Glutamic Acid–Chelated Cobalt Stabilizes G-Quadruplexes and Selectively Suppresses Hepatocellular Carcinoma Growth
Kuan-Hao Lin1, Yu-Ju Lin1, Yu-Bin Hong1, Meng-Huai Hsu1, Zhen-Xiang Liao1, Shuo-Yu Chang1, Chiou-Hwa Yuh1,2,3,4,*
1 Institute of Molecular and Genomic Medicine, National Health Research Institutes, Zhunan, Miaoli, Taiwan
2 Institute of Bioinformatics and Structural Biology, National Tsing Hua University, Hsinchu, Taiwan
3 Department of Biological Science and Technology, National Yang Ming Chiao Tung University, Hsinchu, Taiwan
4 Program in Environmental and Occupational Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan
* Corresponding Author: Chiou-Hwa Yuh. Email: 
Oncology Research https://doi.org/10.32604/or.2026.074144
Received 03 October 2025; Accepted 30 January 2026; Published online 28 February 2026
Abstract
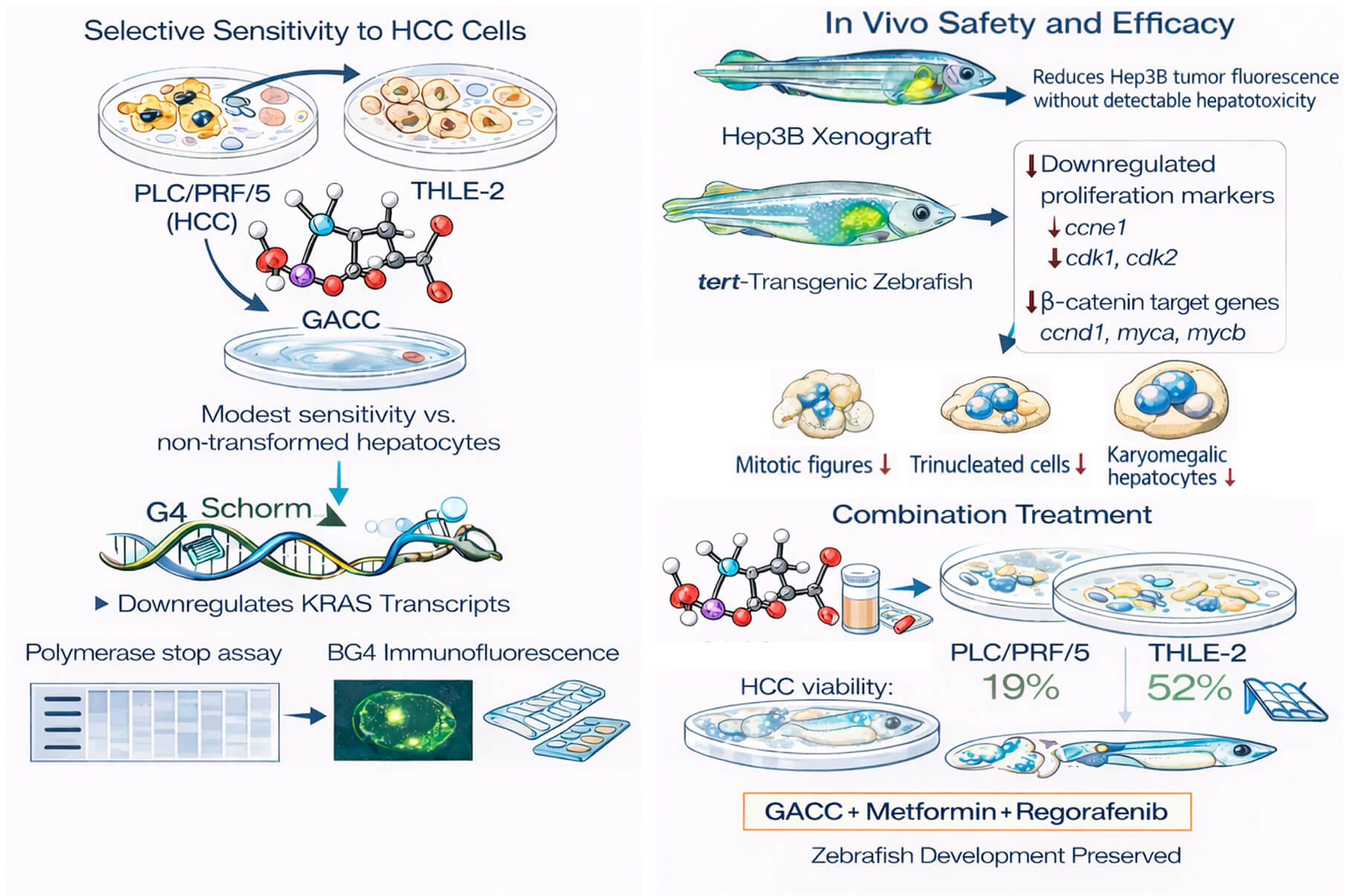
Objectives: Hepatocellular carcinoma (HCC) has limited systemic options with substantial toxicity. G-quadruplex (G4) structures in oncogene promoters are attractive but challenging drug targets. This study aimed to determine whether glutamic acid–chelated cobalt (GACC) is a G4-active scaffold with anti-HCC efficacy and favorable
in vivo safety, and whether an AI-guided phenotypic response surface (PRS) can optimize less toxic combinations.
Methods: Anticancer activity was tested in HCC cell lines (PLC/PRF/5, Hep3B, HepG2) and non-transformed THLE-2 hepatocytes (CCK-8, IC
50).
In vivo safety/efficacy were assessed in zebrafish embryo toxicity assays, a Hep3B xenograft model, and a tert-overexpressing transgenic zebrafish model, with hepatotoxicity monitored in a liver-fluorescent reporter line. Target engagement was examined by docking, native PAGE, a KRAS promoter G4 DNA polymerase stop assay, BG4 immunofluorescence, and KRAS qPCR. PRS was used to optimize GACC–metformin–regorafenib combinations.
Results: GACC reduced HCC viability (IC
50 ~86–115 µM) and showed low embryotoxicity (IC
50 6.87 mM). In zebrafish xenografts, GACC (50 µM) reduced Hep3B tumor fluorescence by ~90% without detectable hepatotoxicity, whereas sorafenib decreased liver size/fluorescence. In tert-overexpressing zebrafish, GACC suppressed proliferation and Wnt/β-catenin–associated transcripts and reduced mitotic figures and nuclear atypia. Mechanistically, GACC increased KRAS promoter polymerase stalling, enhanced nuclear G4 signal, and reduced KRAS transcripts. PRS identified an off-grid triple combination that reduced PLC/PRF/5 viability to 19% while maintaining THLE-2 viability at 52% and preserving zebrafish development.
Conclusion: GACC is a G4-active cobalt–glutamate scaffold with anti-HCC activity and favorable zebrafish safety, and a zebrafish-plus-PRS workflow enables rational, less toxic combination design.
Graphical Abstract
Keywords
Liver cancer; glutamic acid cobalt chelate;
tert transgenic zebrafish; G-quadruplex stabilization; phenotypic response surface;
KRAS promoter; zebrafish xenograft
 Open Access
Open Access