 Open Access
Open Access
ARTICLE
Responses of Osmotic Adjustment and Antioxidant Enzyme Activities in Alfalfa (Medicago sativa L.) Seedlings to Biochar Amendment under Salt Stress
1 College of Grassland Science, Qingdao Agricultural University, Qingdao, China
2 Shandong Key Laboratory for Germplasm Innovation of Saline-alkaline Tolerant Grasses and Trees, Qingdao Agricultural University, Qingdao, China
3 Forestry and Grassland Science Research Institute of Hulunbuir, Hulunbuir, China
4 Laboratory Management Center, Qingdao Agricultural University, Qingdao, China
* Corresponding Authors: Shangzhi Zhong. Email: ; Lulu Sun. Email:
# These authors contributed equally to this work
(This article belongs to the Special Issue: Plant Responses to Stress Factors)
Phyton-International Journal of Experimental Botany 2026, 95(3), 16 https://doi.org/10.32604/phyton.2026.079919
Received 30 January 2026; Accepted 25 February 2026; Issue published 31 March 2026
Abstract
Salt stress has been demonstrated to trigger the overaccumulation of reactive oxygen species (ROS) within plant tissues. This process, in turn, has been shown to induce oxidative damage and further impair the physiological and metabolic processes. As a promising organic soil conditioner, biochar has proven effective in mitigating the adverse effects of salt stress in crops. Nevertheless, the synergistic and interactive effects of salt stress and biochar application on the osmotic adjustment system and antioxidant defense system of alfalfa (Medicago sativa L.) have not been fully elucidated. This study employed a completely randomized design, comprising four NaCl-induced salt stress levels (0, 3, 6, and 9 dS m−1) and three biochar application rates (0, 30, and 60 g kg−1). To evaluate the role and underlying mechanism of biochar in alleviating oxidative damage caused by salt stress in alfalfa, we determined the growth indicators (shoot biomass [SB], root biomass [RB], and total biomass [TB]), physiological indicators (total chlorophyll content and relative water content), and biochemical indicators (osmotic adjustment substances, antioxidant enzyme activities, and oxidative stress-related substances). Results indicated that salt stress significantly suppressed alfalfa biomass accumulation by 16%–84%. With increasing salt stress intensity, the contents of proline (Pro), glycine betaine (GB), soluble sugars (SS), and soluble proteins (SP), along with the activities of catalase (CAT), peroxidase (POD), superoxide dismutase (SOD), ascorbate peroxidase (APX), and polyphenol oxidase (PPO), significantly increased in alfalfa leaves; yet, these defensive responses failed to prevent the decrease in leaf relative water content and the significant accumulation of superoxide anion (O2•−), hydrogen peroxide (H2O2), and malondialdehyde (MDA) contents. These results indicated that the endogenous defense system alone is insufficient to counteract salt stress-induced oxidative damage. In contrast, compared with the non-biochar control, biochar application significantly increased alfalfa chlorophyll content (by ~40%) and biomass accumulation (by ~12%). Notably, under the 30 g kg−1 biochar treatment, the antioxidant enzyme activities and osmotic adjustment substance contents of alfalfa seedlings significantly decreased, accompanied by a marked reduction in O2•− (by ~13%), H2O2 (by ~21%), and MDA (by ~30%) contents. These results indicated that biochar could maintain the dynamic equilibrium of osmotic regulation and antioxidant enzyme systems in alfalfa leaves, and promote the synthesis of photosynthetic pigments and photosynthates, thereby enhancing alfalfa biomass accumulation by 54%–68%. However, under severe salt stress (9 dS m−1), excessive biochar application rate (60 g kg−1) failed to exert a positive effect on alfalfa growth, with only approximately a 1% increase in total biomass of alfalfa seedlings. Collectively, our findings demonstrated that appropriate biochar application can effectively alleviate salinity-induced oxidative damage in alfalfa and thus promote its growth in saline soils. To alleviate salt stress-induced phytotoxicity and optimize the efficacy of soil amendments, it is imperative to rationally regulate the application amount of biochar in saline soils.Keywords
Globally, an estimated 1.4 billion hectares of terrestrial land, accounting for more than 10% of the global total land area, is threatened by soil salinization [1]. In China, there are approximately 7.6 million hectares of salt-affected soils (over 5% of the total arable land area), which is projected to expand continuously in the coming years, driven by unsustainable water and land management practices, along with the intensifying impacts of global climate change [2]. Salt stress is defined as an adverse condition caused by the excessive accumulation of soluble salts in soil, which typically induces a series of adverse effects, including the disruption of ion balance, induction of osmotic stress, and excessive production of reactive oxygen species (ROS) [3,4]. These stress effects severely disturb the normal physiological and biochemical metabolic processes in plants (e.g., impairing chloroplast structure and thereby reducing chlorophyll content), thus inhibiting plant growth and development, and ultimately leading to a significant decline in crop yield and quality [5,6]. Numerous investigations have shown that abiotic stresses, especially salt stress, disrupt the dynamic equilibrium between ROS generation and scavenging in plant cells, and excessive ROS accumulation is the core reason for the decline in global crop yields [3,7,8,9]. Excessive ROS can impair multiple cellular physiological functions by damaging nucleic acid structures, oxidatively modifying proteins, and inducing lipid peroxidation, with the most prominent consequence being the impairment of cell membrane structural integrity [10]. This not only hinders substance transport across cell membranes but also disrupts cellular osmotic homeostasis and ultimately seriously affects the normal physiological functions in plants [11].
The major ROS that accumulate at high concentrations in plants include hydrogen peroxide (H2O2), hydroxyl radical (•OH), singlet oxygen (1O2), and superoxide anion (O2•−) [7]. Given the frequent occurrence of water and salt stresses, which can affect the majority of habitats, plants have evolved a series of sophisticated adaptive strategies to cope with these abiotic stresses [12,13]. Plants possess efficient ROS scavenging systems to counteract oxidative damage induced by excessive ROS accumulation [14]. Studies have confirmed that under salt stress, plants can scavenge stress-induced ROS through the enzymatic antioxidant system, whose core components are superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), ascorbate peroxidase (APX), and polyphenol oxidase (PPO) [15,16]. Specifically, SOD converts O2•− into H2O2 to alleviate cellular damage, while POD, CAT, and APX further catalyze the decomposition of H2O2 into water and oxygen, achieving the cascade scavenging of ROS [7]. Meanwhile, plants can maintain cellular ion homeostasis by limiting excessive Na+ accumulation and sustaining a stable Na+/K+ ratio, thereby mitigating ion toxicity-induced damage to plant tissues, which is primarily because K+ functions as an important cofactor/activator for key antioxidant enzymes (e.g., SOD, POD, CAT, and APX) [17]. Therefore, maintaining cellular ion homeostasis can prevent a marked reduction in antioxidant enzyme activity, which would otherwise lead to the accumulation of ROS and exacerbate oxidative damage. As reported by Urbanavičiūtė et al., this process is coordinately regulated by the Salt Overly Sensitive (SOS) pathway genes (e.g., SOS3, SOS2, and SOS1), the vacuolar Na+/H+ antiporter gene (e.g., NHX1), high-affinity K+ transporters (HKT), and the calcineurin B-like protein (CBL)–CBL-interacting protein kinase (CIPK) signaling complex, which collectively limit intracellular Na+ buildup while sustaining K+ acquisition [18]. Apart from the enzymatic antioxidant system, plant cells can also maintain osmotic homeostasis by accumulating osmotic adjustment substances to combat osmotic damage induced by salt stress [19,20]. The core mechanism involves increasing the concentration of compatible organic solutes in tissues to minimize intracellular water loss, enhance water absorption capacity in saline environments, and thereby avoid metabolic function disruptions from osmotic dehydration [21]. Relevant research has demonstrated that under salt stress, plants can significantly elevate tissue levels of osmotic adjustment substances, such as endogenous glycine betaine (GB), free proline (Pro), soluble sugars (SS), and soluble proteins (SP) [12,19]. This optimization of osmotic balance ultimately alleviates the symptoms of saline-alkaline stress [11]. Furthermore, these substances can protect the bio-membrane integrity, stabilize protein conformations, scavenge ROS, and maintain cellular redox homeostasis, playing a pivotal role in enhancing plant salt stress tolerance [4,22].
Alfalfa (Medicago sativa L.) represents a key high-value species in the economic forage industry. This long-lived leguminous forage is characterized by a deep root system, superior biomass yield, excellent palatability, and excellent digestibility. Its rich nutritional composition, particularly in terms of protein, vitamins, and mineral elements, further enhances its significance in modern agriculture [23]. Meanwhile, alfalfa can adapt to mild saline-alkaline environment, which can effectively lower the groundwater level, remove soil salt, and improve the physicochemical properties of saline soils, thus creating a suitable soil environment for the subsequent cultivation of other economic crops [16]. Previous research by Al-Farsi et al. [19] has shown that under mild saline-alkaline stress, alfalfa can significantly increase fresh forage yield by upregulating antioxidant enzyme activities and enhancing osmotic adjustment substance accumulation. However, this species is relatively sensitive to moderate salt stress, with its growth potential and productivity are significantly inhibited. Research has shown that under either moderate (6 dS m−1) or severe (10 dS m−1) salt stress, alfalfa accumulates higher amounts of free proline in leaves, increases soluble phenolic substances, enhances CAT activity, and raises potassium ion levels in leaves, yet forage fresh weight exhibits no significant difference compared with the control group [24,25]. Therefore, enhancing alfalfa’s soil adaptability and tolerance to salt stress in salt-alkaline environments is a critical issue that urgently requires addressing in current production [23]. Currently, saline-alkaline soil improvement techniques such as substrate drainage, sand mixing and mulching, and field management have limited applications due to their shortcomings, such as high cost, low remediation efficiency, and potential risk of secondary pollution [26]. Therefore, it is imperative to develop efficient, low-cost, and eco-friendly management strategies to remediate saline-alkaline soils, alleviate the adverse impacts of ROS-induced damage by salt stress on alfalfa, and thereby improve its productivity in saline environments.
Biochar is a stable carbonaceous material produced by heating organic materials in an oxygen-limited environment [27]. It exhibits outstanding application prospects for improving the physical and chemical characteristics as well as nutrient availability of salt-affected soils [28]. Its unique porous structure, conferred by a high specific surface area, endows it with outstanding physicochemical stability and a strong ability to adsorb and retain essential soil elements [29,30]. Relevant studies have shown that biochar application could regulate plant growth and alleviate stress damage through direct and indirect dual pathways under saline-alkaline stress: (1) Directly, biochar is rich in mineral elements such as nitrogen (N) and phosphorus (P), which directly enhance plant nutrient uptake efficiency and provide material support for plant growth and development [31]. (2) Indirectly, its regulatory effects mainly lie in two aspects: First, its porous structure can effectively improve soil physicochemical properties; increased application doses of biochar markedly raise the fraction of soil macroaggregates and soil porosity, thereby enhancing soil aeration and optimizing soil water-holding capacity (WHC) [32,33]. Second, biochar modulates the structure, metabolic activity, and diversity of soil bacteria and fungi, thereby indirectly affecting the availability and supply efficiency of soil nutrients (e.g., soil available N and available P), and creating a favorable microenvironment for plant growth [30,34]. Overall, biochar application synergistically optimizes nutrient supply, soil physicochemical properties, and the microecological environment, thereby significantly promoting plant growth in salt-stressed soils and effectively alleviating saline-alkaline stress-induced growth inhibition in plants [29,35]. This provides important technical support for vegetation restoration in saline soils. However, the existing research on the effects of biochar on the osmotic adjustment system and antioxidant enzyme regulation system of alfalfa leaves under gradient salt stress remains relatively limited.
Therefore, the purpose of this study was to investigate the responses of osmotic adjustment and antioxidant enzyme systems in alfalfa seedlings to biochar amendment under salt stress. Specifically, the determined parameters included growth traits (total biomass [TB], shoot biomass [SB], and root biomass [RB]), leaf physiological characteristics (total chlorophyll [Chl] content and relative water content [RWC]), osmotic adjustment substances (Pro, GB, SS, and SP), antioxidant enzyme activities (CAT, POD, SOD, APX, and PPO), along with oxidative stress indicators (H2O2, O2•−, and malondialdehyde [MDA]). We hypothesized two predictions based on earlier reports and our prior pilot test findings: (1) salt stress can inhibit the physiological traits and growth of alfalfa, but alfalfa would mitigate oxidative damage caused by excessive ROS accumulation through the active regulation of its osmotic adjustment and antioxidant enzyme systems; (2) appropriate biochar application rates could significantly enhance the contents of osmotic adjustment substances and activities of antioxidant enzymes in alfalfa, thereby improving its growth performance. This study is expected to provide empirical data support and a theoretical basis for developing a green regulatory strategy to enhance alfalfa salt tolerance under saline conditions.
2.1 Biochar and Soil Preparation
The biochar employed in this study was derived from wheat straw via a 500°C pyrolysis process. The experimental soil was collected from the uppermost layer of soil (0–25 cm depth) of a loam soil in Jiaozhou City, Shandong Province, China (120°04′ E, 36°26′ N, 37 masl). The fundamental physicochemical characteristics of the examined soil and biochar are presented in Table 1.
Table 1: Soil and biochar characteristics in this study. Data are presented as the arithmetic mean of five independent replicates.
| TC | TN | TP | TK | OC | AN | AP | AK | pH | EC | |
|---|---|---|---|---|---|---|---|---|---|---|
| Biochar | 582.39 | 1425.17 | 1357.33 | 19.45 | 524.15 | 134.71 | 697.26 | 13.78 | 8.91 | 2.96 |
| Soil | 23.23 | 673.29 | 818.43 | 8.54 | 20.91 | 41.63 | 17.82 | 2.63 | 7.85 | 1.24 |
2.2 Study Site and Experimental Design
The experiment was conducted within an artificial climate greenhouse at Qingdao Agricultural University (120°39′ E, 36°31′ N, 50 masl), adopting a two-factor fully randomized block design including salt stress (S) and biochar amendment (B). Alfalfa seedlings (Medicago sativa L. cv. Zhongmu No.3) were subjected to four levels of salt stress (0 dS m−1, 3 dS m−1, 6 dS m−1, and 9 dS m−1, induced by NaCl) and three biochar application rates (0 g kg−1, 30 g kg−1, and 60 g kg−1). The seeds of M. sativa were subjected to a process of surface sterilization, employing 75% ethanol as the primary agent, followed by thorough rinsing with deionized (DI) water. The seeds were then subjected to a germination process on moist filter paper at 20°C for a period of 48 h. Before the transplanting of alfalfa seedlings, the weighed biochar was fully mixed with the test soil, and the mixture was filled into plastic pots (24 cm × 23 cm), each containing with 5 kg of soil-biochar mixture in each pot. Five alfalfa seedlings with consistent growth vigor and uniform size were selected and transplanted into each pot, with no seedling injury during the transplanting process. Salinity treatments were initiated two weeks after seedling transplantation, with NaCl-supplemented irrigation water used to ensure uniform application of salt stress. To mitigate acute salt injury to alfalfa seedlings, a gradual increment method was adopted for salt stress imposition. Specifically, the NaCl concentration in the irrigation water was increased by one-third of the final target concentration every 3 days until the predetermined salt stress level was reached. During the seedling establishment period, daily irrigation was applied to maintain the soil water content at 70% of field capacity, ensuring water did not become a limiting factor for growth. The greenhouse environment was controlled throughout the experiment, adhering to a day/night temperature regime of 25/20°C and a relative humidity of 70%.
After five weeks of continuous treatment, alfalfa fresh leaf samples were collected, and leaf RWC measurement and biomass determination were conducted concurrently [36]. Fresh leaf samples were rapidly frozen with liquid nitrogen (LN) to halt metabolic activity and prevent enzymatic degradation, the breakdown of proteins caused by enzymes, which maximized the retention of their inherent physiological and biochemical properties. The samples were subsequently divided into two portions for the purpose of determining the physiological and biochemical parameters: one portion was freeze-dried to constant weight using a freeze dryer, while the other portion was promptly transferred to an ultra-low temperature freezer set at a temperature of −80°C for the purpose of temporary storage. The leaf samples were subjected to a process of freeze-drying, after which they were ground into a uniform fine powder using a ball mill (MM 400, Retsch, Haan, Germany). The powder was then sieved through a 100-mesh sieve for the determination of glycine betaine (GB), proline (Pro), soluble sugar (SS), and malondialdehyde (MDA) contents. The samples temporarily stored in the ultra-low temperature freezer were used for assaying total chlorophyll (Chl) content, soluble protein (SP) content, antioxidant enzyme activities (including catalase [CAT], peroxidase [POD], superoxide dismutase [SOD], ascorbate peroxidase [APX], and polyphenol oxidase [PPO]), and ROS-related indicators (hydrogen peroxide [H2O2] and superoxide anion radical [O2•−]).
In the present study, the shoots and roots of each plant in each treatment group were carefully harvested. Subsequently, the roots were rinsed through mesh sieves (0.2 mm) until free of soil particles. All harvested shoot and root tissues were heat-fixed at 105°C for 30 min to inactivate endogenous enzyme activity in plant tissues, thereby preserving the compositional quality and physicochemical stability of plant samples. Following this, the tissues were dried to a constant weight in a forced-air oven at 65°C, and the weight was subsequently determined using an electronic analytical balance. Shoot biomass (SB) and root biomass (RB) were weighed separately, with total biomass (TB) calculated as the sum of the two components [37].
2.4 Determination of Total Chl Content and RWC in Alfalfa Leaves
The total Chl content was determined and calculated according to the methodology established by Zhong et al. [9]. Briefly, 0.2 g of alfalfa leaf samples were punched into small pieces and transferred to test tubes, to which 2 mL of 95% acetone was added. In order to ensure full extraction and prevent chlorophyll degradation, the test tubes were subjected to an incubation process at 37°C in conditions of darkness for a period of 4 h. After incubation, the mixture was subjected to a centrifugation procedure, and the supernatant was collected to determine absorbances at 663 nm and 645 nm, respectively. The total Chl content was determined through the utilization of the following formula:
The RWC of alfalfa leaves was determined using the method of Barrs and Weatherley [38]. A fresh leaf sample measuring 0.1 g was selected for the purpose of the experiment, and its FW was recorded at the outset. The samples were then soaked in DI water until fully saturated, after which the saturated weight (SW) was measured. Subsequently, the leaf samples were oven-dried at 65°C to a constant dry weight (DW). The leaf RWC of alfalfa was calculated using the following equation: RWC (%) = [(FW − DW)/(SW − DW)] × 100%.
2.5 Determination of Pro, GB, SS, and SP Contents in Alfalfa Leaves
The Pro concentration was determined according to the method previously described by Bates et al. [39]. The absorptivity of the upper organic phase was measured (520 nm), and the Pro content was calculated on the basis of the standard curve, which was generated from a proline standard solution.
The GB content was determined following the method established by Grieve and Grattan [40]. The absorbance of the reaction solution was measured at 365 nm using a microplate reader (Infinite M Plex, Tecan Trading Co., Ltd., Salzburg, Austria). The calculation of GB content was performed utilizing the following formula:
The SS content was determined according to the anthrone-sulfuric acid colorimetric method of Yoshida et al. [41]. In summary, 0.5 g of mature alfalfa leaves was homogenized in 5 mL of 80% anthrone solution. Subsequently, 10 mL of anthrone reagent (2 g anthrone dissolved in 1 L of 95% H2SO4 w/v) was slowly added to the homogenate. The mixture was heated in a water bath at 80°C for 30 min to ensure complete extraction, and then centrifuged at 8000× g for 10 min at 4°C to collect the supernatant. The degree of absorption of the extract was determined at a wavelength of 620 nm using a microplate reader. A calibration curve was generated with glucose as the standard, and the total SS content in alfalfa leaves was consequently quantified. The calculation of SS content was determined by the following formula:
The SP content in alfalfa leaf tissue was assayed following the method reported by Bradford [42]. Following centrifugation (12,000× g, 4°C, 15 min), the supernatant was collected and subjected to absorbance detection at 595 nm via a microplate reader. SP content was quantified via a calibration curve established with bovine serum albumin (BSA) as the standard.
2.6 Determination of CAT, POD, SOD, APX and PPO Activities in Alfalfa Leaves
Briefly, 0.5 g of fresh alfalfa leaf tissue was ground in an ice-cold mortar with 6 mL of 50 mmol L−1 sodium phosphate buffer (pH 7.0), which was pre-supplemented with 0.2 mmol L−1 ethylenediaminetetraacetic acid (EDTA) and 1% polyvinylpyrrolidone (PVP) to scavenge phenolic compounds and maintain enzymatic activity. After filtration through four layers of sterile cheesecloth, the homogenate was centrifuged (15,000× g, 4°C, 20 min). The resulting supernatant was harvested as the crude enzyme extract for later activity measurements. Using the method of Maehly and Chance [43], the activities of CAT (the absorbance at 240 nm) and POD (the absorbance at 470 nm) were measured.
CAT activity was calculated using the formula:
POD activity was calculated using the formula:
SOD activity was determined following the method described by Geng et al. [44]. This method quantifies SOD activity by measuring its capacity to suppress the photochemical reduction of nitro-blue tetrazolium (NBT). SOD activity was computed using the equation below:
APX activity was assayed according to the method described by Nakano and Asada [45], with activity calculated according to the linear region of the absorbance decrease and the molar extinction coefficient of ascorbate. APX activity was calculated using the formula:
PPO activity was assayed according to the protocol established by Gauillard et al. [46]. The absorbance at 495 nm was recorded at 30-s intervals for 3 min using a spectrophotometer. PPO activity was estimated from the linear change in absorbance (ΔOD min−1 mg−1 protein) and normalized to the corresponding protein content.
2.7 Determination of H2O2, and O2•− and MDA Contents in Alfalfa Leaves
The H2O2 concentration in alfalfa leaves was determined according to the protocol of Velikova et al. [47]. Absorbance of the reaction solution was recorded at 390 nm, and the H2O2 concentration was quantified based on a standard curve prepared with known standards. The calculation was performed using the equation shown below:
The generation rate of O2•− was measured with reference to Yan et al. [48]. Since the extraction procedure for O2•− was identical to that of H2O2, the crude extract prepared above was directly used for subsequent reactions. The absorbance of the reaction solution was detected spectrophotometrically at 530 nm. The O2•− concentration was calculated against a calibration curve established with sodium nitrite as the standard, combined with the reaction time. The corresponding value was derived using the equation below:
MDA content in alfalfa leaves was measured using a modified thiobarbituric acid (TBA) method reported by Heath and Packer [49]. In brief, 0.5 g of fresh leaf tissue was homogenized with 5 mL of 5% TBA solution. After centrifugation at 1790× g for 10 min, a 2 mL supernatant fraction was mixed with TBA solution. The mixture was incubated in a 98°C water bath for 10 min, then cooled to ambient temperature to stop the reaction. After a second centrifugation to remove precipitates, the absorbance of the supernatant was measured at 532 nm. Background correction was performed at 600 nm to eliminate nonspecific interference, and the MDA concentration was quantified accordingly. MDA content was computed according to the equation below:
Statistical analysis was performed using SPSS 22.0 (SPSS Inc., Chicago, USA), while graphical visualization was conducted with SigmaPlot 12.5 (Systat Software, San Jose, USA). Normality and homogeneity of variances were tested using the Shapiro–Wilk and Levene’s tests, respectively. Differences among treatments for all determined indices were assessed by one-way ANOVA followed by Duncan’s multiple range test (p < 0.05). Furthermore, two-factor ANOVA was applied to evaluate the individual impacts of salinity (S) and biochar application (B), together with their interactive influence (S × B). Correlation analysis of all quantified indicators was carried out with the GGally and ggplot2 libraries in R (v4.2.2; R Core Team, Vienna, Austria). All experimental data are expressed as mean ± standard deviation (SD) from four replicates (n = 4).
3.1 Impacts of Biochar Application on Biomass, Total Chl Content, and RWC of Salt-Stressed Alfalfa
Our results revealed that both salinity exposure and biochar application exerted substantial effects on the growth status of alfalfa seedlings, including shoot biomass (SB), root biomass (RB), total biomass (TB) of alfalfa seedlings, and total Chl content (p < 0.05, Table 2), but biochar amendment had no significant effect on leaf relative water content () (p > 0.05, Table 2). Compared with non-saline control, SB, RB, TB, total Chl content, and RWC decreased significantly as salt stress intensified (Fig. 1 and Fig. 2). In the present study, biochar application alleviated this salt-induced decline, increasing the values of these parameters, except for RB and RWC under the 9 dS m−1 NaCl stress condition. Based on the average values of all measured indices, the 3% biochar amendment exhibited significantly higher values than the other biochar treatments (p < 0.05). Under the same salt stress levels, the 3% biochar amendment treatment enhanced SB by 65%–73%, RB by 41%–61%, TB by 54%–68%, total Chl content by 8%–23%, and RWC by 1%–7% compared with no biochar addition treatment (non-biochar). However, under the 9 dS m−1 NaCl stress condition, the 6% biochar treatment decreased RB and RWC by 1% and 2% relative to the non-biochar treatment, respectively (Fig. 1B and Fig. 2B). Among all treatments, the 3% biochar treatment under non-saline conditions exhibited the highest values for SB, RB, TB, total Chl content, and RWC. In contrast, the lowest values for these parameters were observed in the non-biochar treatment under the 9 dS m−1 NaCl stress condition.
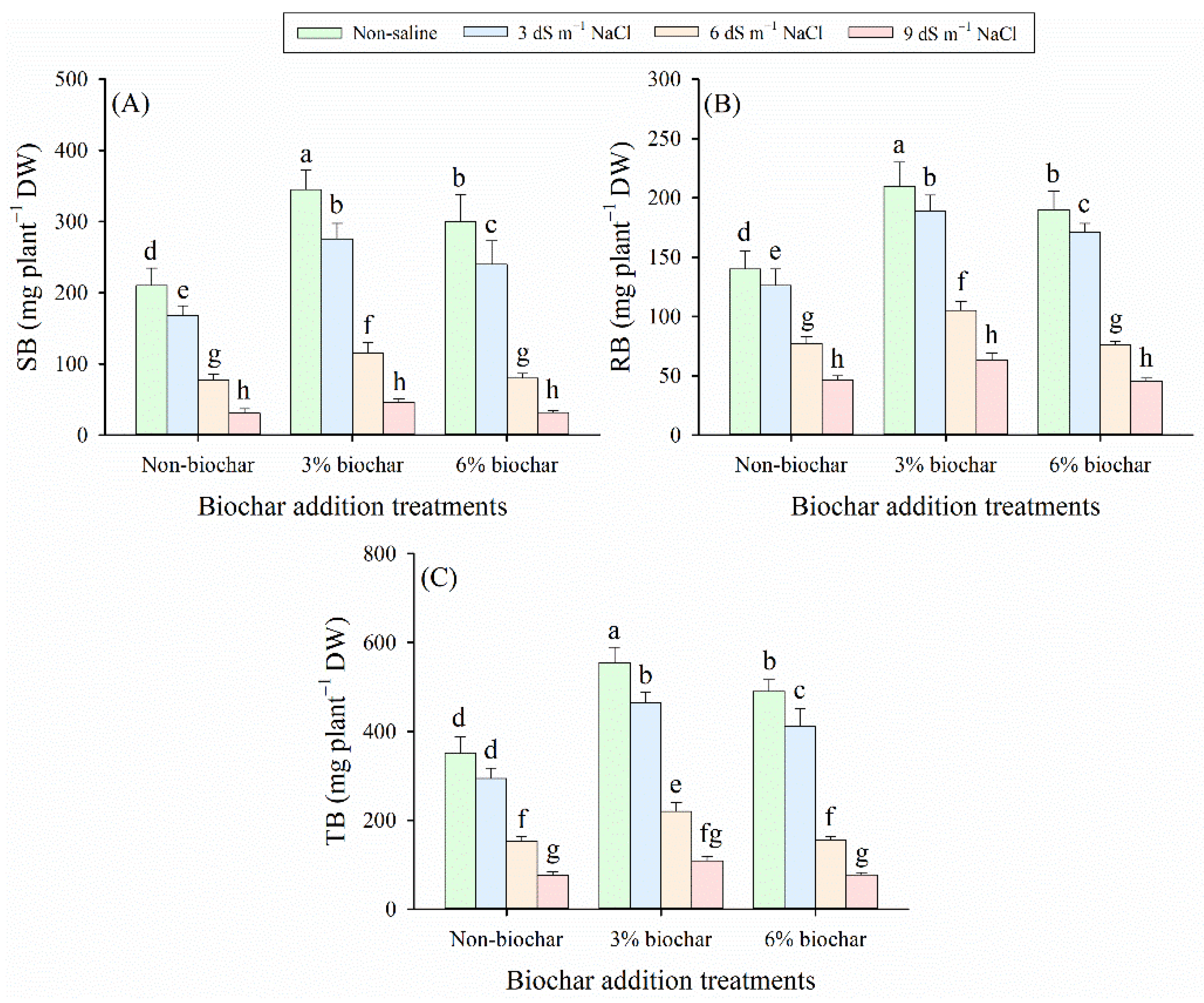
Figure 1: (A) Shoot biomass (SB), (B) root biomass (RB), and (C) total biomass (TB) in alfalfa plants exposed to diverse salinity levels and biochar application regimes. Distinct lowercase letters represent statistically significant variations across all treatment combinations (p < 0.05). Salt stress treatments included NaCl concentrations of 0 dS m−1 (Non-saline), 3 dS m−1 (3 dS m−1 NaCl), 6 dS m−1 (6 dS m−1 NaCl), and 9 dS m−1 (9 dS m−1 NaCl). Biochar amendment treatments included application regimes of 0 g kg−1 (Non-biochar), 30 g kg−1 (3% biochar), and 60 g kg−1 (6% biochar). This treatment notation applies to all subsequent figures.
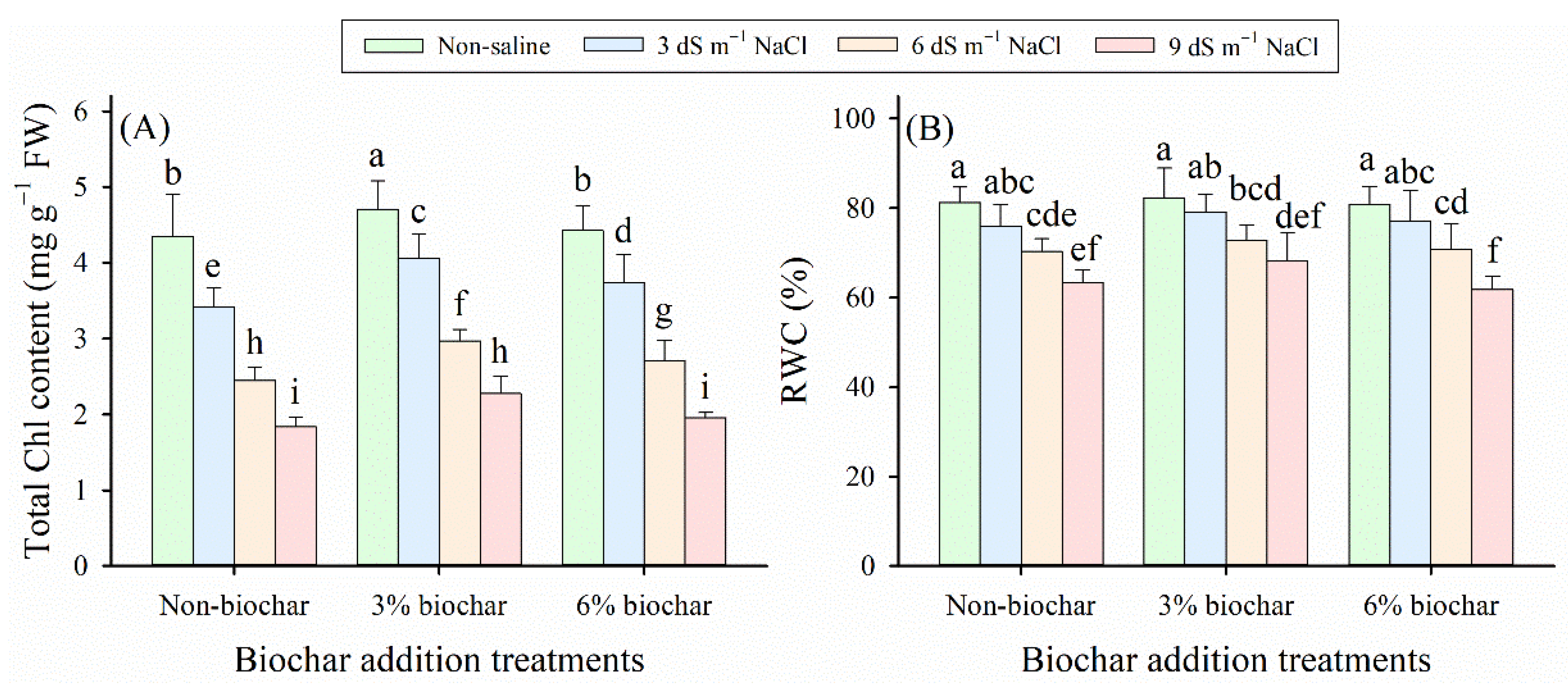
Figure 2: (A) Total chlorophyll (Chl) content and (B) relative water content (RWC) in alfalfa foliage subjected to different salinity levels and biochar application doses. Distinct lowercase letters represent statistically significant variations across all treatment combinations (p < 0.05).
3.2 Effects of Biochar Application on the Concentrations of Pro, GB, SS, and SP in Alfalfa Leaves under Salt Stress
Salinity levels, biochar application, and their interaction significantly affected the concentrations of proline (Pro), glycine betaine (GB), soluble sugar (SS), and soluble protein (SP) in alfalfa leaves (p < 0.05, Table 2), whereas no significant interactive effect was observed between salt stress and biochar amendment on SP content (p > 0.05, Table 2). Specifically, with the intensification of salt stress, the concentrations of Pro, GB, SS, and SP in alfalfa leaves gradually increased (Fig. 3). Notably, biochar application alleviated the salt-induced accumulation of these osmotically active solutes and reduced their levels. Under the same salt stress level, the 3% biochar treatment resulted in significantly lower concentrations of these osmotically active solutes compared with the non-biochar and 6% biochar treatments (p < 0.05). At all salinity levels, compared with non-biochar controls, 3% biochar treatment reduced the concentrations of Pro, GB, SS, and SP by 12%–20%, 12%–36%, 18%–24%, and 7%–12%, respectively. Among all treatments, the highest concentrations of Pro, GB, SS, and SP were detected in the 6% biochar treatment under 9 dS m−1 NaCl stress condition, while the lowest concentrations were recorded in the 3% biochar treatment under non-saline conditions. This further confirmed the dose-dependent regulatory role of biochar in mediating alfalfa’s osmotic adjustment response under salt stress.
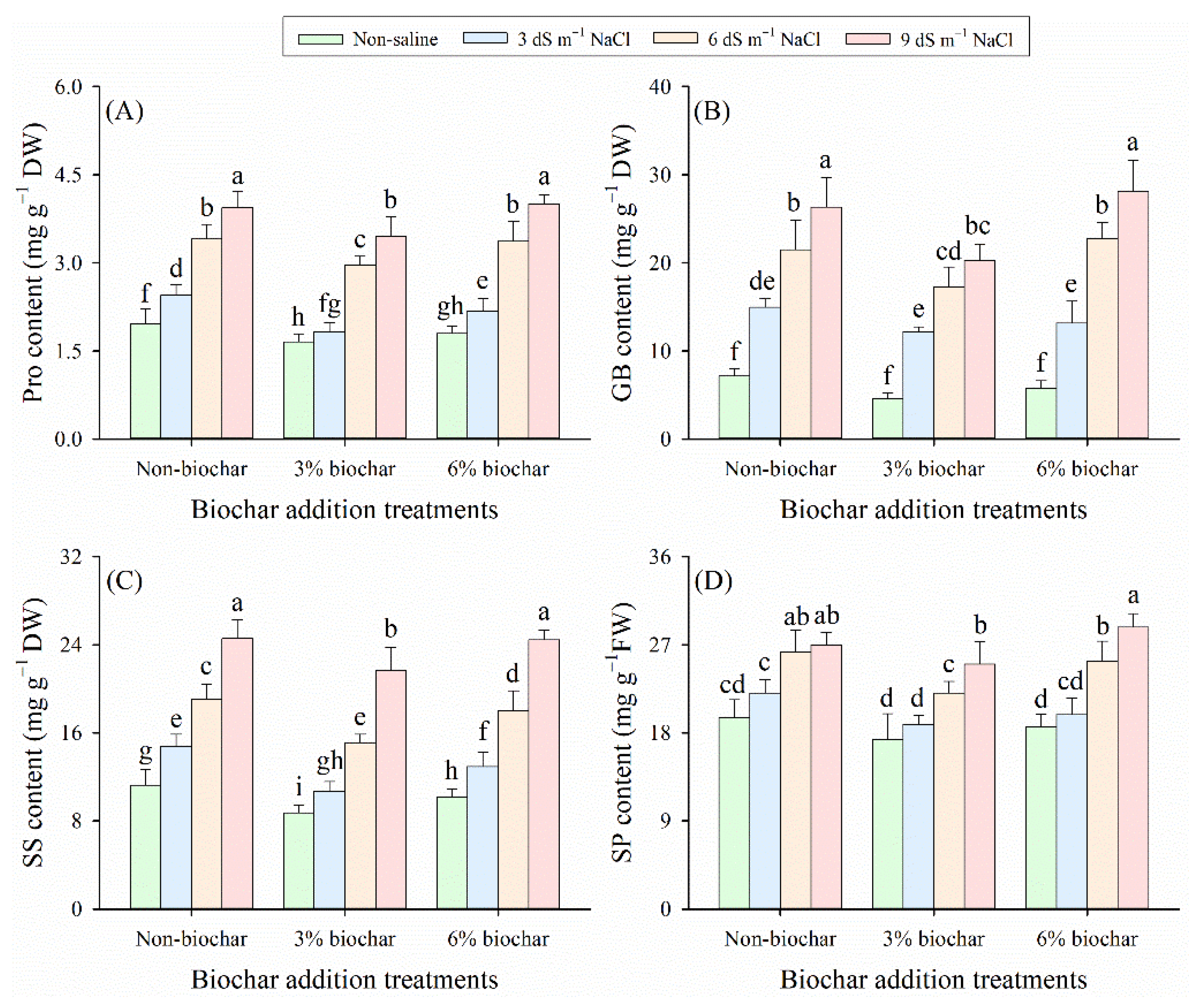
Figure 3: (A) Proline (Pro), (B) glycine betaine (GB), (C) soluble sugar (SS), and (D) soluble protein (SP) contents in alfalfa foliage under different saline conditions and biochar application rates. Distinct lowercase letters represent statistically significant variations across all treatment combinations (p < 0.05).
3.3 Effects of Biochar Addition on the Activities CAT, POD, SOD, APX and PPO in Alfalfa Leaves under Salt Stress
Two-way ANOVA verified that salinity level and biochar addition exerted highly significant effects on the antioxidant enzyme profiles including CAT, POD, SOD, APX and PPO in plant leaves (p < 0.01, Table 2). Specifically, salt stress and biochar amendment had significant interactive effects on the activities of CAT, SOD, and APX (p < 0.01), while no significant interactive effect was observed on the activities of POD and PPO (p > 0.05; Table 2). Under all biochar application regimes, salinity significantly increased the activities of all tested antioxidant enzymes (Fig. 4). Among the biochar treatments, the 3% biochar amendment resulted in the lowest CAT, POD, SOD, APX, and PPO activities under the same salt stress level. Notably, the minimum values of all these enzyme activities were observed in the 3% biochar treatment under non-saline conditions. Specifically, under 9 dS m−1 NaCl stress condition, the 3% biochar treatment reduced CAT, POD, SOD, APX, and PPO activities by 19%, 12%, 24%, 18%, and 2%, respectively, compared with the non-biochar control. However, the 6% biochar treatment increased the activities of CAT, POD, APX, and PPO by 12%, 4%, 7%, and 16% under the same severe salt stress condition, which indicates that excessive biochar application weakened the mitigating effect on salt-induced disorders of antioxidant enzyme activities, thereby exerting a negative regulatory role.
Table 2: Results of two-way analysis of variance (ANOVA) showing F-values and p-values for the effects of salt stress (S), biochar amendment (B), and their interaction (S × B) on the growth, physiological, and biochemical indicators of alfalfa seedlings.
| Salt Stress (S) | Biochar (B) | S × B | ||||
|---|---|---|---|---|---|---|
| F | p | F | p | F | p | |
| SB | 392.96 | <0.001*** | 53.64 | <0.001*** | 8.94 | <0.001*** |
| RB | 368.18 | <0.001*** | 65.01 | <0.001*** | 8.16 | <0.001*** |
| TB | 665.50 | <0.001*** | 99.81 | <0.001*** | 14.82 | <0.001*** |
| Total Chl | 635.27 | <0.001*** | 43.31 | <0.001*** | 0.84 | 0.548n.s. |
| RWC | 29.56 | <0.001*** | 2.01 | 0.140n.s. | 0.29 | 0.940n.s. |
| Pro | 816.41 | <0.001*** | 76.32 | <0.001*** | 3.60 | 0.007** |
| GB | 177.32 | <0.001*** | 17.16 | <0.001*** | 2.37 | 0.049* |
| SS | 965.21 | <0.001*** | 109.93 | <0.001*** | 2.40 | 0.047* |
| SP | 57.76 | <0.001*** | 12.14 | <0.001*** | 1.23 | 0.315n.s. |
| CAT | 731.20 | <0.001*** | 174.39 | <0.001*** | 11.98 | <0.001*** |
| POD | 210.36 | <0.001*** | 9.29 | 0.001** | 0.64 | 0.695n.s. |
| SOD | 1184.64 | <0.001*** | 227.59 | <0.001*** | 7.49 | <0.001*** |
| APX | 1577.24 | <0.001*** | 149.00 | <0.001*** | 6.50 | <0.001*** |
| PPO | 132.06 | <0.001*** | 15.54 | <0.001*** | 1.30 | 0.281n.s. |
| H2O2 | 682.45 | <0.001*** | 102.60 | <0.001*** | 5.92 | <0.001*** |
| O2•− | 43.25 | <0.001*** | 9.79 | <0.001*** | 0.66 | 0.685n.s. |
| MDA | 582.85 | <0.001*** | 204.47 | <0.001*** | 4.63 | 0.001** |
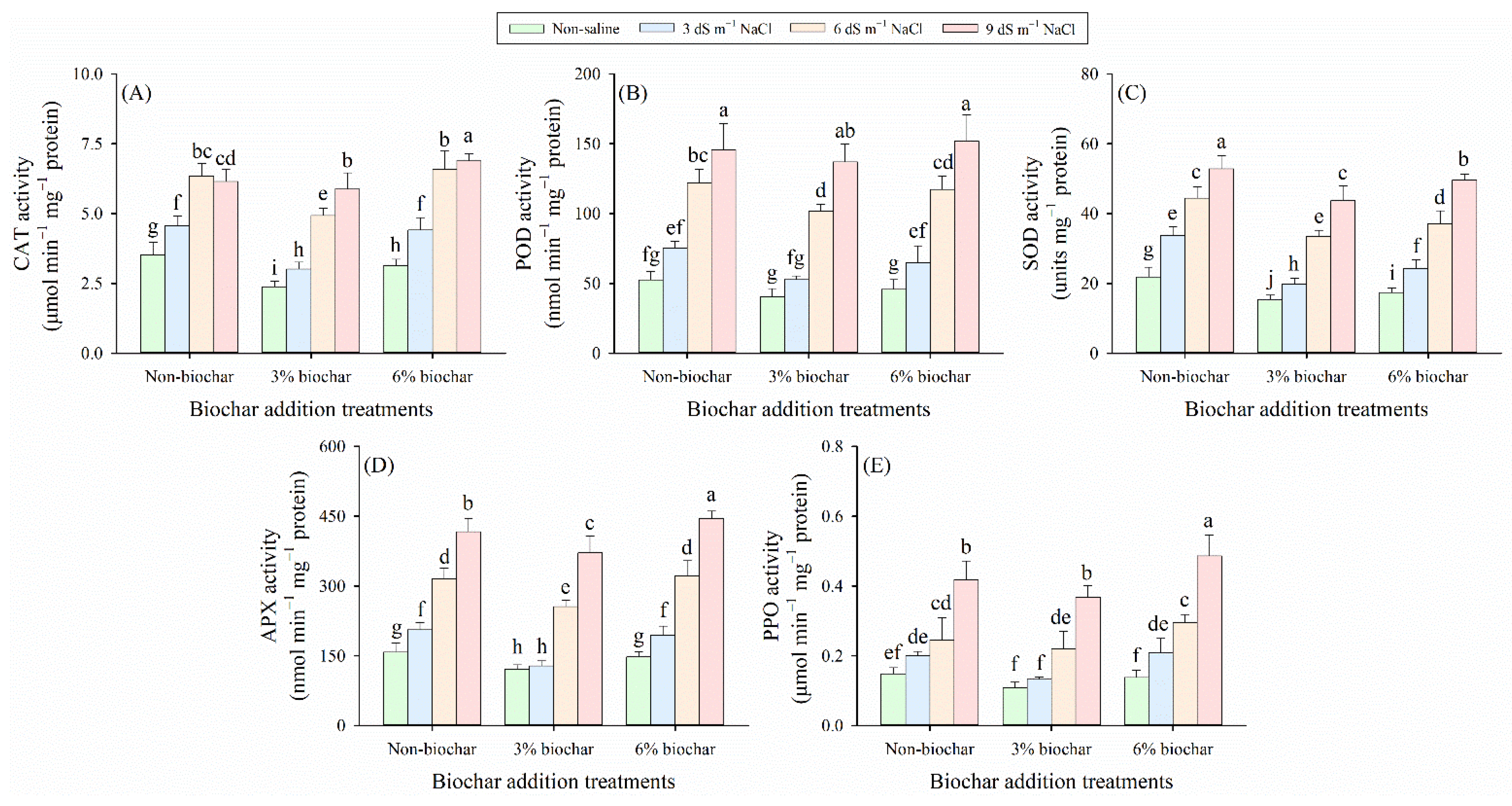
Figure 4: (A) Catalase (CAT) activity, (B) peroxidase (POD) activity, (C) superoxide dismutase (SOD) activity, (D) ascorbate peroxidase (APX) activity, and (E) polyphenol oxidase (PPO) activity in alfalfa foliage under different saline levels and biochar application rates. Distinct lowercase letters represent statistically significant variations across all treatment combinations (p < 0.05).
3.4 Effects of Biochar Addition on the Contents of H2O2, O2•−, and MDA in Alfalfa Leaves under Salt Stress
H2O2, O2•−, and MDA contents were significantly elevated with the intensification of salt stress, in contrast, biochar application lowered their concentrations at the identical salt stress level (p < 0.05, Fig. 5). Salt stress, biochar amendment, and their interaction exerted a significant effect on the concentrations of H2O2, O2•−, and MDA in alfalfa leaves (p < 0.05, Table 2). However, no significant interactive effect between salt stress and biochar amendment was detected on O2•− content (p > 0.05, Table 2). Specifically, under non-saline conditions, the contents of H2O2, O2•−, and MDA treated with 3% biochar were the lowest, which were 23%, 12%, and 32% lower than the non-biochar group, respectively (p < 0.05). On the contrary, under 9 dS m−1 NaCl stress conditions, the concentrations of H2O2 and O2•− were the highest in 6% biochar treatment, showing a significant increase of 85% and 63% in non-saline conditions compared with non-biochar controls. In contrast, the highest MDA concentration was found in the non-biochar treatment under 9 dS m−1 NaCl stress condition, a significant 76% increase compared to the non-biochar control under non-saline conditions. Overall, based on the average of all treatments, 3% biochar treatment exhibited the lowest oxidative stress solute concentrations among all biochar treatments, which further confirmed its best mitigating effect on salt-induced oxidative damage in alfalfa.
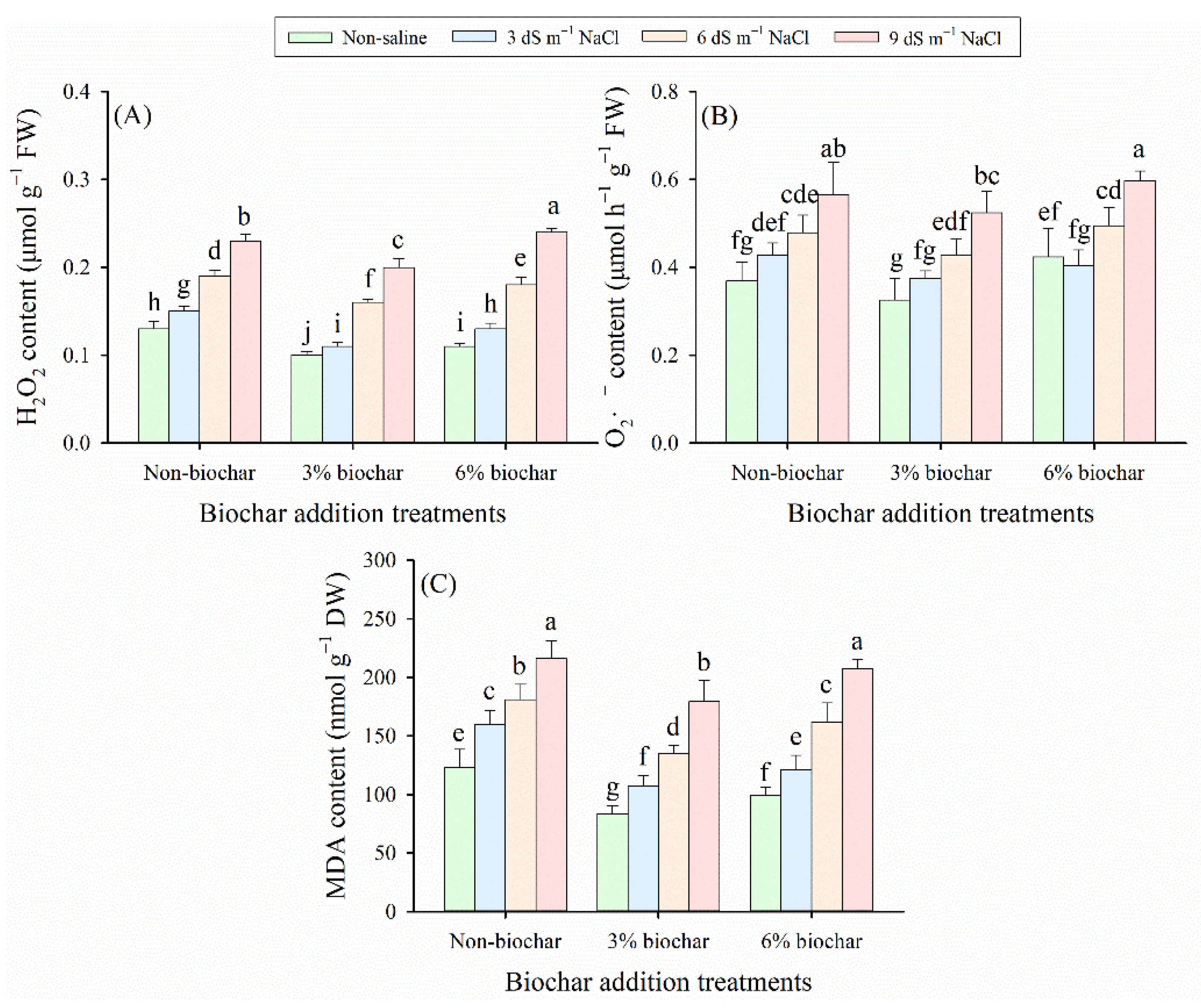
Figure 5: (A) Hydrogen peroxide (H2O2), (B) superoxide anion radical (O2•−), and (C) malondialdehyde (MDA) contents in alfalfa leaves exposed to different salt stress levels and biochar application rates. Distinct lowercase letters represent statistically significant variations across all treatment combinations (p < 0.05).
3.5 The Correlation between Different Growth, Physiological, and Biochemical Indicators
We explored the effect of biochar on alfalfa growth, analyzed the correlations between its growth, physiological, and biochemical indicators (Fig. 6). The TB was significantly positively correlated with both total Chl content and RWC, but negatively correlated with osmotic adjustment substances (Pro, GB, SS, and SP), antioxidant enzyme activities (CAT, POD, SOD, APX, and PPO), and oxidative stress-related substances (H2O2, O2•−, and MDA). Furthermore, total Chl content was positively correlated with RWC and negatively correlated with oxidative stress-related substances. Osmotic adjustment substances and antioxidant enzyme activities were positively correlated with oxidative stress-related substances. Based on these analyses, our results indicated that biochar amendment could enhance RWC and reduce ROS accumulation in alfalfa seedlings under salt stress by regulating the osmotic adjustment and antioxidant enzyme systems.
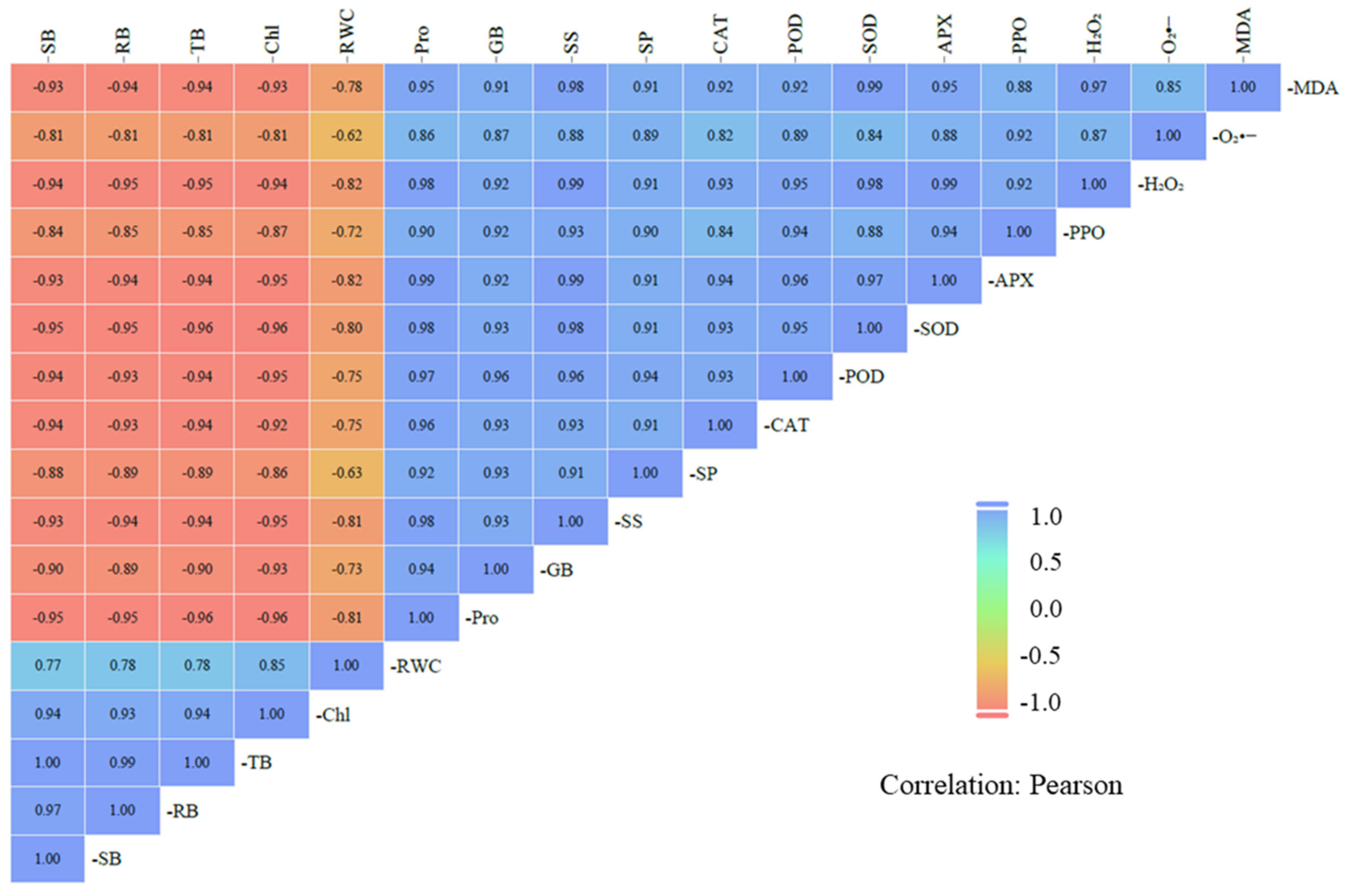
Figure 6: Correlation plots between all measured variables in the experiment. Colour intensity represents the absolute value of the Pearson correlation coefficient.
Salinity stress is one of the primary abiotic stressors threatening global food security [50]. It impairs plant water uptake by establishing an osmotic gradient between the rhizosphere and plant cells, disrupts Na+/K+ homeostasis, perturbs plant physiological and metabolic processes, and ultimately inhibits plant growth, thus culminating in compromised crop yield and quality [17]. Our findings showed that total Chl content in alfalfa leaves decreased significantly with increasing salt stress intensity (Fig. 2A). Consistently, Jabborova et al. [35] also confirmed that total Chl and carotenoid contents in alfalfa were significantly reduced under salt stress conditions. This phenomenon has been widely documented across diverse plant species: Shaheen et al. [51] and He et al. [52] observed significant reductions in Chl content of wheat and maize, respectively, with escalating salt stress, which aligns with the present study's findings. Alfalfa adopted a defensive strategy against salt stress by upregulating the accumulation of osmotic adjustment substances, including leaf Pro, GB, SS, and SP in alfalfa leaves (Fig. 3). Such accumulation increases intracellular solute concentration and reduces cellular water potential, thereby maintaining osmotic balance and preventing irreversible damage induced by excessive cellular dehydration [22]. Despite this adaptive response, the RWC of alfalfa leaves still decreased significantly under salt stress in our research (Fig. 2B). The reduction in alfalfa Chl content is closely associated with two key factors: the decline in leaf water content triggered by osmotic stress and excessive production and accumulation of ROS under salt stress [4]. Relevant studies have corroborated that ROS overaccumulation is typically induced by water and ionic imbalances caused by salt stress [53]. Although our findings confirmed that alfalfa could activate its enzymatic antioxidant system as a defensive strategy by increasing the activities of CAT, POD, SOD, APX, and PPO to scavenge ROS (Fig. 4), the contents of H2O2 and O2•− exhibited a substantial increase (Fig. 5A,B), which further exacerbated chlorophyll degradation (Fig. 2A). Collectively, our findings confirm that the salinity-induced reduction in alfalfa chlorophyll content results from the synergistic effects of decreased leaf RWC (Fig. 2B) and excessive ROS accumulation (Fig. 5). However, the dominant contributor to chlorophyll reduction in salt-stressed alfalfa—whether osmotic imbalance or ROS-mediated damage—requires further clarification in follow-up research.
Notably, our study showed that biochar application significantly increased leaf Chl content (Fig. 2A) and significantly enhanced alfalfa’s shoot, root, and total biomass (Fig. 1), a finding that is consistent with previous studies [29,35]. As a core component of plant photosynthesis, elevated Chl content can enhance photosynthetic capacity, providing material and energy supplies for plant growth and development, thereby promoting biomass accumulation [9,16]. Notably, an interesting observation was that biochar-treated alfalfa showed significantly higher leaf RWC but lower contents of osmotic adjustment substances (Pro, GB, SS, and SP) than the control group (non-biochar) (Fig. 2B, Fig. 3 and Fig. 6). Under salt stress, alfalfa accumulates osmotic adjustment substances to lower osmotic potential and avoid dehydration [19]. The reduced demand for osmotic adjustment following biochar application indicates biochar improves soil water conditions to alleviate salt-induced physiological drought and reduce plant dependence on these substances [28,54]. The underlying mechanism is primarily attributed to biochar's distinctive physicochemical properties, where its large specific surface area and well-developed porous structure enhance the water retention capacity of saline soil and optimize the rhizosphere microdomain, thereby promoting plant water uptake and alleviating physiological drought [9]. Meanwhile, biochar application decreased the activities of antioxidant enzymes (CAT, POD, SOD, APX, and PPO) under salt stress (Fig. 4). Biochar is speculated to reduce ROS generation and accumulation by improving rhizosphere water status and regulating ionic absorption balance, thus diminishing the need for enzymatic antioxidant system activation and alleviating ROS-induced metabolic damage [32,55]. In addition, biochar is rich in plant-available mineral nutrients, and its application can optimize soil nutrient supply to support alfalfa growth—consistent with previous studies [31,54]. Furthermore, the porous structure of biochar improves the rhizosphere microenvironment, promotes root morphological development and function, and stimulates plant-microbe interactions, these effects synergistically improve alfalfa’s nutrient and water acquisition capacity under salt stress, ultimately promoting plant salt stress tolerance and growth performance [55].
A significant finding of this study was the marked capacity of biochar to mitigate the deleterious effects of salt stress on alfalfa (Fig. 1B, Fig. 2B, Fig. 3, Fig. 4A,B,D,E and Fig. 5). Moderate biochar application can remarkably optimize plant osmotic adjustment capacity, maximize the defensive function of the enzymatic antioxidant system, and effectively alleviate salt stress damage [54]. In contrast, high-rate biochar application inversely attenuates these positive effects [56]. This result aligns well with the well-documented finding that biochar exerts a concentration threshold-dependent regulatory effect on plants, thus further confirming that an optimal application rate is critical for maximizing biochar’s remediation efficacy in saline soils [9]. Regarding the mechanism underlying the adverse effects of high-rate biochar application, this study proposes that biochar produced via pyrolysis at high temperature (e.g., >500°C) exhibits strong alkalinity [30]. Excessive application tends to cause a significant increase in rhizosphere microdomain pH, disrupting the original soil acid-base homeostasis. Consequently, the diminished bioavailability of essential mineral nutrients, including phosphorus, zinc, and iron, which inhibits their uptake and translocation by alfalfa roots, is coupled with alterations in soil wettability and pore architecture that induces heterogeneous rhizosphere water distribution, ultimately impairing root water absorption [57]. The combined deficiency of nutrient and water supply synergistically inhibit the synthesis and accumulation of photosynthetic pigments such as chlorophyll, impairs plant photosynthetic efficiency, and decreases photosynthate accumulation, ultimately leading to reduced biomass [5]. Additionally, the strong adsorption capacity of high-rate biochar may excessively immobilize soil available ions, further disrupting ionic homeostasis (e.g., Na+/K+ balance) in plants and exacerbating salt stress-induced cellular damage [56]. The conclusions of this investigation provide significant practical advice concerning the wide-ranging implementation of biochar in the context of forage production in saline-alkaline environments. In practice, to amend saline-alkaline soils and enhance alfalfa productivity with biochar, the application rate should be precisely adjusted according to soil salinization degree and biochar pyrolysis characteristics to determine the optimal threshold and avoid adverse effects from excessive application [58]. This approach not only ensures the practical application efficacy of biochar amendment technology but also reduces biochar resource waste and production costs, significantly enhancing its promotion value and economic viability in saline-alkaline forage cultivation, while improving the economic and practical efficiency of its field application.
The present study was conducted via pot experiments under controlled greenhouse conditions, and it may not fully align with those prevailing in actual field production systems. Nevertheless, through the precise control of experimental variables and sufficient experimental replication, this study ensured the reliability and reproducibility of the results. Consequently, this provided a substantial theoretical and experimental foundation for the extrapolation of these conclusions to actual field production. In fact, the conclusion that optimal biochar application rates can effectively alleviate salt stress and enhance crop yield has been widely validated in numerous field trials conducted on saline-alkali soils [32,56,59]. The present study further provides a clear mechanistic understanding into such regulatory effects at the physiological and biochemical levels of both the osmotic adjustment and antioxidant enzyme systems, thereby deepening the mechanistic understanding of biochar-mediated amelioration in saline-alkali soils. However, we should also acknowledge that environmental conditions (e.g., climate, soil texture, and pH) differ substantially across regions, which is a key limitation of the present study. Furthermore, the physicochemical properties of biochar are dependent on factors such as the type of feedstock and the conditions under which the pyrolysis process occurs [26,30]. In addition, different crop species and even distinct genotypes within the same crop may exhibit different responses to biochar application [16,19]. Therefore, long-term multi-year in situ field positioning trials should be the focus of future research. Through the integration of regional environmental characteristics, biochar physicochemical properties, and crop genotypic differences, researchers should establish and optimize a sustainable biochar application strategy—encompassing optimal application rates, application timings, and combined application methods. This regime holds great potential to markedly enhance the saline-alkaline stress resistance of forage crops, enhancing forage productivity in saline-alkali soils. It also provides a solid technical foundation for restoring saline-alkali lands and efficiently using forage cultivation in such areas.
The experimental findings of this study confirmed that salt stress significantly reduced the RWC and induced a marked accumulation of ROS in alfalfa leaves, along with a pronounced decline in leaf chlorophyll content in alfalfa, ultimately resulting in severe inhibition of plant growth. Our results revealed that alfalfa could partially alleviate these adverse effects by increasing antioxidant enzyme activities and activating osmotic adjustment mechanisms. However, increasing salt stress intensity further promoted the accumulation of ROS (i.e., H2O2 and O2•−) and MDA, which intensified oxidative damage, reduced photosynthetic pigment content, and ultimately impeded the biomass accumulation of alfalfa seedlings. Biochar amendment has been demonstrated to effectively alleviate salt stress in alfalfa by maintaining the stability of the leaf antioxidant enzyme and osmotic adjustment systems. Furthermore, biochar application promoted the synthesis of photosynthetic pigments and photosynthates, thereby enhancing biomass accumulation, with this mitigative effect most pronounced at an application rate (30 g kg−1). Nevertheless, under severe salt stress levels (9 dS m−1), a higher biochar rate (60 g kg−1) did not promote alfalfa growth, potentially because of osmotic stress induced by excessive application. Therefore, appropriate biochar application is essential for mitigating salt toxicity and promoting alfalfa growth in saline soils. Against the backdrop of the pressing demand for environmentally friendly and sustainable approaches to addressing soil salinization, biochar application is a promising agricultural practice to alleviate moderate oxidative stress and improve alfalfa yield, offering theoretical and practical guidance for the scientific and rational use of biochar in saline-alkali soil restoration and sustainable forage production.
Acknowledgement:
Funding Statement: This research was financially supported by National Natural Science Foundation of China (32301496), Shandong Provincial Natural Science Foundation (ZR2022QC174). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author Contributions: The authors confirm contribution to the paper as follows: study conception and design: Shangzhi Zhong, Lulu Sun; data acquisition and curation: Jianghan Ouyang, Yuxin Zhang; experimental assistance: Xiaotong Ma, Xiangjian Tu, Pengxin Hou, Wei Cao; formal analysis: Jianghan Ouyang, Xiaotong Ma; funding acquisition: Shangzhi Zhong, Lulu Sun; experimental supervision and evaluation: Shangzhi Zhong, Lulu Sun; draft manuscript preparation: Jianghan Ouyang, Yuxin Zhang, Shangzhi Zhong, Lulu Sun; manuscript editing and revisions: all authors. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The authors confirm that the data supporting the findings of this study are available within the article.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. FAO . The global status of salt-affected soils. Rome, Italy: FAO; 2024 [cited 2026 Jan 10]. Available from: https://www.fao.org/events/detail/soil-water-forum-2024/en. [Google Scholar]
2. FAO . Global map of salt-affected soils (GSASmap). Rome, Italy: FAO; 2021 [cited 2026 Jan 10]. Available from: https://www.fao.org/documents/card/en/c/cb7247en. [Google Scholar]
3. van Zelm E , Zhang Y , Testerink C . Salt tolerance mechanisms of plants. Annu Rev Plant Biol. 2020; 71: 403– 33. doi:10.1146/annurev-arplant-050718-100005. [Google Scholar] [CrossRef]
4. Acosta-Motos J , Ortuño M , Bernal-Vicente A , Diaz-Vivancos P , Sanchez-Blanco M , Hernandez J . Plant responses to salt stress: adaptive mechanisms. Agronomy. 2017; 7( 1): 18. doi:10.3390/agronomy7010018. [Google Scholar] [CrossRef]
5. Choudhury FK , Rivero RM , Blumwald E , Mittler R . Reactive oxygen species, abiotic stress and stress combination. Plant J. 2017; 90( 5): 856– 67. doi:10.1111/tpj.13299. [Google Scholar] [CrossRef]
6. Kamran M , Wang D , Xie K , Lu Y , Shi C , El Sabagh A , et al. Pre-sowing seed treatment with kinetin and calcium mitigates salt induced inhibition of seed germination and seedling growth of choysum (Brassica rapa var. parachinensis). Ecotoxicol Environ Saf. 2021; 227: 112921. doi:10.1016/j.ecoenv.2021.112921. [Google Scholar] [CrossRef]
7. Chen Z , Wang Q . Graphene ameliorates saline-alkaline stress-induced damage and improves growth and tolerance in alfalfa (Medicago sativa L.). Plant Physiol Biochem. 2021; 163: 128– 38. doi:10.1016/j.plaphy.2021.03.039. [Google Scholar] [CrossRef]
8. Shi S , Tian L , Nasir F , Bahadur A , Batool A , Luo S , et al. Response of microbial communities and enzyme activities to amendments in saline-alkaline soils. Appl Soil Ecol. 2019; 135: 16– 24. doi:10.1016/j.apsoil.2018.11.003. [Google Scholar] [CrossRef]
9. Zhong S , Hou P , Zheng C , Yang X , Tao Q , Sun J . Wheat straw biochar amendment increases salinity stress tolerance in alfalfa seedlings by modulating physiological and biochemical responses. Plants. 2025; 14( 13): 1954. doi:10.3390/plants14131954. [Google Scholar] [CrossRef]
10. Zhang H , Zhu J , Gong Z , Zhu JK . Abiotic stress responses in plants. Nat Rev Genet. 2022; 23( 2): 104– 19. doi:10.1038/s41576-021-00413-0. [Google Scholar] [CrossRef]
11. Bertrand A , Gatzke C , Bipfubusa M , Lévesque V , Chalifour FP , Claessens A , et al. Physiological and biochemical responses to salt stress of alfalfa populations selected for salinity tolerance and grown in symbiosis with salt-tolerant Rhizobium. Agronomy. 2020; 10( 4): 569. doi:10.3390/agronomy10040569. [Google Scholar] [CrossRef]
12. Hou C , Li X , Tian D , Xu B , Zhang C , Ren J , et al. Evaluation of the effects of water and salinity stress on the growth and biochemistry of alfalfa (Medicago sativa L.) at the branching stage. Sustainability. 2022; 14( 16): 10262. doi:10.3390/su141610262. [Google Scholar] [CrossRef]
13. Zhou H , Shi H , Yang Y , Feng X , Chen X , Xiao F , et al. Insights into plant salt stress signaling and tolerance. J Genet Genom. 2024; 51( 1): 16– 34. doi:10.1016/j.jgg.2023.08.007. [Google Scholar] [CrossRef]
14. El-Shal RM , El-Naggar AH , El-Beshbeshy TR , Mahmoud EK , El-Kader NIA , Missaui AM , et al. Effect of nano-fertilizers on alfalfa plants grown under different salt stresses in hydroponic system. Agriculture. 2022; 12( 8): 1113. doi:10.3390/agriculture12081113. [Google Scholar] [CrossRef]
15. Wang WB , Kim YH , Lee HS , Kim KY , Deng XP , Kwak SS . Analysis of antioxidant enzyme activity during germination of alfalfa under salt and drought stresses. Plant Physiol Biochem. 2009; 47( 7): 570– 7. doi:10.1016/j.plaphy.2009.02.009. [Google Scholar] [CrossRef]
16. Feng Y , Chen Z , Chen L , Han M , Liu J , Liu Y , et al. Comprehensive evaluation of physio-morphological traits of alfalfa (Medicago sativa L.) varieties under salt stress. Physiol Plant. 2025; 177( 1): e70044. doi:10.1111/ppl.70044. [Google Scholar] [CrossRef]
17. Yuan J , Cao H , Qin W , Yang S , Zhang D , Zhu L , et al. Genomic and modern biotechnological strategies for enhancing salt tolerance in crops. New Crops. 2025; 2: 100057. doi:10.1016/j.ncrops.2024.100057. [Google Scholar] [CrossRef]
18. Urbanavičiūtė I , Bonfiglioli L , Pagnotta MA . One hundred candidate genes and their roles in drought and salt tolerance in wheat. Int J Mol Sci. 2021; 22( 12): 6378. doi:10.3390/ijms22126378. [Google Scholar] [CrossRef]
19. Al-Farsi SM , Al-Sadi AM , Ullah A , Farooq M . Salt tolerance in alfalfa landraces of Omani origin: morpho-biochemical, mineral, and genetic diversity assessment. J Soil Sci Plant Nutr. 2021; 21( 2): 1484– 99. doi:10.1007/s42729-021-00455-7. [Google Scholar] [CrossRef]
20. Tlahig S , Bellani L , Karmous I , Barbieri F , Loumerem M , Muccifora S . Response to salinity in legume species: an insight on the effects of salt stress during seed germination and seedling growth. Chem Biodivers. 2021; 18( 4): e2000917. doi:10.1002/cbdv.202000917. [Google Scholar] [CrossRef]
21. Babakhani B , Khavari-Nejad R , Hassan Sajedi R , Fahimi H , Saadatmand S . Biochemical responses of Alfalfa (Medicago sativa L.) cultivars subjected to NaCl salinity stress. Afr J Biotechnol. 2013; 10( 55): 11433– 41. [Google Scholar]
22. Singh P , Choudhary KK , Chaudhary N , Gupta S , Sahu M , Tejaswini B , et al. Salt stress resilience in plants mediated through osmolyte accumulation and its crosstalk mechanism with phytohormones. Front Plant Sci. 2022; 13: 1006617. doi:10.3389/fpls.2022.1006617. [Google Scholar] [CrossRef]
23. Liu X , Elzenga JTM , Venema JH , Tiedge KJ . Thriving in a salty future: morpho-anatomical, physiological and molecular adaptations to salt stress in alfalfa (Medicago sativa L.) and other crops. Ann Bot. 2024; 134( 7): 1113– 30. doi:10.1093/aob/mcae152. [Google Scholar] [CrossRef]
24. Lu Q , Ge G , Sa D , Wang Z , Hou M , Jia YS . Effects of salt stress levels on nutritional quality and microorganisms of alfalfa-influenced soil. PeerJ. 2021; 9: e11729. doi:10.7717/peerj.11729. [Google Scholar] [CrossRef]
25. Guo K , Xu Z , Huo Y , Sun Q , Wang Y , Che Y , et al. Effects of salt concentration, pH, and their interaction on plant growth, nutrient uptake, and photochemistry of alfalfa (Medicago sativa) leaves. Plant Signal Behav. 2020; 15( 12): 1832373. doi:10.1080/15592324.2020.1832373. [Google Scholar] [CrossRef]
26. Fakhar A , Galgo SJC , Canatoy RC , Rafique M , Sarfraz R , Farooque AA , et al. Advancing modified biochar for sustainable agriculture: a comprehensive review on characterization, analysis, and soil performance. Biochar. 2025; 7( 1): 8. doi:10.1007/s42773-024-00397-0. [Google Scholar] [CrossRef]
27. Mihoub A , Abu Zied Amin AE , Motaghian HR , Saeed MF , Naeem A . Citric acid (CA)—modified biochar improved available phosphorus concentration and its half-life in a P-fertilized calcareous sandy soil. J Soil Sci Plant Nutr. 2022; 22( 1): 465– 74. doi:10.1007/s42729-021-00662-2. [Google Scholar] [CrossRef]
28. Mihoub A , Mesnoua M , Touzout N , Zeguerrou R , Siabdallah N , Benchikh C , et al. Mitigation of detrimental effects of salinity on sweet pepper through biochar-based fertilizers derived from date palm wastes. Phyton. 2024; 93( 11): 2993– 3011. doi:10.32604/phyton.2024.057536. [Google Scholar] [CrossRef]
29. Su B , Tu Z , Yang Z , Liu N , Bai Z , Deng Z , et al. Enterobacter hormaecheiWu15-loaded biochar enhances the ice plant growth by improving saline soil quality. Plant Soil. 2025; 511( 1): 1503– 15. doi:10.1007/s11104-024-07063-1. [Google Scholar] [CrossRef]
30. Muhammad M , Nazim M , Ghafoor A , Hussain A , Ali M , Tabassum M , et al. Biochar as a climate-smart agricultural practice: reducing greenhouse gas emissions and promoting sustainable farming. Phyton. 2025; 94( 1): 65– 99. doi:10.32604/phyton.2025.058970. [Google Scholar] [CrossRef]
31. Liu M , Zhao Z , Chen L , Wang L , Ji L , Xiao Y . Influences of arbuscular mycorrhizae, phosphorus fertiliser and biochar on alfalfa growth, nutrient status and cadmium uptake. Ecotoxicol Environ Saf. 2020; 196: 110537. doi:10.1016/j.ecoenv.2020.110537. [Google Scholar] [CrossRef]
32. Liu J , Xiong H , Huang S , Li Y , Li C , Li Q , et al. Impact of subsurface drainage and biochar amendment on the coastal soil–plant system: a case study in alfalfa cultivation on saline–alkaline soil. Water. 2025; 17( 10): 1415. doi:10.3390/w17101415. [Google Scholar] [CrossRef]
33. Zhong S , Zhang X , Hou P , Ouyang J , Rakotoson T , Zheng C , et al. Biochar amendment enhances water use efficiency in alfalfa (Medicago sativa L.) under partial root-zone drying irrigation by modulating abscisic acid signaling and photosynthetic performance. Environ Exp Bot. 2025; 238: 106244. doi:10.1016/j.envexpbot.2025.106244. [Google Scholar] [CrossRef]
34. Zhang M , Wang J , Bai SH , Zhang Y , Teng Y , Xu Z . Assisted phytoremediation of a co-contaminated soil with biochar amendment: contaminant removals and bacterial community properties. Geoderma. 2019; 348: 115– 23. doi:10.1016/j.geoderma.2019.04.031. [Google Scholar] [CrossRef]
35. Jabborova D , Abdrakhmanov T , Jabbarov Z , Abdullaev S , Azimov A , Mohamed I , et al. Biochar improves the growth and physiological traits of alfalfa, amaranth and maize grown under salt stress. PeerJ. 2023; 11: e15684. doi:10.7717/peerj.15684. [Google Scholar] [CrossRef]
36. Zhong SZ , Liu XJ , Ouyang JH , Tu XJ , Song WZ , Cao W , et al. Effects of biochar and phosphorus fertilizer combination on the physiological growth characteristics of alfalfa in saline-alkali soil of the Yellow River Delta. Chin J Grassland. 2024; 46( 7): 35– 45. (In Chinese). doi:10.16742/j.zgcdxb.20240017. [Google Scholar] [CrossRef]
37. Zhong S , Hou P , Yu M , Cao W , Tu X , Ma X , et al. Arbuscular mycorrhizal fungi inoculation and different phosphorus fertilizer levels modulate phosphorus acquisition and utilization efficiency of alfalfa (Medicago sativa L.) in saline-alkali soil. Plants. 2025; 15( 1): 114. doi:10.3390/plants15010114. [Google Scholar] [CrossRef]
38. Barrs HD , Weatherley PE . A re-examination of the relative turgidity technique for estimating water deficits in leaves. Aust J Biol Sci. 1962; 15( 3): 413– 28. doi:10.1071/bi9620413. [Google Scholar] [CrossRef]
39. Bates LS , Waldren RP , Teare ID . Rapid determination of free proline for water-stress studies. Plant Soil. 1973; 39( 1): 205– 7. doi:10.1007/BF00018060. [Google Scholar] [CrossRef]
40. Grieve CM , Grattan SR . Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil. 1983; 70( 2): 303– 7. doi:10.1007/BF02374789. [Google Scholar] [CrossRef]
41. Yoshida S , Douglas DA , Cock JH , Kwanchai AG . Laboratory manual for physiological studies of rice. 3rd ed. Los Banos, CA, USA: International Rice Research Institute; 1976. p. 46– 9. [Google Scholar]
42. Bradford MM . A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976; 72( 1–2): 248– 54. doi:10.1016/0003-2697(76)90527-3. [Google Scholar] [CrossRef]
43. Maehly AC , Chance B . The assay of catalases and peroxidases. Methods Biochem Anal. 1954; 1: 357– 424. doi:10.1002/9780470110171.ch14. [Google Scholar] [CrossRef]
44. Geng DM , Shan LS , Li Y . Effect of soil water stress on chlorophyll fluorescence and antioxidant enzyme activity in Reaumuria soongorica seedlings. Chin Bull Bot. 2014; 49( 3): 282– 91. doi:10.3724/SP.J.1259.2014.00282. [Google Scholar] [CrossRef]
45. Nakano Y , Asada K . Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981; 22( 5): 867– 80. doi:10.1093/oxfordjournals.pcp.a076232. [Google Scholar] [CrossRef]
46. Gauillard F , Richard-Forget F , Nicolas J . New spectrophotometric assay for polyphenol oxidase activity. Anal Biochem. 1993; 215( 1): 59– 65. doi:10.1006/abio.1993.1554. [Google Scholar] [CrossRef]
47. Velikova V , Yordanov I , Edreva A . Oxidative stress and some antioxidant systems in acid rain-treated bean plants Protective role of exogenous polyamines. Plant Sci. 2000; 151( 1): 59– 66. doi:10.1016/S0168-9452(99)00197-1. [Google Scholar] [CrossRef]
48. Yan B , Dai Q , Liu X , Huang S , Wang Z . Flooding-induced membrane damage, lipid oxidation and activated oxygen generation in corn leaves. Plant Soil. 1996; 179( 2): 261– 8. doi:10.1007/BF00009336. [Google Scholar] [CrossRef]
49. Heath RL , Packer L . Reprint of: photoperoxidation in isolated chloroplasts I. kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys. 2022; 726: 109248. doi:10.1016/j.abb.2022.109248. [Google Scholar] [CrossRef]
50. Lei S , Jia X , Zhao C , Shao M . A review of saline-alkali soil improvements in China: efforts and their impacts on soil properties. Agric Water Manag. 2025; 317: 109617. doi:10.1016/j.agwat.2025.109617. [Google Scholar] [CrossRef]
51. Shaheen F , Ashraf MY , Hussain MI , Khan ZI , Chaudhry UK , Abideen Z , et al. Co-application of salicylic acid and potassium induce salinity tolerance by modulating ion homeostasis, chlorophyll pigments and photosynthetic apparatus in wheat. Plant Growth Regul. 2025; 105( 6): 2275– 96. doi:10.1007/s10725-025-01373-9. [Google Scholar] [CrossRef]
52. He F , Zhao X , Qi G , Sun S , Shi Z , Niu Y , et al. Exogenous melatonin alleviates NaCl injury by influencing stomatal morphology, photosynthetic performance, and antioxidant balance in maize. Int J Mol Sci. 2024; 25( 18): 10077. doi:10.3390/ijms251810077. [Google Scholar] [CrossRef]
53. He J , Chen Y , Zhang M , Qiu Y , Zhou H , Li M . Current perspectives on improving soybean performance on saline-alkaline lands. New Crops. 2026; 3: 100079. doi:10.1016/j.ncrops.2025.100079. [Google Scholar] [CrossRef]
54. Ghouili E , Muhovski Y , Hogue R , Ouertani RN , Abdelkrim S , Li Z , et al. Compost and biochar application mitigates salt stress on barley: monitoring of agronomic, physiological, biochemical and molecular parameters. Plant Soil. 2025; 513( 2): 1999– 2021. doi:10.1007/s11104-025-07292-y. [Google Scholar] [CrossRef]
55. Murtaza G , Rizwan M , Usman M , Hyder S , Akram MI , Deeb M , et al. Biochar enhances the growth and physiological characteristics of Medicago sativa, Amaranthus caudatus and Zea mays in saline soils. BMC Plant Biol. 2024; 24( 1): 304. doi:10.1186/s12870-024-04957-1. [Google Scholar] [CrossRef]
56. Yan X , Wang Z , Zhao M , Hao J , Liu J , Yan Y , et al. Hydrothermal biochar enhances the photosynthetic efficiency and yield of alfalfa by optimizing soil chemical properties and stimulating the activity of microbial communities. Sci Rep. 2024; 14( 1): 31420. doi:10.1038/s41598-024-83098-9. [Google Scholar] [CrossRef]
57. Yang Y , Ahmed W , Ye C , Yang L , Wu L , Dai Z , et al. Exploring the effect of different application rates of biochar on the accumulation of nutrients and growth of flue-cured tobacco (Nicotiana tabacum). Front Plant Sci. 2024; 15: 1225031. doi:10.3389/fpls.2024.1225031. [Google Scholar] [CrossRef]
58. Ren S , Zhong J , Wang K , Liu R , Feng H , Dong Q , et al. Application of biochar in saline soils enhances soil resilience and reduces greenhouse gas emissions in arid irrigation areas. Soil Tillage Res. 2025; 250: 106500. doi:10.1016/j.still.2025.106500. [Google Scholar] [CrossRef]
59. Hu Y , Sun B , Wu S , Feng H , Gao M , Zhang B , et al. After-effects of straw and straw-derived biochar application on crop growth, yield, and soil properties in wheat (Triticum aestivum L.)-maize (Zea mays L.) rotations: a four-year field experiment. Sci Total Environ. 2021; 780: 146560. doi:10.1016/j.scitotenv.2021.146560. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools