 Open Access
Open Access
REVIEW
Advances in the Construction and Application of Lignin-Based Hydrogels
College of Food Science and Technology, Henan Agricultural University, Zhengzhou, 450002, China
* Corresponding Author: Yanxia An. Email:
Journal of Renewable Materials 2026, 14(2), 2 https://doi.org/10.32604/jrm.2025.02025-0151
Received 29 July 2025; Accepted 04 September 2025; Issue published 25 February 2026
Abstract
Lignin, the most abundant natural aromatic polymer globally, has garnered considerable interest due to its rich and diverse active functional groups and its antioxidant, antimicrobial, and adhesive properties. Recent research has significantly improved the performance of lignin-based hydrogels, suggesting their substantial potential in fields such as biomedicine, environmental science, and agriculture. This paper reviews the process of lignin extraction, systematically introduces synthesis strategies for preparing lignin-based hydrogels, and discusses the current state of research on these hydrogels in biomedical and environmental protection fields. It concludes by identifying the existing challenges in lignin hydrogel research and envisioning future prospects and development trends.Graphic Abstract
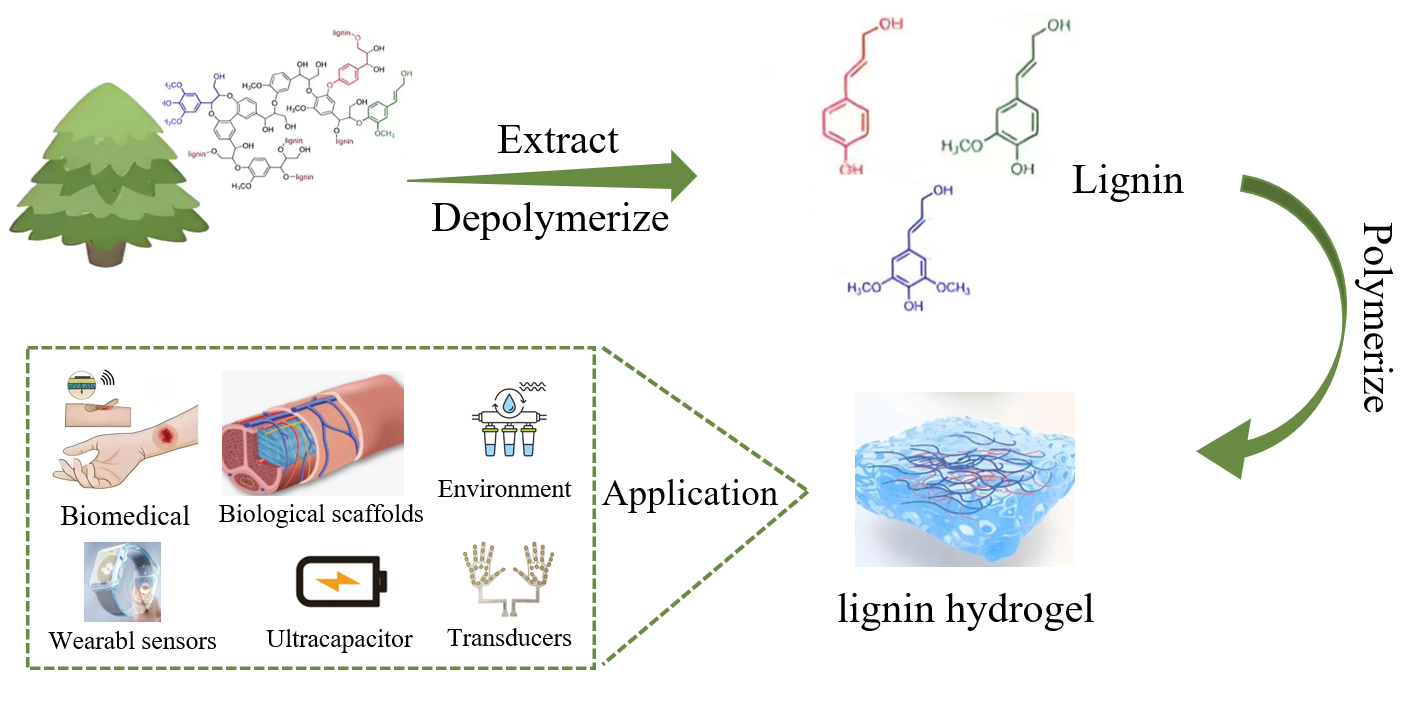
Keywords
Hydrogels are porous polymer materials prepared via physical or chemical crosslinking to form a three-dimensional network structure. Due to their good biocompatibility, non-toxicity, biodegradability, hydrophilicity, and chemical stability, hydrogel materials have been widely used in the fields of health care, agriculture, environment, electronics, and so on [1]. Based on the source of the raw materials used in hydrogel preparation, hydrogels are categorized as natural, synthetic, and hybrid hydrogels. Natural hydrogels are a class of gel materials derived from natural polymers such as agarose, chitosan, cellulose, alginate, hyaluronic acid, gelatin, collagen, and their derivatives [2]. The polymers used to prepare synthetic hydrogels mainly include polyethylene glycol, polyethylene, polyacrylic acid, and poly (methyl methacrylate). Structurally, hydrogels may exhibit crystalline, amorphous, or semi-amorphous structures, and they can also be classified by synthesis method, charge characteristics, or stimulus responsiveness, as shown in Fig. 1. Notably, hydrogels exhibit great versatility—their mechanical and chemical properties can be adjusted according to their application [3]. Traditional hydrogels usually lack mechanical strength and exhibit unstable swelling behavior, poor environmental adaptability, and limited functionality due to their high-water content and weak structural integrity. These constraints have restricted the performance of hydrogels in advanced applications. In recent years, lignin, as a green bio-based component, has been widely studied for the synthesis of hydrogels due to its natural molecular structure and physicochemical properties. The introduction of lignin endows hydrogels with antioxidant and bacteriostatic properties, hydrophobicity, enhanced biocompatibility, and various other functionalities.
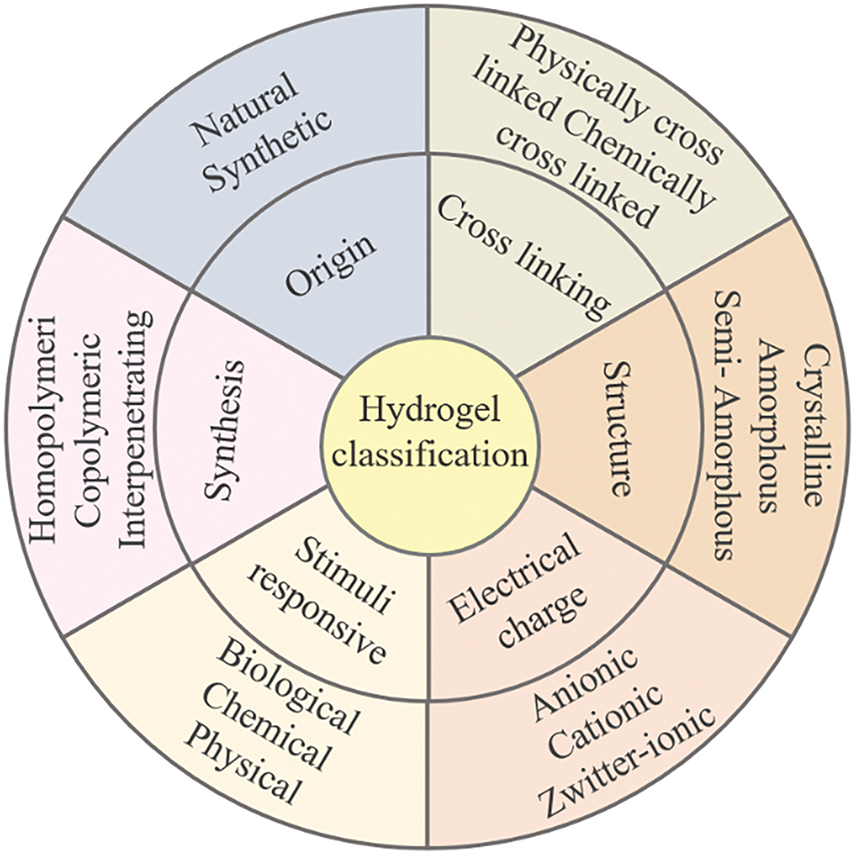
Figure 1: Classification of lignin hydrogels
Lignin, which is the second most abundant natural polymer on earth, is primarily extracted from wood by the pulp and paper industry. While the annual global output of lignin is about ~70 million tons, lignin utilization remains below 10% due to its chemical inertness and structural heterogeneity [4]. Fig. 2 illustrates the presence of lignin in woody plants, intricately intertwined and connected with cellulose and hemicellulose via hydrogen and covalent bonds. This network forms the cell wall’s backbone within a complex, dense structure. The illustration also depicts lignin’s molecular structure, predominantly comprising three types of phenylpropane units linked by ether and carbon-carbon bonds. Noteworthy are reactive groups such as aromatic groups, phenolic hydroxyls, alcoholic hydroxyls, and carbon-based co-choking double bonds within its molecular structure. These groups can participate in various reactions including hydroxymethylation, phenolization, sulfonation, alkylation, polycondensation, and graft copolymerization. In recent years, strategies for the synthesis of hydrogels using lignin have been extensively reported. Lignin can be physically and chemically modified to improve its structure and function, enhancing its usefulness for the preparation of hydrogels [5,6]. Compared with other renewable polymers, lignin possesses antioxidant and antibacterial characteristics, low degradability, and high mechanical strength. Consequently, lignin-based materials are widely utilized in fields such as advanced materials, energy, biomedicine, adsorbents, and environmental protection [7]. These characteristics also indicate the potential value of lignin in the preparation of hydrogels. Cheng et al. [8] successfully developed a lignin-containing cellulose nanofibril hydrogel for sustained levofloxacin release. Within 120 h, this hydrogel effectively released 95.5% of the levofloxacin, and the gel also demonstrated good toughness (2400 J/m2) and adhesion strength (114.6 kPa), providing a reference for the development of high-performance hydrogel biomaterials. Later, Díaz et al. [9] successfully synthesized alkali lignin (AL)-based biocomposite gel via free radical polymerization. This gel had an efficient and stable adsorption capacity for alizarin red, and the initial 81.1% adsorption performance was still maintained after seven cycles, providing a feasible and economically efficient solution for wastewater treatment.
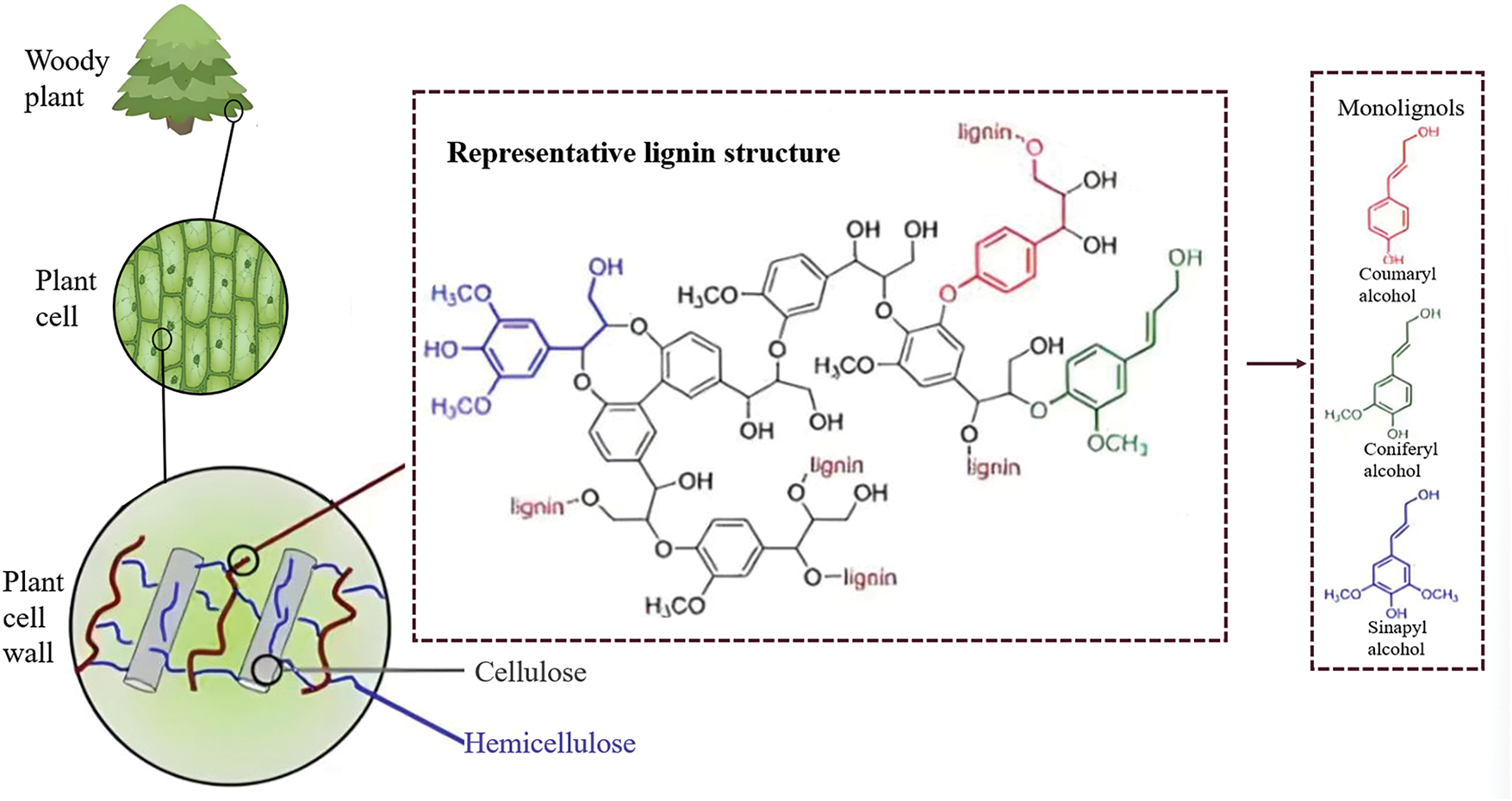
Figure 2: Composition and structure of lignin
2 Extraction and Isolation of Lignin
The structure and properties of lignin primarily depend on the plant source and the extraction method. Firstly, there is a variance in the lignin content across different plants. Timber plants (both coniferous and broadleaf) generally exhibit higher lignin content than herbaceous plants. This difference directly impacts extraction efficiency, with plants having a higher content being easier to extract. Secondly, there are structural differences. Coniferous lignin is mainly comprised of guaiacylpropane units, which have a relatively simple structure and are easy to extract. On the other hand, both broadleaf and herbaceous lignins contain lilac-based propane units, characterized by a complex structure, making them more challenging to extract. Herbaceous lignin also includes p-hydroxyphenyl propane units, which are closely tied with hemicellulose, adding to the difficulty of separation. More importantly, the extraction methods of lignin are mainly physical, chemical, and biological methods. Physical methods primarily involve techniques such as milling, microwave irradiation, extrusion, freezing, ultrasonication, and pyrolysis [10]. However, while these physical methods are operationally simple and avoid the generation of inhibitory molecules, they are often limited by low extraction efficiencies. As shown in Fig. 3, chemical methods typically employ solvents for the extraction of lignin, including acidic solvents (e.g., H2SO4, CH3COOH, H3PO4), alkaline solvents (e.g., NaOH, Ca (OH)2, KOH), and ionic liquids and deep eutectic solvents (DESs). However, these chemical methods are associated with drawbacks such as high costs, lignin structural degradation, and environmental concerns. The biological method employs microorganisms, predominantly enzymes, bacteria, and fungi, to degrade and extract lignin, subsequently converting it into usable monomers or small molecular compounds without generating inhibitors. This technique boasts significant advantages in terms of biodegradability and environmental friendliness. However, its limitations include a prolonged processing cycle and comparatively lower efficiency. Among these methods, chemical methods are the most prevalent. Therefore, the main chemical methods utilized for lignin extraction are introduced and analyzed below.
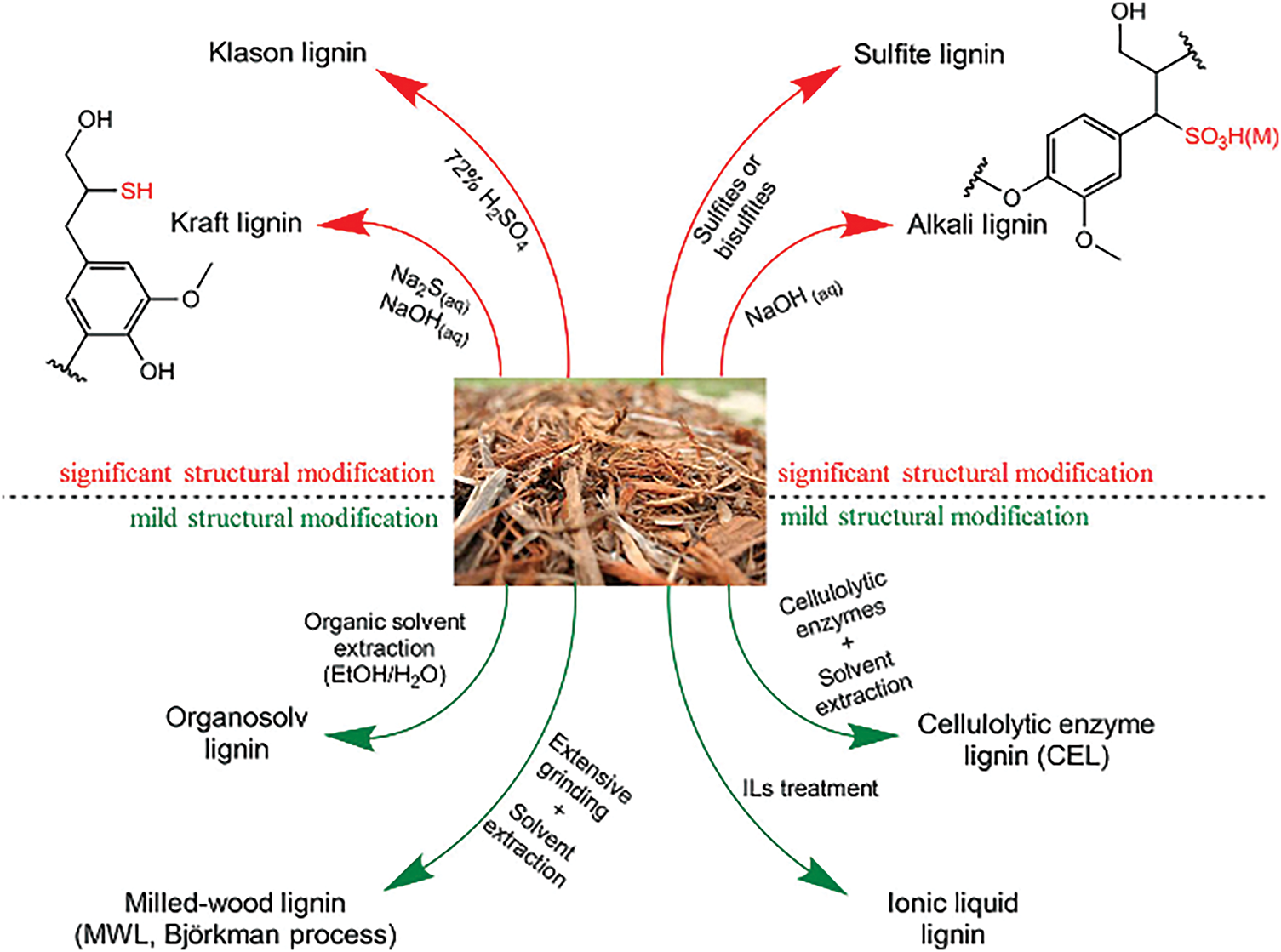
Figure 3: Chemical methods for separating lignin from lignocellulose [11]
Acid-based methods for the extraction of lignin are performed by hydrolyzing the cellulose into glucose using organic or inorganic acids, which enables the isolation of the lignin residue via filtration. These methods are commonly employed to isolate lignin from softwoods. Commonly used acids include organic acids (e.g., formic acid and acetic acid) and inorganic acids (e.g., nitric acid and sulfuric acid). Currently, organic acids are more widely utilized in biomass pretreatment due to their high effectiveness in decomposing lignocellulosic biomass, which minimizes the degradation of by-products and enhances the yield of lignin. Wang et al. [12] employed a formic acid/acetic acid solvent system to extract lignin with a purity of up to 79.83% from wheat straw. The extracted lignin retained its structural integrity and demonstrated favorable thermal stability. The method benefits from a mature process, straightforward operation, and mild reaction conditions. It also yields a high purity of lignin. Nevertheless, the extraction process has certain constraints and the corrosive nature of the acid can damage the lignin structure, as well as corrode equipment and contribute to environmental pollution.
Reagents commonly used in alkaline-based lignin extraction methods are summarized in Table 1. Crucial factors in alkaline-based lignin extraction are the solid-to-liquid ratio, temperature, pressure, and residence time. Among the known alkaline methods, NaOH treatment is particularly notable due to its strong alkalinity, low cost, and relatively straightforward solvent recovery. When combined with other solvents or techniques, NaOH-based treatment strategies can provide enhanced pretreatment efficiency. Wang et al. [13] employed bamboo biomass to evaluate the structural changes in lignin and lignin-carbohydrate complexes caused by alkaline treatment. After the alkaline treatment, the relative content of Klason lignin in their samples reached 84.67%, and the macromolecular lignin linked by β-O-4′ bonds showed minimal degradation. It should be noted that while alkaline extraction methods are highly prevalent, the lignin extracted using these techniques often exhibits lower purity, and these methods pose certain environmental concerns. The Alkaline method is the most frequently used industrial technique due to its high extraction efficiency and suitability for large-scale production. A notable disadvantage is that the extraction process generates a substantial amount of sulfur-containing or alkaline wastewater, which incurs high costs for recycling and treatment.

Organosolv methods utilize organic solvents for lignin extraction. Lignin exhibits good solubility in these solvents, which can effectively disrupt the chemical bonds between the lignin and hemicellulose. After its dissolution, the separated lignin is subsequently separated through processes such as extraction, precipitation, and distillation. Organosolv-based approaches typically require elevated temperatures and pressures, and they can be paired with alkaline or acidic catalysts to enhance the separation efficiency. Common organic solvents include methanol, ethanol, glycerol, γ-valerolactone, and ethylene glycol. Among these solvents, ethanol and methanol are particularly favored due to their low boiling points, cost-effectiveness, and ease of recovery. Agrawal et al. [14] extracted lignin from coconut husk using organic solvents and DES, reporting that the highest extraction efficiency of 71.03% was achieved using an ethanol solvent combined with FeCl3. The organic solvent method offers advantages such as high selectivity, non-toxicity, lack of sulfur contamination, and the ability to recycle the solvent. This method can preserve the natural structure of lignin, making it suitable for high-value applications. However, it presents certain safety hazards, including flammability and toxicity concerns.
Ionic liquids (ILs) are low-temperature molten salts composed of inorganic anions and organic cations. Notably, ILs are capable of efficiently dissolving lignin and carbohydrates. The cations in ILs typically feature an organic core (e.g., ammonium, pyridinium, phosphonium, sulfonium, or cholinium), while the anions can be organic or inorganic. By combining various cations and anions, ILs can be prepared to exhibit special physicochemical properties such as low vapor pressure, high thermal stability, exceptional dissolving power, non-flammability, and low toxicity. Crucially, ILs can be designed to selectively dissolve the components of biomass (cellulose, hemicellulose, and lignin), enhancing both the purity and extraction efficiency of lignin. Furthermore, ILs reduce the structural rigidity of the dissolved components, facilitating their subsequent processing and utilization. Hasanov et al. [15] investigated the effects of three protic ionic liquids (PILs)—pyrrolidinium acetate, pyridinium formate, and pyridinium acetate—on the extraction of lignin from ash wood. Their results indicated that pyridinium formate dissolved lignin closest to its native structure with higher purity, and the recovered pyridinium formate had a purity of more than 98%, suggesting good potential for sustainable reuse. However, despite their compelling properties and promising performance, the large-scale application of ILs is currently hindered by their high production costs and high viscosity.
DESs are an eco-friendly and cost-effective class of green solvents characterized by their low melting points. Typically, DESs are prepared via the combination of a hydrogen bond donor (HBD) and a hydrogen bond acceptor (HBA) at a specific molar ratio. DESs and ILs share certain fundamental characteristics, such as their high stability. However, DESs offer additional advantages, including enhanced environmental friendliness, lower cost, non-toxicity, and recyclability. Consequently, DESs are widely applied in biomass extraction applications. The DESs currently utilized for this purpose can be broadly categorized into four main types, as summarized in Table 2. Among these, Type III DESs (composed of choline chloride (ChCl) and an HBD) are the most prevalent due to their straightforward preparation, excellent dissolving power, high atom utilization efficiency, low production cost, and inherent biocompatibility. Long et al. [16] employed a DES system comprising choline chloride and maleic acid for the extraction of lignin from poplar wood, achieving a lignin yield of 86.5%. This extracted lignin was subsequently utilized to fabricate a biodegradable film that exhibited good sustained-release properties. DES amalgamate the benefits of organic solvents and IL, addressing the issues of high viscosity and toxicity associated with traditional ILs, while augmenting the extraction rate and purity of lignin. Furthermore, DES exerts a minimal impact on lignin’s structure, preserving a greater quantity of phenolic hydroxyls and methoxyls, thereby enhancing its antioxidant properties and broadening its application spectrum.
3 Construction of Lignin-Based Hydrogels
Lignin is endowed with an array of inherent attributes including antibacterial, antioxidant, biocompatibility, and chemical stability. Even minute quantities of lignin can manifest pronounced effects. However, its intricate structure, limited solubility, and inadequate reactivity often restrict its use to enhancing the performance of hydrogels. The preparation methods of lignin-based hydrogels mainly include physical crosslinking and chemical crosslinking, which are discussed in detail in this section.
Physical crosslinking methods typically utilize physical forces (e.g., hydrogen bonding, ionic interactions, hydrophobic interactions) to interconnect lignin and other polymer molecules, leading to the formation of a three-dimensional network structure. Water molecules are entrapped within the pores of this network, resulting in hydrogel formation [17].
Freeze-thaw methods are the most commonly employed physical crosslinking strategy for the synthesis of hydrogels. These methods offer various advantages, including operational simplicity, environmental friendliness, and good biocompatibility. In these processes, lignin is typically combined with a water-soluble polymer (such as polyvinyl alcohol (PVA) or cellulose) in water to form a homogeneous solution. Subsequently, this solution is subjected to low-temperature freezing, causing the water molecules to crystallize and form an ice crystal framework. Next, this frozen structure is thawed by increasing the temperature, causing the ice crystals to melt. During this process, the water molecules are redistributed, leading to the formation of a stable gel network. This freeze-thaw cycle can be repeated multiple times to enhance the crosslinking density and mechanical properties of the hydrogel. In an investigation by Bethel et al. [18], three freeze-thaw cycles were performed on a lignin-PVA solution. This process facilitated the crystallization and subsequent re-crystallization of the solvent, bolstering the interactions between lignin and PVA molecules. This fostered molecular chain rearrangement and aggregation, culminating in a hydrogel material with a lignin concentration reaching 60 wt% and a consistent network structure. The technique promotes the self-assembly of molecular chains into cross-linked networks via successive freezing and thawing cycles, specifically at −20°C for freezing and approximately 20–25°C for thawing. This approach offers a robust control over the porosity and pore size distribution of hydrogels, enhancing their physicochemical attributes [19]. Nevertheless, it necessitates lignin of a comparably elevated molecular weight, and the resultant lignin-based hydrogel exhibits modest mechanical strength with suboptimal long-term stability.
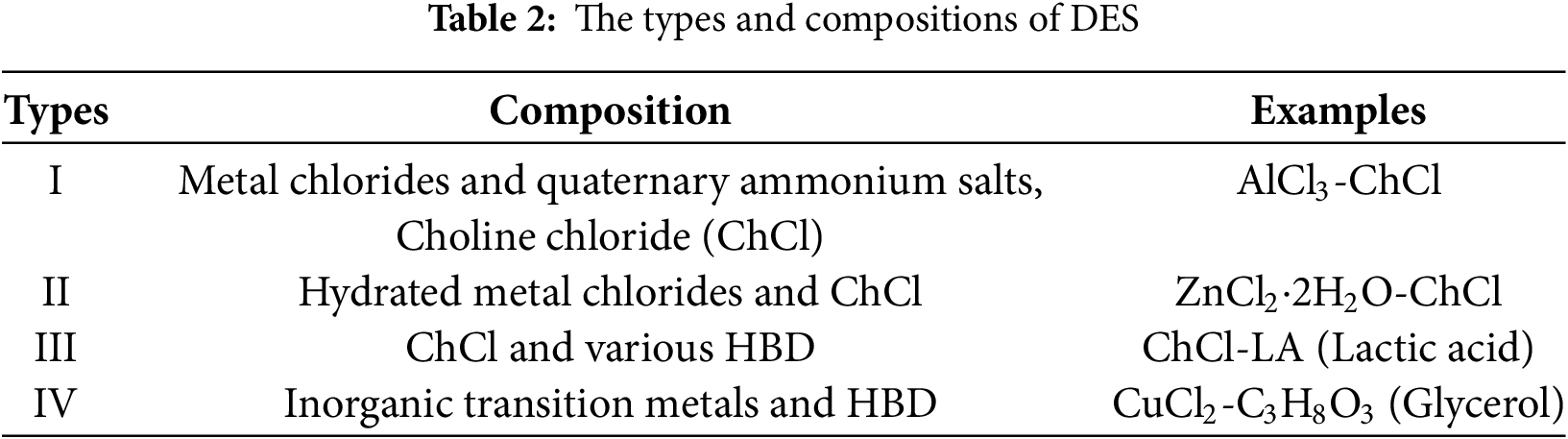
Ionic interactions represent a crucial approach for establishing physical crosslinks within lignin. Utilizing these interactions can significantly enhance the crosslinking between polymer chains, enabling the formation of a stable network structure [20]. Ionic interactions refer to the dynamic attraction between positively and negatively charged groups within the polymer chains of a hydrogel network. Lignin molecules contain abundant active functional groups such as phenolic hydroxyl (–OH), carboxyl (–COOH), and methoxy (–OCH3) groups. In aqueous solutions, these groups can dissociate into negatively charged phenolate (–O−) and carboxylate (–COO−) ions. These negative charges then electrostatically interact with the cations in the system (e.g., Na+, K+, Ca2+, Fe3+), forming stable ionic bonds or coordination bonds [21]. Zhang et al. [22] utilized ionic interactions to fabricate a lignin-chitosan-PVA composite hydrogel. As shown in Fig. 4, the sulfonate groups in the chemical structure of this lignin formed ionic bonds with the amino groups of chitosan, while the hydroxyl groups of the lignin formed hydrogen bonds with the hydroxyl groups of PVA. This hydrogel exhibited good biocompatibility, antibacterial activity, and high mechanical strength. Notably, the hydroxyl groups within lignin molecules can form hydrogen bonds with water molecules or other polar molecules, creating an additional hydrogen bonding network that further reinforces the crosslinking density of hydrogels. This synergy between ionic interaction and hydrogen bonding can enhance the mechanical strength of hydrogels while also improving their adsorption capacity for heavy metalions [23].
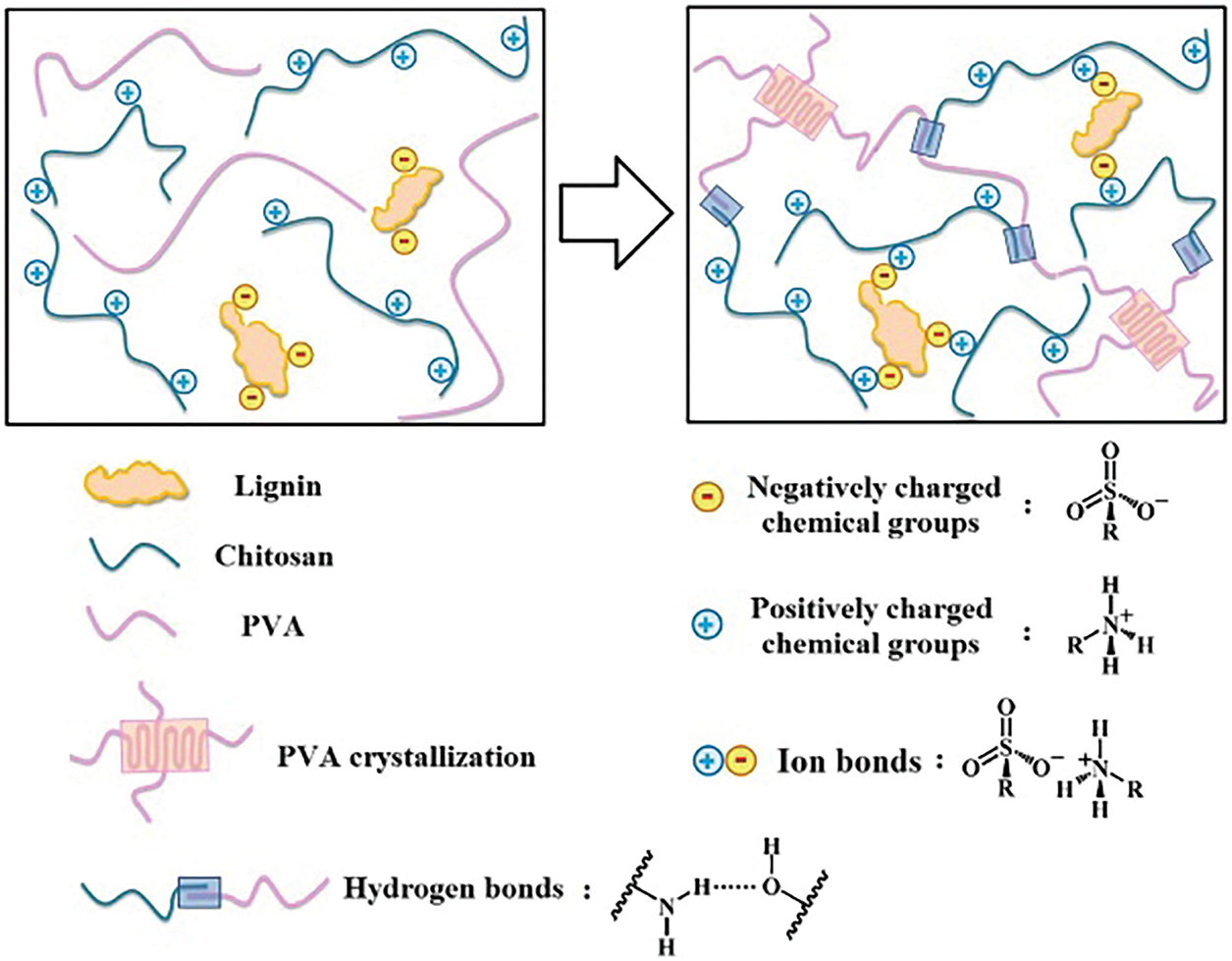
Figure 4: Cross-linking mechanism of lignin-chitosan-PVA hydrogels [22]
3.1.3 Hydrophobic Interactions
Hydrophobic moieties or side chains can act as crosslinking points within hydrogel networks [24]. Consequently, hydrophobic interactions can be utilized to enhance hydrogel structures. In a hydrophilic environment, the extensive benzene ring structure of lignin is easily squeezed together by water molecules to form crosslinking points [25]. You et al. [26] proposed a method involving a solvent exchange strategy to induce multiscale hydrophobic interactions, enabling the preparation of high-strength and high-toughness lignin hydrogels. First, the hydrophilic polymer poly(N,N-dimethylacrylamide) (PDMA), which contains hydrophobic side groups, was combined with lignin. Then, a simple “dissolution-drying-swelling” solvent exchange (DDSSE) process was utilized to generate multiscale hydrophobic interactions and obtain ultra-tough lignin-based hydrogels, as displayed in Fig. 5. Jiang et al. [27] found that incorporating 1-butanol-soluble lignin enhanced the thermal stability of hydrogels and lowered the critical solution temperature. The method is based on non-covalent interactions between polymer chains, exhibiting a strong self-assembly capability. The resultant lignin hydrogels possess a dense network structure and excellent flexibility. However, their stability can be influenced by environmental factors.
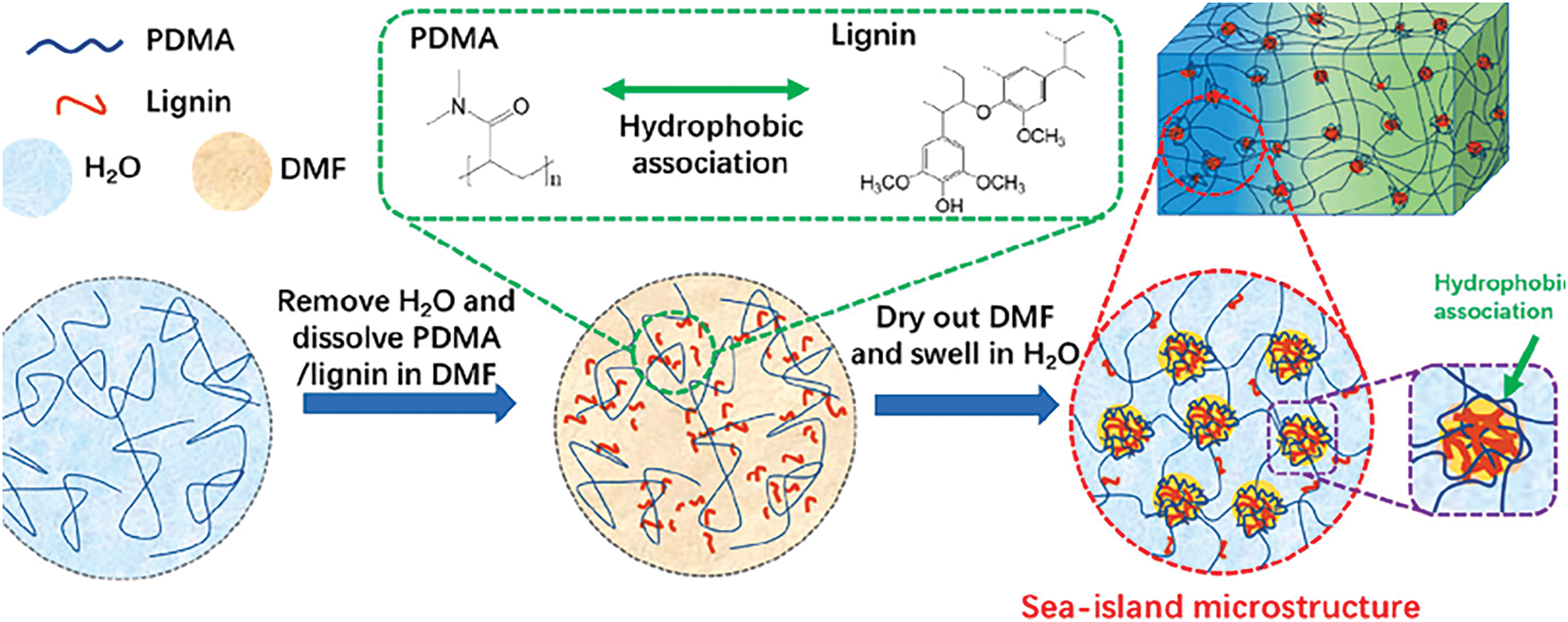
Figure 5: Schematic diagram of lignin hydrogel formation (DDSSE process) [26] (Reprinted with permission from [26] © 2020, American Chemical Society)
Physically crosslinked hydrogels offer advantages such as environmental friendliness, low cost, and mild reaction conditions. However, these hydrogels are susceptible to external factors such as temperature and pH, often resulting in poor long-term stability. The stability of physically crosslinked hydrogels can be improved by tuning the crosslinking density of the hydrogen bonds. For example, Zha et al. [28] utilized a solvent exchange-triggered hydrogen bond activation strategy to prepare highly robust and tough adaptive organohydrogels. An additional strategy for enhancing the network structure of these hydrogels is the introduction of reinforcing phases such as nanoparticles, nanofibers, or rigid polymer chain segments. For instance, Sun et al. [29] exploited the reconfigurable interactions between rigid aromatic nylon nanofibers and flexible PVA to construct a highly oriented composite hydrogel network, mimicking the interactions between collagen fibers and soft proteoglycans at the microscopic level. Improved hydrogels can also be prepared with special microstructures, such as porous structures, interpenetrating network structures, or dual network structures. Alternatively, the stability and mechanical strength of hydrogels can be enhanced by mimicking the structure of natural biological materials, such as the laminar or fibrous structures of shells and bones.
Chemical crosslinking methods such as free radical polymerization, graft copolymerization, and the implementation of interpenetrating networks (IPN) can be utilized for the preparation of lignin-based hydrogels. Under the action of crosslinkers and initiators, lignin forms highly stable covalent bonds with other polymer molecules. This process is irreversible and allows for functional modifications. Common crosslinkers include epichlorohydrin (ECH), polyethylene glycol diglycidyl ether (PEGDGE), and N,N’-methylenebisacrylamide (MBA). These crosslinking agents typically exhibit epoxy groups or double bond structures, enabling them to undergo epoxidation or esterification reactions with the phenolic hydroxyl and alcoholic hydroxyl groups in lignin to form stable covalent bonds [23].
3.2.1 Free Radical Polymerization
Chain-growth polymerization reactions initiated by radicals are known as free radical polymerization. The core mechanism of these reactions involves the decomposition of an initiator to generate primary radicals, which subsequently initiate the chain propagation of monomers. Ammonium persulfate (APS) is commonly employed as an initiator. Under heating, APS decomposes into sulfate radicals (SO4−·), which react with the hydroxyl groups (−OH) in lignin to generate macroradicals. These macroradicals then react with a crosslinker (e.g., MBA) to form a three-dimensional network structure [30]. Furthermore, the functional groups within lignin, such as phenolic hydroxyl and carboxyl groups, can also serve as active sites for chain propagation, enabling the participation of the lignin in the polymerization reaction. Consequently, lignin-based hydrogels prepared via free radical polymerization can exhibit enhanced crosslinking density and improved mechanical properties. Zerpa et al. [31] fabricated kraft lignin hydrogels via free radical polymerization using hardwood kraft lignin and N-isopropylacrylamide as monomers and MBA as the crosslinker. In the procedure, 0.06 g of MBA crosslinking agent and 0.08 g of azobisisobutyronitrile initiator were employed. Dinari et al. [32] synthesized lignin-based nanogel via atom transfer radical polymerization, as shown in Fig. 6. This gel had an average diameter of 150 nm, exhibited dual responsiveness to both temperature and pH, and showed average curcumin loading and encapsulation efficiencies of 49.69% and 92.62%, respectively. This approach offers simplicity in operation, mild reaction conditions, and a broad spectrum of applications. Moreover, the pore size and mechanical attributes of the gel can be finely tuned by modulating the ratio of the initiator and cross-linking agent. A notable limitation is the potential presence of unreacted monomers, which might impact biocompatibility.
Figure 6: Preparation of lignin-based nanogel [32]
Graft copolymerization strategies involve the use of chemical reactions to attach other monomers onto the lignin molecular chain to form graft copolymers, which are then reacted with crosslinkers to produce functional hydrogels [33]. Wei et al. [34] synthesized a biomass trimeric lignin-based hydrogel (LAD) by grafting acrylamide (AM) and acryloyloxyethyl trimethylammonium chloride onto sodium lignosulfonate (LS), as shown in Fig. 7. LAD showed acceptable adsorption properties (maximum adsorption capacity of up to 409.84 mg/g) and conformed to the Langmuir isothermal adsorption model. Rajan et al. [35] grafted 2-hydroxyethyl methacrylate (HEMA) onto lignin via esterification, followed by free radical polymerization with excess HEMA. This strategy yielded a copolymer hydrogel with a high lignin content of 40%, and the prepared hydrogel had an exceptional water retention capacity and good thermal stability. Hydrogels can be multifunctionalized by introducing specific functional groups, such as drug molecules and bioactive molecules, through grafting reactions. However, to enhance the grafting rate, intricate pre-treatment conditions are necessary, such as sulfonation and oxidation. It is crucial to regulate the grafting density to prevent excessive cross-linking which could result in reduced solubility.
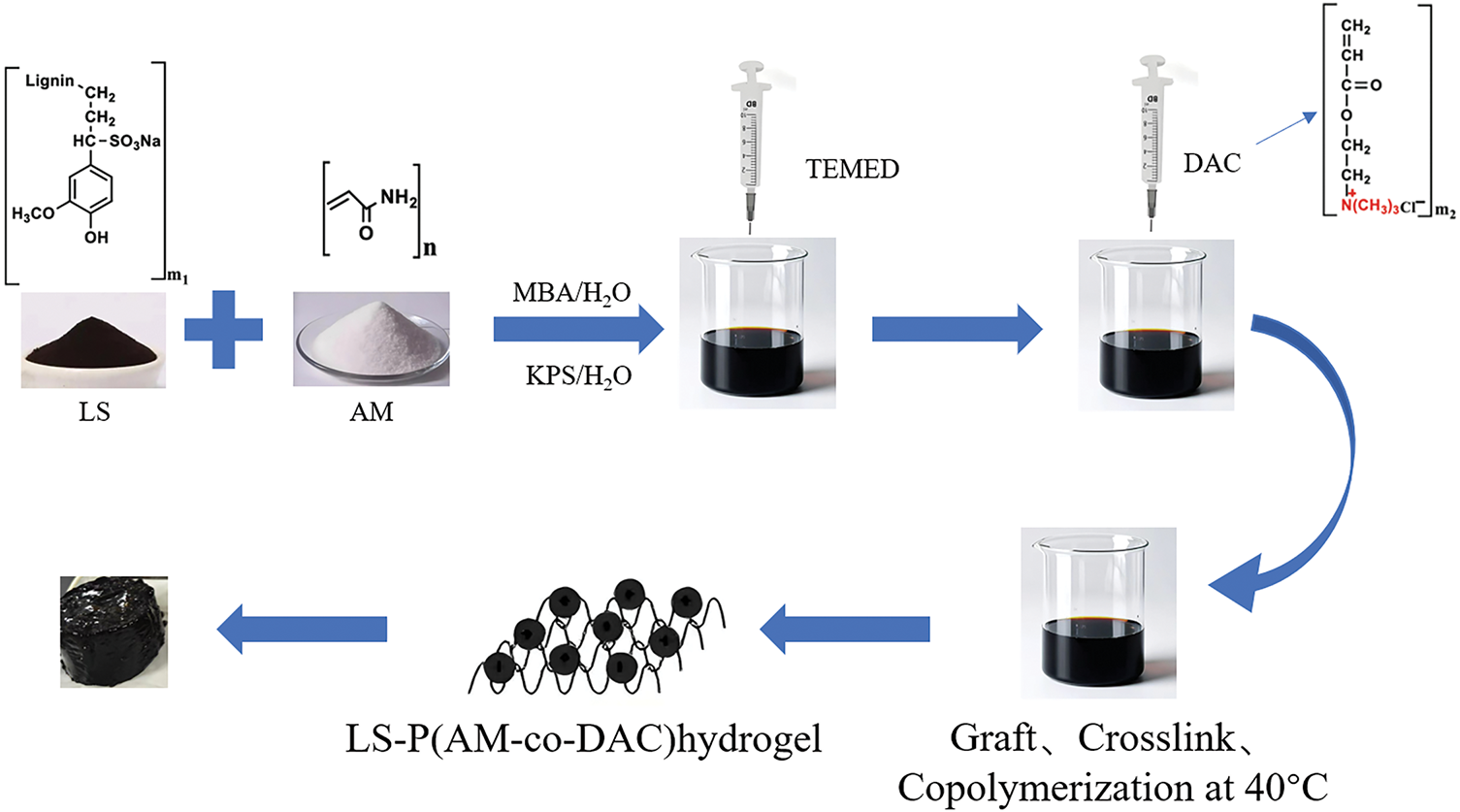
Figure 7: Schematic of the synthesis of LAD hydrogel
3.2.3 Interpenetrating Networks
The interpenetrating network structure method refers to a three-dimensional lattice comprising two or more intertwined polymer chains. These chains, which are intertwined on a molecular scale, do not share covalent bonds and can only be separated through chemical bond breakage. This results in a significant enhancement of the mechanical strength and toughness of lignin gels. Chen et al. [36] successfully prepared aerogels with three-dimensional interpenetrating networks using a wheat straw base as the raw material, PVA as the reinforcing agent, and methyltrimethoxysilane as the hydrophobic modifier. Later, Zhao et al. [37] successfully fabricated a semi-interpenetrating network consisting of LS, carboxymethylcellulose (CMC), and polyacrylamide (PAM) chains as well as a variety of interactions. As shown in Fig. 8, this strategy resulted in the preparation of multifunctional PC4L hydrogels with tensile properties and self-repairing properties. The interpenetration of networks method bolsters the overall attributes of lignin hydrogels by amalgamating the benefits of diverse polymers to achieve complementary properties. The hydrogel network created via this approach exhibits high density and robust mechanical properties. However, the preparation process is intricate and necessitates meticulous control over reaction conditions.
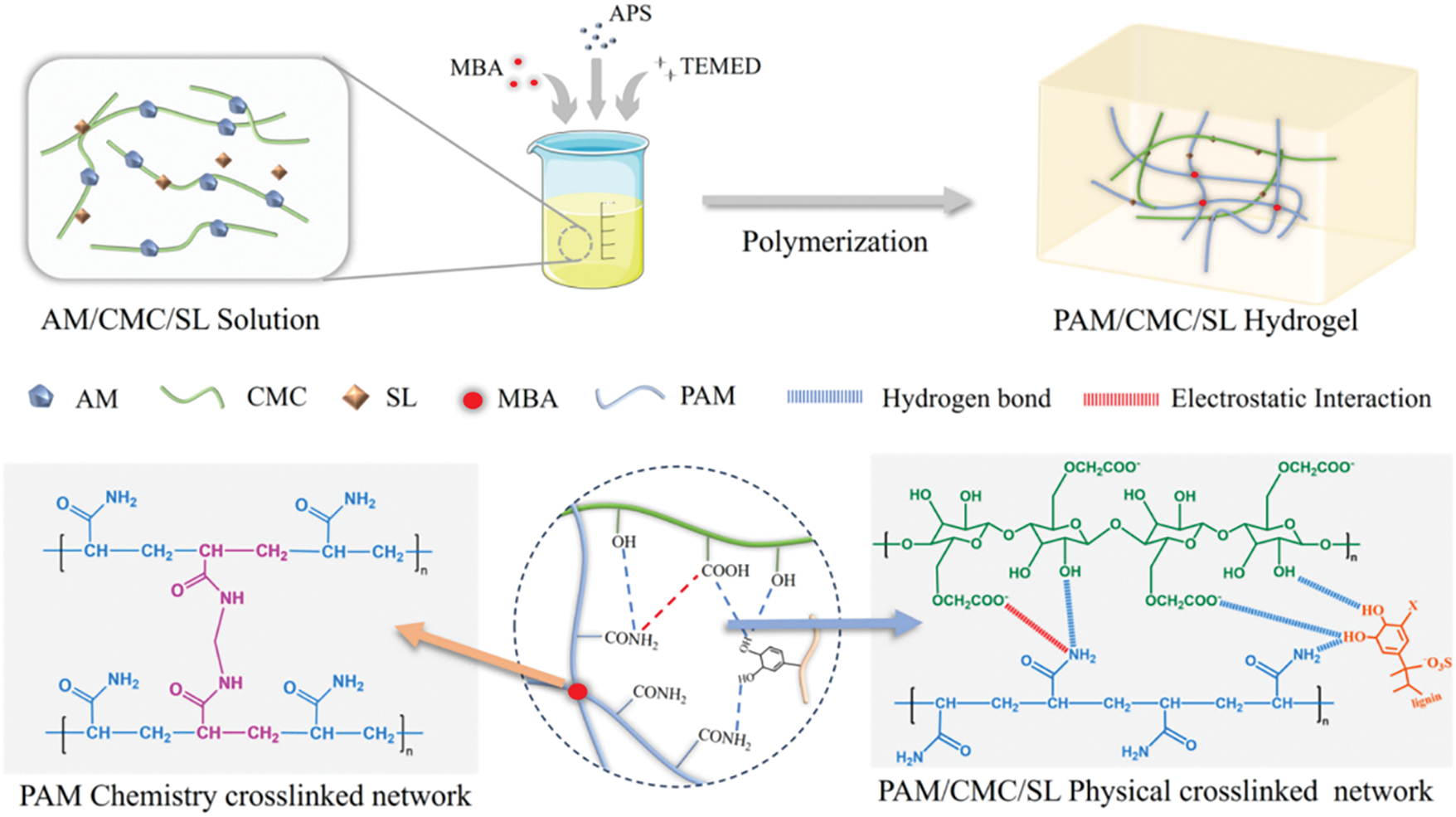
Figure 8: Network structure of PC4L hydrogel [37] (Reprinted with permission from reference [37] © 2025 Elsevier B.V)
In contrast, lignin hydrogels prepared by chemical crosslinking require shorter gelation times and exhibit higher crosslinking density, superior mechanical strength, and better structural stability. However, these gels typically require the introduction of crosslinkers, some of which might pose potential toxicity concerns. Furthermore, residual crosslinkers must be removed after hydrogel preparation, which could increase costs and impact the environmental friendliness of hydrogel production.
4 Properties of Lignin-Based Hydrogels
The properties of lignin-based hydrogels are influenced by a variety of factors. Lignin molecules, rich in active functional groups such as aliphatic hydroxyl, phenolic hydroxyl, and carboxyl groups, can form a porous network structure via methods like graft co-polymerization. This directly impacts the hydrogels’ mechanical strength and water retention characteristics. The cross-linking method is vital to the mechanical attributes of these hydrogels. For example, the dual network structure created through solvent exchange techniques allows for the simultaneous achievement of high modulus and high toughness. This overcomes the conventional limitation where toughness is inversely proportional to stiffness in traditional hydrogels. Additionally, reaction conditions—temperature, reaction time, and the quantity of crosslinking agents—can influence the stability of hydrogels. The integration of composite components such as alkali metal ions can produce a self-catalyzing system that provides rapid gelation and anti-freezing properties to the hydrogel. Conversely, the addition of graphene oxide significantly enhances the adsorption performance. Consequently, by manipulating these influencing factors, lignin-based hydrogels can exhibit a combination of desirable properties: remarkable mechanical performance, water absorption and retention capabilities, antibacterial properties, antioxidant activity, and biodegradability.
The mechanical properties of lignin hydrogels encompass strength, toughness, modulus, and fatigue resistance. Hydrogel strength is directly influenced by the chemical structure and content of the lignin. Lignin molecules have aromatic ring structures, which provide a rigid framework. Moreover, the phenolic hydroxyl and carboxyl groups of lignin act as active crosslinking sites, bonding with other polymers to form a strong crosslinked network. Therefore, increasing the lignin content of hydrogels enhances properties such as strength, toughness, and storage modulus. Ravishankar and Dhamodharan [20] prepared supramolecular hydrogels with adjustable LS content. These hydrogels exhibited a tensile strength of about 20 MPa, a Young’s modulus of about 14 MPa, and a toughness of about 50 MJ m−3, exceeding the performance of natural cartilage. Optimizing the crosslinking within lignin-based hydrogels can further improve their mechanical properties. Gu et al. [38] utilized a multi-solvent high-temperature annealing process to construct a lignin-based supramolecular hydrogel, which demonstrated acceptable self-healing and fatigue resistance. Notably, this hydrogel exhibited structural integrity even after enduring severe compression (up to 0.5 strain) for 500,000 cycles. The mechanical properties of lignin hydrogels are influenced by multiple factors, including the molecular structure and content of lignin, crosslinking method, crosslinker type, and solvent. These factors act through different mechanisms to determine the final mechanical properties of lignin-based hydrogels.
4.2 Water Absorption and Retention
The water absorption and retention capabilities of lignin-based hydrogels are primarily influenced by the pore structure within the hydrogel network encompassing surface morphology, pore size, pore volume ratio, and functional groups [39]. A suitable pore structure provides hydrogels with an enhanced surface area and a greater number of adsorption sites, facilitating the easier absorption and retention of water molecules. Xia et al. [40] employed an interpenetrating polymer network method to prepare a lignin hydrogel with a pore structure that exhibited hydrophilicity at 20°C and hydrophobicity at 45°C. Consequently, this hydrogel showed a significant absorption capacity for oil/water mixtures. Sun et al. [41] used lignin and polyacrylic acid to prepare composite porous hydrogel, which exhibited superior water transport properties compared to pure polyacrylic acid hydrogels. It should be noted that hydrogel networks can be affected by factors such as charge repulsion, excessively high ionic strength, and pH variations. These factors can change the swelling ratio of lignin hydrogel systems [39]. For example, changing the pH alters the protonation state of functional groups (e.g., carboxyl or phenolic hydroxyl groups) within lignin-based hydrogels, causing the network structure to expand or contract. Therefore, the water absorption and retention capabilities of lignin hydrogels are not solely determined by their pore structure and functional group distribution. Instead, they are significantly influenced by external environmental conditions, such as pH levels and ionic strength.
4.3 Antibacterial and Antioxidant Properties
Lignin-based hydrogels possess inherent antibacterial and antioxidant properties, which are primarily attributed to the abundant phenolic structures and functional groups (e.g., hydroxyl, methoxy groups) of the lignin. The phenolic compounds in lignin can disrupt the integrity of bacterial cell membranes, interfere with their metabolic processes, and induce the generation of reactive oxygen species (ROS), leading to oxidative stress and the inhibition of microbial growth [42]. Yun et al. [43] studied a series of lignin materials, reporting that the lignin sample with the lowest molecular weight and highest phenolic hydroxyl content (L3) exhibited significant inhibitory effects against bacteria such as E. coli, Salmonella, Streptococcus, and S. aureus. Notably, the antibacterial activity of this lignin was positively correlated with lignin concentration. In addition to providing an antibacterial effect, the phenolic hydroxyl groups in lignin can scavenge free radicals through electron transfer or hydrogen bonding, leading to remarkable antioxidant capacity. You et al. [26] prepared a lignin/PDMA hydrogel capable of effectively scavenging 1,1-diphenyl-2-picrylhydrazyl (DPPH) radicals. This hydrogel scavenged 80% of the DPPH radicals within 4 min, and a removal rate as high as 93.4% was achieved within 10 min. The antibacterial and antioxidant properties of lignin-based hydrogels can be improved via the inclusion of metal nanoparticles, which can synergistically interact with the lignin. Gan et al. [44] designed a dynamic redox system based on silver-lignin nanoparticles, which endowed their prepared hydrogel with potent antibacterial and antioxidant capabilities suitable for skin wound repair. Lignin can also be compounded with other antibacterial agents (e.g., chitosan, polyurethane, zinc oxide) or antioxidants (e.g., vitamin C) to further enhance its antibacterial and antioxidant properties.
Compared to traditional petroleum-based hydrogels, lignin-based hydrogels offer distinct environmental advantages, including good biodegradability, low toxicity, and renewability. The biodegradation of lignin-based hydrogels is an aerobic metabolism involving ligninolytic enzymes secreted by microorganisms, and at the end of the degradation process, carbon dioxide and water are generated as products. The molecular structure and composition of lignin influence its degradability. Typically, lignin materials with a high molecular weight, high crosslinking density, complex aromatic ring structures, or numerous crosslinked bonds exhibit poorer degradability [45]. However, degradability can be enhanced by the use of chemical modification to introduce hydrophilic groups or physical modification to reduce the molecular weight of lignin. The biodegradability of lignin confers significant advantages across multiple domains. Lignin-based hydrogels can be gradually degraded by enzymes (e.g., laccase, xylanase), enabling their application in controlled drug delivery, tissue engineering, and wound healing [46]. Moreover, the microorganisms in soil can biodegrade these hydrogels, leading to their ultimate transformation into inorganic matter and small-molecule aromatic compounds [47]. Moreover, in wastewater treatment applications, hydrogels can be naturally degraded after the adsorption of pollutants, avoiding the generation of secondary pollution. The biodegradability of lignin hydrogels provides them with broad application potential in fields such as biomedicine, agriculture, and environmental remediation, as shown in Table 3.
5 Application Advances of Lignin-Based Hydrogels
Lignin hydrogel, a composite material derived from the natural polymer lignin, serves multiple functions and has gained significant attention in various research domains [48]. In the biomedical arena, lignin hydrogels have potential applications in controlled drug delivery, tissue engineering, and wound healing, as illustrated in Fig. 9. Within the environmental sector, these hydrogels are effective for water treatment, notably for adsorbing heavy metal ions, dyes, and organic pollutants. They also contribute to soil enhancement and plant growth stimulation. Moreover, lignin hydrogels are apt for flexible sensing and other applications, including capacitors.
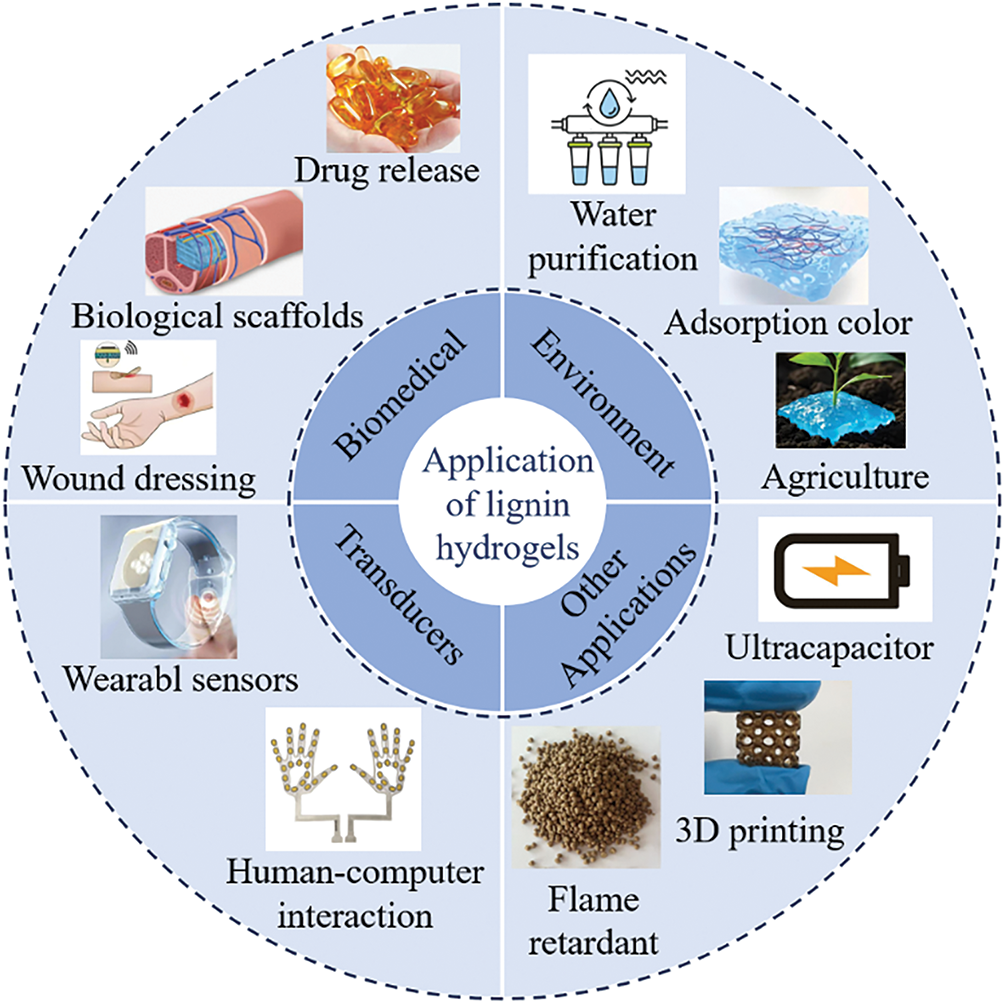
Figure 9: Application of lignin hydrogels
The incorporation of therapeutics within the structure of lignin hydrogels through chemical bonding, physical adsorption, or microencapsulation could enable their utilization as drug carriers. This would enable targeted drug delivery and controlled release, demonstrating their excellent potential for localized or site-specific administration. Thus, therapeutic efficacy could be enhanced while minimizing side effects. Shan et al. [49] designed a composite hydrogel with antibacterial and antioxidant activities based on dynamic covalent bonds between sodium alginate, lignin, and epigallocatechin-3-gallate (EGCG). Fig. 10a illustrates that the hydrogel concurrently exhibited self-healing capabilities, pH-responsiveness, and robust antimicrobial and antioxidant properties. When applied to the skin surface, it significantly mitigated the risk of wound infections and impeded scar formation. Li et al. [50] engineered a multifunctional, bio-based hydrogel suitable for drug delivery. Fig. 10b demonstrates how this hydrogel facilitated pH-responsive drug release through hydrogen bonding and π-π stacking interactions between lignin and the drug. Moreover, this hydrogel exhibited antibacterial activity and promoted wound healing. The inherent antibacterial, antioxidant, pro-angiogenic, and biocompatible properties of lignin hydrogels make them ideal wound dressings. For example, Crivello et al. [51] developed a supramolecular (SM) hydrogel containing embedded cobalt-lignin nanoparticles (LNP) for the treatment of chronic wounds. The optimized SM gel absorbed proteins, alleviated inflammation, and exerted antibacterial effects. Lignin hydrogels can also be utilized as tissue engineering scaffolds to promote cell proliferation and tissue regeneration. Pan et al. [52] confirmed that supramolecular hydrogels constructed via lignin-induced self-assembly possess commendable mechanical properties, and the three-dimensional porous network structure of their hydrogels provided an ideal scaffold for cell growth. Gu et al. [38] used a multi-solvent high-temperature annealing strategy to develop lignin-based hydrogel with excellent mechanical properties. This hydrogel exhibited a high modulus of 74.4 MPa, a toughness of 90 MJ/m3, and a tear strength of 34,000 J/m2—performance metrics approaching those of human cartilage tissue. Therefore, this hydrogel demonstrated significant potential for joint repair applications. Overall, lignin hydrogels are natural biomaterials with broad application prospects, and significant research progress has been made in the application of these hydrogels within the field of biomedicine. Future research should prioritize structural optimization, functional enhancement, and rigorous safety evaluation for clinical applications.
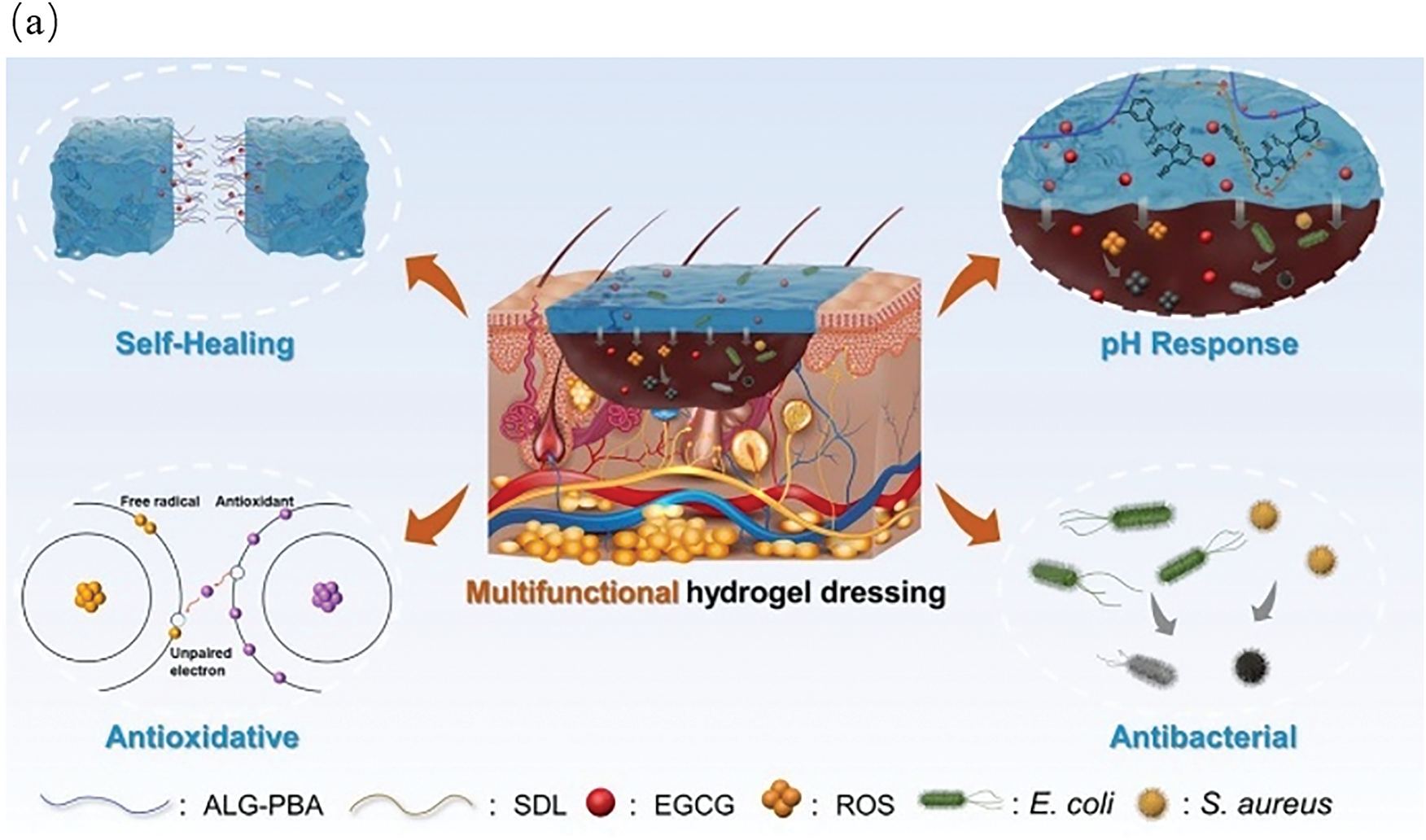
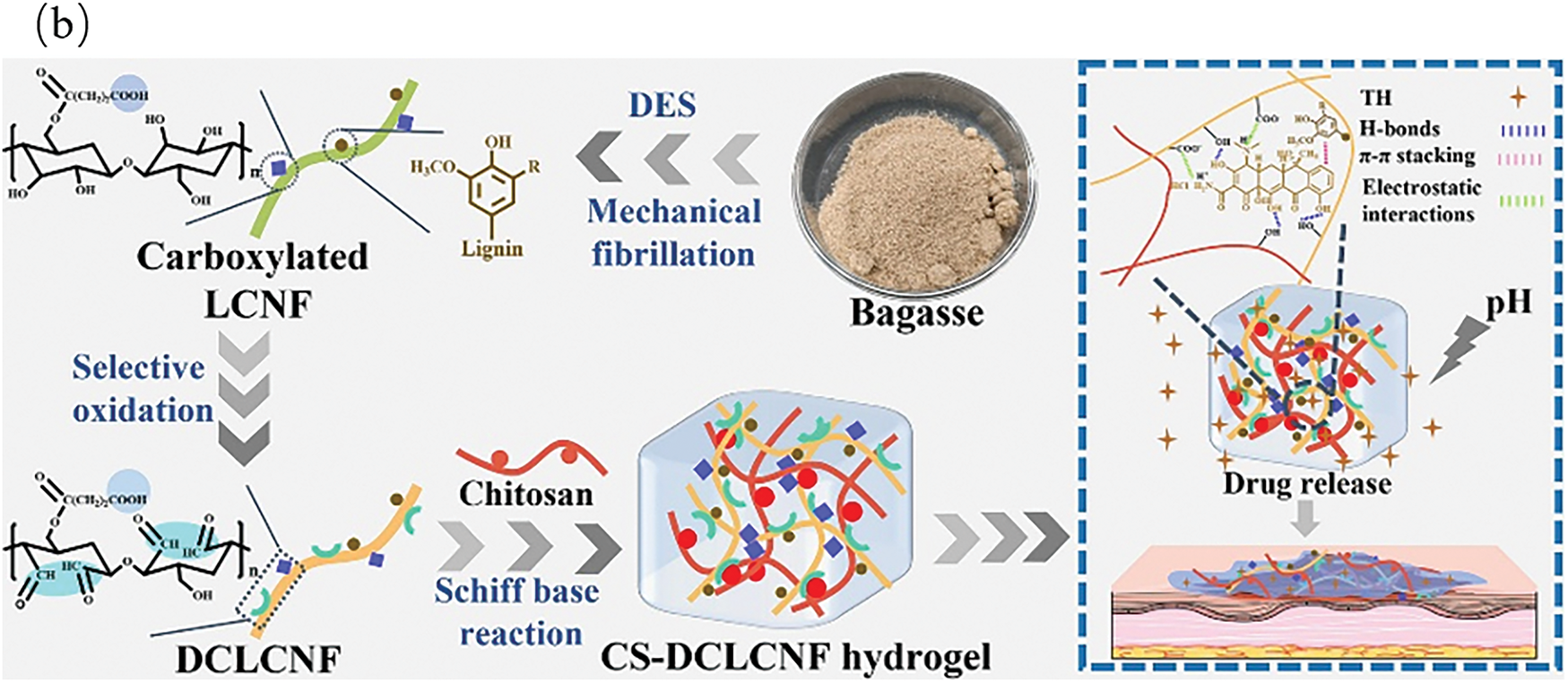
Figure 10: (a) Lignin-based hydrogels in wound healing [49], (b) Multifunctional lignocellulosic nanofiber hydrogels [50] (Reprinted with permission from reference [49] and reference [50] ©2024 Elsevier B.V.)
The functional groups of lignin-based hydrogels (e.g., aliphatic hydroxyl, phenolic hydroxyl, and carboxyl groups) can form coordination bonds with metal ions or adsorb contaminants via hydrogen bonding and electrostatic interactions. Consequently, these hydrogels exhibit good adsorption capacities for pollutants, enabling the effective removal of heavy metal ions and organic dyes from wastewater. Lignin-based hydrogels can form complexes with heavy metal ions (e.g., Pb2+, Hg2+, Cd2+) in soil and wastewater, facilitating the remediation or purification of heavy metal-contaminated environments. Zhao et al. [53] prepared chitosan/lignin hydrogel (CSL) via a simple one-pot synthesis strategy involving Mannich reaction. This gel exhibited ultrafast adsorption rates for Pb2+ and Cu2+, reaching equilibrium in just 1–2 min. Notably, maximum Pb2+ and Cu2+ adsorption capacities of 139.86 mg/g and 98.71 mg/g were respectively achieved. Compared to other reported lignin-based adsorbents, CSL offers the advantages of simple preparation, high adsorption capacity, and rapid equilibrium time. In addition to metal pollutants, lignin hydrogels can be utilized for the adsorption of organic pollutants and dyes due to the large specific surface areas provided by their pore structures. Bashir et al. [54] investigated the utilization of chitosan-lignin hydrogel beads for the adsorption of Direct Blue-218 dye in aqueous solutions. These beads were capable of removing 96% of the Direct Blue-218 within 120 min, offering a cost-effective solution for dye wastewater treatment. Looking beyond water treatment applications, the use of lignin-based hydrogels in soil remediation shows strong potential. Relevant studies have indicated that lignin-based hydrogels can improve the structure, enhance the water retention capacity, and promote the microbial activity and nutrient cycling behavior of soil. Zhang et al. [55] used an inert Ca2+ crosslinking technique to successfully prepare a sodium alginate/LS hydrogel. This hydrogel effectively improved the water holding capacity of soil, increased the stem height and leaf length of crops grown in the soil, and exerted a sustained positive effect on crop growth and development. Therefore, this hydrogel is a promising soil amendment to combat soil degradation. Due to their satisfactory adsorption performance, biocompatibility, and renewability, lignin-based hydrogels hold broad application prospects in environmental protection and are poised to play a greater role in wastewater treatment and related fields.
The utilization of molecular structure design and innovative preparation processes has resulted in the successful preparation of lignin-based hydrogels that simultaneously exhibit high sensitivity, wide strain ranges, and environmental stability, providing ideal solutions for wearable devices and human-machine interaction systems. Yang et al. [56] fabricated a multifunctional green lignin-based hydrogel using LNPs as the raw material and DES/water as a binary solvent. This hydrogel was then utilized for human motion tracking, health monitoring, micro-expression recognition, and speech recognition. Zhao et al. [57] employed a dynamic redox reaction between lignin-coated silica nanoparticles (LSNs) and Fe3+ to prepare a highly elastic conducting hydrogel. This hydrogel was capable of effective operation under harsh conditions, particularly in high-altitude regions with thin ozone layers. As shown in Fig. 11, this hydrogel was successfully employed as a flexible sensor to monitor human motion in various modes. Recently, Wang et al. [58] developed a flexible hydrogel based on silk fibroin and LNPs. This hydrogel exhibited high stress sensitivity, a fast response speed, and cycling stability (≥8000 cycles), showing excellent potential for pressure sensor applications. The natural performance advantages of lignin hydrogels are driving the development of next-generation smart sensor technologies. As hydrogel preparation processes become more mature and costs are optimized, lignin hydrogel sensors are expected to be successfully utilized in large-scale applications within the next five years. This is expected to lead to breakthrough progress, particularly in medical monitoring, motion tracking, and smart robotics applications.
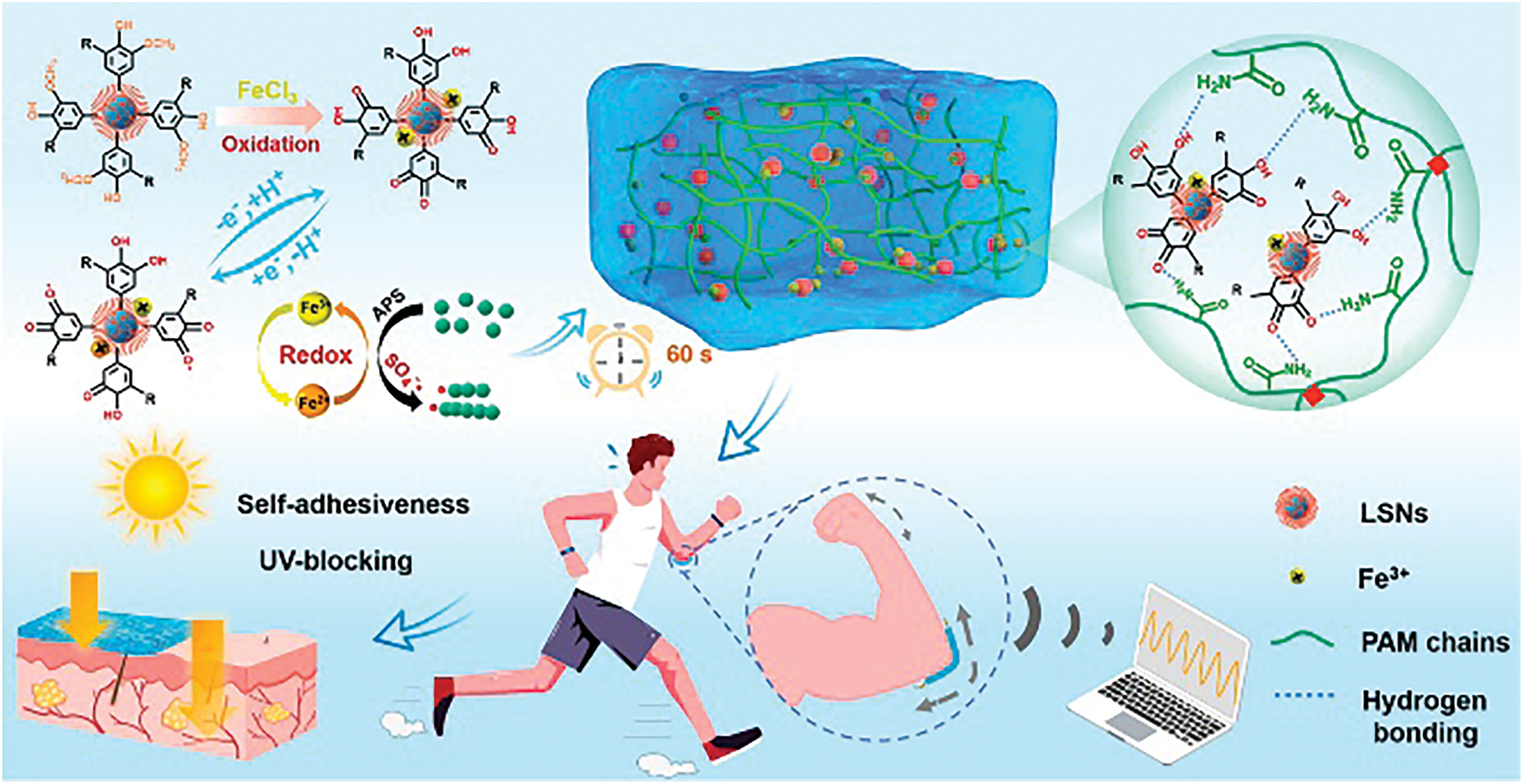
Figure 11: Design of LSN-Fe3+/PAM hydrogel [57] (Reprinted with permission from reference [57] © 2022 American Chemical Society)
Other applications for lignin hydrogels include capacitors, flame retardants, energy conversion, and 3D printing (Table 3). Lignin-based conductive hydrogels have notable ionic conductivity and mechanical properties, enabling their widespread use as electrolytes and electrode materials in the field of capacitors. As shown in Fig. 12, Chen et al. [59] calcined lignocellulose-based hydrogel precursors consisting of alginate (AL), CMC, and KOH under nitrogen to obtain porous carbon materials with tailored pore structures and graphitization levels. Their optimal porous carbon (LHPC) exhibited surprising performance in a zinc-ion hybrid supercapacitor, maintaining 91.90% capacitance after 60,000 cycles. Lignin hydrogels can be designed to exhibit satisfactory fatigue and frost resistance in addition to good flame-retardant properties, making them suitable for flame retardant applications in high- or low-temperature environments. Li et al. [60] successfully developed lignin-reinforced flame-retardant composites by combining lignin as a mechanical reinforcing agent and flame-retardant material with dimethyl methylphosphonate and expanded graphite. This material exhibited flame retardancy, thermal insulation, and mechanical properties. Mai et al. [61] prepared conductive magnetic lignin/graphene oxide aerogel composites for use as energy conversion materials. Due to their good thermal storage capacity and ability to efficiently convert electrical, magnetic, and solar energy, these aerogel composites show extraordinary potential for multi-source-driven thermal conversion. Lignin hydrogels exhibit good rheology and processability, enabling their potential use in the field of 3D printing. These hydrogels could be used as reinforcing agents to improve the comprehensive performance of 3D printed composite products [62]. In summary, lignin hydrogels demonstrate outstanding performance and application potential across multiple domains, providing new directions for the development of sustainable materials.
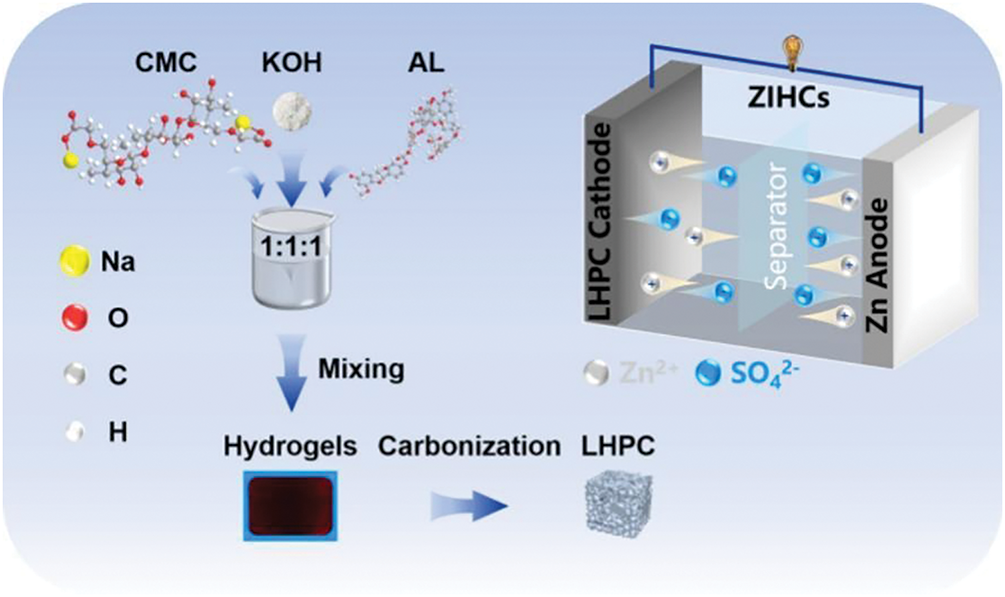
Figure 12: LHPC in zinc-ion hybrid supercapacitors [59] (Reprinted with permission from reference [59] © 2024 Elsevier Ltd.)
Lignin-based hydrogels, as green functional materials, have witnessed considerable progress in their construction strategies and application sectors in recent years. However, several critical challenges remain for their practical utilization. Firstly, the absence of standardized testing criteria and evaluation systems hinders direct comparison and validation of results across various studies. Secondly, the paucity of comprehensive long-term biocompatibility studies casts doubt on the safety of lignin-based hydrogels during extended use. Furthermore, the consistent synthesis of lignin hydrogels poses a significant challenge due to the difficulty in precisely controlling reaction conditions and parameters, resulting in substantial variations in hydrogel properties from one batch to another.
In the future, it is imperative to establish a unified standard for performance evaluation, encompassing standardized testing methodologies for various indicators such as mechanical performance, biocompatibility, drug-release rate, and degradation rate. This will significantly enhance the repeatability and comparability of research findings. It is also crucial to intensify research on long-term biocompatibility to evaluate the degradation behavior, tissue compatibility, and immune response of lignin-based hydrogels in vivo, thereby ensuring their safety for biomedical applications. Moreover, the exploration of more stable and controllable synthetic methods is essential to augment the reproducibility and stability of lignin hydrogels. Such advancements will facilitate their broader application in practice.
Acknowledgement: This study was financially supported by the National Natural Science Foundation of China (21706052, 22278114) and Natural Science Foundation of Henan Province (242300421575).
Funding Statement: Financial support from the National Natural Science Foundation of China (21706052, 22278114) and Natural Science Foundation of Henan Province (242300421575) are gratefully acknowledged.
Author Contributions: Writing—original draft, Visualization, Investigation, Formal analysis, Conceptualization, Jiazi Wang; Writing—review & editing, Supervision, Funding acquisition, Conceptualization, Yanxia An; Investigation, Jingyuan Su and Keke Liu; Supervision, Jian Zhang and Yang Zhao; Investigation, Writing—review & editing, Linlin Li. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Data will be made available on request.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
1. El Sayed MM. Production of polymer hydrogel composites and their applications. J Polym Environ. 2023;31(7):2855–79. doi:10.1007/s10924-023-02796-z. [Google Scholar] [CrossRef]
2. Passauer L, Hallas T, Bäucker E, Ciesielski G, Lebioda S, Hamer U. Biodegradation of hydrogels from oxyethylated lignins in model soils. ACS Sustain Chem Eng. 2015;3(9):1955–64. doi:10.1021/acssuschemeng.5b00139. [Google Scholar] [CrossRef]
3. Zhang A, Liu Y, Qin D, Sun M, Wang T, Chen X. Research status of self-healing hydrogel for wound management: a review. Int J Biol Macromol. 2020;164(1):2108–23. doi:10.1016/j.ijbiomac.2020.08.109. [Google Scholar] [PubMed] [CrossRef]
4. Haq I, Mazumder P, Kalamdhad AS. Recent advances in removal of lignin from paper industry wastewater and its industrial applications—a review. Bioresour Technol. 2020;312(1):123636. doi:10.1016/j.biortech.2020.123636. [Google Scholar] [PubMed] [CrossRef]
5. Zhao L, Ouyang X, Ma G, Qian Y, Qiu X, Ruan T. Improving antioxidant activity of lignin by hydrogenolysis. Ind Crops Prod. 2018;125:228–35. doi:10.1016/j.indcrop.2018.09.002. [Google Scholar] [CrossRef]
6. Xu J, Li C, Dai L, Xu C, Zhong Y, Yu F, et al. Biomass fractionation and lignin fractionation towards lignin valorization. ChemSusChem. 2020;13(17):4284–95. doi:10.1002/cssc.202001491. [Google Scholar] [PubMed] [CrossRef]
7. Collins MN, Nechifor M, Tanasă F, Zănoagă M, McLoughlin A, Stróżyk Mł A, et al. Valorization of lignin in polymer and composite systems for advanced engineering applications—a review. Int J Biol Macromol. 2019;131(1):828–49. doi:10.1016/j.ijbiomac.2019.03.069. [Google Scholar] [PubMed] [CrossRef]
8. Cheng G, Zeng F, Liu X, Yang Q, Wei S, Huang Q. Mussel-inspired adhesive and tough hydrogel for drug release based on lignin-containing cellulose nanofiber. Int J Biol Macromol. 2025;306(Pt 2):141458. doi:10.1016/j.ijbiomac.2025.141458. [Google Scholar] [PubMed] [CrossRef]
9. Díaz J, Roa K, Boulett A, Azócar L, Sánchez J. Reusable aminated lignin-based hydrogel biocomposite for effective dye adsorption in wastewater. Int J Biol Macromol. 2025;304(Pt 2):140842. doi:10.1016/j.ijbiomac.2025.140842. [Google Scholar] [PubMed] [CrossRef]
10. Soltanian S, Aghbashlo M, Almasi F, Hosseinzadeh-Bandbafha H, Nizami AS, Ok YS, et al. A critical review of the effects of pretreatment methods on the exergetic aspects of lignocellulosic biofuels. Energy Convers Manag. 2020;212(7):112792. doi:10.1016/j.enconman.2020.112792. [Google Scholar] [CrossRef]
11. Sun Z, Fridrich B, de Santi A, Elangovan S, Barta K. Bright side of lignin depolymerization: toward new platform chemicals. Chem Rev. 2018;118(2):614–78. doi:10.1021/acs.chemrev.7b00588. [Google Scholar] [PubMed] [CrossRef]
12. Wang Y, Sun XF, Chen J, Hu S, Sun R. Efficient extraction and analysis of wheat straw lignin by response surface methodology. Polymers. 2024;16(20):2935. doi:10.3390/polym16202935. [Google Scholar] [PubMed] [CrossRef]
13. Wang X, Pu J, Xu C, Yao S, Liu Y, Qin C, et al. Effects of alkali-soluble hemicellulose separation on the structure of bamboo lignin and lignin-carbohydrate complexes. Ind Crops Prod. 2024;222:119549. doi:10.1016/j.indcrop.2024.119549. [Google Scholar] [CrossRef]
14. Agrawal D, Tripathi A, Pal P, Hoque M, Bharathi SD, Jacob S. Sustainable extraction strategy for lignin from coconut coir using organosolv and deep eutectic solvents (DES). Waste Biomass Valorizat. 2025;16(4):1599–608. doi:10.1007/s12649-024-02758-z. [Google Scholar] [CrossRef]
15. Hasanov I, Shanmugam S, Kikas T. Extraction and isolation of lignin from ash tree (Fraxinus exselsior) with protic ionic liquids (PILs). Chemosphere. 2022;290:133297. doi:10.1016/j.chemosphere.2021.133297. [Google Scholar] [PubMed] [CrossRef]
16. Long X, Luo Y, Luo Z, Wu Y, Liu B, Qin C, et al. Deep eutectic solvent-mediated extraction of lignin: a novel strategy for producing high-quality biopolymers in controlled-release mulching applications. Int J Biol Macromol. 2025;300(4):140254. doi:10.1016/j.ijbiomac.2025.140254. [Google Scholar] [PubMed] [CrossRef]
17. Thakur VK, Thakur MK. Recent advances in green hydrogels from lignin: a review. Int J Biol Macromol. 2015;72:834–47. doi:10.1016/j.ijbiomac.2014.09.044. [Google Scholar] [PubMed] [CrossRef]
18. Bethel K, Buck A, Tindall G, Thies MC, Davis EM. Fabrication of physically crosslinked lignin-PVA hydrogels containing high concentrations of fractionated and cleaned lignins. MRS Commun. 2022;12(5):624–31. doi:10.1557/s43579-022-00219-z. [Google Scholar] [CrossRef]
19. Abdullah T, İlyasoğlu G, Memić A. Designing lignin-based biomaterials as carriers of bioactive molecules. Pharmaceutics. 2023;15(4):1114. doi:10.3390/pharmaceutics15041114. [Google Scholar] [PubMed] [CrossRef]
20. Ravishankar K, Dhamodharan R. Advances in chitosan-based hydrogels: evolution from covalently crosslinked systems to ionotropically crosslinked superabsorbents. React Funct Polym. 2020;149:104517. doi:10.1016/j.reactfunctpolym.2020.104517. [Google Scholar] [CrossRef]
21. Evstigneyev EI, Grinenko EV, Mazur AS, Vasilyev AV. Study of the formation of lignin hydrogels with metal cations. J Wood Chem Technol. 2021;41(2–3):73–82. doi:10.1080/02773813.2021.1873389. [Google Scholar] [CrossRef]
22. Zhang Y, Jiang M, Zhang Y, Cao Q, Wang X, Han Y, et al. Novel lignin-chitosan-PVA composite hydrogel for wound dressing. Mater Sci Eng C Mater Biol Appl. 2019;104:110002. doi:10.1016/j.msec.2019.110002. [Google Scholar] [PubMed] [CrossRef]
23. Rico-García D, Ruiz-Rubio L, Pérez-Alvarez L, Hernández-Olmos SL, Guerrero-Ramírez GL, Vilas-Vilela JL. Lignin-based hydrogels: synthesis and applications. Polymers. 2020;12(1):81. doi:10.3390/polym12010081. [Google Scholar] [PubMed] [CrossRef]
24. Zhang Z, Fu H, Li Z, Huang J, Xu Z, Lai Y, et al. Hydrogel materials for sustainable water resources harvesting & treatment: synthesis, mechanism and applications. Chem Eng J. 2022;439:135756. doi:10.1016/j.cej.2022.135756. [Google Scholar] [CrossRef]
25. Dai L, Zhu W, Lu J, Kong F, Si C, Ni Y. A lignin-containing cellulose hydrogel for lignin fractionation. Green Chem. 2019;21(19):5222–30. doi:10.1039/C9GC01975H. [Google Scholar] [CrossRef]
26. You X, Wang X, Zhang HJ, Cui K, Zhang A, Wang L, et al. Supertough lignin hydrogels with multienergy dissipative structures and ultrahigh antioxidative activities. ACS Appl Mater Interfaces. 2020;12(35):39892–901. doi:10.1021/acsami.0c10657. [Google Scholar] [PubMed] [CrossRef]
27. Jiang P, Cheng Y, Yu S, Lu J, Wang H. Study on the effect of 1-butanol soluble lignin on temperature-sensitive gel. Polymers. 2018;10(10):1109. doi:10.3390/polym10101109. [Google Scholar] [PubMed] [CrossRef]
28. Zha XJ, Zhang B, Cheng ZC, Zhang S, Pu JH, Huang JG, et al. Solvent-exchange triggered hydrogen bond activation strategy toward self-adaptive strong and tough organohydrogel artificial muscle. Chem Eng J. 2023;476(38):146548. doi:10.1016/j.cej.2023.146548. [Google Scholar] [CrossRef]
29. Sun M, Li H, Hou Y, Huang N, Xia X, Zhu H, et al. Multifunctional tendon-mimetic hydrogels. Sci Adv. 2023;9(7):eade6973. doi:10.1126/sciadv.ade6973. [Google Scholar] [PubMed] [CrossRef]
30. Rashidzadeh A, Olad A, Salari D, Reyhanitabar A. On the preparation and swelling properties of hydrogel nanocomposite based on Sodium alginate-g-Poly (acrylic acid-co-acrylamide)/Clinoptilolite and its application as slow release fertilizer. J Polym Res. 2014;21(2):344. doi:10.1007/s10965-013-0344-9. [Google Scholar] [CrossRef]
31. Zerpa A, Pakzad L, Fatehi P. Hardwood kraft lignin-based hydrogels: production and performance. ACS Omega. 2018;3(7):8233–42. doi:10.1021/acsomega.8b01176. [Google Scholar] [PubMed] [CrossRef]
32. Dinari A, Abdollahi M, Sadeghizadeh M. Design and fabrication of dual responsive lignin-based nanogel via grafting from atom transfer radical polymerization for curcumin loading and release. Sci Rep. 2021;11:1962. doi:10.1038/s41598-021-81393-3. [Google Scholar] [PubMed] [CrossRef]
33. Kaur R, Sharma R, Chahal GK. Synthesis of lignin-based hydrogels and their applications in agriculture: a review. Chem Pap. 2021;75(9):4465–78. doi:10.1007/s11696-021-01712-w. [Google Scholar] [CrossRef]
34. Wei S, Chen W, Tong Z, Jiang N, Zhu M. Synthesis of a functional biomass lignin-based hydrogel with high swelling and adsorption capability towards Acid Red 73. Environ Sci Pollut Res. 2021;28(37):51306–20. doi:10.1007/s11356-021-14324-4. [Google Scholar] [PubMed] [CrossRef]
35. Rajan K, Mann JK, English E, Harper DP, Carrier DJ, Rials TG, et al. Sustainable hydrogels based on lignin-methacrylate copolymers with enhanced water retention and tunable material properties. Biomacromolecules. 2018;19(7):2665–72. doi:10.1021/acs.biomac.8b00282. [Google Scholar] [PubMed] [CrossRef]
36. Chen X, Yang M, Cai X, An L, Lai K, Dou Y. Fabrication of wheat straw-based lignin containing nanofibril aerogels as recyclable absorbents for oil-water separation. Cellulose. 2024;31(1):497–514. doi:10.1007/s10570-023-05636-4. [Google Scholar] [CrossRef]
37. Zhao T, Xin Q, Yang G, Liang S, Lin J. Utilizing carboxymethyl cellulose sodium/sodium lignosulfonate hydrogel to build efficient electrolyte/electrode interfaces for multifunctional high-performance supercapacitors. Int J Biol Macromol. 2025;314(7):144273. doi:10.1016/j.ijbiomac.2025.144273. [Google Scholar] [PubMed] [CrossRef]
38. Gu Y, Xu C, Wang Y, Luo J, Shi D, Wu W, et al. Compressible, anti-fatigue, extreme environment adaptable, and biocompatible supramolecular organohydrogel enabled by lignosulfonate triggered noncovalent network. Nat Commun. 2025;16(1):160. doi:10.1038/s41467-024-55530-1. [Google Scholar] [PubMed] [CrossRef]
39. Meng Y, Lu J, Cheng Y, Li Q, Wang H. Lignin-based hydrogels: a review of preparation, properties, and application. Int J Biol Macromol. 2019;135:1006–19. doi:10.1016/j.ijbiomac.2019.05.198. [Google Scholar] [PubMed] [CrossRef]
40. Xia J, Liu Z, Chen Y, Wang Z, Cao Y. Fabrication of thermo-sensitive lignocellulose hydrogels with switchable hydrophilicity and hydrophobicity through an SIPN strategy. RSC Adv. 2019;9(51):29600–8. doi:10.1039/c9ra05575d. [Google Scholar] [PubMed] [CrossRef]
41. Sun Y, Ma Y, Fang G, Ren S, Fu Y. Controlled pesticide release from porous composite hydrogels based on lignin and polyacrylic acid. BioResources. 2015;11(1):2361–71. doi:10.15376/biores.11.1.2361-2371. [Google Scholar] [CrossRef]
42. Fazeli M, Mukherjee S, Baniasadi H, Abidnejad R, Mujtaba M, Lipponen J, et al. Lignin beyond the status quo: recent and emerging composite applications. Green Chem. 2023;26(2):593–630. doi:10.1039/d3gc03154c. [Google Scholar] [PubMed] [CrossRef]
43. Yun J, Wei L, Li W, Gong D, Qin H, Feng X, et al. Isolating high antimicrobial ability lignin from bamboo kraft lignin by organosolv fractionation. Front Bioeng Biotechnol. 2021;9:683796. doi:10.3389/fbioe.2021.683796. [Google Scholar] [PubMed] [CrossRef]
44. Gan D, Xing W, Jiang L, Fang J, Zhao C, Ren F, et al. Plant-inspired adhesive and tough hydrogel based on Ag-Lignin nanoparticles-triggered dynamic redox catechol chemistry. Nat Commun. 2019;10(1):1487. doi:10.1038/s41467-019-09351-2. [Google Scholar] [PubMed] [CrossRef]
45. Liu C, Li Y, Zhuang J, Xiang Z, Jiang W, He S, et al. Conductive hydrogels based on industrial lignin: opportunities and challenges. Polymers. 2022;14(18):3739. doi:10.3390/polym14183739. [Google Scholar] [PubMed] [CrossRef]
46. Palmese LL, Thapa RK, Sullivan MO, Kiick KL. Hybrid hydrogels for biomedical applications. Curr Opin Chem Eng. 2019;24:143–57. doi:10.1016/j.coche.2019.02.010. [Google Scholar] [PubMed] [CrossRef]
47. Jiménez-Torres HD, Hernández-Olmos SL, Reynaga-Delgado E, Orozco-Guareño E. Study on the degradation of a semi-synthetic lignin-acrylic acid hydrogel with common bacteria found in natural attenuation processes. Polymers. 2023;15(12):2588. doi:10.3390/polym15122588. [Google Scholar] [PubMed] [CrossRef]
48. Pandit S, Sharma P, Prakash A, Lal B, Bhuyan R, Ahmad I, et al. A comprehensive review on technical lignin, lignin hydrogels, properties, preparation, applications & challenges in lab to market transition. Ind Crops Prod. 2024;211(16):118262. doi:10.1016/j.indcrop.2024.118262. [Google Scholar] [CrossRef]
49. Shan Z, Jiang B, Wang P, Wu W, Jin Y. Sustainable lignin-based composite hydrogels for controlled drug release and self-healing in antimicrobial wound dressing. Int J Biol Macromol. 2025;285(6212):138327. doi:10.1016/j.ijbiomac.2024.138327. [Google Scholar] [PubMed] [CrossRef]
50. Li W, Cheng G, Wang S, Jiang Y, Liu X, Huang Q. Bifunctional lignocellulose nanofiber hydrogel possessing intriguing pH-responsiveness and self-healing capability towards wound healing applications. Int J Biol Macromol. 2024;260(Pt 1):129398. doi:10.1016/j.ijbiomac.2024.129398. [Google Scholar] [PubMed] [CrossRef]
51. Crivello G, Orlandini G, Morena AG, Torchio A, Mattu C, Boffito M, et al. Lignin-cobalt nano-enabled poly(pseudo)rotaxane supramolecular hydrogel for treating chronic wounds. Pharmaceutics. 2023;15(6):1717. doi:10.3390/pharmaceutics15061717. [Google Scholar] [PubMed] [CrossRef]
52. Pan X, Pan J, Li X, Wang Z, Ni Y, Wang Q. Tough supramolecular hydrogels crafted via lignin-induced self-assembly. Adv Mater. 2024;36(36):e2406671. doi:10.1002/adma.202406671. [Google Scholar] [PubMed] [CrossRef]
53. Zhao X, Zhao H, Mei A, Peng L, Sun J. Novel chitosan/lignin hydrogel prepared by the Mannich reaction for Pb(II) and Cu(II) removal from aqueous solution. Int J Biol Macromol. 2025;285:138177. doi:10.1016/j.ijbiomac.2024.138177. [Google Scholar] [PubMed] [CrossRef]
54. Bashir T, Dutta J, Masarat S, Rafatullah M, Asthana N, Khan AA, et al. Application of novel Chitosan-Lignin hydrogel beads for optimized removal of textile dye DB-218. J Mol Struct. 2025;1324(1):140892. doi:10.1016/j.molstruc.2024.140892. [Google Scholar] [CrossRef]
55. Zhang X, Yang L, Wang W, Xiang Y, Liu J, An Y, et al. Sodium alginate/sodium lignosulfonate hydrogel based on inert Ca2+ activation for water conservation and growth promotion. Environ Res. 2024;246(6):118144. doi:10.1016/j.envres.2024.118144. [Google Scholar] [PubMed] [CrossRef]
56. Yang J, Feng Y, Wang B, Miao J, Wei S, Li H, et al. Tough, multifunctional, and green double-network binary solvent eutectogel with in situ generation of lignin nanoparticles based on one-step dual phase separations for wearable flexible strain sensors. Chem Eng J. 2023;474(10):145544. doi:10.1016/j.cej.2023.145544. [Google Scholar] [CrossRef]
57. Zhao H, Hao S, Fu Q, Zhang X, Meng L, Xu F, et al. Ultrafast fabrication of lignin-encapsulated silica nanoparticles reinforced conductive hydrogels with high elasticity and self-adhesion for strain sensors. Chem Mater. 2022;34(11):5258–72. doi:10.1021/acs.chemmater.2c00934. [Google Scholar] [CrossRef]
58. Wang L, Wang Y, Peng S, Li S, Wu S. Study on highly sensitive capacitive pressure sensor based on silk fibroin-lignin nanoparticles hydrogel. Biomacromolecules. 2025;26(2):1044–52. doi:10.1021/acs.biomac.4c01334. [Google Scholar] [PubMed] [CrossRef]
59. Chen F, Zheng Y, Jian W, Li Z, Li J, Li H, et al. Graphitic hierarchical porous carbons derived from lignin-hydrogels for high-performance zinc-ion hybrid supercapacitors. J Energy Storage. 2025;108:115163. doi:10.1016/j.est.2024.115163. [Google Scholar] [CrossRef]
60. Li X, Xu Y, An XY, Gong L, Wang R, Liu ZM. Eco-friendly and efficient flame retardant rigid polyurethane foam reinforced with lignin and silica aerogel. Int J Biol Macromol. 2025;304(Pt 2):140947. doi:10.1016/j.ijbiomac.2025.140947. [Google Scholar] [PubMed] [CrossRef]
61. Mai Z, You K, Chen J, Sheng X, Chen Y. Biomass lignin/manganese ferrite-modified reduced graphene oxide aerogel composites for multi-source energy conversion. Int J Biol Macromol. 2025;306(Pt 4):141688. doi:10.1016/j.ijbiomac.2025.141688. [Google Scholar] [PubMed] [CrossRef]
62. Sabbatini B, Cambriani A, Cespi M, Palmieri GF, Perinelli DR, Bonacucina G. An overview of natural polymers as reinforcing agents for 3D printing. ChemEngineering. 2021;5(4):78. doi:10.3390/chemengineering5040078. [Google Scholar] [CrossRef]
63. Yu Z, Min Y, Ouyang Q, Fu Y, Mao Y, Xiang S, et al. Study on an injectable chitosan-lignin/poloxamer hydrogel loaded with platelet-rich plasma for intrauterine adhesion treatment. Polymers. 2025;17(4):474. doi:10.3390/polym17040474. [Google Scholar] [PubMed] [CrossRef]
64. Anand R, Collard D, Thomann JS, Duday D. Antimicrobial sponge: a polyvinyl alcohol, tannic acid and curcumin-loaded nanolignin hydrogel composite scaffold. Gels. 2025;11(3):168. doi:10.3390/gels11030168. [Google Scholar] [PubMed] [CrossRef]
65. Nirbhavane P, Kale SS, Kumar S, Athare T, Magar AG, Chalikwar SS, et al. Cotton stalk derived lignin-based hydrogel and its therapeutic utility in diabetic retinopathy. Biochem Biophys Res Commun. 2025;775(32):152123. doi:10.1016/j.bbrc.2025.152123. [Google Scholar] [PubMed] [CrossRef]
66. Lin J, Chen M, Zhao W, Zhang S, Liu J, Zhou Y, et al. Lignin-mediated dual conductive hydrogels with high conductivity, antibacterial activity and biocompatibility for chronic wound repair. Gels. 2025;11(4):283. doi:10.3390/gels11040283. [Google Scholar] [PubMed] [CrossRef]
67. de Albuquerque TL, Cunha Cavalcante VG, da Silva Rocha W, de Macedo AC, Ponte Rocha MV. Hydrogels based on lignin extracted from cashew apple bagasse and its application in antimicrobial wound dressings. Int J Biol Macromol. 2024;262:130169. doi:10.1016/j.ijbiomac.2024.130169. [Google Scholar] [PubMed] [CrossRef]
68. Li X, Chen R, Tang X, Chen M, Fan Y. A collagen/nanocellulose/lignin hydrogel dressing mimicking to the plant cell wall for enhanced wound healing and bacterial inhibition. Ind Crops Prod. 2025;228:120872. doi:10.1016/j.indcrop.2025.120872. [Google Scholar] [CrossRef]
69. Ma Q, Xu W, Xia J, Zhen Q, She D. Stabilization effects and mechanisms of lignin-based hydrogel-coated sulfide nano-zero-valent iron on lead and cadmium contamination in soil. J Hazard Mater. 2025;492(8):138095. doi:10.1016/j.jhazmat.2025.138095. [Google Scholar] [PubMed] [CrossRef]
70. Gonçalves LR, Ferreira RR, Souza AG, Barbosa RFS, da Costa TB, Rosa DS. Development of lignin biochar and its incorporation in cellulose hydrogels for water decontamination. Polym Bull. 2025;82(10):4479–503. doi:10.1007/s00289-025-05715-5. [Google Scholar] [CrossRef]
71. Kubra MJ, Ahmed T, Rahaman MS, Hasnine SMM, Sultana S, Mortuza F, et al. Eco-friendly lignin/N, N-dimethylacrylamide hydrogel with carrageenan and polyvinylpyrrolidone for amphoteric adsorption of methylene blue and Congo red: optimization, characterization, and adsorption. Int J Biol Macromol. 2025;306(Pt 3):141471. doi:10.1016/j.ijbiomac.2025.141471. [Google Scholar] [PubMed] [CrossRef]
72. Li J, Li L, Zhang Y, Zhang Q, Wang Y, Yu X. Bio-based hydrogel particles with enhanced water retention properties for alleviating drought stress of saline soil. J Clean Prod. 2025;518(11):145960. doi:10.1016/j.jclepro.2025.145960. [Google Scholar] [CrossRef]
73. Kong Q, Wang Y, Tan J, Long S, Li J, Zhao Z, et al. Efficient pesticide degradation through peroxydisulfate activation by magnetic Fe3S4 nanoparticle hydrogel composites: unveiling the pivotal role of electron transfer mechanisms. Appl Catal A Gen. 2025;699:120255. doi:10.1016/j.apcata.2025.120255. [Google Scholar] [CrossRef]
74. Huang J, Zhao L, Xiang P, Zhang F, Yang Y, Chao L, et al. Aminated lignin/cellulose-based hydrogel with high adhesion for wearable sensors. Langmuir. 2025;41(24):15484–93. doi:10.1021/acs.langmuir.5c01389. [Google Scholar] [PubMed] [CrossRef]
75. Shen Q, Xie M, Wang S, Wang L, Song G. C-lignin-enabled multifunctional hydrogels for flexible wearable sensors. Int J Biol Macromol. 2025;309((Pt 2)):142903. doi:10.1016/j.ijbiomac.2025.142903. [Google Scholar] [PubMed] [CrossRef]
76. Ma J, Yang W, Chen J, Zhou Y, Ye M, Xu X, et al. Lignosulfonate-enhanced dispersion and compatibility of liquid metal nanodroplets in PVA hydrogel for improved self-recovery and fatigue resistance in wearable sensors. Int J Biol Macromol. 2025;306(Pt 4):141653. doi:10.1016/j.ijbiomac.2025.141653. [Google Scholar] [PubMed] [CrossRef]
77. Du Z, Zhong X, Qiu J, Zhu L. Hemicellulose-lignin complex reinforcing deep eutectic solvent gels for wearable strain sensing and fire warning monitoring. Polymer. 2025;327:128421. doi:10.1016/j.polymer.2025.128421. [Google Scholar] [CrossRef]
78. Li J, Abdiryim T, Jamal R, Song K, Yang H, Liu J, et al. High-performance supercapacitor based on 3D Ti3C2Tx electrodes and sulfonated lignin gel electrolyte. J Colloid Interface Sci. 2025;698(1):137948. doi:10.1016/j.jcis.2025.137948. [Google Scholar] [PubMed] [CrossRef]
79. Zhong D, Wang K, Wei M, Wang H, Pei P. Enhanced Low-Temperature performance of flexible Zinc-Air batteries via High-Concentration ZnCl2 and lignin modified polyacrylamide hydrogels. Chem Eng J. 2025;510:161596. doi:10.1016/j.cej.2025.161596. [Google Scholar] [CrossRef]
80. Özkan A, Mıhlayanlar E, Turhan EA, Sarıoğlu E, Berlu P, Kaynak E, et al. Role of lignin on microstructure, mechanical properties and flame retardancy of nanocellulose-based composite hydrogels. Int J Biol Macromol. 2025;311((Pt 3)):144007. doi:10.1016/j.ijbiomac.2025.144007. [Google Scholar] [PubMed] [CrossRef]
81. Liu Y, Zhang Z, Wang J, Hu C, Tang J, Shi M, et al. Janus-structured lignin hydrogel evaporator via laser direct writing for high efficiency seawater desalination and solar power generation. Chem Eng J. 2025;513:162853. doi:10.1016/j.cej.2025.162853. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF
 Downloads
Downloads
 Citation Tools
Citation Tools