 Open Access
Open Access
ARTICLE
A Novel Krill Herd Based Random Forest Algorithm for Monitoring Patient Health
1 Department of Health Information Management and Technology, University of Hafr Al-Batin, Saudi Arabia
2 Department of Electrical and Computer Engineering, Faculty of Engineering, King Abdulaziz, Jeddah, 21589, Saudi Arabia
3 Center of Excellence in Intelligent Engineering Systems, King Abdul Aziz University, Jeddah, Saudi Arabia
4 College of Medicine, King Khalid University, Abha, Saudi Arabia
5 Department of Physics, College of Science, Jazan University, P.O. Box. 114, Jazan, 45142, Saudi Arabia
6 Department of Biology, College of Science, Imam Mohammad Ibn Saud Islamic University (IMSIU), Riyadh, 11623, Saudi Arabia
7 Computer Science Department, Faculty of Computing and Information Technology, King Abdulaziz University, Jeddah, 21589, Saudi Arabia
8 Department of Math and Computer Science, Augustana College, IL, 61201, United States
* Corresponding Author: Tauheed Khan Mohd. Email:
Computers, Materials & Continua 2023, 75(2), 4553-4571. https://doi.org/10.32604/cmc.2023.032118
Received 07 May 2022; Accepted 19 November 2022; Issue published 31 March 2023
Abstract
Artificial Intelligence (AI) is finding increasing application in healthcare monitoring. Machine learning systems are utilized for monitoring patient health through the use of IoT sensor, which keep track of the physiological state by way of various health data. Thus, early detection of any disease or derangement can aid doctors in saving patients’ lives. However, there are some challenges associated with predicting health status using the common algorithms, such as time requirements, chances of errors, and improper classification. We propose an Artificial Krill Herd based on the Random Forest (AKHRF) technique for monitoring patients’ health and eliciting an optimal prescription based on their health status. To begin with, various patient datasets were collected and trained into the system using IoT sensors. As a result, the framework developed includes four processes: preprocessing, feature extraction, classification, and result visibility. Additionally, preprocessing removes errors, noise, and missing values from the dataset, whereas feature extraction extracts the relevant information. Then, in the classification layer, we updated the fitness function of the krill herd to classify the patient’s health status and also generate a prescription. We found that the results from the proposed framework are comparable to the results from other state-of-the-art techniques in terms of sensitivity, specificity, Area under the Curve (AUC), accuracy, precision, recall, and F-measure.Keywords
Increasingly, in recent times, entrepreneurs entering the healthcare space have combined traditional and new skills in the solutions they bring to the market [1]. One of the technologies to find the greatest application is artificial intelligence, which has been utilized in clinical, financial, and operational solutions in the healthcare sector [2]. AI is generally defined as a computer system that performs required tasks, such as emulating human intelligence [3]. Moreover, the healthcare industry is undergoing a transformation manifested as rising overall healthcare costs and a growing shortage of healthcare professionals [4,5]. The global healthcare system is beset with massive challenges, including high costs, limited access, an aging population, and a dearth of protective equipment [6]. In recent times, healthcare facilities have undertaken to expand their monitoring and record-management capabilities [7]. As a result, the healthcare system is faced with the challenge of procuring means of securing information and other network communication technology capabilities [8] Utilization of this huge cache of extant data to aid decision making has tremendous scope in this evolving ecosystem. Machine Learning (ML) is one of the AI frameworks that enables an AI system to learn from its surroundings and make intelligent decisions regularly [9]. Fig. 1 illustrates the role of AI in healthcare.
Figure 1: Role of AI in healthcare
Additionally, machine learning is widely used in a variety of industries, including agriculture, finance, media, healthcare, and retail [10,11]. Manual disease prediction using AI-based ML approaches is accurate, early, simple, and timely [12]. Since they collect real-time data via Internet of Things (IoT) sensors, they also reduce the time required to gather patient data from multiple sources. Data from the IoT sensor is collected and shared with other physical devices [13]. Medical practitioners can enhance their computer skills and provide more accurate diagnoses by utilizing ML methods [14]. The use of ML in conjunction with IoT cloud computing concepts improves the accuracy of the performance of the healthcare technique [15]. Fog computing, IoT, machine learning, and cloud computing are all emerging technologies that are attracting the most attention from researchers [16]. Humans are increasingly exposed to newer risk factors, particularly those related to lifestyle and environment, which predispose them to a variety of physiologic disorders at a young age [17]. Early signs of such derangement can be detected by an effective health monitoring system. This can help with better and timely treatment, reducing treatment costs as well as enhancing patient outcomes [18]. Monitoring of health status protects the interests of patients by ensuring that healthcare provider decision-making is more informed by data and hence accurate. Healthcare monitoring systems have been useful in detecting serious illnesses at an early stage, thus playing an instrumental part in providing timely life-saving intervention to patients. Thus, early disease detection is of critical importance, but it is time-consuming owing to the mass of data required [19].
The primary drawbacks of AI-based health monitoring systems are their high time consumption, high cost, and difficulties with disease diagnosis [20]. Numerous techniques, such as the healthcare model, AI, and safety control model, have been developed to improve the performance of AI-based healthcare monitoring systems, but they all suffer from low sensitivity and specificity. The main aim of the design and development of this AI-based healthcare monitoring system is to minimize the cost of healthcare by reducing hospitalizations, and physician office visits as well as minimizing diagnostic testing procedures. This health monitoring system is also supposed to have better sensitivity and accuracy. Hence, this system will help in saving lives by early detection and treatment of potentially dangerous health conditions.
The main components of this research work are summarized as follows:
• Initially, the chest Computed Tomography (CT) scan image dataset is collected using an IoT sensor and trained into the system.
• Thereafter, an AKHRF framework is designed for monitoring and classifying the health status of a patient.
• Consequently, pre-processing and feature extraction are performed to remove the errors, noise, and extract the relevant information from the dataset.
• Thereafter, an update of the fitness of the krill herd in the classification layer for classifying the health status of patient data is undertaken.
• Finally, the validated metrics are compared with other models in terms of accuracy, sensitivity, precision, specificity, recall, F-measure, and AUC.
The following is the order of the parts of this paper: Section 2 discusses the paper’s literature review. Section 3 elaborates on the analyzed problem and system model. In Section 4, the novel technique has been explained. Section 5 will discuss the obtained results and comparative analysis. Section 6 comprises of the conclusion part.
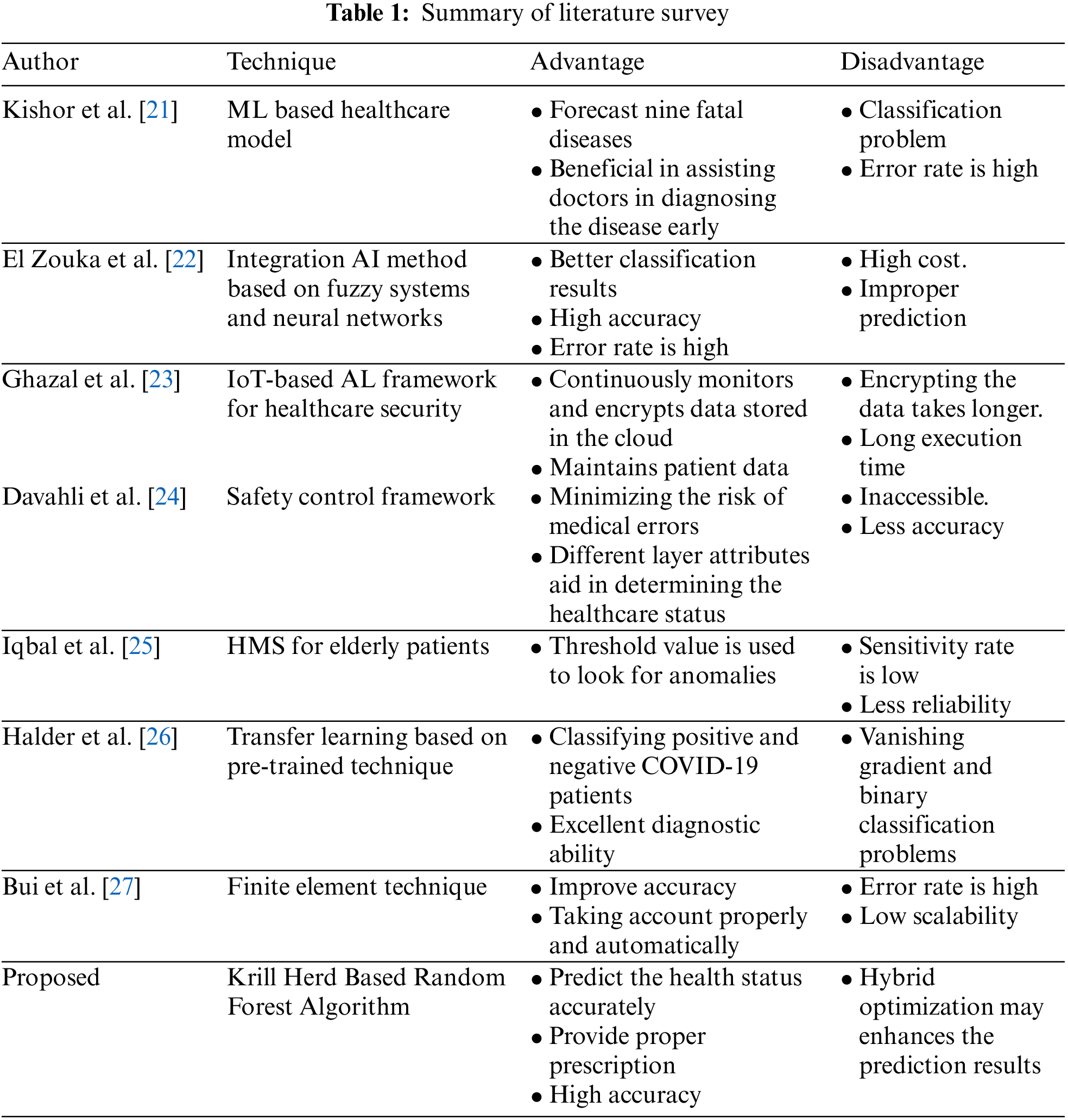
Kishor et al. [21] developed a ML-based healthcare model capable of accurately predicting the early and late outcomes of a variety of diseases. Additionally, this approach employs seven ML classification algorithms to forecast nine fatal diseases. The developed technique performs better, which is beneficial in assisting doctors in diagnosing the disease early. But this system has some problems on the classification front.
Healthcare Monitoring Systems (HMS) is gaining popularity in hospitals and other settings. Thus, El Zouka et al. [22] proposed an integration AI method based on fuzzy systems and neural networks for securing the HMS. Additionally, HMS enables the implementation of a system for intelligent healthcare techniques that prioritizes care based on collected health parameters, but at a high cost.
Ghazal [23] proposed an IoT-based AL framework for healthcare security, and the Wireless Sensor Network (WSN) which is built using IoT technology to connect the digital and physical worlds. Thus, the designed framework continuously monitors and encrypts data stored in the cloud and maintains patient data, but encrypting the data takes longer.
Since 2016, AI based systems have achieved significant successes in healthcare in medical tasks or human performance levels. Davahli et al. [24] developed a safety control framework for minimizing the risk of medical errors. Additionally, different layer attributes aid in determining the healthcare status, but they are inaccessible.
Iqbal et al. [25] created an HMS for elderly patients that utilize an intelligent model to monitor their health status. A wearable sensor is used to monitor heart rate, body temperature, and blood glucose levels. A threshold range is used to detect anomalies. A major drawback of this system is that the sensitivity is low when compared to other methods.
A handful of structural and genetic analyses have identified that surface protein sorts contagious viruses which multiply quickly. Halder et al. [26] proposed a transfer learning-based pre-trained technique for classifying positive and negative COVID-19 patients. Moreover, original and manipulated datasets are tested and analysed with excellent diagnostic ability. However, these systems have vanishing gradient and binary classification problems.
Bui et al. [27] developed a finite element technique for offering background mesh that is executed in real-time surgical simulation. Furthermore, boundary conditions were indirectly imposed on the surface with Lagrange multipliers. The designed model is functional in numerous needle enclosure simulations and attained outcomes improve accuracy through taking account properly and automatically but the error rate is high.
A summary of the literature survey is detailed in Table 1.
3 System Model and Problem Definition
The proposed healthcare system is capable of making decisions about the detected conditions of all patients based on their pulse rate, heart rate, and body temperature. Additionally, the healthcare system is more energy efficient because all the sensors are not activated at all times. It is comprised of an IoT server, embedded sensors, communication channels, a gateway, and cloud storage. Three sensors are connected to an Arduino board to classify and collect patient data in the system model. Additionally, fuzzy neural networks enhance a patient’s collected dataset, but they have some drawbacks, including an error rate, attack vectors, unauthorized access, decreased sensitivity, and decreased specificity. Fig. 2 illustrates the fundamental system model and problem statement.
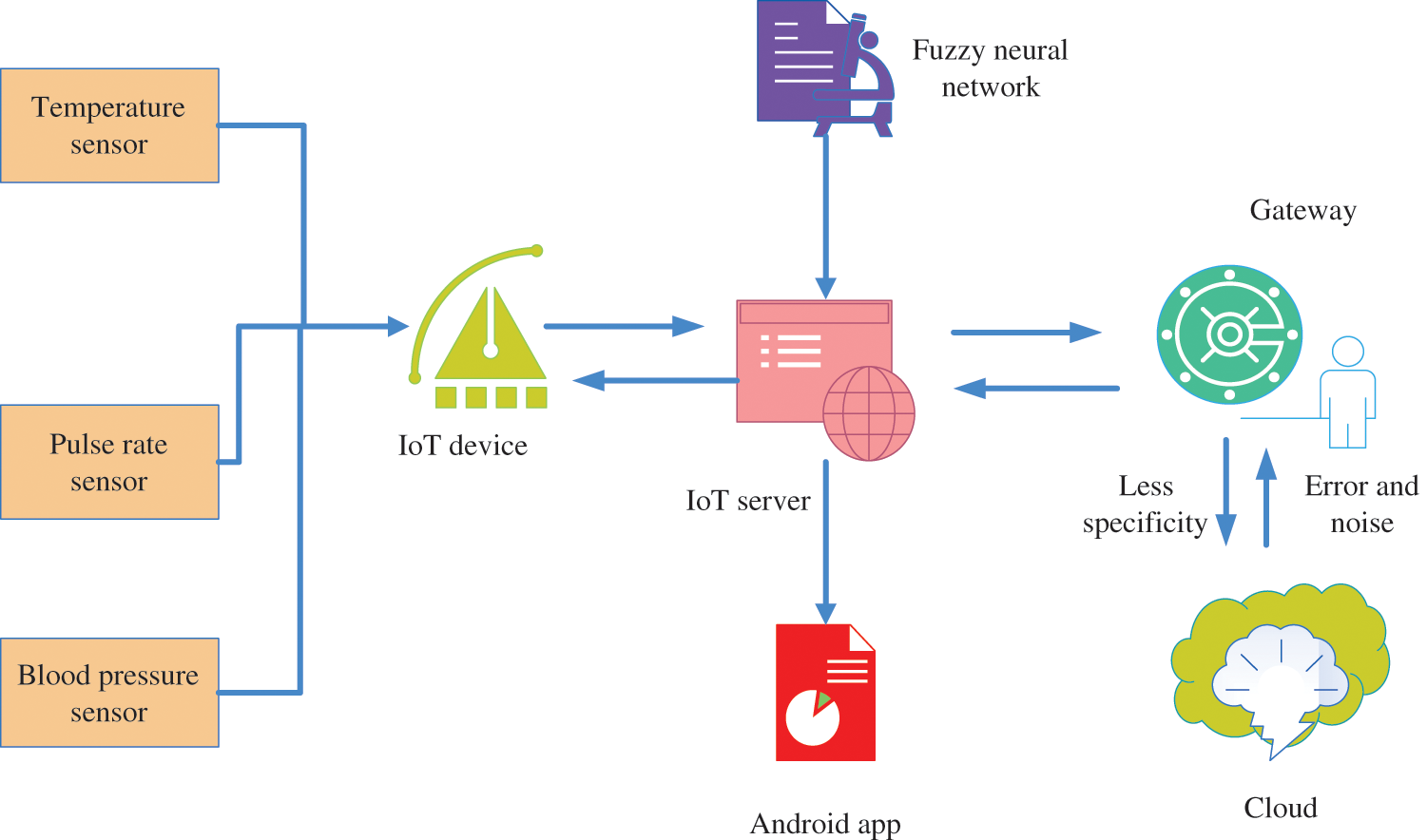
Figure 2: System model and problem definition
The main critical tasks for monitoring the healthcare system are high costs, lack of access, the increasing elderly population requiring greater care and monitoring, and lack of healthcare staff and protective equipment. Due to the vast amount of data, misclassification and detection problems are common. Also, monitoring the health status of a patient takes more time than usual. So, a novel machine learning framework is designed to enhance the performance of healthcare monitoring systems.
The main aim of the design and development of the healthcare monitoring system is to minimize the cost of healthcare by reducing hospitalizations, diagnostic testing procedures, and physician office visits. Initially, various patient datasets are collected using IoT sensors and trained on the system. The data is stored in the cloud. Thus, the developed framework includes four processes such as preprocessing, feature extraction, classification, and visibility of results. Moreover, preprocessing checked and filtered the input data to remove errors, noise, and missing values. Next, feature extraction extracts the relevant features from the dataset, like blood sugar level, heart rate, weight scale, and so on. Then, update the fitness function of the Krill herd in the classification layer, which monitors the status of the patient data with the help of extracted features and then classifies the health status of the patient and also provides a proper prescription based on the health status. The proposed architecture has been detailed in Fig. 3.
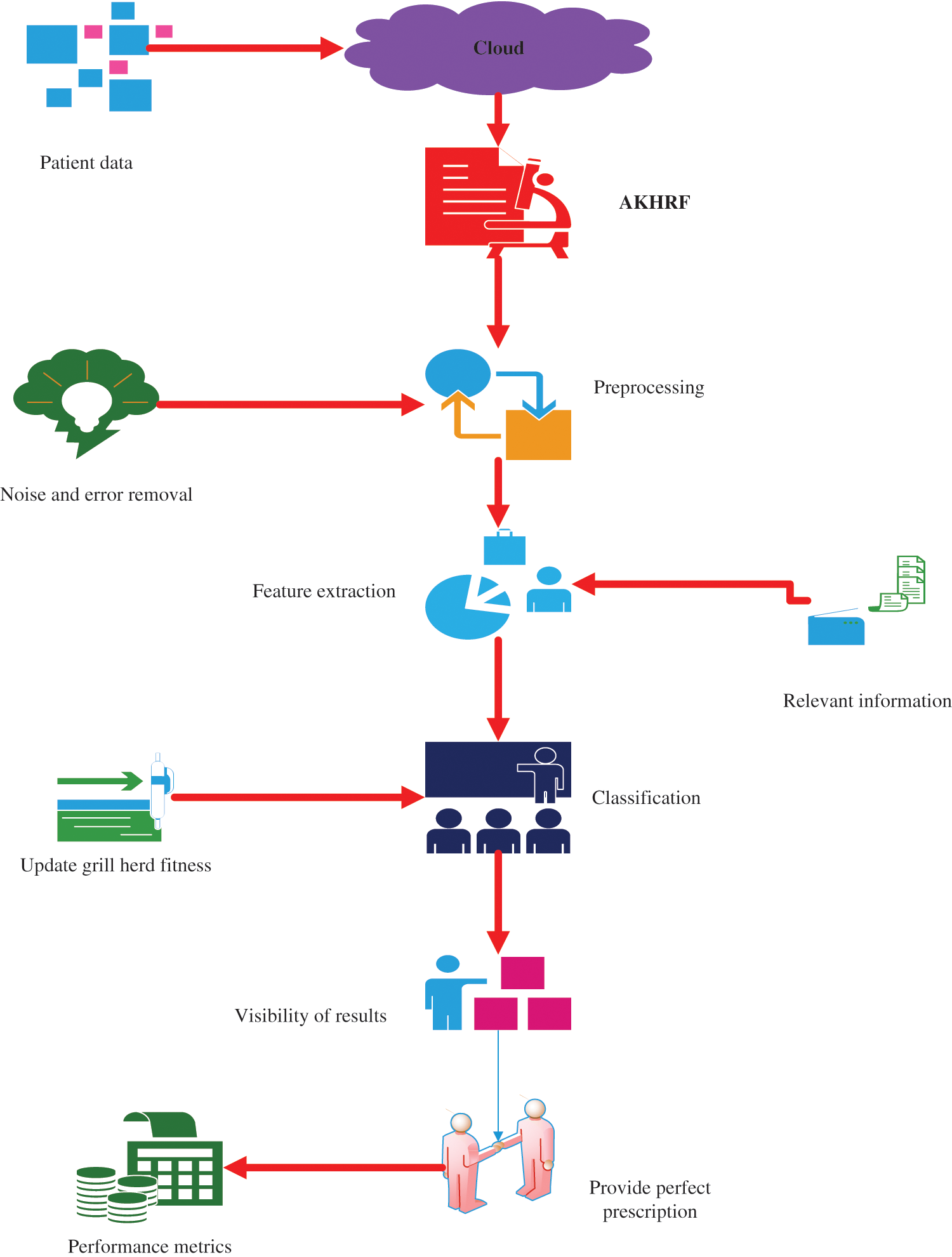
Figure 3: Proposed architecture
First, various patient datasets are collected from different sources like clinics, laboratories, remote sites, and home data. IoT devices and dissimilar sensors are used for collecting different patient datasets in real-time. The IoT device collects the patient health dataset using wearable physiological sensors like temperature sensors, pulse oximeter sensors, heart rate sensors, pressure sensors, and accelerometers. Further, the datasets collected from different patients are stored in the cloud. The novelty of the designed model is in updating of the krill herd fitness in the classification layer for monitoring the health status of the patient by the movement of krill behaviour. It accurately detects and classifies the health status of the patient with high accuracy and incurs lesser execution time. Moreover, feature extraction is useful to enhance the performance of detection, and based on the health status doctors provide a proper description. The timely interventions saves lives and increases the life span of patients.
The developed AKHRF algorithm is performed on the chest CT scan image dataset, which is collected from the website of Kaggle. The dataset can also be found at https://www.kaggle.com/mohamedhanyyy/chest-ctscan-images. Additionally, the dataset is in the format of the image, so one can easily extract the aspect terms. The total dataset used for the developed framework is 916. From that, training used 733 datasets, and testing used 183 datasets. More than 70% of the data is used for training, and 30% of the data is used for training. Thus, the developed framework is trained and tested using krill herd optimization over 200 iterations.
The developed AKHRF framework enhances the classification results of the healthcare monitoring system and attains high accuracy for classifying the health status of a different patient based on the extracted features. Initially, various patient datasets are imported into the input layer. Then classify the health status of each patient based on their blood sugar level, heart rate level, water level, and so on. At that time the health status is visible in the output layer, also doctors provides a proper prescription to the patients. In the classification phase, update the fitness functions of the krill herd because it includes three actions, such as induced movements of other krill, foraging activity, and random diffusion. As a result, the developed framework continuously monitors behavior and changes in the human body, as well as categorizes the health status of all patients.
The various patient datasets were collected, checked and filtered to remove training flaws, errors, and missing values. Moreover, preprocessing helps to enhance the quality of the dataset and filter the dataset using the designed technique. Furthermore, preprocessing is processed using Eq. (1).
Let,
Then feature extraction is utilized to extract the relevant features of various patient datasets for easy classification of health status. Furthermore, extracted features are coordinated and filtered by summation value, and the features are extracted based on time, heart rate, blood glucose level, body temperature, body temperature, activity, blood pressure, and weight scale. The mathematical calculation for extracting relevant information is obtained by Eq. (2).
Let,
In the classification layer, update the fitness function of the krill herd to continuously monitor the health status of all patients. Thus, the fitness of the krill herd monitors the patient data using the movement of krill behaviour. Then the patient’s health status is visible in the output layer. The developed framework attains better performance for predicting the various health statuses of different patients. The classification is performed using Eq. (3):
Let,

Then the visibility of the patient health status is identified using Eq. (4).
Based on the variation of feature extraction, identify, and classify the health status of the patient, and the visibility of patient health is predicted in the output layer. The developed framework attains better performance for monitoring patient health care and provides better outcomes for classifying and providing a proper prescription for the patient.
The developed AKHRF replica is processed using MATLAB. The success rate of the projected model is assessed by current existing mechanisms in terms of accuracy, precision, F-measure, execution time, AUC, and recall. In this approach, various patient datasets are utilized for classifying health status. Here, the proposed AKHRF approach classifies patient diseases based on the extracted health status. Hence, the developed model attained high performance in classification and provided a proper prescription for the patient. Moreover, the gained accuracy and loss of over 200 iterations of the developed technique are shown in Fig. 4.
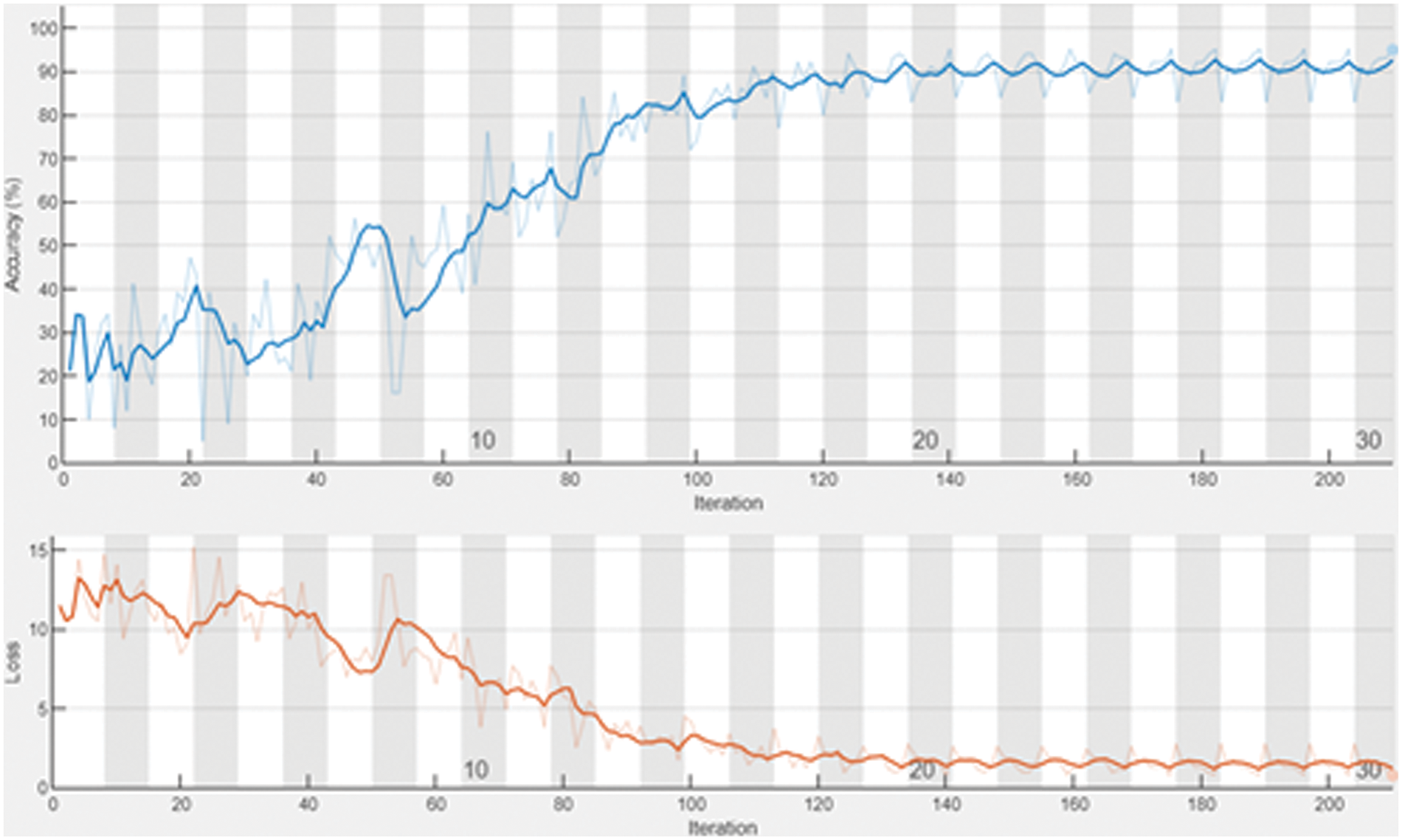
Figure 4: Accuracy vs. Loss
The gained outcome of a developed AKHRF technique is compared with other existing technique metrics. The key metrics of the proposed approach are accuracy, precision, recall, F-measure, sensitivity, specificity, and AUC. Furthermore, the proposed healthcare method efficiency is compared with various conventional methods such as the HMS [21], Intelligent Task Mapping Framework in Healthcare (ITMH) [25], Smart Homes Health Monitoring (SHHM) Method [28], and Intelligent Healthcare Framework (IHF) [29].
Accuracy is defined as the degree of calculation of the efficiency of the proposed model functioning. Also, it is the fraction of properly expected observance of the entire observations that are expressed in Eq. (5):
where,
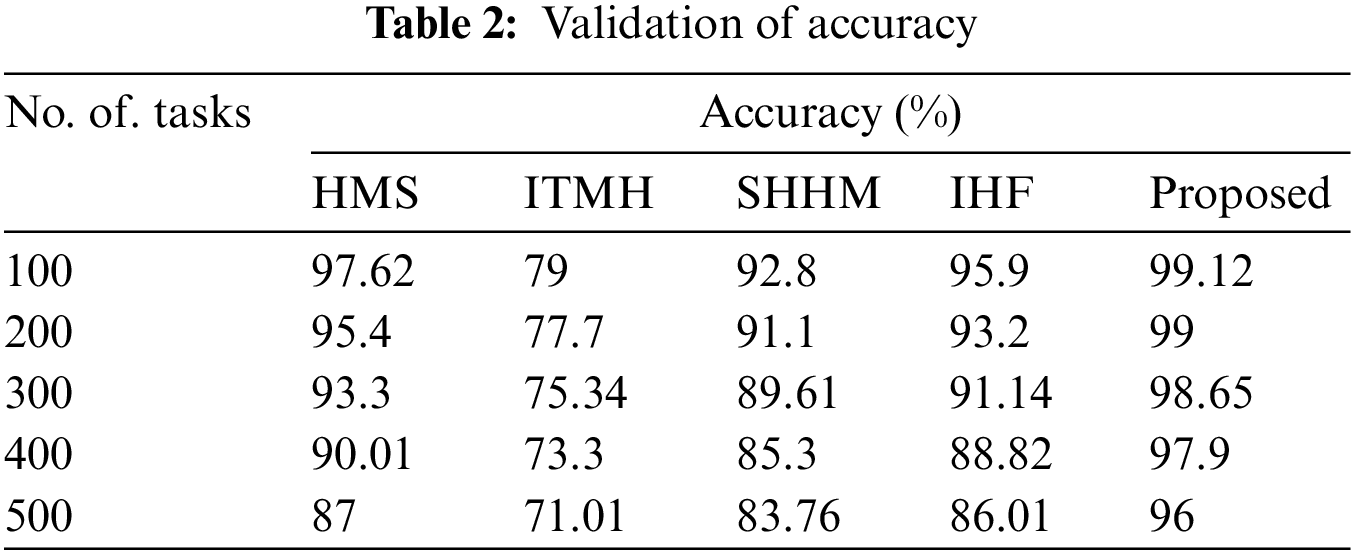
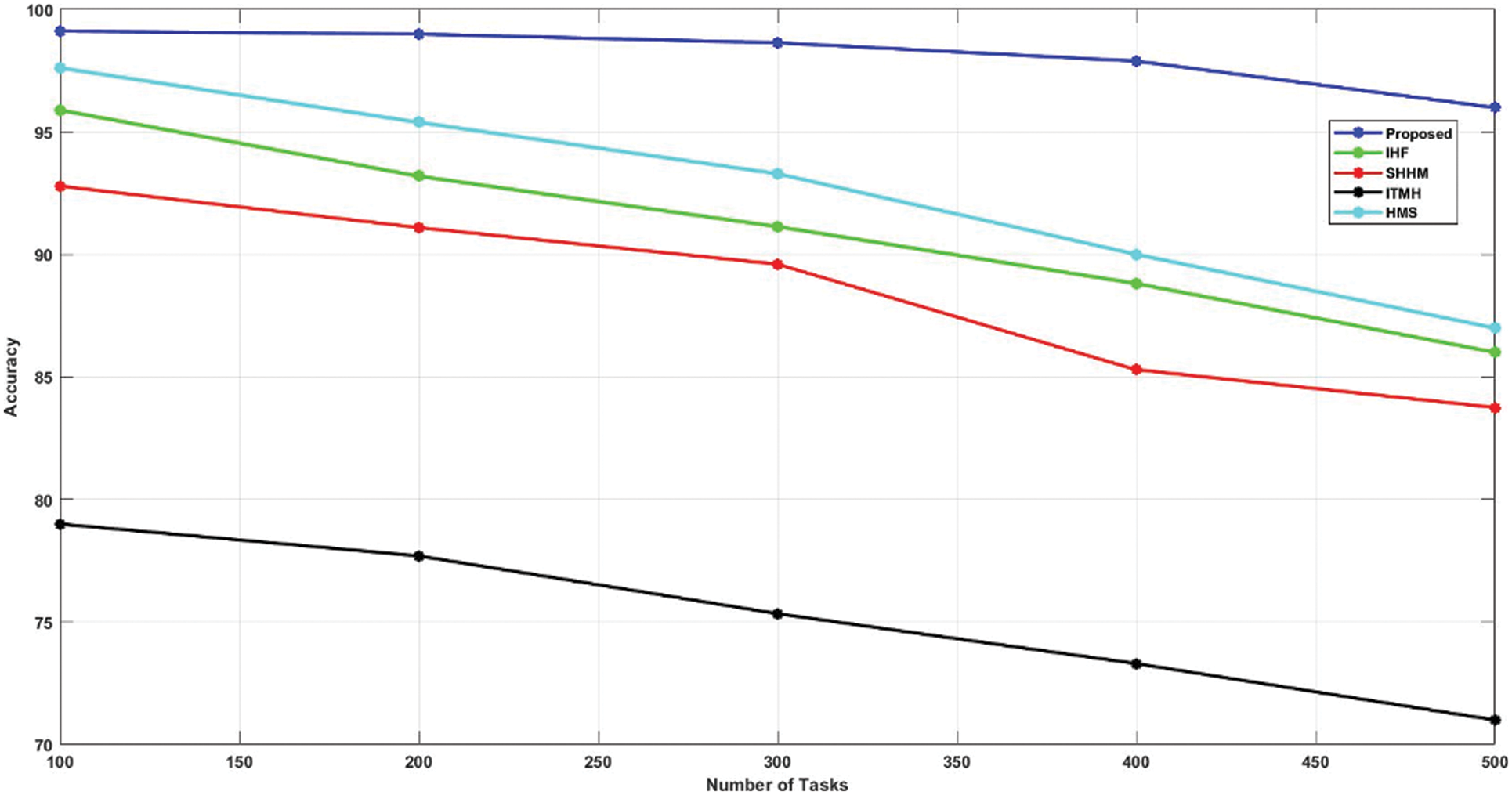
Figure 5: Comparison of accuracy
The achieved accuracy rate of the developed technique is compared with other existing techniques such as HMS, ITMH, SHHM, and IHF. Moreover, the HMS replica attained 97.62% and the ITMH technique gained 79%. Also, SHHM and IHF techniques gained accuracy rates of 92.8% and 95.9%, respectively. The developed AKHRF attained high accuracy while comparing other techniques for classifying patient health status, at 99.12%.
This process was evaluated for identifying the number of correct positive estimates that are aligned with the overall positive estimates. Also, precision is the proportion of classifying patient health status based on the extracted features, which is computed using Eq. (6):
The precision of the proposed AKHRF model is calculated and validated using prevailing methods like HMS, ITMH, SHHM, and IHF approaches which are elaborated in Table 3.
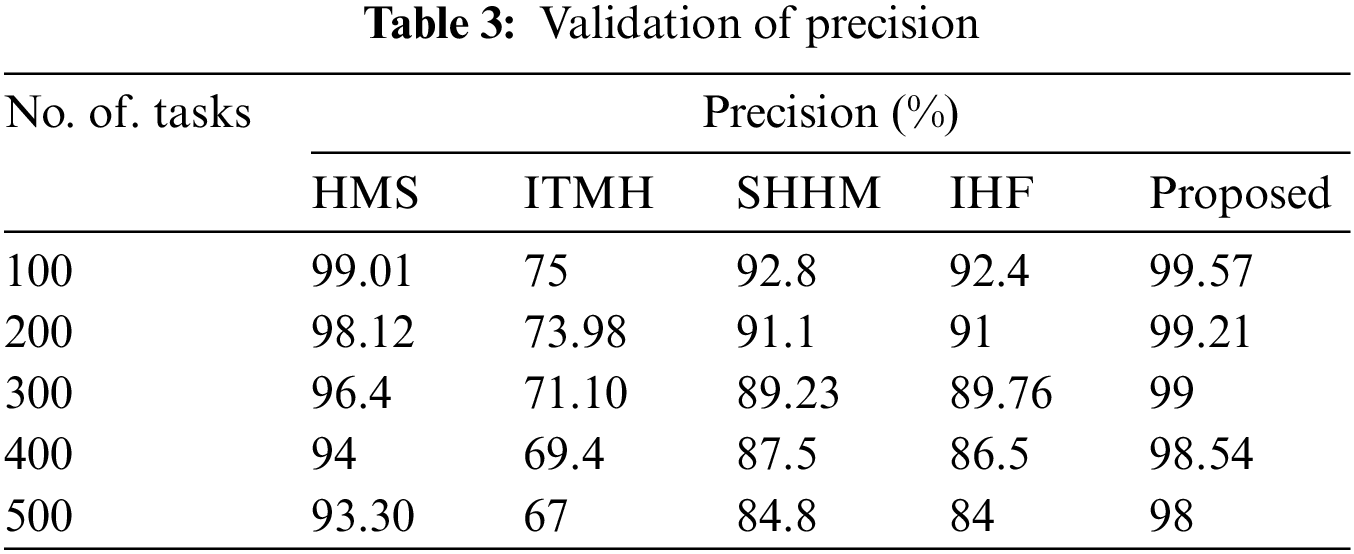
Initially, the HMS technique gained a 99.01% precision rate, and the ITMH replica achieved 75% precision for using 100 tasks. Moreover, the SHHM and IHF techniques gained 92.8% and 92.4% in precision rates, respectively. Finally, the developed AKHRF technique gained a 99.57% precision rate. Thus, the comparison of the precision rate is detailed in Fig. 6.
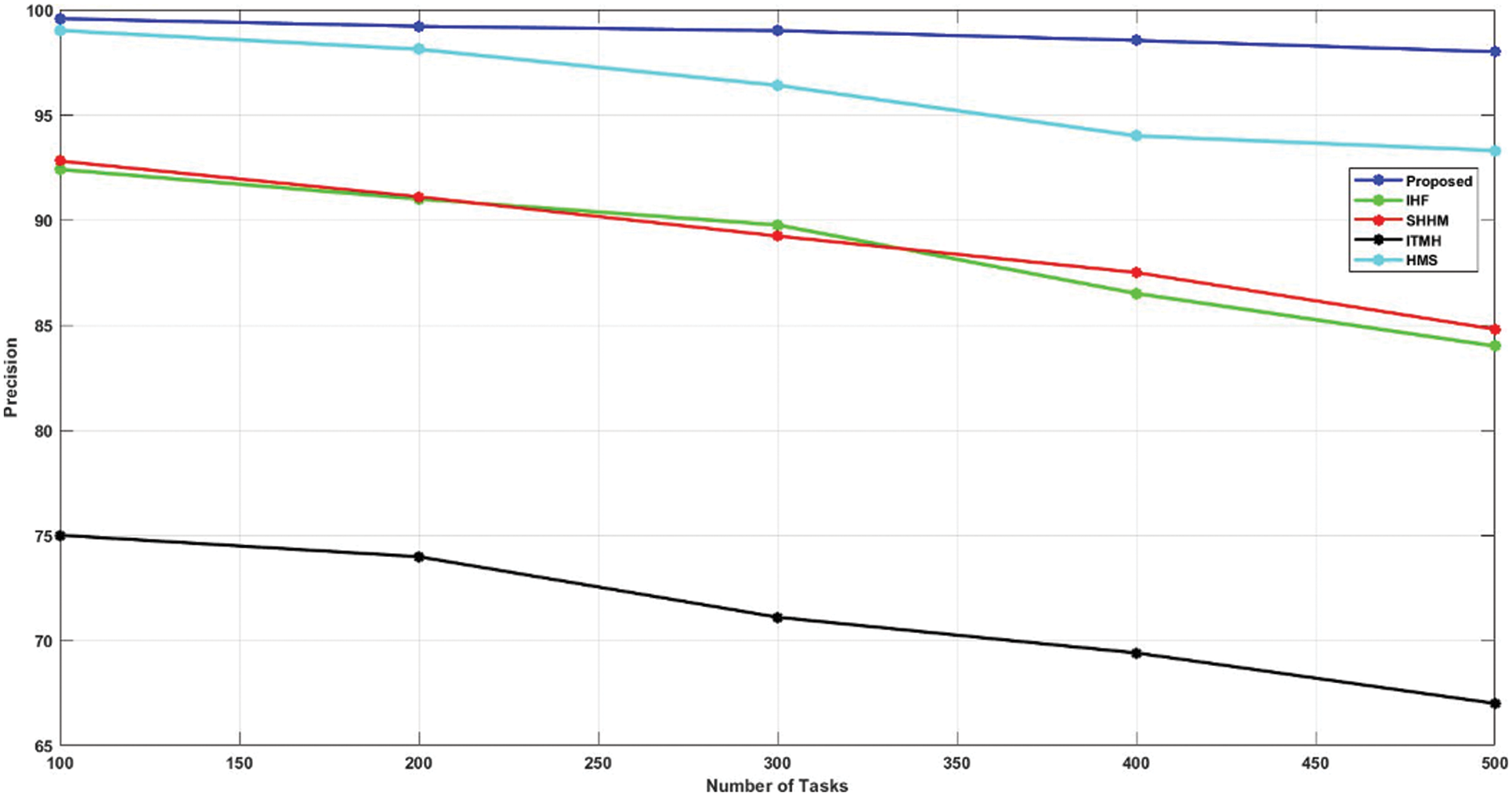
Figure 6: Comparison of precision
The recall is used to calculate the total number of correct positive about the total number of true positives and false negatives. It expresses the proportion of predictions that have correctly diagnosed the health status of the patient and is calculated using Eq. (7).
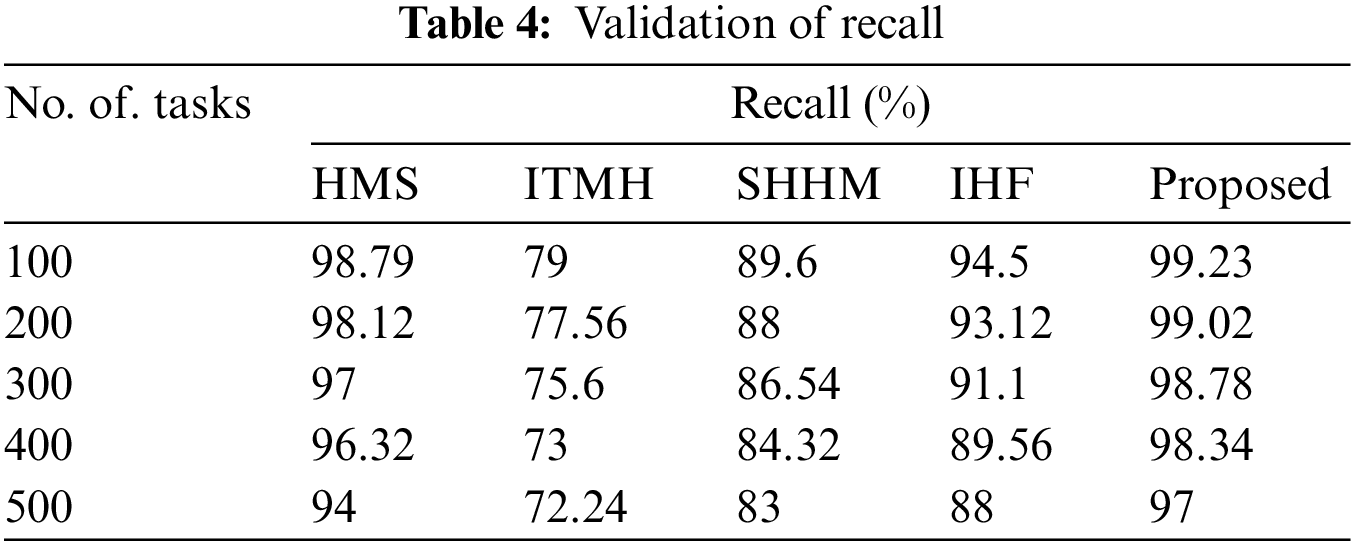
The calculation of the recall by AKHRF technique is compared using prevailing methods like HMS, ITMH, SHHM, and IHF approaches are described in Table 4.
Also, the existing approaches attained a lower recall of almost 98% only. Subsequently, the recall value of the AKHRF approach is higher than other methods. It has achieved a 99.23% recall value, and the comparison of recalls is represented in Fig. 7.
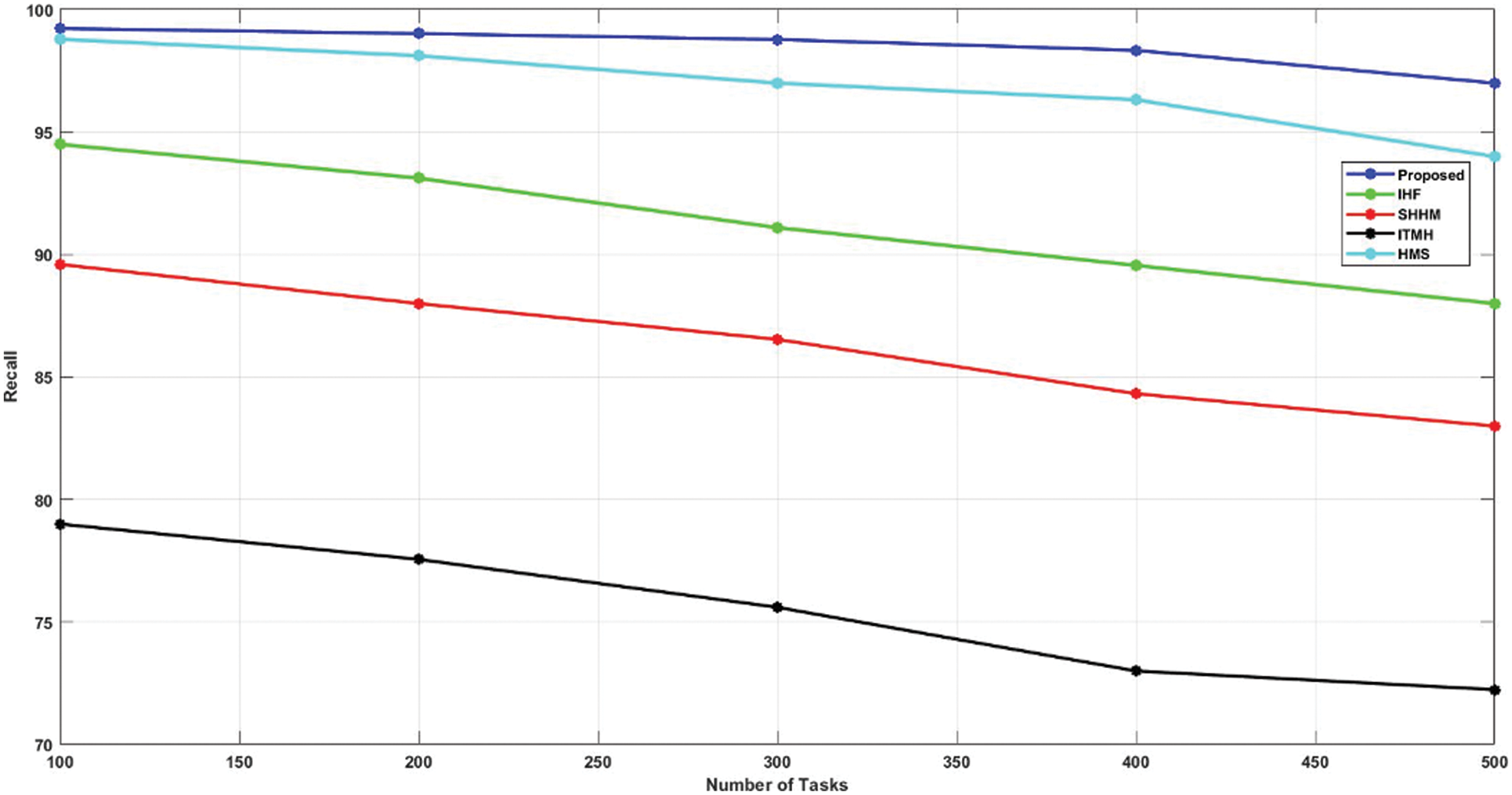
Figure 7: Comparison of recall
The calculation is based on the precision and recall measurements for recognizing the efficiency of classifying the health status of the patient, which is measured using Eq. (8),
where, P denotes the calculated precision value and R represents the calculated recall value.
The F1-score of the proposed AKHRF model is calculated and validated using prevailing methods like HMS, ITMH, SHHM, and IHF approaches. In this case, the existing methods had a lower F1 score, and the proposed model had a higher F1-score value of 98.54% than the other methods, as shown in Fig. 8 and Table 5.
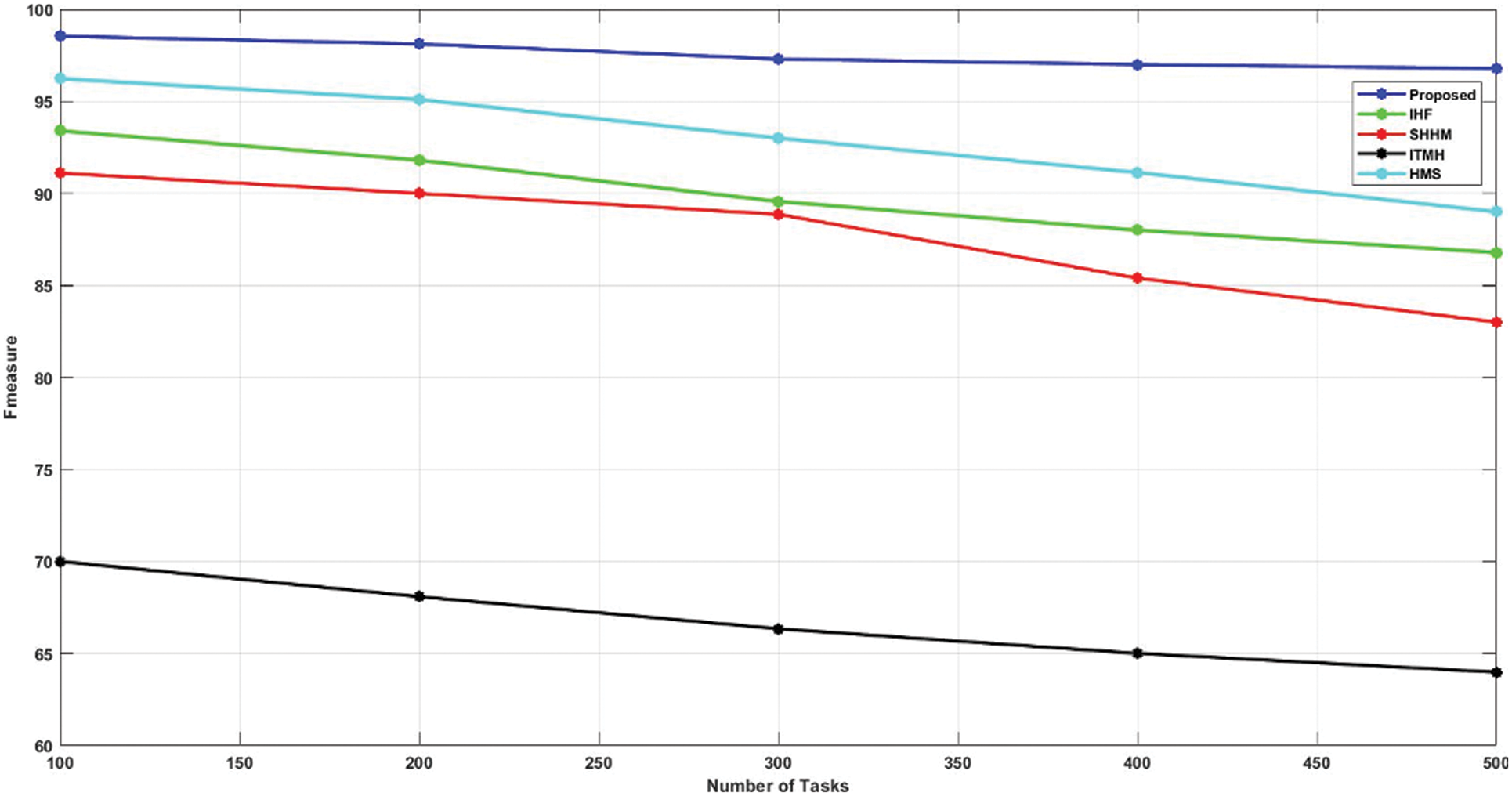
Figure 8: Comparison of F-measure
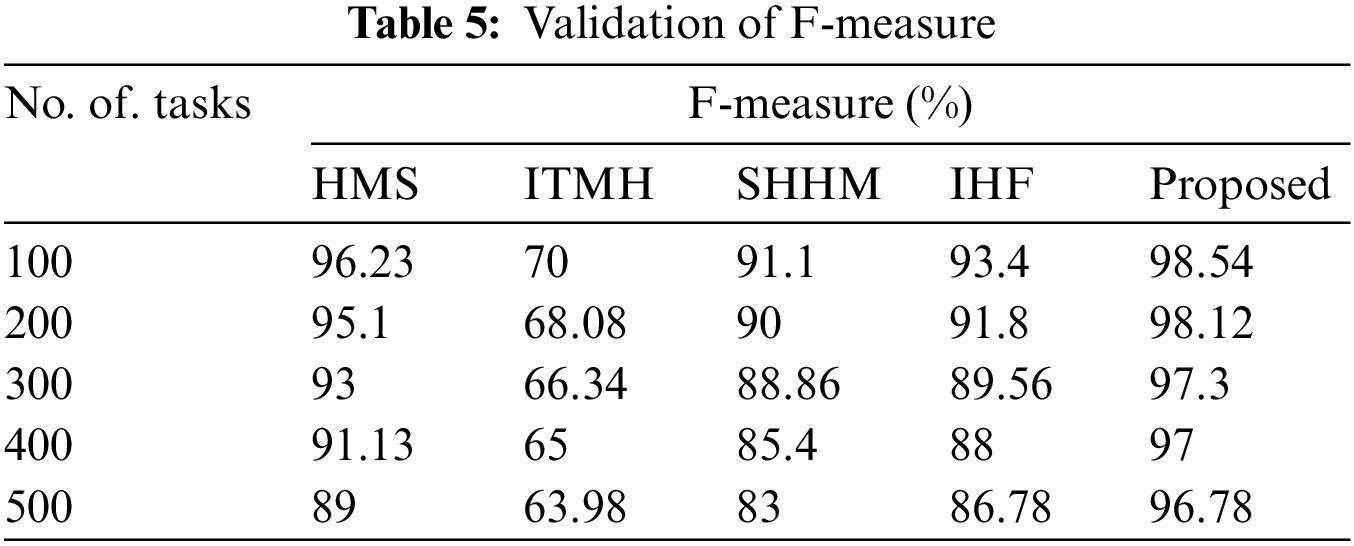
The ratio of
where,
Moreover, the required execution times for HMS and ITMH are 3.5 and 24 s, respectively. Also, SHHM and IHF required 7.02 and 12 s. Furthermore, the IHF technique attained a time of 12 s in execution, and the developed framework attains a time of less than 1.2 s. The comparison of the execution times is shown in Fig. 9.
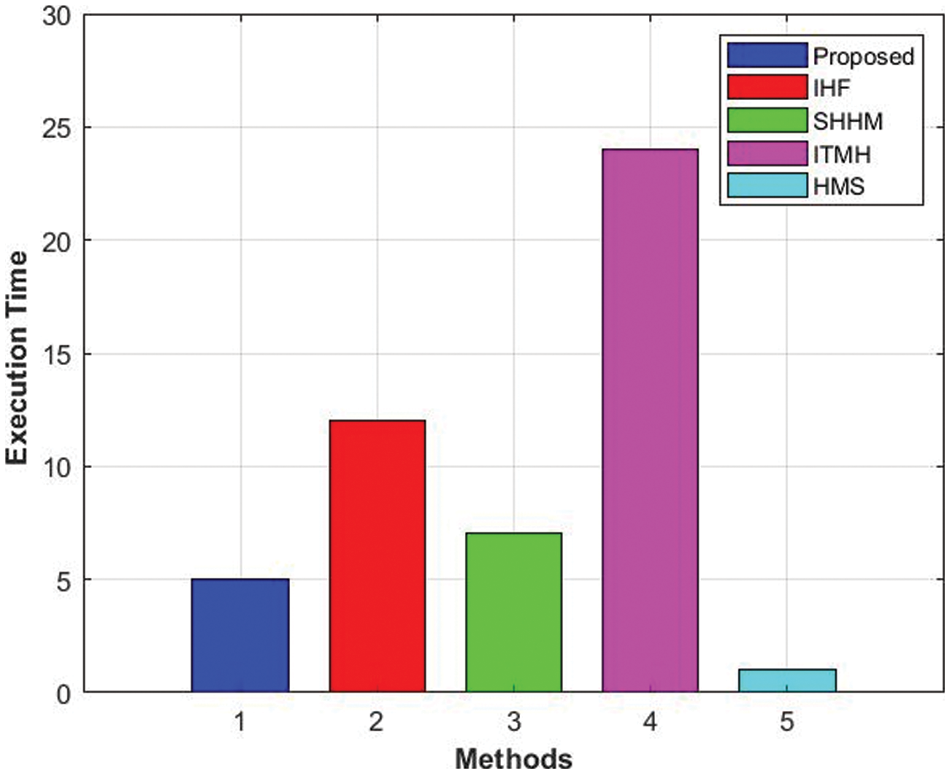
Figure 9: Comparison of F-measure
It is defined as the measure of the capability of the classifier for distinguishing among classes and is also helpful for the summary of the Receiver Operating Characteristic (ROC) curve. The performance of the AUC is measured by the positive and negative segmentation of the affected parts in the skin. Furthermore, AUC denotes the measure or degree of separability, and the high rate of AUC represents the patient with no disease and disease. Furthermore, ROC indicates the performance of classification-based thresholds using a graph. The graphical representation of ROC and AUC is shown in Fig. 10.
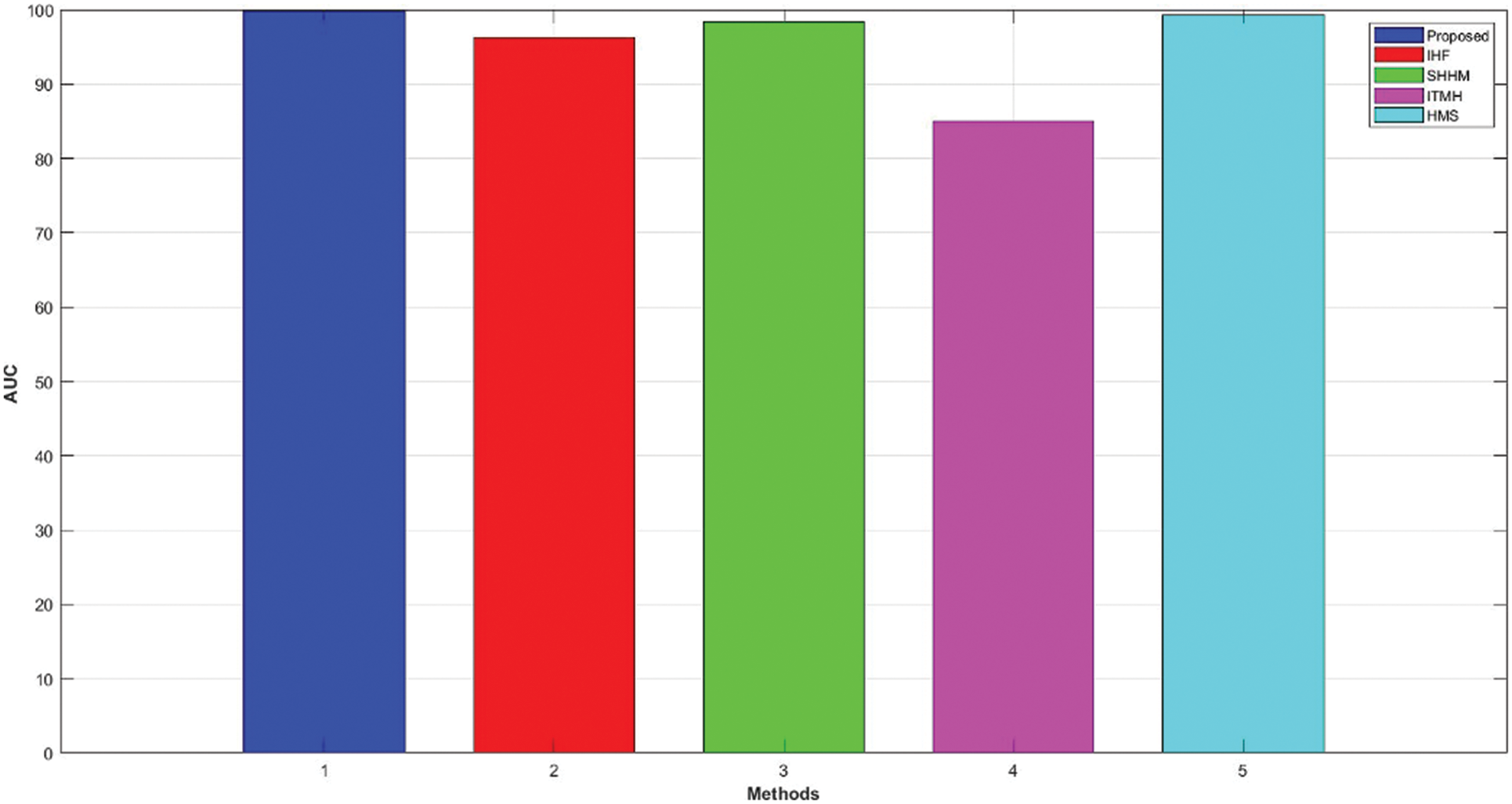
Figure 10: Comparison of AUC
The AUC for the HMS and ITMH was 99.32% and 85%, respectively. Frequently, SHHM and IHF attained 98.4 and 96.3. Moreover, the developed AKHRF technique attained 99.77% in AUC while compared to other existing techniques, the developed framework attained better performance.
The proposed model of AKHRF has shown good performance by attaining the best results in accuracy, sensitivity, specificity, F-measure, precision, AUC, and recall. Thus, the developed scheme removed the training flaws in the initial stage. Next, it was able to extract the features based on blood sugar level, heart rate, weight scale, and so on. It was hence able to classify the health status of the patient as having cancer. Based on the classified results, they are visible in the output, which helps the doctor to provide an appropriate prescription to the patients. Thus, the developed AKHRF technique enhances the prediction performance.
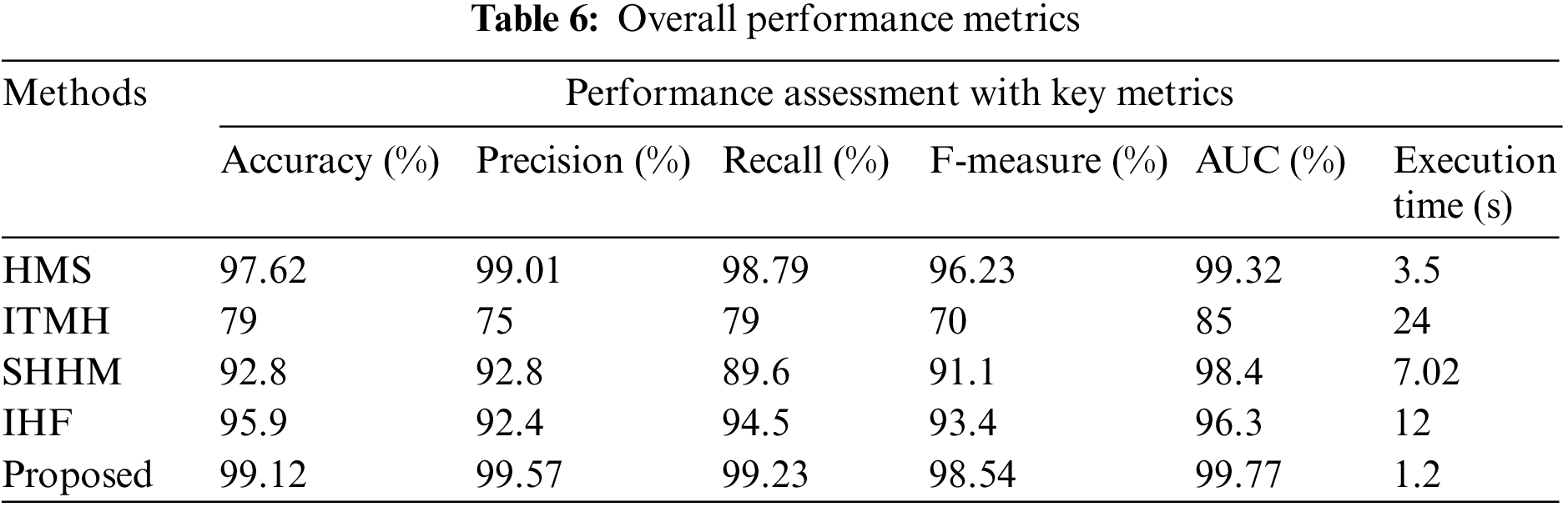
The outstanding metrics comparisons are tabulated in Table 6. In all parameter validation, the proposed AKHRF has gained the finest results. Moreover, the developed framework gained high accuracy of 99.12%, high precision of 99.57%, high recall of 99.23%, high AUC of 99.77%, and an execution time of 1.2 s. Hence, the robustness of the proposed AKHRF has been verified, and it can classify the health status of the patient and provide a proper prescription for the patient. Here, the proposed technique has achieved high performance for classifying and visualizing the health status.
This work develops a novel AKHRF approach for classifying the health status of patients based on their health data. Patient datasets are collected here using an IoT sensor that has been prior tested and trained in the system. The proposed AKHRF model entails preprocessing, feature extraction, classification, and result visualization. Further, the developed model is applied to patient data and, using the extracted features, classifies the health status of all patients. Also, the output layer displays the patient’s health status which guides a doctor in clinical decision making and prescribing. The proposed model achieved improved accuracy, precision, recall, F-measure, execution time, and AUC. When compared to other models, it showed promising results in classifying health status with an accuracy of 99.12 percent with a shorter execution time of 1.2 s.
Funding Statement: The authors extend their appreciation to the Deanship of Scientific Research at King Khalid University for funding this work through Small Research Groups under grant number (RGP.1/62/43).
Conflicts of Interest:: The authors declare that they have no conflicts of interest to report regarding the present study.
References
1. T. Suryavanshi, S. Lambert, S. Lal, A. Chin and T. M. Chan, “Entrepreneurship and innovation in health sciences education: A scoping review,” Medical Science Educator, vol. 30, no. 4, pp. 1797–1809, 2020. [Google Scholar] [PubMed]
2. A. Sharifi, M. Ahmadi and A. Ala, “The impact of artificial intelligence and digital style on industry and energy post-COVID-19 pandemic,” Environmental Science and Pollution Research, vol. 28, no. 34, pp. 46964–46984, 2021. [Google Scholar] [PubMed]
3. R. Dias and A. Torkamani, “Artificial intelligence in clinical and genomic diagnostics,” Genome Medicine, vol. 11, no. 1, pp. 1–12, 2019. [Google Scholar]
4. M. K. Al-Hanawi, S. A. Khan and H. M. Al-Borie, “Healthcare human resource development in Saudi Arabia: Emerging challenges and opportunities—a critical review,” Public Health Reviews, vol. 40, no. 1, pp. 1–16, 2019. [Google Scholar] [PubMed]
5. S. Hermes, T. Riasanow, E. K. Clemons, M. Böhm and H. Krcmar, “The digital transformation of the healthcare industry: Exploring the rise of emerging platform ecosystems and their influence on the role of patients,” Business Research, vol. 13, no. 3, pp. 1033–1069, 2020. [Google Scholar]
6. L. O. Gostin, E. A. Friedman and S. A. Wetter, “Responding to COVID-19: How to navigate a public health emergency legally and ethically,” Hastings Center Report, vol. 50, no. 2, pp. 8–12, 2020. [Google Scholar] [PubMed]
7. K. T. Kadhim, A. M. Alsahlany, S. M. Wadi and H. T. Kadhum, “An overview of patient’s health status monitoring system based on internet of things (IoT),” Wireless Personal Communications, vol. 114, no. 3, pp. 2235–2262, 2020. [Google Scholar]
8. S. Wan, Z. Gu and Q. Ni, “Cognitive computing and wireless communications on the edge for healthcare service robots,” Computer Communications, vol. 149, no. 1, pp. 99–106, 2020. [Google Scholar]
9. T. M. Ghazal, M. K. Hasan, M. T. Alshurideh, H. M. Alzoubi and M. Ahmad, “IoT for smart cities: Machine learning approaches in smart healthcare—A review,” Future Internet, vol. 13, no. 8, pp. 218, 2021. [Google Scholar]
10. M. Cubric, “Drivers, barriers and social considerations for AI adoption in business and management: A tertiary study,” Technology in Society, vol. 62, no. 1, pp. 101257, 2020. [Google Scholar]
11. H. Bhardwaj, P. Tomar, A. Sakalle and U. Sharma, “Principles and foundations of artificial intelligence and internet of things technology,” in Artificial Intelligence to Solve Pervasive Internet of Things Issues, Academic Press, pp. 377–392, 2021. [Google Scholar]
12. M. D. Gurven and D. E. Lieberman, “WEIRD bodies: Mismatch, medicine and missing diversity,” Evolution and Human Behavior, vol. 41, no. 5, pp. 330–340, 2020. [Google Scholar] [PubMed]
13. S. S. Yadav and S. M. Jadhav, “Deep convolutional neural network based medical image classification for disease diagnosis,” Journal of Big Data, vol. 6, no. 1, pp. 1–18, 2019. [Google Scholar]
14. H. Panwar, P. K. Gupta and M. K. Siddiqui, “Application of deep learning for fast detection of COVID-19 in X-Rays using nCOVnet,” Chaos, Solitons & Fractals, vol. 138, no. 3, pp. 109944, 2020. [Google Scholar]
15. B. Ihnaini, M. A. Khan, T. A. Khan, S. Abbas and M. S. Daoud, “A smart healthcare recommendation system for multidisciplinary diabetes patients with data fusion based on deep ensemble learning,” Computational Intelligence and Neuroscience, vol. 2021, no. 6, pp. 11, Article ID 4243700, 2021. [Google Scholar]
16. A. Stanley and J. Kucera, “Smart healthcare devices and applications, machine learning-based automated diagnostic systems, and real-time medical data analytics in COVID-19 screening, testing, and treatment,” American Journal of Medical Research, vol. 8, no. 2, pp. 105–117, 2021. [Google Scholar]
17. J. G. Richens, C. M. Lee and S. Johri, “Improving the accuracy of medical diagnosis with causal machine learning,” Nature Communications, vol. 11, no. 1, pp. 1–9, 2020. [Google Scholar]
18. X. Sui, S. He, S. B. Vilsen, J. Meng and R. Teodorescu, “A review of non-probabilistic machine learning-based state of health estimation techniques for Lithium-ion battery,” Applied Energy, vol. 300, no. 3, pp. 117346, 2021. [Google Scholar]
19. L. Lu, L. Xu, B. Xu, G. Li and H. Cai, “Fog computing approach for music cognition system based on machine learning algorithm,” IEEE Transactions on Computational Social Systems, vol. 5, no. 4, pp. 1142–1151, 2018. [Google Scholar]
20. R. Vashistha, A. K. Dangi, A. Kumar, D. Chhabra and P. Shukla, “Futuristic biosensors for cardiac health care: An artificial intelligence approach,” 3 Biotech, vol. 8, no. 8, pp. 1–11, 2018. [Google Scholar]
21. A. Kishor and C. Chakraborty, “Artificial intelligence and internet of things-based healthcare 4.0 monitoring system,” Wireless Personal Communications, vol. 127, no. 2, pp. 1615–1631, 2022. [Google Scholar]
22. H. A. El Zouka and M. M. Hosni, “Secure IoT communications for smart healthcare monitoring system,” Internet of Things, vol. 13, no. 3, pp. 100036, 2021. [Google Scholar]
23. T. M. Ghazal, “Internet of things with artificial intelligence for health care security,” Arabian Journal for Science and Engineering, vol. 78, no. 4, pp. 1–12, 2021. [Google Scholar]
24. M. R. Davahli, W. Karwowski, K. Fiok, T. Wan and H. R. Parsaei, “Controlling safety of artificial intelligence-based systems in healthcare,” Symmetry, vol. 13, no. 1, pp. 102, 2021. [Google Scholar]
25. N. Iqbal, S. Ahmad and D. H. Kime, “Health monitoring system for elderly patients using intelligent task mapping mechanism in closed loop healthcare environment,” Symmetry, vol. 13, no. 2, pp. 357, 2021. [Google Scholar]
26. A. Halder and B. Datta, “COVID-19 detection from lung CT-scan images using transfer learning approach,” Machine Learning: Science and Technology, vol. 2, no. 4, pp. 045013, 2021. [Google Scholar]
27. H. P. Bui, S. Tomar and S. P. A. Bordas, “Corotational cut finite element method for real-time surgical simulation: Application to needle insertion simulation,” Computer Methods in Applied Mechanics and Engineering, vol. 345, pp. 183–211, 2019. [Google Scholar]
28. P. Verma and S. K. Sood, “Fog assisted-IoT enabled patient health monitoring in smart homes,” IEEE Internet of Things Journal, vol. 5, no. 3, pp. 1789–1796, 2018. [Google Scholar]
29. V. Vijayakumar, D. Malathi, V. Subramaniyaswamy, P. Sarvanan and R. Logesh, “Fog computing-based intelligent healthcare system for the detection and prevention of mosquito-borne diseases,” Computers in Human Behavior, vol. 100, no. 4, pp. 275–285, 2019. [Google Scholar]
Cite This Article
 Copyright © 2023 The Author(s). Published by Tech Science Press.
Copyright © 2023 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools