 Open Access
Open Access
REVIEW
Recent Developments in Super-Hydrophobicity and Fire-Resistance of Tannin-Based Non-Isocyanate Polyurethane Resins for Wood-Based Composites
1 Department of Forest Products, Faculty of Forestry and Environment, IPB University, Bogor, 16680, Indonesia
2 Research Center for Biomass and Bioproducts, National Research and Innovation Agency, South Tangerang, 15314, Indonesia
3 Department of Forestry, Faculty of Agriculture, University of Lampung, Bandar Lampung, 35145, Indonesia
4 Donghwa Enterprise Research and Development Center, Incheon, 22300, Republic of Korea
5 Research Collaboration Center for Biomass and Biorefinery, BRIN and Universitas Padjadjaran, 45363, Indonesia
* Corresponding Authors: Rita Kartika Sari. Email: ; Muhammad Adly Rahandi Lubis. Email:
(This article belongs to the Special Issue: Renewable and Biosourced Adhesives-2023)
Journal of Renewable Materials 2026, 14(3), 4 https://doi.org/10.32604/jrm.2025.02025-0114
Received 16 June 2025; Accepted 05 September 2025; Issue published 25 March 2026
Abstract
Recent advancements in developing tannin-based non-isocyanate polyurethane (NIPU) resins have unlocked new possibilities for sustainable and eco-friendly wood adhesives. Unlike conventional polyurethane, NIPUs eliminate hazardous isocyanates, offering safer alternatives for industrial applications. Tannin, a naturally occurring polyphenolic compound, plays a pivotal role in enhancing these resins’ fire-resistance and super-hydrophobic properties. This review highlights key developments in synthesizing tannin-based NIPU, focusing on various polymerization techniques such as polyaddition, polycondensation, ring-opening polymerization, and rearrangement. These strategies contribute to improved mechanical strength, thermal stability, and resistance to water absorption. Recent studies demonstrate that tannin-based NIPU adhesives meet or exceed international internal bonding strength, elasticity, and durability standards. Their application in wood-based composites like oriented strand boards (OSB) has shown promising results, offering improved resistance to moisture and superior dimensional stability. Furthermore, the cross-linked polymer network, supported by chemical modifications like the introduction of fluoroalkyl groups, significantly enhances hydrophobicity and fire retardancy. Despite these advancements, challenges remain in terms of scalability, cost, and integration with existing manufacturing processes. Future research should focus on optimizing reaction conditions, improving raw material availability, and developing industrial-scale production methods. This review underscores the potential of tannin-based NIPU resins as a viable and sustainable alternative for various wood composite applications.Graphic Abstract
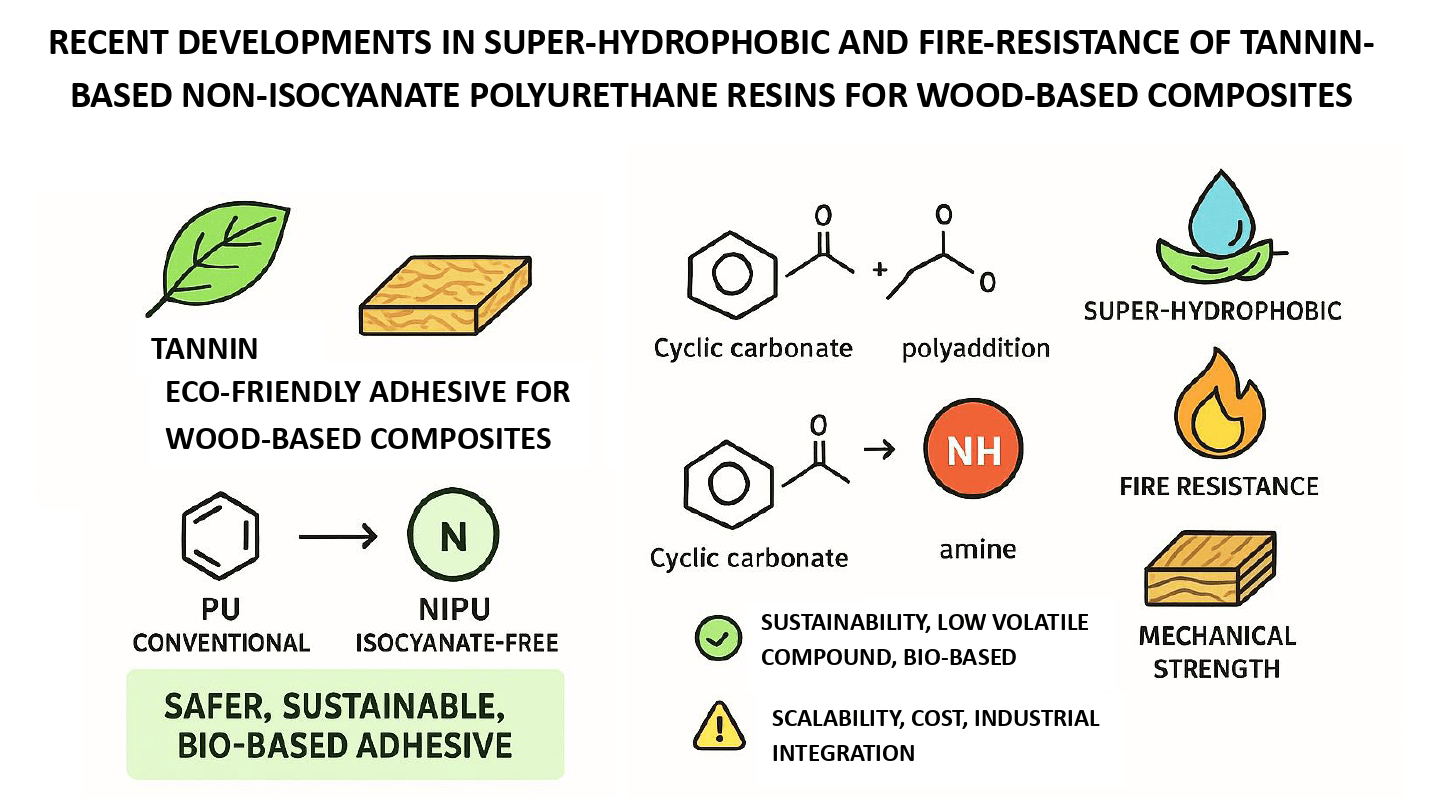
Keywords
Polyurethane adhesives have gained significant attention in various industries due to their excellent mechanical properties, versatility, and potential for sustainability. These adhesives are synthesized through the reaction of polyols with isocyanates, resulting in a polymer exhibiting strong adhesion, flexibility, and durability across various applications, including wood bonding, textiles, and automotive components [1–3]. The unique properties of polyurethane adhesives can be tailored by modifying their chemical composition, such as the type of polyol used, the ratio of isocyanate to hydroxyl groups, and the incorporation of additives like fillers or chain extenders [4,5].
Recent advancements in the formulation of polyurethane adhesives have focused on enhancing their environmental sustainability. For instance, bio-based polyols derived from renewable resources, such as vegetable oils and lignin, have been explored to reduce reliance on petroleum-based materials [6,7]. These bio-based formulations contribute to lower environmental impact and maintain or improve adhesive performance. Studies have shown that polyurethane adhesives made from modified polysaccharides or lignin exhibit superior bonding strength and flexibility compared to traditional formulations [8–10]. Furthermore, developing waterborne polyurethane adhesives has emerged as a promising alternative to solvent-based systems, offering lower volatile organic compound emissions and reduced toxicity [11].
The transition from isocyanate-based polyurethanes (PUs) to non-isocyanate polyurethanes (NIPUs) is driven by the need for safer, more environmentally friendly alternatives. Isocyanates, while effective in PU synthesis, pose significant health risks due to their high reactivity and toxicity, leading to severe respiratory and ocular damage upon exposure [12,13]. Furthermore, the production of isocyanates often involves the use of phosgene, a highly toxic compound, which raises additional safety concerns [14,15]. In contrast, NIPUs utilize alternative raw materials and synthesis methods that mitigate these hazards, making them a compelling option for industrial and academic research [16,17].
Beyond safety and environmental benefits, NIPUs—particularly those formulated with bio-based components such as tannins also offer enhanced functional properties. Tannin-based polyurethane adhesives exhibit remarkable fire-resistance. Research has shown that tannin foams have higher thermal and fire resistance and can self-extinguish the flame, making them a potential alternative to polyurethane foams for fire protection in building engineering and automotive industries [18]. Incorporating tannins into polyurethane formulations enhances their thermal stability and reduces flammability, making them suitable for applications where fire resistance is critical [19]. For instance, a study highlighted the dual functionality of tannin-based coatings that exhibit intumescent and fire-retardant properties, depending on tannin concentration [19]. This versatility allows for tailored applications in various industries, including construction and automotive sectors, where fire safety is paramount.
In addition to their fire-resistant properties, tannin-based polyurethanes can be engineered to achieve super-hydrophobic characteristics. The super-hydrophobicity arises from the unique micro/nano-structural features of the coatings, which can be enhanced by the incorporation of nanoparticles or by manipulating the surface roughness [20,21]. NIPU has also gained much attention because of its improved porosity, water absorption, and thermal and chemical resistance compared to conventional polyurethanes [22]. For example, the surface coating of a medium-density fiberboard (MDF) panel was implemented at 170°C under mild pressure to maintain the minimum filming temp so it could produce good hydrophobicity on the surface [23]. The super-hydrophobic nature of these materials is attributed to the hydrophobic groups in tannins and the structural modifications that create a rough surface texture, which collectively reduce the adhesion of water droplets [24–26].
To better understand the direction and scope of research in this area, a bibliometric analysis was conducted based on 20 publications indexed by Scopus. Fig. 1 presents the visualization of the relationship network between keywords, revealing 32 interconnected terms grouped into several colored clusters. Research on tannin-based materials in the red, blue, and green clusters illustrates an integrated approach to developing environmentally friendly NIPUs with specific applications. The red cluster focuses on applying tannin NIPUs as wood adhesives, utilizing the natural adhesive properties of tannins that can reduce the emission of volatile organic compounds (VOCs). In addition, this cluster highlights the use of analytical techniques, such as Fourier Transform Infrared Spectroscopy (FTIR) and MALDI-TOF, to study the chemical structure and curing mechanism of the adhesive to improve performance.
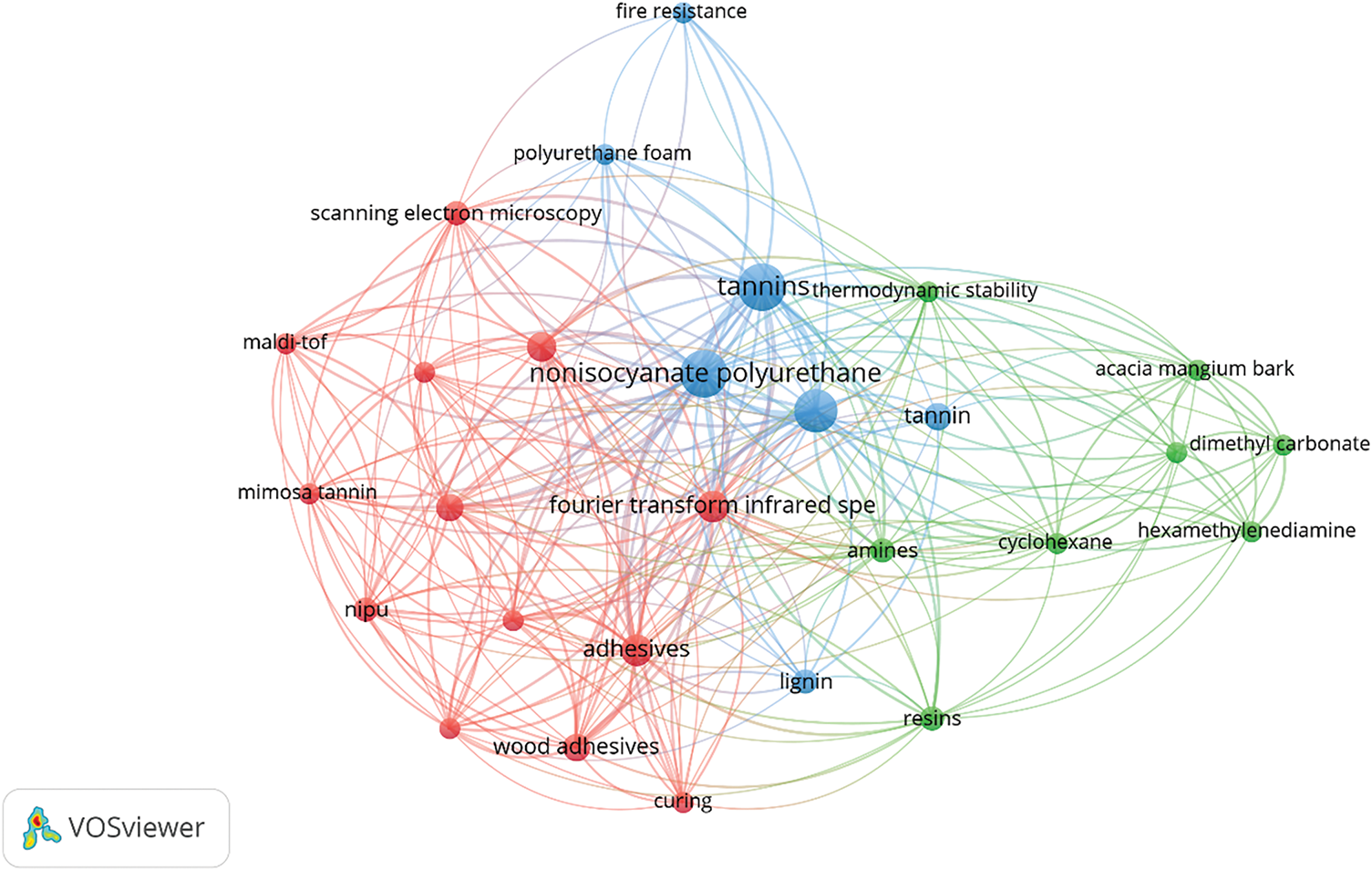
Figure 1: A visual map of keyword occurrences in references considered for “Tannin-based NIPU”. Created with VOSviewers 1.6.20
On the other hand, the blue cluster emphasizes the development of tannin-based polyurethane materials that have superior thermal, mechanical, and fire-resistance properties. The natural content of polyphenols in tannins provides fire-resistance properties, making them attractive for construction and thermal insulation applications. The green cluster complements this with a focus on the use of environmentally friendly raw materials such as dimethyl carbonate (DMC) and hexamethylenediamine (HMDA) for NIPU synthesis, replacing hazardous isocyanates. This combined focus creates a versatile, sustainable, and high-performance tannin-based material solution for modern industrial applications.
2 Synthesis of Bio-Based NIPU Resin
2.1 Bio-Based NIPU Synthesis Pathways
Tailor-made bio-based NIPUs are a form of polyurethane made with bio-based precursors instead of harmful isocyanates (Table 1). These materials are meant to be ecologically friendly and adaptable to specific uses. The production of this form of polyurethane signifies a step toward more sustainable chemistry. There are four primary methods for obtaining an NIPU: polycondensation, rearrangement, ring-opening polymerization, and polyaddition [27], as shown in Fig. 2.
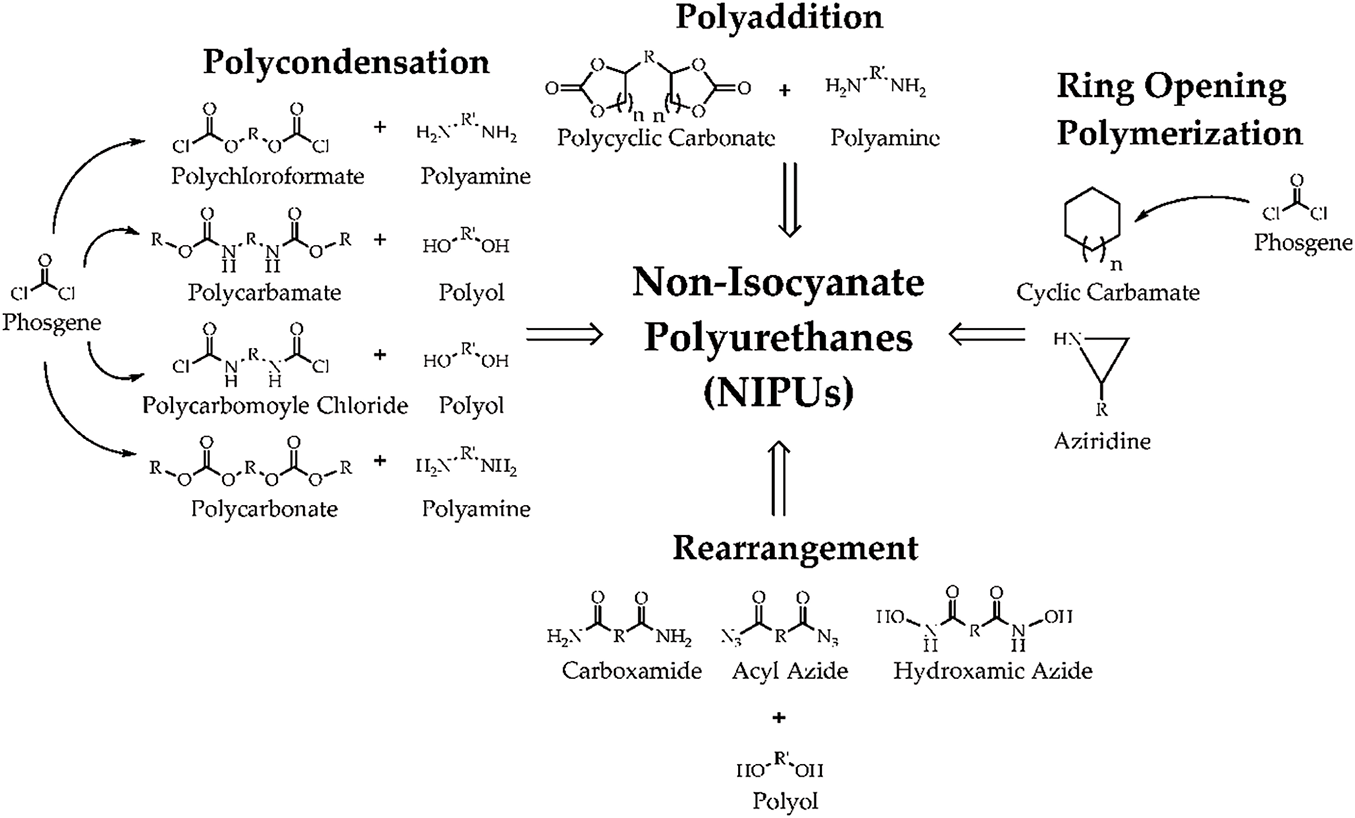
Figure 2: Possible pathways for synthesis of NIPUs [28]
In the first route, polycondensation, the NIPU synthesis process involves a stepwise reaction between polyfunctional materials such as polychloroformate, polycarbonate, or polycarbonate with co-reactant compounds such as polyamine or polyol [27,35,36]. This process results in polymer chain growth through condensation of functional groups, where small molecules such as HCl or water are released during the reaction. For example, polychloroformate reacts with polyamine to form a urethane linkage without the use of isocyanate [37]. Similarly, polycarbamate can interact with polyol, producing a polymer structure with adjustable mechanical properties [38]. This method is considered safer because it avoids toxic materials such as isocyanate or phosgene, which are usually used in conventional polyurethanes.
The polyaddition route in NIPU synthesis is a process in which two main components, namely cyclic carbonate and polyamine, react to form a polymer chain without producing by-products [39,40]. This reaction occurs in a chain-growth polymerization mechanism, where monomers or precursors are added stepwise into the growing polymer chain. In this route, cyclic carbonate (e.g., from vegetable oil or other bio-based materials) acts as a carbonate source. In contrast, polyamine acts as a nucleophilic agent that attacks the carbonate in an addition reaction to form a urethane bridge. One of the advantages of polyaddition is that it can produce polymers with greater control over the desired structure and material properties, making it suitable for applications such as coatings, adhesives, and foams with high mechanical and thermal resistance [41].
Ring-opening polymerization (ROP) is another pathway for synthesizing NIPU that involves opening cyclic monomers to form long polymer chains. In the context of NIPUs, cyclic carbamates (typically derived from phosgene or bio-based sources) react with nucleophilic species like amines to initiate polymerization [42]. Phosgene, or alternatives like cyclic carbonates, are first used to form cyclic monomers, which undergo ring-opening reactions upon adding amines or other suitable nucleophiles [43]. This process does not release by-products such as CO2 or water, making it an environmentally friendly alternative to traditional isocyanate-based polyurethanes. ROP is especially valuable for creating NIPUs with enhanced mechanical properties, thermal stability, and improved biodegradability.
In the rearrangement pathway, NIPUs are synthesized through the chemical rearrangement of functional groups like carboxamides, acyl azides, and hydroxamic azides [44]. These compounds undergo various rearrangements under thermal or catalytic conditions, leading to the formation of urethane-like linkages. For instance, carboxamide groups may rearrange to form isocyanate-like structures or directly react with polyols to form polymers with urethane groups [45]. The rearrangement process typically requires precise control over temperature, pressure, and the presence of catalysts to drive the transformation of the precursor materials into crosslinked polyurethanes. This pathway offers a more flexible and often more straightforward approach than polycondensation or polyaddition, as it can utilize existing chemical species in novel configurations.
Compared to cyclic carbonates, aliphatic carbonates such as DMC or diethyl carbonate (DEC) offer an alternative route in the polyaddition synthesis of NIPU, particularly when combined with polyphenolic compounds like tannins or carbohydrate-based polyols such as glucose and sucrose. While cyclic carbonates react directly with diamines via ring-opening to form urethane linkages in a single-step reaction, aliphatic carbonates typically require a two-step mechanism [46]. In this mechanism, a hydroxyl-rich compound such as tannin, glucose, or polyol is first carbonate-activated by reacting with DMC to form linear or branched carbonate intermediates, which then undergo nucleophilic attack by amines to form urethane bonds. Although less reactive due to the absence of ring strain, this approach offers advantages such as lower toxicity, mild reaction conditions, and high atom economy. Several studies have validated this route: Xi et al. developed glucose- and sucrose-based NIPU adhesives and self-blowing foams with promising mechanical properties [47], while Chen et al. reported glucose-tannin NIPU foams with enhanced fire retardancy and low-temperature curing performance [48]. Therefore, while cyclic carbonate-based NIPUs are generally preferred for their reactivity, the aliphatic carbonate route provides greater feedstock flexibility and better alignment with green chemistry principles, particularly for wood adhesives and insulation foam applications.
2.2 Tannin-Based NIPU Synthesis
The reaction between gallic acid and HMDA produces a compound containing urethane bonds, in which one molecule of gallic acid is bound to HMDA. Notably, the other amino group of HMDA remains free, allowing further cross-linking reactions that can contribute to the formation of a three-dimensional network in foam structures. Gallic acid is often used as a model compound in tannin-based NIPU studies due to its highly reactive structure, characterized by multiple hydroxyl groups and a carboxylic acid functional group. However, it is not the only relevant phenolic compound; other tannin-derived molecules such as catechin, resorcinol, and pyrogallol also significantly influence the reactivity and performance of tannin-based NIPU adhesives. As illustrated in Fig. 3, the formation of non-isocyanate urethane bonds involves not only the carbonation and subsequent reaction with HMDA but also the ability of gallic acid’s active sites to form cross-links, which are crucial for the mechanical strength and structural integrity of NIPU foams.
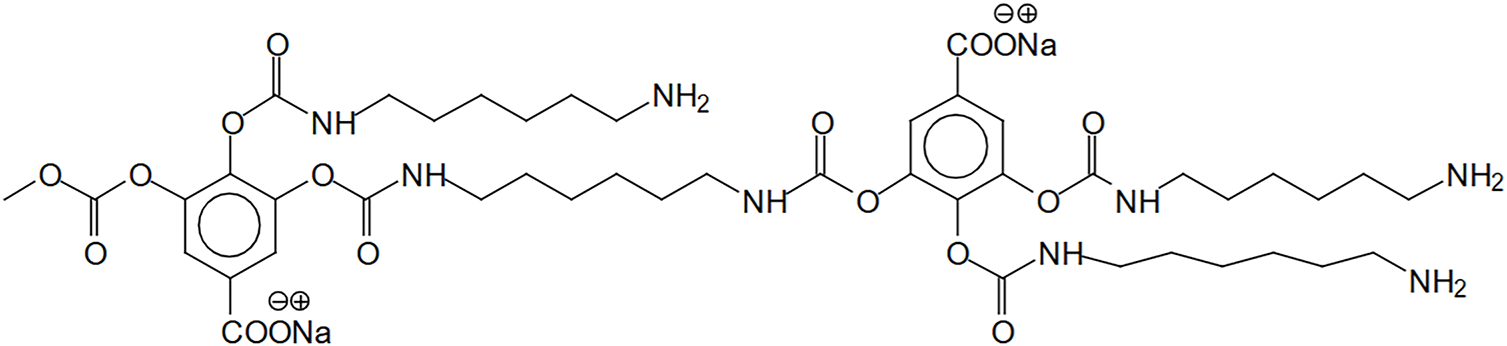
Figure 3: Demonstrating the participation of gallic acid residues in the NIPU formation [49]
The primary reaction in forming NIPU foams begins with carbonating the hydroxyl groups on the tannin structure using DMC and the polycondensation method. Hydroxyl groups from phenolic compounds such as gallic acid or carbohydrates, tightly bound to the tannin structure, react with DMC to form carbonate ester compounds. Next, this carbonation product reacts with HMDA, where the carbonate group interacts with the amino group (–NH2) of HMDA, producing a non-isocyanate urethane bond (-NH-CO-O-). This reaction creates a complex molecular network, with large tannin structures such as pentagalloyl glucose (glucose gallate) participating in the formation of cross-links. In addition, smaller molecular fragments, such as gallic acid, that are separated during the extraction process are also involved in this reaction, resulting in a very complex three-dimensional structure that contributes to the mechanical properties of the resulting foam.
One example of a polyaddition route is Azadeh et al. [49] research in Fig. 4. They analyzed the various chemical species initially present in commercial chestnut tannin extracts and the complex chemical environment formed during hydrolyzable NIPU tannin foam preparation. The results of instrumental chemical analysis showed that gallic acid and gallic acid residues bound to carbohydrates interact with each other through reactions with dimethyl carbonate and diamine. In addition, cross-linking occurs between glucose residues that are integral parts of tannins and mixed cross-linking between gallic acid residues and glucose residues [50,51]. These reactions involve low molecular weight species formed due to the cleavage of tannin polymers during extraction. More interestingly, reactions occur between glucose chains esterified with gallic acid and glucose from similar carbohydrate chains [52,53]. This analysis confirms the formation of NIPU bridges between all these species. Furthermore, it was found that citric acid not only acts as a foaming catalyst but also reacts covalently, participating in the cross-linking of the overall foam system by esterifying glucose residues on hydrolyzable tannins, as well as possibly some phenolic materials.
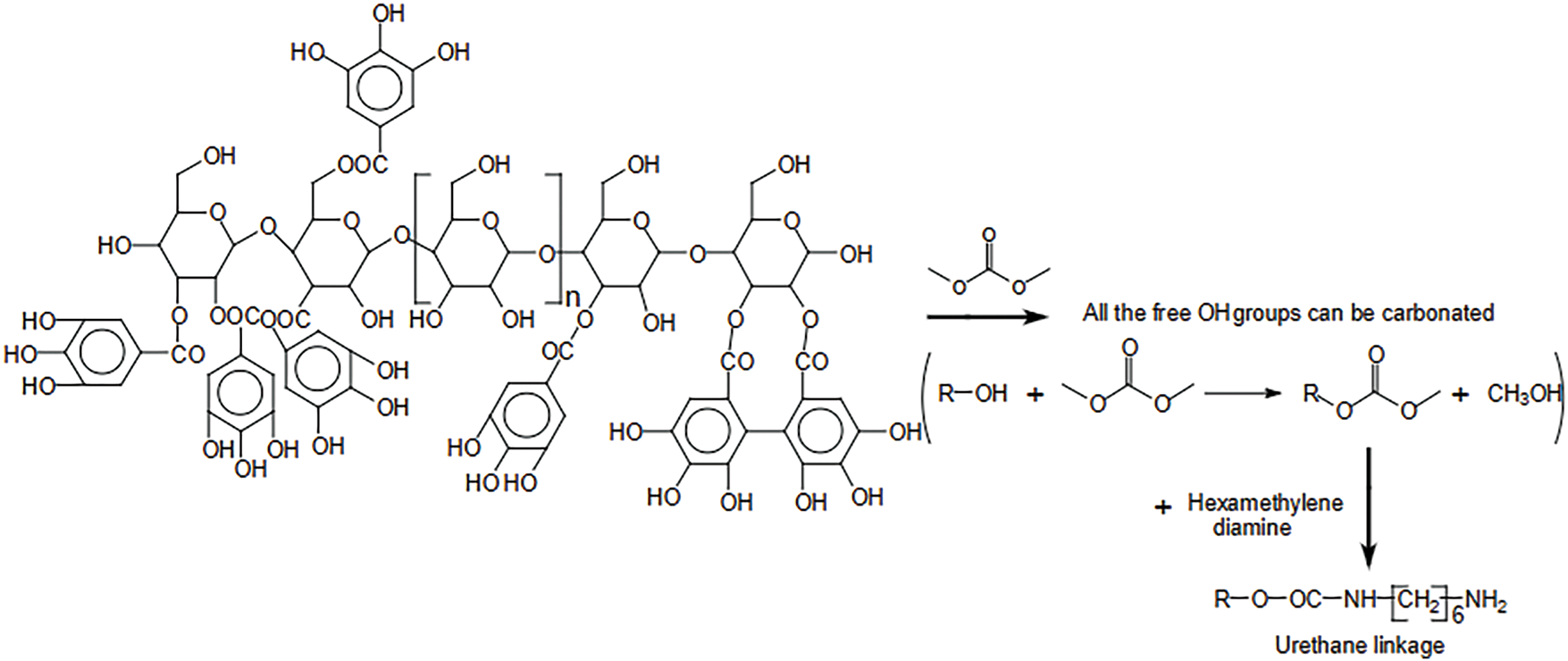
Figure 4: Reaction scheme showing that all the free-OH groups in a glucose chain, with the glucose linked by glycosidic linkages, with on each glucose either no galloyl residue attached, or with a variable number of galloyl residues attached can be first carbonated and the carbonate group reacted with a diamine to form a urethane linkage [49]
A small aliphatic carbonate, DMC, has been shown to offer high flexibility in the synthesis of NIPU. In addition to its reactive chemistry, DMC also provides economic advantages due to its relatively low cost compared to other carbonate precursors [54]. Its use in polyaddition reaction pathways, particularly with diamines such as HMDA, allows the formation of non-isocyanate urethane bonds (-NH-C=O-O-) without the production of harmful byproducts. This reaction between carbonate and amine is environmentally friendly and capable of producing strong and stable polymer networks, making it highly suitable for use as adhesives in wood-based products. The combination of DMC and HMDA in the polyaddition pathway has been widely applied in the development of NIPU adhesives, as it provides fast and strong bonding, important characteristics in the wood panel industry, such as oriented strand board (OSB) and plywood.
3 Super-Hydrophobic Properties of Tannin-Based NIPU Resins
Tannins are plant-derived polyphenols known for their amphiphilic nature, enabling interactions with hydrophilic and hydrophobic molecules. In tannin-based NIPU resins, hydrophobicity is a key functional attribute that contributes to water resistance and durability. This property emerges from several structural and chemical factors inherent to the polymer network. Hydrophobicity in tannin-based NIPU resins arises from multiple synergistic mechanisms. First, the cross-linked polymer network provides mechanical strength and stability, ensuring long-term water resistance [55,56]. Second, hydrophobic functional groups such as long alkyl chains or fluoroalkyl segments lower surface energy and minimize moisture absorption [57,58]. DMC and Hexamethylene Tetraamine (HMTA) serve as non-isocyanate substitutes, while tannin-glyoxal replaces polyols, maintaining performance with reduced environmental impact.
The use of DMC and HMTA as isocyanate-free curing agents, along with tannin-glyoxal as a polyol substitute, not only enhances sustainability but also introduces aliphatic chains and stable methoxy (-OCH2) linkages that improve polymer cohesion and hydrophobic performance [59,60]. Urethane groups (-NHCOO-) further reduce water interactions, reinforcing the hydrophobic effect [61]. Additionally, hierarchical micro- or nanoscale surface roughness increases the contact angle, mimicking natural super-hydrophobic surfaces like lotus leaves, enabling self-cleaning properties [62].
In theory, introducing fluoroalkyl and long alkyl chains into NIPU polymers could significantly enhance hydrophobicity by lowering surface energy and creating a physical barrier to moisture penetration. Fluoroalkyl groups, characterized by strong carbon–fluorine bonds, reduce the attraction of water and air molecules to the polymer surface, contributing to both chemical stability and water repellency [36,63]. Meanwhile, long alkyl chains provide a nonpolar layer that prevents interaction with polar substances such as water. However, such modifications have not yet been widely explored or reported in tannin-based NIPU systems. Most existing studies on fluorinated NIPU resins refer to petroleum-based or synthetic polyol systems, and their direct applicability to tannin-based formulations remains unverified. Therefore, while fluorinated NIPUs exist in synthetic systems, their compatibility and direct transfer to tannin-based formulations remain unverified and represent an open area for future research.
Fig. 5 shows the structure of a polymer modified with fluoroalkyl groups (marked with purple fluorine atoms) and long alkyl chains [36,64]. However, while these modifications are effective, they are not without drawbacks. Fluoroalkyl compounds are costly and environmentally persistent, raising concerns about sustainability and long-term ecological impact. Moreover, achieving uniform distribution of these groups within the polymer matrix can be technically challenging, potentially affecting surface consistency. Despite the enhancements these functional groups provide, it is important to recognize that tannin contributes to the overall hydrophobic behavior. Its aromatic and polyphenolic structure offers reactive sites for modification and can inherently reduce polarity through esterification or cross-linking. Therefore, the synergy between tannin’s native structure and the added hydrophobic components is crucial in achieving durable and water-resistant NIPU materials.
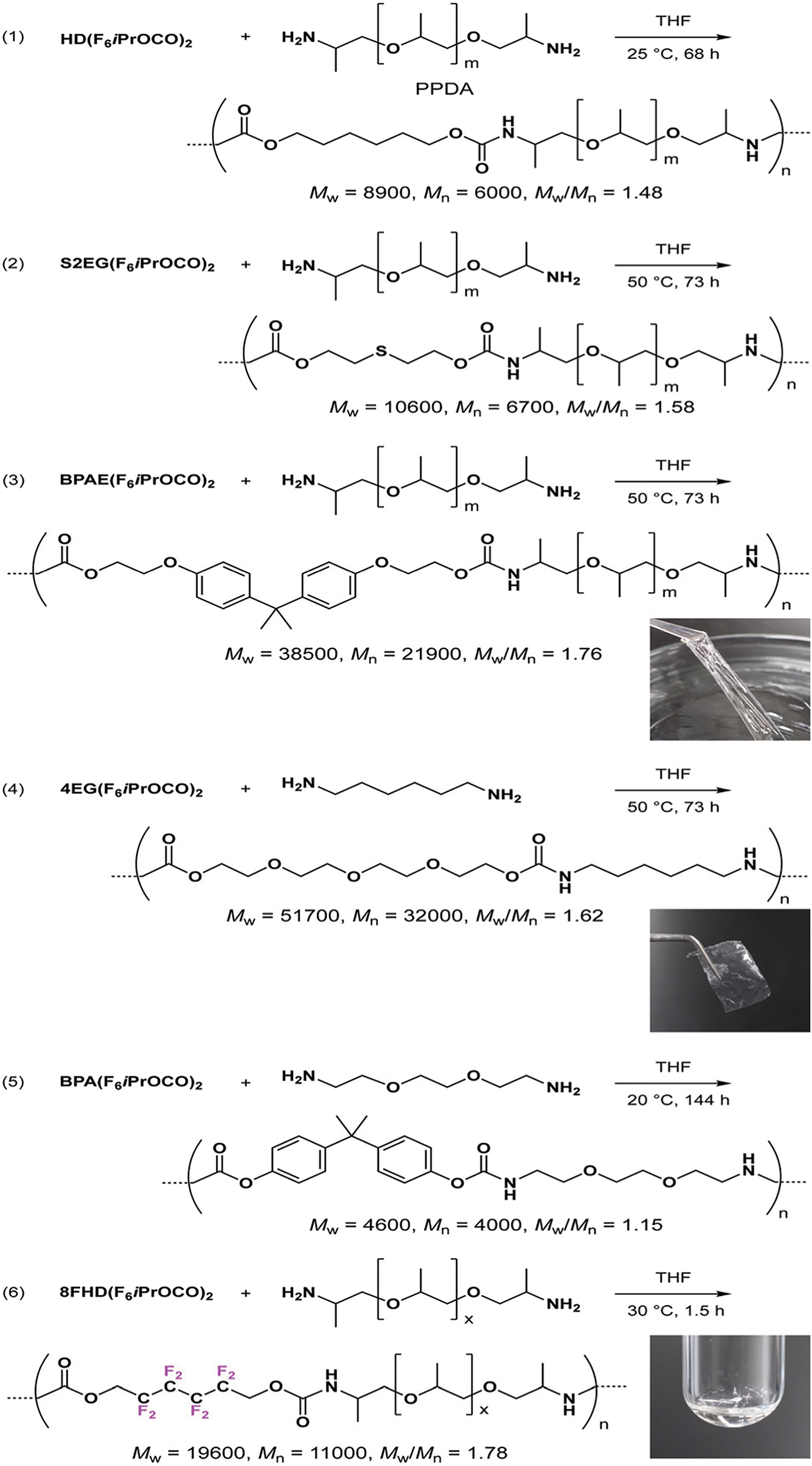
Figure 5: NIPU synthesis from poly(propylene glycol) bis(fluoroalkyl) bis(carbonate)s and diamines [36]
Lastly, the structural integrity of the polymer network sustains hydrophobic performance over time. Rough surface textures amplify water resistance by increasing water contact angles, preventing absorption, and enabling self-cleaning effects [65,66]. This interplay between chemical modification, surface engineering, and polymer cross-linking ensures durable hydrophobicity in tannin-based NIPU adhesives [67].
4 Fire-Resistance Properties of Tannin-Based NIPU Resins
Tannin-based NIPU resins have emerged as a promising alternative to conventional polyurethanes, primarily due to their environmentally friendly nature and the ability to be derived from renewable resources like plant tannins. The fire-resistance properties of these resins are of significant interest, particularly in applications where flame retardancy is critical, such as in coatings, adhesives, and construction materials. Tannin-based NIPU foams exhibit a limiting oxygen index (LOI) value of 24.45%, which is significantly higher than the LOI values of traditional PU foams (17%–19%) [68]. This indicates better fire resistance. The cross-linked structure of NIPU resins can result in a more thermally stable material that is less prone to rapid degradation under high temperatures [69]. This makes it more resistant to ignition and heat propagation than traditional polyurethanes.
Tannin-based NIPU resins exhibit enhanced fire-resistance through multiple mechanisms. Tannins, as polyphenolic compounds, promote the formation of a protective char layer upon exposure to heat, acting as a thermal barrier that slows down heat transfer and material decomposition [70]. Additionally, the cross-linked polymer network formed between tannin and monomers such as cyclic carbonates and amines improves thermal stability by reducing polymer chain mobility and limiting flame propagation [28]. However, the effectiveness of char formation can vary depending on formulation parameters such as the tannin content, curing temperature, or the type of cross-linking agent used. Under suboptimal conditions, incomplete char formation may reduce fire performance. Compared to conventional flame retardants such as halogenated compounds or phosphorus-based additives, tannin-based systems offer a more sustainable and non-toxic alternative. Nevertheless, their performance may still require optimization to match the efficiency of industrial-grade fire retardants, particularly in high-risk applications. Incorporating a systematic formulation approach could help maximize their fire-retardant potential while maintaining their environmental advantages.
Tannin-based NIPU resins also degrade at higher temperatures than conventional polyurethanes, releasing non-flammable gases such as carbon dioxide during thermal decomposition, which dilutes flammable gases in the environment and reduces fire spread [71]. To further enhance fire resistance, flame retardants such as phosphorus-based or halogen-free compounds can be incorporated, promoting char formation and improving flame retardancy. Furthermore, adding nanomaterials or inorganic fillers can create synergistic effects, strengthening the char layer, improving heat barrier properties, and aiding in smoke suppression.
Figs. 6 and 7 collectively illustrate how the chemical structure of tannin-based NIPU resins translates into enhanced thermal and fire-resistant properties. Fig. 6 outlines the synthesis of NIPUs from tannin-based cyclic carbonates and diamines. When a polyphenolic compound combines tannin with cyclic carbonates and diamines, it forms a cross-linked polymer structure. The cyclic carbonate groups react with the amine functional groups of diamines, creating stable, cross-linked networks [72,73]. This cross-linking process enhances the material’s structural integrity, making it more resistant to thermal degradation and the spread of fire [28]. The resulting network acts as a barrier to heat and flame, improving the material’s fire-resistance. Furthermore, the presence of tannin, which can produce a protective char layer during combustion, contributes to the overall fire-retardant behavior of the resin [74]. The high degree of cross-linking, facilitated by these reactions, stabilizes the polymer, preventing it from breaking down easily under heat and thus offering superior fire-resistance compared to traditional polyurethanes. This structure is key in developing bio-based materials with enhanced thermal stability and fire-resistance, particularly in applications requiring sustainable and safe alternatives to conventional polymers.
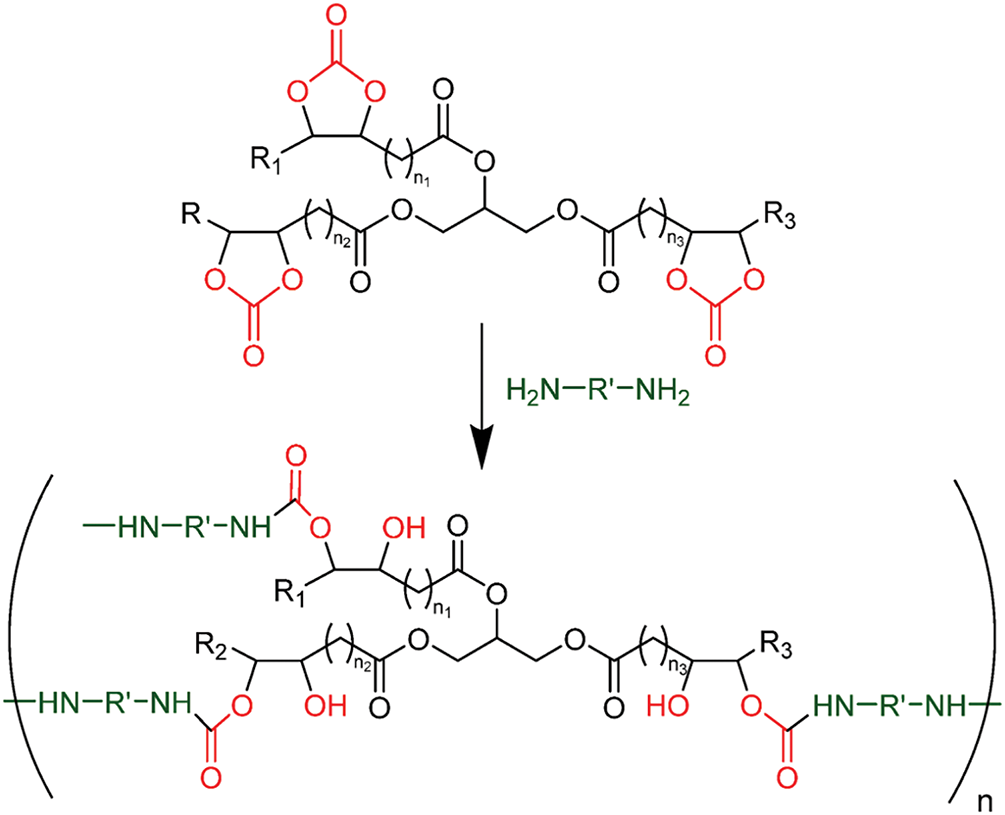
Figure 6: Polyaddition of carbonated triglycerides and diamines. Cyclic carbonate groups and derivatives are in red. Amine groups in green [28]
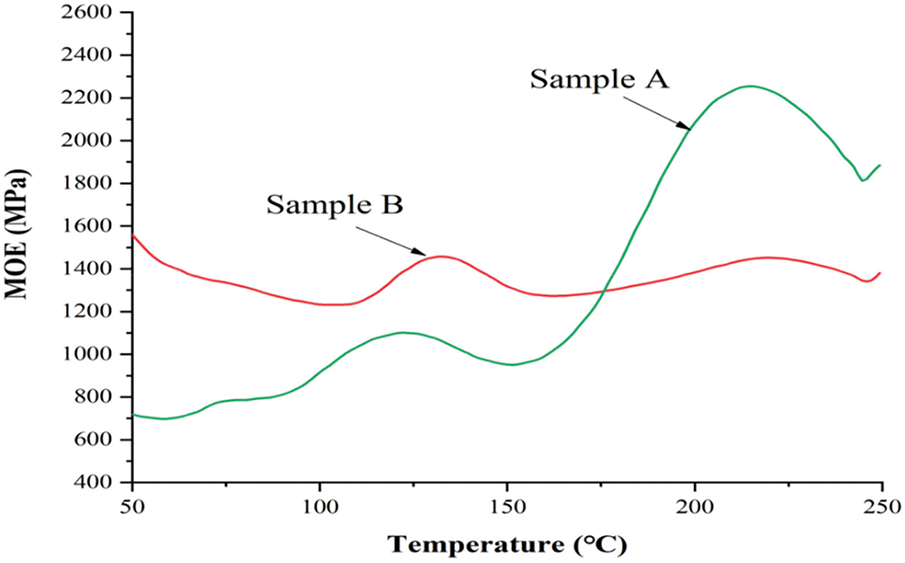
Figure 7: Thermomechanical analysis (TMA) for resins A and B [75]
Fig. 7 illustrates the Thermomechanical Analysis (TMA) traces for resin A (tannin–humin NIPU) and resin B (humin NIPU) [75]. Resin A (green line) shows a higher modulus of elasticity (MOE) at high temperatures than resin B (red line), especially at the second peak around 220°C. This indicates that resin A has a more stable structure against increasing temperatures. In contrast, resin B has a smaller increase in MOE and tends to decrease after 150°C, indicating that its polymer network is more susceptible to deformation and degradation. The higher thermal stability of resin A can be associated with better fire-resistance, since materials that can maintain their stiffness at high temperatures are generally less likely to ignite or experience rapid thermal degradation [76].
Resin A has more cross-linking than resin B due to its highly reactive tannin content. Tannin is essential in increasing thermal stability due to its structure rich in aromatic rings, which tend to form a carbon layer (char) when burning [77]. This layer is a barrier to heat and oxygen, slowing fire spread. In contrast, resin B, which is based solely on humin, has fewer reactive sites; therefore, its polymer network is weaker and more easily degraded at high temperatures. Therefore, resin A, with a tannin-humin composition, has the potential to have better fire resistance than resin B based on pure humin. The graph shows that chemical hardening (cross-linking) occurs at 150°C–160°C, indicating that the adhesive must be heated to a reasonably high temperature to achieve complete stability. After the temperature reaches 200°C, wood degradation occurs, which can be a significant factor in the decrease in MOE after the second peak. Adhesives with a more stable structure at high temperatures can help slow down the wood degradation process, thereby contributing to increased fire-resistance by reducing the burning rate and maintaining material integrity for longer.
TGA and DTG analyses provide insights into the thermal degradation behavior of the tannin-based NIPU resins. The higher thermal stability and greater char residue observed in the TGA curves indicate that the resin possesses inherent fire-retardant characteristics. The formation of a stable char layer during decomposition acts as a thermal barrier, reducing heat release and slowing down the combustion process. Although TGA does not directly assess flammability, the data strongly correlate with improved fire-resistance observed in many char-forming polymer systems. The relatively high char yield observed at 790°C, as reported in Fig. 8, suggests that the T/G-F foams form stable carbonaceous residues upon thermal decomposition. This char acts as a physical barrier that insulates the underlying material from heat and oxygen, thereby slowing down further degradation and combustion. Such behavior is characteristic of materials with intrinsic fire-retardant properties. Although TGA is not a direct flammability test, the multi-step degradation profile and significant char formation indicate that tannin-glucose-based NIPU foams may offer improved flame resistance through enhanced thermal stability and char formation mechanisms.
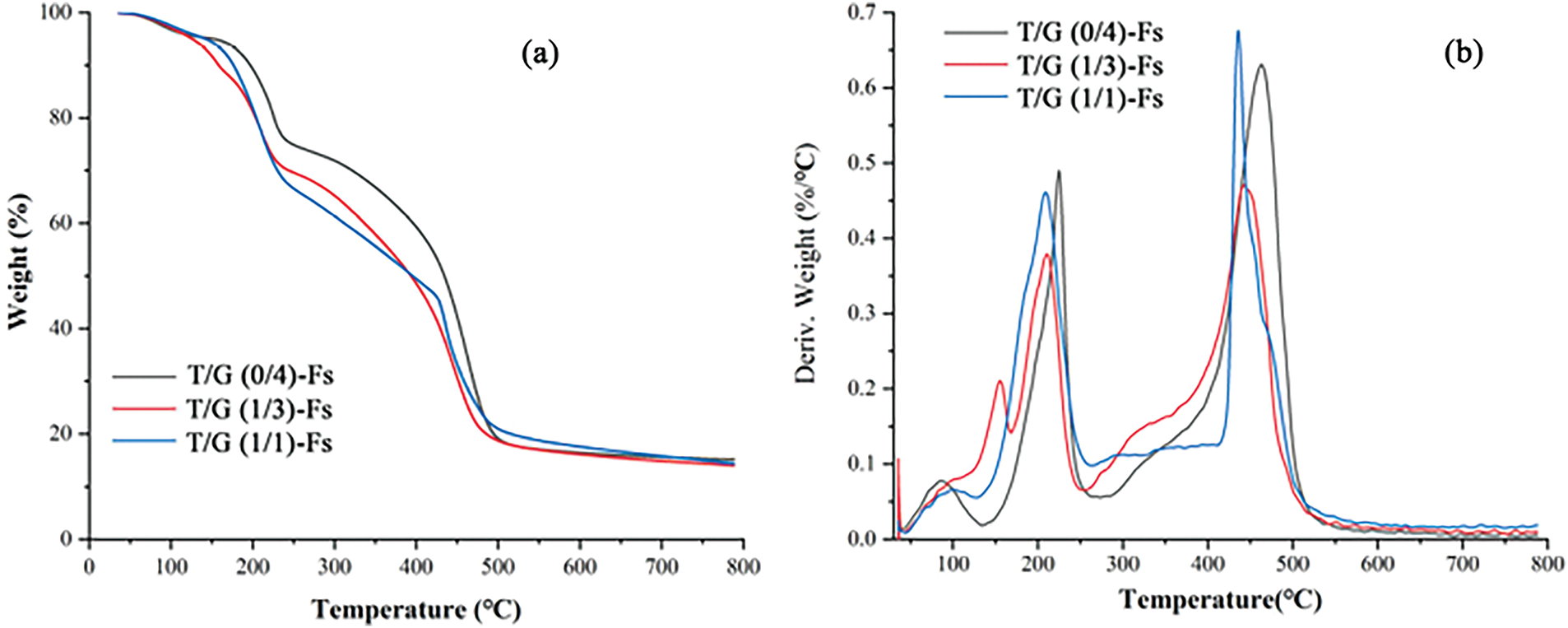
Figure 8: TGA (a) and DTG (b) curves of Tannin-glucose NIPU (T/G): T/G (0/4)-Fs, T/G (1/3)-Fs, and T/G (1/1)-Fs [48]
5 Applications of Tannin-Based NIPU Resins for Wood-Based Composites
Castor oil-based polyurethane (COPU) has been investigated as an alternative adhesive for manufacturing oriented strand boards (Fig. 9). Data shows pure COPU has very low water absorption and minimal swelling, making it a moisture-resistant material. In addition, when used in a composite with luffa mats, COPU provides excellent resistance to water absorption because its matrix can coat and protect the luffa fibers effectively. In contrast, commercial OSB exhibits higher water absorption and swelling levels because the adhesive matrix does not completely encase the wood fragments. This suggests that COPU can be a superior adhesive alternative in improving the water resistance of OSB.
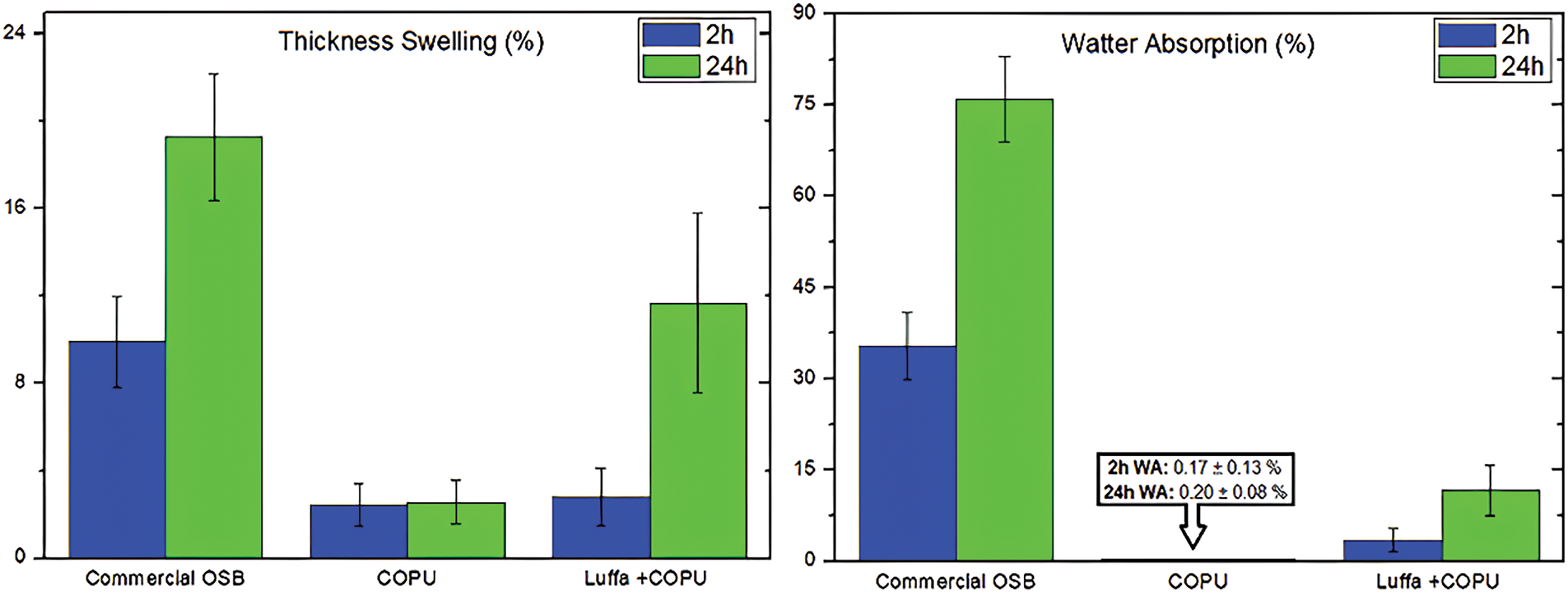
Figure 9: Water absorption (WA) and thickness swelling properties after 2 and 24 h for the commercial oriented strand board, plain COPU, and luffa-reinforced COPU composite [78]
The reaction between castor oil and isocyanate produces a polyurethane structure with a high degree of cross-linking, forming a dense polymer network that is difficult for water molecules to penetrate [78]. This structure contributes to increased resistance to water absorption, making it a more stable material in humid conditions. Conventional polyurethanes often contain hydrophilic groups, such as unreacted hydroxyl groups (-OH), which can attract and retain water in their polymer structure [79]. However, in COPU, most hydroxyl groups react with isocyanate during the polymerization process, resulting in a more hydrophobic polyurethane chain that can interact less with water. This makes COPU an alternative adhesive that is more moisture-resistant than conventional polyurethane.
Sari et al. have studied the use of PVOH-Tannin-Hexamine (PTH) based adhesives for plywood applications [80]. The results showed that the F2 adhesive formulation, which has higher tannin and hexamine content, can produce plywood with strength that meets Japanese standards. As shown in Fig. 10, increasing tannin and hexamine content tends to increase plywood’s moisture content (MC), while increasing pressing time decreases the MC value. The control plywood has a lower moisture content than plywood using PTH-based adhesives, which is 5.3%. Based on the JAS No. 233:2003 standard, the maximum moisture content allowed for all MC levels is 14.0%, so the plywood produced is still within the specified standard limits [81].
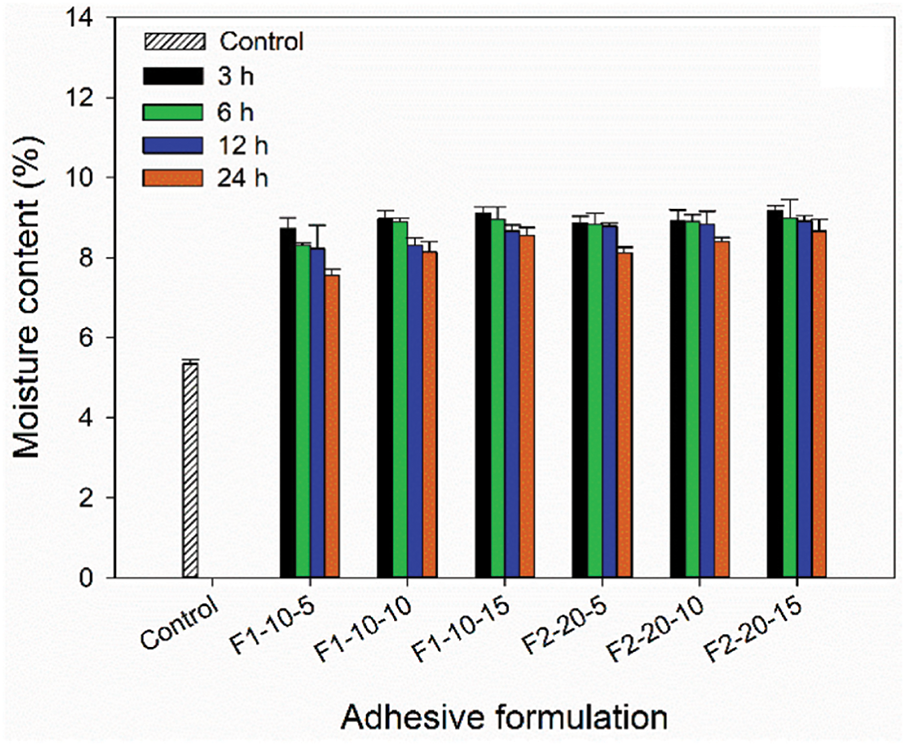
Figure 10: Physical properties of plywood bonded with PVOH–tannin–hexamine-based adhesives: moisture content [80]
The moisture content of plywood using polyurethane adhesive is lower due to the hydrophobic nature of the chemical structure of PU itself. PU is formed from the reaction between polyol and isocyanate, producing a highly cross-linked polymer network that is less able to absorb water than tannin or PVOH-based adhesives that still contain hydrophilic groups. In PU adhesives, most hydroxyl groups (-OH) have reacted to form urethane bonds (-NHCOO-), reducing interactions with water molecules. In addition, polyurethane forms a denser and more uniform adhesive layer between wood fibers, reducing porosity and inhibiting water penetration into the plywood structure. Better dimensional stability is also an advantage of polyurethane adhesives, because low water absorption can prevent swelling and shrinkage due to changes in environmental humidity.
The results of particleboard testing with experimental NIPU adhesives in Table 2 show that all samples meet the European norm requirements for internal bond strength (IB), with values above the minimum limit of 0.35 MPa [75]. Resin A has the highest IB value of 0.70 MPa, likely due to the simultaneous carbonation of tannins and humins, which results in a more extensive urethane bonding in the resin structure. Resin D shows a pretty good IB performance (0.74 MPa), which can be attributed to a more even distribution of urethane bonds between tannins and humins due to the mixing procedure of humin and diamine premix before reaction with tannins. Meanwhile, resin C has a lower IB value (0.65 MPa), which is still above the standard. This can be explained by its preparation procedure, in which tannins are carbonated first before reacting with humins and diamines, causing most humins not to react optimally to form urethane bonds [44]. Resin B had the lowest IB value (0.44 MPa).
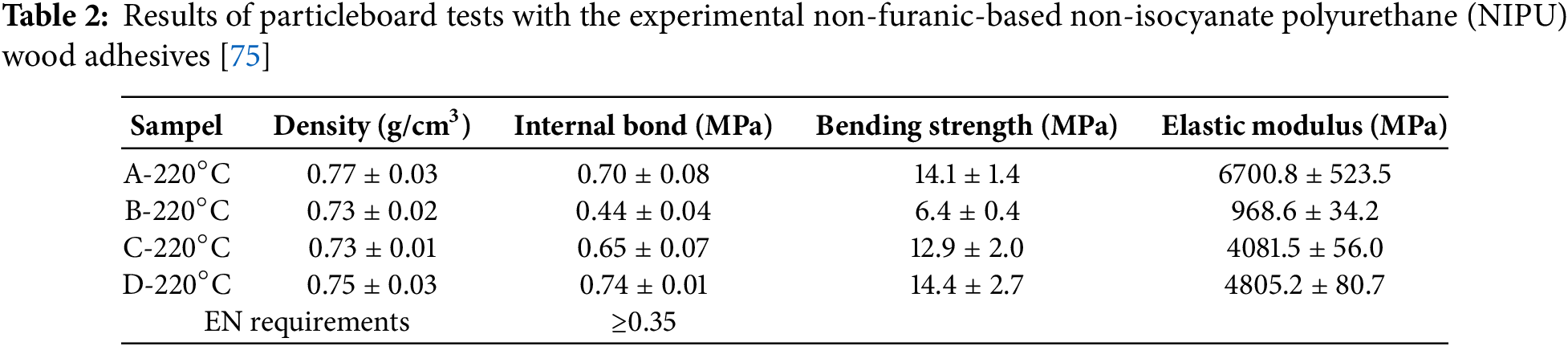
In addition to IB strength, elastic modulus, and bending strength values also showed variations between resins. Resins A and D had higher bending strength values (14.1 and 14.4 MPa), indicating that the simultaneous carbonation method and mixing of humin-diamine premix contributed to the increase in the mechanical strength of the particleboard. In contrast, resin B had the lowest bending strength and elastic modulus values, indicating that humin alone could not provide a strong adhesion structure. Overall, these results confirm that the combination of tannin and humin in the NIPU formulation plays a vital role in improving adhesive performance, with the simultaneous carbonation approach and mixing of premix providing more optimal results than other methods.
6 Challenges and Future Perspectives
PU adhesives are known to have good water resistance and high adhesion performance, but their use still relies on non-renewable petroleum-based compounds [82]. Therefore, more environmentally friendly alternatives are needed, such as biomass-based non-isocyanate polyurethanes (NIPU) [22,83,84]. The use of natural polyols, such as tannins, castor oil, lignin, or humin, is a potential solution to replace fossil-based polyols [78,80,84]. However, one of the main challenges in developing biomass-based NIPUs is to improve the reactivity of the raw materials to form a strong and stable polymer network.
To overcome this challenge, modification of tannins through carbonation with dimethyl carbonate (DMC) has been proposed as a strategy that can increase the reactivity of hydroxyl groups (-OH) in polyols, allowing for more efficient urethane bond formation [59]. In addition, selecting polyols with sufficient hydroxyl groups is essential to ensure optimal reaction with the hardener. With this approach, biomass-based NIPU formulations can be further developed to produce adhesives that have equivalent performance to conventional PU and are more sustainable and environmentally friendly.
While laboratory-scale studies have demonstrated the promising performance of tannin-based NIPU resins, scaling up these materials for industrial applications remains a critical challenge. One of the main obstacles is the variability and limited availability of tannin sources, which may affect the consistency and reactivity of the adhesive formulations. In addition, the synthesis of NIPUs often involves reagents such as dimethyl carbonate (DMC) and multifunctional amines, which, although safer than isocyanates, can increase production costs when used in large quantities. Process parameters such as reaction time, curing temperature, and mixing efficiency also need to be optimized for high-throughput manufacturing without compromising performance. Furthermore, the slower curing rate of some NIPU systems compared to conventional polyurethane adhesives may hinder their direct integration into fast-paced industrial lines. Despite these challenges, the potential for reduced environmental impact, lower VOC emissions, and improved safety makes tannin-based NIPU resins an attractive option for industries aiming to adopt greener materials. Collaborative efforts between academic research and industry, including pilot-scale production trials and techno-economic assessments, are essential to bridge the gap between laboratory innovation and commercial implementation.
Regarding scalability, the biggest challenge is ensuring the NIPU synthesis process can be well integrated into existing adhesive production lines. Current production technology is still mostly tailored for isocyanate-based polyurethanes, so the transition to biomass-based systems requires investment in equipment and modification of manufacturing processes. Therefore, further research is needed to develop more efficient synthesis methods, utilize more economical raw materials, and improve the reactivity of reagents so that NIPU production can be carried out at a more competitive cost and suitable for industrial scale.
The synthesis of tannin-based NIPUs, employing various methods like polyaddition, polycondensation, ring-opening polymerization, and rearrangement, allows fine-tuning of mechanical and thermal properties to meet the specific demands of different industrial applications. Recent studies demonstrate that these bio-based adhesives surpass international internal bonding strength, flexibility, and dimensional stability standards, particularly in oriented strand boards and other wood composite products. Despite these promising results, several challenges remain—most notably, the scalability of production, the cost of raw materials, and the adaptation of existing industrial manufacturing systems.
Tannin-based NIPU resins represent a significant advancement in developing sustainable, eco-friendly adhesives for wood-based composites. These materials offer a safer alternative to conventional polyurethanes, eliminating toxic isocyanates while maintaining comparable or superior mechanical strength, thermal stability, and water resistance. The natural polyphenolic structure of tannins contributes to the resins’ inherent fire resistance, forming a protective char layer during combustion that slows heat transfer and reduces flammability. Additionally, their ability to achieve super-hydrophobic properties through chemical modifications, such as incorporating fluoroalkyl groups or enhancing surface roughness, improves moisture resistance and durability.
Acknowledgement: This study was supported by the Research Program of Research Organization of Nanotechnology and Materials, Fiscal Year 2025, National Research and Innovation Agency, Indonesia. This study was mainly supported by the LPDP-RIIM, Research Grant No. 18/IV/KS/06/2022 and 4830/IT3.L1/PT.01.03/P/B/2022.
Funding Statement: This study was funded by the LPDP-RIIM, Research Grant No. 18/IV/KS/06/2022 and 4830/IT3.L1/PT.01.03/P/B/2022, titled Pengembangan Produk Oriented Strand Board Unggul dari Kayu Ringan dan Cepat Tumbuh dalam Rangka Pengembangan Produk Biokomposit Prospektif.
Author Contributions: The authors confirm their contribution to the paper as follows: study conception and design: Awanda Wira Anggini, Rita Kartika Sari, and Muhammad Adly Rahandi Lubis; data collection: Awanda Wira Anggini, Rita Kartika Sari, and Muhammad Adly Rahandi Lubis; analysis and interpretation of results: Awanda Wira Anggini, Dede Hermawan, and Muhammad Iqbal Maulana; validation: Rita Kartika Sari, Dede Hermawan, and Muhammad Adly Rahandi Lubis; draft manuscript preparation: Awanda Wira Anggini, Rita Kartika Sari, Wahyu Hidayat, Bora Jeong, and Muhammad Adly Rahandi Lubis; review and editing of the manuscript: Dede Hermawan, Muhammad Iqbal Maulana, Wahyu Hidayat, and Bora Jeong. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare that they have no conflicts of interest to report regarding the present study.
References
1. Carbonell-Blasco MP, Moyano MA, Hernández-Fernández C, Sierra-Molero FJ, Pastor IM, Alonso DA, et al. Polyurethane adhesives with chemically debondable properties via Diels–alder bonds. Polymers. 2024;16(1):21. doi:10.3390/polym16010021. [Google Scholar] [PubMed] [CrossRef]
2. Guo G, Liu L, Dang Z, Fang W. Recent progress of polyurethane-based materials for oil/water separation. Nano. 2017;12(4):1730001. doi:10.1142/s1793292017300018. [Google Scholar] [CrossRef]
3. Wang X. Preparation and properties of industrial gelatin wood adhesive modified by polyurethane. Gaofenzi Cailiao Kexue Yu Gongcheng/Polymeric Mater Sci Eng. 2020;36(2):20–6. doi:10.16865/j.cnki.1000-7555.2020.0038. [Google Scholar] [CrossRef]
4. Bockel S, Harling S, Konnerth J, Niemz P, Weiland G, Hogger E, et al. Modifying elastic modulus of two-component polyurethane adhesive for structural hardwood bonding. J Wood Sci. 2020;66(1):69. doi:10.1186/s10086-020-01917-9. [Google Scholar] [CrossRef]
5. Carbonell-Blasco MP, Pérez-Limiñana MA, Ruzafa-Silvestre C, Arán-Ais F, Orgilés-Calpena E. Influence of biobased polyol type on the properties of polyurethane hotmelt adhesives for footwear joints. Appl Adhes Sci. 2021;9(1):8. doi:10.1186/s40563-021-00138-7. [Google Scholar] [CrossRef]
6. Ma C, Tao H, Tan C, Gao S, Wu Z, Guo L, et al. Effects of polyols with different hydroxyl numbers on the structure and properties of starch straws. Carbohydr Polym. 2023;321(2):121297. doi:10.1016/j.carbpol.2023.121297. [Google Scholar] [PubMed] [CrossRef]
7. Peres TLC, Ribeiro FV, Aramburu AB, Barbosa KT, Acosta AP, Missio AL, et al. Polyurethane adhesives for wood based on a simple mixture of Castor oil and crude glycerin. Materials. 2023;16(23):7251. doi:10.3390/ma16237251. [Google Scholar] [PubMed] [CrossRef]
8. Hosseinpourpia R, Eceiza A, Adamopoulos S. Polyurethane wood adhesives prepared from modified polysaccharides. Polymers. 2022;14(3):539. doi:10.3390/polym14030539. [Google Scholar] [PubMed] [CrossRef]
9. Sun M, Bi Y, Zheng M, Wang J, Wang L. Performance of polyurethane mixtures with skeleton-interlocking structure. J Mater Civ Eng. 2020;32(2):04019358. doi:10.1061/(asce)mt.1943-5533.0003015. [Google Scholar] [CrossRef]
10. Ma S, Xiao Y, Zhou F, Schartel B, Chan YY, Korobeinichev OP, et al. Effects of novel phosphorus-nitrogen-containing DOPO derivative salts on mechanical properties, thermal stability and flame retardancy of flexible polyurethane foam. Polym Degrad Stab. 2020;177(86):109160. doi:10.1016/j.polymdegradstab.2020.109160. [Google Scholar] [CrossRef]
11. Fuensanta M, Martín-Martínez JM. Influence of the hard segments content on the structure, viscoelastic and adhesion properties of thermoplastic polyurethane pressure sensitive adhesives. J Adhes Sci Technol. 2020;34(24):2652–71. doi:10.1080/01694243.2020.1780774. [Google Scholar] [CrossRef]
12. Wang T, Deng H, Li N, Xie F, Shi H, Wu M, et al. Mechanically strong non-isocyanate polyurethane thermosets from cyclic carbonate linseed oil. Green Chem. 2022;24(21):8355–66. doi:10.1039/d2gc02910c. [Google Scholar] [CrossRef]
13. Wolfgang JD, White BT, Long TE. Non-isocyanate polyurethanes from 1,1′-carbonyldiimidazole: a polycondensation approach. Macromol Rapid Commun. 2021;42(13):e2100163. doi:10.1002/marc.202100163. [Google Scholar] [PubMed] [CrossRef]
14. Wang P, Liu S, Deng Y. Important green chemistry and catalysis: non-phosgene syntheses of isocyanates—thermal cracking way. Chin J Chem. 2017;35(6):821–35. doi:10.1002/cjoc.201600745. [Google Scholar] [CrossRef]
15. Deng Y, Wang P, Liu S. Green non-phosgene process for manufacturing isocyanates. Sci Sin Chim. 2020;50(2):235–44. [Google Scholar]
16. Ghasemlou M, Daver F, Ivanova EP, Adhikari B. Bio-based routes to synthesize cyclic carbonates and polyamines precursors of non-isocyanate polyurethanes: a review. Eur Polym J. 2019;118(5):668–84. doi:10.1016/j.eurpolymj.2019.06.032. [Google Scholar] [CrossRef]
17. Visser D, Bakhshi H, Rogg K, Fuhrmann E, Wieland F, Schenke-Layland K, et al. Green chemistry for biomimetic materials: synthesis and electrospinning of high-molecular-weight polycarbonate-based nonisocyanate polyurethanes. ACS Omega. 2022;7(44):39772–39781. doi:10.1021/acsomega.2c03731. [Google Scholar] [PubMed] [CrossRef]
18. Rodrigues MBB, Côrrea R, De Cademartori PHG, Ribeiro ACR, Coldebella R, Delucis RA, et al. Bio-based tannin foams: comparing their physical and thermal response to polyurethane foams in lightweight sandwich panels. Compounds. 2024;4(1):1–16. doi:10.3390/compounds4010001. [Google Scholar] [CrossRef]
19. Solis-Pomar F, Díaz-Gómez A, Berrío ME, Ramírez J, Jaramillo AF, Fernández K, et al. A dual active-passive coating with intumescent and fire-retardant properties based on high molecular weight tannins. Coatings. 2021;11(4):460. doi:10.3390/coatings11040460. [Google Scholar] [CrossRef]
20. Zhang W, Wang D, Sun Z, Song J, Deng X. Robust superhydrophobicity: mechanisms and strategies. Chem Soc Rev. 2021;50:4031–61. [Google Scholar] [PubMed]
21. Batool M, Albargi B, Ahmad H, Sarwar A, Khaliq Z, Qadir Z, et al. Nano-silica bubbled structure based durable and flexible superhydrophobic electrospun nanofibrous membrane for extensive functional applications. Nanomaterials. 2023;13(7):1146. doi:10.3390/nano13071146. [Google Scholar] [PubMed] [CrossRef]
22. Sahmim W, Dellarose Boer F, Chapuis H, Obounou-Akong F, Pizzi A, Gérardin P, et al. Feasibility study of the synthesis of isocyanate-free polyurethanes from catechin. J Renew Mater. 2022;10(5):1175–84. doi:10.32604/jrm.2022.016365. [Google Scholar] [CrossRef]
23. Thébault M, Pizzi A, Essawy HA, Barhoum A, Van Assche G. Isocyanate free condensed tannin-based polyurethanes. Eur Polym J. 2015;67(3):513–26. doi:10.1016/j.eurpolymj.2014.10.022. [Google Scholar] [CrossRef]
24. Si C, Cai M, Liu G, Zhang Y, Fan X, Zhu M. PDMS-PI composite coating toward multi-purpose development: hydrophobic, low-friction/wear, and heat-resistance. Tribol Int. 2023;189(1):108919. doi:10.1016/j.triboint.2023.108919. [Google Scholar] [CrossRef]
25. Li W, Yang L, Huang J, Zheng C, Chen Y, Li Y, et al. Progress on fiber engineering for fabric innovation in ecological hydrophobic design and multifunctional applications. Ind Chem Mater. 2024;2(3):393–423. doi:10.1039/d4im00048j. [Google Scholar] [CrossRef]
26. Sfameni S, Rando G, Plutino MR. Sustainable secondary-raw materials, natural substances and eco-friendly nanomaterial-based approaches for improved surface performances: an overview of what they are and how they work. Int J Mol Sci. 2023;24(6):5472. doi:10.3390/ijms24065472. [Google Scholar] [PubMed] [CrossRef]
27. Rokicki G, Parzuchowski PG, Mazurek M. Non-isocyanate polyurethanes: synthesis, properties, and applications. Polym Adv Technol. 2015;26(7):707–61. doi:10.1002/pat.3522. [Google Scholar] [CrossRef]
28. Catalá P, Guerra I, Francisco J. Tailor-made bio-based non-isocyanate polyurethanes (NIPUs). Polymers. 2023;15(6):1589. doi:10.3390/polym15061589. Published online 2023. [Google Scholar] [CrossRef]
29. Yang T, Pizzi A, Xi X, Zhou X, Zhang Q. Preparation and characterization of glucose-based self-blowing non-isocyanate polyurethane (NIPU) foams with different acid catalysts. Polymers. 2024;16(20):2899. doi:10.3390/polym16202899. [Google Scholar] [PubMed] [CrossRef]
30. Maisonneuve L, Lamarzelle O, Rix E, Grau E, Cramail H. Isocyanate-free routes to polyurethanes and poly(hydroxy urethane)s. Chem Rev. 2015;115(22):12407–39. doi:10.1021/acs.chemrev.5b00355. [Google Scholar] [PubMed] [CrossRef]
31. Carré C, Ecochard Y, Caillol S, Avérous L. From the synthesis of biobased cyclic carbonate to polyhydroxyurethanes: a promising route towards renewable non-isocyanate polyurethanes. ChemSusChem. 2019;12(15):3410–30. doi:10.1002/cssc.201900737. [Google Scholar] [PubMed] [CrossRef]
32. Stachak P, Łukaszewska I, Hebda E, Pielichowski K. Recent advances in fabrication of non-isocyanate polyurethane-based composite materials. Materials. 2021;14(13):3497. doi:10.3390/ma14133497. [Google Scholar] [PubMed] [CrossRef]
33. Shu D, Zhang J, Ruan R, Lei H, Wang Y, Moriko Q, et al. Insights into preparation methods and functions of carbon-based solid acids. Molecules. 2024;29(1):247. doi:10.3390/molecules29010247. [Google Scholar] [PubMed] [CrossRef]
34. Shetgaonkar AD. Synthesis, properties and applications of some polycarbonate and polyurethane polymers. Taleigao Plateau, India: Goa University; 2022. [Google Scholar]
35. Wołosz D, Parzuchowski PG, Świderska A. Synthesis and characterization of the non-isocyanate poly(carbonate-urethane)s obtained via polycondensation route. Eur Polym J. 2021;155(7):110574. doi:10.1016/j.eurpolymj.2021.110574. [Google Scholar] [CrossRef]
36. Hosokawa S, Nagao A, Hashimoto Y, Matsune A, Okazoe T, Suzuki C, et al. Non-isocyanate polyurethane synthesis by polycondensation of alkylene and arylene Bis(fluoroalkyl) Bis(carbonate)s with diamines. Bull Chem Soc Jpn. 2023;96(7):663–70. doi:10.1246/bcsj.20230066. [Google Scholar] [CrossRef]
37. Mangal M, Supriya H, Bose S, Banerjee T. Innovations in applications and prospects of non-isocyanate polyurethane bioplastics. Biopolymers. 2023;114(12):e23568. doi:10.1002/bip.23568. [Google Scholar] [PubMed] [CrossRef]
38. Shi J, Zheng T, Zhang Y, Guo B, Xu J. Cross-linked polyurethane with dynamic phenol-carbamate bonds: properties affected by the chemical structure of isocyanate. Polym Chem. 2021;12(16):2421–32. doi:10.1039/d1py00157d. [Google Scholar] [CrossRef]
39. Turnaturi R, Zagni C, Patamia V, Barbera V, Floresta G, Rescifina A. CO2-derived non-isocyanate polyurethanes (NIPUs) and their potential applications. Green Chem. 2023;25(23):9574–602. doi:10.1039/d3gc02796a. [Google Scholar] [CrossRef]
40. Farzan A, Borandeh S, Baniasadi H, Seppälä J. Environmentally friendly polyurethanes based on non-isocyanate synthesis. Express Polym Lett. 2024;18(1):88–101. doi:10.3144/expresspolymlett.2024.7. [Google Scholar] [CrossRef]
41. Lopez de Pariza X, Fanlo P, Polo Fonseca L, Ruiz de Luzuriaga A, Sardon H. Polythiourethanes: synthesis, applications, and opportunities. Prog Polym Sci. 2023;145:101735. doi:10.1016/j.progpolymsci.2023.101735. [Google Scholar] [CrossRef]
42. Mouren A, Avérous L. Sustainable cycloaliphatic polyurethanes: from synthesis to applications. Chem Soc Rev. 2023;52(1):277–317. doi:10.1039/d2cs00509c. [Google Scholar] [PubMed] [CrossRef]
43. Kricheldorf HR, Weidner SM. The ring-opening polymerization-polycondensation (ROPPOC) approach to cyclic polymers. Macromol Rapid Commun. 2020;41(14):e2000152. doi:10.1002/marc.202000152. [Google Scholar] [PubMed] [CrossRef]
44. Iswanto AH, Lubis MAR, Sutiawan J, Al-Edrus SSO, Lee SH, Antov P, et al. Latest advancements in the development of high-performance lignin- and tannin-based non-isocyanate polyurethane adhesive for wood composites. Polymers. 2023;15(19):3864. doi:10.3390/polym15193864. [Google Scholar] [PubMed] [CrossRef]
45. Bobade SK, Paluvai NR, Mohanty S, Nayak SK. Bio-based thermosetting resins for future generation: a review. Polym Plast Technol Eng. 2016;55(17):1863–96. doi:10.1080/03602559.2016.1185624. [Google Scholar] [CrossRef]
46. Yadav N, Seidi F, Crespy D, D’Elia V. Polymers based on cyclic carbonates as trait d’Union between polymer chemistry and sustainable CO2 utilization. ChemSusChem. 2019;12(4):724–54. doi:10.1002/cssc.201802770. [Google Scholar] [PubMed] [CrossRef]
47. Xi X, Pizzi A, Gerardin C, Lei H, Chen X, Amirou S. Preparation and evaluation of glucose based non-isocyanate polyurethane self-blowing rigid foams. Polymers. 2019;11(11):1802. doi:10.3390/polym11111802. [Google Scholar] [PubMed] [CrossRef]
48. Chen X, Li J, Xi X, Pizzi A, Zhou X, Fredon E, et al. Condensed tannin-glucose-based NIPU bio-foams of improved fire retardancy. Polym Degrad Stab. 2020;175(7):109121. doi:10.1016/j.polymdegradstab.2020.109121. [Google Scholar] [CrossRef]
49. Azadeh E, Pizzi A, Gerardin-Charbonnier C, Gerardin P. Hydrolysable chestnut tannin extract chemical complexity in its reactions for non-isocyanate polyurethanes (NIPU) foams. J Renew Mater. 2023;11(6):2823–48. doi:10.32604/jrm.2023.027651. [Google Scholar] [CrossRef]
50. Jayachandran B, Parvin TN, Alam MM, Chanda K, Mm B. Insights on chemical crosslinking strategies for proteins. Molecules. 2022;27(23):8124. doi:10.3390/molecules27238124. [Google Scholar] [PubMed] [CrossRef]
51. Watrelot AA, Norton EL. Chemistry and reactivity of tannins in Vitis spp. a review. Molecules. 2020;25(9):2110. doi:10.3390/molecules25092110. [Google Scholar] [PubMed] [CrossRef]
52. Chen X, Wang X, Wang Q, Cai D, Yu JH, Zhu BW, et al. The controlled two-step release behavior of gallic acid from starch inclusion complexes of short-chain alkyl gallates and their slow starch digestion characteristic. 2024. doi:10.2139/ssrn.4978096. [Google Scholar] [CrossRef]
53. Steverding D, do Nascimento LG, Perez-Castillo Y, de Sousa DP. Gallic acid alkyl esters: trypanocidal and leishmanicidal activity, and target identification via modeling studies. Molecules. 2022;27(18):5876. doi:10.3390/molecules27185876. [Google Scholar] [PubMed] [CrossRef]
54. Santos BAV, Silva VMTM, Loureiro JM, Rodrigues AE. Review for the direct synthesis of dimethyl carbonate. ChemBioEng Rev. 2014;1(5):214–29. doi:10.1002/cben.201400020. [Google Scholar] [CrossRef]
55. Cadenaro M, Maravic T, Comba A, Mazzoni A, Fanfoni L, Hilton T, et al. The role of polymerization in adhesive dentistry. Dent Mater. 2019;35(1):e1–22. doi:10.1016/j.dental.2018.11.012. [Google Scholar] [PubMed] [CrossRef]
56. Ratwani CR, Kamali AR, Abdelkader AM. Self-healing by Diels-Alder cycloaddition in advanced functional polymers: a review. Prog Mater Sci. 2023;131:101001. doi:10.1016/j.pmatsci.2022.101001. [Google Scholar] [CrossRef]
57. Yan Z, Liu W, Gao N, Sun Y, Chen H. Surface properties of the epoxy resin modified by a novel functional fluorinated oligomer. Iran Polym J. 2012;21(10):721–30. doi:10.1007/s13726-012-0074-6. [Google Scholar] [CrossRef]
58. Vitale A, Bongiovanni R, Ameduri B. Fluorinated oligomers and polymers in photopolymerization. Chem Rev. 2015;115(16):8835–66. doi:10.1021/acs.chemrev.5b00120. [Google Scholar] [PubMed] [CrossRef]
59. Kamaruzaman D, Mohamed Mustakim NS, Subki A, Parimon ASR, Yaakob N, Malek MK, et al. Polystyrene Waste-ZnO nanocomposite film for energy harvesting via hydrophobic triboelectric nanogenerator: transforming waste into energy. Mater Today Sustain. 2024;26(15):100726. doi:10.1016/j.mtsust.2024.100726. [Google Scholar] [CrossRef]
60. Chang K, Luo H, Bannon SM, Lin SY, Agata WAS, Geise GM. Methoxy groups increase water and decrease salt permeability properties of sulfonated polysulfone desalination membranes. J Memb Sci. 2021;360:119298. doi:10.1016/j.memsci.2021.119298. [Google Scholar] [CrossRef]
61. León-Campos MI, Claudio-Rizo JA, Cabrera-Munguía DA, Cobos-Puc LE, Caldera-Villalobos M, González-Díaz MO, et al. Development of novel bioactive waterborne polyurethanes: effect of polyurethane chemical structure on its properties. J Polym Res. 2024;31(7):213. doi:10.1007/s10965-024-04055-8. [Google Scholar] [CrossRef]
62. Kumar V, Verma R, Sharma VS, Sharma V. Recent progresses in super-hydrophobicity and micro-texturing for engineering applications. Surf Topogr: Metrol Prop. 2021;9(4):043003. doi:10.1088/2051-672x/ac4321. [Google Scholar] [CrossRef]
63. Ghosh A, Chowdhury SR, Dutta R, Babu R, Rumbo C, Dasgupta N, et al. Polyurethane chemistry for the agricultural applications-recent advancement and future prospects. In: Polyurethanes: preparation, properties, and applications volume 3: emerging applications. Washington, DC, USA: American Chemical Society; 2023. p. 1–36. doi:10.1021/bk-2023-1454.ch001. [Google Scholar] [CrossRef]
64. Zhang C, Yan K, Fu C, Peng H, Hawker CJ, Whittaker AK. Biological utility of fluorinated compounds: from materials design to molecular imaging, therapeutics and environmental remediation. Chem Rev. 2022;122(1):167–208. doi:10.1021/acs.chemrev.1c00632. [Google Scholar] [PubMed] [CrossRef]
65. Bhushan B, Jung YC, Koch K. Micro-, nano- and hierarchical structures for superhydrophobicity, self-cleaning and low adhesion. Philos Trans A Math Phys Eng Sci. 2009;367(1894):1631–72. doi:10.1098/rsta.2009.0014. [Google Scholar] [PubMed] [CrossRef]
66. Wang X, Xu B, Liu Q, Yang Y, Chen Z. Enhancement of vapor condensation heat transfer on the micro- and nano-structured superhydrophobic surfaces. Int J Heat Mass Transf. 2021;177(1):121526. doi:10.1016/j.ijheatmasstransfer.2021.121526. [Google Scholar] [CrossRef]
67. Borrero-López AM, Nicolas V, Marie Z, Celzard A, Fierro V. A review of rigid polymeric cellular foams and their greener tannin-based alternatives. Polymers. 2022;14(19):3974. doi:10.3390/polym14193974. [Google Scholar] [PubMed] [CrossRef]
68. Zhao Y, Zhang Q, Lei H, Zhou X, Du G, Pizzi A, et al. Preparation and fire resistance modification on tannin-based non-isocyanate polyurethane (NIPU) rigid foams. Int J Biol Macromol. 2024;258(Pt 2):128994. doi:10.1016/j.ijbiomac.2023.128994. [Google Scholar] [PubMed] [CrossRef]
69. Bukowczan A, Łukaszewska I, Pielichowski K. Thermal degradation of non-isocyanate polyurethanes. J Therm Anal Calorim. 2024;149(19):10885–99. doi:10.1007/s10973-024-13306-1. [Google Scholar] [CrossRef]
70. Basak S, Raja ASM, Saxena S, Patil PG. Tannin based polyphenolic bio-macromolecules: creating a new era towards sustainable flame retardancy of polymers. Polym Degrad Stab. 2021;189:109603. doi:10.1016/j.polymdegradstab.2021.109603. [Google Scholar] [CrossRef]
71. Aristri MA, Lubis MAR, Yadav SM, Antov P, Papadopoulos AN, Pizzi A, et al. Recent developments in lignin- and tannin-based non-isocyanate polyurethane resins for wood adhesives—a review. Appl Sci. 2021;11(9):4242. doi:10.3390/app11094242. [Google Scholar] [CrossRef]
72. Farkhondehnia M, Maric M. Design of crosslinked networks with hydroxyurethane linkages via bio-based alkyl methacrylates and diamines. J Appl Polym Sci. 2023;140(28):e54039. doi:10.1002/app.54039. [Google Scholar] [CrossRef]
73. Samanta S, Selvakumar S, Bahr J, Wickramaratne DS, Sibi M, Chisholm BJ. Synthesis and characterization of polyurethane networks derived from soybean-oil-based cyclic carbonates and bioderivable diamines. ACS Sustainable Chem Eng. 2016;4(12):6551–61. doi:10.1021/acssuschemeng.6b01409. [Google Scholar] [CrossRef]
74. Bonfatti EAJr, de Barros JMR, Silva GF, Lengowski EC. A comparative analysis of tannin and commercial fire retardants in wood fire protection. Forests. 2024;15(6):951. doi:10.3390/f15060951. [Google Scholar] [CrossRef]
75. Chen X, Pizzi A, Essawy H, Fredon E, Gerardin C, Guigo N, et al. Non-furanic humins-based non-isocyanate polyurethane (NIPU) thermoset wood adhesives. Polymers. 2021;13(3):372. doi:10.3390/polym13030372. [Google Scholar] [PubMed] [CrossRef]
76. Pielichowski K, Njuguna J, Majka TM. Thermal degradation of polymeric materials. Amsterdam, The Netherlands: Elsevier; 2022. [Google Scholar]
77. Wang X, Yang G, Guo H. Tannic acid as biobased flame retardants: a review. J Anal Appl Pyrolysis. 2023;174:106111. doi:10.1016/j.jaap.2023.106111. [Google Scholar] [CrossRef]
78. Neves ACC, Lopes FPD, Simonassi NT, Vieira CMF, Monteiro SN. Properties of Luffa cylindrica mats reinforced castor oil-based polyurethane composite as an alternative for oriented strand board. Polymers. 2022;14(24):5533. doi:10.3390/polym14245533. [Google Scholar] [PubMed] [CrossRef]
79. Sonnenschein MF. Polyurethanes: science, technology, markets, and trends. Hoboken, NJ, USA: John Wiley & Sons, Inc.; 2021. [Google Scholar]
80. Sari RAL, Lubis MAR, Sari RK, Kristak L, Iswanto AH, Mardawati E, et al. Properties of plywood bonded with formaldehyde-free adhesive based on poly(vinyl alcohol)–tannin–hexamine at different formulations and cold-pressing times. J Compos Sci. 2023;7(3):113. doi:10.3390/jcs7030113. [Google Scholar] [CrossRef]
81. Tenorio-Alfonso A, Sánchez MC, Franco JM. A review of the sustainable approaches in the production of bio-based polyurethanes and their applications in the adhesive field. J Polym Environ. 2020;28(3):749–74. doi:10.1007/s10924-020-01659-1. [Google Scholar] [CrossRef]
82. Arias A, González-García S, Feijoo G, Moreira MT. Tannin-based bio-adhesives for the wood panel industry as sustainable alternatives to petrochemical resins. J Ind Ecol. 2022;26(2):627–42. doi:10.1111/jiec.13210. [Google Scholar] [CrossRef]
83. Aristri MA, Sari RK, Lubis MAR, Laksana RPB, Antov P, Iswanto AH, et al. Eco-friendly tannin-based non-isocyanate polyurethane resins for the modification of ramie (Boehmeria nivea L.) fibers. Polymers. 2023;15(6):1492. doi:10.3390/polym15061492. [Google Scholar] [PubMed] [CrossRef]
84. Saražin J, Pizzi A, Amirou S, Schmiedl D, Šernek M. Organosolv lignin for non-isocyanate based polyurethanes (NIPU) as wood adhesive. J Renew Mater. 2021;9(5):881–907. doi:10.32604/jrm.2021.015047. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF
 Downloads
Downloads
 Citation Tools
Citation Tools