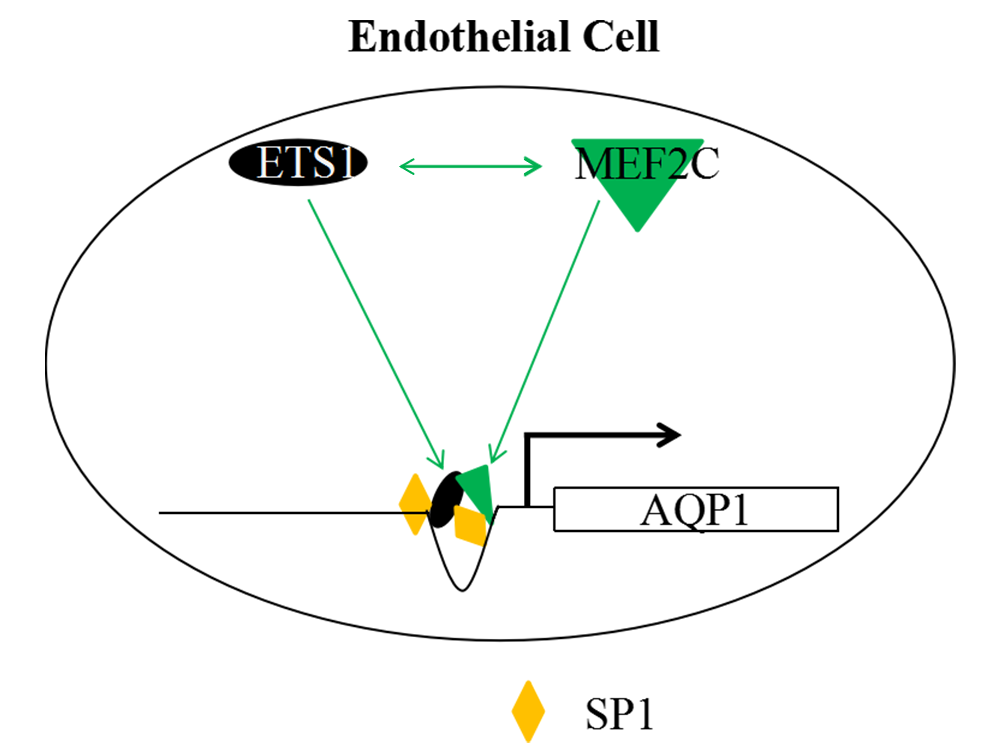
ETS1 Regulates Endothelial AQP1 Expression via Interaction with MEF2C in Endothelial Cells
YONG JIANG*, RUI MA, YU-GE WU, YI-MING HUO, HAN-ZHU ZHOU, JUN-XUAN ZHANG
School of Laboratory Medicine, Jilin Medical University, Jilin, China
* Corresponding Author: YONG JIANG. Email: 
BIOCELL https://doi.org/10.32604/biocell.2026.075982
Received 12 November 2025; Accepted 20 January 2026; Published online 13 February 2026
Abstract
Background: Aquaporin 1 (AQP1) plays a key role in myocardial ischemia-reperfusion (I/R) injury. This study aimed to elucidate the mechanisms by which erythroblast transformation-specific 1 (ETS1) and myocyte enhancer factor 2C (MEF2C) regulated AQP1 transcription.
Methods: Human umbilical vein endothelial cells
(HUVECs) and rats with coronary heart disease were employed for
in vitro and
in vivo experiments, respectively. Expressions of ETS1, MEF2C, and AQP1 were analyzed by western blotting and quantitative reverse transcription polymerase chain reaction (qRT-PCR). Chromatin immunoprecipitation (ChIP) and co-immunoprecipitation (Co-IP) assays were performed to confirm the interactions between ETS1 and MEF2C. Scratch wound healing and transwell assays were performed to assess the migration of HUVECs. Tube formation was conducted to assess the angiogenesis of HUVECs. HE and Masson staining were performed to assess the pathological changes and fibrosis of cardiac tissues; Immunofluorescence of platelet endothelial cell adhesion molecule (CD31) and AQP1 was performed to assess vascular endothelial cell dysfunction in cardiac tissues of rats with coronary heart disease.
Results: ETS1 overexpression upregulated both MEF2C and AQP1 expressions, while ETS1 silencing induced the opposite results. ETS1 and MEF2C interacted with each other and bound to the proximal promoter of AQP1. MEF2C synergized with ETS1 to promote the migration and tube formation of HUVECs, which were abolished by AQP1 silencing. I/R injury caused structural damage and fibrosis of cardiac tissues in rats, which were relieved by ETS1 and MEF2C silencing. Also, the increased AQP1 and CD31 levels in cardiac tissues of rats induced by I/R injury were reversed by ETS1 and MEF2C knockdown.
Conclusion: These results highlight the ETS1/MEF2C/AQP1 pathway as a potential therapeutic target for endothelial and vascular disorders.
Graphical Abstract
Keywords
Erythroblast transformation specific 1; aquaporin 1; myocyte enhancer factor 2C; endothelial cells
 Open Access
Open Access