 Open Access
Open Access
REVIEW
Understanding the Tumor Microenvironmental Mechanisms Driving Immunotherapy Resistance in Colorectal Cancer Liver Metastases
1 Department of Health Sciences, Università del Piemonte Orientale, Novara, 28100, Italy
2 Laboratory of Hepatobiliary Immunopathology, IRCCS Humanitas Research Hospital, Rozzano, Milan, 20089, Italy
3 Department of Biomedical Sciences, Humanitas University, Pieve Emanuele, Milan, 20072, Italy
4 Division of Internal Medicine and Hepatology, Department of Gastroenterology, IRCCS, Humanitas Research Hospital, Rozzano, Milan, 20089, Italy
5 Division of General Surgery, University Maggiore Hospital della Carità, Novara, 28100, Italy
* Corresponding Author: Matteo Donadon. Email:
(This article belongs to the Special Issue: Targeting the Tumor Microenvironment: Emerging Insights into Cancer Progression and Therapeutics)
Oncology Research 2026, 34(4), 6 https://doi.org/10.32604/or.2025.074093
Received 01 October 2025; Accepted 15 December 2025; Issue published 23 March 2026
Abstract
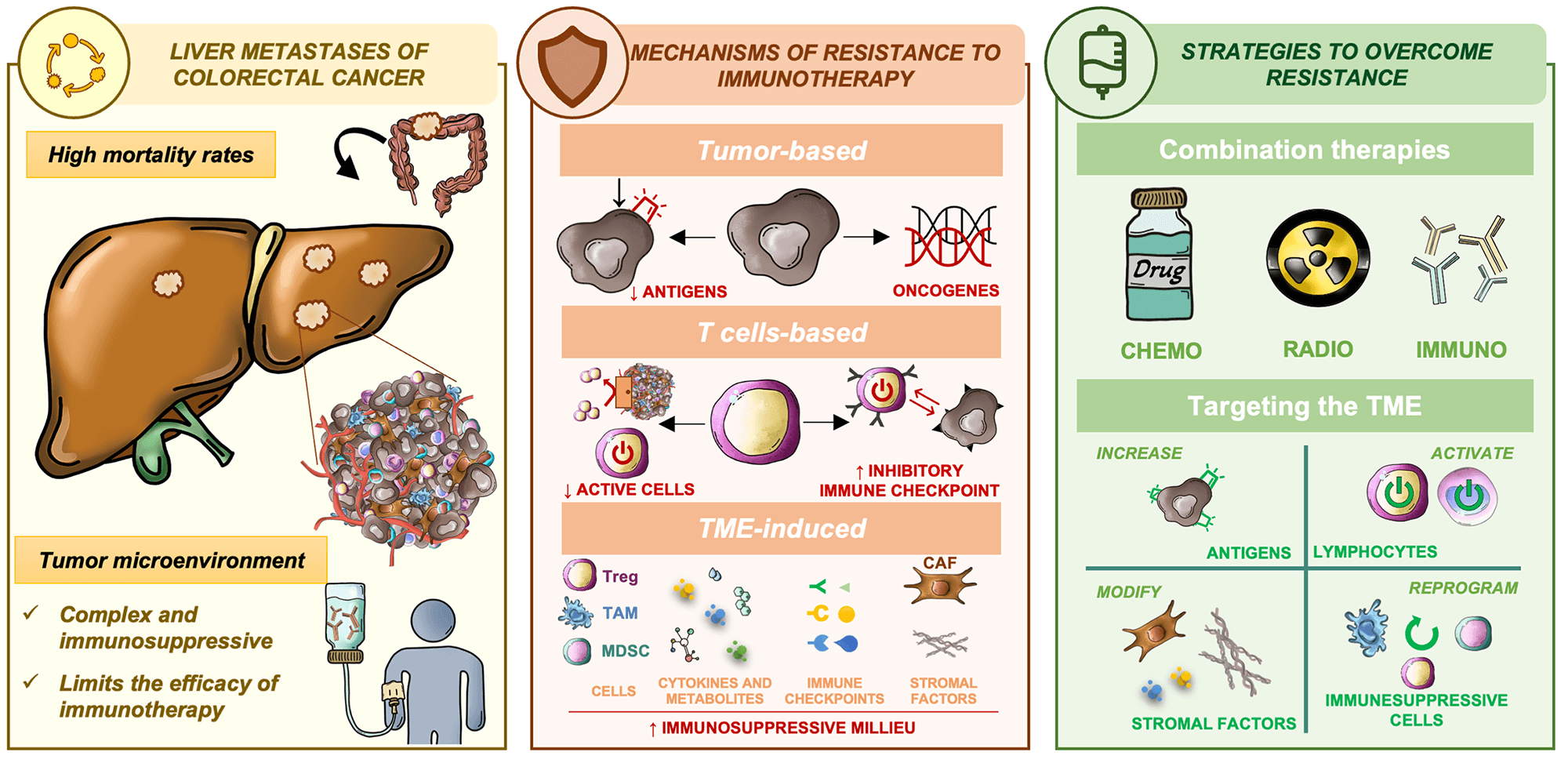
Colorectal cancer (CRC) is the second deadliest cancer worldwide, being the presence of metastasis, mainly in the liver, a major contributor to high mortality rates in affected patients. The tumor microenvironment (TME)—comprised of interacting endothelial, stromal, and immune cells—plays a critical role in creating a supportive niche for tumor cell colonization and immune evasion and, thus, the establishment of metastases. The liver’s intrinsic nature further facilitates the development of immune tolerance, mediated by regulatory T cells, myeloid-derived suppressor cells, and soluble factors such as anti-inflammatory cytokines, which together dampen antitumor immune responses. This immunosuppressive milieu contributes significantly to resistance to immune checkpoint inhibitors, limiting the efficacy of immunotherapy in metastatic CRC. Deciphering the complex crosstalk between metastatic CRC cells and TME within the liver is essential for developing novel, effective immunotherapeutic approaches. Several strategies to overcome this lack of response are under research, including combination therapies, novel compounds, and approaches that target TME components. The scope of this review is to synthesize recent advances in the characterization of the hepatic metastatic microenvironment and emerging therapeutic approaches aimed at overcoming immune resistance in CRC liver metastases.Graphic Abstract
Keywords
Colorectal cancer (CRC) is the third most common cancer worldwide but the second leading cause of cancer-related death [1]. These tumors are highly invasive, as metastases are sometimes found at the time of diagnosis (around 15%–30% of cases) or are lately developed (20%–50%) [2] most commonly in the liver due to its anatomical position, metabolism, and immune microenvironment, but also in the lungs, peritoneum, and distant lymph nodes [3,4]. The occurrence of liver metastases has a severe impact on the patients’ prognosis and quality of life [3].
Surgery is the standard of care for localized CRC, whereas unresectable metastatic CRC (mCRC) requires systemic or localized therapy for disease control. Among the therapeutic options are chemotherapy based on fluoropyrimidines (mainly 5-fluorouracil) in combination with oxaliplatin (FOLFOX) and/or irinotecan (FOLFIRI), therapies targeting either vascular endothelial growth factor (VEGF) pathway (bevacizumab and ramucirumab) or EGFR (cetuximab and panitumumab) [2,5,6], and immunotherapy (pembrolizumab, ipilimumab, or nivolumab) [2,7].
To select the most suitable treatment, it is recommended to test the molecular profile of the tumor, such as mismatch repair (MMR) and microsatellite instability (MSI) status, and mutations of KRAS, NRAS, and BRAF [8]. Indeed, immunotherapy is effective particularly in a subset of patients with deficient MMR and high MSI (12%–20% of locally advanced but only 5% of metastasized tumors [9]), also characterized by high tumor mutational burden (TMB), the consequent formation of neoantigens leading to immune cell recognition [10]. Thus, pembrolizumab is indicated, both in the U.S.A. and Europe, as first-line treatment in these patients, while the combination of nivolumab and ipilimumab is recommended when the tumor progresses after first-line chemotherapy [2,7,11]. Although inhibiting tumor development by stimulating the appropriate immune response through using immune checkpoint inhibitors (ICIs) is a promising treatment strategy, up to 50% of patients with high MSI present with resistance to ICIs [12], while it has shown no benefit in microsatellite stable (MSS) tumors.
Despite the improvement of the survival rates of patients with mCRC in the last two decades, thanks to the identification of molecular traits and great advances in treatment options, the five-year overall survival (OS) remains low (26%) [13], and relapse is frequent (25%–40%) [14]. This showcases its aggressiveness and highlights the need for a more comprehensive understanding of the mechanisms of resistance to enable the development of novel strategies to overcome them.
In fact, growing attention has been devoted to the role of the tumor microenvironment (TME) in shaping patient responses to both chemotherapy and immunotherapy, given its critical influence on tumor growth and progression [15,16]. TME is very intricate as it comprises diverse cellular components, the extracellular matrix (ECM), as well as cytokines and signaling molecules secreted by both tumoral and surrounding cells [17]. Moreover, the complexity of TME increases at metastasis sites, as their characteristics differ from those of the primary tumor. In particular, the one in the liver is prone to minimizing treatment efficacy due to its thicker ECM, certain vasculature design leading to hypoxic areas, and increased evasion of immune surveillance [18–20].
Taking the relevance of this topic into account, the aim of this review is to summarize the available information on the TME of liver-metastasized CRC, its interaction with tumor cells resulting in resistance to immunotherapy, along with new strategies to resolve such a lack of response.
2 Tumor Microenvironment in Colorectal Cancer with Liver Metastasis
Among the different TME cell components are, besides malignant cells, stromal, endothelial, as well as innate and adaptive immune cells. These cells, embedded in dense and highly structured ECM, communicate both through direct cell-to-cell interactions or by indirect signaling mechanisms (release of soluble factors like cytokines and chemokines) (Fig. 1). This complex crosstalk within the liver metastatic niche supports tumor growth, progression, and immune evasion, which undermines the efficacy of anti-tumor immunity and immunotherapeutic strategies [21,22].
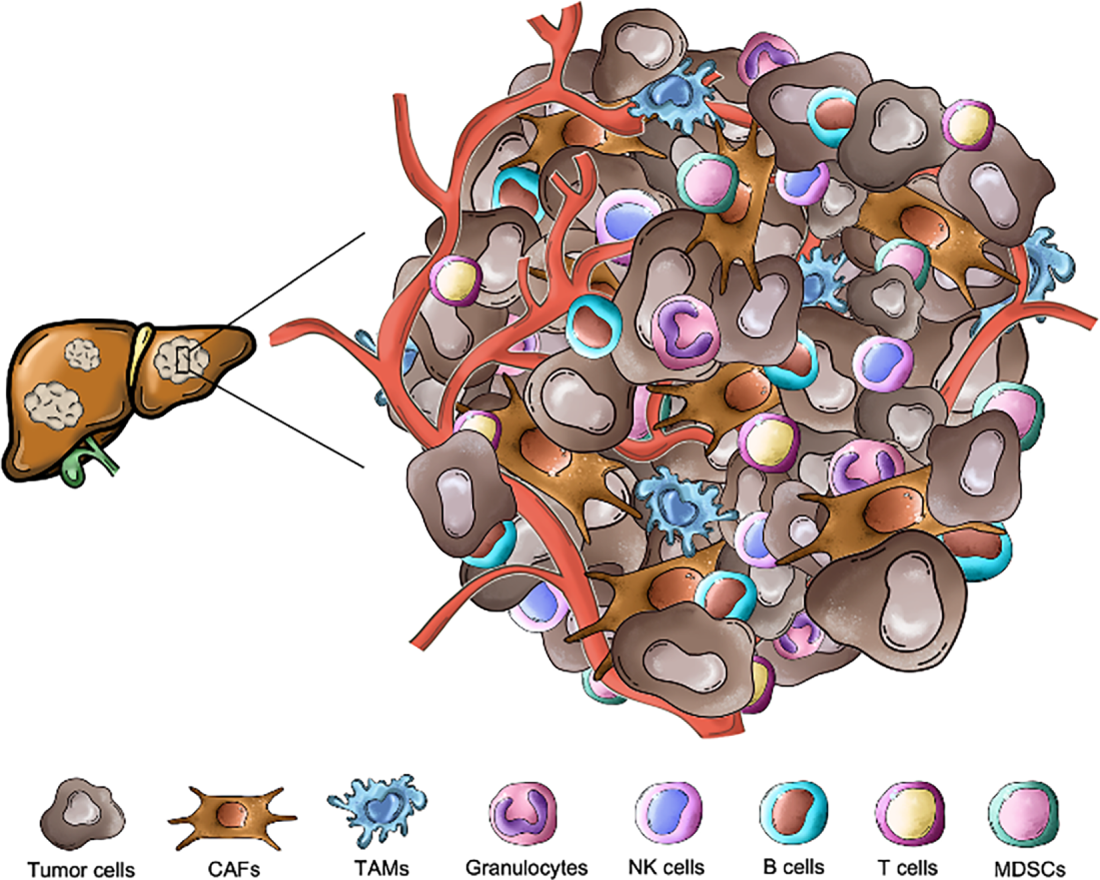
Figure 1: Cellular landscape of the liver metastatic niche in colorectal cancer. The hepatic tumor microenvironment is composed of cancer cells, different immune populations and stromal elements. Key immune subsets include lymphoid cells—T cells, natural killer (NK) cells, B cells and myeloid cells—tumor-associated macrophages (TAMs), myeloid-derived suppressor cells (MDSCs), granulocytes, which exert both pro- and anti-tumor functions. Non-immune stromal components such as cancer-associated fibroblasts (CAFs) and endothelial cells further shape the tolerogenic and immunosuppressive milieu characteristic of colorectal liver metastases. All the pictures present in the figure are original and were hand-drawn
2.1 Cell Types within the Liver Tumor Microenvironment
Stromal cells are actively involved in remodeling the ECM through the production and secretion of components like collagen and fibronectin. Although cancer-associated fibroblasts (CAFs) are the major stromal population, activated hepatic stellate cells (HSCs) are also important contributors to the creation of pro-tumoral immunosuppressive niche in the liver and a great source of CAFs [23,24]. Moreover, they release growth factors and cytokines, such as, CXCL5, TGFβ, and VEGF which favor inflammation, immune escape and angiogenesis, respectively, shaping tumor growth, invasion and metastasis and, thus, the patient’s prognosis [25–27].
Tumor-associated endothelial cells (ECs) play a key role in angiogenesis, however the formed vasculature within the metastatic tissue is abnormal and “leaky”, which affects the amount of nutrients and oxygen that are delivered to tumor cells, contributing to the formation of hypoxic regions. Not only can ECs constitute a barrier for immune cell trafficking, but they can also modulate their activity or metabolism—specially T cells—by expressing certain molecules (PD-L1, FasL...) and/or enzymes, like indoleamine 2,3-dioxygenase 1 (IDO1) or arginase (ARG1) [28].
Another key point in TME is the involvement of the immune system, characterized by many different cell populations among tumor-infiltrating lymphocytes (TILs)—i.e., T cells, B cells, NK cells and myeloid-derived cells—e.g., tumor-associated macrophages (TAMs), myeloid-derived suppressor cells (MDSCs), and dendritic cells (DCs) [29].
Tumor-Infiltrating Lymphocytes (TILs)
Within the T cell compartment, multiple functionally distinct subsets can be distinguished. Cytotoxic T lymphocytes (CD8+) play a central role in mediating anti-tumor immunity, releasing factors like granzyme B, perforin, and interferon γ (IFN-γ) to eliminate tumor cells [30]. In fact, high infiltration was associated with better prognosis in CRC patients [31]. However, these cells are often exhausted due to adverse conditions like high reactive oxygen species (ROS) and long-term antigen exposure in the TME. This dysfunctional state is marked by reduced cytokine production and high expression of immune checkpoints such as PD-1 [10,32]. T helper cells (Th, CD4+) regulate both innate and adaptive immune responses and consist of multiple subsets whose differentiation and function are shaped by the microenvironment cues. Certain subsets support anti-tumor immunity; for example, Th1 cells, which secrete IFN-γ, tumor necrosis factor alpha (TNF-α), and interleukin 2 (IL-2), or cytotoxic CD4+ T cells, which can directly eliminate tumor cells expressing MHC class II [33]. In contrast, subsets such as Th9 (IL-9 producers) and Th17 (IL-17A producers) exhibit context-dependent roles: while they can promote tumor progression by enhancing angiogenesis and suppressing immune responses, they may also facilitate tumor rejection by recruiting and activating cytotoxic immune cells. Th22 cells, characterized by IL-22 expression, contribute to an immunosuppressive microenvironment, partly through the induction of IL-10 production [34]. Regulatory T Cells (Tregs)—characterized by expression of FOXP3 and CD25—suppress immune response by releasing inhibitory factors (IL-10, TGF-β and IL-35) and maintaining immune checkpoints, which reduce cytotoxic T cells activity and inhibit dendritic cells maturation [35]. Their accumulation was associated with poor prognosis in many tumor types, including metastatic CRC [22,36].
B cells are known for presenting antigens and producing antibodies, which can help kill malignant cells and, thus, improve patients’ prognosis. However, a subset called regulatory B (Bregs) cells can enhance angiogenesis, reduce cytotoxic T cell activity by expressing PD-L1 and immunosuppressive cytokines such as IL-10 and TGF-β and, thus, foster immune evasion [17,37].
Natural killer (NK) cells are potent cytotoxic effectors and part of the innate immune system. They can eliminate tumor cells regardless of antigen presentation by producing granzyme B and perforin or facilitating programmed cell death through expression of FasL and TNF-related apoptosis-inducing ligand (TRAIL) [38]. Nonetheless, NK cells can present an exhausted phenotype, characterized by PD-1 expression and impaired function [39].
Myeloid-Derived Cells
TAMs are the most abundant infiltrating immune cells in the TME, often accounting for a substantial proportion—up to 50%—of the total cellular content [40]. Macrophages have elevated plasticity and can adopt diverse phenotypes in response to environmental factors. While M1 phenotype favors inflammation and exhibit anti-tumor activity, M2 supports disease progression due to its immunosuppressive nature. They secrete factors like arginine, IL-10, transforming growth factor-beta (TGF-β) or C-C motif chemokine ligand 5 (CCL5) that inhibit cytotoxic T cell activity and recruit Tregs- and promote ECM remodeling and angiogenesis. In the liver TME, TAMs showed different morphology and localization, which has been correlated with patient prognosis, the poorest outcome in those patients with predominant bigger and pro-tumorigenic M2-like [41–43]. Their presence has also correlated with poor prognosis [22,44,45]. In addition, Kupffer cells, the liver-resident macrophages, can support tumor immune evasion by expressing immune checkpoint molecules which suppress CD8+ T cell effector function [46]. Highly metastatic CRC cells potentiate the polarization of Kupffer cells toward an M2 phenotype, thereby promoting tumor invasion and metastasis [47]. Moreover, tumor-educated Kupffer cells actively contribute to immune tolerance by facilitating Tregs priming [3] and promoting the expansion of MDSCs through the secretion of IL-10 and TGF-β [48–50].
MDSCs are immature myeloid progenitors, often enriched in the metastatic liver, which foster an immunosuppressive environment that supports tumor cell growth and immune escape [51,52]. They can inhibit T and NK cells’ function by direct contact killing and/or depletion of essential amino acids crucial for their viability and they can promote Tregs cell expansion through releasing factors like arginine, ROS and immunosuppressive cytokines [22,53,54].
DCs are specialized antigen-presenting cells (APCs) that capture, process, and present antigens to T cells, thus playing a central role bridging innate and adaptive immunity, orchestrating effective immune surveillance against cancer [55]. Infiltration of mature DCs within the tumor has been correlated with better prognosis in multiple cancer types including CRC [56], however, their function is frequently impaired within the TME of liver metastases, limiting anti-tumor immunity [52].
Granulocytes compile neutrophils, eosinophils, and basophils. Among them, tumor-associated neutrophils (TANs) have attracted more attention in oncology as they can be polarized in response to environmental factors such as TAMs, acquiring pro or anti-tumor activity and influencing different processes like angiogenesis, matrix remodeling, and invasion through signaling molecules [52]. Higher infiltration of TANs was found in the peritumoral area of CRC tumors with mutated KRAS, in comparison with wild-type KRAS [57].
Mast cells (MCs) trigger inflammatory responses through the release of histamine, proteases, cytokines (IL-6, IL-9, IL-13 and TNF) and chemokines (CXCL8, CCL2 and CCL5) when activated. They can modulate angiogenesis, vascular permeability, ECM remodeling and tumor invasion [22]. Indeed, low levels of MCs were associated with better outcomes in CRC patients through decreased vascularity [11].
2.2 Characteristics of the Immune Landscape in Colorectal Cancer with Liver Metastasis
Although genetic heterogeneity is a CRC hallmark, a four-groups consensus molecular subtype (CMS) classification has been developed based on differential gene expressions of tumor and infiltrating cells, thus they also differ in immune TME. CMS1 subtype (14% of cases) is distinguished by hypermutation, high MSI and CpG island methylator phenotype (CIMP). However, the CMS2 “canonical” subtype (37%) and CMS3 “metabolic” subtype (13%) comprise MSS epithelial tumors, but while CMS2 cases are characterized by chromosomal instability, more oncogene copies, with marked WNT and MYC signaling activation, CMS3 are by their metabolic dysregulation and the appearance of KRAS mutations. On the other hand, the CMS4 “mesenchymal” subtype (23% of cases), shows MSS and gene hypermethylation, activation of pathways related to epithelial-mesenchymal transition (EMT) and stemness—including TGF-β—increased stromal invasion, angiogenesis and immunosuppression. Mixed features are shown in 13% cases [58]. Regarding their immune microenvironments, CMS1 tumors have strong immune activation and thus are also known as the “MSI Immune”, with increased T-cell and NK cell infiltration, accompanied by high expression of immune checkpoints. In this context, evaluation of MSI status in both primary CRC tumors and corresponding metastatic sites revealed high concordance in liver ones, although they differed in peritoneal or ovarian metastasis [59]. Alternatively, CMS4 ones are regarded as “immune-inflamed” because, although they also present immune cells infiltration, most are Tregs and Th17 cells and monocyte-derived cells and, which have immunosuppressive functions. Instead, CMS2 and CMS3 are considered as “cold” or “immune desert” tumors, generally PD-L1 negative and with low immune infiltration [60]. Among these subtypes, CMS4 exhibits the worst prognosis, being the TGF-β hyperactivation and the presence of CAFs and ECs important contributors, as they promote invasion, inflammation, angiogenesis and immunosuppression [22,61,62].
The liver’s distinctive anatomical features, including its fenestrated endothelium, and its immune tolerant default state, make it a hospitable niche for metastatic tumor cells’ colonization [3]. Moreover, tumor cell seeding and nutrient delivery are facilitated by the dual blood supply from the portal vein and hepatic artery. Beyond its architecture, the liver inherently presents immunosuppressive microenvironment (evolved to prevent overreaction to gut-derived antigens, to which it is continuously exposed) [63,64] unlike other metastatic sites such as the lung or lymph nodes where immune surveillance is more robust [65]. This immune tolerance is significantly increased in liver metastases, characterized by the presence of Kupffer cells, exhausted CD8+ T cells, Tregs, MDSCs, and TAMs, among other factors. Not only does this contribute to tumor progression but also to drug resistance [22,66], which underscores the need for therapeutic strategies that bypass the liver’s tolerogenic features [67,68]. Besides, the amount of immune infiltrating cells can vary across multiple liver metastases within the same patient [69].
Histological growth patterns (HGPs) of CRLM represent a fundamental determinant of TME interactions and strongly influence immunotherapy sensitivity [70]. CRLM mostly display two overarching HGP categories at the interface of the tumor with the surrounding normal liver: desmoplastic (dHGP) and non-desmoplastic, the latter comprising the replacement (rHGP) and, although less common, pushing (pHGP) subtypes [71]. dHGP lesions form a dense fibrotic frame enriched with CAFs and exhibit robust but aberrant neoangiogenesis, characterized by leaky, dysfunctional vessels with fibrin deposition. This angiogenic phenotype is associated with higher infiltration of immune cells (CD45+ leukocytes, CD8+ T cells, B cells) with an increased CD8+/CD4+ ratio, reflecting a more permissive immune landscape and explaining the comparatively better outcomes reported for patients with dHGP [72–75]. Conversely, rHGP metastases grow by co-opting pre-existing sinusoidal vessels without inducing angiogenesis, maintaining normal hepatic architecture as tumor cells intercalate between hepatocytes whereas pHGP lesions expand by compressing adjacent hepatocytes. This vessel-co-option process, marked by stromal barriers, creates a highly immunosuppressed and immune-excluded niche contributing to poor responsiveness to both anti-angiogenic and ICI-based therapies [75,76].
To further complicate tumor biology, transcriptomic adaptation phenomena have been described, which refers to metastatic tumors gradually adopting expression profiles to resemble their target tissue (in this case, the liver) while retaining traits of their tissue of origin. In CRC, this transition involved changes in pathways related to ECM remodeling and angiogenesis [77].
3 Mechanisms of Immune Resistance
Immunotherapy has emerged as a promising treatment strategy for several solid tumors, including CRC [22]. Its mechanism of action relies on reactivating the patient’s immune system to recognize and eliminate tumor cells by blocking inhibitory pathways that tumors exploit to suppress immune surveillance [78]. Among these pathways, immune checkpoints play a central role. These regulatory molecules, such as programmed cell death protein 1 (PD-1) and cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4), normally function to maintain immune homeostasis and prevent autoimmunity by downregulating T cell activation [12,79]. However, in the TME, these same mechanisms are often hijacked by cancer cells to induce T cell exhaustion and facilitate immune evasion.
PD-1 (CD279), primarily expressed on activated T cells, B cells, and NK cells, is an inhibitory receptor that suppresses T cell proliferation, cytokine secretion, and cytotoxicity upon interaction with its ligands PD-L1 or PD-L2. These ligands are frequently expressed not only by tumor cells but also by immune cells such as macrophages and DCs [80]. In CRC, a higher PD-L1 expression was observed in certain liver metastasis as compared with the primary tumor, this discordance was significantly associated with tumor differentiation [81] and with CD8+ lymphocyte infiltration [65]. Such heterogeneity in PD-L1 expression, which was also reported in other metastasized tumor types [82,83], underscores the complexity of immune evasion in metastatic CRC. The immunosuppressive effect of PD-1 signaling is further enhanced in the presence of TGF-β, which promotes the generation of Tregs, further dampening the antitumor immune response [22].
CTLA-4, another key checkpoint receptor, is constitutively expressed on Tregs and is upregulated on activated T cells. It competes with the co-stimulatory receptor CD28 for binding to B7 ligands (CD80 and CD86) on APCs, but with significantly higher affinity. By outcompeting CD28, CTLA-4 inhibits early T cell activation, limits proliferation, and maintains peripheral tolerance [22].
The most established immunotherapeutic agents are immune checkpoint inhibitors (ICIs), designed to block these inhibitory signals and shift the immune response toward an antitumor phenotype [12]. Monoclonal antibodies such as pembrolizumab and nivolumab target PD-1, preventing its interaction with PD-L1/PD-L2 and restoring T cell activity. Similarly, ipilimumab blocks CTLA-4, enhancing T cell priming and reducing Treg-mediated immunosuppression [22].
Despite the success of ICIs in subsets of patients, particularly those with deficient MMR or high MSI tumors—which are characterized by high neoantigen load and increased immunogenicity—most metastatic CRCs are MSS and do not benefit from these therapies [84]. Nevertheless, a significant proportion of patients still present with intrinsic (“innate” or “primary”) resistance, which constitutes an unresolved and complex issue in immunotherapy [85–87] or develop resistance (“acquired”) during treatment [88,89]. Primary resistance often reflects pre-existing features that prevent effective immune activation, such as defects in antigen presentation, absence of effector T-cell infiltration, or constitutive activation of oncogenic pathways. In contrast, acquired resistance typically emerges under therapeutic pressure, through mechanisms including adaptive secondary mutations that restore immune evasion, such as upregulation of alternative inhibitory receptors which diminish T cell activity [90,91]. From a clinical perspective, distinguishing between these resistance phenotypes is essential: intrinsic resistance demands baseline modulation of the TME to enable immune priming, whereas acquired resistance requires sequential therapeutic strategies and dynamic biomarker monitoring to track treatment response [92–95]. Notably, the presence of liver metastases has been associated with a particularly poor response to ICIs, independent of tumor mutational status [19,96].
Multiple mechanisms have been proposed to explain the limited efficacy of ICIs in liver metastases, including target heterogeneity, tumor-intrinsic alterations, severe T cell exhaustion, and a complex immunosuppressive landscape [87] (Fig. 2). These mechanisms are further exacerbated by features such as stromal fibrosis, hypoxia, and a pro-inflammatory cytokine milieu that collectively foster immune exclusion and dysfunction in metastatic sites [97].
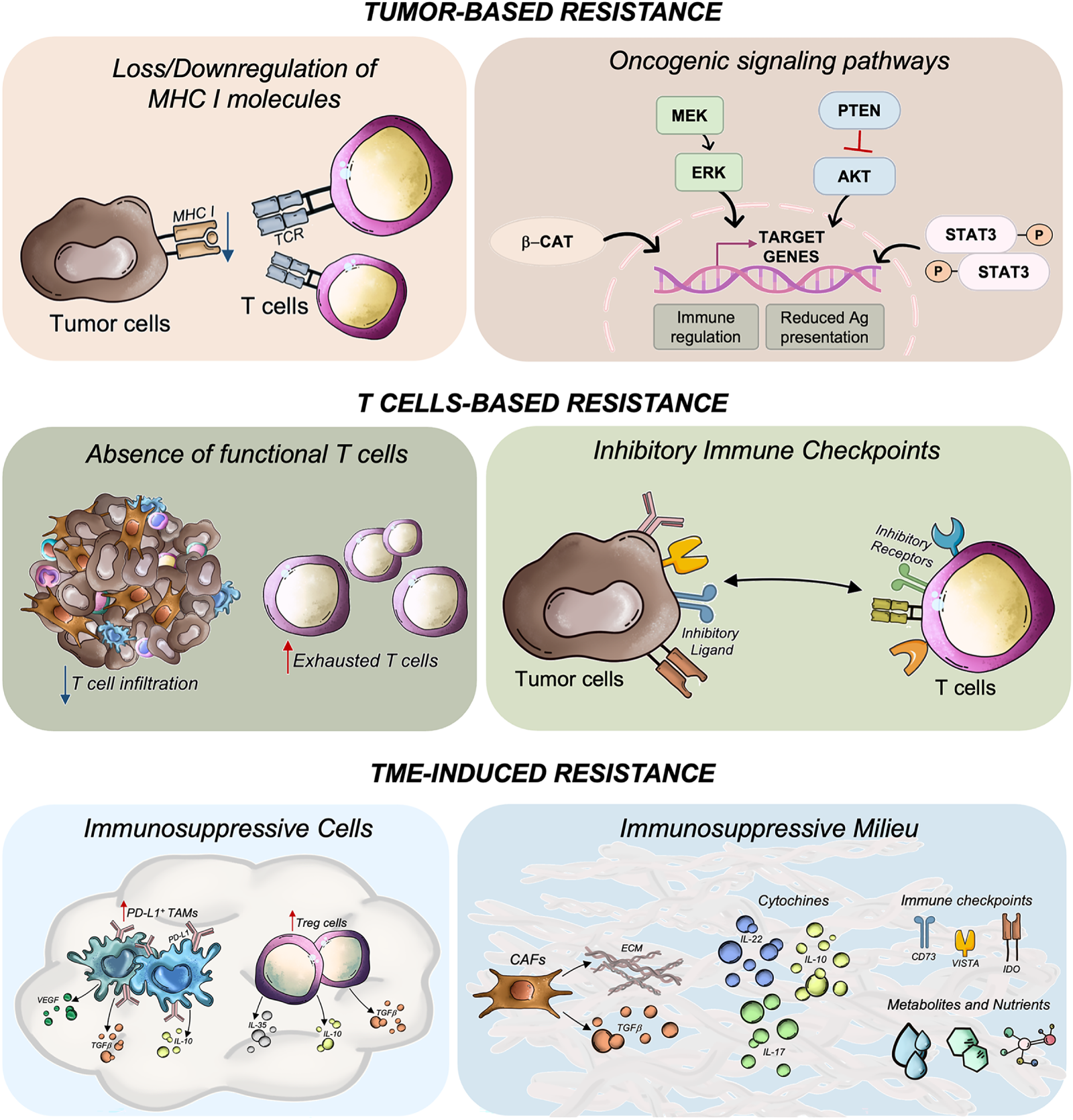
Figure 2: Mechanisms of resistance to immunotherapy in colorectal cancer with liver metastases. Resistance arises from a complex interplay of tumor-intrinsic alterations, dysfunctional T cell responses, and an immunosuppressive tumor microenvironment (TME). Tumor-intrinsic mechanisms include impaired antigen presentation, mutations in oncogenic signaling pathways, and low tumor immunogenicity. T cell-related resistance involves the absence of functional cytotoxic CD8+ T cells due to poor tumor infiltration (immune-cold tumors), accumulation of immunosenescent or exhausted T cells, and upregulation of inhibitory immune checkpoints (i.e., PD-1, CTLA-4, LAG-3, TIGIT), which collectively promote immune tolerance. The TME further contributes through elevated levels of immunosuppressive cells, including regulatory T cells (Tregs) and tumor-associated macrophages (TAMs), which secrete inhibitory cytokines such as TGF-β, IL-10, and IL-17. Cancer-associated fibroblasts (CAFs) also play a key role by releasing immunoregulatory factors and remodeling the extracellular matrix, thereby hindering immune cell infiltration. Additionally, the TME is enriched with immunosuppressive metabolites and nutrient-depleting conditions that impair T cell function, reinforce immune evasion and resistance to immune checkpoint inhibitors. Ag, antigen; AKT, AKT serine/threonine kinase 1; β-cat, beta catenin; CD73, cluster of differentiation 73; ECM, extracellular matrix, ERK, extracellular signal-regulated kinase; IDO, indoleamine 2,3-dioxygenase 1; IL, interleukin; MHC, major histocompatibility complex; MEK, mitogen-activated protein kinase kinase; P, phosphorylated; PD-L1, programmed cell death 1 ligand 1; PTEN, phosphatase and tensin homolog; STAT3, signal transducer and activator of transcription 3; TCR, T-cell receptor; TGF-β, transforming growth factor beta; VEGF, vascular endothelial growth factor; VISTA, V-set immunoregulatory receptor. All the pictures present in the figure are original and were hand-drawn
3.1.1 Absence of Antigenic Proteins and Defective Antigen Presentation
Given the mechanism of action of ICIs, the presence of a sufficiently high TMB along with right antigen presentation is a key determinant of response to PD-1/PD-L1 blockade [98,99].
TMB is typically low (<10 mutations/Mb) in MSS CRC resulting in poor immunogenicity and reduced neoantigen-driven CD8+ T cell activation, which contributes to treatment resistance [100].
Moreover, impaired antigen presentation profoundly impacts immune recognition. Loss or downregulation of MHC class I molecules may occur via β2-microglobulin (β2m) mutations, epigenetic silencing of HLA genes, or deficiencies in antigen-processing components such as TAP1, tapasin, and LMP2/TAP2. For instance, TAP expression is reduced in KRAS-mutated CRC, which compromises peptide presentation to cytotoxic T lymphocytes [80,101]. β2m loss and MHC-I dysfunction can even occur in high MSI CRC with robust T cell infiltration due to immune selection [101].
In the context of liver metastases, antigen presentation may be further impaired by the tolerogenic hepatic microenvironment, which promotes antigen uptake by Kupffer cells and liver sinusoidal endothelial cells, leading to T cell inactivation [80].
3.1.2 Oncogenic Signaling Pathways
Immune resistance can be also mediated by several oncogenic pathways intrinsic to tumor cells, through suppressing T cell infiltration and regulating PD-L1 expression also affected by inflammatory cues.
Aberrant activation of the WNT/β catenin pathway signaling correlates with a reduced CD8+ T cell infiltration in CRC as it decreases the expression of recruiting chemokines such as CCL4 while it increases immune checkpoints like CD47 [102–104]. MAPK pathway activation—secondary to RAS mutations—enhances PD-L1 expression by mRNA stabilization [80]. Moreover, loss of PTEN activates the PI3K/AKT axis, which leads to PD-L1 upregulation and creation of a “cold” TME in multiple tumor types, including CRC [105].
Transcription factors such as STAT3 contribute to the upregulation of PD-L1 on tumor cells, both through intrinsic signaling pathways and in response to external stimuli. Proinflammatory cytokines like IFN-γ and IL-6 enhance this effect via activation of the JAK/STAT1 and STAT3 pathways, respectively. However, defects in IFN-γ signaling, arising from mutations in JAK1/2, IFN-γ receptors, or downstream effectors like IRF1, impair the induction of MHC molecules and antigen presentation machinery, ultimately diminishing the effectiveness of PD-1/PD-L1 blockade [106].
Although over 50% of CRC patients may exhibit tumor PD-L1 positivity (≥10% cutoff), PD-L1 alone remains an unreliable biomarker for predicting ICI response due to challenges such as intratumoral heterogeneity, discordance between primary and metastatic lesions, and a lack of standardized scoring criteria [11,107].
3.2.1 Absence of Functional T-Cells
The reduced presence or dysfunction of T cells within the TME represents a critical factor influencing immunotherapy response. Even when antigens are present, the immune system’s capacity to reach an effective response is hindered by the limited infiltration or senescence of effector T cells [108]. This is particularly challenging in CRC, as MSS tumors typically exhibit a non-inflamed or “cold” TME characterized by low densities of TILs, especially CD8+ cytotoxic T cells, compared to high MSI tumors. Many CD8+ T cells in MSS CRC lack CD28 expression, a critical co-stimulatory molecule, suggesting a senescent-like state with reduced proliferative and cytotoxic capabilities [109].
Additionally, in liver metastases, this T cell dysfunction is exacerbated. Clinical and preclinical studies have shown that the hepatic niche is particularly refractory to ICIs due to reduced marginal CD8+ T cell infiltration, and accumulation of PD-1+ TAMs at the TME of metastases [19]. Notably, metastatic liver disease has been shown to induce systemic deletion or exhaustion of tumor-specific CD8+ T cells, impairing immunotherapy efficacy even at distant tumor sites [96,110,111].
3.2.2 Inhibitory Immune Checkpoints
Besides the immune checkpoints targeted with FDA/EMA-approved ICIs, there are other factors that can cause T-cell exhaustion and modify the patients’ immune response. Indeed, PD-1/PD-L1 blockade efficacy can be undermined in MSS CRC tumors when other inhibitory pathways, including CTLA-4, TIM-3, LAG-3, TIGIT, VISTA, and IDO1, are also activated [80]. While LAG-3, expressed on activated T cells, Tregs, and NK cells, suppresses immune activation by binding MHC class II molecules [112], TIGIT, expressed on T cells and NK cells, binds to CD155 and CD112 on tumor cells or APCs and competes with the co-stimulatory receptor CD226, exerting its inhibitory effect on immune activation [113]. These are frequently simultaneously expressed on exhausted CD8+ and CD4+ T cells, as well as on Tregs, reinforcing a suppressive immune environment. For instance, the presence of LAG-3+ FOXP3+ Tregs in CRC has been associated with poor clinical outcomes, and the co-expression of PD-1 with CTLA-4 or LAG-3 on CD8+ T cells is indicative of deeper functional exhaustion, often refractory to immunotherapy [114,115].
3.3.1 Immunosuppressive Cells: TAMs, MDSCs, Tregs
Liver metastases represent a particularly hostile immune environment, characterized by a unique immunosuppressive milieu which often leads to therapeutic resistance even in tumors with immunogenic features such as high MSI status. Among the challenging factors that promote immune evasion are a marked accumulation of PD L1+ TAMs, expansion of MDSCs, and enrichment of Tregs, working together to create a profoundly immunosuppressive TME that hampers cytotoxic T cell infiltration and function, thereby limiting the efficacy of PD-1/PD-L1 blockade [104].
TAMs in CRC are predominantly inclined toward an M2-like phenotype, while single-cell studies have identified a particularly enriched population of SPP1+ M2-like macrophages, which induce T cell apoptosis through IL-10/STAT3 signaling and Fas/FasL mechanisms and whose presence is associated with poor prognosis [104]. Furthermore, TAMs contribute to immune evasion by secreting immunosuppressive cytokines such as IL-10, TGF-β, and pro-angiogenic factors such as VEGF and by releasing chemokines like MCP 1 and MIP 1α/β that facilitate tumor cell invasion and further immune suppression [80,101,116]. Besides, PD-L1 expression is frequently observed not only in tumor cells but also in surrounding TAMs in liver metastases, contributing to T cell suppression through the PD-1/PD-L1 axis and correlating with resistance to immunotherapy and poorer outcomes [80,117–119]. Interestingly, not only the expression profile but also the morphology of TAMs at the interface of CRC liver metastases correlates with patient survival and has been shown to predict disease recurrence after hepatectomy [42].
MDSCs, particularly polymorphonuclear (PMN-MDSCs) and monocytic (M-MDSCs) subtypes, expand in the metastatic TME, especially under IL 1β–rich conditions [86]. Importantly, this immunosuppressive phenotype is already present in the hepatic pre-metastatic niche and has been clearly associated with tumor progression and invasion. These cells impair T cell function or even deplete them by inducing apoptosis while they also promote Treg differentiation, hindering the antitumor immune response [116,120].
Tregs are highly enriched in CRC compared to healthy tissue and play a critical role in immune evasion. They contribute to immune suppression through various mechanisms. For one, they secrete immunosuppressive cytokines such as IL-10, TGF-β, and IL-35, consume IL-2, depriving effector T cells from this crucial growth factor, and, for another, they can directly induce apoptosis in both APCs and cytotoxic T cells via granzyme B and perforin pathways [116].
3.3.2 Immunosuppressive Cytokines, Metabolites, and Checkpoints in TME
In addition to cellular components, the TME is enriched with soluble factors and metabolites that further hold immune activity back.
Among the most potent of these mediators is TGF-β, secreted by CAFs and TAMs, which promotes Treg induction and drives immune exclusion, particularly in the mesenchymal CMS4 subtype of CRC [104]. IL-10, secreted by both Tregs and TAMs, hampers CD8+ T cell activity by inhibiting T cell receptor signaling and downregulating key effector molecules [116]. IL-17A has been shown to contribute to the creation of a protumor environment by promoting MDSC recruitment and PD-L1 upregulation, while IL-17A blockade could restore sensitivity to PD-1 inhibition in preclinical CRC models by enhancing cytotoxic T cell infiltration [121]. Additionally, elevated serum levels of IL-22 have been associated with chemoresistance in CRC patients, suggesting its potential involvement in broader immune escape mechanisms [34]. Moreover, additional immune checkpoints such as IDO, VISTA, and CD73 can be found within the TME [122], which further reinforces immune resistance.
The presence of specific metabolic byproducts within the TME can also suppress T cell function. In addition to competition with tumor and stromal cells for essential nutrients such as glucose, glutamine, and tryptophan, this metabolic stress results in nutrient-deprived or “starved” T cells, leading to an immunometabolic crisis that impairs their survival and effector functions, including altered cytokine production. For instance, dysregulated glutamine metabolism in TME has been correlated with poor immune infiltration, contributing to immune evasion and resistance to ICIs in CRC tumors [123]. Furthermore, accumulation of lactate and kynurenine further dampens T cell activity [116]. Extracellular adenosine, generated by stimulation of CD39- and CD73-expressing Tregs and TAMs, binds to A2A receptors on T cells and suppresses their function [124,125].
4 Strategies to Overcome Resistance to Immunotherapy
Given the limited efficacy of ICIs in MSS CRC, which represents most metastatic cases, and the relatively small number of patients currently benefiting from these therapies, multiple strategies are being explored to overcome both primary and acquired resistance [126]. One major line of investigation focuses on combination approaches, administrating ICIs alongside other drugs (like targeted therapies), radiotherapy or oncolytic vaccines with the aim to enhance tumor antigenicity and promote immune activation [127]. In parallel, alternative strategies are targeting specific components of the TME aiming to counteract its immunosuppressive characteristics within the liver metastatic niche. Together, these methods seek to convert “cold” (non-inflamed) CRC metastases into “hot” (inflamed) tumors and restore effective T-cell responses [111,126].
Combinatorial regimens are particularly promising in hepatic disease due to the liver’s intrinsic immune tolerance, which often diminishes the effectiveness of ICIs. By simultaneously reducing tumors, remodeling the tumor vasculature, and reactivating immune responses, such multi-target approaches are expected to act synergistically and achieve better therapeutic effects in CRC patients with liver involvement and constitute a promising strategy to overcome ICI resistance [96,111,126,127] (Table 1) (Fig. 3).
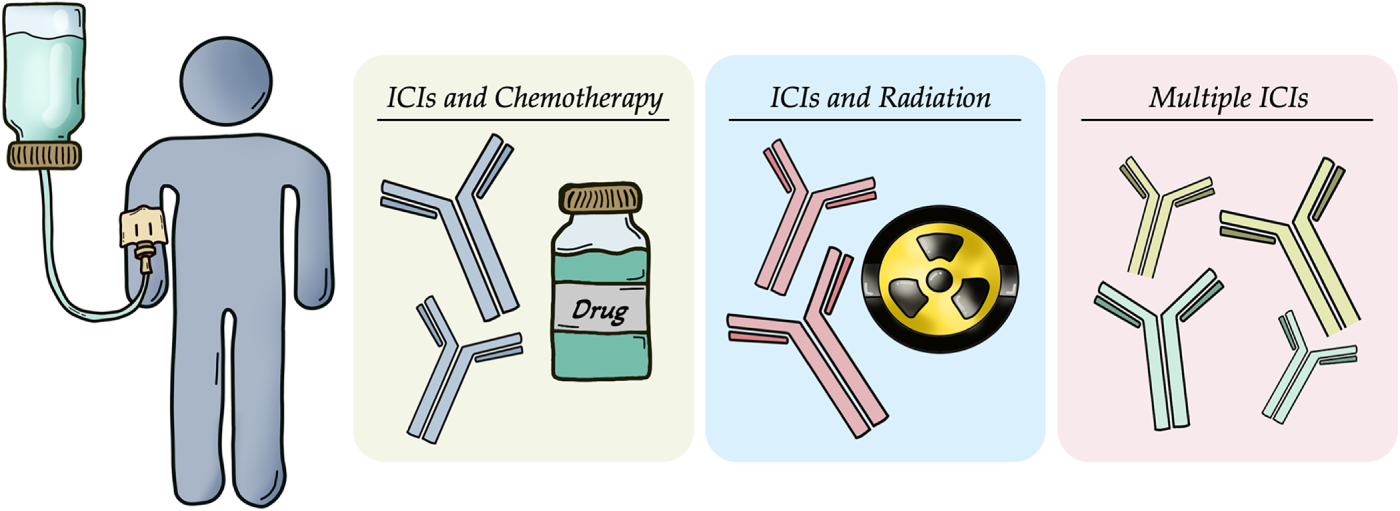
Figure 3: Combination therapies targeting mechanisms of resistance to immunotherapy in colorectal cancer with liver metastases. Approaches include the use of immune checkpoint inhibitors (ICIs) in conjunction with chemotherapeutic agents (cytotoxic and anti-angiogenic drugs), radiotherapy or additional ICIs to enhance antitumor immune responses and overcome immune evasion. All the pictures present in the figure are original and were hand-drawn
Chemotherapy can restore immune surveillance even in immune-depressed tumors by inducing cell death, releasing tumor antigens and danger signals, which enhances DCs activation and T-cell priming [22,128,129]. Indeed, adding ICIs to conventional antitumor drugs (i.e., oxaliplatin) has shown enhanced anti-tumor responses in preclinical models [130] and in the subset of MSS metastasized CRC in randomised METIMMOX phase II trial (NCT03388190) [131]. Similarly, clinical studies have demonstrated the benefit of combining anti-PD-1 therapy with anti-angiogenic such as bevacizumab [132] or tyrosine kinase inhibitors (TKIs) such as regorafenib, as they can normalize tumor vasculature which, in turn, relieves hypoxia and allowing greater T-cell infiltration and a reduction of immunosuppressive populations [126]. Besides, regorafenib has been shown to suppress MDSC and reprogram DCs’ function, sensitizing CRC models to ICIs [126] and CRC patients with advanced MSS CRC without liver metastases in a phase I non-randomized City of Hope Study (NCT04362839) [133]. Notably, the addition of atezolizumab, a PD-L1 inhibitor, to a first-line chemotherapy regimen of FOLFOXIRI with bevacizumab reported survival benefits in both MSS and high MSI CRC in the ongoing AtezoTRIBE trial (NCT03721653), attributed in part to enhanced antigen release and vessel remodeling, allowing greater T-cell infiltration into tumors [126,134,135].
In addition, alternative pathways involved in immune evasion are being investigated as therapeutic targets to improve responses to immune checkpoint inhibitors. For instance, pro-tumorigenic MAPK signaling, commonly activated in CRC, can lead to immune resistance by impairing T cell infiltration and promoting immunosuppressive gene expression. In this context, MEK inhibitors have been used to block MAPK signaling, which in turn preserved CD8+ T cell function and sensitized tumors to PD-L1 blockade in preclinical CRC models [136] and in BRAF-mutated CRC patients within a clinical trial (NCT03668431) [137]. Furthermore, the inhibition of cyclin-dependent kinases 4 and 6 (CDK4/6) with small molecules has proven to increase the response to PD-1 blockade by rising tumor infiltration and activation of effector T cells [138]. Besides, the potential benefit of combining ICIs with inhibitors of immunosuppressive signaling pathways, such as IDO, TGF-β or WNT/β-catenin, is also being explored in ongoing trials to potentiate immune responses and achieve better treatment responses [139,140].
Another relevant mechanism is autophagy, a cellular process essential for maintaining homeostasis, which also shapes the tumor immune microenvironment. Modulation of autophagy has been shown to enhance antigen processing and presentation, leading to improved recognition by cytotoxic T cells. Therefore, pharmacological inhibitors or modulators of autophagy have been combined with anti-PD-1/PD-L1 therapy, showing promising results in both in vitro and in vivo tumor models, including enhanced CD8+ T cell infiltration and tumor regression [141,142].
Liver-directed radiotherapy (RT) can reshape the immunosuppressive liver milieu. Thus, RT combination with ICIs has emerged as a promising strategy to enhance treatment efficacy in liver metastasized CRC [143]. In animal models, liver RT restored systemic antitumor immunity by depleting immunosuppressive TAMs and promoting T cell survival and infiltration when combined with ICIs [111].
The triple combination of RT, anti-PD-1 and anlotinib (anti-angiogenic TKI) was tested in vivo, using a mouse liver metastasis tumor model, to achieve a synergistic effect by acting on different targets: locoregional therapies induce cell death and antigen release, checkpoint blockade reactivates T-cell effector functions and anti-angiogenic agents normalize the vasculature. A significantly higher infiltration of CD4+, CD8+ T cells and less MDSCs were observed in treated mice, which translated into improved disease control. Together, this combination remodeled the liver TME from immunologically “cold” to “hot,” thereby enabling effective antitumor immune responses [144]. Promising results were also observed when combining anlotinib and PD-1 blockers in patients with resistant CRC metastasis [145].
Another triple combination—envafolimab (anti-PD-L1), with short-course RT and CAPEOX-based chemotherapy—was tested on locally advanced rectal cancer within the PRECAM trial (NCT05216653), which resulted in a pathologic complete response rate of 62.5% among MSS patients [126,146].
4.1.3 Blocking Multiple Immune Checkpoints
Targeting more than one immune checkpoint at once could help overcome T-cell exhaustion (i.e., combining anti-PD-1 + anti-CTLA-4 agents) [80]. In the NEST trial (NCT05608044), botensilimab (anti–CTLA-4) and balstilimab (anti–PD-1) achieved durable immunologic responses when used as a neoadjuvant regimen in patients with localized MSS CRC [126]. Furthermore, dual immune checkpoint blockade with durvalumab and tremelimumab (anti-PD-L1 and anti-CTLA-4, respectively) was tested in combination with mFOLFOX6 chemotherapy within the single-arm MEDITREME phase 1b/2 trial (NCT03202758), demonstrating high disease control in RAS-mutated MSS metastatic CRC [129].
Emerging immune checkpoints upregulated in dysfunctional T cells within the TME such as LAG-3, TIGIT, TIM-3, and VISTA are being explored in combination with PD-1/PD-L1 inhibitors aiming to restore effector function and broaden the therapeutic response, particularly in MSS and liver-metastatic CRC, where single-agent immunotherapy has limited efficacy [147–149]. For example, dual PD-1/LAG-3 blockade (pembrolizumab plus favezelimab) has shown synergistic effects by reaching modest clinical responses in MSS CRC within an early-phase clinical trial [149,150]. Similarly, tiragolumab (anti-TIGIT) in combination with atezolizumab (anti-PD-L1) have demonstrated promising efficacy in solid tumors, including high MSI CRC [113,149].
Beyond ICIs’ combination, bispecific antibodies (BsAbs), which bind two different antigens or epitopes, represent an innovative strategy to simultaneously engage immune cells and tumor targets. A PD-L1/CD3 BsAb was designed to activate CD3 on T cells while helping them link to PD-L1+ tumor cells and demonstrating potent antitumor activity in preclinical CRC models. This effect was further enhanced when combined with regorafenib [151,152].
Although combining ICIs with other agents had shown promising response rates, it is also associated with higher frequency and severity of immune-related toxicities compared to single-agent treatments. Therefore, careful patient selection and close monitoring of side effects are critical when implementing these strategies [96,153].
Directly remodeling the TME offers a promising opportunity to improve the efficacy of immunotherapy in liver metastatic CRC, where immunosuppressive signals and structural barriers enable tumors to evade immune surveillance and limit response to current treatments. Among these emerging approaches are enhancing antigen presentation, targeting stromal elements, suppressive cytokines, and immunoregulatory cell populations to disrupt this pro-tumor niche [126] (Table 2).
4.2.1 Enhancing Antigen Presentation
Different therapies to improve antigen presentation and increase tumor “visibility” to the immune system are under investigation, such as restoring MHC-I expression through epigenetic modulation or stimulating antigen-presenting cells [101,116].
Neoantigen-based vaccines could potentially boost tumor recognition and improve survival for patients in immune-cold subtypes. Thus, cancer vaccines and oncolytic viruses are under development to stimulate endogenous DC activation and boost T cell priming [126]. For instance, machine learning was used to identify tumor antigens and immune targets and tailored to immune subtype-specific responses for CRC patients [152].
Other strategies to boost DC function and overcome tolerogenic signaling include in vivo DC activation using GM-CSF or their ex vivo expansion, maturation and reinfusion in the patient [163].
4.2.2 Targeting Matrix and Stromal Factors
The ECM and stromal cells in liver metastases can physically and biochemically exclude immune cells. Hence, strategies that interfere with stromal barriers, such as targeting CAFs or HSCs, or that modulate ECM components (i.e., collagenases), could facilitate T cell infiltration and improve therapeutic efficacy [164]. Interestingly, inhibiting activated HSCs through up-regulating the expression of the relaxin (RLN) resolved liver fibrosis and had anti-metastasis effect with PD-L1 in liver metastasized CRC mouse models [165]. However, a human monoclonal anti-fibroblast activation protein (anti-FAP) antibody showed no clinical activity in CRC patients with liver metastasis within a phase I trial [22].
Hypoxia, common in liver metastases, also drives immunosuppression; novel methods such as manganese-based nanoparticles combined with small-molecule modulators have been proposed to locally increase ROS and provide antitumor treatment and improve the efficacy of immunotherapy [166].
Tumor-derived and stromal cytokines can recruit immunosuppressive cells. Therefore, inhibiting pro-tumor cytokine is a strategy to tilt the balance toward immunity. For instance, preclinical work shows that inhibiting IL-17A or IL-1β can blunt MDSC recruitment and reinvigorate CD8+ T cells in CRC models [80,86].
4.2.3 Activating Innate-Like Lymphocytes
NK cells could be especially useful in immunologically “cold” tumors like MSS CRC due to their ability to recognize “dangerous” cells independently of MHCs and secrete cytokines—such as IFN-γ, IL-4, and TNF-α—which promote cytotoxicity, enhance DC function, and help recruit CD8+ T cells. Strategies using NK cell agonists, α-GalCer–loaded DCs or synthetic glycolipid vaccines are being investigated to activate both NK and CD8+ T cells and synergize with DC-based therapies [163].
Adoptive cell therapies, particularly CAR-T cell approaches, are being adapted to solid tumors like CRC to bypass immune evasion mechanisms and directly engage tumor-specific antigens. In liver metastases of CRC, preclinical studies using CAR-T cells targeting cadherin-17 (CDH17), a cell adhesion molecule highly expressed in CRC but largely absent from normal tissue, have shown potent antitumor activity. Systemically administered CDH17-targeted CAR-T cells significantly reduced tumor burden in humanized mouse models [167]. However, translating CAR-T therapy into CRLM faces unique challenges imposed by the tolerogenic hepatic environment. Liver sinusoids can trap and deactivate CAR-T cells through phagocytosis (mediated by TAMs or Kupffer cell) and immunosuppressive molecules such as IL-10, IDO and adenosine. Moreover, antigen heterogeneity between primary and metastatic lesions predisposes to antigen escape following treatment [168,169]. To overcome these barriers, multiple optimization strategies are under investigation. These include regional delivery approaches, such as hepatic artery infusion to enhance intratumoral CAR-T accumulation; dual-target CAR constructs designed to reduce antigen-escape variants [170]; and “armored” CAR-T cells engineered to resist or remodel the suppressive niche, for example by incorporating TGF-β-resistant signaling domains [171] or by secreting IL-7 and CCL19 to improve local T-cell recruitment and survival [172,173].
4.2.4 Reprogramming Suppressive Immune Cells
Targeting specific immunosuppressive cell populations within TME represents another promising strategy to enhance the efficacy of immune ICIs in metastatic CRC.
Agents such as CSF1R inhibitors can deplete TAMs or reprogram them toward a pro-inflammatory M1 phenotype, restoring antitumor cytokine production such as IFNγ. Additionally, CCR5 blockade, which disrupts the CCL5-CCR5 axis active in both tumor and immune cells, has shown potential to reverse pro-metastatic inflammation and repolarize macrophages in patient-derived CRC models. These findings were supported by early-phase clinical trials demonstrating clinical responses in patients with advanced CRC liver metastases [117,174].
Furthermore, approaches aimed at limiting MDSC recruitment (via CCR2/CCR5 or CXCR2 inhibitors), reprogramming their suppressive phenotype (e.g., with PI3Kγ inhibitors), or depleting them altogether have shown encouraging results in preclinical models [54,175].
Therapeutic strategies to reduce Treg activity or rebalance the Treg/Teffector ratio include TLR agonists, anti-CD25 antibodies, cyclophosphamide, and IDO inhibitors [117].
Despite the promising results of ICIs in the subset of tumors that harbor deficient MMR or high MSI, most CRC patients remain unresponsive to available immunotherapy. Unfortunately, the molecular bases of tumor refractoriness remain poorly understood. This poses a major challenge in advancing immune-based treatments for CRC and has encouraged relevant scientific efforts towards gaining knowledge about said resistance, searching for robust predictive biomarkers (besides MSI status), and developing novel treatment strategies able to overcome it.
Characterizing immune and stromal cell populations present in the TME, and their interactions with tumor cells before and after treatment, can shed some light on how different immunotherapies reshape the immune TME and the counteracting mechanisms of resistance. High-throughput platforms such as transcriptomics, proteomics, and metabolomics can dissect the cellular and molecular composition of the TME, providing insights into immune cell states, stromal dynamics, and signaling pathways. Multiplex methodologies, including multiplex immunohistochemistry/immunofluorescence (mIHC/mIF) and multiplexed imaging, further enhance this analytical capacity by simultaneously detecting and mapping multiple proteins within a single tissue sample. These approaches enable detailed visualization of tumor-immune–stromal interactions, facilitating biomarker discovery, identification of novel drug targets, and improved patient stratification [176–178]. Integrating such complex datasets with artificial intelligence and machine learning holds promise for generating a more comprehensive understanding of the tumor immune landscape and ultimately allowing more precise patient stratification.
The identification of reliable biomarkers that can accurately predict patient response to therapy could significantly enhance clinical decision-making and guide personalized treatment strategies, but remains a challenge in CRC. While TMB has been proposed as a potential predictive marker, its clinical utility is still uncertain. Notably, stromal signatures and cytokine profiles have gained attention due to their role in shaping the immune TME. Circulating tumor DNA (ctDNA) has emerged as a powerful dynamic biomarker in CRLM, offering a non-invasive approach to detect minimal residual disease earlier than conventional imaging, predict recurrence after resection, and quantify treatment response—critical for anticipating timely intervention [179–181]. In addition, ctDNA mutational profiling can reveal newly acquired resistance mechanisms, thereby guiding clinical decision-making and informing alternative therapeutic strategies [182]. In parallel, microRNAs have emerged as promising biomarkers of immune resistance, with specific expression patterns correlating with immune activity and treatment outcomes. Importantly, miRNAs can be detected through liquid biopsy, offering a non-invasive and dynamic approach to monitor the tumor’s immune status in real time. This holds great potential not only for early prediction of immunotherapy response but also for tracking resistance mechanisms during treatment.
Several strategies to overcome the limited response to immunotherapy are currently being investigated. Among them are combination therapies, novel compounds, and approaches specifically designed to target key components of the TME, some being evaluated in clinical trials. A particularly promising path involves multi-targeted approaches that combine ICIs with other therapeutic agents such as chemotherapy, anti-angiogenic agents, radiotherapy, or next-generation immune modulators. These combination strategies are designed to reduce the immunosuppressive milieu, increase tumor immunogenicity, for instance, by facilitating more effective antigen presentation, and enhance the recruitment and activation of effector immune cells. Importantly, the heterogeneous immune landscapes across CMS of CRC suggest that therapeutic approaches should be tailored to subtype-specific biology. CMS1 tumors, characterized by MSI and strong immune activation, are most responsive to ICIs, and combination regimens such as ICI plus anti-angiogenic agents are under investigation. In contrast, CMS4 ones exhibit a fibro-inflammatory milieu dominated by TGF-β signaling and stromal exclusion, where rational strategies include TGF-β blockade and CAF-directed therapies. CMS2 and CMS3 subtypes, generally considered “cold” tumors, may require tumor-intrinsic targeted interventions (for example, EGFR blockade) prior to immune reactivation [29,58,183,184].
Across CRLM-specific trials, meaningful cross-study comparison remains difficult due to heterogeneity in design, patient populations, and endpoints; however, several consistent patterns can be identified. ICI-based combinations with targeted agents yield occasional responses, suggesting partial reversal of hepatic immune tolerance. BsAbss show early clinical activity but are limited by cytokine-associated and hepatotoxic effects. Adoptive cell therapies remain feasible yet face unique barriers in CRLM, including profound immunosuppression within the hepatic niche and risks of off-tumor toxicity. As these trials progress, they hold the potential to broaden the therapeutic reach of immunotherapy beyond the currently responsive deficient MMR/MSI-high subgroup, moving toward more personalized and effective treatment strategies for a larger proportion of CRC patients. Together, these observations underscore the importance of biomarker-guided patient selection and more homogeneous trial frameworks to enable robust evaluation of emerging CRLM-specific immunotherapies.
Despite encouraging preclinical results, the clinical translation of many TME-targeted strategies is still limited in CRLM. Most agents were developed for extrahepatic tumor biology and do not adequately address the liver’s intrinsic immune tolerance. Additionally, spatial heterogeneity between primary CRC tumors and liver metastases complicates biomarker interpretation, and resistance is dynamic and frequently multilayered [185]. These barriers highlight the need for rational, sequential, mechanism-based therapeutic combinations, as moving from experimental models to clinically effective interventions remains one of the central challenges in translational oncology.
In conclusion, overcoming immunotherapy resistance in CRC requires a multifaceted approach that integrates multiple innovative techniques. By increasing our understanding of the immune landscape evolution, personalized immunotherapy to achieve the best possible response rates becomes more reachable.
Acknowledgement: Not applicable.
Funding Statement: This research was funded by the Italian Ministry of Health through the grant “PRIN 2022 PNRR” entitled “Landscape ANalyses of immune CEll signatures associated with early Liver metastatic cOlorecTal cancer” (LANCELOT), P2022N3NC4, which has received funding from European Commission—NextGeneration for the salary of the researcher Candela Cives-Losada.
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization: Candela Cives-Losada, Cristiana Soldani and Matteo Donadon; resources: Candela Cives-Losada and Michela Anna Polidoro; writing—original draft preparation: Candela Cives-Losada and Matteo Donadon; writing—review and editing: Candela Cives-Losada, Cristiana Soldani, Michela Anna Polidoro, Barbara Franceschini, Ana Lleo, Marcello Di Martino and Matteo Donadon; visualization: Candela Cives-Losada, Michela Anna Polidoro and Matteo Donadon; funding acquisition: Matteo Donadon. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
Abbreviations
| APC | Antigen-presenting cell |
| CAF | Cancer-associated fibroblast |
| CMS | Consensus molecular subtype |
| CRC | Colorectal cancer |
| DC | Dendritic cells |
| ECM | Extracellular matrix |
| FOLFIRI | Fluoropyrimidines in combination with irinotecan |
| FOLFOX | Fluoropyrimidines in combination with oxaliplatin |
| HSC | Hepatic stellate cells |
| ICI | Immune checkpoint inhibitor |
| IDO | Indoleamine 2,3-dioxygenase 1 |
| IL | Interleukin |
| mCRC | Metastatic colorectal cancer |
| MDSC | Myeloid-derived suppressor cells |
| MHC | Major histocompatibility complex |
| MMR | Mismatch repair |
| MSI | Microsatellite instability |
| MSS | Microsatellite stable |
| NK | Natural killer |
| ROS | Reactive oxygen species |
| TAM | Tumor-associated macrophage |
| TILs | Tumor-infiltrating lymphocyte |
| TKI | Tyrosine kinase inhibitor |
| TMB | Tumor mutational burden |
| TME | Tumor microenvironment |
| Treg | Regulatory T cell |
| VEGF | Vascular endothelial growth factor |
References
1. Wu S, Zhang Y, Lin Z, Wei M. Global burden of colorectal cancer in 2022 and projections to 2050: incidence and mortality estimates from GLOBOCAN. BMC Cancer. 2025;25(1):1770. doi:10.1186/s12885-025-15138-0. [Google Scholar] [PubMed] [CrossRef]
2. Cervantes A, Adam R, Roselló S, Arnold D, Normanno N, Taïeb J, et al. Metastatic colorectal cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34(1):10–32. doi:10.1016/j.annonc.2022.10.003. [Google Scholar] [PubMed] [CrossRef]
3. Li X, Ramadori P, Pfister D, Seehawer M, Zender L, Heikenwalder M. The immunological and metabolic landscape in primary and metastatic liver cancer. Nat Rev Cancer. 2021;21(9):541–57. doi:10.1038/s41568-021-00383-9. [Google Scholar] [PubMed] [CrossRef]
4. Reboux N, Jooste V, Goungounga J, Robaszkiewicz M, Nousbaum JB, Bouvier AM. Incidence and survival in synchronous and metachronous liver metastases from colorectal cancer. JAMA Netw Open. 2022;5(10):e2236666. doi:10.1001/jamanetworkopen.2022.36666. [Google Scholar] [PubMed] [CrossRef]
5. Vogel JD, Eskicioglu C, Weiser MR, Feingold DL, Steele SR. The American society of colon and rectal surgeons clinical practice guidelines for the treatment of colon cancer. Dis Colon Rectum. 2017;60(10):999–1017. doi:10.1097/DCR.0000000000000926. [Google Scholar] [PubMed] [CrossRef]
6. Inoue Y, Hazama S, Suzuki N, Tokumitsu Y, Kanekiyo S, Tomochika S, et al. Cetuximab strongly enhances immune cell infiltration into liver metastatic sites in colorectal cancer. Cancer Sci. 2017;108(3):455–60. doi:10.1111/cas.13162. [Google Scholar] [PubMed] [CrossRef]
7. Wang Z, Liu Y, Wang K, Ma L. Efficacy and safety of PD-1 and PD-L1 inhibitors in advanced colorectal cancer: a meta-analysis of randomized controlled trials. BMC Gastroenterol. 2024;24(1):461. doi:10.1186/s12876-024-03554-8. [Google Scholar] [PubMed] [CrossRef]
8. Underwood PW, Pawlik TM. Precision medicine for metastatic colorectal cancer: where do we stand? Cancers. 2024;16(22):3870. doi:10.3390/cancers16223870. [Google Scholar] [PubMed] [CrossRef]
9. Battaglin F, Naseem M, Lenz HJ, Salem ME. Microsatellite instability in colorectal cancer: overview of its clinical significance and novel perspectives. Clin Adv Hematol Oncol. 2018;16(11):735–45. [Google Scholar] [PubMed]
10. Kaviyarasan V, Das A, Deka D, Saha B, Banerjee A, Sharma NR, et al. Advancements in immunotherapy for colorectal cancer treatment: a comprehensive review of strategies, challenges, and future prospective. Int J Colorectal Dis. 2024;40(1):1. doi:10.1007/s00384-024-04790-w. [Google Scholar] [PubMed] [CrossRef]
11. Gherman A, Bolundut D, Ecea R, Balacescu L, Curcean S, Dina C, et al. Molecular subtypes, microRNAs and immunotherapy response in metastatic colorectal cancer. Medicina. 2024;60(3):397. doi:10.3390/medicina60030397. [Google Scholar] [PubMed] [CrossRef]
12. Song D, Hou S, Ma N, Yan B, Gao J. Efficacy and safety of PD-1/PD-L1 and CTLA-4 immune checkpoint inhibitors in the treatment of advanced colorectal cancer: a systematic review and meta-analysis. Front Immunol. 2024;15:1485303. doi:10.3389/fimmu.2024.1485303. [Google Scholar] [PubMed] [CrossRef]
13. Zeineddine FA, Zeineddine MA, Yousef A, Gu Y, Chowdhury S, Dasari A, et al. Survival improvement for patients with metastatic colorectal cancer over twenty years. npj Precis Oncol. 2023;7(1):16. doi:10.1038/s41698-023-00353-4. [Google Scholar] [PubMed] [CrossRef]
14. Al Hillan A, Al-Banaa K, Mohamed M, Hawa F, Turtel P. A case of late recurrence of colon cancer after curative treatment. Cureus. 2020;12(5):8083. doi:10.7759/cureus.8083. [Google Scholar] [PubMed] [CrossRef]
15. Halama N, Michel S, Kloor M, Zoernig I, Benner A, Spille A, et al. Localization and density of immune cells in the invasive margin of human colorectal cancer liver metastases are prognostic for response to chemotherapy. Cancer Res. 2011;71(17):5670–7. doi:10.1158/0008-5472.CAN-11-0268. [Google Scholar] [PubMed] [CrossRef]
16. Albini A, Bruno A, Noonan DM, Mortara L. Contribution to tumor angiogenesis from innate immune cells within the tumor microenvironment: implications for immunotherapy. Front Immunol. 2018;9:527. doi:10.3389/fimmu.2018.00527. [Google Scholar] [PubMed] [CrossRef]
17. Lei X, Lei Y, Li JK, Du WX, Li RG, Yang J, et al. Immune cells within the tumor microenvironment: biological functions and roles in cancer immunotherapy. Cancer Lett. 2020;470:126–33. doi:10.1016/j.canlet.2019.11.009. [Google Scholar] [PubMed] [CrossRef]
18. Kubes P, Jenne C. Immune responses in the liver. Annu Rev Immunol. 2018;36:247–77. doi:10.1146/annurev-immunol-051116-052415. [Google Scholar] [PubMed] [CrossRef]
19. Zhou SN, Pan WT, Pan MX, Luo QY, Zhang L, Lin JZ, et al. Comparison of immune microenvironment between colon and liver metastatic tissue in colon cancer patients with liver metastasis. Dig Dis Sci. 2021;66(2):474–82. doi:10.1007/s10620-020-06203-8. [Google Scholar] [PubMed] [CrossRef]
20. Zeng D, Wang M, Wu J, Lin S, Ye Z, Zhou R, et al. Immunosuppressive microenvironment revealed by immune cell landscape in pre-metastatic liver of colorectal cancer. Front Oncol. 2021;11:620688. doi:10.3389/fonc.2021.620688. [Google Scholar] [PubMed] [CrossRef]
21. Peddareddigari VG, Wang D, DuBois RN. The tumor microenvironment in colorectal carcinogenesis. Cancer Microenviron. 2010;3(1):149–66. doi:10.1007/s12307-010-0038-3. [Google Scholar] [PubMed] [CrossRef]
22. Koi M, Carethers JM. The colorectal cancer immune microenvironment and approach to immunotherapies. Future Oncol. 2017;13(18):1633–47. doi:10.2217/fon-2017-0145. [Google Scholar] [PubMed] [CrossRef]
23. Sererols-Viñas L, Garcia-Vicién G, Ruiz-Blázquez P, Lee TF, Lee YA, Gonzalez-Sanchez E, et al. Hepatic stellate cells functional heterogeneity in liver cancer. Semin Liver Dis. 2025;45(1):33–51. doi:10.1055/a-2551-0724. [Google Scholar] [PubMed] [CrossRef]
24. Ezhilarasan D, Najimi M. Deciphering the possible reciprocal loop between hepatic stellate cells and cancer cells in the tumor microenvironment of the liver. Crit Rev Oncol Hematol. 2023;182:103902. doi:10.1016/j.critrevonc.2022.103902. [Google Scholar] [PubMed] [CrossRef]
25. Torres S, Bartolomé RA, Mendes M, Barderas R, Fernandez-Aceñero MJ, Peláez-García A, et al. Proteome profiling of cancer-associated fibroblasts identifies novel proinflammatory signatures and prognostic markers for colorectal cancer. Clin Cancer Res. 2013;19(21):6006–19. doi:10.1158/1078-0432.CCR-13-1130. [Google Scholar] [PubMed] [CrossRef]
26. Nagasaki T, Hara M, Nakanishi H, Takahashi H, Sato M, Takeyama H. Interleukin-6 released by colon cancer-associated fibroblasts is critical for tumour angiogenesis: anti-interleukin-6 receptor antibody suppressed angiogenesis and inhibited tumour-stroma interaction. Br J Cancer. 2014;110(2):469–78. doi:10.1038/bjc.2013.748. [Google Scholar] [PubMed] [CrossRef]
27. Taura K, De Minicis S, Seki E, Hatano E, Iwaisako K, Osterreicher CH, et al. Hepatic stellate cells secrete angiopoietin 1 that induces angiogenesis in liver fibrosis. Gastroenterology. 2008;135(5):1729–38. doi:10.1053/j.gastro.2008.07.065. [Google Scholar] [PubMed] [CrossRef]
28. Leone P, Malerba E, Susca N, Favoino E, Perosa F, Brunori G, et al. Endothelial cells in tumor microenvironment: insights and perspectives. Front Immunol. 2024;15:1367875. doi:10.3389/fimmu.2024.1367875. [Google Scholar] [PubMed] [CrossRef]
29. Binnewies M, Roberts EW, Kersten K, Chan V, Fearon DF, Merad M, et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med. 2018;24(5):541–50. doi:10.1038/s41591-018-0014-x. [Google Scholar] [PubMed] [CrossRef]
30. Koh CH, Lee S, Kwak M, Kim BS, Chung Y. CD8 T-cell subsets: heterogeneity, functions, and therapeutic potential. Exp Mol Med. 2023;55(11):2287–99. doi:10.1038/s12276-023-01105-x. [Google Scholar] [PubMed] [CrossRef]
31. Mlecnik B, Bindea G, Angell HK, Maby P, Angelova M, Tougeron D, et al. Integrative analyses of colorectal cancer show immunoscore is a stronger predictor of patient survival than microsatellite instability. Immunity. 2016;44(3):698–711. doi:10.1016/j.immuni.2016.02.025. [Google Scholar] [PubMed] [CrossRef]
32. Jin X, Zhou K, Zhang R, Li J, Guo M, Qiao H, et al. Construction and validation of prognostic signature for transcription factors regulating T cell exhaustion in hepatocellular carcinoma. Medicine. 2024;103(27):e38713. doi:10.1097/MD.0000000000038713. [Google Scholar] [PubMed] [CrossRef]
33. Thakur A, Schalk D, Sarkar SH, Al-Khadimi Z, Sarkar FH, Lum LG. A Th1 cytokine-enriched microenvironment enhances tumor killing by activated T cells armed with bispecific antibodies and inhibits the development of myeloid-derived suppressor cells. Cancer Immunol Immunother. 2012;61(4):497–509. doi:10.1007/s00262-011-1116-1. [Google Scholar] [PubMed] [CrossRef]
34. Cui G. TH9, TH17, and TH22 cell subsets and their main cytokine products in the pathogenesis of colorectal cancer. Front Oncol. 2019;9:1002. doi:10.3389/fonc.2019.01002. [Google Scholar] [PubMed] [CrossRef]
35. Li C, Jiang P, Wei S, Xu X, Wang J. Regulatory T cells in tumor microenvironment: new mechanisms, potential therapeutic strategies and future prospects. Mol Cancer. 2020;19(1):116. doi:10.1186/s12943-020-01234-1. [Google Scholar] [PubMed] [CrossRef]
36. Betts G, Jones E, Junaid S, El-Shanawany T, Scurr M, Mizen P, et al. Suppression of tumour-specific CD4+ T cells by regulatory T cells is associated with progression of human colorectal cancer. Gut. 2012;61(8):1163–71. doi:10.1136/gutjnl-2011-300970. [Google Scholar] [PubMed] [CrossRef]
37. Sarvaria A, Madrigal JA, Saudemont A. B cell regulation in cancer and anti-tumor immunity. Cell Mol Immunol. 2017;14(8):662–74. doi:10.1038/cmi.2017.35. [Google Scholar] [PubMed] [CrossRef]
38. Coënon L, Geindreau M, Ghiringhelli F, Villalba M, Bruchard M. Natural Killer cells at the frontline in the fight against cancer. Cell Death Dis. 2024;15(8):614. doi:10.1038/s41419-024-06976-0. [Google Scholar] [PubMed] [CrossRef]
39. Xu C, Cao K, Ma A, Zheng M, Xu Y, Tang L. KLRG1 expression induces functional exhaustion of NK cells in colorectal cancer patients. Cancer Immunol Immunother. 2025;74(7):203. doi:10.1007/s00262-025-04059-3. [Google Scholar] [PubMed] [CrossRef]
40. Coffelt SB, Hughes R, Lewis CE. Tumor-associated macrophages: effectors of angiogenesis and tumor progression. Biochim Biophys Acta. 2009;1796(1):11–8. doi:10.1016/j.bbcan.2009.02.004. [Google Scholar] [PubMed] [CrossRef]
41. Donadon M, Torzilli G, Cortese N, Soldani C, Di Tommaso L, Franceschini B, et al. Macrophage morphology correlates with single-cell diversity and prognosis in colorectal liver metastasis. J Exp Med. 2020;217(11):e20191847. doi:10.1084/jem.20191847. [Google Scholar] [PubMed] [CrossRef]
42. Costa G, Sposito C, Soldani C, Polidoro MA, Franceschini B, Marchesi F, et al. Macrophage morphology and distribution are strong predictors of prognosis in resected colorectal liver metastases: results from an external retrospective observational study. Int J Surg. 2023;109(5):1311–7. doi:10.1097/js9.0000000000000374. [Google Scholar] [PubMed] [CrossRef]
43. Soldani C, De Simone G, Polidoro MA, Morabito A, Franceschini B, Colombo FS, et al. Riboflavin-LSD1 axis participates in the in vivo tumor-associated macrophage morphology in human colorectal liver metastases. Cancer Immunol Immunother. 2024;73(4):63. doi:10.1007/s00262-024-03645-1. [Google Scholar] [PubMed] [CrossRef]
44. Gazzillo A, Polidoro MA, Soldani C, Franceschini B, Lleo A, Donadon M. Relationship between epithelial-to-mesenchymal transition and tumor-associated macrophages in colorectal liver metastases. Int J Mol Sci. 2022;23(24):16197. doi:10.3390/ijms232416197. [Google Scholar] [PubMed] [CrossRef]
45. Marzano P, Soldani C, Cazzetta V, Franceschini B, Terzoli S, Carletti A, et al. Tissue-specific immunosuppressive and proliferating macrophages fuel early metastatic progression of human colorectal cancer to the liver. Cancer Immunol Res. 2025;13(11):1783–97. doi:10.1158/2326-6066.CIR-25-0031. [Google Scholar] [PubMed] [CrossRef]
46. Wu K, Kryczek I, Chen L, Zou W, Welling TH. Kupffer cell suppression of CD8+ T cells in human hepatocellular carcinoma is mediated by B7-H1/programmed death-1 interactions. Cancer Res. 2009;69(20):8067–75. doi:10.1158/0008-5472.CAN-09-0901. [Google Scholar] [PubMed] [CrossRef]
47. Tang D, Wang H, Deng W, Wang J, Shen D, Wang L, et al. Mechanism of bufalin inhibition of colon cancer liver metastasis by regulating M2-type polarization of Kupffer cells induced by highly metastatic colon cancer cells. Apoptosis. 2024;29(5–6):635–48. doi:10.1007/s10495-023-01930-5. [Google Scholar] [PubMed] [CrossRef]
48. Heymann F, Peusquens J, Ludwig-Portugall I, Kohlhepp M, Can E, Niemietz P, et al. Liver inflammation abrogates immunological tolerance induced by Kupffer cells. Hepatology. 2015;62(1):279–91. doi:10.1002/hep.27793. [Google Scholar] [PubMed] [CrossRef]
49. Knolle P, Schlaak J, Uhrig A, Kempf P, Meyer zum Büschenfelde KH, Gerken G. Human Kupffer cells secrete IL-10 in response to lipopolysaccharide (LPS) challenge. J Hepatol. 1995;22(2):226–9. doi:10.1016/0168-8278(95)80433-1. [Google Scholar] [PubMed] [CrossRef]
50. Breous E, Somanathan S, Vandenberghe LH, Wilson JM. Hepatic regulatory T cells and Kupffer cells are crucial mediators of systemic T cell tolerance to antigens targeting murine liver. Hepatology. 2009;50(2):612–21. doi:10.1002/hep.23043. [Google Scholar] [PubMed] [CrossRef]
51. Sieminska I, Baran J. Myeloid-derived suppressor cells in colorectal cancer. Front Immunol. 2020;11:1526. doi:10.3389/fimmu.2020.01526. [Google Scholar] [PubMed] [CrossRef]
52. Wang Y, Zhong X, He X, Hu Z, Huang H, Chen J, et al. Liver metastasis from colorectal cancer: pathogenetic development, immune landscape of the tumour microenvironment and therapeutic approaches. J Exp Clin Cancer Res. 2023;42(1):177. doi:10.1186/s13046-023-02729-7. [Google Scholar] [PubMed] [CrossRef]
53. Yang Y, Li C, Liu T, Dai X, Bazhin AV. Myeloid-derived suppressor cells in tumors: from mechanisms to antigen specificity and microenvironmental regulation. Front Immunol. 2020;11:1371. doi:10.3389/fimmu.2020.01371. [Google Scholar] [PubMed] [CrossRef]
54. Zeng W, Liu H, Mao Y, Jiang S, Yi H, Zhang Z, et al. Myeloid-derived suppressor cells: key immunosuppressive regulators and therapeutic targets in colorectal cancer (Review). Int J Oncol. 2024;65(3):85. doi:10.3892/ijo.2024.5673. [Google Scholar] [PubMed] [CrossRef]
55. Legitimo A, Consolini R, Failli A, Orsini G, Spisni R. Dendritic cell defects in the colorectal cancer. Hum Vaccines Immunother. 2014;10(11):3224–35. doi:10.4161/hv.29857. [Google Scholar] [PubMed] [CrossRef]
56. Verneau J, Sautés-Fridman C, Sun CM. Dendritic cells in the tumor microenvironment: prognostic and theranostic impact. Semin Immunol. 2020;48:101410. doi:10.1016/j.smim.2020.101410. [Google Scholar] [PubMed] [CrossRef]
57. Polidoro MA, Milana F, Soldani C, Franceschini B, Anselmo A, Colombo FS, et al. Impact of RAS mutations on the immune infiltrate of colorectal liver metastases: a preliminary study. J Leukoc Biol. 2020;108(2):715–21. doi:10.1002/JLB.5AB0220-608R. [Google Scholar] [PubMed] [CrossRef]
58. Guinney J, Dienstmann R, Wang X, de Reyniès A, Schlicker A, Soneson C, et al. The consensus molecular subtypes of colorectal cancer. Nat Med. 2015;21(11):1350–6. doi:10.1038/nm.3967. [Google Scholar] [PubMed] [CrossRef]
59. He WZ, Hu WM, Wang F, Rong YM, Yang L, Xie QK, et al. Comparison of mismatch repair status between primary and matched metastatic sites in patients with colorectal cancer. J Natl Compr Canc Netw. 2019;17(10):1174–83. doi:10.6004/jnccn.2019.7308. [Google Scholar] [PubMed] [CrossRef]
60. Becht E, de Reyniès A, Giraldo NA, Pilati C, Buttard B, Lacroix L, et al. Immune and stromal classification of colorectal cancer is associated with molecular subtypes and relevant for precision immunotherapy. Clin Cancer Res. 2016;22(16):4057–66. doi:10.1158/1078-0432.ccr-15-2879. [Google Scholar] [PubMed] [CrossRef]
61. Calon A, Lonardo E, Berenguer-Llergo A, Espinet E, Hernando-Momblona X, Iglesias M, et al. Stromal gene expression defines poor-prognosis subtypes in colorectal cancer. Nat Genet. 2015;47(4):320–9. doi:10.1038/ng.3225. [Google Scholar] [PubMed] [CrossRef]
62. Høye E, Kanduri C, Torgunrud A, Lorenz S, Edwin B, Larsen SG, et al. Enrichment of cancer-associated fibroblasts, macrophages, and up-regulated TNF-α signaling in the tumor microenvironment of CMS4 colorectal peritoneal metastasis. Cancer Med. 2025;14(1):e70521. doi:10.1002/cam4.70521. [Google Scholar] [PubMed] [CrossRef]
63. Jenne CN, Kubes P. Immune surveillance by the liver. Nat Immunol. 2013;14(10):996–1006. doi:10.1038/ni.2691. [Google Scholar] [PubMed] [CrossRef]
64. Zheng M, Tian Z. Liver-mediated adaptive immune tolerance. Front Immunol. 2019;10:2525. doi:10.3389/fimmu.2019.02525. [Google Scholar] [PubMed] [CrossRef]
65. Xin H, Zhou C, Wang G, Liu Y, Zhang J, Liu Y, et al. Heterogeneity of PD-L1 expression and CD8 lymphocyte infiltration in metastatic colorectal cancer and their prognostic significance. Heliyon. 2023;9(2):e13048. doi:10.1016/j.heliyon.2023.e13048. [Google Scholar] [PubMed] [CrossRef]
66. He Y, Han Y, Fan AH, Li D, Wang B, Ji K, et al. Multi-perspective comparison of the immune microenvironment of primary colorectal cancer and liver metastases. J Transl Med. 2022;20(1):454. doi:10.1186/s12967-022-03667-2. [Google Scholar] [PubMed] [CrossRef]
67. Milette S, Sicklick JK, Lowy AM, Brodt P. Molecular pathways: targeting the microenvironment of liver metastases. Clin Cancer Res. 2017;23(21):6390–9. doi:10.1158/1078-0432.ccr-15-1636. [Google Scholar] [PubMed] [CrossRef]
68. Burga RA, Thorn M, Point GR, Guha P, Nguyen CT, Licata LA, et al. Liver myeloid-derived suppressor cells expand in response to liver metastases in mice and inhibit the anti-tumor efficacy of anti-CEA CAR-T. Cancer Immunol Immunother. 2015;64(7):817–29. doi:10.1007/s00262-015-1692-6. [Google Scholar] [PubMed] [CrossRef]
69. Halama N, Spille A, Lerchl T, Brand K, Herpel E, Welte S, et al. Hepatic metastases of colorectal cancer are rather homogeneous but differ from primary lesions in terms of immune cell infiltration. Oncoimmunology. 2013;2(4):e24116. doi:10.4161/onci.24116. [Google Scholar] [PubMed] [CrossRef]
70. Garcia-Vicién G, Mezheyeuski A, Bañuls M, Ruiz-Roig N, Molleví DG. The tumor microenvironment in liver metastases from colorectal carcinoma in the context of the histologic growth patterns. Int J Mol Sci. 2021;22(4):1544. doi:10.3390/ijms22041544. [Google Scholar] [PubMed] [CrossRef]
71. Vermeulen PB, Colpaert C, Salgado R, Royers R, Hellemans H, Van Den Heuvel E, et al. Liver metastases from colorectal adenocarcinomas grow in three patterns with different angiogenesis and desmoplasia. J Pathol. 2001;195(3):336–42. doi:10.1002/path.966. [Google Scholar] [PubMed] [CrossRef]
72. Van den Eynden GG, Bird NC, Majeed AW, Van Laere S, Dirix LY, Vermeulen PB. The histological growth pattern of colorectal cancer liver metastases has prognostic value. Clin Exp Metastasis. 2012;29(6):541–9. doi:10.1007/s10585-012-9469-1. [Google Scholar] [PubMed] [CrossRef]
73. Van Dam PJ, Van der Stok EP, Teuwen LA, Van den Eynden GG, Illemann M, Frentzas S, et al. International consensus guidelines for scoring the histopathological growth patterns of liver metastasis. Br J Cancer. 2017;117(10):1427–41. doi:10.1038/bjc.2017.334. [Google Scholar] [PubMed] [CrossRef]
74. Stremitzer S, Vermeulen P, Graver S, Kockx M, Dirix L, Yang D, et al. Immune phenotype and histopathological growth pattern in patients with colorectal liver metastases. Br J Cancer. 2020;122(10):1518–24. doi:10.1038/s41416-020-0812-z. [Google Scholar] [PubMed] [CrossRef]
75. Garcia-Vicién G, Ruiz N, Micke P, Ruffinelli JC, Mils K, Bañuls M, et al. The histological growth patterns in liver metastases from colorectal cancer display differences in lymphoid, myeloid, and mesenchymal cells. MedComm. 2024;5(12):e70000. doi:10.1002/mco2.70000. [Google Scholar] [PubMed] [CrossRef]
76. Kong BT, Fan QS, Wang XM, Zhang Q, Zhang GL. Clinical implications and mechanism of histopathological growth pattern in colorectal cancer liver metastases. World J Gastroenterol. 2022;28(26):3101–15. doi:10.3748/wjg.v28.i26.3101. [Google Scholar] [PubMed] [CrossRef]
77. Sanghvi N, Calvo-Alcañiz C, Rajagopal PS, Scalera S, Canu V, Sinha S, et al. Charting the transcriptomic landscape of primary and metastatic cancers in relation to their origin and target normal tissues. Sci Adv. 2024;10(49):eadn0220. doi:10.1126/sciadv.adn0220. [Google Scholar] [PubMed] [CrossRef]
78. Garg P, Pareek S, Kulkarni P, Horne D, Salgia R, Singhal SS. Next-generation immunotherapy: advancing clinical applications in cancer treatment. J Clin Med. 2024;13(21):6537. doi:10.3390/jcm13216537. [Google Scholar] [PubMed] [CrossRef]
79. Zhang Y, Zheng J. Functions of immune checkpoint molecules beyond immune evasion. Adv Exp Med Biol. 2020;1248:201–26. doi:10.1007/978-981-15-3266-5_9. [Google Scholar] [PubMed] [CrossRef]
80. Xie Q, Liu X, Liu R, Pan J, Liang J. Cellular mechanisms of combining innate immunity activation with PD-1/PD-L1 blockade in treatment of colorectal cancer. Mol Cancer. 2024;23(1):252. doi:10.1186/s12943-024-02166-w. [Google Scholar] [PubMed] [CrossRef]
81. Wei XL, Luo X, Sheng H, Wang Y, Chen DL, Li JN, et al. PD-L1 expression in liver metastasis: its clinical significance and discordance with primary tumor in colorectal cancer. J Transl Med. 2020;18(1):475. doi:10.1186/s12967-020-02636-x. [Google Scholar] [PubMed] [CrossRef]
82. Boman C, Zerdes I, Mårtensson K, Bergh J, Foukakis T, Valachis A, et al. Discordance of PD-L1 status between primary and metastatic breast cancer: a systematic review and meta-analysis. Cancer Treat Rev. 2021;99:102257. doi:10.1016/j.ctrv.2021.102257. [Google Scholar] [PubMed] [CrossRef]
83. Liu DHW, Grabsch HI, Gloor B, Langer R, Dislich B. Programmed death-ligand 1 (PD-L1) expression in primary gastric adenocarcinoma and matched metastases. J Cancer Res Clin Oncol. 2023;149(14):13345–52. doi:10.1007/s00432-023-05142-x. [Google Scholar] [PubMed] [CrossRef]
84. Lizardo DY, Kuang C, Hao S, Yu J, Huang Y, Zhang L. Immunotherapy efficacy on mismatch repair-deficient colorectal cancer: from bench to bedside. Biochim Biophys Acta Rev Cancer. 2020;1874(2):188447. doi:10.1016/j.bbcan.2020.188447. [Google Scholar] [PubMed] [CrossRef]
85. Wang R, Lian J, Wang X, Pang X, Xu B, Tang S, et al. Intrinsic resistance and efficacy of immunotherapy in microsatellite instability-high colorectal cancer: a systematic review and meta-analysis. Biomol Biomed. 2023;23(2):198–208. doi:10.17305/bjbms.2022.8286. [Google Scholar] [PubMed] [CrossRef]
86. Wu T, Zhang X, Liu X, Cai X, Shen T, Pan D, et al. Single-cell sequencing reveals the immune microenvironment landscape related to anti-PD-1 resistance in metastatic colorectal cancer with high microsatellite instability. BMC Med. 2023;21(1):161. doi:10.1186/s12916-023-02866-y. [Google Scholar] [PubMed] [CrossRef]
87. Qin H, Zhou Z, Shi R, Mai Y, Xu Y, Peng F, et al. Insights into next-generation immunotherapy designs and tools: molecular mechanisms and therapeutic prospects. J Hematol Oncol. 2025;18(1):62. doi:10.1186/s13045-025-01701-6. [Google Scholar] [PubMed] [CrossRef]
88. Sahin IH, Akce M, Alese O, Shaib W, Lesinski GB, El-Rayes B, et al. Immune checkpoint inhibitors for the treatment of MSI-H/MMR-D colorectal cancer and a perspective on resistance mechanisms. Br J Cancer. 2019;121(10):809–18. doi:10.1038/s41416-019-0599-y. [Google Scholar] [PubMed] [CrossRef]
89. Normanno N, Caridi V, Fassan M, Avallone A, Ciardiello F, Pinto C. Resistance to immune checkpoint inhibitors in colorectal cancer with deficient mismatch repair/microsatellite instability: misdiagnosis, pseudoprogression and/or tumor heterogeneity? Explor Target Anti Tumor Ther. 2024;5(3):495–507. doi:10.37349/etat.2024.00231. [Google Scholar] [PubMed] [CrossRef]
90. Aldea M, Andre F, Marabelle A, Dogan S, Barlesi F, Soria JC. Overcoming resistance to tumor-targeted and immune-targeted therapies. Cancer Discov. 2021;11(4):874–99. doi:10.1158/2159-8290.CD-20-1638. [Google Scholar] [PubMed] [CrossRef]
91. Rodriguez-Pascual J, Ayuso-Sacido A, Belda-Iniesta C. Drug resistance in cancer immunotherapy: new strategies to improve checkpoint inhibitor therapies. Cancer Drug Resist. 2019;2(4):980–93. doi:10.20517/cdr.2019.61. [Google Scholar] [PubMed] [CrossRef]
92. Leto SM, Trusolino L. Primary and acquired resistance to EGFR-targeted therapies in colorectal cancer: impact on future treatment strategies. J Mol Med. 2014;92(7):709–22. doi:10.1007/s00109-014-1161-2. [Google Scholar] [PubMed] [CrossRef]
93. Sharma P, Siddiqui BA, Anandhan S, Yadav SS, Subudhi SK, Gao J, et al. The next decade of immune checkpoint therapy. Cancer Discov. 2021;11(4):838–57. doi:10.1158/2159-8290.cd-20-1680. [Google Scholar] [PubMed] [CrossRef]
94. Alsaafeen BH, Ali BR, Elkord E. Resistance mechanisms to immune checkpoint inhibitors: updated insights. Mol Cancer. 2025;24(1):20. doi:10.1186/s12943-024-02212-7. [Google Scholar] [PubMed] [CrossRef]
95. Haynes J, Manogaran P. Mechanisms and strategies to overcome drug resistance in colorectal cancer. Int J Mol Sci. 2025;26(5):1988. doi:10.3390/ijms26051988. [Google Scholar] [PubMed] [CrossRef]
96. Jiang C, Zhang ZH, Li JX. Consideration on immunotherapy of liver metastases of malignant tumors. World J Gastrointest Surg. 2024;16(8):2374–81. doi:10.4240/wjgs.v16.i8.2374. [Google Scholar] [PubMed] [CrossRef]
97. Mueller L, Goumas FA, Affeldt M, Sandtner S, Gehling UM, Brilloff S, et al. Stromal fibroblasts in colorectal liver metastases originate from resident fibroblasts and generate an inflammatory microenvironment. Am J Pathol. 2007;171(5):1608–18. doi:10.2353/ajpath.2007.060661. [Google Scholar] [PubMed] [CrossRef]
98. Wang X, Lamberti G, Di Federico A, Alessi J, Ferrara R, Sholl ML, et al. Tumor mutational burden for the prediction of PD-(L)1 blockade efficacy in cancer: challenges and opportunities. Ann Oncol. 2024;35(6):508–22. doi:10.1016/j.annonc.2024.03.007. [Google Scholar] [PubMed] [CrossRef]
99. D’Amico S, Tempora P, Melaiu O, Lucarini V, Cifaldi L, Locatelli F, et al. Targeting the antigen processing and presentation pathway to overcome resistance to immune checkpoint therapy. Front Immunol. 2022;13:948297. doi:10.3389/fimmu.2022.948297. [Google Scholar] [PubMed] [CrossRef]
100. Zhuang Y, Liu C, Liu J, Li G. Resistance mechanism of PD-1/PD-L1 blockade in the cancer-immunity cycle. Onco Targets Ther. 2020;13:83–94. doi:10.2147/OTT.S239398. [Google Scholar] [PubMed] [CrossRef]
101. Wu Y, Jiang X, Yu Z, Xing Z, Ma Y, Qing H. Mechanisms of anti-PD therapy resistance in digestive system neoplasms. Recent Pat Anti Cancer Drug Discov. 2025;20(1):1–25. doi:10.2174/0115748928269276231120103256. [Google Scholar] [PubMed] [CrossRef]
102. Martin-Orozco E, Sanchez-Fernandez A, Ortiz-Parra I, Ayala-San Nicolas M. WNT signaling in tumors: the way to evade drugs and immunity. Front Immunol. 2019;10:2854. doi:10.3389/fimmu.2019.02854. [Google Scholar] [PubMed] [CrossRef]
103. Relecom A, Merhi M, Inchakalody V, Uddin S, Rinchai D, Bedognetti D, et al. Emerging dynamics pathways of response and resistance to PD-1 and CTLA-4 blockade: tackling uncertainty by confronting complexity. J Exp Clin Cancer Res. 2021;40(1):74. doi:10.1186/s13046-021-01872-3. [Google Scholar] [PubMed] [CrossRef]
104. Anderson KG, Braun DA, Buqué A, Gitto SB, Guerriero JL, Horton B, et al. Leveraging immune resistance archetypes in solid cancer to inform next-generation anticancer therapies. J Immunother Cancer. 2023;11(6):e006533. doi:10.1136/jitc-2022-006533. [Google Scholar] [PubMed] [CrossRef]
105. Vidotto T, Melo CM, Castelli E, Koti M, Dos Reis RB, Squire JA. Emerging role of PTEN loss in evasion of the immune response to tumours. Br J Cancer. 2020;122(12):1732–43. doi:10.1038/s41416-020-0834-6. [Google Scholar] [PubMed] [CrossRef]
106. Hargadon KM. Genetic dysregulation of immunologic and oncogenic signaling pathways associated with tumor-intrinsic immune resistance: a molecular basis for combination targeted therapy-immunotherapy for cancer. Cell Mol Life Sci. 2023;80(2):40. doi:10.1007/s00018-023-04689-9. [Google Scholar] [PubMed] [CrossRef]
107. Hou W, Yi C, Zhu H. Predictive biomarkers of colon cancer immunotherapy: present and future. Front Immunol. 2022;13:1032314. doi:10.3389/fimmu.2022.1032314. [Google Scholar] [PubMed] [CrossRef]
108. Ahmed H, Mahmud AR, Siddiquee MF, Shahriar A, Biswas P, Shimul MEK, et al. Role of T cells in cancer immunotherapy: opportunities and challenges. Cancer Pathog Ther. 2023;1(2):116–26. doi:10.1016/j.cpt.2022.12.002. [Google Scholar] [PubMed] [CrossRef]
109. Di J, Liu M, Fan Y, Gao P, Wang Z, Jiang B, et al. Phenotype molding of T cells in colorectal cancer by single-cell analysis. Int J Cancer. 2020;146(8):2281–95. doi:10.1002/ijc.32856. [Google Scholar] [PubMed] [CrossRef]
110. Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pagès C, et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science. 2006;313(5795):1960–4. doi:10.1126/science.1129139. [Google Scholar] [PubMed] [CrossRef]
111. Yu J, Green MD, Li S, Sun Y, Journey SN, Choi JE, et al. Liver metastasis restrains immunotherapy efficacy via macrophage-mediated T cell elimination. Nat Med. 2021;27(1):152–64. doi:10.1038/s41591-020-1131-x. [Google Scholar] [PubMed] [CrossRef]
112. Joller N, Anderson AC, Kuchroo VK. LAG-3, TIM-3, and TIGIT: distinct functions in immune regulation. Immunity. 2024;57(2):206–22. doi:10.1016/j.immuni.2024.01.010. [Google Scholar] [PubMed] [CrossRef]
113. Johnston RJ, Comps-Agrar L, Hackney J, Yu X, Huseni M, Yang Y, et al. The immunoreceptor TIGIT regulates antitumor and antiviral CD8+ T cell effector function. Cancer Cell. 2014;26(6):923–37. doi:10.1016/j.ccell.2014.10.018. [Google Scholar] [PubMed] [CrossRef]
114. Beyrend G, Van der Gracht E, Yilmaz A, Van Duikeren S, Camps M, Höllt T, et al. PD-L1 blockade engages tumor-infiltrating lymphocytes to co-express targetable activating and inhibitory receptors. J Immunotherapy Cancer. 2019;7:217. doi:10.1186/s40425-019-0700-3. [Google Scholar] [PubMed] [CrossRef]
115. Liu J, Zhang S, Hu Y, Yang Z, Li J, Liu X, et al. Targeting PD-1 and tim-3 pathways to reverse CD8 T-cell exhaustion and enhance ex vivo T-cell responses to autologous dendritic/tumor vaccines. J Immunother. 2016;39(4):171–80. doi:10.1097/CJI.0000000000000122. [Google Scholar] [PubMed] [CrossRef]
116. Shan J, Han D, Shen C, Lei Q, Zhang Y. Mechanism and strategies of immunotherapy resistance in colorectal cancer. Front Immunol. 2022;13:1016646. doi:10.3389/fimmu.2022.1016646. [Google Scholar] [PubMed] [CrossRef]
117. Amodio V, Mauri G, Reilly NM, Sartore-Bianchi A, Siena S, Bardelli A, et al. Mechanisms of immune escape and resistance to checkpoint inhibitor therapies in mismatch repair deficient metastatic colorectal cancers. Cancers. 2021;13(11):2638. doi:10.3390/cancers13112638. [Google Scholar] [PubMed] [CrossRef]
118. Li W, Wu F, Zhao S, Shi P, Wang S, Cui D. Correlation between PD-1/PD-L1 expression and polarization in tumor-associated macrophages: a key player in tumor immunotherapy. Cytokine Growth Factor Rev. 2022;67:49–57. doi:10.1016/j.cytogfr.2022.07.004. [Google Scholar] [PubMed] [CrossRef]
119. Yahaya MAF, Lila MAM, Ismail S, Zainol M, Afizan NARNM. Tumour-associated macrophages (TAMs) in colon cancer and how to reeducate them. J Immunol Res. 2019;2019:2368249. doi:10.1155/2019/2368249. [Google Scholar] [PubMed] [CrossRef]
120. Chang L, Xu L, Tian Y, Liu Z, Song M, Li S, et al. NLRP6 deficiency suppresses colorectal cancer liver metastasis growth by modulating M-MDSC-induced immunosuppressive microenvironment. Biochim Biophys Acta Mol Basis Dis. 2024;1870(3):167035. doi:10.1016/j.bbadis.2024.167035. [Google Scholar] [PubMed] [CrossRef]
121. Liu C, Liu R, Wang B, Lian J, Yao Y, Sun H, et al. Blocking IL-17A enhances tumor response to anti-PD-1 immunotherapy in microsatellite stable colorectal cancer. J Immunother Cancer. 2021;9(1):e001895. doi:10.1136/jitc-2020-001895. [Google Scholar] [PubMed] [CrossRef]
122. Topalian SL, Drake CG, Pardoll DM. Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer Cell. 2015;27(4):450–61. doi:10.1016/j.ccell.2015.03.001. [Google Scholar] [PubMed] [CrossRef]
123. Zhang Y, Zhu H, Fan J, Zhao J, Xia Y, Zhang N, et al. A glutamine metabolism gene signature with prognostic and predictive value for colorectal cancer survival and immunotherapy response. Front Mol Biosci. 2025;12:1599141. doi:10.3389/fmolb.2025.1599141. [Google Scholar] [PubMed] [CrossRef]
124. Deaglio S, Dwyer KM, Gao W, Friedman D, Usheva A, Erat A, et al. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med. 2007;204(6):1257–65. doi:10.1084/jem.20062512. [Google Scholar] [PubMed] [CrossRef]
125. Leone RD, Sun IM, Oh MH, Sun IH, Wen J, Englert J, et al. Inhibition of the adenosine A2a receptor modulates expression of T cell coinhibitory receptors and improves effector function for enhanced checkpoint blockade and ACT in murine cancer models. Cancer Immunol Immunother. 2018;67(8):1271–84. doi:10.1007/s00262-018-2186-0. [Google Scholar] [PubMed] [CrossRef]
126. Chen E, Zhou W. Immunotherapy in microsatellite-stable colorectal cancer: strategies to overcome resistance. Crit Rev Oncol. 2025;212:104775. doi:10.1016/j.critrevonc.2025.104775. [Google Scholar] [PubMed] [CrossRef]
127. Werner W, Kuzminskaya M, Lurje I, Tacke F, Hammerich L. Overcoming resistance to immune checkpoint blockade in liver cancer with combination therapy: stronger together? Semin Liver Dis. 2024;44(2):159–79. doi:10.1055/a-2334-8311. [Google Scholar] [PubMed] [CrossRef]
128. Yao S, Han Y, Yang M, Jin K, Lan H. It’s high-time to re-evaluate the value of induced-chemotherapy for reinforcing immunotherapy in colorectal cancer. Front Immunol. 2023;14:1241208. doi:10.3389/fimmu.2023.1241208. [Google Scholar] [PubMed] [CrossRef]
129. Thibaudin M, Fumet JD, Chibaudel B, Bennouna J, Borg C, Martin-Babau J, et al. First-line durvalumab and tremelimumab with chemotherapy in RAS-mutated metastatic colorectal cancer: a phase 1b/2 trial. Nat Med. 2023;29(8):2087–98. doi:10.1038/s41591-023-02497-z. [Google Scholar] [PubMed] [CrossRef]
130. Wang W, Wu L, Zhang J, Wu H, Han E, Guo Q. Chemoimmunotherapy by combining oxaliplatin with immune checkpoint blockades reduced tumor burden in colorectal cancer animal model. Biochem Biophys Res Commun. 2017;487(1):1–7. doi:10.1016/j.bbrc.2016.12.180. [Google Scholar] [PubMed] [CrossRef]
131. Ree AH, Šaltytė Benth J, Hamre HM, Kersten C, Hofsli E, Guren MG, et al. First-line oxaliplatin-based chemotherapy and nivolumab for metastatic microsatellite-stable colorectal cancer-the randomised METIMMOX trial. Br J Cancer. 2024;130(12):1921–8. doi:10.1038/s41416-024-02696-6. [Google Scholar] [PubMed] [CrossRef]
132. Ragusa S, Prat-Luri B, González-Loyola A, Nassiri S, Squadrito ML, Guichard A, et al. Antiangiogenic immunotherapy suppresses desmoplastic and chemoresistant intestinal tumors in mice. J Clin Investig. 2020;130(3):1199–216. doi:10.1172/JCI129558. [Google Scholar] [PubMed] [CrossRef]
133. Fakih M, Sandhu J, Lim D, Li X, Li S, Wang C. Regorafenib, ipilimumab, and nivolumab for patients with microsatellite stable colorectal cancer and disease progression with prior chemotherapy: a phase 1 nonrandomized clinical trial. JAMA Oncol. 2023;9(5):627–34. doi:10.1001/jamaoncol.2022.7845. [Google Scholar] [PubMed] [CrossRef]
134. Antoniotti C, Rossini D, Pietrantonio F, Salvatore L, Lonardi S, Tamberi S, et al. Upfront fluorouracil, leucovorin, oxaliplatin, and irinotecan plus bevacizumab with or without atezolizumab for patients with metastatic colorectal cancer: updated and overall survival results of the ATEZOTRIBE study. J Clin Oncol. 2024;42(22):2637–44. doi:10.1200/jco.23.02728. [Google Scholar] [PubMed] [CrossRef]
135. Antoniotti C, Carullo M, Rossini D, Pietrantonio F, Salvatore L, Lonardi S, et al. Liver metastases do not predict resistance to the addition of atezolizumab to first-line FOLFOXIRI plus bevacizumab in proficient MMR metastatic colorectal cancer: a secondary analysis of the AtezoTRIBE study. ESMO Open. 2025;10(2):104135. doi:10.1016/j.esmoop.2025.104135. [Google Scholar] [PubMed] [CrossRef]
136. Ebert PJR, Cheung J, Yang Y, McNamara E, Hong R, Moskalenko M, et al. MAP kinase inhibition promotes T cell and anti-tumor activity in combination with PD-L1 checkpoint blockade. Immunity. 2016;44(3):609–21. doi:10.1016/j.immuni.2016.01.024. [Google Scholar] [PubMed] [CrossRef]
137. Tian J, Chen JH, Chao SX, Pelka K, Giannakis M, Hess J, et al. Combined PD-1, BRAF and MEK inhibition in BRAFV600E colorectal cancer: a phase 2 trial. Nat Med. 2023;29(2):458–66. doi:10.1038/s41591-022-02181-8. [Google Scholar] [PubMed] [CrossRef]
138. Deng J, Wang ES, Jenkins RW, Li S, Dries R, Yates K, et al. CDK4/6 inhibition augments antitumor immunity by enhancing T-cell activation. Cancer Discov. 2018;8(2):216–33. doi:10.1158/2159-8290.cd-17-0915. [Google Scholar] [PubMed] [CrossRef]
139. Fujiwara Y, Kato S, Nesline MK, Conroy JM, DePietro P, Pabla S, et al. Indoleamine 2, 3-dioxygenase (IDO) inhibitors and cancer immunotherapy. Cancer Treat Rev. 2022;110:102461. doi:10.1016/j.ctrv.2022.102461. [Google Scholar] [PubMed] [CrossRef]
140. Said SS, Ibrahim WN. Cancer resistance to immunotherapy: comprehensive insights with future perspectives. Pharmaceutics. 2023;15(4):1143. doi:10.3390/pharmaceutics15041143. [Google Scholar] [PubMed] [CrossRef]
141. Li Y, Hahn T, Garrison K, Cui ZH, Thorburn A, Thorburn J, et al. The vitamin E analogue α-TEA stimulates tumor autophagy and enhances antigen cross-presentation. Cancer Res. 2012;72(14):3535–45. doi:10.1158/0008-5472.can-11-3103. [Google Scholar] [PubMed] [CrossRef]
142. ALKhemeiri N, Eljack S, Saber-Ayad MM. Perspectives of targeting autophagy as an adjuvant to anti-PD-1/PD-L1 therapy for colorectal cancer treatment. Cells. 2025;14(10):745. doi:10.3390/cells14100745. [Google Scholar] [PubMed] [CrossRef]
143. Procureur A, Simonaggio A, Bibault JE, Oudard S, Vano YA. Enhance the immune checkpoint inhibitors efficacy with radiotherapy induced immunogenic cell death: a comprehensive review and latest developments. Cancers. 2021;13(4):678. doi:10.3390/cancers13040678. [Google Scholar] [PubMed] [CrossRef]
144. Jiang Y, Qiao S, Li L, Zhu X. Combination of radiotherapy and Anlotinib enhances benefit from immunotherapy to liver metastasis and abscopal tumor from lung cancer. Int Immunopharmacol. 2024;128:111441. doi:10.1016/j.intimp.2023.111441. [Google Scholar] [PubMed] [CrossRef]
145. Bai H, Wang WH, Zhou FF, Yang D, Li RJ. Feasibility and tolerability of anlotinib plus PD-1 blockades for patients with treatment-refractory metastatic colorectal cancer: a retrospective exploratory study. Cancer Manage Res. 2024;16:73–86. doi:10.2147/CMAR.S427680. [Google Scholar] [PubMed] [CrossRef]
146. Wang F, Lai C, Lv Y, Zhang F, Shi L, Wang Y, et al. Efficacy and safety of combining short-course neoadjuvant chemoradiotherapy with envafolimab in locally advanced rectal cancer patients with microsatellite stability: a phase II PRECAM experimental study. Int J Surg. 2025;111(1):334–45. doi:10.1097/JS9.0000000000001960. [Google Scholar] [PubMed] [CrossRef]
147. Zhang X, Wu T, Cai X, Dong J, Xia C, Zhou Y, et al. Neoadjuvant immunotherapy for MSI-H/dMMR locally advanced colorectal cancer: new strategies and unveiled opportunities. Front Immunol. 2022;13:795972. doi:10.3389/fimmu.2022.795972. [Google Scholar] [PubMed] [CrossRef]
148. Cai L, Li Y, Tan J, Xu L, Li Y. Targeting LAG-3, TIM-3, and TIGIT for cancer immunotherapy. J Hematol Oncol. 2023;16(1):101. doi:10.1186/s13045-023-01499-1. [Google Scholar] [PubMed] [CrossRef]
149. Karri V, Nimkar S, Dalia SM. Adjuvant immunotherapy in microsatellite instability-high colon cancer: a literature review on efficacy, challenges, and future directions. Discov Med. 2025;37(196):808–15. doi:10.24976/Discov.Med.202537196.71. [Google Scholar] [PubMed] [CrossRef]
150. Garralda E, Sukari A, Lakhani NJ, Patnaik A, Lou Y, Im SA, et al. A first-in-human study of the anti-LAG-3 antibody favezelimab plus pembrolizumab in previously treated, advanced microsatellite stable colorectal cancer. ESMO Open. 2022;7(6):100639. doi:10.1016/j.esmoop.2022.100639. [Google Scholar] [PubMed] [CrossRef]
151. Okpalanwaka IF, Daugherity EA, McCormick AL, Anderson TS, Smith SL, Lawrence C, et al. A PD-L1/CD3 bispecific antibody enhances the antitumor effects of regorafenib against colon cancer. Mol Cancer Ther. 2025;24(8):1240–51. doi:10.1158/1535-7163.MCT-24-1015. [Google Scholar] [PubMed] [CrossRef]
152. Vasudevan K, Dhanushkumar T, Hebbar SR, Selvam PK, Rambabu M, Anbarasu K, et al. Multi-omics and AI-driven immune subtyping to optimize neoantigen-based vaccines for colorectal cancer. Sci Rep. 2025;15(1):19333. doi:10.1038/s41598-025-01680-1. [Google Scholar] [PubMed] [CrossRef]
153. Zhou X, Yao Z, Bai H, Duan J, Wang Z, Wang X, et al. Treatment-related adverse events of PD-1 and PD-L1 inhibitor-based combination therapies in clinical trials: a systematic review and meta-analysis. Lancet Oncol. 2021;22(9):1265–74. doi:10.1016/S1470-2045(21)00333-8. [Google Scholar] [PubMed] [CrossRef]
154. Li Y, Amaladas N, O’Mahony M, Manro JR, Inigo I, Li Q, et al. Treatment with a VEGFR-2 antibody results in intra-tumor immune modulation and enhances anti-tumor efficacy of PD-L1 blockade in syngeneic murine tumor models. PLoS One. 2022;17(7):e0268244. doi:10.1371/journal.pone.0268244. [Google Scholar] [PubMed] [CrossRef]
155. Damato A, Iachetta F, Antonuzzo L, Nasti G, Bergamo F, Bordonaro R, et al. Phase II study on first-line treatment of NIVolumab in combination with folfoxiri/bevacizumab in patients with Advanced COloRectal cancer RAS or BRAF mutated—NIVACOR trial (GOIRC-03-2018). BMC Cancer. 2020;20(1):822. doi:10.1186/s12885-020-07268-4. [Google Scholar] [PubMed] [CrossRef]
156. Damato A, Bergamo F, Antonuzzo L, Nasti G, Iachetta F, Romagnani A, et al. FOLFOXIRI/bevacizumab plus nivolumab as first-line treatment in metastatic colorectal cancer RAS/BRAF mutated: safety Run-In of phase II NIVACOR trial. Front Oncol. 2021;11:766500. doi:10.3389/fonc.2021.766500. [Google Scholar] [PubMed] [CrossRef]
157. Lenz HJ, Parikh A, Spigel DR, Cohn AL, Yoshino T, Kochenderfer M, et al. Modified FOLFOX6 plus bevacizumab with and without nivolumab for first-line treatment of metastatic colorectal cancer: phase 2 results from the CheckMate 9X8 randomized clinical trial. J Immunother Cancer. 2024;12(3):e008409. doi:10.1136/jitc-2023-008409. [Google Scholar] [PubMed] [CrossRef]
158. Men Q, Duan Y, Pei F, Yao Q, He W, Zhao Y, et al. PD-1 blockade combined with chemotherapy and bevacizumab in DNA mismatch repair-proficient/microsatellite stable colorectal liver metastases. J Gastrointest Oncol. 2024;15(4):1534–44. doi:10.21037/jgo-23-940. [Google Scholar] [PubMed] [CrossRef]
159. Ding Y, Feng Y, Ye Y, Shen J, Guo C, He X, et al. High and low dose radiotherapy combined with ICIs for MSS colorectal cancer patients with liver metastases: a phase I study (HaRyPOT). Front Oncol. 2025;15:1503517. doi:10.3389/fonc.2025.1503517. [Google Scholar] [PubMed] [CrossRef]
160. Kasi PM, Hidalgo M, Jafari MD, Yeo H, Lowenfeld L, Khan U, et al. Neoadjuvant botensilimab plus balstilimab response pattern in locally advanced mismatch repair proficient colorectal cancer. Oncogene. 2023;42(44):3252–9. doi:10.1038/s41388-023-02835-y. [Google Scholar] [PubMed] [CrossRef]
161. Morano F, Raimondi A, Pagani F, Lonardi S, Salvatore L, Cremolini C, et al. Temozolomide followed by combination with low-dose ipilimumab and nivolumab in patients with microsatellite-stable, O(6)-methylguanine-DNA methyltransferase-silenced metastatic colorectal cancer: the MAYA trial. J Clin Oncol. 2022;40(14):1562–73. doi:10.1200/JCO.21.02583. [Google Scholar] [PubMed] [CrossRef]
162. Lenz HJ, Van Cutsem E, Luisa Limon M, Wong KYM, Hendlisz A, Aglietta M, et al. First-line nivolumab plus low-dose ipilimumab for microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: the phase II CheckMate 142 study. J Clin Oncol. 2022;40(2):161–70. doi:10.1200/JCO.21.01015. [Google Scholar] [PubMed] [CrossRef]
163. Singer M, Valerin J, Zhang Z, Zhang Z, Dayyani F, Yaghmai V, et al. Promising cellular immunotherapy for colorectal cancer using classical dendritic cells and natural killer T cells. Cells. 2025;14(3):166. doi:10.3390/cells14030166. [Google Scholar] [PubMed] [CrossRef]
164. Prakash J, Shaked Y. The interplay between extracellular matrix remodeling and cancer therapeutics. Cancer Discov. 2024;14(8):1375–88. doi:10.1158/2159-8290.cd-24-0002. [Google Scholar] [PubMed] [CrossRef]
165. Hu M, Wang Y, Xu L, An S, Tang Y, Zhou X, et al. Relaxin gene delivery mitigates liver metastasis and synergizes with check point therapy. Nat Commun. 2019;10(1):2993. doi:10.1038/s41467-019-10893-8. [Google Scholar] [PubMed] [CrossRef]
166. Liang Z, Xu X, Wang N, He X, Han X, Sang L, et al. Manganese-based nanoparticles plus gambogic acid targeted hypoxic tumor microenvironment by enhancing ROS generation and provided antitumor treatment and improved immunotherapy. RSC Adv. 2025;15(15):11283–92. doi:10.1039/d4ra08547g. [Google Scholar] [PubMed] [CrossRef]
167. Greco B, El Khoury R, Balestrieri C, Sirini C, Machado A, De Girardi F, et al. Systemic delivery of cadherin 17-specific CAR T cells allows effective and safe targeting of colorectal cancer liver metastases. Sci Transl Med. 2025;17(800):eadr1928. doi:10.1126/scitranslmed.adr1928. [Google Scholar] [PubMed] [CrossRef]
168. Jaeckel E, Friedman SL, Hudecek M, Protzer U. Chimeric antigen receptor (CAR) T-cell therapy: engineering immune cells to treat liver diseases. J Hepatol. 2025;83(5):1156–71. doi:10.1016/j.jhep.2025.06.007. [Google Scholar] [PubMed] [CrossRef]
169. Li J, Liu C, Zhang P, Shen L, Qi C. Optimizing CAR T cell therapy for solid tumours: a clinical perspective. Nat Rev Clin Oncol. 2025;22(12):953–68. doi:10.1038/s41571-025-01075-1. [Google Scholar] [PubMed] [CrossRef]
170. Gu X, Zhang Y, Zhou W, Wang F, Yan F, Gao H, et al. Infusion and delivery strategies to maximize the efficacy of CAR-T cell immunotherapy for cancers. Exp Hematol Oncol. 2024;13(1):70. doi:10.1186/s40164-024-00542-2. [Google Scholar] [PubMed] [CrossRef]
171. Hou AJ, Chang ZL, Lorenzini MH, Zah E, Chen YY. TGF-β-responsive CAR-T cells promote anti-tumor immune function. Bioeng Transl Med. 2018;3(2):75–86. doi:10.1002/btm2.10097. [Google Scholar] [PubMed] [CrossRef]
172. Adachi K, Kano Y, Nagai T, Okuyama N, Sakoda Y, Tamada K. IL-7 and CCL19 expression in CAR-T cells improves immune cell infiltration and CAR-T cell survival in the tumor. Nat Biotechnol. 2018;36(4):346–51. doi:10.1038/nbt.4086. [Google Scholar] [PubMed] [CrossRef]
173. Hong M, Clubb JD, Chen YY. Engineering CAR-T cells for next-generation cancer therapy. Cancer Cell. 2020;38(4):473–88. doi:10.1016/j.ccell.2020.07.005. [Google Scholar] [PubMed] [CrossRef]
174. Halama N, Zoernig I, Berthel A, Kahlert C, Klupp F, Suarez-Carmona M, et al. Tumoral immune cell exploitation in colorectal cancer metastases can be targeted effectively by anti-CCR5 therapy in cancer patients. Cancer Cell. 2016;29(4):587–601. doi:10.1016/j.ccell.2016.03.005. [Google Scholar] [PubMed] [CrossRef]
175. Collins NB, Al Abosy R, Miller BC, Bi K, Zhao Q, Quigley M, et al. PI3K activation allows immune evasion by promoting an inhibitory myeloid tumor microenvironment. J Immunother Cancer. 2022;10(3):e003402. doi:10.1136/jitc-2021-003402. [Google Scholar] [PubMed] [CrossRef]
176. Parra ER, Uraoka N, Jiang M, Cook P, Gibbons D, Forget MA, et al. Validation of multiplex immunofluorescence panels using multispectral microscopy for immune-profiling of formalin-fixed and paraffin-embedded human tumor tissues. Sci Rep. 2017;7(1):13380. doi:10.1038/s41598-017-13942-8. [Google Scholar] [PubMed] [CrossRef]
177. Rojas F, Hernandez S, Lazcano R, Laberiano-Fernandez C, Parra ER. Multiplex immunofluorescence and the digital image analysis workflow for evaluation of the tumor immune environment in translational research. Front Oncol. 2022;12:889886. doi:10.3389/fonc.2022.889886. [Google Scholar] [PubMed] [CrossRef]
178. Semba T, Ishimoto T. Spatial analysis by current multiplexed imaging technologies for the molecular characterisation of cancer tissues. Br J Cancer. 2024;131(11):1737–47. doi:10.1038/s41416-024-02882-6. [Google Scholar] [PubMed] [CrossRef]
179. Parikh AR, Van Seventer EE, Siravegna G, Hartwig AV, Jaimovich A, He Y, et al. Minimal residual disease detection using a plasma-only circulating tumor DNA assay in patients with colorectal cancer. Clin Cancer Res. 2021;27(20):5586–94. doi:10.1158/1078-0432.ccr-21-0410. [Google Scholar] [PubMed] [CrossRef]
180. Reinert T, Petersen LMS, Henriksen TV, Larsen MØ, Rasmussen MH, Johansen AFB, et al. Circulating tumor DNA for prognosis assessment and postoperative management after curative-intent resection of colorectal liver metastases. Int J Cancer. 2022;150(9):1537–48. doi:10.1002/ijc.33924. [Google Scholar] [PubMed] [CrossRef]
181. Li Y, Xu J, Hu X, Chen Y, Liu F, Chen Y, et al. Personalized circulating tumor DNA monitoring improves recurrence surveillance and management after curative resection of colorectal liver metastases: a prospective cohort study. Int J Surg. 2024;110(5):2776–87. doi:10.1097/JS9.0000000000001236. [Google Scholar] [PubMed] [CrossRef]
182. Topham JT, O’Callaghan CJ, Feilotter H, Kennecke HF, Lee YS, Li W, et al. Circulating tumor DNA identifies diverse landscape of acquired resistance to anti-epidermal growth factor receptor therapy in metastatic colorectal cancer. J Clin Oncol. 2023;41(3):485–96. doi:10.1200/JCO.22.00364. [Google Scholar] [PubMed] [CrossRef]
183. Linnekamp JF, Van Hooff SR, Prasetyanti PR, Kandimalla R, Buikhuisen JY, Fessler E, et al. Consensus molecular subtypes of colorectal cancer are recapitulated in in vitro and in vivo models. Cell Death Differ. 2018;25(3):616–33. doi:10.1038/s41418-017-0011-5. [Google Scholar] [PubMed] [CrossRef]
184. Cohen R, Pudlarz T, Delattre JF, Colle R, André T. Molecular targets for the treatment of metastatic colorectal cancer. Cancers. 2020;12(9):2350. doi:10.3390/cancers12092350. [Google Scholar] [PubMed] [CrossRef]
185. El Hage M, Su Z, Linnebacher M. Addressing challenges in targeted therapy for metastatic colorectal cancer. Cancers. 2025;17(7):1098. doi:10.3390/cancers17071098. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF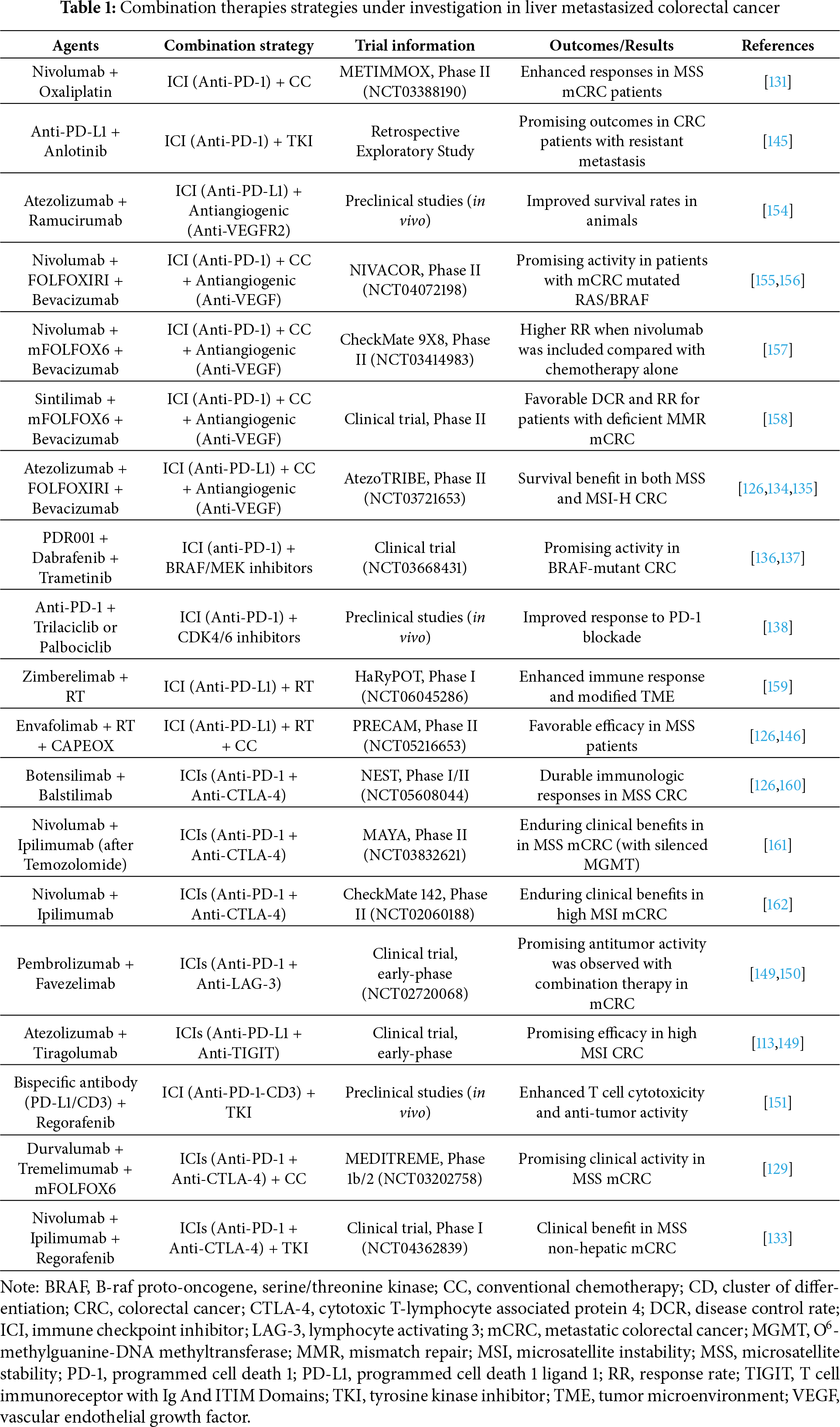
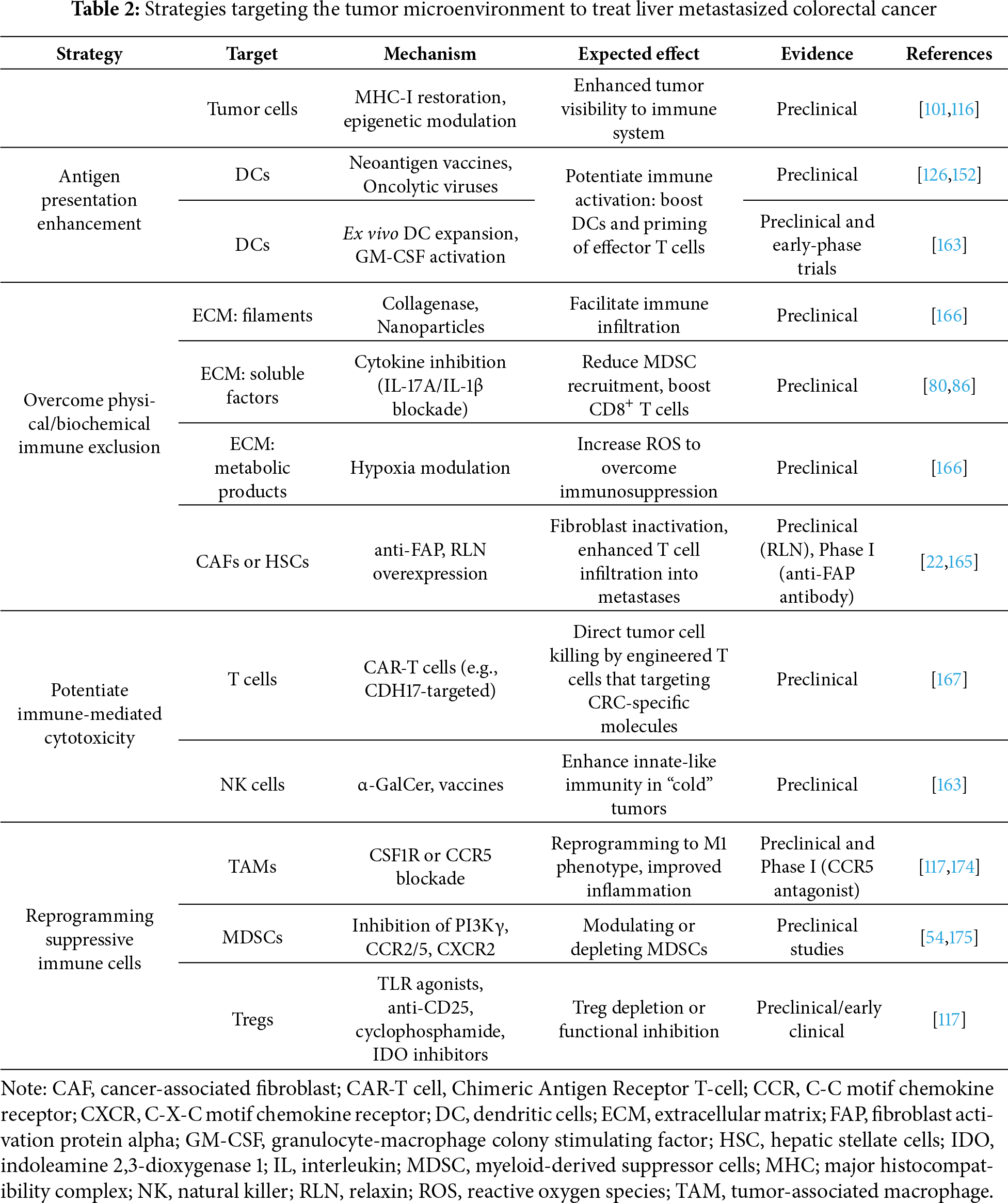
 Downloads
Downloads
 Citation Tools
Citation Tools