Abstract
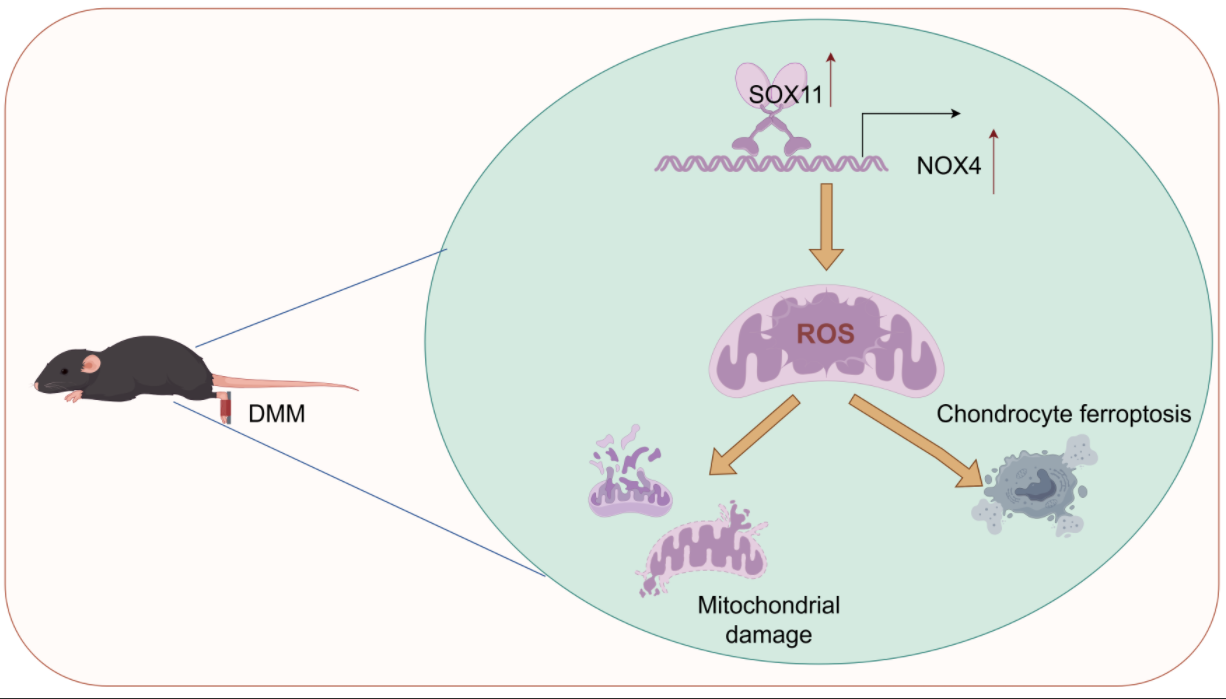
Objectives: Mitochondrial dysfunction and ferroptosis play crucial roles in osteoarthritis (OA), but the mechanisms remain unclear. This study aims to investigate the mechanism of sex-determining region Y-box transcription factor (SOX) 11/nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 4 (NOX4) axis-mediated mitochondrial dysfunction and ferroptosis in OA.
Methods: Destabilization of the medial meniscus (DMM) induced knee OA in mice. Chondrocytes were stimulated with IL-1β. Ferroptosis and mitochondrial function-related indicators were detected by immunofluorescence, 5,5
′,6,6
′-Tetrachloro-1,1
′,3,3
′-tetraethyl-imidacarbocyanine iodide (JC-1) staining, flow cytometry, quantitative real-time polymerase chain reaction (qRT-PCR), and Western blot.
Results: OA mice had 4.4 and 1.1-fold increase in SOX11 and NOX4 proteins (
p < 0.05). ChIP-qPCR and dual-luciferase reporter gene assays confirmed that SOX11 directly bound to the NOX4 promoter. Knocking SOX11 inhibited NOX4 and acyl-CoA synthetase long-chain family member 4 (ACSL4) levels in IL-1β-induced chondrocytes, promoted glutathione peroxidase 4 (GPX4) and solute carrier family 7 member 11 (SLC7A11) expression, and reduced Fe
2+ concentration and cell death. Additionally, SOX11 knockdown inhibited lipid peroxidation and ROS production and malondialdehyde (MDA) level, increased mitochondrial membrane potential, GSH, and SOD levels. NOX4 overexpression interrupted the beneficial effects.
In vivo experiments further validated that SOX11 knockdown reduced cartilage damage and Osteoarthritis Research Society International (OARSI) score, increased the transparent cartilage layer thickness, reduced calcified cartilage thickness, inhibited ferroptosis, and mitochondrial dysfunction.
Conclusion: Our results indicate that SOX11 inhibition reduces mitochondrial dysfunction and ferroptosis in IL-1β-induced chondrocytes by downregulating NOX4, thereby alleviating OA. This finding provides new targets for OA treatment.
Graphical Abstract
Keywords
Osteoarthritis; SRY-box transcription factor 11; nicotinamide adenine dinucleotide phosphate oxidase 4; ferroptosis; mitochondrial dysfunction
 Open Access
Open Access