 Open Access
Open Access
REVIEW
Recent Advances in Natural Polysaccharide-Based Materials for Wound Healing: Chitosan, Konjac Glucomannan and Cellulose
1 School of Public Health, Shaanxi University of Chinese Medicine, Xianyang, China
2 Administration for Drug and Instrument Supervision and Inspection of PLAJLSF, Beijing, China
3 College of Pharmacy, Shaanxi University of Chinese Medicine, Xianyang, China
* Corresponding Author: Liangliang Duan. Email:
# The authors contributed equally to this work and should be considered as co-first authors
Journal of Renewable Materials 2026, 14(4), 3 https://doi.org/10.32604/jrm.2026.02025-0124
Received 27 June 2025; Accepted 26 December 2025; Issue published 24 April 2026
Abstract
Wound healing is a complex and dynamic process essential for restoring the integrity of damaged skin. It requires wound dressings that actively regulate the wound microenvironment by preventing infection, maintaining moisture balance, allowing gas exchange, and managing exudate. Natural polysaccharides, such as konjac glucomannan (KGM), chitosan, and cellulose, are well suited to this role because of their biocompatibility, biodegradability, and intrinsic bioactivity. Extensive research has focused on developing polysaccharide-based wound dressings with enhanced functionality to promote healing. This review examines recent scientific research published mostly in the past five years on the development and application of chitosan, KGM and cellulose-based macromolecules for wound dressing fabrication, including hydrogels, sponges, fibers, and other forms. It explores how their structure-property-function relationships connect primary composition and inter-component interactions (e.g., hydrogen bonding, ionic complexation, covalent crosslinking) to key performance metrics (antibacterial efficacy, hemostatic activity, moisture management such as swelling and water vapor transmission rate, and mechanical robustness). The advantages and disadvantages of various methods for preparing materials of the same type using the same polysaccharide are also discussed with quantitative comparisons across studies to provide clear insights into the healing effects of different wound dressings. For relatively mature cellulose-based wound dressings, we integrate their therapeutic efficacy, functional mechanisms, clinical evidence and marketed products with typical indications. Additionally, we summarize the advantages and limitations of these polysaccharides in wound healing, while identifying future trends and challenges that should guide the rational design of polysaccharide-based wound dressings.Graphic Abstract
Keywords
Wound healing is a complex and dynamic biological process essential for restoring the integrity and function of damaged skin tissue. This intricate cascade progresses through four overlapping yet distinct phases: hemostasis (immediate blood clotting), inflammation (infection control and tissue debridement), proliferation (tissue regeneration, angiogenesis, and extracellular matrix deposition), and remodeling (granulation tissue maturation and scar formation) [1]. An optimal healing environment requires protection from infection, maintenance of proper moisture balance, facilitation of gas exchange, and removal of excess exudate [2]. However, inadequate management of these factors can result in delayed healing, chronic wounds, or excessive scarring.
Wound dressings are a crucial first-line intervention, serving as a mechanical barrier against pathogens and physical trauma, while also acting as an active platform to promote healing. Conventional dressings, such as gauzes, cotton pad and bandages, provide passive coverage and basic exudate management, proving effective for clean and dry wounds [3]. However, their efficacy is often limited by several drawbacks, such as minimal bioactivity, the potential for adhesion to the wound bed—which may cause trauma upon removal—and suboptimal control of the wound microenvironment.
Consequently, numerous studies have focused on the development of advanced dressings derived from natural polymers, which offer several inherent advantages. These include excellent biocompatibility, biodegradability, effective moisture retention, sufficient mechanical strength, an appropriate microstructure, and intrinsic bioactive properties that promote wound healing [4]. Various functional natural polymers, including polysaccharides and proteins, have been extensively explored for their potential in wound healing. Notable examples include chitosan, known for its antibacterial and hemostatic properties [5]; alginate, which excels in exudate absorption, haemostatic activity, and angiogenic properties [6]; hyaluronic acid, which regulates inflammation and promotes cell migration [7]; cellulose and its derivatives, recognized for their excellent absorbency and hemocompatibility [8]; collagen, which supports cell adhesion and proliferation [9]; and silk fibroin, renowned for its biocompatibility and adjustability [3].
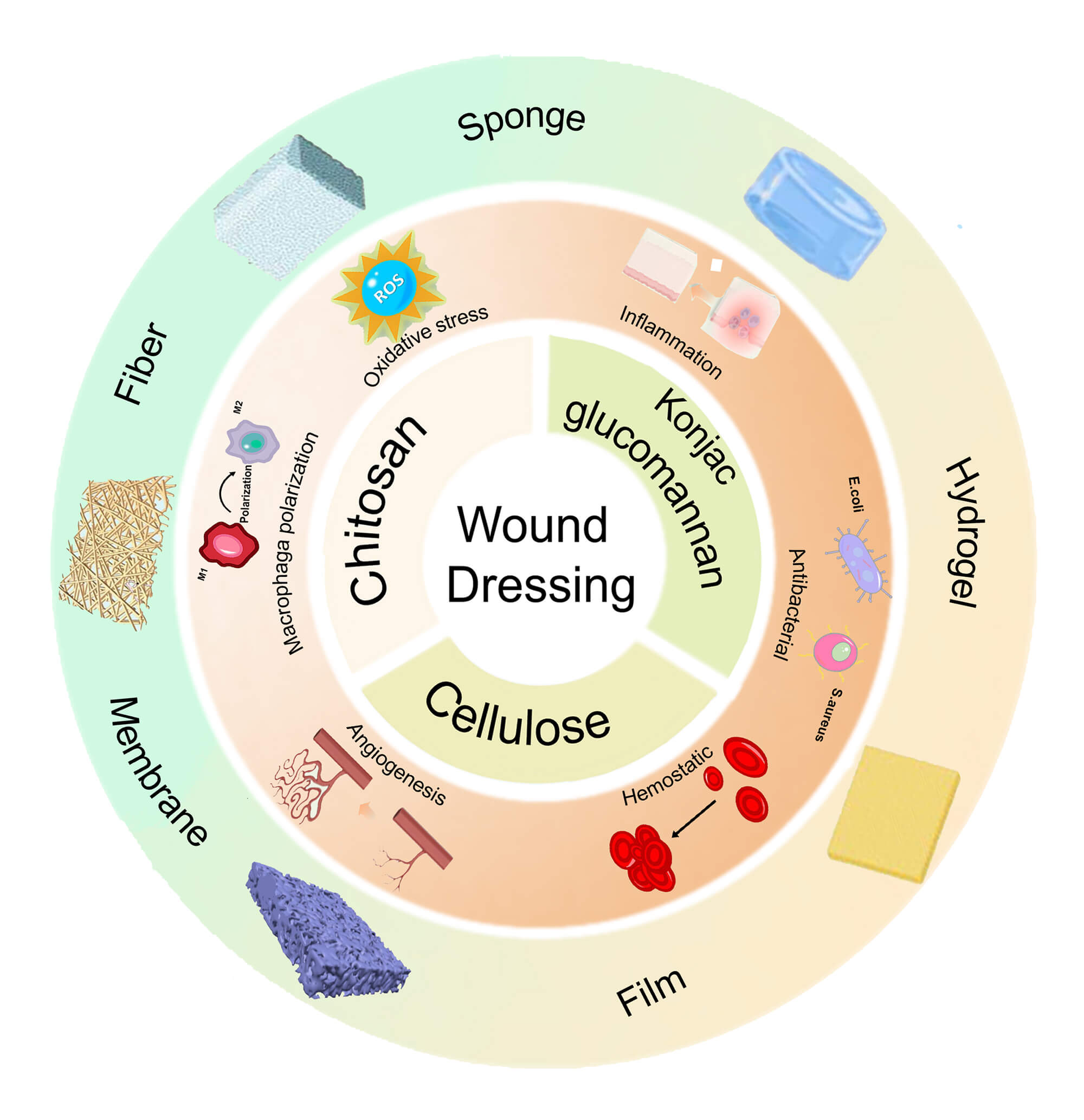
These natural polysaccharides, as biological macromolecules composed of various monosaccharides linked by different glycosidic bonds, are widely utilized in wound dressings due to their tunable physicochemical properties, abundance, and inherent physiological activities that promote wound-healing (e.g., antimicrobial activities [10], immune modulation [11], granulation tissue formation [12], and blood coagulation [13]). In recent years, the use of these polysaccharides in wound dressings has also garnered increasing attention. Within this broad class, we focus on cellulose, chitosan, and konjac glucomannan (KGM) due to their abundance, renewability, and ideal physicochemical properties facilitate wound dressing design. Cellulose, the most abundant biopolymer, offers excellent biocompatibility, chemical modifiability, mechanical robustness, and low-cost scalability, underpinning its use across food, biomedical, environmental, and electronic applications [14]. Chitosan, derived from the second most abundant biopolymer chitin, is the only naturally occurring cationic polysaccharide; its protonated amines confer intrinsic antimicrobial and hemostatic activities, together with biodegradability, making it well suited for wound dressing fabrication [15]. KGM is a highly water-soluble polysaccharide with exceptional hydrophilicity and strong swelling, thickening, and emulsifying capabilities. It has been reported to exhibit antioxidant and immunomodulatory activities, and it readily forms water-rich hydrogels, porous membranes, and films. In aqueous solution, KGM is nonionic, which minimizes electrostatic interactions with charged bioactive components and helps preserve their intrinsic properties. Owing to these attributes, KGM is widely used in the food, pharmaceutical, and cosmetic industries [16]. Collectively, these polymers can be processed into various advanced materials—including hydrogels, films, sponges, and fibers—specially designed to address the challenges associated with wound healing. Table 1 summarizes key properties of KGM, chitosan, and cellulose, as well as main advantages, and limitations for wound healing.
Despite multiple reviews on chitosan-, KGM-, and cellulose-based wound dressings, a comprehensive review that integrates component-interaction-property-function relationships for each type of wound dressing on these polysaccharides remains lacking. This review addresses that gap by applying a unified analytical framework to summarize primary constituents and their interactions (e.g., hydrogen bonding, ionic complexation, covalent crosslinking) in relation to key performance metrics and clinically relevant functions, including mechanical compliance, hemostasis, antibacterial activity, moisture management, and overall wound-healing efficacy. We particularly focus on how the selection of components and interaction mechanisms influence physical properties, physiological functions, and therapeutic effectiveness, as well as the advantages and disadvantages of various methods for preparing materials of the same type using the same polysaccharide. Cellulose-based dressings, which currently have the broadest clinical applications, integrate hemostatic mechanisms, supported by both experimental and clinical evidence, as well as marketed products, including clinical trials and their outcomes. We also compare advantages and limitations of these polysaccharides and provide quantitative cross-study data (e.g., swelling ratio, water vapor transmission rate, blood loss, healing rate, etc.) to clearly display their performance in representative wound dressings. Finally, we outline future research directions and translational challenges to advance these materials towards commercial production and broader clinical application.
Chitosan, a polysaccharide derived from crustaceans or fungi, is primarily obtained through alkaline deacetylation of chitin. It possesses amino and hydroxyl groups that enable it to undergo chemical and physical reactions with other substances. Due to its amino group, chitosan becomes protonated at low pH levels, resulting in positively charged chitosan molecules that readily interact with anionic substances, facilitating chemical modification. The positively charged amines can bind to negatively charged molecules on the surface of microbial structures, thereby inhibiting their growth. Additionally, chitosan promotes the aggregation of platelets and blood cells, effectively aiding in hemostasis, and better regulating inflammation [29]. It is biodegradable and yields non-toxic residues, such as glucosamine and N-acetylglucosamine, which can participate in metabolic processes and are desirable for biomedical applications. Its structure, which is similar to glycosaminoglycans found in the extracellular matrix, makes chitosan a desirable material for use in tissue engineering to promote fibroblast proliferation and accelerate tissue regeneration [30]. Therefore, chitosan is commonly used in wound healing, typically in the form of hydrogels, sponges, films, and fibers.
Because of the good water absorption, water retention, biocompatibility, three-dimensional porous structure, and adhesion property, hydrogel has garnered attention as an ideal dressing. However, the application of pure cationic chitosan-based hydrogels is limited due to their low mechanical strength and high water sensitivity. Additionally, they exhibit ineffective wound repair capabilities, primarily because of weak charge interactions with tissues and inadequate promotion of blood clotting [31]. Significant efforts are currently dedicated to improving the mechanical and physiological properties of chitosan-based hydrogels. A commonly employed approach involves combining chitosan with functional components, such as growth factors, bioactive compounds derived from medicinal plants, or macromolecules that readily form hydrogels. This strategy integrates the unique properties of each ingredient, thereby enhancing the overall performance of the hydrogel. Physical binding, primarily through hydrogen bonds, ionic bonds, and metal coordination, significantly contributes to the blend hydrogel’s mechanical strength, ensuring it maintains its hydrogel state during use. Several chitosan derivatives have been developed to improve solubility and physiological activities, including carboxymethyl, glycol, thiolated, and trimethyl chitosans [32]. The incorporation of quaternary amino groups into chitosan increases its positive charge density, thus broadening its potential for wound healing applications. Carboxymethyl chitosan (CMCS), known for its good biocompatibility, also supports fibroblast growth. A self-healing hydrogel was developed by combining quaternized chitosan and CMCS (Fig. 1a). The hydrogel formation was driven by electrostatic interactions between the negatively charged –COO− groups of CMCS and the positively charged –NH3+ groups of quaternized chitosan, as well as hydrogen bonds between the two polymers. The resulting hydrogel demonstrated an adhesion strength of 6.15 ± 0.14 kPa, along with exceptional self-healing (incision traces markedly diminished within 30 min), good injectability, effective hemostasis (bleeding time decreased to 53.3 ± 11.2 s), and strong antibacterial properties (>80% against S. aureus and E. coli), thereby making it highly suitable for the treatment of infected wounds [21] (Fig. 1b). Once adhered to the wound, it acts as a physical barrier and promotes hemostasis: amino groups are protonated to –NH3+ under physiological conditions, enabling electrostatic interactions that enhance platelet adhesion and accelerate erythrocyte aggregation at the injury site, thereby facilitating thrombus formation. The incorporation of quaternary ammonium groups further increases blood cell adhesion. In addition, the reduced presence of senescent cells and the formation of denser, thicker, and more uniform collagen fibers improve tissue regeneration and accelerate healing in infected wounds.


Figure 1: (a) Preparation mechanism and (b) wound healing efficacy of quaternized chitosan/CMCS hydrogel [21]. (c) Schematic representation of the network structure in CMCS-PAM hydrogel [33] [Adapted with permission from Refs. [21,33]. Copyright © 2025, Elsevier and 2024, Elsevier].
In hydrogel systems with complex functions, multiple crosslinking networks often coexist, integrating two or more networks with distinct physical properties. This approach effectively overcomes the limitations of single-network hydrogels, such as mechanical instability [34]. Self-healing multi-network hydrogels with high strength have garnered significant attention, primarily due to the reversible dissociation and reorganization of their crosslinks. This process necessitates the presence of groups that enable strong and reversible crosslinking [5]. Noncovalent interactions have been widely employed to create chitosan-based self-healing hydrogels. A double-network self-healing hydrogel was synthesized from CMCS, acrylamide, ammonium persulfate, and N,N′-methylenebis (acrylamide). Intermolecular hydrogen bonding between polyacrylamide (PAM) and CMCS formed reversible, dynamic physical crosslinks that enabled self-healing (Fig. 1c). A single CMCS molecule can bind up to three PAM molecules via seven hydrogen bonds, thereby facilitating network rearrangement and resulting in superior adhesion strength (7.52 ± 0.05 kPa), along with exceptional self-healing efficiency (>50%), injectability, and water retention capacity. Furthermore, the hydrogel effectively promoted L929 cells migration and proliferation, achieving a relative wound closure of 59.43 ± 0.36%, which we attribute to CMCS’s ability to promote fibroblast aggregation, underscoring its strong potential as a wound dressing [33].
While physical crosslinking is often preferred for chitosan hydrogels, chemical crosslinking is also widely employed to establish stronger network structures and facilitate the incorporation of active ingredients, thereby significantly enhancing the hydrogels’ physiological properties [35]. Ganoderma lucidum polysaccharide (GLP), known for its anti-inflammatory and antioxidant properties, was oxidized with NaIO4 to produce OGLP, whose aldehyde groups can crosslink with the amino groups of CMCS through a Schiff base reaction. The resulting hydrogel, OGLP/CMCS, exhibited favorable physicochemical properties and enhanced biological activities, making it suitable for promoting fibroblast growth (cell viability 187 ± 11.42%) and migration (healing rate 30.90 ± 2.22%). Additionally, it demonstrated bacteriostatic capacity (98.17 ±1.04% against E. coli and 95.84 ± 0.76% against S. aureus), and antioxidant capacity [36]. Although chemical crosslinking reduced the ROS-scavenging capacity relative to free OGLP, the hydrogel still integrated the physiological functions of CMCS and OGLP, effectively lowering cellular ROS, promoting polarization toward the M2 macrophage phenotype while suppressing M1, accelerating collagen deposition, and promoting wound healing. Li et al. [37] prepared a double-network hydrogel for chronic diabetic wound repair using OGLP, sodium alginate, and CMCS, with genipin as the crosslinking agent. This hydrogel exhibited physicochemical properties comparable to those reported for the OGLP/CMCS hydrogel, owing to the incorporation of OGLP and CMCS. Beyond the Schiff base reaction between OGLP and CMCS, intermolecular hydrogen bonding further reinforced the network, yielding a high swelling ratio—indicative of strong exudate uptake—and an enhanced structural stability. The hydrogel showed 46.25 ± 3.25% mass loss after 21 days immersion in PBS, whereas the OGLP/CMCS fully degraded within 13 days. A multi-chemically crosslinked network hydrogel with strong adhesive and self-healing properties was synthesized by combining polyvinyl alcohol (PVA), chitosan, and gelatin, and incorporating phenylboronic acid (3-CPBA) and tannic acid (TA) as modifying agent. Within the hydrogel, boron ester bonds (B–O bonds) and amide bonds were formed between 3-CPBA and chitosan, boron ester bonds also formed between 3-CPBA and TA, and additional amide bonds between gelatin and chitosan. Hydrogen bonds were also established between PVA and chitosan, as well as among PVA molecules. In the acidic inflammatory infection environment, the hydrogel facilitated the reversible dissociation of boric ester bonds and the sustained release of TA, which contributed to its antibacterial and anti-inflammatory effects. Additionally, the optimized hydrogel with a tensile strength of 0.38 ± 0.0054 MPa and an adhesion strength of 174.44 ± 3 Pa exhibited a high water content (>85%), shape memory behavior, self-healing ability, and excellent hemocompatibility (hemolysis rate < 3.2%), making it a promising material for wound healing applications [38]. The structure is regulated by both covalent and non-covalent bonds, which synergistically govern the physicochemical properties of the hydrogels, resulting in satisfactory performance. This study suggests that certain natural compounds can function simultaneously as crosslinkers and bioactive components in hydrogel wound dressings, allowing them to exert their inherent physiological activities while enhancing overall dressing efficacy. Identifying which natural compounds possess these dual functions and elucidating their mechanisms of action will require further investigation.
To more precisely control the release of bioactive factors and thereby promote cell proliferation and migration in wound tissues, a trilayer coaxial core–shell fibrous hydrogel was fabricated via coaxial microfluidic bioprinting. The core contained platelet-rich plasma and gelatin, the intermediate layer was sodium alginate, and the outer shell was chitosan, with CaCl2 serving as the ionic crosslinker. Upon external diffusion, Ca2+ established dual crosslinking by forming egg-box junctions with the guluronate blocks of alginate and coordinating with the amine groups of chitosan, thereby stabilizing the structure. The hydrogel exhibited high water uptake and retention. Its multilayer architecture increased diffusion tortuosity, reducing burst release and enabling sustained delivery of encapsulated growth factors. For this hydrogel, a biphasic release profile was observed: an initial burst of 37.71% within 48 h, followed by a slower release reaching 65.27% at 168 h [39]. In vivo, this wound dressing promoted granulation tissue formation and angiogenesis and supported a densely organized collagen network (collagen deposition ~96.7% at day 14); high-density hair follicle regeneration was also observed in the animal model. Table 2 shows main composition, water vapor transmission rate, swelling rate, cell viability, antibacterial rate, and wound healing rates of represented wound dressing.
Films are gas-permeable while being impermeable to liquids and bacteria, making them suitable for direct drug delivery to the wound site. However, their main drawbacks include potential pain during removal and limited use on non-highly exuding wounds. Common methods for fabricating wound dressings include solvent casting, spin coating, microfluidic spinning, and three-dimensional printing (as an alternative method to solvent casting) [59], with solvent casting being the most widely employed techniques.
Solvent casting is a straightforward method for producing robust films. It involves pouring a polymer dispersion into a mold, allowing the solvent to evaporate, and then peeling it off the mold [59]. The poor mechanical properties and brittle texture of pure chitosan limit its use as the single material for film fabrication. Blending it with functional components and synthetic polymers can enhance the film’s performance. Carbon quantum dots (CQDs) are carbon-based nanomaterials known for their exceptional antibacterial properties and biocompatibility, making them ideal candidates for various material fabrication applications. In a study, CQDs were synthesized from turmeric powder. Then, a composite film was prepared by combining chitosan, PVA, CQDs and hyaluronic acid. The flexible composite film could adhere to the skin, and exhibited ideal mechanical properties (tensile strength 8.97 ± 0.93 N/m2). These properties were due to the hydrogen bonds formed between chitosan and PVA, which contributed to the formation of a denser structure. As the content of PVA increased, the film became thicker and softer, with a reduction in brittleness. Additionally, the film demonstrated excellent antibacterial activity, high biocompatibility, and effective migration closure capabilities, thereby enhancing its potential for wound healing applications [60]. However, the incorporation of CQDs reduced the film’s water vapor permeability, likely because the CQD-induced, more compact and ordered microstructure narrows membrane pores and increases tortuosity, thereby hindering vapor transport and slowing diffusion. Given the reliance on physical crosslinking, further studies should assess the duration for which the film maintains its structural integrity on moist wounds, as well as its potential to facilitate rapid hemostasis. Effective chemical crosslinking can further enhance the mechanical properties of chitosan-based films, even with the same primary materials. Genipin, extracted from the fruit of Gardenia jasminoides Ellis, has been demonstrated to effectively crosslink chitosan, offering high biocompatibility and low cytotoxicity. A chitosan/PVA film was prepared via genipin-mediated crosslinking. Within the film, hydrogen bonds between chitosan and PVA, coupled with the covalent bonds between genipin and chitosan, reinforced the structure, exhibiting enhanced healing performance, improved water uptake, greater porosity, and higher tensile strength than the uncrosslinked film [61]. However, this compact structure may impede cell penetration into the matrix. Notably, chemical crosslinking did not affect cell viability or migration, nor did it alter the wound closure rate. The underlying mechanism needs further investigation. To overcome this limitation, the development of chitosan-based materials with a porous structure could provide a potential solution.
Sponge, characterized by micrometer-sized pores, exceptional liquid absorbency, and high breathability, is typically processed using techniques such as freeze-drying [62]. However, sponges have certain limitations, such as relatively weak barrier properties and a lack of adhesive qualities, which necessitate the use of secondary dressings, tapes, or bandages to keep them in place. Chitosan sponges have been shown to effectively promote wound healing in diverse and complex wound environments, owing to their rapid fluid absorption capacity and varied pore sizes. Researchers have developed a new method to fabricate pure chitosan sponges with enhanced mechanical properties that do not require a freeze-dryer, thereby making the process simpler, more environmentally sustainable, and cost-effective. In this method, chitosan was dissolved in an acetic acid solution, followed by the addition of a NH4HCO3 solution. The mixture was then stirred and poured into a mold. The subsequent single freeze-thaw cycle, involving freezing at −20°C and thawing at 26°C, resulted in the formation of a sponge (Fig. 2a). The sponge maintained the thermal stability and crystallinity of chitosan, while exhibiting remarkable mechanical strength (26.35 kPa at 1.57% solid content), porosity (86.63%), water vapor transmission rate (1136.5 g/cm2/day), and enhanced wound healing properties (Fig. 2b) [63]. Chitosan’s positive charge facilitates platelet adhesion and red blood cell aggregation. However, when used in its pure form, chitosan displays limited hemostatic efficacy and suboptimal overall performance. As a result, extensive research has been dedicated to enhancing its properties through crosslinking or modification. Silver nanoparticles (AgNP) have gained preference among researchers as a non-toxic, inorganic antimicrobial agent and have been used to fabricate a composite sponge with CMCS and κ-carrageenan (κ-C) through physical crosslinking. Hydrogen bonding and electrostatic interactions between CMCS and κ-C resulted in irregular, pore-like structures. The sponge exhibited a porosity of 70.24 ± 6.84% (lower than the pure chitosan sponge mentioned above), a water vapor transmission rate of 1339.27 ± 32.48 g/cm2/day, and a water absorption of 2690.26 ± 144.60% when the CMCS/κ-C mass ratio was 1:1. The addition of AgNP effectively inhibited the proliferation of E. coli and S. aureus, counteracting the reduced antibacterial activity caused by electrostatic combination between protonated amino groups on CMCS and sulfate groups on κ-C [48].

Figure 2: (a) Schematic illustration of the freeze-thaw preparation process for pure chitosan sponge and (b) its wound healing performance in vivo [63]. (c) Proposed structure and mechanism of crosslinked chitosan/alginate/hyaluronic acid polyelectrolyte composite sponges and (d) its wound contraction in vivo [64]. (e) Schematic representation of the formation of the PLGA-chitosan-tranexamic acid composite sponge and (f) its superficial blood absorption behavior [65]. ** and *** represent p < 0.01 and p < 0.001 vs. the gauze dressing group, respectively; # represents p < 0.05 vs. the CS-HOAc group [Adapted with permission from Refs. [63–65]. Copyright © 2021, Elsevier, 2025, Elsevier and 2025, Elsevier].
A chemically crosslinked composite sponge was fabricated, which integrated the advantages of chitosan, poly(lactic-co-glycolic acid) (PLGA), and tranexamic acid, exhibiting a uniform, fluffy structure with pore sizes ranging from 20 to 30 μm. Amide bond crosslinking between the components enhanced its physical properties, including improved compressive strength (9.67 kPa) high porosity (approximately 85%), excellent water absorption (3197 ± 105% within 30 s, higher than the physical crosslinked chitosan sponge mentioned above). This sponge exhibited good biocompatibility, with a hemolysis rate < 1.2% and a relative cell growth rate > 125%. In vitro and in vivo hemostatic evaluations demonstrated a shorter hemostasis time compared to the unmodified chitosan sponge (Fig. 2e), 72 s in the rat tail amputation model and 27 s in the mouse liver injury model [65]. A composite sponge incorporating both physical and chemical crosslinking was prepared by blending alginate, chitosan, hyaluronic acid, followed by genipin-mediated crosslinking. Its proposed structure and mechanism was shown in Fig. 2c [64]. Increasing the chitosan content reduced the mechanical properties of the sponges while accelerating their biodegradation rate. The final sponge exhibited favorable physical and biological properties, with a uniform network structure, high porosity, and a negatively charged surface, which enhanced its coagulation performance. Animal studies confirmed its ability to promote wound healing (Fig. 2d). The formation of polyelectrolyte complexes using various components is an effective method to improve the mechanical properties of composite sponges. Charged natural macromolecules are expected to have broader applications in hemostasis and the preparation of wound-healing dressing.
Fiber-based materials, such as membranes and mats, are extensively used in wound repair due to their favorable properties, including high exudate absorption, diverse morphologies, large surface area, and high porosity. These physical characteristics mimic the in vivo microenvironments, thereby promoting cell infiltration, adhesion, and proliferation [66]. Additionally, fiber alignment can guide cell growth along a specific direction. However, because chitosan has relatively low spinnability, it is often combined with other polymers to enhance spinnability and facilitate the fabrication of chitosan-based fibers. Micro- or nanoscale polymeric fibers are commonly fabricated using various spinning techniques, with electrospinning, blow spinning, and microfluidic spinning being the most frequently employed methods in recent years.
Electrospinning
Electrospinning is a simple, efficient, and most commonly used technique for producing polymer fibers with diameters ranging from several micrometers to the nanoscale. Over the past decade, it has attracted significant attention for its versatility in controlling fiber structure, orientation, and dimensions, making it particularly well-suited for fabricating wound dressing materials [67]. Electrospinning, an electrohydrodynamic process, typically consists of a high-voltage power supply, a syringe pump, a spinneret, and a collector [68]. In theory, most polymers, when dissolved in an appropriate solvent, can be electrospun into fibers. However, pure chitosan is difficult tor electrospin because it is polycationic in acidic solutions. The high electric field induces strong electrostatic repulsion among charged groups in the polymer backbone, which hinder continuous fibers formation and often yields beads, particles, or flattened structures [69]. Co-electrospinning chitosan with synthetic polymers that exhibit good electrospinnability is commonly employed to improve processability and enhance the mechanical properties of the resulting chitosan-based fibers [70]. This approach preserves the biological advantages of chitosan while integrating the mechanical strengths of synthetic polymers. Poly(ethylene oxide) (PEO), noted for its excellent electrospinnability, was mixed with chitosan, electrospun, and subsequently crosslinked with glutaraldehyde, producing uniform, defect-free mats with an average fiber diameter of 165 ± 35 nm. Glutaraldehyde crosslinking has been reported to improve mechanical durability and wet stability, enhance exudate absorption capacity, and promote fibroblast adhesion and proliferation, thereby supporting wound healing [71]. However, another study indicated that UV irradiation can photo-crosslink PEO, while also partially degrade of chitosan, particularly at higher exposure doses [72]. Therefore, strategies to enhance the physical properties of chitosan-based electrospun fibers while preserving bioactivity require further exploration. Owing to their intrinsic bioactivity, chitosan-based fibrous membranes are promising as wound dressings and drug-delivery platforms capable of releasing therapeutic agents to further promote wound healing. Formulations that rely on physical crosslinking can also be electrospun into nanofibers. For example, a composite of PEO, chitosan, and cellulose nanocrystals (CNC) was electrospun with acacia extract as a model drug. Hydrogen bonds formed between the −OH groups of CNC and the −NH2 groups of chitosan, as well as between chitosan and PEO. The resulting nanofibers exhibited uniform morphology, antibacterial properties, sustained drug release, and an average diameter of 83 nm [73]. In addition to this, co-electrospinning PVA (polyvinyl alcohol), PLA (polylactic acid) [74], and PCL (polycaprolactone) [75] with chitosan was also reported. However, the bioactive properties of chitosan may be altered when combined with synthetic polymers. To mitigate these effects, studies have focused on enhancing the mechanical properties of electrospun chitosan by co-electrospinning it with natural polymers and small amounts of synthetic polymers, such as collagen and honey [76], gelatin [77], pectin [78]. Electrospun nanofibers typically exhibit solid interiors, circular cross-sections, and smooth surfaces.
Notably, coaxial electrospinning enables the fabrication of core-shell nanofibers using a spinneret consisting of two concentric capillaries connected to separate syringe pumps. The electric field exerts a force on the shell solution, causing the ejection of shell fluid. Meanwhile, shear forces, generated by adhesion and friction, act on the core solution, pulling it into the cone and forming a coaxial double jet [79]. Coaxial electrospinning enables the fabrication of core-shell fibers that combine materials with contrasting properties, thereby integrating their respective advantages into a single dressing with sustained release capabilities. Curcumin, a polyphenol compound with anti-inflammatory properties that promote collagen deposition and stimulate angiogenesis, was incorporated into a core solution consisting of chitosan and ZnO nanoparticles in hexafluoroisopropanol (HFIP) to enhance both biological and antimicrobial activity. A separate HFIP solution of polycaprolactone (PCL) formed the shell layer (Fig. 3a). The resulting core-shell nanofibers demonstrated excellent biocompatibility (Fig. 3b) and antibacterial properties. The incorporation of ZnO nanoparticles and curcumin improved the physicomechanical properties of the fibers through the formation of hydrogen bonds with the polymer matrix. In vitro degradation over 1200 h indicated effective encapsulation of the core components [80]. Using a similar approach, Aloe vera extract was loaded into the core solution, while the shell solution contained chitosan, κ-carrageenan, and PVA. The resulting core–shell nanofibrous membrane exhibited a uniform, multilayered structure with smooth surfaces and an average fiber diameter of approximately 180 nm. In addition to its structural uniformity, the membrane demonstrated enhanced wound-healing potential, as well as notable antibacterial and anti-inflammatory activities attributable to the Aloe vera extract, with achieving an encapsulation efficiency that exceeded 80% [73]. Importantly, no toxic reagents or chemical crosslinkers were employed during fabrication, making this method more environmentally friendly and enhancing the safety of the materials.

Figure 3: (a) Schematic illustration of the core-shell nanofibers prepared via coaxial electrospinning and (b) their cellular toxicity [80]. (c) Schematic illustrations of the microfluidic platform, core-shell microfibers generation process, and formation mechanism and (d) their cytotoxicity [81]. *, ** and ns represent p ≤ 0.05, p < 0.01 and p > 0.05, respectively [Adapted with permission from Refs. [80,81]. Copyright © 2025, Elsevier and 2022, Elsevier].
Single-layer wound dressing materials often fail to meet all clinical requirements due to the inherent limitations of the polymers used. To address this, bilayered nanofibrous scaffolds have been developed, comprising a dense outer fiber-based layer and a porous inner layer. The outer layer acts as a protective barrier against external threats, including microorganisms, physical damage, and chemical agents, while the inner layer provides a supportive scaffold that promotes bacterial inhibition, cell adhesion, angiogenesis, tissue regeneration, and re-epithelialization [82,83].
Despite its advantages, electrospinning has several drawbacks: heavy reliance on organic or toxic solvents—especially for hydrophobic polymers; difficulty processing solutions with very low or very high viscousity; challenges in large-scale production; and the necessity for high-voltage power supply [84]. In line with growing environmental and health imperatives, green fabrication strategies for electrospun wound dressings have expanded in recent years [85,86], emphasizing aqueous- or ethanol-based spinning dopes, benign chemical crosslinkers, and more scalable, solvent-efficient processes such as solution blow spinning.
Solution blow spinning
Solution blow spinning (SBS) features lower energy consumption, a simpler process, and higher productivity in fiber fabrication. In a typical SBS setup, a polymer solution and a pressurized gas are co-delivered through concentric nozzles; the high-velocity gas stream stretches the fluid and accelerates solvent evaporation, resulting in fiber formation. Compared to electrospinning, SBS produced thicker, more polydisperse fibers with micro- to nanoscale diameters [87].
In a separate study, chitosan/PEO nanofiber dressings were fabricated using SBS without any crosslinkers agents or additives. A chitosan/PEO solution in acetic acid was blow-spun into nanofibers and vacuum-dried to remove acetic acid and excess water. The fibers were sequentially immersed in anhydrous ethanol and deionized water, and finally freeze-dried. The resulting fiber diameters ranged from 278.6 ± 72.6 nm to 397.4 ± 87.2 nm, with higher chitosan concentrations yielding thicker fibers. The dressing exhibited enhanced crystallinity and a higher chitosan content, and demonstrated excellent wettability, antibacterial properties (>99.99%), high breathability, and notable antioxidant activity. Furthermore, it accelerated wound healing, achieving closure within 10 days [88]. Bilayer membranes can also be produced using the SBS method. Quercetin (QC) is a polyphenolic flavonoid derived from many plants, with functions of antioxidant, anti-inflammatory and bacterial inhibition [89]. A bilayer membrane was explored with an inner chitosan/PEO layer loaded with QC and an outer polylactic acid (PLA) layer; acting as a barrier to bacteria and contaminants. The membrane’s fluffy, porous structure offers excellent air permeability, optimal water vapor transmission (2300 ± 103.21 g/cm2/day), sustained QC release for 24 h, effective microbial infection control (99.5%, 99.1%, 95.1% against E. coli, S. aureus, and P. acnes, respectively), and enhanced antioxidant (antioxidant rate 92 ± 0.06%) and anti-inflammatory properties (IL-6 and TNF dramatically decreased from 34.38 ± 1.57 and 170.74 ± 7.57 pg/mL to 21.40 ± 0.21 and 107.65 ± 1.01 pg/mL), making it ideal for wound healing applications [90]. While bilayer structures combine the advantages of different layers, their preparation process becomes more complex, which can hinder large-scale production.
Microfluidic spinning
Microfluidic spinning is a technology that enables precise fluid manipulation at the microscale, facilitating the continuous fabrication of microfibers with uniform diameters and controlled properties. It employs specialized microchannels to facilitate the flow and interaction of fluids with specific viscosity. By manipulating the driving force of the fluid and the tensile force applied by the receiver, fibers of various sizes and morphologies can be produced. Most natural polymers can be spun into fibers with consistent shapes in a mild spinning environment, without toxic solvents or synthetic polymer additives, using microfluidic spinning. The design of microfluidic devices and the choice of materials enable the production of fibers with various geometries, including core-shell fibers, porous fibers. In a previous study, core-shell microfiber mats were fabricated, with an average diameter of 100.1 ± 1.9 µm, demonstrating the ability to promote wound healing by supporting all four stages of the healing process. The outer phase consisted of a alkyl-chitosan solution and a CaCl2 solution, while the inner phase was composed of a sodium alginate solution. Ionic crosslinking between Ca2+ and alginate, along with interactions between alkylated chitosan and Na-alginate, resulted in the formation of a unique internal structure (Fig. 3c). This structure enhanced the mat’s ability to absorb blood and exudates (192.2 ± 6.2%). Additionally, alkylated chitosan imparted excellent hemostatic performance (within 105.8 ± 2.8 s) and strong antibacterial activity to the microfibers [81]. This approach prevents denaturation or damage during the fabrication process, making it particularly suitable for sensitive materials intended for cell adhesion and culture [91]. The core-shell structure holds significant promise for the controlled release of therapeutic agents at targeted stages, thereby further aiding the healing process. In addition to the physiochemical properties, microfluidic-spun fibers can serve as temporary scaffolds to support cell growth. Researchers have sought to merge blow spinning and microfluidic spinning into a single process known as microfluidic blow-spinning. This fiber fabrication technique involves the integration of two coaxial streams: a polymer spinning solution and a pressurized gas envelops the polymer solution. These combined streams form fibers that are deposited along the direction of the gas flow. In one example, a chitosan solution in formic acid (FA) was blended with silk sericin (SS). The microfluidic pump regulated the flow rates of the alcohol-soluble polyurethane (APU) solution and the chitosan/SS mixture. This process transformed the secondary structure of SS from low to high crystallinity, enhancing its stability and spinnability. The resulting membrane demonstrated outstanding mechanical strength (18.08 ± 1.54 MPa), exceeding that of native mouse skin (11.01 MPa), a water contact angle of 84°, and good cell-adhesion properties. It effectively promoted fibroblast proliferation (cell viability 95.7%), enhanced collagen deposition and neovascularization, and increased granulation tissue thicknesses to 2.98 mm by day 10, owing to the complementary bioactivities of its constituents [92]. Chitosan-based fibers, fabricated through spinning technology, exhibit significant potential in biomedical applications. Future research will likely focus on the integration of various fabrication methods and the optimization of formulations.
The development of a clinical wound dressing typically proceeds through three stages: preclinical studies, regulatory approval, and clinical adoption. Chitosan-based dressings have been extensively investigated in vitro and in vivo, with emphasis on their biocompatibility, biodegradability, hemostasis, and wound closure rate, and these studies collectively support their potential in wound healing.
Compared with other advanced wound-care products, chitosan-based dressings often show comparable—and in some cases superior—performance. In the United States, many have obtained FDA 510(k) clearance; in the European Union, they carry the CE mark, typically as Class IIa or IIb devices; and in China, they are approved by the NMPA as Class I or II devices. Clinically, they are used for a range of wounds—including chronic wounds, diabetic foot ulcers, and pressure ulcers—as well as for hemostasis in traumatic injuries [93]. Representative products available include ChitoFlex®, HemCon®, Axiostat®, Celox®, ChitoGauze® and Chitosan Skin® [24,93–97]. Ongoing clinical studies are assessing chitosan-based dressings for chronic wounds, burn wounds and rapid hemostasis of deep wounds.
Konjac glucomannan is a water-soluble natural polysaccharide derived from konjac tubers, consisting of D-mannose and D-glucose linked by β-1,4 glycosidic bonds, with acetyl groups randomly distributed at the C-6 position; a minor degree of β-1,3 branching occurs at the C-3 position of mannose. KGM exhibits a significant water-absorbing capacity, forming highly viscous gel-like solutions that can transition into either thermally reversible or irreversible hydrogels, depending on the treatment. The unique structure of KGM supports a wide range of biological activities, including antitumor effects [98], cholesterol regulation [99], antidiabetic effects [100], intestinal activity promotion [101], hemostasis, and immune function enhancement [40]. According to previous reports, KGM has also demonstrated properties that promote wound healing, including regulation of anti-inflammatory cytokine secretion [102], facilitation collagen deposition [103], promotion of angiogenesis [41], and enhancement of fibroblast metabolic activity in wounds. As a result, KGM holds significant potential for diverse applications in wound healing.
3.2 Konjac Glucomannan-Based Material
The microenvironment plays a crucial role in regulating wound healing, and KGM-based hydrogels can effectively modulate it. These hydrogels are primarily categorized into three types: single-network, semi-interpenetrating network, and interpenetrating network. Bletilla striata polysaccharide (BSP), a bioactive component derived from the herb Bletilla striata, is known for its specific affinity for macrophages and exhibits anti-inflammatory and hemostasis activities, while promoting cell proliferation, migration, and angiogenesis. It is commonly used in wound healing and wound dressing preparation [104–107]. To prepare a hydrogel with ideal stability and mechanical properties, BSP was crosslinked with KGM using 1,4-butanediol diglycidyl ether (BDDE) under alkaline conditions, resulting in a single-network-hydrogel. Within this hydrogel, hydrogen bonding was formed among the polysaccharide molecular chains, while BDDE reacted with the hydroxyl groups of the polysaccharides through its epoxy groups (Fig. 4a), creating a stable three-dimensional network. The resulting hydrogel exhibited improved properties, including a tensile strength of 1.3 kPa, an adhesive strength of 848.5 ± 31.4 Pa, and rapid self-healing with complete recovery within 2 min (Fig. 4b). Mechanistically, self-healing proceeds through partial rupture of the covalent network while intact BDDE crosslinks preserve overall continuity; high hydration then facilitates polysaccharide chain migration to the fracture interface and physical re-entanglement; finally, rapid reformation of hydrogen bonds and other noncovalent interactions restores local network integrity. The hydrogel preserved the structure and bioactivity of both KGM and BSP, while regulating inflammatory responses, stimulating cell migration and proliferation (cell viability 137.1% at 24 h), promoting collagen accumulation, enhancing blood vessel formation, and promoting wound healing (healing rate 94.55 ± 0.45%) [40].

Figure 4: (a) Schematic illustration of the chemical crosslinking between Bletilla striata polysaccharide, konjac glucomannan, and 1,4-butanediol diglycidyl ether in the KGM/BSP composite hydrogel and (b) its self-healing property [40]. (c) Schematic illustration of the formation of an interpenetrating network in Zinc-induced photocrosslinked konjac glucomannan/glycyrrhizic acid hydrogel and its (d) injectable, moldable and self-healing properties [108] [Adapted with permission from Refs. [40,108]. Copyright © 2025, Elsevier].
KGM exhibits excellent gel-forming ability when subjected to alkali and thermal treatments. Alkali treatment induces deacetylation, which causes the molecules to transition from a random coil conformation to a self-assembled structure, thereby initiating gelation. Upon further heating, a true hydrogel is formed as significant aggregation occurs, leading to the development of a complex supramolecular structure [18]. Therefore, the addition of an alkali solution followed by heating is a common procedure in the preparation of KGM-based hydrogels. Although deacetylation enhances the performance of KGM-based hydrogels, the network is still constrained by limited mechanical strength. Incorporating biopolymers enhances the biological performance of the formulation and yields semi-interpenetrating polymer network hydrogels that can incorporate additional drugs and ions. A hydrogel was prepared using guanosine, 2-formylphenyl boronic acid, K2CO3, deferoxamine mesylate (DFO), ZnSO4 and KGM. DFO, an angiogenic agent, was grafted onto guanosine-stacked (G4) nanofibers through Schiff base crosslinking. Zinc ions enhanced the mechanical properties of the hydrogel by forming coordination crosslinks with DFO (Fig. 5a), while simultaneously conferring antibacterial properties. Strong hydrogen bonds between KGM and G4 nanofibrils further reinforced the network. The resulting hydrogel demonstrated a stable structure with the storage modulus (G′) consistently exceeding the loss modulus (G″), and exhibited sustained drug release, antioxidant properties, increased collagen deposition, reduced inflammation, and enhanced angiogenesis, highlighting its potential as a promising wound dressing material [42] (Fig. 5b). The physiological activity of DFO remains intact within this semi-interpenetrating network, while KGM not only exhibits immunoregulatory effects but also significantly enhances the mechanical properties.

Figure 5: (a) Schematic illustration of the preparation and interactions between components in the G4/DFO-KGM/Zn2+ hydrogel. (b) H&E staining and Masson staining results of wound tissues on day 14 [42] [Adapted with permission from Ref. [42] Copyright © 2025, Elsevier].
KGM is commonly modified to facilitate effective conjugation with other compounds, enabling the formation of composite hydrogels through the interpenetration of two or more independently crosslinked polymer networks (Fig. 4c). Methacrylic anhydride (MA) was grafted onto KGM to prepare KGMMA. A mixture containing KGMMA, glycyrrhizic acid (GA), zinc sulfate, and using phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) as a photoinitiator was heated to form KGMMA hydrogels. GA rapidly formed a primary network under Zn2+ coordination. The network was then photocrosslinked to generate a secondary network based on KGMMA, whose methacrylate vinyl groups served as reactive sites for photopolymerization, thereby providing robust structural support. The resulting hydrogel with interpenetrating network exhibited excellent injectability (Fig. 4d), rapid self-healing (~5 min), and high compressive strength (compression at break 57 ± 3.4 MPa), while intrinsically scavenging reactive oxygen species without additional drug components. It also promoted cell migration (wound closure 80% at 12 h), enhanced angiogenesis (CD31 density 2.18-fold and VEGF coverage 2.52-fold vs. control), supported the regeneration of skin appendages, and accelerated wound healing (57% and 97% closure by days 7 and 14, respectively) [108]. However, its adhesion performance needs to be improved to meet the requirements of tissue engineering. Researchers prepared a self-healing hydrogel with good tissue adhesion, and dual antioxidative-immunomodulatory properties to solve this problem. Gallic acid was grafted onto KGM through esterification between KGM hydroxyls and the gallic acid carboxyl group. Dopamine (DPA) is the key adhesive component in mussel adhesive proteins, ensuring strong adhesion in aqueous environments, which was conjugated to xanthan gum via amide coupling. These two modified polysaccharides then formed a bioadhesive hydrogel through physical crosslinking driven by intermolecular hydrogen bonding. The abundant phenolic hydroxyl and amine groups in DPA interacted with skin surface proteins, yielding an adhesion strength of 17 kPa. The multiple phenolic hydroxyl groups in DPA and gallic acid confer strong antioxidant activity (at 1 mg/mL, the hydrogel scavenged approximately 75% of DPPH) and cytoprotective effects (at 50 μg/mL, it reduced ROS-positive cells from 47% to <30% and maintained cell viability >95% after exposure to 100 μM H2O2 for 48 h). In vivo, the hydrogel accelerated wound closure by 81% within 7 days and enhanced neovascularization by 8.7-fold by day 14 [109].
KGM can form single-networks, semi-interpenetrating networks, and interpenetrating networks through physical crosslinking, chemical crosslinking or their combination. According to the majority of experimental results, various functional components can exert their activities to different extents. KGM not only serves as a functional component but also plays a crucial role in supporting the hydrogel’s structure. However, due to differences in experimental methods, there are multiple testing approaches for the same parameter, which leads to difficulties in comparing the gel properties from different studies. For instance, gel strength may be expressed by tensile strength, compressive strength, adhesion strength, or storage moduli. Additionally, variations in testing time points for the same method can reduce the comparability of results, such as cell viability being tested at 24, 48, or 72 h. Therefore, establishing standardized testing protocols is necessary.
Oxidized konjac glucomannan (OKGM), a derivative of KGM, has demonstrated benefits including improving gel structure, reducing phase separation, and enhancing stability, and can also be utilized for the preparation of wound dressings. OKGM was synthesized by oxidizing KGM with sodium periodate and was then combined with carboxymethyl chitosan (CMCS) to produce a hydrogel with excellent adhesiveness. It has been shown that OKGM can crosslink with CMCS via a Schiff base reaction, resulting in a crosslinked hydrogel with controllable drug-release properties [110]. Honokiol, known for its antibacterial, antioxidative, and anti-inflammatory effects, was encapsulated in stevioside-stabilized micelle particles. CMCS and OKGM were separately dissolved in a stevioside-stabilized honokiol micellar solution, mixed, and subsequently heated to induce gelation. The aldehyde groups of OKGM reacted with the amino groups of CMCS, resulting in the formation of dynamic imine linkages, yielding a single-network structure whose reversible bonds impart self-healing behavior and injectability. The hydrogel’s density increased proportionally with the concentration of OKGM. This hydrogel displayed injectability, excellent adhesiveness, self-healing capabilities, and high transparency, making it highly suitable for wound healing applications. Its potential for wound healing was further demonstrated through its ability to inhibit bacterial growth, reduce the inflammatory response, enhance re-epithelialization, and promote collagen deposition [103]. Similarly, OKGM can also react with other macromolecules via a dynamic Schiff base reaction to form a multifunctional crosslinked hydrogel. This can be either a single-network [18] or an interpenetrating network [111]. Interestingly, researchers have sought to improve the mechanical and physiological properties of hydrogels by combining functionalized nanofibrous membranes loaded with ε-poly-lysine (ε-PL, an antimicrobial peptides) and allantoin with hydrogels. This resulted in a novel hydrogel-based fibrous membrane with exceptional mechanical properties, sustained release, adhesion, antibacterial activity, and biocompatibility. The unique porous structure, formed by the nanopores of the fibrous membranes and the micropores of the hydrogels, facilitates the stable and sustained release of the drug. PCL, allantoin, and ε-PL were dissolved in a mixture solvent of formic acid and acetic acid and then electrospun into nanofiber membranes. The fiber-based membranes were then irradiated and immersed in an OKGM solution. Chitosan oligosaccharide (COS) was added to the blend to form the final hydrogel-membrane combined wound dressing. The dynamic Schiff base bond between COS and OKGM, imparted self-healing properties to the hydrogel. Despite the nanofiber membranes being soaked in OKGM and COS solutions to form a crosslinked network, they retained a relatively compact, stacked structure without noticeable delamination or loosening [112]. The resulting dressing exhibited excellent bactericidal efficacy (99.99% against S. aureus and E. coli), high biocompatibility (cell viability > 100%), and strong adhesion, without visible damage or detachment. OKGM provides good aqueous solubility and thermal stability but has low swelling capacity, whereas deacetylated KGM (DKGM) offers high gel strength at the expense of markedly reduced solubility and excessive hardness that hinder processing. To combine these advantages while mitigating their drawbacks, researchers prepared oxidized deacetylated KGM (ODKGM) by sequential deacetylation followed by ozone oxidation. ODKGM exhibits improved aqueous solubility and thermal stability, low bulk density, and, unlike OKGM or DKGM, forms a soft, processable hydrogel, making it a promising candidate for gel-based dressings [113]. Although its use in dressing development has not yet been reported, its properties indicate strong potential.
Low-molecular-weight degradation products of KGM have been shown to enhance bioactivity, including antioxidant effects and suppression of proinflammatory cytokines, and are also used in the preparation of wound dressings. In a recent study, a composite hydrogel via a Schiff base reaction was prepared. KGM was first degraded into konjac glucomannan oligosaccharides (KOS; 2–3 kDa), which were then modified with 4-carboxybenzaldehyde to afford benzaldehyde-functionalized KOS (BKOS). BKOS subsequently reacted with primary amines on hydrazide-modified polyglutamic acid (HPGA) and ε-polylysine (ε-PL) to form a BKOS/HPGA/ε-PL (BKPP) hydrogel. The resulting hydrogel was injectable and self-healing, exhibiting rapid gelation within 23 s and allowing two freshly cut pieces to fully rejoin within 10 min. BKPP exhibited antibacterial activity against representative G− and G+ bacteria, inhibiting E. coli and S. aureus by 83.3% and 85.0%, respectively. It also modulated macrophage phenotype: in M1-polarized macrophages, IL-10 mRNA increased to 393% compared with control, whereas IL-6 mRNA decreased to 41% of control, changes consistent with attenuated inflammation and potentially contributing to accelerated wound healing. Mechanistically, gel network formation consumes free hydrazide groups on HPGA, thereby reducing any potential proinflammatory effects associated with unreacted hydrazides; moreover, imine/hydrazone linkages and certain hydrazone derivatives have been reported to exhibit anti-inflammatory activity [114].
Films easily conform to the patient’s body, including challenging areas such as joints. They promote moisture evaporation, act as a barrier to external contamination, and enable wound bed inspection without removing the dressing [59]. Solvent casting is a widely employed method for film preparation, involving the application of a polymer solution onto a substrate followed by solvent removal. This process promotes the orientation of polymer molecules, facilitating the formation of a cohesive film. Incorporating KGM with other biopolymers is an effective method to overcome the limitations associated with pure KGM-based films. Tannic acid (TA), with its phenolic hydroxyl group and abundant dihydroxyphenyl units, readily interacts with other polymers through non-covalent forces, imparting antibacterial, anti-inflammatory, and free radical scavenging properties. A blend film was prepared using KGM, TA, and gluconolactone. TA acted as an effective crosslinker, while gluconolactone was physically entrapped within the network. Crosslinking improved cell metabolic activity of the film, with MTT assay results reaching >150% of control. The inclusion of TA improved antibacterial efficacy against S. aureus and E. coli, as evidenced by increased zones of inhibition that enlarged over incubation time (within 6 h, McFarland bacterial growth method). Gluconolactone further improved hemocompatibility (hemolysis degree < 0.8%), as well as enhanced metabolic activity in human dermal fibroblasts (>150% of control, MTT), which is consistent with improved cell proliferation and may contribute to improved wound healing [54]. Bilayer films exhibit a multiphase structure in which the unique characteristics of each component are maintained within its respective layer, resulting in enhanced mechanical, optical, and barrier properties when compared to single-layer films. Therefore, a bilayer film was explored via two-step casting using chitosan and KGM. The resulting transparent bilayer film exhibited a higher crystallinity than pure KGM films and showed a distinct interface between the two layers. The film resisted spontaneous deformation while providing a moist, breathable environment conducive to wound healing [115]. The bilayer film had a water vapor transmission rate of 306 ± 42 g/cm2/day, which is comparable to that of healthy human skin (200–220 g/m2/day).
Titanium dioxide (TiO2) produces reactive oxygen species, such as hydroxyl radicals upon exposure to ultraviolet (UV) light due to its photocatalytic activity, which can confer antimicrobial properties. Buliding on this, researchers integrated inorganic compounds with KGM by using TiO2 as a carrier for curcumin (Cur) and 2-hydroxypropyl-β-cyclodextrin (HPCD) to fabricate a film; HPCD improved Cur solubility/stability and facilitated its loading. Upon 405 nm irradiation, the film demonstrated strong photodynamic bactericidal activity, achieving a 96.62% reduction in viable bacteria (S.aureus, irradiated for 15 min). Theoretically, the TiO2/Cur/HPCD complex was adsorbed onto the bacterial surface, thereby disrupting the bacterial membrane and enhancing antimicrobial efficiency. Despite the incorporation of TiO2, the film exhibited good biocompatibility (hemolysis rate < 2%, cell viability > 80% by Live/Dead). However, most blend films dissolved within 30 min in pure water, indicating limited residence time and insufficient capacity for prolonged wound exudate management without compromising structural integrity. Additionally, the films promoted angiogenesis, reduced the expression of inflammatory markers, and increased the levels of angiogenic factors in a mouse model of bacterial infection, thereby accelerating wound healing (84.6% closure at day 12) [116].
Inspired by natural materials, researchers have sought to develop biomimetic materials with outstanding mechanical properties by combining organic and inorganic components. Layered hydroxyapatite (LHAp) has emerged as an ideal material for composite fabrication due to its superior biocompatibility, biodegradability, and bioactivity, especially when compared to non-biodegradable alternatives. A novel nacre-like, film-type nanocomposite composed of LHAp, graphene oxide (GO), and KGM was fabricated through a simple evaporation-induced assembly process (Fig. 6). Hydrogen bonding between LHAp and GO, GO and KGM, together with electrostatic interactions were between COO- and Ca2+, improved the film’s water resistance, allowing it to maintain structural integrity in water for up to 7 days. Increasing the LHAp content from 0 to 20 wt% increased tensile strength and Young’s modulus by 43.8% and 134.7%, respectively (tested in dry state at a strain rate of 10 mm/min); however, excessive LHAp loading caused cytotoxicity to MC3T3-E1 cells, as evidenced by a decrease in CCK-8 OD450 from ~2.2 to ~0.8 when LHAp increased from 0.05 g to 0.15 g/10 mL. At suitable LHAp levels, the release of Ca2+ and PO43− was associated with enhanced osteogenic bioactivity, evidenced by the highest ALP activity at days 7 and 14 and increased mineralization by alizarin red staining (79.6%), which is consistent with promoted osteoblast differentiation and may support proliferation. The composite film displayed a brick-and-mortar structure with grainy surfaces, a tensile strength of 153.4 MPa and a Young’s modulus of 8.1 GPa [112]. Biomimetic materials accelerate the wound healing process by recapitulating the physical properties of the extracellular matrix and key features of the cellular microenvironment. These materials enhance cell attachment and promote tissue regeneration, thereby improving the performance of wound dressings across all stages of healing. Moreover, they hold great potential for personalized wound care, as they can be tailored to individual patient characteristics, such as wound type, size, and location, ultimately optimizing treatment outcomes.

Figure 6: Schematic representation of the “brick-and-mortar”-like structure of the layered hydroxyapatite/graphene oxide/KGM nanocomposites and the interfacial interactions between their components [117] [Adapted with permission from Ref. [117]. Copyright © 2021, Elsevier].
To better serve as a mechanical barrier, an asymmetric membrane was prepared using single-component KGM. A KGM/NaOH solution was partially dried to form a dense top layer, then freeze-dried to create a porous sublayer, the membrane was subsquently neutralized to remove residual alkali. The resulting membrane consisted of a dense, flat top layer over a porous bottom layer, exhibited high elongation at break (80.8 ± 23.2%) and effectively resisted microbial penetration (C. albicans, S. aureus, P. aeruginosa and E. coli), and low cytotoxicity [118]. However, its tensile strength (0.51 ± 0.12 MPa) and elastic modulus (0.54 ± 0.10 MPa), are much lower than the typical values reported for the human skin (2.5–35 and 4.6–20 MPa, respectively) [119]. Biocomposites have found wide application in wound healing to impart specific functions. An asymmetric composite membrane based on KGM incorporating silver-decorated cellulose nanocrystals (CNC-Ag) was fabricated, featuring a dense occlusive top layer and a porous sublayer. The membrane demonstrated antibacterial activity through a synergic effect of inhibiting bacterial adhesion and contact killing. However, no detectable release of silver nanoparticles was observed over 15 days [120]. Incorporating functional macromolecules into fibrous membranes can accelerate wound healing by mimicking the extracellular matrix, thereby enhancing their performance. Saikosaponins (SSD), active compounds derived from traditional Chinese medicine, are commonly used to regulate oxidative stress in the wound microenvironment due to their potent antioxidant and anti-inflammatory properties. Therefore, fibrous membranes loaded with SSD and further reinforced with pyrogallol (PG) were prepared to enhance their therapeutic efficacy for diabetic wounds. PG not only enhanced the interaction between the KGM and PVA molecular chains but also contributed to the antioxidant capacity of the membrane. As the PG content increased, the tensile strength improved from 2.6 to ~5 MPa, while water adsorption decreased from 13.4 to 10 g/g in water for 30 min. Additionally, higher SSD content promoted cell growth. The membranes demonstrated sustained release of SSD, and antioxidant activity (DPPH radical scavenging of 24.2% at 20 min, increasing over time), which effectively alleviates oxidative stress. The combined properties of biocompatibility, antioxidant activity, breathability, and moisture retention collectively promoted enhanced wound healing [121].
Dressings capable of dynamically removing excess wound exudate while maintaining a suitable moist microenvironment are beneficial for wound healing. The surface wettability of the dressing plays a critical role in the treatment process, and hydrophilic-hydrophobic asymmetric structures have been shown to be particularly effective. To achieve this, multi-layer membranes were developed to facilitate directional water transport, with a hydrophobic inner layer in contact with the skin and a hydrophilic outer layer exposed to the air [122,123]. A composite, hierarchical porous tri-layered membrane was developed to enhance fluid transport and promote wound healing (Fig. 7a). The hydrophilicity of each layer varied, driving the directional movement of exudate from the wound site. The inner layer, a hydrophobic silk fibroin (SF) membrane derived from Bombyx mori cocoons, featured a smaller porous structure to regulate fluid transport. The middle layer, designed to be superabsorbent with stable mechanical properties, was composed by chitosan, KGM, and dialdehyde starch. The outer layer membrane was formed by electrospinning cellulose acetate (CA) that had been soaked with graphene oxide (GO). When the composite dressing was applied to a wound, the hydrophobic inner layer initially directed the bio-liquids away from the wound and into the hydrophilic middle layer due to the differential capillary effect. The middle layer then absorbed and transported the fluids to the hydrophilic outer layer. Finally, the outer layer facilitated evaporation to the external environment through the photothermal effect of GO, preventing excessive moisture buildup and avoiding wound over hydration (Fig. 7b–d). This resulting tri-layered membrane demonstrated excellent wound healing capabilities, on day 3, the remaining wound area was 56.6% compared to 96.9% for the blank control. This improvement is attributed to its hierarchical porous structure, which effectively facilitated fluid transport and, under NIR irradiation, enables photothermal heating that increases the evaporation rate to approximately 6 times that of natural evaporation [124]. Notably, under NIR irradiation, the outer layer temperature increased from 29.1°C to 53.2°C, while the inner layer adjacent to the wound remained near 31.0°C, creating a temperature gradient of about 22.2°C between layer I and layer III. This gradient promotes evaporation. However, with the outer-layer temperature reaching 53.2°C, there is a potential safety risk of burns. Ensuring that the inner layer’s temperature remains controlled, so that the skin-contacting side does not exceed a safe threshold, is crucial.


Figure 7: (a) Schematic illustration of the sandwich-like structure of the silk fibroin/polysaccharide composite dressing. Diffusion and permeability of red ink in the silk fibroin/polysaccharide dressing with (b) initial layer I, (c) layer I treated with ethanol for 1 h and (d) 7 h [124]. (e) Hemostatic mechanism of the composite matrix comprising chitosan, KGM, thrombin-occupied microporous starch particles [125] [Adapted with permission from Refs. [124,125]. Copyright © 2023, Elsevier and 2020, Elsevier].
Biomedical sponges are soft, flexible scaffolds characterized by interconnected porous structures. These sponges can absorb and retain significant amounts of water, creating a moist environment that promotes effective wound healing. The preparation process of sponge is similar to that of hydrogel, typically involving the initial preparation of a solution, followed by drying. The components in the solution may interact with each other through physical or chemical crosslinking. For instance, a physically crosslinked composite sponge incorporating gelatin, KGM, gentamicin sulfate (GS) and gold nanoparticles (AuNPs) was prepared, leveraging their low-cytotoxicity and antibacterial properties. AuNPs, with diameters of 3.55 ± 0.03 nm and 2.86 ± 0.02 nm, acted as nanofillers that enhanced the water-retention capacity, tensile strength (0.29 ± 0.02 MPa), and elongation at break (11.56 ± 1.09%) of the sponge, while also reducing its swelling ratio. The interactions between gelatin, KGM, AuNPs, and GS were mediated by hydrogen bonds rather than chemical bonds. In addition to providing optimal mechanical properties and a moist environment conducive to wound healing, the resulting sponge demonstrated effective antibacterial activity against E. coli, S. aureus and methicillin-resistant S. aureus, without causing cytotoxicity to L929 cells (72 h viability 134.6 ± 6.13%) at the tested concentration [126]. Enhancing the roughness of the porous interface and incorporating coagulation factors can significantly improve the hemostatic efficiency of sponges. To address the need for rapid blood loss control in severe trauma treatment, sponges with hierarchical porous structures were developed based on physical interactions. The sponge comprised a chitosan/KGM matrix physically integrated with thrombin-loaded microporous starch particles (TOMSP). Its hierarchical porous structure (porosity ~86%) facilitated rapid infiltration and entrapment of blood-cell. A high positive surface charge (zeta potential +42.8 mV) contributed electrostatic interactions with negatively charged erythrocytes and platelets, promoting their accumulation at the wound-composite interface (Fig. 7e). The aggregated erythrocyte fraction and the platelet count on the sponge were reported as 6.53% and 7.25 × 106, respectively. These sponges exhibited rapid water uptake (contact angle < 43° at 0.5 s and 0° at 1 s), increased roughness and porosity relative to the control, as well as improved coagulation metrics, including shortened prothrombin time, activated partial thromboplastin time, and whole blood clotting indices [125]. In a rabbit ear injury model, the hemostasis time was approximately 50 s vs. 130 s for the control, and blood loss was 0.23 g vs. 0.4 g for the control.
Researchers aimed to enhance the hemostatic and photothermal antimicrobial properties of the KGM-based sponge by employing a two-step crosslinking method using chitosan, OKGM, tunicate nanocellulose (TCNCs), and polydopamine (PDA NPs). This approach resulted in a self-expanding, interpenetrating macroporous structure, in which both physical and chemical crosslinking coexist. In the first step, OKGM and chitosan crosslinked through dynamic covalent bonds, establishing the primary network. In the second crosslinking step, PDA NPs were incorporated, together with TCNCs, OKGM, and chitosan, they reinforced the matrix via hydrogen bonding and other noncovalent interactions. As a result, the composite sponge exhibited improved physical properties, strong adhesive capabilities, and favorable biocompatibility and hemocompatibility, making it an effective hemostatic agent for rapid bleeding control. Enhanced hemostatic was achieved by the sponge through its ability to adapt its shape and apply pressure to wounds, thereby reducing bleeding and managing excess exudate. Its porous structure further improved adhesion and supported effective barrier formation at the injury site, while promoting platelet activation and the entrapment of blood cells, ultimately resulting in the formation of a stable clot. Additionally, the PDA NPs acted as photothermal conversion centers, which, in combination with the intrinsic antibacterial properties of chitosan, significantly enhanced the antimicrobial efficacy of the sponge. This integrated mechanism are reported to support infection control and to facilitate wound healing [49].
Extensive research has been conducted on KGM-based biomaterials, including aerogels, microneedles, and nanofibers.
Aerogels are characterized by their interconnected networks, ultralight weight, high porosity, and large surface area [127], making them highly promising for applications in wound healing. The preparation of aerogels typically involves sol-gel processing, solvent exchange, and supercritical CO2 drying [128]. A KGM-based composite aerogel with a large-pore structure, favorable water swelling, and applicable mechanical properties was synthesized. KGM was blended with banana-derived cellulose nanofibers (BCNF; fiber diameter < 70 nm) from banana pseudostem and freeze-dried to form composite aerogel. As the KGM content increased from 0.08 to 0.16 wt%, porosity decreased, while mechanical properties improved: tensile strength and elongation at break increased by 96% and 23%, respectively, relative to BCNF aerogels without KGM. The composite displayed a high swelling ratio (2981%) and sustained water retention, remaining at 160% of the dry weight after 400 min of immersion. In vitro studies demonstrated antibacterial activity against E. coli and S. aureus and good biocompatibility (hemolysis rate 3.38%), with no detectable cytotoxicity (cell proliferation > 90%, CCK8) [129].
Microneedle patches hold significant promise for the treatment of chronic wounds, owing to their unique structure, which allows them to penetrate barriers and deliver drugs directly to the wound’s interior, thereby enabling timely therapeutic effects. A microneedle patch was fabricated with OKGM and hyaluronic acid as the main structural material; Cu2+ and gallic acid (GA) were co-dispersed within the matrix for deep-dermal release. The microneedle patch exhibited pronounced hygroscopicity, biodegradability, antibacterial activity, sustained release (~75% of Cu2+ over 72 h), and antioxidant activity, with no significant cytotoxicity toward L929 fibroblasts. Upon application, the released Cu2+ exhibits antibacterial activity and promotes angiogenesis, while GA acts as an antioxidant by scavengering reactive oxygen species, thereby enhancing wound repair. By day 21, microneedle-treated wounds were essentially closed, whereas diabetic controls retained approximately 18% of the original wound area. Notably, OKGM functions both as a drug carrier and a bioactive component that facilitates macrophage polarization toward the M2 phenotype. These synergistic effects collectively enhance the efficiency of wound healing [130]. However, the microneedle patch has poor adhesion and must be secured to the wound with adhesive tape or a bandage.
Electrospun nanofiber scaffolds are beneficial for embedding therapeutic agents and are commonly used in wound healing applications. To improve photothermal conversion and drug delivery efficiency, a three-layer photoactive nanofibrous patch was fabricated by a layer-by-layer assembly, using KGM and polylactic acid (PLA) as matrices, paclitaxel (PTX) as the model drug, and black rice powder (BRP) as the photothermal agent. This approach provides a high surface area and light absorption properties, advancing postoperative care by enhancing the precision of drug delivery. During annealing, starch granules within the BRP/KGM layers underwent further physical reorganization, resulting in a more structured arrangement. The incorporation of BRP within the nanofiber provides dual benefits, offering both photothermal therapy and antibacterial properties, while KGM enhances antibacterial activity and promotes tissue regeneration. The fibers, responsive to near-infrared light, facilitate tissue adhesion and trigger a synergistic effect of photothermal and chemotherapeutic actions, effectively targeting and eliminating residual tumor cells. The nanofiber’s hierarchical structure supports the controlled, sustained release of therapeutic agents, ensuring prolonged therapeutic efficacy. Moreover, the controlled release of BRP/KGM from the patch effectively inhibits tumor cell spread and reduces the risk of metastasis. Photothermal heating further induces contraction of the fiber matrix, accelerating the release rate of PTX. This photothermal nanofiber integrates biomimicry, nanotechnology, and medical science, enabling precise targeting of residual cancer cells while significantly accelerating wound healing. Additionally, enhanced mesh crimp, improved moisture absorption, increased tensile strength, and reduced hydrophilicity were observed [131].
In KGM-based wound dressings, including sponges, hydrogels, and membranes, each material serves a unique function at various stages of wound healing (e.g., hemostasis, inflammation, proliferation). Sponges effectively absorb wound exudate and promote rapid clotting. Self-adhesive and self-healing hydrogels improve ease of use. Asymmetric membranes provide enhanced mechanical barrier properties and facilitate directional water transport, effectively removing exudate while maintaining wound moisture. Films promote moisture evaporation and serve as a barrier to external contamination. Moreover, these materials can be loaded with additional therapeutic agents, further enhancing wound healing efficacy. Despite numerous preclinical investigations, few registered clinical trials specifically assess KGM-based wound dressings, and reports of commercially available KGM dressings for topical use are limited. Current KGM research remains largely at the preclinical stage, emphasizing in vitro and in vivo characterization. In contrast, existing clinical studies primarily address internal/oral indications—such as weight management and gastrointestinal health—rather than wound care.
Cellulose is the most abundant natural polysaccharide produced by plants or bacteria, which is a linear and homogeneous polysaccharide composed of D-glucose units linked by β-1,4-glycosidic bonds. Due to the good renewability, degradability, and excellent biocompatibility, cellulose can be applied in the medical field. Natural cellulose exerts its hemostatic effect mainly through physical adsorption and mechanical compression, while its hemostatic performance can be significantly improved with oxidative modification. The hydroxy groups in cellulose convert into carboxyl groups, forming oxidized cellulose. The anionic functional groups (carboxyl and aldehyde groups) on the surface of the cellulose chain not only lowers the local pH value but also interacts with proteins and metal ions in the blood to form a gel-like substance [132]. Additionally, the natural acidity of oxidized cellulose also causes blood cells to start decomposing, which in turn triggers the coagulation process. The properties of oxidized cellulose, such as biodegradability, biocompatibility, sterilization effect and physical-chemical properties, are closely related to the degree of oxidation and can be matched with the percentage of carboxyl groups introduced into the cellulose chain [133]. As an exemplary hemostatic material, cellulose-based composites present a host of remarkable attributes that render them highly suitable for a wide range of clinical requirements [134]. Their ease of manipulation, rapid clot-forming potential, and excellent biocompatibility set them apart, carving out a promising niche in hemostasis and wound healing application. This is underpinned by their customizable chemical, mechanical, and biological characteristics, as depicted in Fig. 8 [135–137].

Figure 8: Functional properties of cellulose-based composites for hemostasis and wound healing [137] [Adapted with permission from Ref. [137]. Copyright © 2025, MDPI].
Oxidation modification is an important process in the preparation of cellulose hemostatic materials. Specific modified cellulose after oxidation can significantly enhance the hemostatic efficacy of cellulose hemostatic materials. Currently, there is an important research direction to preparing cellulose containing carboxyl groups in cellulose oxidation modification. When the carboxyl content of oxidized cellulose is 0.78 mmol/g, it can dissociate in water to form highly crystalline nanofibers with a width of 5 nm and a length of no less than 2 μm [138]. Oxidized cellulose with a carboxyl content ranging from 0.981 to 1.769 mmol/g can be used to produce water-insoluble fractions [139], which could be applied in surgical treatment and plastic surgery. When applying cellulose materials to surgical hemostasis operations, their solubility mainly depends on the degree of carboxylation, and the solubility depends on the degree of carboxylation. Under normal circumstances, the higher the degree of carboxylation of cellulose materials, the better solubility of cellulose materials. Highly soluble cellulose materials can be prone to degradation in the human body and completely absorbed by the body [140]. Therefore, oxidized cellulose can be used as hemostatic materials and materials for preventing incision adhesion after surgery [141]. Table 3 below shows the representative various forms of oxidized cellulose.
Fabric-based cellulose hemostatic materials are among the most common forms in clinical applications. Such materials mainly include two types: woven structures and non-woven structures. Studies have shown that, at present, oxidized cellulose in the form of woven or non-woven textiles is the most widely used type of hemostatic material in clinical practice [133]. Woven oxidized cellulose possesses excellent toughness and operability. Woven oxidized cellulose strips can be easily cut into any size and shape. And once they react with blood, they swell and spread over the wound site. Due to the large blood contact area, which promotes the aggregation and adhesion of platelets on the fibers, the oxidized cellulose strips can form brown acidic heme thrombi. In addition, the soft yet strong cellulose fibers can adhere tightly to the wound and apply pressure to it, thereby facilitating physical and mechanical hemostasis. Non-woven oxidized cellulose exhibits superior hemostatic performance. Hutchinson et al. reported that non-woven oxidized regenerated cellulose (ORC) achieves hemostasis significantly faster than woven ORC [164], and its hemostatic effect is not affected by the mass of the material used. This performance advantage is mainly attributed to the larger specific surface area and better blood permeability of the non-woven structure. Fabric-based cellulose hemostatic materials also include various modified forms. Studies have indicated that three types of non-woven textiles, which are based on the combination of hyaluronic acid with oxidized cellulose or carboxymethyl cellulose and supplemented with etamsylate, are prepared via the wet spinning method [165]. This composite design integrates the advantages of different materials and enhances the overall hemostatic performance.
The key performance indicators of fabric-based cellulose hemostatic materials include hemostatic time, blood absorption capacity, and mechanical strength. Studies have shown that cellulose-based hemostats with different structures were evaluated in a rat partial nephrectomy model, and the results indicated that the novel sodium carboxymethyl cellulose (CMC) hemostatics required significantly less time to achieve hemostasis (p < 0.001) [166]. Blood absorption capacity is a crucial performance indicator for fabric-based materials. Research has demonstrated that the blood absorption of the original Surgicel® fabric is approximately 359.8% (i.e., absorbing about 3.6 times its own weight in blood), which was lower than that of gelatin-based sponges. But Surgicel® is suitable for small-area, low-pressure bleeding (such as capillary and venous oozing), and exhibits significant advantages especially in minimally invasive surgeries where excessive blood absorption needs to be avoided [167]. In terms of mechanical strength, fabric-based materials exhibit excellent performance. Studies have shown that Equicel® Silk features a double-layer woven structure, providing unparalleled tensile strength for challenging surgical procedures [168]. This high-strength characteristic prevents the material from being easily damaged during use, ensuring the stability of hemostatic efficacy.
Fabric-based cellulose hemostatic materials possess unique advantages in clinical applications. Studies have shown that in gynecological surgeries, hemostatic agents are most effective in areas with oozing or slow bleeding and serve as an auxiliary means to traditional surgical hemostatic methods [169]. Fabric-based materials are particularly suitable for the management of large-area bleeding wounds. Research indicates that the benefits of oxidized regenerated cellulose in vascular flap pedicle management include excellent hemostasis, bactericidal activity, and increased acidity [170]. This increased acidity promotes vasodilation and helps overcome spasms that may occur in important blood vessels deep within the pedicle. These benefits provide patients with the possibility of better outcomes and reduced morbidity [170].
As the most classic dosage form of cellulose-based hemostatic materials, a typical example of ORC is Surgicel® Original by Johnson & Johnson. ORC is an absorbable woven fabric made from regenerated cellulose through oxidation treatment. It features a loose woven structure and soft texture, enabling excellent conformity to irregular wound surfaces [132]. Surgicel® Original consists of loosely woven ORC, which provides a matrix for platelet adhesion [171]. A representative product of oxidized cellulose is Oxycel® by Parke-Davis, which is manufactured by oxidizing natural cellulose (cotton, gauze, or paper) with nitrous oxide. Oxycel® is supplied in forms such as gauze pads, cotton-type tongue depressors, or gauze tongue depressors. Surgicel® Nu-Knit is a high-density woven ORC cloth. Its dense woven structure offers enhanced mechanical strength and superior wrapping performance, making it particularly suitable for areas requiring suture fixation [172]. Surgicel® Fibrillar is a soft, lightweight layered ORC composed of loosely packed fibrils, exhibiting excellent conformability and adherence [173]. Oxidized cellulose cloth produced via non-woven technology has a larger specific surface area and faster blood absorption rate, making it commonly used in disposable hemostatic dressings. And non-woven carboxylated oxidized regenerated cellulose (CORC) exhibits better hemostatic performance than traditional gauze forms [174].
Sponge-based cellulose hemostatic materials, characterized by their unique porous structure, are one of the current research hotspots. Studies have shown that plant-derived TEMPO-oxidized nanocellulose (TOCN)/biopolymer gelatin sponges were successfully prepared via the freeze-drying method. In combination with thrombin, an endogenous blood coagulation enzyme, their application as rapid hemostatic dressings was investigated [175]. Shape memory properties are a unique advantage of certain sponge materials. A shape-adaptive composite sponge was fabricated using oxidized bacterial cellulose, low-acyl gellan gum, and hyaluronic acid via the freeze-drying method. It was then chemically crosslinked by 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide to ensure mechanical stability for hemostasis and grafted with poly(acrylic acid) to decrease its acidity. This sponge, with a pore size of 100–200 μm, promotes effective blood coagulation and the formation of stable blood clots that display fibrin networks without destroying red blood cells. It maintains its shape without fracture after 10 cyclic compression tests and can largely regain its original state. Besides, this sponge demonstrated a hemolysis rate of 0.55 ± 0.14% and could undergo rapid degradation in clinical use. However, the article does not provide specific hemostatic time data [176]. Novel hemostatic sponges with a Janus character were fabricated by conjugating oxidized bacterial cellulose (OBC) with chitosan (CS) and collagen (col). Through heterogeneous mixing and freeze-drying, the materials exhibit surfaces with distinct wettability characteristics. Specifically, during the electrostatic self-assembly process of OBC and CS, col was efficiently incorporated as a functional component through the electrostatic attraction between cationic CS and anionic OBC. The bonding between the hydrophilic and hydrophobic layers was achieved via interpenetrating chemical cross-linking between cellulose nanofibers (CNFs) and organosilanes. Research has shown that, compared to the hydrophilic layer alone, this sponge reduced hemostatic time from 165 ± 20 to 131 ± 26 s in a femoral artery injury model, and from 102 ± 21 to 83 ± 15 s in a hepatic-femoral artery injury model. Compared to hydrophilic samples and commercial gauze, the asymmetric sponge demonstrated effective bleeding control, reducing blood loss by nearly 50% in the femoral artery injury model and prolonging survival time in the carotid artery injury model [177]. Injectable hemostatic sponge is another important category of cellulose-based sponge. Studies have demonstrated the preparation of an injectable hemostatic sponge with shape memory properties, high elasticity, excellent blood absorption capacity, and antibacterial activity. This sponge was engineered using hydroxyethyl cellulose (HEC) and soy protein isolate (SPI), crosslinked via epichlorohydrin, and further modified with dopamine and silver ions. The sponge exhibited high porosity (>85%), with a recovery time of less than 13 s in blood, a high swelling rate (1650 ± 54.23%), and a volume expansion ratio of approximately 90%. The sponge demonstrated a desirable hemostatic effect, with a hemostasis time of 22.75 ± 3.86 s and blood loss of 285.25 ± 24.93 mg, which were more effective than the commercial gelatin sponge (hemostasis time 49.25 ± 3.30 s, blood loss 755.50 ± 24.45 mg). Due to the presence of silver ions (Ag+), the sponge also exhibited effective antibacterial properties against E. coli (92.17 ± 1.42%), S. aureus (84.61 ± 1.74%), and MRSA (90.94 ± 1.75%) [178].
It is worth noting that porosity, liquid absorption capacity, and excellent hemostatic efficacy are key characteristics of sponge-based cellulose hemostatic materials. Studies have indicated that the combined contributions of the freeze-drying method and silica aerogels (used as structural modifiers) led to the production of superabsorbents with a porosity of 70%, which endows the materials with significant blood cell absorption capability [179]. High porosity imparts excellent liquid absorption capacity to the materials. Research shows that agar/oxidized bacterial cellulose (OBC) composite cryogels exhibit superabsorbent properties, with a swelling degree of 4200%, enabling them to absorb large volumes of blood. Additionally, the compressive strength of the composite cryogels is significantly higher than that of pure agar, resulting in a more stable physical structure [180]. Studies have shown that in in vitro thromboelastometry studies, CNFs in gel and sponge forms reduced clotting time by 68 ± 2% and 88 ± 5%, respectively. In an in vivo mouse liver injury model, CNFs sponge significantly reduced blood loss by 38 ± 10% [181]. Their performance in terms of clotting time is particularly prominent. Research indicates that TEMPO-oxidized nanocellulose sponges exhibit excellent hemostatic efficacy, rapid absorbability, and a minimal clotting time (1.37 ± 0.152 min). Moreover, they ensured reduced blood loss in a rat liver biopsy puncture model [175]. Biocompatibility evaluations have yielded favorable results. Studies have shown that neutral-pH CNFs do not damage red blood cells nor hinder the proliferation of fibroblasts or endothelial cells. Histological examination of subcutaneously implanted CNFs revealed that the foreign body reaction subsided as the CNFs degraded, with no scar formation in the skin after 8 weeks [180].
Foams are prepared through foaming processes, featuring a porous structure that allows rapid absorption of large volumes of blood. Fabricated by foaming and solidifying cellulose solutions, it contains numerous interconnected pores and can quickly absorb blood up to dozens of times its own weight. A newly developed superabsorbent (NDS) is synthesized via chemical and physical cross-linking methods. It not only rapidly absorbs large amounts of blood components (60 g/g) but also strongly adheres (~90 KPa) to damaged tissues [182]. Oxidized cellulose foam has larger pores and better elasticity, making it suitable for cavity bleeding (e.g., in the oral cavity and nasal cavity). The advantage of the foam structure lies in its high porosity (up to 70%), which enables significant blood cell absorption capacity [183].
Powdered cellulose hemostatic materials are a category of products that have developed rapidly in recent years. Studies have shown that the newly developed oxidized regenerated cellulose (ORC) powder consists of the potassium salt form of ORC (KORC) and the sodium salt form of ORC (NaORC), which are prepared via NO2-mediated oxidation. Both NaORC and KORC exhibit bactericidal effects against Staphylococcus aureus and demonstrate rapid blood coagulation in in vivo studies on hemostatic activity [184]. Another important type of powdered material is powdered oxidized regenerated cellulose, which is prepared from fibrous structural aggregates that produce ORC microfibers. This design enables it to work effectively over a large surface area in moist regions and endows it with the ability to form adherent and long-lasting thrombi [185]. Microporous polysaccharide hemispheres (MPH) and Per Clot are microporous polysaccharide hemostatic products prepared by extracting from plant starch and subjecting it to further processing. MPH, which contains no polypeptides or proteins, not only rapidly dehydrates blood to form thrombi (thus preventing blood oozing) but also undergoes rapid degradation and absorption in vivo [186]. Micron-sized spherical particles prepared via special processes. They have a large specific surface area and good fluidity and can be injected via a syringe into deep small vessel bleeding sites, avoiding surgical incision and reducing trauma.
Powdered cellulose hemostatic materials possess unique performance advantages. Studies have shown that oxidized regenerated cellulose powder is the most effective hemostatic agent, and the Surgicel® applicator exhibits the highest performance compared with the three starch-based devices under investigation. The efficacy rate of Surgicel® is not inferior to (p ≤ 0.0002) and even superior to (p ≤ 0.004) any of the three starch-based agents, regardless of whether the powder is delivered via its dedicated device or applied directly to the bleeding site. Surgicel® requires fewer applications (p ≤ 0.0002) and a smaller amount of powder (p < 0.0001) to achieve hemostasis. The mechanism of action of powdered materials is mainly based on their high specific surface area and rapid dispersibility. Research indicates that after the first application of the product, hemostasis was achieved in 63% of the sites treated with Surgicel®, while only 21%, 25%, and 13% of the sites treated with AristaTM, PerClot®, and 4DryField® (respectively) achieved successful results. After four consecutive applications, 100% of the Surgicel-treated sites achieved hemostasis, compared with 67%, 71%, and 21% of the sites treated with AristaTM, PerClot®, and 4DryField® (respectively) [185]. Agglomerated granules are the latest generation of cellulose-based hemostatic products, a representative example is Surgicel® Agglomerated Granules by Johnson & Johnson. It is the first domestic granular regenerated oxidized cellulose hemostatic product, approved by the National Medical Products Administration (NMPA) in 2022. This product comprises hemostatic granules formed by compacting fine fibers through a patented process. It includes a 3-g prefilled application device for open surgery and a matching endoscopic application catheter device for endoscopic surgery [187].
Powdered cellulose hemostatic materials have unique advantages in specific clinical scenarios. Studies have shown that the Surgicel® device could treat an average of 9.1 bleeding sites with one 3g device, while the AristaTM, PerClot®, and 4DryField® devices could treat 1.9, 2.2, and 1.2 sites (respectively) with the same weight of device. Oxidized regenerated cellulose powder is available in 3-g packaging and can be used for hard-to-reach bleeding sites [185], such as Surgicel® Powder, an absorbable hemostatic powder made from ORC. Powdered ORC consists of aggregates of fine ORC fibers, providing additional valuable hemostatic properties while retaining the biochemical and bactericidal characteristics of the parent ORC fabric. Powdered materials are particularly suitable for hemostasis in hard-to-reach areas and irregular wounds. Due to their powdery nature, they can be accurately delivered to the bleeding site via various delivery devices to achieve local hemostasis. This characteristic endows powdered materials with important application value in minimally invasive surgeries and endoscopic surgeries [188].
Under the current developmental landscape, the composite modification of cellulose with functional materials can further enhance the inherent advantages of cellulose while imparting multifunctional properties such as hemostatic efficacy, antibacterial activity, and wound healing promotion. Through methods including physical blending, chemical grafting, cross-linking polymerization, or in-situ synthesis, diverse functional materials are rationally integrated with cellulose to achieve synergistic interactions among components. For example, incorporating antibacterial agents—such as silver nanoparticles, chitosan, or plant extracts—enables the resulting composite to effectively inhibit the growth and proliferation of pathogenic bacteria, including Escherichia coli and Staphylococcus aureus, thereby addressing the risk of bacterial infection commonly encountered in wound care and medical device applications. The integration of hemostatic agents (e.g., zeolite, gelatin, or polyvinylpyrrolidone) enhances the material’s capacity for rapid blood absorption, platelet activation, and accelerated clot formation, making it suitable for emergency hemostasis in trauma management, surgical procedures, and similar settings. Furthermore, the incorporation of bioactive components such as growth factors, hyaluronic acid, or collagen helps regulate the tissue repair microenvironment, promote skin cell proliferation and differentiation, and facilitate the regeneration of damaged tissues, thereby supporting wound healing. Beyond these core functionalities, cellulose-based composites can be engineered to exhibit additional tailored properties—such as moisture retention, breathability, enhanced mechanical strength, or pH responsiveness—according to specific application requirements. This multi-functional design enables modified cellulose materials to hold significant promise across a wide range of fields, including healthcare (e.g., antimicrobial wound dressings, hemostatic sponges, tissue engineering scaffolds), consumer products (e.g., antibacterial textiles, skincare formulations), and environmental protection (e.g., antimicrobial water purification materials).
Cellulose hemostatic materials in the form of nanofibers represent the cutting-edge of technological development. Oxidized cellulose nanofibers are materials with a nanofiber-scale structure, which can be fabricated into fiber-like membrane materials or sponge-like porous materials through different preparation processes. Studies have shown that non-oxidized cellulose nanofibers (CNFs) are synthesized via ball milling. In vitro thromboelastometry studies indicate that there exists an optimal balance between the aspect ratio (AR) and specific surface area (SSA) of nanofibers to maximize the promotion of platelet function and plasma coagulation [189]. Optimized CNFs possess a high SSA (17 m²/g) and a high aspect ratio, which shorten the normal whole blood clotting time by 68%—outperforming cellulose-based hemostatic agents. Furthermore, CNFs reduce the clotting time by 80% in platelet-deficient blood and by 54% in heparinized blood [178]. TEMPO-oxidized cellulose nanofibers (TOCNs) are another important nanofiber. Research has demonstrated that the combination of TOCNs with silk fibroin (SF) significantly (p < 0.001) enhances blood absorption capacity and improves the biocompatibility of TOCNs. Thrombin loading enhances the platelet activation and hemostatic performance of the scaffold (TOCN-SF-Th) compared to samples without thrombin. In a rabbit ear artery bleeding model, the hemostatic time of TOCN-SF5-Th was reduced (p < 0.001) from 220 s (for TOCN-SF5) to 114 s [190].
Cellulose hemostatic materials with a hydrogel morphology feature a unique three-dimensional network structure. Studies have shown that hydrogel biomaterials engineered from naturally derived polysaccharides and proteins have gained increasing exploration in preclinical research over the past few years, benefiting from their biocompatibility, degradability, injectability, tunable mechanical properties, and potential ability to promote coagulation, wound healing, and anti-infection [191]. Ca²+-crosslinked nanofiber hydrogels are an important hydrogel morphology. Research indicates that in this work, Ca²+-crosslinked nanofibrillated cellulose hydrogels were investigated as potential hemostatic wound dressings by studying the core interactions between the material and blood—the key component of wounds and wound healing [192]. Evaluations of thrombin and factor XIIa formation, platelet reduction, and the release of complement system activation proteins showed that NFC hydrogels effectively trigger blood coagulation, initiating clot formation rapidly while demonstrating basal complement system activation.
To comprehensively compare the performance of cellulose-based hemostatic materials with different morphologies, the following Table 4 summarizes the comparison of key performance indicators. As can be seen, cellulose-based hemostatic materials with different morphologies each have their own advantages and applicable scenarios. Powder-type materials perform best in terms of hemostatic speed, while sponge-type materials have a significant advantage in blood absorption capacity. Nanofiber-type materials exhibit the highest hemostatic efficiency, but their preparation process is relatively complex. Although fabric-type materials have a longer coagulation time, they have unique advantages in operational convenience and large-area application.

5 Future Trends and Remaining Challenges
Polysaccharides such as chitosan, KGM, and cellulose possess inherent biocompatibility, biodegradability, non-toxicity, and bioactivity, which makes them highly suitable for use in wound dressing fabrication (Table 1). Each polysaccharide also has unique properties. For example, chitosan is a cationic polysaccharide known for its antibacterial and hemostatic effects, while KGM demonstrates strong water-absorbing and gelling properties. However, some inherent characteristics, such as the high viscosity and gelling characteristics of KGM, as well as the insolubility and physicochemical stability of cellulose, pose challenges for the preparation of wound dressings. These limitations can be addressed through structural modifications, which not only enhance their wound healing potential but also facilitate the development of advanced healthcare materials. There has been remarkable progress in advancements in structural modification of these polymer. Beyond common techniques like deacetylation, oxidation, carboxymethylation, quaternization, and ozonation, a host of novel modifications—including methacrylated oxidized KGM, tannic acid modified KGM, lipoic acid-grafted chitosan, phenylboronic acid modified chitosan, catechol-grafted chitosan, decanoic acid-modified chitosan, cationic modified cellulose, quaternized hydroxyethyl cellulose, and oxidized carboxymethyl cellulose—have emerged, significantly elevating the specific performance of wound dressings.
Diverse wound dressings can be fabricated through various bonding mechanisms—such as physical crosslinking, chemical crosslinking, or a combination of both—to meet specific performance requirements. In the case of chemical crosslinking, crosslinkers are essential. However, despite thorough washing with distilled water to remove residual chemicals, it is crucial to select non-toxic crosslinkers to ensure the biocompatibility of the final product and environmentally responsible production. Researchers are exploring sustainable preparation methods by using natural, non-toxic substances—such as tannic acid [40], genipin [61], and citric acid [193]—as crosslinking agents. Additionally, photocrosslinking techniques leverage light-activated intermediates to form covalent bonds within polysaccharide molecules. This photochemical approach offers precise control over modification sites and substitution degrees, while eliminating the need for hazardous catalysts or solvents as crosslinker. The development of these green and efficient modification techniques has significantly enhanced the key physical properties of wound healing materials, as reported in studies on materials based on chitosan, KGM, and cellulose.
An increasing number of novel wound dressings have been developed to combine the strengths of diverse dressing types to enhance overall performance of wound dressing and positively influence the healing process. A particularly promising avenue involves multi-phase combination. By integrating diverse material forms—such as hydrogels and nanofibrous membranes—this approach enables the development of wound dressings with exceptional mechanical properties, sustained release, adhesion, antibacterial activity. Moreover, multilayer materials—including hydrogel microspheres [194], hydrogels [195], nanofiber-based membranes [82,196], and films [197]—synergistically combine the distinct wound-healing advantages of each layer and allow for the directed transfer of liquids, especially for multilayer membranes [124]. These advancements significantly improve both the biological performance and processing adaptability of wound dressings; research in this field is poised to advance further.
Wound healing involves four complex, interconnected stages—hemostasis, inflammation, proliferation, and remodeling. Chitosan has a beneficial effect on all four stages, promoting optimal healing at each phase. In contrast, KGM mainly supports the inflammation and proliferation stages. Cellulose contributes primarily to hemostasis, inflammation, and proliferation. Moreover, chitosan’s antibacterial properties provide significant advantages in the healing of infected wounds. When wound dressings are designed to align with the dynamic needs of each healing phase, they can notably accelerate the recovery process. However, despite these advancements, no dressing has yet been developed that fully meets the requirements of each stage of wound healing. Most studies concentrate on specific stages, while recent research on ‘smart’ wound dressings increasingly emphasizes systems that span multiple stages and respond to factors such as pH, reactive oxygen species (ROS), temperature, light, bacteria, glucose, and their combinations [198,199]. These innovative dressings appear in various forms, including hydrogels, fabrics, scaffolds, and nanofiber-based mats. Such stimuli-responsive systems react to changes at the wound site, enhance microenvironment sensing capabilities, alter the dressing’s physical or chemical properties, and enable precise control over the healing process. Due to the wide variability in patient conditions and wound types, a single universal wound dressing may not be suitable for all cases. Therefore, a promising future direction—distinct from multifunctional integration—is the development of personalized dressings tailored to specific clinical needs. The advancement of 3D printing and artificial intelligence plays a crucial role in realizing this objective. 3D printing delivers the precise fabrication of patient-specific dressings that fit precisely to the wound bed [200]. This technology facilitates the incorporation of hemostatic agents, antimicrobials, and other therapeutics, enabling their controlled release [201]. While numerous studies document 3D-printed dressings utilizing chitosan and cellulose, research on KGM-based systems remains comparatively scarce. Artificial intelligence is driving a shift from passive dressings to smart, bioelectronic wound-care platforms. Recent studies have integrated miniaturized sensors for pH, temperature, and inflammatory biomarkers into wearable dressings, alongside machine learning algorithms, enabling continuous monitoring and prediction of healing trajectories [202–205]. Deep-learning tools for virtual histopathology and automated image analytics provide real-time data feedback for materials optimization [206,207]. Smart wearable systems that enable on-demand drug release provide more precise regulation of wound repair [208]. However, the design and fabrication of AI-enabled wound dressings, particularly those incorporating chitosan, KGM, and cellulose—remain in the early stages, combining polymer chemistry, material science, and clinical medicine. Future advancements will leverage AI-driven inverse design and multiobjective optimization to create multifunctional, controllable dressings with visualizable healing statuses, adaptive release profiles, and seamless human–machine interfaces, thereby accelerating the transition to personalized wound care.
Despite their potential, these polysaccharides face several limitations in wound healing application, including insufficient mechanical strength and significant material variability. Chitosan exhibits batch-to-batch variations in acetylation degree, molecular weight, and viscosity, along with susceptibility to pH-triggered degradation, limited exudate absorption, and a tendency to desiccate. Its antibacterial effectiveness also varies across different wound environments. KGM is characterized by variations in degree of substitution and acetylation ratio, and residual reagents from chemical modification or crosslinking may pose toxicity concerns. Cellulose, while possessing high crystallinity, lacks inherent antibacterial activity and requires modification to enhance its properties. These challenges result in inconsistencies in performance, both in terms of stability and batch-to-batch variation. Furthermore, manufacturing scalability poses challenges, as the complexity of the process, especially when integrating sensitive bioactives or multilayer heterostructures, raises costs and undermines reproducibility. An additional challenge arises from the heterogeneity of test metrics across different types of wound dressing. Each characterization—mechanical property, antimicrobial activity, cytocompatibility/biocompatibility, and hemostasis—can be assessed using non-equivalent assays (for example, tensile strength, adhesion strength, or modulus for hydrogels), each of which is sensitive to different factors. Harmonization—not strict unification—is needed: function- and format-specific core assay sets with minimum reporting standards to enable meaningful cross-study comparisons. Finally, there is a need for long-term assessments of biocompatibility, degradation products, immunogenicity, and systemic exposure—especially for composites with inorganic phases or nanomaterials—together with models that better recapitulate human wound conditions (e.g., infected wounds, realistic exudate and external force).
Chitosan, konjac glucomannan, and cellulose are three widely utilized natural polysaccharides recognized for their favorable physiological functions, biocompatibility, safety, and availability. These polysaccharides have found extensive applications in food preservation, drug delivery, and skin wound healing. This review provides an overview of recent advancements in the utilizing these polysaccharides for wound dressings, focusing on how the selection of components and interaction mechanisms influence physical properties, physiological functions, and therapeutic effectiveness, as well as the advantages and disadvantages of various methods for preparing materials of the same type using the same polysaccharide. The results demonstrate that, through diverse preparation techniques, these wound dressings exhibit a range of physicochemical properties while effectively modulating the wound microenvironment to promote healing. Key functions include maintaining moisture balance, preserving structural integrity, managing exudate, and delivering bioactive functions such as antimicrobial, antioxidant, anti-inflammatory, hemostatic, pro-angiogenic, re-epithelialization, proliferative, and immunomodulatory activities. In addition, some chitosan- and cellulose-based dressings, already applied in clinical settings, have demonstrated superior performance. These results highlight the potential of these polysaccharides in wound healing and emphasize the improtance of ongoing innovation.
Acknowledgement: The authors wish to express their sincere gratitude to Professor Rongqiang Zhang for his valuable suggestions on the manuscript, to Mr. Zhe Wang for his unwavering support throughout the writing of the paper, and to Professor Chusheng Qi for his timely assistance, which enabled the smooth completion of the manuscript revision.
Funding Statement: This study was funded by the Basic Research Project of the Shaanxi Provincial Department of Science and Technology (2025JC-YBMS-1045), Qinchuangyuan Traditional Chinese Medicine Industry Innovation Cluster Project (L2024-QCY-ZYYJJQ-X174, L2024-QCY-ZYYJJQ-X178, L2024-QCY-ZYYJJQ-X63), Special Research Program of the Shaanxi Provincial Department of Education (21JK0600), Undergraduate Training Program for Innovation and Entrepreneurship (202410716026, S202410716126).
Author Contributions: The authors confirm contribution to the paper as follows: Conceptualization, Jin Shang and Liangliang Duan; methodology, Peng Zhao and Weimin Zhang; software, Landuo Zhang and Xiangwen Li; formal analysis, Jin Shang and Weimin Zhang; data curation, Peng Zhao and Xiangwen Li; writing—original draft preparation, Jin Shang and Weimin Zhang; writing—review and editing, Weimin Zhang, Qi Wang and Liangliang Duan; visualization, Landuo Zhang and Qi Wang; project administration, Jin Shang; funding acquisition, Liangliang Duan. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The authors confirm that the data supporting the findings of this study are available within the article.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
References
1. Zhang Y, Qiao N, Liu L, Shang H, Wei D, Ji Z, et al. Advances in the study of polysaccharide-based hydrogel wound dressings. Int J Biol Macromol. 2025;307:142134. doi:10.1016/j.ijbiomac.2025.142134. [Google Scholar] [PubMed] [CrossRef]
2. Vessally E, Rovnag R. Recent advances in polysaccharide-based nanocomposites as wound healing and transdermal drug delivery systems: a review. Int J Biol Macromol. 2025;321(9):144743. doi:10.1016/j.ijbiomac.2025.144743. [Google Scholar] [PubMed] [CrossRef]
3. Dang ZX, Zhang H, Liu QX, Xu WQ, Wang ZY, Wang CY, et al. Silk fibroin-based wound dressings: advances toward smart trauma care platforms. Sens Actuators A Phys. 2025;393:116793. doi:10.1016/j.sna.2025.116793. [Google Scholar] [CrossRef]
4. Liang Y, He J, Guo B. Functional hydrogels as wound dressing to enhance wound healing. ACS Nano. 2021;15(8):12687–722. doi:10.1021/acsnano.1c04206. [Google Scholar] [PubMed] [CrossRef]
5. de Maria Aguiar Carvalho C, da Silva BB, Brianezi SFS, Sanfelice RC, Balogh DT, Assis L, et al. Chitosan-based structures for skin repair: a literature review. Int J Biol Macromol. 2025;306:141426. doi:10.1016/j.ijbiomac.2025.141426. [Google Scholar] [PubMed] [CrossRef]
6. Mazurek Ł., Kuś M, Jurak J, Rybka M, Kuczeriszka M, Stradczuk-Mazurek M, et al. Biomedical potential of alginate wound dressings—from preclinical studies to clinical applications: a review. Int J Biol Macromol. 2025;309:142908. doi:10.1016/j.ijbiomac.2025.142908. [Google Scholar] [PubMed] [CrossRef]
7. Zhang W, Liu Y, Zhang L, Shen X. Development of hyaluronic acid-based hydrogels for chronic diabetic wound healing: a review. Int J Biol Macromol. 2025;308(9):142273. doi:10.1016/j.ijbiomac.2025.142273. [Google Scholar] [PubMed] [CrossRef]
8. Pita-Vilar M, Concheiro A, Alvarez-Lorenzo C, Diaz-Gomez L. Recent advances in 3D printed cellulose-based wound dressings: a review on in vitro and in vivo achievements. Carbohydr Polym. 2023;321(17):121298. doi:10.1016/j.carbpol.2023.121298. [Google Scholar] [PubMed] [CrossRef]
9. Khatoon F, Narula AK, Sehgal P. Efficacy of collagen based biomaterials in diabetic foot ulcer wound healing. Eur Polym J. 2024;217(9):113345. doi:10.1016/j.eurpolymj.2024.113345. [Google Scholar] [CrossRef]
10. Wang Z, Sun Q, Zhang H, Wang J, Fu Q, Qiao H, et al. Insight into antibacterial mechanism of polysaccharides: a review. LET. 2021;150(2):111929. doi:10.1016/j.lwt.2021.111929. [Google Scholar] [CrossRef]
11. He X, Liu L, Gu F, Huang R, Liu L, Nian Y, et al. Exploration of the anti-inflammatory, analgesic, and wound healing activities of Bletilla Striata polysaccharide. Int J Biol Macromol. 2024;261(2):129874. doi:10.1016/j.ijbiomac.2024.129874. [Google Scholar] [PubMed] [CrossRef]
12. Zhang X, Wu Y, Gong H, Xiong Y, Chen Y, Li L, et al. A multifunctional herb-derived glycopeptide hydrogel for chronic wound healing. Small. 2024;20:2400516. doi:10.1002/smll.202400516. [Google Scholar] [PubMed] [CrossRef]
13. Xu G, Cao S, Huang Z, Dong Y, Chu C. Catechol-polysaccharide complex enhancing the detachment and blood coagulation of polyvinyl alcohol/chitosan cryogel for non-compressible bleeding. Int J Biol Macromol. 2025;318(22):145281. doi:10.1016/j.ijbiomac.2025.145281. [Google Scholar] [PubMed] [CrossRef]
14. Liu Y, Hu Y, Wang B, Wei Y, Huang D. Multifunctional cellulose based hemostatic materials: from hemostasis to multi-field applications. Carbohydr Polym. 2025;366(1):123895. doi:10.1016/j.carbpol.2025.123895. [Google Scholar] [PubMed] [CrossRef]
15. Li W. Applications of chitosan-based hydrogels in diabetic wound healing: a review. Int J Biol Macromol. 2025;324(4):147264. doi:10.1016/j.ijbiomac.2025.147264. [Google Scholar] [PubMed] [CrossRef]
16. Shi G, Hong Y, Cheng L, Li L, Gu Z. Unlocking the potential of konjac (Amorphophallus konjac K. Koch) starch: a review on extraction, structure, properties, and potential applications. Food Hydrocoll. 2026;170(2):111659. doi:10.1016/j.foodhyd.2025.111659. [Google Scholar] [CrossRef]
17. Zhou L, Xu T, Yan J, Li X, Xie Y, Chen H. Fabrication and characterization of matrine-loaded konjac glucomannan/fish gelatin composite hydrogel as antimicrobial wound dressing. Food Hydrocoll. 2020;104(5):105702. doi:10.1016/j.foodhyd.2020.105702. [Google Scholar] [CrossRef]
18. Zhuang K, Shu X, Xie W. Konjac glucomannan-based composite materials: construction, biomedical applications, and prospects. Carbohydr Polym. 2024;344(12):122503. doi:10.1016/j.carbpol.2024.122503. [Google Scholar] [PubMed] [CrossRef]
19. Li H, Liang X, Chen Y, Liu K, Fu X, Zhang C, et al. Synergy of antioxidant and M2 polarization in polyphenol-modified konjac glucomannan dressing for remodeling wound healing microenvironment. Bioeng Transl Med. 2023;8(2):e10398. doi:10.1002/btm2.10398. [Google Scholar] [PubMed] [CrossRef]
20. Pan X, Zong Q, Liu C, Wu H, Fu B, Wang Y, et al. Konjac glucomannan exerts regulatory effects on macrophages and its applications in biomedical engineering. Carbohydr Polym. 2024;345(1):122571. doi:10.1016/j.carbpol.2024.122571. [Google Scholar] [PubMed] [CrossRef]
21. Chen G, Xun X, Ao H, Chen Z, Wang D, Wang M, et al. Quaternized chitosan-based injectable self-healing hydrogel for improving wound management in aging populations. Colloids Surf B Biointerfaces. 2025;253(Sup6):114721. doi:10.1016/j.colsurfb.2025.114721. [Google Scholar] [PubMed] [CrossRef]
22. Wu SH, Rethi L, Pan WY, Nguyen HT, Chuang AE-Y. Emerging horizons and prospects of polysaccharide-constructed gels in the realm of wound healing. Colloids Surf B Biointerfaces. 2024;235(1):113759. doi:10.1016/j.colsurfb.2024.113759. [Google Scholar] [PubMed] [CrossRef]
23. Ghattas M, Dwivedi G, Chevrier A, Horn-Bourque D, Alameh M-G, Lavertu M. Chitosan immunomodulation: insights into mechanisms of action on immune cells and signaling pathways. RSC Adv. 2025;15(2):896–909. doi:10.1039/D4RA08406C. [Google Scholar] [PubMed] [CrossRef]
24. Sarkar S, Manna S, Jana P, Das E, Sahu R, Dua TK, et al. Applications of chitosan as a wound dressing material: an updated review. Int J Polym Mater Polym Biomater. 2025;1–23(3):262–84. doi:10.1080/00914037.2025.2521836. [Google Scholar] [CrossRef]
25. Darie-Niţă RN, Frąckowiak S. An overview of potential applications of environmentally friendly hybrid polymeric materials. Polymers. 2025;17(2):252. doi:10.3390/polym17020252. [Google Scholar] [PubMed] [CrossRef]
26. Haririan Y, Elahi A, Shadman-Manesh V, Rezaei H, Mohammadi M, Asefnejad A. Advanced nanostructured biomaterials for accelerated wound healing: insights into biological interactions and therapeutic innovations: a comprehensive review. Mater Des. 2025;258:114698. doi:10.1016/j.matdes.2025.114698. [Google Scholar] [CrossRef]
27. Kumaravel A, Lin SP, Santoso SP, Shanmugasundaram S, Hsu HY, Hsieh CW, et al. Unlocking the potential of bacterial cellulose: synthesis, functionalization, and industrial impact. Int J Biol Macromol. 2025;311(2):143951. doi:10.1016/j.ijbiomac.2025.143951. [Google Scholar] [PubMed] [CrossRef]
28. Wu M, Ding L, Bai X, Cao Y, Rahmaninia M, Li B, et al. Cellulose-based suture: state of art, challenge, and future outlook. J Bioresour Bioprod. 2025;10(3):295–309. doi:10.1016/j.jobab.2024.11.006. [Google Scholar] [CrossRef]
29. Putra PSU, Adhika DR, Asri LATW, Suprijadi S. Performance enhancement of chitosan for food packaging: impact of additives and nanotechnology. J Renew Mater. 2025;13(6):1043–1070. doi:10.32604/jrm.2025.02024-0002. [Google Scholar] [CrossRef]
30. Naveedunissa S, Meenalotchani R, Manisha M, Ankul Singh S, Nirenjen S, Anitha K, et al. Advances in chitosan based nanocarriers for targetted wound healing therapies: a review. Carbohydr Polym Technol Appl. 2025;11:100891. doi:10.1016/j.carpta.2025.100891. [Google Scholar] [CrossRef]
31. Zhao J, Qiu P, Wang Y, Wang Y, Zhou J, Zhang B, et al. Chitosan-based hydrogel wound dressing: from mechanism to applications, a review. Int J Biol Macromol. 2023;244(4):125250. doi:10.1016/j.ijbiomac.2023.125250. [Google Scholar] [PubMed] [CrossRef]
32. El-Saadony MT, Saad AM, Alkafaas SS, Dladla M, Ghosh S, Elkafas SS, et al. Chitosan, derivatives, and its nanoparticles: preparation, physicochemical properties, biological activities, and biomedical applications—a comprehensive review. Int J Biol Macromol. 2025;313(PtB):142832. doi:10.1016/j.ijbiomac.2025.142832. [Google Scholar] [PubMed] [CrossRef]
33. Ma C, Dou Y, Li R, Zhang L, Zhou Z, Guo S, et al. Carboxymethyl chitosan/polyacrylamide double network hydrogels based on hydrogen bond cross-linking as potential wound dressings for skin repair. Int J Biol Macromol. 2024;280(1):135735. doi:10.1016/j.ijbiomac.2024.135735. [Google Scholar] [PubMed] [CrossRef]
34. Cai C, Wang T, Han X, Yang S, Lai C, Yuan T, et al. In situ wound sprayable double-network hydrogel: preparation and characterization. Chin Chem Lett. 2022;33(4):1963–9. doi:10.1016/j.cclet.2021.11.047. [Google Scholar] [CrossRef]
35. Shi X, Fang Q, Ding M, Wu J, Ye F, Lv Z, et al. Microspheres of carboxymethyl chitosan, sodium alginate and collagen for a novel hemostatic in vitro study. J Biomater Appl. 2016;30(7):1092–102. doi:10.1177/0885328215618354. [Google Scholar] [PubMed] [CrossRef]
36. Zou Y, Yang Y, Pei J, Sun P, Wang Y. Ganoderma lucidum polysaccharide/carboxymethyl chitosan hydrogels modulate macrophage polarization for wound healing. Biomacromolecules. 2025;26(4):2675–89. doi:10.1021/acs.biomac.5c00112. [Google Scholar] [PubMed] [CrossRef]
37. Li F, Liu T, Liu X, Han C, Li L, Zhang Q, et al. Ganoderma lucidum polysaccharide hydrogel accelerates diabetic wound healing by regulating macrophage polarization. Int J Biol Macromol. 2024;260(22):129682. doi:10.1016/j.ijbiomac.2024.129682. [Google Scholar] [PubMed] [CrossRef]
38. Zhang X, Li R, Li S, Cui W, Wang D, Zhu Y, et al. Tri-network PVA/chitosan/gelatin hydrogel modified by tannic acid with self-healing, adhesive and anti-inflammatory properties to accelerate wound healing. Int J Biol Macromol. 2025;308(2):142280. doi:10.1016/j.ijbiomac.2025.142280. [Google Scholar] [PubMed] [CrossRef]
39. Huang Q, Wu T, Guo Y, Wang L, Yu X, Zhu B, et al. Platelet-rich plasma-loaded bioactive chitosan@sodium alginate@gelatin shell-core fibrous hydrogels with enhanced sustained release of growth factors for diabetic foot ulcer healing. Int J Biol Macromol. 2023;234:123722. doi:10.1016/j.ijbiomac.2023.123722. [Google Scholar] [PubMed] [CrossRef]
40. Hao Y, Wang J, Zhang H, Liu Q, Wang X, Wei Y, et al. Konjac glucomannan/Bletilla striata polysaccharide composite hydrogel: a promising anti-inflammatory dressing for accelerated wound healing. Carbohydr Polym. 2025;361(11):123639. doi:10.1016/j.carbpol.2025.123639. [Google Scholar] [PubMed] [CrossRef]
41. Pan X, Zong Q, Fu B, Wang Y, Feng X, Sun W, et al. The preparation of methacrylated oxidized konjac glucomannan hydrogel system and its treatment for diabetic wounds. Int J Biol Macromol. 2025;298(10):140005. doi:10.1016/j.ijbiomac.2025.140005. [Google Scholar] [PubMed] [CrossRef]
42. Zhao Z, Wang Y, Jia L, Wei Q, Zhang W, Hu Z, et al. An antioxidant, antibacterial, and immunoregulatory konjac glucomannan-based nanocomposite hydrogel for promoting skin wound healing. Int J Biol Macromol. 2025;307(33):141791. doi:10.1016/j.ijbiomac.2025.141791. [Google Scholar] [PubMed] [CrossRef]
43. Ni J, Dong X, Wang H, Zhang Y, Xu J, Wang T. Oxidized regenerated cellulose-based supermolecular hydrogel for controlled drug release: integrating cyclodextrin-mediated hydrophobic drug distribution, wound-specific degradation, and multifunctional therapeutic effects. Int J Biol Macromol. 2025;310(7):143445. doi:10.1016/j.ijbiomac.2025.143445. [Google Scholar] [PubMed] [CrossRef]
44. Yu H, Chen D, Lu W, Zhang C, Wang H, Peng Z, et al. Characterization of polyvinyl alcohol/chitosan nanofibers loaded with royal jelly by blending electrospinning for potential wound dressings. Int J Biol Macromol. 2025;307(9):141977. doi:10.1016/j.ijbiomac.2025.141977. [Google Scholar] [PubMed] [CrossRef]
45. Jiang T, Wang D, Zhang X, Yang Q, Huang Q, Ju X, et al. Electrospinning of chitosan/polyvinyl alcohol pickering emulsion with tea tree essential oil loaded for anti-infection wound dressings. Mater Chem Phys. 2024;311:128561. doi:10.1016/j.matchemphys.2023.128561. [Google Scholar] [CrossRef]
46. Zhang Y, Li X, Chen H, Xie X, Li C, Hou G, et al. Acetylated yeast mannan endowing biomimetic electrospun nanofibers with hydrophobicity and moisturizing property for accelerated cutaneous defects repair. Int J Biol Macromol. 2025;320(27):145944. doi:10.1016/j.ijbiomac.2025.145944. [Google Scholar] [PubMed] [CrossRef]
47. Hou S, Gou L, Li F, Li C, Yin X, Zhao P, et al. A multifunctional bacterial cellulose wound dressing based on cotton fabric for infected wound healing. Int J Biol Macromol. 2025;322:146850. doi:10.1016/j.ijbiomac.2025.146850. [Google Scholar] [PubMed] [CrossRef]
48. Zheng J, Li K, Li Y, Jiang G. Preparation and characterization of carboxymethyl chitosan/κ-carrageenan/silver nanoparticles sponge for wound dressing. Mater Today Commun. 2023;35(10):105818. doi:10.1016/j.mtcomm.2023.105818. [Google Scholar] [CrossRef]
49. Han J, Lv X, Hou Y, Yu H, Sun Y, Cui R, et al. Multifunctional hemostatic polysaccharide-based sponge enhanced by tunicate cellulose: a promising approach for photothermal antibacterial activity and accelerated wound healing. Int J Biol Macromol. 2023;251(11):126386. doi:10.1016/j.ijbiomac.2023.126386. [Google Scholar] [PubMed] [CrossRef]
50. Zhang H, Tian W, Zhou J, Miao L, Li M, Chen W, et al. Bacterial cellulose cross-linked MXene composite antimicrobial hemostatic sponges with photothermal properties for wound healing. Mater Today Chem. 2025;43(1):102520. doi:10.1016/j.mtchem.2025.102520. [Google Scholar] [CrossRef]
51. Liu L, Hu G, Chen L, Hong FF. A microporous hemostatic and antibacterial multifunctional composite foam wound dressing derived from oxidized bacterial cellulose. Int J Biol Macromol. 2025;327(8):146928. doi:10.1016/j.ijbiomac.2025.146928. [Google Scholar] [PubMed] [CrossRef]
52. Wang X, Zhang J, Shan J, Yang D, Tian G, Dang Y, et al. In-situ loading of Ag nanoparticles and sodium cefoperazone onto bacterial cellulose-based composite antimicrobial films for promoting wound healing. Int J Biol Macromol. 2025;322(33):146707. doi:10.1016/j.ijbiomac.2025.146707. [Google Scholar] [PubMed] [CrossRef]
53. Borse S, Nangare S, Bafna P, Pankaj J, Laxmikant Z. Barbaloin loaded chitosan-gum kondagogu polyelectrolyte complex based biocomposites film for enhanced antibacterial, antioxidant and wound healing activity. Surf Interf. 2025;56(1):105506. doi:10.1016/j.surfin.2024.105506. [Google Scholar] [CrossRef]
54. Beata K, Lidia Z, Ugo DA, Anna P, Anna M, Anna R, et al. Bioactivation of konjac glucomannan films by tannic acid and gluconolactone addition. ACS Appl Mater Interfaces. 2024;16(35):46103–112. doi:10.1021/acsami.4c09909. [Google Scholar] [PubMed] [CrossRef]
55. Silla L, Bethwel T, Lydia K, Thomas K, Yusufu J. Development of a banana stem native cellulose and pine resin-based nanocomposite film for wound dressing applications. Polymer. 2025;340(1):129238. doi:10.1016/j.polymer.2025.129238. [Google Scholar] [CrossRef]
56. Lin Z, Jia J, Liang C, Ma Y, Liu H, Fang K, et al. Shape-recoverable chitosan/sodium alginate aerogels with sustained oxygen release and antibacterial activity for diabetic wound healing. Int J Biol Macromol. 2025;305:141005. doi:10.1016/j.ijbiomac.2025.141005. [Google Scholar] [PubMed] [CrossRef]
57. Wang M, Sun P, Zhang J, Li D, Liu Y, Xia Y, et al. Intelligent and biocompatible cellulose aerogels featured with high-elastic and fast-hemostatic for epistaxis and wound healing. Int J Biol Macromol. 2024;277(8):134239. doi:10.1016/j.ijbiomac.2024.134239. [Google Scholar] [PubMed] [CrossRef]
58. Layth H, Muthanna H, Ahmed O. Chitosan aerogel loaded with biogenic palladium nanoparticles CH/Pd NPs exert antibacterial activity and wound addressing application in vitro and in vivo against bacterial skin infections. J Mol Struct. 2024;1315(6):138983. doi:10.1016/j.molstruc.2024.138983. [Google Scholar] [CrossRef]
59. Savencu I, Iurian S, Porfire A, Bogdan C, Tomuţă I. Review of advances in polymeric wound dressing films. React Funct Polym. 2021;168(6):105059. doi:10.1016/j.reactfunctpolym.2021.105059. [Google Scholar] [CrossRef]
60. Wen F, Su W, Cen L, Chen Y, Huo L, Zhong H, et al. Natural fluorescent carbon quantum dots embedded polyvinyl alcohol/chitosan film with photoregulation and high antibacterial efficiency for infected wound healing. Int J Biol Macromol. 2025;306:141716. doi:10.1016/j.ijbiomac.2025.141716. [Google Scholar] [PubMed] [CrossRef]
61. Panchal R, Mateti T, Likhith K, Rodrigues FC, Thakur G. Genipin cross-linked chitosan-PVA composite films: an investigation on the impact of cross-linking on accelerating wound healing. React Funct Polym. 2022;178(21):105339. doi:10.1016/j.reactfunctpolym.2022.105339. [Google Scholar] [CrossRef]
62. Su J, Liu C, Sun A, Yan J, Sang F, Xin Y, et al. Hemostatic and antimicrobial properties of chitosan-based wound healing dressings: a review. Int J Biol Macromol. 2025;306(7):141570. doi:10.1016/j.ijbiomac.2025.141570. [Google Scholar] [PubMed] [CrossRef]
63. Huang F, Yang D, Bai G, Su D, Zhu Z, Long Y. Rapid organic-solvent-free freeze-thaw fabrication of pure chitosan sponges for scalable potential wound dressing applications. Carbohydr Polym Technol Appl. 2025;10(11):100809. doi:10.1016/j.carpta.2025.100809. [Google Scholar] [CrossRef]
64. Meng X, Lu Y, Gao Y, Cheng S, Tian F, Xiao Y, et al. Chitosan/alginate/hyaluronic acid polyelectrolyte composite sponges crosslinked with genipin for wound dressing application. Int J Biol Macromol. 2021;182(12):512–23. doi:10.1016/j.ijbiomac.2021.04.044. [Google Scholar] [PubMed] [CrossRef]
65. Zhu Y, Chen T, Wan X, Zhu G, Chen Z, Yang Z, et al. Fabrication of PLGA-chitosan-tranexamic acid composite sponge and its mechanism for rapid hemostasis. Int J Biol Macromol. 2025;314(S2):144296. doi:10.1016/j.ijbiomac.2025.144296. [Google Scholar] [PubMed] [CrossRef]
66. Miranda CS, Silva AFG, Evenou C, Lamartine J, Fromy B, Pereira-Lima SMMA, et al. Pioneering wound care solutions: triaxial wet-spun fibers with bioactive agents for chronic wounds, part II (controlled release and biological activity of the active agents). Mater Adv. 2025;6(6):2029–47. doi:10.1039/d4ma01104j. [Google Scholar] [CrossRef]
67. Li Z, Guo Q, Chen R, E Y, Wang Y, Zhu M, et al. Tannic acid coated core-shell fibers with antibacterial and antioxidant properties for diabetic wound healing. Mater Des. 2025;253:113874. doi:10.1016/j.matdes.2025.113874. [Google Scholar] [CrossRef]
68. Chowdhury MFM, Khan MN, Rahman MM. Metal nanoparticles incorporated chitosan-based electrospun nanofibre mats for wound dressing applications: a review. Int J Biol Macromol. 2024;282 Pt 6:137352. doi:10.1016/j.ijbiomac.2024.137352. [Google Scholar] [PubMed] [CrossRef]
69. Augustine R, Rehman SRU, Ahmed R, Zahid AA, Sharifi M, Falahati M, et al. Electrospun chitosan membranes containing bioactive and therapeutic agents for enhanced wound healing. Int J Biol Macromol. 2020;156(9):153–70. doi:10.1016/j.ijbiomac.2020.03.207. [Google Scholar] [PubMed] [CrossRef]
70. Cui C, Sun S, Wu S, Chen S, Ma J, Zhou F. Electrospun chitosan nanofibers for wound healing application. Eng Regen. 2021;2(4):82–90. doi:10.1016/j.engreg.2021.08.001. [Google Scholar] [CrossRef]
71. Khan F, Bairagi B, Mondal B, Mandal D, Nath D. In vivo biocompatibility of electrospun chitosan/PEO nanofiber as potential wound dressing material for diabetic burn wound injury of mice. Chem Pap. 2024;78(16):8833–47. doi:10.1007/s11696-024-03714-w. [Google Scholar] [CrossRef]
72. Kianfar P, Vitale A, Dalle Vacche S, Bongiovanni R. Photo-crosslinking of chitosan/poly(ethylene oxide) electrospun nanofibers. Carbohydr Polym. 2019;217(18):144–51. doi:10.1016/j.carbpol.2019.04.062. [Google Scholar] [PubMed] [CrossRef]
73. Cao-Luu NH, Nguyen TV, Dang HG, Pham HG. Engineered polyvinyl alcohol/chitosan/carrageenan nanofibrous membrane loaded with Aloe vera for accelerating third-degree burn wound healing. Int J Biol Macromol. 2025;311(1–3):143880. doi:10.1016/j.ijbiomac.2025.143880. [Google Scholar] [PubMed] [CrossRef]
74. Ren Y, Huang L, Wang Y, Mei L, Fan R, He M, et al. Stereocomplexed electrospun nanofibers containing poly(lactic acid) modified quaternized chitosan for wound healing. Carbohydr Polym. 2020;247(6):116754. doi:10.1016/j.carbpol.2020.116754. [Google Scholar] [PubMed] [CrossRef]
75. Patole V, Bhosale P, Ingavle G, Behere I, Vyawahare N, Ottoor D, et al. In vitro and in vivo assessment of gallic acid-chitosan/polycaprolactone conjugate electrospun nanofibers for wound healing. J Drug Deliv Sci Technol. 2024;95(9):105569. doi:10.1016/j.jddst.2024.105569. [Google Scholar] [CrossRef]
76. de la Mora-López DS, Madera-Santana TJ, Olivera-Castillo L, Castillo-Ortega MM, López-Cervantes J, Sánchez-Machado DI, et al. Production and performance evaluation of chitosan/collagen/honey nanofibrous membranes for wound dressing applications. Int J Biol Macromol. 2024;275:133809. doi:10.1016/j.ijbiomac.2024.133809. [Google Scholar] [PubMed] [CrossRef]
77. Jinia R, Datta N, Wong S, Li X, Arafat M. Mechanistic study of unoxidized tannic acid crosslinked gelatin-CMC electrospun matrices for combating antibacterial resistance in infected wounds. Int J Biol Macromol. 2025;330(12):147837. doi:10.1016/j.ijbiomac.2025.147837. [Google Scholar] [PubMed] [CrossRef]
78. Guo H, Ran W, Jin X, Huang Y, Long F, Xiao Y, et al. Development of pectin/chitosan-based electrospun biomimetic nanofiber membranes loaded with dihydromyricetin inclusion complexes for wound healing application. Int J Biol Macromol. 2024;278(21):134526. doi:10.1016/j.ijbiomac.2024.134526. [Google Scholar] [PubMed] [CrossRef]
79. Zhao J, Chen L, Ma A, Bai X, Zeng Y, Liu D, et al. Recent advances in coaxial electrospun nanofibers for wound healing. Mater Today Bio. 2024;29(9):101309. doi:10.1016/j.mtbio.2024.101309. [Google Scholar] [PubMed] [CrossRef]
80. Mosallanezhad P, Rajabifar N, Rostami A, Ahmadi Z, Taghdimi R, Nazockdast H. Preparation and structural analyses of antimicrobial core-shell chitosan-polycaprolactone nanofibers for controlled curcumin release. Colloid Surface A. 2025;716(8):136736. doi:10.1016/j.colsurfa.2025.136736. [Google Scholar] [CrossRef]
81. Xu F, Wang S, Cao C, Ma W, Zhang X, Du J, et al. Microfluidic generation of multifunctional core-shell microfibers promote wound healing. Colloids Surf B Biointerfaces. 2022;219:112842. doi:10.1016/j.colsurfb.2022.112842. [Google Scholar] [PubMed] [CrossRef]
82. Cheriyan S, Shin H, Razack SA, Kang M, Boopathi T, Kang HW, et al. In-vitro and in-vivo studies of Tridax procumbens leaf extract incorporated bilayer polycaprolactone/polyvinyl alcohol-chitosan electrospun nanofiber for wound dressing application. Int J Biol Macromol. 2025;299:139920. doi:10.1016/j.ijbiomac.2025.139920. [Google Scholar] [PubMed] [CrossRef]
83. Alizadeh S, Nassiri M, Farahmandian N, Farshi P, Ahovan ZA, Majidi J, et al. Engineering of a bilayer antibacterial wound dressing from bovine pericardium and electrospun chitosan/PVA/antibiotics for infectious skin wounds management: an in vitro and in vivo study. Int J Biol Macromol. 2024;282(5):137055. doi:10.1016/j.ijbiomac.2024.137055. [Google Scholar] [PubMed] [CrossRef]
84. Ramakrishnan P, Ramprasath R, Jalaludeen AM, Jayakumar R, Jolius G, Balu R, et al. Electrospun nanofibers of collagen and chitosan for tissue engineering and drug delivery applications: a review. Int J Biol Macromol. 2025;296:139663. doi:10.1016/j.ijbiomac.2025.139663. [Google Scholar] [PubMed] [CrossRef]
85. Gholizadeh R, Mosleh-Shirazi S, Amani AM. Investigation of electrospun chitosan nanofibers reinforced with mesoporous silicon oxide decorated with silver nanoparticles in diabetic wound treatment. Int J Biol Macromol. 2025;322(9):146432. doi:10.1016/j.ijbiomac.2025.146432. [Google Scholar] [PubMed] [CrossRef]
86. Gundhavi Devi M, Arun Karthick S. Fabrication of chitosan/sericin/polyvinyl alcohol nanofiber with silver nanoparticles for wound dressing applications. Polym Eng Sci. 2024;64(9):4128–43. doi:10.1002/pen.26837. [Google Scholar] [CrossRef]
87. Marin L, Andreica B-I, Anisiei A, Cibotaru S, Bardosova M, Materon EM, et al. Quaternized chitosan (nano)fibers: a journey from preparation to high performance applications. Int J Biol Macromol. 2023;242:125136. doi:10.1016/j.ijbiomac.2023.125136. [Google Scholar] [PubMed] [CrossRef]
88. Jia J, Lin Z, Zhu J, Liu Y, Hu Y, Fang K. Anti-adhesive and antibacterial chitosan/PEO nanofiber dressings with high breathability for promoting wound healing. Int J Biol Macromol. 2024;261:129668. doi:10.1016/j.ijbiomac.2024.129668. [Google Scholar] [PubMed] [CrossRef]
89. Nguyen TLA, Bhattacharya D. Antimicrobial activity of quercetin: an approach to its mechanistic principle. Molecules. 2022;27(8):2494. doi:10.3390/molecules27082494. [Google Scholar] [PubMed] [CrossRef]
90. Yang B, Tang B, Wang Z, Feng F, Wang G, Zhao Z, et al. Solution blow spun bilayer chitosan/polylactic acid nanofibrous patch with antibacterial and anti-inflammatory properties for accelerating acne healing. Carbohydr Polym. 2024;326(8):121618. doi:10.1016/j.carbpol.2023.121618. [Google Scholar] [PubMed] [CrossRef]
91. Cheng J, Jun Y, Qin J, Lee SH. Electrospinning versus microfluidic spinning of functional fibers for biomedical applications. Biomaterials. 2017;114:121–43. doi:10.1016/j.biomaterials.2016.10.040. [Google Scholar] [PubMed] [CrossRef]
92. Liu J, Cui T, Xu X, Du Y, Wang L, Chen S, et al. Robust alcohol soluble polyurethane/chitosan/silk sericin (APU/CS/SS) nanofiber scaffolds toward artificial skin extracellular matrices via microfluidic blow-spinning. Adv Fiber Mater. 2023;5(1):349–61. doi:10.1007/s42765-022-00227-7. [Google Scholar] [CrossRef]
93. Gerling KA, Kersey AJ, Lauria AL, Mares JA, Hutzler JD, White PW, et al. Evaluation of novel hemostatic agents in a coagulopathic swine model of junctional hemorrhage. J Trauma Acute Care Surg. 2023;95(2S):S144–51. doi:10.1097/TA.0000000000004071. [Google Scholar] [PubMed] [CrossRef]
94. Thakare NR, Hazarika S. Eco-friendly biopolymers and their biomedical applications: a review. Int J Biol Macromol. 2025;327(4):147225. doi:10.1016/j.ijbiomac.2025.147225. [Google Scholar] [PubMed] [CrossRef]
95. Roy P, Saha R, Dattaray D, Saha S, Mandal TK, Srivastava P, et al. Bioactive glass incorporated dressing matrix for rapid hemostatic action with antibacterial activity. Mater Chem Phys. 2024;315(2):128942. doi:10.1016/j.matchemphys.2024.128942. [Google Scholar] [CrossRef]
96. Zhang W, Geng X, Qin S, Xie Z, Li W, Li J. Research progress and application of chitosan dressings in hemostasis: a review. Int J Biol Macromol. 2024;282(10):136421. doi:10.1016/j.ijbiomac.2024.136421. [Google Scholar] [PubMed] [CrossRef]
97. Huang ZH, Zhang D, Tong LQ, Gao F, Zhang SZ, Wang XQ, et al. Protonated-chitosan sponge with procoagulation activity for hemostasis in coagulopathy. Bioact Mater. 2024;41(10):174–92. doi:10.1016/j.bioactmat.2024.07.012. [Google Scholar] [PubMed] [CrossRef]
98. Li K, Qi H, Liu Q, Li T, Chen W, Li S, et al. Preparation and antitumor activity of selenium-modified glucomannan oligosaccharides. J Funct Foods. 2020;65(6):103731. doi:10.1016/j.jff.2019.103731. [Google Scholar] [CrossRef]
99. Jian X, Jian S, Deng B. Konjac Glucomannan: a functional food additive for preventing metabolic syndrome. J Funct Foods. 2024;115(3):106108. doi:10.1016/j.jff.2024.106108. [Google Scholar] [CrossRef]
100. Shah BR, Li B, Wang L, Liu S, Li Y, Wei X, et al. Health benefits of konjac glucomannan with special focus on diabetes. Bioact Carbohydr Diet Fibre. 2015;5(2):179–87. doi:10.1016/j.bcdf.2015.03.007. [Google Scholar] [CrossRef]
101. Kapoor DU, Sharma H, Maheshwari R, Pareek A, Gaur M, Prajapati BG, et al. Konjac glucomannan: a comprehensive review of its extraction, health benefits, and pharmaceutical applications. Carbohydr Polym. 2024;339(1):122266. doi:10.1016/j.carbpol.2024.122266. [Google Scholar] [PubMed] [CrossRef]
102. Zhu L, Chen J, Mao X, Tang S. A γ-PGA/KGM-based injectable hydrogel as immunoactive and antibacterial wound dressing for skin wound repair. Mater Sci Eng C. 2021;129:112374. doi:10.1016/j.msec.2021.112374. [Google Scholar] [PubMed] [CrossRef]
103. Xu S, Yan SR, You J, Wu XC. Antibacterial micelles-loaded carboxymethyl chitosan/oxidized konjac glucomannan composite hydrogels for enhanced wound repairing. ACS Appl Mater Interfaces. 2024;16(11):13563–72. doi:10.1021/acsami.3c19268. [Google Scholar] [PubMed] [CrossRef]
104. Shang J, Duan L, Zhang W, Li X, Ma C, Xin B. Characterization and evaluation of Bletilla striata polysaccharide/konjac glucomannan blend hydrogel for wound healing. J Appl Biomater Funct Mater. 2023;21:22808000231176202. doi:10.1177/22808000231176202. [Google Scholar] [PubMed] [CrossRef]
105. Shang J, Duan L, Zhang W, Zhuang Q, Ren X, Gu D. The effect of Bletilla striata polysaccharide on the physical and healing properties of curdlan-based hydrogel for wound healing. J Biomater Appl. 2024;38(9):943–56. doi:10.1177/08853282241238409. [Google Scholar] [PubMed] [CrossRef]
106. He Z, He Y, Meng X, Ge Z, Sun H. Structural characteristics and wound-healing effects of Bletilla striata fresh tuber polysaccharide. Int J Biol Macromol. 2024;278:134679. doi:10.1016/j.ijbiomac.2024.134679. [Google Scholar] [PubMed] [CrossRef]
107. Han X, Liu H, Song Z, Yang L, Liu X, Zhang J, et al. Biocompatible Bletilla striata polysaccharide-based hemostatic sponges enhance wound healing and reduce the risk for infections. Carbohydr Polym Technol Appl. 2024;7(10):100492. doi:10.1016/j.carpta.2024.100492. [Google Scholar] [CrossRef]
108. Li J, Wu D, Su Z, Guo J, Cui L, Su H, et al. Zinc-induced photocrosslinked konjac glucomannan/glycyrrhizic acid hydrogel promotes skin wound healing in diabetic mice through immune regulation. Carbohydr Polym. 2025;348(1):122780. doi:10.1016/j.carbpol.2024.122780. [Google Scholar] [PubMed] [CrossRef]
109. Li H, Ma L, Zhu N, Liang X, Liu K, Fu X, et al. Natural-origin bioadhesive hydrogel with dual antioxidative and immunoregulatory properties for enhanced angiogenesis and wound healing. Bioact Mater. 2025;53(11):507–21. doi:10.1016/j.bioactmat.2025.07.023. [Google Scholar] [PubMed] [CrossRef]
110. Wu Y, Xu W, Li J, Zhong Z, Huang L, Li S, et al. Influence of tannic acid post-treatment on the degradation and drug release behavior of Schiff base crosslinked konjac glucomannan/chitosan hydrogel. Eur Polym J. 2024;202(6):112592. doi:10.1016/j.eurpolymj.2023.112592. [Google Scholar] [CrossRef]
111. Yang J, Du L, Guo J, Zhang L, Wang S, Wang X. Injectable carboxymethyl chitosan/konjac glucomannan/catechin hydrogel with free radical-scavenging, antimicrobial, and pro-healing abilities for infected wound repair. Int J Biol Macromol. 2025;308(14):142572. doi:10.1016/j.ijbiomac.2025.142572. [Google Scholar] [PubMed] [CrossRef]
112. Wang M, Gu J, Hao Y, Qin X, Yu Y, Zhang H. Adhesive, sustained-release, antibacterial, cytocompatible hydrogel-based nanofiber membrane assembled from polysaccharide hydrogels and functionalized nanofibers. Cellulose. 2023;30(1):323–37. doi:10.1007/s10570-022-04894-y. [Google Scholar] [CrossRef]
113. Wang H, Fan T, Zeng Z, Chen Z, Lu M, Zhou M, et al. Use of ozone oxidation in combination with deacetylation for improving the structure and gelation properties of konjac glucomannan. Food Chem. 2024;453(1):139599. doi:10.1016/j.foodchem.2024.139599. [Google Scholar] [PubMed] [CrossRef]
114. Huang F, Chen J, Tang X, Li Y, Bao H, Mao X, et al. Preparation and wound repair of injectable and self-healing benzaldehyde-modified konjac glucomannan oligosaccharide/polyglutamic ccid/ε-polylysine hydrogel. Biomacromolecules. 2024;26(1):609–22. doi:10.1021/acs.biomac.4c01407. [Google Scholar] [PubMed] [CrossRef]
115. Neto RJG, Genevro GM, de Almeida Paulo L, Lopes PS, de Moraes MA, Beppu MM. Characterization and in vitro evaluation of chitosan/konjac glucomannan bilayer film as a wound dressing. Carbohydr Polym. 2019;212(1):59–66. doi:10.1016/j.carbpol.2019.02.017. [Google Scholar] [PubMed] [CrossRef]
116. Su R, Su W, Cai J, Cen L, Huang S, Wang Y, et al. Photodynamic antibacterial application of TiO2/curcumin/hydroxypropyl-cyclodextrin and its konjac glucomannan composite films. Int J Biol Macromol. 2024;254(3):127716. doi:10.1016/j.ijbiomac.2023.127716. [Google Scholar] [PubMed] [CrossRef]
117. Wan Y, Zhu X, Huang Z, Peng M, Luo H. Incorporation of dual nanoplatelets to a natural polymer for foldable, robust, bioactive, and biocompatible nacre-like nanocomposites. Compos Part B Eng. 2021;214(1):108747. doi:10.1016/j.compositesb.2021.108747. [Google Scholar] [CrossRef]
118. Genevro GM, Neto RJG, de Almeida Paulo L, Lopes PS, de Moraes MA, Beppu MM. Glucomannan asymmetric membranes for wound dressing. J Mater Res. 2020;35(2):216–6. doi:10.1557/JMR.2019.315. [Google Scholar] [CrossRef]
119. Annaidh AN, Bruyère K, Destrade M, Gilchrist MD, Otténio M. Characterization of the anisotropic mechanical properties of excised human skin. J Mech Behav Biomed Mater. 2012;5(1):139–48. doi:10.1016/j.jmbbm.2011.08.016. [Google Scholar] [PubMed] [CrossRef]
120. Lopes LM, Germiniani LGL, Neto JBMR, Andrade PF, da Silveira GAT, Taketa TB, et al. Preparation and characterization of porous membranes of glucomannan and silver decorated cellulose nanocrystals for application as biomaterial. Int J Biol Macromol. 2023;250(18):126236. doi:10.1016/j.ijbiomac.2023.126236. [Google Scholar] [PubMed] [CrossRef]
121. Yu M, Tang P, Tang Y, Wei C, Wang Z, Zhang H. Breathable, moisturizing, anti-oxidation SSD-PG-PVA/KGM fibrous membranes for accelerating diabetic wound tissue regeneration. ACS Appl Bio Mater. 2022;5(6):2894–901. doi:10.1021/acsabm.2c00255. [Google Scholar] [PubMed] [CrossRef]
122. Zhang Z, Li W, Liu Y, Yang Z, Ma L, Zhuang H, et al. Design of a biofluid-absorbing bioactive sandwich-structured Zn-Si bioceramic composite wound dressing for hair follicle regeneration and skin burn wound healing. Bioact Mater. 2021;6(7):1910–20. doi:10.1016/j.bioactmat.2020.12.006. [Google Scholar] [PubMed] [CrossRef]
123. Hu R, Wang N, Hou L, Liu J, Cui Z, Zhang C, et al. Bilayer nanoporous polyethylene membrane with anisotropic wettability for rapid water transportation/evaporation and radiative cooling. ACS Appl Mater Interfaces. 2022;14(7):9833–43. doi:10.1021/acsami.1c22974. [Google Scholar] [PubMed] [CrossRef]
124. Wang Y, Wang H, Lu B, Yu K, Xie R, Lan G, et al. A sandwich-like silk fibroin/polysaccharide composite dressing with continual biofluid draining for wound exudate management. Int J Biol Macromol. 2023;253(2):127000. doi:10.1016/j.ijbiomac.2023.127000. [Google Scholar] [PubMed] [CrossRef]
125. Shi Z, Lan G, Hu E, Lu F, Qian P, Liu J, et al. Puff pastry-like chitosan/konjac glucomannan matrix with thrombin-occupied microporous starch particles as a composite for hemostasis. Carbohydr Polym. 2020;232(2):115814. doi:10.1016/j.carbpol.2019.115814. [Google Scholar] [PubMed] [CrossRef]
126. Zou Y, Xie R, Hu E, Qian P, Lu B, Lan G, et al. Protein-reduced gold nanoparticles mixed with gentamicin sulfate and loaded into konjac/gelatin sponge heal wounds and kill drug-resistant bacteria. Int J Biol Macromol. 2020;148(2):921–31. doi:10.1016/j.ijbiomac.2020.01.190. [Google Scholar] [PubMed] [CrossRef]
127. Kiani S, Dutta A, Yong SA, Karamikamkar S, Behzadfar E. Emerging trend of carbon aerogel synthesis for biomedical applications. Curr Opin Green Sustain Chem. 2025;54:101033. doi:10.1016/j.cogsc.2025.101033. [Google Scholar] [CrossRef]
128. Karamikamkar S, Yalcintas EP, Haghniaz R, de Barros NR, Mecwan M, Nasiri R, et al. Aerogel-based biomaterials for biomedical applications: from fabrication methods to disease-targeting applications. Adv Sci. 2023;10(23):2204681. doi:10.1002/advs.202370154. [Google Scholar] [CrossRef]
129. Zhang M, Guo N, Sun Y, Shao J, Liu Q, Zhuang X, et al. Nanocellulose aerogels from banana pseudo-stem as a wound dressing. Ind Crops Prod. 2023;194:116383. doi:10.1016/j.indcrop.2023.116383. [Google Scholar] [CrossRef]
130. Zong Q, Peng X, Wu H, Ding Y, Ye X, Gao X, et al. Copper-gallate metal-organic framework encapsulated multifunctional konjac glucomannan microneedles patches for promoting wound healing. Int J Biol Macromol. 2024;257:128581. doi:10.1016/j.ijbiomac.2023.128581. [Google Scholar] [PubMed] [CrossRef]
131. Zheng H, Chen K, Dun Y, Xu Y, Zhou A, Ge H, et al. Harnessing nature’s ingenuity to engineer butterfly-wing-inspired photoactive nanofiber patches for advanced postoperative tumor treatment. Biomaterials. 2025;314(25):122808. doi:10.1016/j.biomaterials.2024.122808. [Google Scholar] [PubMed] [CrossRef]
132. Mecwan M, Li J, Falcone N, Ermis M, Torres E, Morales R, et al. Recent advances in biopolymer-based hemostatic materials. Regen Biomater. 2022;9:rbac063. doi:10.1093/rb/rbac063. [Google Scholar] [PubMed] [CrossRef]
133. Zhang S, Li J, Chen S, Zhang X, Ma J, He J. Oxidized cellulose-based hemostatic materials. Carbohydr Polym. 2020;230(4):115585. doi:10.1016/j.carbpol.2019.115585. [Google Scholar] [PubMed] [CrossRef]
134. Meng S, Wu H, Xiao D, Lan S, Dong A. Recent advances in bacterial cellulose-based antibacterial composites for infected wound therapy. Carbohydr Polym. 2023;316(8):121082. doi:10.1016/j.carbpol.2023.121082. [Google Scholar] [PubMed] [CrossRef]
135. Kontturi E, Laaksonen P, Linder MB, Nonappa, Gröschel AH, Rojas OJ, et al. Advanced materials through assembly of nanocelluloses. Adv Mater. 2018;30(24):e1703779. doi:10.1002/adma.201703779. [Google Scholar] [PubMed] [CrossRef]
136. Zheng M, Su H, Xiao R, Chen J, Chen H, Tan KB, et al. Effects of polygonatum cyrtonema extracts on the antioxidant ability, physical and structure properties of carboxymethyl cellulose-xanthan gum-flaxseed gum active packaging films. Food Chem. 2023;403(1):134320. doi:10.1016/j.foodchem.2022.134320. [Google Scholar] [PubMed] [CrossRef]
137. Bukatuka CF, Mbituyimana B, Xiao L, Qaed Ahmed AA, Qi F, Adhikari M, et al. Recent Trends in the application of cellulose-based hemostatic and wound healing dressings. J Funct Biomater. 2025;16(5):151. doi:10.3390/jfb16050151. [Google Scholar] [PubMed] [CrossRef]
138. Saito T, Hirota M, Tamura N, Kimura S, Fukuzumi H, Heux L, et al. Individualization of nano-sized plant cellulose fibrils by direct surface carboxylation using TEMPO catalyst under neutral conditions. Biomacromolecules. 2009;10(7):1992–6. doi:10.1021/bm900414t. [Google Scholar] [PubMed] [CrossRef]
139. Lei D, Zhu L, Yong L, Qun F. The role of formic acid pretreatment in improving the carboxyl content of TEMPO-oxidized cellulose. Cellul Chem Technol. 2014;48(5):469–75. [Google Scholar]
140. Galgut P. Oxidized cellulose mesh: I. Biodegradable membrane in periodontal surgery. Biomaterials. 1990;11(8):561–4. doi:10.1016/0142-9612(90)90078-5. [Google Scholar] [PubMed] [CrossRef]
141. Watanabe J, Ishida F, Ishida H, Fukunaga Y, Watanabe K, Naito M, et al. A prospective multi-center registry concerning the clinical performance of laparoscopic colorectal surgery using an absorbable adhesion barrier (INTERCEED®) made of oxidized regenerated cellulose. Surgery Today. 2019;49(10):877–84. doi:10.1007/s00595-019-01816-7. [Google Scholar] [PubMed] [CrossRef]
142. Jalvo B, Aguilar-Sanchez A, Ruiz-Caldas MX, Mathew AP. Water filtration membranes based on non-woven cellulose fabrics: effect of nanopolysaccharide coatings on selective particle rejection, antifouling, and antibacterial properties. Nanomaterials. 2021;11(7):1752. doi:10.3390/nano11071752. [Google Scholar] [PubMed] [CrossRef]
143. Yang X, Du Y, Jiang P, Fu R, Liu L, Miao C, et al. Woven agarose-cellulose composite aerogel fibers with outstanding radial elasticity for personal thermal management. ACS Appl Mater Interfaces. 2024;16(20):26757–67. doi:10.1021/acsami.4c03509. [Google Scholar] [PubMed] [CrossRef]
144. Wagenhäuser M, Mulorz J, Ibing W, Simon F, Spin J, Schelzig H, et al. Oxidized (non)-regenerated cellulose affects fundamental cellular processes of wound healing. Sci Rep. 2016;6(1):32238. doi:10.1038/srep32238. [Google Scholar] [PubMed] [CrossRef]
145. Wu S, Applewhite AJ, Niezgoda J, Snyder R, Shah J, Cullen B, et al. Oxidized regenerated cellulose/collagen dressings: review of evidence and recommendations. Adv Skin Wound Care. 2017;30(11S):S1–18. doi:10.1097/01.ASW.0000525951.20270.6c. [Google Scholar] [PubMed] [CrossRef]
146. Kleine J, Leisz S, Ghadban C, Hohmann T, Prell J, Scheller C, et al. Variants of oxidized regenerated cellulose and their distinct effects on neuronal tissue. Int J Mol Sci. 2021;22(21):11467. doi:10.3390/ijms222111467. [Google Scholar] [PubMed] [CrossRef]
147. Wu Y, Wang F. Study of the degradation mechanisms of oxidized cellulose-derivative hemostatic agent by using model compound. J Biomater Sci Polym Ed. 2019;30(7):580–91. doi:10.1080/09205063.2019.1592797. [Google Scholar] [PubMed] [CrossRef]
148. Wang G, Zeng Y. Progress in application of medical absorbable haemostatic materials for haemostasis in orthopaedic surgery. Chin J Repar Reconstr Sur. 2024;38:1421–6. (In Chinese). doi:10.7507/1002-1892.202405068. [Google Scholar] [PubMed] [CrossRef]
149. Gulmez M, Aktekin A, Aker F, Sanko V, Sezer S. Evaluation of in vivo adhesion properties of new generation polyglactin, oxidized regenerated cellulose and chitosan-based meshes for hernia surgery. Cureus. 2021;13:e18755. doi:10.7759/cureus.18755. [Google Scholar] [PubMed] [CrossRef]
150. Cheng F, Liu C, Li H, Wei X, Yan T, Wang Y, et al. Carbon nanotube-modified oxidized regenerated cellulose gauzes for hemostatic applications. Carbohydr Polym. 2018;183(37):246–53. doi:10.1016/j.carbpol.2017.12.035. [Google Scholar] [PubMed] [CrossRef]
151. Zhang B, Chakoli AN, He JM, Huang YD, Aleshin AN. Hemostatic effect of aminated multiwalled carbon nanotubes/oxidized regenerated cellulose nanocomposites. J Nanoscience Nanotechnol. 2019;19(11):7410–5. doi:10.1166/jnn.2019.16617. [Google Scholar] [PubMed] [CrossRef]
152. Slezak P, Keibl C, Labahn D, Schmidbauer A, Genyk Y, Gulle H. A comparative efficacy evaluation of recombinant topical thrombin (RECOTHROM®) with a gelatin sponge carrier versus topical oxidized regenerated cellulose (TABOTAMP®/SURGICEL®) in a porcine liver bleeding model. J Investig Surg. 2021;34(8):862–8. doi:10.1080/08941939.2019.1705444. [Google Scholar] [PubMed] [CrossRef]
153. Hsueh W, Shah-Khan SM, Vallabh H, Cardinal JS, Nasr JY. Endoscopic use of oxidized regenerated cellulose and topical thrombin: a novel approach for control of bleeding pancreatic pseudocyst wall after endoscopic cystgastrostomy. VideoGIE. 2017;2(8):193–4. doi:10.1016/j.vgie.2017.04.006. [Google Scholar] [PubMed] [CrossRef]
154. Cidreira ACM, de Castro KC, Hatami T, Linan LZ, Mei LHI. Cellulose nanocrystals-based materials as hemostatic agents for wound dressings: a review. Biomed Microdevices. 2021;23(4):43. doi:10.1007/s10544-021-00581-0. [Google Scholar] [PubMed] [CrossRef]
155. Ndlovu SP, Ngece K, Alven S, Aderibigbe BA. Gelatin-based hybrid scaffolds: promising wound dressings. Polymers. 2021;13(17):2959. doi:10.3390/polym13172959. [Google Scholar] [PubMed] [CrossRef]
156. Chiulan I, Panaitescu DM, Serafim A, Radu ER, Ioniţă G, Rădiţoiu V, et al. Sponges from plasma treated cellulose nanofibers grafted with poly(ethylene glycol)methyl ether methacrylate. Polymers. 2022;14(21):4720. doi:10.3390/polym14214720. [Google Scholar] [PubMed] [CrossRef]
157. Li H, Cheng W, Liu K, Chen L, Huang Y, Wang X, et al. Reinforced collagen with oxidized microcrystalline cellulose shows improved hemostatic effects. Carbohydr Polym. 2017;165:30–8. doi:10.1016/j.carbpol.2017.02.023. [Google Scholar] [PubMed] [CrossRef]
158. Nascimento RE, Monte J, Cadima M, Alves VD, Neves LA. Rendering banana plant residues into a potentially commercial byproduct by doping cellulose films with phenolic compounds. Polymers. 2021;13(5):843. doi:10.3390/POLYM13050843. [Google Scholar] [PubMed] [CrossRef]
159. Dan J, Ma Y, Yue P, Xie Y, Zheng Q, Hu P, et al. Microcrystalline cellulose-carboxymethyl cellulose sodium as an effective dispersant for drug nanocrystals: a case study. Carbohydr Polym. 2016;136(1):499–506. doi:10.1016/j.carbpol.2015.09.048. [Google Scholar] [PubMed] [CrossRef]
160. Seguchi N, Sakamoto Y, Kikuchi A, Kishi K. Hemostatic efficacy of oxidized regenerated cellulose powder in Le Fort 1 osteotomy. J Craniofac Surg. 2024;35(1):189–91. doi:10.1097/SCS.0000000000009772. [Google Scholar] [PubMed] [CrossRef]
161. Ishikura H, Nishiwaki T, Fujita S, Masuyama Y, Hatano M, Tanaka T, et al. Oxidized regenerated cellulose powder reduces perioperative bleeding and thigh swelling in total hip arthroplasty: a prospective interventional study. J Orthop Surg Res. 2025;20(1):505. doi:10.1186/s13018-025-05913-x. [Google Scholar] [PubMed] [CrossRef]
162. Li S, Gong L, Chen J, Wu X, Liu X, Shou Q, et al. Fabricating the multibranch carboxyl-modified cellulose for hemorrhage control. Mater Today Bio. 2023;24(6 Suppl 5):100878. doi:10.1016/j.mtbio.2023.100878. [Google Scholar] [PubMed] [CrossRef]
163. Paternò VA, Bisin A, Addis A. Comparison of the efficacy of five standard topical hemostats: a study in porcine liver and spleen models of surgical bleeding. BMC Surg. 2020;20(1):215. doi:10.1186/s12893-020-00874-w. [Google Scholar] [PubMed] [CrossRef]
164. Hutchinson W, George K, Johns D, Craven L, Zhang G, Shnoda P. Hemostatic efficacy and tissue reaction of oxidized regenerated cellulose hemostats. Cellulose. 2013;20(1):537–45. doi:10.1007/s10570-012-9828-8. [Google Scholar] [CrossRef]
165. Suchý P, Paprskářová A, Chalupová M, Marholdová L, Nešporová K, Klusáková J, et al. Composite hemostatic nonwoven textiles based on hyaluronic acid, cellulose, and etamsylate. Materials. 2020;13(7):1627. doi:10.3390/ma13071627. [Google Scholar] [PubMed] [CrossRef]
166. Chalupová M, Suchý P, Pražanová G, Bartošová L, Sopuch T, Havelka P. Local tissue reaction after the application of topical hemostatic agents in a rat partial nephrectomy model. J Biomed Mater Res A. 2012;100(6):1582–90. doi:10.1002/jbm.a.34098. [Google Scholar] [PubMed] [CrossRef]
167. Cheng F, He J, Yan T, Liu C, Wei X, Li J, et al. Antibacterial and hemostatic composite gauze of N,O-carboxymethyl chitosanoxidized regenerated cellulose. RSC Adv. 2016;6(97):94429–36. doi:10.1039/c6ra15983d. [Google Scholar] [CrossRef]
168. Equicel® Silk - Fast Hemostatic Solution for Challenging Surgical Procedures [Internet]. Zurich Corp; 2023 [cited 2025 Nov 29]. Available from: https://zurichcorp.com/products/disposable/hemostatics-gauzes/equicel-silk-advanced-oxidized-regenerated-cellulose-for-fast-hemostasis/. [Google Scholar]
169. Wysham WZ, Roque DR, Soper JT. Use of topical hemostatic agents in gynecologic surgery. Obstet Gynecol Surv. 2014;69(9):557–63. doi:10.1097/OGX.0000000000000106. [Google Scholar] [PubMed] [CrossRef]
170. Christenson LJ, Otley CC, Roenigk RK. Oxidized regenerated cellulose gauze for hemostasis of a two-stage interpolation flap pedicle. Dermatol Surg. 2004;30(12p2):1593–4. doi:10.1111/j.1524-4725.2004.30574.x. [Google Scholar] [PubMed] [CrossRef]
171. Tam T, Harkins G, Dykes T, Gockley A, Davies M. Oxidized regenerated cellulose resembling vaginal cuff abscess. JSLS. 2014;18(2):353–6. doi:10.4293/108680813X13693422518597. [Google Scholar] [PubMed] [CrossRef]
172. SURGICE NU-KNIT® Absorbable Hemostat [Internet]. ETHICON US, LLC; 2022 [cited 2025 Nov 29]. Available from: https://www.jnjmedtech.com/en-US/product/surgicel-nu-knit-absorbable-hemostat. [Google Scholar]
173. SURGICEL® FIBRILLAR™ Absorbable Hemostat [Internet]. ETHICON US, LLC; 2022 [cited 2025 Nov 29]. Available from: https://www.jnjmedtech.com/en-US/product/surgicel-fibrillar-absorbable-hemostat. [Google Scholar]
174. Jing L, Wei D. Research progress of absorbable oxidized regenerated cellulose hemostatic materials. J Anesth Clin Res. 2021;12(8):10001017. [Google Scholar]
175. Ibne Mahbub MS, Sultana T, Gwon JG, Lee BT. Fabrication of thrombin loaded TEMPO-oxidized cellulose nanofiber-gelatin sponges and their hemostatic behavior in rat liver hemorrhage model. J Biomater Sci Polym Ed. 2022;33(4):499–516. doi:10.1080/09205063.2021.1992877. [Google Scholar] [PubMed] [CrossRef]
176. Pinyakit Y, Trachoo V, Suwanwong Y, Chanamuangkon T, Sae-ear P, Srijunbarl A, et al. Shape adaptive bacterial cellulose-based sponge for hemostasis applications. Int J Biol Macromol. 2025;306(1–2):141694. doi:10.1016/j.ijbiomac.2025.141694. [Google Scholar] [PubMed] [CrossRef]
177. Cheng H, Xiao D, Tang Y, Wang B, Vancso GJ, Sui X, et al. Sponges with janus character from nanocellulose: preparation and applications in the treatment of hemorrhagic wounds. Adv Healthc Mater. 2020;9(17):e1901796. doi:10.1002/adhm.201901796. [Google Scholar] [PubMed] [CrossRef]
178. Dong Q, Liang X, Chen F, Ke M, Yang X, Ai J, et al. Injectable shape memory hydroxyethyl cellulose/soy protein isolate based composite sponge with antibacterial property for rapid noncompressible hemorrhage and prevention of wound infection. Int J Biol Macromol. 2022;217:367–80. doi:10.1016/j.ijbiomac.2022.07.051. [Google Scholar] [PubMed] [CrossRef]
179. Ni L, Ye H, Chang Y, Yang K, Luo Y, Yan L, et al. Anisotropic and lightweight silica aerogel-doped copolymerized polyimide foams fabricated by combining microwave-assisted foaming and freeze-drying for high-temperature thermal insulation and hydrophobic applications. ACS Appl Mater Interfaces. 2025;17(31):44952–65. doi:10.1021/acsami.5c09574. [Google Scholar] [PubMed] [CrossRef]
180. Shakya KR, Nigam K, Sharma A, Jahan K, Tyagi AK, Verma V. Preparation and assessment of agar/TEMPO-oxidized bacterial cellulose cryogels for hemostatic applications. J Mater Chem B. 2024;12(14):3453–68. doi:10.1039/D4TB00047A. [Google Scholar] [PubMed] [CrossRef]
181. Mohamed E, Wang Y, Crispin PJ, Fitzgerald A, Dahlstrom JE, Fowler S, et al. Superior hemostatic and wound-healing properties of gel and sponge forms of nonoxidized cellulose nanofibers: in vitro and in vivo studies. Macromol Biosci. 2022;22(10):e2200222. doi:10.1002/mabi.202200222. [Google Scholar] [PubMed] [CrossRef]
182. Zhang Z, Abidi N, Lucia L, Chabi S, Denny CT, Parajuli P, et al. Cellulose/nanocellulose superabsorbent hydrogels as a sustainable platform for materials applications: a mini-review and perspective. Carbohydr Polym. 2023;299(5):120140. doi:10.1016/j.carbpol.2022.120140. [Google Scholar] [PubMed] [CrossRef]
183. Wassgren J, Clarke BR, Messikh MB, Ho CH, Crosby AJ, Tew GN, et al. Enhancement of mechanical properties of nanocellulose xerogels using TEMPO-oxidized fibers. Carbohydr Polym. 2025;348(Pt B):122839. doi:10.1016/j.carbpol.2024.122839. [Google Scholar] [PubMed] [CrossRef]
184. Mahmoodzadeh A, Moghaddas J, Jarolmasjed S, Ebrahimi Kalan A, Edalati M, Salehi R. Biodegradable cellulose-based superabsorbent as potent hemostatic agent. Chem Eng J. 2021;418(1):129252. doi:10.1016/j.cej.2021.129252. [Google Scholar] [CrossRef]
185. Stark M, Wang AY, Corrigan B, Woldu HG, Azizighannad S, Cipolla G, et al. Comparative analyses of the hemostatic efficacy and surgical device performance of powdered oxidized regenerated cellulose and starch-based powder formulations. Res Pract Thromb Haemost. 2024;9(1):102668. doi:10.1016/j.rpth.2024.102668. [Google Scholar] [PubMed] [CrossRef]
186. Guo Y, Cheng N, Sun H, Hou J, Zhang Y, Wang D, et al. Advances in the development and optimization strategies of the hemostatic biomaterials. Front Bioeng Biotechnol. 2023;10:1062676. doi:10.3389/fbioe.2022.1062676. [Google Scholar] [PubMed] [CrossRef]
187. SURGICEL™ powder absorbable hemostat [Internet]. ETHICON US, LLC; 2022 [cited 2025 Nov 29]. Available from: https://www.jnjmedtech.com/en-US/product/surgicel-powder-absorbable-hemostat. [Google Scholar]
188. Al-Attar N, de Jonge E, Kocharian R, Ilie B, Barnett E, Berrevoet F. Safety and hemostatic effectiveness of SURGICEL® powder in mild and moderate intraoperative bleeding. Clin Appl Thromb Hemost. 2023;29(11):10760296231190376. doi:10.1177/10760296231190376. [Google Scholar] [PubMed] [CrossRef]
189. Mohamed E, Coupland LA, Crispin PJ, Fitzgerald A, Nisbet DR, Tsuzuki T. Non-oxidized cellulose nanofibers as a topical hemostat: In vitro thromboelastometry studies of structure vs. function. Carbohydr Polym. 2021;265(1):118043. doi:10.1016/j.carbpol.2021.118043. [Google Scholar] [PubMed] [CrossRef]
190. Shefa AA, Taz M, Lee SY, Lee BT. Enhancement of hemostatic property of plant derived oxidized nanocellulose-silk fibroin based scaffolds by thrombin loading. Carbohydr Polym. 2019;208(3):168–79. doi:10.1016/j.carbpol.2018.12.056. [Google Scholar] [PubMed] [CrossRef]
191. Cheng J, Liu J, Li M, Liu Z, Wang X, Zhang L, et al. Hydrogel-based biomaterials engineered from natural-derived polysaccharides and proteins for hemostasis and wound healing. Front Bioeng Biotechnol. 2021;9:780187. doi:10.3389/fbioe.2021.780187. [Google Scholar] [PubMed] [CrossRef]
192. Basu A, Hong J, Ferraz N. Hemocompatibility of Ca2+-crosslinked nanocellulose hydrogels: toward efficient management of hemostasis. Macromol Biosci. 2017;17(11):293. doi:10.1002/mabi.201700236. [Google Scholar] [PubMed] [CrossRef]
193. Shahzad S, Ghouse G, Zubair M, Yaqoob A, Mujahid M, Ahmed F, et al. Citric acid cross-linked honey and aloe vera gel loaded chitosan hydrogels for wound dressing applications. RSC Adv. 2025;15(48):40745–59. doi:10.1039/d5ra05550d. [Google Scholar] [PubMed] [CrossRef]
194. Chen Z, Jiang B, Bi H, Wang C, Liu Y, Wang J, et al. Construction of multilayer multifunctional hydrogel microspheres with natural macromolecules, tannic acid, and copper for enhanced diabetic wound healing. Chem Eng J. 2025;520(9):165896. doi:10.1016/j.cej.2025.165896. [Google Scholar] [CrossRef]
195. Ding S, Jingbin H, Siyu L, Md R, Shuaihang W, Sameer J. 3D-printed multilayer GelMA-PVA dual-network hydrogels with gradient andrographolide loading for antimicrobial wound healing: fabrication, characterization, and in vitro evaluation. Mater Today Commun. 2025;48(15):113638. doi:10.1016/j.mtcomm.2025.113638. [Google Scholar] [CrossRef]
196. Mousavi-Ejarestaghi N, Shaabani A, Milani S, Motedayen M, Maghsoudian S, Fatahi Y, et al. Smart multilayer chitosan-based nanofibers for burn wounds: pH-responsive dual release of diclofenac sodium & Hamamelis virginiana extract: from design to in vitro & in vivo evaluation. Int J Biol Macromol. 2025;320(Pt 3):145826. doi:10.1016/j.ijbiomac.2025.145826. [Google Scholar] [PubMed] [CrossRef]
197. Yao S, Chen T, Liu Z, Fan L, Xie Y, You S, et al. A dual-layer chitosan-based conductive hydrogel and piezoelectric film for ultrasound-driven wound repair and scar reduction. Carbohydr Polym. 2025;370(1):124305. doi:10.1016/j.carbpol.2025.124305. [Google Scholar] [PubMed] [CrossRef]
198. Cillis AD, Garzarelli V, Foscarini A, Gigli G, Turco A, Primiceri E, et al. 3D-printed barriers with machine learning powered image analysis for enhanced wound healing assays. Mater Des. 2025;259:114746. doi:10.1016/j.matdes.2025.114746. [Google Scholar] [CrossRef]
199. Kolipaka T, Pandey G, Abraham N, Srinivasarao DA, Raghuvanshi RS, Rajinikanth PS, et al. Stimuli-responsive polysaccharide-based smart hydrogels for diabetic wound healing: design aspects, preparation methods and regulatory perspectives. Carbohydr Polym. 2024 15;324(3):121537. doi:10.1016/j.carbpol.2023.121537. [Google Scholar] [PubMed] [CrossRef]
200. Andam S, Borhani F. 3D-printed hyaluronic acid-based hydrogel in wound healing applications: a review of environmentally friendly approaches. Eur Polym J. 2025;236:114140. doi:10.1016/j.eurpolymj.2025.114140. [Google Scholar] [CrossRef]
201. Chen X, Zhou Y, Zhou S, Zhang D, Zeng L, Zhou C, et al. 3D printing of chitosan hydrogel reinforced with tubular nanoclay for hemostasis and infected wound healing. Bioact Mater. 2025;54(36):404–22. doi:10.1016/j.bioactmat.2025.08.024. [Google Scholar] [PubMed] [CrossRef]
202. Thanha KK, Sana PA, Ajesh J, Naseef P, Tharayil H, Lubaib P, et al. Advanced smart bioelectronics for wound healing: biosensing, drug delivery, and artificial intelligence. Int J Pharm. 2025;684:126098. doi:10.1016/j.ijpharm.2025.126098. [Google Scholar] [PubMed] [CrossRef]
203. Qu Z, Wang Y, Dong Y, Li X, Hao L, Sun L, et al. Intelligent electrospinning nanofibrous membranes for monitoring and promotion of wound healing. Mater Today Bio. 2024;26(Suppl 2):101093. doi:10.1016/j.mtbio.2024.101093. [Google Scholar] [PubMed] [CrossRef]
204. Deng D, Liang L, Su K, Gu H, Wang X, Wang Y, et al. Smart hydrogel dressing for machine learning-enabled visual monitoring and promote diabetic wound healing. Nano Today. 2025;60(4):102559. doi:10.1016/j.nantod.2024.102559. [Google Scholar] [CrossRef]
205. Foltynski P, Kruszewska K, Krakowiecki A, Czarkowska-Paczek B, Ladyzynski P. Artificial intelligence models for wound infection recognition and their comparison with human results. Biocybern Biomed Eng. 2025;45(3):572–9. doi:10.1016/j.bbe.2025.08.003. [Google Scholar] [CrossRef]
206. Deng J, Shi G, Ye Z, Xiao Q, Zhang X, Ren L, et al. Unveiling and swift diagnosing chronic wound healing with artificial intelligence assistance. Chin Chem Lett. 2025;36(3):110496. doi:10.1016/j.cclet.2024.110496. [Google Scholar] [CrossRef]
207. Lemonnier D, Sumpio BJ, Crouse M, Mezghani I, Theocharidis G, Jakus MA, et al. Quantitative ultrasound augmented with machine learning to assess tissue microstructure during wound healing. Biomed Signal Process Control. 2025;103(7):107420. doi:10.1016/j.bspc.2024.107420. [Google Scholar] [CrossRef]
208. Pang Q, Yang F, Jiang Z, Wu K, Hou R, Zhu Y. Smart wound dressing for advanced wound management: real-time monitoring and on-demand treatment. Mater Des. 2023;229(4):111917. doi:10.1016/j.matdes.2023.111917. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF


 Downloads
Downloads
 Citation Tools
Citation Tools