 Open Access
Open Access
ARTICLE
Sodium Alginate Nanoemulsions for the Delivery of Eucalyptus staigeriana Essential Oil: Production, Characterization, and Ovicidal and Larvicidal Effects against Haemonchus contortus
1 Programa de Pós-graduação em Ciências Veterinárias, Faculdade de Veterinária, Campus do Itaperi, Universidade Estadual do Ceará, 60714-903, CE, Brasil
2 Centro Universitário, Faculdade Metropolitana, Campus Fortaleza, 60040-531, CE, Brasil
3 Pós-graduçâo em Ciências Naturais, Centro de Ciências e Tecnologia, Campus do Itaperi, Universidade Estadual do Ceará, Fortaleza, 60714-903, CE, Brasil
4 Departamento de Patologia e Medicina Legal, Faculdade de Medicina, Campus do Porangabuçu, Universidade Federal do Ceará, Fortaleza, 60430-160, CE, Brazil
5 Departamento de Fisiologia e Farmacologia, Faculdade de Medicina, Campus do Parangaboçu, Universidade Federal do Ceará, Fortaleza, 60430-275, CE, Brazil
* Corresponding Author: Claudia M. L. Bevilaqua. Email:
(This article belongs to the Special Issue: Biobased Nanoemulsions for a Sustainable Future)
Journal of Renewable Materials 2026, 14(2), 7 https://doi.org/10.32604/jrm.2025.02025-0138
Received 06 June 2025; Accepted 27 October 2025; Issue published 25 February 2026
Abstract
Eucalyptus staigeriana essential oil (EsEO) has well-known anthelmintic activity in small ruminants. However, its volatility limits its therapeutic action. The aim of this study was to develop a water-in-oil sodium alginate-based nanoemulsion with an effective in vitro effect on the eggs and larvae of Haemonchus contortus, a gastrointestinal parasite of sheep and goats. Four oil-in-water sodium alginate-based emulsions were prepared using a high-energy method with different proportions of Tween 80, EsEO, and sodium alginate (ALG) 4%. The physical-chemical characterization included stability, particle size, zeta potential and infrared spectra. The effects of the emulsions were evaluated against H. contortus via the egg hatching test (EHT) and larval development test (LDT). The results showed that the emulsions were stable over 7 days, nanometer scale particles (218.8 to 371.5 nm) predominating and with negative zeta potentials (−28.9 to −46.9 mV). All four emulsions were more effective than EsEO in the EHT, with 50% effective concentrations (EC50) of 0.088 to 0.15 mg/mL for the emulsions and 0.308 mg/mL for EsEO. However, in the LDT, the emulsions and essential oil had similar effects, with EC50 values of 3.91 to 4.60 mg/mL for the emulsions and 4.17 mg/mL for EsEO. Emulsion 2, with low Tween 80/EsEO and ALG/EsEO ratios, was considered better overall in terms of physical, chemical and anthelmintic assessment and is a promising candidate for further in vivo assays against adult H. contortus.Graphic Abstract
Keywords
Supplementary Material
Supplementary Material FileParasitic nematodes are important pathogens that cause diseases in humans and other animals. They are responsible for significant economic losses in all animal production systems. In small ruminant production, gastrointestinal nematodes (GINs), mainly those belonging to the order Strongylida and superfamily Trichostrongyloidea, cause subclinical and clinical infections, with effects ranging from a decrease in appetite to the death of susceptible hosts with heavier worm burdens [1–3].
Control of GIN parasitism has almost exclusively been accomplished with anthelmintic drugs. However, the widespread and frequent use of these molecules has led to the emergence and spread of multidrug-resistant nematode populations. In this context, anthelmintic resistance has motivated many studies on the identification of molecular mechanisms related to the development of resistance and its diagnosis and has encouraged the search for alternative methods to control helminthiasis to avoid economic loss [4–6].
Plants produce chemical substances with different biological properties for different purposes. The use of plant extracts and isolated compounds has been widely studied as an environmentally friendly method for controlling GIN. In vitro and in vivo assays have been performed, and many reports the potential of natural bioactive compounds against the eggs, larvae and adults of small ruminant parasitic nematodes [7–10].
The genus Eucalyptus belongs to the family Myrtaceae, which is native to Australia and cultivated worldwide. Many Eucalyptus species are essential oil producers. These essential oils are complex mixtures of volatile compounds, mainly terpenes and terpenoids, and are produced by secondary metabolism in plants. The essential oils of Eucalyptus spp., including Eucalyptus staigeriana essential oil (EsEO), have diverse therapeutic properties and often contain citral and limonene as major constituents [11,12]. EsEO has been shown to affect bacteria [13], fungi [12], insects [14,15], and nematodes [15] among other taxa.
Macedo et al. [15] demonstrated that EsEO has activity against H. contortus, inhibiting 99.27% of egg hatching at 1.35 mg/mL and 99.2% of larval development at 5.4 mg/mL. In goats, 500 mg/kg EsEO reduced the fecal egg count by 76.57% 15 days after treatment. Although these results were not as efficient as those of synthetic anthelmintic drugs, the anthelmintic potential of EsEO, especially in vivo, is noteworthy. Thus, strategies that enhance essential oil effects, such as encapsulation for drug delivery, are needed [16,17].
Essential oils are unstable and can easily deteriorate if exposed to light, temperature, humidity and oxygen. The encapsulation of essential oil in a polymeric matrix can reduce the risk of deterioration, improve the stability of the resulting emulsion, and increase its bioavailability [18,19].
Biopolymers are well-known wall materials used in the development of oil/water (O/W) nanoemulsions. Among them, sodium alginate, which is a natural anionic polysaccharide extracted from brown algae, has been widely evaluated as a wall matrix for the encapsulation of lipophilic compounds such as carvacrol [20], sunflower seed oil [21], peppermint oil [22] and Serenoa repens oil [23]. Ong et al. [24] demonstrated that sodium alginate-based O/W emulsions with nonionic surfactants (Tween 80 and Span 20) were stable and had high oil contents [25,26].
Polysaccharides such as sodium alginate, gum Arabic, Xanthan gum, and their derivatives have gained traction as emulsifying agents and encapsulating matrices in oil-in-water nanoemulsion formulations owing to their amphiphilic character, biocompatibility, and ability to form electrosteric interfacial films that stabilize oil droplets against coalescence and gravitational separation [27,28]. By adsorbing at the oil–water interface, these macromolecules sharply decrease interfacial tension and retard Ostwald ripening; in concert with increased continuous-phase viscosity, they maintain kinetically stable dispersions with mean droplet diameters below 200 nm and polydispersity indices under 0.3 for extended storage periods [29,30]. Encapsulation within hydrated polysaccharide networks shields volatile, oxidation-prone actives—such as essential oils—from thermal, photolytic, and oxidative degradation, thus preserving their chemical integrity and bioactivity during storage and processing [28]. Additionally, the dense polymeric shell imposes a tortuous diffusion path that enables controlled, sustained release of encapsulated agents, thereby enhancing antimicrobial or antioxidant efficacy at reduced dosages [27,30]. These integrated properties underscore the potential of polysaccharide-stabilized nanoemulsions for applications in veterinary parasitology and functional foods, where long-term stability, bioavailability, and precise release profiles are essential.
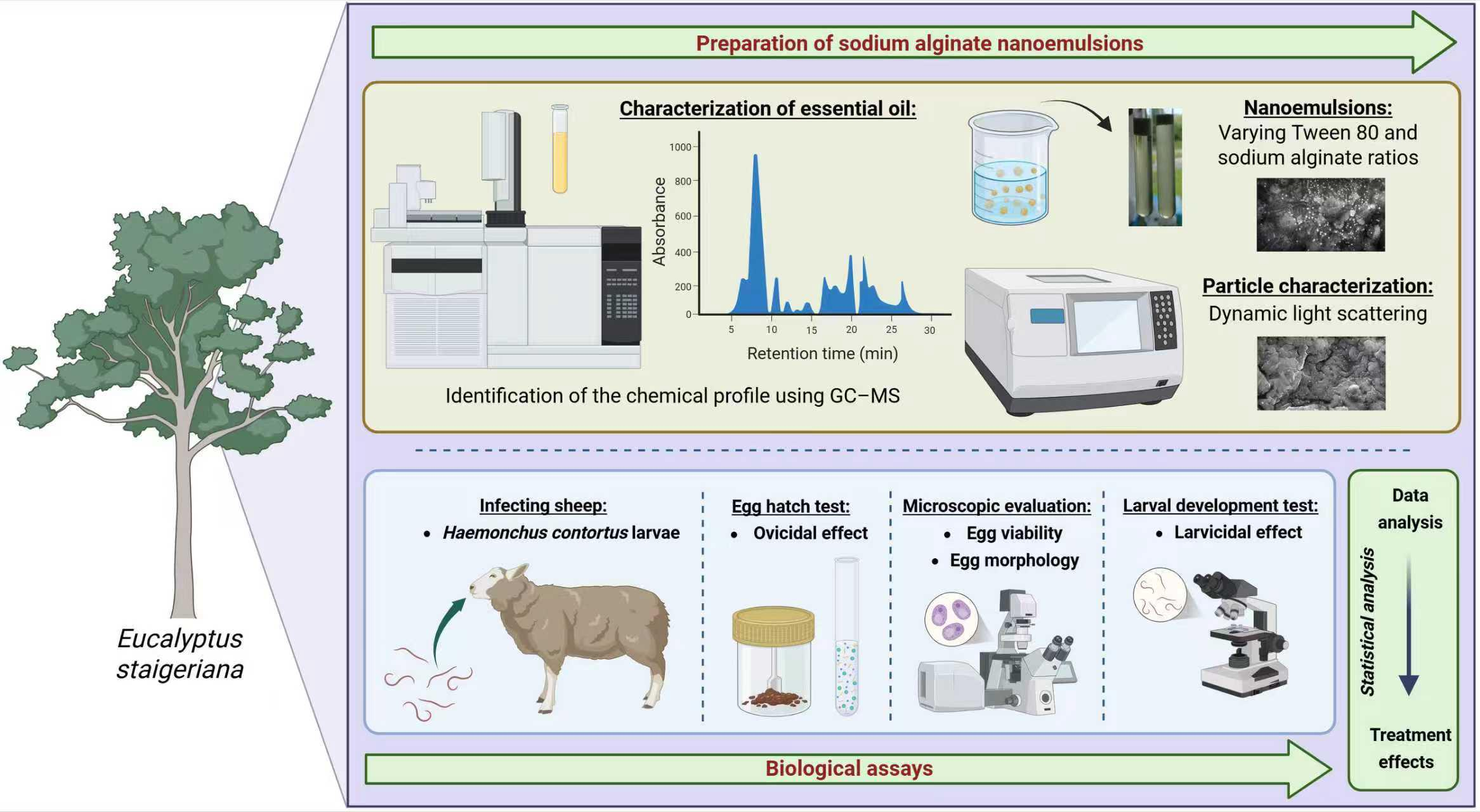
Thus, to optimize the performance of EsEO, the aims of this study were to develop sodium alginate-based EsEO emulsions, physicochemically characterize them, evaluate their effects on egg hatching and larval development in H. contortus and determine the emulsion with the best performance.
2.1 Essential Oil Characterization
The polysaccharide sodium alginate (DINÂMICA, São Paulo, Brazil), surfactant Tween 80® (VETEC, Rio de Janeiro, Brazil), were used as received. EsEO was purchased from Ferquima® (São Paulo, Brazil). The chemical composition of the oil was determined via gas chromatography-mass spectrometry (GC-MS) (GCMS-QP2010S, Shimadzu®, Kyoto, Japan). The following experimental conditions were used: an RTX-5 (30 m × 0.25 mm) capillary column, a helium carrier gas, an injector temperature of 250°C, a detector temperature of 260°C, and a column temperature of 50°C–150°C at 2.5°C/min followed by 150°C–250°C at 25°C/min. For mass spectrometry, the electron impact energy was 70 eV.
The components of EsEO were identified according to their GC retention time, as expressed by Kovats index, which was calculated using the van den Dool and Kratz equation [31]. Additionally, the mass spectra of the test compounds were compared with spectra from the National Institute for Standard Technology computer database and published spectra. The EsEO compounds were quantified using the relative percentage of peak areas from the chromatogram.
2.2 Preparation of Nanoemulsions
A two-level factorial experimental design was employed to evaluate the effects of two independent variables—Tween 80/EsEO ratio (Factor A) and sodium alginate/EsEO ratio (Factor B)—on the stability of nanoemulsions. The dependent variables (responses) were particle size and zeta potential (Table S1).
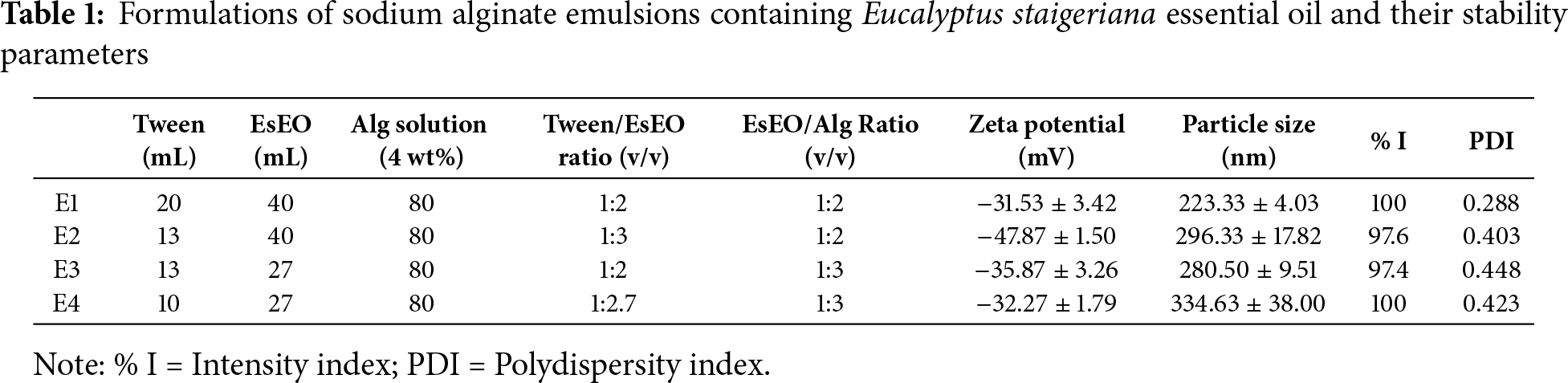
Four oil-in-water (O/W) nanoemulsions were prepared in duplicate using a high-speed homogenization method adapted from [32], by varying the proportions of Tween 80, EsEO, and sodium alginate solution (ALG, 4% w/v in distilled water). The formulations were prepared at the following volume ratios (v/v): 1:2:4 (E1), 1:3:6 (E2), 1:2:6 (E3), and 1:2.5:7.5 (E4). Table 1 presents the quantities used in each formulation, along with the relative volume ratios of Tween 80 to essential oil (Tween/EsEO) and essential oil to alginate solution (EsEO/ALG).
Initially, Tween 80 was added to the EsEO (oil phase), and sodium alginate (aqueous phase) was prepared separately. Both phases were subjected to ultrasonic treatment for 10 min. Subsequently, the oil and aqueous phases were combined using an ULTRA-380 mechanical homogenizer (Ultra Stirrer) at 18,000 rpm for 3 min to produce the emulsions. Approximately 140 mL of each emulsion was obtained, stored in closed vials, and refrigerated at 8°C–10°C. For attenuated total reflectance–Fourier transform infrared spectroscopy (ATR-FTIR) analysis, approximately 20 mL of each emulsion was frozen and subsequently freeze-dried using a L101 freeze dryer (Liobras, São Paulo, SP, Brasil).
Statistical analysis was performed using one-way analysis of variance (ANOVA) in Microsoft Excel® (version 2010). The planning matrix and ANOVA results containing A, B and interaction factor AB independent variables are available in supplementary materials (Table S1).
2.3 Nanoemulsion Characterization
2.3.1 Dynamic Light Scattering (DLS)
Dynamic Light Scattering (DLS) was used to assess the long-term physicochemical stability of the nanoemulsions by measuring particle size, polydispersity index (PDI), and zeta potential 45 days post-formulation. Measurements were conducted using a Malvern Zetasizer (Malvern Instruments, Worcestershire, UK). Given the metastable nature of nanoemulsions, early-stage measurements—particularly within the first week—may not accurately reflect the stabilized physicochemical profile of the system. Prior to analysis, samples were diluted in distilled water to a final concentration of 0.1% (v/v) and stirred continuously for 10 h to ensure complete dispersion of the matrix in the aqueous medium.
2.3.2 Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy (ATR-FTIR)
ATR-FTIR was employed to identify and characterize the functional groups present in the compounds forming nanoemulsion. Spectral data were acquired using a Perkin-Elmer 2000 spectrophotometer over the wavenumber range of 600–4000 cm−1. Prior to analysis, samples were homogenized with potassium bromide (KBr) at a 1:10 ratio and compressed into translucent pellets to ensure optimal spectral resolution.
2.3.3 Scanning Electron Microscopy (SEM)
Scanning Electron Microscopy (SEM) was performed to investigate the morphological characteristics of nanoemulsions. Approximately 200 μL of each sample was deposited onto metallic substrates (Brazilian 25-cent coins) and dehydrated in a drying oven at 50°C for approximately 2 h, resulting in the formation of a thin film. The dried samples were sputter-coated with a thin layer of gold using a coating system (Electron Microscopy Sciences, Hatfield, PA, USA) and subsequently examined under a Zeiss 940A microscope at an accelerating voltage of 20 kV and magnifications ranging from 5000× to 30,000×.
2.4 Experimental Infection with H. contortus
Two 4 months-old-sheep males weighing 15 to 16 kg, of undefined breed were placed in metabolic cages and dewormed with levamisole (Ripercol®, Campinas, SP, Brasil), ivermectin (Ivomec®, Itapecerica da Serra, SP, Brasil) and monepantel (Zolvix®, Bragança Paulista, SP, Brasil) according to the manufacturer’s instructions. One animal was monospecifically infected with 5000 H. contortus third-stage larvae (L3) and used as a source of eggs and larvae. The other animal remained uninfected as a source of nematode-free feces. The H. contortus Kokstad isolate, which is resistant to benzimidazoles, levamisole and macrocyclic lactones [33,34], was used for in vitro trials, including the egg hatch test (EHT) and the larval development test (LDT).
The EHT was performed according to [35]. Initially, feces from the animal harboring H. contortus were collected directly from the rectum and processed to obtain a suspension containing parasite eggs. Briefly, 250 μL of the egg suspension containing approximately 100 eggs was incubated for 48 h at 25°C with 250 μL of the following treatments: EsEO (0.2, 0.4, 0.5, 0.6, 0.8, 1.0 and 2 mg/mL) and the four emulsions (0.05, 0.1, 0.125, 0.15, 0.2, 0.25 and 0.5 mg/mL). The EsEO and emulsions were diluted in 3% Tween 80 (v/v in distilled water) and distilled water, respectively. This test was conducted with three controls: 3% Tween 80 and ALG:Tween 80 matrix (0.5 mg/mL) as negative controls and 0.05 mg/mL thiabendazole as a positive control.
After incubation, drops of Lugol solution were added to stop egg hatching, and eggs and first-stage larvae (L1) were counted under a microscope. Three repetitions with five replicates for each treatment and for each control were performed.
The effect of each treatment on the EHT was determined following the formula: Ovicidal effect = (number of eggs/number of eggs + number of L1) × 100.
2.6 Microscopic Evaluation of H. contortus Eggs
Confocal laser scanning microscopy (CLSM) of H. contortus eggs was performed to evaluate changes in morphology, viability and larval formation induced by the treatment. The samples were prepared similarly to the EHT, with some modifications. The incubation time was approximately 24 h (25°C) and after this period, the samples were refrigerated (8°C) until analysis within 24 h post-incubation. The highest concentrations of EsEO (2 mg/mL) and sodium alginate emulsions (0.5 mg/mL) were evaluated. The negative and positive controls were the same as those for the EHT.
The eggs were stained with Live/DeadTM (Invitrogen, São Paulo, SP, Brasil) containing SYTO9 and propidium iodide. The samples were subsequently evaluated with a Nikon C2 confocal microscope (NIKON, Tokyo, Japan) and visualized with a 40× objective lens at 488 nm to detect SYTO9, for live or viable eggs/larvae, and at 561 nm, to detect propidium iodide, for dead or damaged eggs/larvae. An aliquot of each sample was also evaluated using an optical microscope (OLYMPUS) with a 40× objective lens, for comparison with the CLSM results.
2.7 Larval Development Test (LDT)
The LDT was performed as described by [35,36]. An egg suspension was incubated for 24 h at 25°C to obtain an H. contortus L1 solution. Next, 500 μL of larval suspension containing approximately 250 L1 and 500 μL EsEO at 2–8 mg/mL or the emulsions at the same concentrations were incubated with 1 g of nematode-free feces for six days at 27°C.
After incubation, L3 were recovered according to the method described by [37] and drops of Lugol solution were added. The L3 were counted under a light microscope. The following controls were employed: Tween 80 3% and ALG:Tween 80 matrix (8 mg/mL) as negative controls and 0.008 mg/mL ivermectin as a positive control. Three repetitions with five replicates for each treatment and for each control were performed.
In the LDT, the following formula was used: Larvicidal effect = [(number of L3 in the negative control − number of L3 in the treated group)/number of L3 in the negative control] × 100.
The EHT and LDT results were analyzed using an analysis of variance (ANOVA) in GraphPad Prism® 5.0.
A one-way ANOVA followed by Tukey’s test was used to compare data from the same treatment (p < 0.05).
Unpaired Student’s t-tests were performed for analysis of the same concentration between emulsions. The results are expressed as the mean percentage effect on egg hatching and larval development inhibition ± standard deviation. The effective concentration to inhibit 50% (EC50) of egg hatching and the EC50 of larval development were determined by linear regression using the SPSS 17.0 program and the ratio between the EC50 of EsEO and emulsions was calculated as follows: EC50 ratio = EC50 of EsEO/EC50 of emulsion.
To confirm the hypothesis regarding the enhanced physicochemical stability and biological efficacy of sodium alginate-based nanoemulsions containing E. staigeriana essential oil, a series of analytical techniques was employed. ATR-FTIR was used to identify the functional groups of the compounds forming the nanoemulsion, while SEM provided insights into the morphological characteristics of the emulsified system. Importantly, DLS measurements were conducted 45 days post-formulation to evaluate whether the nanoemulsions maintained their colloidal stability over time, considering the metastable nature of such systems.
3.1 Essential Oil Characterization
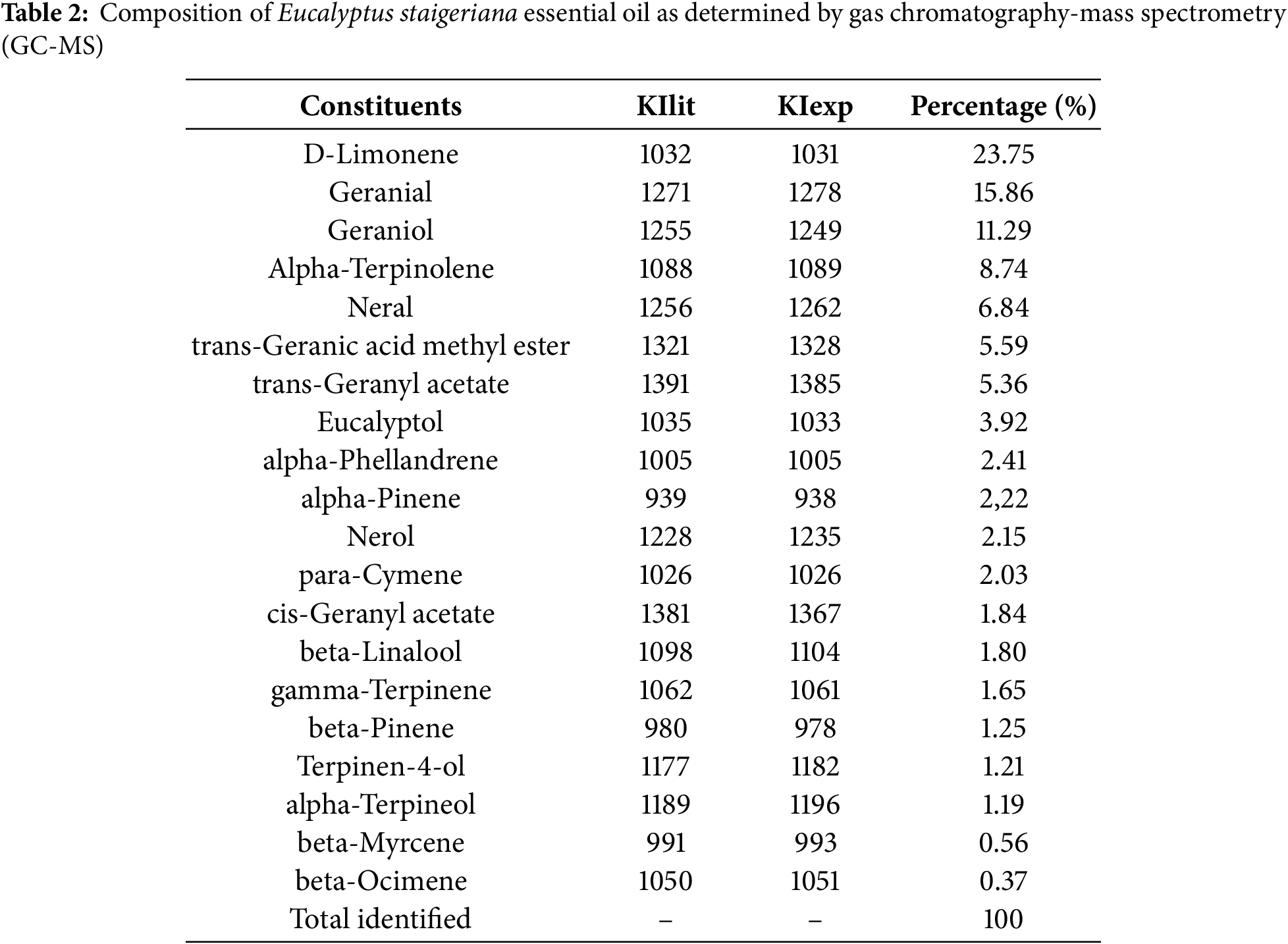
The chemical composition of EsEO is shown in Table 2. D-limonene (23.7%), geranial (15.9%) and trans-geraniol (11.3%) were the three main constituents identified in the GC-MS analysis.
The stability parameters of emulsions formulated with different Tween/EsEO and ALG/EsEO ratios showed notable variation across the experimental conditions. The average particle size ranged from 223.33 nm (E1) to 334.63 nm (E3), indicating that the formulations produced emulsions within the nanometric scale. Most samples exhibited particle sizes below 300 nm, except for E4. The polydispersity index (PDI) varied between 0.288 (E1) and 0.448 (E3), suggesting a relatively homogeneous particle size distribution across formulations, with E1 showing the lowest PDI. Zeta potential values ranged from −31.53 mV (E1) to −47.87 mV (E2). All samples exhibited negative surface charges, with magnitudes greater than −30 mV, which may indicate good electrostatic stability of the emulsions.
The FTIR spectra of EsEO, ALG, Tween 80 and the four nanoemulsions are shown in Fig. 1. The essential oil showed bands at 2962 cm−1 corresponding to the stretching vibration of methyl groups (–CH3), at 2929 cm−1 due to methylene groups (–CH2), at 1730 cm−1 corresponding to carbonyl C=O stretching, at 1676 cm−1 due to C=C stretching and at 1445 cm−1 due to C–H deformation [12,38]. ALG showed bands approximately 1800 and 1620 cm−1 corresponding to COO− stretching, and at 1234 cm−1, attributed to COH stretching [39,40]. Tween 80 showed a methyl group absorption band at 2918 cm−1, a band approximately 2860 cm−1 due to –CH2 stretching, a band at 1735 cm−1 due to C=O and a band at 1095 cm−1 corresponding to C–O–C stretching [41,42]. The nanoemulsions exhibited some bands corresponding to their main constituents. The presence of the essential oil at 1676 cm−1 was attributed to C=C stretching. The peaks at 2920 cm−1 and 1735 cm−1 were attributed to the –CH2 stretching and C=O stretching of Tween 80, respectively. Additionally, the peak at 1240 cm−1 may indicate the presence of ALG due to COH stretching.
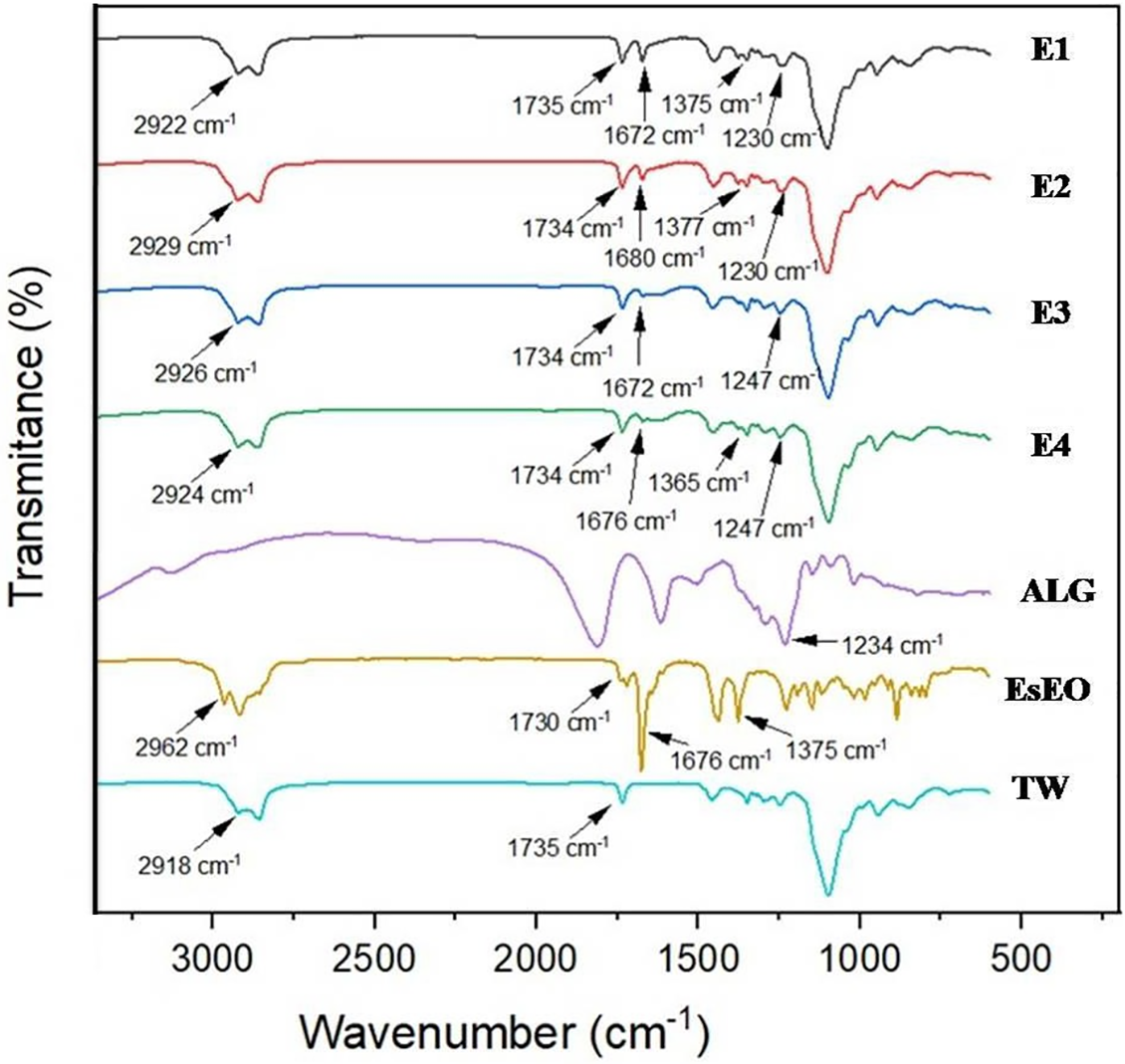
Figure 1: FTIR spectra of Tween 80 (TW), Eucalyptus staigeriana essential oil (EsEO), sodium alginate (ALG) and the four essential oil emulsions (E1, E2, E3 and E4)
Scanning Electron Microscopy (SEM)
SEM images of the emulsions are shown in Fig. 2. Spherical oil droplets with diameters below 200 nm were observed dispersed throughout the sodium alginate matrix. The droplets exhibited uniform morphology and were well distributed within the polymeric network, confirming the nanoscale structure and effective encapsulation of the essential oil.

Figure 2: Scanning electron microscopy surface images of the Eucalyptus staigeriana essential oil emulsions: emulsion 1 (A), emulsion 2 (B), emulsion 3 (C) and emulsion 4 (D)
The effects of EsEO and sodium alginate emulsions (E1, E2, E3 and E4) in the EHT against H. contortus are shown in Table 3.

The essential oil and emulsions exhibited ovicidal activity in a dose-dependent manner. Even the lowest concentrations (0.2 EsEO and 0.05 mg/mL emulsion) differed from the negative control (p < 0.05). The EC50 values and the ratios between the EC50 values of the oil and emulsions in the EHT are summarized in Table 4. All four sodium alginate emulsions had lower EC50 values than that of EsEO, with the lowest EC50 of 0.088 mg/mL and the highest EC50 ratio (3.5) among the emulsions.
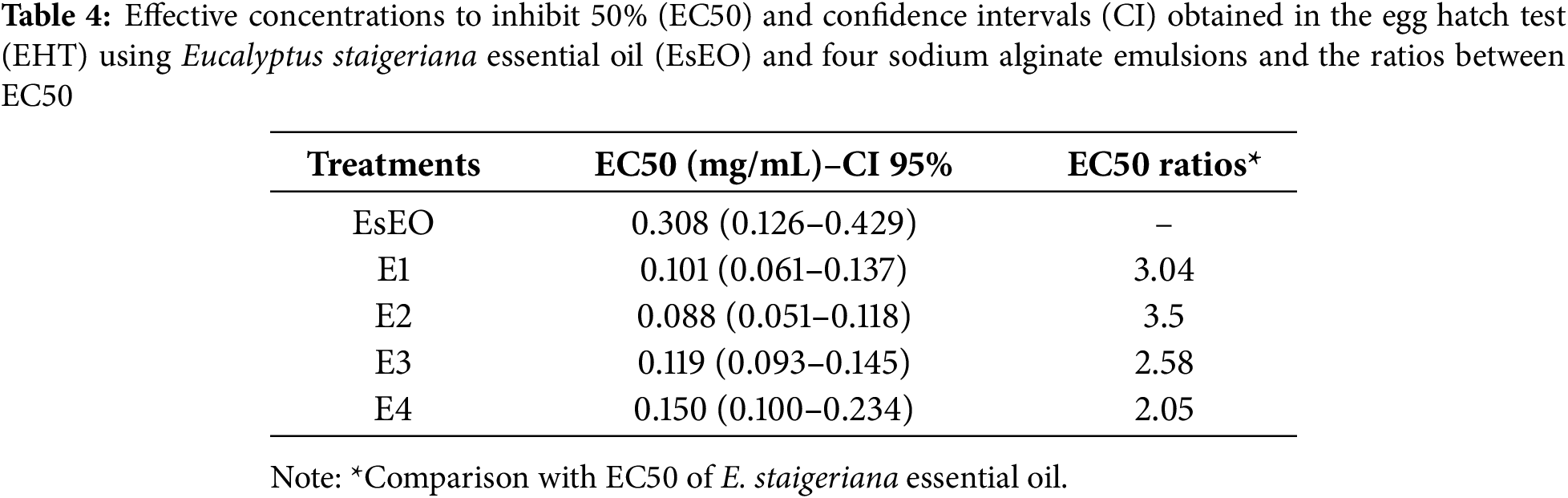
Microscopic Evaluation of H. contortus Eggs
The H. contortus eggs exposed to EsEO and sodium alginate emulsions and evaluated via CLSM, and optical microscopy are shown in Fig. 3. In the positive and negative controls, the eggs were weakly stained. In the negative control, the larvae were fully formed and mobile. The larvae stained green and red, but this could only be observed after hatching, indicating larval integrity with damaged areas. In the positive control, which was exposed to thiabendazole, there was no larval formation, and only an amorphous mass of cells inside the eggs. In the eggs exposed to EsEO and sodium alginate emulsions, larvae were formed but immobile. The larvae were also stained green, but in the emulsion group, there were also red-stained spots along the larvae.
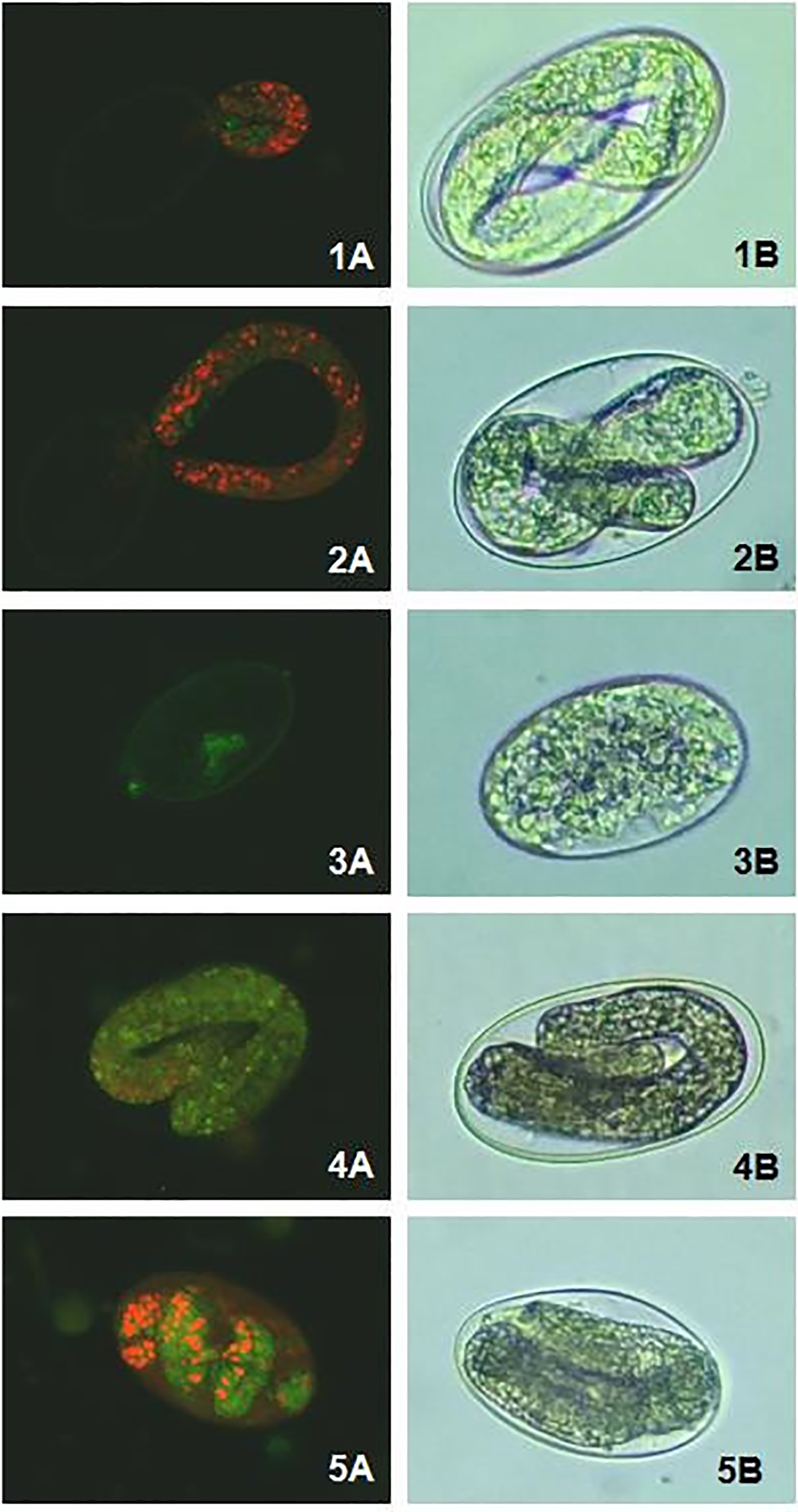
Figure 3: Confocal laser scanning microscopy (A) and optical microscopy (B) images of Haemonchus contortus eggs exposed to: Tween 80 (1), the encapsulating matrix (2), thiabendazole (3), Eucalyptus staigeriana essential oil (4) and essential oil emulsions (5)
3.3 Larval Development Test (LDT)
EsEO and the emulsions inhibited the larval development of H. contortus in a dose-dependent manner as shown in Table 5.
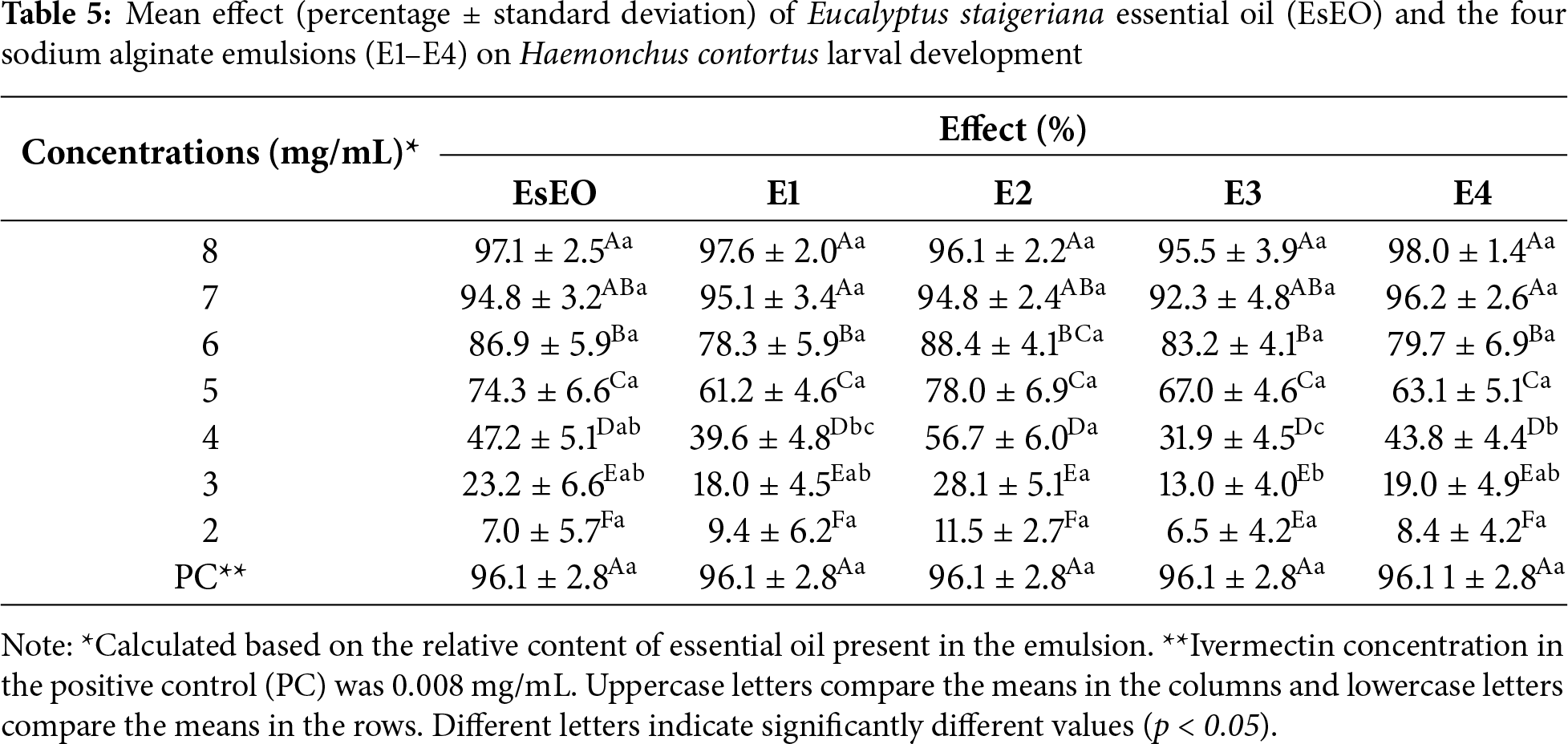
At higher concentrations (7 and 8 mg/mL), the effect was greater than 90%. These results were similar to those of positive control. The EC50 values and the ratios between the EC50 values of oil and emulsions in LDT are summarized in Table 6. Only E2 had a lower EC50 than the essential oil. All emulsions showed EC50 ratios close to 1. Thus, based on the physical-chemical characterization and effects on H. contortus eggs and larvae, E2, which had the highest proportion of essential oil: (Tween 80)/3 (EsEO)/6 (ALG) (v/v), was considered the most promising emulsion.
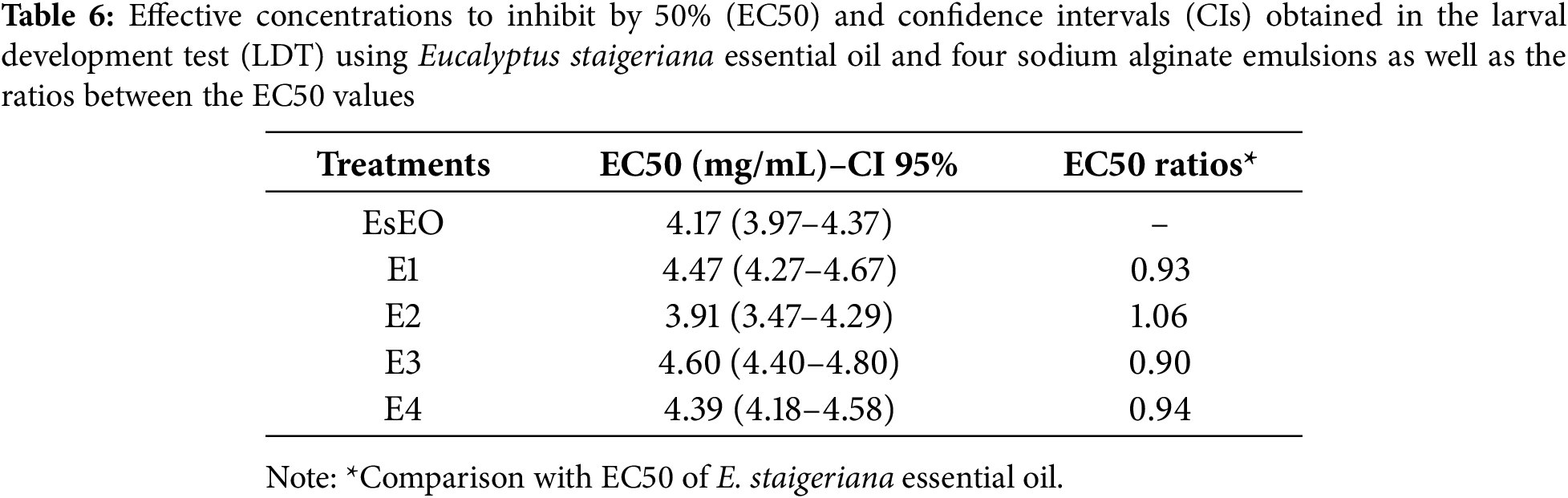
The essential oils are characterized by large quantities of volatile compounds, mainly terpenes and terpenoids. Phytochemical analysis has shown that the constituents of an essential oil may differ within the same Eucalyptus species in response to factors such as climate, soil composition, age, the part of the plant used and the extraction method. This variation in the qualitative and quantitative composition of essential oils directly influences their medicinal properties and therefore their therapeutic applications [10,11,24,43].
In this study, the EsEO was rich in D-limonene (23.7%) and geranial (15.9%). Limonene and geranial (E-citral/trans-citral) are often found as major constituents of EsEO, ranging from 14.83% to 72.9% (limonene) and from 7.98% to 23.48% (geranial) of the essential oil content [11,44,45]. However, EsEO compositions where limonene is not a major compound (7%) [43] or were not identified [11,32], have also been reported.
Limonene and geranial have anthelmintic activities. D-limonene had an EC50 value of 2.62 mg/mL for motility inhibition of the Caenorhabditis elegans Bristol N2 strain [46]. However, it was less effective against H. contortus eggs, with an EC50 value of 207.56 mg/mL, presenting the lowest effect among the ten essential oil constituents [47].
Moreover, geranial exhibited the highest anthelmintic activity against adult C. elegans among thirteen compounds from Cinnamomum bejolghota with an EC50 value of 57.55 μg/mL in the adult worm mortality assay and 47.65 μg/mL in the EHT [48].
Plant essential oils are lipophilic and have limited solubility in water. However, their solubility can be altered by the use of surfactants, such as Tween 80, which is considered one of the best surfactants for the preparation of nanoemulsions due to its relatively high hydrophilic-lipophilic balance. Surfactants or emulsifiers are molecules capable of adsorbing at the oil-water interface, reducing the interfacial tension and sustaining the stability of oil droplets [49].
With respect to the nanoemulsion composition, the Tween 80/EsEO ratio is the main factor that directly influences particle size stability. At a higher content a significant decrease in particle size was observed. In this context, a higher content of Tween provides stronger stabilization of the essential oil in the micellar domains, which favors a lower particle size. There is also an effect of the ALG/EsEO ratio, which increases sodium alginate content resulting in larger particle sizes, because of the external coating of the polysaccharide. The zeta potential results are related to the particle surface charge, and all formulations presented values below −30 mV, a sign of stabilization of the nanoemulsion.
There is some uncertainty about the droplet size cutoff of an emulsion that is considered a nanoemulsion. Systems with mean droplet sizes ranging from 20–100 nm, <200 nm, and <1000 nm were classified as nanoemulsions [50]. Therefore, in this study, the four essential oil emulsions were considered nanoemulsions, as most of the particles were smaller than 350 nm. A smaller droplet size results in greater stability with respect to gravitational separation and aggregation [51,52].
The emulsions E2 (1:3:6-Tween 80:EsEO:sodium alginate) and E3 (1:2:6-Tween 80:EsEO:sodium alginate) presented small fractions of 2.56% and 2.43% intensity, respectively, of the oil droplet size on a micrometric scale of approximately 5.000 nm. This increase in particle size may be due to the coalescence process in which smaller droplets collide and merge, resulting in larger droplets [53,54]. However, approximately 98% of the droplets were on the nanoscale 45 days after the development of the emulsions, suggesting that the ratios of Tween 80:EsEO:ALG were sufficient for the emulsion formulation.
The PDI value indicates the width of the particle size distribution ranging from 0 (dispersed) to 1 (aggregated). The PDIs of the four sodium alginate emulsions ranged from 0.288 to 0.448, demonstrating that the droplets were distributed throughout the dispersion media [55].
The zeta potential of the four emulsions ranged from −31.3 to −47.9. Typically, particles with zeta potentials greater than +30 mV or less than −30 mV are considered stable [56]. The electric charge of the droplets is due to the adsorption of ionized emulsifiers or biopolymers. Because Tween 80 is a nonionic surfactant, the negative zeta potential values are attributed to the sodium alginate carboxyl groups (-COO) on the surface of the droplet, indicating that the coating was successful [57].
SEM analysis revealed the presence of uniformly spherical oil droplets with diameters below 200 nm, finely dispersed throughout the sodium alginate matrix. The dehydration method employed, drying the emulsions on metallic substrates, was effective in preserving the native morphology of the nanodroplets. This structural integrity is likely maintained by the surface charge of the particles, as indicated by zeta potential values below –30 mV. Such negative surface potential promotes electrostatic repulsion between droplets, preventing aggregation and contributing to the long-term colloidal stability of the nanoemulsion system.
The EsEO and the four sodium alginate emulsions inhibited egg hatching and larval development in H. contortus. However, the expected increase in effect for the emulsions, in comparison with the essential oil, was observed only in the EHT, as indicated by EC50 values for the alginate emulsions that were at least 2 times lower than those of the EsEO EC50 values. In comparison, in the LDT, the EC50 values of the essential oil and the emulsions were similar. Similar findings were reported by [58] when comparing the ovicidal and larvicidal effects of free Eucalyptus citriodora essential oil, EsEO and its emulsions with a chitosan:Tween 80 encapsulating matrix. The EC50 values of egg hatching for the free EsEO and E. citriodora essential oil were 0.7 and 1.3 mg/mL, respectively, and those for the emulsions were 0.2 and 0.4 mg/mL, moreover, the EC50 values of larval development for the free EsEO and E. citriodora essential oil were 1.8 and 1.7 mg/mL, respectively, and those for the emulsions were 2.3 and 1.7 mg/mL [58].
The effect of the emulsions on the LDT did not increase as expected. This result may be due to the conditions under which the test was performed. The incubation period for the EHT is 48 h and it takes place in an aqueous medium [36]. In contrast, the samples in the LDT are incubated for 7 days in a solid medium composed of sheep feces [37]. In this context, the aqueous medium favored oil release, whereas in the solid medium, the dehydration that occurs via the absorption of water by feces and via evaporation may impact the stability of the droplets, and consequently oil release. However, the abomasum, an organ that harbors H. contortus, secretes water, enzymes, hormones and HCl, and is likely a favorable environment for the release of the oil [59].
As observed in the CLSM and optical microscopy images, the eggs exposed to the highest EsEO and sodium alginate emulsion concentrations did not hatch, but the L1 inside them developed. This is due mainly to the eggshell, which is composed of three layers: the external vitelline layer, the medial chitinous layer and the basal lipid/protein layer. As development progresses from morula to larva and with the proximity of hatching, the permeability of the eggshell to water and other compounds increases [60,61]. However, as observed via CLSM, the eggs exposed to EsEO and sodium alginate emulsions were more permeable to the dyes and stained distinctly from the eggs exposed to the negative and positive controls. Thus, we assume that EsEO is responsible for promoting deterioration in the outer layer and/or interacting with the inner lipidic layer, changing eggshell fluidity, which is important for maintaining egg impermeability [62,63].
The effective control of gastrointestinal nematodes in small ruminants remains a major challenge in livestock production, particularly due to the rapid development of resistance to conventional anthelmintic drugs. This study presents a promising alternative based on a natural product with global distribution—E. staigeriana essential oil (EsEO) encapsulated in sodium alginate-based nanoemulsions produced via a high-energy method. These formulations successfully addressed the volatility limitations of EsEO, demonstrating enhanced physicochemical stability and significant biological activity against H. contortus. Among the four emulsions tested, formulation E2 (Tween 80:EsEO: sodium alginate at a 1:3:6 ratio) emerged as the most promising candidate, combining optimal droplet size distribution, high colloidal stability (zeta potential <–30 mV), and superior ovicidal efficacy, with an EC50 value of 0.088 mg/mL, more than three times lower than that of free EsEO. Although larvicidal effects were comparable between emulsions and the essential oil, the enhanced inhibition of egg hatching by E2 suggests its potential to disrupt the parasite’s life cycle more effectively. The aqueous nature of the abomasum, where H. contortus resides, is likely to favor the release of EsEO from the nanoemulsion, further supporting its suitability for in vivo application. By integrating principles of green chemistry and advanced materials nanotechnology, this work exemplifies a sustainable and innovative approach to veterinary therapeutics, leveraging biodegradable polymers and plant-derived bioactives to develop efficient, eco-friendly anthelmintic delivery systems.
From a scientific perspective, this study contributes to the advancement of nanotechnology in veterinary parasitology by demonstrating the feasibility of using biopolymer-based nanoemulsions for targeted delivery of botanical anthelmintics. Practically, formulation E2 offers a sustainable, cost-effective, and environmentally friendly alternative for parasite control in small ruminants, with the potential to reduce reliance on synthetic drugs. Therefore, E2 should be prioritized for future in vivo studies targeting adult H. contortus, with the ultimate goal of integrating this formulation into comprehensive parasite management strategies in livestock systems.
Acknowledgement: The authors would like to thank the Central Analítica-UFC (funded by Finep-CT-INFRA, CAPES-Pró-Equipamentos, and MCTI-CNPq-SisNano2.0) for microscopy measures.
Funding Statement: This work has been supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (grant No. 142165/2018-2). Mr. Araújo-Filho had a doctoral scholarship and Dr. Bevilaqua has a research fellowship (grant No. 305911/2019-8) from CNPq.
Author Contributions: Conceptualization: Claudia M. L. Bevilaqua, Flávia O. M. da Silva Abreu, Lorena M. B. de Oliveira; methodology: Claudia M. L. Bevilaqua, Flávia O. M. da Silva Abreu, Débora S. C. M. Castelo Branco; validation: Ana Carolina F. L. Melo; investigation, José Vilemar de Araújo-Filho, Livia F. Ximenes, Henety N. Pinheiroo, Wesley L. C. Ribeiro; formal analysis: José Vilemar de Araújo-Filho, Henety N. Pinheiroo, Weibson P. P. André; resources: Claudia M. L. Bevilaqua; writing—original draft preparation: José Vilemar de Araújo-Filho; writing—review and editing: Claudia M. L. Bevilaqua; visualization: Ana Carolina F. L. Melo, Wesley L. C. Ribeiro; funding acquisition supervision, Claudia M. L. Bevilaqua, Lorena M. B. de Oliveira. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Materials: Data supporting the results are available upon request from the Laboratorio de Doenças Parasitárias do Programa de Pós-graduação da Faculdade de Veterinária da Universidade Estadual do Ceara.
Ethics Approval: This study was approved by the Ethics Commission on Animal Use of the Universidade Estadual do Ceará and registered under the number 9472634/2018.
Conflicts of Interest: The authors declare no conflicts of interest to report regarding the present study.
Supplementary Materials: The supplementary material is available online at https://www.techscience.com/doi/10.32604/jrm.2025.02025-0138/s1.
References
1. Sluijs LV, Geisen S, Lozano-Torres JL, Sterken MG, Vervoort MTW, Wilbers RHP. Unearthing roundworms: nematodes as determinants of human health. One Health. 2025;21:101103. doi:10.1016/j.onehlt.2025.101103. [Google Scholar] [PubMed] [CrossRef]
2. Zheng Y, Young ND, Wang T, Chang BCH, Song J, Gasser RB. Systems biology of Haemonchus contortus—advancing biotechnology for parasitic nematode control. Biotechnol Adv. 2025;81(3):108567. doi:10.1016/j.biotechadv.2025.108567. [Google Scholar] [PubMed] [CrossRef]
3. Zajac AM, Garza J. Biology, epidemiology, and control of gastrointestinal nematodes of small ruminants. Vet Clin North Am Food Anim Pract. 2020;36(1):73–87. doi:10.1016/j.cvfa.2019.12.005. [Google Scholar] [PubMed] [CrossRef]
4. Baudinette E, O’Handley R, Trengove C. Anthelmintic resistance of gastrointestinal nematodes in goats: a systematic review and meta-analysis. Vet Parasitol. 2022;312(7414):109809. doi:10.1016/j.vetpar.2022.109809. [Google Scholar] [PubMed] [CrossRef]
5. Rodrigues JFV, Monteiro JP, Almeida TM, Molento MB. A systematic review of the molecular mechanisms related to anthelmintic resistance in Haemonchus contortus: a contemporary narrative. Vet Parasitol. 2025;334:110394. doi:10.1016/j.vetpar.2025.110394. [Google Scholar] [PubMed] [CrossRef]
6. Ureña MJE, Katchborian-Neto A, Garcez Neto JR, da Silva Lopes FF, de Morais SM, Narciso dos Reis VE, et al. Simarouba berteroana Krug & Urb. extracts and fractions possess anthelmintic activity against eggs and larvae of multidrug-resistant Haemonchus contortus. Vet Sci. 2025;12(2):90. doi:10.3390/vetsci12020090. [Google Scholar] [CrossRef]
7. Hoste H, Meza-OCampos G, Marchand S, Sotiraki S, Sarasti K, Blomstrand BM, et al. Use of agro-industrial by-products containing tannins for the integrated control of gastrointestinal nematodes in ruminants. Parasite. 2022;29(7):10. doi:10.1051/parasite/2022010. [Google Scholar] [PubMed] [CrossRef]
8. Ali R, Rooman M, Mussarat S, Norin S, Ali S, Adnan M, et al. A systematic review on comparative analysis, toxicology, and pharmacology of medicinal plants against Haemonchus contortus. Front Pharmacol. 2021;12:644027. doi:10.3389/fphar.2021.644027. [Google Scholar] [PubMed] [CrossRef]
9. Bakkali F, Averbeck S, Averbeck D, Idaomar M. Biological effects of essential oils—a review. Food Chem Toxicol. 2008;46(2):446–75. doi:10.1016/j.fct.2007.09.106. [Google Scholar] [PubMed] [CrossRef]
10. Chandorkar N, Tambe S, Amin P, Madankar C. A systematic and comprehensive review on current understanding of the pharmacological actions, molecular mechanisms, and clinical implications of the genus Eucalyptus. Phytomed Plus. 2021;1(4):100089. doi:10.1016/j.phyplu.2021.100089. [Google Scholar] [CrossRef]
11. da Silva PPM, de Oliveira J, dos Mares Biazotto A, Parisi MM, da Glória EM, Spoto MHF. Essential oils from Eucalyptus staigeriana F. Muell. ex Bailey and Eucalyptus urograndis W. Hill ex Maiden associated to carboxymethylcellulose coating for the control of Botrytis cinerea Pers. Fr. and Rhizopus stolonifer (Ehrenb.: Fr.) Vuill. in strawberries. Ind Crops Prod. 2020;156:112884. doi:10.1016/j.indcrop.2020.112884. [Google Scholar] [CrossRef]
12. Herculano ED, de Paula HCB, de Figueiredo EAT, Dias FGB, de Pereira VA. Physicochemical and antimicrobial properties of nanoencapsulated Eucalyptus staigeriana essential oil. LWT Food Sci Technol. 2015;61(2):484–91. doi:10.1016/j.lwt.2014.12.001. [Google Scholar] [CrossRef]
13. Braga BCF, Alves DS, Lima AF, Oliveira JAC, Figueiredo KG, Carvalho VC, et al. Lethal effect and two-sex life table of Tuta absoluta (meyrick) treated with Melaleuca alternifolia and Eucalyptus staigeriana essential oils. Horticulturae. 2025;11(8):951. doi:10.3390/horticulturae11080951. [Google Scholar] [CrossRef]
14. Kalaivani K. Efficacy of Eucalyptus oil as a sustainable biopesticide for population suppression of Sylepta derogata Fab (Lepidoptera: pyralidaean okra pest. J Nat Pestic Res. 2025;13(2):100139. doi:10.1016/j.napere.2025.100139. [Google Scholar] [CrossRef]
15. Macedo ITF, Bevilaqua CML, de Oliveira LMB, Camurça-Vasconcelos ALF, Vieira LS, Oliveira L, et al. Anthelmintic effect of Eucalyptus staigeriana essential oil against goat gastrointestinal nematodes. Vet Parasitol. 2010;173(1–2):93–8. doi:10.1016/j.vetpar.2010.06.004. [Google Scholar] [PubMed] [CrossRef]
16. Tian Q, Zhou W, Cai Q, Ma G, Lian G. Concepts, processing, and recent developments in encapsulating essential oils. Chin J Chem Eng. 2021;30(11):255–71. doi:10.1016/j.cjche.2020.12.010. [Google Scholar] [CrossRef]
17. Chen H, Su X, Luo Y, Liao Y, Wang F, Huang L, et al. Natural-derived porous nanocarriers for the delivery of essential oils. Chin J Nat Med. 2024;22(12):1117–33. doi:10.1016/S1875-5364(24)60731-4. [Google Scholar] [PubMed] [CrossRef]
18. Sousa VI, Parente JF, Marques JF, Forte MA, Tavares CJ. Microencapsulation of essential oils: a review. Polymers. 2022;14(9):1730. doi:10.3390/polym14091730. [Google Scholar] [PubMed] [CrossRef]
19. Pavoni L, Perinelli DR, Bonacucina G, Cespi M, Palmieri GF. An overview of micro- and nanoemulsions as vehicles for essential oils: formulation, preparation and stability. Nanomater. 2020;10(1):135. doi:10.3390/nano10010135. [Google Scholar] [PubMed] [CrossRef]
20. Zhang Y, Wang QC, Yu H, Zhu J, de Lange K, Yin Y, et al. Evaluation of alginate-whey protein microcapsules for intestinal delivery of lipophilic compounds in pigs. J Sci Food Agric. 2016;96(8):2674–81. doi:10.1002/jsfa.7385. [Google Scholar] [PubMed] [CrossRef]
21. Marquis M, Alix V, Capron I, Cuenot S, Zykwinska A. Microfluidic encapsulation of Pickering oil microdroplets into alginate microgels for lipophilic compound delivery. ACS Biomater Sci Eng. 2016;2(4):535–43. doi:10.1021/acsbiomaterials.5b00522. [Google Scholar] [PubMed] [CrossRef]
22. Azad AK, Doolaanea AA, Al-Mahmood SMA, Kennedy JF, Chatterjee B, Bera H. Electro-hydrodynamic assisted synthesis of lecithin-stabilized peppermint oil-loaded alginate microbeads for intestinal drug delivery. Int J Biol Macromol. 2021;185(2):861–75. doi:10.1016/j.ijbiomac.2021.07.019. [Google Scholar] [PubMed] [CrossRef]
23. Lopedota AA, Arduino I, Lopalco A, Iacobazzi RM, Cutrignelli A, Laquintana V, et al. From oil to microparticulate by prilling technique: production of polynucleate alginate beads loading Serenoa repens oil as intestinal delivery systems. Int J Pharm. 2021;599:120412. doi:10.1016/j.ijpharm.2021.120412. [Google Scholar] [PubMed] [CrossRef]
24. Ong WD, Tey BT, Quek SY, Tang SY, Chan ES. Alginate-based emulsion template containing high oil loading stabilized by nonionic surfactants. J Food Sci. 2015;80(1):E93–100. doi:10.1111/1750-3841.12729. [Google Scholar] [PubMed] [CrossRef]
25. Rehman A, Tong Q, Sharif HR, Korma SA, Karim A, Manzoor MF, et al. Biopolymer based nanoemulsion delivery system: an effective approach to boost the antioxidant potential of essential oil in food products. Carbohydr Polym Technol Appl. 2021;2(46):100082. doi:10.1016/j.carpta.2021.100082. [Google Scholar] [CrossRef]
26. Silva KFCE, da Silva Carvalho AG, Santos Rabelo R, Hubinger MD. Sacha inchi oil encapsulation: emulsion and alginate beads characterization. Food Bioprod Process. 2019;116(1–3):118–29. doi:10.1016/j.fbp.2019.05.001. [Google Scholar] [CrossRef]
27. do Nascimento JF, da Silva Abreu FOM, Holanda T, Castelo RM, dos Santos HS, de Menezes JESA, et al. Evaluation of stability and anxiolytic potential of oil-in-water polysaccharide nanoemulsions loaded with chalcone (1E,4E)-1,5-bis(4-methoxyphenyl) penta-1,4-dien-3-one. Chem Pap. 2024;78(17):9205–22. doi:10.1007/s11696-024-03738-2. [Google Scholar] [CrossRef]
28. Pandey VK, Islam RU, Shams R, Dar AH. A comprehensive review on the application of essential oils as bioactive compounds in nano-emulsion based edible coatings of fruits and vegetables. Appl Food Res. 2022;2(1):100042. doi:10.1016/j.afres.2022.100042. [Google Scholar] [CrossRef]
29. Karsli GT, Sahin S, Oztop MH. High-pressure-homogenized clove and thyme oil emulsions: formulation, stability, and antioxidant capacity. ACS Food Sci Technol. 2022;2(12):1832–9. doi:10.1021/acsfoodscitech.2c00231. [Google Scholar] [CrossRef]
30. da Silva Abreu FOM, Lima MF, de Oliveira BP, Lima MR, de Araújo Filho JV, Moisés Aguiar AAR, et al. Production of carboxymethylated xanthan gum microemulsions for eugenol encapsulation. Macromol Symp. 2022;406(1):2200035. doi:10.1002/masy.202200035. [Google Scholar] [CrossRef]
31. Adams RP. Identification of essential oil components by gas chromatography/quadrupole mass spectroscopy. Oak Park, IL, USA: Allured; 2007. [Google Scholar]
32. da Silva Abreu FOM, Costa EF, Cardial MRL, André WPP. Polymeric nanoemulsions enriched with Eucalyptus citriodora essential oil. Polímeros. 2020;30(2):e2020024. doi:10.1590/0104-1428.00920. [Google Scholar] [CrossRef]
33. Neveu C, Charvet C, Fauvin A, Cortet J, Castagnone-Sereno P, Cabaret J. Identification of levamisole resistance markers in the parasitic nematode Haemonchus contortus using a cDNA-AFLP approach. Parasitology. 2007;134(8):1105–10. doi:10.1017/S0031182007000030. [Google Scholar] [PubMed] [CrossRef]
34. Fauvin A, Charvet C, Issouf M, Cortet J, Cabaret J, Neveu C. cDNA-AFLP analysis in levamisole-resistant Haemonchus contortus reveals alternative splicing in a nicotinic acetylcholine receptor subunit. Mol Biochem Parasitol. 2010;170(2):105–7. doi:10.1016/j.molbiopara.2009.11.007. [Google Scholar] [PubMed] [CrossRef]
35. Coles GC, Bauer C, Borgsteede FHM, Geerts S, Klei TR, Taylor MA, et al. World Association for the advancement of veterinary parasitology (WAAVP) methods for the detection of anthelmintic resistance in nematodes of veterinary importance. Vet Parasitol. 1992;44(1–2):35–44. doi:10.1016/0304-4017(92)90141-U. [Google Scholar] [PubMed] [CrossRef]
36. Frota GA, Dos Santos VO, Rodrigues JFV, Oliveira BR, Albuquerque LB, de Vasconcelos FRC, et al. Biological activity of cinnamaldehyde, citronellal, geraniol and anacardic acid on Haemonchus contortus isolates susceptible and resistant to synthetic anthelmintics. Rev Bras Parasitol Vet. 2023;32(2):e006023. doi:10.1590/S1984-29612023027. [Google Scholar] [PubMed] [CrossRef]
37. Roberts F, O’Sullivan PJ. Methods for egg counts and larval cultures for strongyles infesting the gastro-intestinal tract of cattle. Aust J Agric Res. 1950;1(1):99–102. doi:10.1071/ar9500099. [Google Scholar] [CrossRef]
38. Ribeiro WLC, Camurça-Vasconcelos ALF, dos Santos JML, Macedo ITF, Ribeiro JC, de Oliveira J, et al. The use of Eucalyptus staigeriana nanoemulsion for control of sheep haemonchosis. Pesq Vet Bras. 2017;37(3):221–6. doi:10.1590/s0100-736x2017000300004. [Google Scholar] [CrossRef]
39. Abreu FOMS, Bianchini C, Forte MMC, Kist TBL. Influence of the composition and preparation method on the morphology and swelling behavior of alginate-chitosan hydrogels. Carbohydr Polym. 2008;74(2):283–9. doi:10.1016/j.carbpol.2008.02.017. [Google Scholar] [CrossRef]
40. Chen J, Ouyang J, Cai X, Xing X, Zhou L, Liu Z, et al. Removal of ciprofloxacin from water by millimeter-sized sodium alginate/H3PO4 activated corncob-based biochar composite beads. Sep Purif Technol. 2021;276:119371. doi:10.1016/j.seppur.2021.119371. [Google Scholar] [CrossRef]
41. Kura AU, Hussein-Al-Ali SH, Hussein MZ, Fakurazi S. Preparation of Tween 80-Zn/Al-levodopa-layered double hydroxides nanocomposite for drug delivery system. Sci World J. 2014;2014(1-2):104246. doi:10.1155/2014/104246. [Google Scholar] [PubMed] [CrossRef]
42. Tai K, He X, Yuan X, Meng K, Gao Y, Yuan F. A comparison of physicochemical and functional properties of icaritin-loaded liposomes based on different surfactants. Colloids Surf A Physicochem Eng Aspects. 2017;518:218–31. doi:10.1016/j.colsurfa.2017.01.019. [Google Scholar] [CrossRef]
43. Almeida Barbosa LC, Filomeno CA, Teixeira RR. Chemical variability and biological activities of Eucalyptus spp. essential oils. Molecules. 2016;21(12):1671. doi:10.3390/molecules21121671. [Google Scholar] [PubMed] [CrossRef]
44. Pedrotti C, Franzoi C, Rosa MTS, Trentin TR, Vilasboa J, Scariot FJ, et al. Antifungal activity of essential oil from Eucalyptus staigeriana against Alternaria alternata causing of leaf spot and black rot in table grapes. An Acad Bras Cienc. 2022;94(2):e20200394. doi:10.1590/0001-3765202220200394. [Google Scholar] [PubMed] [CrossRef]
45. Ribeiro WLC, Camurça-Vasconcelos ALF, Macedo ITF, dos Santos JML, de Araújo-Filho JV, de Carvalho Ribeiro J, et al. In vitro effects of Eucalyptus staigeriana nanoemulsion on Haemonchus contortus and toxicity in rodents. Vet Parasitol. 2015;212(3–4):444–7. doi:10.1016/j.vetpar.2015.07.019. [Google Scholar] [CrossRef]
46. Lima AS, Costa Junior HNP, Costa-Junior LM, Monteiro OS, Maia JGS, da Rocha CQ. Anthelmintic effect of essential rhizome oil from Hedychium coronarium Koenig (Zingiberaceae) introduced in Northeastern Brazil. Acta Trop. 2021;218(2):105912. doi:10.1016/j.actatropica.2021.105912. [Google Scholar] [PubMed] [CrossRef]
47. Katiki LM, Barbieri AME, Araujo RC, Veríssimo CJ, Louvandini H, Ferreira JFS. Synergistic interaction of ten essential oils against Haemonchus contortus in vitro. Vet Parasitol. 2017;243(3):47–51. doi:10.1016/j.vetpar.2017.06.008. [Google Scholar] [CrossRef]
48. Gogoi B, Tamuli KJ, Neipihoi, Bordoloi M, Sharma HK. Isolation and characterization of chemical constituents with in vitro anti-hypertensive and anthelmintic activities of Cinnamomum bejolghota (Buch.-Ham.) sweet leaves: an ethno medicinal plant of North East India. S Afr N J Bot. 2021;140:161–6. doi:10.1016/j.sajb.2021.04.004. [Google Scholar] [CrossRef]
49. Das S, Vishakha K, Banerjee S, Mondal S, Ganguli A. Sodium alginate-based edible coating containing nanoemulsion of Citrus sinensis essential oil eradicates planktonic and sessile cells of food-borne pathogens and increased quality attributes of tomatoes. Int J Biol Macromol. 2020;162:1770–9. doi:10.1016/j.ijbiomac.2020.08.086. [Google Scholar] [PubMed] [CrossRef]
50. Shishir MRI, Xie L, Sun C, Zheng X, Chen W. Advances in micro and nano-encapsulation of bioactive compounds using biopolymer and lipid-based transporters. Trends Food Sci Technol. 2018;78:34–60. doi:10.1016/j.tifs.2018.05.018. [Google Scholar] [CrossRef]
51. Jaiswal M, Dudhe R, Sharma PK. Nanoemulsion: an advanced mode of drug delivery system. 3 Biotech. 2015;5(2):123–7. doi:10.1007/s13205-014-0214-0. [Google Scholar] [PubMed] [CrossRef]
52. McClements DJ. Emulsion design to improve the delivery of functional lipophilic components. Annu Rev Food Sci Technol. 2010;1(1):241–69. doi:10.1146/annurev.food.080708.100722. [Google Scholar] [PubMed] [CrossRef]
53. Tadros T, Izquierdo P, Esquena J, Solans C. Formation and stability of nano-emulsions. Adv Colloid Interface Sci. 2004;108:303–18. doi:10.1016/j.cis.2003.10.023. [Google Scholar] [PubMed] [CrossRef]
54. McClements DJ, Rao J. Food-grade nanoemulsions: formulation, fabrication, properties, performance, biological fate, and potential toxicity. Crit Rev Food Sci Nutr. 2011;51(4):285–330. doi:10.1080/10408398.2011.559558. [Google Scholar] [PubMed] [CrossRef]
55. Singh S, Lohani A, Mishra AK, Verma A. Formulation and evaluation of carrot seed oil-based cosmetic emulsions. J Cosmet Laser Ther. 2019;21(2):99–107. doi:10.1080/14764172.2018.1469769. [Google Scholar] [PubMed] [CrossRef]
56. McNeil SE. Characterization of nanoparticles intended for drug delivery. New York, NY, USA: Humana Press; 2011. DOI 10.1007/978-1-60327-198-1_6. [Google Scholar] [CrossRef]
57. Paiva Filho JC, de Morais SM, Nogueira Sobrinho AC, Cavalcante GS, da Silva NA, da Silva Abreu FOM. Design of chitosan-alginate core-shell nanoparticules loaded with anacardic acid and cardol for drug delivery. Polímeros. 2019;29(4):e2019060. doi:10.1590/0104-1428.08118. [Google Scholar] [CrossRef]
58. Ribeiro JC, Ribeiro WLC, Camurça-Vasconcelos ALF, Macedo ITF, Santos JML, Paula HCB, et al. Efficacy of free and nanoencapsulated Eucalyptus citriodora essential oils on sheep gastrointestinal nematodes and toxicity for mice. Vet Parasitol. 2014;204(3–4):243–8. doi:10.1016/j.vetpar.2014.05.026. [Google Scholar] [PubMed] [CrossRef]
59. Fubini SL, Ducharme N. Farm animal surgery. 2nd ed. Amsterdam, The Netherlands: Elsevier; 2016. 667 p. [Google Scholar]
60. Perry RN, Clarke AJ. Hatching mechanisms of nematodes. Parasitology. 1981;83(2):435–49. doi:10.1017/s0031182000085413. [Google Scholar] [CrossRef]
61. Curtis RHC, Jones JT, Davies KG, Sharon E, Spiegel Y. Plant nematode surfaces. in: Biological control of plant-parasitic nematodes: building coherence between microbial ecology and molecular mechanisms. Dordrecht, The Netherlands: Springer; 2011. p. 115–44. doi:10.1007/978-1-4020-9648-8_5. [Google Scholar] [CrossRef]
62. Chen Q, Peng D. Nematode chitin and application. In: Targeting chitin-containing organisms. Singapore: Springer; 2019. p. 209–19. doi:10.1007/978-981-13-7318-3_10. [Google Scholar] [CrossRef]
63. Riou M, Guégnard F, Le Vern Y, Grasseau I, Koch C, Blesbois E, et al. Effects of cholesterol content on activity of P-glycoproteins and membrane physical state, and consequences for anthelmintic resistance in the nematode Haemonchus contortus. Parasite. 2020;27(3):3. doi:10.1051/parasite/2019079. [Google Scholar] [PubMed] [CrossRef]
Cite This Article
 Copyright © 2026 The Author(s). Published by Tech Science Press.
Copyright © 2026 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools