 Open Access
Open Access
ARTICLE
Cold-Induced Accumulation of Low-Molecular-Weight Dehydrins in Etiolated Wheat Seedlings: Relationship with Oxidative Stress Protection and Frost Tolerance
1 Yuriev Plant Production Institute, National Academy of Agrarian Sciences of Ukraine, Kharkiv, Ukraine
2 Czech Agrifood Research Center, Prague, Czech Republic
3 Department of Plant Protection, Poltava State Agrarian University, Poltava, Ukraine
* Corresponding Author: Yuriy E. Kolupaev. Email:
(This article belongs to the Special Issue: Plant Responses to Stress Factors)
Phyton-International Journal of Experimental Botany 2026, 95(3), 18 https://doi.org/10.32604/phyton.2026.079882
Received 30 January 2026; Accepted 04 March 2026; Issue published 31 March 2026
Abstract
Low temperature is a major abiotic stress factor inducing the accumulation of dehydrins in plants. Dehydrins are hydrophilic, heat-stable proteins implicated in plant stress responses; however, their synthesis under cold conditions during the early stages of wheat development has not been sufficiently studied. This study investigated the relationship between cold-induced dehydrin accumulation in etiolated seedlings and frost tolerance in wheat cultivars differing in their level of frost tolerance. Three-day-old seedlings of high frost-tolerant (high-FT) cultivars (Antonivka, Doskonala, and Nordika) and low frost-tolerant (low-FT) cultivars (Tobak, Tonnage, and Altigo) of Triticum aestivum L. were hardened at +3°C for six days. Dehydrin accumulation was analyzed by electrophoretic separation, while frost tolerance was assessed based on seedling survival following freezing at −4, −9, and −12°C. Cold-induced oxidative damage was evaluated by determining malondialdehyde (MDA) content in seedling shoots after freezing at −4°C. In control seedlings, dehydrins were barely detectable in all cultivars. Cold hardening at +3°C induced pronounced accumulation of dehydrins with molecular masses of approximately 46, 49.6, and 68 kDa in both high-FT and low-FT cultivars. In contrast, low-molecular-weight dehydrins (14–16 kDa) were detected predominantly in high-FT cultivars. Seedling survival after freezing at −12°C showed a strong positive correlation with total dehydrin content (r = 0.82). Even stronger correlations were observed between the content of low-molecular-weight dehydrins (14–16 kDa) and seedling survival after freezing at −9 and −12°C (r = 0.84 and 0.94, respectively). An inverse correlation was found between 14–16 kDa dehydrin content and MDA accumulation following freezing at −4°C (r = −0.87). These results indicate that low-molecular-weight dehydrins play an important role in protecting etiolated wheat seedlings from cold-induced oxidative stress and may serve as reliable biochemical markers of frost tolerance.Graphic Abstract
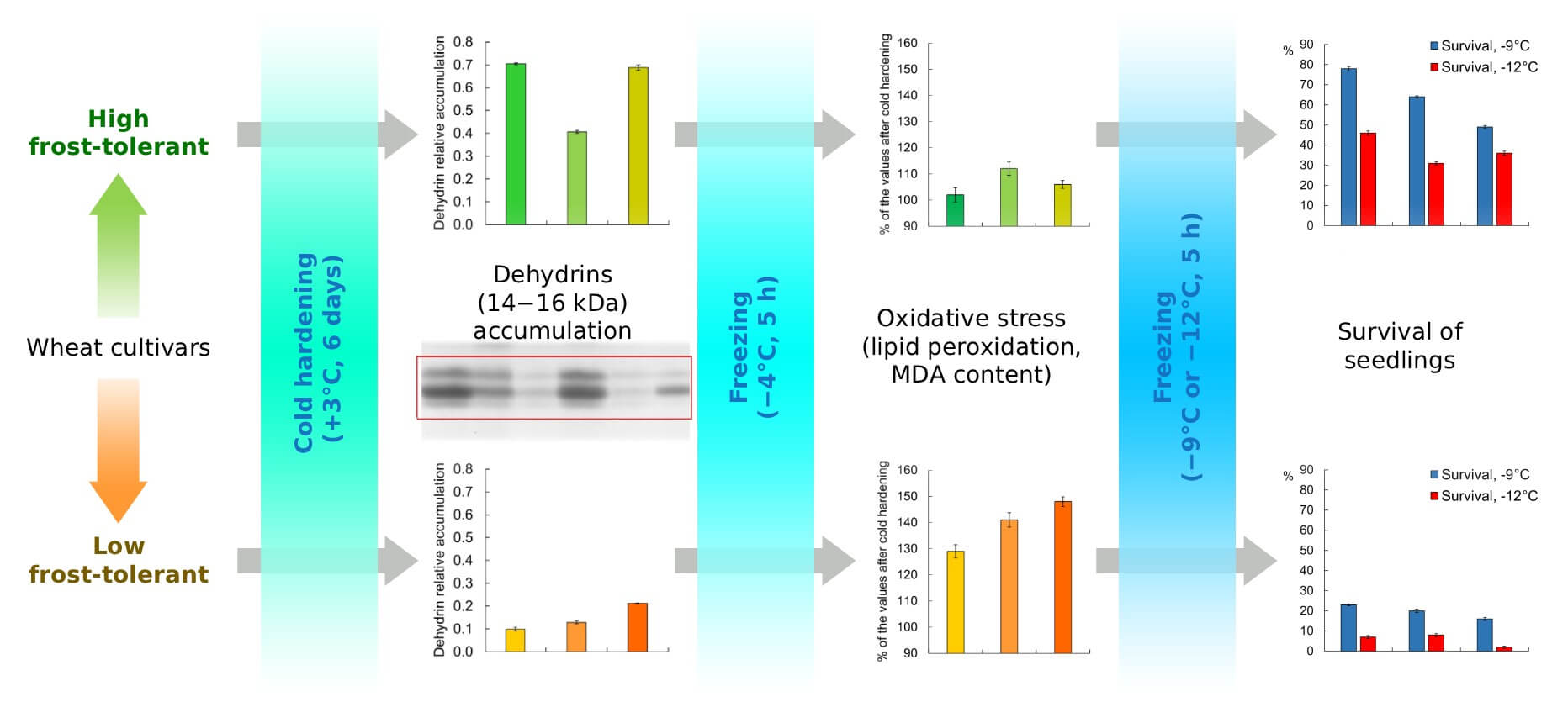
Keywords
Supplementary Material
Supplementary Material FileIn most regions where wheat is cultivated, including Europe, China, the United States, and Australia, plants are exposed to stress temperatures [1,2,3]. Despite the reduction in winter duration due to global warming, plant ecologists have noted a paradoxical relationship between rising air temperatures and the likelihood of cold damage to plants [4]. Global climate change poses a significant threat to the winter hardiness and grain yield of winter crops in temperate regions. Longer and warmer autumns followed by numerous abiotic stresses during the winter negatively affect the long-term survival of winter crops [5]. Since plants may be exposed to sudden low temperatures after warm periods, winter conditions can result in the loss of up to 90% of the winter wheat crop [6]. Additionally, warm winters prevent the selection of highly winter-hardy breeding lines, resulting in the accumulation of lines with low winter hardiness when developing new cultivars [7]. Thus, increasing the frost resistance of winter crops through classical breeding and genetic engineering methods remains an extremely relevant problem.
Plant frost resistance is a complex trait that depends on the functioning of many genes [8]. To date, approximately 450 genes involved in the cold response have been identified in wheat. However, their specific functions have yet to be determined [9]. The regulation of adaptive responses of plants to low temperatures is carried out by a multicomponent network of signaling molecules and plant hormones [10,11,12]. One of the main triggers for activating signaling processes appears to be the increase in membrane lipid bilayer rigidity induced by low temperatures. This change affects calcium channel state [10,13], modulates redox homeostasis [9,14], and develops other processes that transduce cold signals to the cell’s genetic apparatus [14,15,16].
The functioning of the aforementioned stress protection systems requires specific reprogramming of gene expression, which triggers transcriptional, biochemical, and physiological events [8,17,18,19]. Meanwhile, developing plant cold tolerance typically requires prolonged exposure to low, yet non-lethal, temperatures [8,20,21]. For instance, studies have shown that in winter wheat, genes that respond to cold stress typically begin to express one day after exposure to low (near-zero) temperatures [22].
The importance of primary stress-protective systems that ensure cold resistance of winter cereals has been established to date. Due to the increased reactive oxygen species (ROS) generation in plant cells when exposed to cold, activating the antioxidant system is critical for adaptation [11,23,24]. This system includes enzymes and low-molecular-weight antioxidants [23,25]. Another system involved in cold adaptation is the osmoprotective system, which prevents excessive cell dehydration during extracellular ice formation [23,26]. This system consists mainly of multifunctional low-molecular-weight compounds that perform osmoprotective, membrane-protective, and antioxidant functions [27]. Among such compounds, proline and certain other amino acids, as well as soluble carbohydrates, accumulate to the greatest extent under the influence of cold [11,26]. Another obligatory adaptive response to low temperatures is an increase in desaturase activity. Desaturases convert saturated fatty acids into unsaturated ones, allowing membranes to maintain necessary fluidity at low temperatures [28,29]. Finally, one more set of adaptive responses is associated with significant changes in the proteome [9,30,31].
Cold-induced changes in the proteome of plant cells are mediated by the increased expression of cold-regulated (COR) genes and genes that encode so-called late embryogenesis abundant (LEA) proteins [32]. The most significant protein group synthesized in response to cold is dehydrins, belonging to group 2 of the LEA family [33]. These intrinsically disordered proteins are highly hydrophilic and contain a conserved lysine-rich domain (K-segment) [34,35]. Dehydrin synthesis is usually induced by dehydrating stresses, such as drought, salinity, and exposure to low positive or negative temperatures [36]. Dehydrins appear to be multifunctional proteins [37]. They can perform chaperone functions associated with the presence of K-segments. Through their amphipathic α-helical structure, these segments can prevent protein aggregation [38,39,40]. The cryoprotective properties of dehydrins result from the presence of functional groups that interact electrostatically with negatively charged groups of membrane phospholipids. This helps maintain the fluidity of the lipid base and prevents membrane fusion [34,37,41]. Recently, data has emerged on the nuclear localization of certain dehydrins and their capacity to protect transcription factors and other nuclear proteins from damage, primarily from ROS, under stressful conditions [42]. Additionally, there is evidence suggesting that dehydrins are involved in regulating antioxidant enzyme gene expression, which may be another mechanism for preventing oxidative stress development [43].
In recent years, the relationship between dehydrin synthesis and the resistance of certain plant species to low temperatures has been actively studied. For instance, various wheat cultivars at the three-leaf stage exhibited cold-induced accumulation of substantial quantities of dehydrins with molecular weights of 40, 50, and 66 kDa in response to temperatures of 4°C and 9°C [44]. Previously, Kosová et al. [45] found a link in barley plants between the accumulation of dehydrin DHN5 (molecular weight 82 kDa) in leaves and frost resistance.
Etiolated wheat seedlings are used as model organisms to assess the frost resistance of cultivars and study their cold adaptation strategies [46]. However, the synthesis of dehydrins in these seedlings is not well studied. It has been shown that etiolated shoots accumulate virtually no dehydrins under optimal conditions. However, in response to stress factors causing dehydration, they accumulate dehydrins of various molecular weights, similar to green leaves [47]. Borovskii et al. [48] demonstrated the cold-induced accumulation of dehydrin-like proteins with molecular weights ranging from 41 kDa to 209 kDa in etiolated seedlings of winter and spring wheat. On the other hand, Stupnikova et al. [49] found seasonal accumulation of low-molecular-weight dehydrin proteins with molecular weights ranging from 12 kDa to 24 kDa in the crowns of wheat plants grown in field conditions, coinciding with the development of the highest frost resistance. Nevertheless, the relationship between etiolated wheat tissues’ ability to accumulate different dehydrin fractions and cultivars’ frost resistance has not yet been studied. The relationship between the accumulation of dehydrins by different wheat genotypes and their resistance to cold-induced oxidative stress remains unexplored, although information on the direct and indirect antioxidant effects of dehydrins [43] suggests its existence.
Our study aimed to investigate the relationship between the accumulation of dehydrins in etiolated seedlings of six winter common wheat cultivars in response to cold temperatures, their survival at below-zero temperatures, and their resistance to cold-induced oxidative stress.
2.1 Plant Material and Treatments
We used common winter wheat (Triticum aestivum L.) from the Gene Bank of the Czech Agrifood Research Center in Prague, Czech Republic, and the collection of the National Center for Plant Genetic Resources of Ukraine. The experiments were carried out on six cultivars of different ecological and geographical origins. According to the scientific literature and the Ukrainian Institute for Plant Variety Examination’s resources (Table 1), the cultivars differ significantly in frost tolerance. Antonivka, Doskonala, and Nordika cultivars exhibit high frost tolerance (high-FT cultivars). Tonnage, Tobak, and Altigo show low frost tolerance (low-FT cultivars).
Table 1: Data on wheat cultivars used in the study.
| Cultivar | Country | Frost resistance | Source |
|---|---|---|---|
| Antonivka | Ukraine | high (8.7 points*) | http://sort.sops.gov.ua/cultivar/view/12198 |
| Doskonala | Ukraine | high (8.8 points) | http://sort.sops.gov.ua/cultivar/view/12253 |
| Nordika | Czech Republic | above average (7.2 points) | http://sort.sops.gov.ua/cultivar/view/4802 |
| Tonnage | Denmark | below average (4.5 points) | http://sort.sops.gov.ua/cultivar/view/14669 |
| Tobak | Germany | low (2.5 points) | http://sort.sops.gov.ua/cultivar/view/3599 |
| Altigo | France | low (2.0 points) | http://cryo.org.ua/journal/index.php/probl-cryobiol-cryomed/article/view/2093 |
The seeds were disinfected for 15 min in a 1% sodium hypochlorite solution, thoroughly washed with distilled water, and germinated on paper in Petri dishes in a thermostat at 24°C. To cold-harden them, three-day-old, etiolated seedlings were incubated in a cold chamber (Frigera, Czech Republic) (without light) at 3°C for six days. The duration of exposure to low positive temperatures required for maximum development of frost resistance in etiolated seedlings was established in our previous experiments [46]. After cold hardening, one group of seedlings was used to analyze dehydrin content, while another group was used to evaluate cultivar frost resistance and oxidative stress levels caused by exposure to negative temperatures. The hardened seedlings were frozen for five hours in a dark (low temperature freezer VT146, Vestfrost, Denmark) at temperatures of −4, −9, or −12°C. To achieve these temperatures, the chamber temperature was lowered at a rate of 1°C/h. After the freezing period, the samples were thawed by gradually raising the temperature to 5°C at a rate of 1°C/h.
Seedlings frozen at −4°C were thawed according to the above procedure and used to determine the main marker of oxidative stress, the content of the lipid peroxidation (LPO) product malondialdehyde (MDA). Hardened seedlings that were not frozen served as a control for this analysis. As our preliminary experiments showed, exposing seedlings to −4°C increased MDA content (indicating oxidative stress), yet all the seedlings survived. Thus, MDA content was determined in living tissues to avoid errors in analysis associated with necrosis that occurs during freezing at lower temperatures.
Seedlings that were frozen at −9°C and −12°C were used to assess survival. After thawing, the seedlings were transferred to a chamber with a temperature of 24°C and an illumination of 150 W/m2, where they were grown for two days. Their survival was then assessed. Seedlings with green leaves that retained their ability to grow were classified as alive.
2.2 Assessment of Malondialdehyde Levels in Seedling Shoots
The amount of LPO products reacting with 2-thiobarbituric acid (mainly MDA) was analyzed by homogenizing the shoots in a reaction medium containing 0.25% 2-thiobarbituric acid in 10% trichloroacetic acid [46]. The resulting homogenate was placed in foil-covered tubes and boiled for 30 min in a water bath. After cooling, the samples were centrifuged at 10,000× g for 15 min using an MPW 350R centrifuge (Med-Instruments). The absorbance of the supernatant was measured at 532 nm, which is the maximum light absorption wavelength of MDA. Additionally, the absorbance was measured at 600 nm to correct for non-specific light absorption. MDA levels were assessed by comparing results from frozen seedlings to those of the corresponding controls.
2.3 Electrophoretic Separation and Estimation of Dehydrin Content
To extract the proteins, the collected shoot samples were homogenized in liquid nitrogen with 0.1 M Tris-HCl buffer (pH 8.8) and protease inhibitors (Complete EDTA-free protease inhibitor cocktail tablets, Roche, Basel, Switzerland). The weight of the plant material in samples differed by no more than 5–10%. In all cases, the ratio of plant material to buffer volume was strictly maintained at 1:5. The fraction enriched in dehydrins, which are boiling-resistant proteins, was isolated after the extract was centrifugated (20 min at 14,000× g at 4°C). The samples were boiled in a water bath for 15 min, followed by repeated centrifugation. The proteins in the supernatant were precipitated using ice-cold acetone supplemented with 1% (v/v) mercaptoethanol. The resulting pellets were then dried and dissolved in Laemmli buffer. The proteins were separated by SDS-PAGE on 12.5% resolving gels. A portion of the samples equivalent to 5 μg of fresh shoot weight was added to each well. The gels were run at 15 mA for the stacking gel and 25 mA for the resolving gel, according to the standard procedure [50]. Precision Plus Protein™ All Blue Standards (Bio-Rad) were used for molecular weight estimation.
The results of the electrophoretic separation were transferred to a 0.45-mm nitrocellulose membrane (Pharmacia Biotech) at 300 mA for 60 min. The membrane was blocked with 4.5% nonfat dry milk in Tris-buffered saline (TBS) and then incubated with a primary anti-dehydrin antibody (ADI-PLA-100-F Dehydrin (plant) polyclonal antibody, Enzo Life Sciences, Farmingdale, NY, USA). The antibody was diluted in 1.5% nonfat dry milk, 0.05% Tween-20, and TBS to a final dilution of 1:1000. To visualize the results, an immunoblot kit with goat anti-rabbit IgG alkaline phosphatase (Bio-Rad) and BCIP/NBT staining (Bio-Rad) were used. The membranes were scanned using a GS-800 densitometer (Bio-Rad), and the images underwent analysis with Quantity One v. 4.6.2 (Bio-Rad). The quantity of dehydrin protein was expressed in relative units, with the total dehydrin content in the mixed sample (internal standard) serving as the base unit.
To confirm the specificity of the antibody’s binding to the dehydrins, the gels of the control variant (without hardening) and the experimental variant (cold hardening) were stained with Bio-Safe Coomassie G-250 Stain (Bio-Rad). The absence of differences in the staining of the gels containing the control and experimental variants’ proteins was considered evidence of the antibody’s specific action.
2.4 Replication of Experiments and Mathematical Processing of Results
All experiments were independently repeated three times, with each treatment repeated three times. Survival was assessed using 70 seedlings per treatment for each biological repetition. For biochemical analyses, 12–15 seedlings were sampled from each repetition of each treatment.
The significance of the differences among treatments of the studied parameters was determined using a one-factor analysis of variance (ANOVA), followed by a Tukey’s multiple comparisons test. The mean values from three biological replicates, along with their standard errors, are displayed in the figures. Different letters indicate significant differences (p ≤ 0.05). Pearson’s correlation coefficients and their significance were calculated in Excel.
3.1 Survival of Seedlings after Freezing
As our previous experiments have shown, unhardened etiolated wheat seedlings were almost completely killed when frozen at temperatures below −6°C, regardless of the cultivar’s frost tolerance [46]. Therefore, to evaluate frost resistance levels, we used seedlings that had been hardened for six days at 3°C.
After freezing at −9°C, the survival rate of the high-FT cultivars (Antonivka, Doskonala, and Nordika) ranged from 49% to 78%. In contrast, among the low-FT cultivars (Tonnage, Tobak, and Altigo), only 16–23% of the seedlings remained viable (Fig. 1). The results were even more contrasting after freezing the seedlings at −12°C. In this case, the survival rate of the high-FT cultivars was 31–46%, whereas the survival rate of the low-FT cultivars did not exceed 8% (Fig. 1).
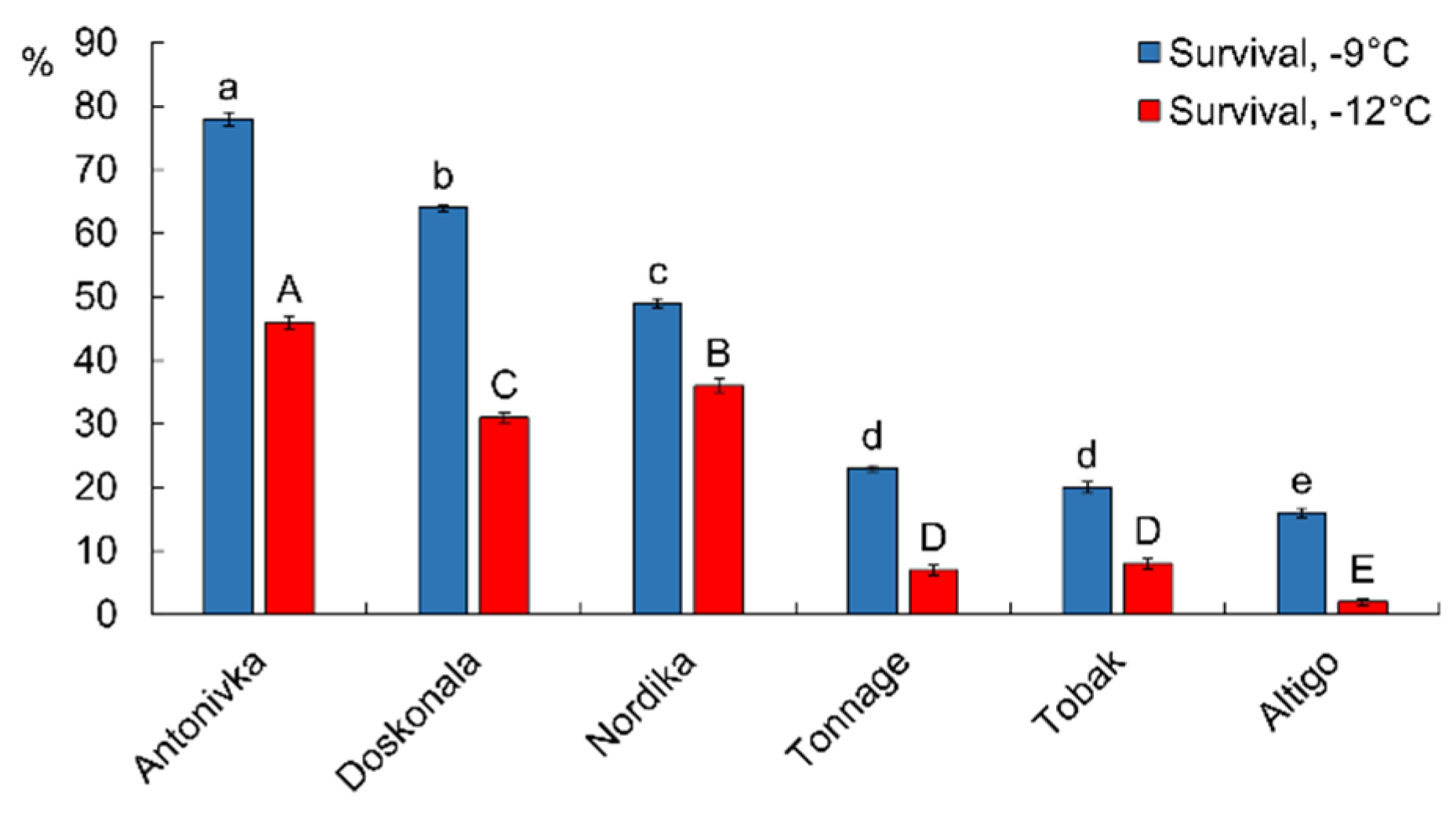
Figure 1: Survival of wheat seedlings (%) after 5 h of freezing at −9°C and −12°C. Different letters denote values with significant differences at p ≤ 0.05.
3.2 Content of the LPO Product MDA in Wheat Seedling Shoots
After freezing at −4°C, all seedlings remained alive. The content of the oxidative stress marker MDA remained virtually unchanged in the high-FT cultivars Antonivka and Nordika, while it increased slightly in the Doskonala cultivar (Fig. 2). In contrast, the MDA content in the low-FT cultivars Tonnage, Tobak, and Altigo increased by 29%, 41%, and 48%, respectively, compared to the hardened but not frozen samples.
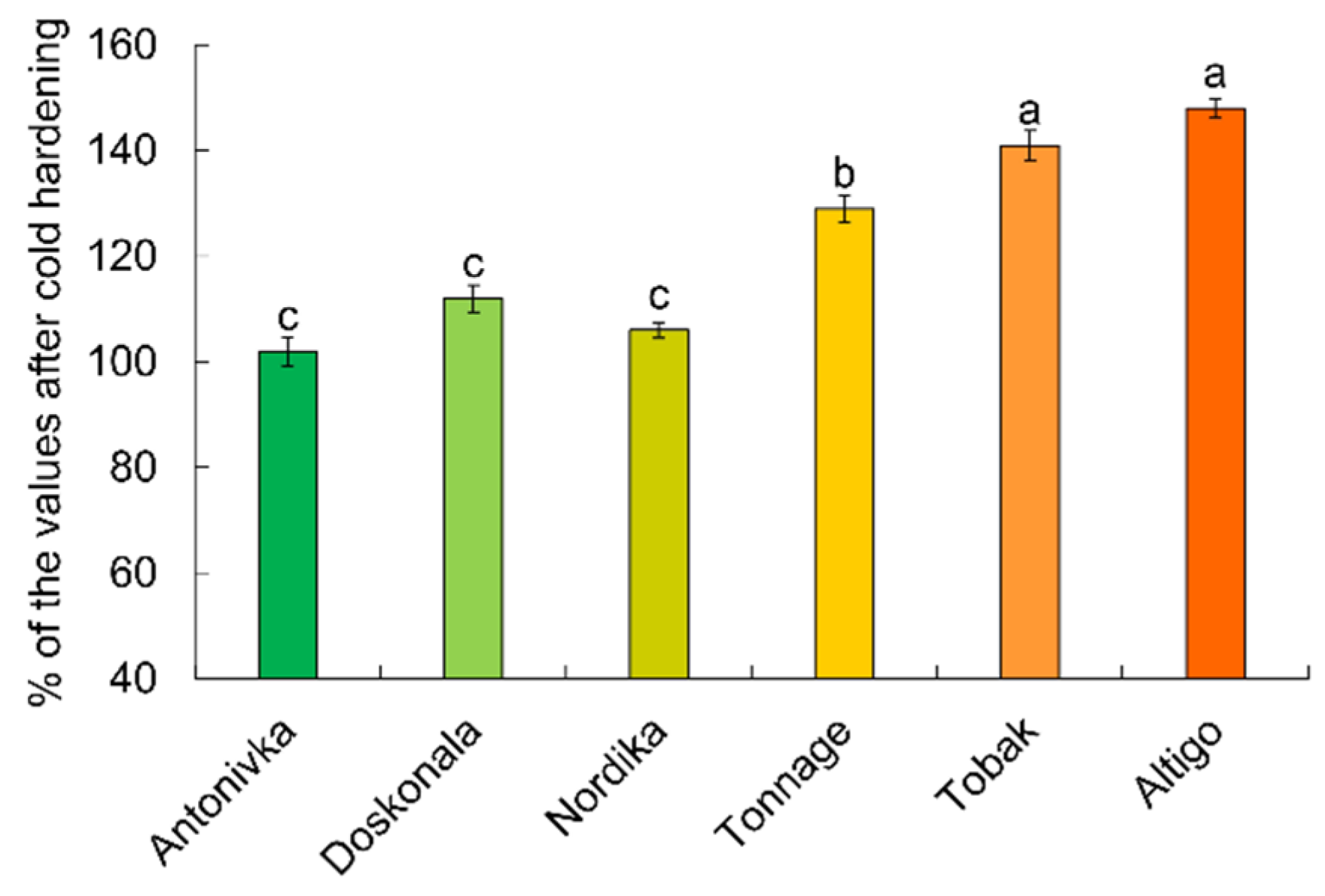
Figure 2: MDA content in wheat seedling shoots after 5 h of freezing at −4°C (% relative to values after cold hardening). Different letters denote values with significant differences at p ≤ 0.05.
3.3 Dehydrin Content in Wheat Seedling Shoots
Dehydrins were barely detected in the shoots of unhardened seedlings. Only proteins with molecular weights of approximately 49 kDa and 46 kDa showed faint bands in all cultivars, not allowing for a quantitative assessment (Fig. 3A). Meanwhile, electrophoregrams of proteins extracted from hardened seedlings revealed at least nine to eleven dehydrin bands with molecular weights ranging from 14 kDa to 68 kDa in all cultivars (Fig. 3B). The protein with a molecular weight of 49.6 kDa accumulated in the largest quantities in all cultivars. Cold hardening also resulted in significant accumulation of a protein with a molecular weight of approximately 68 kDa in all cultivars studied (Fig. 3B).
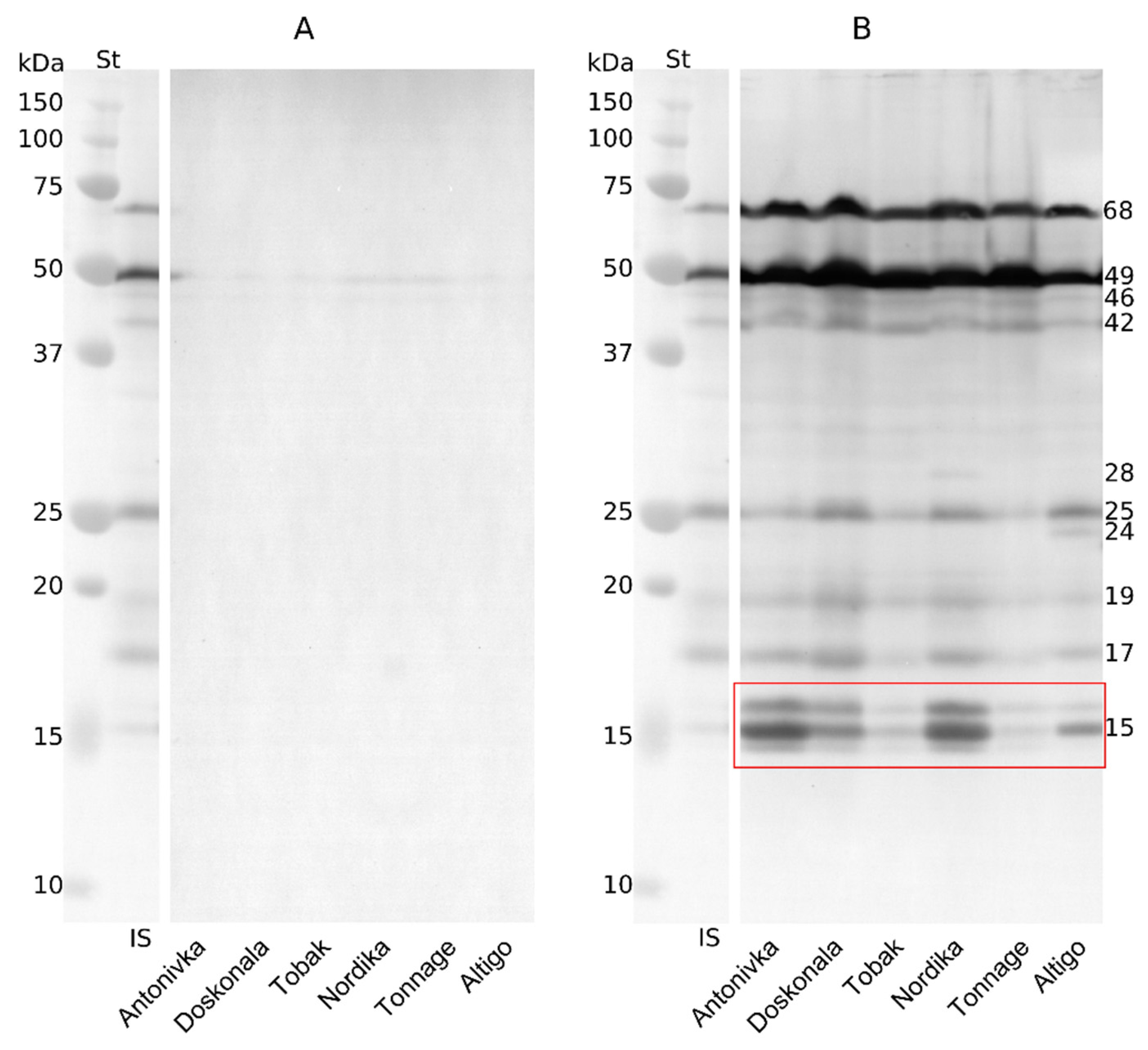
Figure 3: Electrophoretic spectrum of dehydrins in shoots of etiolated wheat seedlings. (A) control (3-day-old seedlings); (B) hardening (3-day-old seedlings hardened at 3°C for 6 days). St—Precision Plus Protein™ All Blue Standards (Bio-Rad); IS—inner standard (mixed). The red frame outlines the dehydrins with a molecular weight of approximately 14–16 kDa.
Although low-molecular-weight dehydrins were present in smaller quantities, the spectrum of these proteins was quite diverse (Fig. 3B and Fig. S1). The dehydrin with the lowest molecular weight (14.6 kDa) was only found in three high-FT cultivars: Antonivka, Doskonala, and Nordika (Fig. 3B and Fig. S1). Bands corresponding to dehydrins with molecular weights of 15.2 kDa and 15.8 kDa were observed in the shoots of hardened seedlings of all cultivars, but the quantities were significantly higher in the high-FT cultivars Antonivka, Nordika, and, to a lesser extent, Doskonala. Conversely, the amount of these proteins was minimal in the low-FT cultivars Tonnage and Tobak. The band with a molecular weight of about 15.2 kDa was more noticeable in the Altigo cultivar, but the overall content of this group of proteins was low (Fig. 3B and Fig. S1).
The 17.5-kDa protein was minor but present in all cultivars, with slightly higher levels in the high-FT cultivars Doskonala, Nordika, and Antonivka (Fig. 3B and Fig. S1). Dehydrin with a molecular weight of 19.5 kDa was also present in small amounts in all cultivars. A 24-kDa protein was found only in the Altigo cultivar and was absent in all other cultivars studied. Conversely, a protein with a molecular weight of 25.5 kDa was found in all cultivars, albeit in varying amounts. The highest amounts were found in the Altigo, Doskonala, and Nordika cultivars. In other words, no connection was found between the accumulation of this protein and the cultivars’ frost resistance (Fig. 3B and Fig. S1).
The total amount of all detected dehydrins varied greatly among different cultivars. In high-FT cultivars, the amount was approximately 2.2–2.6 relative units, whereas in low-FT cultivars, the amount was 1.6–1.9 relative units (Fig. 4). The difference in the content of the lowest molecular weight group of dehydrins, with molecular weights ranging from 14 kDa to 16 kDa, was even more noticeable. In three resistant cultivars, the average amount of dehydrins was approximately four times higher than in non-resistant ones (Fig. 4).
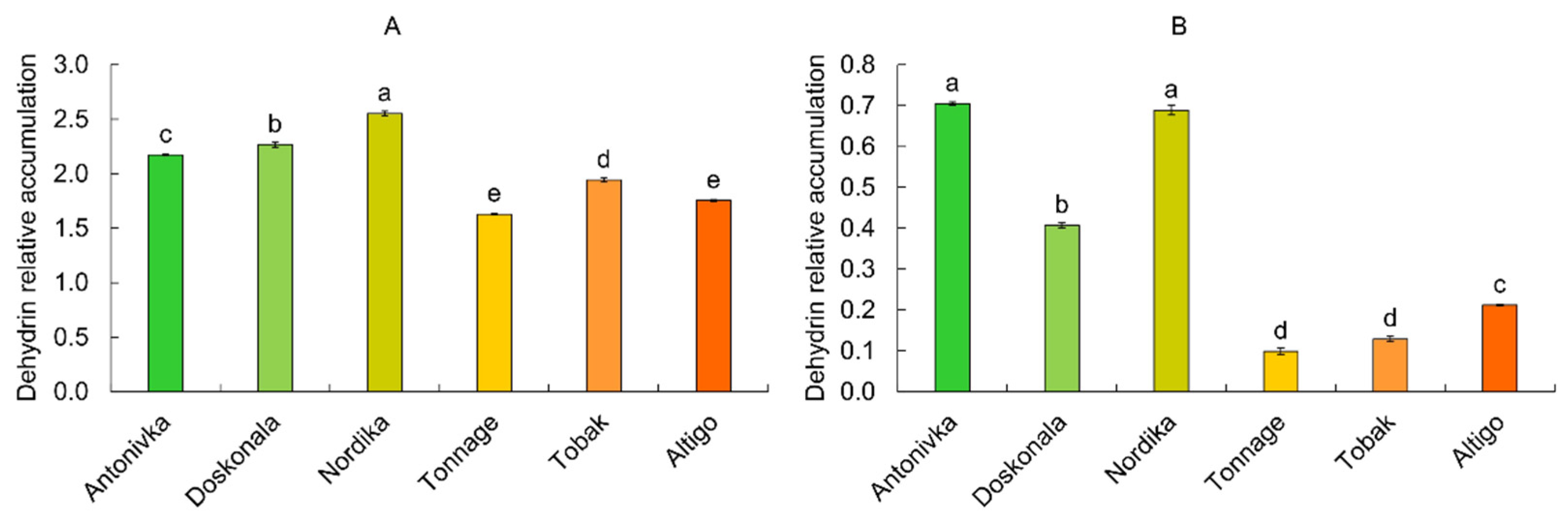
Figure 4: Total content of dehydrins (A) and total content of low-molecular-weight (14–16 kDa) dehydrins (B) in shoots of etiolated wheat seedlings after cold hardening (6 days at 3°C). Different letters denote values with significant differences at p ≤ 0.05.
In a parallel experiment, gels were stained with Coomassie G-250 after electrophoretic separation of boiling-resistant proteins to test the specificity of the antibody interaction with dehydrins. Only subtle changes in the protein spectrum isolated from nonhardened and hardened seedlings were observed (Fig. S2). These changes may be related to general transformations in the composition of different protein groups, including boiling-resistant proteins, in response to cold exposure [21]. However, such changes were generally much weaker than those observed in the immunoblot with specific detection of dehydrins. Furthermore, these changes did not coincide with changes in the content of proteins identified with the ADI-PLA-100-F antibody (see Fig. 3 and Fig. S2). This indicates the specificity with which ADI-PLA-100-F antibodies bind to dehydrins.
3.4 Correlations between the Frost Tolerance of Cultivars, Their Oxidative Stress Levels, and Dehydrin Content
The survival rates of seedlings after freezing at two different temperatures were closely correlated (r = 0.960 at p ≤ 0.01; 95%CI [0.671; 0.996]), indicating an absence of methodological artifacts in the frost resistance assessment (Fig. 5). Seedling survival after freezing was inversely correlated with MDA content (r = −0.918 at p ≤ 0.01; 95%CI [–0.991; −0.419] for −9°C and r = −0.960 at p ≤ 0.01; 95%CI [–0.996; −0.673] for −12°C).
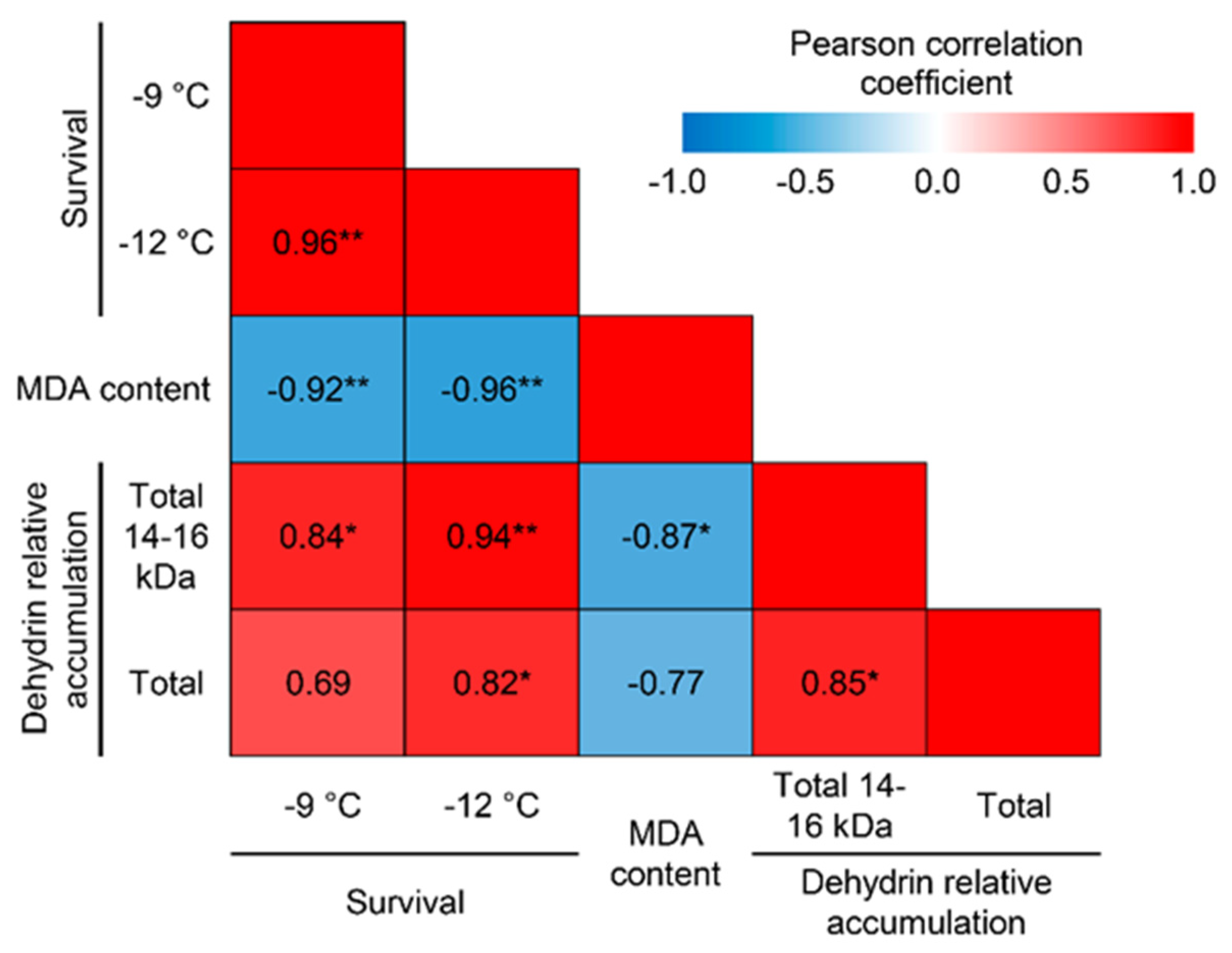
Figure 5: Correlations between wheat seedling survival, MDA content in seedlings after freezing, and the amount of total and low-molecular-weight (14–16 kDa) dehydrins. *Significant at p ≤ 0.05, **Significant at p ≤ 0.01.
Total dehydrin content positively correlated with seedling survival (r = 0.692; 95%CI [–0.273; 0.963] at −9°C and r = 0.817, p ≤ 0.05; 95%CI [0.016; 0.979] at −12°C). The content of the lowest molecular weight dehydrins (with a molecular weight range of 14–16 kDa) was even more closely correlated with seedling survival after exposure to −9°C (r = 0.841 at p ≤ 0.05; 95%CI [0.092; 0.982]) and −12°C (r = 0.940 at p ≤ 0.01; 95%CI [0.542; 0.994]) (Fig. 5 and Fig. S1). Conversely, no significant correlation with seedling survival was found for other groups of dehydrins (Fig. S3). Overall, there is a high level of correlation between dehydrin content with molecular weights ranging from 15 kDa to 20 kDa.
The total content of dehydrins in the shoots of seedlings was inversely correlated with the level of MDA observed after freezing; however, the correlation was not significant at p ≤ 0.05 (r = −0.771; 95%CI [–0.973; 0.110]). Meanwhile, the total content of low-molecular-weight dehydrins with molecular weights ranging from 14 kDa to 16 kDa was closely and inversely correlated with MDA content (r = −0.872 at p ≤ 0.05; 95%CI [–0.986; −0.206;]) (Fig. 5). No significant correlations with MDA content were found for other dehydrins (Fig. S3).
Our study revealed the accumulation of a diverse set of dehydrins with various molecular weights in the etiolated shoots of wheat seedlings from different ecological and geographical groups in response to hardening temperatures (Fig. 3). Notably, dehydrins were hardly detected in the shoots of non-hardened seedlings. This fact alone may indicate their special role in developing cold resistance in etiolated seedlings. There are only a few studies in the literature that have investigated the ability of etiolated cereal seedlings to accumulate dehydrins. Borovskii et al. [48] demonstrate the accumulation of dehydrins with molecular weights of 41, 50, and 66 kDa in etiolated seedlings of highly frost-resistant cultivars developed in Russia’s Siberian region, which were induced by cold hardening.
However, in our experiments, there was no correlation between the amount of dehydrins with molecular weights of 50, 66, and 42 kDa and seedling survival after freezing (Fig. S3). Conversely, a significant correlation was found between total dehydrin content and seedling survival at −12°C (Fig. 5).
Additionally, our study is the first to demonstrate the cold-induced accumulation of low-molecular-weight dehydrins with molecular weights of 14–16 kDa in etiolated wheat seedlings (Fig. 3B and Fig. S1). The total amount of these dehydrins was closely and positively correlated with the survival of seedlings after freezing (Fig. 5). The high correlation between the content of dehydrins with molecular weights of 14–16 kDa and frost tolerance of cultivars likely determined the high level of correlation between total dehydrin content and the survival of cultivars with different frost tolerance after exposure to −12°C. However, our study was conducted on a relatively small set of samples (six wheat cultivars), so the correlations found between the content of low-molecular-weight dehydrins and the frost tolerance of seedlings should be interpreted with caution. Nevertheless, the study revealed significant differences in the levels of dehydrins with molecular weights of 15.2 kDa and 15.8 kDa between high-FT and low-FT cultivars (Fig. S1). Furthermore, an absence of dehydrins with a molecular weight of 14.6 kDa was observed in low-FT cultivars. These facts clearly indicate a link between the accumulation of low-molecular-weight dehydrins and the frost tolerance of wheat cultivars. Further studies examining the relationship between the expression of genes of different groups of dehydrins in etiolated seedlings and their frost resistance are needed for more definitive conclusions.
It should be noted that the accumulation of low-molecular-weight dehydrins associated with cold adaptation has been found in various species of herbaceous and woody plants. Under natural conditions, for example, dehydrin accumulation with molecular weights of 12, 15, 17, 22, and 24 kDa was observed in winter wheat crowns in midwinter, whereas these dehydrins were completely absent in the first half of autumn [49]. In the buds of silver birch (Betula pendula Roth), it has been shown that dehydrins with molecular weights of 14–21 kDa are the most sensitive to seasonal changes. Their content increases significantly during the autumn-winter period [51]. A clear accumulation of dehydrin with a molecular weight of 15 kDa was found in the needles of Scots pine (Pinus sylvestris L.) during the cold season, with a maximum from November to April, and this dehydrin was completely absent during the summer months. Meanwhile, dehydrins with a molecular weight of 66 kDa were detected throughout the year, decreasing slightly during the summer [52]. In blueberry plants (Vaccinium corymbosum L.), a decrease in the level of 14-kDa dehydrin correlated with an increase in air temperature and a loss of frost resistance [53].
Thus, it can be assumed that low-molecular-weight dehydrins, particularly those with a molecular weight of approximately 15 kDa, play a crucial role in maintaining the frost tolerance of etiolated plants. It is noteworthy that dehydrins are found in a wide variety of organisms, including angiosperms, gymnosperms, mosses, fungi, algae, and cyanobacteria [54]. Their protective role likely manifests in different taxonomic groups and organs, including chlorophyll-lacking tissues such as etiolated seedlings and cereal crowns. At the same time, it cannot be ruled out that the accumulation of a specific group of dehydrins is important for certain plant organs. It can also be assumed that low-molecular-weight dehydrin accumulation is specifically regulated in response to cold hardening of etiolated wheat seedlings. In our previous study, which examined etiolated wheat seedlings in response to osmotic stress caused by PEG 8000, we observed the accumulation of dehydrins with molecular weights ranging from 17 kDa to 67 kDa. However, none of the cultivars exhibited the accumulation of lower-molecular-weight dehydrins with molecular weights between 14 kDa and 16 kDa [47].
In our study, for the first time in etiolated seedlings, a close inverse correlation was found between the accumulation of low-molecular-weight dehydrins and the manifestation of cold-induced oxidative stress, which was recorded by the content of the LPO product MDA (Fig. 5 and Fig. S2). Simultaneously, the MDA content was in a clear inverse correlation with the frost tolerance of the seedlings (Fig. 5). These results indicate the antioxidant role of dehydrin. There is literature indicating the direct antioxidant effect of dehydrins. In particular, K-segments in dehydrin structure may cause the “shielding effect” associated with histidine residues that chelate transition metal ions and prevent their participation in Fenton reactions [54]. The possibility of dehydrin binding to ROS and the participation of these proteins in the regulation of antioxidant enzyme gene expression has also been considered [54]. In transgenic tobacco plants overexpressing dehydrin genes under oxidative stress, lower levels of hydroxyl and superoxide radicals were observed, as well as higher expression of antioxidant enzyme genes compared to wild-type plants [55].
Another important property of dehydrins that contributes to cold tolerance development is their ability to interact with membrane lipids [34,56]. For instance, the dehydrin Lti30, which was isolated from Arabidopsis thaliana, has been demonstrated to electrostatically interact with vesicles containing both zwitterionic phospholipids (e.g., phosphatidylcholine) and negatively charged phospholipids (e.g., phosphatidylglycerol, phosphatidylserine, and phosphatidic acid) [57]. This interaction lowers the temperature of the main phase transition of lipids, which is important for cold tolerance. Additionally, dehydrins’ role in cold adaptation can be explained by their cryoprotective effect on enzymes and other proteins. These properties are due to the presence of a hydrophobic K-segment and, according to the latest data, an F-segment in dehydrins [58]. These segments can insert themselves between denaturing enzyme molecules, interact temporarily with hydrophobic regions, and prevent macromolecule aggregation. These effects have mainly been studied on a model enzyme (lactate dehydrogenase), but active research is currently underway on the molecular mechanisms of the cryoprotective action of dehydrins on proteins [34].
In practical terms, the presence of different types of dehydrins is considered an indicator of plant genotypes’ resistance to certain abiotic stresses, including cold stress. For example, when five wheat cultivars were exposed to temperatures of 4°C or 9°C, it was shown that a greater amount of dehydrin WCS120 (with a molecular weight of about 50 kDa) accumulated in the leaves of frost-resistant genotypes [44]. The results of the present study indicate the important role of low-molecular-weight dehydrins, with a molecular weight of approximately 15 kDa, in the frost resistance of etiolated wheat seedlings. A close direct correlation exists between the amount of dehydrin with a molecular weight of 14–16 kDa and seedling survival after freezing. There is also an inverse correlation between the content of these dehydrins and the manifestation of oxidative stress. This indicates the possibility of using their content in screening the frost tolerance of cultivars and lines. The presence of these correlations in etiolated seedlings makes them a convenient and cost-effective model for the rapid primary assessment of frost tolerance. Certainly, the data obtained in this study on the relationship between the accumulation of low-molecular-weight dehydrins and the frost tolerance of etiolated seedlings of different cultivars can only be extrapolated with great caution to the frost tolerance of adult plants. However, the long-term data available on the frost tolerance of the cultivars used in our study during the tillering phase (Table 1) are quite consistent with our data on the survival of etiolated seedlings of the corresponding cultivars after freezing (Fig. 1).
Undoubtedly, to gain a deeper understanding of the contribution of dehydrins to the frost tolerance of seedlings, it is necessary to study the links between the content of dehydrins and the functioning of other important components of stress protection systems, particularly by studying the same genotypes for the characteristics of osmolite accumulation and the functioning of the enzymatic antioxidant system. Which properties of the lowest molecular weight dehydrins (14–16 kDa) are associated with their special role in the frost tolerance of etiolated wheat seedlings remains to be resolved.
This study is the first to demonstrate a close positive correlation between the accumulation of low-molecular-weight dehydrin proteins with a molecular weight of 14–16 kDa in wheat seedlings of different ecological and geographical groups during cold hardening and their tolerance to frost. A significant inverse correlation was also established between the accumulation of this group of dehydrins and the intensity of cold-induced oxidative stress, indicating their antioxidant effects. Thus, the ability of etiolated wheat seedlings to accumulate dehydrins (primarily low-molecular-weight dehydrins) in response to cold exposure may be a marker of frost tolerance for its rapid assessment. However, to clarify the specific cryoprotective roles of low-molecular-weight dehydrins, further studies are needed.
Acknowledgement:
Funding Statement: This work is part of the project “Comprehensive scientific study of mechanisms of resistance of crop plants to biotic, abiotic, and anthropogenic stress, use of genetic diversity, and creation of stress-resistant cultivars and hybrids”, funded by the Ministry of Education and Science of Ukraine (registration number 0125U003530). The work was also supported by the Ministry of Agriculture of the Czech Republic, Grant number QL26010208.
Author Contributions: Conceptualization, Pavel Vítámvás, Ilja T. Prášil and Yuriy E. Kolupaev; methodology, Tetiana O. Yastreb, Ilja T. Prášil and Zdeněk Cit; investigation, Tetiana O. Yastreb, Ivan V. Shakhov and Yuriy E. Kolupaev; data curation, Tetiana O. Yastreb and Ivan V. Shakhov; writing—original draft preparation, Yuriy E. Kolupaev and Tetiana O. Yastreb; writing—review and editing Tetiana O. Yastreb; supervision, Yuriy E. Kolupaev and Pavel Vítámvás. All authors reviewed and approved the final version of the manuscript.
Availability of Data and Materials: The authors confirm that the data supporting the findings of this study are available within the article.
Ethics Approval: Not applicable.
Conflicts of Interest: The authors declare no conflicts of interest.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/phyton.2026.079882/s1. Supplementary Figure S1: Content of different forms of dehydrin in shoots of etiolated wheat seedlings after cold hardening (6 days at 3°C). Different letters denote values with significant differences at p ≤ 0.05. Supplementary Figure S2: Electrophoretic spectrum of boiling-resistant proteins after their staining with Coomassie G-250. Supplementary Figure S3: Correlations between the content of individual dehydrins, wheat seedling survival after freezing, and MDA content. *Significant at p ≤ 0.05, **Significant at p ≤ 0.01.
References
1. Hassan MA , Xiang C , Farooq M , Muhammad N , Yan Z , Hui X , et al. Cold stress in wheat: Plant acclimation responses and management strategies. Front Plant Sci. 2021; 12: 676884. doi:10.3389/fpls.2021.676884. [Google Scholar] [CrossRef]
2. Wang Y , Bo C , Wang X , Yang X , Wang H . Analysis of the physiological parameters of cold resistance in core winter and spring wheat cultivars. Agronomy. 2024; 14( 10): 2438. doi:10.3390/agronomy14102438. [Google Scholar] [CrossRef]
3. Ahmed U , Arif M , Abbas W , Saeed MF , Radicetti E , Jamal A , et al. Optimizing wheat yield and quality through the selection of climate-resilient varieties and appropriate planting dates. Global NEST J. 2025; 27( 8): 1– 14. doi:10.30955/gnj.07453. [Google Scholar] [CrossRef]
4. Gu L , Hanson PJ , Post W , Mac Kaiser DP , Yang B , Nemani R , et al. The 2007 eastern US spring freeze: Increased cold damage in a warming world? Bioscience. 2008; 58: 253– 62. doi:10.1641/B580311. [Google Scholar] [CrossRef]
5. Vaitkevičiūtė G , Aleliūnas A , Gibon Y , Armonienė R . The effect of cold acclimation, deacclimation and reacclimation on metabolite profiles and freezing tolerance in winter wheat. Front Plant Sci. 2022; 13: 959118. doi:10.3389/fpls.2022.959118. [Google Scholar] [CrossRef]
6. Vico G , Hurry V , Weih M . Snowed in for survival: Quantifying the risk of winter damage to overwintering field crops in northern temperate latitudes. Agric For Meteorol. 2014; 197: 65– 75. doi:10.1016/j.agrformet.2014.06.003. [Google Scholar] [CrossRef]
7. Prášil IT , Musilová J , Prášilová P , Janáček J , Coufová M , Kosová K , et al. Effect of geographical origin, regional adaptation, genotype, and release year on winter hardiness of wheat and triticale accessions evaluated for six decades in trials. Sci Rep. 2025; 15( 1): 5961. doi:10.1038/s41598-025-89291-8. [Google Scholar] [CrossRef]
8. Li L , Han C , Yang J , Tian Z , Jiang R , Yang F , et al. Comprehensive transcriptome analysis of responses during cold stress in wheat (Triticum aestivum L.). Genes. 2023; 14: 844. doi:10.3390/genes14040844. [Google Scholar] [CrossRef]
9. Ahad A , Gul A , Batool TS , Huda NU , Naseeer F , Abdul Salam U , et al. Molecular and genetic perspectives of cold tolerance in wheat. Mol Biol Rep. 2023; 50( 8): 6997– 7015. doi:10.1007/s11033-023-08584-1. [Google Scholar] [CrossRef]
10. Cao Y , Hwarari D , Radani Y , Guan Y , Yang L . Molecular mechanism underlying plant response to cold stress. Phyton-Int J Exp Bot. 2023; 92( 9): 2665– 83. doi:10.32604/phyton.2023.024929. [Google Scholar] [CrossRef]
11. Kolupaev YE , Yastreb TO , Ryabchun NI , Kuzmyshyna NV , Marenych MM , Ryabchun VK . Signaling and protection systems in the adaptation of plants to cold. J Central Eur Agricult. 2023; 24( 1): 202– 15. doi:10.5513/JCEA01/24.1.3776. [Google Scholar] [CrossRef]
12. Zhang Y , Xu J , Li R , Ge Y , Li Y , Li R . Plants’ response to abiotic stress: Mechanisms and strategies. Int J Mol Sci. 2023; 24: 10915. doi:10.3390/ijms241310915. [Google Scholar] [CrossRef]
13. Iqbal Z , Memon AG , Ahmad A , Iqbal MS . Calcium mediated cold acclimation in plants: Underlying signaling and molecular mechanisms. Front Plant Sci. 2022; 13: 855559. doi:10.3389/fpls.2022.855559. [Google Scholar] [CrossRef]
14. Qi W , Sun W , Ma L , Li X , Liu H , Yang C , et al. Pathways related to ROS production, clearance, and signal transduction during cold response in Brassica napus L. with strong cold resistance. Phyton-Int J Exp Bot. 2025; 94( 3): 739– 62. doi:10.32604/phyton.2025.060712. [Google Scholar] [CrossRef]
15. Nuotio S , Heino P , Palva ET . Signal transduction under low-temperature stress. In: Basra A , editor. Crop responses and adaptations to temperature stress. Boca Raton, FL, USA: CRC Press; 2023. p. 151– 75. doi:10.1201/9781003421221-5. [Google Scholar] [CrossRef]
16. Kolupaev YE , Ryabchun NI , Relina LI , Yastreb TO , Yegorova NY , Kolomatska VP . Exogenous nitric oxide and hydrogen sulfide as biotechnological tools for enhancing plant adaptation to cold. Not Sci Biol. 2024; 16( 3): 12064. doi:10.55779/nsb16312064. [Google Scholar] [CrossRef]
17. Kosová K , Vítámvás P , Planchon S , Renaut J , Vanková R , Prášil IT . Proteome analysis of cold response in spring and winter wheat (Triticum aestivum) crowns reveals similarities in stress adaptation and differences in regulatory processes between the growth habits. J Proteome Res. 2013; 12( 11): 4830– 45. doi:10.1021/pr400600g. [Google Scholar] [CrossRef]
18. Lv L , Dong C , Liu Y , Zhao A , Zhang Y , Li H , et al. Transcription-associated metabolomic profiling reveals the critical role of frost tolerance in wheat. BMC Plant Biology. 2022; 22( 1): 333. doi:10.1186/s12870-022-03718-2. [Google Scholar] [CrossRef]
19. Caccialupi G , Milc J , Caradonia F , Nasar MF , Francia E . The triticeae CBF gene cluster—To frost resistance and beyond. Cells. 2023; 12( 22): 2606. doi:10.3390/cells12222606. [Google Scholar] [CrossRef]
20. Jaškūnė K , Armonienė R , Liatukas Ž , Statkevičiūtė G , Cesevičienė J , Brazauskas G . Relationship between freezing tolerance and leaf growth during acclimation in winter wheat. Agronomy. 2022; 12( 4): 859. doi:10.3390/agronomy12040859. [Google Scholar] [CrossRef]
21. Stachurska J , Sadura-Berg I , Rys M . When warm breaks cold: Understanding deacclimations and reacclimations cycles as a key to winter crop resilience. Int J Mol Sci. 2025; 26( 22): 11080. doi:10.3390/ijms262211080. [Google Scholar] [CrossRef]
22. Kurepin LV , Dahal KP , Savitch LV , Singh J , Bode R , Ivanov AG , et al. Role of CBFs as integrators of chloroplast redox, phytochrome and plant hormone signaling during cold acclimation. Int J Mol Sci. 2013; 14( 6): 12729– 63. doi:10.3390/ijms140612729. [Google Scholar] [CrossRef]
23. Kumar N , Verma P , Singh SK , Bakshi M , Singh S , Mirza AA . Understanding plant resilience: Biochemical and physiological responses to low temperature stress in plants. CABI Rev. 2025; 20( 1): 0073. doi:10.1079/cabireviews.2025.0073. [Google Scholar] [CrossRef]
24. Sardoei AS , Fazeli-Nasab B . Physiological and molecular mechanisms of freezing in plants. Phyton-Int J Exp Bot. 2025; 94( 6): 1601– 30. doi:10.32604/phyton.2025.064729. [Google Scholar] [CrossRef]
25. García-Caparrós P , De Filippis L , Gul A , Hasanuzzaman M , Ozturk M , Altay V , et al. Oxidative stress and antioxidant metabolism under adverse environmental conditions: A review. Bot Rev. 2021; 87( 4): 421– 66. doi:10.1007/s12229-020-09231-1. [Google Scholar] [CrossRef]
26. Manasa LS , Panigrahy M , Panigrahi KCS , Rout GR . Overview of cold stress regulation in plants. Bot Rev. 2022; 88( 3): 359– 87. doi:10.1007/s12229-021-09267-x. [Google Scholar] [CrossRef]
27. Roychowdhury R , Das SP , Sarkar P , Khan Z , Kumar A , Sarker U , et al. Physiological, biochemical and molecular signaling basis of cold stress tolerance in plants. Front Plant Sci. 2025; 16: 1707204. doi:10.3389/fpls.2025.1707204. [Google Scholar] [CrossRef]
28. Theocharis A , Clement C , Barka EA . Physiological and molecular changes in plants grown at low temperatures. Planta. 2012; 235( 6): 1091– 105. doi:10.1007/s00425-012-1641-y. [Google Scholar] [CrossRef]
29. Bhattacharya A . Lipid metabolism in plants under low-temperature stress: A review. In: Physiological processes in plants under low temperature stress. Singapore: Springer; 2022. p. 409– 516. doi:10.1007/978-981-16-9037-2_5. [Google Scholar] [CrossRef]
30. Habibpourmehraban F . Plant proteome in response to abiotic stress. In: Hasanuzzaman M , Nahar K , editors. Plant stress physiology—Perspectives in agriculture. London, UK: IntechOpen; 2022. p. 1– 20. doi:10.5772/intechopen.102875. [Google Scholar] [CrossRef]
31. Zakariya M , Akbar F , Iqbal A , Ali SS , Ali Z , Suleman M , et al. Understanding abiotic stress tolerance in plants by proteomic approach. In: Prakash CS , Fiaz S , Fahad S , editors. Principles and practices of OMICS and genome editing for crop improvement. Berlin/Heidelberg, Germany: Springer; 2022. p. 257– 77. doi:10.1007/978-3-030-96925-7_11. [Google Scholar] [CrossRef]
32. Xu K , Zhao Y , Gu J , Zhou M , Gao L , Sun RX , et al. Proteomic analysis reveals the molecular mechanism underlying the cold acclimation and freezing tolerance of wheat (Triticum aestivum L.). Plant Sci. 2022; 318: 111242. doi:10.1016/j.plantsci.2022.111242. [Google Scholar] [CrossRef]
33. Szlachtowska Z , Rurek M . Plant dehydrins and dehydrin-like proteins: Characterization and participation in abiotic stress response. Front Plant Sci. 2023; 14: 1213188. doi:10.3389/fpls.2023.1213188. [Google Scholar] [CrossRef]
34. Smith MA , Graether SP . The disordered dehydrin and its role in plant protection: A Biochemical perspective. Biomolecules. 2022; 12: 294. doi:10.3390/biom12020294. [Google Scholar] [CrossRef]
35. Sun Z , Li S , Chen W , Zhang J , Zhang L , Sun W , et al. Plant dehydrins: Expression, regulatory networks, and protective roles in plants challenged by abiotic stress. Int J Mol Sci. 2021; 22: 12619. doi:10.3390/ijms222312619. [Google Scholar] [CrossRef]
36. Kosová K , Vítámvás P , Prášil IT . Wheat and barley dehydrins under cold, drought, and salinity—What can LEA-II proteins tell us about plant stress response? Front Plant Sci. 2014; 5: 343. doi:10.3389/fpls.2014.00343. [Google Scholar] [CrossRef]
37. Liu Y , Song Q , Li D , Yang X , Li D . Multifunctional Roles of plant dehydrins in response to environmental stresses. Front Plant Sci. 2017; 8: 1018. doi:10.3389/fpls.2017.01018. [Google Scholar] [CrossRef]
38. Drira M , Saibi W , Brini F , Gargouri A , Masmoudi K , Hanin M . The K-segments of the wheat dehydrin DHN-5 are essential for the protection of lactate dehydrogenase and β-glucosidase activities in vitro. Mol Biotechnol. 2013; 54( 2): 643– 50. doi:10.1007/s12033-012-9606-8. [Google Scholar] [CrossRef]
39. Shi J , Liu M , Chen Y , Wang J , Lu C . Heterologous expression of the dehydrin-like protein gene AmCIP from Ammopiptanthus mongolicus enhances viability of Escherichia coli and tobacco under cold stress. Plant Growth Regul. 2016; 79( 1): 71– 80. doi:10.1007/s10725-015-0112-4. [Google Scholar] [CrossRef]
40. Tiwari P , Chakrabarty D . Dehydrin in the past four decades: From chaperones to transcription co-regulators in regulating abiotic stress response. Curr Res Biotechnol. 2021; 3: 249– 59. doi:10.1016/j.crbiot.2021.07.005. [Google Scholar] [CrossRef]
41. Hao Y , Hao M , Cui Y , Kong L , Wang H . Genome-wide survey of the dehydrin genes in bread wheat (Triticum aestivum L.) and its relatives: Identification, evolution and expression profiling under various abiotic stresses. BMC Genom. 2022; 23( 1): 73. doi:10.1186/s12864-022-08317-x. [Google Scholar] [CrossRef]
42. Boddington KF , Graether SP . Binding of a Vitis riparia dehydrin to DNA. Plant Sci. 2019; 287: 110172. doi:10.1016/j.plantsci.2019.110172. [Google Scholar] [CrossRef]
43. Riyazuddin R , Nisha N , Singh K , Verma R , Gupta R . Involvement of dehydrin proteins in mitigating the negative effects of drought stress in plants. Plant Cell Rep. 2022; 41( 3): 519– 33. doi:10.1007/s00299-021-02720-6. [Google Scholar] [CrossRef]
44. Vítámvás P , Prášil IT , Vítámvás J , Klíma M . Relationship between WCS120 protein family accumulation and frost tolerance in wheat cultivars grown under different temperature treatments. Plants. 2021; 10: 1114. doi:10.3390/plants10061114. [Google Scholar] [CrossRef]
45. Kosová K , Vítámvás P , Prášilová P , Prášil IT . Accumulation of WCS120 and DHN5 proteins in differently frost-tolerant wheat and barley cultivars grown under a broad temperature scale. Biol Plant. 2013; 57: 105– 12. doi:10.1007/s10535-012-0237-5. [Google Scholar] [CrossRef]
46. Yastreb TO , Kolupaev YE , Kokorev AI , Маkaova BE , Ryabchun NI , Zmiievska OA , et al. Indices of antioxidant and osmoprotective systems in seedlings of winter wheat cultivars with different frost resistance. Ukr Biochem J. 2023; 95( 1): 73– 84. doi:10.15407/ubj95.01.073. [Google Scholar] [CrossRef]
47. Yastreb TO , Kosová K , Vítámvás P , Cit Z , Shakhov IV , Kolupaev YE . Dehydrin content and status of antioxidant and osmoprotective systems of etiolated Triticum aestivum seedlings of different genotypes under osmotic stress. J Crop Health. 2025; 77( 4): 115. doi:10.1007/s10343-025-01190-0. [Google Scholar] [CrossRef]
48. Borovskii GB , Stupnikova IV , Antipina AI , Voinikov VK . Accumulation of dehydrins and ABA-inducible proteins in wheat seedlings during low-temperature acclimation. Russ J Plant Physiol. 2002; 49( 2): 229– 34. doi:10.1023/A:1014857708301. [Google Scholar] [CrossRef]
49. Stupnikova IV , Borovskii GB , Voinikov VK . Seasonal changes in the composition and content of dehydrins in winter wheat plants. Russ J Plant Physiol. 2004: 51( 5): 636– 41. doi:10.1023/B:RUPP.0000040750.10102.72. [Google Scholar] [CrossRef]
50. Laemmli UK . Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970; 227( 5259): 680– 5. doi:10.1038/227680a0. [Google Scholar] [CrossRef]
51. Tatarinova TD , Bubyakina VV . Vetchinnikova LV, Perk AA, Ponomarev AG, Vasilieva IV. Dehydrin stress proteins in birch buds in regions with contrasting climate. Cell Tiss Biol. 2017; 11: 483– 8. doi:10.1134/S1990519X17060098. [Google Scholar] [CrossRef]
52. Tatarinova TD , Perk AA , Bubyakina VV , Vasilieva IV , Ponomarev AG , Maximov TC . Dehydrin stress proteins in Pinus sylvestris L. needles under conditions of extreme climate of Yakutia. Doklady Biochem Biophys. 2017; 473( 1): 98– 101. doi:10.1134/S160767291702003X. [Google Scholar] [CrossRef]
53. Dhanaraj AL , Slovin JP , Rowland LJ . Isolation of a cDNA clone and characterization of expression of the highly abundant, cold acclimation-associated 14 kDa dehydrin of blueberry. Plant Sci. 2005; 168( 4): 949– 57. doi:10.1016/j.plantsci.2004.11.007. [Google Scholar] [CrossRef]
54. Güler NS , Terzi R . Dehydrins: An overview of current approaches and advancement. Turk J Bot. 2020; 44( 5): 481– 92. doi:10.3906/bot-2005-78. [Google Scholar] [CrossRef]
55. Halder T , Upadhyaya G , Basak C , Das A , Chakraborty C , Ray S . Dehydrins impart protection against oxidative stress in transgenic tobacco plants. Front Plant Sci. 2018; 9: 136. doi:10.3389/fpls.2018.00136. [Google Scholar] [CrossRef]
56. Andersson JM , Pham QD , Mateos H , Eriksson S , Harryson P , Sparr E . The plant dehydrin Lti30 stabilizes lipid lamellar structures in varying hydration conditions. J Lipid Res. 2020; 61( 7): 1014– 24. doi:10.1194/jlr.RA120000624. [Google Scholar] [CrossRef]
57. Eriksson SK , Kutzer M , Procek J , Gröbner G , Harryson P . Tunable membrane binding of the intrinsically disordered dehydrin Lti30, a cold-induced plant stress protein. Plant Cell. 2011; 23( 6): 2391– 404. doi:10.1105/tpc.111.085183. [Google Scholar] [CrossRef]
58. Ohkubo T , Kameyama A , Kamiya K , Kondo M , Hara M . F-segments of Arabidopsis dehydrins show cryoprotective activities for lactate dehydrogenase depending on the hydrophobic residues. Phytochemistry. 2020; 173: 112300. doi:10.1016/j.phytochem.2020.112300. [Google Scholar] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools