 Open Access
Open Access
ARTICLE
Uretero-enteric strictures after cystectomy: revealing the modifiable risk factors
Department of Urology, Tours Regional University Hospital, Tours, 37000, France
* Corresponding Author: Ali Bourgi. Email:
(This article belongs to the Special Issue: Advances in Diagnosis and Management of Bladder Cancer: From Molecular Insights to Therapeutic Innovations)
Canadian Journal of Urology 2026, 33(1), 155-163. https://doi.org/10.32604/cju.2025.069546
Received 25 June 2025; Accepted 25 September 2025; Issue published 28 February 2026
Abstract
Objectives: Uretero-enteric strictures are feared complications following cystectomy. Despite surgical advancements, particularly the rise of robot-assisted approaches, the risk factors associated with these strictures remain poorly defined. This study aimed to identify the risk factors associated with uretero-enteric anastomotic strictures after cystectomy, according to the surgical approach and type of urinary diversion (extracorporeal vs. intracorporeal). Methods: We conducted a single-center retrospective study including 340 patients who underwent cystectomy between 2016 and 2024 at Tours University Hospital. Clinical, biological, perioperative, and postoperative data were analyzed. The occurrence of a uretero-ileal anastomotic stricture was defined radiologically by a uretero-hydronephrosis ≥20 mm. We constructed and analyzed a learning curve for robotic surgery with intracorporeal urinary diversion. Results: Strictures occurred in 60 patients (17.6%). On multivariable analysis, reduced preoperative glomerular filtration rate (odds ratio [OR] = 1.45 per 10 mL/min decrease, 95% CI [1.12–1.87], p = 0.004), elevated creatinine (OR = 1.30 per 10 µmol/L increase, 95% CI [1.05–1.61], p = 0.018), prior myocardial infarction (OR = 2.25, 95% CI [1.10–4.62], p = 0.027), and postoperative urinary tract infection (OR = 3.10, 95% CI [1.65–5.82], p < 0.001) were independent predictors. Most strictures were left-sided. Intracorporeal robotic diversion had a higher, though non-significant, stricture rate (21.5% vs. 15.2%, OR = 1.52, p = 0.12). Stricture rates fell markedly after 20 robotic cases per surgeon (23.8% vs. 12.1%). Conclusion: Uretero-enteric strictures are multifactorial, strongly influenced by baseline renal function, cardiovascular comorbidity, and postoperative infection. Robotic intracorporeal diversion shows a learning curve effect, underlining the importance of surgical expertise and infection prevention in reducing risk.Keywords
Cystectomy with urinary diversion remains the standard treatment for muscle-invasive bladder tumors,1 but it is also indicated in benign conditions such as treatment-refractory neurogenic bladders.2,3 Regardless of the indication, cystectomy is associated with significant postoperative morbidity, often exceeding 50% in some series.4–7 While some observational studies suggest fewer major complications with robotic approaches, randomized trials have not demonstrated significant differences.8 Among the medium to long-term complications, there is uretero-enteric anastomotic stricture, which can lead to repeated hospitalizations, upper urinary tract infections, or long-term deterioration of renal function. Known predictors of uretero-enteric strictures include urine leak and a complicated postoperative course, as reported by Richards et al. 9
The rate of uretero-enteric stricture ranges from 3% to 10% with the open approach10–12 and can reach up to 25% with robotic surgery, depending on whether the urinary diversion is performed extracorporeally or intracorporeally.13,14 It most often occurs within 5 to 10 months,15,16 and in most cases on the left side.16 The gold standard treatment for uretero-enteric anastomotic stricture remains open uretero-enteric reimplantation. However, this technique is associated with significant morbidity. The anastomotic technique does not appear to significantly influence the incidence of strictures.10,11 The main hypotheses proposed include tension on the anastomosis, ureteral ischemia, extensive ureterolysis, or postoperative infections.12,17
The literature remains heterogeneous, with small sample sizes and predominantly single-center studies. At present, no clear consensus exists regarding the predictive factors for uretero-enteric strictures. In this context, our study aimed to explore the risk factors associated with the occurrence of uretero-enteric strictures following cystectomy, through a retrospective single-center analysis conducted at Tours University Hospital. The primary objective of the study was to identify the risk factors for uretero-enteric stricture according to the surgical approach used. The secondary objectives were to determine the risk factors for uretero-enteric stricture based on whether the urinary diversion was performed extracorporeally or intracorporeally.
We retrospectively collected data on all patients who underwent cystectomy, regardless of the surgical indication (oncologic or non-oncologic) and the surgical approach used (open or robotic), at Tours University Hospital between January 2016 and December 2024.
This study was approved by the Institutional Review Board of Tours University Hospital and was declared to the French National Commission on Informatics and Liberty (CNIL, approval number 2025_034). It was conducted in accordance with the principles of the Declaration of Helsinki. Given its retrospective design, the requirement for individual informed consent was waived.
Data collection and study design
Demographic data (age, sex, BMI [body mass index], smoking status), medical history (diabetes, hypertension, myocardial infarction), ongoing antiplatelet or anticoagulant treatments, Charlson comorbidity index, ASA (American Society of Anesthiologists) score, history of radiotherapy or neoadjuvant chemotherapy for bladder cancer, as well as biological data (serum creatinine, glomerular filtration rate, hemoglobin, and urine culture results) were collected.
Perioperative and postoperative variables (surgeon experience, oncologic vs. non-oncologic indication, type of urinary diversion, surgical approach, type of urinary anastomosis, operative time, and postoperative complications) were also collected in an anonymized manner from medical records using the Common Classification of Medical Procedures (CCAM) coding system.
Non-oncologic indications included neurogenic bladder and complex lower urinary tract dysfunction.
The surgical techniques for cystectomy were similar among surgeons regardless of whether the approach was open or robotic; however, differences existed in the method of urinary diversion. All patients received intraoperative ureteral stents for anastomotic support. Suture materials included PDS 4.0 or 5.0 and Monocryl 4.0 (Ethicon, Inc., Somerville, NJ, USA), applied using either running or interrupted techniques based on surgeon preference. The length of the ureter resected was not uniformly recorded, but generally aimed to preserve distal vascularization while ensuring tension-free anastomosis. In the robotic group, the urinary diversion was performed either extracorporeally or intracorporeally. The choice of neobladder type (Studer, Hautmann, Foch, and Camey) was based on surgeon expertise and intraoperative anatomical considerations.
We defined a uretero-enteric stricture as the presence of postoperative uretero-hydronephrosis, identified on computed tomography (CT) imaging by dilation of the ureter and renal pelvis with an anteroposterior diameter greater than 20 mm, associated with delayed excretion on delayed-phase CT.
Bilateral strictures were identified when imaging demonstrated hydronephrosis and delayed excretion on both sides. All patients included in the study underwent a preoperative CT scan and a postoperative scan at 3 months if the indication for cystectomy was oncologic. For patients who underwent cystectomy for non-oncologic indications, a postoperative CT scan was performed only if they presented with flank pain, recurrent urinary tract infections, impaired renal function, or unexplained fever.
Postoperative urinary tract infection was defined as a positive urine culture with clinical signs of infection.
Statistical analysis was performed using RStudio software (version 2023.06.1+524). Continuous quantitative variables were described as mean ± standard deviation (SD) when normally distributed (assessed by graphical inspection and the Shapiro-Wilk test), or as median values when the distribution was non-Gaussian. Group comparisons were conducted using the Student’s t-test for normally distributed continuous variables, or the Mann–Whitney test for non-parametric variables.
Qualitative variables were expressed as counts and percentages. Comparisons were made using Pearson’s chi-squared test or Fisher’s exact test when expected frequencies were less than 5 in more than 20% of the cells. To analyze stricture rates according to surgical experience, a learning curve was constructed, and the area under the curve (AUC) was calculated. A p-value of <0.05 was considered statistically significant for all tests.
Among the 340 patients included, 60 (17.6%) developed a uretero-enteric anastomotic stricture (Table 1). The mean age of the patients was 67.2 ± 11.6 years, with no significant difference between those who developed a stricture and those who did not. No significant differences were observed regarding sex, BMI, Charlson Comorbidity Index, or ASA score. In univariate analysis, preoperative glomerular filtration rate (GFR) was significantly lower in patients who developed a stricture (68.5 ± 23.6 mL/min/1.73 m2 vs. 75.4 ± 27.1 mL/min/1.73 m2; p = 0.048), and preoperative serum creatinine was higher (113.0 ± 68.2 µmol/L vs. 96.5 ± 47.7 µmol/L; p = 0.074) (Table 1).
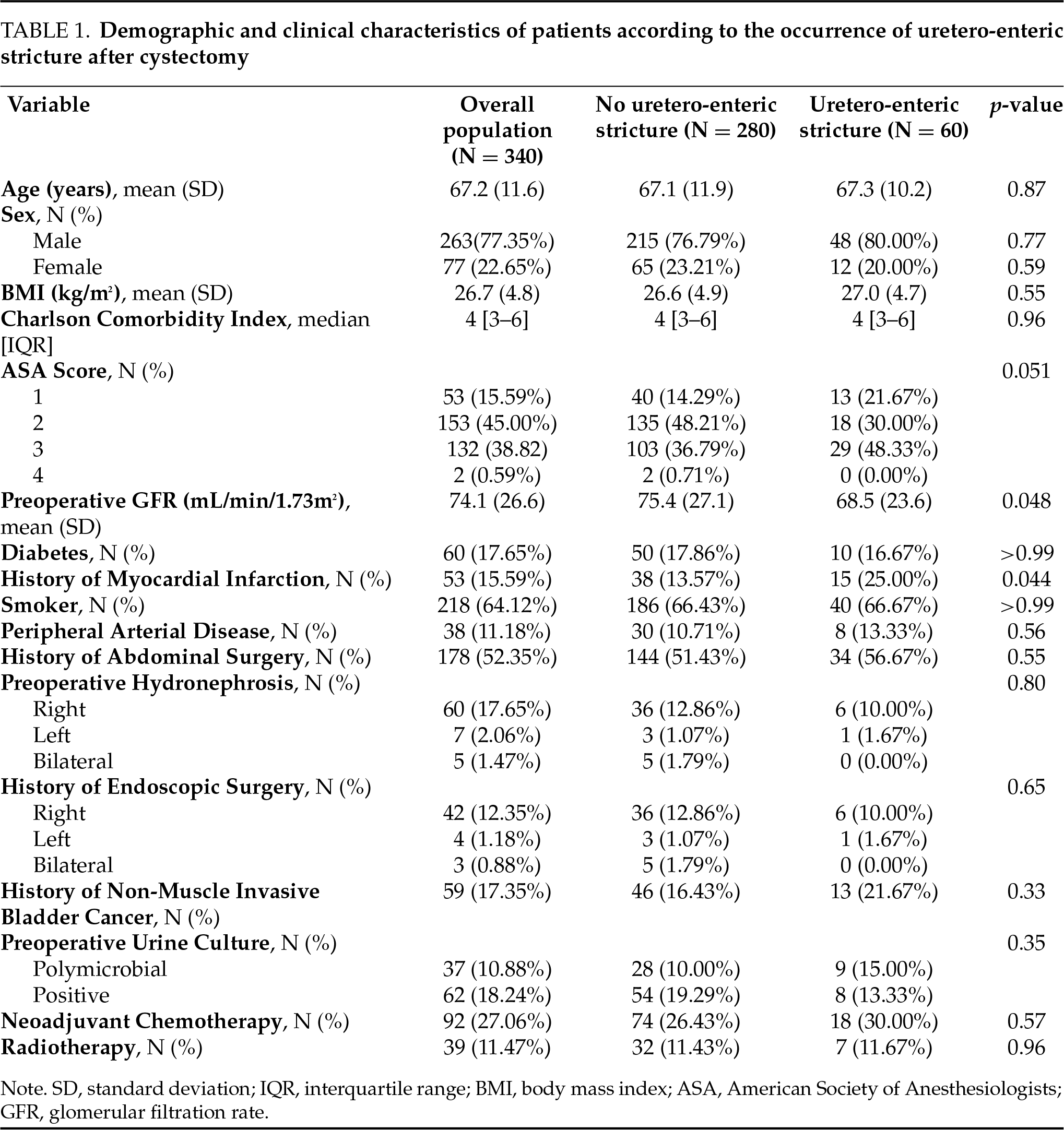
Patients who developed a uretero-enteric anastomotic stricture more frequently had a history of myocardial infarction (25% vs. 14%, p = 0.044) (Table 1). Perioperative and postoperative characteristics showed no significant differences between the groups.
Among the 170 patients who underwent robotic surgery, 36 (21.2%) developed a uretero-enteric stricture. Among the 170 patients treated via open surgery, 24 (14.1%) developed a stricture. Although the stricture rate was numerically higher in the robotic group, this difference did not reach statistical significance (p = 0.088) (Table 2). Postoperative characteristics revealed a significantly higher incidence of urinary tract infections in the stricture group (48% vs. 34%, p = 0.035) (Table 3).
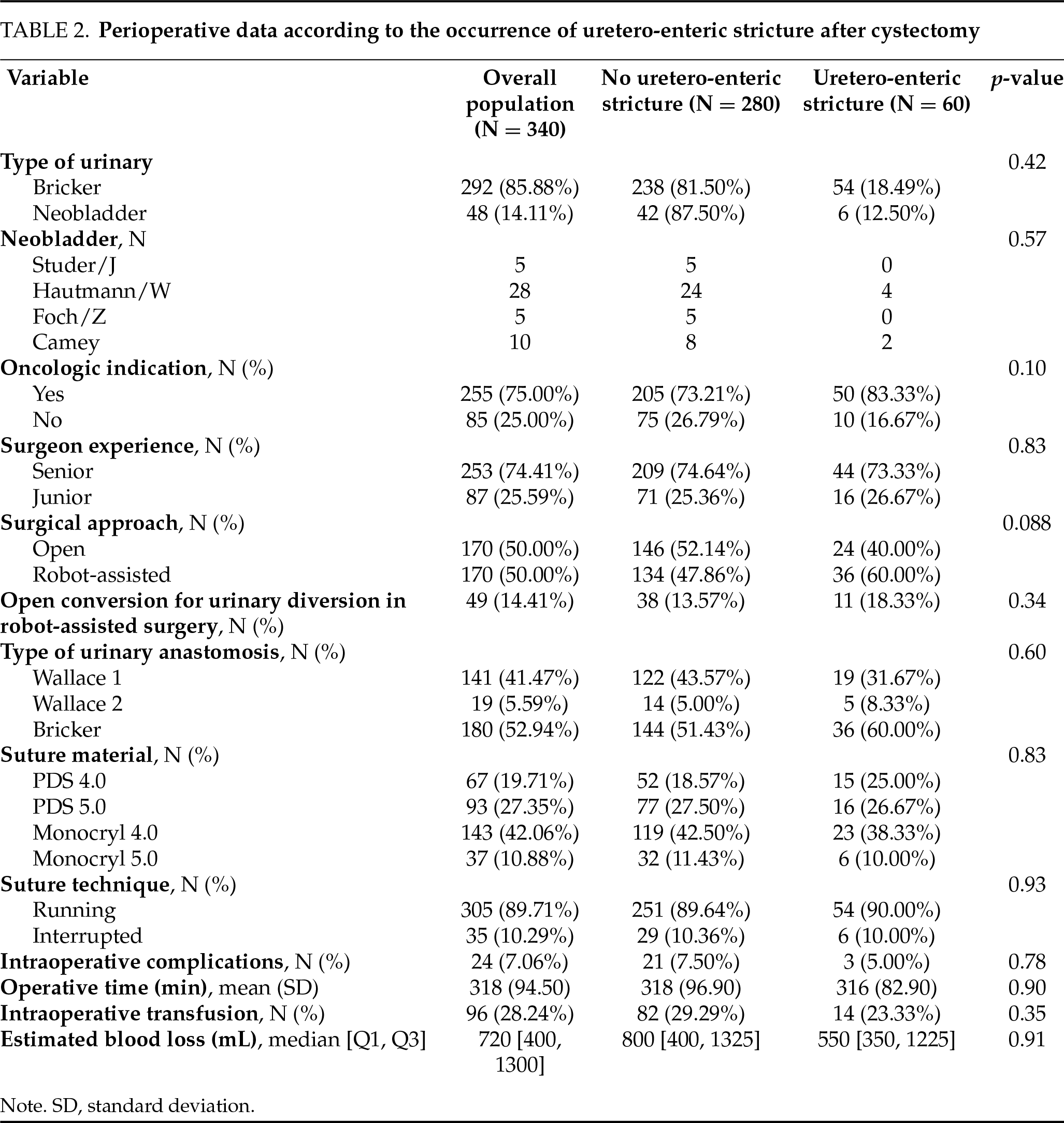
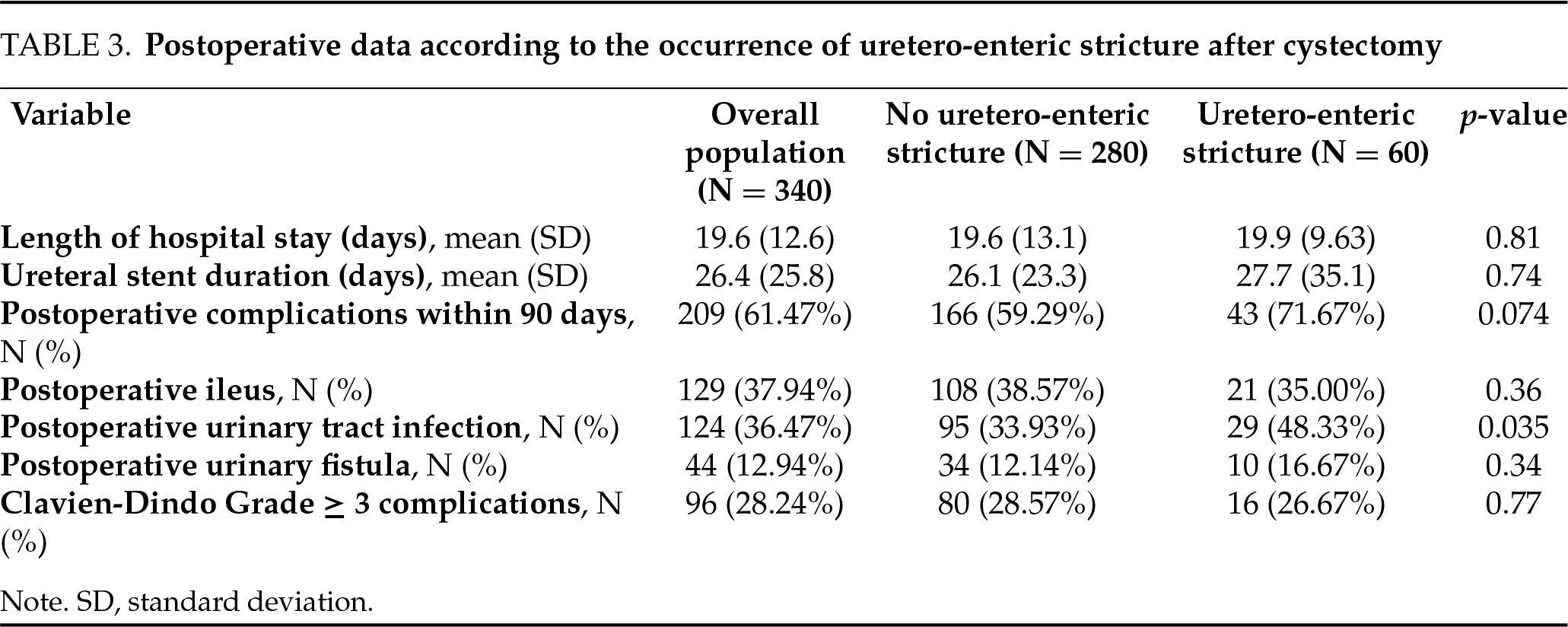
On multivariable analysis, reduced preoperative glomerular filtration rate was associated with an increased risk of stricture (OR = 1.45 per 10 mL/min decrease, 95% CI [1.12–1.87], p = 0.004). Elevated creatinine was also predictive (OR = 1.30 per 10 µmol/L increase, 95% CI [1.05–1.61], p = 0.018). A history of myocardial infarction independently increased the risk (OR = 2.25, 95% CI [1.10–4.62], p = 0.027). Finally, postoperative urinary tract infection strongly predicted stricture formation (OR = 3.10, 95% CI [1.65–5.82], p < 0.001).
Stricture location was categorized as right-sided, left-sided, or bilateral based on radiological findings. These were then stratified according to surgical approach (open, intracorporeal robotic, extracorporeal robotic) (Table 4). Strictures were more frequently located on the left side, particularly after robot-assisted cystectomy with intracorporeal urinary diversion (65.4%) or extracorporeal diversion (60%), compared to 37.5% in open surgery. Bilateral strictures were observed in 37.5% of open cases, 15.4% in intracorporeal robotic cases, and 40% in extracorporeal robotic cases. Right-sided strictures were less common, especially in the extracorporeal robotic group, where no cases were reported. The distribution of uretero-enteric strictures by location (right, left, bilateral) did not differ significantly among the three surgical approaches (open, intracorporeal robotic, extracorporeal robotic), with p-values of >0.99 for right-sided strictures, 0.128 for left-sided strictures, and 0.149 for bilateral strictures.
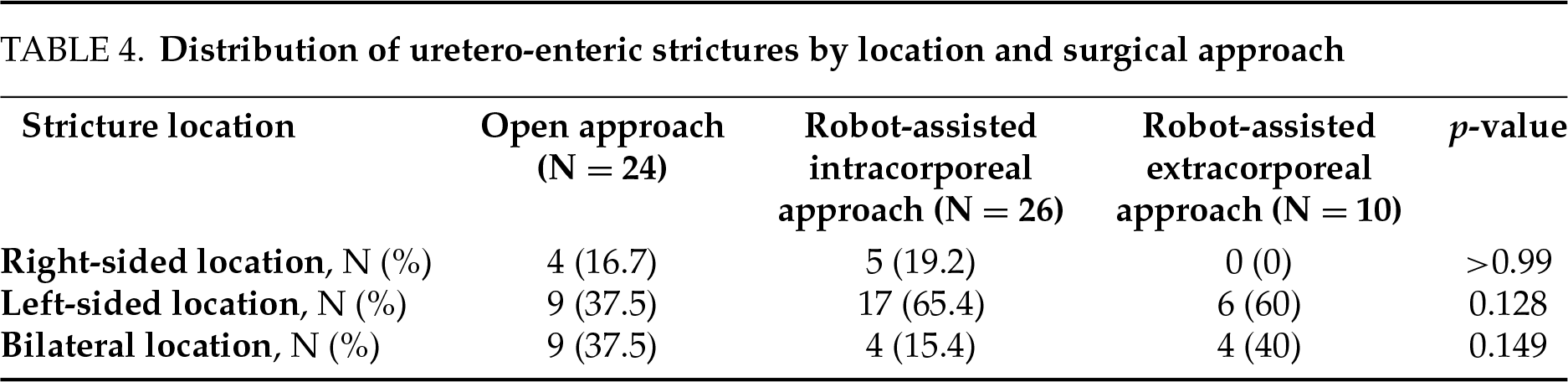
When comparing the stricture locations between the two robotic techniques (intracorporeal vs. extracorporeal), no significant differences were observed for right-sided (p = 0.293), left-sided (p > 0.99), or bilateral strictures (p = 0.179). However, it is noteworthy that no right-sided strictures were observed in the extracorporeal robotic group. The overall distribution of uretero-enteric strictures (right, left, bilateral) did not differ significantly according to the surgical approach (p = 0.175).
The rate of uretero-enteric strictures decreased after the initial learning phase, defined here as the first 20 cases performed by each surgeon. For surgeon 1, the stricture rate dropped from 35% during the first 20 cases to 10% thereafter. A similar trend was observed for surgeon 2, with a decrease from 30% to 9% (Table 5). The graphical representation of this trend (Figure 1) demonstrates an overall reduction in stricture rate with increasing experience, consistent with a favorable learning curve. The AUC for stricture rate were 0.225 for surgeon 1 and 0.195 for surgeon 2. However, this trend did not reach statistical significance (p = 0.130).
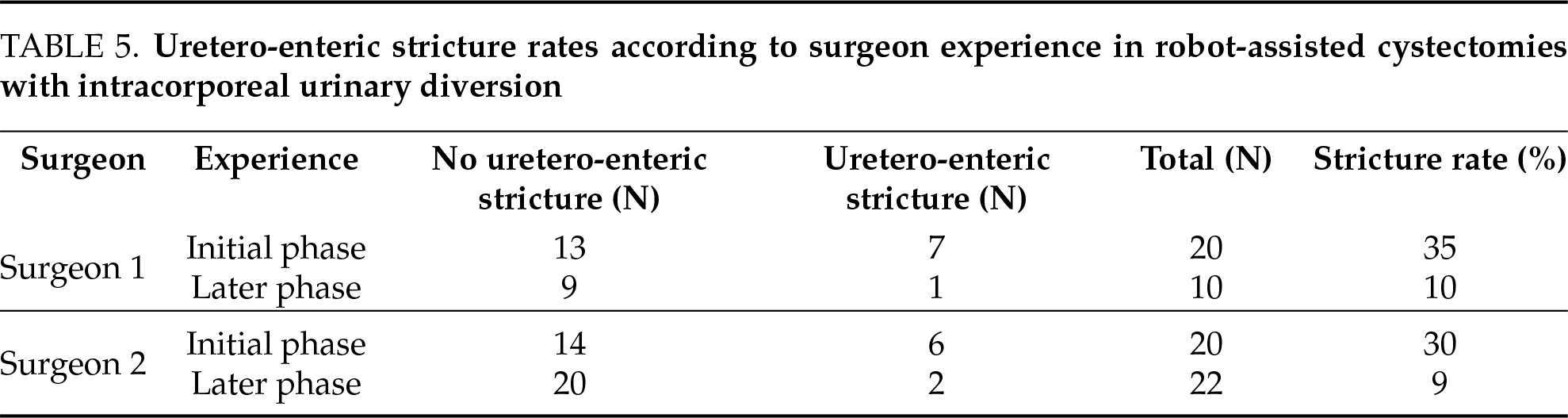
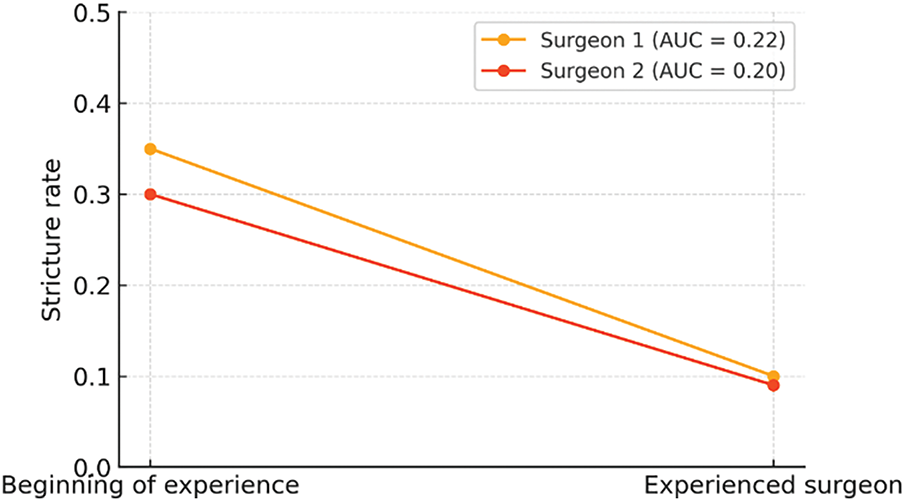
FIGURE 1. Learning curve and stricture rate in robot-assisted surgery with intracorporeal urinary diversion
Our study confirms that uretero-enteric strictures following cystectomy are a common complication, occurring in 17.6% of cases. Despite technical advancements—particularly the development of robot-assisted surgery—several risk factors remain concerning. Cystectomy, regardless of the surgical approach, remains a procedure associated with significant morbidity, affecting up to one in two patients.4,6,7
Recent data show that while robot-assisted radical cystectomy (RARC) is associated with lower intraoperative blood loss, its trifecta and pentafecta outcomes—encompassing oncological and perioperative criteria—are comparable to those of open radical cystectomy.18 Uretero-enteric anastomotic strictures represent one of the most feared medium- to long-term complications. They typically occur within 5 to 10 months postoperatively, with reported frequencies ranging from 3% to 25% depending on the series.15,16 In our cohort, we observed similar findings, with an early postoperative complication rate of 61% and a stricture rate of 17.6%.
Bizzarri et al.19 reported that low preoperative albumin levels, elevated fibrinogen, and a history of abdominal surgery were significantly associated with an increased risk of benign uretero-enteric strictures following radical cystectomy. Several pathophysiological hypotheses have been proposed, including distal ureteral ischemia, urinary tract infections, fistulas, surgical approach, obesity, and neoadjuvant chemotherapy.
In our series, we observed a significant association between reduced preoperative glomerular filtration rate and the occurrence of uretero-enteric strictures. Similarly, a history of myocardial infarction was also associated with a higher risk of stricture.
These findings support the ischemic and vascular hypothesis in the development of strictures, likely linked to distal ureteral ischemia. This aligns with the findings of Brandt et al.,20 who reported a correlation between left-sided strictures and elevated BMI, and between bilateral strictures and smoking.
Although urinary tract infections (UTIs) were more common in patients who developed strictures, the retrospective nature of our study does not allow us to establish causality. It is possible that early subclinical narrowing of the anastomosis may have led to impaired drainage and subsequently to infection, rather than infection being the initial cause. However, the inflammatory and fibrotic cascade triggered by infection may further exacerbate stricture progression. The anastomotic site is particularly vulnerable to such complications due to surgical manipulation, devascularization, and urine exposure—factors that do not typically apply to the native ureter.
Most uretero-enteric strictures are reported to occur on the left side in the literature,15,16,21,22 and this was also the case in our study. This left-sided predominance may be explained by the more extensive mobilization required for the left ureter, which must pass beneath the sigmoid mesentery, thereby increasing the risk of distal ischemia. This observation is supported by the study of Ramahi et al.,23 which reported a left-sided predominance (46%) regardless of the type of urinary diversion performed.
The choice between a Wallace or Bricker uretero-enteric anastomosis was based on surgeon preference. A retrospective meta-analysis including three open cystectomy series and one robot-assisted series found no significant difference in the uretero-enteric stricture rate according to the type of anastomosis, consistent with our findings.10 Additional data, such as that from Das et al.,21 indicate that longer distal resections—resulting in shorter ureters—were associated with a lower risk of uretero-enteric stricture (OR = 0.73, p = 0.028). As also reported in the study by Ramahi et al.,23 our series showed a significantly higher rate of postoperative urinary tract infections in patients who developed a uretero-enteric anastomotic stricture. UTIs (urinary tract infections) may act both as early markers of underlying strictures and as contributing factors through inflammation and fibrosis.
Our findings suggest a steep learning curve for robotic intracorporeal diversion, highlighting the importance of supervision during early training phases.
The stricture rate was higher among patients who underwent robotic cystectomy (21.2%) compared to those who underwent open surgery (14.1%), although this difference did not reach statistical significance (p = 0.088). This trend may reflect a learning curve effect in robotic intracorporeal urinary diversion, as supported by our surgeon-specific data. However, the analysis of the learning curve revealed a marked reduction in stricture rates beyond the first 20 cases for our two most experienced surgeons. These findings are consistent with those of Ericson et al.,13 who identified a threshold of 75 cases. In their study, the stricture rate in the intracorporeal robot-assisted group dropped from 17.5% to 4.9%. Moreover, they found that for every additional 10 cases performed, the risk of stricture decreased further.
In addition to experience, the absence of haptic feedback in robotic surgery—which limits the tactile perception of tissue resistance—may lead to a more aggressive dissection of the ureter, increasing the risk of thermal injury or vascular devascularization, particularly during the early learning phase.23 These findings highlight the technical complexity of robot-assisted cystectomy with intracorporeal urinary diversion, particularly during the early stages of the learning curve.
The literature reports that intraoperative use of indocyanine green (ICG) may help assess ureteral perfusion. However, data from Carbonell et al.,24 involving more than 220 patients who underwent robot-assisted cystectomy with intracorporeal diversion, did not show a significant reduction in stricture rates with the use of ICG (18.3% vs. 19% in the non-ICG group; p = 0.901). These results contrast with other studies,25,26 which demonstrated a significant reduction in stricture rates with ICG, particularly in open surgery.26
Our practice of using intraoperative ureteral stents can also be questioned. Although stents are often used to secure uretero-enteric anastomoses, data from Donat et al.27 showed an increased rate of infectious complications in stented patients (32% vs. 14% in the non-stented group; p = 0.003). In the study by Ramahi et al.,23 stent placement was significantly associated with uretero-enteric strictures (OR = 2.27, p = 0.05).
Moreover, a 2021 meta-analysis that included four comparative studies evaluating uretero-enteric strictures and urinary fistulas in patients with or without ureteral stents showed a higher stricture rate in stented patients (OR = 1.64, 95% CI [0.88–3.05], p = 0.12), although statistical significance was not reached.28 These findings suggest that ureteral stents may represent a potential risk factor for stricture formation. Donat et al. also demonstrated that 30-day complication rates were significantly lower in patients who did not receive ureteral stents during open cystectomy.27
Our study does present several limitations and potential biases, primarily due to its retrospective design. Being a single-center study, the generalizability of our findings is limited. In addition, heterogeneous follow-up and variability in surgical practices represent further limitations. Nevertheless, the size of our cohort, the comprehensive analysis of clinical, perioperative, and postoperative factors, and the consistency of our findings with the existing literature support the relevance of our study in evaluating the risk of uretero-enteric strictures.
As all patients in our cohort received double-J stents, our findings cannot inform the debate on stentless anastomosis. Future studies specifically designed to compare stented vs. stentless techniques are warranted.
Uretero-enteric strictures following cystectomy are common, multifactorial, and sometimes preventable complications. Our study highlights the importance of addressing modifiable risk factors such as postoperative urinary tract infections and underlying vascular conditions, alongside the role of surgical experience. Supervision of junior surgeons, standardization of techniques, and critical evaluation of intraoperative ureteral stent use appear to be key areas for improvement.
Acknowledgement
The authors would like to thank all the teams at Tours University Hospital for their involvement in conducting this study.
Funding Statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author Contributions
Conceptualization and study design: Abdelkader Akkad, Ali Bourgi; Data collection: Abdelkader Akkad; Statistical analysis: Abdelkader Akkad, Ali Bourgi; Manuscript drafting: Abdelkader Akkad, Franck Bruyere; Critical revision of the manuscript: Abdelkader Akkad, Franck Bruyere, Ali Bourgi; Final approval of the version to be published: Abdelkader Akkad, Franck Bruyere, Ali Bourgi. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials
The datasets generated and analyzed during the current study are not publicly available due to patient confidentiality, but are available from the corresponding author on reasonable request.
Ethics Approval
This study was approved by the Institutional Review Board of Tours University Hospital and was declared to the French National Commission on Informatics and Liberty (CNIL, approval number 2025_034). It was conducted in accordance with the principles of the Declaration of Helsinki.
Informed Consent
Given its retrospective design, the requirement for individual informed consent was waived.
Conflicts of Interest
The authors declare no conflicts of interest to report regarding the present study.
References
1. Roumiguié M, Marcq G, Neuzillet Y et al. Recommandations françaises du Comité de cancérologie de l’AFU – Actualisation 2024–2026: tumeurs de la vessie infiltrant le muscle (TVIM). Prog Urol FMC 2024;34(7):F296–F312. (In French). doi:10.1016/j.fpurol.2024.07.211. [Google Scholar] [CrossRef]
2. European Association of Urology. EAU Guidelines. In: EAU Annual Congress; 2025; Madrid, Spain. [Google Scholar]
3. Chkir S, Michel F, Akakpo W et al. Non-continent urinary diversion (ileal conduit) as salvage therapy in patients with refractory lower urinary tract dysfunctions due to multiple sclerosis: results of a national cohort from the French Association of Urology (AFU) Neurourology Committee and the French-speaking Neurourology Study Group (GENULF). Urology 2022;168:216–221. doi:10.1016/j.urology.2022.06.014. [Google Scholar] [PubMed] [CrossRef]
4. Albisinni S, Oderda M, Fossion L et al. The morbidity of laparoscopic radical cystectomy: analysis of postoperative complications in a multicenter cohort by the European Association of Urology (EAU)-Section of Uro-Technology. World J Urol 2016;34(2):149–156. doi:10.1007/s00345-015-1633-1. [Google Scholar] [PubMed] [CrossRef]
5. Stein JP, Lieskovsky G, Cote R et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1054 patients. J Clin Oncol 2001;19(3):666–675. doi:10.1200/jco.2001.19.3.666. [Google Scholar] [PubMed] [CrossRef]
6. Sakhri R, Seigle-Murandi F, Jacqmin D, Lang H, Saussine C. Cystectomie cœlioscopique et urétérostomie transiléale pour vessies neurologiques et assimilées: morbidité et amélioration de la qualité de vie des patients. Prog Urol 2015;25(6):342–347. (In French). doi:10.1016/j.purol.2015.01.022. [Google Scholar] [CrossRef]
7. Shabsigh A, Korets R, Vora KC et al. Defining early morbidity of radical cystectomy for patients with bladder cancer using a standardized reporting methodology. Eur Urol 2009;55(1):164–174. doi:10.1016/j.eururo.2008.07.031. [Google Scholar] [PubMed] [CrossRef]
8. Parekh DJ, Reis IM, Castle EP et al. Robot-assisted radical cystectomy versus open radical cystectomy in patients with bladder cancer (RAZORan open-label, randomised, phase 3, non-inferiority trial. Lancet 2018;391:2525–2536. doi:10.1016/j.eururo.2018.09.014. [Google Scholar] [PubMed] [CrossRef]
9. Richards KA, Cohn JA, Large MC, Bales GT, Smith ND, Steinberg GD. The effect of length of ureteral resection on benign ureterointestinal stricture rate in ileal conduit or ileal neobladder urinary diversion following radical cystectomy. Urol Oncol 2015;33:65.e1–65.e6.11. doi:10.1016/j.urolonc.2014.05.015. [Google Scholar] [PubMed] [CrossRef]
10. Davis NF, Burke JP, McDermott TED, Flynn R, Manecksha RP, Thornhill JA. Bricker versus Wallace anastomosis: a meta-analysis of ureteroenteric stricture rates after ileal conduit urinary diversion. Can Urol Assoc J 2015;9(5–6):E284–E290. doi:10.5489/cuaj.2692. [Google Scholar] [PubMed] [CrossRef]
11. Delaume A, Védrine N, Guandalino M et al. Comparaison des anastomoses Bricker et Wallace dans les urétérostomies cutanées trans-iléales: étude rétrospective, multicentrique. Prog Urol 2016;26(1):58–64. doi:10.1016/j.purol.2015.09.009. [Google Scholar] [CrossRef]
12. Alonso Mediavilla E, Campos-Juanatey F, Azcárraga Aranegui G et al. Ureteroileal anastomosis stricture after urinary diversions performed by open, laparoscopic and robotic approaches. Actas Urol Esp (Engl Ed) 2022;46(1):49–56. doi:10.1016/j.acuroe.2021.06.010. [Google Scholar] [PubMed] [CrossRef]
13. Ericson KJ, Thomas LJ, Zhang JH et al. Uretero-enteric anastomotic stricture following radical cystectomy: a comparison of open, robotic extracorporeal, and robotic intracorporeal approaches. Urology 2020;144:130–135. doi:10.1016/j.urology.2020.06.047. [Google Scholar] [PubMed] [CrossRef]
14. Sahni DS, Oberoi AS, Ramsaha S, Teahan S, Morton S. Incidence of ureteroenteric anastomotic strictures after robotic vs open cystectomy in adults. Urology 2024;185:100–108. doi:10.1016/j.urology.2024.01.010. [Google Scholar] [PubMed] [CrossRef]
15. Shah SH, Movassaghi K, Skinner D et al. Ureteroenteric strictures after open radical cystectomy and urinary diversion: the University of Southern California experience. Urology 2015;86(1):87–91. doi:10.1016/j.urology.2015.03.014. [Google Scholar] [PubMed] [CrossRef]
16. Ahmed YE, Hussein AA, May PR et al. Natural history, predictors and management of ureteroenteric strictures after robot assisted radical cystectomy. J Urol 2017;198(3):567–574. doi:10.1016/j.juro.2017.02.3339. [Google Scholar] [PubMed] [CrossRef]
17. Benson CR, Ajay D, Barrett-Harlow BL et al. Ureteroenteric anastomosis in orthotopic neobladder creation: do urinary tract infections impact stricture rate? World J Urol 2021;39(4):1171–1176. doi:10.1007/s00345-020-03266-0. [Google Scholar] [PubMed] [CrossRef]
18. Gavi F, Foschi N, Fettucciari D et al. Assessing trifecta and pentafecta success rates between robot-assisted vs open radical cystectomy: a propensity score-matched analysis. Cancers 2024;16:1270. doi:10.3390/cancers16071270. [Google Scholar] [PubMed] [CrossRef]
19. Bizzarri FP, Campetella M, Russo P et al. Risk factors for benign uretero-enteric anastomotic strictures after open radical cystectomy and ileal conduit. Urologia 2025;92:224–230. doi:10.1177/03915603241303634. [Google Scholar] [PubMed] [CrossRef]
20. Brandt SB, Ibsen L, Lam GW, Bøttcher M, Kingo PS, Jensen JB. Ureteroenteric strictures after cystectomy: side-specific risk factors and radiological assessment. BJUI Compass 2024;5(7):813–822. [Google Scholar]
21. Das A, Zeng E, Risk M et al. Shorter ureters lead to fewer strictures following cystectomy and urinary diversion. Urology 2024;184:272–277. doi:10.1016/j.urology.2023.12.005. [Google Scholar] [PubMed] [CrossRef]
22. McNicholas DP, El-Taji O, Siddiqui Z, Hanchanale V. Systematic review comparing uretero-enteric stricture rates between open cystectomy with ileal conduit, robotic cystectomy with extra-corporeal ileal conduit and robotic cystectomy with intra-corporeal ileal conduit formation. J Robot Surg 2024;18(1):100. doi:10.1007/s11701-024-01850-9. [Google Scholar] [PubMed] [CrossRef]
23. Ramahi YO, Shiekh M, Shah AA et al. Uretero-enteric strictures after robot assisted radical cystectomy: prevalence and management over two decades. Clin Genitourin Cancer 2023;21(2):e19–e26. doi:10.1016/j.clgc.2022.10.006. [Google Scholar] [PubMed] [CrossRef]
24. Carbonell E, Sierra A, Mercader C et al. Intravenous indocyanine green to evaluate distal ureteral vascularity during robot-assisted radical cystectomy with intracorporeal urinary diversion. World J Urol 2024;42(1):568. doi:10.1007/s00345-024-05284-8. [Google Scholar] [PubMed] [CrossRef]
25. Ahmadi N, Ashrafi AN, Hartman N et al. Use of indocyanine green to minimise uretero-enteric strictures after robotic radical cystectomy. BJU Int 2019;124(2):302–307. doi:10.1111/bju.14733. [Google Scholar] [PubMed] [CrossRef]
26. Yeaman C, Ignozzi G, Kazeem A, Isharwal S, Krupski TL, Culp SH. Impact of SPY fluorescence angiography on incidence of ureteroenteric stricture after urinary diversion. J Urol 2024;212(6):844–850. doi:10.1097/ju.0000000000004198. [Google Scholar] [PubMed] [CrossRef]
27. Donat SM, Tan KS, Jibara G, Dalbagni G, Carlon VA, Sandhu J. Intra-operative ureteral stent use at radical cystectomy is associated with higher 30-day complications rates. J Urol 2021;205(2):483–490. doi:10.1097/ju.0000000000001451. [Google Scholar] [PubMed] [CrossRef]
28. Peng YL, Ning K, Wu ZS et al. Ureteral stents cannot decrease the incidence of ureteroileal anastomotic stricture and leakage: a systematic review and meta-analysis. Int J Surg 2021;93:106058. doi:10.1016/j.ijsu.2022.106657. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools