 Open Access
Open Access
REVIEW
HBx Protein in Hepatitis B Virus-Related Hepatocellular Carcinoma: Pathogenic Mechanisms and Emerging Interventions
1 Department of Research, Taipei Tzu Chi Hospital, The Buddhist Tzu Chi Medical Foundation, New Taipei City, 231, Taiwan
2 Department of Internal Medicine, Cathay General Hospital, Taipei, 106, Taiwan
3 Taipei Fuhsing Private School, Taipei, 106, Taiwan
4 Department of Dentistry, Taipei Tzu Chi Hospital, The Buddhist Tzu Chi Medical Foundation, New Taipei City, 231, Taiwan
5 Taipei American School, Taipei, 111, Taiwan
6 Division of Nephrology, Department of Internal Medicine, Sijhih Cathay General Hospital, New Taipei City, 221, Taiwan
7 Department of Medical Research, Cathay General Hospital, Taipei, 106, Taiwan
8 Institute of Oral Medicine and Materials, College of Medicine, Tzu Chi University, Hualien, 970, Taiwan
* Corresponding Authors: Po-Chih Hsu. Email: ; Chan-Yen Kuo. Email:
# These authors contributed equally to this work
BIOCELL 2026, 50(3), 1 https://doi.org/10.32604/biocell.2025.073698
Received 23 September 2025; Accepted 21 November 2025; Issue published 23 March 2026
Abstract
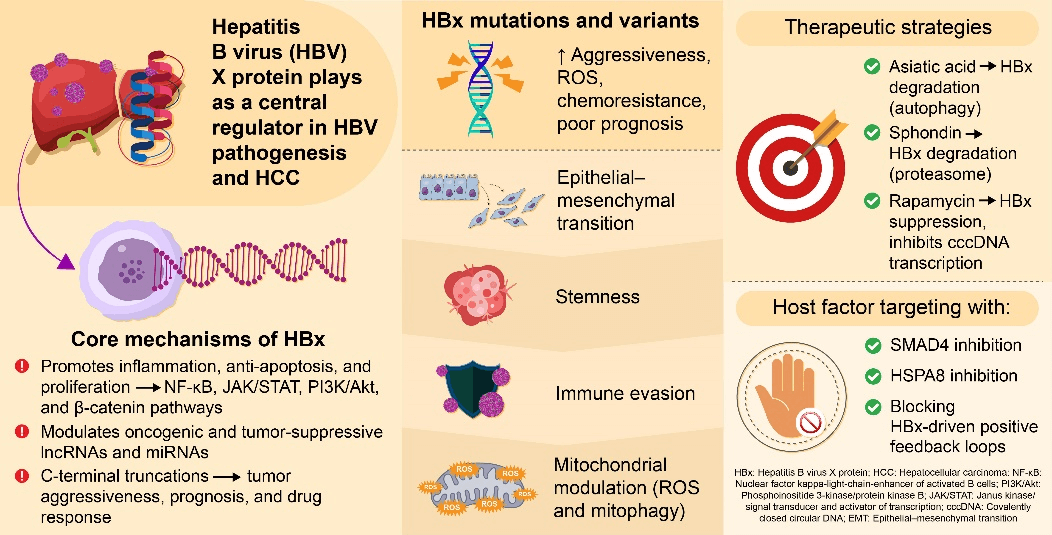
Hepatocellular carcinoma (HCC) is a leading cause of cancer-related death worldwide, most commonly driven by chronic hepatitis B virus (HBV) infection. The HBV X protein (HBx) plays a central role in hepatocarcinogenesis by regulating transcription, signal transduction, epigenetic modification, and interactions with noncoding RNAs. This review summarizes current advances in HBx-mediated signaling pathways and mutation-specific functions, highlighting its potential as a prognostic biomarker and therapeutic target, and providing insights for future strategies in HCC treatment and HBV eradication. Activation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), cAMP response element binding protein/activating transcription factor (CREB/ATF), and phosphatidylinositol 3′-kinase/AKT serine-threonine protein kinase family (PI3K/Akt) pathways by HBx promotes tumor proliferation, epithelial–mesenchymal transition, and immune evasion. Mutation- and truncation-specific variants of HBx, such as C1485T, C1653T, and K130M/V131I, further enhance oxidative stress, inflammatory signaling, and chemoresistance, contributing to poor prognosis. Emerging preclinical evidence indicates that natural compounds, including asiatic acid, sphondin, and rapamycin, can suppress HBx stability and transcriptional activity, offering novel antiviral and antitumor strategies. Understanding HBx-driven molecular mechanisms and mutation-specific effects may guide the development of precise diagnostic, prognostic, and therapeutic approaches for HBV-related HCC.Graphic Abstract
Keywords
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools