 Open Access
Open Access
ARTICLE
Oxidative Stress Footprints in Bone Marrow Mesenchymal Stem Cells from Untreated Advanced Breast Cancer
1 Laboratorio de Inmunohematología, Instituto de Biología y Medicina Experimental (IBYME), Fundación IBYME, Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Ciudad Autónoma de Buenos Aires, Argentina
2 Laboratorio de Medicina Regenerativa Cardiovascular, Instituto de Medicina Traslacional, Trasplante y Bioingeniería (IMETTyB-Universidad Favaloro-CONICET), Ciudad Autónoma de Buenos Aires, Argentina
3 Instituto de Bioquímica y Medicina Molecular (IBIMOL), Facultad de Farmacia y Bioquímica, CONICET, Universidad de Buenos Aires, Ciudad Autónoma de Buenos Aires, Argentina
4 Division of Hematology and Medical Oncology, Department of Medicine, Weill Cornell Medical College, New York, NY, USA
5 Facultad de Ciencias de la Salud, Universidad Nacional del Centro de la Provincia de Buenos Aires (UNCPB), Tandil, Provincia de Buenos Aires, Argentina
6 Servicio de Hematología, Hospital Militar Central, Ciudad Autónoma de Buenos Aires, Argentina
7 Laboratorio de Genética Hematológica, Instituto de Medicina Experimental (IMEX)-CONICET, Academia Nacional de Medicina, Ciudad Autónoma de Buenos Aires, Argentina
8 Servicio de Oncología, Hospital Militar Central, Ciudad Autónoma de Buenos Aires, Argentina
9 Laboratorio de Mutagénesis, IMEX-CONICET, Academia Nacional de Medicina, Ciudad Autónoma de Buenos Aires, Argentina
* Corresponding Authors: Francisco Raúl Borzone. Email: ; Norma Alejandra Chasseing. Email:
Oncology Research 2026, 34(4), 20 https://doi.org/10.32604/or.2026.074321
Received 08 October 2025; Accepted 22 January 2026; Issue published 23 March 2026
Abstract
Backgrounds: Breast cancer metastasis remains the leading cause of mortality and frequently targets the bone. Breast cancer cells release soluble factors and extracellular vesicles that disrupt bone marrow (BM)/bone homeostasis, promoting osteoclastogenesis and the accumulation of senescent cells. In line with updated cancer hallmarks, senescent mesenchymal stem/ stromal cells (MSCs), osteoblasts, and osteocytes contribute to remodeling of the BM microenvironment, thereby favoring pre-metastatic niche (PMN) formation and subsequent bone metastasis. We previously demonstrated that untreated stage III-B breast cancer patients (BCPs) exhibit increased oxidative stress and elevated reactive oxygen species (ROS) levels, accompanied by senescent and functionally impaired BM-MSCs—key regulators of BM/bone homeostasis. In the present study, we sought to identify the molecular targets affected by oxidative stress that drive MSC senescence in these patients. Methods: BM-MSCs were isolated from untreated stage III-B BCPs and healthy volunteers (HVs). Oxidative stress responses were evaluated by quantitative real-time PCR (qRT-PCR) analysis of stress- and antioxidant-related genes. Oxidative damage to DNA, proteins, and lipids was assessed using alkaline comet assay, chromosomal aberration (CAs) analysis, micronuclei (MN) and nuclear blebs (NBs) quantification, protein carbonyl content, and detection of 4-hydroxynonenal (4-HNE) adducts. The MSC secretome was analyzed by label-free quantitative proteomics followed by Gene Ontology enrichment analysis. Results: Our results show that elevated oxidative stress in BCPs induces the overexpression of oxidative stress–related and antioxidant response genes in BM-MSCs; however, this response is insufficient to prevent extensive ROS-induced damage to deoxyribonucleic acid (DNA), proteins, and lipids. In addition, proteomic analysis of the BM-MSC secretome revealed a distinct protein expression profile in BCPs compared with HVs. Conclusions: Together, these findings highlight oxidative stress–induced MSC damage as a key mechanism contributing to PMN formation and suggest potential therapeutic targets to mitigate bone metastasis in advanced breast cancer.Graphic Abstract
Keywords
Supplementary Material
Supplementary Material FileNomenclature
| α-MEM | α-minimal essential medium |
| ACs | Abnormal cells |
| Abs | Antibodies |
| AE1–AE3 | Cytokeratins |
| AF | Acentric fragments |
| BM | Bone Marrow |
| BMP | Bone morphogenetic proteins |
| BCPs | Breast Cancer Patients |
| BSA | Bovine serum albumin |
| CAs | Chromosomal aberrations |
| CAT | Catalase |
| CB | Chromatid breaks |
| CCL-2 | C-C motif chemokine 2 |
| CCR2 | C-C motif chemokine receptor 2 |
| cDNA | Complementary DNA |
| CFU-F | Fibroblastic Colony-Forming Unit |
| CM | Conditioned Media |
| DIC | Dicentric chromosomes |
| DNA | Deoxyribonucleic Acid |
| DNPH | 2,4-dinitrophenylhydrazine |
| DTT | Dithiothreitol |
| EDTA | Ethylene diamine tetraacetic acid |
| EMA | Epithelial membrane antigen |
| FBS | Fetal Bovine Serum |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase |
| GO | Gene Ontology |
| GPx1 | Glutathione Peroxidase 1 |
| HRP | Horseradish Peroxidase |
| HSP90 | Heat Shock Protein 90 |
| HUC-MSCs | Human Umbilical Cord Mesenchymal Stem Cells |
| HVs | Healthy volunteers |
| TERT | Telomerase Reverse Transcriptase |
| IL-6 | Interleukin 6 |
| LFQ | Label-Free Quantification |
| LMP | Low-melting-point agarose |
| MN | Micronuclei |
| MNCs | Mononuclear cells |
| MSCs | Mesenchymal Stem/ Stromal Cells |
| NBs | Nuclear blebs |
| NMP | Normal-melting-point agarose |
| Nrf2 | Nuclear Factor Erythroid 2–Related Factor 2 |
| OCT4 | Octamer-binding transcription factor 4 |
| PBS | Phosphate-Buffered Saline |
| PFA | Paraformaldehyde |
| PMN | Pre-Metastatic Niche |
| RC | Ring chromosomes |
| RNA | Ribonucleic acid |
| ROS | Reactive Oxygen Species |
| RT-PCR | Reverse Transcription Polymerase Chain Reaction |
| SASP | Senescence-Associated Secretory Phenotype |
| SDS-PAGE | Sodium dodecyl sulfate-polyacrylamide |
| SE | Standard Error |
| SOD1 | Superoxide Dismutase 1 |
| SOD2 | Superoxide Dismutase 2 |
| SOX2 | Sex determining region Y-box 2 |
| TAS | Telomeric associations |
| TBARS | 2-thiobarbituric acid-reactive substances |
| TBS | Tris-Buffered Saline |
| T-20 | Tween 20 |
| TGF-β | Transforming growth factor beta |
| TNF-α | Tumor Necrosis Factor Alpha |
| VEGF | Vascular Endothelial Growth Factor |
| 4-HNE | 4-Hydroxynonenal |
Distant metastasis remains the leading cause of mortality among BCPs. Breast cancer metastases display marked organotropism, with bone representing the most frequent initial site, followed by lung and, to a lesser extent, liver and brain [1,2]. Importantly, bone metastases not only drive skeletal-related complications but also facilitate secondary dissemination of tumor cells through horizontal cross-metastatic seeding [3,4].
Primary breast tumors secrete soluble factors and extracellular vesicles that play a critical role in preparing and conditioning distant tissues for PMN formation [5–8]. In the BM/bone microenvironment, this paracrine signaling alters the behavior of osteoblasts and osteoclasts, promoting changes that support tumor cell colonization [9–11]. In parallel, it modulates MSC phenotype and function, inducing alterations that further contribute to the establishment of a permissive PMN [12–14].
Recent evidence highlights cellular senescence as a key driver of bone metastatic disease. In established breast cancer bone metastases, osteocytes undergo premature senescence and acquire a distinct senescence-associated secretory phenotype (SASP) that promotes bone degradation. These findings identify cellular senescence as an initiating event in lytic bone disease associated with metastatic breast cancer [15].
MSCs are central regulators of BM/bone homeostasis and key components of the PMN. In previous work, we demonstrated that MSCs isolated from the BM of untreated advanced BCPs (stage III-B, without detectable BM/bone metastasis) exhibit pronounced morphological alterations, including enlarged and flattened morphology driven by cytoskeletal rearrangements—early hallmarks of cellular senescence [16–18]. These cells also display increased senescence-associated β-galactosidase activity, reduced self-renewal and proliferative capacity, and impaired cell cycle progression, consistent with a senescent phenotype [16,19].
In addition to these functional changes, MSCs from advanced BCPs exhibit elevated oxidative stress, characterized by increased total and mitochondrial ROS production, reduced expression of human telomerase reverse transcriptase (TERT), and impaired telomere maintenance. This phenotype is accompanied by marked transcriptional alterations, including decreased expression of stemness-associated genes (OCT4, SOX2, and TERT), reduced fibroblastic colony-forming unit (CFU-F) capacity, impaired osteogenic and adipogenic differentiation, and enhanced pro-inflammatory and pro-osteoclastogenic activity [16,20–22].
Consistent with these findings, we previously detected increased expression of genes associated with osteoclastogenesis, inflammation, and tumor promotion, including C-C motif chemokine 2 (CCL2), macrophage colony-stimulating factor, and interleukin-6 (IL-6) [16]. Notably, IL-6 and CCL2 are prominent components of the SASP and are abundantly secreted by senescent cells [23]. In parallel, alterations in soluble factor levels were observed in both peripheral blood and BM plasma from BCPs, implicating these factors in breast cancer cell intravasation, extravasation, migration, proliferation, and bone resorption [21]. Moreover, conditioned media derived from MSC-CFU-F cultures of untreated advanced BCPs enhanced the migratory capacity of human breast cancer cell lines, supporting a direct role for MSCs in facilitating tumor cell homing to the BM/bone microenvironment [21].
Among the various stressors capable of inducing senescence in MSCs, oxidative stress represents a major driver. Oxidative stress arises from an imbalance between ROS production and antioxidant defenses [24,25]. We previously reported that untreated advanced BCPs exhibit a heightened oxidative state, as assessed by elevated thiobarbituric acid–reactive substances (TBARS) and increased ROS levels in both BM plasma and peripheral blood plasma [26]. Excessive ROS generation—largely originating from mitochondrial dysfunction—damages DNA, proteins, and lipids, thereby activating oxidative stress response pathways, including heat shock protein 90 alpha (HSP90) and nuclear factor erythroid 2-related factor 2 (Nrf2), as well as downstream antioxidant genes such as superoxide dismutase 1 (SOD1), superoxide dismutase 2 (SOD2), catalase (CAT), and glutathione peroxidase 1 (GPx1). When these protective responses are insufficient, sustained molecular damage promotes MSC dysfunction and senescence [27,28].
The BM is composed of multiple interconnected compartments that must remain functionally integrated to preserve overall BM/bone homeostasis. MSCs are essential to this process, as they regulate hematopoiesis and bone metabolism, including both bone formation and resorption. Accordingly, the maintenance of a sufficient and functional MSC pool is critical to prevent PMN establishment and subsequent colonization by breast cancer cells [12].
Although oxidative stress-induced damage in the BM microenvironment of BCPs is gaining increasing attention, its impact on MSCs remains poorly understood. This study aims to characterize the metabolic and molecular alterations in BM-derived MSCs from untreated advanced BCPs, including damage to DNA, proteins, and lipids, as well as the expression of genes involved in the oxidative stress response. Furthermore, it seeks to investigate how these alterations are reflected in the MSC secretome and their potential contribution to a pro-tumoral microenvironment in this patient group. Thus, we hypothesized that oxidative stress promotes molecular damage in BM-derived MSCs, facilitating PMN formation in breast cancer.
Targeting oxidative stress and its consequences on MSCs could offer new therapeutic avenues to counteract pro-tumoral microenvironment development.
We conducted a prospective study including 10 patients diagnosed with breast cancer confirmed by histology and 10 HVs as controls. Inclusion criteria was women aged 50 to 65 years with invasive ductal breast carcinoma, clinical-pathological stage III-B, non-inflammatory (according to the TNM staging system of the International Union Against Cancer), with no prior surgery for the primary tumor, free of oncological treatment (chemotherapy, hormone therapy, immunotherapy, and/or radiotherapy), and in a menopausal state without hormonal treatment. The exclusion criteria included metastasis to the bone and/or BM or other organs, the presence of another primary tumor of the same or different origin, and diseases that could alter bone metabolism, such as osteoporosis, vitamin D deficiency, thyroid disease, parathyroid disease, and kidney damage. The absence of bone metastasis was confirmed by X-ray and bone scintigraphy, following the recommendations of the clinical guidelines established by the European Society for Medical Oncology (ESMO) [29]. Patients were selected without considering the molecular subtype of the breast tumor, given that the study group is very limited according to all the previously described inclusion criteria.
The control cohort consisted of women 45–65 years old, donors for allogeneic BM transplantation or HVs who attended the Hematology Service and had no involvement of the BM, immuno-hematological disorders, or bone issues, meaning they had no diseases that could affect bone metabolism. HVs were matched for menopausal status with breast cancer patients.
This study was conducted in accordance with the ethical principles of the Declaration of Helsinki. Ethical approval was obtained from the relevant institutional ethics committees of all participating institutions. Specifically, the study was approved by the Comité de Ética Dr. Enrique T. Segura, IBYME-CONICET (Project CE 051/2015), and by the Comité Institucional de Revisión de Ensayos Clínicos (CIREC), Hospital Militar Central (Acta 326/2014). Written informed consent was obtained from all individual participants included in the study.
2.2 Isolation and Preparation of BM-MSCs
BM aspirates were obtained from the posterior iliac crest under local anesthesia and collected in preservative-free heparinized saline (25 units/mL; cat. 15077-019, Gibco, Grand Island, NY, USA). Mononuclear cells were isolated from BM samples using a Histopaque (density = 1.075 g/cm3; cat. 10771, Sigma Aldrich, St. Louis, MO, USA) density gradient. After centrifugation for 25 min at 340× g, the light-density BM mononuclear cell (MNC) fraction was recovered from the interface, washed twice with phosphate-buffered saline (PBS; 137 mM NaCl [cat. 2000164600, Biopack Productos Químicos, Zárate, BA, Arg]; 2.7 mM KCl [cat. 2000934600, Biopack Productos Químicos, Zárate, BA, Arg]; 10 mM Na2HPO4 [cat. 2000163900, Biopack Productos Químicos, Zárate, BA, Arg]; 1.8 mM KH2PO4 [cat. 2000168907, Biopack Productos Químicos, Zárate, BA, Arg]; pH 7.4), and resuspended in supplemented α-minimal essential medium (α-MEM; cat. 11900024, Gibco, Grand Island, NY, USA) containing 2 mM L-glutamine (cat. 25030081, Gibco, Grand Island, NY, USA), 100 IU/mL antibiotic-antimycotic (cat.15240062, Gibco, Grand Island, NY, USA), and 20% heat-inactivated fetal bovine serum (FBS; cat.16000044, Gibco, Grand Island, NY, USA) [Sup-α-MEM].
Total MNC number was determined using a 3% acetic acid solution and cell viability was assessed by 0.04% trypan blue dye exclusion. Viable MNCs (10 × 106) from BM samples of HVs and BCPs were seeded into 25 cm2 tissue culture flasks containing 10 mL of Sup-α-MEM and maintained at 37°C in a humidified atmosphere with 5% CO2. After 24 h, non-adherent cells were removed and fresh medium was added. Primary cultures were expanded until reaching 75%–80% sub-confluence, with medium replacement every 7 days.
Stromal cells from one first-passage flask were washed twice with PBS, detached using a 0.05% trypsin/EDTA solution in PBS (cat. 15400054, Gibco, Grand Island, NY, USA), and replated in two new 25 cm2 flasks. Cells were allowed to proliferate until reaching 75%–80% confluence again. Following the second passage, stromal cells were plated at low density (240 cells/cm2) in 25 cm2 flasks and cultured in Sup-α-MEM for 12 days, with medium renewal on day 6. This low-density plating strategy favors enrichment of MSCs with high self-renewal and multipotency, as described by Ylöstalo et al. [30].
Surface phenotype analysis was performed to confirm that the cells from the 3rd subculture met the minimal criteria for defining MSCs established by The International Society for Cellular Therapy [31]. After this period, cells were washed twice with PBS, trypsinized, washed again, and replated in 25 cm2 flasks at a density of 3000 viable cells/cm2. Cells were cultured under the conditions described above, with medium renewal every 7 days until reaching 75%–80% confluence (4th subculture). Subsequently, MSCs were washed twice with PBS, trypsinized, counted, and used for various assays. This change in plating density was intended to increase the number of MSCs available for different assays. Importantly, this change in deeding density does not affect the characteristics of MSCs, as the same density is used in osteogenic and adipogenic plasticity assays [21,32].
2.3 Study of Bone Marrow Infiltration with Neoplastic Cells
BM infiltration by neoplastic cells was evaluated by immunocytochemistry in combination with morphological assessment. The aspirates were processed using an immunocytochemistry detection system (cat. K0673, Dako, Carpinteria, CA, USA) and cell morphology was evaluated using the Pappenheim technique. For epithelial markers detection, samples were incubated with antibodies (Abs) directed against cytokeratins AE1–AE3 (cat. IR053, Dako, Carpinteria, CA, USA) and epithelial membrane antigen (EMA; cat. M0613, Dako, Carpinteria, CA, USA). Samples were classified as positive for metastasis only when cells exhibited both cytokeratin AE1–AE3 and EMA expression together with malignant morphological features. Isotype control Abs (cat. 08-6599, ZYMED Laboratories, South San Francisco, CA, USA, and cat. X0943, Dako, Carpinteria, CA, USA) were included in parallel, using the same antibody concentrations as the corresponding primary antibodies. Each experiment was performed twice for every sample.
2.4 Phenotypic Characterization of Mesenchymal Stem/ Stromal Cells
MSCs from the 3rd subculture were suspended in PBS containing 1% bovine serum albumin (BSA; cat. A7030, Sigma Aldrich, St. Louis, MO, USA) and incubated with fluorochrome-conjugated primary Abs directed against the following human surface markers: CD105 (cat. FAB10971V, R&D Systems Inc., Minneapolis, MN, USA), CD90 (cat. FAB2067G, R&D Systems Inc., Minneapolis, MN, USA), CD73 (cat. FAB5795A, R&D Systems Inc., Minneapolis, MN, USA), CD79a (cat. FAB69201G, R&D Systems Inc., Minneapolis, MN, USA), CD11b (cat. FAB1699V, R&D Systems Inc., Minneapolis, MN, USA), CD34 (cat. FAB7227P, R&D Systems Inc., Minneapolis, MN, USA) and C-C motif chemokine receptor 2 (CCR2) (cat. FAB151P, R&D Systems Inc., Minneapolis, MN, USA). Corresponding isotype-matched control Abs (cat. IC002V, IC003G, IC0041A, IC002G, IC002V, IC002P, IC0041P, R&D Systems Inc., Minneapolis, MN, USA) were processed in parallel at identical concentrations.
After incubation for 30 min at room temperature, samples were acquired on a FACScanto II flow cytometer (BD Biosciences, San Diego, CA, USA). A minimum of 10,000 events per sample were recorded and analyzed relative to the corresponding isotype controls. Data analysis and histogram generation were performed using FlowJo software (v. X, Tree Star Inc., Ashland, OR, USA). All experiments were conducted in duplicate using independent MSC preparations.
2.5 Quantitative Real-Time PCR
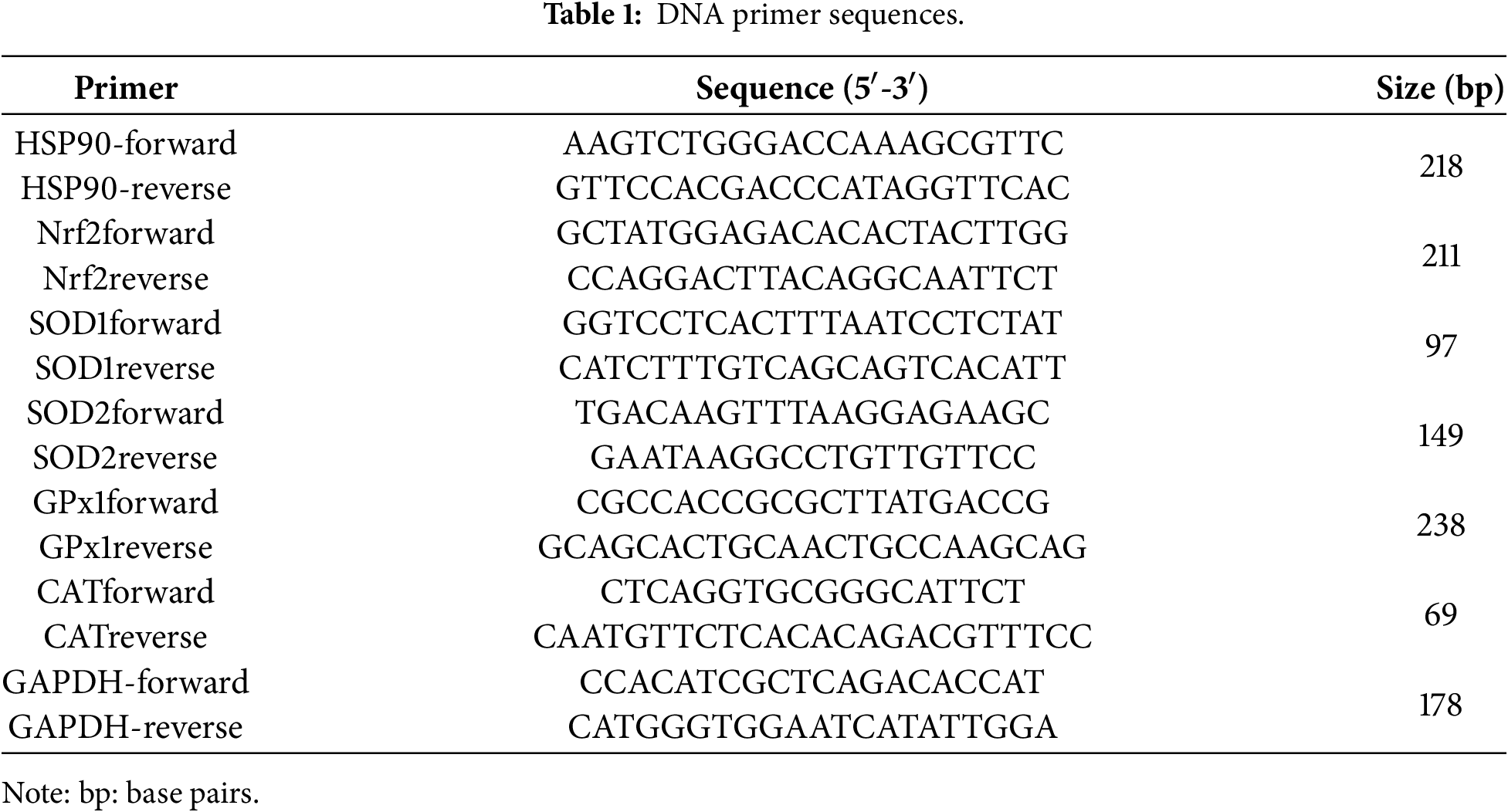
Total ribonucleic acid (RNA) was extracted from MSCs of the 4th subculture (BCPs, n = 10 and HVs, n = 10) using TRI Reagent® (cat. TR 118, Molecular Research Center Inc., Cincinnati, OH, USA). Reverse transcription was carried out using 1 µg RNA and random primers (cat. 4368814, Applied Biosystems, Foster City, CA, USA) to generate copy DNA (cDNA). Quantitative PCR was performed in duplicate using SYBR Green master mix (cat. 04913850001, ROCHE, Mannheim, Germany) on a real-time PCR system (CFX96™ TOUCH REAL-TIME PCR, Bio-Rad, Hercules, CA, USA) under standard cycling conditions. Amplification specificity was verified by melting curve analysis. The threshold cycle (Ct) values were normalized against the reference gene glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and data are presented as the fold change in gene expression, relative to the HVs group. Primer sequences are provided in Table 1.
2.6 A Single-Cell Gel Electrophoresis Assay (Comet Assay)
To detect DNA damage, the comet assay was performed under alkaline conditions, following the protocol described in Collins et al. [33]. Briefly, 2 × 104 MSCs (BCPs, n = 7 and HVs, n = 5) from the 4th subculture were resuspended in 75 μL of 1% low-melting-point (LMP) agarose (cat. V2111, Promega, Madison, WI, USA) solution and seeded onto a pre-treated glass slide (cat. D100002, Eurotubo Deltalab SL, Barcelona, Spain) with 1% normal-melting-point (NMP) agarose (cat. A4718, Sigma Aldrich, St. Louis, MO, USA). Subsequently, a third layer of LMP agarose was applied. The seeded slides were placed in a cold lysis solution (0.5 M NaCl, 100 mM EDTA [cat. E5134, Sigma Aldrich, St. Louis, MO, USA], 10 mM Tris [cat. 2000166800, Biopack Productos Químicos, Zárate, BA, Arg.], pH 10, 1% Triton X-100 [cat. X100, Sigma Aldrich, St. Louis, MO, USA]) and maintained at 4°C for at least 18 h. After this period, they were stabilized in PBS at 4°C for 1 h. Immediately after, the slides were transferred to the electrophoretic tank containing alkaline solution (300 mM NaOH [cat. 1223, Laboratorios Cicarelli, San Lorenzo, SF, Arg], 1 mM EDTA, pH 13) and kept for 20 min at 4°C in the dark. For the electrophoresis run, a current of 20 V (constant voltage) was applied for 30 min in the dark. The slides were then removed and incubated in neutralization solution (0.4 M Tris, pH 7.5) for 5 min and stained with 20 μL of ethidium bromide (20 μg/mL; cat. H5041, Promega, Madison, WI, USA). The nucleoids (cytoplasmic area containing the cell’s genetic material) were evaluated using a fluorescence microscope. A total of 100 comets per sample were randomly scored based on the intensity of fluorescence, tail length, and nucleoid integrity. The comets were classified into 5 categories, ranging from C0 (Class 0, no DNA damage, no detectable tail) to C4 (Class 4, high level of DNA damage, almost all DNA in the comet tail). Based on this classification, a score was calculated according to the following formula: score [arbitrary units] = nC1 + nC2 · 2 + nC3 · 3 + nC4 · 4, resulting in values between 0 and 400, where n is the number of comets counted in each category.
Untreated human umbilical cord MSCs (HUC-MSCs) were used as negative controls, and 0.1 mM H2O2-treated HUC-MSCs were used as positive controls (kindly donated by Dr. Esteban Fiore, Instituto de Investigaciones en Medicina Traslacional-CONICET, Facultad de Ciencias Biomédicas, Universidad Austral, Provincia de Buenos Aires, Argentina) [33]. The experiments were repeated twice for each sample.
2.7 Chromosomal Aberrations Analysis
The methodology used was described by de Campos-Nebel et al. [34]. MSCs (BCPs, n = 5 and HVs, n = 3) from the 3rd subculture were seeded at a density of 3000 cells/cm2 in 25 cm2 flasks (4th subculture) and cultured in Sup-α-MEM until reaching 50% confluence (exponential growth phase). Subsequently, the culture medium was changed to Sup-α-MEM containing, in this opportunity, 30% FBS and 0.04 μg/mL of colcemid (N-desacetyl-N-methylcolchicine; cat. 15212012, Gibco, Grand Island, NY, USA) for 18 h. Afterwards, cells were washed twice with PBS and treated with a solution of 0.05% trypsin-0.02% EDTA (cat. 15400054, Gibco, Grand Island, NY, USA). To the cell suspension, 10% FBS was added and centrifuged at 448× g for 10 min. Cells were re-suspended in a hypotonic solution of 75 mM KCl for 35 min, fixed in a mixture of methanol (cat. 2000160900, Biopack Productos Químicos, Zárate, BA, Arg) and acetic acid (3:1; cat. 1025110, Laboratorios Cicarelli, San Lorenzo, SF, Arg) for 24 h at −20°C, dropped onto slides, and stained with 10% Giemsa (cat. 2000110000, Biopack Productos Químicos, Zárate, BA, Arg) in water for 4 min. Fifty metaphase cells were analyzed per sample to determine the number of CAs under a bright-field microscope with a 100× objective (CX31, Olympus, Tokyo, Japan). Any metaphase cell presenting at least one CA was considered abnormal. The CAs were classified as chromatid breaks (CB), acentric fragments (AF) and chromatid exchanges [telomeric associations (TAS), ring chromosomes (RC), and dicentric chromosomes (DIC)] [35].
2.8 Quantification of Nuclear Blebs and Micronuclei
MSCs (BCPs, n = 6 and HVs, n = 6) from the 3rd subculture were seeded at a density of 3000 cells/cm2 on glass coverslips in 6-well plates (cat. 4430300, Orange Scientific, Braine-l’Alleud, Belgium) and cultured in Sup-α-MEM until reaching 50% confluence (4th subculture). After this period, the culture was washed twice with PBS and fixed with a 4% paraformaldehyde (PFA) solution in PBS for 15 min at room temperature. They were then stained with pure Giemsa for 5 min, mounted and analyzed under a bright-field microscope with a 100× objective (Eclipse E200, Nikon Instruments Inc., Tokyo, Japan). The frequency of NBs and MN was evaluated per 1000 cells [36].
2.9 Oxidative Damage to Proteins
Protein carbonyls are a general marker of protein oxidation generated by various radicals. The experiments were conducted with protein lysates from MSCs (BCPs, n = 4 and HVs, n = 5) of the 4th subculture. Viable cells (1 × 105) were resuspended in 100 μL of Thermo Scientific™ NP-40 lysis buffer (cat. J60766.AP, Thermo Scientific™, Waltham, MA, USA) supplemented with 1× Complete™ Protease Inhibitor Cocktail (cat. 11873580001, Roche, Mannheim, Germany). The lysates were incubated on ice for 30 min and then centrifuged at 10,000× g for 20 min at 4°C. The supernatant was collected, and protein concentrations were measured using the DC Protein Assay (cat. 5000111, Bio-Rad, Hercules, CA, USA). Carbonyl concentrations can be assessed via their reaction with 2,4-dinitrophenylhydrazine (DNPH; cat. D199303, Sigma Aldrich, St. Louis, MO, USA) to form hydrazone, a yellow product that can be quantified spectrophotometrically. To quantify total protein carbonyl concentrations, the protocol described by Hawkins et al. “Quantification of protein carbonyls: Total carbonyls on proteins” was followed [37]. Protein oxidation was also assessed in the conditioned medium of MSCs (secretome). For this assay, once reaching 75%–80% confluence at the 4th subculture, some of the cell cultures were washed twice with PBS and incubated at 37°C, 5% CO2, and high humidity for 48 h in α-MEM with 2 mM L-glutamine, 100 IU/mL antibiotic/antimycotic, but without FBS supplementation. After this period, the conditioned media (CM) were collected, centrifuged at 250× g for 10 min, aliquoted, and stored at −80°C until evaluation of carbonyl protein. All measurements were performed in duplicate.
2.10 Oxidative Damage to Lipids
Lipid peroxidation was indirectly assessed through the study of the presence of 4-hydroxynonenal (4-HNE) protein adducts in protein lysates from MSCs (BCPs, n = 4 and HVs, n = 3) of the 4th subculture. The 4-HNE protein adducts refer to the chemical modifications that occur in proteins due to the binding with this byproduct. 4-HNE is a product of lipid peroxidation, a process in which lipids present in cell membranes are damaged by ROS, producing reactive compounds [38,39]. Protein lysates were obtained from viable cells following the protocol previously described. A total of 25 µg of protein per sample was resolved in a 12% sodium dodecyl sulfate-polyacrylamide (SDS-PAGE) gel. The resolved protein bands were electroblotted onto a polyvinylidene fluoride membrane (PVDF; cat. 1620177, Bio-Rad, Hercules, CA, USA). Molecular weights were determined using the PageRuler™ (cat. 26616, Thermo Scientific™, Vilnius, LT, Lithuania). Membranes were then blocked for 45 min in 1× Tris-buffered saline (TBS; 50 mM Tris-HCl [cat. H5123, Promega, Madison, WI, USA]; 150 mM NaCl; pH 7.4) containing 0.1% TWEEN 20 (T-20) and 5% (w/v) non-fat dried milk, and subsequently incubated overnight at 4°C with primary Abs against 4-HNE monoclonal Abs (cat. Ab243070, ABCAM, Cambridge, MA, USA) and β-actin (cat. A5441, Sigma Aldrich, St. Louis, MO, USA), used as a loading control. Subsequently, the membranes were washed with washing buffer (0.01% T-20 in TBS) and incubated with the horseradish peroxidase (HRP)-conjugated secondary Abs (cat. 170-6516, BIORAD, Hercules, California, USA) for 1 h at room temperature. Finally, the membranes were washed three times with washing buffer and the bound Abs were detected using the chemiluminescent detection system (cat. 1705060, BioRad, Hercules, California, USA) according to the manufacturer’s instructions and visualized by exposure to photographic films. The developed films were subsequently digitized using the ChemiDoc MP Imaging System (BIORAD, Hercules, CA, USA). The bands were quantified by densitometry using ImageJ (V 1.5.4, National Institutes of Health, Bethesda, MD, USA). The density of the protein bands was normalized to β-actin expression.
2.11 Collection of Conditioned Media for Proteomic Analysis
After the collection and storage of the CM, 10 mL of each secretome were taken and pooled into BCPs-CM (BCPs, n = 6) and HVs-CM (HVs, n = 6) according to their respective groups. The total 60 mL from each study group was then lyophilized for 48 h in a laboratory freeze-drying system, following the manufacturer’s instructions (Modulyod 115, Thermo Fisher, Carlsbad, CA, USA). The lyophilized samples were reconstituted in 4 mL of sterile H2O (cat. 956-A, Rivero, CABA, Arg). The corresponding concentrated media (~15-fold) from each study group was used for proteomic analysis. Protein concentration was determined using the Bradford method with BSA (cat. sc-2323, Santa Cruz, CA, USA) as the standard [40].
2.12 Protein Content Analysis of Conditioned Media (Secretome) from Mesenchymal Stem/Stromal Cells
Thirty micrograms of proteins per lane were subjected to 1D electrophoresis on a 10% SDS-polyacrylamide gel under reducing conditions, with each sample (BCPs-CM and HVs-CM) loaded in triplicate. Electrophoresis was carried out at a constant voltage of 80 V until the front reached 1 cm below the stacking gel.
Immediately after electrophoresis, triplicates of each sample were fixed with fixing buffer (30% v/v ethanol [cat. E7023, Sigma Aldrich, St. Louis, MO, USA] and 2% v/v phosphoric acid [cat. 93752, Sigma Aldrich, St. Louis, MO, USA] in H2O) with agitation overnight at room temperature.
After fixation, gels were washed three times for 30 min with H2O and equilibrated in equilibration buffer (18% v/v methanol, 17% w/v ammonium sulfate (cat. AX1385, Sigma Aldrich, St. Louis, MO, USA), and 2% v/v phosphoric acid in sterile H2O) with agitation for 1 h at room temperature. Subsequently, Coomassie Brilliant Blue G-250 (cat. 1610406, BioRad, Hercules, California, USA) was added to the buffer by sprinkling, reaching a final concentration of 0.5 g/L (staining solution). The gel was incubated in this solution overnight with agitation at room temperature. The next day, the gel was washed three times for 30 min with H2O, and protein bands were excised from the polyacrylamide gel using a sterile scalpel and placed in individual tubes for further analysis. Proteomic analysis was conducted by the Centro de Estudios Químicos y Biológicos por Espectrometría de Masa (CEQUIBIEM) at the Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, using the label-free protein quantification (LFQ) proteomic method. Briefly, the samples were reduced with 20 mM Ditiotreitol (DTT; cat. 43816, Sigma Aldrich, St. Louis, MO, USA) for 45 min at 56°C and alkylated with 50 mM iodoacetamide (cat. I1149, Sigma Aldrich, St. Louis, MO, USA) for 45 min in the dark. They were then digested with trypsin (cat. T7575, Sigma Aldrich, St. Louis, MO, USA) overnight. Peptides were extracted using acetonitrile (cat. 100029; Merck, Darmstadt, Germany). The samples were lyophilized using a Speed Vac (Thermo Fisher Scientific, Waltham, MA, USA) and then resuspended in 30 µL of 0.1% trifluoroacetic acid (cat. 80457, Sigma Aldrich, St. Louis, MO, USA). Desalting was performed using a ZipTip C18 (cat. ZTC18S096, Merck Millipore Ltd., Germany) and separated by nanoLC (EASY-nLC 1000, Thermo Fisher Scientific, Waltham, MA, USA) before being analyzed by tandem mass spectrometry (spectrometer Q-Exactive, Thermo Fisher Scientific, Waltham, MA, USA) with Orbitrap technology (nano HPLC-ESI-MS/MS). A voltage of 1.5–3.5 kV was used for ElectroSpray Ionization (EASY-SPRAY, Thermo Fisher Scientific, Waltham, MA, USA). The MS equipment features a high collision dissociation cell (HCD) for fragmentation and an Orbitrap analyzer (Q-Exactive, Thermo Fisher Scientific, Waltham, MA, USA). XCalibur software (V 3.0.63, Thermo Fisher Scientific, Waltham, MA, USA) was used for data acquisition. Raw spectra were analyzed using Proteome Discoverer software (version 2.2, Thermo Fisher Scientific, Waltham, MA, USA) for protein identification. Spectra were searched against the UniProt proteome reference from the Homo sapiens database UP000005640 using standard settings. Statistically significant differences in relative abundances between the two analyzed groups (BCPs-CM and HVs-CM) were assessed using Perseus software (V 2.0.11, available at https://maxquant.net/perseus/). Differentially expressed proteins (DEPs) in BCPs and HVs were identified using a two-sided Student’s t-test in combination with a permutation-based false discovery rate correction (FDR).
To gain molecular insight into the DEPs identified, a Gene Ontology (GO) enrichment analysis was performed for biological processes using the Database for Annotation, Visualization, and Integrated Discovery (DAVID; v2023q4, NIH, Bethesda, MD, USA) [41], applying an EASE threshold of 0.05 and a Count threshold of 2 in all cases.
Results are given as the mean ± standard error (SE) when appropriate. Data normality was assessed using the Shapiro–Wilk test. For comparisons between two groups, an unpaired Student’s t-test with Welch’s correction was applied when data followed a normal distribution, whereas the Mann–Whitney U test was used for non-normally distributed variables. Categorical data were analyzed using the Chi-square test when expected frequencies met test assumptions. For proteomic analyses, a permutation-based FDR = 0.05 correction was used. Differences were considered statistically significant when p < 0.05.
3.1 Study of Bone Marrow Infiltration with Neoplastic Cells
Bone marrow aspirates from the posterior iliac crest showed no evidence of infiltration by neoplastic cells in any of the cancer patients studied.
3.2 Mesenchymal Stem/ Stromal Cell Phenotypic Characterization
MSCs at the third passage were phenotypically characterized by flow cytometry using classic surface markers. Over 95% of MSCs from both groups (BCPs, n = 10 and HVs, n = 10) expressed CD73, CD105, and CD90, while showing no expression of CD11b, CD34, or CD79a (Fig. 1). Thus, the MSCs employed in this study meet the minimal criteria established for defining MSCs [31].
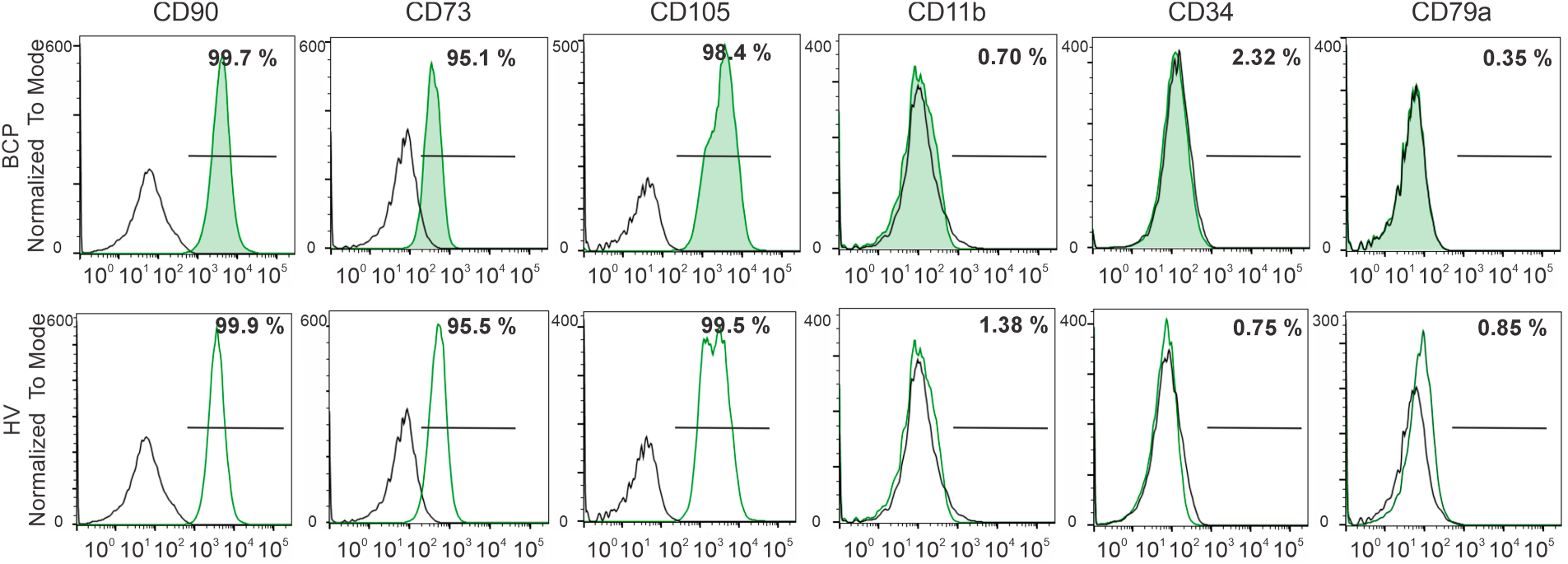
Figure 1: Phenotypic characterization of bone marrow-derived mesenchymal stem/ stromal cells (BM-MSCs). Representative flow cytometry histograms of MSC surface antigens from a breast cancer patient (BCP) and healthy volunteer (HV). Isotype controls are shown as open black histograms, whereas specific surface antigen staining is shown in green histograms (shaded in BCP samples and open in HV samples). The percentage of positive cells for each marker is indicated. CD: Cluster of Differentiation.
3.3 Expression of CCR2 in Mesenchymal Stem/Stromal Cells
It has been reported that soluble factors such as CCL-2, via CCR2, drive MSC senescence through ROS [42]. These observations led us to investigate CCR2 expression in MSCs from BCPs, as previous studies showed elevated CCL-2 levels in their conditioned media compared with HVs [20]. Thus, the CCL2/CCR2 interaction may enhance ROS accumulation, thereby promoting oxidative modifications of lipids, proteins, and DNA. Notably, although only a small subset of BM-MSCs expressed CCR2, the proportion of CCR2+ cells was significantly higher in BCPs than in HVs (Fig. 2a), while the mean fluorescence intensity of CCR2+ cells remained comparable between groups (Fig. 2b), indicating that the difference arises from a higher frequency of positive cells rather than from increased receptor density per cell.
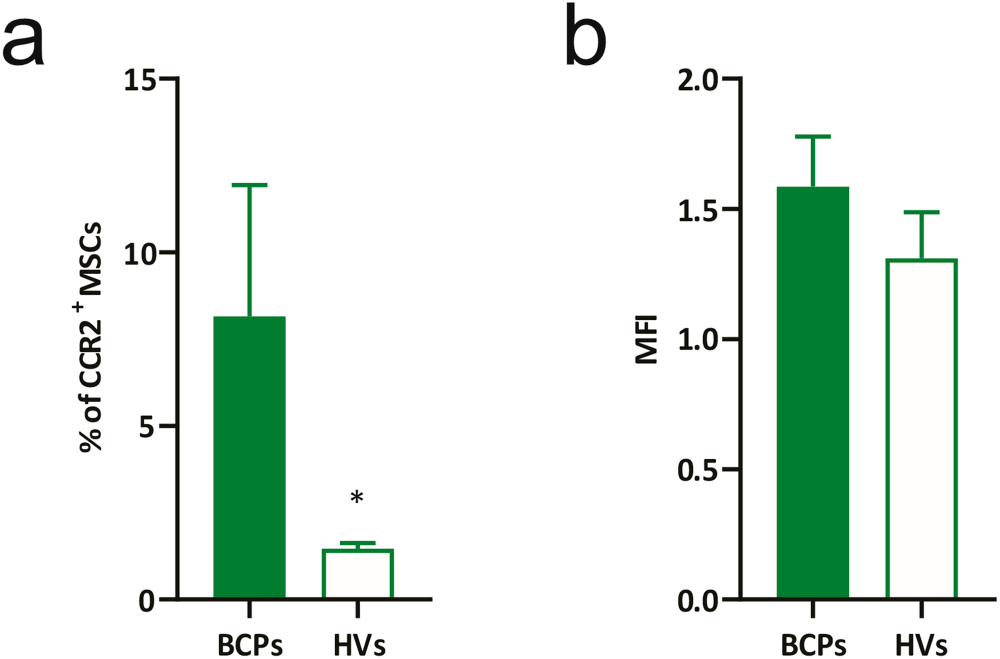
Figure 2: Expression of CCR2 in mesenchymal stem/ stromal cells (MSCs). (a) Percentage of CCR2+ MSCs from breast cancer patients (BCPs, n = 6) and healthy volunteers (HVs, n = 6). Values are expressed as mean ± standard error. Statistical analysis: Mann–Whitney U test. Asterisks indicate a significant difference (*p < 0.05). (b) Relative fluorescence intensity of CCR2 in MSCs from BCPs (n = 6) and HVs (n = 6). Median fluorescence intensity (MFI), values were normalized to the HV group. Values are expressed as mean ± standard error. CCR2: C-C Motif Chemokine Receptor 2.
3.4 Increased Expression of Oxidative Stress Response Genes in Mesenchymal Stem/ Stromal Cells from Breast Cancer Patients
Our recent studies demonstrated that BM-MSCs from this patient group exhibit increased oxidative stress [16]. In this regard, we assessed the expression of genes related to oxidative stress response, such as HSP90 and Nrf2, as well as antioxidant response genes, including SOD1, SOD2, CAT, and GPx1. Our findings revealed that MSCs from BCPs showed overexpression of both oxidative stress-related genes and antioxidant response genes (Fig. 3).
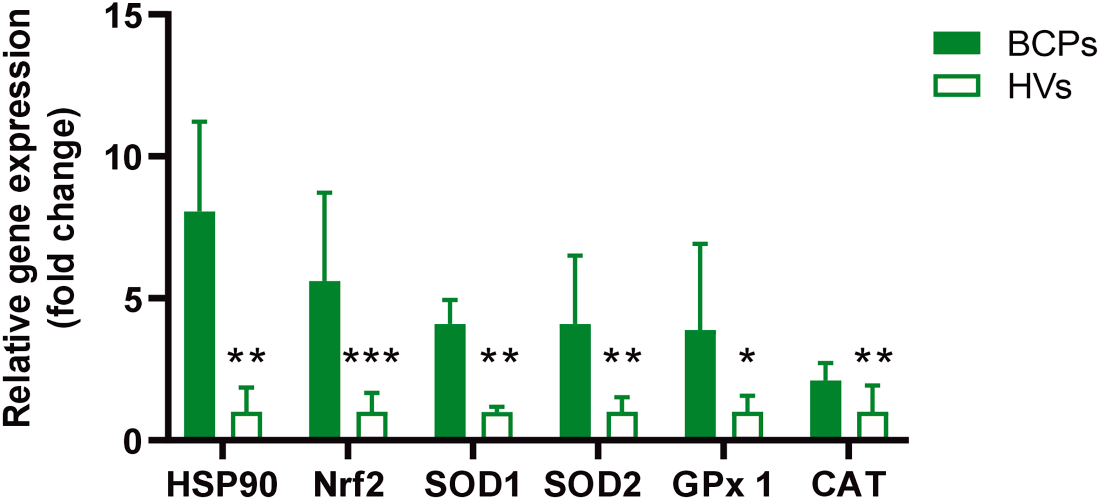
Figure 3: Expression of oxidative stress response factors in bone marrow mesenchymal stem/ stromal cells (MSCs). Samples of MSCs were obtained from breast cancer patient (BCPs, n = 10) and healthy volunteers (HVs, n= 10). The expression of specific genes (HSP90, Nrf2, SOD1, SOD2, GPx1, and CAT) was quantitative by real-time polymerase chain reaction (PCR) assay. All results were normalized to a reference gene GAPDH and are presented relative to the expression level in MSCs from HVs. Values are expressed as mean ± standard error. Statistical analysis: unpaired t-test with Welch’s correction. Asterisks indicate a significant difference (*p < 0.05, **p < 0.01 and ***p < 0.001). HSP90: heat shock protein 90 alpha; Nrf2: nuclear factor erythroid 2-related factor 2; SOD1: superoxide dismutase 1; SOD2: superoxide dismutase 2; GPx1: glutathione peroxidase 1; CAT: catalase.
3.5 Enhanced Oxidative Damage in Mesenchymal Stem/ Stromal Cells from Breast Cancer Patients
The comet assay was used to evaluate DNA damage in MSCs from BCPs and HVs. In this technique, damaged DNA migrates from the cell nucleus in a manner resembling the tail of a comet [43]. The more substantial the DNA breakage/damage, the more pronounced the comet tail will be (Fig. 4a). Positive and negative technical controls were performed in parallel using HUC-MSCs treated or not with 0.1 mM H2O2 (Fig. 4b). The results of this assay show that MSCs from BCPs form comets with morphologies more associated with DNA damage compared to those from HV samples (Fig. 4c). The comet score calculation revealed an increase for BCPs, indicating a higher average DNA damage in MSCs from this group (Fig. 4d).
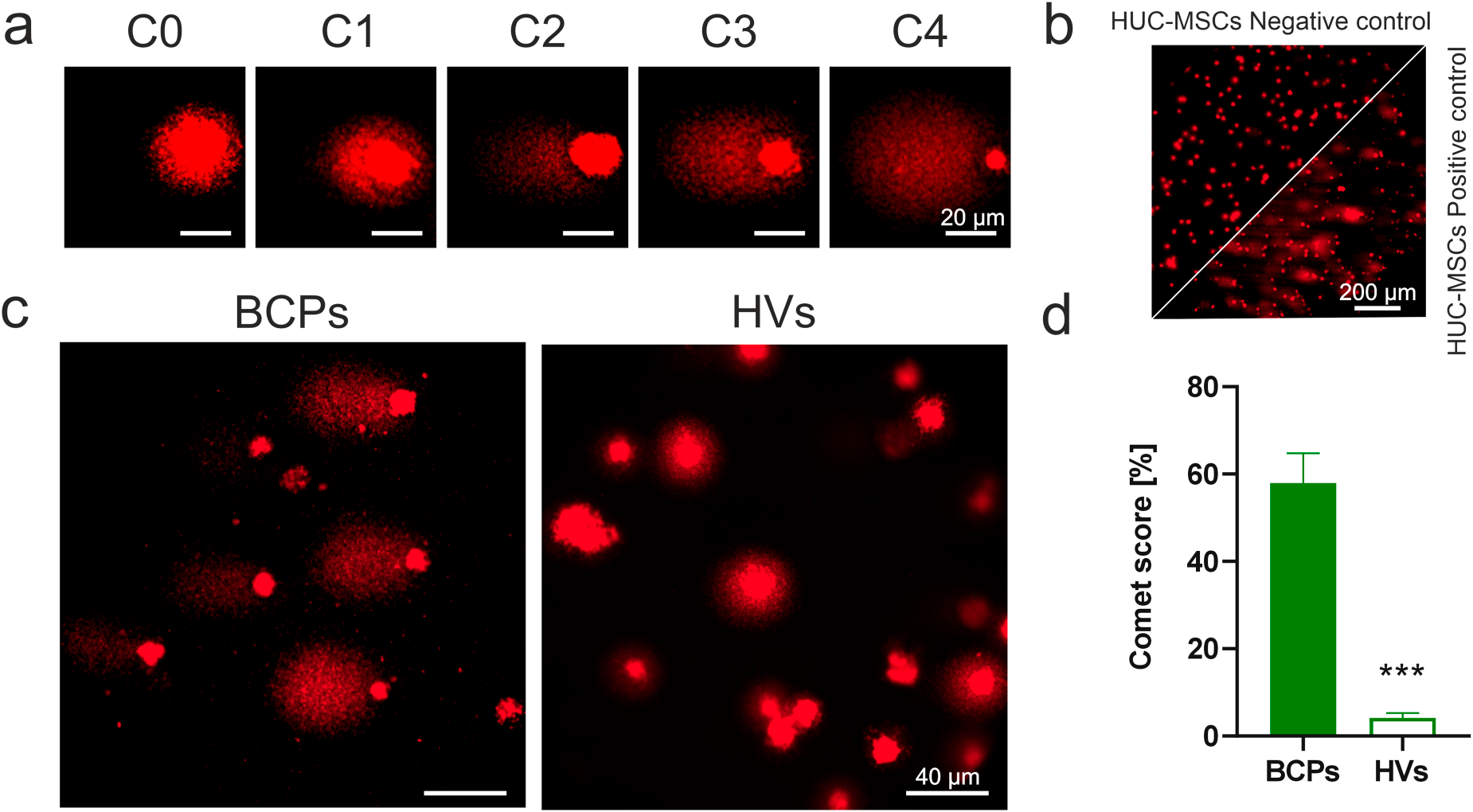
Figure 4: DNA oxidative damage in mesenchymal stem/stromal cells (MSCs) from bone marrow (BM) in breast cancer patients (BCPs). (a) BM-MSCs from BCPs processed using comet assay and stained with Ethidium Bromide and analyzed by fluorescence microscopy. Comets were classified into five categories (C0–C4). (b) Technical controls: untreated human umbilical cord MSCs (HUC-MSCs; negative control, left) and HUC-MSCs treated with 0.1 mM H2O2 (positive control, right). (c) Representative panel, MSCs from a BCP and a healthy volunteer (HV). (d) Comet score expressed as a percentage of BCPs (n = 7) and HVs (n = 5). Values are expressed as mean ± standard error. Statistical analysis: unpaired t-test with Welch’s correction. Asterisk indicates a significant difference (***p < 0.001).
3.5.2 Breast Cancer Patients Exhibit a Higher Proportion of Bone Marrow Mesenchymal Stem/ Stromal Cells with Chromosomal Aberrations, Nuclear Blebs, and Micronuclei
ROS can cause DNA strand breaks and other types of damage, which, if not properly repaired, can lead to chromosomal aberrations (Fig. 5a,b). When evaluating the number of abnormal cells, defined as the number of metaphases with at least one CA out of the total metaphase cells assessed, we found a trend towards an increased number of abnormal MSCs in the BCPs group compared to the HVs group (Fig. 5c).
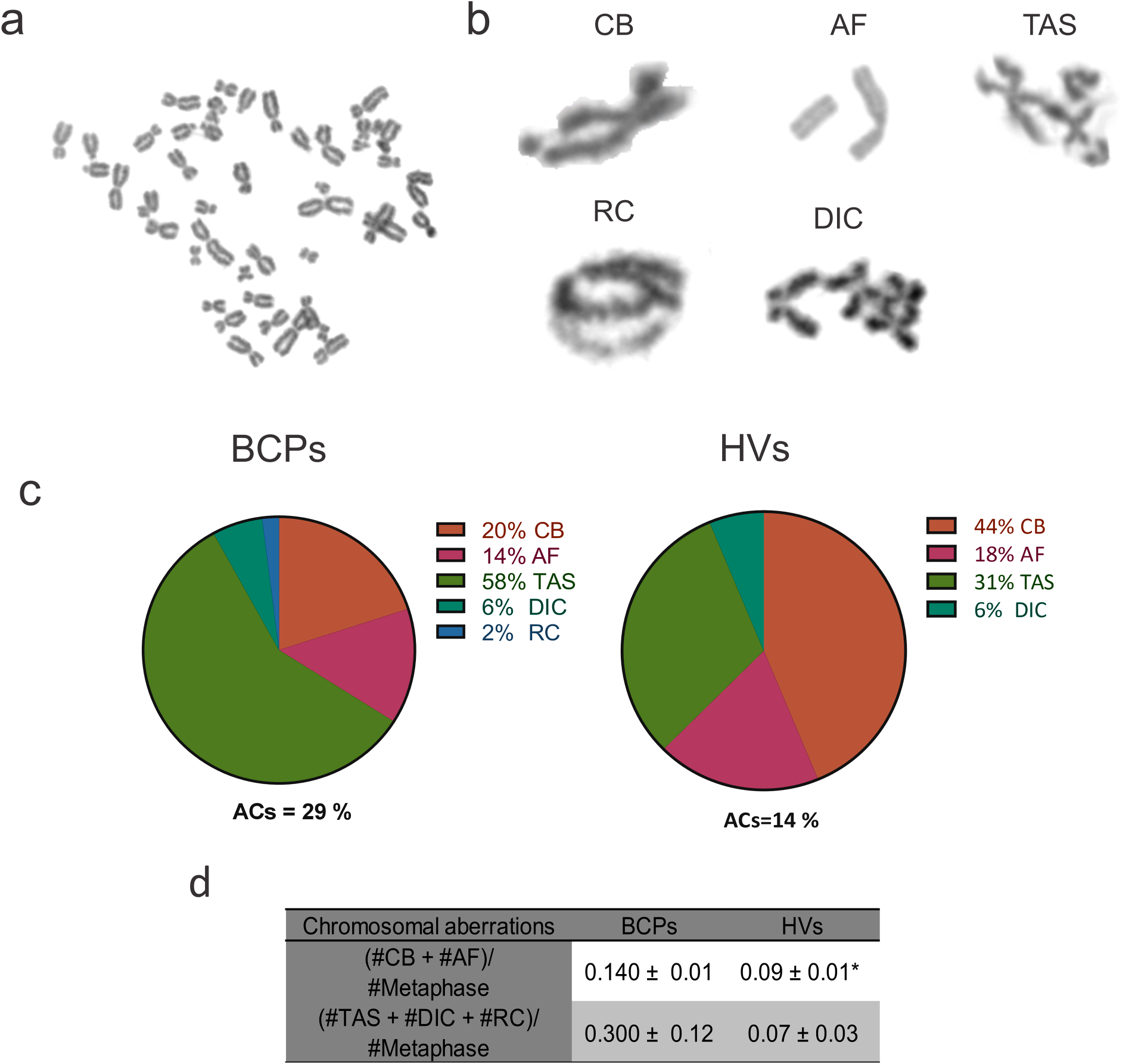
Figure 5: Structural chromosomal aberrations observed in mesenchymal stem/stromal cells (MSCs) from breast cancer patients (BCPs). (a) Normal metaphase (without chromosomal aberrations) in a BCP, 2n = 46, XX. (b) Examples of observed structural chromosomal aberrations: (i) Chromatid break (CB), (ii) Acentric fragment (AF), (iii) Telomeric association (TAS), (iv) Ring chromosome (RC) and (v) Dicentric chromosome (DIC). (c) Diagram of the percentage distribution of structural chromosomal aberrations in abnormal MSCs with chromosomal aberrations in metaphase in BCPs (n = 5) and healthy volunteers (HVs, n = 3). ACs: abnormal cells. (d) Clustered chromosomal aberration analysis. Values are expressed as mean ± standard error. Statistical analysis: Chi-square test. Asterisks indicate a significant difference (*p < 0.05).
Furthermore, when analyzing the frequency of aberrations categorized by the number of breaks required for their occurrence—one break for CB and AF, and two breaks for TAS, RC, and DIC—we observed that the frequencies were significantly higher for the BCPs compared to the HVs (Fig. 5c,d).
Another indicator of damage and oxidative stress is the formation of NBs and/or MN. In this regard, we found NBs and MN in all MSC samples observed from both BCP and HV groups (Fig. 6a,b). It is noteworthy that multinucleated cells and metaphase cells with lagging chromosomes were also identified (Fig. 6c,d). Moreover, when evaluating the percentage of MSCs with NBs and MN, a higher percentage was observed in the BCPs group compared to the HVs group (Fig. 6e,f).
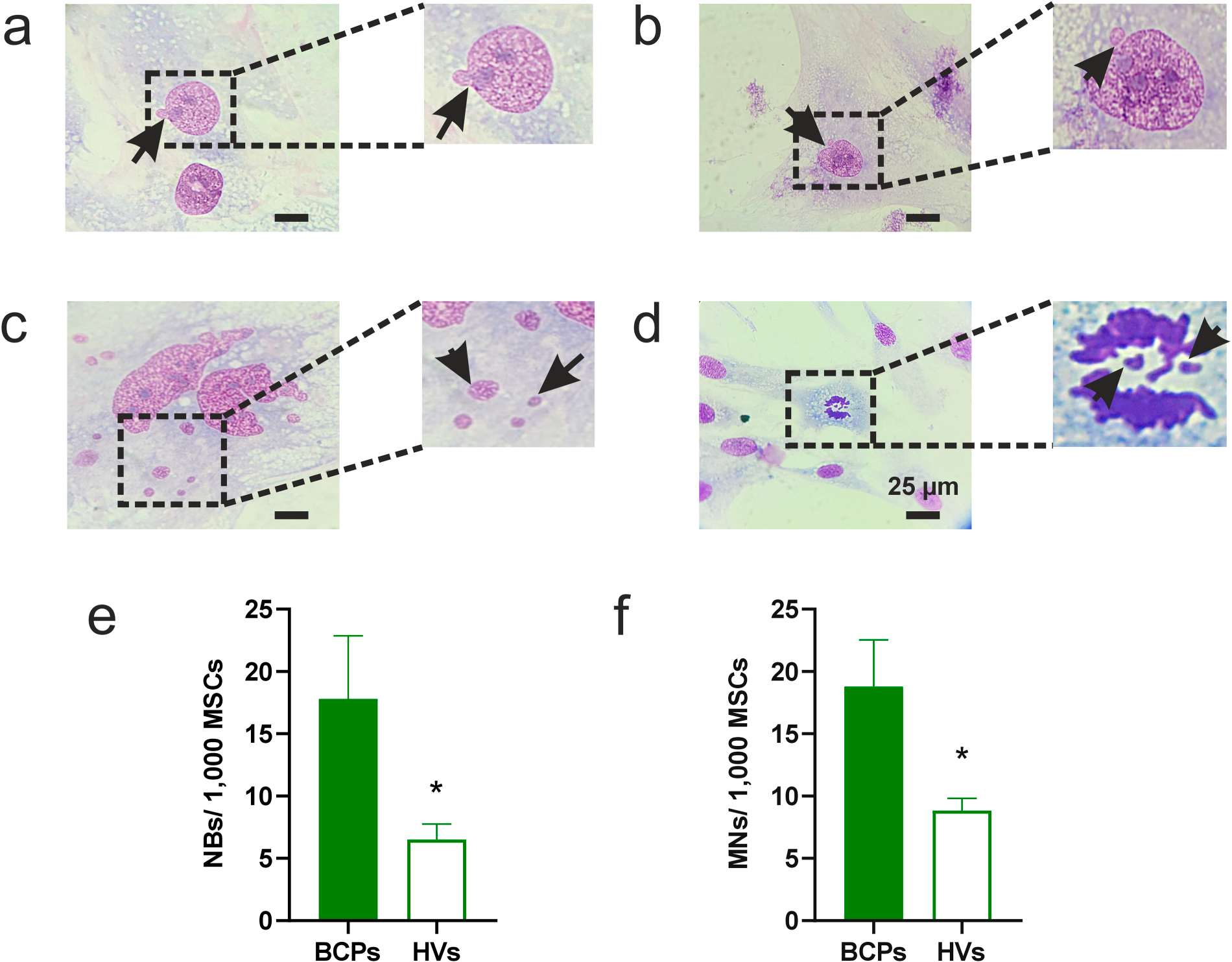
Figure 6: Frequency of mesenchymal stem/stromal cells (MSCs) with nuclear blebs (NBs) and micronuclei (MN) in breast cancer patients (BCPs). (a) NBs. The arrow indicates the NBs observed in an MSC from a BCP. (b) MN. The arrow indicates the MN observed in an MSC from a BCP. (c) Multimicro-nucleated MSC from a BCP. Arrows show the multiple MN. (d) Anaphase lag in MSC from a BCP under standard culture conditions. Arrows indicate lagging chromosomes, which lead to the formation of MN. Microphotographs were taken at a total magnification of 1000×, localized zoom 2×. (e) Differences observed for NBs in the BCPs (n = 6) and healthy volunteers (HVs, n = 6). (f) Differences observed for MN in the BCPs (n = 6) and HVs (n = 6). Values are expressed as mean ± standard error. Statistical analysis: unpaired t-test. Asterisk indicates a significant difference (*p < 0.05).
3.5.3 Protein and Lipid Damage
Interestingly, and in accordance with the previous results, when evaluating oxidative damage in protein extracts of MSCs, increased values of carbonylated proteins were found in the BCPs group compared to the HVs group (Fig. 7a). At the same time, the assessment of protein oxidation in the CM of MSCs (Secretome) revealed an increase in carbonylated proteins for the BCPs group (Fig. 7b). Regarding oxidative damage in lipids, it was assessed through the presence of 4-HNE, a byproduct of lipid peroxidation formed when lipids in cell membranes undergo oxidative damage. SDS-PAGE analysis revealed an increase in the abundance of adducts in a subset of proteins of various sizes, ranging from approximately 250 to 75 kDa, in MSCs from BCPs compared to the HVs (Fig. 7c,d).
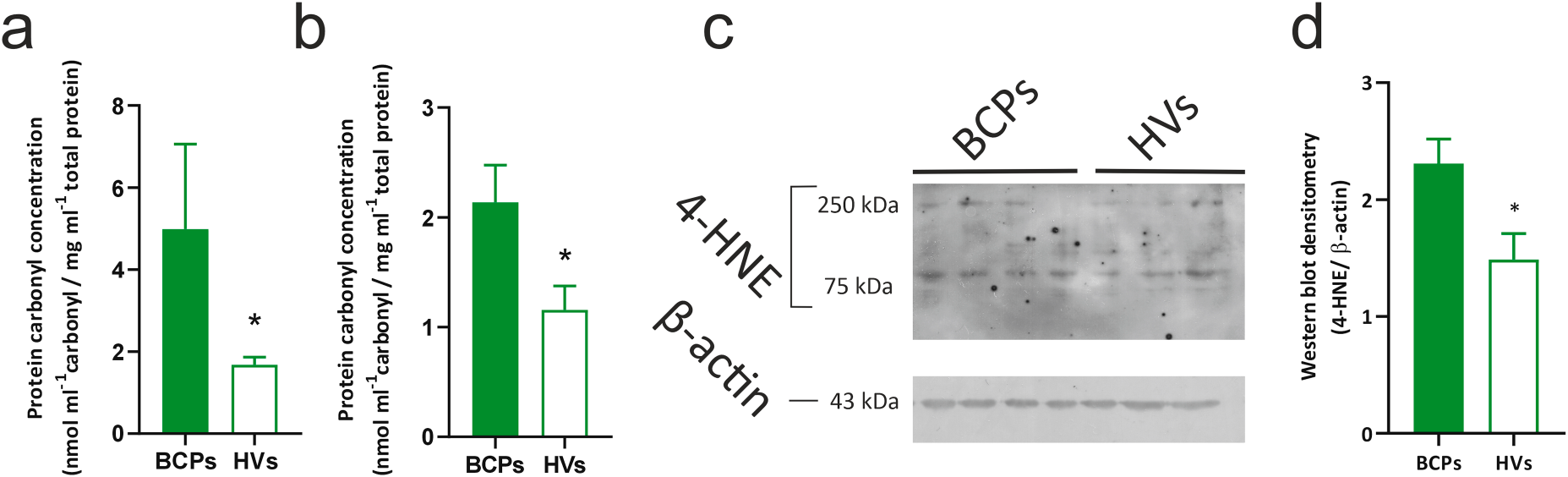
Figure 7: Oxidative damage to proteins and lipids in bone marrow-derived mesenchymal stem/ stromal cells (MSCs) from breast cancer patients (BCPs) and conditioned media from BCPs and healthy volunteers (HVs). (a) Carbonylated protein content in protein extracts from MSCs of BCPs (n = 4) and HVs (n = 5). Values are expressed as mean ± standard error (SE). (b) Carbonylated protein content in conditioned media from MSCs of BCPs (n = 4) and HVs, (n = 5). Values are expressed as mean ± SE. (c) Immunoblot of bone marrow-derived MSC proteins extracts from BCPs and HVs, using antibodies against 4-hidroxinonenal (4-HNE) and β-actin. (d) Abundance of 4-HNE protein in MSC protein extracts from BCPs (n = 4) and HVs (n = 3). The expression of 4-HNE and β-actin was analyzed by immunoblotting, and the images were analyzed by densitometry. Values are expressed as mean ± SE. Statistical analysis: unpaired t-test with Welch’s correction. Asterisks indicate a significant difference (*p < 0.05).
3.5.4 Characterization of the Secretome of Mesenchymal Stem/Stromal Cells from Breast Cancer Patients
It is well known that MSCs release a variety of paracrine factors, including cytokines, growth factors, and extracellular vesicles, which influence the surrounding microenvironment. The study of the secretome of BM-MSCs in the PMN context of these BCPs is crucial, as the factors they release play a key role in creating an optimal environment for extravasation and interaction with circulating cancer cells.
In this study, a total of 141 proteins were identified (Fig. 8a,b), of which 7 were differentially expressed in the BCPs group and 34 in the HVs group (Tables 2 and 3). The 100 proteins commonly expressed in both groups are listed in Supplementary Table S1.
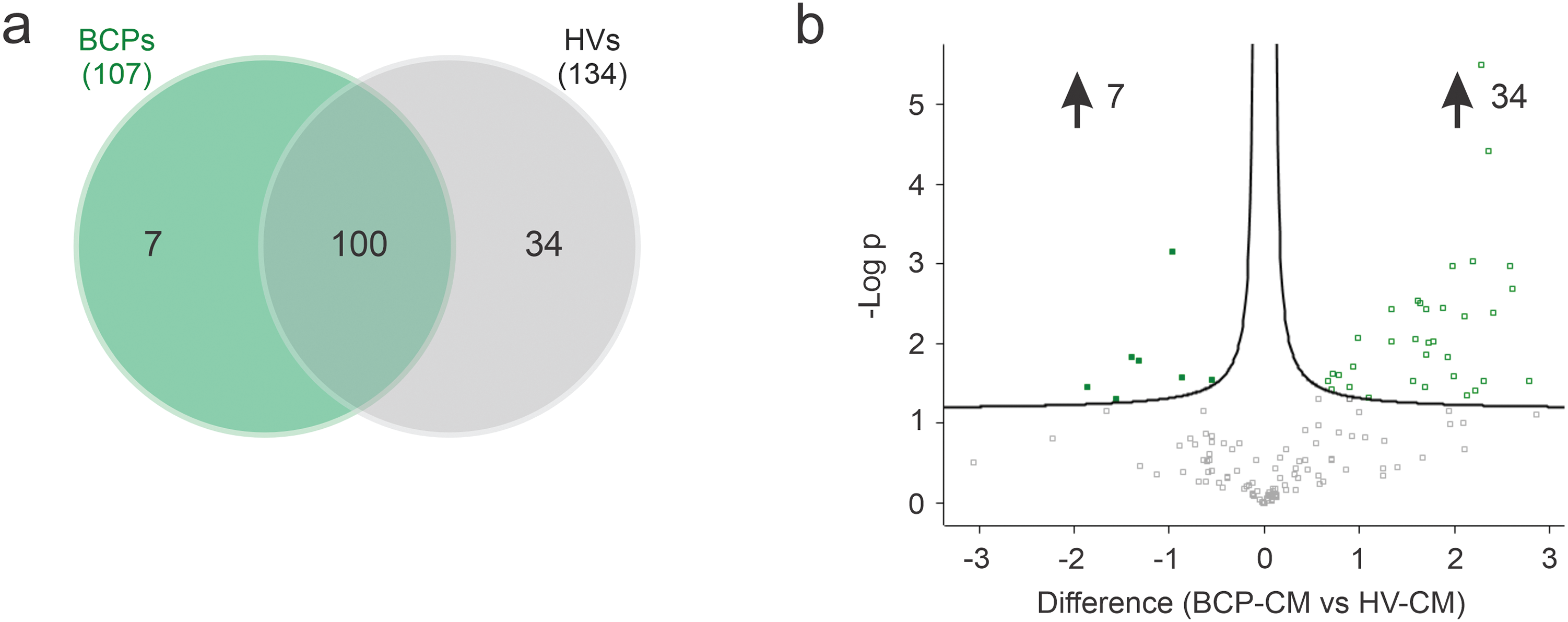
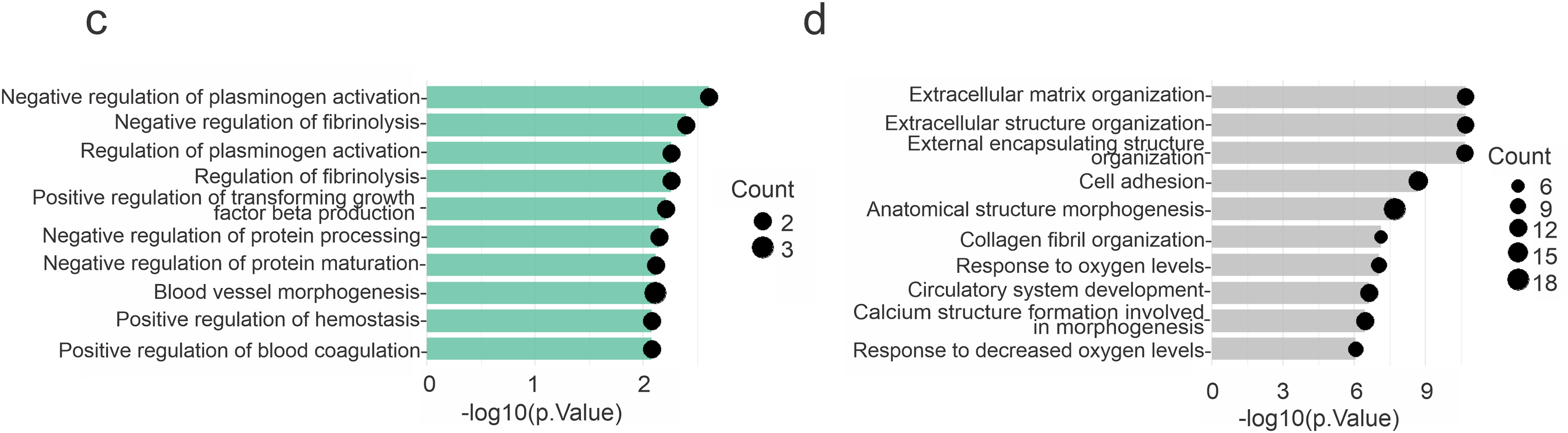
Figure 8: Analysis of differentially expressed proteins (DEPs) in breast cancer patients (BCPs)-conditioned media (CM) and healthy volunteer (HV)-CM. (a) Venn diagram illustrating the number of DEPs for each CM group. (b) Volcano plot showing statistical significance of DEPs (adjusted p-value) vs. magnitude of expression change (log2 fold change); numbers indicate the upregulated proteins in BCPs-CM (DEPs = 7) and HV-CM (DEPs = 34). (c) Gene Ontology (GO) biological process enrichment analysis of DEPs secreted by BCPs-MSCs. The top 10 enriched biological processes are shown. (d) GO biological process enrichment analysis of DEPs secreted by HV-MSCs. The top 10 enriched biological processes are shown. Bubble sizes correspond to the number of proteins associated with each process. All analyses were conducted using DAVID Bioinformatics Resources (v2023q4). DAVID: Database for Annotation, Visualization, and Integrated Discovery.
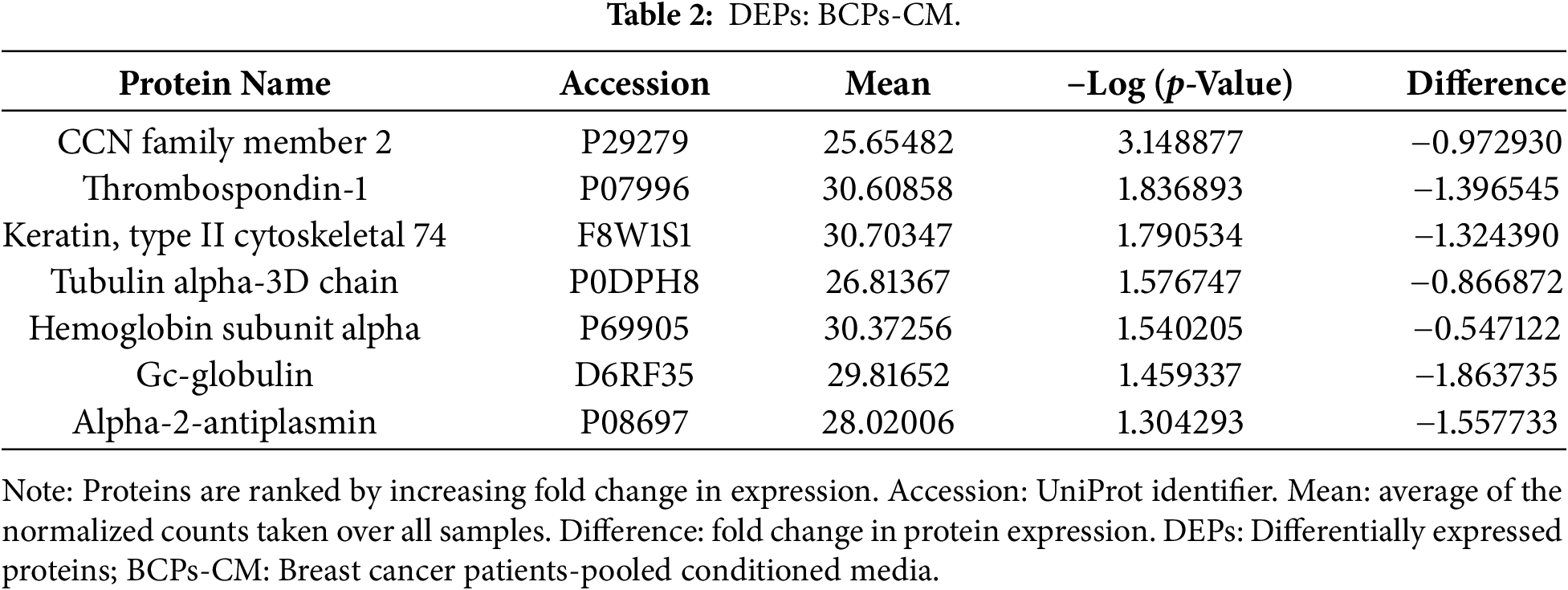
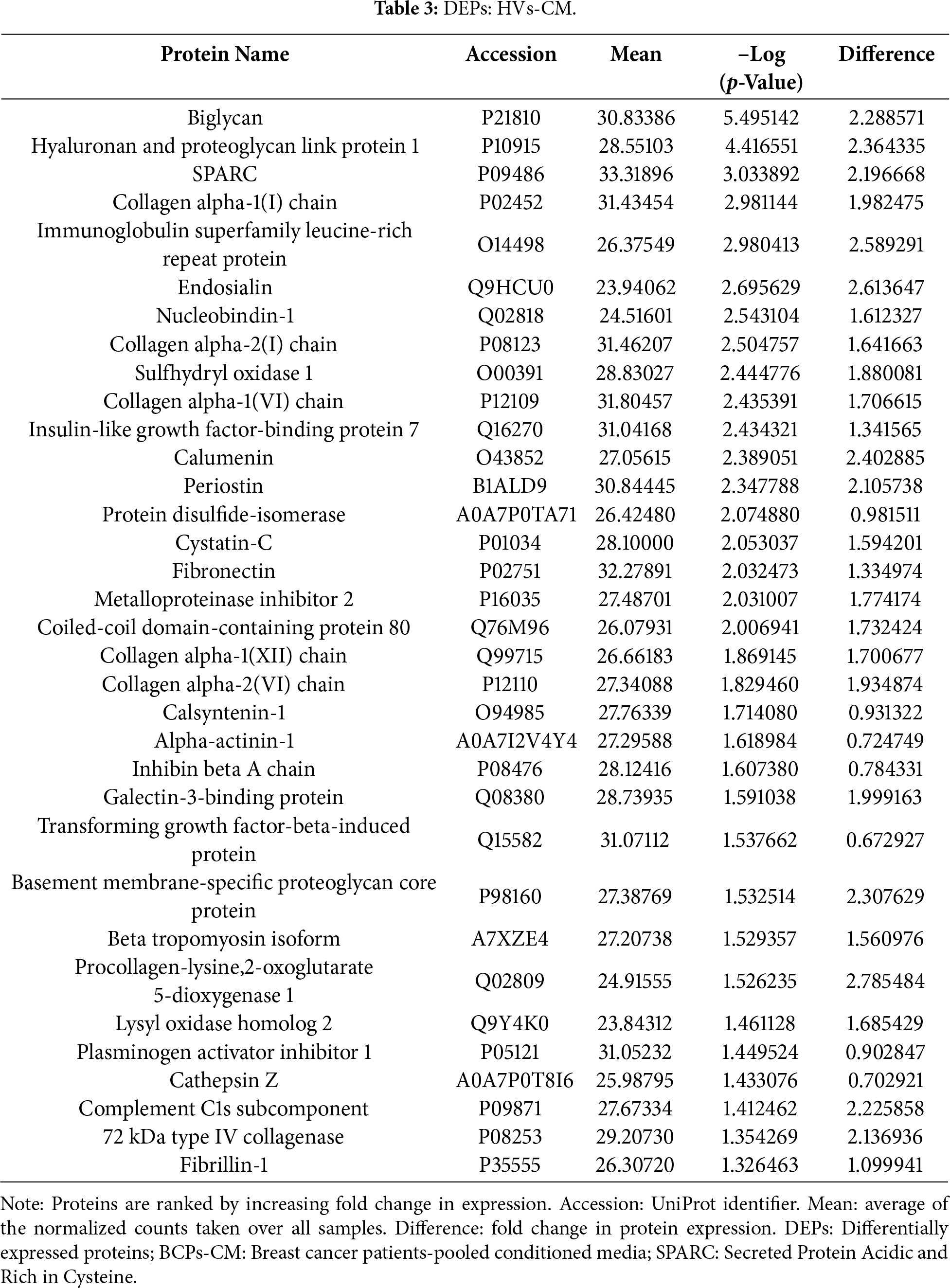
Interestingly, among the differentially secreted proteins in BCPs, cellular communication network factor 2 (CCN2), thrombospondin-1 (THBS1), α-2-antiplasmin (SERPINF2) and type II cytoskeletal keratin 74 (KRT74) stood out due to their known roles in MSC dysfunction and oxidative stress responses [20,44,45].
The top 10 significantly enriched terms (GO biological process) for BCPs and HVs are shown in Fig. 8c,d, respectively. In BCPs, additional terms, reactive oxygen species metabolic process and positive regulation of MAPK cascade, were identified, which is of particular interest due to its link to the oxidative stress previously reported in patient-derived MSCs. In HVs, additional terms such as direct ossification, intramembranous ossification, ossification, and bone trabecula formation were identified, which are noteworthy for their association with healthy MSCs and bone formation. Complete lists of enriched terms are provided in Supplementary Tables S2 and S3.
Dysfunctional MSCs promote the metastatic cascade by creating the BM/ bone PMN [16,21,46,47].
Investigating the mechanisms underlying PMN formation and their role in tumor invasion and outgrowth remains an active area of research [22,48]. However, the impact of increased oxidative stress on MSCs in advanced BCPs, and how it alters their physiological functions remains poorly understood. Here, we provide compelling evidence of such alterations in MSCs from untreated advanced BCPs. It has been reported that soluble factors such as CCL2, acting through its receptor CCR2, reinforce senescence in MSCs by upregulating p53 and p21 protein levels via ROS-mediated or p38-MAPK signaling pathways [42]. Notably, p53 activation in senescent cells further promotes CCL2 secretion, establishing a feed-forward loop that sustains the senescent phenotype. Consistent with this mechanism, we previously showed that in advanced BCPs (clinic-pathological stage III-B) the BM microenvironment shifts toward a pro-osteoclastogenic and inflammatory state, characterized by elevated CCL-2 levels even before detectable bone or BM metastasis [20]. In line with these findings, we now report an increased proportion of patient-derived MSCs expressing CCR2, indicative of a microenvironment prone to oxidative stress and ROS generation. Given that cellular senescence can propagate in a paracrine manner, a relatively small CCR2+ MSCs subpopulation may be sufficient to initiate a feed-forward loop in which senescent cells induce senescence in neighboring MSCs [49], leading to a progressive expansion of the senescent MSCs pool.
Consistently, MSCs from BCPs exhibited elevated expression of oxidative stress response genes, including HSP90 and Nrf2, which are induced by oxidative stress and play a key role in protein protection and repair, as well as in the regulation of antioxidant genes such as SOD1, SOD2, CAT, and GPx-1. Although activation of these antioxidant pathways helps counteract ROS and preserve cellular homeostasis [50–52], sustained oxidative stress ultimately leads to cumulative biomolecular damage [26,53]. Consistent with this, we observed significantly increased DNA damage in MSCs from BCPs compared with those from HVs, as assessed by the alkaline comet assay. This sensitive technique detects DNA strand breaks and alkali-labile sites associated with ROS-induced genomic damage [33,54]. The extent of DNA damage critically influences cell fate decisions, as both DNA repair and apoptotic pathways are initiated by cell cycle arrest, in agreement with our previous observations of impaired cell cycle progression in these MSCs [16].
Despite the presence of DNA repair mechanisms, cells do not always successfully reverse damage, and some lesions may remain unrepaired or be misrepaired. Such residual lesions can trigger apoptosis; however, when apoptotic pathways are compromised, the risk of chromosomal instability increases. This instability includes numerical chromosomal alterations and structural CAs, such as the TAS predominantly observed in MSCs from BCPs. The formation of these structural CAs requires breakage of chromatids at chromosome ends followed by end-to-end fusion [55].
In this context, telomeres are especially vulnerable to oxidative damage due to their high guanine content [56,57]. Under conditions of increased oxidative stress, as observed in BCPs, single-strand DNA breaks preferentially accumulate in telomeres. This leads to instability in this chromosomal region [58], which could explain why TAS represent the most frequent CA observed in MSCs from these patients.
As a consequence of defective DNA repair under oxidative stress, MSCs from BCPs also exhibit NBs and MN. These nucleus-like structures arise from lagging chromosomes or chromosomal fragments that fail to be reincorporated into the primary nucleus following cell division [59]. Both NBs and MN are widely recognized as biomarkers of genomic instability and are frequently associated with impaired DNA repair mechanisms [60]. Excessive ROS can damage DNA and cell-cycle–associated proteins, thereby compromising genetic stability and proper chromosomal segregation [33,61], which likely explains the increased frequency of NBs and MN observed in MSCs from BCPs. Notably, Sharma et al. demonstrated that elevated NB and MN frequencies impair MSC proliferation and alter cell cycle progression through differential regulation of cell cycle checkpoint genes associated with cell cycle arrest [62]. These findings are consistent with our previous observations showing reduced self-renewal capacity, prolonged G0/G1 phase, and shortened S phase in MSCs from this patient group [16].
Similarly, lipid oxidation was increased, as evidenced by elevated levels of 4-HNE.This reactive lipid peroxidation product inhibits DNA, RNA, and protein synthesis and exerts both genotoxic and cytotoxic effects [63,64]. Beyond serving as a biomarker of lipid peroxidation, 4-HNE is highly reactive and forms adducts with DNA and proteins, thereby constituting an additional indicator of ROS-induced molecular damage [65]. Notably, it has been reported that an increase in 4-HNE is dose-dependently linked to the increase in Nrf2 expression, which leads to an increase in the expression of antioxidant genes, as demonstrated in BCP samples. Moreover, in vitro experiments showed that 4-HNE induced significantly elevated levels of CAs and MN, in certain cell types [64]. This last observation partially explains the increase in the number of CAs and MN observed in MSCs from BCPs, as it links them to the elevated levels of 4-HNE present in these stromal cells.
Notably, protein carbonylation, the primary product of ROS-mediated oxidation reactions in proteins, has been found to be increased in protein isolates from the lysates of BCPs-MSCs, as well as in their CM. This observation is consistent with oxidative stress affecting multiple components of the proteome in these cells. Krisko Radman described that protein carbonylation serves as an indicator of diminished functionality of a damaged proteome [66]. Particularly, telomerase, one of the least abundant cellular proteins, is among the human proteins most susceptible to carbonylation [67], which could help explain the reduced telomerase activity previously reported in MSCs from these patients [16]. Moreover, telomere shortening and genomic instability can be considered secondary consequences of widespread proteome damage [68,69].
Our findings reveal substantial damage to DNA, proteins, and lipids in BCPs-MSCs, alterations that will inevitably impact the relationship between these cells and their surrounding environment [70]. Accordingly, characterization of the MSC secretome is essential to understand how BCP-derived MSCs contribute to PMN formation within the BM/bone microenvironment, as well as how MSCs from HVs regulate normal BM homeostasis.
Among the differentially expressed proteins identified in the secretome of BCPs-MSCs, the GO analysis highlighted biological processes relevant to the pathophysiological context of the PMN. These included pathways related to cellular signaling (positive regulation of transforming growth factor beta production and positive regulation of MAPK cascade), oxidative stress (reactive oxygen species metabolic process), and protein homeostasis (negative regulation of protein processing and negative regulation of protein maturation). Collectively, these processes reinforce the notion that BCPs-MSCs actively contribute to the remodeling of the BM microenvironment into PMN. These associations were primarily driven by the expression of CCN2, THBS1, SERPINF2, KRT74, and the vitamin D-binding protein (GC).
Regarding CCN2, the members of the CCN family are proteins present in the extracellular matrix. Overexpression of CCN2 is closely related to cell adhesion, proliferation, and migration, directly contributing to tumor development and metastasis in various types of cancer. Specifically in breast cancer, CCN2 can regulate the expression of matrix metalloproteinase-1, transforming growth factor-beta (TGF-β), N-cadherin, Snail, β-catenin, and bone morphogenetic protein (BMP), promoting metastasis and angiogenesis [71,72]. TGF-β signaling is particularly relevant, as it drives epithelial mesenchymal transition and promotes tumor cell invasion and metastasis in advanced stages of breast cancer. Under hypoxic conditions, TGF-β enhances the hypoxia-inducible factor-1 signaling pathway within the bone microenvironment, stimulating osteoclast formation and activity while inhibiting the osteogenic differentiation of MSCs [73–77]. Notably, TGF-β1 promotes senescence of BM-MSCs through elevated mitochondrial ROS production [78]. In the BM, CCN2 is mainly expressed by MSCs, where it supports early osteogenic differentiation but inhibits Wnt3a- and BMP-induced differentiation when persistently expressed. Moreover, by interacting with both receptor-receptor activator of nuclear factor kappa-B ligand osteoprotegerin, CCN2 enhances receptor activator of nuclear factor kappa-B ligand signaling, thereby promoting osteoclast formation and activity [72,79,80].
In summary, CCN2 expression may partly account for the impaired osteogenic differentiation of BCP-derived MSCs and the enhanced osteoclastic activity previously reported in the PMN of these patients, as well as for the promotion of MSC senescence through mitochondrial ROS production [20,21].
With regard to THBS1, functions as an an inhibitor of stem cell self-renewal. Acting through its cell surface receptor CD47, THBS1 suppresses the expression of important self-renewal transcription factors, including OCT4, SOX2, KLF4, and MYC in non-malignant cells [45]. This mechanism could underlie the decreased colony-forming ability and the reduced expression of stemness-associated genes observed in MSCs from these patients [16]. In addition, THBS1 may contribute to the PMN formation by disrupting the endothelial intercellular junctions integrity, a process known to facilitate breast cancer metastasis [81].
Both THBS1 and SERPINF2 have been reported as plasma biomarkers of breast disease [82,83]. Our findings suggest that BM-derived MSCs from these patients could represent a potential source of these elevated factors.
In addition to these proteins, keratins, including KRT74, are epithelial markers associated with cancer progression, diagnosis, and treatment, and their expression is often elevated in cancer or oxidative stress conditions [44], which is relevant given the increased oxidative stress observed in BCPs-MSCs [16].
Finally, GC, a vitamin D-related protein involved in bone metabolism, can bind C5a, extending its half-life and activity. C5a promotes osteoclast activation and, in the presence of IL-1β, induces IL-6 and IL-8 expression [84,85], which may contribute to the pro-inflammatory PMN and increased osteoclastogenesis observed in these patients [16,20].
The remaining two proteins identified in the BCP-derived MSC secretome lack well-defined roles in cancer or BM biology and were therefore not discussed further.
The secretome of MSCs derived from HVs was was enriched in proteins whose GO annotations highlighted biological processes fundamental to maintaining a healthy tissue microenvironment, including extracellular matrix organization, extracellular structure organization, cell adhesion, regulation of cell population proliferation, regulation of cell migration, and ossification.
Among the identified proteins are collagen alpha-2(I) chain (COL1A2), secreted protein acidic and rich in cysteine (SPARC), and quiescin sulfhydryl oxidase 1 (QSOX1) were particularly notable. We focused on these proteins given their direct relevance to bone homeostasis, whereas the remaining proteins, although important for general tissue maintenance, have less clearly defined roles in these processes.
Regarding COL1A2, this protein supports type I collagen synthesis, fibroblast proliferation, elastin production, and wound healing, thereby playing a key role in bone formation [86]. SPARC regulates bone remodeling, inhibits breast cancer cell migration, and suppresses osteoclast activation; notably, reduced stromal SPARC levels have been associated with bone metastasis [87,88]. QSOX1 is essential for laminin incorporation, cell adhesion, and migration, stabilizes tissue inhibitors of metalloproteinases (TIMPs) to inhibit matrix metalloproteinase activity, and protects against oxidative stress. Importantly, higher QSOX1 expression in BCPs has been correlated with improved patient survival [89–91]. This profile contrasts markedly with the secretome of MSCs derived from BCPs, in which the microenvironment is remodeled to support tumor progression. These findings suggest that alterations in BCP-derived MSCs drive a shift in the secreted protein profile, leading to dysfunction in key processes such as bone formation and angiogenesis—both essential for maintaining BM/bone homeostasis. Disruption of these functions appears sufficient to promote PMN formation, as observed in this patient group. Nevertheless, future quantitative and functional assays will be required to validate and further characterize the secretome-associated changes in BCP-derived MSCs. Taken together, the alterations observed in the secretome of BCP-derived MSCs reflect a broader dysfunctional phenotype characterized by cellular senescence, metabolic reprogramming, and molecular changes that favor PMN establishment [16,20,21]. In this context, senescent MSCs from these patients exhibit the metabolic and molecular features required to support PMN formation within the BM/bone microenvironment. Because disease progression largely depends on PMN characteristics—the so-called “fertile soil”—which are in turn shaped by MSCs, therapeutic strategies aimed at normalizing MSC function are of particular interest. Restoration of this altered MSC phenotype could delay BM/bone metastasis and improve patient quality of life. However, reversing the damage caused by chronic oxidative stress in patients at this stage of the disease (IIIB) remains challenging [92]. Future studies should explore antioxidants—such as resveratrol, ascorbic acid, and idebenone—or their combinations, as well as their potential use alongside additional interventions, including anti-CCL2 or anti-CCR2 therapies [93,94]. In addition, senolytic approaches, such as treatment with quercetin, which has been shown to effectively eliminate senescent MSCs, may represent a promising strategy to purify the MSC pool by selectively removing senescent cells [95–98].
Nevertheless, the temporal interval between primary tumor diagnosis and metastatic dissemination highlights the potential of early therapeutic strategies aimed at preserving BM-MSC health to prevent senescence-driven niche formation.
1. Small sample size.
This study is limited by the relatively small number of patient-derived MSC samples (n = 10), which may affect statistical power and reflects the strict inclusion and exclusion criteria applied for patient selection.
2. Variable sample numbers across assays.
The number of samples included in each experimental assay varied due to the intrinsically low self-renewal capacity of MSCs obtained from untreated advanced BCPs, which limited cell expansion and the availability of sufficient material for all downstream analyses.
Despite these constraints, the converging evidence obtained from multiple complementary analyses strengthens the reliability of the observed trends.
Our findings define a distinct oxidative stress signature in BM–derived MSCs from untreated advanced BCPs, characterized by the accumulation of DNA damage, genomic instability, increased protein carbonylation, and enhanced lipid peroxidation. These oxidative stress–associated alterations are accompanied by dysregulated expression of oxidative stress response genes and marked changes in the MSCs secretome compared with HVs. Collectively, this molecular footprint provides mechanistic insight into PMN formation and may support the development of prognostic biomarkers and personalized therapeutic strategies aimed at delaying or preventing bone metastasis.
Acknowledgement: The authors would like to thank the Florencio Fiorini Foundation, Argentina; René Barón Foundation, Argentina; Bigand Foundation, Argentina and Williams Foundation, Argentina for their generous donations supporting IBYME, which made this work possible. We are grateful to Dr. Esteban Fiore from Instituto de Investigaciones en Medicina Traslacional, Universidad Austral-CONICET, Derqui-Pilar, Buenos Aires, Argentina for the kind donation of HUC-MSCs.
Funding Statement: This research was supported by the FONCYT, PICT 2016-#1093, Argentina. CONICET, PIP2014-#300, Argentina. Florencio Fiorini Foundation (#2021-2022), Argentina.
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, Norma Alejandra Chasseing; methodology, Francisco Raúl Borzone; software, Francisco Raúl Borzone; validation, Francisco Raúl Borzone, María Belén Giorello, Agustina Freire, Marcela Beatriz González Cid; formal analysis, Francisco Raúl Borzone, María Belén Giorello, Agustina Freire, Marcela Beatriz González Cid; investigation, Francisco Raúl Borzone, María Belén Giorello, Agustina Freire, Marcela Beatriz González Cid; resources, Leonardo Feldman, Federico Dimase, Emilio Batagelj; data curation, Francisco Raúl Borzone, María Belén Giorello, Agustina Freire, Marcela Beatriz González Cid; writing—original draft preparation, Francisco Raúl Borzone; writing—review and editing, Francisco Raúl Borzone, María Belén Giorello, Marcela Beatriz González Cid, Pablo Evelson, Leandro Marcelo Martinez, Irene Larripa, Norma Alejandra Chasseing; visualization, Francisco Raúl Borzone; supervision, Norma Alejandra Chasseing; project administration, Norma Alejandra Chasseing; funding acquisition, Pablo Evelson, Irene Larripa, Norma Alejandra Chasseing. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The datasets generated or analyzed during the current study are available from the corresponding authors on reasonable request.
Ethics Approval: This study was conducted in accordance with the ethical principles of the Declaration of Helsinki. Ethical approval was obtained from the relevant institutional ethics committees of all participating institutions. Specifically, the study was approved by the Comité de Ética Dr. Enrique T. Segura, IBYME-CONICET (Project CE 051/2015) and by the Comité Institucional de Revisión de Ensayos Clínicos (CIREC), Hospital Militar Central (ACTA 326/2014). Written informed consent was obtained from all individual participants included in the study.
Conflicts of Interest: The authors declare no conflicts of interest.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/or.2026.074321/s1.
References
1. Zhang M, Deng H, Hu R, Chen F, Dong S, Zhang S, et al. Patterns and prognostic implications of distant metastasis in breast Cancer based on SEER population data. Sci Rep. 2025;15(1):26717. doi:10.1038/s41598-025-12883-x. [Google Scholar] [PubMed] [CrossRef]
2. Liu F, Ding Y, Xu Z, Hao X, Pan T, Miles G, et al. Single-cell profiling of bone metastasis ecosystems from multiple cancer types reveals convergent and divergent mechanisms of bone colonization. Cell Genom. 2025;5(7):100888. doi:10.1016/j.xgen.2025.100888. [Google Scholar] [PubMed] [CrossRef]
3. Huang Y, Wang H, Yue X, Li X. Bone serves as a transfer station for secondary dissemination of breast cancer. Bone Res. 2023;11(1):21. doi:10.1038/s41413-023-00260-1. [Google Scholar] [PubMed] [CrossRef]
4. Mishra A, Shiozawa Y, Pienta KJ, Taichman RS. Homing of cancer cells to the bone. Cancer Microenviron. 2011;4(3):221–35. doi:10.1007/s12307-011-0083-6. [Google Scholar] [PubMed] [CrossRef]
5. Peinado H, Lavotshkin S, Lyden D. The secreted factors responsible for pre-metastatic niche formation: old sayings and new thoughts. Semin Cancer Biol. 2011;21(2):139–46. doi:10.1016/j.semcancer.2011.01.002. [Google Scholar] [PubMed] [CrossRef]
6. Wang Y, Jia J, Wang F, Fang Y, Yang Y, Zhou Q, et al. Pre-metastatic niche: formation, characteristics and therapeutic implication. Sig Transduct Target Ther. 2024;9(1):236. doi:10.1038/s41392-024-01937-7. [Google Scholar] [PubMed] [CrossRef]
7. Nadeau A, Tsering T, Abdouh M, Kienzle L, Cleyle J, Taylor L, et al. Characterization of extracellular vesicle-associated DNA and proteins derived from organotropic metastatic breast cancer cells. J Exp Clin Cancer Res. 2025;44(1):157. doi:10.1186/s13046-025-03418-3. [Google Scholar] [PubMed] [CrossRef]
8. Tang R, Luo S, Liu H, Sun Y, Liu M, Li L, et al. Circulating tumor microenvironment in metastasis. Cancer Res. 2025;85(8):1354–67. doi:10.1158/0008-5472.can-24-1241. [Google Scholar] [PubMed] [CrossRef]
9. Lu X, Kang Y. Chemokine (C-C motif) ligand 2 engages CCR2+ stromal cells of monocytic origin to promote breast cancer metastasis to lung and bone. J Biol Chem. 2009;284(42):29087–96. doi:10.1074/jbc.m109.035899. [Google Scholar] [PubMed] [CrossRef]
10. Huang G, Hou T, Song D, Meng T. The regulatory networks and mechanisms of bone microenvironment in tumorigenesis and metastasis. J Bone Oncol. 2025;55(1):100729. doi:10.1016/j.jbo.2025.100729. [Google Scholar] [PubMed] [CrossRef]
11. Briggs EN, Lynch ME. The role of osteocytes in pre-metastatic niche formation. Curr Osteoporos Rep. 2024;22(1):105–14. doi:10.1007/s11914-023-00857-9. [Google Scholar] [PubMed] [CrossRef]
12. Sanmartin MC, Borzone FR, Giorello MB, Pacienza N, Yannarelli G, Chasseing NA. Bone marrow/bone pre-metastatic niche for breast cancer cells colonization: the role of mesenchymal stromal cells. Crit Rev Oncol Hematol. 2021;164(8):103416. doi:10.1016/j.critrevonc.2021.103416. [Google Scholar] [PubMed] [CrossRef]
13. Barcellos-de-Souza P, Gori V, Bambi F, Chiarugi P. Tumor microenvironment: bone marrow-mesenchymal stem cells as key players. Biochim Biophys Acta Rev Cancer. 2013;1836(2):321–35. doi:10.1016/j.bbcan.2013.10.004. [Google Scholar] [PubMed] [CrossRef]
14. Malanchi I, Santamaria-Martínez A, Susanto E, Peng H, Lehr HA, Delaloye JF, et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature. 2012;481(7379):85–9. doi:10.1038/nature10694. [Google Scholar] [PubMed] [CrossRef]
15. Kaur J, Adhikari M, Sabol HM, Anloague A, Khan S, Kurihara N, et al. Single-cell transcriptomic analysis identifies senescent osteocytes that trigger bone destruction in breast cancer metastasis. Cancer Res. 2024;84(23):3936–52. doi:10.1158/0008-5472.can-24-0857. [Google Scholar] [PubMed] [CrossRef]
16. Borzone FR, Giorello MB, Martinez LM, Sanmartin MC, Feldman L, Dimase F, et al. Senescent mesenchymal stem/stromal cells in pre-metastatic bone marrow of untreated advanced breast cancer patients. Oncol Res. 2023;31(3):361–74. doi:10.32604/or.2023.028104. [Google Scholar] [PubMed] [CrossRef]
17. Hernandez-Segura A, Nehme J, Demaria M. Hallmarks of cellular senescence. Trends Cell Biol. 2018;28(6):436–53. doi:10.1016/j.tcb.2018.02.001. [Google Scholar] [PubMed] [CrossRef]
18. Li Y, Wu Q, Wang Y, Li L, Bu H, Bao J. Senescence of mesenchymal stem cells (Review). Int J Mol Med. 2017;39(4):775–82. doi:10.3892/ijmm.2017.2912. [Google Scholar] [PubMed] [CrossRef]
19. Jin HJ, Kwon JH, Kim M, Bae YK, Choi SJ, Oh W, et al. Downregulation of melanoma cell adhesion molecule (MCAM/CD146) accelerates cellular senescence in human umbilical cord blood-derived mesenchymal stem cells. Stem Cells Transl Med. 2016;5(4):427–39. doi:10.5966/sctm.2015-0109. [Google Scholar] [PubMed] [CrossRef]
20. Fernández Vallone V, Borzone FR, Martinez LM, Giorello MB, Choi H, Dimase F, et al. Spontaneous Osteoclastogenesis, a risk factor for bone metastasis in advanced luminal A-type breast cancer patients. Front Oncol. 2023;13:1073793. doi:10.3389/fonc.2023.1073793. [Google Scholar] [PubMed] [CrossRef]
21. Fernández Vallone VB, Hofer EL, Choi H, Bordenave RH, Batagelj E, Feldman L, et al. Behaviour of mesenchymal stem cells from bone marrow of untreated advanced breast and lung cancer patients without bone osteolytic metastasis. Clin Exp Metastasis. 2013;30(3):317–32. doi:10.1007/s10585-012-9539-4. [Google Scholar] [PubMed] [CrossRef]
22. Martinez LM, Vallone VBF, Labovsky V, Choi H, Hofer EL, Feldman L, et al. Changes in the peripheral blood and bone marrow from untreated advanced breast cancer patients that are associated with the establishment of bone metastases. Clin Exp Metastasis. 2014;31(2):213–32. doi:10.1007/s10585-013-9622-5. [Google Scholar] [PubMed] [CrossRef]
23. Kizilay Mancini Ö, Lora M, Shum-Tim D, Nadeau S, Rodier F, Colmegna I. A proinflammatory secretome mediates the impaired immunopotency of human mesenchymal stromal cells in elderly patients with atherosclerosis. Stem Cells Transl Med. 2017;6(4):1132–40. doi:10.1002/sctm.16-0221. [Google Scholar] [PubMed] [CrossRef]
24. Turinetto V, Vitale E, Giachino C. Senescence in human mesenchymal stem cells: functional changes and implications in stem cell-based therapy. Int J Mol Sci. 2016;17(7):1164. doi:10.3390/ijms17071164. [Google Scholar] [PubMed] [CrossRef]
25. Elsayed Azab A, A Adwas A, Ibrahim Elsayed AS, A Adwas A, Ibrahim Elsayed AS, Quwaydir FA. Oxidative stress and antioxidant mechanisms in human body. J Appl Biotechnol Bioeng. 2019;6(1):43–7. doi:10.15406/jabb.2019.06.00173. [Google Scholar] [CrossRef]
26. de Cavanagh EMV, Honegger AE, Hofer E, Bordenave RH, Bullorsky EO, Chasseing NA, et al. Higher oxidation and lower antioxidant levels in peripheral blood plasma and bone marrow plasma from advanced cancer patients. Cancer. 2002;94(12):3247–51. doi:10.1002/cncr.10611. [Google Scholar] [PubMed] [CrossRef]
27. Neri S. Genetic stability of mesenchymal stromal cells for regenerative medicine applications: a fundamental biosafety aspect. Int J Mol Sci. 2019;20(10):2406. doi:10.3390/ijms20102406. [Google Scholar] [PubMed] [CrossRef]
28. Arulselvan P, Fard MT, Tan WS, Gothai S, Fakurazi S, Norhaizan ME, et al. Role of antioxidants and natural products in inflammation. Oxid Med Cell Longev. 2016;2016(1):5276130. doi:10.1155/2016/5276130. [Google Scholar] [PubMed] [CrossRef]
29. Cardoso F, Kyriakides S, Ohno S, Penault-Llorca F, Poortmans P, Rubio IT, et al. Early breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2019;30(8):1194–220. doi:10.1093/annonc/mdz173. [Google Scholar] [PubMed] [CrossRef]
30. Ylöstalo J, Bazhanov N, Prockop DJ. Reversible commitment to differentiation by human multipotent stromal cells in single-cell-derived colonies. Exp Hematol. 2008;36(10):1390–402. doi:10.1016/j.exphem.2008.05.003. [Google Scholar] [PubMed] [CrossRef]
31. Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini FC, Krause DS, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. Int Soc Cell Ther Position Statement Cytotherapy. 2006;8(4):315–7. doi:10.1080/14653240600855905. [Google Scholar] [PubMed] [CrossRef]
32. Grotheer V, Skrynecki N, Oezel L, Windolf J, Grassmann J. Osteogenic differentiation of human mesenchymal stromal cells and fibroblasts differs depending on tissue origin and replicative senescence. Sci Rep. 2021;11(1):11968. doi:10.1038/s41598-021-91501-y. [Google Scholar] [PubMed] [CrossRef]
33. Collins A, Møller P, Gajski G, Vodenková S, Abdulwahed A, Anderson D, et al. Measuring DNA modifications with the comet assay: a compendium of protocols. Nat Protoc. 2023;18(3):929–89. doi:10.1038/s41596-022-00754-y. [Google Scholar] [PubMed] [CrossRef]
34. de Campos-Nebel M, Larripa I, González-Cid M. Non-homologous end joining is the responsible pathway for the repair of fludarabine-induced DNA double strand breaks in mammalian cells. Mutat Res Fundam Mol Mech Mutagen. 2008;646(1–2):8–16. doi:10.1016/j.mrfmmm.2008.08.013. [Google Scholar] [PubMed] [CrossRef]
35. Savage JR. Classification and relationships of induced chromosomal structual changes. J Med Genet. 1976;13(2):103–22. doi:10.1136/jmg.13.2.103. [Google Scholar] [PubMed] [CrossRef]
36. Elguero ME, de Campos-Nebel M, González-Cid M. DNA-PKcs-dependent NHEJ pathway supports the progression of topoisomerase II poison-induced chromosome aberrant cells. Environ Mol Mutagen. 2012;53(8):608–18. doi:10.1002/em.21729. [Google Scholar] [PubMed] [CrossRef]
37. Hawkins CL, Morgan PE, Davies MJ. Quantification of protein modification by oxidants. Free Radic Biol Med. 2009;46(8):965–88. doi:10.1016/j.freeradbiomed.2009.01.007. [Google Scholar] [PubMed] [CrossRef]
38. Murphy MP, Bayir H, Belousov V, Chang CJ, Davies KJA, Davies MJ, et al. Guidelines for measuring reactive oxygen species and oxidative damage in cells and in vivo. Nat Metab. 2022;4(6):651–62. doi:10.1038/s42255-022-00591-z. [Google Scholar] [PubMed] [CrossRef]
39. Garcés M, Magnani ND, Pecorelli A, Calabró V, Marchini T, Cáceres L, et al. Alterations in oxygen metabolism are associated to lung toxicity triggered by silver nanoparticles exposure. Free Radic Biol Med. 2021;166(7):324–36. doi:10.1016/j.freeradbiomed.2021.02.008. [Google Scholar] [PubMed] [CrossRef]
40. Kruger NJ. The Bradford method for protein quantitation. In: Springer protocols handbooks. Totowa, NJ, USA: Humana Press; 2009. p. 17–24. doi:10.1007/978-1-59745-198-7_4. [Google Scholar] [CrossRef]
41. Sherman BT, Hao M, Qiu J, Jiao X, Baseler MW, Lane HC, et al. DAVID a web server for functional enrichment analysis and functional annotation of gene lists (2021 update). Nucleic Acids Res. 2022;50(W1):W216–21. doi:10.1093/nar/gkac194. [Google Scholar] [PubMed] [CrossRef]
42. Jin HJ, Lee HJ, Heo J, Lim J, Kim M, Kim MK, et al. Senescence-associated MCP-1 secretion is dependent on a decline in BMI1 in human mesenchymal stromal cells. Antioxid Redox Signal. 2016;24(9):471–85. doi:10.1089/ars.2015.6359. [Google Scholar] [PubMed] [CrossRef]
43. Fuchs R, Stelzer I, Drees CMP, Rehnolt C, Schraml E, Sadjak A, et al. Modification of the alkaline comet assay with human mesenchymal stem cells. Cell Biol Int. 2012;36(1):113–7. doi:10.1042/cbi20110251. [Google Scholar] [PubMed] [CrossRef]
44. Ogunnigbagbe O, Bunick CG, Kaur K. Keratin 1 as a cell-surface receptor in cancer. Biochim Biophys Acta BBA Rev Cancer. 2022;1877(1):188664. doi:10.1016/j.bbcan.2021.188664. [Google Scholar] [PubMed] [CrossRef]
45. Isenberg JS, Roberts DD. THBS1 (thrombospondin-1). Atlas Genet Cytogenet Oncol Haematol. 2020;8(8):1–17. doi:10.4267/2042/70774. [Google Scholar] [PubMed] [CrossRef]
46. Chasseing NA, Trejo YG, Bordenave RH, Bullorsky EO, Diaz NB, Rumi LS. Bone marrow fibroblastic progenitors in patients with advanced breast cancer. Breast Cancer Res Treat. 1997;45(3):211–8. doi:10.1023/A:1005757218114. [Google Scholar] [PubMed] [CrossRef]
47. Hofer EL, Labovsky V, La Russa V, Vallone VF, Honegger AE, Belloc CG, et al. Mesenchymal stromal cells, colony-forming unit fibroblasts, from bone marrow of untreated advanced breast and lung cancer patients suppress fibroblast colony formation from healthy marrow. Stem Cells Dev. 2010;19(3):359–70. doi:10.1089/scd.2008.0375. [Google Scholar] [PubMed] [CrossRef]
48. Peinado H, Zhang H, Matei IR, Costa-Silva B, Hoshino A, Rodrigues G, et al. Pre-metastatic niches: organ-specific homes for metastases. Nat Rev Cancer. 2017;17(5):302–17. doi:10.1038/nrc.2017.6. [Google Scholar] [PubMed] [CrossRef]
49. Chou LY, Ho CT, Hung SC. Paracrine senescence of mesenchymal stromal cells involves inflammatory cytokines and the NF-κB pathway. Cells. 2022;11(20):3324. doi:10.3390/cells11203324. [Google Scholar] [PubMed] [CrossRef]
50. Pole A, Dimri M, Dimri GP. Oxidative stress, cellular senescence and ageing. AIMS Mol Sci. 2016;3(3):300–24. doi:10.3934/molsci.2016.3.300. [Google Scholar] [CrossRef]
51. Ma Q. Role of Nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol. 2013;53(1):401–26. doi:10.1146/annurev-pharmtox-011112-140320. [Google Scholar] [PubMed] [CrossRef]
52. Szyller J, Bil-Lula I. Heat shock proteins in oxidative stress and ischemia/reperfusion injury and benefits from physical exercises: a review to the current knowledge. Oxid Med Cell Longev. 2021;2021(1):6678457. doi:10.1155/2021/6678457. [Google Scholar] [PubMed] [CrossRef]
53. Halliwell B, Gutteridge JMC. Free radicals in biology and medicine. 3rd ed. Oxford, UK: Oxford University Press; 1999. 936 p. [Google Scholar]
54. Ravanat JL, Sauvaigo S, Caillat S, Martinez GR, Medeiros MHG, Di Mascio P, et al. Singlet oxygen-mediated damage to cellular DNA determined by the comet assay associated with DNA repair enzymes. Biol Chem. 2004;385(1):17–20. doi:10.1515/bc.2004.003. [Google Scholar] [PubMed] [CrossRef]
55. Scassa ME, Marazita MC, Ceruti JM, Carcagno AL, Sirkin PF, González-Cid M, et al. Cell cycle inhibitor, p19INK4d, promotes cell survival and decreases chromosomal aberrations after genotoxic insult due to enhanced DNA repair. DNA Repair. 2007;6(5):626–38. doi:10.1016/j.dnarep.2006.12.003. [Google Scholar] [PubMed] [CrossRef]
56. Fouquerel E, Lormand J, Bose A, Lee HT, Kim GS, Li J, et al. Oxidative guanine base damage regulates human telomerase activity. Nat Struct Mol Biol. 2016;23(12):1092–100. doi:10.1038/nsmb.3319. [Google Scholar] [PubMed] [CrossRef]
57. Barnes RP, Fouquerel E, Opresko PL. The impact of oxidative DNA damage and stress on telomere homeostasis. Mech Ageing Dev. 2019;177:37–45. doi:10.1016/j.mad.2018.03.013. [Google Scholar] [PubMed] [CrossRef]
58. Srinivas N, Rachakonda S, Kumar R. Telomeres and telomere length: a general overview. Cancers. 2020;12(3):558. doi:10.3390/cancers12030558. [Google Scholar] [PubMed] [CrossRef]
59. Krupina K, Goginashvili A, Cleveland DW. Causes and consequences of micronuclei. Curr Opin Cell Biol. 2021;70:91–9. doi:10.1016/j.ceb.2021.01.004. [Google Scholar] [PubMed] [CrossRef]
60. Gisselsson D, Björk J, Höglund M, Mertens F, Dal Cin P, Åkerman M, et al. Abnormal nuclear shape in solid tumors reflects mitotic instability. Am J Pathol. 2001;158(1):199–206. doi:10.1016/s0002-9440(10)63958-2. [Google Scholar] [PubMed] [CrossRef]
61. Bonassi S, Znaor A, Ceppi M, Lando C, Chang WP, Holland N, et al. An increased micronucleus frequency in peripheral blood lymphocytes predicts the risk of cancer in humans. Carcinogenesis. 2006;28(3):625–31. doi:10.1093/carcin/bgl177. [Google Scholar] [PubMed] [CrossRef]
62. Sharma S, Bhonde R. Influence of nuclear blebs and micronuclei status on the growth kinetics of human mesenchymal stem cells. J Cell Physiol. 2015;230(3):657–66. doi:10.1002/jcp.24789. [Google Scholar] [PubMed] [CrossRef]
63. Eckl PM, Ortner A, Esterbauer H. Genotoxic properties of 4-hydroxyalkenals and analogous aldehydes. Mutat Res Fundam Mol Mech Mutagen. 1993;290(2):183–92. doi:10.1016/0027-5107(93)90158-c. [Google Scholar] [PubMed] [CrossRef]
64. Karlhuber GM, Bauer HC, Eckl PM. Cytotoxic and genotoxic effects of 4-hydroxynonenal in cerebral endothelial cells. Mutat Res Fundam Mol Mech Mutagen. 1997;381(2):209–16. doi:10.1016/s0027-5107(97)00170-x. [Google Scholar] [PubMed] [CrossRef]
65. Li Y, Zhao T, Li J, Xia M, Li Y, Wang X, et al. Oxidative stress and 4-hydroxy-2-nonenal (4-HNEimplications in the pathogenesis and treatment of aging-related diseases. J Immunol Res. 2022;2022(8):2233906. doi:10.1155/2022/2233906. [Google Scholar] [PubMed] [CrossRef]
66. Krisko A, Radman M. Protein damage, ageing and age-related diseases. Open Biol. 2019;9(3):180249. doi:10.1098/rsob.180249. [Google Scholar] [PubMed] [CrossRef]
67. de Graff AMR, Hazoglou MJ, Dill KA. Highly charged proteins: the Achilles’ heel of aging proteomes. Structure. 2016;24(2):329–36. doi:10.1016/j.str.2015.11.006. [Google Scholar] [PubMed] [CrossRef]
68. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194–217. doi:10.1016/j.cell.2013.05.039. [Google Scholar] [PubMed] [CrossRef]
69. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: an expanding universe. Cell. 2023;186(2):243–78. doi:10.1016/j.cell.2022.11.001. [Google Scholar] [PubMed] [CrossRef]
70. Block TJ, Marinkovic M, Tran ON, Gonzalez AO, Marshall A, Dean DD, et al. Restoring the quantity and quality of elderly human mesenchymal stem cells for autologous cell-based therapies. Stem Cell Res Ther. 2017;8(1):239. doi:10.1186/s13287-017-0688-x. [Google Scholar] [PubMed] [CrossRef]
71. Ahmed KA, Al Hasib T, Paul SK, Saddam M, Mimi A, Saikat ASM, et al. Potential role of CCN proteins in breast cancer: therapeutic advances and perspectives. Curr Oncol. 2021;28(6):4972–85. doi:10.3390/curroncol28060417. [Google Scholar] [PubMed] [CrossRef]
72. Leguit RJ, Raymakers RAP, Hebeda KM, Goldschmeding R. CCN2 (Cellular Communication Network factor 2) in the bone marrow microenvironment, normal and malignant hematopoiesis. J Cell Commun Signal. 2021;15(1):25–56. doi:10.1007/s12079-020-00602-2. [Google Scholar] [PubMed] [CrossRef]
73. McAndrews KM, McGrail DJ, Ravikumar N, Dawson MR. Mesenchymal stem cells induce directional migration of invasive breast cancer cells through TGF-β. Sci Rep. 2015;5(1):16941. doi:10.1038/srep16941. [Google Scholar] [PubMed] [CrossRef]
74. Xie F, Ling L, Van Dam H, Zhou F, Zhang L. TGF-β signaling in cancer metastasis. Acta Biochim Biophys Sin. 2018;50(1):121–32. doi:10.1093/abbs/gmx123. [Google Scholar] [PubMed] [CrossRef]
75. Hiraga T, Kizaka-Kondoh S, Hirota K, Hiraoka M, Yoneda T. Hypoxia and hypoxia-inducible factor-1 expression enhance osteolytic bone metastases of breast cancer. Cancer Res. 2007;67(9):4157–63. doi:10.1158/0008-5472.can-06-2355. [Google Scholar] [PubMed] [CrossRef]
76. McMahon S, Charbonneau M, Grandmont S, Richard DE, Dubois CM. Transforming growth factor β1 induces hypoxia-inducible factor-1 stabilization through selective inhibition of PHD2 expression. J Biol Chem. 2006;281(34):24171–81. doi:10.1074/jbc.m604507200. [Google Scholar] [PubMed] [CrossRef]
77. Maeda S, Hayashi M, Komiya S, Imamura T, Miyazono K. Endogenous TGF-β signaling suppresses maturation of osteoblastic mesenchymal cells. EMBO J. 2004;23(3):552–63. doi:10.1038/sj.emboj.7600067. [Google Scholar] [PubMed] [CrossRef]
78. Wu J, Niu J, Li X, Wang X, Guo Z, Zhang F. TGF-β1 induces senescence of bone marrow mesenchymal stem cells via increase of mitochondrial ROS production. BMC Dev Biol. 2014;14(1):21. doi:10.1186/1471-213X-14-21. [Google Scholar] [PubMed] [CrossRef]
79. Aoyama E, Kubota S, Khattab HM, Nishida T, Takigawa M. CCN2 enhances RANKL-induced osteoclast differentiation via direct binding to RANK and OPG. Bone. 2015;73(3):242–8. doi:10.1016/j.bone.2014.12.058. [Google Scholar] [PubMed] [CrossRef]
80. Barbe MF, Amin M, Gingery A, Lambi AG, Popoff SN. Blocking CCN2 preferentially inhibits osteoclastogenesis induced by repetitive high force bone loading. Connect Tissue Res. 2021;62(1):115–32. doi:10.1080/03008207.2020.1788546. [Google Scholar] [PubMed] [CrossRef]
81. Patwardhan S, Mahadik P, Shetty O, Sen S. ECM stiffness-tuned exosomes drive breast cancer motility through thrombospondin-1. Biomaterials. 2021;279:121185. doi:10.1016/j.biomaterials.2021.121185. [Google Scholar] [PubMed] [CrossRef]
82. Barrón-Gallardo CA, Garcia-Chagollán M, Morán-Mendoza AJ, Delgadillo-Cristerna R, Martínez-Silva MG, Villaseñor-García MM, et al. A gene expression signature in HER2+ breast cancer patients related to neoadjuvant chemotherapy resistance, overall survival, and disease-free survival. Front Genet. 2022;13:991706. doi:10.3389/fgene.2022.991706. [Google Scholar] [PubMed] [CrossRef]
83. Suh EJ, Kabir MH, Kang UB, Lee JW, Yu J, Noh DY, et al. Comparative profiling of plasma proteome from breast cancer patients reveals thrombospondin-1 and BRWD3 as serological biomarkers. Exp Mol Med. 2012;44(1):36–44. doi:10.3858/emm.2012.44.1.003. [Google Scholar] [PubMed] [CrossRef]
84. Ignatius A, Schoengraf P, Kreja L, Liedert A, Recknagel S, Kandert S, et al. Complement C3a and C5a modulate osteoclast formation and inflammatory response of osteoblasts in synergism with IL-1β. J Cell Biochem. 2011;112(9):2594–605. doi:10.1002/jcb.23186. [Google Scholar] [PubMed] [CrossRef]
85. Ikeda T, Kasai M, Tatsukawa E, Kamitakahara M, Shibata Y, Yokoi T, et al. A bone substitute with high affinity for vitamin D-binding protein―relationship with niche of osteoclasts. J Cell Mol Med. 2014;18(1):170–80. doi:10.1111/jcmm.12180. [Google Scholar] [PubMed] [CrossRef]
86. Hwang SJ, Ha GH, Seo WY, Kim CK, Kim K, Lee SB. Human collagen alpha-2 type I stimulates collagen synthesis, wound healing, and elastin production in normal human dermal fibroblasts (HDFs). BMB Rep. 2020;53(10):539–44. doi:10.5483/bmbrep.2020.53.10.120. [Google Scholar] [PubMed] [CrossRef]
87. Rosset EM, Bradshaw AD. SPARC/osteonectin in mineralized tissue. Matrix Biol. 2016;52-54(1):78–87. doi:10.1016/j.matbio.2016.02.001. [Google Scholar] [PubMed] [CrossRef]
88. Ma J, Gao S, Xie X, Sun E, Zhang M, Zhou Q, et al. SPARC inhibits breast cancer bone metastasis and may be a clinical therapeutic target. Oncol Lett. 2017;14(5):5876–82. doi:10.3892/ol.2017.6925. [Google Scholar] [PubMed] [CrossRef]
89. Ilani T, Alon A, Grossman I, Horowitz B, Kartvelishvily E, Cohen SR, et al. A secreted disulfide catalyst controls extracellular matrix composition and function. Science. 2013;341(6141):74–6. doi:10.1126/science.1238279. [Google Scholar] [PubMed] [CrossRef]
90. Morel C, Adami P, Musard JF, Duval D, Radom J, Jouvenot M. Involvement of sulfhydryl oxidase QSOX1 in the protection of cells against oxidative stress-induced apoptosis. Exp Cell Res. 2007;313(19):3971–82. doi:10.1016/j.yexcr.2007.09.003. [Google Scholar] [PubMed] [CrossRef]
91. Pernodet N, Hermetet F, Adami P, Vejux A, Descotes F, Borg C, et al. High expression of QSOX1 reduces tumorogenesis, and is associated with a better outcome for breast cancer patients. Breast Cancer Res. 2012;14(5):R136. doi:10.1186/bcr3341. [Google Scholar] [PubMed] [CrossRef]
92. Weng Z, Wang Y, Ouchi T, Liu H, Qiao X, Wu C, et al. Mesenchymal stem/stromal cell senescence: hallmarks, mechanisms, and combating strategies. Stem Cells Transl Med. 2022;11(4):356–71. doi:10.1093/stcltm/szac004. [Google Scholar] [PubMed] [CrossRef]
93. Lim SY, Yuzhalin AE, Gordon-Weeks AN, Muschel RJ. Targeting the CCL2-CCR2 signaling axis in cancer metastasis. Oncotarget. 2016;7(19):28697–710. doi:10.18632/oncotarget.7376. [Google Scholar] [PubMed] [CrossRef]
94. Jin Y, Wang Y, Yang R. Chemokine ligand 2: beyond chemotaxis—a multifaceted role in tumor progression. Front Immunol. 2025;16:1685474. doi:10.3389/fimmu.2025.1685474. [Google Scholar] [PubMed] [CrossRef]
95. Chapman J, Fielder E, Passos JF. Mitochondrial dysfunction and cell senescence: deciphering a complex relationship. FEBS Lett. 2019;593(13):1566–79. doi:10.1002/1873-3468.13498. [Google Scholar] [PubMed] [CrossRef]
96. Suda M, Tchkonia T, Kirkland JL, Minamino T. Targeting senescent cells for the treatment of age-associated diseases. J Biochem. 2025;177(3):177–87. doi:10.1093/jb/mvae091. [Google Scholar] [PubMed] [CrossRef]
97. Kirkland JL, Tchkonia T. Senolytic drugs: from discovery to translation. J Intern Med. 2020;288(5):518–36. doi:10.1111/joim.13141. [Google Scholar] [PubMed] [CrossRef]
98. Gasek NS, Kuchel GA, Kirkland JL, Xu M. Strategies for targeting senescent cells in human disease. Nat Aging. 2021;1(10):870–9. doi:10.1038/s43587-021-00121-8. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools