 Open Access
Open Access
ARTICLE
Preparation of ZnO Nanoparticles Incorporated into Cross-Linked Poly (Methyl Methacrylate) (PMMA) Polymer and Its Biological Application
Chemistry Department, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia
* Corresponding Author: Nazeeha S. Alkayal. Email:
(This article belongs to the Special Issue: Cellulose and Nanocellulose in Polymer Composites: Sustainable Engineering Approach)
Journal of Polymer Materials 2026, 43(1), 19 https://doi.org/10.32604/jpm.2026.073588
Received 21 September 2025; Accepted 23 January 2026; Issue published 03 April 2026
Abstract
The development of polymer nanoparticle composites with enhanced thermal and antibacterial properties is essential for next-generation biomedical materials. However, conventional polymers often exhibit limited bioactivity and poor resistance to degradation, restricting their functional applications. The novelty of this study involves the combination of the bio-derived cross-linker 2,5-bis(aminomethyl)furan (BAF) into poly (methyl methacrylate) PMMA to form a cross-linked network incorporated with various ratios of ZnO nanoparticles (ZnO NPs), resulting in improved biological and thermal properties. The surface morphologies, material crystallinity, and thermal degradation properties of the synthesized BAF–PMMA/ZnO were investigated using Scanning Electron microscopy (SEM), Energy-Dispersive X-ray spectroscopy (EDX), X-ray diffraction (XRD), and Thermogravimetric characterization technique (TGA), respectively. The prepared BAF-PMMA/ZnO nanocomposites showed an enhancement in the crystallinity after increasing the ratio of ZnO NPs compared to the amorphous cross-linked BAF–PMMA polymer. The thermal stability of nanocomposites was significantly enhanced after the introduction of ZnO NPs into cross-linked BAF–PMMA polymer. The resultant nanocomposites BAF–PMMA/ZnO were examined as antibacterial agents against the Escherichia coli (E. coli) and Staphylococcus aureus (S. aureus) bacterial strains. The results showed that most BAF–PMMA/ZnO nanocomposites have antibacterial activity against both bacterial species compared to the pure cross-linked BAF-PMMA polymer. The BAF–PMMA/ZnO 10 wt.% sample shows the highest inhibition zone of (16.3 ± 0.33) against E. coli. These outcomes demonstrate that such nanocomposites offer a viable pathway toward multipurpose biomaterials with exceptional structural and biological features.Keywords
Supplementary Material
Supplementary Material FilePoly (methyl methacrylate) (PMMA) is a multifunctional thermoplastic that is prized for its biocompatibility, chemical resistance, optical clarity, and thermal stability [1]. It is extensively utilized in drug delivery, dentistry, orthopedics, biomedical devices, and optoelectronics. Nevertheless, PMMA has limited mechanical strength and brittleness, especially at high temperatures. These restrictions are improved by crosslinking with multifunctional agents, opening new possibilities for wastewater treatment, electronics, sensors, and pharmaceuticals [1,2].
Nanocomposites have become an important class of innovative materials because they combine the functional benefits of inorganic nanoparticles with the unique features of polymers [1]. These hybrid materials, which are created by introducing nanoscale fillers into polymeric matrices, frequently have improved rigidity, thermal resistance, optical properties, and biological activity when compared to their pure counterparts [3,4]. The high surface-to-volume ratio and distinct physicochemical characteristics of nanoparticles, which permit robust interfacial interactions with the host polymer, are the causes of these enhancements [5]. Nanocomposites have found widespread uses in environmental remediation, biomedical engineering, catalysis, and antimicrobial coatings as a result of these synergies [6,7].
Metal oxide nanoparticles were used as fillers for the enhancement of polymer-based nanocomposites [8,9]. Zinc Oxide nanoparticles (ZnO NPs), as one of the versatile inorganic nanomaterials, have drawn more attention recently because of their numerous significant physical and chemical characteristics, such as chemical stability [10,11], remarkable optical transparency [12], high photocatalytic activity [13], and potent antibacterial and bactericide properties [14,15]. Due to their small size, high specific area, quantum influence, and strong interactions between the organic polymer and inorganic nanoparticles, ZnO NPs have the potential to enhance the thermal and optical performance of PMMA polymer matrices [16,17]. Therefore, these nanocomposites could be employed in dental prosthetics, bone cement, contact lenses, and antimicrobial coatings [18,19]. There are numerous examples that clarify the effectiveness of modifying polymeric materials with ZnO NPs. One study, for example, focused on creating a novel polymer with ZnO NPs [20]. It was discovered that the ZnO nanoparticles and the different polymer matrices had excellent interfacial contact, which improved the material’s mechanical qualities [21]. Additionally, another work produced a PMMA/ZnO hybrid system and validated its advantageous application for photocatalytic activity [7]. Furthermore, researchers examined the antibacterial properties of polyacrylate/ZnO nanocomposites comprising various ZnO nanoparticle morphologies and discovered that those having sphere-like and flower-like ZnO NPs had superior antibacterial properties [22].
In this work, 2,5-bis(aminomethyl)furan (BAF) was proposed as a bifunctional cross-linking agent to modify the properties of PMMA and incorporated with different concentrations of ZnO NPs ranging from 2% to 20% into the polymer matrix. ZnO nanoparticles were selected over other metal oxide nanoparticles, such as Ag, TiO2, or Cu, due to their antimicrobial activity, environmental safety, low toxicity, and cost. This modification improves rigidity, thermal stability, and biological performance. As a result, the selection of BAF–PMMA/ZnO nanocomposites in this study was based on making it a promising material for potential biomedical and environmental applications.
PMMA was procured from Across Organics, Geel, Belgium. BAF, 98% was supplied by Baoji Guokang Biotechnology, Ltd., Baoji, China, while tetrahydrofuran (THF, 99.9%) was obtained from Merck KGaA, Darmstadt, Germany. ZnO nanoparticles were commercially obtained (Sigma-Aldrich, St. Louis, MO, USA <100 nm, purity > 99%). All compounds were utilized without additional purification.
Thermo-gravimetric analysis (TGA) was performed on a TGA4724 with a heating rate of 10°C/min between 25°C and 500°C under N2 atmosphere. Fourier transform infrared spectra (FTIR) with a Nicolet Magna 6700 FT spectrometer were conducted in a wavenumber region (400–4000 cm−1). X-ray diffraction (XRD) patterns were studied using a Bruker D8 Advance with Cu Kα radiation (wavelength 1.5418 Å) at 40 kV and 40 mA. The patterns were collected between 2θ of 10° and 60°, and the scan speed was 1.5°/min. Scanning electron microscopy (SEM) imaging was performed with a FEI TENEO VS microscope equipped with an EDAX detector.
2.3 Preparation of BAF–PMMA/ZnO Nanocomposites
BAF–PMMA/ZnO nanocomposite materials were prepared by an in-situ polymerization technique as follows: 1 g of PMMA was dissolved in THF solvent (50 mL). 2,5-Bis(aminomethyl)furan (BAF) was used as a cross-linking agent. A fixed amount of 0.20 g of BAF (1.59 mmol), corresponding to 13.7 mol% relative to PMMA repeat units (1.00 g, 9.99 mmol), was added to the reaction mixture prior to in-situ polymerization. Then ZnO nanoparticles with different ratios (2, 5, 10, 20 wt.%) were dispersed through the PMMA solution by stirring, before being ultrasonicated for 10 min. Finally, the cross-linker BAF was added, and the mixture was refluxed for about 8 h at 70°C with regular shaking. The resulting mixture was then poured into petri dishes and left to dry for 24 h at 25°C. The samples were designated as presented in Table 1.
2.4 The Antibacterial Activities of BAF–PMMA/ZnO Nanocomposites
A Disk diffusion susceptibility test was utilized to examine the antibacterial effects of BAF-PMMA and BAF-PMMA/ZnO nanoparticles compared to Tetrahydrofuran (THF), ZnO as negative controls, and Gentamicin (Mast diagnostics, Ltd., Bootle, UK) as a positive control using Müeller-Hinton agar (Difco, Laboratories, Detroit, MI, USA) plates. Gentamicin was selected as it inhibits a wide spectrum, affecting both Gram-negative and Gram-positive bacteria. Gram-negative bacteria, Escherichia coli (ATCC 11775), and Gram-positive bacteria, Staphylococcus aureus (ATCC 12600), were selected. The bacterial strains were subcultured by inoculating them in Nutrient Broth (Oxoid Ltd., Basingstoke, UK) for 18 h to ensure the bacteria were in the log phase. Then, around 8 mm of nanoparticles were prepared in Disks, followed by adding them to Müeller-Hinton agar after streaking with bacteria. Plates were incubated at 37°C for 24 h before measuring the inhibition zones in millimeters (mm) using a caliper (Memmert GmbH + Co. KG, Schwabach, Germany). The charts were created using GrapPad Prism (version 10.6.0), which included triple readings. Two-way ANOVA was performed, followed by Dunnett’s multiple comparisons test to compare between groups. Using Gentamicin as a positive control to compare with all other bacterial treatments. The dried films were cut into circular discs (8 mm in diameter); each treatment was tested on at least three independent plates (n = 3), and the areas of inhibition were measured in millimeters using a calibrated digital scale (Mitutoyo Corporation, Kawasaki, Japan).
3.1 Morphological and Elemental Investigation
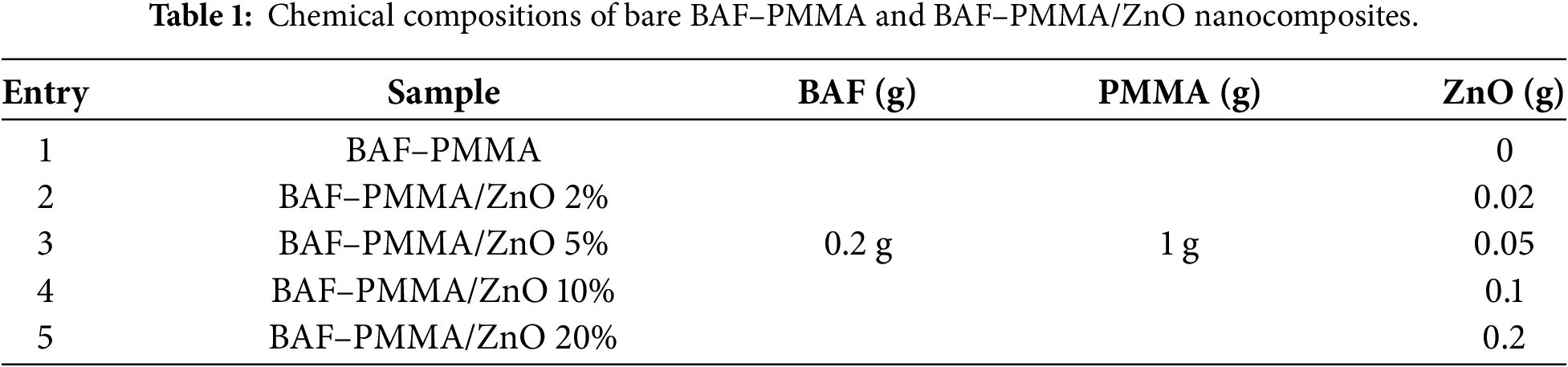
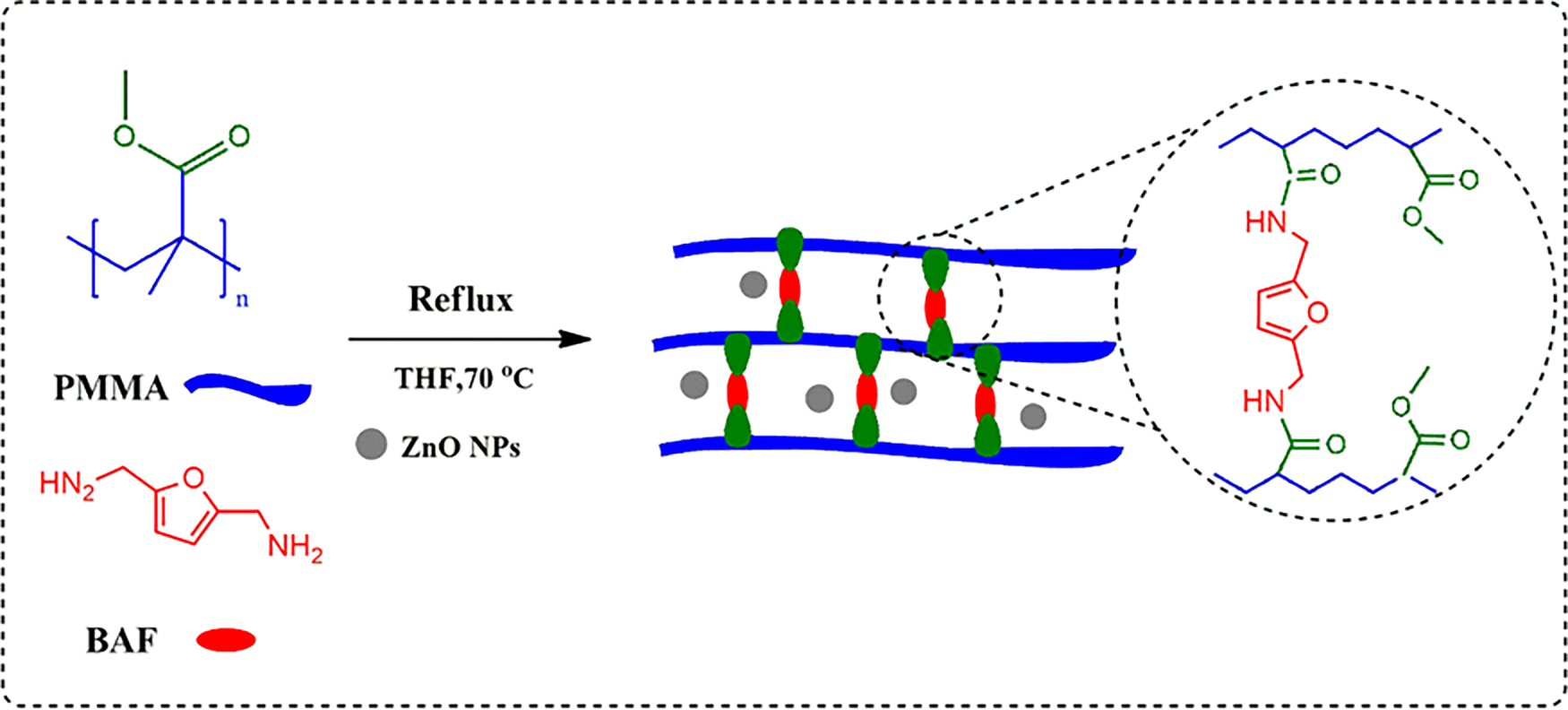
BAF–PMMA/ZnO nanocomposite materials were prepared by an in-situ polymerization technique as illustrated in scheme 1. The surface morphologies of pure BAF–PMMA cross-linked polymer exhibited a microporous structure as illustrated in Fig. 1a,b. Therefore, the SEM images of the prepared nanocomposites BAF–PMMA/ZnO revealed porous morphology on the polymer matrix, which can increase the surface area and help with the good distribution of ZnO NPs. The porosity measurement was not quantitative in this work and is planned for future study to correlate the effect of porosity with the application. The SEM micrographs (Fig. 1c–j) illustrate the effect of varying ZnO NPs ratios (2–20 wt.%). In the BAF–PMMA/ZnO nanocomposites, the bright regions correspond to ZnO NPs, indicating their presence both on the surface and within the pores of the polymeric network, as shown in Fig. 1c–j. When the concentration of ZnO NPs increases from 2 to 20 wt.%, noticeable agglomeration is observed. This aggregation is attributed to the high surface energy and strong interparticle attractions of the nanoparticles, which promote clustering at higher concentrations [23].
Scheme 1: Schematic diagram for the BAF–PMM/ZnO nanocomposites polymer fabrication.


Figure 1: SEM images at two magnifications for (a,b) BAF–PMMA, (c,d) BAF–PMMA/ZnO 2%, (e,f) BAF–PMMA/ZnO 5%, (g,h) BAF–PMMA/ZnO 10%, and (i,j) BAF–PMMA/ZnO 20%.
The elemental composition of the prepared BAF–PMMA/ZnO nanocomposites was investigated using Energy Dispersive X-ray (EDX) analysis. The EDX of the bare BAF-PMMA was illustrated in Fig. S1, which demonstrates the characteristic signals corresponding to carbon (C), nitrogen (N), and oxygen (O). As shown in Fig. 2a,b for samples containing 2 and 5 wt.% ZnO, respectively, the EDX spectra exhibited only two characteristic signals corresponding to carbon (C) and oxygen (O), with no detectable Zn peaks. This absence may be attributed to the low concentration of ZnO nanoparticles and their inhomogeneous dispersion within the cross-linked polymer matrix, as well as the surface-sensitive and localized nature of EDX analysis [24,25]. In contrast, at higher ratios from ZnO 10 and 20 wt.% (Fig. 2c,d), distinct Zn signals appeared alongside the C and O peaks, confirming the presence of ZnO nanoparticles in the nanocomposites. These findings reveal the outstanding purity of the synthesized materials and show that their elemental composition is in line with the intended formula. Table 2 summarizes the elemental contents of the BAF–PMMA/ZnO (1–4) nanocomposites.
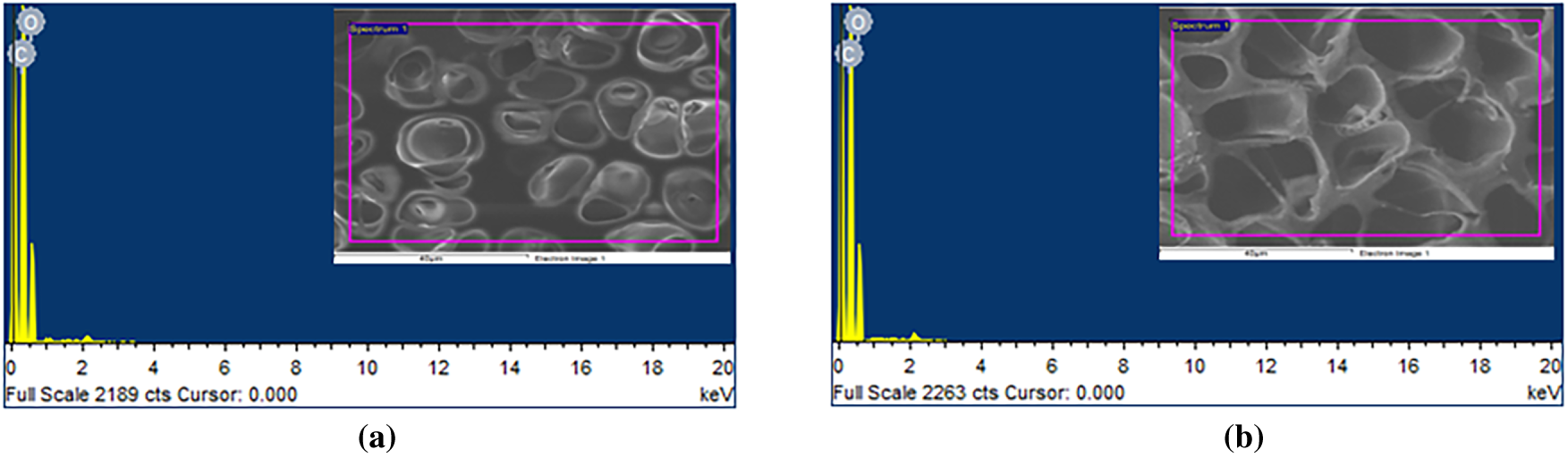
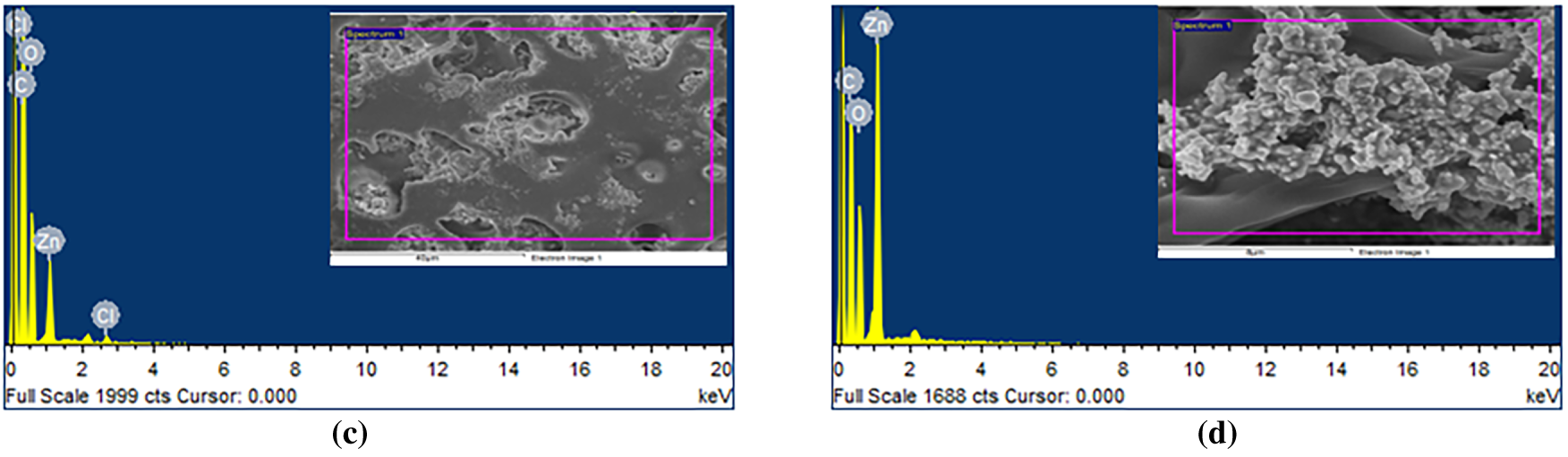
Figure 2: EDX analysis of (a) BAF–PMMA/ZnO 2%, (b) BAF–PMMA/ZnO 5%, (c) BAF–PMMA/ZnO 10%, and (d) BAF–PMMA/ZnO 20%.
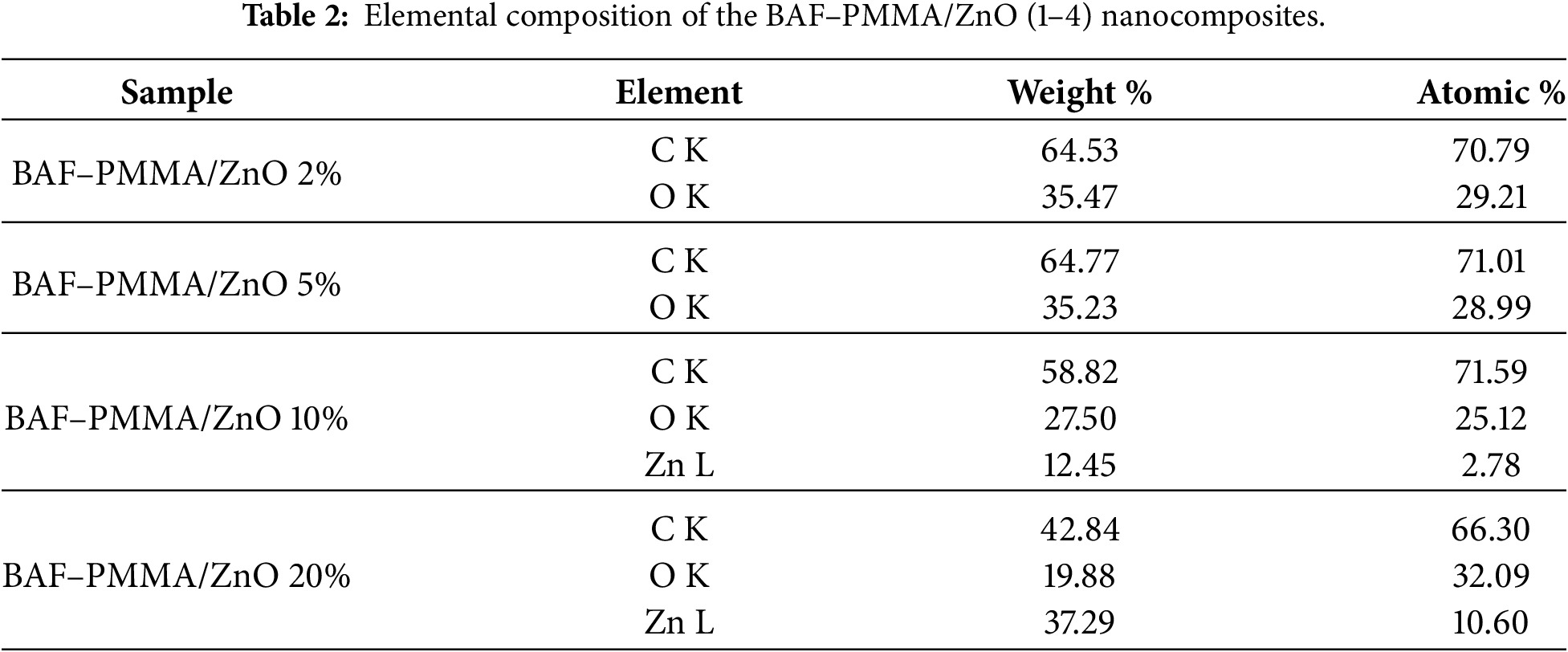
X-ray diffraction analysis was performed to examine the crystal structure of the prepared BAF–PMMA/ZnO nanocomposites over a 2θ range of 5°–60°. The diffraction patterns for BAF-PMMA and all prepared BAF-PMMA/ZnO nanocomposites are presented in Fig. 3. The bare BAF–PMMA exhibits an amorphous nature, as indicated by the broad peaks centered around 2θ values of 15°, 30°, and 43°. Upon the incorporation of ZnO nanofillers, notable changes in the crystallinity of the cross-linked BAF–PMMA polymer were observed. Specifically, sharp and intense diffraction peaks appeared at 2θ values of 31.7°, 34.4°, 36.1°, 47.5°, 56.5°, 62.8°, 66.3°, 67.9°, and 69°, which are consistent with the standard pattern of hexagonal wurtzite ZnO (JCPDS Card No. 36-1451) [26]. The intensity of these peaks progressively increased with rising ZnO ratios (2–20 wt.%) in the polymer matrix. These observations not only confirm the successful incorporation of ZnO nanoparticles into the cross-linked BAF–PMMA polymer but also demonstrate that ZnO contributes to enhancing the overall crystallinity of the nanocomposite matrix. The crystallinity % are 5.44, 3.14, 1.83, and 1.07 for BAF-PMMA/Zno 20%, BAF-PMMA/ZnO 10%, BAF-PMMA/ZnO 5% and BAF-PMMA/ZnO 2%, respectively. In this study, the XRD discussion demonstrated that the crystallinity of ZnO NPs in the polymer matrix increases with the rise in the ratios of ZnO NPs. The crystalline size and lattice strain behavior of ZnO NPs within the BAF–PMMA matrix will be calculated using the Scherrer equation or Williamson-Hall analysis for further investigation in future work.
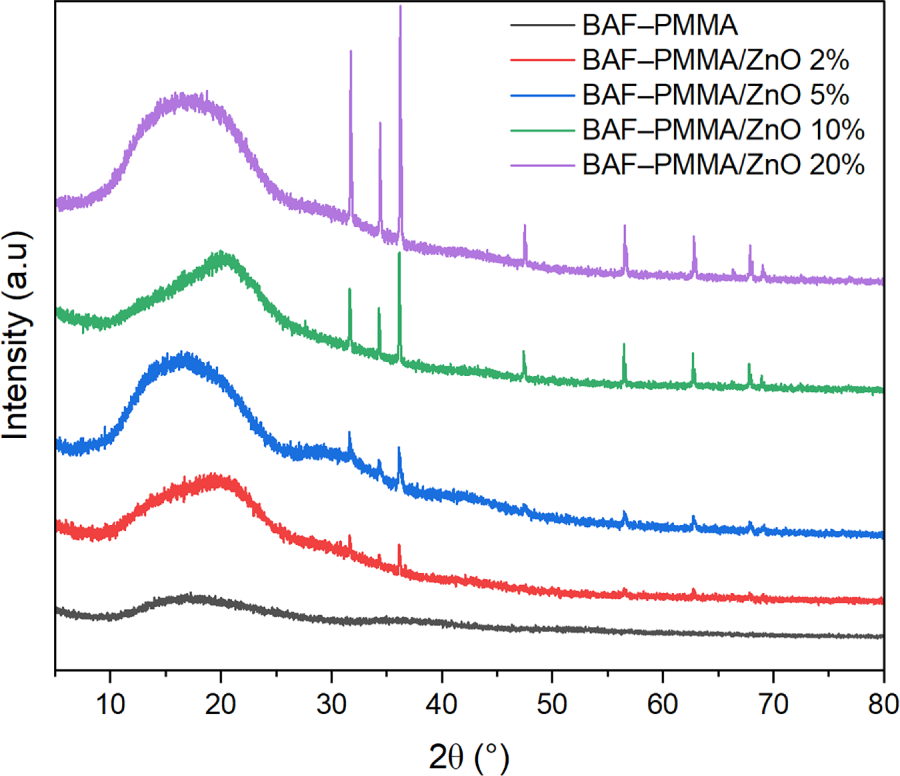
Figure 3: The XRD patterns of bare BAF–PMMA and BAF–PMMA/ZnO nanocomposites.
The Fourier-Transform Infrared spectra of the prepared BAF–PMMA/ZnO nanocomposites were recorded in the range of 4000–500 cm−1, as presented in Fig. 4. The spectra revealed characteristic absorption bands corresponding to the polymer precursors, namely PMMA and the cross-linker aminomethyl furan [27]. The successful formation of the cross-linked polymer was confirmed by the sharp absorption band at 1721 cm−1, attributed to the C=O stretching vibration of the acrylate ester group in PMMA. Furthermore, the transformation of primary amino groups (R–NH2) in aminomethyl furan into secondary amino groups (N–H) was verified by the appearance of a singlet stretching vibration band at 3383 cm−1 in the final cross-linked polymer. FT-IR analysis confirmed that the polymer retained its original bonding framework, showing no evidence of bond cleavage or the formation of new functional groups after ZnO NPs incorporation.
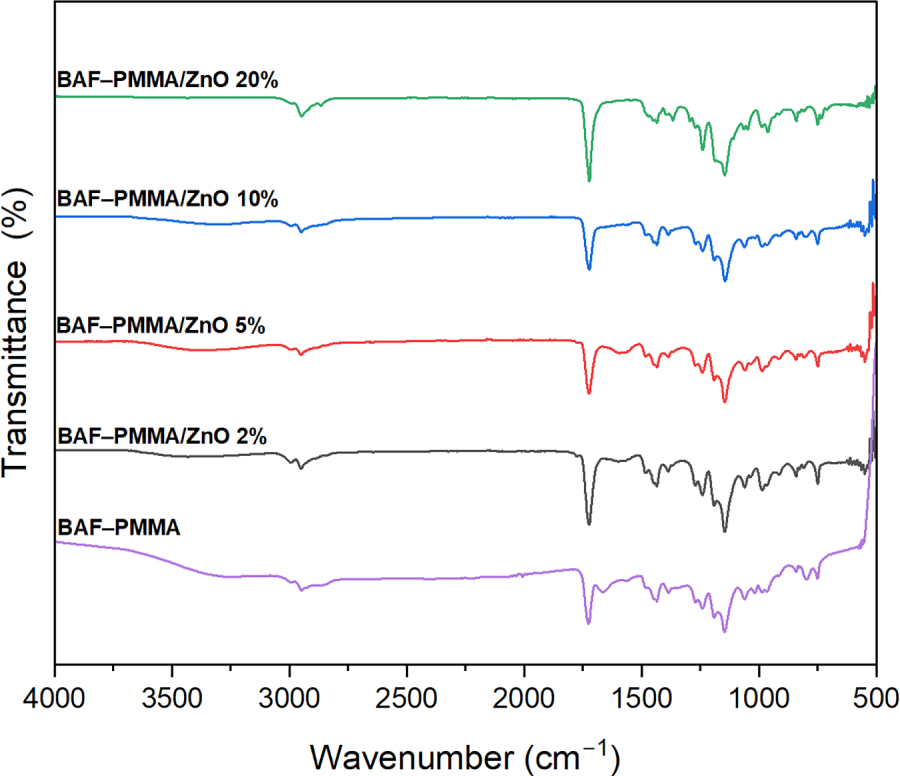
Figure 4: FT-IR spectra of bare BAF–PMMA and BAF–PMMA/ZnO nanocomposites.
3.3 Thermogravimetric Analysis
The thermal stability of the prepared BAF–PMMA/ZnO nanocomposites was investigated to evaluate the influence of ZnO nanoparticle incorporation and concentration on the stability of the BAF–PMMA polymer. As shown in Fig. 5, the TGA curves of all nanocomposites revealed three distinct degradation stages, observed at both low and high ZnO. At lower ZnO contents (2 and 5 wt.%), the first stage corresponds to a 5% weight loss of around 152°C, followed by a 10% weight loss near 184°C in the second stage, and a major 50% weight loss at approximately 353°C in the third stage. At higher ZnO (10 and 20 wt.%), degradation also occurred in three stages, with 5% weight loss in the temperature range of 153°C–165°C, 10% weight loss in the 235°C–246°C range, and a significant 60% weight loss at around 406°C. The initial degradation step is attributed to the evaporation of absorbed water molecules. The second degradation stage is attributed to the breakdown of both cross-linking bonds and the polymer backbone. The third stage involves extensive decomposition and combustion, resulting in a 50%–60% weight loss. The maximum polymer decomposition temperature (Tmax), which indicates the temperature at which the highest rate of weight loss occurs, was determined for all samples. In addition, the characteristic degradation temperatures T10, T30, and T50—representing the temperatures at which 10%, 30%, and 50% mass loss occur, respectively—were also evaluated for the BAF–PMMA/ZnO nanocomposites as illustrated in Table 3.
Figure 5: The TGA analysis of BAF–PMMA and BAF–PMMA/ZnO nanocomposites.
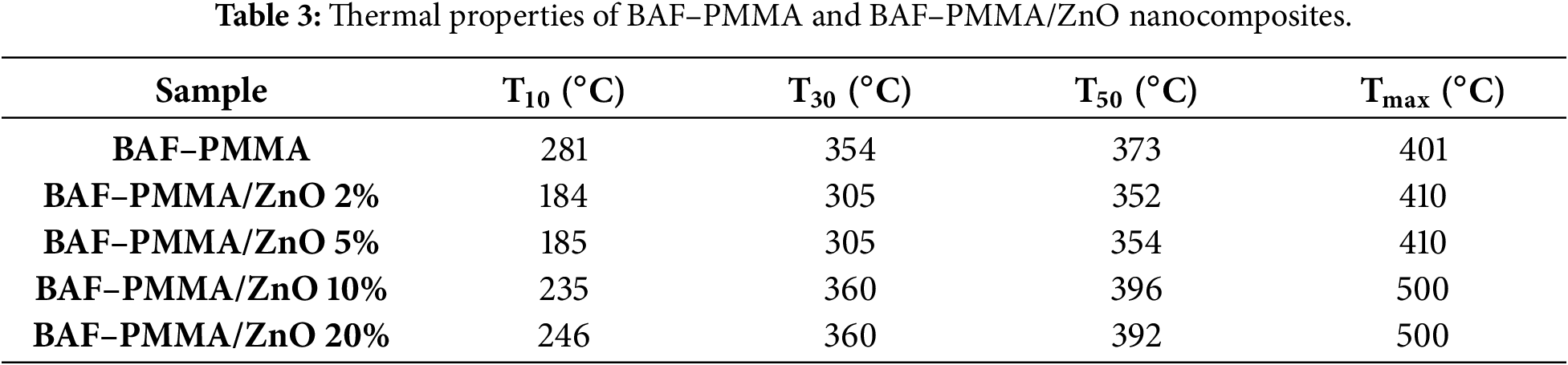
The Tmax, T10, T30, and T50 values of BAF–PMMA/ZnO nanocomposites with higher ZnO were consistently greater than those observed at lower contents, reflecting the higher decomposition resistance of the inorganic phase [27]. Moreover, the Tmax values of the high ratio of ZnO NPs (500°C) were significantly higher than those reported for the cross-linked polymer without ZnO NPs in our previous work (393°C–412°C) [28]. Overall, these findings demonstrate a marked enhancement in the thermal stability of BAF–PMMA/ZnO nanocomposites compared with pure cross-linked polymer.
3.4 Antibacterial Activity of Polymers
The antibacterial performance of the prepared nanocomposites was evaluated against two representative bacterial strains: Escherichia coli ATCC 11775 (Gram-negative) and Staphylococcus aureus ATCC 12600 (Gram-positive) [28–30]. The antibacterial effects of BAF-PMMA in the presence or absence of increasing concentrations of ZnO (BAF-PMMA/ZnO) were assessed against gram-negative E. coli and gram-positive S. aureus bacteria. As shown in Fig. 6, BAF-PMMA alone exhibited no inhibitory effect on either bacterial strain, evidenced by the absence of inhibition zones around the polymer disks; therefore, a fixed concentration of 20% BAF-PMMA was selected for subsequent experiments.

Figure 6: The effect of BAF-PMMA on E. coli and S. aureus. Different concentrations of BAF-PMMA (20%, 30%, 40%, 50%, and 60%) were examined using a Disk diffusion test for 24 h at 37°C.
Interestingly, increasing concentrations of ZnO (BAF-PMMA/ZnO) from 2% to 20% have inhibited bacterial growth. Results in Fig. 7 showed that the BAF-PMMA/ZnO nanoparticles have created an increasing diameter inhibiting zone of E. coli from (11.2 ± 0.6 mm) in 2% ZnO to (16.3 ± 0.33 mm) in 10% ZnO, with a sudden decrease in the inhibiting zone diameter of (9.6 ± 0.33 mm) in 20% ZnO. Interestingly, comparing BAF-PMMA/ZnO treated E. coli with Gentamicin as a positive control (17.4 ± 0.33 mm) shows no significance between the 2 groups, indicating that BAF-PMMA/ZnO 10% as strong as Gentamicin in inhibiting E. coli, with a p value = 0.723. Unlike all the other E. coli-treated cultures with THF, ZnO, BAF-PMMA, BAF-PMMA/ZnO 2%, BAF-PMMA/ZnO 5%, and BAF-PMMA/ZnO 20% separately, which show a p value of <0.0001, <0.0001, 0.0001, 0.001, 0.002, 0.0001, respectively.

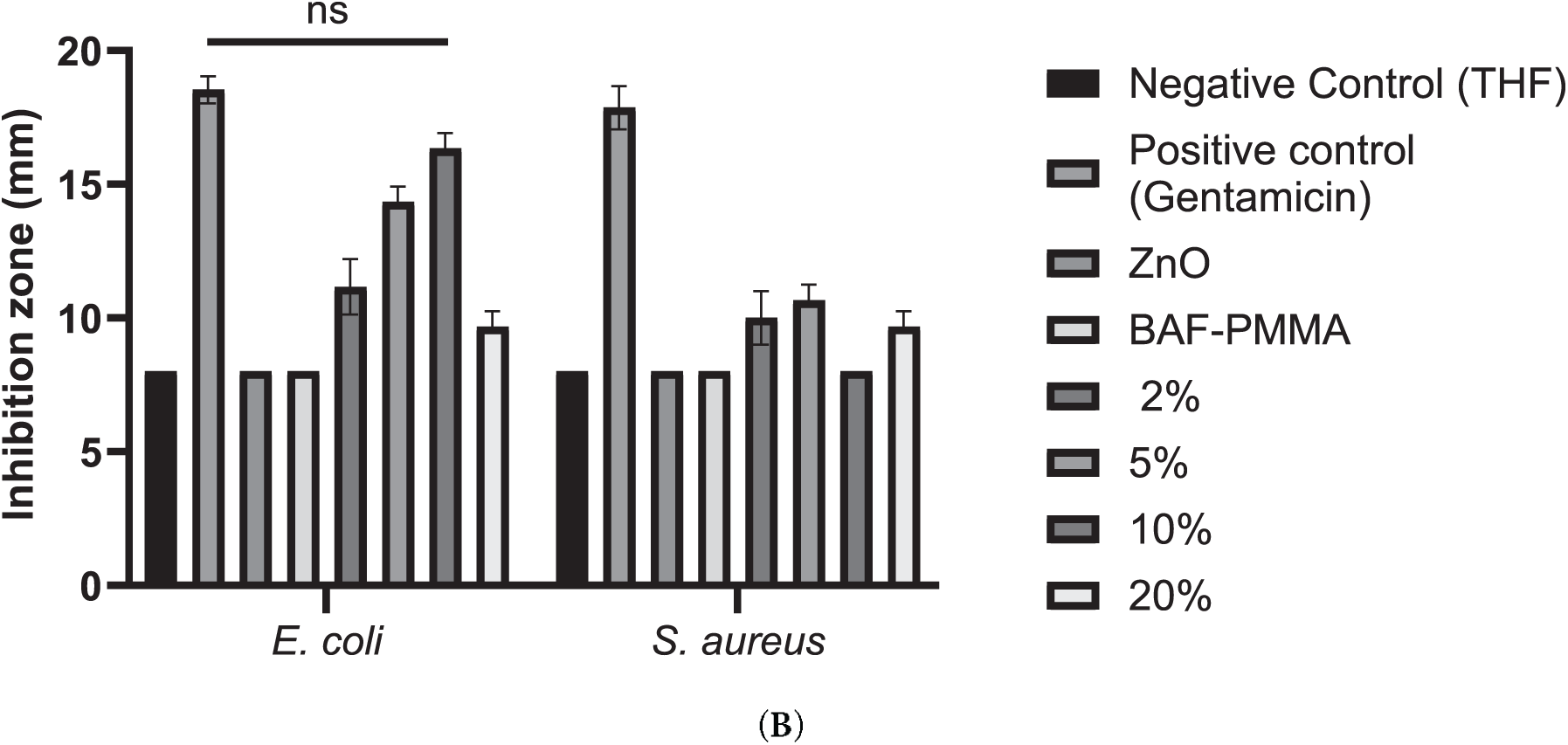
Figure 7: Antibacterial activity of BAF-PMMA/ZnO nanocomposites prepared using 20% BAF-PMMA cross-linked with varying ZnO concentrations (2%, 5%, 10%, and 20%) against E. coli and S. aureus. Treatments were compared with a negative control (THF), a positive control (Gentamicin), ZnO alone, and BAF-PMMA alone. Disk diffusion assays were performed on Müeller–Hinton agar plates inoculated with each bacterial strain, followed by incubation for 24 h at 37°C. (A) Representative inhibition zones produced by BAF-PMMA/ZnO against both bacterial species. (B) Bar graphs showing the mean inhibition zone diameters (mm), generated using GraphPad Prism. The data represents the average of three independent measurements. Statistical analysis was conducted using two-way ANOVA followed by Dunnett’s multiple comparisons test, with Gentamicin used as the reference treatment. Significant differences were observed for all treatments compared with Gentamicin (p < 0.002–0.0001), except for E. coli treated with 10% BAF-PMMA/ZnO, which showed no significant (ns) difference relative to Gentamicin (p = 0.723).
The inhibition zones of S. aureus affected by BAF-PMMA/ZnO nanoparticles were less compared to E. coli. The highest inhibition zone of S. aureus was detected at (10.66 ± 0.33 mm) at 5% ZnO, and no effect at 10% ZnO. The distinct effects that BAF-PMMA/ZnO demonstrated against the two species of bacteria are due to structural variations between them. At higher loading of ZnO, the irregular response is most likely attributed to the aggregation/blocking of zinc oxide molecules, which reduces the proportion of surface-exposed molecules and limits the diffusion of active species in the agar diffusion assay. This interpretation will be confirmed by dispersion mapping and zinc (Zn2+) ion release measurements in future studies.
The equilibrium and integrity of the bacterial cell wall, which displays anti-microbial activity towards microorganisms, may be disrupted by the BAF-PMMA/ZnO resin due to the electrostatic interaction between the positively charged resin and the negatively charged bacterial cell membrane. Furthermore, the free radicals on the resin surface may disrupt the lipids and structure of the microorganism cells’ membranes, impairing their ability to function. Because of ZnO is incorporated within a PMMA cross-linked polymer network, the antibacterial effect is expected to be controlled by surface-exposed ZnO and limited Zn+2 diffusion; direct ROS/Zn+2 release assays were not achieved in this study and are planned for future work.
A comparative study of various commercial metal oxide nanoparticles was presented in Table 4. Common metal oxide nanoparticles have strong photocatalytic and antimicrobial properties. However, they often have challenges such as cytotoxicity, agglomeration, or the need for UV activation. In contrast, the prepared BAF-PMMA/ZnO nanocomposite approach a balanced combination of thermal stability, bio-derived cross-linker, biocompatibility, enhanced ZnO dispersion, and antimicrobial activity. The present findings display that the BAF–PMMA/ZnO nanocomposite is a promising material for potential biomedical and environmental applications.
In this study, cross-linked BAF–PMMA/ZnO nanocomposites were successfully synthesized via an in-situ polymerization approach and evaluated for antibacterial activity against Staphylococcus aureus (Gram-positive) and Escherichia coli (Gram-negative). Comprehensive structural, thermal, and morphological characterizations demonstrated that ZnO nanoparticles were effectively incorporated within the polymer matrix, as clearly shown by the XRD diffraction peaks and EDX compositional mapping. The addition of ZnO nanoparticles significantly improved the thermal stability of the BAF–PMMA polymer. Moreover, the nanocomposites exhibited superior antibacterial performance compared to pristine BAF–PMMA, with particularly strong inhibition against E. coli, showing inhibition zones ranging from 9.6 ± 0.33 to 16.3 ± 0.33 mm.
Acknowledgement: The author acknowledges Dr. Fatemah Salem Basingab from the biology department at King Abdulaziz University for her support in biological study.
Funding Statement: Not applicable.
Availability of Data and Materials: This article does not involve data availability, and this section is not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The author declares no conflicts of interest.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/jpm.2026.073588/s1.
References
1. Alkayal NS. Fabrication of cross-linked PMMA/SnO2 nanocomposites for highly efficient removal of chromium (III) from wastewater. Polymers. 2022;14(10):2101. doi:10.3390/polym14102101. [Google Scholar] [PubMed] [CrossRef]
2. Poddar MK, Sharma S, Moholkar VS. Sonochemical synthesis of PMMA/cloisite 30B nanocomposites: a mechanistic investigation. Macromol Symp. 2016;361(1):82–100. doi:10.1002/masy.201500009. [Google Scholar] [CrossRef]
3. Liu P, Su Z. Preparation and characterization of PMMA/ZnO nanocomposites via in-situ polymerization method. J Macromol Sci Part B Phys. 2006;45(1):131–8. doi:10.1080/00222340500408085. [Google Scholar] [CrossRef]
4. Poddar MK, Sharma S, Moholkar VS. Investigations in two-step ultrasonic synthesis of PMMA/ZnO nanocomposites by in-situ emulsion polymerization. Polymer. 2016;99(1):453–69. doi:10.1016/j.polymer.2016.07.052. [Google Scholar] [CrossRef]
5. Khlifi K, Atallah MS, Cherif I, Karkouch I, Barhoumi N, Attia-Essaies S. Synthesis of ZnO nanoparticles and study of their influence on the mechanical properties and antibacterial activity of PMMA/ZnO composite for orthotic devices. Surf Interfaces. 2023;41(2):103279. doi:10.1016/j.surfin.2023.103279. [Google Scholar] [CrossRef]
6. Di Mauro A, Cantarella M, Nicotra G, Pellegrino G, Gulino A, Brundo MV, et al. Novel synthesis of ZnO/PMMA nanocomposites for photocatalytic applications. Sci Rep. 2017;7(1):40895. doi:10.1038/srep40895. [Google Scholar] [PubMed] [CrossRef]
7. Di Mauro A, Farrugia C, Abela S, Refalo P, Grech M, Falqui L, et al. Synthesis of ZnO/PMMA nanocomposite by low-temperature atomic layer deposition for possible photocatalysis applications. Mater Sci Semicond Process. 2020;118:105214. doi:10.1016/j.mssp.2020.105214. [Google Scholar] [CrossRef]
8. Canché-Escamilla G, Duarte-Aranda S, Toledano M. Synthesis and characterization of hybrid silica/PMMA nanoparticles and their use as filler in dental composites. Mater Sci Eng C. 2014;42:161–7. doi:10.1016/j.msec.2014.05.016. [Google Scholar] [PubMed] [CrossRef]
9. Wahab R, Hwang IH, Kim YS, Musarrat J, Siddiqui MA, Seo HK, et al. Non-hydrolytic synthesis and photo-catalytic studies of ZnO nanoparticles. Chem Eng J. 2011;175:450–7. doi:10.1016/j.cej.2011.09.055. [Google Scholar] [CrossRef]
10. Sadeghi B. Preparation of ZnO/Ag nanocomposite and coating on polymers for anti-infection biomaterial application. Spectrochim Acta Part A Mol Biomol Spectrosc. 2014;118:787–92. doi:10.1016/j.saa.2013.09.022. [Google Scholar] [PubMed] [CrossRef]
11. Khairy M, Amin NH, Kamal R. Optical and kinetics of thermal decomposition of PMMA/ZnO nanocomposites. J Therm Anal Calorim. 2017;128(3):1811–24. doi:10.1007/s10973-016-6062-x. [Google Scholar] [CrossRef]
12. Dadi R, Azouani R, Traore M, Mielcarek C, Kanaev A. Antibacterial activity of ZnO and CuO nanoparticles against gram positive and gram-negative strains. Mater Sci Eng C. 2019;104:109968. doi:10.1016/j.msec.2019.109968. [Google Scholar] [PubMed] [CrossRef]
13. Mendes CR, Dilarri G, Forsan CF, de Moraes Ruy Sapata V, Lopes PRM, de Moraes PB, et al. Antibacterial action and target mechanisms of zinc oxide nanoparticles against bacterial pathogens. Sci Rep. 2022;12:2658. doi:10.1038/s41598-022-06657-y. [Google Scholar] [PubMed] [CrossRef]
14. Sirelkhatim A, Mahmud S, Seeni A, Kaus NHM, Ann LC, Bakhori SKM, et al. Review on zinc oxide nanoparticles: antibacterial activity and toxicity mechanism. Nano Micro Lett. 2015;7(3):219–42. doi:10.1007/s40820-015-0040-x. [Google Scholar] [PubMed] [CrossRef]
15. Behera SK, Khan GA, Singh SS, Jena B, Sashank K, Patnaik S, et al. Antibacterial efficacy of ZnO/bentonite (clay) nanocomposites against multidrug-resistant Escherichia coli. ACS Omega. 2024;9(2):2783–94. doi:10.1021/acsomega.4c00630. [Google Scholar] [PubMed] [CrossRef]
16. Motelica L, Vasile BS, Ficai A, Surdu AV, Ficai D, Oprea OC, et al. Antibacterial activity of zinc oxide nanoparticles loaded with essential oils. Pharmaceutics. 2023;15(10):2470. doi: 10.3390/pharmaceutics15102470. [Google Scholar] [PubMed] [CrossRef]
17. Barua E, Deoghare AB, Chatterjee S, Sapkal P. Effect of ZnO reinforcement on the compressive properties, in vitro bioactivity, biodegradability and cytocompatibility of bone scaffold developed from bovine bone-derived HAp and PMMA. Ceram Int. 2019;45(16):20331–45. doi:10.1016/j.ceramint.2019.07.006. [Google Scholar] [CrossRef]
18. Ahmad N, Ahmad SI, Ahmedi S, Yadav P, Manzoor N, Parwaz M, et al. Structural, optical and antifungal properties of the PMMA-ZnO nanocomposites: potential applications in odontology. Mater Chem Phys. 2023;309:128382. doi:10.1016/j.matchemphys.2023.128382. [Google Scholar] [CrossRef]
19. Ben Aissa C, Barhoumi N, Khlifi K, Bousslama W, Karkouch I, Majid F. Nanocomposite dental restorations resin doped with smart antimicrobial agents: mechanical properties, wear resistance and antimicrobial activities. Mater Today Commun. 2025;48:113489. doi:10.1016/J.MTCOMM.2025.113489. [Google Scholar] [CrossRef]
20. Rehner AMG, Tudorache DI, Bîrcă AC, Nicoară AI, Niculescu AG, Holban AM, et al. Antibacterial properties of PMMA/ZnO(NanoAg) coatings for dental implant abutments. Materials. 2025;18(2):382. doi:10.3390/ma18020382. [Google Scholar] [PubMed] [CrossRef]
21. Ponnamma D, Cabibihan JJ, Rajan M, Pethaiah SS, Deshmukh K, Gogoi JP, et al. Synthesis, optimization and applications of ZnO/polymer nanocomposites. Mater Sci Eng C. 2019;98:1210–40. doi:10.1016/j.msec.2019.01.081. [Google Scholar] [PubMed] [CrossRef]
22. Liu J, Ma J, Bao Y, Wang J, Zhu Z, Tang H, et al. Nanoparticle morphology and film-forming behavior of polyacrylate/ZnO nanocomposite. Compos Sci Technol. 2014;98(4):64–71. doi:10.1016/j.compscitech.2014.02.019. [Google Scholar] [CrossRef]
23. Algamdi B, Alam MM, Tashkandi N, Bahaidarah EA, Alshareef FM, Alzahrani KA, et al. New furan-cross-linked PMMA for efficient removal of cationic dyes and detection of cadmium ions. Polym Adv Technol. 2025;36(2):e70107. doi:10.1002/pat.70107. [Google Scholar] [CrossRef]
24. Hammani S, Barhoum A, Bechelany M. Fabrication of PMMA/ZnO nanocomposite: effect of high nanoparticles loading on the optical and thermal properties. J Mater Sci. 2018;53(3):1911–21. doi:10.1007/s10853-017-1654-9. [Google Scholar] [CrossRef]
25. Permyakova ES, Manakhov AM, Kiryukhantsev-Korneev PV, Leybo DV, Konopatsky AS, Makarets YA, et al. Electrospun polycaprolactone/ZnO nanocomposite membranes with high antipathogen activity. Polymers. 2022;14(24):5364. doi:10.3390/polym14245364. [Google Scholar] [PubMed] [CrossRef]
26. Alamgeer, Tahir M, Sarker MR, Ali S, Ibraheem, Hussian S, et al. Polyaniline/ZnO hybrid nanocomposite: morphology, spectroscopy and optimization of ZnO concentration for photovoltaic applications. Polymers. 2023;15(2):363. doi:10.3390/polym15020363. [Google Scholar] [PubMed] [CrossRef]
27. Zhang Y, Zhuang S, Xu X, Hu J. Transparent and UV-shielding ZnO@PMMA nanocomposite films. Opt Mater. 2013;36(2):169–72. doi:10.1016/j.optmat.2013.08.021. [Google Scholar] [CrossRef]
28. Farha AH, Al Naim AF, Mansour SA. Thermal degradation of polystyrene (PS) nanocomposites loaded with sol gel-synthesized ZnO nanorods. Polymers. 2020;12(9):1935. doi:10.3390/polym12091935. [Google Scholar] [PubMed] [CrossRef]
29. Ng AMC, Chan CMN, Guo MY, Leung YH, Djurišić AB, Hu X, et al. Antibacterial and photocatalytic activity of TiO2 and ZnO nanomaterials in phosphate buffer and saline solution. Appl Microbiol Biotechnol. 2013;97(12):5565–73. doi:10.1007/s00253-013-4889-7. [Google Scholar] [PubMed] [CrossRef]
30. Rodrigues AS, Batista JGS, Rodrigues MÁV, Thipe VC, Minarini LAR, Lopes PS, et al. Advances in silver nanoparticles: a comprehensive review on their potential as antimicrobial agents and their mechanisms of action elucidated by proteomics. Front Microbiol. 2024;15:1440065. doi:10.3389/fmicb.2024.1440065. [Google Scholar] [PubMed] [CrossRef]
31. Gudkov SV, Burmistrov DE, Fomina PA, Validov SZ, Kozlov VA. Antibacterial properties of copper oxide nanoparticles (review). Int J Mol Sci. 2024;25(21):11563. doi:10.3390/ijms252111563. [Google Scholar] [PubMed] [CrossRef]
32. Abd El-Kader MFH, Elabbasy MT, Adeboye AA, Zeariya MGM, Menazea AA. Morphological, structural and antibacterial behavior of eco-friendly of ZnO/TiO2 nanocomposite synthesized via Hibiscus rosa-sinensis extract. J Mater Res Technol. 2021;15(5):2213–20. doi:10.1016/j.jmrt.2021.09.048. [Google Scholar] [CrossRef]
33. de Lima MS, Schio AL, Aguzzoli C, de Souza WV, Roesch-Ely M, Leidens LM, et al. Visible light-driven photocatalysis and antibacterial performance of a Cu-TiO2 nanocomposite. ACS Omega. 2024;9(47):47122–34. doi:10.1021/acsomega.4c07515. [Google Scholar] [PubMed] [CrossRef]
34. Govindasamy GA, Mydin RBSMN, Gadaime NKR, Sreekantan S. Phytochemicals, biodegradation, cytocompatibility and wound healing profiles of chitosan film embedded green synthesized antibacterial ZnO/CuO nanocomposite. J Polym Environ. 2023;31(10):4393–409. doi:10.1007/s10924-023-02902-1. [Google Scholar] [CrossRef]
35. Gouyau J, Duval RE, Boudier A, Lamouroux E. Investigation of nanoparticle metallic core antibacterial activity: gold and silver nanoparticles against Escherichia coli and Staphylococcus aureus. Int J Mol Sci. 2021;22(4):1905. doi:10.3390/ijms22041905. [Google Scholar] [PubMed] [CrossRef]
36. Modi SK, Gaur S, Sengupta M, Singh MS. Mechanistic insights into nanoparticle surface-bacterial membrane interactions in overcoming antibiotic resistance. Front Microbiol. 2023;14:1135579. doi:10.3389/fmicb.2023.1135579. [Google Scholar] [PubMed] [CrossRef]
37. More PR, Pandit S, De Filippis A, Franci G, Mijakovic I, Galdiero M. Silver nanoparticles: bactericidal and mechanistic approach against drug resistant pathogens. Microorganisms. 2023;11(2):369. doi:10.3390/microorganisms11020369. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF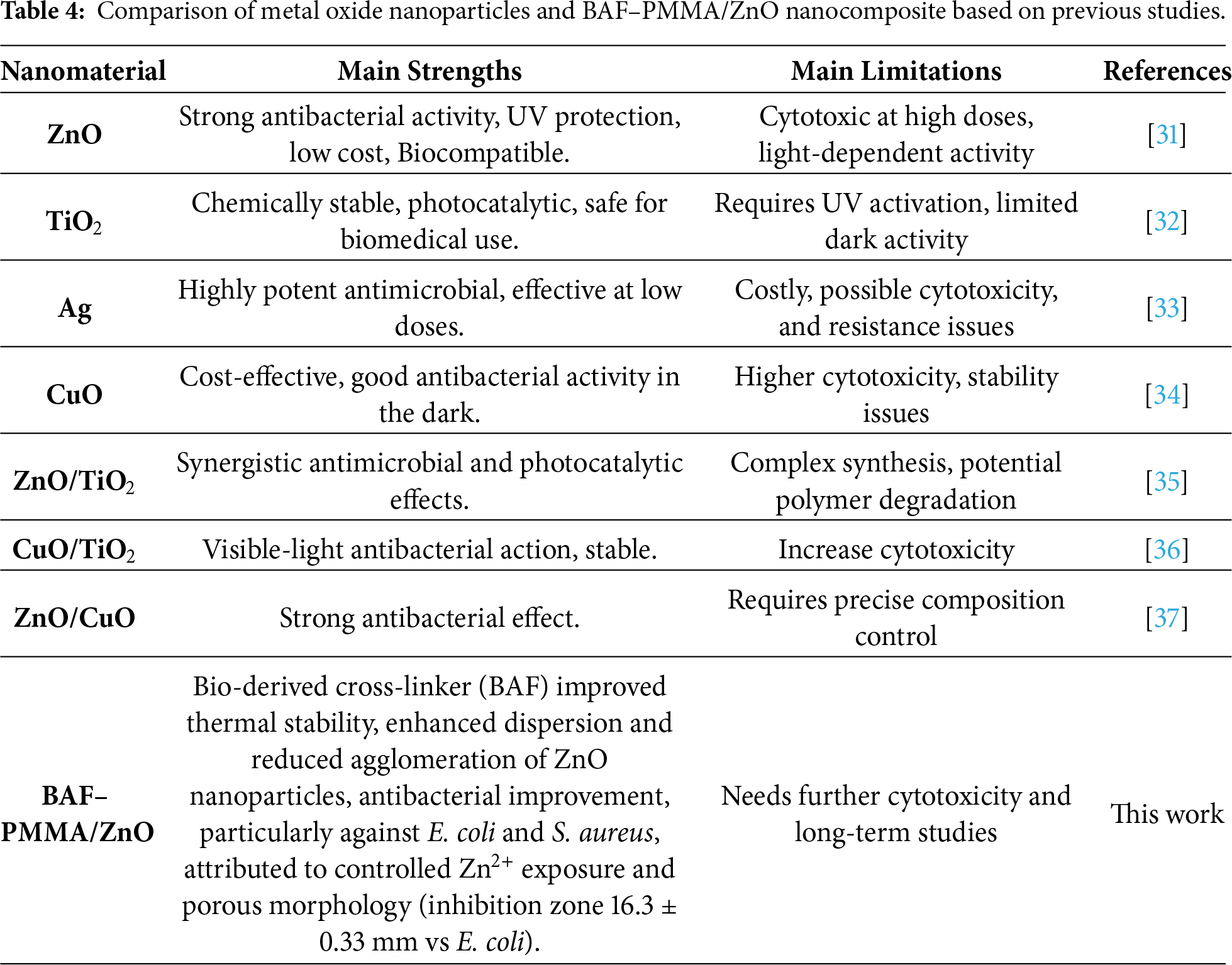
 Downloads
Downloads
 Citation Tools
Citation Tools