 Open Access
Open Access
ARTICLE
Maleated Deep Eutectic Solvent (DES) Lignin-Glyoxal-Chitosan Wood Adhesives
1 Department of Wood and Paper Sciences, Faculty of Natural Resources, Semnan University, Semnan, Iran
2 Laboratoire d’Etude et de Recherche sur le Materiau Bois (LERMAB), University of Lorraine, Blvd des Aiguiellettes, Nancy, France
3 Department of Chemistry, Science and Research Branch, Islamic Azad University, Tehran, Iran
* Corresponding Authors: Hamed Younesi-Kordkheili. Email: ; Antonio Pizzi. Email:
(This article belongs to the Special Issue: Renewable and Biosourced Adhesives-3rd Edition)
Journal of Renewable Materials 2026, 14(4), 1 https://doi.org/10.32604/jrm.2026.02026-0013
Received 28 January 2026; Accepted 27 March 2026; Issue published 24 April 2026
Abstract
In this research, an effective method has been developed to bond wood panel adhesives of good performance using biosourced lignin and chitosan as the two main raw materials. Lignin was thus modified by a deep eutectic solvent (DES) and maleic anhydride (MA), respectively. Then DES/MA modified lignin was reacted with glyoxal to obtain a new environmentally friendly lignin-glyoxal (LG) resin. Next, chitosan was added as a bio-crosslinker to the synthesised LG resin. Finally, the performance of the resulting adhesives was evaluated by bonding plywood panels with them. Fourier Transform Infra Red (FTIR) analysis was used to confirm the chemical modification of lignin as well as the cross-links between lignin and chitosan. 13C Nuclear Magnetic Resonance (13C NMR) and Matrix Assisted Laser Desorption Ionization Time of Flight (MALDI TOF) mass spectrometry analysis show that both DES lignin and chitosan readily react and link with glyoxal and indicate the type of structures that are formed. Differential Scanning Calorimetry (DSC) showed that the glass transition temperature (Tg) in pristine lignin decreased after DES treatment but it increases by functionalizing it with maleic anhydride. The DES/MA-modified lignin resin has higher solid content, viscosity and density compared to those prepared with either DES-modified lignin or pristine lignin. The results also indicated that the addition of chitosan to the LG resin prepared had consequent effects on improving the bond strength of the plywood panels as their dry shear strength can meet the required adhesion level of the EN-314 standard. Meanwhile, the dimensional stability of the plywood panels bonded with lignin-glyoxal resin was decreased by the addition of chitosan. Finally, the results suggest that the crosslinking of DES/MA-treated lignin with glyoxal in the presence of chitosan as a bio-crosslinker is a promising approach for developing lignin-based bioadhesives for wood bonding.Graphic Abstract
Keywords
The increasing concerns regarding environmental and health safety have today focused the growing interest in green chemistry. Green chemistry focuses on the design, production, and eco-friendly application of products that minimise harm to humans and the environment by avoiding or minimising as much as possible the use of hazardous materials [1]. Some of the hazardous materials comprise formaldehyde-based wood adhesives such as phenol-formaldehyde (PF) and urea-formaldehyde (UF) resins. Formaldehyde is classified as a carcinogen, and previous research has confirmed that it can cause respiratory issues, eye irritation, and skin irritation [2]. For this reason, today, the trend towards the use of adhesives totally, or in the great majority bio-sourced in the wood industry is growing. In this context, extracted lignin from black liquors of the pulp and paper industry is one of the biopolymers being considered for use as wood adhesives. In fact, the lignin phenolic nature renders it a potential substitute for formaldehyde-based synthetic wood adhesives. So far, the use of lignin as a partial substitute for phenol and urea in combination with formaldehyde has been investigated by several researchers [3,4]. However, research on crosslinking lignin alone as the primary component of wood adhesives remains limited. There are significant challenges which limit the application of lignin alone for resin synthesis. The greatest limitations include chemical variations depending on the lignin origin, high dispersion in its molecular weight, complex structures, and low reactivity [5]. Thus, the objective of this work was to develop wood adhesives with lignin as their main component.
To obtain lignin-based wood adhesive with acceptable properties, the chemical structure of lignin should first be modified. So far, several methods for lignin modification such as phenolation, glyoxalation, hydrolysis, etc., have been proposed. One of the best proposed methods for lignin modification is the use of deep eutectic solvents (DESs). ChCl–ZnCl2 (Choline Chloride-Zinc Chloride) DES has already been used as a green DES to modify the lignin structure [6,7]. The most obvious advantages of using ChCl-ZnCl2 as a DES-based are the appropriate performance of the desired products, short reaction time with maximum synthesis efficiency, low cost of raw materials for preparation, and a mild and environmentally friendly reaction environment. Also, Zn-based DESs could provide a greener platform for the transformation of lignin compared to other DESs. The indication was that with DES treatment, the reactive sites of lignin increased and the molecular weight decreased. However, to obtain an adhesive with lignin in even higher proportions more modifications are needed to increase its reactivity even further.
Conversely, the reactive maleic anhydride (MA) graft to lignin does significantly increase lignin reactivity. MA and lignin react together causing an increase in the lignin reactive sites to form oligomers as already shown previously [8,9]. Previous research showed that the modification of lignin by MA can significantly increase the reactive sites of lignin. The consequence of this is the bettering of the physicochemical characteristics of the resin. Maleation of lignin units and formation of maleated lignin are showed at Fig. 1. The aromatic nuclei and C=C double bonds in MA’s structure consequently would lead to grafting to DES-modified lignin. Hence, in this research, lignin was firstly modified by DES and then functionalized by maleic anhydride to obtain very reactive lignin molecules.

Figure 1: Maleation of lignin units and formation of maleated lignin [7,8].
Conversely, to obtain wood adhesives based on lignin, formaldehyde, being classed as toxic, should be substituted by non or much less toxic aldehydes. So far, several nontoxic aldehydes have been proposed to substitute for formaldehyde in wood adhesive preparation. Among the reactive aldehydes, glyoxal is an excellent choice for the synthesis of bio-sourced adhesive from lignin and from other bioresources. Glyoxal is of human very low toxicity, has no carcinogenic properties, is nonvolatile, is inexpensive and fully biodegradable. Hence, in the research presented here, DES-treated lignin was functionalized by MA and by reacting it with glyoxal a new adhesive based on lignin has been synthesized, see Fig. 2, where some of the lignin sites liable of reaction with glyoxal are indicated.

Figure 2: Indication of some of the lignin sites available for reaction with glyoxal.
Conversely, to obtain higher performance lignin adhesives with comparable properties to synthetic wood adhesive, adding bio-crosslinkers appears to be necessary. Chitosan is a bio-sourced, abundant, antimicrobial, biodegradable, nontoxic, inexpensive and biocompatible polysaccharide. Previous work has shown that due to a high proportion of its main structural chain of amino and hydroxyl groups available for reaction, chitosan can be used as a bio-crosslinker to increase the bonding strengths in wood adhesives [10]. Previous work has also shown that glyoxal and glyoxalated material can readily cross-link in several different ways with chitosans [11–13] as shown in Fig. 3.

Figure 3: Example of bonds formed by the reaction of glyoxal with chitosan.
When chitosan is then added to modified lignin, the amine groups of chitosan can react with the still reactive residual glyoxal aldehyde groups attached onto lignin, in the same manner shown in Fig. 3, as well as with the residual maleic anhydride groups in modified lignin, resulting in an improvement in the properties of the resin prepared. Hence, it is expected that by functionalizing lignin with maleic anhydride and adding chitosan, the properties of the prepared resins will be significantly improved. So far, several researchers have been focused on the lignin-glyoxal and lignin–chitosan resins but there are no reports about integrating DES–MA lignin with glyoxal and chitosan to obtain a new wood adhesive of acceptable quality. Hence, in the research work presented here a new mostly bio-sourced wood panel adhesive designed through DES/maleated lignin, glyoxal and chitosan has been prepared.
Bagasse Soda black liquor with a pH of 13% and 40% solid content as a source of lignin was used in the current study. Black liquor sulfuric acid extracted soda lignin was used. All the other chemicals have been obtained from the Sigma Aldrich Chemical Co. (Burlington, NC, USA). Degree of deacetylation, molecular weight and viscosity grade of the prepared chitosan are reported in Table 1:

2.2.1 DES-Modification of Lignin
Lignin was modified according to a method reported by Hong et al. [14]. Dried choline chloride and zinc chloride were mixed in an Erlenmeyer flask (the molar ratio of choline chloride to zinc chloride was 1: 2, ChCl–ZnCl2). Then the mixture was transferred to a 90°C water bath with magnetic stirring up to homogeneous transparency. The DES-modified lignin (DL) was then subjected first to precipitation and then to regeneration by adding water. This was followed by separation by centrifugation and distilled water washing of the lignin regenerated. This process was continued up to its conductivity being close to that of distilled water. Followed first a 45°C drying, and then vacuum drying for 24 h at 50°C. A percentage yield of 66% by mass of DL lignin was obtained.
2.2.2 Preparation of DES/MA-Modified Lignin
First, 15 g constant weight DES-modified lignin was dissolved with DMSO (50 mL) in a four-necked flask fitted with a mechanical stirrer and a reflux condenser. Then, 200 μL 1-methylimidazole was added dropwise to the lignin solution as a catalyst for the esterification reaction with maleic anhydride (20 g) which followed immediately. The reaction mixture has then been heated to 80°C under mechanical stirring for 3 h. The maleated lignin was then recovered by cooling and precipitation at pH 3 and the excess maleic anhydride was removed by washing with water. The maleated lignin solids recovered underwent a final drying at 60°C.
2.2.3 Synthesis of Lignin-Glyoxal (LG) DES-Lignin-Glyoxal (DLG), DES/MA-Lignin-Glyoxal (DLMG) Resins
The lignin resins were prepared according to the method presented in [15]. First, 25 g of different types of lignin (pristine lignin, DES-modified lignin and DES/MA-modified lignin) was separately dissolved in 85 mL of 1 M NaOH in a beaker and transferred to a 250 mL three-necked round-bottom flask equipped with a thermometer, mechanical stirring, and a condenser. For maintaining a uniform temperature around the flask, a dry bath stacker was used. Then, glyoxal was gradually added to the lignin solution. The temperature was increased steadily from room temperature to 65°C in 30 min while stirring at 600 rpm using a digital hot-plate stirrer. Once the solution temperature reached 65°C, it was kept at 65°C for 30 min, after which 40 mL of 1 M NaOH solution was added to the flask. Then, the temperature was increased gradually to 90°C, where it was held constant for 2 h. The formulated resin was next cooled down to room temperature. It should be noted that the molar ratio of glyoxal to lignin was kept constant at 2:1 for all the resins.
2.2.4 Addition of Chitosan to LG Adhesives
First, 30 g of chitosan in 30% citric acid solution at 25°C with stirring at 600 rpm. After that, chitosan solution was added to the LG resin and mixed well at 40°C and 500 rpm for 1 h.
The synthesis of the Phenol formaldehyde (PF) adhesive control has already been described in detail [16,17]. To a sufficient amount of molten phenol, formalin 37% was added dropwise and each of distilled water and methanol was put in a flask. The mixture was then heated to 80°C. Then, sodium hydroxide was added and the reaction was continued for 4 h at 80°C. After 4 h of resinification reaction, the flask was cooled to room temperature to stop the reaction. The methanol in the flask was removed by rotary evaporation at 60°C under reduced pressure to produce a dark-brown and viscous aqueous resin solution. The molar ratio of formaldehyde to phenol was 2:1.
2.2.6 Physico-Chemical Characterization of the Synthesized Resins
The resins’ physico-chemical characteristics, namely viscosity, density and percentage solids content, have been determined according to relevant standards. Their percentage solids content has been measured according to China’s National Standard GB/T 14074-2017 [18], while the viscosity has been obtained by Ford cup Nr 4, and the resin solution density by hydrometer.
2.2.7 Fourier Transform Infrared Spectrometry
The changes in the chemical structure of lignin after DES modification and MA functionalization have been analyzed by Fourier Transform Infrared spectrometry (FTIR) (Shimadzu FTIR 8400S, Nakagyo, Japan). The chemical structure of chitosan and modified lignin-–chitosan mixture was also analyzed by FTIR in the 400 to 4000 cm−1 range with the spectra normalized to a constant peak. KBR pellets containing 1% weight of the powdered resins were prepared and used for the purpose.
2.2.8 CP-MAS 13C NMR Spectrometry
After glyoxalation, and hardening for 2 h at 105°C the lignin resins were finely ground for NMR spectroscopy and examined by 13C NMR on a 400 MHz spectrometer Bruker AVANCE II (Bruker, Wissembourg, France). The parameters used were: 100.6 MHz frequency, 12 kHz spin, 1 s-depending 1H times of relaxation (t1), 1 ms contact time with the inversion-recovery pulse sequence, 15000 transients, and 78 kHz decoupling. Tetramethyl silane (TMS) was used as control to determine the chemical shifts. Spectra accuracy was +1 ppm. Suppression of spinning sidebands was used.
2.2.9 Matrix-Assisted Laser Desorption Ionization Time of Flight (MALDI TOF) Mass Spectrometry
A KRATOS Kompact MALDI AXIMA TOF 2 spectrometer (Kratos Analytical, Shimadzu, Manchester, UK) has been used in a positive polarity tuning mode. A 337 nm wavelength pulsed nitrogen laser for a 3 ns pulse was the irradiation source. The parameters used were: positive polarity, linear ions flight path, accelerate volt of 20 kV, with pulse numbers in each spectrum between 100–150 pulses. Acetone was used to dissolve the lignin water solution specimens (4 mg/mL, 50/50% volume). 2,5-dihydroxy benzoic acid was the matrix used. To an acetone solution to which were added the sample solutions (10 mg/mL in acetone) the matrix was added. 10 mg/mL of NaCl in water was added to the matrix to enhance ion generation. 3 parts matrix + 3 parts resin solution + 1 part NaCl solution were premixed. 0.5 to 1 µL of this mix was placed on the MALDI target, the sample was then evaporated on the supporting target and this with the dry droplet was finally placed in the mass spectrometer.
2.2.10 Differential Scanning Calorimetry (DSC) Analysis
The changes in glass transition temperature (Tg) of oven-dried lignin before and aftermodification were determined by DSC using a NETZSCH DSC 200 F3 Model thermal analyzer (Netzsch-Geratebau GmbH, Selb, Germany). Also, the changes in curing temperature of the prepared resins were determined by a DSC analyzer. For the DSC scans 5 mg of freeze-dried samples were placed on the aluminum support followed by heating under a nitrogen 60 mL/min flow rate in the 25°C–250°C range with a 10°C/min rate of heating, to determine the lignin resins curing temperature.
2.2.11 Gel Permeation Chromatography
Gel permeation chromatography (GPC) (LC-20A, Shimadzu, Kyoto, Japan) was used to measure the lignin samples’ dispersion and molecular mass before and after modifications. The lignin was acetylated before the molecular mass test and placed as a 5 mg/mL of lignin in tetrahydrofuran (THF). The tests were conducted at a 40°C temperature of the column, with THF as the eluent at 1 mL/min rate of flow. An external standard method using a standard of polystyrene monodisperse was used to determine the molecular weight average for lignins pristine and modified.
An already reported and codified method was used to prepare plywood [15,16] bonded with the resins prepared above. 6% moisture content 400 mm × 400 mm × 2 mm Fagus orientalis (beech) veneers were spread single layer with 310 gm−2 of gluemix, assembled in 3 layers, and hot pressed at 180°C for 7 min at 6 MPa pressure. The 3-layer plywood panels were then stacked for 24 h. Three random samples of each panel bonded with each adhesive were chosen and their dry shear strength was then tested.
Shear specimens were prepared from each board to test them dry according to EN 314-2 [19]. Short-term (24 h) water absorption tests were performed according to ASTM D4442-07 [20]. They underwent two weeks of conditioning at 23°C +/−2°C and at a relative humidity of 60% ± 5% and then 5 randomly selected specimens for each treatment were selected. The dry shear strengths were measured in tensile with an Instron 1186 universal testing machine (Instron, High Wycombe, UK).
The effects of the prepared resins on the plywood properties were examined by SPSS software with a two way 95% level of confidence ANOVA (two-way variance analysis). The Duncan Test was used to compare the values obtained when significant differences were remarked by the ANOVA analysis.
The infrared spectrum of the pristine lignin (PL), DES-modified lignin (DL), and DES-MA modified lignin (DML) are shown in Fig. 4. It can be seen that the chemical modifications of lignin shift the vibration signals slightly to the right and/or left. The modification of lignin by DES increases the proportion of its aromatic hydroxyls (1210 cm−1) by a decrease in its methoxyl (2817 cm−1) proportions when compared to their proportions in pristine lignin, as shown in Fig. 4. Thus, this confirms that by modifying lignin by a DES does in effect cleave some of the lignin ether (β-O-4′) bridges as well as demethylate a proportion of the lignin methoxy groups. The DES treatment of lignin thus influences its side chains and does not affect the aromatic ring. Previous research indicated that modification by DES is an effective and convenient way to increase the phenolic hydroxyls content of lignin [21,22]. These works indicated that the phenolic monomer content of the DES-treated lignin is the same as obtained by the lignin phenolation method [21,22].

Figure 4: FTIR analysis of the pristine lignin (L), DES-modified lignin (DL), and DES-MA modified lignin (DML).
Conversely, it can be seen in Fig. 4 that the DES-MA-modified lignin (DML) presents the highest peak intensities for the 1700 , 2800 and 1200 cm−1 bands compared to the non-maleate DES-treated lignin and pristine lignin, respectively. This means that the content of −COOH, C=C and C-O groups in the lignin markedly increases after the DES-MA treatment. Thus, the grafting of the maleic anhydride increases the number of reactive sites of the material by introducing –COOH, C=O and C=C linkages in the lignin structure. The DES-maleated lignin then with a higher proportion of reactive sites can then be linked to other monomers to form improved prepared adhesives [23]. The DML-treated lignin also showed the lowest peak at 3420 cm−1 of all the lignins tested. This means that a proportion of hydroxyl groups in lignin were consumed by the reaction with the ChCl–ZnCl2 DES and with maleic anhydride. This reduction is also due to the esterification reaction. In general, the reduction of these bands can reduce the polarity of the lignin as maleic groups or carbonyl groups replace the hydroxyl groups. Cleavage of the bonds in lignin by the DES treatment helps by increasing the reaction of the functional groups of maleic anhydride with the DES-modified lignin.
Conversely, Fig. 5 shows the FTIR analysis of chitosan and DML-modified lig-nin-chitosan mixture. It can be observed that some of the main chitosan and DML modified lignin-chitosan mixture peaks are different. The intensity of the peaks related to the amine groups, these being the main peaks of importance for chitosan have been decreased in the mixture. Thus, the FTIR results confirm the formation of chemical bonds between the amine (NH2) groups of chitosan and two unsaturated double bonds (carboxylic acid) of MA-DES modified lignin. Moreover, the hydroxyl groups of the chitosan can crosslink by hydrogen bond to the functional groups of modified lignin and produce a mixture with excellent cohesion properties.

Figure 5: FTIR analysis of chitosan and DML—chitosan mix.
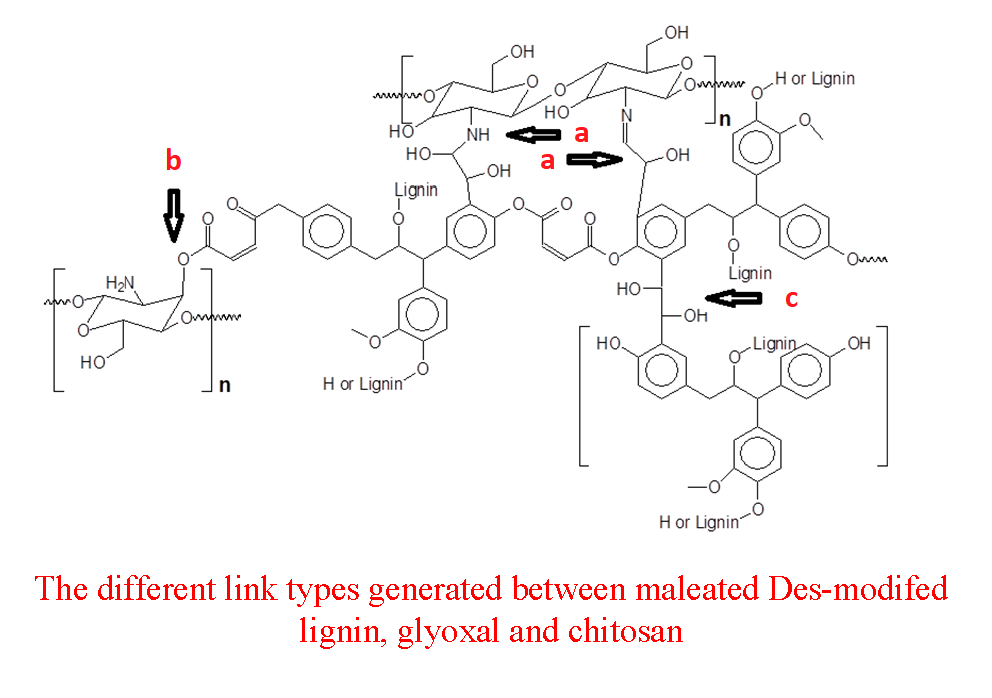
Thus, from the indications from the FTIR and the following analysis the type of cross-links occurring between DES-treated maleated glyoxalated lignin and chitosan can schematically be represented as in Fig. 6.

Figure 6: A schematic example of the most likely cross-linking bridges of glyoxalated maleated lignin with chitosans, indicated by arrows, such as: (a) Glyoxal bridges between the aminogroups of chitosan and the aromatic ring of lignin. (b) Ester bridges between the unreacted maleic acid moieties of maleated lignin and the hydroxyl groups of chitosan (c) Glyoxal bridges between lignin aromatic rings.
3.2.1 Reaction of DES Lignin with Glyoxal
In Table A1 (Appendix A) the assignment to the molecular structures formed are reported as guaiacyl (G), siringyl (S) units, and as H for lignin aromatic units without methoxyls. The “classical” manner to indicate the interunit bridges as β-O-4, α-O-4, β-β, β-5 and 5-5′ is used. These bridges are more easily named according to the Table A2 (Appendix B) as G-HF. With HF, GF and SF are indicated:

The three fractions (I), (II), and (III) linked -β-1 to another lignin unit or fraction of lignin unit.
Comparing the results of Table A2 and Fig. 7 with Fig. A1 and Table A1 (Appendices A and B) and Fig. 8, evident changes appear to occur by glyoxalating lignin. Thus, some of the original lignin compounds have disappeared due to both the effects of the long period of heating and the markedly alkaline environment used. For example, the 231 Da S-type monomer has disappeared. Conversely, the HF, GF and SF fractions do appear, thus inferring that a proportion of methoxyls have been demethylated and that the C3 aliphatic chain has also been cleaved from some lignin units aromatic ring. All this being just internal lignin rearrangements independent from any reaction with glyoxal. A marked increase can also be noticed in dimers in which β1-bridged to not cleaved lignin units of SF, GF and HF fractions occur. This is a well-known cleaving and recombining mechanism of lignin rearrangement [24]. Different types of self-condensation rearrangement have also occurred in just the lignin as a consequence of its treatment by heat. Thus, even structures undetected in the original lignin like the 750.3 Da tetramer of structure G-G-G-G, not detected in the original lignin appear to have been generated by this route.

Figure 7: MALDI TOF spectra of Glyoxalated DES-treated lignin (a) 60 to 280 Da range (b) 260 to 1100 Da range.

Figure 8: 13C CP-MAS NMR of glyoxalated lignin.
Conversely, several different condensation compounds appear to have been generated by the reaction of glyoxal with the lignin. Thus, aldehyde groups from glyoxal have reacted with ease with phenolic structures such as (I), (II) and (III) generating a number of different oligomers. High proportions of species presenting a hydroxyglyoxyl group still reactive such as structure (IV) have thus been generated, as observed from the relative peak intensities in Table A2 (Appendix B) and Fig. 7a,b, like the 197.6, 198.7 and 199.9 Da species.

These are all assigned to type (IV) structures in which one of the glyoxal aldehyde groups occurs as the hydroxymethyl group as in general is the case in water.
Type (IV) structures being present, these generated by precipitation due to variations of pH after lignin glyoxalation, are also confirmed by the 13C NMR. The 61.9 ppm shift in Fig. 3 is assigned to the hydroxymethyl (−CH2OH) group in structure (IV). The split peak at the 72.9 ppm shift is instead assigned to the aliphatic hydroxymethyl group (−CH2OH) of the Cγ of unreacted lignin units (Fig. A2, NMR of unreacted lignin, Appendix A), which generates the additional 70.3 ppm shift attesting to the existence of the reacted glyoxal >CHOH groups in structure (IV).
Oligomers of higher molecular weight and also hydroxyglyoxalated appear to be present like the 602.2 and 661.6 Da trimers and the 817.5 and 971.0 Da tetramers (Fig. 7). All these appear to be present in relatively small proportions, as attested by the intensity of their peaks. The peaks at 197.6, 198.7, 199.9 and 255.9 Da, are assigned to hydroxyglyoxalated monomers while dimers hydroxyglyoxalated like the 398.5 Da peak predominate judging by the intensities in Table A2 and Fig. 7 a,b. Even compounds that are twice hydroxyglyoxalated are present like the one at 255.9 Da although they are relatively rare.
Compounds where glyoxalene bridges have formed linking the aromatic rings of two separate lignin units also occur. The peaks at 284.6, 312.1, 372.6 and 430.6 Da are assigned to species presenting glyoxalene bridges of this type, predominantly those linking the (I), (II) and (III) fractions, judging from the relative intensities of the 284.6, 312.1, 372.6 and 430.6 peaks mentioned above. Structure (V) is an example of the species assigned to the 312.1 Da peak.

Such types of structure as (V) are confirmed in the 13C NMR Structures in Fig. 8 by the unreacted lignin 32.7–33.3 ppm shift increase to an intensity higher than the -CH- glyoxalene bridges 30 ppm shift between two aromatic rings in structure (V). The structure (V) hydroxymethyl group (−CH2OH) contributes to the 70.3 ppm band as well.
Present are also oligomers of lignin units bridged by one or several glyoxalene bridges like: (i) the notable proportion of trimers generated by a lignin dimer condensation through a glyoxalene bridge with a monomer of lignin such as the 552.5, 574.4 and 606.3 Da species, equally supported by the NMR spectra as structure (V); (ii) the lower proportion of differently generated tetramers by bridging by a glyoxalene link either two lignin units dimers or even by bridging a monomer with a trimer as for 794.9 Da species; (iii) two glyoxalene bridges linking two lignin oligomers to generate pentamers as for like the case of the 993.5 Da species.
Confirmation of other types of structural intermediates generated by glyoxalation of lignin is obtained by 13C NMR The 105–115 ppm shift of unreacted ortho and para lignin aromatic ring sites showed a marked decrease in intensity by glyoxalation when comparing them to the 125–135 ppm shift. The unreacted ortho site shift at 105 ppm appears to be particularly affected by its decrease (see Figs. 8 and A2). Concurrent with this is the 125–135 ppm shift marked intensity increase assigned to reacted ortho and para sites, but especially of the ortho sites of the lignin aromatic ring, showing that this is a major site for glyoxal reaction. All this confirms and supports the MALDI TOF findings. After glyoxalation the methylene groups pattern also differs due to the appearance of the 13.9–14.3 ppm small shift assigned to -CH2-groups sterically crowded. The high intensity 181.8 ppm shift is also interesting, as it is absent in lignin not reacted. It is attributed to the generation of quinones from the phenolic hydroxyls of lignin. The shift assigned to the −OCH3 methoxyls on the aromatic ring of lignin units at 55.3–55.5 ppm also decreases markedly (Figs. 8 and A2). The increased reactivity of lignin’s phenolic rings is inferred by the simultaneous, although moderate 148–150 ppm shift increase assigned to the aromatic carbons supporting lignin hydroxyls.
Finally, a lower molecular weight DES lignin reaction with glyoxal also generates depolymerisations and recombinations just due to the lignin itself, glyoxal condensing with cleaved fractions and uncleaved lignin units rather than generating different hydroxy-glyoxalated and glyoxalene bridged compounds. An increased viscosity of the reaction mix is the logical consequence of this as the reaction invariably leads to the generation of oligomers of ever increasing molecular mass as inferred by the average masses shown in Table 1. Consequently, reaction lengthening yields glyoxalated lignin oligomers of progressively higher viscosity as condensation by glyoxalene bridges increases. The viscosity results in Table 2 confirm the results inferred by Table 1 that the viscosity increases of the lignin used as the time of glyoxalation become progressively longer. Too extensive a glyoxalation, for too long a period, however, does not improve the performance of glyoxalated lignin as an adhesive, as this relies on the reaction of the hydroxyglyoxyl groups [25] with the still reactive sites of lignin aromatic nuclei [26]. This is also shown with even more reactive phenol nuclei such as those in tannin-lignin-based formulations [26]. The conclusion is that the time of glyoxylation of the lignin for optimal performance might well depend quite closely on the type of lignin used, according to the level of reactivity this presents. Hence, more reactive lignins might need shorter glyoxalation times than less reactive lignins for optimal performance.

3.2.2 Reaction of Chitosan with Glyoxal
Structures with chitosan oligomers reacted with one or more glyoxal molecules or with glyoxal oligomers due to aldol condensation can also be observed, such as the species at 617, 634, 652, 671, 685, 698, 708, 778, 795 and 885 Da as listed in Table A3 (Appendix B) from the spectrum in Fig. 9, such as for example the structure VI at 617 Da and structures VII and VIII at 652 and 795 Da respectively:

Figure 9: MALDI TOF spectrum of the reaction of chitosan with glyoxal.
Structures VII and VIII indicate clearly that glyoxal not only reacts with chitosan, but that it also cross-links separate chitosan chains through the main type of cross-linking bonds already described in the literature [13]. It must be pointed out that these species have also been observed for the first time by MALDI TOF spectrometry, adding further confirmation to the NMR-determined previous literature.
Glyoxal may also undergo aldol condensation, giving several species that can also be observed by MALDI TOF in the reaction between chitosan and glyoxal as shown in the relevant literature [27], although this has not been observed in the present work.
In the same way, the aldehyde groups in the structure can further react with another chitosan molecule to form tridimensional spatial network structures, thereby providing good bonding performance. In addition, it needs to be pointed out that the structure of -N-CHOH-from the reaction of amino and aldehyde groups easily occurs by water elimination in hot pressing conditions to also obtain C=N structures, as already proven in the literature [27,28].
The glass transition temperature (Tg) of the pristine lignin (PL), DES-modified lignin (DL) and DES-MA modified lignin (DML) is shown in Fig. 10. The DSC curves show that the Tg of pristine lignin is 92°C while DES-treated lignin has a lower Tg value (82°C). In fact, the treatment of lignin by ChCl–ZnCl2 causes a significant decrease in the lignin Tg. The molecular weight (MW) is a determinant factor having an influence on lignin’s Tg. For this reason, the molecular weight of lignin before and after the different treatments was examined by GPC. Number average molecular weights (Mn), weight-average molecular weights (Mw) and Polydispersity Indexes (PDI = Mw/Mn) of the PL, DL and DML are shown in Table 3.

Figure 10: DSC analysis of pristine lignin (L), DES-modified lignin (DL) and DES-MA modified lignin (DML).

It can be seen that compared to L and DML, DL had both the lowest average molecular weight indexes (Mw and Mn) and polydispersity coefficient. Lignin inherently is heterogeneous but the homogeneity of lignin can be significantly increased by the DES treatment, based on the GPC results obtained. Previous research [6] has already demonstrated that deep eutectic solvents can markedly cleave a proportion of some of the chemical bridges linking lignin units. Thus, the Tg of DES-modified lignin significantly decreases by the DES-induced reduction of molecular size and weight.
Conversely, it can be noted that by reacting maleic anhydride with the DES-treated lignin to obtain DML causes an increase of both the average molecular weight (Mw and Mn) as well as of the polydispersity index. Table 1 shows that Mw and Mn reached respectively 2921 and 2242 by esterification of the DES-treated lignin. The polydispersity index of DML also decreased to 1.3. These results show that maleic anhydride can link to DES-treated lignin by reactions involving either the C=C, COOH or C-O groups, or several of them, and thus increase the molecular size and weight of lignin. In fact, the generation of a more extensive and stiff network formed by the reaction of DES-treated lignin and maleic anhydride can decrease the mobility of lignin. Moreover, the covalent bonds formed between lignin and maleic anhydride are also rather strong. Hence, DML needs more heat (Tg) to change to a solid state or to a rubbery state than L and DL. The hydroxyl functional groups present in the DES-treated lignin structures have already been shown to provide the possibility for esterification with maleic anhydride [29].
DSC analysis of pristine lignin/glyoxal (PLG), DES-modified lignin/glyoxal (DLG), DES-maleated lignin/glyoxal (DMLG) and DES-maleated lignin/glyoxal/chitosan (DMLGC) resins was also carried out (Fig. 11). Fig. 11 shows that the LG resins containing pristine lignin had the highest temperature peak (119°C) among all prepared resins. The temperature peak respectively decreased to 113°C and 109°C by the treatment of lignin by DES and DES/MA. The reaction of DES with lignin decreases the proportion of lignin methoxy groups by cleaving the inter-unit bridges as well as a number of methoxy groups generating a higher proportion of aromatic hydroxyl groups. This results in a decrease in the lignin molecular weight. For this reason, the chemical reactivity of DES-modified lignin improves significantly and it shows a lower temperature peak of the resins prepared from it than the resins synthesized from pristine lignin. Conversely, DMLG has a higher curing rate than the DLG and PLG resins indicates that the incorporation of DES-maleated lignin accelerates the curing of the resin. This can be attributed to the higher reactivity of the functional groups in DES-maleated lignin. There are highly reactive sites in DES-maleated lignin, which cause lignin to easily full cross-link with ease during reaction. One of the main defects of bio-sourced wood adhesives, especially of wood adhesives based on lignin, is their higher temperature of curing than for synthetic resins. Nonetheless, DES-maleated lignin can produce a lignin-based wood adhesive with a sensibly lower curing temperature, as shown by the results of the present work.

Figure 11: DSC analysis of LG, DLG, DLMG and DLMGC resins.
Fig. 11 also indicates, moreover, that after the addition of chitosan as a crosslinker to the DLMG resin the peak temperature (Tp) of the resin decreased to 103°C. These results confirm the good crosslinking reaction of C=C, C=O and COOH groups of the DES-maleated lignin with the amine groups (NH2) of chitosan, and perhaps also with its hydroxyl groups. This high proportion of bridges formed among lignin, glyoxal and chitosan led to the reaction taking place at a lower temperature. Decreasing the peak temperature of lignin-based adhesives by the addition of chitosan has a positive effect on the development of their applications.
3.4 Physicochemical Properties of Resins
The physicochemical properties of the resins prepared are reported in Table 4. The viscosity, solids content and density are lowest of all the resins for the control LG resin at respectively 376 mPa.s, 28% and 1.05. The low chemical reactivity of the LG resin due to both lignin and glyoxal is the cause of its weak physicochemical properties of the LG resin as already reported by a number of research groups [30]. Based on the physicochemical properties obtained, the DLG resin containing DES-modified lignin has higher viscosity, solid content and density than those made from pristine lignin. The higher measured physicochemical properties of the DLG resins are due to chemical effects such as the increasing proportion of cross-linkages between DES-modified lignin and glyoxal. The use of the lignin DES treatment has been shown that it can cause an increase in the lignin-based resin viscosity and solid content [6].

Conversely, it can be seen in Table 4 that the DMLG resins have higher viscosity, higher solids content and density than the DLG and PLG resins. The aliphatic and aromatic hydroxyl groups present in lignin provide a possibility for an esterification reaction with maleic anhydride, to introduce maleate half-ester groups into a number of lignin units. Previous research confirmed that the double bond of the maleate half-esters is also very reactive [29]. The cleavage of the β-O-4 linkages in the lignin structure by the ChCl–ZnCl2 treatment before functionalizing by MA can also increase the amount of reactive sites of the lignin structure compared to pristine lignin. The reason for the improvement of the physicochemical characteristics of the resin is the consequence of the high reactivity of the MA C-C double bond allowing its graft on the available reaction sites in DES-treated lignin.
Table 3 also shows that all measured physicochemical properties (viscosity, solid content and density) of the resins were increased by the addition of chitosan. This can be attributed to chitosan inducing an increased density of cross-linking in the resin. Chitosan has been shown to form several types of strong chemical bonds, as well as electrostatics and hydrogen bonds with lignin [13]. These interactions can clearly improve the properties of lignin-based resins. Based on finding of this research, some physiochemical properties of the DLMGC resins are close to those of phenol-formaldehyde (PF) resin (Table 4).
3.5 The Mechanical Properties of the Bonded Panels
The plywood dry and wet strength and wood failure percentage glued with the synthesized resins have been reported in Table 5. The plywood panels bonded with the LG resin presented the lowest dry and wet shear strength and wood failure percentage among all the panels prepared. Using the DES-modified lignin in the PLG resin increases the shear strength and wood failure percentage of plywood. Breaking the β–O–4 and β–β linkages within the lignin by the DES treatment increases the phenolic hydroxyl content of the lignin. As regards the high reactivity of these phenolic hydroxyl groups, DES-treated lignin can react more extensively with glyoxal than pristine lignin. For this reason, DES-treated lignin can increase cross-linking between the different constituents of the adhesive. The differences in physicochemical properties of these types of resin can also help to explain the results obtained. So far, the positive effect of lignin modification by deep eutectic solvent on the mechanical properties of the panels bonded with such resins has been reported by several researchers [31,32].

Conversely, the plywood panels bonded with DMLG resin had higher shear strength and wood failure percentage than those bonded with DLG and PLG resins, respectively. The plywood bonded with the LG adhesive presented a lower dry and wet shear strength than those using the DMLG adhesive, the advantage of this latter on the former being of 66%. The DMLG bonded plywood has greater shear than those made with the DLG and PLG resins, which can be related to the difference in the type and proportion of chemical bonds and to the physicochemical quality of the resins. Maleation of DES-treated lignin causes maleic esterification with the available aromatic and aliphatic hydroxyl groups (generated on lignin by the DES treatment). Maleate cross-linking bridges between modified lignin and glyoxal, as well as between two lignin units can probably also occur, which can contribute to increasing the mechanical strength of the final adhesive network. Younesi-Kordkheili and Pizzi investigated the most important methods for lignin modification to allow its use as a phenol substitute in PF resins [33]. This work showed that even particleboard panels bonded when using a maleated lignin-phenol-formaldehyde resin had the highest mechanical strength among all the lignin modification methods.
The addition of chitosan as a cross-linker has a significantly positive effect on the mechanical strength of the resins prepared. In particular, the dry shear strength and wood failure percentage of the plywood panels bonded using the DMLGC resin containing chitosan are found to be 1.3 ± 0.08 MPa and 85%, respectively. Based on the findings of this research, there are some significant differences between the results obtained with the DMLG resin containing chitosan and those with a commercial PF resin (Table 4) but especially in the case of the dry shear strength these are still tolerable. This is due to the amide bond formation in the cross-linked system, along with the ability of chitosan to form electrostatic interactions and hydrogen bonds with lignin due to its cationic nature and the presence of polar functional groups [10]. The interactions between the DES-maleated lignin and chitosan can stabilize the lignin network and further enhance its structural integrity. Patel et al. also suggested that chitosan can interact with wood primarily through hydrogen bonding and electrostatic interactions, with the electrostatic charges aiding its retention on the wood surface [34]. Thus, chitosan not only can act as a cross-linker but it can also improve the adhesion strength through electrostatic and hydrogen bond interactions. Previous work has indicated that chitosan can form strong bonds with wood due to the intermolecular, electrostatic and other secondary interactions that occur between them [34]. The shear strength differences in Table 5 of the plywood bonded with the DMLG and DMLGC resins appear not to be significant. It means that hydrophilic intrinsic nature of chitosan can influence the wet shear strength.
3.6 Physical Properties of the Panels Prepared
The water resistance of wood-based panels is one of the important parameters for evaluating the bond strength and applicability of wood adhesives. Figs. 12 and 13 show the short-term (24 h) water absorption and thickness swelling of the plywood panels bonded with PLG, DLG and DMLG resins. The plywood bonded with the LG resin presented the highest thickness swelling and water absorption of all the other resins. The low-dimensional stability of the panels bonded with the PLG resin can be ascribed to poorer and weaker cross-linking between pristine lignin and glyoxal. An improvement is also observed in the plywood bonded with lignin resins pre-treated with ChCl–ZnCl2 DES and the use of modified lignin. This is probably due to the higher proportion of reactive sites in DES-treated lignin compared to pristine lignin.

Figure 12: Water absorption (24 h) of the plywood panels prepared. Different letters for the bars indicate that they are significantly different (p < 0.05).

Figure 13: Thickness swelling (24 h) of the prepared plywood panels. Different letters for the bars indicate that they are significantly different (p < 0.05).
Conversely, the DMLG bonded plywood had lower water absorption and thickness swelling percentages than those bonded with DLG and PLG resins, respectively. Different bridge types between maleated DES-treated lignin with glyoxal compared to pristine and DES-treated lignin with glyoxal can influence the dimensional stability of the plywood panels prepared. The higher level of cross-linking of the maleated-DES-treated lignin with glyoxal also helps to reduce water absorption and thickness swelling in plywood panels. Fig. 13 also shows that the addition of chitosan as a cross-linker to the DMLG resin decreases the plywood water adsorption-dependant dimensional stability. Unlike lignin, chitosan has a hydrophilic nature and can absorb water and swell markedly reducing the wood-based panels’ dimensional stability.
In this research, bio-sourced adhesives designed through lignin for wood bonding have been investigated. To achieve this aim, lignin was modified in two stages with DES and maleic anhydride, respectively. Then, DES-maleated lignin was reacted with glyoxal as a nontoxic aldehyde to prepare lignin-glyoxal resin. Chitosan was also added as a bio-cross-linker to prepare adhesives for improving the mechanical properties of the panels bonded with the resin. The following conclusions can drawn from the results:
• DSC analysis indicated that althoughconclusions DES-treatment can decrease Tg, molecular weight (Mw and Mn) and polydispersity coefficient of lignin the reaction of DES-treated lignin with maleic anhydride increases Tg, molecular weight and polydispersity coefficient.
• FTIR analysis of lignin indicates that DES-modified lignin has a higher proportion of phenolic hydroxyl groups compared to oxidised lignin. Also, the content of COOH, C=C and C-O groups in lignin units markedly increases after DES-MA treatment.
• 13C NMR and MALDI TOF analysis be drawn from show that both DES lignin and chitosan readily react and link with glyoxal and indicate the type of structures that are formed.
• Due to maleic anhydride having a highly reactive aromatic ring and unsaturated double bonds, it can easily graft to available reaction sites in DES-modified lignin. For this reason, based on the physicochemical properties obtained, the DMLG resin presents higher viscosity, solid content and density than the DLG and LG resins, respectively.
• Plywood panels bonded with the DMLG resin and chitosan had excellent shear strength, satisfying the relevant European Norm 314-2 (1MPa).
• The panels bonded with the DMLG resin showed good dimensional stability, although the addition of chitosan, due to its intrinsic hydrophilic nature can reduce short-term water absorption stability.
• This research indicates the potential of DES-maleated lignin for the preparation of wood adhesives to reduce the use of toxic synthetic adhesives and to develop bio-sourced and lower-cost bonding solutions for the wood industry.
• Both chitosan and lignin are inexpensive, renewable, and biodegradable, and the easy preparation of wood adhesives based on DES—modified lignin, glyoxal and chitosan can make the process scalable and eco-friendly for industrial applications.
Acknowledgement: Not applicable.
Funding Statement: This research has been funded by Semnan University, research grant no. 226/1404/T14041123. The MALDI and NMR spectra have been funded by the LERMAB, University of Lorraine, which is supported by a grant overseen by the French National Research Agency (ANR) as part of the ‘‘Investissements d’Avenir’’ program (ANR-11-LABX-0002-001) Lab of Excellence ARBRE).
Author Contributions: Conceptualization, methodology, investigation, testing, data interpretation, writing, Hamed Younesi-Kordkheili, Antonio Pizzi and Ghorban Niyatzadeh; instrumental chemical analysis and interpretation A. Pizzi. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: All contained in this article.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.


Figure A1: MALDI TOF Spectrum of untreated lignin in the 260 Da 750 Da range.

Figure A2: 13C CP-MAS NMR of unreacted lignin.


References
1. Amoneit M, Weckowska D, Spahr S, Wagner O, Adeli M, Mai I, et al. Green chemistry and responsible research and innovation: moving beyond the 12 principles. J Clean Prod. 2024;484(1):144011. doi:10.1016/j.jclepro.2024.144011. [Google Scholar] [CrossRef]
2. Sun X, Yang C, Zhang W, Zheng J, Ou J, Ou S. Toxicity of formaldehyde, and its role in the formation of harmful and aromatic compounds during food processing. Food Chem X. 2025;25(18):102225. doi:10.1016/j.fochx.2025.102225. [Google Scholar] [PubMed] [CrossRef]
3. Younesi-Kordkheili H, Pizzi A. Nanolignin modified by amino acid-derived deep eutectic solvent as formaldehyde scavenger in urea-formaldehyde resin. Int J Adhes Adhes. 2026;147(1):104283. doi:10.1016/j.ijadhadh.2026.104283. [Google Scholar] [CrossRef]
4. Younesi-Kordkheili H, Pizzi A. Reduction of formaldehyde emission from urea-formaldehyde resins with nanolignin modified by deep eutectic solvent. Eur J Wood Wood Prod. 2025;83(5):160. doi:10.1007/s00107-025-02308-x. [Google Scholar] [CrossRef]
5. Georgs V, Piili H, Gustafsson J, Xu C. A critical review on lignin structure, chemistry, and modification towards utilisation in additive manufacturing of lignin-based composites. Ind Crops Prod. 2025;233:121416. doi:10.1016/j.indcrop.2025.121416. [Google Scholar] [CrossRef]
6. Younesi-Kordkheili H, Pizzi A. Wood bio-adhesives made by polymerizing oxidized starch with deep eutectic solvent-modified lignin. Polymers. 2025;17(22):3023. doi:10.3390/polym17223023. [Google Scholar] [PubMed] [CrossRef]
7. Younesi-Kordkheili H, Pizzi A. Plywood bio-adhesives by oxidized lignin urea bridged with oxidized starch. J Renew Mater. 2026;14(1):1–10. doi:10.32604/jrm.2025.02025-0179. [Google Scholar] [CrossRef]
8. Subbotina E, Olsén P, Lawoko M, Berglund LA. Maleated technical lignin thermosets and biocomposites designed for degradation. ACS Sustaini Chem Eng. 2024;12(9):3632–42. doi:10.1021/acssuschemeng.3c06741. [Google Scholar] [CrossRef]
9. Pizzi A. Recent developments in advanced lignin-based adhesives and binders. In: Advanced lignin technologies. London: IntechOpen; 2024. doi:10.5772/intechopen.1004627. [Google Scholar] [CrossRef]
10. Ghahri S, Park BD. Bio-crosslinking of oxidized hardwood kraft lignin as fully bio-based adhesives for wood bonding. Int J Biol Macromol. 2025;309(Pt 3):142907. doi:10.1016/j.ijbiomac.2025.142907. [Google Scholar] [PubMed] [CrossRef]
11. Taha AG, Hezma AM. Synthesis and characterization of glyoxal-crosslinked chitosan with N-amino anthracene succinimide polymers for antimicrobial applications. BMC Chem. 2025;19(1):139. doi:10.1186/s13065-025-01505-2. [Google Scholar] [PubMed] [CrossRef]
12. Kaczmarek-Szczepańska B, Mazur O, Michalska-Sionkowska M, Łukowicz K, Osyczka AM. The preparation and characterization of chitosan-based hydrogels cross-linked by glyoxal. Materials. 2021;14(9):2449. doi:10.3390/ma14092449. [Google Scholar] [PubMed] [CrossRef]
13. Abdulhameed AS, Mohammad AT, Jawad AH. Modeling and mechanism of reactive orange 16 dye adsorption by chitosan-glyoxal/TiO2 nanocomposite: application of response surface methodology. Desalin Water Treat. 2019;164(2):346–60. doi:10.5004/dwt.2019.24384. [Google Scholar] [CrossRef]
14. Hong S, Lian H, Sun X, Pan D, Carranza A, Pojman JA, et al. Zinc-based deep eutectic solvent-mediated hydroxylation and demethoxylation of lignin for the production of wood adhesive. RSC Adv. 2016;6(92):89599–608. [Google Scholar]
15. Siahkamari M, Emmanuel S, Hodge DB, Nejad M. Lignin-glyoxal: a fully biobased formaldehyde-free wood adhesive for interior engineered wood products. ACS Sustain Chem Eng. 2022;10(11):3430–41. [Google Scholar]
16. Younesi-Kordkheili H, Pizzi A. Modification of nanolignin by deep eutectic solvent to improve the properties of phenol-formaldehyde resin. Eur J Wood Wood Prod. 2024;82(6):2099–108. doi:10.1007/s00107-024-02147-2. [Google Scholar] [CrossRef]
17. Younesi-Kordkheili H, Pizzi A. Properties of plywood panels bonded with ionic liquid-modified lignin-phenol–formaldehyde resin. J Adhes. 2018;94(2):143–54. doi:10.1080/00218464.2016.1263945. [Google Scholar] [CrossRef]
18. GB/T 14074-2017. Testing methods for wood adhesives and resins. Beijing, China: Beijing COC Tech Co., Ltd; 2017. [Google Scholar]
19. EN 314-2:1993. Plywood—bonding quality. Brussels, Belgium: European Committee for Standardization; 1993. [Google Scholar]
20. ASTMD4442-07. Standard test methods for directmoisturemeasurement of wood and wood basematerials. PA, USA: West Conshohoken; 2007. [Google Scholar]
21. Li P, Lu Y, Long G, Li S, Li K, Jiang B, et al. Structural characterization of acid DES-modified alkaline lignin and evaluation of antioxidant properties. Forests. 2023;14(3):550. doi:10.3390/f14030550. [Google Scholar] [CrossRef]
22. Zhang HN, Ren H, Zhai HM. Analysis of phenolation potential of spruce kraft lignin and construction of its molecular structure model. Ind Crops Prod. 2021;167:113506. doi:10.1016/j.indcrop.2021.113506. [Google Scholar] [CrossRef]
23. Lei H, Pizzi A, Despres A, Pasch H, Du G. Ester acceleration mechanisms in phenol-formaldehyde resin adhesives. J Appl Polym Sci. 2006;100(4):3075–93. doi:10.1002/app.23714. [Google Scholar] [CrossRef]
24. Fengel D, Wegener G. Wood: chemistry, ultrastructure, reactions. Berlin, Germany: Walter de Gruyter; 1989. [Google Scholar]
25. El Mansouri NE, Salvadó J. Analytical methods for determining functional groups in various technical lignins. Ind Crops Prod. 2007;26(2):116–24. doi:10.1016/j.indcrop.2007.02.006. [Google Scholar] [CrossRef]
26. Navarrete P, Pizzi A, Pasch H, Delmotte L. Study on lignin-glyoxal reaction by MALDI-TOF and CP-MAS 13C-NMR. J Adhes Sci Technol. 2012;26(8–9):1069–82. doi:10.1163/016942410X550030. [Google Scholar] [CrossRef]
27. Xi X, Pizzi A, Lei H, Zhang B, Chen X, Du G. Environmentally friendly chitosan adhesives for plywood bonding. Int J Adhes Adhes. 2022;112(2–3):103027. doi:10.1016/j.ijadhadh.2021.103027. [Google Scholar] [CrossRef]
28. Bajić M, Ročnik T, Oberlintner A, Scognamiglio F, Novak U, Likozar B. Natural plant extracts as active components in chitosan-based films: a comparative study. Food Packag Shelf Life. 2019;21(4):100365. doi:10.1016/j.fpsl.2019.100365. [Google Scholar] [CrossRef]
29. Younesi-Kordkheili H. Reduction of formaldehyde emission from urea-formaldehyde resin by maleated nanolignin. Int J Adhes Adhes. 2024;132(11):103677. doi:10.1016/j.ijadhadh.2024.103677. [Google Scholar] [CrossRef]
30. Younesi-Kordkheili H, Pizzi A. Preparation and properties of a modified corn flour-lignin-glyoxal as a Green wood adhesive. Int Wood Prod J. 2022;13(2):119–26. doi:10.1080/20426445.2022.2048338. [Google Scholar] [CrossRef]
31. Xian X, Wu S, Wei W, Zhang F. Pretreatment of kraft lignin by deep eutectic solvent and its utilization in preparation of lignin-based phenolic formaldehyde adhesive. BioResources. 2021;16(2):3103–20. doi:10.15376/biores.16.2.3103-3120. [Google Scholar] [CrossRef]
32. Younesi-Kordkheili H, Pizzi A. Synthesis of choline chloride-zinc chloride deep eutectic solvent-modified nanolignin/poly vinyl alcohol/glyoxal resin as a new green wood adhesive. Wood Mater Sci Eng. 2025;2025:1–7. doi:10.1080/17480272.2025.2523579. [Google Scholar] [CrossRef]
33. Younesi-Kordkheili H, Pizzi A. A comparison among lignin modification methods on the properties of lignin-phenol-formaldehyde resin as wood adhesive. Polymers. 2021;13(20):3502. doi:10.3390/polym13203502. [Google Scholar] [PubMed] [CrossRef]
34. Patel AK, Michaud P, Petit E, de Baynast H, Grédiac M, Mathias JD. Development of a chitosan‐based adhesive. Application to wood bonding. J Appl Polym Sci. 2013;127(6):5014–21. doi:10.1002/app.38097. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools