Tissue-Agnostic Targeting in Solid Tumors: A PRISMA-Compliant Meta-Analysis of Efficacy, Safety, and Resistance Determinants Across Histologies
Marwa Balaha1, Saad A. Aldosari2, Ahmed A. Alamer2, Nehad Ahmed2, Mohamed F. Balaha2,*
1 Department of Pharmacy, “G. d’Annunzio” University of Chieti-Pescara, Chieti, Italy
2 Department of Clinical Pharmacy, College of Pharmacy, Prince Sattam Bin Abdulaziz University, Al-Kharj, Saudi Arabia
* Corresponding Author: Mohamed F. Balaha. Email: 
Oncology Research https://doi.org/10.32604/or.2026.077965
Received 20 December 2025; Accepted 02 April 2026; Published online 17 April 2026
Abstract
Objectives: Tissue-agnostic oncology personalizes treatments based on shared molecular biomarkers, addressing challenges like assay variability, control-arm rigor, and non-proportional hazards. Integrating efficacy, safety, and resistance factors with consistent estimands is essential for evaluating biomarker-matched therapies across histologies. This review aims to quantify and compare their efficacy and safety, and to identify determinants of resistance, using PRISMA-compliant methods.
Methods: We conducted a systematic review and random-effects meta-analysis of 38 studies (15,018 participants), employing dual screening, standardized bias assessment, and evaluations of heterogeneity and small-study effects. Hazard ratios (HRs) with 95% CIs were estimated for time-to-event outcomes, and restricted mean survival times were used when the proportional hazards assumption was violated.
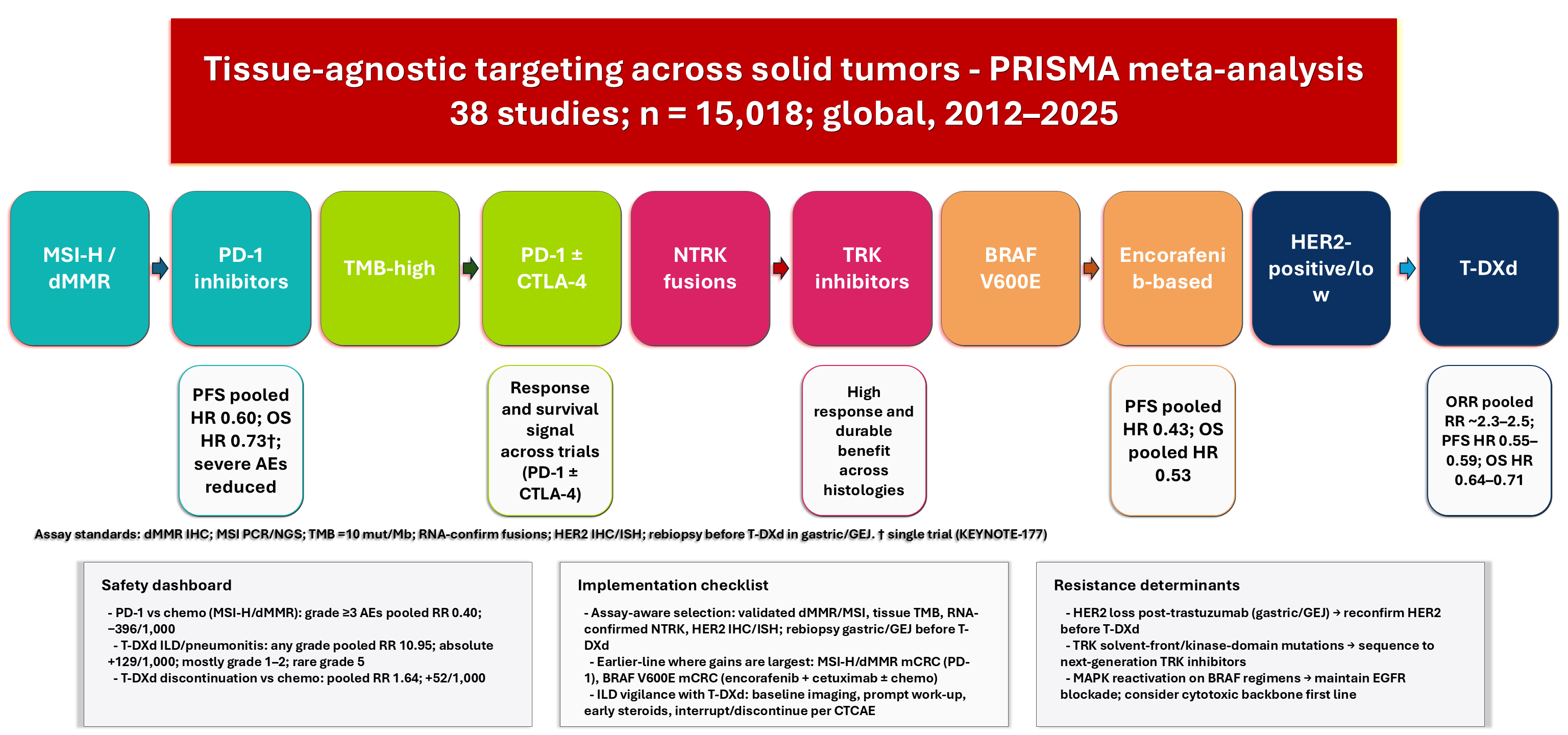
Results: Trastuzumab deruxtecan improved objective response rates and extended progression-free survival (PFS) and overall survival (OS) in HER2-positive gastric and gastroesophageal junction cancers and in HER2-low metastatic breast cancer, showing longer response durations. In metastatic colorectal cancer with microsatellite instability-high (MSI-H) or deficient mismatch repair (dMMR), PD-1 blockade significantly increased PFS and five-year OS despite crossover, with restricted mean survival time gains of about 11 months. In endometrial cancer, dostarlimab combined with chemotherapy improved PFS in both dMMR/MSI-H and mismatch repair–proficient disease and increased OS overall. Encorafenib-based therapies reduced progression and death in BRAF V600E metastatic colorectal cancer. Safety profiles were class-specific: PD-1 inhibitors caused fewer grade 3 or higher adverse events than chemotherapy, whereas trastuzumab deruxtecan was associated with increased interstitial lung disease (ILD) or pneumonitis and higher rates of treatment discontinuation.
Conclusion: Biomarker-matched therapies confer significant survival benefits with predictable toxicities. Confidence is strongest for PD-1 inhibitors in MSI-H/dMMR tumors, trastuzumab deruxtecan in HER2-low or HER2-positive cancers, and encorafenib-based regimens in BRAF V600E metastatic colorectal cancer. Implementation should include validated assays (including reconfirmation of HER2 status), prioritize earlier treatment lines where gains are greatest, and require vigilant ILD monitoring. Head-to-head trials and assay standardization, especially for tumor mutational burden, remain priorities.
Graphical Abstract
Keywords
Tissue-agnostic therapy; PRISMA; meta-analysis; MSI-H/dMMR; PD-1 inhibitor; HER2-low; antibody-drug conjugate; interstitial lung disease; BRAF V600E; NTRK fusion
 Open Access
Open Access