 Open Access
Open Access
ARTICLE
Improved Performance and Compost Biodegradation of PLA/PBAT Blend and PLA/PBAT Compatibilized Blends with Algae as a Reinforcer
1 Department of Chemical, Metallurgical and Materials Engineering, Tshwane University of Technology, Pretoria, 0001, South Africa
2 Centre for Nanostructures and Advanced Materials, DSI-CSIR Nanotechnology Innovation Centre, Council for Scientific and Industrial Research, Pretoria, 0001, South Africa
3 Department of Chemistry, Faculty of Science, Nelson Mandela University, Port Elizabeth, 6001, South Africa
* Corresponding Author: Sudhakar Muniyasamy. Email:
(This article belongs to the Special Issue: Recent Advances on Renewable Materials)
Journal of Renewable Materials 2026, 14(2), 6 https://doi.org/10.32604/jrm.2025.02025-0132
Received 02 June 2025; Accepted 04 October 2025; Issue published 25 February 2026
Abstract
Melt blending of biodegradable polyesters such as poly (lactic acid) (PLA) and poly (butylene adipate co-terephthalate) (PBAT) with a compatibilizer and natural filler offers a chance to develop biodegradable bio-composites with improved performance. In this study, we examined how PLA/PBAT blends behave during ultimate biodegradation (mineralization), both with and without compatibilizer and algae as a reinforcement, under controlled composting conditions using carbon dioxide (CO2) respirometry techniques. Throughout the biodegradation process, the disintegration behaviour, thermal, chemical, and morphological properties of test samples before and after biodegradation were analyzed using FTIR, TGA, DSC, and SEM techniques. The results from CO2 biodegradation showed that PLA/PBAT blend exhibits a higher rate of biodegradation compared to neat PLA and PBAT. The addition of algae to a compatibilized PLA/PBAT blend showed an enhanced biodegradation rate due to hydrolytic cleavage and microbial assimilation. This was further supported by the disintegration test, where algae-reinforced composites showed fragmentation within 30 days. FTIR, TGA and SEM analysis revealed the structural changes that occurred during biodegradation, highlighting the role of algae in affecting the thermal stability and surface morphology. After the compost biodegradation step, eco-toxicity seed germination was conducted on the test samples. Plant seed germination test results confirmed that all test samples achieved maximum germination. This indicates there were no toxic residues, suggesting that the degraded materials are environmentally safe. Overall, this study contributes to the understanding of biodegradation mechanisms and the ecological impact of bio-based polymer composites as eco-friendly materials and products.Graphic Abstract
Keywords
Petroleum-based plastics are the major source of plastic pollution globally. These plastics are resistant to degradation and are durable, which leads to serious environmental issues. They can harm wildlife by introducing toxic substances into the food chain, which can ultimately pose risks to human health [1]. Therefore, it is important to develop eco-friendly materials to address the problem of single-use plastics that are not recycled and contribute to plastic pollution. The transition to biobased plastics has a greater advantage over fossil-based non-biodegradable plastics. The push for developing bio-based biodegradable plastics stems from their unique characteristics, including mechanical properties, versatile physicochemical properties and biodegradability after their end-of-life across various industrial applications. This type of plastic, when disposed of, is easily chemically converted into natural components such as carbon dioxide (CO2), methane CH3, and biomass by the action of natural microorganisms and leaving no toxic residues. This helps reduce the pressure on overflowing landfills and minimizes plastic pollution [2]. Additionally, using biodegradable plastics in various applications helps lower greenhouse gas emissions and decrease dependence on fossil fuels. Recently, efforts to adopt biodegradable plastics have mainly focused on producing materials and managing waste effectively [3]. The key drive on biodegradable plastics aligns with the materials design to meet sustainability criteria, namely, regulations, innovation, and effective use of resources. One of the drawbacks of biodegradable plastics is that they are expensive, costing three to five times more than plastic made from petroleum [4]. Thus, there is a need for the development of biodegradable-based products with the desired properties of the biodegradation products for various applications.
Among biodegradable polymers, poly (lactic acid) (PLA) and poly (butylene adipate-co-terephthalate) (PBAT) are types of semicrystalline biodegradable polyesters that can be produced using traditional industrial techniques [5]. Both PLA and PBAT have inherited limitations that make them less suitable for some uses. For instance, PLA is known for being very brittle, having low toughness, and poor processability [6]. On the other hand, PBAT suffers from low heat resistance, higher production cost, and lower stiffness [7]. To overcome these issues and achieve better overall properties, blending PLA and PBAT is an effective solution. Research shows that combining PLA with PBAT can improve PLA toughness and crystallization [8–10]. However, blending PLA and PBAT tends to form immiscible blends, which compromise the desired properties of the blends due to inadequate compatibilization between the polymers. Adding a compatibilizer, such as poly (lactic acid) grafted maleic anhydride (PLA-g-MA), can enhance the compatibility between PLA and PBAT. Cai et al. [11] showed that introducing a micro-crosslinked PBAT phase containing the initiator bis (1-(tert-butylperoxy)-1-methylethyl) benzene and 1,3,5-tri-2-propenyl-1,3,5-triazine-2,4,6 (1H, 3H, 5H)-trione effectively improved impact strength while maintaining tensile strength and modulus. Similarly, Hong et al. [12] developed reactive micro-crosslinked PBAT tougheners via melt blending with glycidyl methacrylate (GMA) and dicumyl peroxide (DCP), which enhanced the toughness of the blends.
PBAT is a type of biodegradable polyester derived from petroleum [13]. It is known for being flexible and having good ductility. Generally, PBAT has low strength compared to other plastics, which can be improved by blending it with other biodegradable materials such as PLA and polyhydroxybutyrate (PHB). One of the effective methods to enhance the properties of a bio-polymer blend involves reinforcing a compatibilized binary blend (PLA/PBAT) with low-cost renewable additives such as calcium carbonate, sawdust, chitosan, algae, and others. Many reviews explored efforts to improve the tensile strength and modulus of PLA/PBAT blends by incorporating fillers such as calcium carbonate [14,15], straw fibre [16], micro zinc oxide [17], and algae [1]. For instance, Correa-Pacheco et al. [18] investigated the effect of adding prickly pear flour to a biodegradable PLA/PBAT polymer blend. Their result indicated that after six months, the composites containing prickly pear flour and coupling agent resulted in accelerated biodegradation. Similarly, Mhd Ramle et al. [19] examined the biodegradation of PLA/PBAT blends reinforced with cellulose from bamboo. Their findings indicated that the biodegradability was enhanced at higher loading of cellulose after a time frame of 15 days. Typically, PLA undergoes a two-step degradation process. First, it undergoes hydrolytic degradation caused by environmental abiotic factors such as humidity, temperature and ultraviolet radiation or by enzymes from living organisms. This process breaks the polymer down into water-soluble products that microorganisms further decompose into carbon dioxide and water [20]. In contrast, PBAT biodegradation mainly relies on microbial activity that helps break down the PBAT ester bonds rather than just environmental factors [21]. During this breakdown process, the amorphous structure of butylene adipate (BA) degrades faster than the crystalline portion of the butylene terephthalate (BT) structure [20].
Numerous studies indicate that PLA is one of the most extensively researched bio-based polymers compared to other renewable-based polymers. However, PLA degrades best in industrial composting conditions and not as effectively in natural environments such as soil, anaerobic digesters and marine water [19]. It is widely recognized that composting of materials in a closed system helps lower methane gas emissions, which significantly contribute to climate change, when compared to uncontrolled landfills. The biodegradation of PLA, PBAT and PLA/PBAT composite materials has been evaluated using weight loss and various analytical techniques to assess different physical, chemical, thermal and surface properties [22,23]. Weight loss and other polymer analysis techniques do not directly measure the ultimate biodegradation (mineralization) process. Rather, changes in weight loss or surface structure may indicate partial degradation instead of the complete breakdown of a polymer into simpler substances like carbon dioxide and water. Therefore, conducting complete mineralization tests to monitor CO2 and CH4 evolution is an important criterion to meet claims of eco-friendly material, including the eco-toxicity after biodegradation. The rate at which PLA and PBAT degrade in natural environments is slow. Besides the microorganisms found in biological environments (like soil, compost and water), several abiotic factors (such as temperature and humidity), along with the physical and chemical characteristics of the polymer (like crystallinity and molecular weight), also significantly influence how these materials biodegrade.
Among others, algae are emerging as a promising and affordable biodegradable accelerator used as a filler. They can absorb a large amount of CO2 from the atmosphere, even up to five times more than any other land plant [24]. Nowadays, algae farming is on the rise and is seen as the future of materials and resources. It provides highly nutritious food for a growing population and helps create crops that are carbon negative, which can aid in mitigating climate change. Using algae as a filler in biopolymer composites could be profitable and enhance biodegradability. Kalita et al. [25] assessed the effect of algae biomass as a potential degradation accelerator on PLA matrix. Their findings showed that adding 5 wt% algae to PLA improved its biodegradation. Tsou et al. [26] prepared PBAT/corn stalk composites and evaluated their biodegradability. Both PLA and PBAT are biodegradable polymers, and numerous studies have investigated their blending processes and their resulting properties. Recent efforts have focused on developing PLA/PBAT materials with enhanced degradation characteristics. However, few studies have comprehensively investigated the full mineralization behavior (ultimate biodegradation) of compatibilized PLA/PBAT blend in the presence of algae as a functional reinforcement. Algae functioned both as a structural filler and as a biodegradation accelerator due to their protein-rich, hydrophilic nature and the ability to promote microbial colonization. This study presented a novel formulation and evaluation of algae-reinforced compatibilized PLA/PBAT bio-composite under composting conditions. Unlike most prior work that focused on the mechanical and thermal properties of PLA/PBAT material, this study emphasizes the ultimate biodegradation of the PLA/PBAT blends, with and without a coupling agent, using a significant amount of algae as reinforcement. A comprehensive evaluation of the biodegradation process was carried out to determine the degradation rate of the test samples, considering the influence of physical, thermal, chemical and morphological changes. This was assessed using TGA, DSC, FTIR, SEM, disintegration analysis and CO2 evolution measurements. The eco-toxicity of the final compost was validated to ensure environmental safety. Furthermore, this study offers new insights into the design of fully compostable, eco-safe bio-polymer systems suitable for scalable applications such as packaging and agriculture.
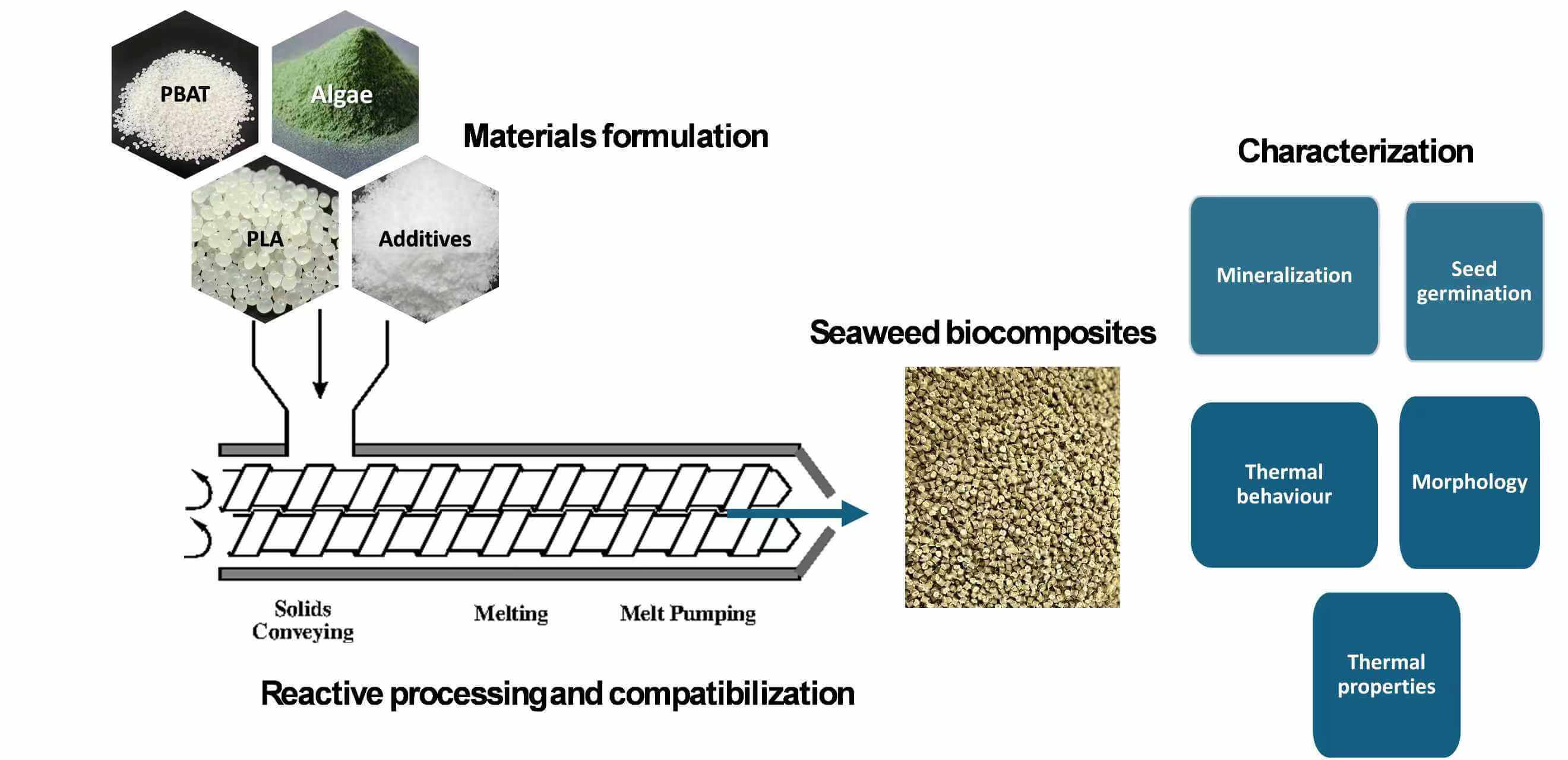
PLA with a commercial grade of Luminy Lx 175 was purchased from Total Energies Corbion. PLA has a glass transition temperature of ~60°C, a melting temperature of 155°C and a density of 1.24 g/cm3. PBAT (C1200) was obtained from BASF, Ludwigshafen, Germany. It has a melting temperature of 120°C and a glass transition temperature of −32°C. The compatibilizer, maleic anhydride-grafted poly (lactic acid), was fabricated in-house with a grafting degree of 3.9%, following the described procedure [27]. Reactive extrusion was employed for the production of a compatibilizer by facilitating a chemical reaction between PLA, dicumyl peroxide and maleic anhydride. During the reactive process, the concentration of maleic anhydride was kept constant at 4 wt%, while DCP ranged between 0 and 0.4 wt%. Algae, sourced from 2M Biotech Engineering Polymer, served as a filler in its as-received state. The algae powder exhibited a particle size distribution (PSD) with D10 = 5.7 µm, D50 = 14.8 µm and D90 =29.5 µm, which were determined in previous work [27]. Biodegradation studies were conducted using 100% organic compost, which was sourced from a local nursery in Tshwane, South Africa.
2.2 Sample Preparation and Analysis
Before the melt extrusion process, PLA and PBAT were dried at 50°C in a desiccant dryer to eliminate moisture and prevent potential hydrolysis of the matrix. Algae was oven-dried at 110°C for 20 h. Table 1 provides a breakdown of the precursors used in the formulations. The PLA/PBAT blend ratio was kept steady at 80:20 for all samples.
The developed formulations were fabricated using a co-rotating twin screw extruder (Model: TE-30/600-11-40, China and L/D = 40). The barrel temperature profile is set in a gradient from the feeding section at 120°C to the die at 160°C. The screw speed rotated at 100 rpm. Subsequently, the extrudate was dissolved in chloroform at room temperature and formed into test film samples that had an average thickness of 15–20 μm.
The biodegradability of the prepared test samples was tested using American Society for Testing and Materials (ASTM) D6400, European Standard (EN) 13432 and Organization for Standardization (ISO) 17088 standard methods under industrial composting conditions at a temperature of 58 ± 2.0°C. The compost used was an aerated mixture that was three months old and rich in organic material, sourced from GardenMaster Compost in Pretoria, South Africa. To ensure a consistent particle size for testing, the compost was passed through a sieve with openings smaller than 0.8 cm. Its physical and chemical characteristics are listed in Table 2. The activity of the compost, measured by volatile solids, resulted in 63.0 mg of CO2 per gram of volatile solids in the first 10 days, satisfying standard criteria.
Echo Automated closed respirometer connected to CO2, CH4 and oxygen (O2) sensors was employed to study the ultimate biodegradability (CO2 evolution) of the test samples and microcrystalline cellulose (positive reference) under controlled composting conditions at 58°C–60°C. 2 L bioreactors were filled with the mixture of 300 g total dry solids of compost and 50 g dry solids of the test samples at the ratio of 6:1. The bioreactors were placed in a system with diffuse light and maintained at a constant temperature of 58 ± 2°C. Throughout the experiment, total CO2 released from each reactor was measured and taken as an indication of how much the test samples degraded. Biodegradation percentages were determined from the total organic carbon (Ct) obtained via elemental analysis, while theoretical CO2 (CO2(t)) was calculated from the dry weight of the test material using Eq. (1).
Mt represents the total dry weight of plastic material incorporated into the compost, while Ct indicates the amount of total organic carbon found in the dry plastic material. The proportion of organic carbon mineralized into CO2, reflecting the level of biodegradation, was determined using Eq. (2). In this equation, (CO2)s refers to the carbon dioxide evolved from the test sample (compost combined with the specimen), (CO2)c is the carbon dioxide released from the blank compost and (CO2)t is the theoretical maximum amount of carbon dioxide that could be generated from the material. A biodegradation profile was generated by plotting the percentage values against incubation time.
Disintegration testing is an initial step in assessing degradation, focusing on whether the material physically breaks down into smaller fragments. This test was conducted using the same compost as in the CO2 biodegradation experiments and was carried out under controlled conditions at a temperature of 58 ± 2.0°C. Approximately 5 g of polymer pellets were measured and placed in a glass watch dish, followed by the addition of 20 mL of chloroform to dissolve the polymer. The chloroform was allowed to evaporate in a fume hood, which created a thin film. The film samples were positioned on the compost surface inside the bioreactor, and their disintegration or fragmentation was monitored periodically for up to 70 days.
2.2.3 Fourier Transform Infrared Spectroscopy (FTIR) Analysis
Changes in chemical properties of the produced samples were evaluated by a Spectrum 100 Fourier transform infrared spectroscopy (FTIR) (Perkin Elmer, Waltham, MA, USA). The test was performed in the attenuated total reflection (ATR) spectra mode within the spectrometer range of 400–4000 cm−1. Measurements were taken at three stages, namely, prior to biodegradation, after 30 and 60 days of biodegradation.
The carbonyl index (CI) was determined as the ratio of the area under the carbonyl absorption peak to that of the CH bending peak, serving as an indicator of ester bond hydrolysis, as in Eq. (3) [28,29].
2.2.4 Thermogravimetry Analysis (TGA)
The thermal behaviour of the bio-composites was carried out on a Q500 Thermogravimetric Analyzer (TA Instrument, New Castle, USA). Data analysis was performed using universal analysis software. Approximately 10 mg of each specimen was placed on a platinum pan and subjected to heating from room temperature to 900°C at a constant heating rate of 10°C/min under a nitrogen atmosphere. Each reported value represents the mean of three replicate measurements per sample.
2.2.5 Differential Scanning Calorimetry (DSC)
Differential Scanning Calorimetry (DSC) was employed to investigate the thermal properties of the produced samples using DSC-2500 (TA instrument, New Castle, USA), under a constant nitrogen purge of 20 mL/min. Samples weighing 5–6 mg were stabilized at −65°C for 1 min, then heated at 10°C/min to 200°C, and held for 1 min. The cooling cycle was subsequently performed at 10°C/min to −65°C to monitor crystallization behaviour, followed by reheating to 200°C to determine melting characteristics. The results reported represent the average of three replicates per sample.
The microstructure of the bio-composites before and after biodegradation was examined using a field-emission scanning electron microscope (JSM–7500, JOEL, Tokyo, Japan) at an acceleration voltage of 3 kV. Prior to imaging, samples were sputter–coated with gold to enhance conductivity.
The seed germination test was used to assess the possible effects of test samples mixed into compost on the growth and development of land plants, in accordance with the Organization for Economic Co-operation and Development (OECD) guideline 208 for the terrestrial Plant Test. After the compost biodegradation tests, 5 g of compost from each reactor were mixed with 5 g of fresh agricultural soil and 5 tomato seeds were planted in it. The pH of the compost-soil mixture was determined, and the results were between 7.2–7.4. Water was added to maintain the relative humidity of the compost-soil mixture between 50%–55%. Experiments were carried out in triplicate under controlled conditions of temperature, light, and moisture, to simulate natural growth environments. Observations are made over a specific period to assess parameters like seed germination rate, root and shoot growth, and any visible signs of toxicity.
Biodegradation studies were carried out on the test samples for 180 days of composting under controlled conditions, using cellulose as the comparative reference. The amount of CO2 evolved from the test samples was measured to assess the extent of biodegradation (mineralization), as illustrated in Fig. 1. The cellulose reference sample reached 40% biodegradation with an acceleration phase within 15 days and reached more than 70% biodegradation within 45 days. This demonstrates that the biodegradation setup used complied with the test method validity criteria outlined in the ASTM D5338. The cellulose reached 90% biodegradation after 80 days and exhibited a plateau phase afterwards, as expected. It is known that cellulose is a readily biodegradable material under composting conditions.
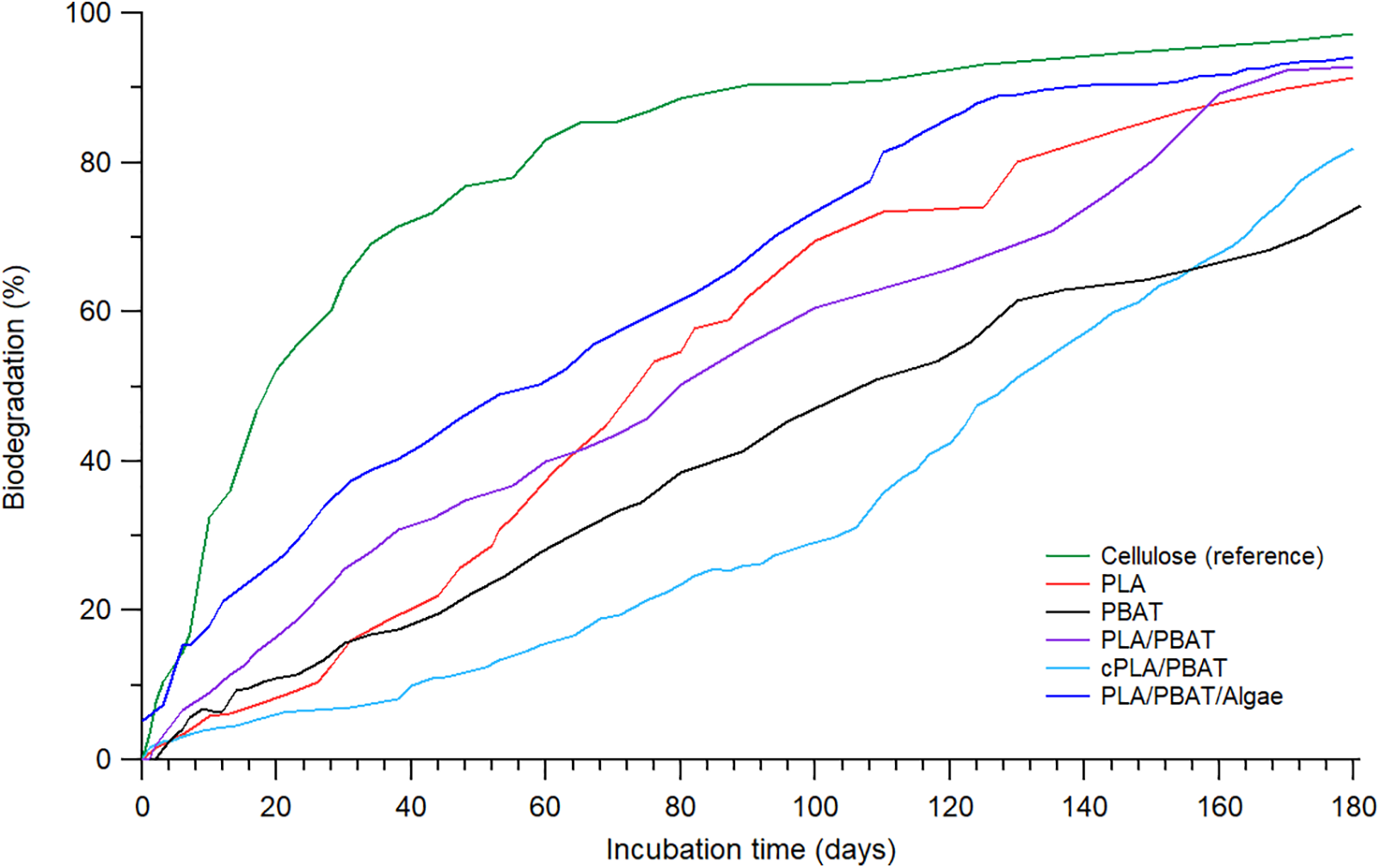
Figure 1: Biodegradation characteristics of cellulose and the test samples under composting conditions as measured by CO2 respirometric analysis for 180 days
Biodegradation under composting conditions proceeded through a multi-step mechanism, starting with abiotic hydrolysis, followed by microbial assimilation and enzymatic cleavage. The biodegradation behaviours of PLA began slowly over the first 30 days and thereafter started an acceleration phase, achieving 70% biodegradation within 90 days. After that, it levelled off, reaching 85% biodegradation by day 160. It is well known that PLA breaks down through hydrolytic biodegradation in industrial composting conditions. Factors like relative humidity and temperatures between 58°C–60°C significantly help break long-chain molecules into smaller ones, and followed by microbial assimilation into final products such as CO2, H2O and microbial biomass [25]. On the other hand, neat PBAT showed a slow biodegradation process, with only 20% degradation in the first 60 days. This was followed by an acceleration phase that led to 70% degradation after 180 days. The slow degradation may be attributed to the crystalline butylene terephthalate units, which resist enzymatic attack, while the aliphatic butylene adipate segments degrade more readily through hydrolysis [20]. In contrast, PLA/PBAT blend samples demonstrated a faster biodegradation rate of 25% within the first 30 days compared to both neat PLA and neat PBAT. The improved degradation of the PLA/PBAT blend likely results from breaking down its crystalline structure into more amorphous areas, allowing for ester cleavage between water and nearby carbonyl groups around benzene rings. However, the compatibilized cPLA/PBAT blend degraded more slowly than neat PLA and neat PBAT because the compatibilizer assists in facilitating reactions and compatibility between maleic anhydride and PLA/PBAT, rearranging amorphous groups into a more crystalline structure, which restricts water diffusion and microbial access. Interestingly, the algae-reinforced compatibilized PLA/PBAT blend showed a biodegradation rate due to protein molecules and other water-soluble compounds distributed throughout the biopolymer matrix. These compounds influence hydrolytic chain cleavage and microbial assimilation processes. Table 3 provides a summary of the biodegradation performance of the tested samples and similar materials. Studies focusing on compatibilized PLA/PBAT reinforced composites evaluated using mineralization methods remain scarce. Most literature reports emphasize weight loss or visual disintegration, while systematic mineralization (CO2 evolution) studies are limited. In this context, the present work fills an important gap by providing mineralization data for a reinforced compatibilized PLA/PBAT composite and shows the role of algae as a bio-filler that supports biodegradation performance.
Disintegration is the first stage in the breakdown process of polymer materials. This process is catalysed due to environmental factors or enzymes from living organisms or the combination of both factors. During this stage, the polymer material breaks down into oligomeric units. Therefore, a disintegration test provides insight into whether a plastic material experiences the primary phase of biodegradation. During this step, plastics may fragment into lower molecular weight components, which can be accessible for microbial biodegradation in subsequent stages. In this study, film samples of neat PLA, neat PBAT, PLA/PBAT blend, cPLA/PBAT blend and cPLA/PBAT-Algae bio-composite were subjected to disintegration testing. Fig. 2 shows the disintegration of these test samples during a 60-day incubation period.


Figure 2: Photographs of disintegrated films. Neat PLA, neat PBAT, PLA/PBAT blend, cPLA/PBAT blend and cPLA/PBAT-Algae bio-composite
PLA started to undergo disintegration and fragmentation steps after 30 days, as shown in Fig. 2. After 50 days, the sample became very fragile. However, no visible cracks or pieces were observed on the PBAT film during the 50-day incubation period. The PLA/PBAT film started to become fragile and break apart after day 30, and it continued to weaken after day 50. It was observed that cPLA/PBAT became thinner but remained intact beyond day 50. This disintegration behaviour corresponded with its biodegradation rate, showing a slow onset of degradation that accelerated after day 60. In contrast, the addition of algae into cPLA/PBAT blend began to crack and fragment as early as day 20, becoming increasingly brittle after day 50 (Fig. 2).
Fig. 3 presents the FTIR functional groups characterization of test samples before and after biodegradation. Table 4 summarizes the positions of the transmittance peaks along with their related functional groups. In Fig. 3a, the FTIR spectrum of PLA shows distinct absorption bands at 3002 and 2985 cm−1, attributed to CH and CH3. A prominent peak at 1750 cm−1 corresponds to C = O stretching from the ester group, while peaks between 1500–1301 cm−1 are linked to CH3 and CH bending vibrations. Additionally, peaks at 955 and 855 cm−1 correspond to C–C and C–COO vibration of stretching [34]. After PLA undergoes biodegradation, an increase in the peak at 1750 cm−1 corresponding to the carbonyl group is observed over time (Fig. 3a). This increase is due to the hydrolytic breakdown of the ester group into carboxyl groups [35]. PBAT exhibited similar characteristic peaks as PLA because their chemical structure are similar, except for a peak at 730 cm−1 that corresponds to CH-plane bending vibration in the benzene ring [18]. After biodegradation of PBAT, an increase in the band peak at 3110 cm−1 is observed (Fig. 3b), indicating bond cleavage and the formation of -OH groups, characteristic of the hydrolysis process during composting [36].
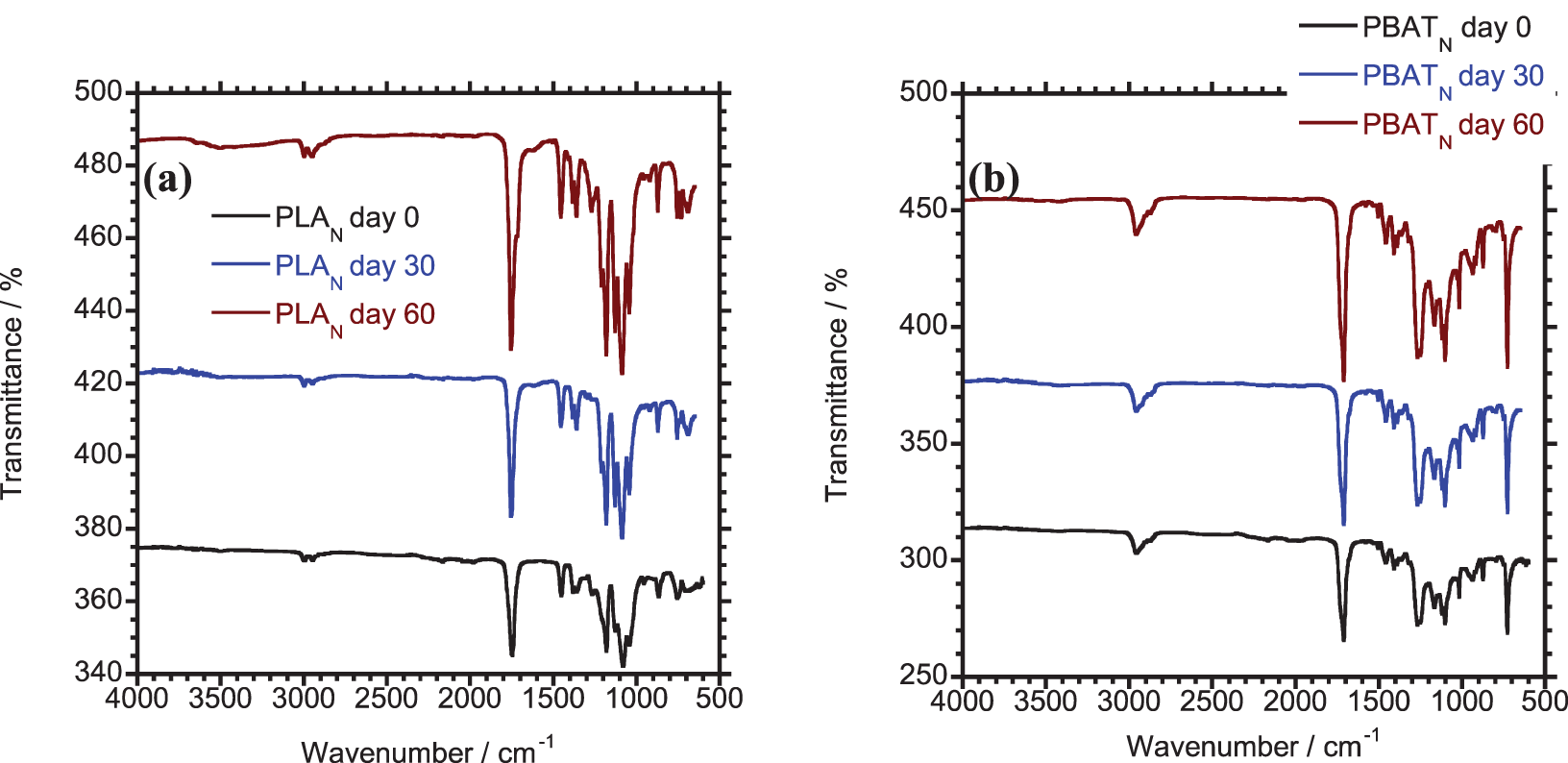
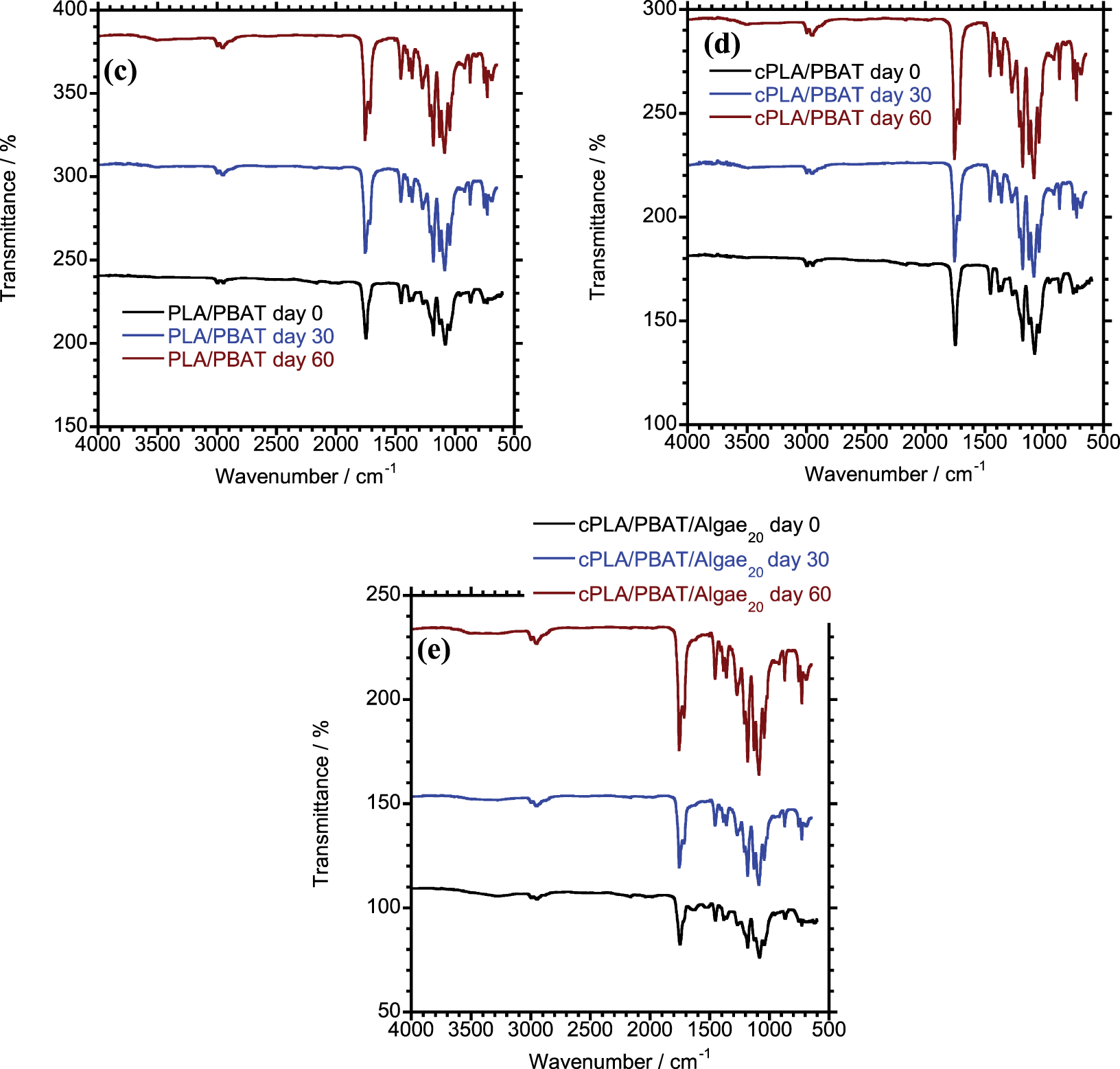
Figure 3: FTIR spectra of (a) PLA (b) PBAT (c) PLA/PBAT (d) cPLA/PBAT (e) cPLA/PBAT/Algae20 before and after biodegradation
Before biodegradation, the characteristic position of PLA/PBAT blend, compatibilized cPLA/PBAT blend and algae reinforced cPLA/PBAT bio-composite reflect the respective peaks for PLA and PBAT (Fig. 3c and d). In comparison, the addition of a coupling agent led to a slight increase in the intensity of the peak band at 1750 cm−1. This improvement is attributed to the effect of MA grafting, which improves the interfacial bonding and compatibility between PLA and PBAT.
As shown in Table 5, the carbonyl index value increased significantly in all composted samples. These findings indicate that the degradation of the samples was initiated by hydrolytic chain scission [37,38]. The chain scission produced shorter polymer chains and consequently generated a high concentration of carboxylic acid end groups [39,40]. The increase was most prominent in the algae-reinforced composite, indicating accelerated degradation. The significantly higher CI in the algae composite suggests enhanced chain scission and ester bond cleavage, supporting the observed increase in CO2 evolution during composting.
Thermogravimetric analysis was performed to investigate the thermal stability and thermal degradation behaviours of neat PLA, PBAT, PLA/PBAT blend, compatibilized blend and algae-filled composites before and during composting. Fig. 4 shows weight loss and derivative curves of the test samples before and after biodegradation. Table 6 shows the thermal properties of the test samples, namely the onset temperature at 5 wt% weight loss (T5), maximum degradation temperature (Tmax), minor degradation temperature (Tminor) and decomposition temperature of the volatile compounds (Td).
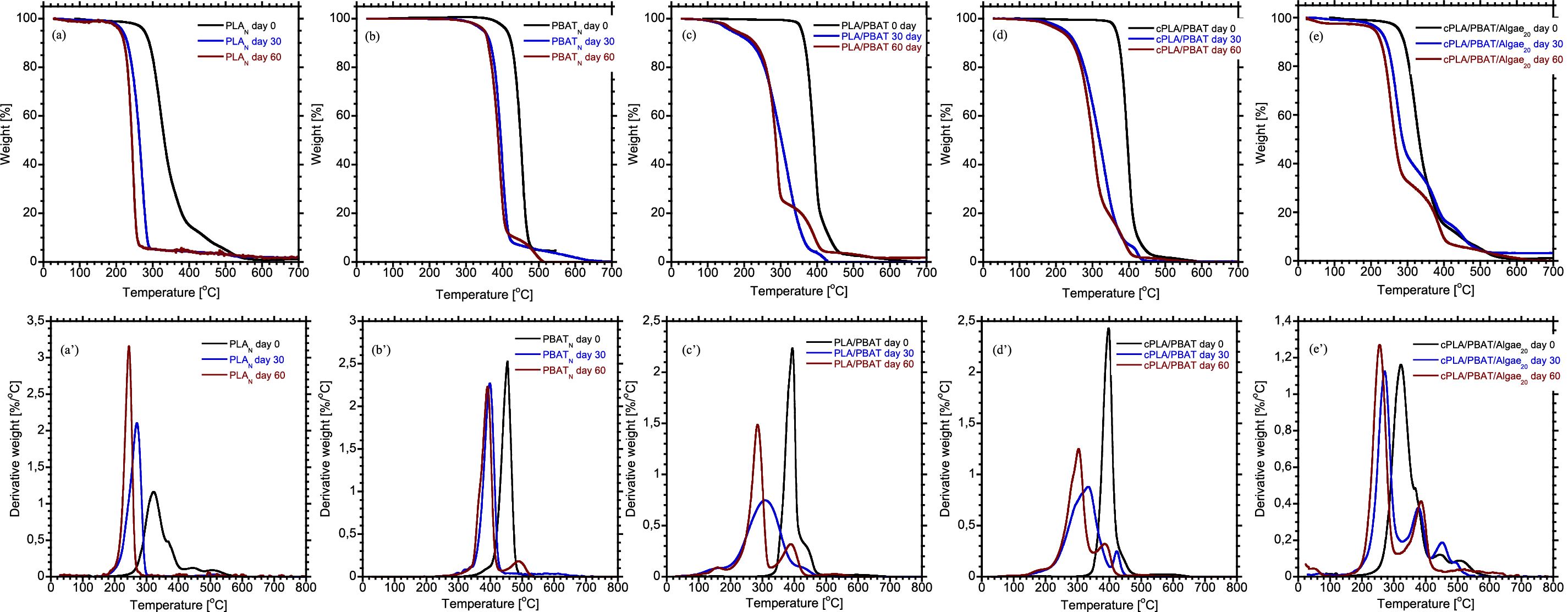
Figure 4: Thermogravimetry analysis (a–e) and derivative thermogravimetric (DTG) analysis (a’–e’)
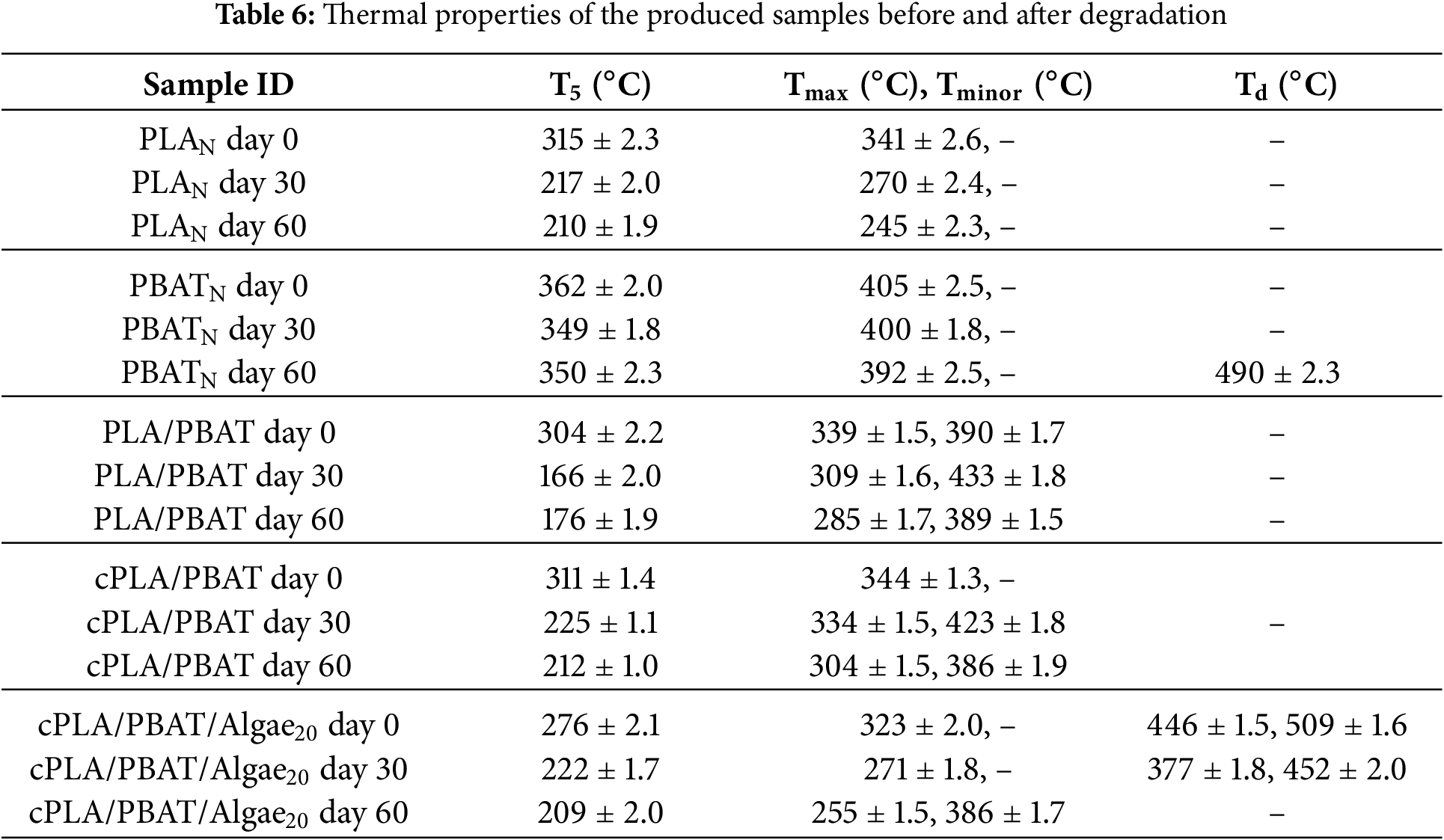
Pure PLA displayed a one-step thermal degradation behaviour, and the maximum degradation temperatures for PLA before and after biodegradation at 30 and 60 days were 341°C, 270°C and 245°C, respectively. These findings show that the long-chain molecules of PLA break down into shorter chains, resulting in reduced molecular weight [41]. Similarly, the thermal decomposition of PBAT decreased during the biodegradation process, affecting both the onset and maximum degradation temperature. However, PBAT maintained a more stable thermal degradation behavior compared to PLA over time. This stability is likely because of abiotic hydrolytic degradation of PLA in industrial composting conditions [5,42]. After 30 days of biodegradation, PBAT showed a second-step weight reduction, characterized by decomposition temperature peaks at 392°C and 490°C, respectively (Fig. 4b’). These temperatures correspond to the breakdown of aromatic and aliphatic components. The blends of PLA/PBAT exhibited two steps in thermal degradation, corresponding temperatures of the PLA and PBAT molecular chains. The incorporation of a compatibilizer into the blend exhibited a similar degradation behaviour to that of the unmodified blend over the degradation period. The compatibilized cPLA/PBAT blend demonstrated improved thermal stability as Tonset, Tmax and Tminor increased compared to those in the binary PLA/PBAT blend. This implies that incorporating PLA-g-MA could have created a barrier effect that restricts the movement of agents that promote degradation into the polymer matrix. Similar results from Teamsinsungvon et al. [43] indicated that adding a coupling agent into the PLA/PBAT blend enhanced the thermal stability of the system. The thermal stability of the biocomposites was altered by adding algae to the compatibilized binary blend. In particular, adding algae reduced the thermal stability compared to the compatibilized blend without algae. This decline in thermal stability could be attributed to weakened interfacial bonding between the components caused by the algae. A poorly bonded interface is more susceptible to hydrolytic degradation and enzymatic attack, which can lead to a faster biodegradation rate. Letwaba et al. [27] in their previous work found that adding algae affects how the compatibilized binary blends behave thermally. The results indicated that at algae content exceeded 10 wt%, the thermal stability of the bio-composites decreased noticeably. Both the onset and maximum degradation temperatures of cPLA/PBAT/Algae decreased progressively with degradation time (Table 6). Furthermore, peaks corresponding to the degradation of volatile compounds in the algae reinforced bio-composites were detected at both the initial stage and after 30 days of degradation. In contrast, these peaks were no longer present after 60 days of degradation. This could suggest that by 60 days, most of the organic components that were prone to volatile decomposition might have decomposed.
Fig. 5 shows the DSC first cooling and their corresponding second heating curves of the produced samples before and after bio-degradation. Meanwhile, Table 7 presents a summary of DSC results of test samples, including the glass transition temperature (Tg), cold crystallization temperature (Tcc), melting temperature (Tm), crystallization temperature (Tc) and percentage crystallinity (Xc). During the biodegradation processes, the PLA biodegradation glass transition temperature and the melting temperature before and after biodegradation were reduced from 58°C and 152°C to 49°C and 136°C, respectively. Additionally, the crystallinity of PLA reduced as a function of biodegradation duration. This behaviour is primarily attributed to the degradation of PLA polymer chains, leading to a less ordered polymer structure. Fu et al. [5] reported the same behaviour of degradation of PLA in fresh water. For PBAT, the melting and crystallization temperatures before biodegradation were at 123°C and 74°C, respectively. After undergoing biodegradation, the melting temperature slightly increased to 126°C while the crystallization temperature increased to 84°C. However, despite these changes in Tm and Tc, the overall crystallinity of PBAT remained stable during biodegradation. This suggests that a more stable and organized crystalline structure forms due to chain breaking and reorganization.
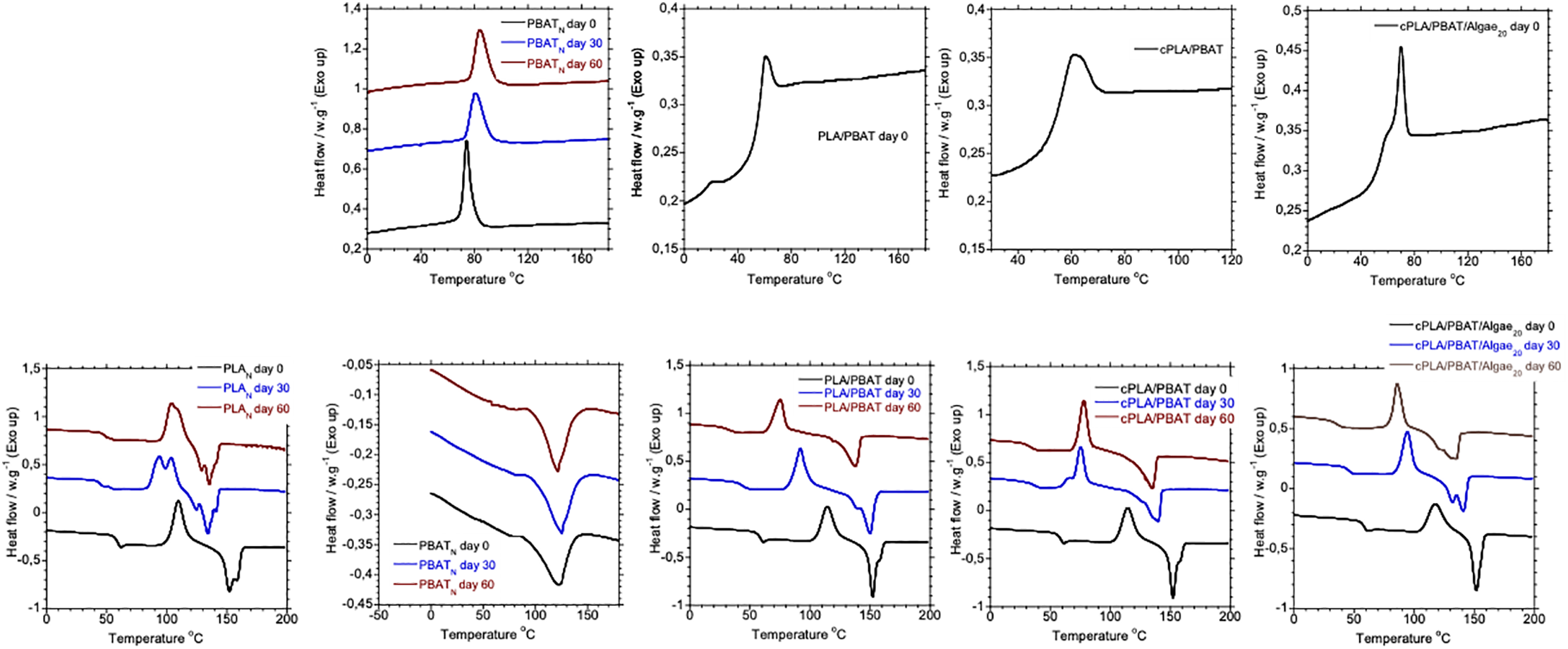
Figure 5: DSC first cooling curves (Top) and the corresponding second heating curves (Bottom)
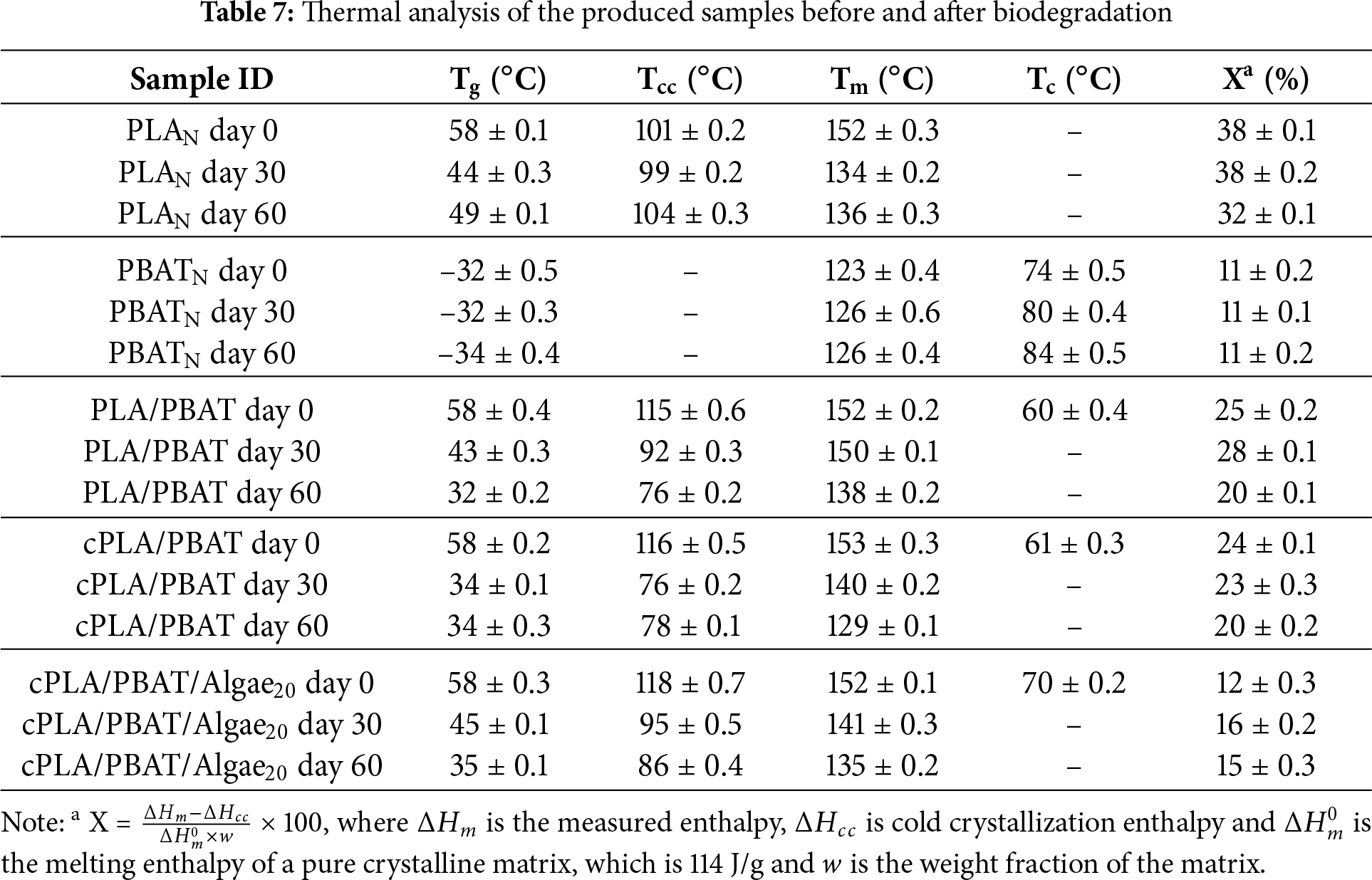
The thermal properties of PLA/PBAT blend and compatibilized cPLA/PBAT did not show significant changes. However, after 30 days of biodegradation, no crystallization temperature was detected in the blends and algae-reinforced PLA/PBAT bio-composite. This phenomenon may indicate a reduction in the molecular weight of the polymers in the blend, causing the chains to become too short to crystallize effectively. Consequently, this chain shortening can prevent the formation of new crystalline regions, resulting in no detectable crystallization temperature. Adding a compatibilizer to the PLA/PBAT blend did not significantly change its thermal properties after 60 days of biodegradation when compared to the uncompatibilized blend. Additionally, after 60 days of biodegradation, adding algae to the compatibilized blend resulted in Tg, Tcc, Tm, and XC values of 35°C, 86°C, 135°C, and 15%, respectively. The presence of algae in the compatibilized blend reduced the degree of crystallization over time compared to both the neat and the compatibilized blends. This may occur because the algae interfere with the crystalline regions of the polymers, making them more amorphous and easier for microbes to break down.
Fig. 6 displays the surface structure of the tested samples both before and after biodegradation, as analyzed by SEM. The results show that all samples developed cracks and holes as the biodegradation time increased. For PLA, the defects became more severe over the biodegradation period (Fig. 6). PLA breaks down more easily in its amorphous regions compared to crystalline regions because water and enzymes can access the softer amorphous structure more easily. This quicker access leads to faster breakdown through hydrolysis and microbial action, resulting in the disintegration of the polymer chain [25]. After biodegradation, the surface of PBAT exhibited a noticeable change, with numerous protrusions and eroded regions. This change is due to the biodegradation process, where the amorphous regions of PBAT are more rapidly degraded, leading to a rough and uneven surface (Fig. 6).
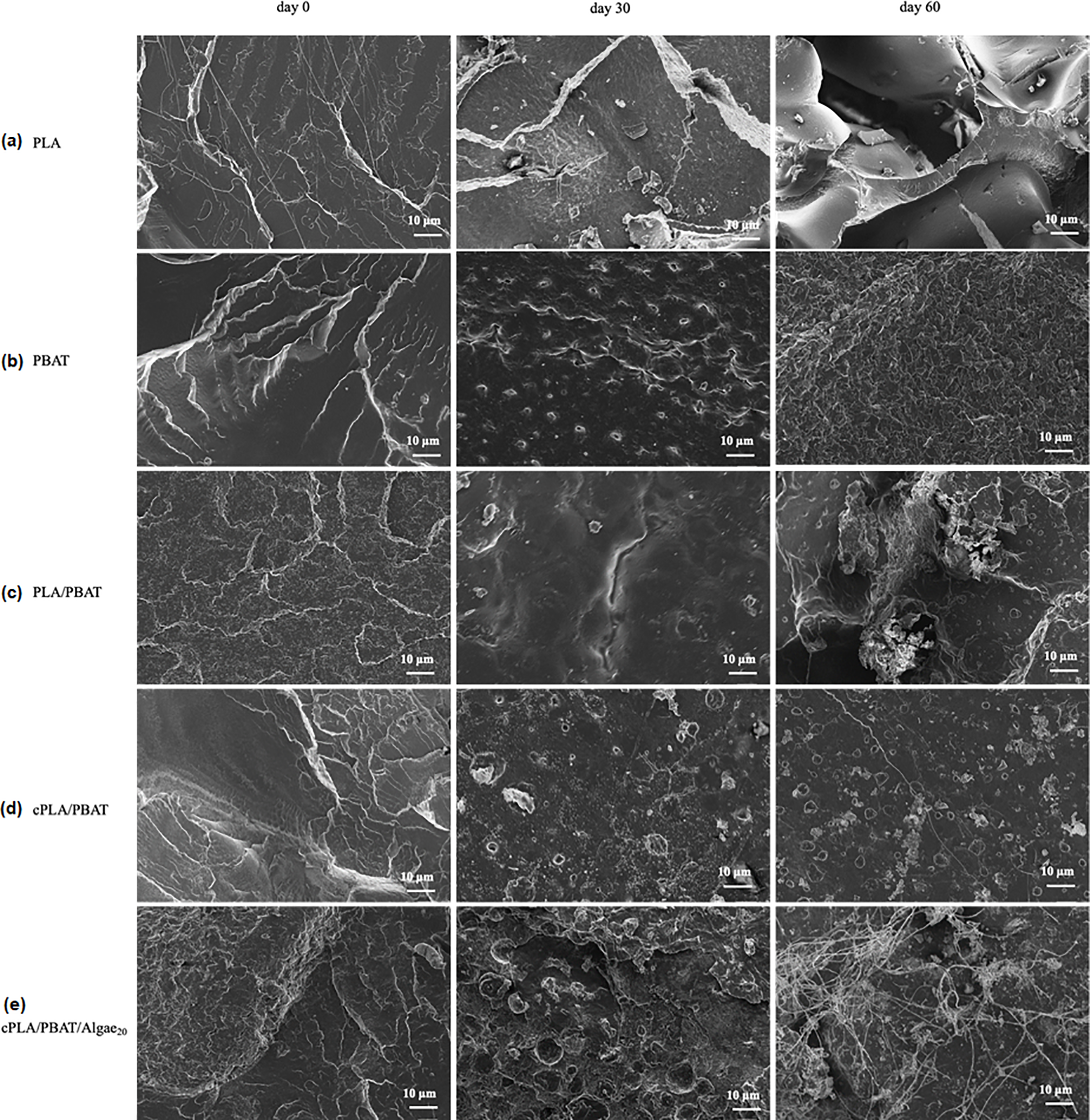
Figure 6: SEM micrographs of (a) PLA (b) PBAT (c) PLA/PBAT (d) cPLA/PBAT (e) cPLA/PBAT/Algae20 before and after biodegradation
Fig. 6 shows the surface morphology of PLA/PBAT blend before and after biodegradation. Before biodegradation, the surface of this blend shows that it has an immiscible two-phase system. After 30 days of biodegradation, noticeable surface defects such as cracks and voids appear on the blend (Fig. 6). After 60 days of biodegradation, the test sample tends to become more fragile (Fig. 6). This suggests that the amorphous regions in the blend are susceptible to hydrolytic breakdown. Adding a coupling agent to the blend improved compatibility between PLA and PBAT, resulting in a less porous and compact structure (Fig. 6). The coupling agent helps reduce the interfacial space of the binary blend caused by the enhanced interfacial interaction [43,44]. As a result, the compatibilized blend shows fewer surface imperfections compared to the neat blend during biodegradation. This improvement may be due to its compact structure, which limits microorganisms’ access to the polymer chains and slows down biodegradation. The incorporation of algae into the compatibilized blend altered the morphology of the compatibilized binary blend as expected (Fig. 6). As the biodegradation incubation time increases, surface defects, namely corrosive holes and the formation of protruding ribs, are noticeable. This indicates that microorganisms are breaking down the material and altering its structure. Similarly, Kalita et al. [25] observed that adding algae to the PLA matrix induces microbial attachment on the surface of bio-composites.
3.6 Evaluation of the Environmental Safety Assessment of Compost
The compost obtained after biodegradation testing was visually and functionally evaluated to assess its basic environmental safety and potential for reuse in soil-related applications. This assessment focused on seed germination, plant development and physical observation, including the presence of abnormal coloration or toxic odors.
Fig. 7 and Table 3 show the germination of tomato seeds within 15 days and the growth of transplanted seedlings after 6 weeks. According to the OECD 208 standard test method, at least 50% of the seeds should germinate in the blank compost–soil mixture. Table 3 and Fig. 7 show that 90%–100% of seeds germinated in the compost–soil mixture in the reactor at the end of the biodegradation tests. These results suggest that there was no negative effect on plant growth after compost biodegradation.

Figure 7: Seed germination testing of compost after biodegradation test
While these results are promising, it is important to note that the current study did not quantify specific bioavailable degradation products (e.g., oligomers, acids, etc). Therefore, this analysis should not be interpreted as a complete ecotoxicological profile.
This study demonstrated that algae reinforced PLA/PBAT bio-composites break down faster than compared to neat PLA, PBAT and PLA/PBAT blends when placed in controlled composting conditions. The enhanced biodegradation behaviour of the composites is primarily attributed to the biochemical composition of algae, particularly their protein and polysaccharide content, which promotes hydrolytic chain scission and facilitates microbial assimilation. These biological components act not only as fillers but also as active agents, accelerating both abiotic and biotic biodegradation processes. Disintegration tests provided visual and structural confirmation of earlier fragmentation in algae-reinforced composite, consistent with the CO2 evolution rate observed during composting. Thermal analysis (TGA and DSC) further supported these findings, revealing a decrease in thermal stability compared to the neat material. These effects are attributed to the inherently unstable structure of algae, which weakens interfacial bonding within the polymer matrix and contributes to increased molecular mobility and volatile mass loss at lower temperatures, especially at high algae loading. Chemical and morphological assessments using FTIR and SEM confirmed the progression of biodegradation at the molecular and microstructural level. FTIR spectra showed increased carbonyl and hydroxyl band intensities over time, indicating ester bond cleavage, while SEM images revealed pronounced surface erosion and fragmentation, particularly in the algae-filled sample. Importantly, eco-toxicity analysis via seed germination demonstrated that the post-composted materials were non-toxic, affirming the environmental safety of the final biodegradation products. This work contributes to the broader development of sustainable, compostable materials suitable for packaging, agricultural films and single-use items.
Acknowledgement: The authors extend their appreciation to the Centre for Nanostructures and Advanced Materials (CeNAM), for providing the essential facilities and resources for conducting this research.
Funding Statement: The authors received no specific funding for this study.
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, John Letwaba and Sudhakar Muniyasamy; Design of research work, John Letwaba, Nagarethinam Rakku, Lucey Mavhungu and Sudhakar Muniyasamy; Material processing and characterization, John Letwaba and Sudhakar Muniyasamy; Writing—review and editing, John Letwaba, Nagarethinam Rakku, Lucey Mavhungu and Sudhakar Muniyasamy. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: The data that support the findings of this study are available from the corresponding author, Sudhakar Muniyasamy, upon reasonable request.
Ethics Approval: Not applicable. For studies not involving humans and animals.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
References
1. Letwaba J, Muniyasamy S, Lekalakala R, Mavhungu L, Mbaya R. Design of compostable toughened PLA/PBAT blend with algae via reactive compatibilization: the effect of algae content on mechanical and thermal properties of bio-composites. J Appl Polym Sci. 2024;141:e55204. doi:10.1002/app.55204. [Google Scholar] [CrossRef]
2. Moshood TD, Nawanir G, Mahmud F, Mohamad F, Ahmad MH, AbdulGhani A. Biodegradable plastic applications towards sustainability: a recent innovations in the green product. Cleaner Eng Technol. 2022;2022(6):100404–17. doi:10.1016/j.clet.2022.100404. [Google Scholar] [CrossRef]
3. Mehta N, Cunningham E, Roy D, Cathcart A, Dempster M, Berry E, et al. Exploring perceptions of environmental professionals, plastic processors, students and consumers of bio-based plastics: informing the development of the sector. Sustain Prod Consumption. 2021;26:574–87. doi:10.1016/j.spc.2020.12.015. [Google Scholar] [CrossRef]
4. Yaguchi Y, Takeuchi K, Waragai T, Tateno T. Durability evaluation of an additive manufactured biodegradable composite with continuous natural fiber in various conditions reproducing usage environment. Int J Automot Technol. 2020;14:959–65. doi:10.20965/ijat.2020.p0959. [Google Scholar] [CrossRef]
5. Fu Y, Wu G, Bian X, Zeng J, Weng Y. Biodegradation behavior of poly(butylene adipate-co-terephthalate) (PBATpoly(lactic acid) (PLAand their blend in freshwater with sediment. Molecules. 2020;25:3946. doi:10.3390/molecules25173946. [Google Scholar] [PubMed] [CrossRef]
6. Plamadiala I, Croitoru C, Pop MA, Roata IC. Enhancing polylactic acid (PLA) performance: a review of additives in fused deposition modelling (FDM) filaments. Polymers. 2025;17:191. doi:10.3390/polym17020191. [Google Scholar] [PubMed] [CrossRef]
7. Venkatesan R, Mayakrishnan V, Alrashed MM, Kim SC. Recent Advances in PBAT (nano) composite materials for food packaging: a comprehensive review. J Appl Polym Sci. 2025;142(28):e57163. doi:10.1002/app.57163. [Google Scholar] [CrossRef]
8. Ding Y, Lu B, Wang P, Wang G, Ji J. PLA-PBAT-PLA tri-block copolymers: effective compatibilizers for promotion of the mechanical and rheological properties of PLA/PBAT blends. Polymer Degrad Stab. 2018;147:41–8. doi:10.1016/j.polymdegradstab.2017.11.012. [Google Scholar] [CrossRef]
9. Chen J, Rong C, Lin T, Chen Y, Wu J, You J, et al. Stable co-continuous PLA/PBAT blends compatibilized by interfacial stereocomplex crystallites: toward full biodegradable polymer blends with simultaneously enhanced mechanical properties and crystallization rates. Macromolecules. 2021;54:2852–61. doi:10.1021/acs.macromol.0c02861. [Google Scholar] [CrossRef]
10. Nofar M, Salehiyan R, Ciftci U, Jalali A, Durmus A. Ductility improvements of PLA-based binary and ternary blends with controlled morphology using PBAT, PBSA, and nanoclay. Compos Part B Eng. 2020;182:107661. doi:10.1016/j.compositesb.2019.107661. [Google Scholar] [CrossRef]
11. Cai K, Wang Q, Liu X, Tu S, Wang J, Feng J. PLA/PBAT/CaCO3 composites with balanced super-toughness and stiffness through dynamic vulcanization and double interfacial compatibilization. ACS Appl Polym Mater. 2024;6:13378–88. doi:10.1021/acsapm.4c02812. [Google Scholar] [CrossRef]
12. Hong J, Zhang J, Cao X, Wang H, Li Y. Designing of reactive micro-crosslinked PBAT as the efficient biodegradation toughener for PLLA. Polymer. 2023;282:126167. doi:10.1016/j.polymer.2023.126167. [Google Scholar] [CrossRef]
13. Letwaba J, Motloung MP, Muniyasamy S, Mavhungu L, Mbaya R, Okpuwhara R. Algae infused enhancement of PBAT stiffness: investigating the influence of algae content on mechanical and thermal propertis. J Appl Polym Sci. 2024;141:e55679. doi:10.1002/app.55679. [Google Scholar] [CrossRef]
14. Teamsinsungvon A, Ruksakulpiwat Y, Jarukumjorn K. Poly(lactic acid)/Poly(butylene adipate-co-terephthalate) blend and its composite: effect of maleic anhydride grafted poly(lactic acid) as a compatibilizer. Adv Mater Res. 2011;410:51–4. doi:10.4028/www.scientific.net/amr.410.51. [Google Scholar] [CrossRef]
15. Jia X, Wen Q, Sun Y, Chen Y, Gao D, Ru Y, et al. Preparation and performance of PBAT/PLA/CaCO3 composites via solid state shear milling technology. Polymers. 2024;16:2942. doi:10.3390/polym16202942. [Google Scholar] [PubMed] [CrossRef]
16. Feng J, Zhang W, Wang L, He C. Performance comparison of four kinds of straw/PLA/PBAT wood plastic composites. BioResources. 2020;15:2596–604. doi:10.15376/biores.15.2.2596-2604. [Google Scholar] [CrossRef]
17. Hussain M, Khan SM, Shafiq M, Khan SU, Raza MR, Tlili I. A study on mechanical strength and biodegradability for sustainable packaging applications. J Korean Phys Soc. 2025;86:180–8. doi:10.1007/s40042-024-01244-y. [Google Scholar] [CrossRef]
18. Correa-Pacheco ZN, Bautista-Banos S, Benitez-Jimenez JJ, Ortega-Gudino P, Cisneros-Lopez EO, Hernandez-Lopez M. Biodegradable assessment of prickly pear waste polymer fibers under soil composting. Polymers. 2023;15:4164. doi:10.3390/polym15204164. [Google Scholar] [PubMed] [CrossRef]
19. Mhd Ramle SF, Ahmad NA, Mohammad Rawi NF, Zahidan NS, Geng BJ. Physical properties and soil degradation of PLA/PBAT blends film reinforced with bamboo cellulose. IOP Conf Ser Earth Sci. 2020;596(1):01202. doi:10.1088/1755-1315/596/1/012021. [Google Scholar] [CrossRef]
20. Weng YX, Jin YJ, Meng QY, Wang L, Zhang M, Wang YZ. Biodegradation behaviour of poly(butylene adipate co terephthalate) (PBATpoly(lactic acid) (PLAand their blend under soil conditions. Polym Test. 2013;32:918–26. doi:10.1016/j.polymertesting.2013.05.001. [Google Scholar] [CrossRef]
21. Kijchavengkul T, Auras R, Rubino M, Selke S, Ngouajio M, Fernandez RT. Biodegradation and hydrolysis rate of aliphatic aromatic polyester. Polym Degrad Stab. 2010;95:2641–7. doi:10.1016/j.polymdegradstab.2010.07.018. [Google Scholar] [CrossRef]
22. Tavares LB, Ito NM, Salvadori MC, Santos DJD, Rosa DS. PBAT/kraft lignin blend in flexible laminated food packaging: peeling resistance and thermal degradability. Polym Test. 2018;67:169–76. doi:10.1016/j.polymertesting.2018.03.004. [Google Scholar] [CrossRef]
23. Lors C, Leleux P, Park CH. State of the art on biodegradability of bio-based plastics containing polylactic acid. Front Mater. 2025;11:1476484. doi:10.3389/fmats.2024.1476484. [Google Scholar] [CrossRef]
24. Sudhakar K, Suresh S, Premalatha M. An overview of CO2 mitigation using algae cultivation technology. Int J Chem Res. 2011;3:110–7. [Google Scholar]
25. Kalita NK, Damare NA, Hazarika D, Bhagabati P, Kalamdhad A. Biodegradation and characterization study of compostable PLA bioplastic containing algae biomass as potential degradation accelerator. Environ Chall. 2021;3(4):100067. doi:10.1016/j.envc.2021.100067. [Google Scholar] [CrossRef]
26. Tsou CH, Chen ZJ, Yuan S, Ma ZL, Wu CS, Yang T, et al. The preparation and performance of poly(butylene adipate) terephthalate/corn stalk composites. Curr Res Green Sustain Chem. 2022;5:100329. doi:10.1016/j.crgsc.2022.100329. [Google Scholar] [CrossRef]
27. Letwaba J, Makwakwa D, Muniyasamy S, Motloung M, Moropeng L. Melt extruded polylactic acid (PLA)/algae bio-composites: effect of grafting level and filler loading on thermal and mechanical properties. J Appl Polym Sci. 2023;46:e54669. doi:10.1002/app.54669. [Google Scholar] [CrossRef]
28. Palsikowski PA, Kuchnier CN, Pinheiro IF, Morales AR. Biodegradation in soil of PLA/PBAT blends compatibilized with chain extender. J Polym Environ. 2018;26:330–41. doi:10.1007/s10924-017-0951-3. [Google Scholar] [CrossRef]
29. Nomadolo N, Dada OE, Swanepoel A, Mokhena T, Muniyasamy S. A comparative study on the aerobic biodegadation of the biopolymer blends of poly (butylene succinatepoly (butylene adipate terephthalate) and poly(lactic acid). Polymers. 2022;14:1894. doi:10.3390/polym14091894. [Google Scholar] [PubMed] [CrossRef]
30. Drumright RE, Gruber PR, Henton DE. Polylactic acid technology. Adv Mater. 2000;12:1841–6. doi:10.1002/1521-4095(200012)12:23<1841::aid-adma1841>3.0.co;2-e. [Google Scholar] [CrossRef]
31. Muniyasamy S, Anstey A, Reddy MM, Misra M, Mohanty A. Biodegradability and compostability of lignocellulosic based composite materials. J Renew Mater. 2013;1:253–72. doi:10.7569/jrm.2013.634117. [Google Scholar] [CrossRef]
32. Parida M, Jena T, Mohanty S, Nayak SK. Advancing sustainable agriculture: evaluation of Poly (lactic acid) (PLA) based mulch films and identification of biodegrading microorganisms among soil microbiota. Int J Biol Macromol. 2024;269(10):132085. doi:10.1016/j.ijbiomac.2024.132085. [Google Scholar] [PubMed] [CrossRef]
33. Harada J, de Souza AG, de Macedo JR, Rosa DS. Soil culture: influence of different natural fillers incorporated in biodegradable mulching film. J Mol Liq. 2019;273:33–6. doi:10.1016/j.molliq.2018.09.109. [Google Scholar] [CrossRef]
34. Verginio GEA, Montanheiro TLdo A, Montagna LS, Marini J, Passador FR. Effectiveness of the preparation of maleic anhydride grafted poly(lactic acid) by reactive processing for poly(lactic acid)/carbon nanotubes nanocomposites. J Appl Polym Sci. 2020;138:e50087. doi:10.1002/app.50087. [Google Scholar] [CrossRef]
35. Chen W, Qi C, Li Y, Tao H. The degradation investigation of biodegradable PLA/PBAT blend: thermal stability, mechanical properties and PALS analysis. Radiat Phys Chem. 2021;180:109239. doi:10.1016/j.radphyschem.2020.109239. [Google Scholar] [CrossRef]
36. Xu J, Feng K, Li Y, Xie J, Wang Y, Zhang Z, et al. Enhanced biodegradation rate of poly(butylene adipate-co-terephthalate) composites using reed fiber. Polymers. 2024;16:411. doi:10.3390/polym16030411. [Google Scholar] [PubMed] [CrossRef]
37. Limsukon W, Rubino M, Rabnawaz M, Lim LT, Auras R. Hydrolytic degradation of poly(lactic acidunravelling correlations between temperature and the three phase structures. Polym Degrad Stab. 2023;217:110537. doi:10.1016/j.polymdegradstab.2023.110537. [Google Scholar] [CrossRef]
38. Scaffaro R, Mario A, Sutera F, Gulino EF, Morreale M. Degradation and recycling of films based on biodegradable polymers: a short review. Polymers. 2019;11:651. doi:10.3390/polym11040651. [Google Scholar] [PubMed] [CrossRef]
39. Alex A, Llango NK, Ghosh P. Comparative role of chain scission and solvation in the biodegradation of polylactic acid (PLA). J Phys Chem B. 2018;122:9516–26. doi:10.1021/acs.jpcb.8b07930. [Google Scholar] [PubMed] [CrossRef]
40. Tham WL, Poh BT, Mohd Ishak ZA, Chow WS. Water absorption kinetics and hygrothermal aging of poly(lactic acid) containing halloysite nanoclay and meleated rubber. J Polym Environ. 2015;23:242–50. doi:10.1007/s10924-014-0699-y. [Google Scholar] [CrossRef]
41. Al-Itry R, Lamnawar K, Maazouz A. Improvement of thermal stability, rheological and mechanical properties of PLA, PBAT and their blends by reactive extrusion with functionalized epoxy. Polym Degrad Stab. 2012;97:1898–914. doi:10.1016/j.polymdegradstab.2012.06.028. [Google Scholar] [CrossRef]
42. Trofimchuk E, Ostrikova V, Ivanova O, Moskvina M, Plutalova A, Grokhovskaya T, et al. Degradation of structurally modified polylactide under the controlled composting of food waste. Polymers. 2023;15(19):4017. doi:10.3390/polym15194017. [Google Scholar] [PubMed] [CrossRef]
43. Teamsinsungvon A, Ruksakulpiwat Y, Jarukumjorn K. Preparation and characterization of poly(lactic acid)/poly(butylene adipate-co-terepthalate) blends and their composite. Polymer Plast Technol Eng. 2013;52:1362–7. doi:10.1080/03602559.2013.820746. [Google Scholar] [CrossRef]
44. Jang H, Kwon S, Kim SJ, Park S. Maleic anhydride-grafted PLA preparation and characteristics of compatibilized PLA/PBSeT blend films. Int J Molecul Sci. 2022;23(13):7166. doi:10.3390/ijms23137166. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF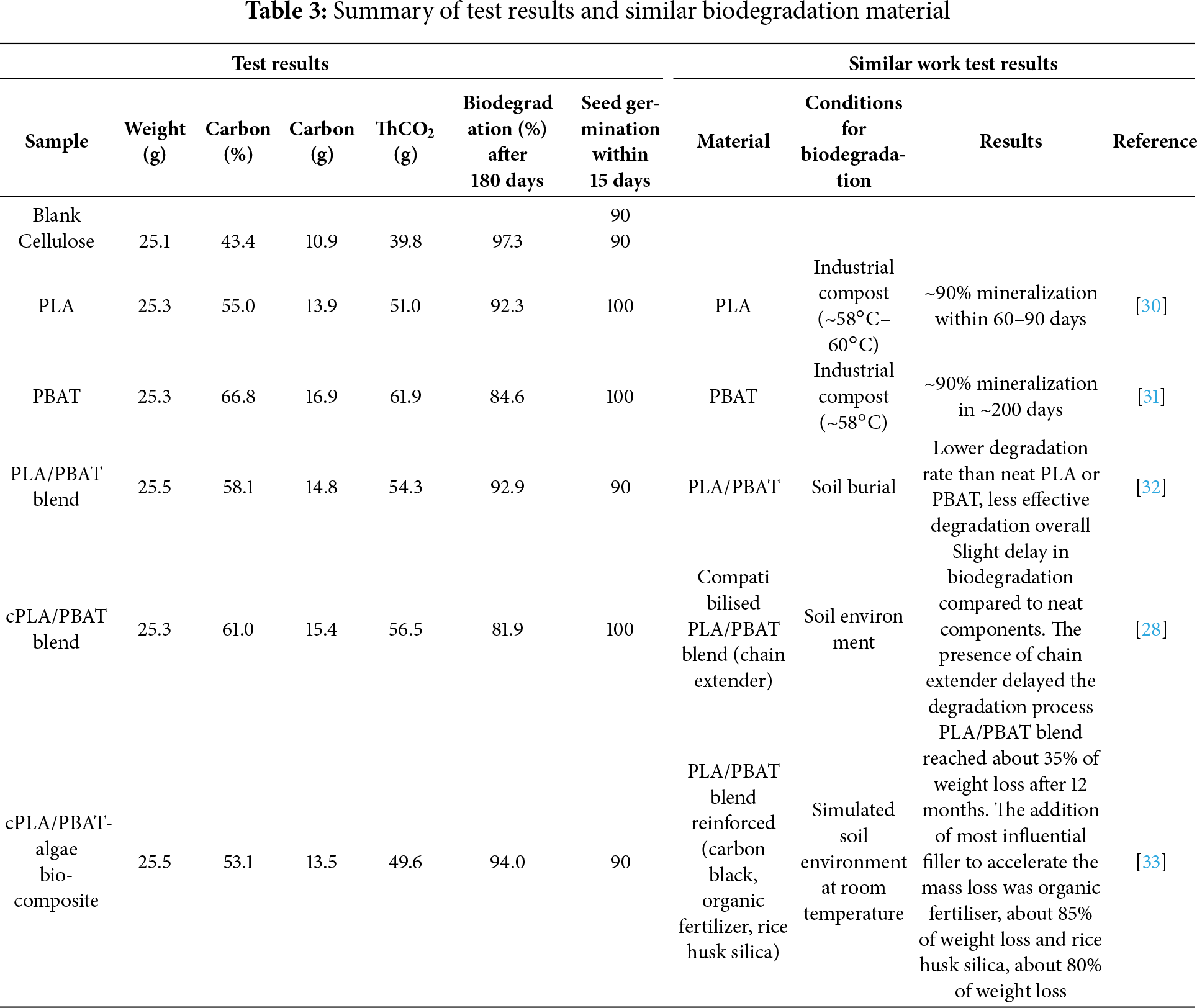
 Downloads
Downloads
 Citation Tools
Citation Tools