 Open Access
Open Access
ARTICLE
Oxidative Stress Footprints in Bone Marrow Mesenchymal Stem Cells from Untreated Advanced Breast Cancer
1 Laboratorio de Inmunohematología, Instituto de Biología y Medicina Experimental (IBYME), Fundación IBYME, Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Ciudad Autónoma de Buenos Aires, Argentina
2 Laboratorio de Medicina Regenerativa Cardiovascular, Instituto de Medicina Traslacional, Trasplante y Bioingeniería (IMETTyB-Universidad Favaloro-CONICET), Ciudad Autónoma de Buenos Aires, Argentina
3 Instituto de Bioquímica y Medicina Molecular (IBIMOL), Facultad de Farmacia y Bioquímica, CONICET, Universidad de Buenos Aires, Ciudad Autónoma de Buenos Aires, Argentina
4 Division of Hematology and Medical Oncology, Department of Medicine, Weill Cornell Medical College, New York, NY, USA
5 Facultad de Ciencias de la Salud, Universidad Nacional del Centro de la Provincia de Buenos Aires (UNCPB), Tandil, Provincia de Buenos Aires, Argentina
6 Servicio de Hematología, Hospital Militar Central, Ciudad Autónoma de Buenos Aires, Argentina
7 Laboratorio de Genética Hematológica, Instituto de Medicina Experimental (IMEX)-CONICET, Academia Nacional de Medicina, Ciudad Autónoma de Buenos Aires, Argentina
8 Servicio de Oncología, Hospital Militar Central, Ciudad Autónoma de Buenos Aires, Argentina
9 Laboratorio de Mutagénesis, IMEX-CONICET, Academia Nacional de Medicina, Ciudad Autónoma de Buenos Aires, Argentina
* Corresponding Authors: Francisco Raúl Borzone. Email: ; Norma Alejandra Chasseing. Email:
Oncology Research 2026, 34(4), 20 https://doi.org/10.32604/or.2026.074321
Received 08 October 2025; Accepted 22 January 2026; Issue published 23 March 2026
Abstract
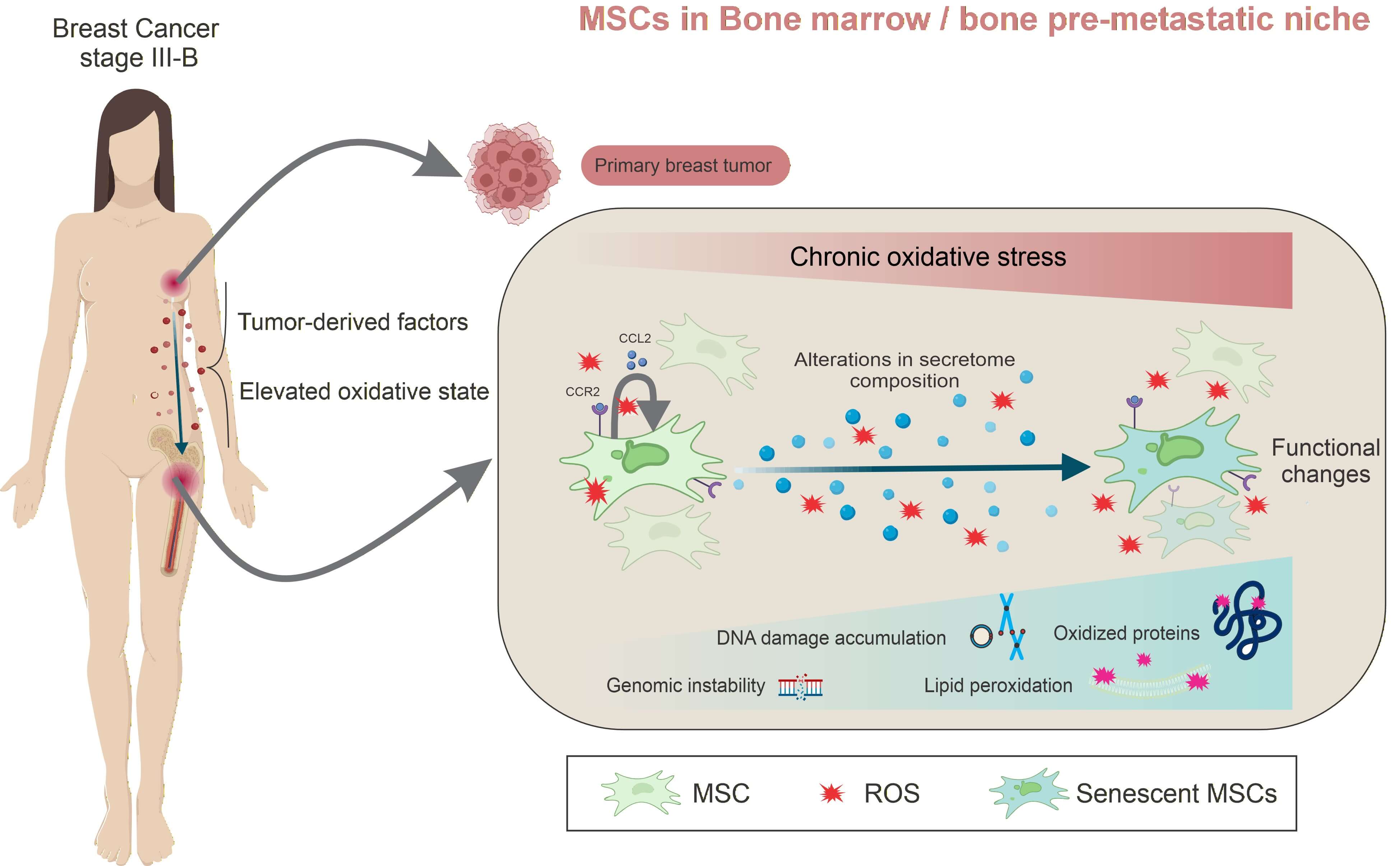
Backgrounds: Breast cancer metastasis remains the leading cause of mortality and frequently targets the bone. Breast cancer cells release soluble factors and extracellular vesicles that disrupt bone marrow (BM)/bone homeostasis, promoting osteoclastogenesis and the accumulation of senescent cells. In line with updated cancer hallmarks, senescent mesenchymal stem/ stromal cells (MSCs), osteoblasts, and osteocytes contribute to remodeling of the BM microenvironment, thereby favoring pre-metastatic niche (PMN) formation and subsequent bone metastasis. We previously demonstrated that untreated stage III-B breast cancer patients (BCPs) exhibit increased oxidative stress and elevated reactive oxygen species (ROS) levels, accompanied by senescent and functionally impaired BM-MSCs—key regulators of BM/bone homeostasis. In the present study, we sought to identify the molecular targets affected by oxidative stress that drive MSC senescence in these patients. Methods: BM-MSCs were isolated from untreated stage III-B BCPs and healthy volunteers (HVs). Oxidative stress responses were evaluated by quantitative real-time PCR (qRT-PCR) analysis of stress- and antioxidant-related genes. Oxidative damage to DNA, proteins, and lipids was assessed using alkaline comet assay, chromosomal aberration (CAs) analysis, micronuclei (MN) and nuclear blebs (NBs) quantification, protein carbonyl content, and detection of 4-hydroxynonenal (4-HNE) adducts. The MSC secretome was analyzed by label-free quantitative proteomics followed by Gene Ontology enrichment analysis. Results: Our results show that elevated oxidative stress in BCPs induces the overexpression of oxidative stress–related and antioxidant response genes in BM-MSCs; however, this response is insufficient to prevent extensive ROS-induced damage to deoxyribonucleic acid (DNA), proteins, and lipids. In addition, proteomic analysis of the BM-MSC secretome revealed a distinct protein expression profile in BCPs compared with HVs. Conclusions: Together, these findings highlight oxidative stress–induced MSC damage as a key mechanism contributing to PMN formation and suggest potential therapeutic targets to mitigate bone metastasis in advanced breast cancer.Graphic Abstract
Keywords
Supplementary Material
Supplementary Material FileCite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools