 Open Access
Open Access
REVIEW
Wild Edible Plants As Overlooked Models for Plant Stress Tolerance: Physiological, Metabolic and Applied Perspectives
1 Departamento de Agronomía, CIAMBITAL, University of Almería, Almería, Spain
2 Food Technology Division, ceiA3, CIAMBITAL, University of Almería, Almería, Spain
3 Department of Engineering, University of Almería, Almería, Spain
* Corresponding Author: Mohamed Ezzaitouni. Email:
(This article belongs to the Special Issue: Plant Responses to Stress Factors)
Phyton-International Journal of Experimental Botany 2026, 95(3), 6 https://doi.org/10.32604/phyton.2026.079255
Received 18 January 2026; Accepted 13 March 2026; Issue published 31 March 2026
Abstract
Wild edible plants have evolved in response to persistent and often severe environmental pressures, including salinity, drought, extreme temperatures, high light intensity and nutrient-poor soils. Despite the considerable physiological flexibility and adaptive capacity exhibited by these species, they remain underrepresented in contemporary plant stress research, which has traditionally focused on a limited number of model species and major crops. The present review proposes a conceptual framework that positions wild edible plants as physiological and ecological reference systems for studying naturally evolved plant stress tolerance, rather than as alternative genetic model species. The synthesis of current knowledge on the ecological contexts that shape their stress adaptation is conducted, and the major abiotic stressors influencing wild edible plants across diverse environments are examined. This study focuses on the physiological mechanisms involved in water regulation, osmotic adjustment, photosynthetic performance, and antioxidant defence. The role of metabolic plasticity and stress-induced production of secondary metabolites is also discussed, highlighting how chronic or moderate environmental stress may, under specific conditions, enhance nutritional and functional attributes without necessarily compromising metabolic stability. Furthermore, the potential of wild edible plants as reservoirs of stress-resilient traits relevant to sustainable agriculture, crop diversification, and functional food systems in marginal environments is evaluated. The identification of key research gaps is the final stage of the research process. These gaps include the limited application of omics-based approaches, the lack of standardised experimental methodologies, and the scarcity of direct comparative studies between wild edible and domesticated plant species. The present review underscores the potential benefits and current constraints associated with the utilisation of wild edible plants in promoting plant stress biology and informing climate-resilient agricultural strategies.Graphic Abstract
Keywords
Stressors that affect the growth, development and productivity of a plant are both biotic and abiotic stresses that disrupt metabolic and physiological homeostatic balance and therefore can adversely impact the quality of the crop [1]. Plants are generally exposed to multiple stresses arising both in natural environments and in artificial human-made systems. These stresses require coordinated adaptive responses at the physiological, biochemical, and molecular levels to maintain metabolic balance and ensure healthy growth [2]. For many years, research on plant stresses has tended to be dominated by the use of model species (primarily Arabidopsis thaliana) and a small number of major food crops [3]. Nevertheless, the heavy reliance on domesticated and model species creates limitations for understanding plant stresses. Domesticated crops have been bred intensively for increased yield and uniformity. In addition, Arabidopsis thaliana occupies a narrow ecological niche that does not represent the full range of adaptive responses observed in other higher plant species [4]. In contrast to cultivated edible plants, which have evolved under relatively stable and human-managed conditions, wild edible plants have been shaped by long-term exposure to highly variable ecological stressors. These environments reduce their dependence on controlled conditions and contribute to the development of high phenotypic plasticity and strong adaptive capacity. This highlights their ecological, physiological, and nutritional importance as model systems for linking stress physiology to functional quality in both wild and cultivated species [5,6]. However, existing studies on wild edible plants have largely focused on ethnobotanical documentation or chemical characterization, with comparatively fewer investigations employing these species as model systems for plant stress physiology [7]. Research on wild edible plants (e.g., Sonchus oleraceus, Asparagus acutifolius, Rumex acetosa) examining salinity tolerance and physiological adaptability (i.e., ion regulation) is just beginning to provide the missing pieces of information necessary to connect wild edible plants with sustainable agricultural systems and stress physiology [8,9]. Given their long-term exposure to natural stressors, wild edible plants constitute highly resilient biological systems shaped by evolutionary selection rather than agronomic optimization. Synthesizing current knowledge on their ecological background and stress responses may therefore reveal adaptive mechanisms that are underrepresented in conventional crop-based research and provide valuable insights for climate-resilient agriculture and functional food development [10,11,12]. Unlike previous reviews that focus mainly on ethnobotanical uses or nutritional attributes, the present work integrates ecological adaptation, stress-response mechanisms, and metabolic plasticity. Our objective is to position wild edible plants as physiologically and ecologically informative systems shaped by natural selection. This framework allows us to move beyond descriptive accounts and better interpret naturally evolved stress tolerance. The objective of this conceptual review is thus to position wild edible plants as physiologically and ecologically informative reference systems shaped by long-term natural selection. By integrating ecological context, physiological and metabolic mechanisms, and applied perspectives, this review aims to move beyond descriptive accounts and provide a unifying framework for interpreting stress adaptation in wild plants. Rather than viewing these plants as marginal food resources, we argue that they constitute naturally evolved experimental systems capable of complementing classical model plants and crop-based approaches. Specifically, we first examine the ecological settings and major abiotic stress factors affecting wild edible plants. We then review the key physiological and metabolic mechanisms underlying stress tolerance. Finally, we discuss the implications of these traits for sustainable agriculture, food systems, and future research directions.
2 Ecological Background of Wild Edible Plants
Wild edible plant species have developed in many different types of marginal and heterogeneous environments, where environmental stress is an ongoing and continuous selection pressure rather than a one-time occurrence. These environments include saline soil (salty), long periods of water deficit (drought), continuous high levels of high light intensity (UV), ranges of high and low temperature (cold/heat shocks), and low nutrient levels [13]. Wild edible plant species have a much more direct and ongoing association with abiotic stressors during their entire life cycle, as opposed to cultivated agricultural crops, which are buffered from many of the factors of environmental variability through agronomic practices. In addition, long-term exposure to abiotic stressors has created adaptations that give physiological resilience and ecological success [14]. In arid and semi-arid areas, such as the Mediterranean basin, many wild edible species occupy environments characterized by low water availability and high light intensity, rather than UV radiation, creating strong selection pressures associated with photosynthetic regulation, reduced water loss, and enhanced oxidative stress tolerance. Halophytes and facultative salt-tolerant plant species also occupy environments that have high soil salinity, which increases the need for efficient strategies for ion homeostasis and osmotic adjustment [15]. Because all these environmental factors occur simultaneously, they create conditions that favour the evolution of adaptive syndromes rather than individual stressor responses [16].
An important difference between wild and cultivated plants arises from their respective evolution. The domestication process favoured characteristics such as yield, taste, and uniformity; these traits were often selected for in an optimal growing environment. Although domesticated plants have increased yield, they generally have reduced genetic diversity and decreased stress tolerance compared to many wild-collected edible species [17]. On the other hand, wild edible plant species have been primarily selected through natural selection, where survival and reproduction are based on their ability to cope with fluctuating and challenging environments. As a result, wild edible plants have often developed greater ecological versatility and, in many cases, higher stress tolerance levels relative to cultivated edible plants [18]. The divergence between wild and cultivated plants is directly related to phenotypic plasticity, which denotes the ability of a genotype to produce different phenotypes under varying environmental conditions. High levels of phenotypic plasticity enable wild plants to alter their morphological, physiological, and metabolic characteristics in response to changing conditions, thus enhancing their growth and productivity under various environmental conditions [19]. Results of comparative studies have demonstrated that wild-collected plants grown under natural conditions often have equal or greater amounts of bioactive compounds when compared to those grown under artificially controlled conditions and selected based on their appearance [20,21] Along with plasticity is the concept of ecological resiliency which is defined as the ability of an organism to absorb disturbance and maintain its functional integrity. Wild edible plants demonstrate ecological resilience by sustaining growth, reproduction, and secondary metabolism even under repeated or chronic exposure to stress. Research studies comparing species such as Amaranthus viridis, Sisymbrium irio and Sonchus tenerrimus have shown that the adaptability of these traits is not fully represented in controlled settings and thus provides an example of the potential of wild edible plants as models for research on stress tolerance [21,22,23]. However, these responses remain species and context dependent and should not be generalized across all wild edible plants. Ethnobotanical research supports the ecological perspective. Many of the wild edible plants traditionally harvested and consumed in Mediterranean and arid areas are found on marginal lands specifically because they can thrive under less than desirable soil and growing conditions; therefore, they have historically been utilized for this purpose. In addition, the continued use of these species by humans reflects the cumulative body of cultural knowledge, as well as their inherent biological resiliency, thereby placing these species at the nexus of ecology, nutrition, and stress biology [11,24].
The interplay between ecological, evolutionary and ethnobotanical factors indicates that wild edible plants represent a naturally adapted system for coping with stress due to their prolonged exposure to various types of environmental constraints. In addition, these ecological adaptations create the basis on which physiological and metabolic mechanisms of stress tolerance operate. Thus, it is important to be aware of these ecological adaptations when assessing and interpreting stress responses for the purpose of utilizing these adaptive traits in sustainable agriculture and food systems [25,26].
3 Major Abiotic Stress Factors Affecting Wild Edible Plants
As illustrated in Fig. 1, wild edible plants are continually exposed to multiple abiotic stressors, particularly salinity, drought, extreme temperatures, and high light intensity under natural conditions. These factors often act simultaneously and persist over long periods, creating complex environmental pressures that shape the physiological resilience of these species.
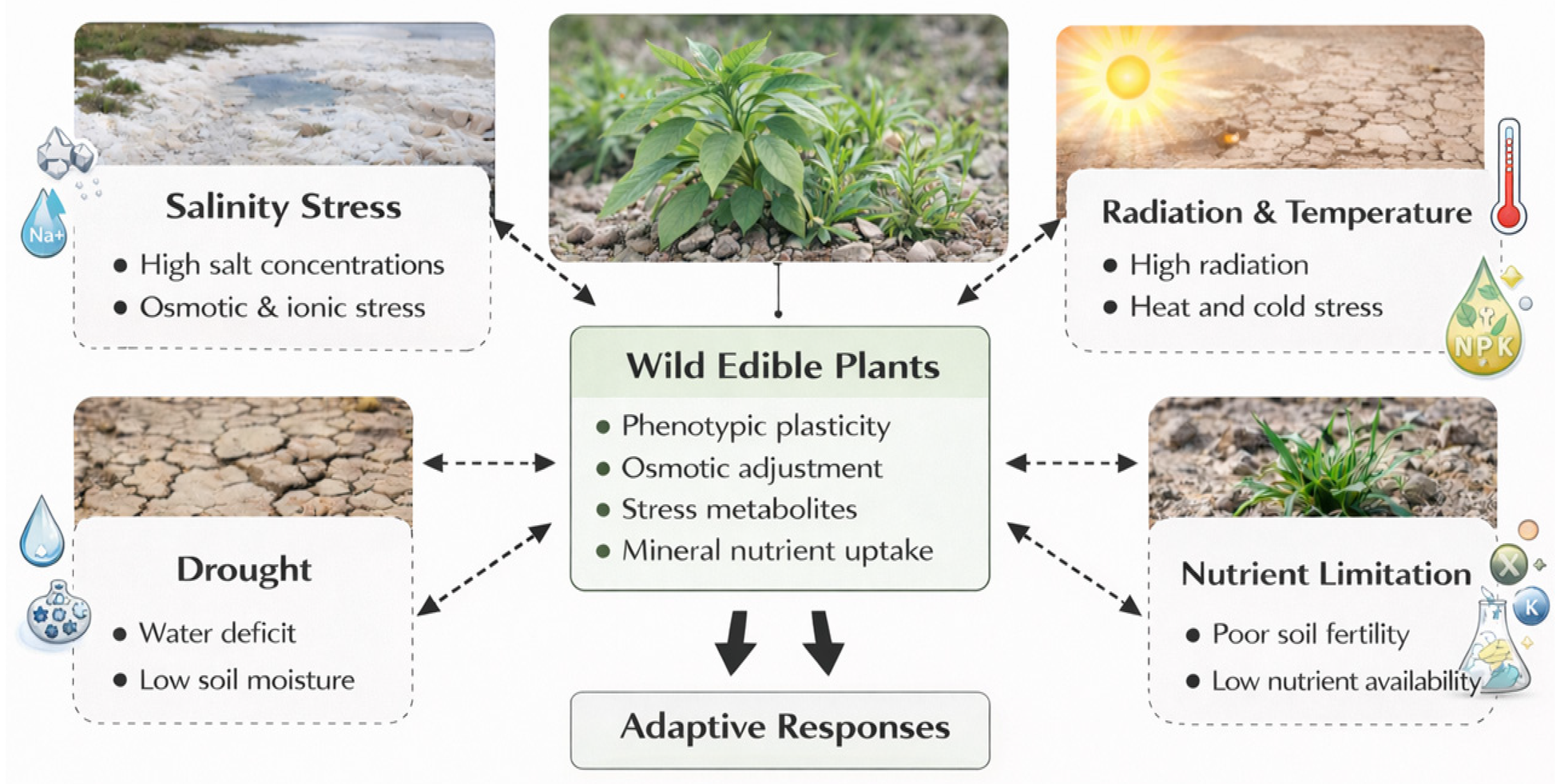
Figure 1: Major abiotic stress factors affecting wild edible plants and their key physiological adaptive responses. This schematic summarizes the main stressors (salinity, drought, high light intensity, temperature extremes, and nutrient limitation) that shape natural environments. It also highlights the core physiological responses osmotic adjustment, phenotypic plasticity, and mineral regulation enabling resilience under chronic stress.
The salt concentration in the soil or irrigation water increases, thereby causing osmotic stress, ion toxicity, and nutrient imbalance, which in turn all contribute to the development of salinity stresses as well as reducing the ability of plants to uptake water and maintain cellular homeostasis. Salinity stress presents a significant barrier to the plant growth of many coastal and arid regions. Salinity exposure in most naturally occurring wild edible plants occurs chronically, as these species typically inhabit persistently saline environments [27]; however, this also occurs acutely, as in the case of seawater intrusion or soil evaporation, both of which require both long-term acclimatization and short-term physiological adjustment [28,29]. Many wild edible plants demonstrate an inherent capacity to tolerate salinity, particularly halophytic and facultatively salt-tolerant species. It has been demonstrated that certain wild plants, which are edible and native to the Mediterranean region, (e.g., Sonchus oleraceus), in addition to specific leafy greens, possess the capacity to thrive and sustain their physiological functions under conditions of moderate salinity [8]. Furthermore, their capacity for adaptation enables them to persist for extended periods in saline environments. The occurrence of edible halophytes (e.g., Salicornia fruticosa, Atriplex halimus) thriving in saline soils offers valuable models for studying the mechanisms of salt tolerance, which have evolved over millennia through continuous natural selection driven by environmental pressures [30]. At the broadest scope, salinity tolerance of wild plant species results from effective osmotic regulation and includes, but is not limited to, a combination of the three major mechanisms of compatible solute accumulation, compartmentalization of ions, and photosynthetic performance under ionic stress. The presence of larger and more effective antioxidants has also been suggested as an additional factor in the alleviation of oxidative damage caused by salt and salt-related stresses [31], enabling the maintenance of metabolic processes in high-salinity environments, especially when the plant is exposed to this chronic stress [32].
Drought-related effects occur when an area has no water for prolonged periods of time, as this leads to lower soil moisture levels and higher levels of drought stress on the growing plant. Areas such as drylands, areas where the average amount of rain falls during a given year and Mediterranean climates are particularly susceptible to experiencing drought stress because they have low levels of annual rainfall and very high evapotranspiration rates [33,34]. In natural ecosystems, wild edible plants experience not only chronic water deficits but also acute drought events; the chronic and acute events work in concert with one another to develop strategies for how wild edible plants adapt to water limitations [35]. Evidence suggests that wild edible plants occurring in dryland and Mediterranean climates often exhibit greater drought adaptability than cultivated plants, as they are routinely exposed to water limitations [36]. These plants have adapted to survive and grow even when there is little available water by regulating how much water they lose from their foliage, making osmotic adjustments, and developing physical structures that reduce transpiration losses [14,37]. The general mechanisms of drought tolerance as found in wild edible and other wild plants consist of regulating stomatal conductance, maintaining cellular moisture level by osmotic adjustment, and maintaining the photosynthetic process in water-limited situations [37]. These processes are augmented by a strong antioxidant defense system [38], which helps maintain a healthy cellular environment and promote cellular growth in response to the drought-induced oxidative stress [39,40].
3.3 High Light Intensity and Temperature Extremes
In many ways, the wild edible plants that are exposed to both high light exposure and hot temperatures are under significant environmental stress due to significant exposure to solar radiation, as well as extreme thermal temperatures. High solar light exposure and thermal extremes can inhibit the machinery of photosynthesis, increase the rate of transpiration and produce reactive oxygen species, especially when combined with limitations in either water or nutrient availability [41,42,43]. Wild edible plants that grow at high light intensity and high-temperature environments exhibit a high degree of tolerance due to long-term exposure to high light intensity/temperature fluctuations. Studies comparing the performance of wild edible plants in natural populations with those cultivated in controlled environments indicate that, under high light intensity, wild edible plants (e.g., Sonchus oleraceus) can maintain photosynthetic efficiency and produce protective metabolites [20,21]. Tolerance to high light intensity and temperature extremes is typically mediated by photoprotective processes, stabilization of photosynthetic efficiency, and activation of antioxidant systems that reduce oxidative damage caused by excessive light intensity and thermal stress. The integration of these responses enables wild edible plants to sustain metabolic activity and growth under the challenging conditions characteristic of natural environments [44,45].
4 Physiological Mechanisms of Stress Tolerance in Wild Plants
Wild edible plants have developed several different ways to cope with environmental stress over a long period of time. This contrasts with domesticated crops that have been selectively bred under more stable and controlled conditions. For example, many domesticated crops are bred for maximum growth and yield, while wild species develop a range of physiological adjustments that assist them in maintaining their internal water status, metabolic balance, and cellular structure when faced with an adverse environment [46]. The ability of wild edible plants to survive under extreme environments results from the combined effects of evolutionary selection and repeated exposure to interacting stressors [47]. However, the intensity and expression of these responses vary widely among species and should not be generalized [8].
4.1 Regulation of Plant Water Status
Regulation of plant water balance is a primary determinant of stress tolerance, particularly under drought and salinity conditions. Wild plants frequently display enhanced control over stomatal conductance, allowing them to limit transpirational water loss while maintaining sufficient carbon assimilation. This balance contributes to the maintenance of leaf water potential and delays the onset of hydraulic failure under water-limited conditions [48,49,50]. Comparative studies indicate that wild species often maintain more stable water potential than cultivated plants under similar stress intensities, reflecting superior regulation of water uptake and loss. Such control is critical in marginal habitats, where fluctuations in soil moisture are frequent and unpredictable [14]. In contrast, many cultivated crops show earlier hydraulic imbalance under comparable stress levels, highlighting reduced flexibility in water-use regulation. However, these responses are species-specific and should not be generalized across all wild edible plants.
4.2 Osmoregulation and Ion Homeostasis
Osmoregulation represents a central physiological strategy in wild plants exposed to salinity and drought. The accumulation of compatible solutes such as proline, soluble sugars, and organic acids contributes to osmotic adjustment, enabling cells to retain water without compromising metabolic processes. In saline environments, the regulation of ion uptake and compartmentalization, particularly sodium and potassium, prevents ionic toxicity and sustains cellular homeostasis [51,52]. Wild edible plants frequently exhibit efficient ion regulation mechanisms, allowing growth and metabolic activity under moderate to high salinity. In contrast, many domesticated crops display reduced tolerance due to limited capacity for ion exclusion or vacuolar sequestration, underscoring a key physiological divergence shaped by domestication [8].
4.3 Photosynthetic Adjustments under Environmental Stress
Stress conditions commonly impair photosynthesis through stomatal limitation, damage to the photosynthetic apparatus, or disruption of electron transport [53]. Wild plants adapted to high light stress, salinity, or water deficit often exhibit pronounced photosynthetic plasticity, including adjustments in light harvesting, photochemical efficiency, and carbon fixation pathways [47,54]. Experimental comparisons between natural environment and controlled-environment cultivation indicate that wild plants can maintain higher photosystem II efficiency and sustain photosynthetic performance under stress conditions [47]. This capacity reflects both structural and biochemical acclimation mechanisms that mitigate photoinhibition and preserve energy balance [55]. By contrast, cultivated plants frequently exhibit stronger declines in photosynthetic efficiency when exposed to similar stress intensities [21,56,57].
4.4 Antioxidant Defense Systems
Abiotic stress is commonly associated with excessive production of reactive oxygen species (ROS), which can damage lipids, proteins, and nucleic acids. Wild plants typically possess robust antioxidant defense systems, encompassing both enzymatic components (e.g., superoxide dismutase, catalase, and peroxidases) and non-enzymatic molecules such as phenolic compounds, flavonoids, and carotenoids [46,58]. Enhanced antioxidant capacity has been repeatedly observed in wild edible plants grown under natural stress conditions, contributing to oxidative stress mitigation and metabolic stability. Compared with many cultivated species, wild plants often show higher baseline levels of antioxidant metabolites, reflecting an adaptive strategy for coping with recurrent oxidative challenges [20]. However, maintaining enhanced antioxidant capacity and physiological flexibility may involve metabolic costs that can constrain growth or reproductive output under non-stress conditions [59].
4.5 Wild Versus Domesticated Plants: a Physiological Perspective
Taken together, these physiological mechanisms highlight fundamental differences between wild and domesticated plants. While domestication has favored traits related to productivity and uniformity under managed environments, it has often resulted in reduced physiological flexibility and stress resilience. Wild edible plants, in contrast, maintain a broader repertoire of adaptive responses, including tighter water regulation, effective osmotic adjustment, flexible photosynthetic behavior, and strong antioxidant defenses [12,60]. These differences suggest that wild plants represent not only alternative species, but alternative stress-response paradigms, shaped by natural selection under chronic environmental pressure rather than by agronomic optimization. As such, wild edible plants constitute valuable biological models for understanding naturally evolved stress tolerance and for informing strategies aimed at improving crop resilience under increasingly variable and unpredictable environmental conditions [18,61]. Table 1 synthesizes these physiological contrasts, reinforcing the argument that wild edible plants represent alternative stress-response paradigms shaped by natural selection rather than agronomic optimization.
Table 1: Comparative overview of stress-response mechanisms in wild edible plants and domesticated crops.
| Trait | Wild Edible Plants | Domesticated Crops | References |
|---|---|---|---|
| Water regulation | Tight stomatal control; stable leaf water potential under stress | Earlier hydraulic imbalance; reduced flexibility | [62,63] |
| Osmoregulation | Efficient ion compartmentalization; high compatible solute accumulation | Limited Na+ exclusion; lower osmotic adjustment capacity | [64] |
| Photosynthetic performance | High photochemical flexibility; reduced photoinhibition | Greater decline in PSII efficiency under stress | [65] |
| Antioxidant systems | Elevated baseline antioxidant activity; stable ROS scavenging | More inducible but often lower baseline antioxidant capacity | [66] |
| Phenotypic plasticity | High environmental responsiveness | Reduced plasticity due to breeding selection | [67] |
Nevertheless, the expression and effectiveness of these physiological traits remain strongly dependent on species identity, environmental context, and experimental conditions.
5 Metabolic Plasticity and Secondary Metabolite Accumulation
One of the most distinctive features of wild edible plants under environmental stress is their pronounced metabolic plasticity, which enables dynamic modulation of secondary metabolism in response to adverse conditions. Unlike primary metabolism, which is directly linked to growth and reproduction, secondary metabolism plays a central role in plant defense, stress mitigation, and ecological interactions. In wild plants, long-term exposure to abiotic stressors has favored the evolution of metabolic strategies that not only ensure survival but also promote the accumulation of bioactive compounds with nutritional and functional relevance [68,69,70]. While Fig. 1 summarises the environmental stressors, Fig. 2 integrates the corresponding physiological pathways.
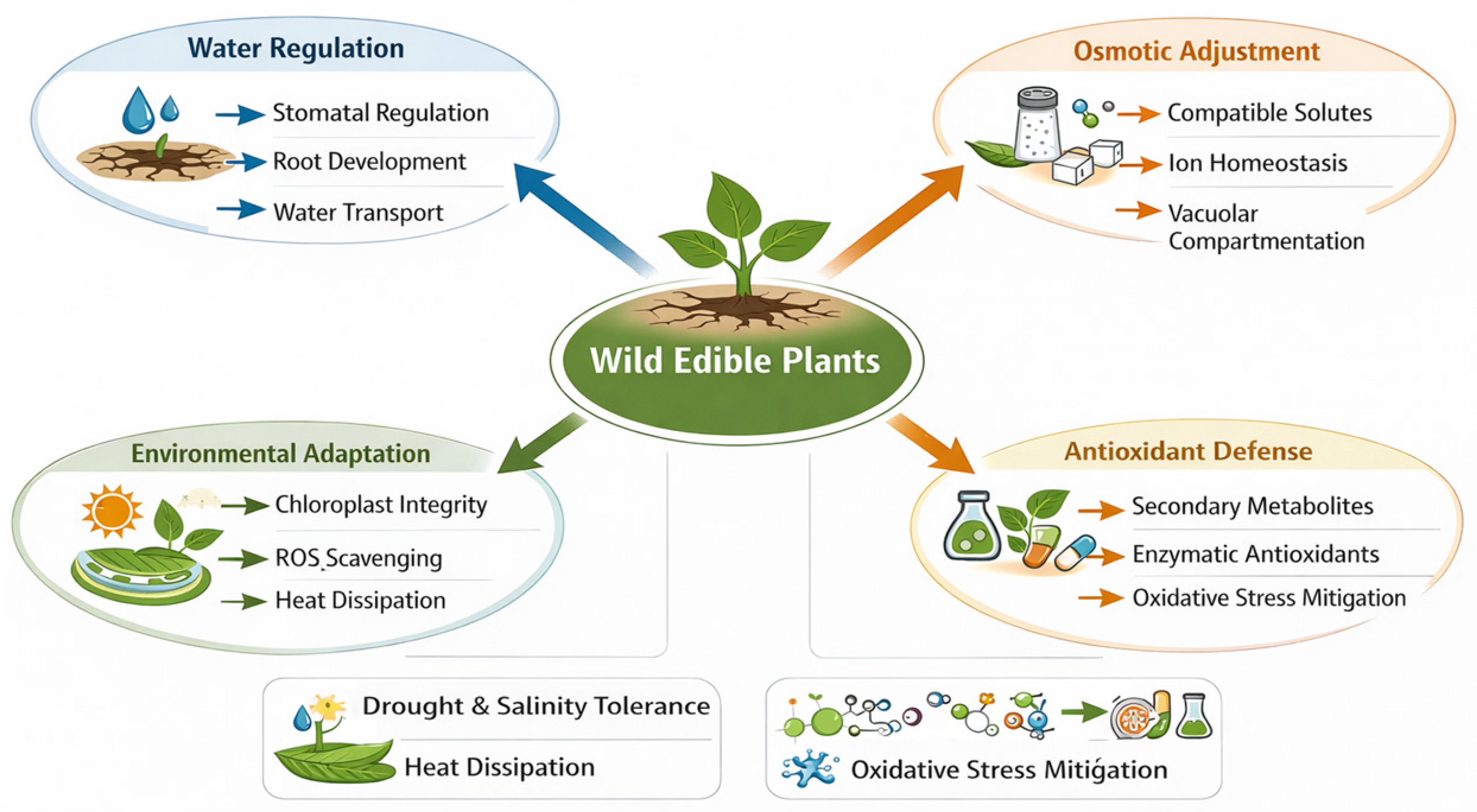
Figure 2: Integrated physiological responses of wild edible plants to abiotic stress. The diagram integrates water regulation, osmotic adjustment, ROS scavenging, and chloroplast protection as central mechanisms supporting stress tolerance. These processes collectively maintain metabolic stability when plants face salinity, drought, or high light intensity.
Abiotic stresses such as salinity, drought high light intensity and temperature extremes stimulate the biosynthesis of phenolic compounds, flavonoids, and terpenoids. These metabolites contribute to stress tolerance through antioxidant activity, membrane stabilization, and protection of the photosynthetic apparatus [71]. In wild edible plants, stress-induced metabolic responses are often more pronounced and consistent than in cultivated species, reflecting adaptive regulation rather than stress-related metabolic impairment [72,73]. Phenolic compounds constitute one of the most responsive metabolite classes under stress conditions. Increased total phenolic content (TPC) has been consistently reported in wild plants growing under natural field stress, where enhanced phenylpropanoid pathway activity is closely associated with oxidative stress mitigation [74]. Similarly, flavonoids perform a dual function as antioxidants and photoprotective agents, particularly under high light and drought conditions. Terpenoids, including carotenoids and other isoprenoids, further contribute to stress resilience by supporting photoprotection, membrane stabilization, and reactive oxygen species scavenging [75,76].
This adaptive metabolic behaviour supports the idea that moderate or chronic stress can enhance nutritional and functional quality [8]. However, not all stress levels promote metabolite accumulation. Stress intensity and duration strongly influence metabolic outcomes, and excessively severe stress may reduce plant quality [74]. However, not all stress levels enhance metabolite accumulation, highlighting the importance of stress intensity and duration in determining metabolic outcomes. Comparative studies between natural and controlled-environment cultivation have demonstrated that wild plants frequently accumulate higher levels of antioxidants and other bioactive metabolites under adverse environmental conditions [20]. Consequently, plants grown under controlled conditions where stress factors are minimized typically exhibit lower antioxidant concentrations, underscoring the value of wild species both as functional foods and as biological models for stress-responsive metabolism [21,77]. Importantly, this metabolic plasticity contrasts with the responses typically observed in domesticated crops, where stress often results in yield penalties and more variable or transient metabolite accumulation. As illustrated in Table 2, stress-associated increases in phenolics, flavonoids, and antioxidant capacity are recurrent across multiple wild edible species [8]. In these plants, the induction of secondary metabolism represents a stable and naturally selected adaptive trait, rather than a transient damage response [20]. These characteristics highlight the relevance of wild edible plants not only as functional food sources but also as experimental systems for understanding stress-responsive metabolic regulation [77].
Table 2: Examples of stress-associated increases in bioactive compounds in wild edible plants.
| Species (Wild-Grown) | Stress Context/Growth Condition | Key Metabolite Indicator | Reported Value* | Reference |
|---|---|---|---|---|
| Asparagus spp. (wild shoots) | Natural field conditions (Mediterranean) | Total phenolic content (TPC) | ~18–22 mg GAE g−1 dw | [20] |
| Amaranthus viridis L. | Natural vs. controlled environment | Antioxidant activity (DPPH) | Higher in wild plants (↑ ~30%) | [21] |
| Sisymbrium irio L. | Growth in nature under high light intensity | (TPC) | ~12–15 mg GAE g−1 dw | [22] |
| Salicornia fruticosa | Saline soils | Antioxidant capacity (FRAP/DPPH) | Increased antioxidant activity under salinity | [78] |
| Atriplex halimus/Salicornia fruticosa/Cakile maritima | Saline conditions | Total phenolic content | Increased under salinity vs. non-saline | [79] |
Metabolic Trade-Offs and Resource Allocation under Stress
While enhanced antioxidant capacity and secondary metabolite accumulation contribute to stress tolerance, these adaptive responses may incur significant metabolic costs. The allocation of carbon, energy, and reducing power towards defensive pathways, such as phenylpropanoid biosynthesis, ROS scavenging, and stress responsive hormone networks, often results in a reduction of investment in growth and reproduction under non-stress conditions [74]. This reflects a fundamental trade-off between resilience and productivity. Recent research into plant specialised metabolism under stress highlights how multi-omics-integrated regulatory networks orchestrate increased biosynthesis of defensive compounds at the expense of primary growth pathways, linking stress signalling with resource partitioning in plants [71]. Analyses of trait trade-offs indicate that integrative adaptation strategies, including coordination between above- and below-ground traits, contribute to ecological resilience. These strategies may also influence resource distribution patterns [80]. In wild edible plants, prolonged exposure to fluctuating environmental pressures appears to have fine-tuned balance, thereby enabling sustained metabolic performance and defence capacity without complete inhibition of growth [8]. It is imperative to comprehend these metabolic trade-offs in order to assess the transferability of stress-resilient traits into high-yield cropping systems. In such systems, excessive allocation of resources to defence may potentially compromise productivity [74]. These trade-offs illustrate how wild plants balance resilience and resource allocation, making them valuable comparative models for improving crop stress tolerance [77].
6 Wild Edible Plants As Reservoirs of Stress-Resilient Traits
Wild edible plants (e.g., Sonchus oleraceus, Amaranthus viridis) represent an important and still underutilized reservoir of stress-resilient traits that can be leveraged to enhance agricultural sustainability under increasingly challenging environmental conditions [8]. Having evolved under persistent abiotic constraints, these species harbor adaptive features that confer tolerance to salinity, water scarcity, and oxidative stress-traits that are increasingly valuable in modern crop systems facing climate change and land degradation [12]. It is important to note that the transferability of these traits is species-specific and strongly modulated by environmental conditions, which limits broad generalizations [81].
6.1 Edible Halophytes As Sources of Salinity Tolerance
Edible halophytes (e.g., Salicornia fruticosa, Atriplex halimus) are among the most emblematic examples of naturally evolved salt tolerance. These species thrive in saline soils and coastal environments where conventional crops frequently fail, making them valuable models for understanding and exploiting salinity resilience [27]. Their adaptive traits include efficient ion compartmentalization, osmotic adjustment through compatible solutes, and sustained photosynthetic activity under elevated salt concentrations [30,51]. Beyond their ecological relevance, edible halophytes also exhibit significant nutritional and functional value, often characterized by high levels of phenolic compounds and antioxidant capacity. These combined agronomic and nutritional attributes position halophytes as strategic biological resources for saline agriculture and as physiological and ecological references for the development of salt-tolerant cropping systems [14,82].
6.2 Mediterranean Wild Plants and Drought Resilience
Mediterranean wild edible plants (e.g., Sisymbrium irio, Rumex vesicarius, Cichorium intybus) are adapted to environments characterized by seasonal drought, high light intensity and nutrient-poor soils. These conditions have been selected for traits associated with efficient water use, pronounced phenotypic plasticity, and resilience to oxidative stress [22]. Compared with domesticated crops, wild Mediterranean species often maintain physiological stability and metabolic activity under prolonged water limitation [27,83]. Comparative studies between natural and controlled cultivated conditions indicate that species such as Amaranthus viridis, Sisymbrium irio, and Sonchus tenerrimus exhibit robust performance and enhanced phytochemical accumulation when grown under natural stress conditions. These responses reflect intrinsic adaptive traits that could inform crop improvement strategies or support the development of alternative and resilient crop systems [21,22,23]. Although wild edible plants frequently show higher levels of phytochemicals than cultivated species, experimental validation is required in each specific case to exclude the influence of abiotic factors (e.g., drought, fungal infections, high light intensity, poor soil nutrient availability, and similar stressors) that may artificially enhance the phytochemical content of wild-grown plants [84]. Therefore, additional controlled experimentation is needed to determine whether these differences truly reflect intrinsic species-specific traits or simply environmentally induced responses [77].
6.3 Traditional Wild Vegetables As Models of Metabolic Robustness
Traditional wild vegetables (e.g., Portulaca oleracea, Amaranthus viridis, Cichorium intybus) consumed for generations in marginal regions represent an additional pool of stress-resilient traits. Their continued inclusion in local diets reflects not only accumulated cultural knowledge but also consistent biological performance under adverse environmental conditions [85]. These species frequently exhibit metabolic robustness, characterized by stable or enhanced production of bioactive compounds under stress, rather than stress-induced metabolic collapse [86,87]. Such robustness is particularly relevant for agricultural systems targeting functional food production, where nutritional quality is as critical as yield stability. Traditional wild vegetables therefore provide valuable insights into how stress-responsive metabolism can be integrated into sustainable food systems without compromising plant performance [77,85].
6.4 Transferable Traits and Agricultural Implications
The adaptive traits exhibited by wild edible plants (e.g., Sisymbrium irio, Rumex vesicarius) can be regarded as transferable assets for agriculture through various pathways, including the direct cultivation of wild or semi-domesticated species, conventional breeding methods, and management strategies that exploit stress-induced resilience [81]. Salinity tolerance, water-use efficiency and metabolic stability under environmental stress are three key traits of interest, all of which are increasingly relevant for future crop systems [85,88]. However, the direct genetic transfer of stress-resilient traits from wild to cultivated species is often constrained by reproductive barriers, complex trait inheritance, and trade-offs between stress tolerance and yield. Consequently, wild edible plants should be regarded not only as potential genetic donors but also as physiological and ecological references capable of informing breeding priorities and adaptive management strategies [81,89]. As illustrated in Table 2, the principal stress-resilient traits identified in the species under consideration are synthesised, with particular emphasis on their relevance for agricultural systems facing salinity, drought, and climatic instability. Table 3 further summarizes the key stress-resilient traits identified in wild edible plants and highlights their potential applications in agriculture. The concrete applications of this theory provide further evidence for its translational value. Edible halophytes such as Salicornia fruticosa and Atriplex halimus are cultivated in saline agriculture systems [79,82], while stress-related traits from wild relatives have informed breeding programmes targeting improved ion regulation and drought tolerance [81,89]. Concurrently, controlled moderate stress (e.g., deficit irrigation) has been investigated to enhance phenolic content and antioxidant capacity [90], thereby establishing a correlation between physiological resilience and the quality of functional food. For instance, traits related to ion compartmentalization in Atriplex halimus have been used to design saline agriculture systems, and drought-resilient features of Sisymbrium irio have informed stress-tolerant crop ideotypes [27].
Table 3: Wild edible plants as reservoirs of stress-resilient traits with agricultural relevance.
| Plant Group | Representative Examples | Main Stress Tolerated | Key Adaptive Traits | Agricultural Relevance | Reference |
|---|---|---|---|---|---|
| Edible halophytes | Atriplex halimus, Salicornia fruticosa, Cakile maritima | Salinity | Ion compartmentalization, osmotic adjustment, antioxidant capacity | Saline agriculture, marginal lands | [79] |
| Mediterranean wild plants | Amaranthus viridis, Sisymbrium irio | Drought, high light intensity | Water-use efficiency, phenotypic plasticity | Climate-resilient crops | [21,22] |
| Wild leafy vegetables | Sonchus spp. | Salinity, drought | Physiological stability, metabolic plasticity | Alternative leafy crops | [8] |
According to studies, wild edible plants have been shown to exhibit tighter stomatal regulation, more efficient ion compartmentalisation, greater photochemical stability under highlight, and higher baseline antioxidant capacity in comparison to domesticated crops [8]. Conversely, domesticated species characteristically demonstrate diminished physiological adaptability and accelerated functional deterioration in response to stress, which is indicative of a selective focus on yield over resilience [58,71]. These recurring patterns underscore the significance of wild edible plants as valuable complementary reference systems for understanding naturally evolved stress tolerance [77].
7 Implications for Sustainable Agriculture and Food Systems
The growing body of evidence on stress tolerance in wild edible plants (e.g., Sisymbrium irio, Rumex vesicarius) has important implications for the development of more resilient agricultural systems and food chains. As climate change intensifies environmental constraints such as salinity, drought, and temperature extremes, conventional crop systems increasingly face limitations in maintaining productivity under marginal conditions [37,52]. In this context, wild edible plants provide biologically grounded insights that support the re-evaluation of agricultural strategies beyond yield centred approaches [27]. The broader implications of their physiological and metabolic traits are summarised in Fig. 3, which integrates ecological adaptation, crop diversification, and functional food development into a unified sustainability framework.
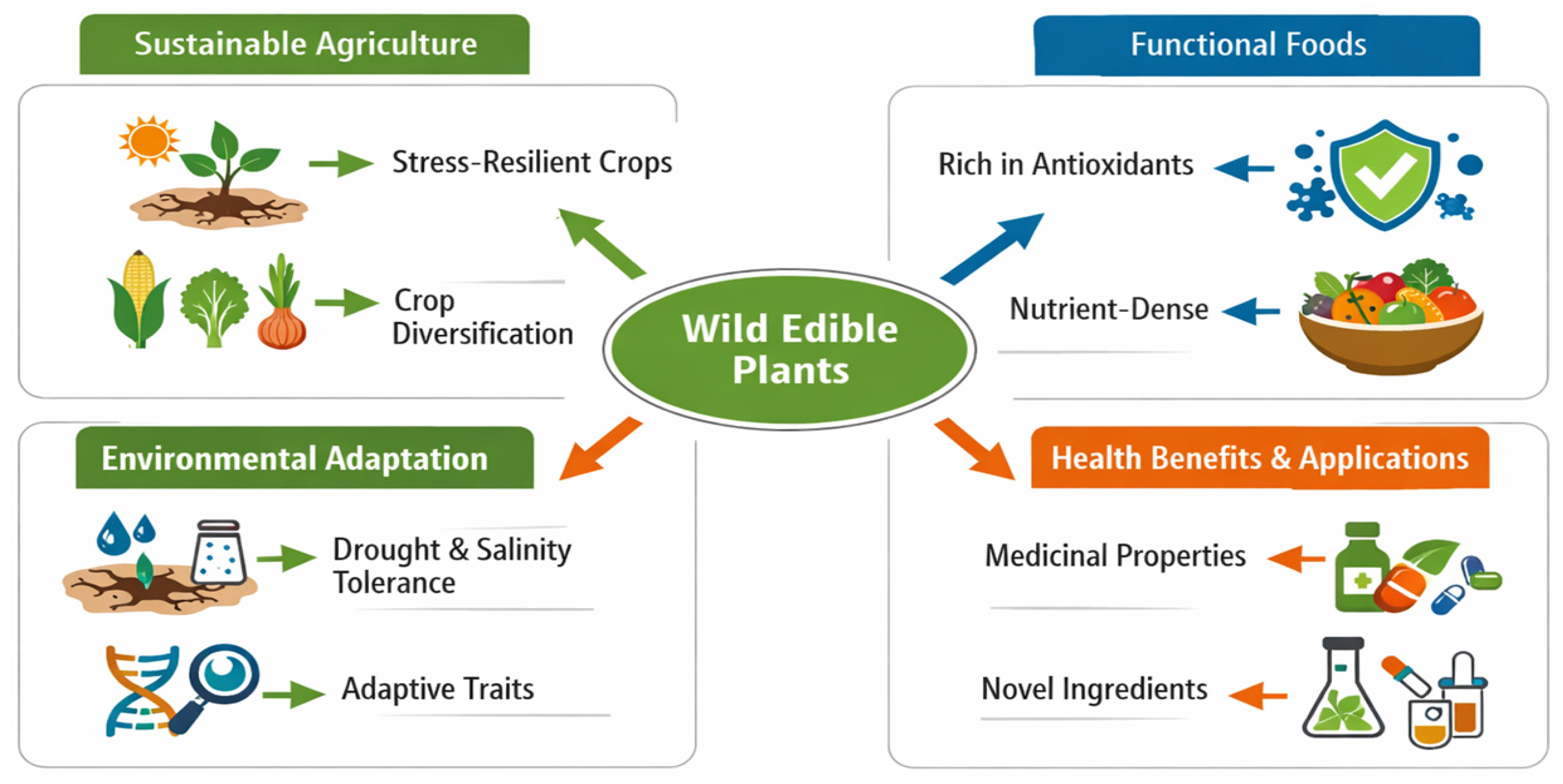
Figure 3: Implications of wild edible plants for sustainable agriculture and resilient food systems. The figure illustrates how stress-resilient traits can support crop diversification, low-input farming, and the development of antioxidant-rich functional foods. It highlights the relevance of wild edible plants as complementary resources for climate-adaptive agriculture.
The resilience of wild edible plants is underpinned by key physiological and metabolic traits, including efficient water-use regulation, robust antioxidant systems, and flexible secondary metabolism [37]. These characteristics can inform the design of low-input agricultural strategies. One direct application lies in farming systems established on marginal lands. These include saline soils, arid regions, and low-fertility environments [27]. Many wild edible species are naturally adapted to such conditions and can be cultivated with reduced water and nutrient inputs, thereby lowering external resource dependence while maintaining functional performance [14,82,91]. Wild edible plants may also serve as alternative or complementary crops within diversified production systems. Their potential domestication or semi-domestication may broaden the crop portfolio, reduce reliance on a limited number of intensively bred species, and enhance agroecosystem stability [27]. Unlike highly improved crops, wild plants often retain greater genetic diversity and phenotypic plasticity, traits closely linked to physiological adaptability under unpredictable climatic conditions [12,92]. Comparative studies between natural conditions and controlled cultivation further indicate that several species can sustain productivity while preserving elevated phytochemical levels, supporting their feasibility for future cropping systems [21]. Nevertheless, the successful integration of wild edible plants into agricultural and food systems requires careful consideration of agronomic feasibility, consumer acceptance, and supply-chain constraints [85]. Beyond agronomic factors, the stress-induced accumulation of bioactive compounds in wild edible plants has clear implications for functional food production. Enhanced levels of phenolics, flavonoids, and antioxidant capacity under moderate stress conditions reflect metabolically regulated responses rather than stress damage [77]. These characteristics link physiological stress tolerance with nutritional quality, reinforcing the relevance of wild edible plants at the interface between sustainable agriculture and nutrition science [59,90]. At a broader systems level, the integration of wild edible plants into agricultural and food systems may contribute to climate-change adaptation by increasing biological diversity and reducing vulnerability to environmental shocks [93]. Knowledge derived from the physiological and metabolic basis of stress tolerance in wild plants therefore extends beyond crop improvement and supports the development of more diversified, resilient, and sustainable food systems [12,93]. Together, these considerations highlight the relevance of wild edible plants not as universal solutions, but as complementary components of more diversified and resilient agri-food systems.
8 Knowledge Gaps and Future Research Directions
Despite the growing interest in wild edible plants as sources of stress tolerance and functional value, significant knowledge gaps remain that currently limit their full integration into plant stress biology and sustainable agriculture. For example, omics-based studies on wild species are still scarce, restricting our understanding of the molecular networks underpinning their stress tolerance [71,94]. Addressing these gaps represents not only a scientific challenge but also a strategic opportunity to expand current paradigms in plant stress biology [77]. Future research should adopt experimental approaches that quantify adaptive plasticity and establish links between physiological and metabolic responses under controlled conditions. Common-garden and stress-gradient assays, which are widely utilised to characterise differential plasticity between wild and domesticated species [95]. Have the potential to facilitate the delineation of species-specific tolerance thresholds. In a similar manner, the implementation of controlled salinity or irradiance gradients would facilitate the identification of physiological trade-offs and limits of plasticity. Integrated multi-omics frameworks, which have proven highly effective for the analysis of regulatory networks involved in plant stress responses [94,96]. Could further clarify the mechanisms underpinning stress-induced metabolic plasticity in wild edible plants.
8.1 Limited Application of Omics Approaches in Wild Edible Plants
Most omics-based studies, including genomics, transcriptomics, proteomics, and metabolomics, have been conducted on model species and major crops. In contrast, wild edible plants remain largely unexplored at the systems biology level [84]. This limitation restricts our understanding of the molecular networks and regulatory pathways underlying their stress tolerance and metabolic plasticity. Expanding omics approaches to wild species would enable the identification of novel stress-responsive genes and metabolites shaped by natural selection [94,96,97]. For example, integrating metabolomics and transcriptomics in species such as Sisymbrium irio or Atriplex halimus would help identify stress-inducible pathways that cannot be detected using classical physiological measurements [58].
8.2 Scarcity of Experiments under Controlled Environments
A large proportion of studies on wild edible plants rely on field observations or naturally grown samples, where environmental variability can complicate mechanistic interpretation [93]. Controlled-environment experiments remain scarce, yet they are essential for isolating the effects of individual stress factors and ensuring experimental reproducibility. Future research should prioritize well-designed controlled studies to complement field-based evidence and to enable robust cross-species comparisons [61,98]. For instance, salinity-gradient hydroponic trials could be used to isolate specific ion-regulation mechanisms, while controlled irradiance chambers would help quantify photoprotection thresholds in wild versus domesticated plants [42].
8.3 Lack of Methodological Standardization
Research on wild edible plants is characterized by substantial heterogeneity in experimental protocols, including stress application, growth conditions, and analytical methods for metabolite quantification [78]. This lack of standardization hinders direct comparison among studies and limits the potential for meta-analytical approaches [78]. The development of standardized experimental frameworks would substantially improve data comparability and scientific robustness in this emerging field [61,99,100]. As an example, adopting unified protocols for measuring antioxidant activity (e.g., DPPH, FRAP) or standardized electrical conductivity levels for salinity trials would facilitate direct comparison across studies [21].
8.4 Insufficient Comparative Studies with Domesticated Crops
Direct comparative studies between wild edible plants and domesticated crops grown under identical stress conditions remain limited. Such comparisons are critical for identifying traits that are genuinely associated with wild stress adaptation and for evaluating their potential transferability to agricultural systems. Future research should incorporate parallel assessments of wild and cultivated species to bridge the gap between ecological adaptation and agronomic application [101,102,103]. Parallel cultivation of wild and domesticated species under identical conditions, such as comparing Amaranthus viridis with domesticated spinach, would help determine which traits are truly intrinsic versus environmentally induced [69].
8.5 Need for Integrative and Interdisciplinary Approaches
Current research efforts are often fragmented across disciplines such as ecology, plant physiology, nutrition, and agronomy. Integrative approaches that combine these perspectives are required to fully capture the complexity of stress tolerance in wild edible plants and to translate biological insights into practical solutions for sustainable agriculture and resilient food systems [12,104,105]. Addressing these research gaps is imperative for the advancement of wild edible plants as informative biological models and for the translation of stress tolerance knowledge into sustainable agri-food solutions. For example, combining ecological field surveys with controlled physiological assays and metabolomic profiling would provide a more complete understanding of how wild edible plants respond to both natural and simulated stress conditions [97].
Wild edible plants offer a compelling and still underexploited perspective in plant stress biology. Shaped by long-term exposure to environmental constraints such as salinity, drought, high light intensity and nutrient limitation, these species have developed integrated physiological and metabolic strategies that support resilience and functional performance under stress. The evidence reviewed here indicates that wild plants frequently maintain water balance, photosynthetic efficiency, and metabolic stability while simultaneously enhancing the accumulation of bioactive compounds in response to adverse environmental conditions. Unlike many domesticated crops, whose stress responses are often constrained by breeding for high-input agricultural systems, wild edible plants retain high levels of phenotypic plasticity and adaptive flexibility. This characteristic makes them particularly valuable for understanding naturally evolved stress tolerance mechanisms and for identifying traits relevant to climate-resilient agriculture. Their ability to couple stress tolerance with enhanced nutritional and functional quality further positions wild edible plants at the intersection of sustainability, nutrition science, and plant physiology. Wild edible plants therefore represent untapped biological models for understanding and exploiting plant stress tolerance. Future research integrating omics-based approaches, standardized experimental designs, and direct comparisons with cultivated crops will be essential to fully harness their potential. Advancing this knowledge will contribute not only to crop diversification and sustainable agriculture but also to the development of resilient food systems capable of addressing the challenges imposed by global environmental change. Recognizing wild edible plants as informative biological models rather than marginal species represents an important conceptual shift in plant stress research.
Acknowledgement:
Funding Statement: The authors received no specific funding for this study.
Author Contributions: Conceptualization, Hajiba Benteima and Mohamed Ezzaitouni; methodology, Hajiba Benteima, Mohamed Ezzaitouni, Tarik Chileh-Chelh, Carlos Galindo and José Luis Guil-Guerrero; software, Hajiba Benteima and Mohamed Ezzaitouni; validation, José Luis Guil-Guerrero; formal analysis, Hajiba Benteima and Mohamed Ezzaitouni; investigation, Hajiba Benteima and Mohamed Ezzaitouni, Tarik Chileh-Chelh, Carlos Galindo and José Luis Guil-Guerrero; resources, Mohamed Ezzaitouni and José Luis Guil-Guerrero; data curation, Hajiba Benteima, Mohamed Ezzaitouni and José Luis Guil-Guerrero; writing—original draft preparation, Hajiba Benteima, Mohamed Ezzaitouni, Tarik Chileh-Chelh, Carlos Galindo and José Luis Guil-Guerrero; writing—review and editing, Hajiba Benteima, Mohamed Ezzaitouni and José Luis Guil-Guerrero; visualization, Mohamed Ezzaitouni and José Luis Guil-Guerrero; supervision, José Luis Guil-Guerrero; project administration, José Luis Guil-Guerrero. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: Not applicable.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Suzuki N , Rivero RM , Shulaev V , Blumwald E , Mittler R . Abiotic and biotic stress combinations. New Phytol. 2014; 203( 1): 32– 43. doi:10.1111/nph.12797. [Google Scholar] [CrossRef]
2. Zandalinas SI , Fichman Y , Mittler R . Vascular bundles mediate systemic reactive oxygen signaling during light stress. Plant Cell. 2020; 32( 11): 3425– 35. doi:10.1105/tpc.20.00453. [Google Scholar] [CrossRef]
3. Koornneef M , Meinke D . The development of Arabidopsis as a model plant. Plant J. 2010; 61( 6): 909– 21. doi:10.1111/j.1365-313X.2009.04086.x. [Google Scholar] [CrossRef]
4. Exposito-Alonso M , Burbano HA , Bossdorf O , Nielsen R , Weigel D . Natural selection on the Arabidopsis thaliana genome in present and future climates. Nature. 2019; 573( 7772): 126– 9. doi:10.1038/s41586-019-1520-9. [Google Scholar] [CrossRef]
5. Khare S , Singh NB , Singh A , Hussain I , Niharika KM , Yadav V , et al. Plant secondary metabolites synthesis and their regulations under biotic and abiotic constraints. J Plant Biol. 2020; 63( 3): 203– 16. doi:10.1007/s12374-020-09245-7. [Google Scholar] [CrossRef]
6. Ntakoulas N , Karkanis A , Martins N , Petropoulos SA . Nutritional and functional properties of wild edible greens. Foods. 2025; 14( 2): 198. doi:10.3390/foods14020198. [Google Scholar] [CrossRef]
7. Ramos-Muñoz M , Castellanos MC , Blanco-Sánchez M , Pías B , Ramírez-Valiente JA , Benavides R , et al. Drivers of phenotypic variation and plasticity to drought in populations of a Mediterranean shrub along an environmental gradient. Environ Exp Bot. 2024; 228: 106011. doi:10.1016/j.envexpbot.2024.106011. [Google Scholar] [CrossRef]
8. Gkotzamani A , Ipsilantis I , Menexes G , Katsiotis A , Mattas K , Koukounaras A . The impact of salinity in the irrigation of a wild underutilized leafy vegetable, Sonchus oleraceus L. Plants. 2024; 13( 11): 1552. doi:10.3390/plants13111552. [Google Scholar] [CrossRef]
9. Bharucha Z , Pretty J . The roles and values of wild foods in agricultural systems. Philos Trans R Soc Lond B Biol Sci. 2010; 365( 1554): 2913– 26. doi:10.1098/rstb.2010.0123. [Google Scholar] [CrossRef]
10. Clemente-Villalba J , Burló F , Hernández F , Carbonell-Barrachina ÁA . Valorization of wild edible plants as food ingredients and their economic value. Foods. 2023; 12( 5): 1012. doi:10.3390/foods12051012. [Google Scholar] [CrossRef]
11. Alemayehu G , Awoke A , Kassa Z . Wild edible plant species and their role in nutrition and health in Korahe Zone, Eastern Ethiopia. Trop Med Health. 2025; 53( 1): 182. doi:10.1186/s41182-025-00867-6. [Google Scholar] [CrossRef]
12. Polyzos N , Liava V , Antoniadis V , Garcia P , Alexopoulos AA , Petropoulos SA . The new trends in wild edible plants valorization: Commercial cultivation protocols, agronomic practices and future challenges. Front Hortic. 2025; 4: 1638703. doi:10.3389/fhort.2025.1638703. [Google Scholar] [CrossRef]
13. Lovett-Doust J . Plant strategies, vegetation processes, and ecosystem properties. J Veg Sci. 2002; 13( 2): 294. doi:10.1658/1100-9233(2002)013[0294:PSVPAE]2.0.CO;2. [Google Scholar] [CrossRef]
14. Christoforidi I , Kollaros D , Manios T , Daliakopoulos IN . Drought- and salt-tolerant plants of the Mediterranean and their diverse applications: The case of Crete. Land. 2022; 11( 11): 2038. doi:10.3390/land11112038. [Google Scholar] [CrossRef]
15. Flowers TJ , Colmer TD . Plant salt tolerance: Adaptations in halophytes. Ann Bot. 2015; 115( 3): 327– 31. doi:10.1093/aob/mcu267. [Google Scholar] [CrossRef]
16. Meyer RS , Purugganan MD . Evolution of crop species: Genetics of domestication and diversification. Nat Rev Genet. 2013; 14( 12): 840– 52. doi:10.1038/nrg3605. [Google Scholar] [CrossRef]
17. Qiao X , Hao Z , Liu H , Liu Y . A global meta-analysis of the effects of irrigation with water containing salts on saline-alkali soils. Agric Water Manag. 2026; 325: 110202. doi:10.1016/j.agwat.2026.110202. [Google Scholar] [CrossRef]
18. Nicotra AB , Atkin OK , Bonser SP , Davidson AM , Finnegan EJ , Mathesius U , et al. Plant phenotypic plasticity in a changing climate. Trends Plant Sci. 2010; 15( 12): 684– 92. doi:10.1016/j.tplants.2010.09.008. [Google Scholar] [CrossRef]
19. Kumar BM , Bhavya G , De Britto S , Jogaiah S . Wild edible plants for food security, dietary diversity, and nutraceuticals: A global overview of emerging research. Front Sustain Food Syst. 2025; 9: 1686446. doi:10.3389/fsufs.2025.1686446. [Google Scholar] [CrossRef]
20. Chileh Chelh T , Rincon-Cervera MA , Gomez-Mercado F , Lopez-Ruiz R , Gallon-Bedoya M , Ezzaitouni M , et al. Wild Asparagus shoots constitute a healthy source of bioactive compounds. Molecules. 2023; 28( 15): 5786. doi:10.3390/molecules28155786. [Google Scholar] [CrossRef]
21. da Cunha-Chiamolera TPL , Chileh-Chelh T , Urrestarazu M , Ezzaitouni M , López-Ruiz R , Gallón-Bedoya M , et al. Crop productivity, phytochemicals, and bioactivities of wild and grown in controlled environment slender amaranth (Amaranthus viridis L.). Agronomy. 2024; 14( 9): 2038. doi:10.3390/agronomy14092038. [Google Scholar] [CrossRef]
22. Chileh-Chelh T , da Cunha-Chiamolera TPL , Urrestarazu M , Ezzaitouni M , López-Ruiz R , Nájera C , et al. London rocket (Sisymbrium irio L.) as healthy green: Bioactive compounds and bioactivity of plants grown in wild and controlled environments. Molecules. 2025; 30( 1): 31. doi:10.3390/molecules30010031. [Google Scholar] [CrossRef]
23. Cunha-Chiamolera TPL , Chileh-Chelh T , Ezzaitouni M , Guil-Guerrero JL , Urrestarazu M . Effect of electrical conductivity of nutrient solution and light spectra on the main phytochemical content of Sonchus tenerrimus L. under wild and controlled environments. Plants. 2025; 14( 17): 2811. doi:10.3390/plants14172811. [Google Scholar] [CrossRef]
24. Alrhmoun M , Sulaiman N , Pieroni A . Phylogenetic perspectives and ethnobotanical insights on wild edible plants of the Mediterranean, middle east, and north Africa. Foods. 2025; 14( 3): 465. doi:10.3390/foods14030465. [Google Scholar] [CrossRef]
25. Aryendu A , Dikshit D , Ramamurthy S . Cross-talk of signaling pathways and metabolic networks in plant stress resilience. J Plant Nutr. 2025: 1– 12. doi:10.1080/01904167.2025.2591261. [Google Scholar] [CrossRef]
26. Awoke A , Siyum Y , Gudesho G , Akmel F , Abate KS . Ethnobotanical study of wild and semi-wild edible plants in Yeki district, Sheka Zone, Southwest Ethiopia. J Ethnobiol Ethnomed. 2025; 21( 1): 54. doi:10.1186/s13002-025-00810-3. [Google Scholar] [CrossRef]
27. Lombardi T , Bedini S , Bertacchi A , Ventura I . Exploring the potential of Mediterranean edible halophytes as novel crops: Ecological and nutritional insights from Tuscany’s salt marshes. Agronomy. 2025; 15( 3): 634. doi:10.3390/agronomy15030634. [Google Scholar] [CrossRef]
28. Bernstein N . Plants and salt: Plant response and adaptations to salinity. In: Model ecosystems in extreme envi-ronments. Cambridge, MA, USA: Academic Press; 2019. p. 101– 12. doi:10.1016/B978-0-12-812742-1.00005-2. [Google Scholar] [CrossRef]
29. Kalaivani J , Jegadeeswari V , Vijayalatha KR , Arulmozhiyan R , Meena S , Selvarajan R , et al. Impact of salinity in tropical fruit crops–a review. Commun Soil Sci Plant Anal. 2024; 55( 21): 3286– 306. doi:10.1080/00103624.2024.2390478. [Google Scholar] [CrossRef]
30. Meng X , Zhou J , Sui N . Mechanisms of salt tolerance in halophytes: Current understanding and recent advances. Open Life Sci. 2018; 13( 1): 149– 54. doi:10.1515/biol-2018-0020. [Google Scholar] [CrossRef]
31. Hasanuzzaman M , Raihan MRH , Masud AAC , Rahman K , Nowroz F , Rahman M , et al. Regulation of reactive oxygen species and antioxidant defense in plants under salinity. Int J Mol Sci. 2021; 22( 17): 9326. doi:10.3390/ijms22179326. [Google Scholar] [CrossRef]
32. Ullah A , Tariq A , Sardans J , Graciano C , Zeng F , Noor J , et al. Calligonum mongolicum employs a variety of physiological and biochemical strategies to acclimatize to hyperarid saline deserts. Acta Physiol Plant. 2025; 47: 9. doi:10.1007/s11738-024-03757-2. [Google Scholar] [CrossRef]
33. Siebielec S , Siebielec G , Klimkowicz-Pawlas A , Gałązka A , Grządziel J , Stuczyński T . Impact of water stress on microbial community and activity in sandy and loamy soils. Agronomy. 2020; 10( 9): 1429. doi:10.3390/agronomy10091429. [Google Scholar] [CrossRef]
34. Yu M , Zhang J , Wei L , Wang G , Dong W , Liu X . Impact of soil textures on agricultural drought evolution and field capacity estimation in humid regions. J Hydrol. 2023; 626: 130257. doi:10.1016/j.jhydrol.2023.130257. [Google Scholar] [CrossRef]
35. Nour MM , Aljabi HR , AL-Huqail AA , Horneburg B , Mohammed AE , Alotaibi MO . Drought responses and adaptation in plants differing in life-form. Front Ecol Evol. 2024; 12: 1452427. doi:10.3389/fevo.2024.1452427. [Google Scholar] [CrossRef]
36. Guadarrama-Escobar LM , Hunt J , Gurung A , Zarco-Tejada PJ , Shabala S , Camino C , et al. Back to the future for drought tolerance. New Phytol. 2024; 242( 2): 372– 83. doi:10.1111/nph.19619. [Google Scholar] [CrossRef]
37. Abo Amin S , Hayat F , Alyafei M . Drought tolerance strategies of some native plants in the UAE desert: Growth, biochemical, and antioxidant insights. BMC Plant Biol. 2025; 25: 1374. doi:10.1186/s12870-025-07305-z. [Google Scholar] [CrossRef]
38. Kumar V , Srivastava AK , Sharma D , Pandey SP , Pandey M , Dudwadkar A , et al. Antioxidant defense and ionic homeostasis govern stage-specific response of salinity stress in contrasting rice varieties. Plants. 2024; 13( 6): 778. doi:10.3390/plants13060778. [Google Scholar] [CrossRef]
39. Mahmood T , Khalid S , Abdullah M , Ahmed Z , Shah MKN , Ghafoor A , et al. Insights into drought stress signaling in plants and the molecular genetic basis of cotton drought tolerance. Cells. 2020; 9( 1): 105. doi:10.3390/cells9010105. [Google Scholar] [CrossRef]
40. Jain D , Phurailatpam L , Mishra S . Microbes-mediated mitigation of drought stress in plants: Recent trends and future challenges. In: Advances in plant microbiome and sustainable agriculture. Singapore: Springer; 2020. p. 199– 218. doi:10.1007/978-981-15-3204-7_9. [Google Scholar] [CrossRef]
41. Pérez-Romero JA , Barcia-Piedras JM , Redondo-Gómez S , Mateos-Naranjo E . Impact of short-term extreme temperature events on physiological performance of Salicornia ramosissima J. Woods under optimal and sub-optimal saline conditions. Sci Rep. 2019; 9: 659. doi:10.1038/s41598-018-37346-4. [Google Scholar] [CrossRef]
42. Zhu Y , Cheng Z , Feng K , Chen Z , Cao C , Huang J , et al. Influencing factors for transpiration rate: A numerical simulation of an individual leaf system. Therm Sci Eng Prog. 2022; 27: 101110. doi:10.1016/j.tsep.2021.101110. [Google Scholar] [CrossRef]
43. Zahra N , Hafeez MB , Ghaffar A , Kausar A , Al Zeidi M , Siddique KHM , et al. Plant photosynthesis under heat stress: Effects and management. Environ Exp Bot. 2023; 206: 105178. doi:10.1016/j.envexpbot.2022.105178. [Google Scholar] [CrossRef]
44. Kumari A , Lakshmi GA , Krishna GK , Patni B , Prakash S , Bhattacharyya M , et al. Climate change and its impact on crops: A comprehensive investigation for sustainable agriculture. Agronomy. 2022; 12( 12): 3008. doi:10.3390/agronomy12123008. [Google Scholar] [CrossRef]
45. Šola I , Poljuha D , Pavičić I , Jurinjak Tušek A , Šamec D . Climate change and plant foods: The influence of environmental stressors on plant metabolites and future food sources. Foods. 2025; 14( 3): 416. doi:10.3390/foods14030416. [Google Scholar] [CrossRef]
46. Mishra N , Jiang C , Chen L , Paul A , Chatterjee A , Shen G . Achieving abiotic stress tolerance in plants through antioxidative defense mechanisms. Front Plant Sci. 2023; 14: 1110622. doi:10.3389/fpls.2023.1110622. [Google Scholar] [CrossRef]
47. Jiang Z , van Zanten M , Sasidharan R . Mechanisms of plant acclimation to multiple abiotic stresses. Commun Biol. 2025; 8( 1): 655. doi:10.1038/s42003-025-08077-w. [Google Scholar] [CrossRef]
48. Blum A . Osmotic adjustment is a prime drought stress adaptive engine in support of plant production. Plant Cell Environ. 2017; 40( 1): 4– 10. doi:10.1111/pce.12800. [Google Scholar] [CrossRef]
49. Kuromori T , Fujita M , Takahashi F , Yamaguchi-Shinozaki K , Shinozaki K . Inter-tissue and inter-organ signaling in drought stress response and phenotyping of drought tolerance. Plant J. 2022; 109( 2): 342– 58. doi:10.1111/tpj.15619. [Google Scholar] [CrossRef]
50. Pamungkas SST , Farid N . Drought stress: Responses and mechanism in plants. Rev Agric Sci. 2022; 10: 168– 85. doi:10.7831/ras.10.0_168. [Google Scholar] [CrossRef]
51. Martins TS , Da-Silva CJ , Shabala S , Striker GG , Carvalho IR , de Oliveira ACB , et al. Understanding plant responses to saline waterlogging: Insights from halophytes and implications for crop tolerance. Planta. 2024; 259: 24. doi:10.1007/s00425-023-04275-0. [Google Scholar] [CrossRef]
52. Waheed A , Zhuo L , Wang M , Xu H , Tong Z , Wang C , et al. Integrative mechanisms of plant salt tolerance: Biological pathways, phytohormonal regulation, and technological innovations. Plant Stress. 2024; 14: 100652. doi:10.1016/j.stress.2024.100652. [Google Scholar] [CrossRef]
53. Tripathi JM , Khan BR , Gaur R , Yadav D , Verma KK , Gupta R . Gibberellic acid improves photosynthetic electron transport and stomatal function in crops that are adversely affected by salinity exposure. Plants. 2025; 14( 21): 3388. doi:10.3390/plants14213388. [Google Scholar] [CrossRef]
54. Alghamdi SA . Drought and salinity effects on plant growth: A comprehensive review. SABRAO J Breed Genet. 2024; 56( 6): 2231– 340. doi:10.54910/sabrao2024.56.6.14. [Google Scholar] [CrossRef]
55. Didaran F , Kordrostami M , Ghasemi-Soloklui AA , Pashkovskiy P , Kreslavski V , Kuznetsov V , et al. The mechanisms of photoinhibition and repair in plants under high light conditions and interplay with abiotic stressors. J Photochem Photobiol B. 2024; 259: 113004. doi:10.1016/j.jphotobiol.2024.113004. [Google Scholar] [CrossRef]
56. Hernández-González O , Romo-Campos RL , Muñiz-Castro MÁ , Flores J . Light and irrigation effects on chlorophyll fluorescence depend on seedling provenance in Magnolia pugana endemic of Mexico. Flora. 2020; 271: 151684. doi:10.1016/j.flora.2020.151684. [Google Scholar] [CrossRef]
57. Yasin S , Zavala-García F , Niño-Medina G , Rodríguez-Salinas PA , Gutiérrez-Diez A , Sinagawa-García SR , et al. Morphological and physiological response of maize (Zea mays L.) to drought stress during reproductive stage. Agronomy. 2024; 14( 8): 1718. doi:10.3390/agronomy14081718. [Google Scholar] [CrossRef]
58. Rao MJ , Duan M , Zhou C , Jiao J , Cheng P , Yang L , et al. Antioxidant defense system in plants: Reactive oxygen species production, signaling, and scavenging during abiotic stress-induced oxidative damage. Horticulturae. 2025; 11( 5): 477. doi:10.3390/horticulturae11050477. [Google Scholar] [CrossRef]
59. Chico-Peralta A , Villamiel M , Angulo-Bejarano PI , Ramírez-Jiménez AK . Abiotic stress alters the nutritional, metabolomic, and glycomic profiles of Piper auritum kunth. Foods. 2025; 14( 20): 3543. doi:10.3390/foods14203543. [Google Scholar] [CrossRef]
60. Hernández JA , Diaz-Vivancos P , Acosta-Motos JR , Barba-Espín G . Where biotic and abiotic stress responses converge: Common patterns in response to salinity and Plumpox virus infection in pea and peach plants. Ann Appl Biol. 2021; 178( 2): 281– 92. doi:10.1111/aab.12667. [Google Scholar] [CrossRef]
61. Gebre A , Gitima G , Berhanu Y . Ethnobotanical study of wild edible plants in Goba District Southwest Ethiopia. Sci Rep. 2025; 15( 1): 27689. doi:10.1038/s41598-025-11862-6. [Google Scholar] [CrossRef]
62. Witzel K , Acosta Motos JR , Atay E , Çiçek N , Mistríková V , Oney-Birol S , et al. Leveraging microorganisms and biostimulants: Mitigating salinity stress in crops with agricultural biologicals. Plant Soil. 2025: 1– 23. doi:10.1007/s11104-025-07578-1. [Google Scholar] [CrossRef]
63. Õunapuu-Pikas E , Venisse JS , Hõrak H , Tamm I , Label P , Sellin A . Elevated environmental humidity modulates aquaporin expression and impacts leaf hydraulic efficiency in silver birch (Betula pendula). New Phytol. 2026; 249( 2): 860– 77. doi:10.1111/nph.70702. [Google Scholar] [CrossRef]
64. Liu C , Jiang X , Yuan Z . Plant responses and adaptations to salt stress: A review. Horticulturae. 2024; 10( 11): 1221. doi:10.3390/horticulturae10111221. [Google Scholar] [CrossRef]
65. Ren Y , Yan H , Ma A . Plant genomic and microbial interplay in the rhizosphere under salt stress: A review. Front Plant Sci. 2026; 16: 1667328. doi:10.3389/fpls.2025.1667328. [Google Scholar] [CrossRef]
66. Acharya BR , Gill SP , Kaundal A , Sandhu D . Strategies for combating plant salinity stress: The potential of plant growth-promoting microorganisms. Front Plant Sci. 2024; 15: 1406913. doi:10.3389/fpls.2024.1406913. [Google Scholar] [CrossRef]
67. Silva ALJ , de Farias OR , Corrêa ÉB , de Lacerda CF , de Melo AS , de Mello Oliveira MD . Biostimulant modulate the physiological and biochemical activities, improving agronomic characteristics of bell pepper plants under salt stress. Sci Rep. 2025; 15( 1): 14969. doi:10.1038/s41598-025-99414-w. [Google Scholar] [CrossRef]
68. Prinsloo G , Nogemane N . The effects of season and water availability on chemical composition, secondary metabolites and biological activity in plants. Phytochem Rev. 2018; 17( 4): 889– 902. doi:10.1007/s11101-018-9567-z. [Google Scholar] [CrossRef]
69. Yang J , Li Y , He Y , He H , Chen X , Liu T , et al. Wild vs. cultivated Zingiber striolatum Diels: Nutritional and biological activity differences. Plants. 2023; 12( 11): 2180. doi:10.3390/plants12112180. [Google Scholar] [CrossRef]
70. Patil D , Patil A . Impact of abiotic stress signals on secondary metabolites. In: Plant secondary metabolites and abiotic stress. Hoboken, NJ, USA: Wiley; 2024. p. 173– 94. doi:10.1002/9781394186457.ch7. [Google Scholar] [CrossRef]
71. Pérez-Sánchez LE , Ayala-Guerrero LM , Mendieta-Moctezuma A , Villalobos-López MA , Ríos-Meléndez S . Stress-induced plant specialized metabolism: Signaling, multi-omics integration, and plant-derived antimicrobial metabolites to combat antimicrobial resistance. Plants. 2026; 15( 2): 193. doi:10.3390/plants15020193. [Google Scholar] [CrossRef]
72. Gaude AA , Jalmi SK . Environmental stress induced biosynthesis of plant secondary metabolites-transcriptional regulation as a key. Crop Des. 2025; 4( 2): 100100. doi:10.1016/j.cropd.2025.100100. [Google Scholar] [CrossRef]
73. Khatoon S , Maheshwari C , Kumari S , Mahajan M , Kaur H , Ahmad Chaudhary A , et al. Phytomelatonin and secondary metabolites: Operative strategies to induce plant abiotic stress tolerance. Plant Stress. 2026; 19: 101178. doi:10.1016/j.stress.2025.101178. [Google Scholar] [CrossRef]
74. Nicolas-Espinosa J , Garcia-Ibañez P , Lopez-Zaplana A , Yepes-Molina L , Albaladejo-Marico L , Carvajal M . Confronting secondary metabolites with water uptake and transport in plants under abiotic stress. Int J Mol Sci. 2023; 24( 3): 2826. doi:10.3390/ijms24032826. [Google Scholar] [CrossRef]
75. Bautista I , Boscaiu M , Lidón A , Llinares JV , Lull C , Donat MP , et al. Environmentally induced changes in antioxidant phenolic compounds levels in wild plants. Acta Physiol Plant. 2016; 38: 9. doi:10.1007/s11738-015-2025-2. [Google Scholar] [CrossRef]
76. Ray A , Kundu S , Mohapatra SS , Sinha S , Khoshru B , Keswani C , et al. An insight into the role of phenolics in abiotic stress tolerance in plants: Current perspective for sustainable environment. J Pure Appl Microbiol. 2024; 18( 1): 64– 79. doi:10.22207/jpam.18.1.09. [Google Scholar] [CrossRef]
77. Araque Gelves DI , Andreoli de Souza GC , Salvador MJ , Díaz-Rodríguez AM , Campos-Avelar I , de los Santos-Villalobos S . Impact of plant–microorganism interaction: A key driver for the production of bioactive metabolites of interest in the pharmaceutical, agricultural, cosmetic, and food industries. J Plant Growth Regul. 2026; 45: 833– 60. doi:10.1007/s00344-025-11921-2. [Google Scholar] [CrossRef]
78. Ngxabi S , Jimoh MO , Sogoni A , Laubscher CP , Rautenbach F , Kambizi L . Salinity influenced proximate, minerals, anti-nutrients and phytochemical composition of Trachyandra ciliata kunth (wild cabbage): A promising edible halophyte. Food Sci Nutr. 2025; 13: e4755. doi:10.1002/fsn3.4755. [Google Scholar] [CrossRef]
79. Agudelo A , Carvajal M , Martinez-Ballesta MDC . Halophytes of the Mediterranean Basin as nutritious crops. Foods. 2021; 10( 1): 119. doi:10.3390/foods10010119. [Google Scholar] [CrossRef]
80. Schoutens K , Pelckmans I , Gebruers S , Schoelynck J , Temmerman S . Plant trait trade-offs regulate tidal marsh resistance to combined hydrodynamic and erosion stress. J Appl Ecol. 2026; 63( 1): e70281. doi:10.1111/1365-2664.70281. [Google Scholar] [CrossRef]
81. Kapazoglou A , Gerakari M , Lazaridi E , Kleftogianni K , Sarri E , Tani E , et al. Crop wild relatives: A valuable source of tolerance to various abiotic stresses. Plants. 2023; 12( 2): 328. doi:10.3390/plants12020328. [Google Scholar] [CrossRef]
82. Rosales-Nieblas AC , Yamada M , Murillo-Amador B , Yamada S . Salinity tolerance in halophytes in aquaponics. Horticulturae. 2025; 11( 6): 623. doi:10.3390/horticulturae11060623. [Google Scholar] [CrossRef]
83. Mohanta TK , Mohanta YK , Kaushik P , Kumar J . Physiology, genomics, and evolutionary aspects of desert plants. J Adv Res. 2024; 58: 63– 78. doi:10.1016/j.jare.2023.04.019. [Google Scholar] [CrossRef]
84. Kaur A , Sangha MK , Devi V , Pathak M , Singla D . Nutritional, antinutritional and antioxidant assessment of immature Abelmoschus pods: Implications for crop improvement from cultivated and wild varieties. S Afr N J Bot. 2024; 174: 485– 94. doi:10.1016/j.sajb.2024.09.022. [Google Scholar] [CrossRef]
85. Carrascosa A , Pascual JA , Ros M , Petropoulos SA , del Mar Alguacil M . Agronomical practices and management for commercial cultivation of Portulaca oleracea as a crop: A review. Plants. 2023; 12( 6): 1246. doi:10.3390/plants12061246. [Google Scholar] [CrossRef]
86. Yildirim E , Ekinci M , Turan M , Maskan E , Ibrahim Binici H . Comparative analysis of antioxidant activities and phenolic contents in selected wild edible plants. Ital J Food Sci. 2025; 37( 4): 209– 18. doi:10.15586/ijfs.v37i4.3149. [Google Scholar] [CrossRef]
87. Mpolokeng TG , Mokgau K , Nko KI , Asong JA , Struwig M , Aremu AO . Ethnobotanical uses of traditional African vegetables among three selected villages of Madibeng Local Municipality, South Africa. Genet Resour Crop Evol. 2025; 72: 6337– 47. doi:10.1007/s10722-025-02331-0. [Google Scholar] [CrossRef]
88. Bae N , Shim SH , Alavilli H , Do H , Park M , Lee DW , et al. Enhanced salt stress tolerance in plants without growth penalty through increased photosynthesis activity by plastocyanin from Antarctic moss. Plant J. 2025; 121: e17168. doi:10.1111/tpj.17168. [Google Scholar] [CrossRef]
89. Mino M , Tezuka T . Overcoming reproductive barriers: Genetic and molecular insights for enhanced crop breeding. Euphytica. 2025; 221( 7): 110. doi:10.1007/s10681-025-03564-x. [Google Scholar] [CrossRef]
90. Dujmović M , Opačić N , Radman S , Fabek Uher S , Voća S , Šic Žlabur J . Accumulation of stinging nettle bioactive compounds as a response to controlled drought stress. Agriculture. 2023; 13( 7): 1358. doi:10.3390/agriculture13071358. [Google Scholar] [CrossRef]
91. Mustafa A , Mehmood A , Ahmad KS , Hussain S , Khan MF , Ahmad M . Exploring the diversity and ethnomedicinal uses of wild edible plants in District Poonch, Azad Jammu and Kashmir. Genet Resour Crop Evol. 2025; 72( 7): 9043– 67. doi:10.1007/s10722-025-02499-5. [Google Scholar] [CrossRef]
92. Cordero S , Gálvez F , Carvallo GO . Biodiversity-productivity relationship in urban vascular flora: A comparison between wild edible and non-edible plants. Bot Sci. 2022; 100( 1): 107– 19. doi:10.17129/botsci.2892. [Google Scholar] [CrossRef]
93. Biri S , Ayenew B , Dida G , Sebsibe A , Gurmessa F , Woldeab B , et al. Ethnobotanical study of wild edible plants in Arsi Robe district of East Arsi Zone, Ethiopia. J Ethnobiol Ethnomed. 2024; 20: 70. doi:10.1186/s13002-024-00703-x. [Google Scholar] [CrossRef]
94. Sarfraz Z , Zarlashat Y , Ambreen A , Mujahid M , Iqbal MS , Fatima SA , et al. Plant biochemistry in the era of omics: Integrated omics approaches to unravel the genetic basis of plant stress tolerance. Plant Breed. 2025: 1– 23. doi:10.1111/pbr.13277. [Google Scholar] [CrossRef]
95. Matesanz S , Milla R . Differential plasticity to water and nutrients between crops and their wild progenitors. Environ Exp Bot. 2018; 145: 54– 63. doi:10.1016/j.envexpbot.2017.10.014. [Google Scholar] [CrossRef]
96. Hassan SH , Simiele M , Scippa GS , Morabito D , Trupiano D . Omics advancements towards exploring arsenic toxicity and tolerance in plants: A review. Planta. 2025; 261( 4): 79. doi:10.1007/s00425-025-04646-9. [Google Scholar] [CrossRef]
97. Cheng L , Mo C , Ma P , Li Y , Xiong Y . Comparative metabolomics profiling reveals the aroma and nutritional diversity of eight wild edible plants. Food Chem. 2025; 491: 145202. doi:10.1016/j.foodchem.2025.145202. [Google Scholar] [CrossRef]
98. Oluoch WA , Borgemeister C , de Deus Vidal J Jr , Fremout T , Gaisberger H , Whitney C , et al. Predicted changes in distribution and richness of wild edible plants under climate change scenarios in northwestern Kenya. Reg Environ Change. 2024; 24: 11. doi:10.1007/s10113-023-02175-3. [Google Scholar] [CrossRef]
99. Tebkew M , Gebremariam Y , Mucheye T , Alemu A , Abich A , Fikir D . Uses of wild edible plants in Quara district, northwest Ethiopia: Implication for forest management. Agric Food Secur. 2018; 7: 12. doi:10.1186/s40066-018-0163-7. [Google Scholar] [CrossRef]
100. Zhang S , He C , Su L , Wang H , Lin J , Li Y . An ethnobotanical study on wild edible plants in Taishan County, Guangdong, China. Front Sustain Food Syst. 2025; 9: 1606420. doi:10.3389/fsufs.2025.1606420. [Google Scholar] [CrossRef]
101. Alseekh S , Klemmer A , Yan J , Guo T , Fernie AR . Embracing plant plasticity or robustness as a means of ensuring food security. Nat Commun. 2025; 16( 1): 461. doi:10.1038/s41467-025-55872-4. [Google Scholar] [CrossRef]
102. Yolcu S , Alavilli H , Lee BH . Natural genetic resources from diverse plants to improve abiotic stress tolerance in plants. Int J Mol Sci. 2020; 21( 22): 8567. doi:10.3390/ijms21228567. [Google Scholar] [CrossRef]
103. Petereit J , Bayer PE , Thomas WJW , Tay Fernandez CG , Amas J , Zhang Y , et al. Pangenomics and crop genome adaptation in a changing climate. Plants. 2022; 11( 15): 1949. doi:10.3390/plants11151949. [Google Scholar] [CrossRef]
104. Choudhary R , Ahmad F , Kaya C , Upadhyay SK , Muneer S , Kumar V , et al. Decrypting proteomics, transcriptomics, genomics, and integrated omics for augmenting the abiotic, biotic, and climate change stress resilience in plants. J Plant Physiol. 2025; 305: 154430. doi:10.1016/j.jplph.2025.154430. [Google Scholar] [CrossRef]
105. Ali Khan N , Owens L , Nuñez MA , Khan AL . Complexity of combined abiotic stresses to crop plants. Plant Stress. 2025; 17: 100926. doi:10.1016/j.stress.2025.100926. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools