32. doi:10.1080/14620316.2017.1381045. [Google Scholar] [CrossRef]
22. Zhao J , Li W , Jiang F , Zhang T , Yan X , Liu X , et al. PoSCPL61 as a negative regulator of seed size in tree peony of Paeonia ostii revealing by genome-wide identification of SCPL genes and functional validation. Ind Crops Prod. 2025; 237: 122191. doi:10.1016/j.indcrop.2025.122191. [Google Scholar] [CrossRef]
23. Chang M , Wang Z , Zhang T , Wang T , Liu R , Wang Y , et al. Characterization of fatty acids, triacylglycerols, phytosterols and tocopherols in peony seed oil from five different major areas in China. Food Res Int. 2020; 137: 109416. doi:10.1016/j.foodres.2020.109416. [Google Scholar] [CrossRef]
24. Zhang T , Zhang Z , Qiao Q , Liu W , Hou X . Effect of cultivation methods on seed yield traits, oil yield, and fatty acid compositions of cultivated Paeonia ostii. HortScience. 2021; 56( 12): 1505– 12. doi:10.21273/hortsci16215-21. [Google Scholar] [CrossRef]
25. He C , Cheng F . PrDA1-1 and its interacting proteins PrTCP1/PrTCP9 in flare tree peony affect yield by regulating seed weight and number. J Integr Agric. 2025; 24( 2): 610– 22. doi:10.1016/j.jia.2024.10.001. [Google Scholar] [CrossRef]
26. Shen GH , Wang XQ , Luo J , Zhang SL , Qian PH , Jin FL . Effects of greenhouse culture on pear pollen quantity per anther and pollen viability. Acta Agric Shanghai. 2008; 24( 3): 54– 7. (In Chinese). [Google Scholar]
27. Barman C , Singh VK , Tandon R . Reproductive biology of Salvadora oleoides Decne. (Salvadoraceae). Int J Plant Reprod Biol. 2018; 10( 1): 69– 76. doi:10.14787/ijprb.201810.1.69-76. [Google Scholar] [CrossRef]
28. Bao J , Zhang K , He X , Chen Z , Wang J , He C , et al. Comparison of pollen-collecting abilities between Apis mellifera L. and Bombus terrestris L. in the oil tree peony field. Horticulturae. 2023; 9( 6): 658. doi:10.3390/horticulturae9060658. [Google Scholar] [CrossRef]
29. Luo CW , Chen Y , Liu Y . Pollen viability, stigma receptivity and reproductive characteristics of Paeonia ostia (Paeoniaceae). J Henan Agric Sci. 2019; 48( 07): 48– 53. (In Chinese). doi:10.15933/j.cnki.1004-3268.2019.07.007. [Google Scholar] [CrossRef]
30. Arenas-de-Souza MD , Rossi AAB , Varella TL , Da Silveira GF , Souza SAM . Stigmatic receptivity and pollen viability of Theobroma subincanum Mart.: Fruit species from the Amazon region. Rev Bras Frutic. 2016; 38( 4): e-757. doi:10.1590/0100-29452016757. [Google Scholar] [CrossRef]
31. Lankinen Å , Lindström SAM , D’Hertefeldt T . Variable pollen viability and effects of pollen load size on components of seed set in cultivars and feral populations of oilseed rape. PLoS One. 2018; 13( 9): e0204407. doi:10.1371/journal.pone.0204407. [Google Scholar] [CrossRef]
32. Isiaka AI , Agossou COA , Agbolade JO , Adje CAO , Fassinou Hotegni NV , Tossou MG , et al. Phenology, floral organs ultra-structure, traits correlation, stigma receptiveness, pollen viability and germinability in horsegram (Macrotyloma uniflorum Lam.) Verdc. S Afr N J Bot. 2023; 161: 444– 53. doi:10.1016/j.sajb.2023.08.033. [Google Scholar] [CrossRef]
33. Du G , Xu J , Gao C , Lu J , Li Q , Du J , et al. Effect of low storage temperature on pollen viability of fifteen herbaceous peonies. Biotechnol Rep. 2019; 21: e00309. doi:10.1016/j.btre.2019.e00309. [Google Scholar] [CrossRef]
34. Wang X , Liang H , Guo D , Guo L , Duan X , Jia Q , et al. Integrated analysis of transcriptomic and proteomic data from tree peony (P. ostii) seeds reveals key developmental stages and candidate genes related to oil biosynthesis and fatty acid metabolism. Hortic Res. 2019; 6( 1): 111. doi:10.1038/s41438-019-0194-7. [Google Scholar] [CrossRef]
35. Duan X , Liu W , Wang X , Zhang L , Liu S , Guo L , et al. Effects of Phosphorus Fertilization on Growth Characteristics, Fatty Acid Composition, and Seed Yields of Fengdan (Paeonia ostii T. Hong & J. X. Zhang). HortScience. 2022; 57( 6): 733– 40. doi:10.21273/HORTSCI16555-22. [Google Scholar] [CrossRef]
36. Wang D , Ma Y , Zhao X , Wang L . Genetic diversity of Rhododendron dauricum based on morphological traits and SSR markers. Front Plant Sci. 2025; 16: 1533824. doi:10.3389/fpls.2025.1533824. [Google Scholar] [CrossRef]
37. Bavec F , Bavec M . Effects of plant population on leaf area index, cob characteristics and grain yield of early maturing maize cultivars (FAO 100–400). Eur J Agron. 2002; 16( 2): 151– 9. doi:10.1016/S1161-0301(01)00126-5. [Google Scholar] [CrossRef]
38. Li S , Cao Y , Wang C , Yan C , Sun X , Zhang L , et al. Genome-wide association mapping for yield-related traits in soybean (Glycine max) under well-watered and drought-stressed conditions. Front Plant Sci. 2023; 14: 1265574. doi:10.3389/fpls.2023.1265574. [Google Scholar] [CrossRef]
39. Zhang K , Li Y , Bao J , He X , Chen L , Guo L , et al, Bee-mediated pollination enhances fruit set and seed yield in Paeonia ostii ‘Fengdan’: insights into physiological and molecular mechanisms. Hortic Res, 2025; 12( 11): uhaf224. doi:10.1093/hr/uhaf224. [Google Scholar] [CrossRef]
40. He C , Zhang K , Han D , Wang S , Hou X , Zhu C . Foraging Behavior of Honeybees (Apis Mellifera L.) and Ground Bumblebees (Bombus Terrestris L.) and its Influence on Seed Yield and Oil Quality of Oil Tree Peony Cultivar ‘Fengdan’ (Paeonia Ostii T. Hong et J. X. Zhang). J Apic Sci. 2020; 64( 1): 131– 42. doi:10.2478/jas-2020-0014. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools