miR-216a-5p Improves Macrophage M1 Polarization and Pyroptosis in URSA via Modulating HMGB1 to Regulate TLR4/NF-κB/NLRP3 Axis
Weina Xu1, Yi Xia1, Qing Shen1, Ling Ai1, Yingye Lu2,*
1 Department of Obstetrics, Jiaxing Maternity and Child Health Care Hospital, Jiaxing, China
2 Department of Traditional Chinese Medicine, Jiaxing Maternity and Child Health Care Hospital, Jiaxing, China
* Corresponding Author: Yingye Lu. Email: 
BIOCELL https://doi.org/10.32604/biocell.2026.076001
Received 12 November 2025; Accepted 27 February 2026; Published online 23 March 2026
Abstract
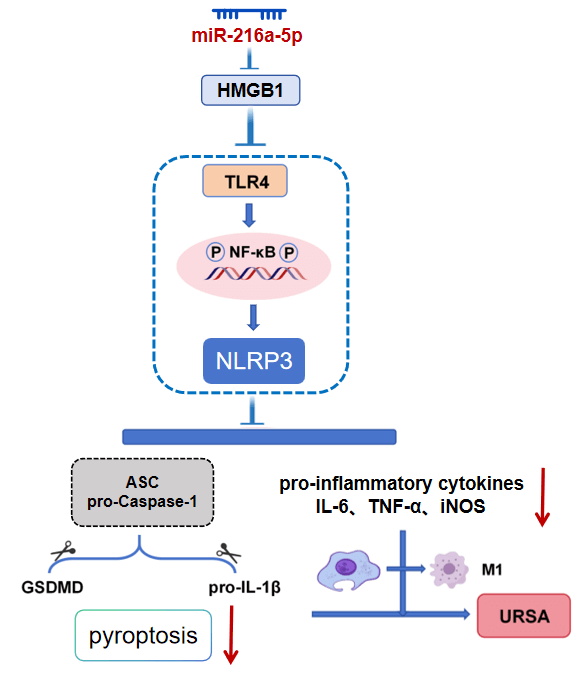
Background: Dysfunction of decidual macrophages (dMφ) mediated by high mobility group box 1 (HMGB1) is related to unexplained recurrent spontaneous abortion (URSA), but its upstream regulatory mechanism remains unclear. The research explores whether miR-216a-5p regulates the toll-like receptor 4/nuclear factor-κB/nucleotide-binding oligomerization domain-like receptor protein 3 (TLR4/NF-κB/NLRP3) signaling axis by targeting HMGB1, thereby affecting the M1 polarization and pyroptosis of dMφ in URSA.
Methods: The URSA mouse model was established, and primary dMφ was isolated and cultured. HMGB1 and miR-216a-5p were overexpressed or knocked down. Their expressions were examined. Their targeting relationship was verified through a bioinformatics database and dual luciferase assays. Immunofluorescence and Western blot detected macrophage M1/M2 phenotype and key indicators of pyroptosis. The TLR4/NF-κB/NLRP3 signal was evaluated. Additionally, HE staining was used to evaluate the tissue damage of the maternal-fetal interface (MFI).
Results: HMGB1 was enhanced, and miR-216a-5p was decreased within URSA mice. Overexpression of HMGB1 aggravated the inflammatory injury of decidual tissue in URSA mice, increased M1 macrophage proportion, and activated the TLR4/NF-κB/NLRP3 signal and pyroptosis pathway. miR-216a-5p inhibited HMGB1 expression, and its overexpression significantly reversed the above pathological changes, including reducing decidual inflammatory injury, inhibiting macrophage M1 polarization and pyroptosis, and inhibiting TLR4/NF-κB/NLRP3 signal.
Conclusion: miR-216a-5p inhibited TLR4/NF-κB/NLRP3 signal by targeting HMGB1, reduced M1 polarization and pyroptosis of dMφ, and alleviated inflammatory damage at the MFI of URSA. This finding offers novel bases for the mechanism study and targeted therapy of URSA.
Graphical Abstract
Keywords
miR-216a-5p; high mobility group box 1; decidual macrophages; unexplained recurrent spontaneous abortion
 Open Access
Open Access