 Open Access
Open Access
ARTICLE
Efficacy of vaginal androgen combined with pelvic floor muscle training for stress urinary incontinence in postmenopausal women: a randomized-controlled trial
1 Department of Urology, Hainan Western Central Hospital, Danzhou, 571700, China
2 Department of Urology, Zhejiang Provincial Hospital of Chinese Medicine, The First Affiliated Hospital of Zhejiang Chinese Medical University, Hangzhou, 310006, China
* Corresponding Author: Yang Yu. Email:
Canadian Journal of Urology 2026, 33(1), 143-154. https://doi.org/10.32604/cju.2025.069041
Received 12 June 2025; Accepted 02 September 2025; Issue published 28 February 2026
Abstract
Objectives: Postmenopausal women with stress urinary incontinence (SUI) exhibit low androgen expression. This study aimed to evaluate the efficacy and safety of vaginal androgen combined with pelvic floor muscle training (PFMT) in the treatment of SUI in postmenopausal women. Methods: Postmenopausal women with SUI were recruited from Hainan West Central Hospital between January 2024 and March 2025. Participants were randomly assigned in a double-blind manner to receive either vaginal androgen cream combined with PFMT (treatment group) or a visually identical placebo cream (without androgens) combined with PFMT (control group). The vaginal cream was applied to the vaginal wall at a dose of 0.5 g per application, twice weekly for a total of 10 applications, while PFMT was conducted for 8 weeks. The clinical efficacy and safety were compared between the two groups. Results: A total of 61 patients were finally enrolled, with 31 in the treatment group and 30 in the control group. At both 3-month and 6-month follow-ups, the treatment group demonstrated significantly lower values in daily pad usage (p < 0.05), 24-h pad test scores (p < 0.05), and ICIQ-UI SF scores (p < 0.05) compared to the control group. The improvement rate of urinary incontinence was significantly higher in the treatment group (p < 0.05). Compared to baseline, the treatment group showed statistically significant reductions in all three outcome measures (all p < 0.05). No severe adverse events were reported in either group during the treatment period. Conclusions: Androgen therapy combined with PFMT significantly improved the urinary incontinence remission rate in postmenopausal women with SUI, with no severe adverse effects observed. These findings suggest that androgen therapy may represent a novel therapeutic approach for SUI management in postmenopausal women. Clinical trial registration number: ChiCTR2300078752 in the Chinese Clinical Trial Registry.Keywords
Stress urinary incontinence (SUI) is defined as any involuntary leakage of urine associated with physical activity,1 characterized by the involuntary loss of urine during exertion (e.g., physical effort, coughing, exercise, or sneezing) when intravesical pressure exceeds urethral closure pressure.2 SUI is the most prevalent form of urinary incontinence, with reported prevalence rates ranging from 10% to 70%. Its incidence increases with age and is more commonly observed in middle-aged and elderly women.2 SUI imposes a significant burden on affected individuals, impairing their quality of life and causing embarrassment due to involuntary urine leakage during activities such as sneezing or coughing.3 With the continued global aging of the population, the prevalence and clinical significance of SUI are expected to rise correspondingly.4
The severity of SUI typically worsens postmen opausally.5 Current evidence suggests a strong association between menopause and SUI, with menopause being recognized as a major risk factor for this condition.6 The primary etiology of SUI in postmenopausal women involves alterations in the structure and function of urethral supportive tissues, though the precise pathogenic mechanisms remain incompletely understood.4 Menopause is typically accompanied by changes in circulating sex hormone levels, which subsequently induce atrophic changes in the urogenital tract.7 Recent studies have increasingly suggested that the development of SUI may be associated with alterations in testosterone levels, demonstrating an inverse correlation between testosterone concentration and SUI incidence.8,9 Furthermore, studies suggest that alterations in testosterone levels in elderly women may potentially increase the risk of urinary incontinence, rendering it an ideal hormonal target for investigating incontinence pathogenesis in this population.9 Our preliminary studies have similarly demonstrated that postmenopausal women with SUI exhibit significantly reduced serum testosterone levels, establishing testosterone deficiency as one of the risk factors for SUI in this population.10 Both animal studies and clinical trials indicate a significant association between testosterone and urinary incontinence, with evidence showing that testosterone supplementation or modulation of androgen receptor density and sensitivity may positively influence SUI prevention and symptom alleviation, while exhibiting minimal adverse effects.11,12
However, due to study heterogeneity—limited by variations in androgen types, dosage intensity, treatment duration, administration routes, and small sample sizes—it remains challenging to ascertain the definitive benefits of testosterone supplementation for SUI. Further, whether androgen therapy combined with pelvic floor muscle training (PFMT) could serve as a novel reference and adjunctive approach for treating SUI remains clinically unverified. Specifically, future studies should explore testosterone supplementation therapy in conjunction with PFMT in postmenopausal SUI patients (who typically exhibit low endogenous testosterone levels) to systematically evaluate its clinical efficacy and safety profile. Therefore, this randomized double-blind controlled trial aims to preliminarily investigate the clinical efficacy and safety of androgen therapy combined with PFMT for SUI in postmenopausal women, seeking to establish a potential novel therapeutic approach and provide initial solutions to the aforementioned scientific challenges.
This study enrolled postmenopausal female patients diagnosed with SUI who sought treatment at Hainan West Central Hospital between July 2023 and October 2024, with complete medical records available for analysis. The study protocol received ethical approval from the Ethics Committee of Hainan West Central Hospital (Approval No. LLKY-2023-13), and written informed consent was obtained from all participants prior to their inclusion. This investigation was conducted in accordance with the World Medical Association’s Declaration of Helsinki (revised 2013) and has been registered with the Chinese Clinical Trial Registry (Registration No. ChiCTR2300078752).
Inclusion and exclusion criteria
Inclusion criteria for SUI patients: 1) postmenopausal women with clinically confirmed SUI, diagnosed according to the 2017 American Urological Association (AUA) Guideline: Surgical Treatment of Female Stress Urinary Incontinence; 2) patients opting for non-surgical management approaches; 3) patients with complete medical records available for review.
Exclusion criteria: 1) patients with prior surgical intervention for stress urinary incontinence; 2) candidates actively seeking surgical management for SUI; 3) history of abdominal or pelvic surgery within six months preceding enrollment; 4) active urinary tract infection; 5) documented neurological or psychiatric disorders; 6) indeterminate menopausal status; 7) diagnosis of urge urinary incontinence or mixed urinary incontinence; 8) malignancy (any cancer diagnosis); 9) unexplained vaginal bleeding; 10) current use of systemic or local hormone therapy (including oral contraceptives, vaginal estrogen, selective estrogen receptor modulators, or androgen receptor modulators); 11) inability to comprehend and sign informed consent documents.
Determination of menopausal status
Menopausal status was ascertained through structured clinical interviews. Participants were initially queried: “Have you experienced at least one menstrual period in the past 12 months?” (with explicit instruction to exclude bleeding episodes attributable to medical conditions, hormonal therapy, or surgical procedures). Subjects responding affirmatively were excluded from study participation. Negative responders underwent secondary assessment: “What is the primary reason for your absence of menstruation during the past 12 months?” (Response options: pregnancy; lactation; hysterectomy; menopausal transition/life change; other). Women were classified as postmenopausal if they had experienced 12 consecutive months of amenorrhea, with etiological categorization as either natural menopause or hysterectomy. While naturally postmenopausal women were included in the study cohort, those with hysterectomy were analyzed separately. Postmenopausal women with a history of hysterectomy were classified as naturally menopausal and included in the study if the surgical procedure was performed after establishing 12 consecutive months of amenorrhea. Postmenopausal women undergoing hysterectomy without prior documentation of 12 consecutive months of amenorrhea were required to meet all the following criteria to be considered as having achieved postmenopausal status and thus eligible for study inclusion: 1) postmenopausal women with amenorrhea for the preceding 12 months; 2) clearly documented evidence attributing the cause of amenorrhea to hysterectomy; 3) definitive confirmation that: i) amenorrhea onset occurred following hysterectomy, and ii) a minimum of 12 months had elapsed since the surgical procedure; and 4) age between 45–55 years with concurrent menopausal syndrome manifestations including: i) vasomotor symptoms (spontaneous episodes of facial/neck/chest flushing and diaphoresis unrelated to physical activity); ii) autonomic dysfunction (palpitations, vertigo, cephalgia, or tinnitus); iii) neuropsychiatric manifestations (impaired concentration, emotional lability including irritability, anxiety, or depression); iv) sexual dysfunction (decreased libido and vaginal dryness); and v) other symptoms (e.g., sleep disturbances). Women who reported 12-month amenorrhea without documented etiology were excluded from study participation.
Termination and withdrawal criteria
Termination criteria: 1) cases experiencing severe adverse events, complications, or special physiological changes that make continued participation in the study inadvisable; 2) subjects with poor compliance who are unable to adhere to the prescribed treatment protocol.
Withdrawal criteria: 1) patients who discontinue treatment due to personal reasons; 2) patients undergoing surgical intervention during follow-up; 3) patients lost to follow-up without prior notice.
Postmenopausal women with SUI were randomly allocated to either the treatment group or the control group using a SAS software-generated randomization method. The treatment group received intravaginal testosterone cream combined with PFMT, whereas the control group received a visually identical placebo cream (without active testosterone) in addition to PFMT.
For the treatment group, patients applied Testosterone Hormone Ointment (Brand: Harasawa Pharmaceuticals, Product Name: Testosterone Hormone Ointment 10 g, Specification: 10 g/tube) 0.5 g per dose intravaginally twice weekly for a total of 10 applications. Concurrently, they performed PFMT in a standing position, consisting of sustained pelvic floor muscle contractions (Kegel exercises) for 5 s followed by 5 s of relaxation. This cycle was repeated 15 times per set, with 8 sets performed daily for 8 weeks.
For the control group, patients applied a visually matched placebo cream (0.5 g per dose, devoid of active testosterone) intravaginally twice weekly for a total of 10 applications. PFMT was conducted identically to the treatment group.
This study was conducted as a randomized, double-blind, placebo-controlled trial. The treating physicians, data collectors, statisticians, and telephone follow-up personnel were all blinded to group allocation, with no communication permitted regarding treatment assignment among these parties.
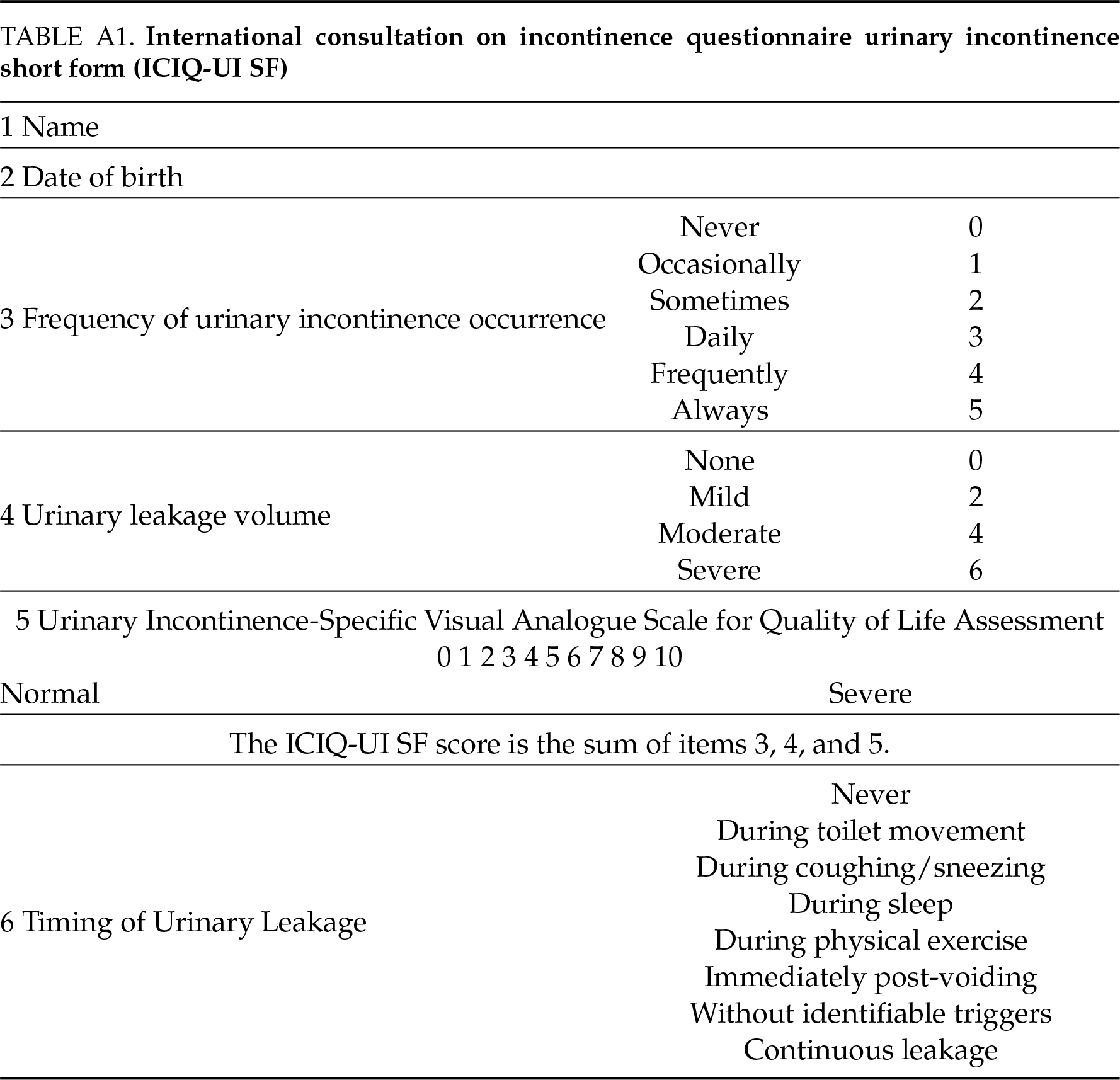
Patients in both groups were followed up via telephone or clinic visits at baseline (pre-treatment), 3 months post-treatment, and 6 months post-treatment. The following parameters were recorded: 1) number of required sanitary pads; 2) International Consultation on Incontinence Questionnaire Urinary Incontinence Short Form score (ICIQ-UI SF) (see Table A1); 3) basic physical condition.
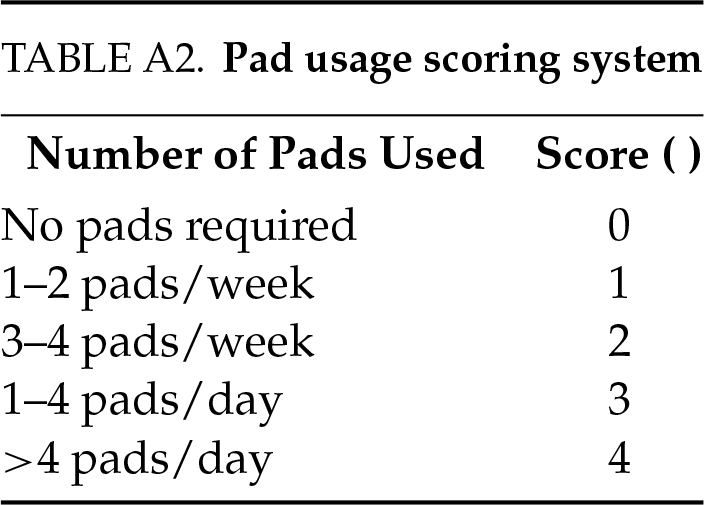
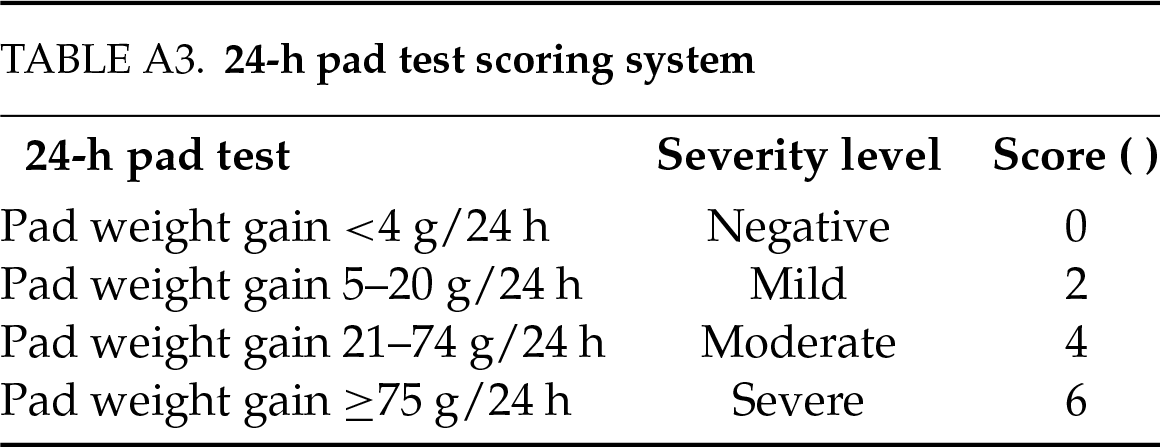
Primary outcome measures: 1) pad usage score (scored according to the criteria in Table A2): 0 points: no pad required; 1 point: 1–2 pads/week; 2 points: 3–4 pads/week; 3 points: 1–4 pads/day; 4 points: >4 pads/day; 2) 24-h pad test (with scoring system detailed in Table A3): 0 points (negative): <4 g weight increase in 24 h; 2 points (mild): 5–20 g weight increase in 24 h; 4 points (moderate): 21–74 g weight increase in 24 h; 6 points (Severe): ≥75 g weight increase in 24 h.
Secondary outcome measures: ICIQ-UI SF score.
Efficacy evaluation criteria and safety assessment
Efficacy evaluation criteria: the primary efficacy endpoint was defined as the improvement rate of urinary incontinence, calculated as: [(total baseline score − total post-treatment score) ÷ total baseline score] × 100%. The assessment incorporated three parameters: 1) pad usage count, 2) 24-h pad test results, and 3) ICIQ-UI SF scores. Comparative analyses were performed for all parameters (including the improvement rate) using: 1) inter-group comparisons (treatment vs. control); 2) intra-group comparisons (pre- vs. post-treatment).
Safety assessment: 1) vital signs monitoring: blood pressure, heart rate, respiratory rate, and consciousness status were evaluated and recorded before and after each treatment session to detect any acute changes. 2) adverse events (AEs): patients were closely monitored following each intervention. Any treatment-emergent AEs (e.g., vaginal pruritus, erythema, or pain) were systematically documented and assessed using standardized criteria (see Table A4).
The sample size was estimated using MedCalc software (version 19.0.7), with a minimum required sample size of 40 cases at α= 0.05 and β= 0.1. After strict screening based on the inclusion and exclusion criteria, a total of 80 postmenopausal women with stress urinary incontinence (SUI) were enrolled in the study, among whom 19 cases were excluded due to loss to follow-up, opting for surgical intervention, or non-compliance with the prescribed treatment protocol.
The quantitative data were analyzed using SPSS 28.0 statistical software (IBM Corporation, Armonk, NY, USA). Normally distributed measurement data were expressed as mean ± standard deviation (
Baseline demographic and clinical characteristics of both patient cohorts
A total of 61 postmenopausal women with SUI were ultimately enrolled (see Figure 1). Participants were allocated as follows: 1) treatment group (n = 31): received intravaginal testosterone cream combined with PFMT; 2) control group (n = 30): received an identical-appearing placebo cream (without active testosterone component) combined with PFMT. The baseline clinical characteristics of both groups are presented in Table 1.
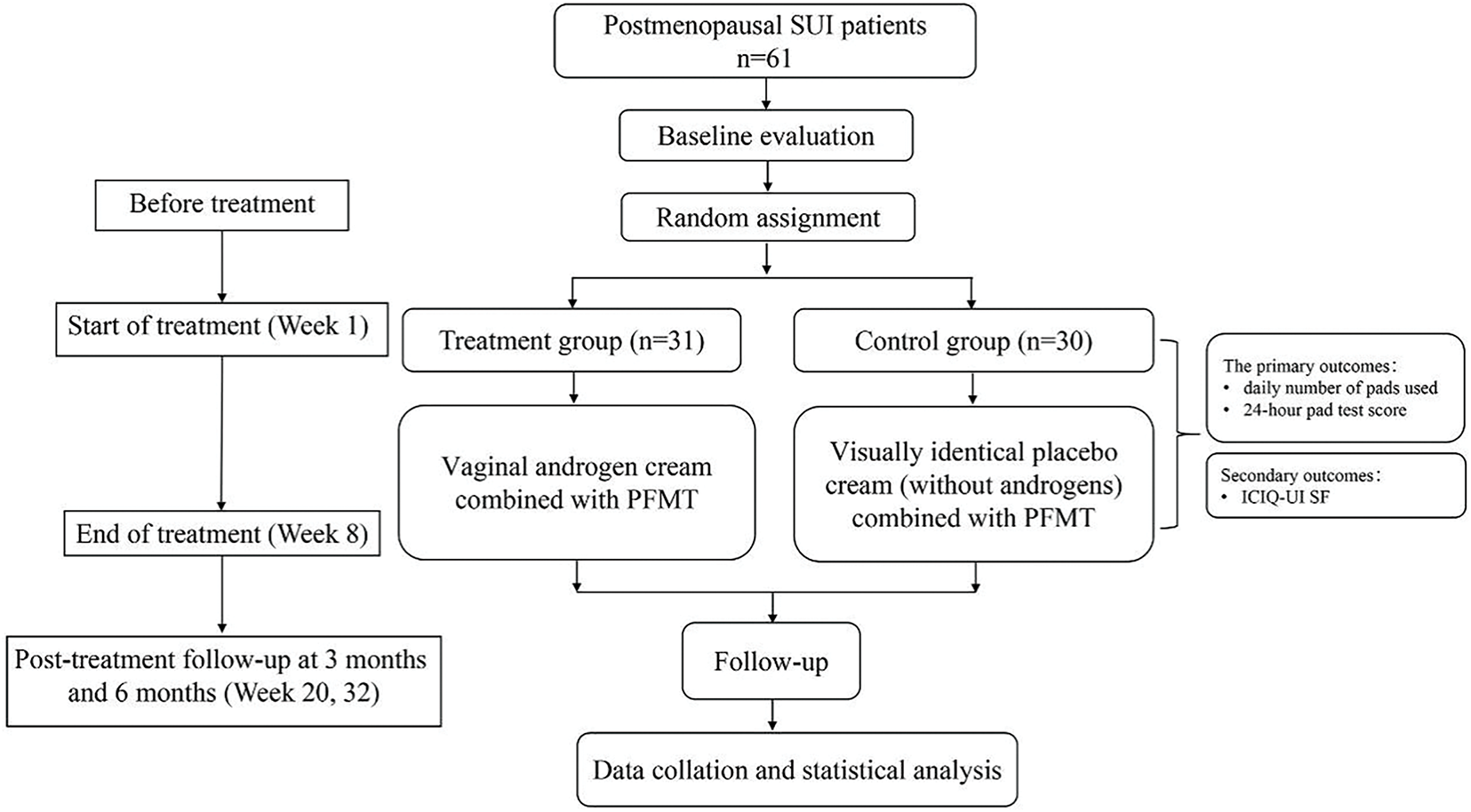
FIGURE 1. Flowchart of the study
Comparative analysis of clinical efficacy between groups
The follow-up data at 3 months and 6 months post-treatment showed that the treatment group demonstrated significantly better outcomes than the control group (p < 0.05) in the following measures: significantly lower pad count, 24-h pad test scores, and ICIQ-UI SF scores, along with a significantly higher urinary incontinence improvement rate. All intergroup differences were statistically significant (p < 0.05), as detailed in Table 2 and Figure 2. Furthermore, within-group analysis revealed statistically significant reductions (p < 0.05) in all observed parameters (pad usage, 24-h pad test scores, and ICIQ-UI SF scores) from baseline levels in the treatment group, as shown in Table 3 and Figure 3.
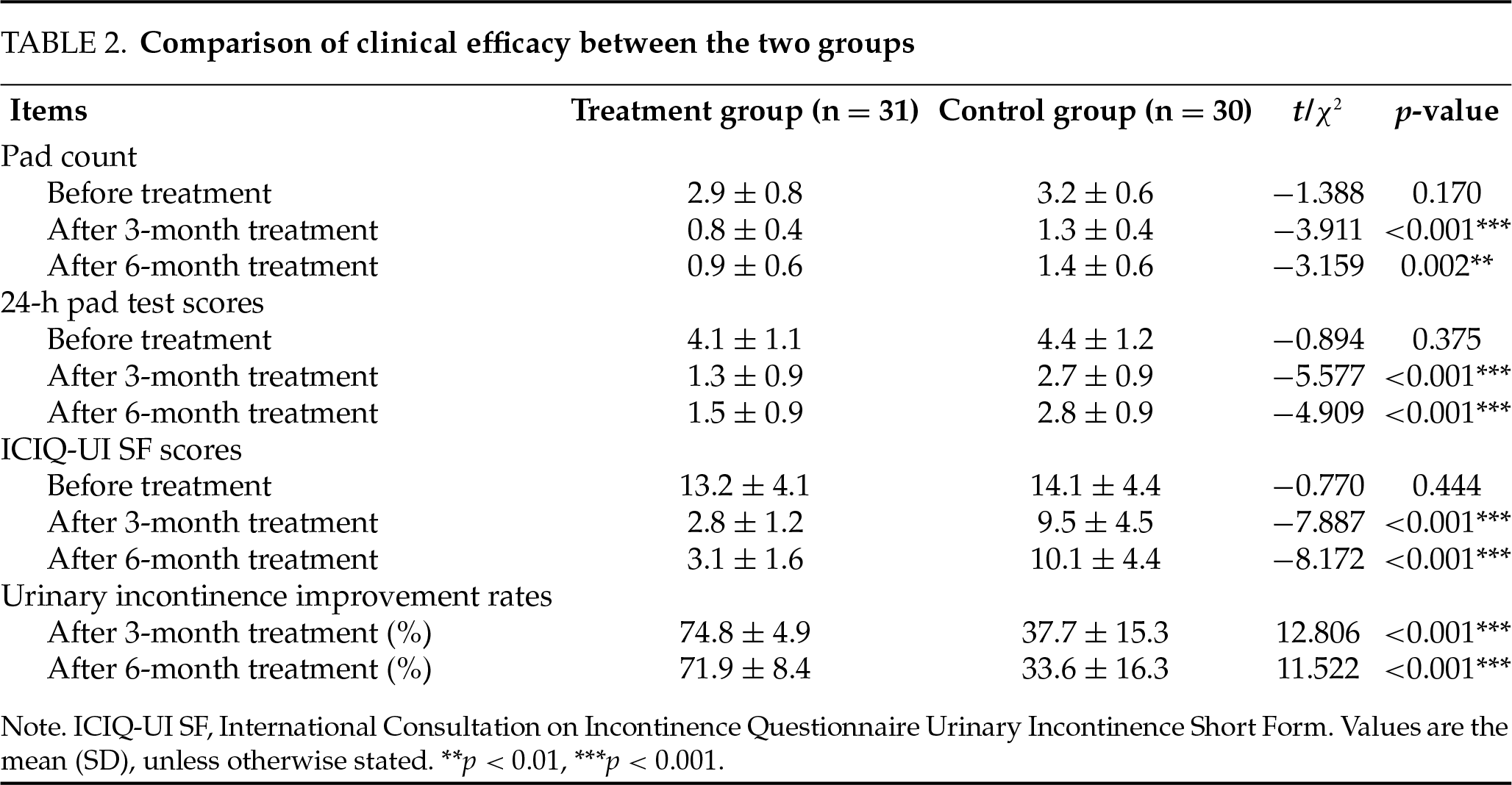
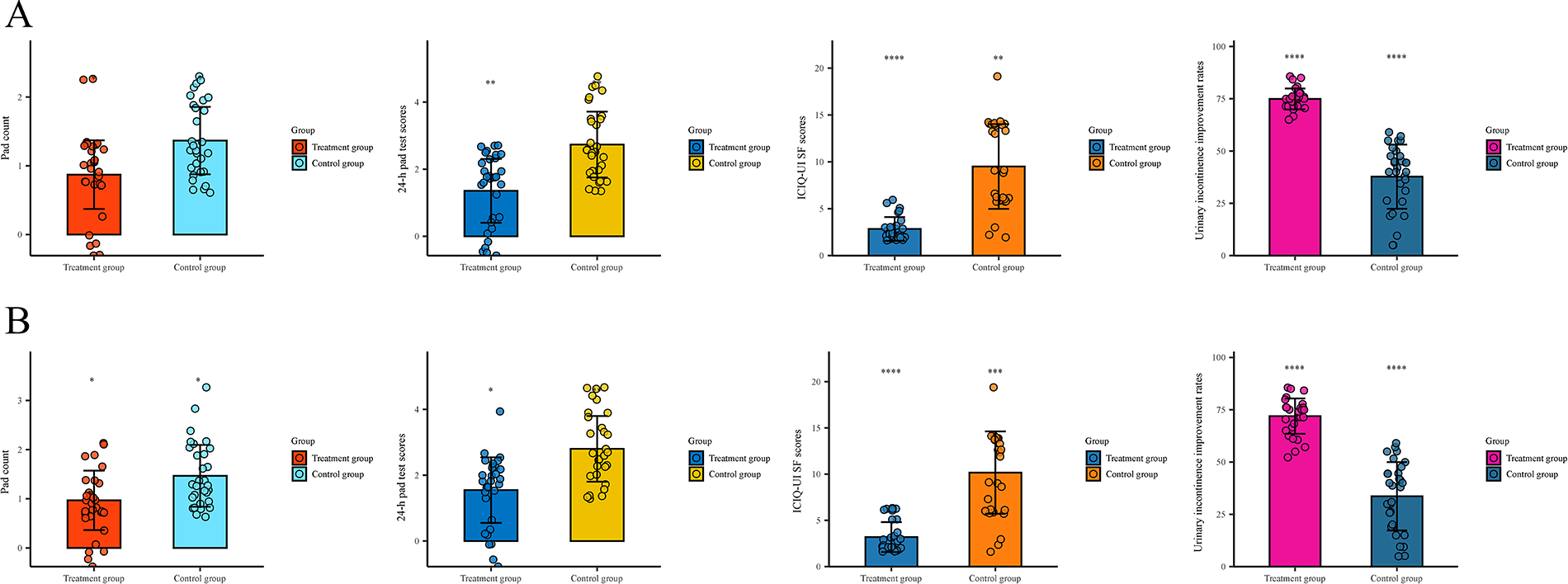
FIGURE 2. Comparison of clinical efficacy between the two patient groups. (A) At the 3-month follow-up, the treatment group demonstrated significantly lower values in pad usage (p < 0.05), 24-h pad test scores (p < 0.01), and ICIQ-UI SF scores (p < 0.01) compared with the control group, along with a significantly higher urinary incontinence improvement rate (p < 0.0001). (B) At the 6-month follow-up, the treatment group maintained lower values in pad usage (p < 0.05), 24-h pad test scores (p < 0.01), and ICIQ-UI SF scores (p < 0.001) compared with controls, with persistently superior urinary incontinence improvement rates (p < 0.0001). *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001

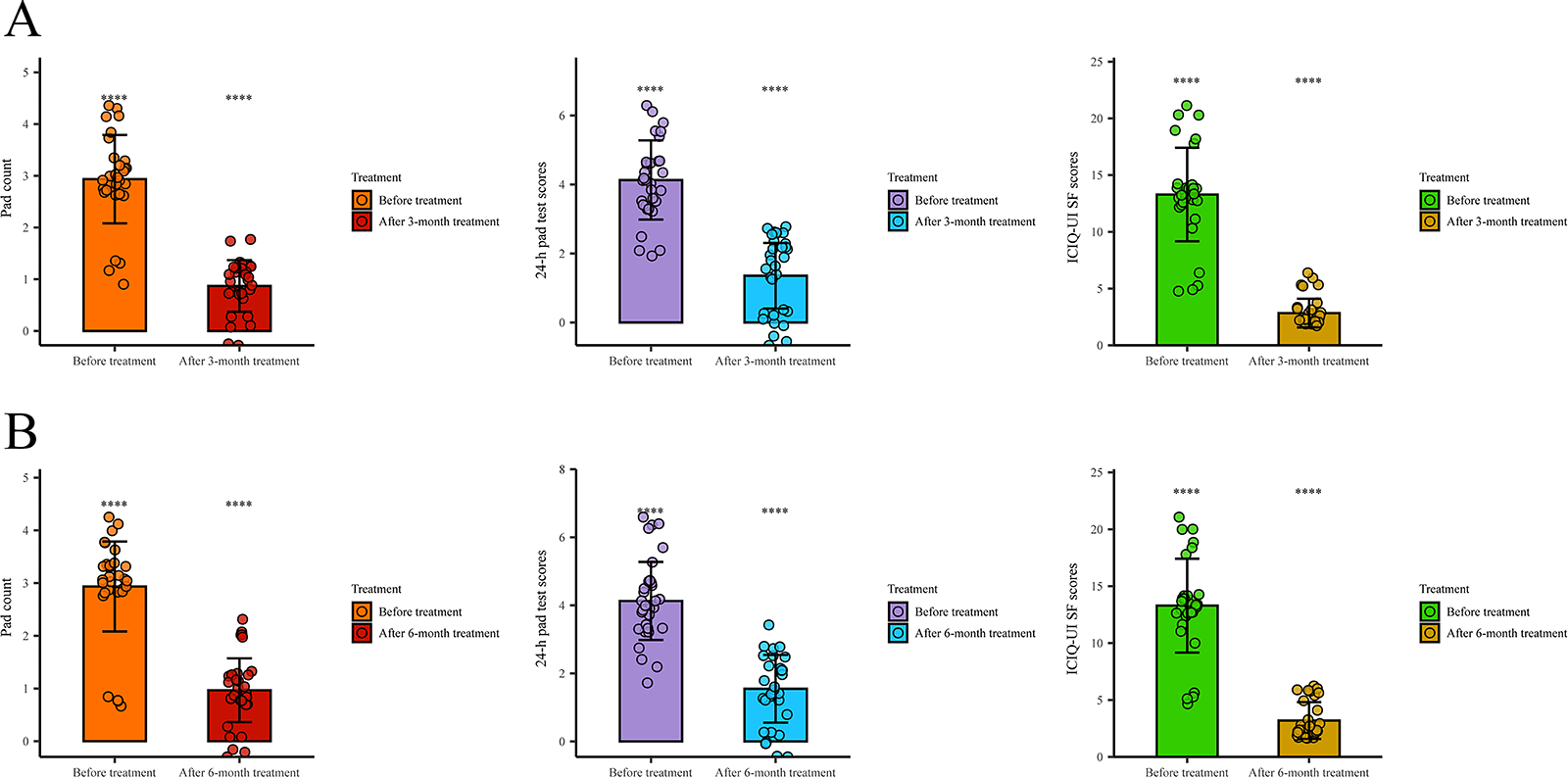
FIGURE 3. Comparison of clinical parameters before and after treatment in the treatment group. (A) At the 3-month follow-up, the treatment group exhibited statistically significant reductions in daily pad count (p < 0.0001), 24-h pad test score (p < 0.0001), and ICIQ-UI SF score (p < 0.0001) compared to pretreatment baseline values. (B) These significant improvements persisted at the 6-month follow-up, with maintained reductions in daily pad count (p < 0.0001), 24-h pad test score (p < 0.0001), and ICIQ-UI SF score (p < 0.0001) compared to baseline measurements. ****p < 0.0001
Safety profile comparison between groups
Both groups showed no severe adverse events. Mild complications (1 erythema and 1 pruritus in treatment group vs. 1 erythema in controls) showed no statistical significance (p > 0.05, Table 3).
SUI represents a significant global health challenge for women.13,14 Among various subtypes of urinary incontinence, SUI predominates, clinically characterized by involuntary urine leakage during activities that increase intra-abdominal pressure, including sneezing, coughing, laughing, or physical exertion. This condition demonstrates particular prevalence among women during pregnancy or menopausal transition. SUI imposes profound physical and psychological consequences on affected individuals, while simultaneously creating substantial medical and socioeconomic burdens for society.13 The pathophysiology of SUI is primarily driven by two distinct mechanisms: urethral hypermobility resulting from compromised support structures, and intrinsic sphincteric deficiency (ISD) characterized by deterioration of urethral mucosal coaptation and muscular tone. Current therapeutic options for SUI encompass both conservative management strategies and surgical interventions. Conservative approaches, while widely employed, demonstrate substantial dependence on patient compliance and are associated with elevated recurrence rates. Surgical alternatives, including midurethral sling procedures, provide effective anatomical restoration but entail significant healthcare expenditures and potential adverse effects. These inherent limitations underscore the pressing need for more efficacious and comprehensive treatment modalities.2
Current evidence suggests an inverse correlation between advancing age-related androgen decline and increasing SUI prevalence, implying potential pathophysiological causation. Longitudinal data demonstrate that circulating testosterone levels initiate their descent around the age of 25 in women, reaching a plateau during postmenopause—a temporal pattern paralleling SUI risk progression. These clinical observations posit androgens as putative pathogenic contributors to SUI development, warranting their reconsideration as “the overlooked female hormones”.15 A large-scale cohort study involving 2321 female participants demonstrated a significant association between markedly decreased serum testosterone levels and elevated susceptibility to SUI.16 This finding corroborates prior research evidence indicating that menopausal women exhibit diminished testosterone concentrations, which appear clinically correlated with SUI pathogenesis.17 Our preliminary studies have similarly demonstrated significantly reduced serum testosterone levels in postmenopausal women with SUI, establishing hypogonadism as an independent risk factor for SUI in this population.10 However, the aforementioned studies present several notable limitations, most significantly the lack of clinical validation regarding androgen supplementation in postmenopausal women with SUI and low endogenous serum testosterone levels. Future research should specifically investigate the therapeutic efficacy and safety profile of testosterone administration in this patient population.
Previous investigations have demonstrated that testosterone exerts both direct and indirect modulatory effects on micturition physiology.18 Androgen receptors (ARs), which exhibit responsiveness to testosterone stimulation, have been histologically identified across multiple pelvic structures, including the levator ani muscle and its fascial attachments, urethral epithelium, and detrusor smooth muscle in both human and animal models.19–21 The expression of androgen receptors in urethral and bladder epithelium, as well as smooth muscle, suggests direct androgen-mediated regulation of micturition. Mechanistically, diminished testosterone levels may potentiate detrusor contractility, potentially inducing detrusor overactivity and consequent voiding dysfunction. Conversely, testosterone may exert inhibitory effects on detrusor contraction through post-junctional non-genomic receptor pathways.18 Furthermore, androgens likely demonstrate anabolic effects on periurethral musculature and connective tissues, thereby contributing to urinary continence mechanisms.5 Our results demonstrated significant between-group differences at both 3- and 6-month follow-ups (all p < 0.05). The treatment group demonstrated significantly lower values than controls in pad count (p < 0.05), 24-h pad test scores (p < 0.05), ICIQ-UI SF scores (p < 0.05), and significantly higher urinary incontinence improvement rates (p < 0.05). Furthermore, intra-group analyses revealed statistically significant reductions (p < 0.05) in all measured parameters (pad usage, 24-h pad test scores, and ICIQ-UI SF scores) from baseline within the treatment group. Our preliminary investigation into the therapeutic efficacy of androgen supplementation in postmenopausal women with SUI demonstrated clinically significant improvements in continence rates.
Multiple studies have provided further histological, animal experimental, and clinical evidence supporting the association between serum circulating testosterone levels and the development of stress urinary incontinence (SUI).22,23 First, androgen receptors (ARs) are widely distributed across various muscular tissues, including the pelvic floor muscles.19–21 The ARs in the pelvic floor and lower urinary tract muscles exhibit sensitivity to testosterone stimulation.19–21 All anatomical structures involved in urinary continence are richly supplied with ARs, suggesting a functional role of androgens in these tissues.15 Current evidence indicates that: Androgens may upregulate nitric oxide synthase (NOS) via AR expression, thereby increasing nitric oxide (NO) production to induce bladder muscle relaxation and contribute to continence mechanisms; Given that ARs are expressed in the striated muscle fibers, stromal cells, and fascia of the female levator ani muscle, androgens may regulate pluripotent cells through AR-mediated pathways to increase levator ani muscle mass while reducing fat content, indirectly supporting continence; Through the anabolic effects of ARs, androgens may stimulate collagen synthesis while inhibiting its degradation, increase collagen fiber density, promote striated muscle hypertrophy, and ultimately enhance urinary continence.19–21 Furthermore, studies have demonstrated that androgens can effectively strengthen human pelvic floor and lower urinary tract muscles.11 Mammadov et al. showed in a female SUI rat model that a single testosterone injection increased myofiber cross-sectional area and improved SUI symptoms.23 Ponnusamy et al. reported that selective androgen receptor modulators (SARMs) could regulate AR sensitivity, increase pelvic floor muscle mass in ovariectomized SUI mice, and alleviate SUI symptoms. Animal studies suggest that testosterone supplementation or modulation of AR quantity and sensitivity may have beneficial effects on SUI prevention and symptom improvement.19 A Phase II clinical trial demonstrated that SARMs effectively relieved SUI symptoms in postmenopausal women with a favorable safety profile.12
Our study further revealed a favorable safety profile for androgen therapy in postmenopausal SUI patients: the treatment group had 1 case of vaginal erythema and 1 case of vaginal pruritus, and the control group had 1 case of vaginal erythema. The incidence of adverse events did not differ significantly between groups (p > 0.05). In a prospective observational pilot study, 34 female patients with vaginal atrophy and moderate SUI were enrolled. All participants received daily intravaginal dehydroepiandrosterone (DHEA) 6.5 mg for 12 weeks. The results demonstrated that intravaginal DHEA treatment not only significantly alleviated urinary symptoms, improved quality of life, and enhanced pelvic floor muscle function in SUI patients but also did not induce any adverse events.5 Our findings are consistent with the above results, providing preliminary evidence on the clinical efficacy of androgen therapy combined with pelvic floor muscle training in postmenopausal women with SUI. Furthermore, this study reinforces the safety profile of androgen supplementation in the treatment of SUI in postmenopausal women, suggesting that testosterone supplementation may play a crucial role in managing SUI in this population.
Our study has several limitations. First, the mechanism of androgen action in women with urinary incontinence is complex. Existing studies have demonstrated that androgens can induce muscle hypertrophy while reducing fat mass. Additional research has found that the levator ani muscle, along with other pelvic floor and lower urinary tract muscles, exhibits sensitivity to the anabolic effects of testosterone.21 Moreover, emerging evidence suggests that androgens may adversely affect urogenital tissues by modulating collagen metabolism—particularly through the inhibition of matrix metalloproteinase (MMP) synthesis or activity.24 This study did not thoroughly investigate the specific pathogenic mechanisms of testosterone in postmenopausal women with SUI. Second, limitations in the route of administration. This study employed vaginal delivery of the drug to minimize systemic exposure and avoid potential virilizing effects, thereby restricting its pharmacological actions to localized effects. Our study has a relatively short follow-up period and lacks long-term follow-up data. Additionally, while the primary focus was on treatment efficacy during therapy, serum testosterone levels were not monitored in a timely manner. The findings of our study may also be influenced by the “postmenopausal” condition, and thus may not be applicable to surgically menopausal or perimenopausal women. Given that testosterone levels in surgically menopausal and perimenopausal women may differ from those in postmenopausal women, further research is needed to determine whether our results extend to these populations. Furthermore, this study warrants the inclusion of larger clinical cohorts, and future investigations should consider a multicenter design. Despite these limitations, our findings provide a valuable addition to the currently limited body of research examining the association between serum testosterone and SUI.
In conclusion, this study demonstrated that the treatment group showed significantly lower values in pad usage, 24-h pad test scores, and ICIQ-UI SF scores compared to the control group, along with a significantly higher urinary incontinence improvement rate, with statistically significant intergroup differences. Furthermore, the treatment group exhibited significant reductions in pad usage, 24-h pad test scores, and ICIQ-UI SF scores from baseline levels, all of which were statistically significant. Furthermore, both groups experienced minimal side effects during treatment. In summary, our study demonstrates that androgen therapy combined with PFMT significantly improves the urinary incontinence remission rate in postmenopausal women with SUI, without any severe adverse effects. Therefore, androgen therapy may serve as a novel therapeutic approach for SUI in postmenopausal women, improving their physical and mental health as well as quality of life.
Acknowledgement
Not applicable.
Funding Statement
This study was supported by Joint Program on Health Science & Technology Innovation of Hainan Province (WSJK2024QN093); Discipline Leader Development Program for Outstanding Talents of Hainan West Central Hospital.
Author Contributions
Yingxiu Chen and Yang Yu designed the study. Yingxiu Chen, Zhongyu Ren and Yang Yu provided treatment to the patients and conducted follow-up, recording their clinical data. Yingxiu Chen and Jiasheng Yan analyzed the data. Yingxiu Chen and Yang Yu wrote the manuscript. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
Data is not available due to ethical and legal restrictions. Due to the nature of this research, participants of this study did not agree to their data being shared publicly, so supporting data is not available.
Ethics Approval
The study involved human participants and was approved by the Ethics Committee of Hainan West Central Hospital (Medical ethics approval number: LLKY-2023-13), and all patients signed consent forms before the start of the trial. The procedures followed in this study followed the Declaration of Helsinki of the World Medical Association, as amended in 2013 and the study was registered with Chinese Clinical Trial Registry (ChiCTR2300078752).
Conflicts of Interest
All authors have not any conflicts of interest to declare.
Appendix A
(a) Causality assessment of adverse event to vaginal androgen cream therapy:
1 = Definitely related
2 = Probably related
3 = Possibly related
4 = Probably unrelated
5 = Definitely unrelated
(b) Interventions taken:
1 = None
2 = Hospitalization/prolonged hospitalization
3 = Other (specify): _____
(c) Adverse event outcomes:
1 = Recovered without sequelae
2 = Recovered with sequelae
3 = Improved
4 = Unchanged/persistent
5 = Worsened
6 = Life-threatening/death

References
1. Moris L, Heesakkers J, Nitti V et al. Prevalence, diagnosis, and management of stress urinary incontinence in women: a collaborative review. Eur Urol 2025;87(3):292–301. doi:10.1016/j.eururo.2024.12.017. [Google Scholar] [PubMed] [CrossRef]
2. Nasrollahzadeh Saravi M, Mohseni M, Oskouie M et al. Exosome therapy in stress urinary incontinence: a comprehensive literature review. Biomedicines 2025;13(2):1229. doi:10.3390/biomedicines13051229. [Google Scholar] [PubMed] [CrossRef]
3. Pourebrahimi A, Khalili A, Behzadi S et al. Platelet-rich plasma for treatment of female stress urinary incontinence. Int Urol Nephrol 2025;57(2):313–321. doi:10.1007/s11255-024-04229-9. [Google Scholar] [PubMed] [CrossRef]
4. Nevine I, Katie H, Steven J et al. The effect of 12 weeks of estriol cream on stress urinary incontinence post-menopause: a prospective multinational observational study. Neurourol Urodyn 2023;42(4):799–806. doi:10.1002/nau.25165. [Google Scholar] [PubMed] [CrossRef]
5. Giulia M, Eleonora R, Maria Magdalena MG et al. Effects of vaginal DHEA on stress urinary incontinence in postmenopausal women with vulvovaginal atrophy. Observat Study 2025;196:108232. doi:10.1016/j.maturitas.2025.108232. [Google Scholar] [PubMed] [CrossRef]
6. Minassian VA, Drutz HP, Al-Badr A. Urinary incontinence as a worldwide problem. Int J Gynecol Obset 2003;82(3):327–338. doi:10.1016/s0020-7292(03)00220-0. [Google Scholar] [PubMed] [CrossRef]
7. Smith P, Heimer G, Norgren A, Ulmsten U. Localization of steroid hormone receptors in the pelvic muscles. Eur J Obstet Gynecol Repord Biol 1993;50(1):83–85. doi:10.1159/000293207. [Google Scholar] [CrossRef]
8. Zhao C, Wang Z, Xiang RY, Li L. Interaction between pelvic bone mineral density and sex steroid hormone on the risk of urinary incontinence from national health and nutrition examination survey 2013–2014. Urol Int 2022;16:1–14. doi:10.1159/000527747. [Google Scholar] [PubMed] [CrossRef]
9. Nobuo O, Machiko O. Frailty and the correlation between total testosterone levels and urinary incontinence among elderly women. Int Urogynecol J 2024;35(9):1909–1919. doi:10.1007/s00192-024-05906-0. [Google Scholar] [PubMed] [CrossRef]
10. Chen Y, Song X, Fang W et al. Correlation of serum circulating testosterone levels with stress urinary incontinence in postmenopausal women. World J Urol 2023;41:3651–3656. doi:10.1007/s00345-023-04681-9. [Google Scholar] [PubMed] [CrossRef]
11. Tapper J, Huang G, Pencina KM et al. The effects of testosterone administration on muscle areas of the trunk and pelvic floor in hysterectomized women with low testosterone levels: proof-of-concept study. Menopause 2019;26(12):1405–1414. doi:10.1097/gme.0000000000001410. [Google Scholar] [PubMed] [CrossRef]
12. Peters KM, Newman D, Belkoff L et al. Oral enobosarm shows promising activity in post-menopausal woman with stress urinary incontinence: results of a phase 2 study. In: AUA Annual Meeting; San Francisco, CA, USA; 2018. doi:10.1016/j.juro.2018.02.417 [Google Scholar] [CrossRef]
13. Luo C, Niu X. A comprehensive review of conservative therapies for female stress urinary incontinence: advancements, efficacy, and future directions. Curr Urol 2025;19(2):84–89. doi:10.1097/cu9.0000000000000270. [Google Scholar] [PubMed] [CrossRef]
14. Yang X, Wang X, Gao Z et al. The anatomical pathogenesis of stress urinary incontinence in women. Medicine 2022;59(1):5. doi:10.3390/medicina59010005. [Google Scholar] [PubMed] [CrossRef]
15. Nicholas S, Eboo V. Stress urinary incontinence and the forgotten female hormones. Int Urogynecol J 2022;33(7):1711–1716. doi:10.1007/s00192-022-05178-6. [Google Scholar] [PubMed] [CrossRef]
16. Michelle MK, Evgeniy IK. The association of serum testosterone levels and urinary incontinence in women. J Urol 2018;199(2):522–527. doi:10.1016/j.juro.2017.08.093. [Google Scholar] [PubMed] [CrossRef]
17. Kwon JK, Kim JH, Choi H et al. Voiding characteristics and related hormonal changes in peri-menopausal and post-menopausal women: a preliminary study. Maturitas 2014;79(3):311–315. doi:10.1016/j.maturitas.2014.07.011. [Google Scholar] [PubMed] [CrossRef]
18. Li W, Huang J, Chen J et al. Associations between serum testosterone levels and overactive bladder in women: a cross-sectional research of NHANES, 2011 to 2016. Int Urol Nephrol 2024;56(8):2521–2529. doi:10.1007/s11255-024-03996-9. [Google Scholar] [PubMed] [CrossRef]
19. Ponnusamy S, Sullivan RD, Thiyagarajan T, Tillmann H, Getzenberg RH, Narayanan R. Tissue selective androgen receptor modulators (SARMs) increase pelvic floor muscle mass in ovariectomized mice. J Cell Biochem 2017;118(3):640–646. doi:10.1002/jcb.25751. [Google Scholar] [PubMed] [CrossRef]
20. Copas P, Bukovsky A, Asbury B, Elder RF, Caudle MR. Estrogen, progesterone, and androgen receptor expression in levator ani muscle and fascia. J Womens Health Gend Based Med 2001;10(8):785–795. doi:10.1089/15246090152636541. [Google Scholar] [PubMed] [CrossRef]
21. Ho MH, Bhatia NN, Bhasin S. Anabolic effects of androgens on muscles of female pelvic floor and lower urinary tract. Curr Opin Obster Gynecol 2004;16(5):405–409. doi:10.1097/00001703-200410000-00009. [Google Scholar] [PubMed] [CrossRef]
22. Kudish BI, Shveiky D, Gutman RE et al. Hysterectomy and urinary incontinence in postmenopausal women. Int Urogynecol J 2014;25(11):1523–1531. doi:10.1007/s00192-014-2422-x. [Google Scholar] [CrossRef]
23. Mammadov R, Simsir A, Tuglu I, Evren V, Gurer E, Özyurt C. The effect of testosterone treatment on urodynamic findings and histopathomorphology of pelvic floor muscles in female rats with experimentally induced stress urinary incontinence. Int Urol Nephrol 2011;43(4):1003–1008. doi:10.1007/s11255-011-9938-5. [Google Scholar] [PubMed] [CrossRef]
24. Edwall L, Carlstrom K, Jonasson AF. Endocrine status and markers of collagen synthesis and degradation in serum and urogenital tissue from women with and without stress urinary incontinence. Neurourol Urodyn 2007;26(3):410–415. doi:10.1002/nau.20335. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools